Abstract
Background
Natriuretic peptides are recognized as important predictors of cardiovascular events in patients with heart failure, but less is known about their prognostic importance in patients with acute coronary syndrome. We sought to determine whether B‐type natriuretic peptide (BNP) and N‐terminal prohormone B‐type natriuretic peptide (NT‐proBNP) could enhance risk prediction of a broad range of cardiovascular outcomes in patients with acute coronary syndrome and type 2 diabetes mellitus.
Methods and Results
Patients with a recent acute coronary syndrome and type 2 diabetes mellitus were prospectively enrolled in the ELIXA trial (n=5525, follow‐up time 26 months). Best risk models were constructed from relevant baseline variables with and without BNP/NT‐proBNP. C statistics, Net Reclassification Index, and Integrated Discrimination Index were analyzed to estimate the value of adding BNP or NT‐proBNP to best risk models. Overall, BNP and NT‐proBNP were the most important predictors of all outcomes examined, irrespective of history of heart failure or any prior cardiovascular disease. BNP significantly improved C statistics when added to risk models for each outcome examined, the strongest increments being in death (0.77–0.82, P<0.001), cardiovascular death (0.77–0.83, P<0.001), and heart failure (0.84–0.87, P<0.001). BNP or NT‐proBNP alone predicted death as well as all other variables combined (0.77 versus 0.77).
Conclusions
In patients with a recent acute coronary syndrome and type 2 diabetes mellitus, BNP and NT‐proBNP were powerful predictors of cardiovascular outcomes beyond heart failure and death, ie, were also predictive of MI and stroke. Natriuretic peptides added as much predictive information about death as all other conventional variables combined.
Clinical Trial Registration
URL: http://www.clinicaltrials.gov. Unique identifier: NCT01147250.
Keywords: acute coronary syndrome, biomarker, brain natriuretic peptide, cardiac outcomes, diabetes mellitus, Evaluation of Lixisenatide in Acute Coronary Syndrome trial, glucagon‐like peptide‐1, natriuretic peptide, N‐terminal prohormone B‐type natriuretic peptide, risk model
Subject Categories: Cardiovascular Disease; Epidemiology; Heart Failure; Diabetes, Type 2
Introduction
Patients admitted with an acute coronary syndrome (ACS) are at increased risk of subsequent cardiovascular events, especially those with type 2 diabetes mellitus,1, 2 who constitute ≈30% of all ACS patients.3 Determining the predictors of death, myocardial infarction (MI), heart failure (HF), and stroke among these patients is important as it assists in identifying the individuals at highest risk of these various outcomes. These individuals should be the focus of the most intensive secondary preventive strategies. In the same way, risk stratification may help motivate both patients and clinicians in secondary preventive efforts. Lastly, predictive models can be used to select the highest risk individuals for trials of new secondary preventive therapies.
B‐type natriuretic peptide (BNP) and N‐terminal prohormone B‐type natriuretic peptide (NT‐proBNP) levels are well established predictors of HF hospitalization and mortality in patients with HF.4, 5, 6, 7 However, as the incidences of outcomes differ between patients with HF8 and patients with a recent ACS9 in a stable phase, it is less established how natriuretic peptides are associated with cardiovascular outcomes, especially subsequent MI and stroke, in the latter population.3, 10, 11, 12, 13, 14 Concentrations of natriuretic peptides may be affected by both asymptomatic myocardial ischemia15, 16 and atrial fibrillation,17, 18 which could make BNP and NT‐proBNP relevant as predictors of MI and stroke. However, trials investigating patients at high risk of atherosclerotic events have found conflicting results regarding both the predictive ability of natriuretic peptides and cardiovascular outcomes—including MI and stroke—as well as the predictive strength of BNP versus NT‐proBNP.19, 20, 21, 22
We wanted to expand knowledge of these natriuretic peptides as predictors of death, cardiovascular death, myocardial infarction, heart failure, and stroke in patients with a recent coronary event and type 2 diabetes mellitus enrolled in the Evaluation of Lixisenatide in Acute Coronary Syndrome trial (ELIXA, NCT01147250). Data from the ELIXA trial allowed us to compare the predictive strength of baseline BNP and NT‐proBNP in a high risk ACS patient cohort with type 2 diabetes mellitus. In addition, prospective ascertainment during a reasonable follow‐up period, and adjudication of a variety of cardiovascular events, ensured detailed and validated data for analyses.
Methods
The ELIXA trial (Evaluation of Lixisenatide in Acute Coronary Syndrome) trial included 6068 patients with type 2 diabetes mellitus and an acute coronary event within 180 days from randomization (index event).23 The study was approved by the appropriate national and institutional regulatory and ethics boards, and all subjects gave informed consent. The objective of the ELIXA trial was to assess the safety and efficacy of lixisenatide, a glucagon‐like peptide‐1 receptor agonist, on cardiovascular morbidity and mortality. Details of the trial design and the demographic and clinical characteristics of the included patients have been reported previously.24 In summary, patients were included in this randomized, double‐blind, placebo‐controlled, parallel‐group study, between 2010 and 2013, from 49 countries, and followed for a median of 25 months. Key exclusion criteria were percutaneous coronary intervention within 15 days of screening or planned percutaneous coronary intervention within 90 days after screening, coronary artery bypass graft treatment at the index event, an estimated glomerular filtration rate of less than 30 mL/min per 1.73 m2 of body‐surface area, a glycated hemoglobin level of less than 5.5% or more than 11.0%, or an inability to provide written informed consent. Patients were randomized to subcutaneous injections of either lixisenatide (maximum 20 μg daily) or placebo (volume matched) in addition to locally determined standards of care. The ELIXA trial showed that lixisenatide had a neutral effect with regard to the occurrence of the primary outcome (cardiovascular death, MI, stroke, or hospitalization for unstable angina) and HF hospitalization.23
Covariates and Outcomes
All data pertaining to baseline variables including demographics, anthropometrics, cardiovascular risk factors, and prior medical history were obtained at the time of randomization in the study. All events were reported to a centralized and independent adjudication committee who classified events according to prespecified definitions.24 Data on adjudicated time‐to‐event for outcomes of all‐cause death (death), cardiovascular death (cardiovascular death), heart failure hospitalization (HF), fatal and nonfatal myocardial infarction (MI), and fatal and nonfatal stroke (stroke) were used for analyses.
BNP and NT‐proBNP sampling was carried out at baseline. Samples were collected and analyzed at a core laboratory (Covance Central Laboratory Services, Meyrin, Switzerland). The Triage BNP assay was used to analyze BNP. The intra‐assay coefficient of variation was 1.1% to 3.1%. The interassay coefficient of variation was 1.8% to 6.6%. The Immulite NT‐proBNP assay was used to analyze NT‐proBNP. The intra‐assay coefficient of variation was 2.3% to 5.4%. The interassay coefficient of variation was 4.0% to 6.4%. BNP and NT‐proBNP samples from 5925 patients (98%) were obtained.
Statistical Analyses
Baseline characteristics shown in Table 1 were selected for best risk models. Patients without data on all these relevant variables, including BNP and NT‐proBNP measurements, were excluded (n=543, 9%). The distributions of baseline BNP, NT‐proBNP, and C‐reactive protein were found to be right‐skewed and were therefore log‐transformed prior to analysis. Continuous variables were included in the models unless there was clear evidence of nonlinearity.
Table 1.
Characteristics of All Included Patients
| No Cardiovascular Events (n=4626) | Cardiovascular Events (n=899) | P Value | |
|---|---|---|---|
| Randomized to lixisenatide | 2327 (50.3%) | 449 (49.9%) | 0.84 |
| Age, y | 59.7±9.5 | 63.3±9.7 | <0.001 |
| Male (%) | 3238 (70.0%) | 627 (69.7%) | 0.88 |
| BMI, kg/m2 | 30.0±5.6 | 30.3±6.1 | 0.24 |
| Race | <0.001 | ||
| Asian | 648 (14.0%) | 82 (9.1%) | |
| Black | 14 (3.1%) | 47 (5.2%) | |
| Other | 385 (8.3%) | 86 (9.6%) | |
| White | 3451 (74.6%) | 684 (76.1%) | |
| Ethnicity—Hispanic | 1396 (30.2%) | 250 (27.8%) | 0.16 |
| Region | <0.001 | ||
| Africa/Near East | 215 (4.6%) | 54 (6.0%) | |
| Asia Pacific | 597 (12.9%) | 72 (8.0%) | |
| Eastern Europe | 1172 (25.3%) | 241 (26.8%) | |
| North America | 563 (12.2%) | 153 (17.0%) | |
| South and Centr. America | 1551 (33.5%) | 273 (30.4%) | |
| Western Europe | 528 (11.4%) | 106 (11.8%) | |
| Systolic blood pressure, mm Hg | 129±17 | 131±19 | 0.86 |
| Diastolic blood pressure, mm Hg | 77±10 | 77±11 | 0.18 |
| Heart rate, bpm | 70±10 | 71±11 | 0.001 |
| Current smoker | 511 (11.0%) | 117 (13.0%) | 0.09 |
| Former smoker | 2113 (45.7%) | 409 (45.5%) | 0.92 |
| Medical history | |||
| MI | 918 (19.8%) | 340 (37.7%) | <0.001 |
| HF | 905 (19.5%) | 330 (36.7%) | <0.001 |
| Atrial fibrillation/flutter | 240 (5.2%) | 121 (13.5%) | <0.001 |
| PAD | 271 (5.9%) | 142 (15.8%) | <0.001 |
| TIA | 83 (1.8%) | 44 (4.9%) | <0.001 |
| Ventricular tachycardia | 57 (1.2%) | 17 (1.9%) | 0.12 |
| Stroke | 201 (4.3%) | 91 (10.1%) | <0.001 |
| CABG | 309 (6.7%) | 151 (16.8%) | <0.001 |
| Implanted pacemaker | 102 (2.2%) | 41 (4.6%) | <0.001 |
| Carotid disease | 87 (1.9%) | 52 (5.8%) | <0.001 |
| Hypertension | 3449 (74.6%) | 761 (84.6%) | <0.001 |
| Index event | <0.001 | ||
| STEMI | 2146 (46.4%) | 297 (33.0%) | |
| NSTEMI | 1702 (36.8%) | 432 (48.1%) | |
| Unstable angina pectoris | 778 (16.8%) | 170 (18.9%) | |
| PCI at index event | 2943 (63.6%) | 463 (51.5%) | <0.001 |
| Insulin‐treated | 1699 (36.7%) | 460 (51.2%) | <0.001 |
| Duration of diabetes mellitus, y | 8.8±7.9 | 11.9±9.5 | <0.001 |
| Retinopathy | 452 (9.8%) | 139 (15.5%) | <0.001 |
| Neuropathy | 714 (15.4%) | 205 (22.8%) | <0.001 |
| Asthma | 114 (2.5%) | 40 (4.4%) | <0.001 |
| COPD | 173 (3.7%) | 76 (8.5%) | <0.001 |
| HbA1c, % | 7.6±1.3 | 7.9±1.3 | <0.001 |
| HDL, mg/dL | 43±11 | 43±11 | 0.65 |
| LDL, mg/dL | 77±34 | 83±39 | <0.001 |
| eGFR, mL/min per 1.73 m2 | 77.5±21.1 | 68.1±20.6 | <0.001 |
| Albuminuria | <0.001 | ||
| Normoalbuminuria | 3558 (76.9%) | 544 (60.5%) | |
| Microalbuminuria | 829 (17.9%) | 234 (26.0%) | |
| Macroalbuminuria | 239 (5.2%) | 121 (13.5%) | |
| Hemoglobin, g/dL | 13.8±1.4 | 13.5±1.5 | <0.001 |
| Na, mmol/L | 140.4±2.9 | 140.3±3.1 | 0.75 |
| Albumin, g/dL | 4.1±0.3 | 3.9±0.4 | <0.001 |
| CRP, mg/dL | 2.0 (1.9–2.0) | 2.7 (2.4–2.9) | <0.001 |
| BNP, pg/mL | 95 (92–98) | 198 (184–213) | <0.001 |
| NT‐proBNP, pg/mL | 285 (274–295) | 703 (644–766) | <0.001 |
BMI indicates body mass index; bpm, beats per minute; BNP, B‐type natriuretic peptide; CABG, coronary artery bypass grafting; COPD, chronic obstructive pulmonary disease; CRP, C‐reactive protein; eGFR, estimated glomerular filtration rate; HbA1c, glycated hemoglobin; HDL, high‐density lipoproteins; HF, heart failure; LDL, low‐density lipoproteins; MI, myocardial infarction; NSTEMI, non‐ST elevation myocardial infarction; NT‐proBNP, N‐terminal prohormone B‐type natriuretic peptide; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; STEMI, ST elevation myocardial infarction; TIA, transient ischemic attack.
Cox proportional hazard modeling was used to create best risk models without BNP or NT‐proBNP using forward selection with a cut‐off value of 0.05. Separate base risk models were created for the following outcomes; death, cardiovascular death, fatal or nonfatal MI, fatal or nonfatal HF hospitalization, as well as fatal or nonfatal stroke. The variables selected were ordered according to their χ2 value and sorted in descending order for each outcome. The predictive ability of base risk models were assessed using Harrell's C statistics. Selected 30‐day models were made for comparison with previous studies. Using the selected variables from the base model, comparison between the predictive ability of the base model compared to the base model with log2BNP/log2NT‐proBNP was assessed for all outcomes. Changes in C statistics, Net Reclassification Index (NRI), and Integrated Discrimination Index (IDI) were estimated to evaluate the incremental value of adding BNP or NT‐proBNP to best risk models using a set time of 2 years comparable to the average follow‐up time (somersd package, STATA 13. survIDINRI package, R 2.3.2).
Identification of baseline variables independently associated with BNP/NT‐proBNP were obtained using forward selection regression models with P<0.001 as a cut‐off. The 5 variables with the highest χ2 value were listed along with the r 2 values.
To identify the most significant predictive threshold of BNP/NT‐proBNP values, we divided the continuous BNP/NT‐proBNP concentrations into arbitrary threshold concentrations (ie, 35, 100, 125, 200, 300…1000, 2000, 5000). Then using HF hospitalization as an outcome, a fully adjusted Cox model with forward selection identified the BNP threshold concentration that most significantly separated patients into a lower versus a higher risk group. A univariate approach was also carried out using receiver operating characteristic analysis (SENSPEC package, STATA 13) with binary outcome of HF hospitalization to determine the optimal cut‐off value with respect to Youden index (ie, sensitivity+specificity−1). Interaction analyses between natriuretic peptides and timing of the baseline sample in temporal relation to the index ACS event for outcome of death was also performed, as was interaction between natriuretic peptides and type of ACS index event (ST‐elevation MI [STEMI], Non‐ST elevation MI [NSTEMI], Unstable Angina Pectoris [UAP]).
To assess whether the relationship between baseline BNP/NT‐proBNP and hazard was linear, fully adjusted Cox spline models for each outcome with transformed BNP/NT‐proBNP were analyzed. Concentrations below 35 pg/mL (BNP) or 125 pg/mL (NT‐proBNP) were considered normal, as these concentrations are commonly referenced as diagnostic thresholds for excluding HF in patients presenting in a nonacute manner.25 Hence, the risk of death in patients with these levels were used as references. Both unadjusted and adjusted models were used to confirm findings. Risk modeling, including interaction analyses (history of HF and log2BNP/log2NT‐proBNP), discriminatory statistics, and Cox spline models were also analyzed in patients stratified according to medical history of HF for BNP.
All BNP/NT‐proBNP values were summarized as geometric mean±95% CI. A significance level of 0.05 was considered statistically significant.
Results
Baseline Characteristics
Our study included 5525 patients comprising 91% of the included patients in the ELIXA trial. The median follow‐up time was 26 months. In our population, 4626 (84%) patients did not experience any cardiovascular event confirmed by adjudication. Baseline characteristics of patients with or without a cardiovascular event are listed in Table 1 (Baseline characteristics according BNP quartiles and as linear covariates are listed in Tables S1 and S2). Compared with patients not experiencing a cardiovascular event, those that did were in general older and more burdened with comorbidity, and were more likely to have micro‐ or macroalbuminuria, and a lower estimated glomerular filtration rate. Blood pressure was similar in both groups. Baseline BNP and NT‐proBNP were elevated in those subsequently experiencing any cardiovascular event.
Predictive Variables
In separate models, BNP and NT‐proBNP were the most significant predictors for each of death from any cause, death from a cardiovascular cause, HF, and stroke among the studied variables. The natriuretic peptides were the second most significant predictors for MI (Tables 2 and 3). Apart from BNP/NT‐proBNP, the 14 other variables that conferred the greatest information were the following: Prior MI, body mass index, NSTEMI (index event), heart rate (HR), glycated hemoglobin, percutaneous coronary intervention at the index event (percutaneous coronary intervention), cerebrovascular disease (prior stroke/transient ischemic attack), atrial fibrillation, prior HF, sodium concentration, macroalbuminuria, peripheral artery disease (PAD), age, and LDL concentration. Fifteen and 16 variables were independently associated with concentrations of BNP/NT‐proBNP at the α=0.001 level and accounted for 26% and 34% of patient‐level variability, respectively. The 5 strongest associated variables are listed in Tables S3 through S5.
Table 2.
Predictors of Outcomes Ranked 1 to 5 According to χ2 Value Using Base Variables and BNP (n=5525)
| Outcome | 1st | 2nd | 3rd | 4th | 5th |
|---|---|---|---|---|---|
| Death (397 events) | log2BNP (χ2:203, HR 1.67) | AF (χ2:11, HR 1.60) | NSTEMI (χ2:11, HR 1.41) | Naa (χ2:10, HR 1.08) | HR per 10 (χ2:10, HR 1.17) |
| Cardiovascular death (286 events) | log2BNP (χ2:201, HR 1.82) | HbA1c (χ2:13, HR 1.18) | AF (χ2:13, HR 1.79) | NSTEMI (χ2:9, HR 1.46) | HR per 10 (χ2:9, HR 1.19) |
| MI (473 events) | Prior MI (χ2:46, HR 1.96) | log2BNP (χ2:44, HR 1.23) | NSTEMI (χ2:29, HR 1.66) | Prior stroke (χ2:13, HR 1.69) | PAD (χ2:10, HR 1.52) |
| HF (221 events) | log2BNP (χ2:135, HR 1.80) | BMI per 5 (χ2:34, HR 1.33) | HR per 10 (χ2:17, HR 1.30) | Prior HF (χ2:13, HR 1.82) | Prior MI (χ2:12, HR 1.67) |
| Stroke (115 events) | log2BNP (χ2:23, HR 1.35) | Prior TIA (χ2:13, HR 3.12) | Macroalbuminuria (χ2:11, HR 2.35) | Age per 10 (χ2:9, HR 1.36) | LDL per 10 (χ2:4, HR 1.06) |
Hazard ratios reflect 1 unit changes if nothing else is stated. For log2BNP that translates into a doubling of the untransformed BNP concentrations. Macroalbuminuria: >300 mg albumin excretion/24 hours. All variables are significant, P<0.05. AF indicates atrial fibrillation/flutter; BNP, B‐type natriuretic peptide; HbA1c, glycated hemoglobin; HR, heart rate; LDL, low‐density lipoproteins; MI, myocardial infarction; NSTEMI, non‐ST elevation myocardial infarction at index event; PAD, peripheral artery disease; TIA, transient ischemic attack.
1 mmol/L decreases below 140 mmol/L.
Table 3.
Predictors of Outcomes Ranked 1 to 5 According to χ2 Value Using Base Variables and NT‐proBNP (n=5525)
| Outcome | 1st | 2nd | 3rd | 4th | 5th |
|---|---|---|---|---|---|
| Death (397 events) | log2NT‐proBNP (χ2:215, HR 1.52) | NSTEMI (χ2:10, HR 1.40) | PCI (χ2:10, HR 0.71) | DBPa (χ2:8, HR 1.02) | Nab (χ2:8, HR 1.04) |
| Cardiovascular death (286 events) | log2NT‐proBNP (χ2:226, HR 1.65) | HbA1c (χ2:14, HR 1.20) | NSTEMI (χ2:10, HR 1.47) | AF (χ2:9, HR 1.62) | Prior HF (χ2:8, HR 1.44) |
| MI (473 events) | Prior MI (χ2:49, HR 2.00) | log2NT‐proBNP (χ2:37, HR 1.17) | NSTEMI (χ2:29, HR 1.66) | Prior stroke (χ2:13, HR 1.71) | PAD (χ2:9, HR 1.48) |
| HF (221 events) | log2NT‐proBNP (χ2:132, HR 1.61) | BMI per 5 (χ2:31, HR 1.31) | NSTEMI (χ2:14, HR 1.90) | Prior HF (χ2:14, HR 1.73) | Prior MI (χ2:12, HR 1.69) |
| Stroke (115 events) | log2NT‐proBNP (χ2:19, HR 1.25) | Prior TIA (χ2:13, HR 3.09) | Macroalbuminuria (χ2:10, HR 2.34) | Age per 10 (χ2:7, HR 1.32) | LDL per 10 (χ2:4, HR 1.05) |
Hazard ratios reflect 1 unit changes if nothing else is stated. For log2NT‐proBNP that translates into a doubling of the untransformed NT‐proBNP concentrations. Macroalbuminuria: >300 mg albumin excretion/24 hours. All variables are significant, P<0.05. AF indicates atrial fibrillation/flutter; BMI, body mass index; HbA1c, glycated hemoglobin; HF, heart failure; HR, heart rate; LDL, low‐density lipoproteins; MI, myocardial infarction; NT‐proBNP, N‐terminal prohormone B‐type natriuretic peptide; NSTEMI, non‐ST elevation myocardial infarction at index event; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; TIA, transient ischemic attack.
1 mm Hg decreases below 70 mm Hg.
1 mmol/L decreases below 140 mmol/L.
Enhanced Prediction With BNP and NT‐proBNP
To estimate the predictive strength of BNP alone, C statistics were compared between base models without BNP versus BNP alone. This showed that the discriminatory ability of base models without BNP versus BNP was similar in outcomes of death (Harrell's C statistics: 0.77 both models [0–30 days: 0.82 versus 0.88, P=0.26]) and in cardiovascular death (Harrell's C statistics: 0.77 versus 0.79, P=0.17).
In contrast, BNP was significantly less discriminatory compared with best risk models without BNP for the outcomes of MI (Harrell's C statistics: 0.71 versus 0.62), HF (Harrell's C statistics: 0.84 versus 0.77) and stroke (Harrell's C statistics: 0.75 versus 0.67) (all P≤0.01). Similar estimates and trends were evident for NT‐proBNP.
The strength of BNP and NT‐proBNP as contributors to risk prediction translated into augmented predictive ability of risk models for all outcomes, as summarized in the increases in C statistics, NRI, and Integrated Discrimination Index (Table 4). Although BNP and NT‐proBNP did not increase the C statistics for the risk model for stroke, both peptides improved NRI significantly.
Table 4.
Discriminatory Changes in Best Risk Models With and Without BNP and NT‐proBNP
| C Statistics in Each Model (n=5525) | ||||
|---|---|---|---|---|
| Base Model | BNP/NT‐proBNP in Model | NRI | IDI | |
| Death (397 events) | ||||
| BNP | 0.77 (74–0.79) | 0.82 (0.80–0.84)a | 30.6% (25.2–36.8)a | 5.0% (3.5–7.2)a |
| NT‐proBNP | 0.81 (0.79–0.83)a | 24.3% (17.4–29.3)a | 3.3% (2.1–5.0)a | |
| Cardiovascular death (286 events) | ||||
| BNP | 0.77 (0.74–0.80) | 0.83 (0.81–0.86)a | 36.0% (27.4–41.4)a | 5.6% (3.4–8.6)a |
| NT‐proBNP | 0.83 (0.80–0.86)a | 30.9% (21.7–36.9)a | 4.0% (2.4–6.3)a | |
| MI (473 events) | ||||
| BNP | 0.71 (0.68–0.73) | 0.72 (0.70–0.75)a | 14.3% (9.3–19.5)a | 1.2% (0.6–2.1)a |
| NT‐proBNP | 0.72 (0.67–0.74)a | 10.6% (5.7–16.6)a | 0.8% (0.3–1.6)a | |
| HF (221 events) | ||||
| BNP | 0.84 (0.81–0.86) | 0.87 (0.85–0.89)a | 35.4% (24.7–40.6)a | 5.0% (3.0–7.6)a |
| NT‐proBNP | 0.87 (0.84–0.89)a | 29.9% (21.8–36.6)a | 3.8% (2.2–5.8)a | |
| Stroke (115 events) | ||||
| BNP | 0.74 (0.70–0.79) | 0.76 (0.72–0.80) | 19.3% (8.8–29.9)a | 0.4% (0–1.2) |
| NT‐proBNP | 0.76 (0.72–0.80) | 17.2% (6.3–28.1)a | 0.2% (0–0.8) | |
NRI and IDI summarized as mean percent improvement ±95% CI. BNP indicates B‐type natriuretic peptide; HF, heart failure; IDI, Integrated Discrimination Index; MI, myocardial infarction; NRI, Net Reclassification Index; NT‐proBNP, N‐terminal prohormone B‐type natriuretic peptide.
P<0.05, comparison between base model and BNP/NT‐proBNP model.
BNP and NT‐proBNP were the most predictive variables in risk models of type of cardiovascular death; however, when added to best risk models, BNP and NT‐proBNP only significantly improved the predictive ability (Harrel's C statistics) in outcomes of fatal HF and sudden death; fatal HF (n=39) (Base model: 0.850, BNP: +0.085, NT‐proBNP: +0.086, both P<0.001), and sudden death (n=116) (Base model: 0.773, BNP: +0.024, NT‐proBNP: +0.037, P=0.20 and 0.046, respectively), but not in outcome of fatal MI (n=52) (Base model: 0.827, BNP: +0.025, NT‐proBNP: +0.016, P=0.24 and 0.37, respectively).
In univariate analysis, a BNP concentration of 228 pg/mL best separated patients into a lower versus higher group at risk of subsequent HF (sensitivity: 0.62, specificity: 0.77, Youden index: 0.39), with a corresponding threshold for NT‐proBNP of 751 pg/mL (sensitivity: 0.67, specificity: 0.74, Youden index: 0.41). In adjusted Cox models, a BNP concentration of 500 pg/mL provided the most significant threshold by which to further identify patients at lower versus higher risk of subsequent HF (HR 3.0 [2.1–4.1], P<0.0001), with a corresponding threshold for NT‐proBNP of 700 pg/mL (HR 2.5 [1.7–3.5], P<0.0001).
Predictive Strength of BNP Compared to NT‐proBNP
There was no significant increase in C statistics when BNP was included in the best risk models compared to NT‐proBNP in outcomes of death +0.002 (P=0.55), cardiovascular death +0.0002 (P=0.97), MI +0.002 (P=0.50), HF +0.0001 (P=0.98), stroke +0.002 (P=0.88). The comparable estimates were confounded by a significant correlation between BNP and NT‐proBNP (Spearman's rho 0.86, P<0.0001).
Subgroup and Sensitivity Analyses
In our population, 22% had a history of HF. As sensitivity analysis, patients were stratified according to history of HF at baseline to provide ranking and estimates of important risk factors, although there was no significant interaction with natriuretic peptides and history of HF (death; BNP, P=0.57. NT‐proBNP, P=0.21, Figure S1). BNP was the strongest prognostic variable for all outcomes examined in both groups (±prior HF; Tables S6 through S8). Sensitivity analysis was also done in the subset of patients (52%) without any prior cardiovascular disease at baseline (HF, atrial fibrillation, peripheral artery disease, transient ischemic attack/stroke, ventricular tachycardia, coronary artery bypass graft or MI apart from index event). The same trends were also evident in this subset (Tables S9 and S10).
Analysis in the subset of patients that had information on left ventricular ejection fraction present at their index ACS was performed (n=3390). Left ventricular ejection fraction was not among the 3 most significant predictors across all outcomes when added to the variable list. The predictive ability of BNP and NT‐proBNP was comparable in this subset compared to the entire cohort (Tables S11 through S13).
The timing of the sampling in relation to the index ACS event did not affect the risk estimates for death for BNP (P=0.63) or NT‐proBNP (P=0.46), nor did the type of ACS (BNP, P=0.30; NT‐proBNP, P=0.32).
There was no significant interaction between sex and concentrations of BNP (P=0.17) or NT‐proBNP (P=0.58) when tested in a fully adjusted model.
Discussion
Our goal was to examine the strength of BNP and NT‐proBNP in predicting a range of cardiovascular outcomes in high risk ACS patients with type 2 diabetes mellitus. We found that baseline levels of these natriuretic peptides were elevated in patients with a subsequent cardiovascular event during follow‐up compared with those not having an event. The levels of natriuretic peptides most likely reflect that all patients recently suffered a coronary event and on average had a high comorbid burden.
The significance of elevated natriuretic peptides was reiterated when BNP and NT‐proBNP were added to risk models of major cardiovascular outcomes. Ranked according to the strength of prediction, BNP and NT‐proBNP were the primary predictive variables in all outcomes examined, except MI, where they were the second most predictive variable. The predictive strength of BNP and NT‐proBNP was also evident when viewing the χ2 values, which were magnitudes higher than other significant variables in death, cardiovascular death and HF, moderately higher in stroke, and comparable to having had a prior MI in outcome of MI. This was also mirrored in the ability of BNP and NT‐proBNP to predict causes of death, where a significant predictive contribution of BNP and NT‐proBNP was present when added to risk models in outcome of sudden death and HF death, whereas none was found for fatal MI. The cut‐off point that most significantly divided patients into a higher versus lower risk group of subsequent HF hospitalization was 500 and 700 pg/mL for BNP and NT‐proBNP, in multivariate analysis.
Natriuretic peptides are primarily recognized as predictors of mortality and HF hospitalization, whereas our finding of a strong predictive ability of natriuretic peptide levels in outcomes of MI and stroke is less validated, especially in ACS patients. This ability could be attributed to higher levels of natriuretic peptides in patients with asymptomatic myocardial ischemia15, 16 and paroxysmal atrial fibrillation,17, 18 which could predispose to both MI and stroke.
Our risk models also identified other important predictors apart from BNP and NT‐proBNP. Risk models in other diabetic populations (TREAT [Trial to Reduce Cardiovascular Events with Aranesp (darbepoetin‐alfa) Therapy]),26 UKPDS [UK Prospective Diabetes Study]27) have yielded results that also highlight the importance of the risk factors we identified, such as prior HF, glycated hemoglobin, age, heart rate, albuminuria, and cardiac arrhythmias.28, 29 Important differences were that these studies either used composite end points28 or only single outcomes29 when examining predictors. Furthermore, the diabetic patients on which these risk models were based were either at higher risk (TREAT: 81.1 deaths per 1000 PY) or lower risk of death (UKPDS: 18.9 deaths per 1000 PY) than in the present study (32.3 deaths per 1000 PY). Nonetheless, traditional cardiovascular risk factors combined with markers of chronic dysglycemia seem to persist as predictors of adverse cardiovascular events despite the differences in the diabetic populations studied.
When BNP or NT‐proBNP was used to predict death or cardiovascular death alone compared to using all variables available excluding these natriuretic peptides, estimates of C statistics were comparable, but the stand‐alone natriuretic peptide receiver operating characteristic area under the curve values exceeded those reported earlier in patients with HF or coronary artery disease.19, 30 Thus, a single measurement of BNP or NT‐proBNP contains the same predictive information about risk of death as all other variables listed in Table 1. Of note, the reverse was seen in cardiovascular morbidity (MI, HF, stroke) where best risk models outperformed a single BNP or NT‐proBNP measurement. As visually depicted in the figure, differences in natriuretic peptides conferred ≈20‐fold changes in risk of death. This large risk gradient enables easier identification of patients at higher versus lower risk and reinforces the prognostic information contained in BNP and NT‐proBNP levels.
Figure 1.
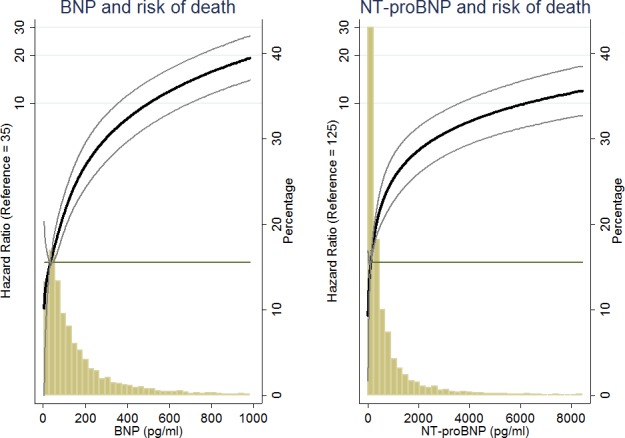
The association of BNP and NT‐proBNP concentrations and risk of all‐cause death. The hazard of death is depicted with 95% CIs. The reference of hazard ratio=1.0 corresponds to a BNP concentration of 35 pg/mL, and a NT‐proBNP concentration of 125 pg/mL. BNP indicates B‐type natriuretic peptide; NT‐proBNP, N‐terminal prohormone BNP.
The addition of either natriuretic peptide to best risk models improved the discriminatory ability significantly. This was evident in the changes in C statistics, NRI, and Integrated Discrimination Index. C statistics increased 0.01 to 0.06 depending on outcome, which was reflected in NRI increases of 10.6% to 35.4%. These changes make both BNP and NT‐proBNP valuable prognostic determinants that should be considered in future risk determination in a comparable population. Furthermore, history of HF did not influence the predictive ability of BNP or NT‐proBNP across all outcomes, including MI and stroke.
Having diabetes mellitus seems to influence levels of natriuretic peptides in both the absence and presence of cardiovascular disease,31, 32 which potentially could change the relationship between levels of natriuretic peptides and risk of cardiovascular events. Whether natriuretic peptides predict outcomes differently in ACS patients with type 2 diabetes mellitus compared to similar patients without diabetes mellitus is not known. The stand‐alone discriminatory strength of BNP and NT‐proBNP in cardiovascular death was somewhat lower (receiver operating characteristic area under the curve: BNP 0.58; NT‐proBNP 0.68) in patients with coronary artery disease from the Prevention of Events With Angiotensin Converting Enzyme (PEACE) trial (≈16% patients had diabetes mellitus). In comparison, when BNP was sampled 2 to 4 days after the infarct in STEMI patients included in the Enoxaparin Tenecteplase‐Tissue‐Type Plasminogen Activator With or Without Glycoprotein IIb/IIIa Inhibitor as Reperfusion Strategy in ST‐Segment Elevation Myocardial Infarction‐Thrombolysis In Myocardial Infarction‐23 (ENTIRE‐TIMI‐23) trial (≈13% patients had diabetes mellitus), the receiver operating characteristic area under the curve for death after 30 days was 0.81,11 which is comparable to our results (area under the curve30 days: 0.88). This could suggest that the severity of the coronary pathology influences the predictive strength of natriuretic peptides and/or the timing of the sample used for risk determination is important, as shown by Lindahl et al.33 Earlier studies have shown that levels of natriuretic peptides are dynamic in the subacute phase following an MI,34 and smaller studies suggest that patients are at higher risk of death and left ventricular remodeling if natriuretic peptides continue to be elevated after the MI, compared with those with decreasing levels.35, 36 In our study, patients were randomized within 180 days from their ACS. The timing of the baseline sample in relation to their index ACS did not affect the risk estimates of BNP or NT‐proBNP, nor did the type of index event (STEMI, NSTEMI, or UAP). This suggests that the predictive value of natriuretic peptides is retained from shortly after the event until at least 6 months after the ACS in patients with type 2 diabetes mellitus, irrespective of coronary pathology.
As the availability of BNP and NT‐proBNP analyses differs between institutions and regions, we also assessed the predictive value of BNP compared to NT‐proBNP. In the PEACE trial, BNP was only a predictor of HF, while NT‐proBNP was a predictor of cardiovascular death, HF, and stroke in coronary artery disease patients.19 Neither biomarker predicted MI. In the Valsartan Heart Failure Trial (Val‐Heft), NT‐proBNP proved superior to BNP in predicting a composite of morbidity and mortality and HF hospitalization in chronic HF patients; however, the incremental discriminatory value of NT‐proBNP versus BNP was small.30 In our study of ACS patients with type 2 diabetes mellitus, both natriuretic peptides had comparable predictive strength in all outcomes studied (death, cardiovascular death, MI, HF, and stroke), albeit NRI and Integrated Discrimination Index values increased slightly more with BNP across all outcomes.
Our results expand knowledge of earlier findings of enhanced risk prediction using natriuretic peptides in patients with a recent ACS3, 10, 11, 12 to also include patients with a recent ACS and type 2 diabetes mellitus. This prevalent population has only marginally been studied in this context.37, 38, 39, 40 The use of BNP or NT‐proBNP for risk prediction in all ACS patients, irrespective of diabetes mellitus status, is now further substantiated. Whether drugs that directly influence natriuretic peptide concentrations (eg, sacubitril41) can modify the incidence of the cardiovascular outcomes examined in this study remains speculative.
Limitations
To learn more about how diabetes mellitus per se affects the predictive ability of natriuretic peptides, a similar study design with ACS patients with and without type 2 diabetes mellitus would have been optimal. Furthermore, our average follow‐up time of ≈2 years precludes any estimates on the long‐term predictive ability of BNP and NT‐proBNP. Data on the severity of the index ACS were not obtained (eg, troponin), which as a marker of myocardial damage could have attenuated the prognostic ability of BNP/NT‐proBNP. Extrapolating from the present study to other ACS patients with type 2 diabetes mellitus should be done cautiously, as inclusion criteria may have led to selection bias compared to patients not included in this trial. Importantly, patients with estimated glomerular filtration rate <30 mL/min per 1.73 m2 were excluded from this study, and renal function is shown to affect levels of natriuretic peptides.42
Conclusion
In a population of patients with a recent ACS and type 2 diabetes mellitus, BNP and NT‐proBNP are important and comparable predictors of death, cardiovascular death, MI, HF, as well as stroke. Both natriuretic peptides improve the discriminatory ability significantly when added to best risk models with known predictors of adverse cardiovascular outcomes, irrespective of prior history of HF or prior cardiovascular disease.
Sources of Funding
Sanofi funded the ELIXA trial. EW was supported by an unrestricted grant from The Danish Council for Independent Research (DFF—4183‐00550).
Disclosures
Wolsk: supported by unrestricted grants from The Danish Council for Independent Research (DFF – 4183‐00550), Fonden af 17‐12‐1981, Eva & Henry Frænkels Mindefond, Kong Christian den Tiendes Fond, Knud Højgaards Fond, and Direktør Ib Henriksens Fond. Claggett: None. Pfeffer: Received research Grant Support from Novartis, Sanofi. Consultant: AstraZeneca, Bayer, Boehringer Ingelheim, DalCor, Genzyme, Gilead, GlaxoSmithKline, Janssen, Lilly, Medicines Company, Merck, Novartis, Novo Nordisk, Relypsa, Sanofi, Teva and Thrasos. Stock Options: DalCor. Other: Patent awarded to BWH regarding the use of inhibitors of the renin‐angiotensin system in MI. Licensed by Novartis, Dr. Pfeffer's share irrevocably assigned to charity. Diaz: Received grants from Sanofi. Dickstein: Member of the ELIXA Executive Steering Committee. Gerstein: is supported by the McMaster‐Sanofi Population Health Institute Chair in Diabetes Research and Care. Gerstein has received research grant support from Sanofi, Lilly, AstraZeneca and Merck, honoraria for speaking from Sanofi, Novo Nordisk, and AstraZeneca, and Berlin Chemie, and consulting fees from Sanofi, Lilly, AstraZeneca, Merck, Novo Nordisk, Abbot, Amgen, Boehringer Ingelheim, and Kaneq Bioscience. Lawson: Sanofi employee. Lewis: received research support from Sanofi. Maggioni: Honoraria for participation in the ELIXA Executive/Steering Committee sponsored by Sanofi. McMurray: None. Probstfield: None. Riddle: AstraZeneca: Research support through my institution and honoraria for consulting. Elcelyx: Honoraria for consulting. Eli Lilly: Research support through my institution and Honoraria for consulting. GlaxoSmithKline: Honoraria for consulting. NovoNordisk: Research support through my institution. Sanofi: Research support through my institution and honoraria for consulting and for speaking at professional meetings. Theracos: Honoraria for consulting. Valeritas: Honoraria for consulting. Solomon: Received research support from Sanofi. Tardif: Has recevied research grants from Amarin, AstraZeneca, DalCor, EliLilly, Esperion, Merck, Pfizer, Sanofi and Servier; honoraria from DalCor, Pfizer, Sanofi and Servier; and holds a minor equity interest in DalCor. Køber: Personal fees from Sanofi and Novartis as speaker, outside the submitted work.
Supporting information
Table S1. Characteristics of All Included Patients Grouped According to Quartiles of BNP
Table S2. Linear Regression of BNP and NT‐proBNP With All Variables Listed in the Model
Table S3. Variables Independently Associated With BNP/NT‐proBNP Concentrations, and Corresponding r 2 Values for Regression Models With the 5 Listed Variables
Table S4. Unadjusted Estimates of Predictors of Outcomes Found Significant in Multivariate Models Using Base Variables and BNP (n=5525)
Table S5. Unadjusted Estimated of Predictors of Outcomes Found Significant in Multivariate Models Using Base Variables and NT‐proBNP (n=5525)
Table S6. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and BNP and Stratified According to History of Heart Failure (No Prior HF, n=4290; Prior HF, n=1235)
Table S7. Discriminatory Changes in Best Risk Models With and Without BNP Stratified According to History of Heart Failure (No Prior HF, n=4290; Prior HF, n=1235)
Table S8. Discriminatory Changes in Best Risk Models Without BNP Compared to BNP Alone, in All Patients (n=5525) and Stratified According to History of Heart Failure (No Prior HF, n=4290; Prior HF, n=1235)
Table S9. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and BNP in Patients Without Prior CV Disease (n=2899)
Table S10. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and NT‐proBNP in Patients Without Prior CV Disease (n=2899)
Table S11. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and BNP With and Without Adding Information on LVEF (n=3390)
Table S12. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and NT‐proBNP With and Without Adding Information on LVEF at Index ACS (n=3390)
Table S13. Discriminatory Changes in Best Risk Models With and Without BNP and NT‐proBNP With LVEF and Coronary Intervention Procedure Added to Base Model
Figure S1. Association of BNP Concentrations and Risk of All‐Cause Death According to History of Heart Failure.
(J Am Heart Assoc. 2017;6:e004743 DOI: 10.1161/JAHA.116.004743.)28554908
References
- 1. Haffner SM, Lehto S, Rönnemaa T, Pyörälä K, Laakso M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med. 1998;339:229–234. [DOI] [PubMed] [Google Scholar]
- 2. Beckman JA, Creager MA, Libby P. Diabetes and atherosclerosis: epidemiology, pathophysiology, and management. JAMA. 2002;287:2570–2581. [DOI] [PubMed] [Google Scholar]
- 3. Scirica BM, Kadakia MB, de Lemos JA, Roe MT, Morrow DA, Li S, Wiviott SD, Kontos MC. Association between natriuretic peptides and mortality among patients admitted with myocardial infarction: a report from the ACTION Registry®–GWTGTM . Clin Chem. 2013;59:1205–1214. [DOI] [PubMed] [Google Scholar]
- 4. Jhund PS, Anand IS, Komajda M, Claggett BL, McKelvie RS, Zile MR, Carson PE, McMurray JJV. Changes in N‐terminal pro‐B‐type natriuretic peptide levels and outcomes in heart failure with preserved ejection fraction: an analysis of the I‐Preserve study. Eur J Heart Fail. 2015;17:809–817. [DOI] [PubMed] [Google Scholar]
- 5. Kristensen SL, Jhund PS, Køber L, McKelvie RS, Zile MR, Anand IS, Komajda M, Cleland JGF, Carson PE, McMurray JJV. Relative importance of history of heart failure hospitalization and N‐terminal pro‐B‐type natriuretic peptide level as predictors of outcomes in patients with heart failure and preserved ejection fraction. JACC Heart Fail. 2015;3:478–486. [DOI] [PubMed] [Google Scholar]
- 6. Lourenço P, Ribeiro A, Pintalhão M, Silva S, Bettencourt P. Predictors of six‐month mortality in BNP‐matched acute heart failure patients. Am J Cardiol. 2015;116:744–748. [DOI] [PubMed] [Google Scholar]
- 7. Berger R, Huelsman M, Strecker K, Bojic A, Moser P, Stanek B, Pacher R. B‐type natriuretic peptide predicts sudden death in patients with chronic heart failure. Circulation. 2002;105:2392–2397. [DOI] [PubMed] [Google Scholar]
- 8. Pfeffer MA, Swedberg K, Granger CB, Held P, McMurray JJV, Michelson EL, Olofsson B, Ostergren J, Yusuf S, Pocock S; CHARM Investigators and Committees . Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM‐Overall programme. Lancet. 2003;362:759–766. [DOI] [PubMed] [Google Scholar]
- 9. Pfeffer MA, McMurray JJV, Velazquez EJ, Rouleau J‐L, Køber L, Maggioni AP, Solomon SD, Swedberg K, Van de Werf F, White H, Leimberger JD, Henis M, Edwards S, Zelenkofske S, Sellers MA, Califf RM; Valsartan in Acute Myocardial Infarction Trial Investigators . Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N Engl J Med. 2003;349:1893–1906. [DOI] [PubMed] [Google Scholar]
- 10. Omland T, Aakvaag A, Bonarjee VV, Caidahl K, Lie RT, Nilsen DW, Sundsfjord JA, Dickstein K. Plasma brain natriuretic peptide as an indicator of left ventricular systolic function and long‐term survival after acute myocardial infarction. Comparison with plasma atrial natriuretic peptide and N‐terminal proatrial natriuretic peptide. Circulation. 1996;93:1963–1969. [DOI] [PubMed] [Google Scholar]
- 11. Mega JL, Morrow DA, De Lemos JA, Sabatine MS, Murphy SA, Rifai N, Gibson CM, Antman EM, Braunwald E. B‐type natriuretic peptide at presentation and prognosis in patients with ST‐segment elevation myocardial infarction: an ENTIRE‐TIMI‐23 substudy. J Am Coll Cardiol. 2004;44:335–339. [DOI] [PubMed] [Google Scholar]
- 12. Morrow DA, de Lemos JA, Sabatine MS, Murphy SA, Demopoulos LA, DiBattiste PM, McCabe CH, Gibson CM, Cannon CP, Braunwald E. Evaluation of B‐type natriuretic peptide for risk assessment in unstable angina/non‐ST‐elevation myocardial infarction: B‐type natriuretic peptide and prognosis in TACTICS‐TIMI 18. J Am Coll Cardiol. 2003;41:1264–1272. [DOI] [PubMed] [Google Scholar]
- 13. Scirica BM, Bhatt DL, Braunwald E, Raz I, Cavender MA, Im K, Mosenzon O, Udell JA, Hirshberg B, Pollack PS, Steg PG, Jarolim P, Morrow DA. Prognostic implications of biomarker assessments in patients with type 2 diabetes at high cardiovascular risk: a secondary analysis of a randomized clinical trial. JAMA Cardiol. 2016;1:989–998. [DOI] [PubMed] [Google Scholar]
- 14. James SK, Lindahl B, Siegbahn A, Stridsberg M, Venge P, Armstrong P, Barnathan ES, Califf R, Topol EJ, Simoons ML, Wallentin L. N‐terminal pro‐brain natriuretic peptide and other risk markers for the separate prediction of mortality and subsequent myocardial infarction in patients with unstable coronary artery disease: a Global Utilization of Strategies To Open occluded arteries (GUSTO)‐IV substudy. Circulation. 2003;108:275–281. [DOI] [PubMed] [Google Scholar]
- 15. Sabatine MS, Morrow DA, de Lemos JA, Omland T, Desai MY, Tanasijevic M, Hall C, McCabe CH, Braunwald E. Acute changes in circulating natriuretic peptide levels in relation to myocardial ischemia. J Am Coll Cardiol. 2004;44:1988–1995. [DOI] [PubMed] [Google Scholar]
- 16. Wiersma JJ, van der Zee PM, van Straalen JP, Fischer JC, van Eck‐Smit BLF, Tijssen JGP, Trip MD, Piek JJ, Verberne HJ. NT‐pro‐BNP is associated with inducible myocardial ischemia in mildly symptomatic type 2 diabetic patients. Int J Cardiol. 2010;145:295–296. [DOI] [PubMed] [Google Scholar]
- 17. Wachter R, Lahno R, Haase B, Weber‐Krüger M, Seegers J, Edelmann F, Wohlfahrt J, Gelbrich G, Görlitz A, Kermer P, Vollmann D, Hasenfuß G, Gröschel K, Stahrenberg R. Natriuretic peptides for the detection of paroxysmal atrial fibrillation in patients with cerebral ischemia—the Find‐AF Study. PLoS One. 2012;7:e34351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Sinner MF, Stepas KA, Moser CB, Krijthe BP, Aspelund T, Sotoodehnia N, Fontes JD, Janssens ACJW, Kronmal RA, Magnani JW, Witteman JC, Chamberlain AM, Lubitz SA, Schnabel RB, Vasan RS, Wang TJ, Agarwal SK, McManus DD, Franco OH, Yin X, Larson MG, Burke GL, Launer LJ, Hofman A, Levy D, Gottdiener JS, Kääb S, Couper D, Harris TB, Astor BC, Ballantyne CM, Hoogeveen RC, Arai AE, Soliman EZ, Ellinor PT, Stricker BHC, Gudnason V, Heckbert SR, Pencina MJ, Benjamin EJ, Alonso A. B‐type natriuretic peptide and C‐reactive protein in the prediction of atrial fibrillation risk: the CHARGE‐AF Consortium of community‐based cohort studies. Europace. 2014;16:1426–1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Omland T, Sabatine MS, Jablonski KA, Rice MM, Hsia J, Wergeland R, Landaas S, Rouleau JL, Domanski MJ, Hall C, Pfeffer MA, Braunwald E; PEACE Investigators . Prognostic value of B‐type natriuretic peptides in patients with stable coronary artery disease: the PEACE Trial. J Am Coll Cardiol. 2007;50:205–214. [DOI] [PubMed] [Google Scholar]
- 20. Blankenberg S. Comparative impact of multiple biomarkers and N‐terminal pro‐brain natriuretic peptide in the context of conventional risk factors for the prediction of recurrent cardiovascular events in the Heart Outcomes Prevention Evaluation (HOPE) Study. Circulation. 2006;114:201–208. [DOI] [PubMed] [Google Scholar]
- 21. Mishra RK, Beatty AL, Jaganath R, Regan M, Wu AHB, Whooley MA. B‐type natriuretic peptides for the prediction of cardiovascular events in patients with stable coronary heart disease: the Heart and Soul Study. J Am Heart Assoc. 2014;3:e000907 DOI: 10.1161/JAHA.114.000907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Eggers KM, Kempf T, Venge P, Wallentin L, Wollert KC, Lindahl B. Improving long‐term risk prediction in patients with acute chest pain: the Global Registry of Acute Coronary Events (GRACE) risk score is enhanced by selected nonnecrosis biomarkers. Am Heart J. 2010;160:88–94. [DOI] [PubMed] [Google Scholar]
- 23. Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, Lawson FC, Ping L, Wei X, Lewis EF, Maggioni AP, McMurray JJV, Probstfield JL, Riddle MC, Solomon SD, Tardif J‐C; ELIXA Investigators . Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015;373:2247–2257. [DOI] [PubMed] [Google Scholar]
- 24. Bentley‐Lewis R, Aguilar D, Riddle MC, Claggett B, Diaz R, Dickstein K, Gerstein HC, Johnston P, Køber LV, Lawson F, Lewis EF, Maggioni AP, McMurray JJV, Ping L, Probstfield JL, Solomon SD, Tardif J‐C, Wu Y, Pfeffer MA; ELIXA Investigators . Rationale, design, and baseline characteristics in Evaluation of LIXisenatide in Acute Coronary Syndrome, a long‐term cardiovascular end point trial of lixisenatide versus placebo. Am Heart J. 2015;169:631–638.e7. [DOI] [PubMed] [Google Scholar]
- 25. McMurray JJV, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, Falk V, Filippatos G, Fonseca C, Gomez‐Sanchez MA, Jaarsma T, Køber L, Lip GYH, Maggioni AP, Parkhomenko A, Pieske BM, Popescu BA, Rønnevik PK, Rutten FH, Schwitter J, Seferovic P, Stepinska J, Trindade PT, Voors AA, Zannad F, Zeiher A; Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology , Bax JJ, Baumgartner H, Ceconi C, Dean V, Deaton C, Fagard R, Funck‐Brentano C, Hasdai D, Hoes A, Kirchhof P, Knuuti J, Kolh P, McDonagh T, Moulin C, Popescu BA, Reiner Z, Sechtem U, Sirnes PA, Tendera M, Torbicki A, Vahanian A, Windecker S, McDonagh T, Sechtem U, Bonet LA, Avraamides P, Ben Lamin HA, Brignole M, Coca A, Cowburn P, Dargie H, Elliott P, Flachskampf FA, Guida GF, Hardman S, Iung B, Merkely B, Mueller C, Nanas JN, Nielsen OW, Orn S, Parissis JT, Ponikowski P; ESC Committee for Practice Guidelines . ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14:803–869. [DOI] [PubMed] [Google Scholar]
- 26. Pfeffer MA, Burdmann EA, Chen C‐Y, Cooper ME, de Zeeuw D, Eckardt K‐U, Feyzi JM, Ivanovich P, Kewalramani R, Levey AS, Lewis EF, McGill JB, McMurray JJV, Parfrey P, Parving H‐H, Remuzzi G, Singh AK, Solomon SD, Toto R; TREAT Investigators . A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med. 2009;361:2019–2032. [DOI] [PubMed] [Google Scholar]
- 27. Intensive blood‐glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;352:837–853. [PubMed] [Google Scholar]
- 28. McMurray JJV, Uno H, Jarolim P, Desai AS, de Zeeuw D, Eckardt K‐U, Ivanovich P, Levey AS, Lewis EF, McGill JB, Parfrey P, Parving H‐H, Toto RM, Solomon SD, Pfeffer MA. Predictors of fatal and nonfatal cardiovascular events in patients with type 2 diabetes mellitus, chronic kidney disease, and anemia: an analysis of the Trial to Reduce cardiovascular Events with Aranesp (darbepoetin‐alfa) Therapy (TREAT). Am Heart J. 2011;162:748–755.e3. [DOI] [PubMed] [Google Scholar]
- 29. Stevens RJ, Kothari V, Adler AI, Stratton IM; United Kingdom Prospective Diabetes Study (UKPDS) Group . The UKPDS risk engine: a model for the risk of coronary heart disease in Type II diabetes (UKPDS 56). Clin Sci (Lond). 2001;101:671–679. [PubMed] [Google Scholar]
- 30. Masson S, Latini R, Anand IS, Vago T, Angelici L, Barlera S, Missov ED, Clerico A, Tognoni G, Cohn JN; Val‐HeFT Investigators . Direct comparison of B‐type natriuretic peptide (BNP) and amino‐terminal proBNP in a large population of patients with chronic and symptomatic heart failure: the Valsartan Heart Failure (Val‐HeFT) data. Clin Chem. 2006;52:1528–1538. [DOI] [PubMed] [Google Scholar]
- 31. Luchner A, Behrens G, Stritzke J, Markus M, Stark K, Peters A, Meisinger C, Leitzmann M, Hense H‐W, Schunkert H, Heid IM. Long‐term pattern of brain natriuretic peptide and N‐terminal pro brain natriuretic peptide and its determinants in the general population: contribution of age, gender, and cardiac and extra‐cardiac factors. Eur J Heart Fail. 2013;15:859–867. [DOI] [PubMed] [Google Scholar]
- 32. Peng Q, Hu W, Su H, Yang Q, Cheng X. Levels of B‐type natriuretic peptide in chronic heart failure patients with and without diabetes mellitus. Exp Ther Med. 2013;5:229–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Lindahl B, Lindbäck J, Jernberg T, Johnston N, Stridsberg M, Venge P, Wallentin L. Serial analyses of N‐terminal pro‐B‐type natriuretic peptide in patients with non‐ST‐segment elevation acute coronary syndromes: a Fragmin and fast Revascularisation during In Stability in Coronary artery disease (FRISC)‐II substudy. J Am Coll Cardiol. 2005;45:533–541. [DOI] [PubMed] [Google Scholar]
- 34. White M, Rouleau JL, Hall C, Arnold M, Harel F, Sirois P, Greaves S, Solomon S, Ajani U, Glynn R, Hennekens C, Pfeffer M. Changes in vasoconstrictive hormones, natriuretic peptides, and left ventricular remodeling soon after anterior myocardial infarction. Am Heart J. 2001;142:1056–1064. [DOI] [PubMed] [Google Scholar]
- 35. Sandhu S, Har BJ, Aggarwal SG, Kavanagh KM, Ramadan D, Exner DV; REFINE Investigators . Predictive value of repeated versus single N‐terminal pro B‐type natriuretic peptide measurements early after‐myocardial infarction. Pacing Clin Electrophysiol. 2009;32(suppl 1):S86–S89. [DOI] [PubMed] [Google Scholar]
- 36. Fertin M, Hennache B, Hamon M, Ennezat PV, Biausque F, Elkohen M, Nugue O, Tricot O, Lamblin N, Pinet F, Bauters C. Usefulness of serial assessment of B‐type natriuretic peptide, troponin I, and C‐reactive protein to predict left ventricular remodeling after acute myocardial infarction (from the REVE‐2 study). Am J Cardiol. 2010;106:1410–1416. [DOI] [PubMed] [Google Scholar]
- 37. Wannamethee SG, Welsh P, Lowe GD, Gudnason V, Di Angelantonio E, Lennon L, Rumley A, Whincup PH, Sattar N. N‐terminal pro‐brain natriuretic peptide is a more useful predictor of cardiovascular disease risk than C‐reactive protein in older men with and without pre‐existing cardiovascular disease. J Am Coll Cardiol. 2011;58:56–64. [DOI] [PubMed] [Google Scholar]
- 38. Pfister R, Tan D, Thekkanal J, Hellmich M, Erdmann E, Schneider CA. NT‐pro‐BNP measured at discharge predicts outcome in multimorbid diabetic inpatients with a broad spectrum of cardiovascular disease. Acta Diabetol. 2007;44:91–97. [DOI] [PubMed] [Google Scholar]
- 39. James SK, Lindahl B, Timmer JR, Ottervanger JP, Siegbahn A, Stridsberg M, Armstrong P, Califf R, Wallentin L, Simoons ML. Usefulness of biomarkers for predicting long‐term mortality in patients with diabetes mellitus and non‐ST‐elevation acute coronary syndromes (a GUSTO IV substudy). Am J Cardiol. 2006;97:167–172. [DOI] [PubMed] [Google Scholar]
- 40. Vergès B, Zeller M, Desgrès J, Dentan G, Laurent Y, Janin‐Manificat L, L'Huillier I, Rioufol G, Beer J‐C, Makki H, Rochette L, Gambert P, Cottin Y. High plasma N‐terminal pro‐brain natriuretic peptide level found in diabetic patients after myocardial infarction is associated with an increased risk of in‐hospital mortality and cardiogenic shock. Eur Heart J. 2005;26:1734–1741. [DOI] [PubMed] [Google Scholar]
- 41. Vardeny O, Miller R, Solomon SD. Combined neprilysin and renin‐angiotensin system inhibition for the treatment of heart failure. JACC Heart Fail. 2014;2:663–670. [DOI] [PubMed] [Google Scholar]
- 42. Vickery S, Price CP, John RI, Abbas NA, Webb MC, Kempson ME, Lamb EJ. B‐type natriuretic peptide (BNP) and amino‐terminal proBNP in patients with CKD: relationship to renal function and left ventricular hypertrophy. Am J Kidney Dis. 2005;46:610–620. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Characteristics of All Included Patients Grouped According to Quartiles of BNP
Table S2. Linear Regression of BNP and NT‐proBNP With All Variables Listed in the Model
Table S3. Variables Independently Associated With BNP/NT‐proBNP Concentrations, and Corresponding r 2 Values for Regression Models With the 5 Listed Variables
Table S4. Unadjusted Estimates of Predictors of Outcomes Found Significant in Multivariate Models Using Base Variables and BNP (n=5525)
Table S5. Unadjusted Estimated of Predictors of Outcomes Found Significant in Multivariate Models Using Base Variables and NT‐proBNP (n=5525)
Table S6. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and BNP and Stratified According to History of Heart Failure (No Prior HF, n=4290; Prior HF, n=1235)
Table S7. Discriminatory Changes in Best Risk Models With and Without BNP Stratified According to History of Heart Failure (No Prior HF, n=4290; Prior HF, n=1235)
Table S8. Discriminatory Changes in Best Risk Models Without BNP Compared to BNP Alone, in All Patients (n=5525) and Stratified According to History of Heart Failure (No Prior HF, n=4290; Prior HF, n=1235)
Table S9. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and BNP in Patients Without Prior CV Disease (n=2899)
Table S10. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and NT‐proBNP in Patients Without Prior CV Disease (n=2899)
Table S11. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and BNP With and Without Adding Information on LVEF (n=3390)
Table S12. Predictors of Outcomes Ranked According to χ2 Value Using Base Variables and NT‐proBNP With and Without Adding Information on LVEF at Index ACS (n=3390)
Table S13. Discriminatory Changes in Best Risk Models With and Without BNP and NT‐proBNP With LVEF and Coronary Intervention Procedure Added to Base Model
Figure S1. Association of BNP Concentrations and Risk of All‐Cause Death According to History of Heart Failure.


