Abstract
Aim: To evaluate the efficacy of bioglass (BAG-S53P4) in the treatment of patients with chronic osteomyelitis and compare the results with calcium sulphate antibiotic beads in one medical centre.
Methods: Retrospective analysis of 25 cases. Inclusion criteria: patients diagnosed clinically and radiographically of osteomyelitis and treated surgically (Group 1: cavitary bone defects treated with bioglass and Group 2: cavitary bone defects treated with calcium sulphate antibiotic beads) during the period of 2014 and 2015 in one medical centre.
Results: Patients in group 1 (bioglass treatment): total of 12 patients (11 males and 1 female) with mean age: 50 years (30-86). Average length of hospital stay was 22 days and mean follow-up time: 23 months (16-33). Mean erythrocyte sedimentation rate (ESR) and mean c-reactive protein (CRP) before surgery: 55mm/hr and 54 mg/L, respectively. Mean ESR and mean CRP in last blood exam: 18 mm/hr and 8 mg/L, respectively. There were 2 postoperative complications: seroma formation and delayed wound healing. Only 1 patient had recurrence of infection.
Patients in group 2 (calcium sulphate antibiotic beads treatment): total of 13 patients (9 males and 4 females) with mean age: 48 years (17-67). Average length of hospital stay was 21 days and mean follow-up time 22 months (16-29). Mean ESR and mean CRP before surgery: 51mm/hr and 41 mg/L, respectively. Mean ESR and mean CRP in last blood test: 15 mm/hr and 11 mg/L. 2 postoperative complications were registered: chronic expanding hematoma of the muscle flap donor site and seroma formation. 1 patient had recurrence of infection. Overall, there were no differences in recurrence of infection, p=0.740 and in complication rate, p=0.672. 11 (91,7%) patients in group 1 and 12 (92,3%) patients in group 2 showed no signs of recurrence of infection both clinically and radiologically at final follow-up.
The most frequent cause of osteomyelitis in group 1 was post traumatic while a postsurgical aetiology was more frequent in group 2. The distal tibia was the most common location. The most frequent pathogen isolated in both groups was methicillin sensible staphylococcus aureus.
Conclusions: An advance in treatment of patients with cavitary bone defects in chronic osteomyelitis is the use of synthetic bone substitutes although current evidence is low. In this study, we demonstrate how bioglass without local antibiotics and calcium sulphate antibiotic beads are both equally effective treatment options. Overall, there were no differences between groups in mean hospital stay, complication rates and recurrence of infection.
Keywords: bioactive glass, osteomyelitis, calcium sulphate, bone substitute, cavitary defect.
Background
Osteomyelitis represents one of the most challenging conditions in orthopedic surgery. It consists of a destructive inflammatory process in the bone caused by infectious microorganisms. Staphylococcus aureus and other Gram-positive pathogens are usually the most commonly involved.1 The primary cause of infection has multiple etiologies; it can arise from haematogenous, post-traumatic or post-operative colonisation.2,3
Acute osteomyelitis is characterized by the cardinal features of inflammation along with soft tissue edema, locally decreased blood supply and pus formation. When this condition persists over time due to not-treatment or treatment failure, the infection can progress to a chronic phase, with formation of large areas of necrotic bone called sequestrum, providing the perfect place for infection recurrence.4
Given the limited ability of antibiotics to penetrate poorly vascularized or devitalized bone and surrounding tissues, treatment often requires aggressive surgical debridement, although unfortunately this often results in large bone defects. 3,5
The management of this bone defect has been addressed in various ways, which includes local or soft-tissue flaps, bone transport or local administration of antibiotics, delivered via polymethylmethacrylate (PMMA), bone graft or bone substitutes.6-8
For several years, bone defects in osteomyelitis have been treated through debridement in combination with gentamicin-loaded PMMA. However this method isn´t free of drawbacks, such as thermal damage to the antibiotic, the need of a second intervention to remove PMMA and facilitation of biofilm formation by multiresistant colonies due to inadequate antibiotic concentration.9,10
With the development of synthetic bone substitutes, such as calcium sulphate (CS) and bioglass, new possibilities of adjuvant treatment in osteomyelitis have arisen. On the one hand, CS is a biodegradable ceramic used as bone graft material since 189211 and is a well-established antibiotic carrier12. It allows obliteration of the dead space and release of the associated antibiotic after its entire degradation. However, CS has intrinsic disadvantages. First, it cannot provide structural support due to its quick hydrolysis, 6-12 weeks in bone13. In addition it has been related to wound issues owing to serous exudate14-16.
On the other hand, Bioactive glasses are synthetic materials that have been shown to have antibacterial, osteoconductive and angiogenic properties, making them ideal for treating bone defects in osteomyelitis.9,17-21 Particularly S53P4, composed by SiO2, Na2O, CaO and P2O5, has been reported to facilitate tissue growth by chemically binding to the bone matrix, thereby promoting new bone formation.22 Sodium and calcium ions are released from the surface and increase the local pH and osmotic pressure granting the antibacterial properties without the need of local antibiotics while calcium phosphates crystalize to natural hydroxyapatite, which starts the activation of osteoblasts for the formation of new bone.23,24 At present, no induction of bacterial resistance to S53P4 has been reported.
Aim
Due to the different properties of these two synthetic bone substitutes, we conducted a study to evaluate their efficacy in the treatment of patients with cavitary defects due to chronic osteomyelitis.
Patients and methods
This was a retrospective study of 25 consecutive patients with chronic osteomyelitis operated during the period of 2014-2015 in one medical center specialized in bone and joint infections. Patients were diagnosed of osteomyelitis clinically and were confirmed by radiology and laboratory results. MRI and CT scans were ordered preoperatively only in specific patients for surgical planning (to measure bone void and sinus tract extension). Patients susceptible to bone void filling were allocated into the two treatment groups (group 1: bioglass without local antibiotics, group 2: antibiotic loaded calcium sulphate) by order of consultation at the outpatient clinic in a 1:1 fashion by senior surgeon (J.B). Patients that could require a concomitant plastic surgical procedure were consulted with the Plastic Surgery Division and surgery was planned (1 stage). All patients gave informed consent and the study was conducted in accordance with the ethics committee of our institution. The primary endpoint of the study was absence of recurrence of osteomyelitis based on clinical and radiological findings and no need for further surgery. The secondary endpoints were mean hospital stay and complication rates when using the two bone substitutes.
Group 1 consisted of 12 patients that were treated with surgical debridement, systemic antibiotics and local application of Bioactive glass S53P4 (BonAlive, Bone Alive Biomaterial Ltd, Turku, Finland). Meanwhile, group 2 was composed of 13 patients treated with surgical debridement, systemic antibiotics and local application of calcium sulphate antibiotic beads (Stimulan, Biocomposite Ltd, Staffordshire, England).
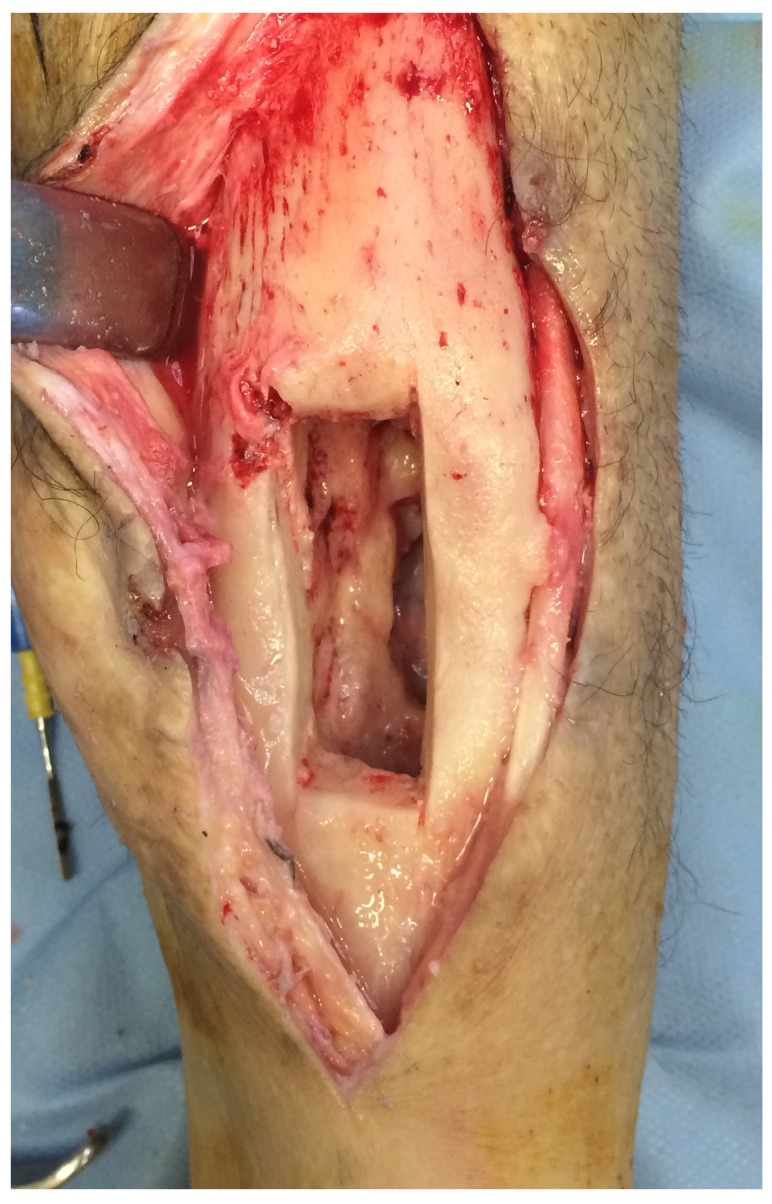
Surgery was carried out through usual technique by senior surgeon (J.B). First, incision was centered at the focus of osteomyelitis/ sinus tract. A bone window was marked first with a drill guide. The drill holes were connected by osteotomes and lastly an oscillating saw was used to perform the finishing cut (Figure 2). A thorough debridement of the medullary canal was performed with different size curettes and 6-7 samples of the infected medullary site and soft tissues (sinus tract) were collected and sent to the Microbiology Department. Posteriorly, the medullary canal (if osteomyelitis of long bone) was reamed in its entirety with a motorized burr if suspicion of infection was present (preoperative images of cement in the canal or sequestrae). Lastly, a waterjet hydrosurgery system was used copiously at primary site of osteomyelitis. After re-gloving and a new sterile field set-up, cavities were filled with bone substitutes, group 1 with bioactive glass granules and no local antibiotics, while group 2 with calcium sulphate beads with a combination of local antibiotics: vancomycin and gentamycin. Special attention was taken to completely fill the defects with the bone substitutes. To prevent the biomaterial from being distributed outside the bone, a layer of bioglass in putty form (paste solution) was used to create a physical barrier to cover the bone window in the cases in group 1. No steps were taken in the cases of group 2 with calcium sulphate beads other than skin closure. Suction drains were not used in any group.
Figure 2.
Rectangular bone window.
At discharge, patients received 6-12 weeks of systemic antibiotic therapy established by the Infectious Disease Department and according to culture results.
Patient follow-up at outpatient clinic took place at weeks 2 and 4 to check skin closure and at 3, 6 and 12 months. Radiographs were obtained in the immediate postoperative period and at 4 weeks, 6 months and yearly thereafter. CT scans were ordered in specific cases to evaluate bone substitute integration and bone void filling and were evaluated by the Musculoskeletal Radiology Section. Laboratory evaluation was conducted by the Infectious Disease Department at week 4, 6, 8, 12(depending on the microorganisms isolated and antibiotic therapy) and at 6 months and yearly thereafter. Only patients with more than one year follow-up were included in the study for comparison.
Statistical analysis
Comparison of preoperative and postoperative values was performed using the unpaired t-test. Comparison of qualitative data was performed using the chi-square test. The Kolmogorov-Smirnov test was used to assess the normality of the variables' distribution. A p-value less than 0.05 was considered significant. All statistical analyses were performed using IBM SPSS1 statistics software (Version 20.0; Chicago, IL, USA).
Results
Pre-operative data did not differ between groups compared (Table 1). Patients in group 1 (bioglass granules) had a mean age of 50 years (11 males, 1 female). Majority of patients were classified as ASA II (7 patients). The most frequent location of osteomyelitis was the tibia (7 patients), followed by the femur (4 patients) and calcaneus (1 patient). Regarding osteomyelitis etiology, six of the patients had sustained a traumatic event in the lower extremity, four patients had undergone previous operative treatments at their regional hospital and two cases had an hematogenous origin. Six patients presented an active draining sinus at index surgery (swab samples were sent for preoperative microbiological analysis). Mean erythrocyte sedimentation rate (ESR) and mean c-reactive protein (CRP) before surgery were: 55 mm/hr and 54 mg/L respectively.
Table 1.
Preoperative data.
| Group 1 (Bio active glass) | Group 2 (calcium sulphate) | P-value | |
|---|---|---|---|
| Total number of patients (n) | 12 | 13 | |
| Mean age (years), SD | 50± 18 | 48± 17 | 0.845 |
| Male/female | 11/1 | 9/4 | 0.186 |
| ASA Classification (I/II/III/IV) | 4/7/1/0 | 3/8/2/0 | 0.852 |
| Pathogenesis (haematogenous, post-traumatic, post-operative) | 2/6/4 | 0/2/11 | 0.027 |
| Location (tibia/femur/calcaneus/other) | 7/4/1/0 | 6/2/4/1 | 0.320 |
| Draining fistula at time of surgery | 6 | 7 | 0.848 |
| Mean pre-op ESR (mm/h), SD | 55± 26 | 51± 29 | 0.717 |
| Mean pre-op serum C-reactive protein (mg/l), SD | 54± 52 | 41± 31 | 0.473 |
Meanwhile, patients in group 2 (calcium sulphate antibiotic beads) had a mean age of 48 years (9 males and 4 females). Majority of patients were classified as ASA II (8 patients). The most frequent location was also the tibia (6 patients), followed by the calcaneus (4 patients), femur (2 patient) and humerus (1 patient). The most frequent etiology in this group was postsurgical (11 patients had undergone previous treatments including revisions and arthrodesis at their regional hospital) followed by posttraumatic (2 cases). Seven patients presented an active draining sinus at time of surgery and swab samples were sent for microbiological analysis. Mean ESR and mean CRP before surgery were: 51 mm/hr and 41 mg/L, respectively. The most frequent microorganism isolated for both groups was methicillin-sensitive S. aureus.
The estimated sizes of the bone defects were estimated preoperatively with CT images when available, but final amount was decided intraoperatively after adequate debridement. In the case of group 1, the mean amount of bioglass required to fill the bone defect was 17,7cc (granule size was always chosen medium,1-2mm, except in 1 patient where the bone window was very small and was mixed with size small 0,5-0,8mm for better filling) while the mean amount of calcium sulphate used was 9.4cc (pellet size 4.8mm, 10cc kit in 9 patients and 5cc kit in one patient) in combination with 1 gr of vancomycin and 240 mg of gentamycin with the 10cc kit and 500mg vancomycin and 160mg of gentamycin with the 5 cc kit. Patients in both groups had similar mean hospital stay, similar to published literature, which was not statistically significant: 22 days in group 1 and 21 days in group 2, p=0.83110. Also, patient mean follow-up at the outpatient clinic was similar in both groups, 23,3 months in group 1 (range 16-33 months) and 22 months in group 2 (range 16- 29), p=0.578. In the last laboratory test, mean ESR and mean CRP in group 1 were: 18 mm/hr and 8 mg/L respectively. Meanwhile, mean ESR and mean CRP at final follow-up in group 2 were: 15 mm/hr and 11 mg/L.
Intramedullary reaming of long bones (tibia and femur) using the Reamer-Irrigator-Aspirator system was performed in 11 patients in group 1 and 8 patients in group 2, p=0.160. Planned single stage muscle flap reconstruction was performed in 2 patients in group 1 and 1 patient in group 2, p=0.593. After aggressive debridement, in all three cases, Plastic Surgery Division designed an anterolateral thigh flap for soft tissue coverage in the tibia.
Postoperative complications were similar in both groups, p=0.672. In group 1 there were two postoperative complications. In one patient, with calcaneal osteomyelitis, the surgical wound re-opened (1cm x 1cm) in the immediate postoperative period and bioglass granules extruded from the skin upon pressure. Wound was treated during hospital stay by applying pressure and eliminating excessive granules and serous fluid. At discharge, after 10 days, wound closed. However, at the 6-month follow-up visit, patient had developed a new sinus tract. Patient is currently in the waiting list to re-operate. Another patient in group 1 with distal tibia osteomyelitis presented a delay in wound closure and bone void filling. Patient had to be re-operated by the plastic surgery department and wound was covered with lateral thigh free flap. Currently, at last follow-up, patient is asymptomatic.
In group 2, there were also 2 postoperative complications. One patient developed a chronic expanding hematoma of the muscle flap donor site which required surgical drainage. The other patient with distal tibia osteomyelitis developed a mild seroma which was treated by applying pressure and wound healed in 7 days during hospital stay. However, last hospital visit at 1 year follow-up, patient is symptomatic with pain and altered laboratory parameters.
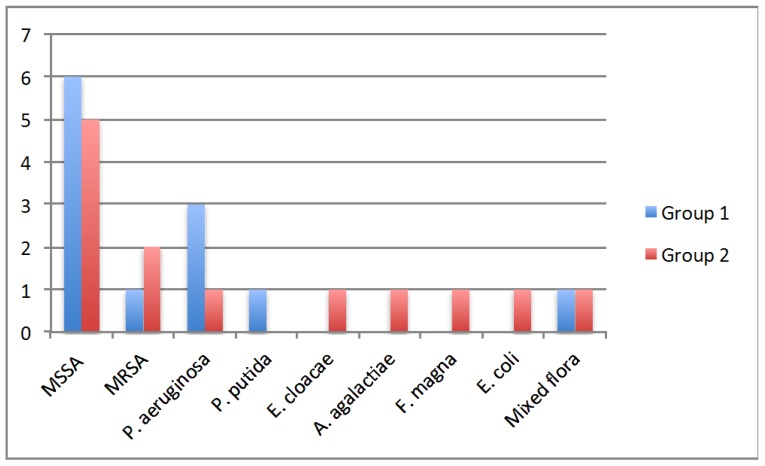
There were no differences between groups in microorganisms isolated from osteomyelitis site, p=0.576. Methicillin‐sensitive Staphylococcus aureus (MSSA) was the most frequent pathogen isolated in both groups (6/12 cases in group 1 vs 5/13 cases in group 2). Methicillin‐ resistant Staphylococcus aureus was the second most frequent isolated pathogen in group 2 (2 cases) while P. Aeruginosa was the second most frequent in group 1 (3 cases). Other micoorganisms isolated in individual cases included P. putida, E.cloacae, S. agalactiae, F. magna, E.coli and one case of mixed flora.
Overall, there were no differences in recurrence of infection, p=0.740 and in complication rate, p=0.672. 11 (91,7%) patients in group 1 and 12 (92,3%) patients in group 2 showed no signs of recurrence of infection both clinically and radiologically at final follow-up.
Discussion
This comparative study demonstrates that both bioglass and calcium sulphate are equally effective bone substitutes useful for filling bone defects. Our primary endpoint was absence of recurrence at minimum one year follow-up, and we obtained results similar to those published in literature (around 90% absence of infection in both groups)10.
Another key aspect is that we found no differences in hospital stay or complication rate when comparing both groups. We do take note, that our mean hospital stay is longer than previous studies but this can be attributed to the fact that we did not exclude patients that had local plastic surgery procedures (1 stage)10. Nevertheless, since bone substitute treatment was assigned in a 1:1 fashion between the two groups, authors assumed that patients that would require skin or muscle flaps would be distributed equally among both groups. Also, like mentioned before, wound complication rate was similar in both groups. Seroma formation appeared in both groups which resolved in a similar fashion with repeated drainage of serum fluid in alternative days in a period of one to two weeks. It is interesting to note, that both cases at last follow-up had recurrence of infection. Due to the small sample size, we cannot attribute with certainty seroma formation to recurrence of infection. Authors believe that the key for successful treatment lies in aggressive surgical debridement and correct soft tissue coverage. Probably in these two cases, surgical debridement was inadequate and a focus of osteomyelitis was not addressed.
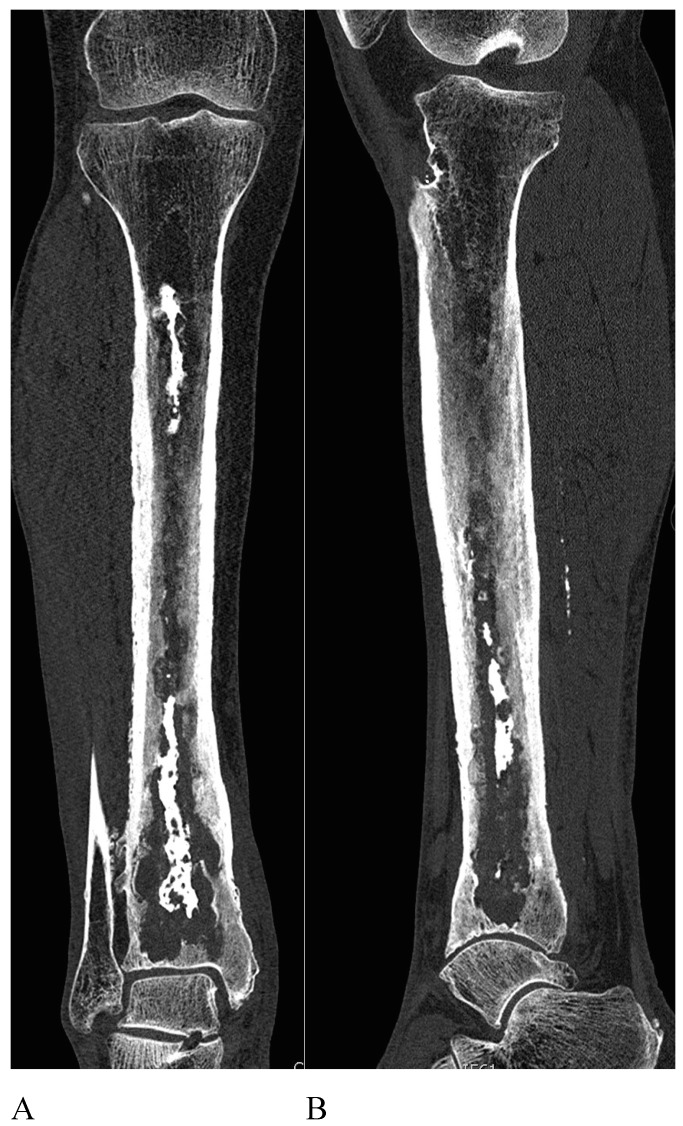
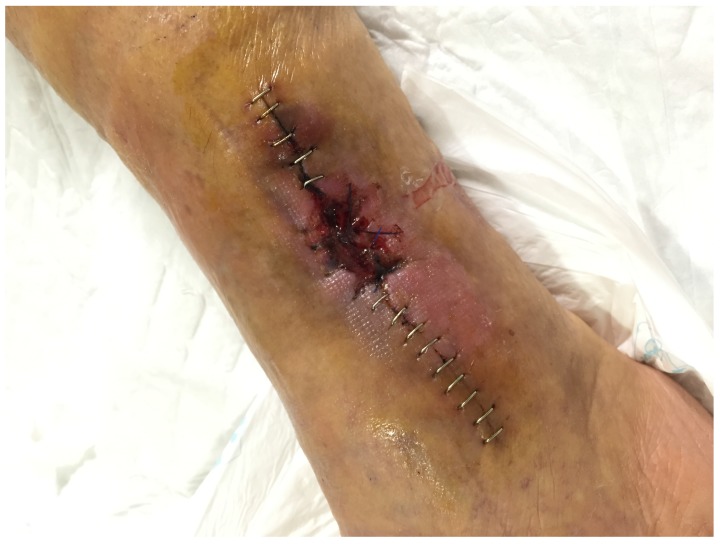
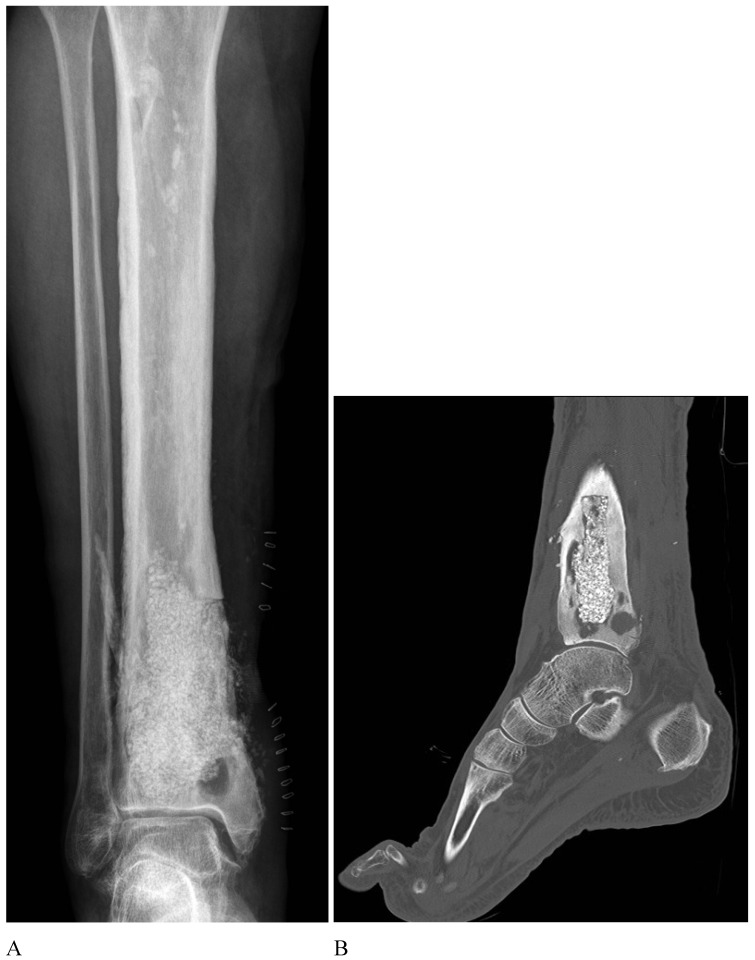
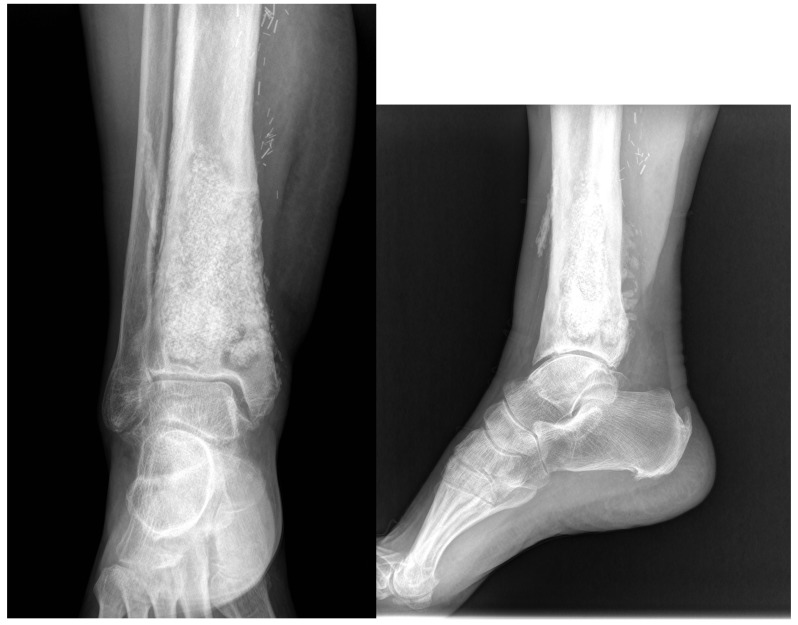
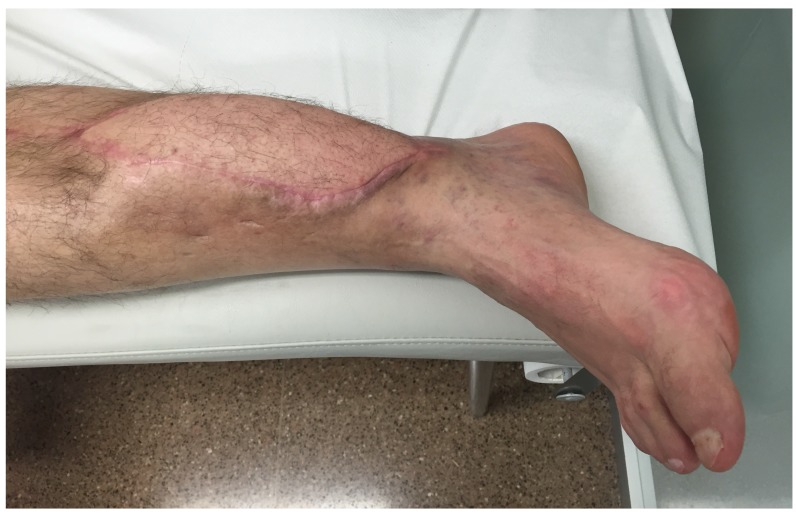
This leads us to present one case to emphasize the importance of surgical debridement of all focus. A 68-year-old patient with posttraumatic osteomyelitis of the tibia was transferred from his regional hospital to our Bone and Joint Infection Department for treatment decision. He referred several previous surgeries at his regional hospital but all had failed. On examination, patient presented an active draining sinus on medial aspect of distal tibia. Preoperative CRP and ESR were 65,3mg/L and 36mm/h respectively. X ray images and CT scan were ordered for surgical planning and bone void filling. After image analysis, patient was consented for surgery and treatment with bioglass. As evidenced with the CT scan, large areas of sequestrae were present in the whole medullary canal (Figure 3). 30cc of bioglass was estimated that would be necessary to fill the cavity. Surgery was carried out in regular fashion as described above. After debridement and medullary canal reaming, 45 cc of medium size bioglass was used to fill the cavity. Upon closure, surgical team was able to close skin but with excessive tension. Plastic surgery department had not been consulted for this case since senior surgeon had believed that a primary closure would be possible. In the immediate postoperative period, there was a delay in wound healing with border necrosis (Figure 4). In addition, post-op X ray and CT scan showed a cavity which had not been addressed during the surgical procedure even though fluoroscopy had been used (Figure 5). Consequently, the plastic surgery department was contacted, patient was consented and taken back to the surgery room. Intraoperatively, we noticed that the not-addressed cavity was independent of the primary cavity. We performed a new bone window, aggressive surgical debridement, irrigation and filled the defect with 5cc of bone glass. Then, the Plastic Surgery Team performed a lateral thigh free flap to cover the surgical wound. Postoperatively, x ray images showed adequate filling (Figure 6) of the cavity and the free muscle flap healed correctly. Patient was discharged. At 2 year follow-up patient is asymptomatic with no active draining sinus (Figure 7). In addition, similar to other studies4,10,25,26, bioglass can still be seen on the plain radiographs at 2 year post-operatively unlike calcium sulphate which has complete incorporation in all of our cases at the 6 month follow-up. We believe that this case is particularly important to demonstrate the importance of aggressive surgical debridement of all osteomyelitis focus and adequate skin coverage without tension to prevent infection recurrence. If correct filling of the second cavity had not been performed, infection recurrence would have been inevitable.
Figure 3.
Visible sequestrae along the whole medullary canal.
Figure 4.
Skin necrosis and wound dehiscence probably due to excessive tension.
Figure 5.
A. Small posteromedial cavity not filled with bioglass B. Second cavity is independent of main cavity.
Figure 6.
Correct bone void filling of all the cavities with bioglass.
Figure 7.
Last follow-up, lateral thigh free muscle flap has survived completely with absence of recurrence of infection.
Limitations of this study consist primarily of the small sample size and retrospective nature. However, this is the second largest study4,10,25 to compare two bone substitutes in one centre by same surgical team with an adequate follow-up (at least one year, mean 23 months for both groups). Also, we compared only two bone substitutes when there are many more commercially available with different efficacy profiles. Larger studies, with longer follow-up and different substitutes are desirable to establish conclusions.
Conclusions
In the antibiotic era, chronic osteomyelitis remains difficult to treat. Aggressive surgical debridement and pathogen-specific antibiotics is key to eradicate infection. An advance in treatment is the use of synthetic bone substitutes although current evidence is low. In this study, we demonstrate how bioglass and calcium sulphate antibiotic beads are both equally effective treatment options for cavitary bone defects in osteomyelitis. Both treatment groups showed similar absence of recurrence, mean hospital stay and complication rates.
Figure 1.
Microorganisms isolated in the two different treatment groups.
Table 2.
Postoperative data.
| Group 1 (Bio active glass) | Group 2 (calcium sulphate) | P-value | |
|---|---|---|---|
| Mean follow-up (months), SD | 23,3 ± 6,6 | 22± 4,4 | 0.578 |
| Mean hospital stay (days), SD | 22,5 ± 10,6 | 21,3 ± 16,1 | 0.831 |
| Mean systemic antibiotic treatment duration (weeks), SD | 8,7 ± 1,6 | 8,9 ± 1,7 | 0.704 |
| Mean post-op ESR (mm/h), SD | 18± 15 | 15± 9 | 0.576 |
| Mean post-op serum C-reactive protein (mg/l), SD | 8 ± 6 | 11± 18 | 0.547 |
| Microorganisms isolated ( MSSA, MRSA, P. aeruginosa, P. putida, E. cloacae, S. agalactiae, F. magna, E. coli, mixed flora) | 6/1/3/1/0/0/0/1 | 5/2/1/0/1/1/1/1/1 | 0.576 |
| Intramedullary 0 of long bones | 11 | 8 | 0.160 |
| Single stage muscle flap reconstruction | 2 | 1 | 0.593 |
| Complications | 2 | 2 | 0.672 |
| Infection recurrence | 1 | 1 | 0.740 |
References
- 1.Romano CL, Romano D, Logoluso N, Drago L. Bone and joint infections in adults: a comprehensive classification proposal. Eur Orthop Traumatol. 2011;1:207–217. doi: 10.1007/s12570-011-0056-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lazzarini L, Mader JT, Calhoun JH. Osteomyelitis in long bones. J Bone Joint Surg Am. 2004;86A:2305–2318. doi: 10.2106/00004623-200410000-00028. [DOI] [PubMed] [Google Scholar]
- 3.Lew DP, Waldvogel FA. Osteomyelitis. Lancet (London, England) 2004;364:369–379. doi: 10.1016/S0140-6736(04)16727-5. [DOI] [PubMed] [Google Scholar]
- 4.Lindfors NC. Bioactive glass S53P4 as a bone graft substitute in the treatment of osteomyelitis. Bioact Glas Mater Prop Appl. 2011;47:209–216. doi: 10.1016/j.bone.2010.05.030. [DOI] [PubMed] [Google Scholar]
- 5.Haas DW, McAndrew MP. Bacterial osteomyelitis in adults: evolving considerations in diagnosis and treatment. Am J Med. 1996;101:550–561. doi: 10.1016/s0002-9343(96)00260-4. [DOI] [PubMed] [Google Scholar]
- 6.Webb JCJ, Spencer RF. The role of polymethylmethacrylate bone cement in modern orthopaedic surgery. J Bone Joint Surg Br. 2007;89:851–857. doi: 10.1302/0301-620X.89B7.19148. [DOI] [PubMed] [Google Scholar]
- 7.Bridgens J, Davies S, Tilley L, Norman P, Stockley I. Orthopaedic bone cement: do we know what we are using? J Bone Joint Surg Br. 2008;90:643–647. doi: 10.1302/0301-620X.90B5.19803. [DOI] [PubMed] [Google Scholar]
- 8.Parsons B, Strauss E. Surgical management of chronic osteomyelitis. Am J Surg. 2004;188:57–66. doi: 10.1016/S0002-9610(03)00292-7. [DOI] [PubMed] [Google Scholar]
- 9.Hench LL, Paschall HA. Direct chemical bond of bioactive glass-ceramic materials to bone and muscle. J Biomed Mater Res. 1973;7:25–42. doi: 10.1002/jbm.820070304. [DOI] [PubMed] [Google Scholar]
- 10.Romanò CL, Logoluso N, Meani E, Romanò D, De Vecchi E, Vassena C. et al. A comparative study of the use of bioactive glass S53P4 and antibiotic-loaded calcium-based bone substitutes in the treatment of chronic osteomyelitis: A retrospective comparative study. Bone Jt J. 2014;96B:845–850. doi: 10.1302/0301-620X.96B6.33014. [DOI] [PubMed] [Google Scholar]
- 11.Fillingham Y, Jacobs J. Bone grafts and their substitutes. Bone Joint J. 2016;98B:6–9. doi: 10.1302/0301-620X.98B.36350. [DOI] [PubMed] [Google Scholar]
- 12.Ferguson J, Diefenbeck M, McNally M. Ceramic Biocomposites as Biodegradable Antibiotic Carriers in the Treatment of Bone Infections. J Bone Jt Infect. 2017;2:38–51. doi: 10.7150/jbji.17234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ochsner PE, Borens O, Bodler P-M. Infections of the musculoskeletal system: basic principles, prevention, diagnosis and treatment. 2014.
- 14.McKee MD, Wild LM, Schemitsch EH, Waddell JP. The use of an antibiotic-impregnated, osteoconductive, bioabsorbable bone substitute in the treatment of infected long bone defects: early results of a prospective trial. J Orthop Trauma. 2002;16:622–627. doi: 10.1097/00005131-200210000-00002. [DOI] [PubMed] [Google Scholar]
- 15.Ferguson JY, Dudareva M, Riley ND, Stubbs D, Atkins BL, McNally MA. The use of a biodegradable antibiotic-loaded calcium sulphate carrier containing tobramycin for the treatment of chronic osteomyelitis: a series of 195 cases. Bone Joint J. 2014;96B:829–836. doi: 10.1302/0301-620X.96B6.32756. [DOI] [PubMed] [Google Scholar]
- 16.Borrelli JJ, Prickett WD, Ricci WM. Treatment of nonunions and osseous defects with bone graft and calcium sulfate. Clin Orthop Relat Res. 2003:245–254. doi: 10.1097/01.blo.0000069893.31220.6f. [DOI] [PubMed] [Google Scholar]
- 17.Munukka E, Lepparanta O, Korkeamaki M, Vaahtio M, Peltola T, Zhang D. et al. Bactericidal effects of bioactive glasses on clinically important aerobic bacteria. J Mater Sci Mater Med. 2008;19:27–32. doi: 10.1007/s10856-007-3143-1. [DOI] [PubMed] [Google Scholar]
- 18.Lindfors NC, Aho AJ. Granule size and composition of bioactive glasses affect osteoconduction in rabbit. J Mater Sci Mater Med. 2003;14:365–372. doi: 10.1023/a:1022988117526. [DOI] [PubMed] [Google Scholar]
- 19.Lepparanta O, Vaahtio M, Peltola T, Zhang D, Hupa L, Hupa M. et al. Antibacterial effect of bioactive glasses on clinically important anaerobic bacteria in vitro. J Mater Sci Mater Med. 2008;19:547–551. doi: 10.1007/s10856-007-3018-5. [DOI] [PubMed] [Google Scholar]
- 20.Day RM. Bioactive glass stimulates the secretion of angiogenic growth factors and angiogenesis in vitro. Tissue Eng. 2005;11:768–777. doi: 10.1089/ten.2005.11.768. [DOI] [PubMed] [Google Scholar]
- 21.Andersson OH, Kangasniemi I. Calcium phosphate formation at the surface of bioactive glass in vitro. J Biomed Mater Res. 1991;25:1019–1030. doi: 10.1002/jbm.820250808. [DOI] [PubMed] [Google Scholar]
- 22.Valimaki V V, Aro HT. Molecular basis for action of bioactive glasses as bone graft substitute. Scand J Surg. 2006;95:95–102. doi: 10.1177/145749690609500204. [DOI] [PubMed] [Google Scholar]
- 23.Jones JR. Review of bioactive glass: from Hench to hybrids. Acta Biomater. 2013;9:4457–4486. doi: 10.1016/j.actbio.2012.08.023. [DOI] [PubMed] [Google Scholar]
- 24.Van Gestel NAP, Geurts J, Hulsen DJW, Van Rietbergen B, Hofmann S, Arts JJ. Clinical Applications of S53P4 Bioactive Glass in Bone Healing and Osteomyelitic Treatment: A Literature Review. Biomed Res Int 2015; 2015. doi:10.1155/2015/684826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McAndrew J, Efrimescu C, Sheehan E, Niall D. Through the looking glass; Bioactive glass S53P4 (BonAlive®) in the treatment of chronic osteomyelitis. Ir J Med Sci. 2013;182:509–511. doi: 10.1007/s11845-012-0895-5. [DOI] [PubMed] [Google Scholar]
- 26.Aurégan J-C, Bégué T. Bioactive glass for long bone infection: a systematic review. Injury. 2015;46:S3–S7. doi: 10.1016/S0020-1383(15)30048-6. [DOI] [PubMed] [Google Scholar]