Abstract
MicroRNAs (miRNAs) have been proven to be involved in cell metastasis and angiogenesis by interaction with the target mRNAs. Evidence has been confirmed that miR-140-5p is a tumor suppressor in human cancers such as breast cancer. However, the potential molecular mechanism of miR-140-5p in breast cancer invasion and angiogenesis is still poorly understood. According to our study, we reported that miR-140-5p inhibited the tumor invasion and angiogenesis of breast cancer cells both in vitro and in vivo by targeting VEGF-A. The mRNA amount of miR-140-5p was decreased in the breast cancer clinical samples and breast cancer with metastasis compared with the corresponding adjacent normal tissues and cancer without metastasis. MiR-140-5p mimics and a negative control were transfected into human MCF-7 and MDA-MB-231 cells. Transwell chambers were used to detect the invasive ability of the cells, and the angiogenic ability was assessed by tube-formation assay. The markers of invasion and angiogenesis, VEGF-A, CD31 and MMP-9, were detected by using immunohistochemistry and western blot analysis in vivo. VEGF-A was verified as a possible target gene of miR-140-5p, and corroborated by dual-luciferase reporter and ELISA. Taken together, the study elucidates the molecular mechanisms by which miR-140-5p inhibits breast cancer metastasis and angiogenesis, and provides a potent evidence for the development of a novel microRNA-targeting anticancer strategy for breast cancer patients.
Subject terms: Targeted therapies, Cell biology
Introduction
MicroRNAs (miRNAs) are small noncoding RNA sequences of 19–25 nucleotides that play a role in the post-transcriptional control of gene expression.1 Recently, it has been reported that miRNAs are involved in the control of cell proliferation, apoptosis, cell cycle distribution, cellular migration and angiogenesis.2, 3, 4, 5 Accumulating evidence shows that miRNAs are often abnormally expressed in diverse cancers, including leukemias and lymphomas,6 colorectal cancer,7 liver cancer,8 head and neck cancer9 and breast cancers.10
Breast cancer is the main cause for death related to cancer for women all over the world. In China, a total of 1 779 500 new female cancer cases were reported in 2015; breast cancer itself is predicted to take 15% of all new cancers in women.11 Breast cancer patients frequently develop metastases at multiple sites,12 commonly to the lung, brain and bone.13, 14, 15, 16, 17 Previous studies have reported that miR-34a, miR-502 and miR-22 play the role as tumor suppressors in breast cancer,18, 19, 20 but miR-32 and miR-191 function as oncogenes in breast cancer and lung cancer.21, 22 miR-140 appeared to be positively controlled by retinoblastoma and to antagonize the effect of Rb depletion on spherogenesis and tumorigenesis,23 and it can regulate the cell invasion of esophageal cancer via controlling Slug expression.24 However, the correlation of miR-140 expression with invasion and angiogenesis in breast cancer has not been reported.
Vascular endothelial growth factor (VEGF), a main supervisor of angiogenesis, is a dimeric glycoprotein secreted by many kinds of cells, including cancer cells and peripheral blood mononuclear cells.25 VEGF is a member of the family of platelet-derived growth factors, including VEGF-A, VEGF-B, VEGF-C, VEGF-D and VEGF-E, and placental growth factors.26 On one hand, expansion of the VEGF-A gene is an essential mechanism for elevated VEGF-A protein expression, and on the other hand it is also a poor prognostic element for tumor-free survival in osteosarcomas.27 Thus, VEGF-A is an appealing objective for cancer therapy.
The findings of this study suggested that miR-140-5p downregulation was greatly related to clinical grades and metastasis in breast cancer tissues, and we aim to study the mechanisms of miR-140-5p in angiogenesis by a series of studies in vitro and vivo. By analyzing bioinformatics information (TargetScan) and validating with experiences, it was found that VEGF-A was a direct objective of miR-140-5p and miR-140-5p, which functioned as a tumor suppressor in breast cancer metastasis and angiogenesis by inhibiting the expression of VEGF-A.
Materials and methods
Patient samples
A total of 40 breast cancer tissues (Stage I: 12 cases; Stage II: 15 cases; Stage III: 13 cases) from 40 patients who did not receive chemotherapy or radiotherapy before the surgery, including 25 cases with lymph node metastasis, and another 30 normal breast tissues obtained from the area adjacent to the cancer were used. After the surgery, tissue samples were immediately deposited at −80 °C until RNA was extracted. All of the samples were confirmed by three pathologists. This study was approved by the Dalian medical university’s ethics committee.
Cell culture
MCF-7 and MDA-MB-231 cells were obtained from ATCC (Manassas, VA, USA) and cultivated in Dulbecco's Modified Eagle's Medium (Gibco Laboratories, Frederick, MD, USA) with 10% fetal bovine serum (Gibco) and one percent penicillin/streptomycin (Gibco) at 37 °C and 5% CO2; the medium was changed every 2 days.
Vector construction and dual-luciferase reporter assay
The dual-luciferase reporter plasmid fused with the wild-type or mutant 3′-UTR segment of human VEGF-A was obtained from GenePharma (Shanghai, China). For verification of the regulation of VEGF-A by miR-140 mimics, a total of 7 × 104 HEk-293T cells stably transfected with -miR-140-5p mimics or miR-NC/N-control (scramble mimic) were seeded into 24-well plates. On the following day, the cells were transfected with 100 ng of the VEGF-A Wt or Mt 3′-UTR reporter plasmids, and 50 nM miR-140-5p mimics or scramble mimics with Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA, USA). After 2 days, cells were harvested with passive lysis buffer (Invitrogen) and the luciferase activity was calculated by the Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA) according to the manufacturer’s instructions.
qRT-PCR analysis of miR-140-5p and VEGF-A
Lentiviral vectors were used for transfection. The vectors mediated miR-14-5p were performed and result was constructed along with the protocol (Invitrogen). miR-140-5p mimics and Negative controls (Invitrogen) were transfected into breast cancer cells with Lipofectamine 2000 reagent (Invitrogen). MiR-140-5p and VEGF-A mRNA expression levels were determined by quantitative real-time RT–PCR (qRT–PCR) with the 7900HT Real-Time PCR System (Applied Biosystems, Thermo Fisher Scientific, Carlsbad, CA, USA). qRT–PCR analysis was performed to detect miRNA or mRNA expression. A measure of 1 μg of total RNA was transcribed to cDNA with a RevertAid Premium First Strand cDNA Synthesis Kit (Fermentas, Burlington, Canada). The cDNA was intensified by immediate PCR with a SYBR Green PCR master mix kit (Invitrogen, California, USA). The following primers were used: miR-140-5p forward primer 5′-GAGTGTCAGTGGTTTTACCCT-3′, reverse primer 5′-GCAGGGTCCGAGGTATTC-3′; VEGF-A forward primer 5′-TTTCTGCTGTCTTGGGTGCATTGG-3′, reverse primer 5′-ACCACTTCGTGATGATTCTGCCCT-3′; β-actin forward primer 5′-TCAAGATCATT-GCTCCTCCTG-3′, reverse primer 5′-CTG-CTTGCTGATCCACATCTG-3′; U6 forward primer 5′-TGCGGGTGCTCGCTTCGGCAGC-3′, reverse primer 5′-CCAGTGCAGGGTCCGAGGT-3′.28 U6 or β-actin was applied as an endogenous regulator. Gene expression was calculated in triplicate, and the results were analyzed using the 2−ΔΔCt method and normalized to the controls.
Preparation of tumor-conditioned medium
MCF-7 cells were transfected with miR-140-5p mimics or miR-NC within 48 h, and then the cells were washed with cold phosphate-buffered saline (PBS) three times and maintained in medium without serum. After 2 days, tumor-conditioned medium (TCM) was collected after centrifugation at 12 000 g for 10 min at 4 °C and then kept at −80 °C.
ELISA
MCF-7 and MDA-MB-231 cells (5 × 103, 6 × 103 cells per well, respectively) in 96-well plates were transfected with miR-140-5p mimics and N-Control for 48 h. Culture medium was collected and secreted VEGF-A was detected with the human VEGF-A ELISA kit (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions. The VEGF-A secretion was measured with a standard curve.
HUVEC tube-formation assay
HUVECs (7 × 105) were added into the mixture of TCM (300 μl) and Dulbecco's Modified Eagle's Medium (300 μl) with 10% fetal bovine serum and seeded on a 24-well plate coated with matrigel (300 μl per well, BD Biosciences, San Jose, CA, USA). After 2 days of incubation at 37 °C, tube construction was viewed under a microscope (Nikon Corporation, Tokyo, Japan). The number of branches of the associated tubes was calculated and compared within different groups.
Western immunoblot analysis
At 48 h after transfection, cells were lysed with RIPA buffer (Invitrogen) and western immunoblotting was performed according to the standard process. The major antibodies applied to the analysis were anti-human and mouse VEGF-A antibody (1:500; Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit anti-human CD31 antibody (1:500, Abcam, Cambridge, MA, USA), rabbit anti-Ki-67 (1:500, Santa Cruz Biotechnology), anti-MMP-9 mouse antibody (1:700, Abcam) and mouse anti-human GAPDH antibody (1:1000, Santa Cruz Biotechnology) as a loading regulator.
Tumor cell metastasis and invasion assay
A density of 2 × 104 MCF-7 and MDA-MB-231 cells transfected with miR-140 mimic or N-regulators were added into the top chambers with or without matrigel. After 12 h, cells at the bottom were stained with crystal violet stain and were observed and counted under the microscope in five different fields.
Cell proliferation assay
MCF-7 and MDA-MB-231 cells were added into 96-well plates at a density of 3 × 104 cells per well with 100 μl of medium. Cell Counting Assay Kit-8 reagent (CCK-8, Diojindo Laboratories, Kumamoto, Japan) was added as per protocol. The absorbance at 490 nm was calculated on a spectrophotometer. All experiments were repeated with three independent experiments.
Colony formation assay
Single suspended cells were seeded in six-well plates at 300 cells per well. After 10 days, the colonies were stained with 0.02% crystal violet for 10 min and washed with PBS three times. The number of colonies was counted under the microscope. All experiments were repeated three times.
Tumor xenograft assay
MCF-7 cells transfected with miR-140-5p mimics or negative regulator vector (1 × 106) were injected into 8-week-old nude mice (n=5 per group). Tumor volume was calculated as follows: Tumor volume=(width2 × length)/2. After 8 weeks, all mice were killed, and tumor volume and weight were determined. The research was supervised by the Dalian medical university’s ethics committee.
Immunohistochemistry
The xenograft tumor slices from blocked formalin-fixed and paraffin-embedded tissue were checked for immunostaining for VEGF-A and CD31. After deparaffinization, hydration and heat-induced antigen retrieval, the tissue parts were nurtured for 1 h at room temperature with VEGF-A and CD31 antibodies both diluted at 1:200. Biotinylated secondary antibody (DAKO, Carpinteria, CA, USA) was used and binding was found with the substrate DAB against a hematoxylin counterstain.
Statistical analysis SPSS
Standard version 18.0 (SPSS, Chicago, IL, USA) was applied to analyze the data. The data were expressed as mean±s.d., and all results were studied. P-value <0.05 was considered to be statistically significant. For comparison of two independent data sets, Student’s t-test was used.
Results
Downregulated miR-140-5p in primary breast cancer tissues, and reduced expression of miR-140-5p connected with advanced clinical procedure and poor patient prognosis
To investigate the role of miR-140-5p expression in patients with breast cancer, we examined the expression level of miR-140-5p in 40 breast cancer tissues and 30 adjacent normal tissues. The whole RNA was extracted from breast samples and analyzed by RT–PCR in real time. As the data demonstrated, breast cancer had reduced miR-140-5p expression by at least fivefold as compared with the normal tissues in Figure 1a (P<0.05). In addition, miR-140-5p was associated with tumor progression as it was decreased in the sequence from stage I to III (Figure 1b, P<0.05). Meanwhile, it was shown that the expression of miR-140-5p was lower in lymph node metastasis tissues compared with breast cancer tissues without metastasis (Figure 1c, P<0.05). In addition, the expression levels of VEGF-A mRNA dominated by qRT–PCR in breast cancer tissue samples were further analyzed. As shown in the data, VEGF-A expression levels were inversely correlated with those of miR-140-5p in breast cancer tissues (R2=−0.834, P<0.05; Figure 1d).
Figure 1.
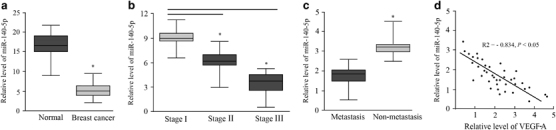
miR-140-5p is downregulated in breast cancer clinical samples. (a) The miR-140-5p expression level is decreased in breast tumor tissues compared with normal tissues. (b) miR-140-5p is differentially expressed in the early-stage (stages I and II) group compared with the stage III group. (c) MiR-140-5p is differentially expressed in the lymph node metastasis group compared with the group without metastasis. (d) Expression patterns of VEGF-A with miR-140-5p exhibited a significant negative correlation.
MiR-140-5p exactly inhibits VEGF-A gene expression via targeting its 3′-UTR
TargetScan was used to study putative protein-coding gene objectives of miR-140-5p; the VEGF-A gene was recognized as a possible target and a dual-luciferase reporter assay was conducted. The wild-type (WT) and mutant (MUT) versions of the VEGF-A 3′-UTR containing site-directed mutations in the putative miR-140-5p target places were cloned into the reporter plasmids (Figure 2a). Luciferase reporter assay showed that the luciferase activity of the wild-type VEGF-A 3′-UTR reporter gene was significantly reduced in HEK-293T cells transfected with miR-140-5p mimics, whereas the mutant reporter gene was not affected (Figure 2b), which confirmed that VEGF-A is an exact aim of miR-140-5p. We further analyzed the transcript and protein expression of VEGF-A in MCF-7 and MDA-MB-231 cells transfected with scrambled regulation or miR-140-5p mimics. The data showed that overexpression of miR-140-5p suppresses the expression mRNA and intracellular protein of VEGF-A (Figures 2c and d). In addition, the secreted VEGF-A was analyzed by ELISA. VEGF-A protein expression was reduced in cells transfected with miR-140-5p mimics in TCM (Figure 2e).
Figure 2.
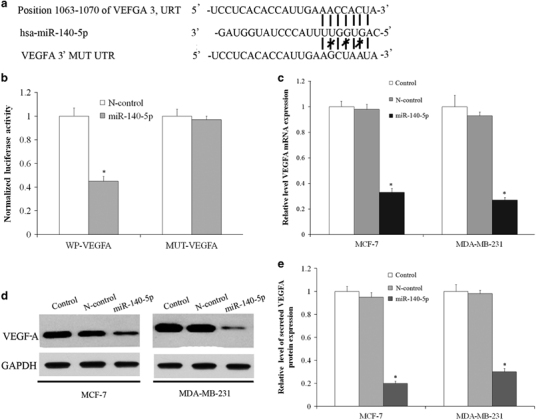
VEGF-A is the direct target of miR-140-5p. (a) A putative miR-140-5p-binding site exists in the 3′-UTR of VEGF-A mRNA, and three point mutations were generated in the binding site. (b) Luciferase reporter assay for HEK-293T WT and Mut 3′-UTRs. (c) miR-140-5p re-expression reduced VEGF-A mRNA levels as compared with the negative control in MCF-7 and MDA-MB-231 cells. (d) The expression of miR-140-5p in MCF-7 and MDA-MB-231 cells decreased the intracellular VEGF-A protein levels, as shown by western blot analysis. (e) The expression of miR-140-5p in MCF-7 and MDA-MB-231 cells decreased secreted VEGF-A protein levels, as shown by ELISA.
miR-140-5p regulates biological behavior in breast cancer cell lines
Overexpression of miR-140-5p was observed to markedly reduce the cell increase compared with its negative controls, as shown in CCK-8 assays (Figure 3a). Colony-formation assays demonstrated that ectopic expression of miR-140-5p led to nearly 70 and 60% reduction in colony number in MCF-7 and MDA-MB-231 cells, separately, after 10 days of culturing (Figure 3b). Given our initial observation that miR-140-5p expression correlated with breast cancer invasiveness, we investigated whether miR-140-5p regulated cellular migration and invasion in vitro. After transfecting with miR-140-5p mimics, it was found to be greatly inhibited for the movement and invasion of these cells compared with N-control cells (Figures 3c and d). Hence, it can be concluded that miR-140 acts on some crucial genes controlling proliferation and metastases in breast cancer.
Figure 3.
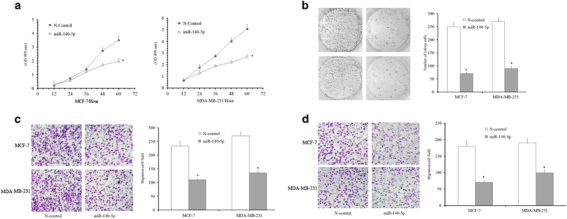
Overexpression of miR-140-5p suppresses the proliferation and invasion in breast cancer. (a) Cells transfected with miR-140-5p oligo exhibit a significant reduction in cellular proliferation compared with cells transfected with miRNA scramble control at 60 h after plating. (b) The growth of MCF-7 and MDA-MB-231 cells was detected by colony formation after transfection with miR-140-5p or a negative control after 10 days. (c and d) Migration and invasion assays were performed. Cells migrating into lower chambers were stained with crystal violet and imaged. Experiments were repeated three times.
Reduced capacity of cells to advance HUVEC tube construction in vitro by MiR-140
HUVEC tube construction assays were performed with TCM from MCF-7 and MDA-MB-231 cells. HUVECs were dispersed in TCM and cultivated on the matrigel-coated wells to build capillary tubes in 2 days. Obviously, well-associated and more mature capillary tubes that were derived from cells were built in the TCM transfected with N-control, as illustrated in Figure 4a. However, less well-formed capillary-like structures were built for HUVECs in the TCM derived from cells transfected using miR-140 mimics (Figure 4b).
Figure 4.
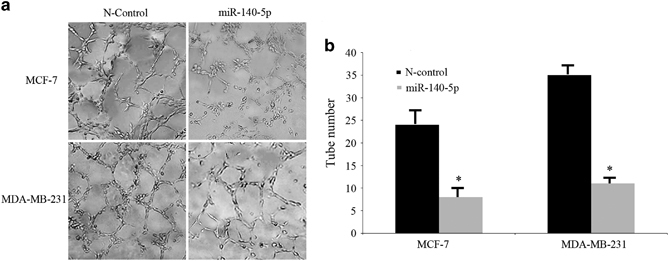
MiR-140-5p reduces the capability of tumor cells to promote HUVEC tube formation. (a and b) Representative images of tube formation and the relative tube number were measured.
miR-140-5p suppresses breast cancer metastasis and angiogenesis in vivo
We injected MCF-7 cells with miR-140-5p stably expressing mimics or negative regulator vector into the nude mice. After 8 weeks, the mice were killed, and the tumors were examined (Figure 5a). The results showed that the weight and volume of tumor nodules derived from miR-140-5p-overexpressing cells were less than those of the negative control group (Figure 5b). A histologic analysis of the tumors confirmed the angiogenesis by immunohistochemistry. As shown in Figure 5c, the protein-level expression of VEGF-A and MMP-9 in N-control group was more than the miR-140-5p mimics group. Compared with the N-control and miR-140-5p groups, the positive cell percentage of VEGF-A is 73.2±3.3% and 24.4±4.1%, and the positive cell percentage of MMP-9 is 62.2±2.1% and 22.1±6.1%, respectively. Furthermore, the protein expression stage of VEGF-A, CD31, Ki-67 and MMP-9 compared with the negative regulation group by western blot analysis (Figure 5d), as the data showed that VEGF-A, CD31, Ki-67 and MMP-9 were greatly decreased in the miR-140-5p group.
Figure 5.
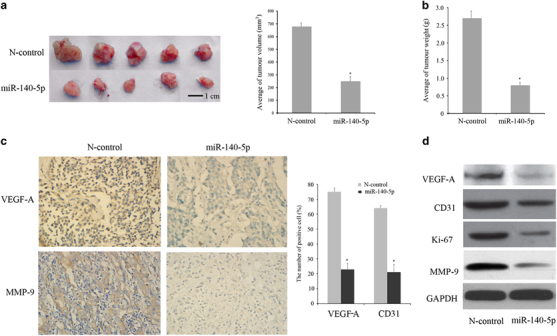
miR-140-5p inhibits tumor growth and angiogenesis in vivo. (a) Compared with the N-control group, miR-140-5p mimics inhibited the tumor volume (bar=10mm). (b) miR-140-5p mimics inhibited the weight of the tumor. (c) The expression of VEGF-A and MMP-9 in tumor tissues and the number of positive cells. (d) The protein expression of VEGF-A, CD31, Ki-67 and MMP-9 in N-control and miR-140-5p mimics groups by western blot analysis.
Discussion
MiRNAs function mainly in carcinogenic programs by controlling genes at the post-transcriptional level, such as tumor cell development, differentiation, metastasis and angiogenesis.29 MiRNAs play the role as either tumor suppressors or oncogenes in regulating the expression of immigration and angiogenesis-related genes. For example, miR-101 inhibits nasopharyngeal carcinoma cell angiogenesis and metastasis by directly repressing ITGA3,30 and tumor growth and angiogenesis is suppressed by miR-203 targeting VEGF-A in cervical cancer.31 While, MicroRNA-30d boosts tumor angiogenesis development by MYPT1/c-JUN/VEGF-A in prostate cancer.32
Here focusing on the current study, it showed that miR-140-5p expression was greatly lack of regulation in breast cancer tissues. Meanwhile, decreased expression of miR-140-5p was found in advanced clinical stage and metastatic cancer tissues. The precise mechanism of miR-140 deregulation is still unclear. However, we speculate that the function of miR-140 is fundamentally linked with cancer stem cell (CSC).33 CSC was usually known for its high tumorigenicity and metastasis.34 Some reports indicated that miR-140-5p was a key regulator in CSC progression and metastasis, and it has been described to target the stem cell pathways to modulate cancer stem cell formation, such as breast cancer stem cell and colorectal cancer stem cell.35, 36 In particular, miR-140 functions greatly in controlling stem cell signaling in common breast epithelium and in ductal carcinoma in situ (DCIS), and miR-140 loss can overthrow a completely important regulator of breast tissue differentiation possibilites.35
Our studies demonstrated that miR-140-5p mimics reduced cell proliferation and colony formation, and the findings are consistent with previously published reports in colorectal cancer.37 In order to consist with the results in vitro, the tumor xenografts were analyzed. In our study, MCF-7 transfected with miR-140 mimics in the matrigel plug bioassay in vivo greatly reserved the weight and volume of the tumor compared with the N-Control group. We found the expression of Ki-67 to be downregulated in the miR-140-5p group after overexpression of miR-140-5p. Usually, the highest expression of Ki-67 was considered as an indicator of poor prognosis in many kinds of cancers, such as breast cancer. In addition, it is a useful tool to determine the potential of tumor proliferation.38 Previous findings indicated that Ki-67 protein expression is closely related to the cell cycle. Coincidentally, miR-140-5p may be a cyclin regulator. In colorectal cancer, 140-5p overexpression led to a growth in G1 stage with a decrease in S phase, and Yan et al. reported that miR-140-5p had other targets such as Pin1 and cyclin D1 to reduce tumor growth in hepatocellular carcinoma.39 However, further study is necessary to determine the relationship between miR-140-5p and cyclin protein.
Furthermore, there is an inverse relationship between the expression of miR-140-5p and VEGF-A in breast cancer tissues. VEGF-A is a well-known regulator of the angiogenesis and metastasis of human cancers. Metastasis is the main reason for the unsatisfactory prognosis of breast cancer patients. Metastasis and blood vessels generally go along with each other and share many characteristics, including molecular pathways.40 In this study, we further determined whether miR-140-5p could affect angiogenesis by targeting VEGA-A by using the HUVEC assay. We found that MiR-140-5p blocked the tube-forming ability of HUVECs in vitro. In vivo, the expression of CD31 was downregulated as miR-140-5p was overexpressed. Generally, CD31 expression on the endothelial cells is estimated on the basis of the number of the Micro Vessel Density (MVD),41 and adequate blood supply is essential for tumor growth, invasion and metastasis.42 Therefore, we speculate that it is a valid way to inhibit tumor angiogenesis by upregulating miR-140-5p. Although the MCF-7 cell unsuccessfully migrated to other parts of the nude mice in the xenograft model, we detected the expression of VEGF-A and MMP-9 in the tumor tissues. MMP-9 has been constantly associated with migration, invasion and angiogenesis in many cancer subtypes such as breast cancer.43 The immunohistochemical analysis of VEGF-A and MMP-9 in tumor xenografts derived from animals treated with miR-140 mimics showed a clear decrease in positive density, with a 50 and 40% reduction in tumor. The latest study revealed that, compared with regulators, which signifies its function in angiogenesis, the FAK activity is demonstrated to be exactly connected with the VEGF-mediated vasculature in mammary tumors. That CuB suppressed cancer cell metastasis and angiogenesis through the downregulation of VEGF/FAK/MMP signaling with both in vitro and in vivo models.44 However, the interrelationship between CuB/FAK and miR-140-5p should be investigated in further work.
In conclusion, here in this work, the potential clinical relevance of miR-140-5p in breast cancer has been identified, wherein it promotes a variety of functions such as decreased cell viability, colony formation and invasion. MiR-140-5p suppresses VEGF-A expression, which then leads to inhibit angiogenesis formation, along with the downregulation of proteins such as MMP-9. Taken together, miR-140-5p may act as a biomarker in the diagnosis and treatment of breast cancer.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 81502561), and Educational Department of Liaoning Province (No. L2015152).
Competing interests
The authors declare no conflict of interest.
References
- 1.Giordano S, Columbano A. MicroRNAs: new tools for diagnosis, prognosis, and therapy in hepatocellular carcinoma? Hepatology. 2013;57:840–847. doi: 10.1002/hep.26095. [DOI] [PubMed] [Google Scholar]
- 2.Pan C, Wang D, Zhang Y, Yu W. MicroRNA-1284 inhibits cell viability and induces apoptosis of ovarian cancer cell line OVCAR3. Oncol Res. 2016;24:429–435. doi: 10.3727/096504016X14685034103518. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 3.Hydbring P, Wang Y, Fassl A, Li X, Matia V, Otto T. Cell-Cycle-Targeting MicroRNAs as Therapeutic Tools against Refractory Cancers. Cancer Cell. 2017;31:576–590.e8. doi: 10.1016/j.ccell.2017.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cai H-K, Chen X, Tang Y-H, Deng Y-C. MicroRNA-194 modulates epithelial–mesenchymal transition in human colorectal cancer metastasis. Oncotargets Ther. 2017;10:1269. doi: 10.2147/OTT.S125172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guan B, Wu K, Zeng J, Xu S, Mu L, Gao Y. Tumor-suppressive microRNA-218 inhibits tumor angiogenesis via targeting the mTOR component RICT OR in prostate cancer. Oncotarget. 2017;8:8162. doi: 10.18632/oncotarget.14131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Valleron W, Laprevotte E, Gautier E, Quelen C, Demur C, Delabesse E. Specific small nucleolar RNA expression profiles in acute leukemia. Leukemia. 2012;26:2052–2060. doi: 10.1038/leu.2012.111. [DOI] [PubMed] [Google Scholar]
- 7.Fantini S, Salsi V, Reggiani L, Maiorana A, Zappavigna V. The miR-196b miRNA inhibits the GATA6 intestinal transcription factor and is upregulated in colon cancer patients. Oncotarget. 2016;8:4747–4759. doi: 10.18632/oncotarget.13580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang Z, Li X, Sun W, Yue S, Yang J, Li J. Loss of exosomal miR-320a from cancer-associated fibroblasts contributes to HCC proliferation and metastasis. Cancer Lett. 2017;397:33–42. doi: 10.1016/j.canlet.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 9.Guan G-f, Zhang D-j, Wen L-j, Xin D, Liu Y, Yu D-j. Overexpression of lncRNA H19/miR-675 promotes tumorigenesis in head and neck squamous cell carcinoma. Int J Med Sci. 2016;13:914. doi: 10.7150/ijms.16571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ke J, Zhao Z, Hong S-H, Bai S, He Z, Malik F. Role of microRNA221 in regulating normal mammary epithelial hierarchy and breast cancer stem-like cells. Oncotarget. 2015;6:3709. doi: 10.18632/oncotarget.2888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F. CA Cancer J Clin. 2015;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 12.Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’hypothesis revisited. Nat Rev Cancer. 2003;3:453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- 13.Hu J, Li G, Zhang P, Zhuang X, Hu G. A CD44v+ subpopulation of breast cancer stem-like cells with enhanced lung metastasis capacity. Cell Death Dis. 2017;8:e2679. doi: 10.1038/cddis.2017.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jackson W, Sosnoski DM, Ohanessian SE, Chandler P, Mobley A, Meisel KD. Role of Megakaryocytes in Breast Cancer Metastasis to Bone. Cancer Res. 2017;77:1942–1954. doi: 10.1158/0008-5472.CAN-16-1084. [DOI] [PubMed] [Google Scholar]
- 15.Gordon AC, Salem R, Lewandowski RJ. Yttrium-90 radioembolization for breast cancer liver metastases. J Vasc Interv Radiol. 2016;27:1316. doi: 10.1016/j.jvir.2016.06.016. [DOI] [PubMed] [Google Scholar]
- 16.Custódio-Santos T, Videira M, Brito MA. Brain metastasization of breast cancer. Biochim Biophys Acta. 2017;1868:132–147. doi: 10.1016/j.bbcan.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 17.Rhu J, Lee SK, Kil WH, Lee JE, Nam SJ. Surgery of primary tumour has survival benefit in metastatic breast cancer with single‐organ metastasis, especially bone. ANZ J Surg. 2015;85:240–244. doi: 10.1111/ans.12548. [DOI] [PubMed] [Google Scholar]
- 18.Do Yeon Kim EYP, Chang E, Kang H-G, Koo Y, Lee EJ, Ko JY. A novel miR-34a target, protein kinase D1, stimulates cancer stemness and drug resistance through GSK3/β-catenin signaling in breast cancer. Oncotarget. 2016;7:14791. doi: 10.18632/oncotarget.7443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu B, Zhang X, Song F, Zheng H, Zhao Y, Li H. MiR-502/SET8 regulatory circuit in pathobiology of breast cancer. Cancer Lett. 2016;376:259–267. doi: 10.1016/j.canlet.2016.04.008. [DOI] [PubMed] [Google Scholar]
- 20.Xia S, Zhang G, Liu Z, Tian H, He Y, Meng C. MicroRNA-22 suppresses the growth, migration and invasion of colorectal cancer cells through a Sp1 negative feedback loop. Oncotarget. 2017;8:36266–36278. doi: 10.18632/oncotarget.16742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xia H, Long J, Zhang R, Yang X, Ma Z. MiR-32 contributed to cell proliferation of human breast cancer cells by suppressing of PHLPP2 expression. Biomed Pharmacother. 2015;75:105–110. doi: 10.1016/j.biopha.2015.07.037. [DOI] [PubMed] [Google Scholar]
- 22.Zhao J, Qiao CR, Ding Z, Sheng YL, Li XN, Yang Y. A novel pathway in NSCLC cells: miR-191, targeting NFIA, is induced by chronic hypoxia, and promotes cell proliferation and migration. Mol Med Rep. 2017;15:1319–1325. doi: 10.3892/mmr.2017.6100. [DOI] [PubMed] [Google Scholar]
- 23.Yoshida A, Kitajima S, Li F, Cheng C, Takegami Y, Kohno S. MicroRNA-140 mediates RB tumor suppressor function to control stem cell-like activity through interleukin-6. Oncotarget. 2017;8:13872. doi: 10.18632/oncotarget.14681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li W, Jiang G, Zhou J, Wang H, Gong Z, Zhang Z. Down-regulation of miR-140 induces EMT and promotes invasion by targeting Slug in esophageal cancer. Cell Physiol Biochem. 2014;34:1466–1476. doi: 10.1159/000366351. [DOI] [PubMed] [Google Scholar]
- 25.Petrovic N. Targeting angiogenesis in cancer treatments: where do we stand? J Pharm Pharm Sci. 2016;19:226–238. doi: 10.18433/J30033. [DOI] [PubMed] [Google Scholar]
- 26.Shinkaruk S, Bayle M, Lain G, Deleris G. Vascular endothelial cell growth factor (VEGF), an emerging target for cancer chemotherapy. Curr Med Chem. 2003;3:95–117. doi: 10.2174/1568011033353452. [DOI] [PubMed] [Google Scholar]
- 27.Yang J, Yang D, Sun Y, Sun B, Wang G, Trent JC. Genetic amplification of the vascular endothelial growth factor (VEGF) pathway genes, including VEGFA, in human osteosarcoma. Cancer. 2011;117:4925–4938. doi: 10.1002/cncr.26116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kang H-W, Wang F, Wei Q, Zhao Y-F, Liu M, Li X. miR-20a promotes migration and invasion by regulating TNKS2 in human cervical cancer cells. FEBS Lett. 2012;586:897–904. doi: 10.1016/j.febslet.2012.02.020. [DOI] [PubMed] [Google Scholar]
- 29.Sun X, Charbonneau C, Wei L, Chen Q, Terek RM. miR-181a targets RGS16 to promote chondrosarcoma growth, angiogenesis, and metastasis. Mol Cancer Res. 2015;13:1347–1357. doi: 10.1158/1541-7786.MCR-14-0697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tang X-R, Wen X, He Q-M, Li Y-Q, Ren X-Y, Yang X-J. MicroRNA-101 inhibits invasion and angiogenesis through targeting ITGA3 and its systemic delivery inhibits lung metastasis in nasopharyngeal carcinoma. Cell Death Dis. 2017;8:e2566. doi: 10.1038/cddis.2016.486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhu X, Er K, Mao C, Yan Q, Xu H, Zhang Y. miR-203 suppresses tumor growth and angiogenesis by targeting VEGFA in cervical cancer. Cell Physiol Biochem. 2013;32:64–73. doi: 10.1159/000350125. [DOI] [PubMed] [Google Scholar]
- 32.Lin Z-y, Chen G, Zhang Y-q, He H-c, Liang Y-x, Ye J-h. MicroRNA-30d promotes angiogenesis and tumor growth via MYPT1/c-JUN/VEGFA pathway and predicts aggressive outcome in prostate cancer. Mol Cancer. 2017;16:48. doi: 10.1186/s12943-017-0615-x. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 33.Zhang Y, Eades G, Zhou Q. Examining estrogen regulation of cancer stem cells through multicolor lineage tracing. Methods Mol Biol. 2014;1204:35–44. doi: 10.1007/978-1-4939-1346-6_4. [DOI] [PubMed] [Google Scholar]
- 34.Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008;8:755–768. doi: 10.1038/nrc2499. [DOI] [PubMed] [Google Scholar]
- 35.Wolfson B, Eades G, Zhou Q. Roles of microRNA-140 in stem cell-associated early stage breast cancer. World J Stem Cells. 2014;6:591–597. doi: 10.4252/wjsc.v6.i5.591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhai H, Fesler A, Ba Y, Wu S, Ju J. Inhibition of colorectal cancer stem cell survival and invasive potential by hsa-miR-140-5p mediated suppression of Smad2 and autophagy. Oncotarget. 2015;6:19735–19746. doi: 10.18632/oncotarget.3771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang W, Zou C, Pan L, Xu Y, Qi W, Ma G. MicroRNA-140-5p inhibits the progression of colorectal cancer by targeting VEGFA. Cell Physiol Biochem. 2015;37:1123–1133. doi: 10.1159/000430237. [DOI] [PubMed] [Google Scholar]
- 38.Falato C, Lorent J, Tani E, Karlsson E, Wright PK, Bergh J. Ki67 measured in metastatic tissue and prognosis in patients with advanced breast cancer. Breast Cancer Res Treat. 2014;147:407–414. doi: 10.1007/s10549-014-3096-2. [DOI] [PubMed] [Google Scholar]
- 39.Yan X, Zhu Z, Xu S, Yang L-n, Liao X-H, Zheng M. MicroRNA-140-5p inhibits hepatocellular carcinoma by directly targeting the unique isomerase Pin1 to block multiple cancer-driving pathways. Sci Rep. 2017;7:45915. doi: 10.1038/srep45915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jun HY, Lee YH, Juhng SK, Lee MS, Oh J, Yoon KH. Micro-CT measurements of tumoral vessels supplied by portal circulation in hepatic colorectal metastasis mouse model. Microsc Res Tech. 2014;77:415–421. doi: 10.1002/jemt.22361. [DOI] [PubMed] [Google Scholar]
- 41.Adelipour M, Babaei F, Mirzababaei M, Allameh A. Correlation of micro vessel density and c-Myc expression in breast tumor of mice following mesenchymal stem cell therapy. Tissue Cell. 2017;49(2PtB):315–322. doi: 10.1016/j.tice.2017.01.007. [DOI] [PubMed] [Google Scholar]
- 42.Pulford E, Hocking A, Griggs K, McEvoy J, Bonder C, Henderson DW, Klebe S . Vasculogenic mimicry in malignant mesothelioma: an experimental and immunohistochemical analysis. Pathology 2016; 48. [DOI] [PubMed]
- 43.Xu X-D, Shen H-B, Zhu L, Lu J-Q, Zhang L, Luo Z-Y. Anti-rhoc sirnas inhibit the proliferation and invasiveness of breast cancer cells via modulating the Kai1, MMP9, and cXcr4 expression. Oncotargets Ther. 2017;10:1827. doi: 10.2147/OTT.S93164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sinha S, Khan S, Shukla S, Lakra AD, Kumar S, Das G. Cucurbitacin B inhibits breast cancer metastasis and angiogenesis through VEGF-mediated suppression of FAK/MMP-9 signaling axis. Int J Biochem Cell Biol. 2016;77:41–56. doi: 10.1016/j.biocel.2016.05.014. [DOI] [PubMed] [Google Scholar]


