Abstract
Application of cytostatics in cancer patients’ chemotherapy results in a number of side effects, including the inhibition of various parts of hematopoiesis. Two sulfated polysaccharides, fucoidan from the seaweed Chordaria flagelliformis (PS-Fuc) and fucosylated chondroitin sulfate from the sea cucumber Massinium magnum (PS-FCS), were studied as stimulators of hematopoiesis after cyclophosphamide immunosuppression in mice. Recombinant granulocyte colony-stimulating factor (r G-CSF) was applied as a reference. Both tested polysaccharides PS-Fuc and PS-FCS have a similar activity to r G-CSF, causing pronounced neutropoiesis stimulation in animals with myelosuppression induced by cyclophosphamide (CPh). Moreover, these compounds are also capable to enhance thrombopoiesis and erythropoiesis. It should be noted that PS-FCS demonstrated a greater activity than r G-CSF. The results indicate the perspective of further studies of PS-Fuc and PS-FCS, since these compounds can be considered as potentially promising stimulators of hematopoiesis. Such drugs are in demand for the accompanying treatment of cancer patients who suffer from hematological toxicity during chemo and/or radiation therapy.
Keywords: granulocyte colony-stimulating factor, fucoidan, fucosylated chondroitin sulfate, hematopoiesis, immunosuppression, cyclophosphamide
1. Introduction
Chemotherapy of cancer patients with cytostatics results in a number of side effects, including hematopoietic toxicity. One of the widely applied chemotherapy drugs with a large spectrum of antitumor activity is cyclophosphamide (CPh). It is an alkylating agent used in the treatment of various forms of malignant neoplasms [1].
The main toxic effect of CPh is an acute inhibition of hematopoiesis, which manifests by the suppression of rapidly proliferating hematopoietic progenitor cells and results in the form of neutropenia. However, lymphopenia and erythropenia are also significant toxic side effects of CPh [2,3]. In clinical practice, cancer patients’ treatment with CPh and other cytotoxic agents is accompanied by marked thrombocytopenia, which was manifested by the inhibitory effect of CPh on later megakaryocyte progenitors [4].
At present, recombinant granulocyte colony-stimulating factor (r G-CSF) is applied to combat the most severe complication of chemotherapy-induced neutropenia. Currently, there are various r G-CSF dosage forms that are used to treat neutropenia caused by chemo- and/or radiotherapy and to stimulate the recovery of neutrophils after bone marrow transplantation in cancer patients [5]. For preclinical evaluation of neutropoiesis stimulation efficiency under the action of r G-CSF, small laboratory animals (mice and rats) with induced myelosuppression are used [6]. Experimental studies in mice with induced myelosuppression have shown that r G-CSF stimulates not only the colony formation of bone marrow precursor cells but also causes marked stimulation of hematopoiesis in the spleen [7]. However, r G-CSF drugs do not have a stimulating effect on platelet and erythrocyte germ. Therefore, in patients with combined neutro-thrombocytopenia and erythrocytopenia, additional stimulation of thrombopoietin (erythropoietin) factors or transfusion of blood components (platelets, erythrocytes) are demanded.
Various compounds of polysaccharide nature were found to stimulate hematopoiesis similarly to colony-stimulating factor [8,9,10]. At the same time, in experimental models it was shown that, unlike r G-CSF, some of these compounds on the background of cyclophosphamide-induced myelosuppression not only neutralize neutropenia, but also contribute to the restoration of the number of lymphocytes and red blood cells (RBC) in animals. In particular, sulfated polysaccharide fucoidan from the seaweed Fucus vesiculosis caused pronounced mobilization of progenitor cells when administered to intact mice at a dose of 25 mg/kg. The colony-stimulating effect of the fucoidan was associated with its ability to inhibit P-selectin and L-selectin but not E-selectin [8,11].
Recently, various biological properties similar to those of fucoidan were shown for another type of sulfated polysaccharides, namely, fucosylated chondroitin sulfates [12,13,14,15]. These compounds from different species of sea cucumbers vary in structure, including degree and pattern of sulfation, position of branches and molecular weight [14,15,16,17]. It is known that the fine structure of O-sulfated polysaccharides significantly influences their biological properties [17,18,19].
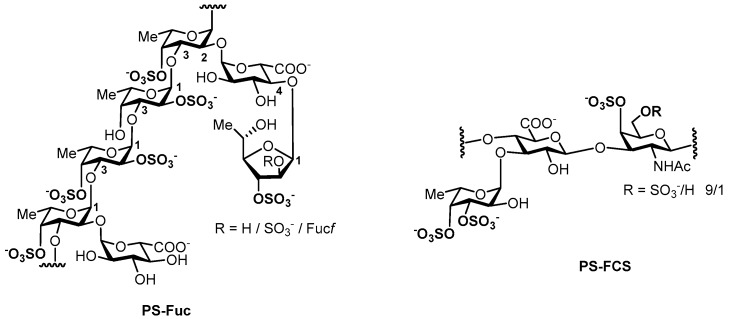
In this communication, the results of the study of two sulfated polysaccharides as stimulators of hematopoiesis are presented. The tested compounds were fucoidan from the seaweed Chordaria flagelliformis (PS-Fuc) [20] and fucosylated chondroitin sulfate from the sea cucumber Massinium magnum (PS-FCS) [14] (Figure 1). Both polysaccharides were structurally characterized recently using chemical and physicochemical methods including comparison of their NMR spectra with those of synthetic related oligosaccharides [20,21,22,23,24,25]. PS-FCS represents one of the most regular and structurally simple cases of biopolymers of this group while fucoidan PS-Fuc was selected for its unusually complex structure.
Figure 1.
A dominant structural fragment of fucoidan (PS-Fuc) from the seaweed Chordaria flagelliformis [20] and the repeating unit of fucosylated chondroitin sulfate (PS-FCS) from the sea cucumber Massinium magnum [14].
2. Results
Both compounds PS-Fuc and PS-FCS are related to sulfated polysaccharides of marine origin. However, the fine structures of these biopolymers are quite different (Figure 1). The backbone of fucoidan PS-Fuc is built from (1→3)-linked sulfated α-l-fucopyranosyl residues, some of which bear α-d-GlcA or more complicated sulfated α-l-Fucf(1→4)-α-d-GlcA fragments as branches at O-2. Polysaccharide PS-FCS consists of the chondroitin core [→4)-β-d-GlcA-(1→3)-β-d-GalNAc-(1→]n decorated by 3,4-di-O-sulfated α-l-fucosyl branches attached to O-3 of GlcA units and sulfates at O-4 and/or O-6 of GalNAc.
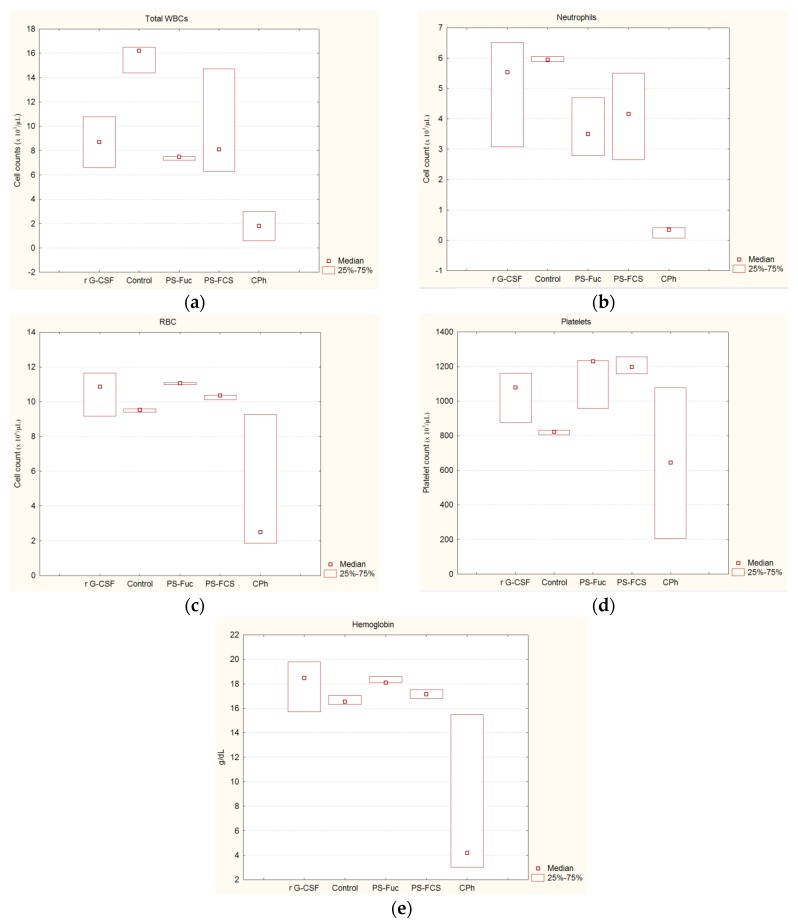
The study of the influence of polysaccharides PS-Fuc and PS-FCS on hematopoiesis was performed on the model of CPh-induced immunosuppression in mice. Recombinant r G-CSF (Leicyta) was applied as a reference. Intact animals were characterized as a control. Used active concentrations were selected on the basis of our previous data [18,26]. The results of the investigation of hematological parameters in various groups of mice are presented in Table 1 and Figure 2. The levels of white blood cells (WBC), neutrophils, RBC, platelets and hemoglobin were determined.
Table 1.
Hematologic parameters of mice with cyclophosphamide (CPh)-induced immunosuppression after treatment with PS-Fuc, PS-FCS and recombinant granulocyte colony-stimulating factor (r G-CSF).
| Groups | WBC (×103/µL) |
Neutrophils (×103/µL) |
Neutrophils (%) | RBC (×106/µL) |
Hemoglobin (g/dL) |
Platelets (×103/µL) |
|---|---|---|---|---|---|---|
| CPh + r G-CSF | 8.7 1 | 5.5 | 51 | 10.9 | 18.5 | 1079 |
| 6.9–10.9 2 | 3.0–6.9 | 45–77 | 9.0–11.9 | 14.9–19.9 | 870–1169 | |
| CPh + PS-Fuc | 7.5 1 | 3.5 | 47 | 11.1 | 18.1 | 1231 |
| 7.3–7.7 2 | 2.1–4.9 | 36–69 | 11.0–11.6 | 18.0–18.9 | 951–1244 | |
| CPh + PS-FCS | 8.1 1 | 4.2 | 42 | 10.4 | 17.2 | 1197 |
| 6.1–14.9 2 | 2.4–5.8 | 30–54 | 10.0–10.8 | 16.1–17.7 | 1149–1261 | |
| CPh | 1.8 1 | 0.4 | 12 | 2.5 | 4.2 | 645 |
| 0.3–3.4 2 | 0.1–0.9 | 11–25 | 1.5–9.4 | 3.0–15.9 | 203–1084 | |
| Control | 16.2 1 | 6.0 | 37 | 9.5 | 16.6 | 823 |
| 13.9–16.9 2 | 5.5–6.9 | 34–44 | 9.0–9.9 | 16.0–17.2 | 800–845 |
1 Median, 2 Minimum–maximum.
Figure 2.
Hematologic parameters of mice with CPh-induced immunosuppression after the treatment with PS-Fuc, PS-FCS and r G-CSF: (a) WBC; (b) neutrophils; (c) red blood cells (RBC); (d) platelets; (e) hemoglobin. Intact animals were characterized as a control.
The data of Table 1 indicated that under the influence of PS-Fuc, PS-FCS and r G-CSF there was a significant (p ≤ 0.041) increase of WBC in comparison with the group CPh: 4.2, 4.5 and 4.8 times, respectively (by the ratio of medians). This indicated the leveling of CPh-induced leukopenia after the administration of PS-Fuc and PS-FCS. At the same time, the level of their activity was not significantly different from that of r G-CSF (p ≥ 0.345). The recovery of total WBC count under the influence of both tested compounds was due to an increase in the neutrophils count in the blood of mice with CPh-induced leukopenia (p ≤ 0.01). This trend was more pronounced under the influence of PS-FCS and r G-CSF than under the influence of PS-Fuc: the neutrophils count increased by 10.5, 13.8 and 8.8 times, respectively, compared with the group of CPh.
An increase of the concentration of RBC, hemoglobin and platelets in the blood of mice with CPh-induced immunosuppression was observed after the treatment with r G-CSF, PS-Fuc and PS-FCS. It was noticeable that in the case of the use of PS-Fuc and PS-FCS increasing of RBC, hemoglobin and platelets was statistically significant (p < 0.05) comparing to a control, while in the case of r G-CSF the effect was not significant because the p values remarkably exceeded 0.05 (0.226 for RBC, 0.328 for hemoglobin, and 0.063 for platelets, Table S1).
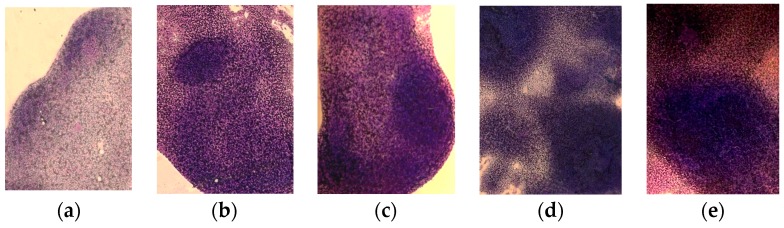
Analysis of the morphology of the spleen on the smears-prints showed that after repeated administration of CPh, myelosuppression is accompanied by depletion of the cellular composition of the white pulp, represented mainly by WBC (Figure 3a). Thus, the follicles (B-dependent areas) and periarteriolar sheaths (T-dependent areas) disappeared. The architecture of the spleen was destroyed, and the interstitial tissue was composed of a dense and uniform layer of lymphoid cells. After the course of r G-CSF and both tested polysaccharides PS-Fuc and PS-FCS, cell repair was observed (Figure 3b–d). In particular, intensive recovery was noted after the course of PS-FCS administration.
Figure 3.
Morphology of the spleen of mice with CPh-induced immunosuppression after treatment by tested substances compared to intact animals (hematoxylin-eosin staining): (a) CPh; (b) CPh + PS-Fuc; (c) CPh + PS-FCS; (d) CPh + r G-CSF; (e) control. Original magnification × 400.
3. Discussion
It was found that polysaccharides PS-Fuc and PS-FCS, similarly to r G-CSF, are able of stimulating neutropoiesis in systemic administration to mice with myelosuppression induced by CPh. At the same time, only PS-FCS in some animals led to a complete recovery of the level of total WBC to the initial values. Therefore, PS-FCS is more active than PS-Fuc in neutralization of CPh-induced neutropenia.
The investigated compounds also stimulated lymphopoiesis. This is evidenced by the normalization of the spleen white pulp cell composition in CPh-induced mice.
Concerning the effect of tested compounds on erythropoiesis, the following can be noted: the application of all three substances led to an increase in the number of RBC in peripheral blood of animals exposed to CPh by more than 4 times. In this case, PS-Fuc and PS-FCS acted more reproducibly than r G-CSF alone, leading to an increase in the concentration of RBC in the blood of animals, even above the basis level (p < 0.002).
Similar dynamics were observed in the analysis of the concentration of platelets in the blood of animals. It was shown that PS-Fuc and PS-FCS were more active (by 91% and 86%, respectively), compared to r G-CSF (by 67%) in the stimulation of thrombopoiesis in mice on the background of immunosuppression induced by CPh. This resulted in a significant increase in the platelet count in the blood after the administration of the test compounds in comparison with the baseline level (p = 0.025 and p = 0.001, respectively).
4. Materials and Methods
4.1. Sulfated Polysaccharides
Fucoidan PS-Fuc was isolated from the seaweed Chordaria flagelliformis as described [20]. Fucosylated chondroitin sulfate PS-FCS was isolated from the sea cucumber Massinium magnum as described [14].
4.2. Animal Model
The study was approved by the local ethical committee of the N.N. Blokhin Medical Research Center of Oncology, Russia, Moscow.
Thirty mice of the CBA line (males, weight 25 ± 1 g) were divided into five groups of six animals in each. Before and during the experiment, the animals were in standardized vivarium conditions (T of air was 20 ± 2 °C, under conditions of free access to food and water). For the inducing of myelosuppression, CPh (Endoxan, Baxter, Halle, Germany) in a dosage of 100 mg/kg was injected to animals of 4 groups 1 time daily intraperitoneally for 4 days. Then the following sterile solutions (0.2 mL) were administered subcutaneously to all animals for 3 days (1 time daily): 0.5 mg/mL of PS-Fuc in isotonic sodium chloride solution (group CPh + PS-Fuc), 0.5 mg/mL of PS-FCS in isotonic sodium chloride solution (group CPh + PS-FCS), and 3 nmol/mL of r G-CSF (Leucita, Sygardis AqVida, Lich, Germany) in isotonic sodium chloride solution (group CPh + r G-CSF), sterile isotonic sodium chloride solution (groups CPh). A sterile isotonic sodium chloride solution was administered to the mice of the control group in the same regime. The animals were euthanized by decapitation after 2 days. Blood of each animal was collected in the tubes with ethylenediaminetetraacetic acid (EDTA), the spleen was removed from the animals, and smears were imprinted on the polyethylene coated glasses (Gerhard Menzei GmbH, Termo Fisher Scientific, Waltham, MA, USA). The fingerprints were fixed in May-Grunwald solution, stained with hematoxylin-eosin (HE) and analyzed by light microscopy. Hematologic parameters of blood were analyzed on an automatic analyzer, determining the concentration of WBC, platelets and RBC. In a blood smear stained with HE, a neutrophil count of 100 leukocytes was counted using a light microscope, then the number of neutrophils in the blood of mice was calculated.
4.3. Statistical Analysis
The data in the group was represented in the format of medians and range of minimum–maximum values. The differences between multiple groups were compared with an ANOVA. The Wald-Wolfowitz runs test was used to compare the two groups. Differences were considered significant at p < 0.05.
5. Conclusions
Two sulfated polysaccharides as stimulators of hematopoiesis have been studied in vivo. The tested compounds were the fucoidan from the seaweed C. flagelliformis (PS-Fuc) and the fucosylated chondroitin sulfate from the sea cucumber M. magnum (PS-FCS). Both tested biopolymers have a comparable level of activity with r G-CSF with regard to stimulation of pronounced neutropoiesis in animals with myelosuppression induced by CPh. Additionally, these compounds were shown to be capable of stimulating thrombopoiesis and erythropoiesis. It should be noted that PS-FCS demonstrated a greater activity than r G-CSF.
The obtained results argue for the necessity of further studies into PS-Fuc- and PS-FCS-like polysaccharides and synthetic oligosaccharides which represent their fragments to form the basis for further development of novel hematopoiesis stimulating drugs. They are in demand for the accompanying treatment of cancer patients who suffer from hematological toxicity during chemo and/or radiation therapy. Availability of parent natural fucoidans [18,20,21] and fucosylated chondroitin sulfates [12,13,14,15,16,17] of very different structure as well as the possibility of chemical synthesis of oligosaccharides which represent fucoidans [22,25,26,27] and fucosylated chondroitin sulfates [23,24] fragments as well as the methods for their conformational analysis [27,28,29,30,31] permits systematic structure-activity study, to assess the pharmacophore fragments in the structures of sulfated polysaccharides and to undertake the next steps—the search for target cellular receptors and rational design of their druggable inhibitors. These studies are in progress and will be reported in due course.
Acknowledgments
This work was supported by the Russian Science Foundation (grant 14-50-00126—studies of fucoidan from the seaweed Chordaria flagelliformis; grant 14-13-01325—studies of fucosylated chondroitin sulfate from the sea cucumber Massinium magnum).
Supplementary Materials
The following are available online at www.mdpi.com/1660-3397/15/10/301/s1, Table S1: Comparative analysis of hematological parameters in groups of mice after treatment of CPh with r G-CSF, PS-Fuc, PS-FCS.
Author Contributions
N.A., N.U., M.K. and N.N. conceived and designed the experiments; N.A. and F.D. performed the experiments; N.A. and M.K. analyzed the data; M.B., N.U. and A.U. prepared and characterized the tested compounds; all the authors wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Fishman P., Bar-Yehuda S., Barer F., Madi L., Multani A.S., Pathak S. The A3 adenosine receptor as a new target for cancer therapy and chemoprotection. Exp. Cell Res. 2001;269:230–236. doi: 10.1006/excr.2001.5327. [DOI] [PubMed] [Google Scholar]
- 2.Schirmer J.H., Bremer J.P., Moosig F., Holle J.U., Lamprecht P., Wieczorek S., Haenisch S., Cascorbi I. Cyclophosphamide treatment-induced leukopenia rates in ANCA-associated vasculitis are influenced by variant CYP450 2C9 genotypes. Pharmacogenomics. 2016;17:367–374. doi: 10.2217/pgs.15.176. [DOI] [PubMed] [Google Scholar]
- 3.Im S.A., Kim K.H., Kim H.S., Lee K.H., Shin E., Do S.G., Jo T.H., Park Y.I., Lee C.K. Processed Aloe vera gel ameliorates cyclophosphamide-induced immunotoxicity. Int. J. Mol. Sci. 2014;15:19342–19354. doi: 10.3390/ijms151119342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kuter D.J. Managing thrombocytopenia associated with cancer chemotherapy. Oncology (Williston Park) 2015;29:282–294. [PubMed] [Google Scholar]
- 5.Crobu D., Spinetti G., Schrepfer R., Tonon G., Jotti G.S., Onali P., Dedoni S., Orsini G., Di Stefano A. Preclinical and clinical phase I studies of a new recombinant Filgrastim (BK0023) in comparison with Neupogen®. BMC Pharmacol. Toxicol. 2014;15:7. doi: 10.1186/2050-6511-15-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Romero-Weaver A.L., Wan X.S., Diffenderfer E.S., Lin L., Kennedy A.R. Kinetics of neutrophils in mice exposed to radiation and/or granulocyte colony-stimulating factor treatment. Radiat. Res. 2013;180:177–188. doi: 10.1667/RR3055.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Molineux G., Pojda Z., Dexter T.M. A comparison of hematopoiesis in normal and splenectomized mice treated with granulocyte colony-stimulating factor. Blood. 1990;75:563–569. [PubMed] [Google Scholar]
- 8.Frenette P.S., Weiss L. Sulfated glycans induce rapid hematopoietic progenitor cell mobilization: Evidence for selectin-dependent and independent mechanisms. Blood. 2000;96:2460–2468. [PubMed] [Google Scholar]
- 9.Chen X., Nie W., Fan S., Zhang J., Wang Y., Lu J., Jin L. A polysaccharide from Sargassum fusiforme protects against immunosuppression in cyclophosphamide-treated mice. Carbohydr. Polym. 2012;90:1114–1119. doi: 10.1016/j.carbpol.2012.06.052. [DOI] [PubMed] [Google Scholar]
- 10.Kubonishi S., Kikuchi T., Yamaguchi S., Tamamura H., Fujii N., Watanabe T., Arenzana-Seisdedos F., Ikeda K., Matsui T., Tanimoto M., et al. Rapid hematopoietic progenitor mobilization by sulfated colominic acid. Biochem. Biophys. Res. Commun. 2007;355:970–975. doi: 10.1016/j.bbrc.2007.02.069. [DOI] [PubMed] [Google Scholar]
- 11.Hidalgo A., Peired A.J., Weiss L.A., Katayama Y., Frenette P.S. The integrin alphaMbeta2 anchors hematopoietic progenitors in the bone marrow during enforced mobilization. Blood. 2004;104:993–1001. doi: 10.1182/blood-2003-10-3702. [DOI] [PubMed] [Google Scholar]
- 12.Pomin V.H. Holothurian Fucosylated Chondroitin Sulfate. Mar. Drugs. 2014;12:232–254. doi: 10.3390/md12010232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Borsig L., Wang L., Cavalcante M.C., Cardilo-Reis L., Ferreira P.L., Mourão P.A., Esko J.D., Pavão M.S. Selectin blocking activity of a fucosylated chondroitin sulfate glycosaminoglycan from sea cucumber. Effect on tumor metastasis and neutrophil recruitment. J. Biol. Chem. 2007;282:14984–14991. doi: 10.1074/jbc.M610560200. [DOI] [PubMed] [Google Scholar]
- 14.Ustyuzhanina N.E., Bilan M.I., Dmitrenok A.S., Borodina E.Y., Stonik V.A., Nifantiev N.E., Usov A.I. A highly regular fucosylated chondroitin sulfate from the sea cucumber Massinium magnum: Structure and effects on coagulation. Carbohydr. Polym. 2017;167:20–26. doi: 10.1016/j.carbpol.2017.02.101. [DOI] [PubMed] [Google Scholar]
- 15.Ustyuzhanina N.E., Bilan M.I., Dmitrenok A.S., Shashkov A.S., Kusaykin M.I., Stonik V.A., Nifantiev N.E., Usov A.I. Structure and biological activity of a fucosylated chondroitin sulfate from the sea cucumber Cucumaria japonica. Glycobiology. 2016;26:449–459. doi: 10.1093/glycob/cwv119. [DOI] [PubMed] [Google Scholar]
- 16.Ustyuzhanina N.E., Bilan M.I., Dmitrenok A.S., Shashkov A.S., Nifantiev N.E., Usov A.I. The structure of a fucosylated chondroitin sulfate from the sea cucumber Cucumaria frondosa. Carbohydr. Polym. 2017;165:7–12. doi: 10.1016/j.carbpol.2017.02.003. [DOI] [PubMed] [Google Scholar]
- 17.Chen S., Xue C., Yin L., Tang Q., Yu G., Chai W. Comparison of structures and anticoagulant activities of fucosylated chondroitin sulfates from different sea cucumbers. Carbohydr. Polym. 2011;83:688–695. doi: 10.1016/j.carbpol.2010.08.040. [DOI] [Google Scholar]
- 18.Cumashi A., Ushakova N.A., Preobrazhenskaya M.E., D’Incecco A., Piccoli A., Totani L., Tinari N., Morozevich G.E., Berman A.E., Bilan M.A., et al. Comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology. 2007;17:541–552. doi: 10.1093/glycob/cwm014. [DOI] [PubMed] [Google Scholar]
- 19.Ustyuzhanina N.E., Bilan M.I., Ushakova N.A., Usov A.I., Kiselevskiy M.V., Nifantiev N.E. Fucoidans: Pro- or antiangiogenic agents? Glycobiology. 2014;24:1265–1274. doi: 10.1093/glycob/cwu063. [DOI] [PubMed] [Google Scholar]
- 20.Bilan M., Vinogradova E.V., Tsvetkova E.A., Grachev A.A., Shashkov A.S., Nifantiev N.E., Usov A.I. A sulfated glucuronofucan containing both fucofuranose and fucopyranose residues from the brown alga Chordaria flagelliformis. Carbohydr. Res. 2008;343:2605–2612. doi: 10.1016/j.carres.2008.06.001. [DOI] [PubMed] [Google Scholar]
- 21.Ustyuzhanina N.E., Ushakova N.A., Preobrazhenskaya M.E., Bilan M.I., Tsvetkova E.N., Krylov V.B., Anisimova N.A., Kiselevskiy M.V., Krukovskaya N.V., Li C., et al. Fucoidans as a platform for new anticoagulant drugs discovery. Pure Appl. Chem. 2014;86:1365–1375. doi: 10.1515/pac-2014-0404. [DOI] [Google Scholar]
- 22.Khatuntseva E.A., Ustuzhanina N.E., Zatonskii G.V., Shashkov A.S., Usov A.I., Nifant’ev N.E. Synthesis, NMR and conformational studies of fucoidan fragments. Part 1. Desulfated 2,3- and 3,4-branched trisaccharide fragments and costituing disaccharides. J. Carbohydr. Chem. 2000;19:1151–1173. doi: 10.1080/07328300008544140. [DOI] [Google Scholar]
- 23.Ustyuzhanina N.E., Fomitskaya P.A., Gerbst A.G., Dmitrenok A.S., Nifantiev N.E. Synthesis of the oligosaccharides related to branching sites of fucosylated chondroitin sulfates from sea cucumbers. Mar. Drugs. 2015;13:770–787. doi: 10.3390/md13020770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vinnitskiy D.Z., Ustyuzhanina N.E., Dmitrenok A.S., Shashkov A.S., Nifantiev N.E. Synthesis and NMR analysis of model compounds related to fucosylated chondroitin sulfates: GalNAc and Fuc(1→6)GalNAc derivatives. Carbohydr. Res. 2017;438:9–17. doi: 10.1016/j.carres.2016.11.015. [DOI] [PubMed] [Google Scholar]
- 25.Krylov V.B., Kaskova Z.M., Vinnitskiy D.Z., Ustyuzhanina N.E., Grachev A.A., Chizhov A.O., Nifantiev N.E. Synthesis, NMR and conformational studies of fucoidan fragments. Part 11. Acid-promoted synthesis of per-O-sulfated fucooligosaccharides related to fucoidan fragments. Carbohydr. Res. 2011;346:540–550. doi: 10.1016/j.carres.2011.01.005. [DOI] [PubMed] [Google Scholar]
- 26.Anisimova N.Y., Ustyuzhanina N.E., Donenko F.V., Bilan M.I., Ushakova N.A., Usov A.I., Nifantiev N.E., Kiselevskiy M.V. Influence of fucoidans and their derivatives on antitumor and phagocytic activity of human blood leucocytes. Biochemistry. 2015;80:925–933. doi: 10.1134/S0006297915070111. [DOI] [PubMed] [Google Scholar]
- 27.Gerbst A.G., Ustuzhanina N.E., Grachev A.A., Tsvetkov D.E., Khatuntseva E.A., Shashkov A.S., Usov A.I., Preobrazhenskaya M.E., Ushakova N.A., Nifantiev N.E. Synthesis, NMR and conformational studies of fucoidan fragments. Part 5. Linear 4,4′,4′′-tri-O-sulfated and parent non-sulfated (1→3)-fucotrioside fragments. J. Carbohydr. Chem. 2003;22:109–122. doi: 10.1081/CAR-120020481. [DOI] [Google Scholar]
- 28.Grachev A.A., Gerbst A.G., Ustuzhanina N.E., Shashkov A.S., Usov A.I., Nifantiev N.E. Synthesis, NMR and conformational studies of fucoidan fragments. 9. NMR investigation of the influence of sulfate group at C-2 and C-4 on the conformational behaviour of fucoidan fragments with homo-(1→3)-linked backbone. J. Carbohydr. Chem. 2006;25:315–330. doi: 10.1080/07328300600770493. [DOI] [Google Scholar]
- 29.Gerbst A.G., Dmitrenok A.S., Ustyuzhanina N.E., Nifantiev N.E. Conformational analysis of the oligosaccharides related to side chains of holothurian fucosylated chondroitin sulfates. Mar. Drugs. 2015;13:936–947. doi: 10.3390/md13020936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gerbst A.G., Ustuzhanina N.E., Grachev A.A., Tsvetkov D.E., Khatuntseva E.A., Nifantiev N.E. Synthesis, NMR and conformational studies of fucoidan fragments. Part 4. 4-Mono- and 4,4′-disulfated (1→3)-α-l-fucobioside and 4-sulfated fucoside fragments. J. Carbohydr. Chem. 2002;21:313–324. doi: 10.1081/CAR-120013500. [DOI] [Google Scholar]
- 31.Ustuzhanina N.E., Krylov V.B., Grachev A.A., Gerbst A.G., Nifantiev N.E. Synthesis, NMR and Conformational Studies of Fucoidan Fragments. VIII. Convergent block-wise synthesis of long chain linear and 2,3-branched oligosaccharides. Synthesis. 2006;2006:4017–4031. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.