ABSTRACT
Laterality is a basic characteristic of all life forms, from single cell organisms to complex plants and animals. For many metazoans, consistent left-right asymmetric patterning is essential for the correct anatomy of internal organs, such as the heart, gut, and brain; disruption of left-right asymmetry patterning leads to an important class of birth defects in human patients. Laterality functions across multiple scales, where early embryonic, subcellular and chiral cytoskeletal events are coupled with asymmetric amplification mechanisms and gene regulatory networks leading to asymmetric physical forces that ultimately result in distinct left and right anatomical organ patterning. Recent studies have suggested the existence of multiple parallel pathways regulating organ asymmetry. Here, we show that an isoform of the hyperpolarization-activated cyclic nucleotide-gated (HCN) family of ion channels (hyperpolarization-activated cyclic nucleotide-gated channel 4, HCN4) is important for correct left-right patterning. HCN4 channels are present very early in Xenopus embryos. Blocking HCN channels (Ih currents) with pharmacological inhibitors leads to errors in organ situs. This effect is only seen when HCN4 channels are blocked early (pre-stage 10) and not by a later block (post-stage 10). Injections of HCN4-DN (dominant-negative) mRNA induce left-right defects only when injected in both blastomeres no later than the 2-cell stage. Analysis of key asymmetric genes' expression showed that the sidedness of Nodal, Lefty, and Pitx2 expression is largely unchanged by HCN4 blockade, despite the randomization of subsequent organ situs, although the area of Pitx2 expression was significantly reduced. Together these data identify a novel, developmental role for HCN4 channels and reveal a new Nodal-Lefty-Pitx2 asymmetric gene expression-independent mechanism upstream of organ positioning during embryonic left-right patterning.
KEY WORDS: HCN4, Bioelectricity, Ion channels, Laterality, Xenopus
Summary: We identify a novel, developmental role for HCN4 channels and reveal a new Nodal-Lefty-Pitx2-independent, non-canonical mechanism upstream of organ positioning during embryonic left-right patterning. This article has an associated First Person interview with the first author of the paper as part of the supplementary information.
INTRODUCTION
Invariant left-right asymmetry is a fundamental aspect of all life, from single cell organisms to plants and animals with complex body plans like humans (Chen et al., 2012; Coutelis et al., 2008; Davison et al., 2016; Dimonte et al., 2016; Gros et al., 2009; Hashimoto, 2002; Kuroda et al., 2009; Naganathan et al., 2014; Petzoldt et al., 2012; Pohl, 2011; Spéder et al., 2007; Thitamadee et al., 2002; Wan et al., 2011; Xu et al., 2007; Yost, 1990, 1991). Consistent orientation of the left-right (LR) axis is a difficult problem for an embryo to solve in a universe that does not macroscopically distinguish left from right, and must be done reliably and accurately to achieve correct organization of internal organ structures. Errors in left-right asymmetry form a large and important class of human birth defects, affecting almost all major visceral organs, including the heart and the brain (Burn, 1991; Cohen et al., 2007; Hoffman and Kaplan, 2002; Peeters and Devriendt, 2006; Ramsdell, 2005). Hence, understanding the establishment of consistent laterality in the organization of body plans is a fundamental question in evolutionary and developmental biology, with important implications for addressing birth defects via regenerative medicine.
It is becoming clear that the origins of left-right asymmetry lie in physical aspects of cytoskeletal chirality (Naganathan et al., 2016; Suzuki et al., 2017; Tee et al., 2015; Wan et al., 2011), amplified immediately post-fertilization in the early embryos of many species (McDowell et al., 2016a,b; Okumura et al., 2008; Spéder et al., 2007; Vandenberg et al., 2013a; Vandenberg and Levin, 2013). The cytoskeletal chirality-mediated initiation of laterality is highly conserved across phyla, and even kingdoms (Levin and Nascone, 1997; Levin and Palmer, 2007; Lobikin et al., 2012; Okumura et al., 2008; Spéder et al., 2007). Multiple amplification and reinforcement mechanisms transmit this early left-right asymmetry across the entire developing embryo. These include asymmetric transport of ion translocators (Adams et al., 2006; Aw et al., 2008; Bessodes et al., 2012; Levin et al., 2002), charged molecules through gap junctions (Fukumoto et al., 2005a,b; Garic-Stankovic et al., 2008; Oviedo and Levin, 2007; Vandenberg et al., 2013b), and ciliary flow (Basu and Brueckner, 2008; Schweickert et al., 2007). Collectively, these signals trigger an embryo-wide asymmetric gene regulatory network, with a left-side expression of Nodal-Lefty-Pitx2 as the primary node (Levin, 1998; Mercola and Levin, 2001; Nakamura and Hamada, 2012; Ramsdell and Yost, 1998; Raya and Izpisua Belmonte, 2004a,b). Finally, asymmetric generation of mechanical forces results in asymmetric organ structure and placement (Granados-Riveron and Brook, 2012; Voronov et al., 2004; Welsh et al., 2013).
Previous work has shown an important role of ion fluxes-mediated regulation of membrane voltage in determination of left-right laterality (Adams et al., 2006; Aw et al., 2008, 2010; Garic-Stankovic et al., 2008; Hibino et al., 2006; Levin et al., 2006, 2002; Morokuma et al., 2008; Oviedo and Levin, 2007; Shimeld and Levin, 2006). Like other mechanisms involved in left-right laterality determination, these also largely feed into the Nodal-Lefty-Pitx2 node of gene regulatory networks. Recent evidence has uncovered a non-linearity between the cytoskeletal initiation of left-right asymmetry and establishment of organ situs, where errors in key aspects of the gene regulatory network, like the sidedness of Nodal expression, are bypassed to establish correct left-right organ situs despite randomized expression of upstream laterality determinant genes (Cota et al., 2006; McDowell et al., 2016a,b). These alternate pathways provide redundancy and robustness to the establishment of proper left-right organ situs, but are poorly understood. Here we report a new element of the endogenous bioelectric toolbox of left-right patterning, the hyperpolarization-activated cyclic nucleotide-gated channel 4 (HCN4), and present data suggesting that it mediates alternative pathways (bypassing the Nodal-Lefty-Pitx2 cassette) involved in the establishment of left-right asymmetry in Xenopus embryos.
Hyperpolarization-activated cyclic nucleotide-gated (HCN) channels are a unique group of voltage-gated channels where the threshold voltage for opening of the channel is modulated by the metabolic state of the cell (levels of cyclic nucleotides like cAMP) (Biel et al., 2009; Wahl-Schott and Biel, 2009). They open at hyperpolarized membrane voltages (negative), giving rise to currents which are a mix of sodium and potassium ion fluxes. Among the four HCN isoforms (HCN1-4), HCN4 channels have been primarily studied in adult hearts as pacemaker channels (Scicchitano et al., 2012; Verkerk and Wilders, 2015), but recent evidence has demonstrated their presence in human and mouse embryonic cells (Cerbai et al., 1999; Qu et al., 2008; Robinson et al., 1997; Später et al., 2013; Vicente-Steijn et al., 2011; Yasui et al., 2001), and implicated them in cardiac patterning (Pitcairn et al., 2017). Because these channels have not been studied in the context of developmental bioelectricity (Adams and Levin, 2013; Levin, 2013, 2014a,b) or control of embryonic axial patterning, we characterized the role of HCN4 channels in embryonic left-right asymmetry establishment in Xenopus embryos.
Here we extend our recent work on HCN4 channels in cardiogenesis (Pitcairn et al., 2017) using a different set of misexpression conditions to target earlier events. We show that HCN4 channels are present in Xenopus embryos beginning at the first cleavage event (2-cell stage). Pharmacological inhibition of HCN channels (Ih currents) by ZD7288 causes heterotaxia (randomization of organ situs) in Xenopus tadpoles, but only if embryos are exposed early (pre-stage 10, prior to the onset of gastrulation). Similarly, injections of mRNA encoding a dominant-negative protein (which blocks HCN4 channel function) randomize organ asymmetry if injected into both blastomeres at the 2-cell stage. Remarkably, despite the early period of action (prior to asymmetric gene expression), HCN4 channel disruption does not affect the laterality of subsequent Nodal, Lefty, or Pitx2 expression, exhibiting significant latency in its effects on later organ situs. Together, these results are the first, to our knowledge, to show an early embryonic axial patterning role of this important ion channel, and implicate it in a pathway bypassing the asymmetric gene expression of Nodal, Lefty, and Pitx2 in regulating left-right asymmetry.
RESULTS
Early exposure to pharmacological inhibitor ZD7288 induces heterotaxia
Our prior work demonstrated that interference with four different ion translocators (two channels and two pumps) induces heterotaxia, the independent randomization of organ positioning along the LR axis (Adams et al., 2006; Aw et al., 2008, 2010; Levin et al., 2002; Morokuma et al., 2008). To test whether HCN4 channels also play a role in embryonic left-right laterality determination, we first used the pharmacological inhibitor ZD7288 (100 µM), which blocks HCN channels (Ih currents) via a trapping mechanism (Shin et al., 2001). To probe the timing of the endogenous role of HCN4 channels in development, we compared the results of exposures beginning immediately after fertilization to those that began during gastrulation. Xenopus embryos were exposed to ZD7288 (100 µM) from stage 1 through stage 10 or starting at stage 10 until stage 40, followed by anatomical laterality analysis at stage 45 (swimming tadpoles). Any instance of mirror image reversal of organ position, in the context of normal organ patterning and normal dorso-anterior index was counted as an instance of heterotaxia. Untreated embryos served as controls.
ZD7288 exposure during stages 1-10 induced a significantly high incidence (∼26%, P<0.001, χ2) of heterotaxia in comparison to controls (∼6%) (Fig. 1A,B). In sharp contrast, ZD7288 exposure during stages 10-40 did not induce heterotaxia (∼6%) in comparison to controls (∼6%) (Fig. 1A,B). Similar results were observed with another, more-specific HCN4 channel inhibitor ivabradine (Fig. S1). Distribution of heterotaxia outcomes from the early ZD7288 exposure showed various combinations of asymmetric placement of gut, heart and gallbladder (Fig. 1C). However, the majority (∼86%) of left-right asymmetric tadpoles showed situs inversus – full mirroring of all three organs (gut, heart and gallbladder) (Fig. 1B,C). Overall, percentage of ZD7288-treated tadpoles showing incorrect placement of each of the three organs were approximately equal (heart ∼34%, gut ∼32%, gallbladder ∼34%). Together, these results suggest that HCN4 function is required prior to embryonic stage 10 in establishing left-right asymmetry.
Fig. 1.
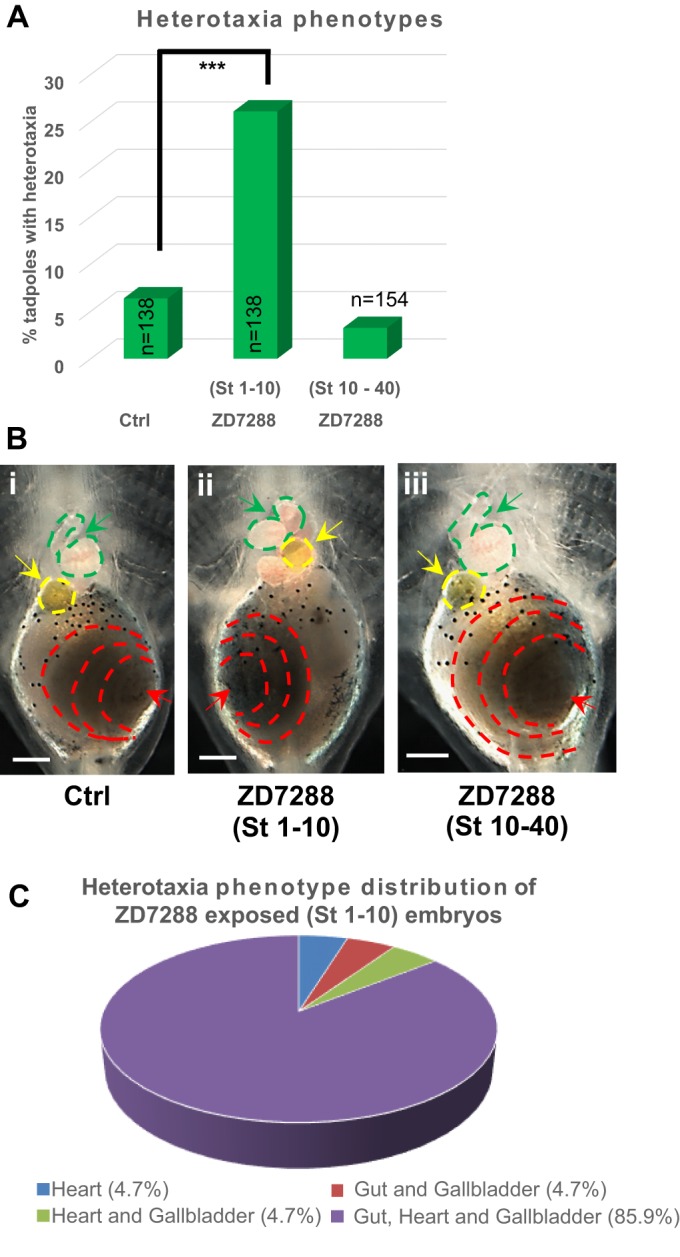
HCN4 channel inhibitor ZD7288 affects left-right organ laterality only upon early exposure of embryos (St 1-10). (A) Quantification of stage 45 tadpoles for left-right organ (heart, gut and gallbladder) laterality with or without exposure to 100 µM ZD7288 at 22°C at the indicated stages. A significantly high incidence of heterotaxia was observed in embryos exposed to ZD7288 between stages 1-10 in comparison to controls. Embryos exposed to ZD7288 late (St 10-40) did not show any significant increase in the incidence of left-right organ misplacement. The experiment was conducted in triplicates and data was pooled to run a χ2 analysis, ***P<0.001. (B) Representative images of stage 45 tadpoles: (i) Control tadpole showing rightward coiling gut as indicated by the red dotted lines and red arrow, rightward coiling heart as indicated by green dotted lines and green arrow, and leftward placed gallbladder as indicated by yellow dotted line and yellow arrow, (ii) tadpoles from embryos exposed to ZD7288 (100 µM – St 1-10) showing inversion of gut coiling as indicated by the red dotted lines and red arrow, inversion of the heart as indicated by green dotted line and green arrow, and inversion of gallbladder placement as indicated by yellow dotted lines and yellow arrow, (iii) tadpoles from embryos exposed to ZD7288 (100 µM – St 10-40) showing normal gut coiling as indicated by the red dotted lines and red arrow, normal heart as indicated by green dotted line and green arrow, and normal gallbladder placement as indicated by yellow dotted lines and yellow arrow. Scale bar: 0.25 mm. (C) Pie chart showing the incidence of various left-right phenotypes seen in the tadpoles from embryos exposed to ZD7288 (100 µM) between stages 1-10.
These studies took advantage of the temporal control afforded by pharmacological experiments beginning at different timepoints. Treatment with ZD7288, which inhibits HCN (Ih currents), resulted in high incidence of situs inversus. These data implicate the HCN4 channel in early (prior to stage 10) processes involved in determination of left-right patterning in the Xenopus embryo.
HCN4-DN (dominant-negative) causes heterotaxia
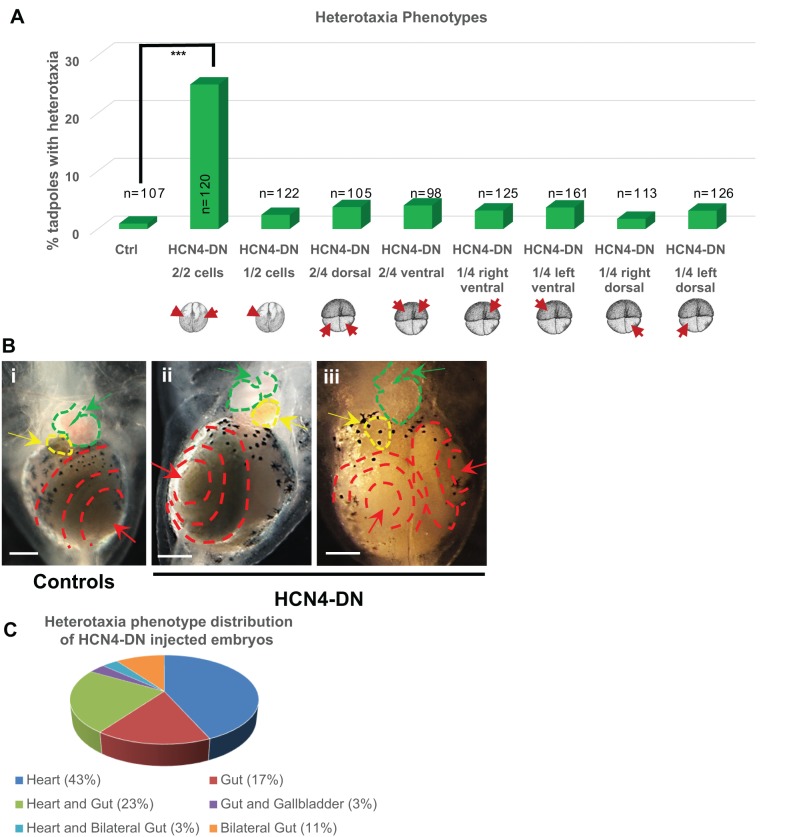
To molecularly validate whether HCN4 channels are specifically involved in organizing left-right organ laterality, we used an HCN4-DN (hyperpolarization-activated cyclic nucleotide-gated channel 4-dominant negative) mRNA construct that has been previously molecularly characterized and shown to inhibit the HCN4 channel current in mammalian cell culture by direct electrophysiology (Pitcairn et al., 2017). We confirmed that expression of HCN4-DN protein blocks HCN4 channel function and depolarizes the membrane voltage in Xenopus embryos (Fig. S2). We used a dominant-negative construct because many channels are present in Xenopus as maternal proteins (Adams et al., 2006; Aw et al., 2008, 2010; Levin et al., 2002; Morokuma et al., 2008; Qiu et al., 2005), and thus cannot be targeted by morpholinos or RNAi. Xenopus embryos were injected with HCN4-DN mRNA at 2- and 4-cell stage, in various blastomeres as indicated in Fig. 2A (red arrows), followed by anatomical laterality analysis at stage 45 (swimming tadpoles). As with the pharmacological inhibitor experiments, any deviation from the normal laterality of the three organs, in the context of otherwise normal patterning, was counted as an instance of heterotaxia. Uninjected embryos served as controls [as neither water nor non-specific mRNA, e.g. β–galactosidase, injections affect left-right asymmetry endpoints (McDowell et al., 2016b)]. HCN4-DN mRNA-injected tadpoles showed a significantly high incidence (25%, P<0.001, χ2) of heterotaxia only when injected in both blastomeres at 2-cell stage in comparison to controls (1%) (Fig. 2A,B). Distribution of all the different combinations of the three organs among the heterotaxic population is shown in Fig. 2C. The majority of phenotypes were inverted gut (∼17%), inverted gut and heart (∼23%), and inverted heart (∼43%) (Fig. 2C), with a few cases (∼10%) of bilateral gut (Fig. 2B,C). Overall, in HCN4-DN mRNA-injected tadpoles, gut (44%) and heart (54%) were the most affected organs. In contrast to HCN4-DN, HCN2-DN mRNA-injected tadpoles showed no effect on laterality (Fig. S3). Taken together, these data validate, in a gene-specific manner, the loss-of-function pharmacological inhibitor data implicating a role for HCN4 in LR patterning.
Fig. 2.
Early injection of HCN4-DN affects left-right organ laterality in Xenopus laevis. (A) Quantification of stage 45 tadpoles for left-right organ (heart, gut and gallbladder) laterality with or without microinjecting HCN4-DN mRNA apically (∼0.5-1 ng/injection/blastomere) in both blastomeres at 2-cell stage as indicated in the illustrations. A significantly high incidence of heterotaxia was observed in only when HCN4-DN mRNA was injected in both blastomeres at 2-cell stage, in comparison to controls. The experiment was conducted in triplicate and data was pooled to run a χ2 analysis, ***P<0.001. (B) Representative ventral images of stage 45 tadpoles: (i) Control tadpole showing rightward coiling gut as indicated by the red dotted lines and red arrow, rightward coiling heart as indicated by green dotted lines and green arrow, and leftward placed gallbladder as indicated by yellow dotted line and yellow arrow, (ii) HCN4-DN mRNA injected (both blastomeres at 2-cell stage) tadpoles showing inversion of gut coiling as indicated by the red dotted lines and red arrow, inversion of the heart as indicated by green dotted line and green arrow, and inversion of gallbladder placement as indicated by yellow dotted lines and yellow arrow, (iii) HCN4-DN mRNA injected (both blastomeres at 2-cell stage) tadpoles showing bilateral gut coiling as indicated by the red dotted lines and red arrow, with normal heart (green dotted lines and green arrow) and gallbladder (yellow dotted line and yellow arrow). Scale bar: 0.25 mm. (C) Pie chart showing the incidence of various left-right phenotypes seen in the HCN4-DN mRNA injected (in both blastomeres at 2-cell stage) tadpoles.
We then probed the time-dependency of HCN4 activity in left-right patterning by injecting at subsequent cleavage stages. We found that, even when adjusted for identical amounts and distribution of mRNA, injections performed after the second cell cleavage did not result in any significant incidence of left-right organ laterality defects in comparison to controls (Fig. 2A). We conclude that HCN4 channel activity is involved in determining heart and visceral organ laterality; moreover, its activity must occur very early during cleavage stages, as HCN4-DN mRNA introduced even at 4-cell stage is no longer effective at randomizing the left-right axis.
The HCN4 channel is present very early in Xenopus embryonic development
The presence and distribution of HCN4 channels during early embryonic developmental stages is not currently known. To assess this, we analyzed the spatio-temporal distribution of maternal HCN4 channels using whole embryo immunofluorescence on Xenopus embryos. HCN4 channel immunohistochemistry signal was detected as early as cleavage-stage (2-cell stage) and was present through stage 9 in Xenopus embryos (Fig. 3). No primary antibody-stained embryos were used as controls (Fig. 3). These results show that HCN4 channel is present very early 2-cell stage in developing frog embryos, and together with the bioelectric imaging and functional studies, are consistent with early exposure to HCN4-blocking reagents targeting endogenous physiological machinery operating at the earliest stages of LR patterning. Thus, we next sought to examine the transcriptional mechanisms known to be downstream of early bioelectric states.
Fig. 3.
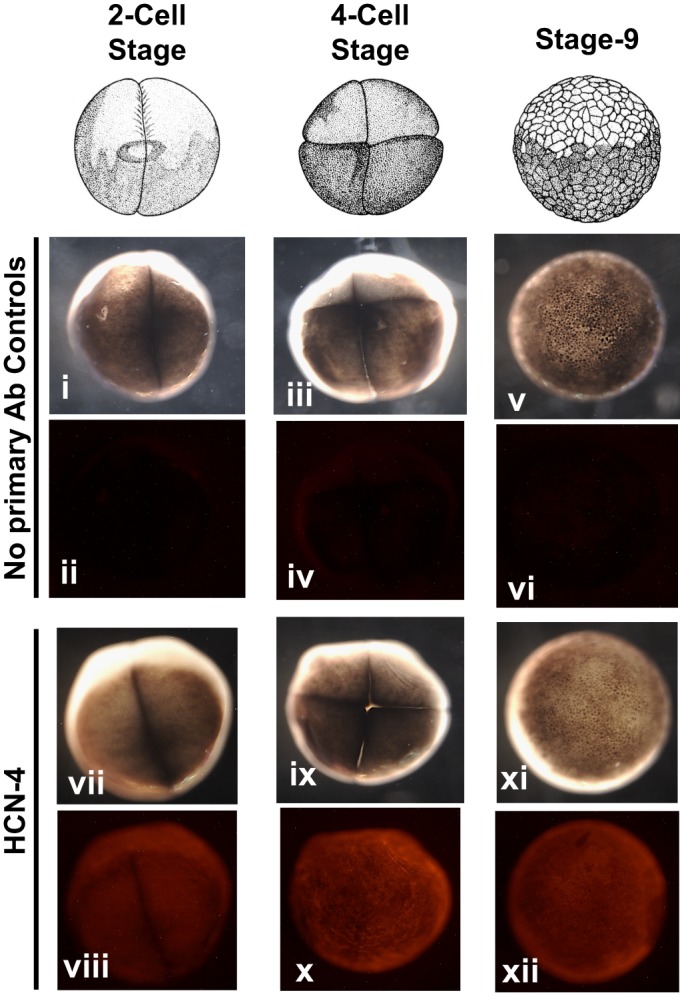
Xenopus laevis embryos express endogenous HCN4 channel during early development. Immunofluorescence analysis of whole Xenopus embryos for HCN4 channel protein at indicated stages of development. (i-vi) No primary antibody controls, (vii – xii) HCN4 immunofluorescence, (i, iii, v, vii, ix, xi) bright field images of immunofluorescent embryos, (ii, iv, vi, viii, x, xii) fluorescence images of immunofluorescence embryos. Xenopus embryos at the indicated stage of development showed a prominent HCN4 channel protein (n=15).
Neither HCN4-DN nor an HCN channel (Ih current) inhibitor affect the sidedness of Nodal and Lefty transcriptional asymmetry
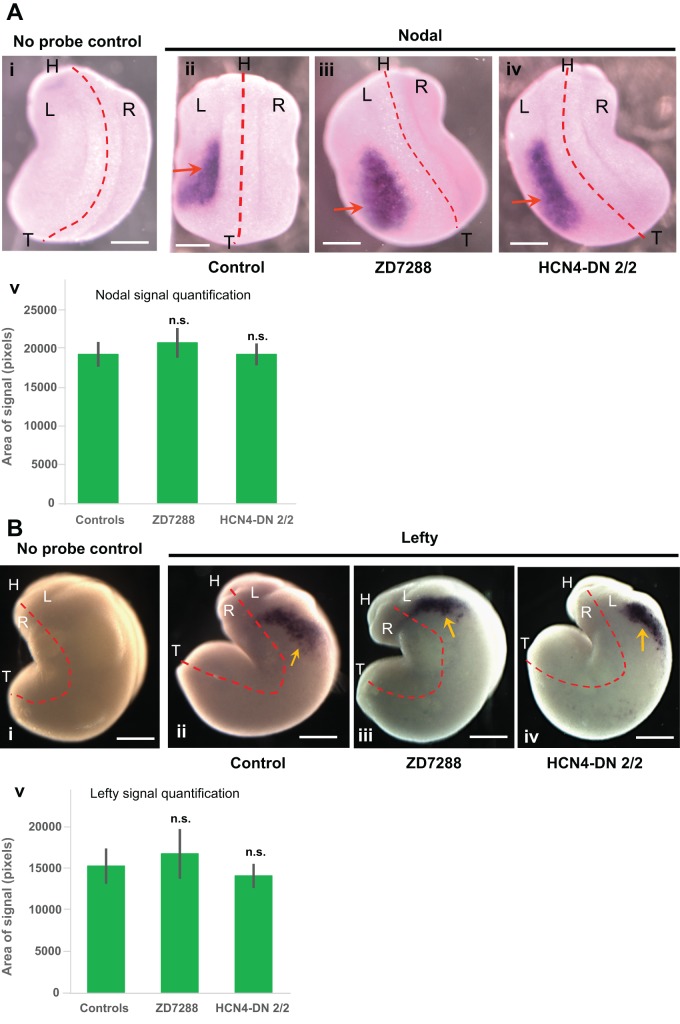
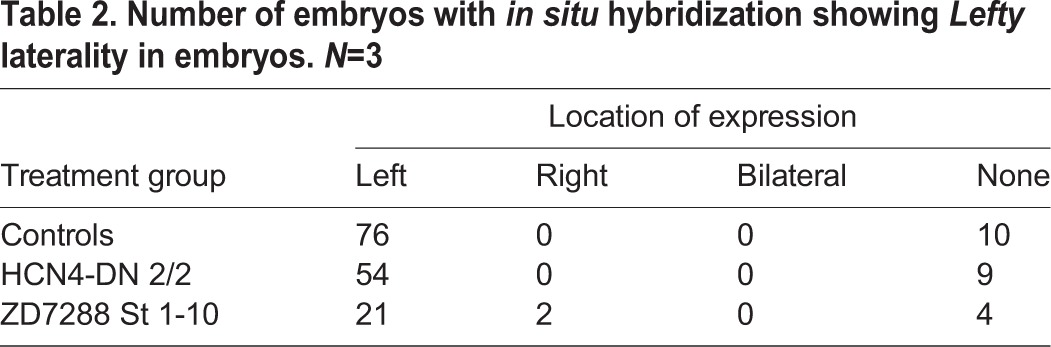
The asymmetric position of the organs is regulated by a cascade of asymmetric gene expression (Levin, 2005; Ramsdell and Yost, 1998; Raya and Izpisua Belmonte, 2006); especially crucial are the left-side markers Nodal, Lefty, and Pitx2, and indeed the other known early mechanisms of asymmetry all act by randomizing the expression of these left-side determinant genes. Thus, we asked if HCN4 channel inhibition induces randomization by perturbing the normally consistent sidedness of this transcriptional cascade. To assess the epistasis between the Nodal-Lefty-Pitx2 cassette and HCN4 function, we first performed whole embryo in situ hybridization with antisense probes against Nodal and Lefty. Xenopus embryos were either injected with HCN4-DN mRNA in both blastomeres at 2-cell stage (the misexpression condition that leads to heterotaxia, Fig. 2), or treated with the HCN channel (Ih current) pharmacological inhibitor ZD7288 (100 µM) between stages 1-10. Uninjected and untreated embryos were used as controls. The embryos were fixed at stage 21 for Nodal (Fig. 4A, Table 1) and at stage 23 for Lefty (Fig. 4B, Table 2) in situ hybridization analysis.
Fig. 4.
Localization of the asymmetric gene Xnr-1 (nodal) is not affected by HCN4-DN and ZD7288. (A) Representative images of approximately stage 21 embryos assayed for Xnr-1 (nodal) expression by in situ hybridization and quantification of area of nodal expression. Red dotted line is midline and L representing left-side, R representing right-side, H representing head and T representing tail of embryos. Scale bar: 0.25 mm. (i) No probe (negative) untreated control, (ii) control embryos with Xnr-1 signal – red arrow, (iii) ZD7288-treated (from stage 1-10) embryos with Xnr-1 signal – red arrow, (iv) HCN4-DN mRNA injected (in both blastomeres at 2-cell stage) embryos with Xnr-1 signal – red arrows, and (v) quantification of area of Nodal expression in embryos showed no significant change in the area of Nodal expression in HCN4-DN mRNA-injected and ZD7288-treated embryos. N>10; data was analyzed by one-way ANOVA; n.s., non-significant. (B) Representative images of approximately stage 23 embryos assayed for Lefty expression by in situ hybridization and quantification of area of lefty expression. Red dotted line is midline and L representing left-side, R representing right-side, H representing head and T representing tail of embryos. Scale bar: 0.25 mm. (i) No probe (negative) untreated control, (ii) control embryos with Lefty signal – yellow arrow, (iii) ZD7288-treated (from stage 1-10) embryos with Lefty signal – yellow arrow, (iv) HCN4-DN mRNA-injected (in both blastomeres at 2-cell stage) embryos with Lefty signal – yellow arrows, and (v) quantification of area of Lefty expression in embryos showed no significant change in the area of Lefty expression in HCN4-DN mRNA-injected and ZD7288-treated embryos. N=10; data was analyzed by one-way ANOVA; n.s., non-significant.
Table 1.
Number of embryos with in situ hybridization showing Xnr-1 (Nodal) laterality in embryos. N=3
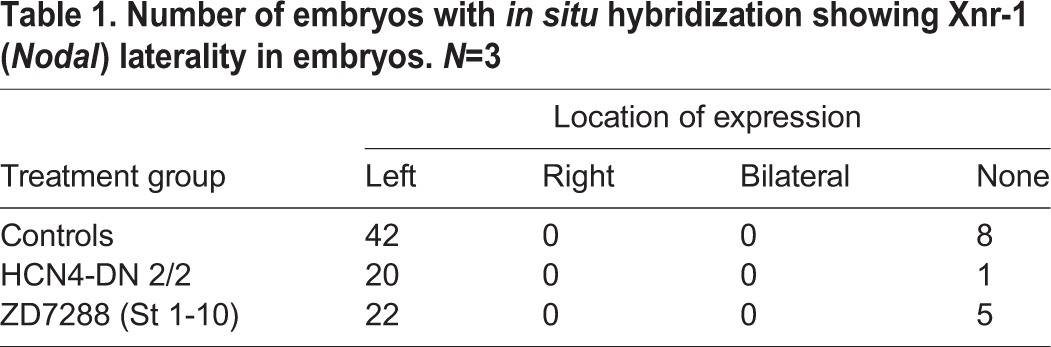
Table 2.
Number of embryos with in situ hybridization showing Lefty laterality in embryos. N=3
In controls, as expected, Nodal was expressed on the left side of the embryo in the majority (∼84%) of the embryos (Fig. 4A, Table 1). HCN4-DN mRNA-injected and ZD7288-treated embryos showed no significant change in the laterality of Nodal signal, with the majority of embryos (∼95% and ∼81%, respectively) expressing Nodal on the left side (Table 1). Quantification of area of Nodal expression domain showed no significant difference between the HCN4-DN mRNA-injected and ZD7288-treated embryos, and control embryos (Fig. 4Av, ANOVA). A subset of control and treated embryos from the same batch used for in situ analysis were raised to stage 45 and scored for left-right organ placement, confirming that HCN4-DN mRNA and ZD7288 were still effectively inducing heterotaxia in this cohort of animals, just as seen in Figs 1 and 2 (data not shown). We conclude that very early disruption of HCN4 function randomizes organ situs, bypassing the normal sequence of asymmetric Nodal expression.
In control embryos, Lefty was expressed on the left side in the majority (∼88%) of the embryos (Fig. 4B, Table 2). Strikingly, HCN4-DN mRNA-injected and ZD7288-treated embryos showed no significant change from wild-type embryos in the laterality of Lefty signal with majority of embryos (∼86% and ∼78%, respectively), showing Lefty on left side as in the set of control embryos (Table 2). Quantification of area of Lefty expression domain showed no significant difference between the HCN4-DN mRNA-injected and ZD7288-treated embryos, and control embryos (Fig. 4Bv, ANOVA). A subset of control and treated embryos from the same batch used for in situ analysis were raised until stage 45 and scored for left-right organ placement to verify that HCN4-DN mRNA and ZD7288 were still inducing heterotaxia in this cohort, as seen in Figs 1 and 2 (data not shown).
Thus, HCN4-DN mRNA and ZD7288 induce left-right organ randomization without altering the normal left-sided expression of Nodal and Lefty. These results reveal that HCN4 channel function affects organ placement, but bypasses the major asymmetry-regulating gene cassette, Nodal-Lefty.
HCN4-DN and HCN channel (Ih current) inhibition affect Pitx2 expression
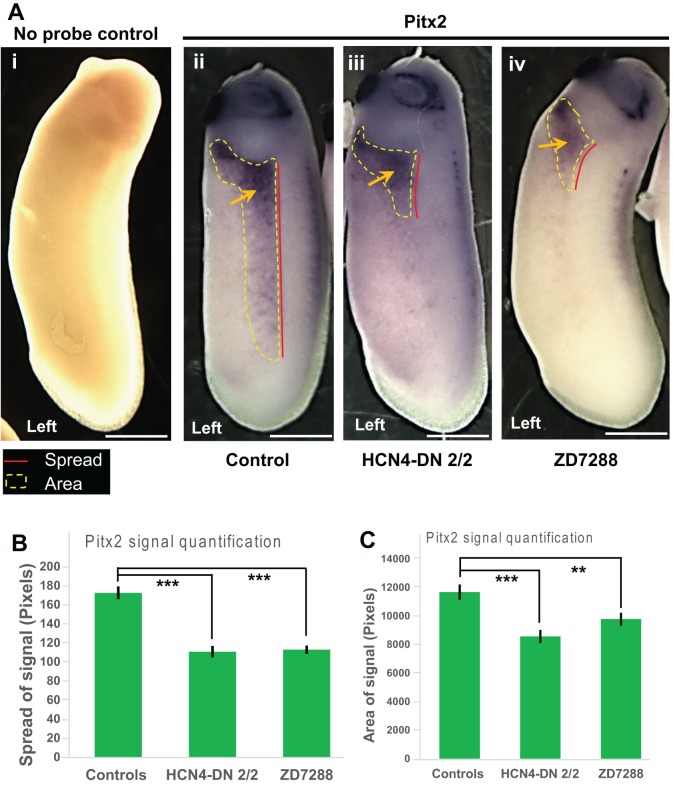
To assess if HCN4 channel inhibition affects the normally left-sided expression of the late marker Pitx2, we performed whole embryo in situ hybridization against Pitx2. Xenopus embryos were either injected with HCN4-DN mRNA in both blastomeres at 2-cell stage or treated with a pharmacological inhibitor of HCN channels (Ih currents), ZD7288 (100 µM), between stages 1-10. Uninjected and untreated embryos were used as controls [as these are known to be equivalent to water- or nonspecific mRNA-injected embryos (McDowell et al., 2016b)]. The embryos were fixed at stage 28 for Pitx2 (Fig. 5, Table 3) in situ hybridization analysis. In controls, as expected, Pitx2 is present on the left side of the embryo in majority (∼87%) of the embryos (Fig. 5A, Table 3). HCN4-DN mRNA-injected and ZD7288-treated embryos caused no significant change in the laterality of Pitx2 signal, with a majority of embryos (∼81% and ∼87%, respectively) showing Pitx2 on the left side of the embryos similar to controls (Table 3). A subsection of control and treated embryos from the same batch used for in situ analysis were raised to stage 45 and scored for left-right organ placement, confirming that HCN4-DN mRNA and ZD7288 were still inducing heterotaxia as seen in Figs 1 and 2 (data not shown). We conclude that the randomizing effects of HCN4 inhibition can bypass asymmetric Pitx2 gene expression.
Fig. 5.
Pitx2 expression is affected by HCN4-DN and ZD7288. (A) Representative images of approximately stage 28 embryos assayed for Pitx2 expression by in situ hybridization. Left orientation of the embryo is indicated at the bottom of the image. Red line indicates the anterior-posterior spread of the Pitx2 expression and yellow dotted line indicates the area of the Pitx2 expression. (i) No probe (negative) untreated control, (ii) control embryos with Pitx2 signal – yellow arrow, (iii) embryos injected with HCN4-DN mRNA in both blastomeres at 2-cell stage with Pitx2 expression - yellow arrow, (iv) ZD7288-treated (100 µM stage1-10) embryo with Pitx2 expression - yellow arrow. Scale bar: 0.25 mm. (B) Quantification of anterior-posterior spread of Pitx2 expression (as indicated by red lines in A) in embryos showed a significant reduction in the spread of Pitx2 expression in HCN4-DN mRNA-injected and ZD7288-treated embryos. N=20; data was analyzed by one-way ANOVA; ***P<0.001. (C) Quantification of area of Pitx2 expression (as indicated by yellow dotted lines in A) in embryos showed a significant reduction in the area of Pitx2 expression in HCN4-DN mRNA-injected and ZD7288-treated embryos. N>25; data was analyzed by one-way ANOVA; ***P<0.001, **P<0.01.
Table 3.
Number of embryos with in situ hybridization showing Pitx2 laterality in embryos. N=3
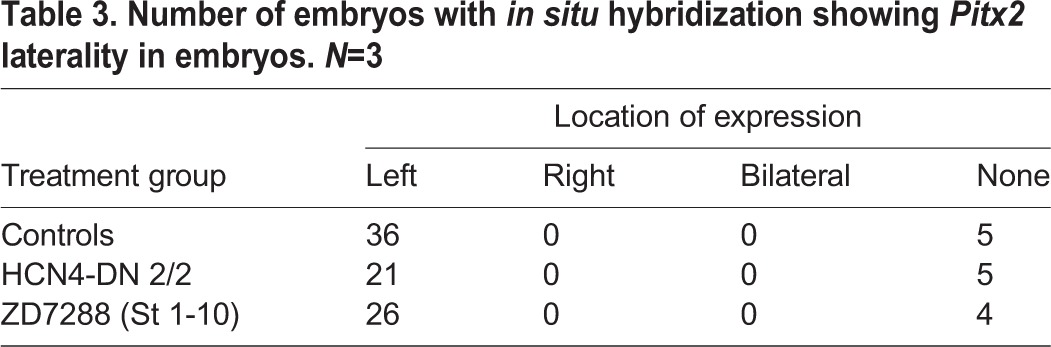
Examining closely the results of treatments that randomized organs but not Pitx2 situs, we observed that in contrast to the normal sidedness of expression, there was a significant difference in the spatial extent (pattern) of the Pitx2 transcriptional domain in the treated embryos in comparison to controls. To analyze this, we quantified the anterior to posterior spread distance and area of the Pitx2 signal in embryos (Fig. 5A-C). The anterior-posterior spread of Pitx2 signal was significantly reduced (***P<0.001, ANOVA) in HCN4-DN mRNA-injected and ZD7288-treated embryos (Table 3). Similarly, the total area of Pitx2 signal was significantly reduced (***P<0.001, **P<0.01, ANOVA) in HCN4-DN mRNA-injected and ZD7288-treated embryos (Fig. 5C). A similar analysis of the Nodal and Lefty expression domains revealed no significant changes in the signal pattern (Fig. 4). These results show that while HCN4-DN mRNA and ZD7288 treatments do not affect the laterality of Pitx2 gene expression, both treatments significantly reduce the anterior to posterior spread distance and the area of Pitx2 expression, while inducing left-right organ patterning defects in tadpoles.
DISCUSSION
Here, we show that HCN4 channels are present in Xenopus embryos from the earliest stages of development, and play an important role in establishing left-right organ situs in Xenopus tadpoles. Blocking HCN4 channels results in heterotaxia and incorrect organ situs in tadpoles. This is effective only during early embryogenesis (pre-stage 10) as post stage 10, HCN4 channel blocking has no effect on organ situs. Even within the first 10 stages of embryogenesis, the HCN4-DN construct induces heterotaxia only when introduced in all blastomeres during early cleavage stages, further suggesting a very early role of HCN4 in establishing left-right asymmetry. Interestingly, an early block of HCN4 with ZD7288 and HCN4-DN does not have a significant effect on the sidedness of Nodal-Lefty-Pitx2 gene expression, even though it leads to incorrect organ situs, suggesting it is acting either via a canonical pathway but bypassing Nodal and Lefty and directly affecting downstream targets or an alternative pathway of left-right asymmetry that is able to bypass the Nodal-Lefty-Pitx2 cassette completely. Blocking of HCN4 channels (both pharmacologically and physiologically) induces a significant decrease in Pitx2 signal in its anterior-posterior spread and area of the signal.
Pleotropic actions of HCN4 in Xenopus embryonic development
Our recent study explored HCN4 function in embryonic cardiac tissue development (Pitcairn et al., 2017). That study and the experiments presented here used drastically different injection and culture conditions to target two different lineages/tissues: the animal cap versus the mesodermal heart lineage. The major differences were as follows. (1) In Pitcairn et al. (2017), where the role of HCN4 during heart development was the focus of study, the HCN4-DN was injected medially (along the equitorial plane of the embryo) which targets mostly mesodermal cardiac and kidney tissues. Here our injections were apical, to achieve a more global expression in the embryo, especially targeting the animal cap ectoderm and derived structures. (2) In Pitcairn et al. (2017), embryos were injected (equatorially) in one blastomere at the two-cell stage and kept in relatively high salt (1× MMR) until stage 9 (gastrulation). Under these high salt conditions, the ion gradients (and hence ion flux) for K+ and Na+ are reversed, affecting all K+ and Na+ ion channel fluxes: blocking HCN4 channel using HCN4-DN is a hyperpolarizing treatment and leads to subsequent incorrect Nodal-Lefty-Pitx2 expression and gross morphology defects of heart. In contrast, the experiments reported here made use of injections (apical) into both blastomeres at the 2-cell stage, and the embryos were kept in low salt 0.1× MMR (mimicking its natural environment) throughout development. Under these conditions, an HCN4-DN-mediated block of HCN4 ion flux leads to depolarization (Fig. S2) with normal sidedness and pattern of Nodal-Lefty-Pitx2 expression (Figs 4 and 5, Tables 1-3), but randomized organ situs (with normal organ morphology) (Figs 1 and 2).
The two different conditions help tease apart two different functions of HCN4: an early activity involved in establishing laterality of organs and another in developmental morphology of heart. Previously, it has been shown that moving the membrane voltage in either direction away from wild type leads to defects (Pai et al., 2012, 2015). Since early embryos already have quite depolarized membrane potentials, the HCN4-DN-mediated depolarization perturbation reported here may be too subtle to affect the asymmetry of the Nodal-Lefty-Pitx2 expression, but is sufficient to affect a parallel pathway that feeds into organ situs. However, injections targeted to the developing heart in high salt conditions, combined with HCN4 blockade, induce a strong hyperpolarization perturbation (Pitcairn et al., 2017), which is a more drastic change that affects several gene regulatory networks involved in organ morphology, including feedback loops between Pitx2 and HCN4 during cardiac morphgenesis (Christoffels et al., 2010; Clauss and Kaab, 2011; Wang et al., 2010). Work is currently ongoing in our laboratories to construct a detailed, testable, physiologically-realistic, and spatialized model of the early frog embryo's bioelectric circuits, chemical gradients, and relevant gene-regulatory circuitry (Pietak and Levin, 2016).
HCN4 channels can act in a Nodal-Lefty asymmetric gene expression-independent manner
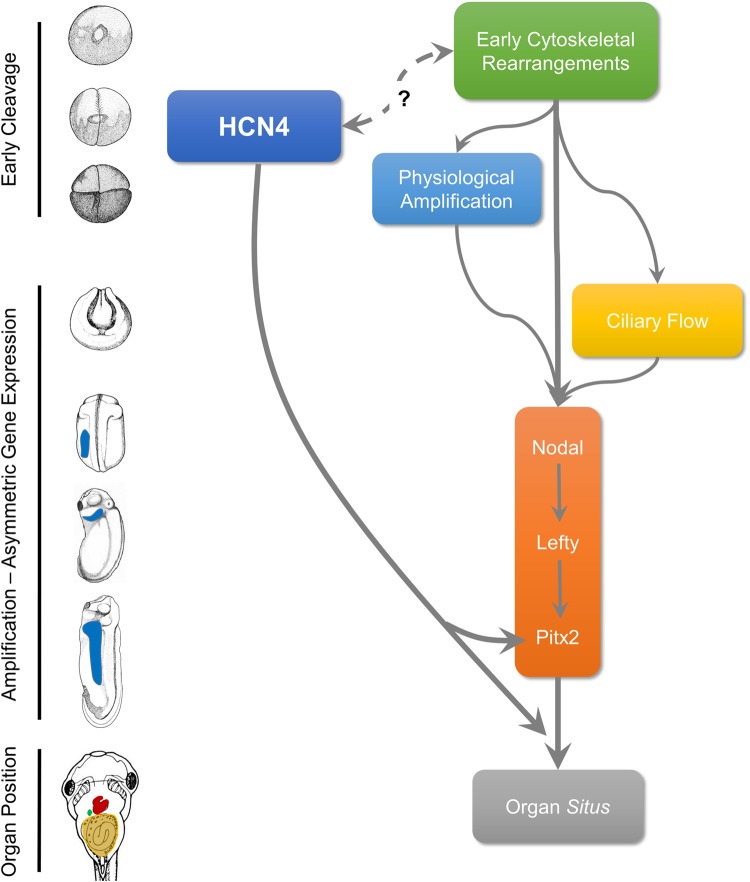
A recent series of studies and meta-analyses looking at the relation between causes of left-right abnormality, asymmetric gene expression of Nodal-Lefty-Pitx2, and organ situs, strongly suggested non-linearity of the laterality pathway (McDowell et al., 2016a,b; Vandenberg, 2012). Methods leading to disruption of asymmetric Nodal expression can still show correct organ situs, suggesting the presence of flexible gene regulatory networks and the presence of alternative pathways conferring redundancy and robustness to laterality and particularly organ situs. Some laterality defects were corrected even past Pitx2 expression. Perturbation of early cytoskeleton, motor proteins, gap-junctions, and serotonin signaling, all showed significant repair [high incidence of mispatterned Nodal, but a lower incidence of organ situs defects (McDowell et al., 2016a)] (Fig. 3). However, a particularly interesting observation was that defects induced by perturbing laterality-relevant ion fluxes did not correct over time (incidence of organ situs defects were higher than incidence of incorrect sidedness of Nodal), strongly suggesting possible bioelectrically mediated Nodal-independent pathway that could correct for errors in the normal laterality-establishing transcriptional pathway.
Here we observed the opposite phenomenon: correct expression of asymmetric genes Nodal and Lefty but incorrect organ situs after HCN4 inhibition (Figs 1, 2, 4 and 5). In case of Pitx2 the sidedness of expression was correct (left side) but the area of expression was significantly reduced. Although Nodal and Lefty expression was unaltered, it is possible that HCN4 inhibition interferes with the function of Nodal and Lefty proteins, resulting in decreased Pitx2 expression and/or affecting other downstream pathways involved in left-right determination, thus acting through the pathway but bypassing the asymmetric gene expression of nodal and lefty genes. Alternatively, the decreased Pitx2 gene expression may be due to interference in its intronic enhancer binding function. Although Pitx2 is induced by Nodal (which is transiently expressed), its expression is maintained long after Nodal by binding of its intronic enhancer ASE with enhancers, including Nkx2 and Foxh1 (Shiratori et al., 2001, 2006). It is possible that HCN4 inhibition may interfere with this enhancer binding, leading to decreased Pitx2 gene expression.
Previous studies have shown that Pitx2 plays two important roles during organogenesis; the left-sided expression in the lateral plate mesoderm is important for overall embryonic left-right determination, while the asymmetric (left side) expression within individual organ's tissues (particularly gut and heart) guides asymmetric morphogenesis of these organs (Campione et al., 1999; Davis et al., 2017). Studies (both in mouse and Xenopus) (Clauss and Kaab, 2011; Davis et al., 2017; Poelmann et al., 2008) have shown that eliminating asymmetric Pitx2 expression within organs like heart and gut disrupts asymmetric organ morphogenesis and also leads to disrupted organ morphologies. In our studies, the reduced (but correctly-sided) Pitx2 signal in the lateral plate mesoderm may be affecting the Pitx2 expression in the heart and gut organs. However, unlike the heart and gut Pitx2 expression studies, we do not see morphological abnormalities in organ patterning. The gut and heart are placed in (left-right orientation) either normal or revered orientation but with fully formed morphologies.
Taken together, these data suggest that HCN4 channel-mediated ion flux as part of the pathways that bypass Nodal-Lefty asymmetric gene expression (Fig. 6). To our knowledge, there is only one other report of such a reagent: the Mahogunin (ubiquitin ligase) mutant (Mgrn1-C314D) causes left-right perturbation that bypasses asymmetric Nodal gene expression in mouse (Cota et al., 2006) and bypasses Nodal-Lefty-Pitx2 axis asymmetric gene expression in Xenopus (McDowell et al., 2016b). It is not yet known whether Mahogunin and HCN4 are part of the same or different alternative pathways conferring robustness to laterality. It is also not yet known whether HCN4-mediated laterality pathways are present in other animals.
Fig. 6.
Model for HCN4 function in establishing laterality. The developmental timeline along the left illustrates early cleavage stages to post-gastrulation asymmetric gene expression of the Nodal-Lefty-Pitx2 cascade leading to final organ situs. Previously established important laterality events are outlined adjacent to the developmental time line and portray very early laterality events of cytoskeletal rearrangement and physiological amplification, as well as later events such as gastrulation-stage ciliary flows, all funnel into the canonical Nodal-Lefty-Pitx2 gene regulatory network to bring about invariant asymmetric organ situs. HCN4 action is required during early cleavage stages and can largely bypass the Nodal-Lefty asymmetric gene expression cascade to affect organ situs. HCN4-mediated Nodal-Lefty asymmetric gene expression-independent effect could be due to directly acting on downstream factors of the Nodal-Lefty pathway or through a non-canonical pathway. Players in this HCN4-mediated laterality patterning remain to be discovered.
HCN4 channel: downstream consequences of its inhibition
Situs inversus is the most frequent phenotype observed upon pharmacologically blocking HCN4 during early embryogenesis (Fig. 1C). In contrast, HCN4-DN mRNA injection-mediated HCN4 block (Fig. 2C) resulted in a wider spectrum of defects. This difference is most likely because injected HCN4-DN mRNA persists long beyond stage 9 and this effect on organ morphology may be due to HCN4-DN action in laterality overlapping with later organ morphogenesis steps (Pitcairn et al., 2017).
Left-right asymmetry establishment can be broadly categorized into three steps: (1) symmetry breaking, (2) orientation of the axes, and (3) amplification steps (Levin, 2005, 2006; Vandenberg et al., 2011). Disrupting each of these three steps is specifically predicted to have three different outcomes on organ situs. Disrupting the first step of symmetry breaking (cytoskeletal chirality, the directional action of motor proteins and transport) will cause the right and left halves of the embryos to be mirror-images of each other (isomerism), often observed in mice but rarely observed in Xenopus (Levin, 2005, 2006; Vandenberg et al., 2011). Disrupting the second step of the orientation of left-right axis with respect to anterior-posterior and dorso-ventral axis (which include asymmetric ion translocators and ion fluxes) will lead to a majority of individuals with complete reversal of asymmetric organs (situs inversus), as the left-right axis is randomly oriented with respect to the anterior-posterior and dorso-ventral axis (Levin, 2005, 2006; Vandenberg et al., 2011). Lastly, disruption of the third amplification step (which includes gap-junction communication, serotonin transport, ciliary flow, and Nodal-lefty-Pitx2 cascade) will result in each organ making an independent decision leading to predominantly heterotaxic individuals.
Interestingly, blocking the HCN4 channel early leads to a major incidence of situs inversus and a small incidence of the bilateral gut (isomerism) (Figs 1 and 2); it should be noted that gut isomerism is extremely rare in the extensive Xenopus literature on left-right-randomizing treatments. This is consistent with HCN4 acting at the level of the second step of orientation of left-right axis in relation to the other two (anterior-posterior and dorso-ventral axes). This is further supported by the observation that HCN4 channels are present from early cleavage stages, and their role in laterality seems to be executed during early cleavage stages. Similar to HCN4, disrupting cytoskeletal dynamics causes a major incidence of situs inversus (Vandenberg et al., 2011). It is not yet clear whether cytoskeletal dynamics are upstream of HCN4 or if they are part of two independent mechanisms. The later is likely the case as we do not see consistent asymmetry in the distribution of HCN4 channel within the early embryo. What then could be upstream of HCN4 that is causing them to act physiologically in a different manner? A recent landmark set of studies showed that, as early as 8-cell stage, the left and right blastomeres are metabolically very different (Onjiko et al., 2016). It is possible that metabolic differences among blastomeres at the 2-cell stage lead to different physiology of HCN4 channel function.
The timing and action of HCN4 channels in development: a very early role
Here we have identified a novel role of HCN4 channels during embryonic left-right patterning. Many previous reports indicate a critical role of ion translocators and ion fluxes in determining embryonic left-right asymmetry (Adams et al., 2006; Aw et al., 2008, 2010; Levin et al., 2002; Morokuma et al., 2008). In particular, asymmetric functions of H+/K+-ATPase and V-ATPase (Hibino et al., 2006; Kawakami et al., 2005; Levin et al., 2002; Shimeld and Levin, 2006), as well as two other potassium channels (KCNQ1 and KATP) have been implicated. For the majority of these ion translocators, their asymmetric localization and action is post 4-cell stage, but before gastrulation. Crucially, HCN4 inhibition affects asymmetry during early embryogenesis – an observation that is incompatible with potential hypotheses about roles in regulating much later events like ciliary motion at gastrulation (Basu and Brueckner, 2008) since all of those events would be targeted by inhibitor exposure starting at stage 10. The same is true of many other highly-conserved elements of the left-right symmetry breaking machinery, such as cytoskeletal proteins (Davison et al., 2016; Lobikin et al., 2012), and reinforces the importance of focusing on intracellular, biophysical events as the earliest components of left-right pattering.
We found that HCN4 channels are already present in 2-cell embryos (most likely maternally loaded) and are uniformly expressed throughout the embryo all the way through gastrulation (Fig. 3). Hence, the action of HCN4 in establishing laterality is most likely at the physiological (post-translational gating) level of its function. This is not unprecedented, as in zebrafish the H+/K+-ATPase is uniformly expressed throughout the embryo and still is involved in laterality determination by its actions at the physiological level (Kawakami et al., 2005). Moreover, HCN4 is gated by a number of ligands (e.g. cAMP) which can be differentially localized to result in differential bioelectrical activity even if HCN4 protein is ubiquitous. Future studies using fluorescent reporters of cAMP and individual ion concentrations (being developed by a number of groups but not yet available in Xenopus), as well as transgenic Xenopus in which native HCN4 is labeled with a fluorescent tag, will dissect the very early steps of HCN4 activity. New techniques for introducing material into Xenopus eggs prior to fertilization may also be useful in manipulating this process, since injections even at 1-cell stage may be attenuated in their effects by the amount of time needed to make protein from the mRNA introduced then.
Conclusion
Establishing invariant laterality is a fundamental aspect of most life forms across the tree of life. It is becoming clear that the mechanisms of establishing and executing laterality are redundant and highly robust to ensure correct organ situs even in presence of certain errors in the pathway. Many fascinating questions remain about the physiological processes that transmit and amplify physical chirality of intracellular cytoskeletal structures into embryo-wide programs of gene expression, and ultimately to the consistent asymmetry of organogenesis. The characterization of a novel player, the HCN4 channel, provides a new entry point into pathways which act very early during embryogenesis and then bypass the canonical asymmetric gene expression cascade of nodal-lefty-pitx2 to exert their effects much later. Moreover, the discovery of new ion channels that underlie the endogenous bioelectric signaling that is increasingly seen to be an important component of developmental (Bates, 2015; Levin, 2012; Levin and Stevenson, 2012) and regenerative (Chifflet et al., 2005; Levin, 2014b; Wang and Zhao, 2010) patterning, adds to the toolbox of available targets for understanding and control of growth and form. The investigation of the dynamic interplay between early bioelectrics, subsequent transcriptional regulation, and resultant anatomical patterning presents exciting opportunities for understanding developmental and evolutionary dynamics. It is also possible that the study of compensatory redundant pathways will reveal new approaches for harnessing the robustness of developmental mechanisms for regenerative medicine.
MATERIALS AND METHODS
Animal husbandry
Xenopus laevis embryos were fertilized in vitro according to standard protocols (Sive et al., 2000) in 0.1× Marc's Modified Ringer's (MMR; 10 mM Na+, 0.2 mM K+, 10.5 mM Cl−, 0.2 mM Ca2+, pH 7.8). Xenopus embryos were housed at 14-18°C (14°C overnight after injection and subsequently at 18°C), except during drug exposure which was at 22°C, and staged according to Nieuwkoop and Faber (1967). All experiments were approved by the Tufts University Animal Research Committee (M2014-79) in accordance with the guide for care and use of laboratory animals.
Microinjections
Capped synthetic mRNAs generated using mMessage mMachine kit (Ambion) were dissolved in nuclease free water and injected into embryos immersed in 3% Ficoll using standard methods (Sive et al., 2000). Each injection delivered between ∼0.5-1 nl (0.5-1 ng) of mRNA (per blastomere) into the embryos, at the indicated stages into the middle of the cell in the animal pole. HCN4-DN was a mammalian (mouse) HCN4, modified as per Pitcairn et al. (2017). Briefly, a standard approach was used for generating dominant-negative channel subunit (Kuzhikandathil and Oxford, 2000; Preisig-Muller et al., 2002; Xue et al., 2002). The HCN4 channel function was abolished by altering the highly conserved cation selective sequence in the pore domain (changed from GYG349-351 to AAA349-351) to generate HCN4-(AAA)-DN mutant from HCN4-WT using primers: 5′CACATGCTGTGCATTGAGGACGAACGTCAGGCA-3′ (forward) and 5'-TGCCTGACGTTCGTCCTCAATGCACAGCATGTG-3′ (reverse). The construct was subcloned into a pCS2 vector to transcribe into mRNAs for Xenopus microinjections.
Anatomical laterality assays
Xenopus embryos were analyzed as in Levin and Mercola (1998) for position (situs) of three organs: heart, gut and gallbladder at stage 45 (Nieuwkoop and Faber, 1967) using fiber light illumination from the ventral side. Heterotaxic embryos were defined as ones having a reversal in one or more organs. Treatments were titered to levels that gave rise to >90% embryos with normal dorso-anterior development and correctly-formed organs. All reported left-right inversions are embryos with clear (unambiguous) left-right organ situs. It is important to note that our analysis is extremely stringent – it underestimates the overall effect of any given treatment. Even if 100% of embryos are affected by some manipulation, the maximum observable effect will still be capped at 87.5% as some percentage of embryos will have all three organs randomly land in correct orientation making them indistinguishable from the wild type and hence will be scored as normal.
Drug exposure
Xenopus embryos were incubated in pharmacological blocker of HCN4 channel ZD7288 (Tocris biosciences) (100 mM stock solution in water) dissolved in 0.1× MMR (final concentration 100 µM) during the stages indicated in respective experiments followed by several washes with 0.1× MMR. Note that these experiments were performed at 22°C since ZD7288 effect on embryonic left-right asymmetry was found stronger at 22°C and not at 14°C and 18°C (data not shown) (Yanagida et al., 2000). Untreated embryos reared at 22°C served as controls.
Imaging Vmem using CC2-DMPE: DiBAC4(3)
CC2-DMPE and DiBAC4(3) voltage reporter dyes were obtained from Invitrogen and used as per the standard protocol, including dark-field and flat-field correction (Adams and Levin, 2012). Briefly, the use of two dyes with opposite emission profiles simultaneously provides an internal control and allows ratiometric normalization. CC2-DMPE stock (5 mM) was dissolved 1:1000 in 0.1× MMR and the embryos were incubated in dark in this solution for at least 1 h followed by washes with 0.1× MMR. DiBAC4(3) stock (1.9 mM) was dissolved 1:4000 in 0.1× MMR and the CC2-DMPE-stained embryos were then incubated in dark in this solution for at least 30 min followed by visualization under the microscope. An Olympus BX-61 microscope equipped with a Hamamatsu ORCA AG CCD camera, and controlled by Metamorph software (Molecular Devices), was used to collect signal. NIH Image J software was used to quantify the fluorescence intensities of the CC2-DMPE:DiBAC signal.
Intracellular recordings from embryo cells
Membrane potentials were measured using an oocyte clamp OC-725C amplifier (Warner Instruments, Hamden, CT, USA) with a single voltage electrode. Microelectrodes were made from thin-walled borosilicate glass pulled with a flaming/brown micropipette puller (p-97, Sutter Instruments, Novato, CA, USA) and back filled with electrode solution (2 M potassium acetate, 10 mM KCl, 5 mM HEPES pH 7.5). Tip resistances were 80-100 MΩ. Electrode penetration of ectodermal cells was by visual guidance on a fixed-stage microscope (Zeiss) using a three-axis micromanipulator.
Immunofluorescence
Spatial distribution of HCN4 channel in embryos was detected by immunofluorescence for the HCN4 channel on whole embryos. Briefly, embryos were fixed overnight in MEMFA at 4°C (Sive et al., 2000). The embryos were permeabilized in PBS 0.1%Triton-X-100, blocked with 10% goat serum in PBST for 1 h at room temperature, and incubated at 4°C overnight with primary antibody (Anti-HCN4 – rabbit polyclonal; Abcam ab66501) for HCN4 at 1:500 dilution in PBST+10% goat serum (blocking buffer). Embryos were washed six times in PBST and incubated with Alexa Fluor-conjugated fluorescent secondary antibody (Invitrogen) at 1:500 dilution in PBST+10% goat serum overnight at 4°C. Embryos were washed six times in PBST and photographed using Nikon SMZ-1500 scope with Q-capture software.
In situ hybridization
Xenopus embryos were collected and fixed in MEMFA (Sive et al., 2000) and in situ hybridization was performed as previously described (Harland, 1991; Sive et al., 2000). The embryos were washed with phosphate buffered saline 0.1% Tween-20 (PBST) and transferred through series of methanol washes (25%, 50%, 75%, 100%). In situ anti-sense probes were generated in vitro from linearized templates using a DIG labeling mix (Roche). Chromogenic reaction times were optimized for signal to background ratio. Probes used were: Xnodal (Sampath et al., 1997), Xlefty (Meno et al., 1997), and Xpitx2 (Campione et al., 1999). NIH Image J software was used to quantify the in situ signal.
Statistics
All statistical analysis was performed using Microsoft Excel. As appropriate for each case, data were either pooled from multiple repeat experiments, with χ2 analysis performed on them, or data from various iterations was analyzed by t-test (for 2 groups) or ANOVA (for more than two groups), as indicated with each experiment.
Acknowledgements
This work is dedicated to the memory of Amar Klar. We thank Erin Switzer, and Rakela Colon for Xenopus husbandry and general lab assistance, Dany Adams for help with microscopy, Chris Wright for Nodal and Lefty antisense probe, and H. Joseph Yost for Pitx2 antisense probes.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
Conceptualization: M.L., V.P.P., K.A.M.; Methodology: V.P.P., E.J.P., J.M.L., J.-F.P.; Investigation: V.P.P., V.W., E.J.P., J.M.L., J.-F.P., N.-Q.S.; Writing - original draft: V.P.P.; Writing - review & editing: M.L., V.P.P., E.J.P., J.M.L., J.-F.P., K.A.M.; Supervision: M.L., K.A.M.; Project administration: M.L., K.A.M.; Funding acquisition: M.L.
Funding
We gratefully acknowledge support of the John Templeton Foundation (TWCF0089/AB55), and an Allen Discovery Center award from The Paul G. Allen Family Foundation (12171).
Supplementary information
Supplementary information available online at http://bio.biologists.org/lookup/doi/10.1242/bio.025957.supplemental
This article has an associated First Person interview with the first author(s) of the paper available online at http://bio.biologists.org/lookup/doi/10.1242/bio.025957.supplemental.
References
- Adams D. S. and Levin M. (2012). Measuring resting membrane potential using the fluorescent voltage reporters DiBAC4(3) and CC2-DMPE. Cold Spring Harb. Protoc. 2012, 459-464. 10.1101/pdb.prot067702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adams D. S. and Levin M. (2013). Endogenous voltage gradients as mediators of cell-cell communication: strategies for investigating bioelectrical signals during pattern formation. Cell Tissue Res. 352, 95-122. 10.1007/s00441-012-1329-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adams D. S., Robinson K. R., Fukumoto T., Yuan S., Albertson R. C., Yelick P., Kuo L., McSweeney M. and Levin M. (2006). Early, H+-V-ATPase-dependent proton flux is necessary for consistent left-right patterning of non-mammalian vertebrates. Development 133, 1657-1671. 10.1242/dev.02341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aw S., Adams D. S., Qiu D. and Levin M. (2008). H,K-ATPase protein localization and Kir4.1 function reveal concordance of three axes during early determination of left-right asymmetry. Mech. Dev. 125, 353-372. 10.1016/j.mod.2007.10.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aw S., Koster J. C., Pearson W., Nichols C. G., Shi N.-Q., Carneiro K. and Levin M. (2010). The ATP-sensitive K(+)-channel (K(ATP)) controls early left-right patterning in Xenopus and chick embryos. Dev. Biol. 346, 39-53. 10.1016/j.ydbio.2010.07.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basu B. and Brueckner M. (2008). Cilia multifunctional organelles at the center of vertebrate left-right asymmetry. Curr. Top. Dev. Biol. 85, 151-174. 10.1016/S0070-2153(08)00806-5 [DOI] [PubMed] [Google Scholar]
- Bates E. (2015). Ion channels in development and cancer. Annu. Rev. Cell Dev. Biol. 31, 231-247. 10.1146/annurev-cellbio-100814-125338 [DOI] [PubMed] [Google Scholar]
- Bessodes N., Haillot E., Duboc V., Röttinger E., Lahaye F. and Lepage T. (2012). Reciprocal signaling between the ectoderm and a mesendodermal left-right organizer directs left-right determination in the sea urchin embryo. PLoS Genet. 8, e1003121 10.1371/journal.pgen.1003121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biel M., Wahl-Schott C., Michalakis S. and Zong X. (2009). Hyperpolarization-activated cation channels: from genes to function. Physiol. Rev. 89, 847-885. 10.1152/physrev.00029.2008 [DOI] [PubMed] [Google Scholar]
- Burn J. (1991). Disturbance of morphological laterality in humans. Ciba Found Symp. 162, 282-296; discussion 296-289. [DOI] [PubMed] [Google Scholar]
- Campione M., Steinbeisser H., Schweickert A., Deissler K., van Bebber F., Lowe L. A., Nowotschin S., Viebahn C., Haffter P., Kuehn M. R. et al. (1999). The homeobox gene Pitx2: mediator of asymmetric left-right signaling in vertebrate heart and gut looping. Development 126, 1225-1234. [DOI] [PubMed] [Google Scholar]
- Cerbai E., Pino R., Sartiani L. and Mugelli A. (1999). Influence of postnatal-development on If occurrence and properties in neonatal rat ventricular myocytes. Cardiovasc. Res. 42, 416-423. 10.1016/S0008-6363(99)00037-1 [DOI] [PubMed] [Google Scholar]
- Chen T.-H., Hsu J. J., Zhao X., Guo C., Wong M. N., Huang Y., Li Z., Garfinkel A., Ho C.-M., Tintut Y. et al. (2012). Left-right symmetry breaking in tissue morphogenesis via cytoskeletal mechanics. Circ. Res. 110, 551-559. 10.1161/CIRCRESAHA.111.255927 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chifflet S., Hernandez J. A. and Grasso S. (2005). A possible role for membrane depolarization in epithelial wound healing. Am. J. Physiol. Cell Physiol. 288, C1420-C1430. 10.1152/ajpcell.00259.2004 [DOI] [PubMed] [Google Scholar]
- Christoffels V. M., Smits G. J., Kispert A. and Moorman A. F. M. (2010). Development of the pacemaker tissues of the heart. Circ. Res. 106, 240-254. 10.1161/CIRCRESAHA.109.205419 [DOI] [PubMed] [Google Scholar]
- Clauss S. and Kaab S. (2011). Is Pitx2 growing up? Circ. Cardiovasc. Genet. 4, 105-107. 10.1161/CIRCGENETICS.111.959791 [DOI] [PubMed] [Google Scholar]
- Cohen M. S., Anderson R. H., Cohen M. I., Atz A. M., Fogel M., Gruber P. J., Lopez L., Rome J. J. and Weinberg P. M. (2007). Controversies, genetics, diagnostic assessment, and outcomes relating to the heterotaxy syndrome. Cardiol. Young 17 Suppl. 2, 29-43. 10.1017/S104795110700114X [DOI] [PubMed] [Google Scholar]
- Cota C. D., Bagher P., Pelc P., Smith C. O., Bodner C. R. and Gunn T. M. (2006). Mice with mutations in Mahogunin ring finger-1 (Mgrn1) exhibit abnormal patterning of the left-right axis. Dev. Dyn. 235, 3438-3447. 10.1002/dvdy.20992 [DOI] [PubMed] [Google Scholar]
- Coutelis J. B., Petzoldt A. G., Spéder P., Suzanne M. and Noselli S. (2008). Left-right asymmetry in Drosophila. Semin. Cell Dev. Biol. 19, 252-262. 10.1016/j.semcdb.2008.01.006 [DOI] [PubMed] [Google Scholar]
- Davis A., Amin N. M., Johnson C., Bagley K., Ghashghaei H. T. and Nascone-Yoder N. (2017). Stomach curvature is generated by left-right asymmetric gut morphogenesis. Development 144, 1477-1483. 10.1242/dev.143701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davison A., McDowell G. S., Holden J. M., Johnson H. F., Koutsovoulos G. D., Liu M. M., Hulpiau P., Van Roy F., Wade C. M., Banerjee R. et al. (2016). Formin is associated with left-right asymmetry in the pond snail and the frog. Curr. Biol. 26, 654-660. 10.1016/j.cub.2015.12.071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dimonte A., Adamatzky A., Erokhin V. and Levin M. (2016). On chirality of slime mould. Biosystems 140, 23-27. 10.1016/j.biosystems.2015.12.008 [DOI] [PubMed] [Google Scholar]
- Fukumoto T., Blakely R. and Levin M. (2005a). Serotonin transporter function is an early step in left-right patterning in chick and frog embryos. Dev. Neurosci. 27, 349-363. 10.1159/000088451 [DOI] [PubMed] [Google Scholar]
- Fukumoto T., Kema I. P. and Levin M. (2005b). Serotonin signaling is a very early step in patterning of the left-right axis in chick and frog embryos. Curr. Biol. 15, 794-803. 10.1016/j.cub.2005.03.044 [DOI] [PubMed] [Google Scholar]
- Garic-Stankovic A., Hernandez M., Flentke G. R., Zile M. H. and Smith S. M. (2008). A ryanodine receptor-dependent Ca(i)(2+) asymmetry at Hensen's node mediates avian lateral identity. Development 135, 3271-3280. 10.1242/dev.018861 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Granados-Riveron J. T. and Brook J. D. (2012). The impact of mechanical forces in heart morphogenesis. Circ. Cardiovasc. Genet. 5, 132-142. 10.1161/CIRCGENETICS.111.961086 [DOI] [PubMed] [Google Scholar]
- Gros J., Feistel K., Viebahn C., Blum M. and Tabin C. J. (2009). Cell movements at Hensen's node establish left/right asymmetric gene expression in the chick. Science 324, 941-944. 10.1126/science.1172478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harland R. M. (1991). In situ hybridization: an improved whole mount method for Xenopus embryos. In Xenopus laevis: Practical Uses in Cell and Molecular Biology (ed. Kay B. K. and Peng H. B.), pp. 685-695. San Diego: Academic Press. [DOI] [PubMed] [Google Scholar]
- Hashimoto T. (2002). Molecular genetic analysis of left-right handedness in plants. Philos. Trans. R. Soc. Lond. B Biol. Sci. 357, 799-808. 10.1098/rstb.2002.1088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hibino T., Ishii Y., Levin M. and Nishino A. (2006). Ion flow regulates left-right asymmetry in sea urchin development. Dev. Genes Evol. 216, 265-276. 10.1007/s00427-005-0051-6 [DOI] [PubMed] [Google Scholar]
- Hoffman J. I. E. and Kaplan S. (2002). The incidence of congenital heart disease. J. Am. Coll. Cardiol. 39, 1890-1900. 10.1016/S0735-1097(02)01886-7 [DOI] [PubMed] [Google Scholar]
- Kawakami Y., Raya A., Raya R. M., Rodríguez-Esteban C. and Izpisua Belmonte J. C. (2005). Retinoic acid signalling links left-right asymmetric patterning and bilaterally symmetric somitogenesis in the zebrafish embryo. Nature 435, 165-171. 10.1038/nature03512 [DOI] [PubMed] [Google Scholar]
- Kuroda R., Endo B., Abe M. and Shimizu M. (2009). Chiral blastomere arrangement dictates zygotic left-right asymmetry pathway in snails. Nature 462, 790-794. 10.1038/nature08597 [DOI] [PubMed] [Google Scholar]
- Kuzhikandathil E. V. and Oxford G. S. (2000). Dominant-negative mutants identify a role for GIRK channels in D3 dopamine receptor-mediated regulation of spontaneous secretory activity. J. Gen. Physiol. 115, 697-706. 10.1085/jgp.115.6.697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M. (1998). Left-right asymmetry and the chick embryo. Semin. Cell Dev. Biol. 9, 67-76. 10.1006/scdb.1997.0192 [DOI] [PubMed] [Google Scholar]
- Levin M. (2005). Left-right asymmetry in embryonic development: a comprehensive review. Mech. Dev. 122, 3-25. 10.1016/j.mod.2004.08.006 [DOI] [PubMed] [Google Scholar]
- Levin M. (2006). Is the early left-right axis like a plant, a kidney, or a neuron? The integration of physiological signals in embryonic asymmetry. Birth Defects Res. C Embryo Today 78, 191-223. 10.1002/bdrc.20078 [DOI] [PubMed] [Google Scholar]
- Levin M. (2012). Molecular bioelectricity in developmental biology: new tools and recent discoveries: control of cell behavior and pattern formation by transmembrane potential gradients. BioEssays 34, 205-217. 10.1002/bies.201100136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M. (2013). Reprogramming cells and tissue patterning via bioelectrical pathways: molecular mechanisms and biomedical opportunities. Wiley Interdiscipl. Rev. Syst. Biol. Med. 5, 657-676. 10.1002/wsbm.1236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M. (2014a). Endogenous bioelectrical networks store non-genetic patterning information during development and regeneration. J. Physiol. 592, 2295-2305. 10.1113/jphysiol.2014.271940 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M. (2014b). Molecular bioelectricity: how endogenous voltage potentials control cell behavior and instruct pattern regulation in vivo. Mol. Biol. Cell 25, 3835-3850. 10.1091/mbc.E13-12-0708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M. and Mercola M. (1998). Gap junctions are involved in the early generation of left-right asymmetry. Dev. Biol. 203, 90-105. 10.1006/dbio.1998.9024 [DOI] [PubMed] [Google Scholar]
- Levin M. and Nascone N. (1997). Two molecular models of initial left-right asymmetry generation. Med. Hypotheses 49, 429-435. 10.1016/S0306-9877(97)90092-X [DOI] [PubMed] [Google Scholar]
- Levin M. and Palmer A. R. (2007). Left-right patterning from the inside out: widespread evidence for intracellular control. BioEssays 29, 271-287. 10.1002/bies.20545 [DOI] [PubMed] [Google Scholar]
- Levin M. and Stevenson C. G. (2012). Regulation of cell behavior and tissue patterning by bioelectrical signals: challenges and opportunities for biomedical engineering. Annu. Rev. Biomed. Eng. 14, 295-323. 10.1146/annurev-bioeng-071811-150114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M., Thorlin T., Robinson K. R., Nogi T. and Mercola M. (2002). Asymmetries in H+/K+-ATPase and cell membrane potentials comprise a very early step in left-right patterning. Cell 111, 77-89. 10.1016/S0092-8674(02)00939-X [DOI] [PubMed] [Google Scholar]
- Levin M., Buznikov G. A. and Lauder J. M. (2006). Of minds and embryos: left-right asymmetry and the serotonergic controls of pre-neural morphogenesis. Dev. Neurosci. 28, 171-185. 10.1159/000091915 [DOI] [PubMed] [Google Scholar]
- Lobikin M., Wang G., Xu J., Hsieh Y.-W., Chuang C.-F., Lemire J. M. and Levin M. (2012). Early, nonciliary role for microtubule proteins in left-right patterning is conserved across kingdoms. Proc. Natl. Acad. Sci. USA 109, 12586-12591. 10.1073/pnas.1202659109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDowell G., Rajadurai S. and Levin M. (2016a). From cytoskeletal dynamics to organ asymmetry: a nonlinear, regulative pathway underlies left-right patterning. Philos. Trans. R. Soc. Lond. B Biol. Sci. 371, 20150409 10.1098/rstb.2015.0409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDowell G. S., Lemire J. M., Paré J.-F., Cammarata G., Lowery L. A. and Levin M. (2016b). Conserved roles for cytoskeletal components in determining laterality. Integr. Biol. 8, 267-286. 10.1039/C5IB00281H [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meno C., Ito Y., Saijoh Y., Matsuda Y., Tashiro K., Kuhara S. and Hamada H. (1997). Two closely-related left-right asymmetrically expressed genes, lefty-1 and lefty-2: their distinct expression domains, chromosomal linkage and direct neuralizing activity in Xenopus embryos. Genes Cells 2, 513-524. 10.1046/j.1365-2443.1997.1400338.x [DOI] [PubMed] [Google Scholar]
- Mercola M. and Levin M. (2001). Left-right asymmetry determination in vertebrates. Annu. Rev. Cell Dev. Biol. 17, 779-805. 10.1146/annurev.cellbio.17.1.779 [DOI] [PubMed] [Google Scholar]
- Morokuma J., Blackiston D. and Levin M. (2008). KCNQ1 and KCNE1 K+ channel components are involved in early left-right patterning in Xenopus laevis embryos. Cell. Physiol. Biochem. 21, 357-372. 10.1159/000129628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naganathan S. R., Fürthauer S., Nishikawa M., Jülicher F. and Grill S. W. (2014). Active torque generation by the actomyosin cell cortex drives left-right symmetry breaking. Elife 3, e04165 10.7554/eLife.04165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naganathan S. R., Middelkoop T. C., Fürthauer S. and Grill S. W. (2016). Actomyosin-driven left-right asymmetry: from molecular torques to chiral self organization. Curr. Opin. Cell Biol. 38, 24-30. 10.1016/j.ceb.2016.01.004 [DOI] [PubMed] [Google Scholar]
- Nakamura T. and Hamada H. (2012). Left-right patterning: conserved and divergent mechanisms. Development 139, 3257-3262. 10.1242/dev.061606 [DOI] [PubMed] [Google Scholar]
- Nieuwkoop P. D. and Faber J. (1967). Normal Table of Xenopus laevis (Daudin) 2nd edn. Amsterdam: North-Holland Publishing Company. [Google Scholar]
- Okumura T., Utsuno H., Kuroda J., Gittenberger E., Asami T. and Matsuno K. (2008). The development and evolution of left-right asymmetry in invertebrates: lessons from Drosophila and snails. Dev. Dyn. 237, 3497-3515. 10.1002/dvdy.21788 [DOI] [PubMed] [Google Scholar]
- Onjiko R. M., Morris S. E., Moody S. A. and Nemes P. (2016). Single-cell mass spectrometry with multi-solvent extraction identifies metabolic differences between left and right blastomeres in the 8-cell frog (Xenopus) embryo. Analyst 141, 3648-3656. 10.1039/C6AN00200E [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oviedo N. J. and Levin M. (2007). Gap junctions provide new links in left-right patterning. Cell 129, 645-647. 10.1016/j.cell.2007.05.005 [DOI] [PubMed] [Google Scholar]
- Pai V. P., Aw S., Shomrat T., Lemire J. M. and Levin M. (2012). Transmembrane voltage potential controls embryonic eye patterning in Xenopus laevis. Development 139, 313-323. 10.1242/dev.073759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pai V. P., Lemire J. M., Pare J.-F., Lin G., Chen Y. and Levin M. (2015). Endogenous gradients of resting potential instructively pattern embryonic neural tissue via notch signaling and regulation of proliferation. J. Neurosci. 35, 4366-4385. 10.1523/JNEUROSCI.1877-14.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peeters H. and Devriendt K. (2006). Human laterality disorders. Eur. J. Med. Genet. 49, 349-362. 10.1016/j.ejmg.2005.12.003 [DOI] [PubMed] [Google Scholar]
- Petzoldt A. G., Coutelis J.-B., Geminard C., Speder P., Suzanne M., Cerezo D. and Noselli S. (2012). DE-Cadherin regulates unconventional Myosin ID and Myosin IC in Drosophila left-right asymmetry establishment. Development 139, 1874-1884. 10.1242/dev.047589 [DOI] [PubMed] [Google Scholar]
- Pietak A. and Levin M. (2016). Exploring instructive physiological signaling with the Bioelectric Tissue Simulation Engine (BETSE). Front. Bioeng. Biotechnol. 4, 55. 10.3389/fbioe.2016.00055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitcairn E., Harris H., Epiney J., Pai V. P., Lemire J. M., Ye B., Shi N.-Q., Levin M. and McLaughlin K. A. (2017). Coordinating heart morphogenesis: a novel role for hyperpolarization-activated cyclic nucleotide-gated (HCN) channels during cardiogenesis in Xenopus laevis. Commun. Integr. Biol. 10, e1309488 10.1080/19420889.2017.1309488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poelmann R. E., Jongbloed M. R. M. and Gittenberger-de Groot A. C. (2008). Pitx2: a challenging teenager. Circ. Res. 102, 749-751. 10.1161/CIRCRESAHA.108.174847 [DOI] [PubMed] [Google Scholar]
- Pohl C. (2011). Left-right patterning in the C. elegans embryo: unique mechanisms and common principles. Commun. Integr. Biol. 4, 34-40. 10.4161/cib.14144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preisig-Muller R., Schlichthorl G., Goerge T., Heinen S., Bruggemann A., Rajan S., Derst C., Veh R. W. and Daut J. (2002). Heteromerization of Kir2.x potassium channels contributes to the phenotype of Andersen's syndrome. Proc. Natl. Acad. Sci. USA 99, 7774-7779. 10.1073/pnas.102609499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu D., Cheng S.-M., Wozniak L., McSweeney M., Perrone E. and Levin M. (2005). Localization and loss-of-function implicates ciliary proteins in early, cytoplasmic roles in left-right asymmetry. Dev. Dyn. 234, 176-189. 10.1002/dvdy.20509 [DOI] [PubMed] [Google Scholar]
- Qu Y., Whitaker G. M., Hove-Madsen L., Tibbits G. F. and Accili E. A. (2008). Hyperpolarization-activated cyclic nucleotide-modulated ‘HCN’ channels confer regular and faster rhythmicity to beating mouse embryonic stem cells. J. Physiol. 586, 701-716. 10.1113/jphysiol.2007.144329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramsdell A. F. (2005). Left-right asymmetry and congenital cardiac defects: getting to the heart of the matter in vertebrate left-right axis determination. Dev. Biol. 288, 1-20. 10.1016/j.ydbio.2005.07.038 [DOI] [PubMed] [Google Scholar]
- Ramsdell A. F. and Yost H. J. (1998). Molecular mechanisms of vertebrate left-right development. Trends Genet. 14, 459-465. 10.1016/S0168-9525(98)01599-6 [DOI] [PubMed] [Google Scholar]
- Raya A. and Izpisua Belmonte J. C. (2004a). Sequential transfer of left-right information during vertebrate embryo development. Curr. Opin. Genet. Dev. 14, 575-581. 10.1016/j.gde.2004.07.011 [DOI] [PubMed] [Google Scholar]
- Raya A. and Izpisua Belmonte J. C. (2004b). Unveiling the establishment of left-right asymmetry in the chick embryo. Mech. Dev. 121, 1043-1054. 10.1016/j.mod.2004.05.005 [DOI] [PubMed] [Google Scholar]
- Raya A. and Izpisua Belmonte J. C. (2006). Left-right asymmetry in the vertebrate embryo: from early information to higher-level integration. Nat. Rev. Genet. 7, 283-293. 10.1038/nrg1830 [DOI] [PubMed] [Google Scholar]
- Robinson R. B., Yu H., Chang F. and Cohen I. S. (1997). Developmental change in the voltage-dependence of the pacemaker current, if, in rat ventricle cells. Pflügers Archiv. 433, 533-535. 10.1007/s004240050309 [DOI] [PubMed] [Google Scholar]
- Sampath K., Cheng A. M., Frisch A. and Wright C. V. (1997). Functional differences among Xenopus nodal-related genes in left-right axis determination. Development 124, 3293-3302. [DOI] [PubMed] [Google Scholar]
- Schweickert A., Weber T., Beyer T., Vick P., Bogusch S., Feistel K. and Blum M. (2007). Cilia-driven leftward flow determines laterality in Xenopus. Curr. Biol. 17, 60-66. 10.1016/j.cub.2006.10.067 [DOI] [PubMed] [Google Scholar]
- Scicchitano P., Carbonara S., Ricci G., Mandurino C., Locorotondo M., Bulzis G., Gesualdo M., Zito A., Carbonara R., Dentamaro I. et al. (2012). HCN channels and heart rate. Molecules 17, 4225-4235. 10.3390/molecules17044225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimeld S. M. and Levin M. (2006). Evidence for the regulation of left-right asymmetry in Ciona intestinalis by ion flux. Dev. Dyn. 235, 1543-1553. 10.1002/dvdy.20792 [DOI] [PubMed] [Google Scholar]
- Shin K. S., Rothberg B. S. and Yellen G. (2001). Blocker state dependence and trapping in hyperpolarization-activated cation channels: evidence for an intracellular activation gate. J. Gen. Physiol. 117, 91-101. 10.1085/jgp.117.2.91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiratori H., Sakuma R., Watanabe M., Hashiguchi H., Mochida K., Sakai Y., Nishino J., Saijoh Y., Whitman M. and Hamada H. (2001). Two-step regulation of left-right asymmetric expression of Pitx2: initiation by nodal signaling and maintenance by Nkx2. Mol. Cell 7, 137-149. 10.1016/S1097-2765(01)00162-9 [DOI] [PubMed] [Google Scholar]
- Shiratori H., Yashiro K., Shen M. M. and Hamada H. (2006). Conserved regulation and role of Pitx2 in situs-specific morphogenesis of visceral organs. Development 133, 3015-3025. 10.1242/dev.02470 [DOI] [PubMed] [Google Scholar]
- Sive H. L., Grainger R. M. and Harland R. M. (2000). Early Development of Xenopus Laevis. New York: Cold Spring Harbor Laboratory Press. [Google Scholar]
- Später D., Abramczuk M. K., Buac K., Zangi L., Stachel M. W., Clarke J., Sahara M., Ludwig A. and Chien K. R. (2013). A HCN4+ cardiomyogenic progenitor derived from the first heart field and human pluripotent stem cells. Nat. Cell Biol. 15, 1098-1106. 10.1038/ncb2824 [DOI] [PubMed] [Google Scholar]
- Spéder P., Petzoldt A., Suzanne M. and Noselli S. (2007). Strategies to establish left/right asymmetry in vertebrates and invertebrates. Curr. Opin. Genet. Dev. 17, 351-358. 10.1016/j.gde.2007.05.008 [DOI] [PubMed] [Google Scholar]
- Suzuki K., Miyazaki M., Takagi J., Itabashi T. and Ishiwata S. (2017). Spatial confinement of active microtubule networks induces large-scale rotational cytoplasmic flow. Proc. Natl. Acad. Sci. USA 114, 2922-2927. 10.1073/pnas.1616001114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tee Y. H., Shemesh T., Thiagarajan V., Hariadi R. F., Anderson K. L., Page C., Volkmann N., Hanein D., Sivaramakrishnan S., Kozlov M. M. et al. (2015). Cellular chirality arising from the self-organization of the actin cytoskeleton. Nat. Cell Biol. 17, 445-457. 10.1038/ncb3137 [DOI] [PubMed] [Google Scholar]
- Thitamadee S., Tuchihara K. and Hashimoto T. (2002). Microtubule basis for left-handed helical growth in Arabidopsis. Nature 417, 193-196. 10.1038/417193a [DOI] [PubMed] [Google Scholar]
- Vandenberg L. N. (2012). Laterality defects are influenced by timing of treatments and animal model. Differentiation 83, 26-37. 10.1016/j.diff.2011.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenberg L. N. and Levin M. (2013). A unified model for left-right asymmetry? Comparison and synthesis of molecular models of embryonic laterality. Dev. Biol. 379, 1-15. 10.1016/j.ydbio.2013.03.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenberg L. N., Pennarola B. W. and Levin M. (2011). Low frequency vibrations disrupt left-right patterning in the Xenopus embryo. PLoS ONE 6, e23306 10.1371/journal.pone.0023306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenberg L. N., Lemire J. M. and Levin M. (2013a). It's never too early to get it Right: a conserved role for the cytoskeleton in left-right asymmetry. Commun. Integr. Biol. 6, e27155 10.4161/cib.27155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenberg L. N., Lemire J. M. and Levin M. (2013b). Serotonin has early, cilia-independent roles in Xenopus left-right patterning. Dis. Model. Mech. 6, 261-268. 10.1242/dmm.010256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkerk A. O. and Wilders R. (2015). Pacemaker activity of the human sinoatrial node: an update on the effects of mutations in HCN4 on the hyperpolarization-activated current. Int. J. Mol. Sci. 16, 3071-3094. 10.3390/ijms16023071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vicente-Steijn R., Passier R., Wisse L. J., Schalij M. J., Poelmann R. E., Gittenberger-de Groot A. C. and Jongbloed M. R. M. (2011). Funny current channel HCN4 delineates the developing cardiac conduction system in chicken heart. Heart Rhythm 8, 1254-1263. 10.1016/j.hrthm.2011.03.043 [DOI] [PubMed] [Google Scholar]
- Voronov D. A., Alford P. W., Xu G. and Taber L. A. (2004). The role of mechanical forces in dextral rotation during cardiac looping in the chick embryo. Dev. Biol. 272, 339-350. 10.1016/j.ydbio.2004.04.033 [DOI] [PubMed] [Google Scholar]
- Wahl-Schott C. and Biel M. (2009). HCN channels: structure, cellular regulation and physiological function. Cell. Mol. Life Sci. 66, 470-494. 10.1007/s00018-008-8525-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan L. Q., Ronaldson K., Park M., Taylor G., Zhang Y., Gimble J. M. and Vunjak-Novakovic G. (2011). Micropatterned mammalian cells exhibit phenotype-specific left-right asymmetry. Proc. Natl. Acad. Sci. USA 108, 12295-12300. 10.1073/pnas.1103834108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang E. T. and Zhao M. (2010). Regulation of tissue repair and regeneration by electric fields. Chin. J. Traumatol. 13, 55-61. 10.3760/cma.j.issn.1008-1275.2010.01.011 [DOI] [PubMed] [Google Scholar]
- Wang J., Klysik E., Sood S., Johnson R. L., Wehrens X. H. T. and Martin J. F. (2010). Pitx2 prevents susceptibility to atrial arrhythmias by inhibiting left-sided pacemaker specification. Proc. Natl. Acad. Sci. USA 107, 9753-9758. 10.1073/pnas.0912585107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh I. C., Thomsen M., Gludish D. W., Alfonso-Parra C., Bai Y., Martin J. F. and Kurpios N. A. (2013). Integration of left-right Pitx2 transcription and Wnt signaling drives asymmetric gut morphogenesis via Daam2. Dev. Cell 26, 629-644. 10.1016/j.devcel.2013.07.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Van Keymeulen A., Wakida N. M., Carlton P., Berns M. W. and Bourne H. R. (2007). Polarity reveals intrinsic cell chirality. Proc. Natl. Acad. Sci. USA 104, 9296-9300. 10.1073/pnas.0703153104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue T., Marban E. and Li R. A. (2002). Dominant-negative suppression of HCN1- and HCN2-encoded pacemaker currents by an engineered HCN1 construct: insights into structure-function relationships and multimerization. Circ. Res. 90, 1267-1273. 10.1161/01.RES.0000024390.97889.C6 [DOI] [PubMed] [Google Scholar]
- Yanagida H., Inoue R., Tanaka M. and Ito Y. (2000). Temperature-sensitive gating of cation current in guinea pig ileal muscle activated by hyperpolarization. Am. J. Physiol. Cell Physiol. 278, C40-C48. [DOI] [PubMed] [Google Scholar]
- Yasui K., Liu W., Opthof T., Kada K., Lee J.-K., Kamiya K. and Kodama I. (2001). If current and spontaneous activity in mouse embryonic ventricular myocytes. Circ. Res. 88, 536-542. 10.1161/01.RES.88.5.536 [DOI] [PubMed] [Google Scholar]
- Yost H. J. (1990). Inhibition of proteoglycan synthesis eliminates left-right asymmetry in Xenopus laevis cardiac looping. Development 110, 865-874. [DOI] [PubMed] [Google Scholar]
- Yost H. J. (1991). Development of the left-right axis in amphibians. Ciba Found Symp. 162, 165-176; discussion 176-181. [DOI] [PubMed] [Google Scholar]