Abstract
Introduction
Only 45% of people currently living with HIV infection in sub-Saharan Africa are aware of their HIV status. Unmet testing needs may be addressed by utilizing the Emergency Department (ED) as an innovative testing venue in low and middle-income countries (LMICs). The purpose of this review is to examine the burden of HIV infection described in EDs in LMICs, with a focus on summarizing the implementation of various ED-based HIV testing strategies.
Methodology and results
We performed a systematic review of Pubmed, Embase, Scopus, Web of Science and the Cochrane Library on June 12, 2016. A three-concept search was employed with emergency medicine (e.g., Emergency department, emergency medical services), HIV/AIDS (e.g., human immunodeficiency virus, acquired immunodeficiency syndrome), and LMIC terms (e.g., developing country, under developed countries, specific country names).
The search returned 2026 unique articles. Of these, thirteen met inclusion criteria and were included in the final review. There was a large variation in the reported prevalence of HIV infection in the ED population ranging from to 2.14% in India to 43.3% in Uganda. The proportion HIV positive patients with previously undiagnosed infection ranged from 90% to 65.22%.
Conclusion
In the United States ED-based HIV testing strategies have been front and center at curbing the HIV epidemic. The limited number of ED-based studies we observed in this study may represent the paucity of HIV testing in this venue in LMICs. All of the studies in this review demonstrated a high prevalence of HIV infection in the ED and an extraordinarily high percentage of previously undiagnosed HIV infection. Although the numbers of published reports are few, these diverse studies imply that in HIV endemic low resource settings EDs carry a large burden of undiagnosed HIV infections and may offer a unique testing venue.
Introduction
Despite significant strides in combating HIV worldwide, the AIDS pandemic continues. The UNAIDS Gap Report declares that it will be impossible to end the epidemic without bringing HIV treatment to all who need it [1]. Consequently, UNAIDS has adopted ambitious treatment targets called “90-90-90” that stipulate by 2010 90% of those infected with HIV will be aware of their diagnosis, of which 90% will be successful linked to acre (LTC) and of which 90% will achieve viral suppression [2].
Significant challenges lie ahead with regard to reaching these goals. Currently there are 22 million people worldwide who either do not have access to life-saving treatment due to coverage gaps within the health care system [2, 3]. Globally the HIV epidemic is concentrated, with 15 countries accounting for nearly 75% of all individuals living with HIV. Two thirds of these countries are located in sub-Saharan Africa (South Africa, Nigeria, Kenya, Mozambique, Uganda, Tanzania, Zimbabwe, Zambia, Malawi, Ethiopia); the remaining five countries include India, China, Russia, Brazil, and the United States [1]. Many of these countries (all except Russia and the USA) fall under the definition of low-income economies (either designated as low-income countries (LICs) (less than $1,045 USD GNI per capita) or lower-middle income countries (LMICs) ($1,045-$4,125 USD GNI per capita) as defined by the World Bank [4]. It is in these economies where healthcare resources are limited, that the number of people with unknown HIV infection, new HIV infections, and AIDS related deaths remain the highest [5].
Early detection of undiagnosed HIV infection with subsequent effective HIV treatment is widely recognized to extend life expectancy, improves life quality, and reduces HIV transmission, making it a cost-effective public health intervention [3]. Accordingly, the key to early treatment is early recognition with the first stage of the HIV care continuum (or “cascade”) beginning with diagnosis [6]. Unfortunately, only 45% of people currently living with HIV in sub-Saharan Africa are aware of their HIV status (about half of the UNAIDS goal) [5]. While promising results have emerged from innovative mobile and home testing initiatives to identify key/under-tested populations a wider sustained testing strategy is required in venues that provides care to key populations and are able to effectively integrate HIV screening into routine care [7].
The Emergency Department (ED) provides care large volumes of individuals who present for episodic care, many of whom use emergency care services as their sole source of care [8]. In the United States (US), HIV testing was expanded to non-traditional venues such as EDs and non-clinical settings. This highly successful strategy has made significant strides in curbing the HIV epidemic in the US [9]. The rise of integrated HIV testing into US emergency departments (which serve over 140 million patients/year) has been shown to parallel declines in rates undiagnosed HIV infection, and increases in rates of antiretroviral use [9, 10]. The ED is now recognized by the Centers for Disease Control to be central to the national HIV testing strategy, and the implementation of ED-based HIV screening programs is recommended by the US Preventive Services Task Force (USPSTF) when the local prevalence of HIV infection is >0.1% and has been adopted by the American College of Emergency Physicians [11–13]. As ED-based HIV testing has gathered momentum, numerous studies have also sought to understand the best approaches to implementing HIV testing in the ED for example opt-in versus opt-out approaches, it is unclear what strategies are feasible in low resource EDs [14] [15].
While there is, strong evidence supporting the role and impact of ED-based HIV testing in the US, there is a dearth of knowledge regarding what the scope of missed HIV infection may be in EDs across low resource settings. The purpose of this review is to quantify the burden of HIV infection described in EDs in low resource settings and examine the acceptance and feasibility of ED-based HIV testing strategies in LMICs.
Materials and methods
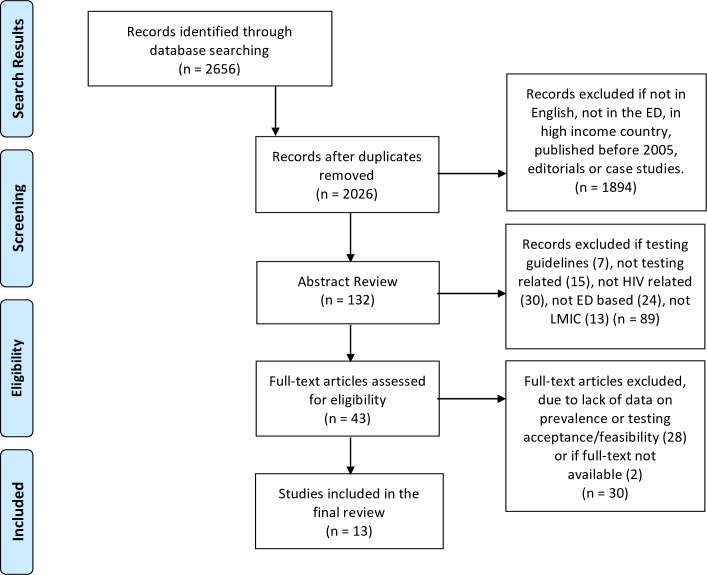
We performed an initial search of Pubmed, Embase, Scopus, Web of Science and the Cochrane Library on June 12th 2016 with an updated search on January 1st 2017. A three-concept search was employed with emergency medicine (e.g., Emergency department, emergency medical services), HIV/AIDS (e.g., human immunodeficiency virus, acquired immunodeficiency syndrome), and low and middle-income country terms (e.g., developing country, under developed countries, and specific country names employing the Cochrane Collaboration's low and middle-income filter) [16]. A full search strategy is presented in S1 Appendix. All applicable controlled vocabularies and keyword terms were searched. The concepts for the search strategy were developed in collaboration with a professional librarian (KL) and clinicians (BH, TD, SH). The search was run without any restrictions and two authors screened each result. Only articles published from Jan 1st 2005 to Jan 1st 2017 were included. Studies were included if they described a specific ED-based testing strategy, presented data on HIV prevalence in the ED, or presented data on the acceptability/feasibility of an ED-based testing program. Studies were excluded if they were conducted in high income settings, were purely descriptive, editorials, or case based studies (defined as an n less than 5. A final breakdown of our search strategy is presented in Fig 1. Authors further evaluated the studies for data on HIV prevalence (including undiagnosed), LMIC status, and ED-based testing strategies. Two independent authors reviewed each study with identified discrepancies resolved by a third senior author (BH).
Fig 1. PRISMA flow diagram of articles included in the systematic review.
Results
The search returned 2026 unique articles. Of these, thirteen articles met inclusion criteria and were included in the final review. Three studies were in LICs [17–19], nine in LMICs [20–29]. Seven studies examined adult populations, one study examined both adult and pediatric populations, and two studies examined only the pediatric population. A summary of all the adult studies included in the review are provided in Table 1 and pediatric studies included in the review are provided in Table 2.
Table 1. Summary of all adult studies included in the systematic review and reported HIV prevalence, proportion of undiagnosed HIV infection and the testing strategy utilized.
| Quantitative Studies including adult patients | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Country | Year | Journal | Author | Type of study | Tested (N)/total offered | Sampling Method |
Point of Testing | Total HIV + (%) | Total new HIV diagnosis (%) | Type of HIV test |
| India | 2010 | Ind J of STDs | Devi [26] |
Cross sectional | 400 | Unclear | Unclear | 23 (5.75%) |
15 (65.22%) |
COMB AIDS-RS (HIV 1 and 2 Immunodot test kit) confirmed by Retroqic HIV and HIV Tridot for HIV I/II antibodies |
| India++ | 2010 | Indian J Pathol Microbiol | Minz [25] |
Cross sectional | 607 | Symptoms of HIV (Targeted) | Pediatric, Surgical, and Medical EDs | 13 (2.14%) |
Unknown | Rapid Kits (Signal HIV test, Comb AIDS test, Immunocomb II HIV test). Confirmed using micro-ELISA. |
| India | 2014 | Int J of STD and AIDS | Minz [24] |
Retrospe-ctive review | 654 | Targeted | ED | 30 (4.60%) |
Unknown | 4th generation micro-ELISA, confirmation w/ GENEDIA and TRIDOT |
| Uganda* | 2006 & 2007 | Afr Health Sci | Nakan- jako [17, 19] |
Cross sectional | 198/233 | Every 6th Patient | Medical ED | 86 (43.40%) |
86 (77.00%) |
3 sequential rapid tests; Determine, HIV ½ Stat-Pak, Unigold. |
| Kenya | 2007 | EMJ | Ranney [21] |
Retrospe-ctive review | 285/321 | Sexually assaulted women | ED | 7 (2.46%) |
5 (71.40%) |
not defined |
| India | 2004 | Indian J Med Microbiol. | Teja [22] |
Retrospe-ctive review | 1187 | Known HIV positive | Inpatient | Unknown | 90% | not defined |
| India | 2008 | Indian J Med Microbiol. | Teja [23] |
Retrospe-ctive review |
10752 | Provider initiated (Targeted) | ED (medical patients only) | 317 (2.90%) |
211 (84.75%) |
Rapid HIV for emergent surgery (HIV Tri dot) + ELISA for all Vironostika HIV ag/ab Combi, reactive specimens reevaluated by 2nd assay; discordants confirmed by 4th Gen VIDAS HIV Ultra Duo |
| Kenya | 2007 | AIDS Patient Care and STDs | Wax -man [20] |
Retrospe-ctive review |
1339/ 1371 |
Symptoms of HIV, Opt-out testing | Medical ED | 312 (22.70%) |
Unknown | Two rapid tests: Uni-GoldTM Recombigen(R) and Determine(R) HIV-1/2 |
* 2 papers by Nakajanko et al. present data from the same dataset
** 419 patients (25.7% had no HIV result documented in the chart)
Table 2. Summary of all pediatric studies included in the systematic review and reported HIV prevalence, proportion of undiagnosed HIV infection and the testing strategy utilized.
| Country | Year | Journal | Author | Type of study | Tested (N)/total offered | Sampling Method |
Point of Testing | Total HIV + (%) | Total new HIV diagnosis (%) | Type of HIV test |
|---|---|---|---|---|---|---|---|---|---|---|
| Malawi* | 2010 | EMJ | Ahmad [18] |
Cross sectional | 576 | Critically ill children only | ED | 152 (26%) |
Unknown | Bedside antibody test for HIV ½, confirmed by HIV RNA PCR. |
| India** | 2010 | Ind J of Path and Micro | Minz [25] |
Cross sectional | 239 | Symptoms of HIV (Targeted) | ED | 13 (2.14%) |
Unknown | Rapid Kits (Signal HIV test, Comb AIDS test, Immunocomb. HIV test). Confirmed w/ micro-ELISA. |
| Tanz -ania |
2016 | BMJ Open | Sawe [29] |
Retrospe-ctive review | 1632/ 5540 |
Charts reviewed | ED and Inpatient | 239 (14.5%)** |
Unknown | not defined |
* In this study children under 18 months excluded
** Same article presents data on both adults and children, thus has been included twice in the table above
HIV prevalence
Nine studies quantified the burden of HIV infection in ED patients [17, 18, 20, 21, 23–26, 29]. The countries included in this review were: India [22–26], Kenya [20, 21, 28], Uganda [17, 19], Malawi [18], Tanzania [29] and Guyana [27]. There was a large variation in the reported prevalence of HIV infection in the ED population ranging from to 2.14% in India to 43.3% in Uganda [19, 25]. Only five studies were able to provide data on the burden of undiagnosed HIV infection [17, 21–23, 26]. The burden of previously undiagnosed infection reported ranged from 90% to 65.22% [23, 26].
ED-based HIV testing strategies
The majority of studies (6) were conducted as retrospective chart reviews of all ED patients that presented for care during the study period, only four studies attempted to perform a cross-sectional prevalence study, i.e., they implemented a testing strategy over a defined period and then assessed testing acceptance and reactivity rates.[17–19, 25, 26] Two of the studies used fourth generation lab based testing and conducted on samples already collected from patients, both of these studies were also conducted in India [23, 25, 26]. The remaining studies that report testing strategies used rapid point of care tests with confirmatory ELISA or PCR [17–19, 28].
Acceptance and feasibility of ED-based HIV testing
In four studies, it was possible to calculate the proportion of patients who accepted HIV testing when offered. In two studies the testing strategy was also defined, Waxman et al. offered opt-out testing and Nakanjako et al. targeted, they respectively reported a 97.7% and 85.0% acceptance of HIV testing in the ED [17, 19, 20]. A study in rape survivors by Ranney et al. reported testing acceptance at 88.7%, the lowest testing acceptance was reported by Sawe et al. who conducted a study in pediatric patient and reported a testing acceptance of only 29.5% [21, 29]. Linkage to care feasibility was assessed by Waxman et al. who reported that 85% of newly diagnosed HIV positive patients, compliant with their initial HIV clinic visits and 65% complaint with an additional 1-month follow-up visit [20].
Three studies conduct patient surveys to evaluate the acceptance of an ED-based HIV testing strategy (Table 3). Nakanjako et al. interviewed 233 patients in the ED and reported that 99% of patients support HIV testing in the ED and 86% believed that ED-based testing would improve linkage to care [19]. Christensen el al. sought to determine the acceptability of ED based testing using a closed-question survey [27]. Out of the 343 patients interviewed (using a convenience sample of patients who presented for care in the ED during the study), 75% were found to be amenable to opt-out testing if offered in the ED [27]. Patients greater then 50 years old, females, and those who had not been previously tested were more likely to refuse hypothetical HIV testing [27]. Potential reasons for declining testing were also evaluated. The two most common reasons for declining were, “I have had an HIV test recently enough” (85%, 95% CI 74.0–91.4%) and “I am not at risk for HIV/AIDS” (83%, 95% CI 73.0–90.4%) [27]. Additionally, Christensen and colleagues found that over 30% of patients in their study had never been tested for HIV, with 40% reporting that the ED was their only access to health care [27, 30]. Fear and stigma also played a role in refusal. People cited embarrassment (19%, 11.7–30.4%), rejection (30%, 20.3–41.5%), and being afraid (21%, 12.7–31.8%) as reasons they would decline hypothetical HIV testing in the ED [27].
Table 3. A summary of qualitative studies evaluating ED-based HIV testing acceptance.
| Country | Year | Journal | Author | Type of study | Description |
|---|---|---|---|---|---|
| Guyana | 2012 | Int. Health | Christensen [27] |
Survey | Four-part survey administered to 343 non-critical adult patients |
| Uganda | 2007 | AIDS Behav | Nakanako [19] |
Survey | A convenience sample of 245 patients were screened of which 233 adults were interviewed and offered HIV testing, data was collected on reasons of prior HIV testing, acceptance to take a test and their current HIV sero-status. |
| Kenya | 2008 | AIDS Patient Care and STDs | Waxman [28] |
Survey | Descriptive study of staff experience regarding the implementation of an ED-based HIV testing program. |
Only a single paper which evaluated the cost implications of ED based testing strategies in our review. Due to the readily available provision of low cost tests, Minz et al. found that testing costs in LMICs are likely to be significantly lower than in high income settings [24].
Discussion
The HIV epidemic remains a significant contributor to the burden of disease in low resource settings. In the US, ED-based HIV testing strategies have been front and center at curbing the HIV epidemic [31]. This systematic review yielded a surprisingly low number of studies focused on ED-based HIV testing in LMICs (in particular there were no studies from South America or South-East Asia). This may represent the fact that emergency medicine is a relatively new specialty, and in many countries (particularly in LMICs) yet to be formally recognized [32]. In nascent healthcare systems, EDs are often a small, underfunded, and under resourced components of the healthcare delivery model [33]. The limited number of studies we observed may represent the paucity of HIV testing in this venue in LMICs. EDs in LMICs also report significantly higher morbidity and mortality compared to their high-income country counterparts [34]. Even though many LMICs recommend routine HIV testing in all healthcare facilities it may be difficult to implement and sustain testing given the already high burden of healthcare needs of patients in the ED [28, 35, 36]. The lack of ED-based testing studies thus appears to be representative of an overall absence of ED-based testing programs in LMICs where they are most desperately needed.
It was difficult to compare HIV prevalence across studies. Two of the studies (Minz et al. 2010 and Teja et al. 2004), based ED HIV prevalence estimates on patients that had a routine HIV testing done prior to emergent surgery [22, 25]. Other studies based their decision to offer ED based testing on whether patients were symptomatic [20, 23, 24]. Both of these proxies likely mis-represent the true prevalence of HIV infection in ED. The one study that used a unbiased approach, sampled every 6th patient and reported the highest prevalence of HIV infection at 50% [19].
A variety of testing strategies were identified in this review. The advent of inexpensive rapid point of care HIV tests in the mid-1990s shifted the HIV testing paradigm and allowed for decentralized testing without sophisticated laboratory equipment [37]. Traditional laboratory based testing using ELISA only to detect HIV antibodies were challenged by long laboratory delays. Thus, it is not surprising that the majority of studies in this review, utilized rapid point of care tests to offer readily available testing in the ED [17, 19, 20, 25, 26]. Rapid tests on average require 10–20 minutes to complete, with reactive results permitting patient counseling during the ED visit. Unfortunately, they usually require sequential confirmatory testing with ELISA or PCR which can be time consuming. While point of care HIV testing allows for mobility the overall process is more time consuming and labor intensive for the ED staff.
Fourth generation laboratory based ‘combo’ tests were introduced in the late 1990s and include both an ELISA, which tests for HIV antibodies, and the p24 antigen. The advantage to adding the p24 antigen is the ability for near real-time detection of HIV infection before antibodies are produced thus, improving the detection of acute infections that were previously missed during the window period (i.e., a period when patients test negative (by antibody) despite being infected. Recent advances in diagnostics have shortened assay turnaround time to approximately 20 minutes, such that tests can now be added to routinely sent laboratory testing from the ED, and permit high volume and high throughput testing without additional ED labor time and costs. Rapid lab-based assays also lend themselves to an opt-out consent approach (i.e., testing without pre-and post-test counseling, reflexed onto routine laboratory blood tests). While integrated laboratory based testing in the US has already been shown to yield higher testing volumes (given that the majority of ED patients receive blood draws), this may not apply to resource-limited settings, where laboratory service may be unreliable or underutilized due to cost and resource constraints.
In the low resource settings, there is often a paucity of health care workers to provide testing in the ED. An innovative solution in the ED may be the provision of self-testing [38, 39]. Many patient in the ED present with stable injuries, in these cases patients are likely to have long wait times, and are unlikely to receive a blood draw. Shifting the self-testing innovation from community based settings to the ED, may provide an elegant solution to providing 24 hours testing availability in settings where there are not enough providers perform rapid tests and blood based tests are not possible due to delays in getting results. The concern however is that while self-testing has shown high acceptability it may be associated with low rates of linkage to care [40, 41].
Successful linkage to care from the ED was demonstrated by Waxman et al [28]. Timely linkage to care is even more critical in the current HIV treatment climate which advocates changing the threshold of initiating ARV therapy (from a CD4 count <350 cells/mm3 to a CD4 count >500 cells/mm3) [42]. While the findings from the Waxman et al. study are encouraging, they are equally surprising given that most LMICs EDs are often located in centralized tertiary care hospitals and see patients from a wide catchment area [34]. It is unclear how linkage to care can be sustainably maintained from a transient care facility such as the ED and future work should focus on the feasibility and efficacy of ARV initiation in the ED and the impact of such a strategy both on delivery of sustainable treatment and development of antiviral resistance within the community.
Given the high burden of HIV infection in many of these settings, coupled with the high patient turnover in the ED, the introduction of HIV testing into EDs has serious financial implications on the institution. Despite national recommendations from the CDC in 2006 mandating universal HIV testing, many EDs in the US have not implemented a testing strategy at their institution [43]. In the US, institutions must foot the bill for HIV testing and are unlikely to see the downstream benefits of morbidity and mortality avoided by early treatment initiation. Many healthcare systems in LMICs are government funded, and thus the inducement of population level benefits may allow for the provision for resources required to successfully implement testing in these settings.
Limitations
The studies presented in this review vary great by scope and sampling strategy. No studies tested all patients, and thus samples were likely not representative of the true ED population, limiting the ability to have a more complete understanding of the true burden of disease. Most studies were performed at single hospitals, and may lack external validity. It is possible that due to limited resources, patients with histories, symptoms, or signs suggestive of HIV were prioritized in the testing algorithms, artificially boosting prevalence findings. The testing strategy also varied greatly among studies and some did not require confirmatory testing as is standard in many developed countries. Nonetheless, despite this variability, these diverse few studies did show a relatively high burden of HIV infection.
Conclusions
Although the numbers of published reports are few, these diverse studies imply that in HIV endemic low resource settings, EDs carry a large burden of undiagnosed HIV infection. The ED is a strategic venue for targeting testing in low resource settings for early HIV diagnosis. ED-based testing has been shown to be both feasible and acceptable within this environment. The true burden of HIV infection in the ED and the unique demographic features of the HIV positive ED population still remain largely unknown given the lack of a blanket testing strategy. Further research is required on the implementation and cost effectiveness of ED-based testing strategies given the resource constraints and high prevalence of HIV infection in LMICs.
Supporting information
(DOCX)
(DOC)
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
The authors received no specific funding for this work.
References
- 1.HIV/AIDS JUNPo. The gap report. Geneva: UNAIDS. 2014.
- 2.HIV/Aids JUNPo, HIV/Aids JUNPo. 90-90-90: an ambitious treatment target to help end the AIDS epidemic. Geneve: UNAIDS. 2014.
- 3.Girardi E, Sabin CA, Antonella d'Arminio Monforte M. Late diagnosis of HIV infection: epidemiological features, consequences and strategies to encourage earlier testing. JAIDS Journal of Acquired Immune Deficiency Syndromes. 2007;46:S3–S8. doi: 10.1097/01.qai.0000286597.57066.2b [DOI] [PubMed] [Google Scholar]
- 4.New Country Classifications | Data 2015. Available from: http://data.worldbank.org/news/new-country-classifications-2015.
- 5.UNAIDS report on the global AIDS epidemic 2013. accessed online: http://www.unaids.org/en/media/unaids/contentassets/documents/epidemiology/2013/gr2013/UNAIDS_Global_Report_2013_en.pdf.
- 6.Hsieh YH, Kelen GD, Laeyendecker O, Kraus CK, Quinn TC, Rothman RE. HIV Care Continuum for HIV-Infected Emergency Department Patients in an Inner-City Academic Emergency Department. Ann Emerg Med. 2015;66(1):69–78. doi: 10.1016/j.annemergmed.2015.01.001 ; PubMed Central PMCID: PMC4478148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Maheswaran H, Thulare H, Stanistreet D, Tanser F, Newell ML. Starting a home and mobile HIV testing service in a rural area of South Africa. J Acquir Immune Defic Syndr. 2012;59(3):e43–6. doi: 10.1097/QAI.0b013e3182414ed7 ; PubMed Central PMCID: PMCPMC4239475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tang N, Stein J, Hsia RY, Maselli JH, Gonzales R. Trends and characteristics of US emergency department visits, 1997–2007. Jama. 2010;304(6):664–70. doi: 10.1001/jama.2010.1112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rothman RE, Ketlogetswe KS, Dolan T, Wyer PC, Kelen GD. Preventive care in the emergency department: should emergency departments conduct routine HIV screening? a systematic review. Acad Emerg Med. 2003;10(3):278–85. . [DOI] [PubMed] [Google Scholar]
- 10.Kelen GD, Rothman RE. Emergency department–based HIV testing: too little, but not too late. Annals of emergency medicine. 2009;54(1):65–71. doi: 10.1016/j.annemergmed.2009.03.027 [DOI] [PubMed] [Google Scholar]
- 11.Force USPST. Final Update Summary: Human Immunodeficiency Virus (HIV) Infection: Screening—US Preventive Services Task Force. 2016.
- 12.Rothman RE. Current Centers for Disease Control and Prevention guidelines for HIV counseling, testing, and referral: critical role of and a call to action for emergency physicians. Ann Emerg Med. 2004;44(1):31–42. doi: 10.1016/S0196064404000678 . [DOI] [PubMed] [Google Scholar]
- 13.American College of Emergency P. HIV testing and screening in the emergency department. Ann Emerg Med. 2007;50(2):209 doi: 10.1016/j.annemergmed.2007.04.019 . [DOI] [PubMed] [Google Scholar]
- 14.Montoy JCC, Dow WH, Kaplan BC. Patient choice in opt-in, active choice, and opt-out HIV screening: randomized clinical trial. bmj. 2016;352:h6895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Haukoos JS, Hopkins E, Conroy AA, Silverman M, Byyny RL, Eisert S, et al. Routine opt-out rapid HIV screening and detection of HIV infection in emergency department patients. Jama. 2010;304(3):284–92. doi: 10.1001/jama.2010.953 [DOI] [PubMed] [Google Scholar]
- 16.Cochrane. LMIC Filters | Cochrane Effective Practice and Organisation of Care. 2016.
- 17.Nakanjako D, Kyabayinze DJ, Mayanja-Kizza H, Katabira E, Kamya MR. Eligibility for HIV/AIDS treatment among adults in a medical emergency setting at an urban hospital in Uganda. African health sciences. 2007;7(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ahmad S, Ellis JC, Kamwendo H, Molyneux EM. Impact of HIV infection and exposure on survival in critically ill children who attend a paediatric emergency department in a resource-constrained setting. Emerg Med J. 2010;27(10):746–9. doi: 10.1136/emj.2009.085191 . [DOI] [PubMed] [Google Scholar]
- 19.Nakanjako D, Kamya M, Daniel K, Mayanja-Kizza H, Freers J, Whalen C, et al. Acceptance of routine testing for HIV among adult patients at the medical emergency unit at a national referral hospital in Kampala, Uganda. AIDS and Behavior. 2007;11(5):753–8. doi: 10.1007/s10461-006-9180-9 [DOI] [PubMed] [Google Scholar]
- 20.Waxman MJ, Kimaiyo S, Ongaro N, Wools-Kaloustian KK, Flanigan TP, Carter EJ. Initial outcomes of an emergency department rapid HIV testing program in western Kenya. AIDS Patient Care STDS. 2007;21(12):981–6. doi: 10.1089/apc.2007.0075 . [DOI] [PubMed] [Google Scholar]
- 21.Ranney ML, Rennert-May E, Spitzer R, Chitai MA, Mamlin SE, Mabeya H. A novel ED-based sexual assault centre in western Kenya: description of patients and analysis of treatment patterns. Emerg Med J. 2011;28(11):927–31. doi: 10.1136/emj.2010.096412 . [DOI] [PubMed] [Google Scholar]
- 22.Teja VD, Lakshmi V, Sudha T. Impact of the human immunodeficiency virus infection on emergency medicine department in a tertiary care hospital in India. Indian J Med Microbiol. 2004;22(3):159–65. . [PubMed] [Google Scholar]
- 23.Teja VD, Sudha T, Lakshmi V. Emergency department based HIV screening: An opportunity for early diagnosis in high prevalent areas. Indian Journal of Medical Microbiology. 2008;26(2):167 doi: 10.4103/0255-0857.40535 [DOI] [PubMed] [Google Scholar]
- 24.Minz RW, Khairwa A, Aggarwal R, Kaur H, Singh S, Sharma A, et al. Cost analysis and benefits of opt-out HIV testing at a tertiary care centre in northern India. Int J STD AIDS. 2014;25(5):341–7. doi: 10.1177/0956462413507442 . [DOI] [PubMed] [Google Scholar]
- 25.Minz RW, Singh S, Varma S, Mathuria S, Aggrawal R, Sehgal S. Relevance of opt-out screening for HIV in emergency and pre-surgery patients in a tertiary care center in Northern India: A pilot study. Indian Journal of Pathology and Microbiology. 2010;53(2):287 doi: 10.4103/0377-4929.64334 [DOI] [PubMed] [Google Scholar]
- 26.Devi P, Arora U, Yadav S, Malhotra S. Seroprevalence of HIV infection among the patients attending various emergency departments in a tertiary care hospital. Indian journal of sexually transmitted diseases. 2010;31(1):27 doi: 10.4103/0253-7184.68997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Christensen A, Russ S, Rambaran N, Wright SW. Patient perspectives on opt-out HIV screening in a Guyanese emergency department. Int Health. 2012;4(3):185–91. doi: 10.1016/j.inhe.2012.03.001 . [DOI] [PubMed] [Google Scholar]
- 28.Waxman MJ, Muganda P, Carter EJ, Ongaro N. The role of emergency department HIV care in resource-poor settings: lessons learned in western Kenya. Int J Emerg Med. 2008;1(4):317–20. doi: 10.1007/s12245-008-0065-8 ; PubMed Central PMCID: PMCPMC2657254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sawe HR, Mfinanga JA, Ringo FH, Mwafongo V, Reynolds TA, Runyon MS. HIV counselling and testing practices for children seen in an urban emergency department of a tertiary referral hospital in Dar es Salaam, Tanzania: a retrospective cross-sectional study. BMJ open. 2016;6(2):e010298 doi: 10.1136/bmjopen-2015-010298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cox M, Shao J. Emergency medicine in a developing country: experience from Kilimanjaro Christian Medical Centre, Tanzania, East Africa. Emerg Med Australas. 2007;19(5):470–5. doi: 10.1111/j.1742-6723.2007.01012.x . [DOI] [PubMed] [Google Scholar]
- 31.Laeyendecker O, Rothman RE, Henson C, Horne BJ, Ketlogetswe KS, Kraus CK, et al. The effect of viral suppression on cross-sectional incidence testing in the johns hopkins hospital emergency department. J Acquir Immune Defic Syndr. 2008;48(2):211–5. doi: 10.1097/QAI.0b013e3181743980 ; PubMed Central PMCID: PMC2738975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nowacki AK, Landes M, Azazh A, Ritchie MLP. A review of published literature on emergency medicine training programs in low- and middle-income countries. International Journal of Emergency Medicine. 2013;6(1):1–10. doi: 10.1186/1865-1380-6-26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wallis LA, Garach SR, Kropman A. State of emergency medicine in South Africa. Int J Emerg Med. 2008;1(2):69–71. doi: 10.1007/s12245-008-0033-3 ; PubMed Central PMCID: PMC2657239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Obermeyer Z, Abujaber S, Makar M, Stoll S, Kayden SR, Wallis LA, et al. Emergency care in 59 low-and middle-income countries: a systematic review. Bulletin of the World Health Organization. 2015;93(8):577–86. doi: 10.2471/BLT.14.148338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Council S. National strategic plan on HIV, STIs and TB 2012Á2016. Pretoria, South Africa: South African National AIDS Council. 2012.
- 36.Gilks CF, Floyd K, Otieno LS, Adam AM, Bhatt SM, Warrell DA. Some effects of the rising case load of adult HIV-related disease on a hospital in Nairobi. J Acquir Immune Defic Syndr Hum Retrovirol. 1998;18(3):234–40. . [DOI] [PubMed] [Google Scholar]
- 37.Branson BM, Viall A, Marum E. Expanding HIV testing: back to the future. JAIDS Journal of Acquired Immune Deficiency Syndromes. 2013;63:S117–S21. doi: 10.1097/QAI.0b013e3182986f25 [DOI] [PubMed] [Google Scholar]
- 38.Krause J, Subklew-Sehume F, Kenyon C, Colebunders R. Acceptability of HIV self-testing: a systematic literature review. BMC public health. 2013;13(1):735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Choko AT, Desmond N, Webb EL, Chavula K, Napierala-Mavedzenge S, Gaydos CA, et al. The uptake and accuracy of oral kits for HIV self-testing in high HIV prevalence setting: a cross-sectional feasibility study in Blantyre, Malawi. PLoS Med. 2011;8(10):e1001102 doi: 10.1371/journal.pmed.1001102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Walensky RP, Bassett IV. HIV self-testing and the missing linkage. PLoS Med. 2011;8(10):e1001101 doi: 10.1371/journal.pmed.1001101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pai NP, Sharma J, Shivkumar S, Pillay S, Vadnais C, Joseph L, et al. Supervised and unsupervised self-testing for HIV in high-and low-risk populations: a systematic review. PLoS Med. 2013;10(4):e1001414 doi: 10.1371/journal.pmed.1001414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lodi S, Phillips A, Touloumi G, Geskus R, Meyer L, Thiébaut R, et al. Time from human immunodeficiency virus seroconversion to reaching CD4+ cell count thresholds< 200,< 350, and< 500 cells/mm3: assessment of need following changes in treatment guidelines. Clinical infectious diseases. 2011;53(8):817–25. doi: 10.1093/cid/cir494 [DOI] [PubMed] [Google Scholar]
- 43.Hardwicke R, Malecha A, Lewis ST, Grimes RM. HIV testing in emergency departments: a recommendation with missed opportunities. Journal of the Association of Nurses in AIDS Care. 2008;19(3):211–8. doi: 10.1016/j.jana.2008.03.005 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOC)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.