Abstract
Purpose The aim of the study was to evaluate the clinical effects of HYADD® 4, an hydrogel based on a hyaluronic acid derivative, in patients with symptomatic knee osteoarthritis, on symptoms, and joint function.
Methods This retrospective study of patients with Kellgren–Lawrence grade II to IV knee osteoarthritis (American College of Rheumatology criteria) enrolled patients who had received two infiltrations of HYADD® 4, (24 mg/3 mL) 1 week apart, and evaluated: pain at rest, pain with movement, change in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score; change in nonsteroidal anti-inflammatory drugs (NSAIDs)/acetaminophen use; satisfaction with therapy; tolerability. Study duration was 6 months for all predefined endpoints, with a 6-month extension for pain symptoms only.
Results After 6 months, all predefined endpoints were evaluable in 698 of 937 enrolled patients (74.5%). Mean WOMAC scores were reduced by 56.3% from baseline ( p < 0.05). NSAIDs/acetaminophen use ≥2 times/week (48.8% of patients at baseline) was substantially reduced after 1 month and was 19.6% after 6 months. After 6 months, 85.6% of patients were satisfied about efficacy. There were no significant adverse effects. The effect on resting pain was rapid, strong, and lasting: reduction from baseline was 45.1% at 1 month ( p < 0.05), 56.8% at 6 months ( p < 0.05), and 53.6% at 12 months ( p < 0.05). Pain on moving was reduced by 47.4% after 6 months ( p < 0.05) and 46.0% after 12 months ( p < 0.05), results at 6 and 12 months were similar.
Conclusion HYADD® 4 is a new-generation hyaluronic acid with distinctive viscoelastic and rheological properties. In patients with mild-to-severe knee OA (Kellgren–Lawrence grades II–IV), two consecutive infiltrations 1 week apart reduced WOMAC scores and NSAIDs/acetaminophen consumption for at least 6 months. In a subpopulation ( n = 106), efficacy on pain lasted approximately 12 months. Adverse events were reported in 11.2% of patients; the most frequent were arthralgias. No cases of allergic reactions or systemic effects were recorded.
Level of Evidence Level IV, retrospective case series.
Keywords: hyaluronic acid, osteoarthritis, knee, viscosupplementation, Hymovis®, viscoelastic
Introduction
Osteoarthritis (OA) is the most common form of arthritis in industrialized countries, with prevalence in the general population of Caucasian ethnicity largely stable for decades between 3.4 and 6.1%. 1 The prevalence increases progressively with age and becomes significant after 60 years (range, 10–15%), with women affected more frequently. 2 The most commonly affected joints are the hip and knee. Patients with OA experience functional disability and progressive deterioration in the quality of life (QoL). The impact on function and QoL depend on the number of joints involved and the severity of joint damage. Mobility can be seriously impaired in patients with severe bilateral knee and/or hip OA. Such patients are at an increased risk of accidental falls that can result in fractures, emergency surgery, permanent disability, and premature death. 3 Recent studies show that functional disability from hip or knee OA is associated with a higher incidence of metabolic disease (e.g., diabetes, obesity) and cardiovascular disease (e.g., heart attack, stroke, atherothrombotic vascular disease) due to physical inactivity and weight gain. 4 5
The causes of OA are only partially known, even though the major risk factors and pathogenic mechanisms of cartilage damage have been identified in recent decades. 6 7 Articular cartilage wear is a common denominator, into which the effects of various risk factors are summed. For example, knee OA is approximately 10 times more common in weight lifters and 5 times more common in runners, 8 as a result of repeated microtrauma to cartilage. These values further increase in individuals with a history of the ruptured meniscus or cruciate ligament. In such patients, altered biomechanics can cause dynamic loads to impinge on the cartilage along nonphysiological pressure lines. 9 Recently, obesity has emerged as a potent promoter of hyaline cartilage degeneration. When overweight begins in childhood, the risk of early (<50 years) knee and hip OA is much higher compared with that in the normal weight population. 10 11 12
OA treatment may comprise conservative interventions (physical therapy), medication (local or systemic) or surgery. 13 The guidelines of the Osteoarthritis Research Society International (OARSI) suggest that interventions should be progressive and proportionate to disease severity. 14 Pharmacological interventions may include nonsteroidal anti-inflammatory drugs (NSAIDs), pure analgesics (acetaminophen) and disease-modifying therapies. 15 16
The costs associated with pharmaceutical therapy and arthroplasty for OA are substantial and increasing. Piscitelli et al have calculated that between 2001 and 2005, the knee arthroplasty interventions in Italy increased by 16.6% for men and 12.4% for women, with an exponential increase in direct and indirect economic costs. 17 Consequently, also on the basis of economic considerations, there is a tendency to use measures that prevent or delay recourse to arthroplasty.
Intra-articular infiltration (viscosupplementation) with derivatives of hyaluronic acid (HA), a polysaccharide polymer naturally present in articular cartilage, is a therapeutic measure that restores the rheological properties of synovial fluid and stimulates the chondrocytes that produce endogenous HA. 18 19 Whereas intra-articular HA injection is included in several major guidelines on managing OA, 14 20 conflicting evidence for its efficacy has resulted in a lack of agreement among various guidelines regarding its use. 21 The discrepancy between the beneficial effects of this procedure observed in clinical practice and guideline recommendations may result from study characteristics, such as inclusion criteria and the form of HA used. 22 23 24 Currently, several HA formulations are approved for clinical use in Europe and the United States. These formulations differ in the origin of the HA and production methods used, in their chemical–physical properties, joint space half-life, rheological properties, as well as their administration schedules and cost. 25 26
HYADD® 4 (Hymovis®; Fidia Farmaceutici, Abano Terme, Italy) is a derivative of HA obtained by controlled chemical synthesis, with a molecular weight between 500 and 730 kDa. In Hymovis®, 2% of the carboxy radicals on the glucuronic acid present in the polysaccharide chain are conjugated with an aliphatic amine (hexadecylamine). This chemical structure makes the three-dimensional lattice of Hymovis® particularly hygroscopic and forms a hydrogel with excellent viscoelastic, lubricating, and rheological properties.
Hymovis® has been shown to significantly delay cartilage degeneration in a rabbit model of anterior cruciate ligament rupture. 27 Moreover, controlled clinical studies with Hymovis® have demonstrated its superiority to Hyalgan (Fidia Farmaceutici) both in terms of reduction in synovial hyperplasia and increased synthesis of high-molecular-weight HA by chondrocytes, with fewer Hymovis® infiltrations. 28 29 The present study investigated the efficacy and safety of viscosupplementation with Hymovis® in patients with knee OA. The hypothesis of the study was that viscosupplementation with Hymovis® in patients with knee OA improves subjective outcome and knee function with no significant adverse events.
Methods
Study Design
This was a retrospective observational study designed to evaluate the clinical efficacy and tolerability of two intra-articular Hymovis® infiltrations (24 mg/3 mL) administered 1 week apart in everyday clinical practice. Data were obtained from the ANTIAGE national register, a nonprofit database of clinical data on the effects of ultrasound-guided intra-articular viscosupplementation. All patients were evaluated at 1 month and subsequently at 3, 6, 9, and 12 months ( Table 1 ).
Table 1. Monitoring of clinical parameters during the study.
| End point | Baseline ( n = 937) |
Month 1 ( n = 821) |
Month 3 ( n = 743) |
Month 6 ( n = 698) |
Month 9 ( n = 137) |
Month 12 ( n = 106) |
|---|---|---|---|---|---|---|
| Pain at rest (VAS score) | 55.9 ± 11.4 | 31.8 ± 9.7 | 24.9 ± 10.2 | 24.1 ± 9.1 | 25.3 ± 7.3 | 25.9 ± 6.9 |
| Pain on movement (VAS score) | 72.1 ± 9.8 | 48.3 ± 6.7 | 38.2 ± 7.7 | 37.9 ± 7.3 | 38.4 ± 6.5 | 38.9 ± 11.5 |
| WOMAC score | 56.8 ± 8.7 | 45.7 ± 7.9 | 27.6 ± 10.3 | 24.8 ± 8.8 | N/A | N/A. |
| Acetaminophen/NSAIDs use | 48.8% | 27.4% | 19.4% | 19.6% | N/A | N/A |
| Patient satisfaction | ||||||
| Worsening (%) | 2.4 | 3.2 | 4.8 | |||
| Unchanged (%) | 9.3 | 8.9 | 9.6 | |||
| Improved (%) | 88.3 | 87.9 | 85.6 | |||
Abbreviations: N/A, not available; NSAID, nonsteroidal anti-inflammatory drug; VAS, visual analog scale; WOMAC, Western Ontario McMasters University Osteoarthritis Index.
Patients
Eligible patients met the following evaluation criteria: age ranging from 41 to 80 years; symptomatic knee arthritis diagnosed according to the American College of Rheumatology (ACR) criteria; 30 Kellgren–Lawrence disease severity grades II to IV; disease duration of ≥2 years; and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) total score >30 points. Patients were excluded if they had other chronic rheumatic or autoimmune diseases, cancers, joint line collapse, a history of allergy to HA or its derivatives. Also, patients receiving anticoagulants and those who had received viscosupplementation or corticosteroids (CS) infiltration in the previous 12 months were excluded.
Outcomes Measures
The study duration was 6 months for all preestablished end points, with a 12-month extension for the parameters pain at rest and pain in motion. Data on these two parameters were available at month 9 ( n = 137) and month 12 ( n = 106).
The reference standard for evaluating patients with hip or knee OA is the WOMAC questionnaire, which detects clinically relevant data in three specific areas: joint pain at rest and in motion, articular function, and joint stiffness. The total WOMAC score can range from 0 (no impairment) to 96 points (maximum impairment). Conventionally, treatments are considered efficacious if they provide a clinically significant reduction in WOMAC score of ≥20 points. 31 The WOMAC questionnaire measures pain using a Likert scale, where 0 corresponds to no pain and 4 to maximum pain; it also provides an accurate assessment of functionality and joint stiffness by measuring the ability to perform specific movements. Scores below 25 indicate mild impairment, while scores above 50 indicate moderate-to-severe impairment. Scores above 75 indicate severe joint impairment.
Pain at rest and pain in motion were also evaluated on a visual analog scale (VAS) in millimeters, with scores from 0 to 100 mm.
Because 48.8% of included patients were using acetaminophen and/or NSAIDs at baseline, a reduction in the number of patients requiring these medications was considered a reliable endpoint of clinical efficacy.
All patients were asked to answer a question on a subjective evaluation of the results, in terms of pain reduction and joint function. The three options were: (1) improvement; (2) worsening; and (3) no change.
All local and systemic adverse events (AEs) were recorded. AE associated with the first infiltration were recorded during the first week, whereas those after the second infiltration were recorded at months 1, 3, and 6.
Statistical Analysis
The changes from baseline for each end point under study were analyzed with the chi-square test. Two measurements were performed for each parameter, and the mean ± standard deviation (SD) reported. Excel spreadsheets were used for data management (Microsoft, Redmond, Washington, United States). Statistical analysis was performed with Sigma Stat software (SPSS 9; SPSS, Chicago, Illinois, United States). Significance was set for values of p < 0.05 (5%).
Results
Data from 937 eligible patients, 534 women (57%), were extracted from the database ( Table 2 ). Of these, 210 had bilateral disease. In total, 1,147 knee joints were treated, for a total of 2,294 infiltrations. Mean disease duration was 6.8 ± 6.6 years. Disease severity according to the Kellgren–Lawrence classification ranged between grade II (38%) and grade IV (7%). Most patients (55%) had grade III disease, and 39.2% had received the previous infiltration with HA or CS.
Table 2. Clinical and demographic characteristics of the study population.
| Item | Value |
|---|---|
| Age (y) (mean ± SD) | 66 ± 11.4 |
| Women ( n [%]) | 534 (57%) |
| Men ( n [%]) | 403 (43%) |
| BMI (kg/m 2 ) (mean ± SD) | 27.09 ± 3.6 |
| Duration OA (y) (mean ± SD) | 6.8 ± 6.6 |
| Kellgren–Lawrence grade | |
| Grade II ( n [%]) | 356 (38%) |
| Grade III ( n [%]) | 515 (55%) |
| Grade IV ( n [%]) | 66 (7%) |
| Right knee ( n [%]) | 484 (51.7%) |
| Left knee ( n [%]) | 243 (25.9%) |
| Bilateral ( n [%]) | 210 (22.4%) |
| Previous use of HA or CS (mean ± SD) | 39.2 ± 5.6 |
| Pain at rest (VAS score) (mean ± SD) | 55.9 ± 11.3 |
| Pain on movement (VAS score) (mean ± SD) | 72.1 ± 9.8 |
| Acetaminophen/NSAIDs use ≥2 times/wk (%) (mean ± SD) | 48.8 ± 6.1 |
| Comorbidity (%) (mean ± SD) | 19.5 ± 3.9 |
| WOMAC | |
| Pain (Likert 0–4) (mean ± SD) | 11.5 ± -2.7 |
| Physical function (mean ± SD) | 39.8 ± 8.3 |
| Stiffness (mean ± SD) | 5.5 ± 3.7 |
| Total score (mean ± SD) | 56.8 ± 9.9 |
Abbreviations: CS, corticosteroids; HA, hyaluronic acid; NSAID, nonsteroidal anti-inflammatory drug; SD, standard deviation; VAS, visual analog scale; WOMAC, Western Ontario McMasters University Osteoarthritis Index.
Impairment at baseline measured with the WOMAC questionnaire was 56.8 ± 9.9 points out of a possible 96. Accordingly, 48.8% ± 6.1% made regular use (≥2 times/week) of NSAIDs and/or acetaminophen to relieve pain when performing daily activities.
At the first follow-up (1 month), 821/937 patients (87.6%) were evaluable; at the second follow-up, 743/937 patients (79.3%) were evaluated; in the sixth month, the 698/937 patient data were available (74.5%). By month 12, it was possible to examine the data relating to only two parameters (pain at rest and in motion) in a subpopulation of 106 patients ( Table 1 ). The dropout was due to the failure of patients to present for scheduled visits or incomplete compilation of medical records, with missing data relevant to the clinical evaluation (e.g., missing annotation about acetaminophen/NSAIDs use). Patients with bilateral knee OA were excluded if results were discordant between the two affected joints because the protocol was set to report only one answer per patient. After the 12-month extension, 106 patients had evaluable data for the pain outcomes.
The effect on pain was rapid, with a reduction of 24.1 points (43.1%) after the first month ( Fig. 1 ). The analgesic action lasted until the third month, a decrease of 31 points (55.4%), remaining largely stable between the second and third visits. At the end of the first phase of the study (6 months), pain at rest was reduced by 56.8%. The same trend was observed for pain on moving, with a slightly smaller reduction from baseline in the sixth month of 47.4%. These changes were statistically significant ( p < 0.05) for both parameters already after the first month.
Fig. 1.
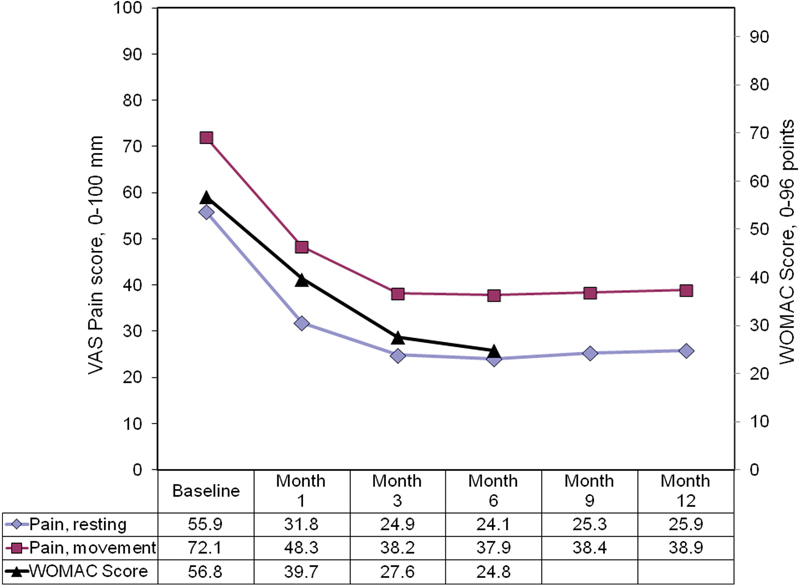
Trend in pain scores and WOMAC scores after two Hymovis® infiltrations. WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
For the pain parameters, available data allowed assessments to a total of 12 months for a small cohort of patients ( n = 106). Neither of the pain parameters had clinically or statistically significant changes between months 6 and 12. Compared with baseline, the reduction in pain at rest was 54.7% at month 9 ( p < 0.05) and 53.6% at month 12 ( p < 0.05). Compared with the maximum reduction in resting pain recorded at month 3 (56.8%), the loss of efficacy through month 12 was 3.2% ( p = nonsignificant). A similar trend was observed for pain in motion, with reductions equal to 47.4 and 46.0% at months 6 and 12, respectively. Also, in this case, the difference between months 6 and 12 is not statistically significant ( p > 0.91).
The mean WOMAC (SD) score was reduced by 17.1 points (30.1%) 1 month after the second infiltration ( p < 0.05), and was reduced by 24.8 points (56.3%) after 6 months ( p < 0.05). The mean (SD) score after 6 months was 24.8 ( ± 8.8) points, corresponding to mild impairment. It was not possible to assess the WOMAC score during the extension to 12 months. At the first visit, only 225/821 evaluable patients (27.4%) continued to take acetaminophen or NSAIDs at least twice a week ( Fig. 2 ).
Fig. 2.
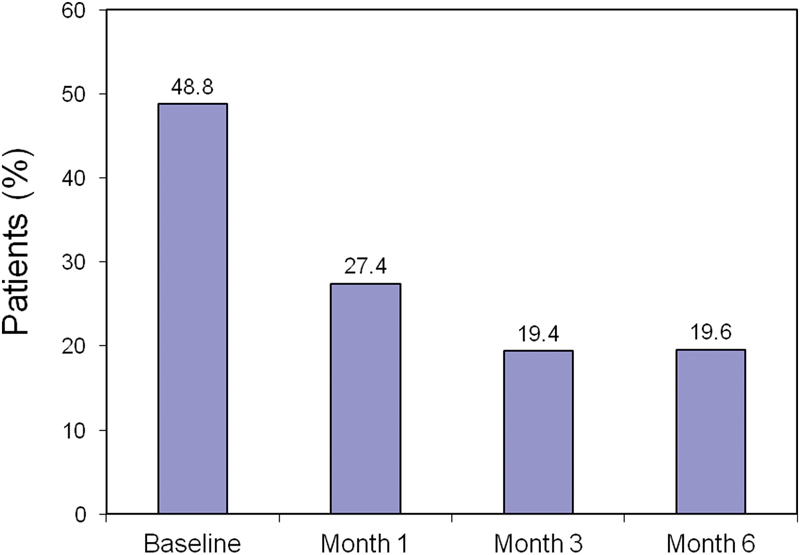
The use of NSAIDs or acetaminophen during the study. NSAID, nonsteroidal anti-inflammatory drug.
At 6 months, only 137 patients continued to take NSAIDs or acetaminophen (19.6%) with a frequency of more than two times per week. A subanalysis conducted on 561 patients who had stopped taking painkillers with this frequency, revealed that 178 patients had not taken any such medication from the second month until study end; these were classified as “drug-free.” The reduction from baseline in use was highest in the third month (19.4%). These data suggest that the effect of viscosupplementation on the use of anti-inflammatory and analgesic drugs is both rapid and durable.
Data on the pain response and the number of patients taking NSAIDs/acetaminophen correlate well with the degree of subjective satisfaction with therapy: 3 months after infiltration, 87.9% of the patients reported an improvement in symptoms ( Fig. 3 ). This improvement remained largely stable over the first 6 months of observation.
Fig. 3.
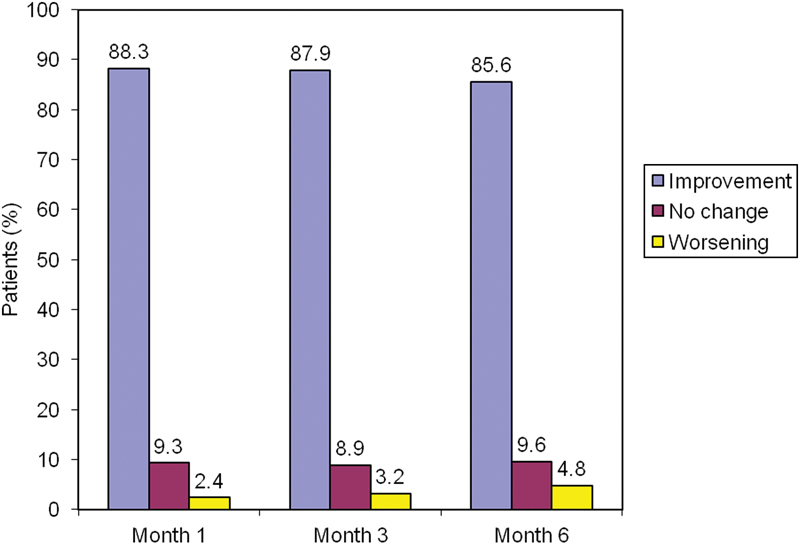
The degree of patient satisfaction with therapy.
AEs were reported in 105 patients (11.2%). Arthralgias were the most frequent AEs, and they had a relatively rapid onset of 12 to 36 hours, rarely lasting more than 2 days. No AEs were observed after the first 4 weeks from infiltration, suggesting that surveillance should be focused in this time span. None of the AEs prevented the second infiltration, which was planned at a distance of 1 week from the first. There are no recorded cases of allergic reactions or systemic effects of the drug.
Discussion
The effects of viscosupplementation with HA are supported by numerous experimental and clinical studies and several meta-analyses, although there is no unanimous agreement on the effect size. 25 32 In a recent consensus organized in Italy, 52 clinicians with expertise in intra-articular infiltration agreed that the viscosupplementation with HA is effective and appropriate in patients with Kellgren–Lawrence grade II to IV knee OA. 33 In vitro studies and experiments in animal models have demonstrated the efficacy and tolerability of Hymovis®. 27 Our retrospective study demonstrated that two administrations of Hymovis® at a distance of 1 week not only reduce pain at rest and pain in the movement for approximately 1 year but also provide considerable sparing of NSAIDs and/or acetaminophen use. This sparing effect was documented for at least 6 months. However, the favorable effects on the pain that we documented for up to 12 months suggest that the medication-sparing effect may last longer. The lack of corroborating data from the WOMAC score at 12 months does not invalidate the significant results observed on resting and movement-related pain.
Because patients with symptomatic OA are elderly and receiving multiple drug therapies, a reduction in NSAID consumption may results in a lower risk of AEs, both direct events (e.g., gastrointestinal bleeding, ulcers from NSAIDs) or indirect events caused by pharmacological interference (e.g., with antihypertensive drugs, cortisone). Based on these results, it may be appropriate to consider treating also patients with grade I knee OA who are at an increased risk of pharmacological interference or NSAID-induced gastropathy, in accordance with the opinions expressed by a significant proportion of the participants in the aforementioned clinical consensus. 33 Regarding overall tolerability, we recorded a slightly lower AE rate than that reported by other authors, although the average age of our sample is higher. 16
As with all retrospective studies, ours also has limitations related to design, but these are offset by a large number of evaluable patients and the long duration of the observation period.
Conclusion
In conclusion, our results indicate that Hymovis® is effective and safe in patients with mild-to-severe knee OA. Prospective studies with adequate population size and design are needed to better assess clinical effect size, optimal infiltration schedule, and criteria for patient selection. We await the results of the ongoing prospective randomized, double-blind efficacy study (Safety and Effectiveness Study of a Non-Crosslinked HA Alkylamide HYADD® 4 Hydrogel for Osteoarthritis of the Knee; ClinicalTrials.gov NCT02187549).
References
- 1.Cross M, Smith E, Hoy D et al. The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis. 2014;73(07):1323–1330. doi: 10.1136/annrheumdis-2013-204763. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Y, Jordan J M. Epidemiology of osteoarthritis. Clin Geriatr Med. 2010;26(03):355–369. doi: 10.1016/j.cger.2010.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Barbour K E, Lui L Y, Nevitt M C et al. Hip osteoarthritis and the risk of all-cause and disease-specific mortality in older women: a population-based cohort study. Arthritis Rheumatol. 2015;67(07):1798–1805. doi: 10.1002/art.39113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nüesch E, Dieppe P, Reichenbach S, Williams S, Iff S, Jüni P. All cause and disease specific mortality in patients with knee or hip osteoarthritis: population based cohort study. BMJ. 2011;342:d1165. doi: 10.1136/bmj.d1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Veronese N, Cereda E, Maggi S et al. Osteoarthritis and mortality: A prospective cohort study and systematic review with meta-analysis. Semin Arthritis Rheum. 2016;46(02):160–167. doi: 10.1016/j.semarthrit.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 6.Felson D T, Zhang Y, Hannan M T et al. Risk factors for incident radiographic knee osteoarthritis in the elderly: the Framingham Study. Arthritis Rheum. 1997;40(04):728–733. doi: 10.1002/art.1780400420. [DOI] [PubMed] [Google Scholar]
- 7.Muraki S, Akune T, Oka H et al. Incidence and risk factors for radiographic knee osteoarthritis and knee pain in Japanese men and women: a longitudinal population-based cohort study. Arthritis Rheum. 2012;64(05):1447–1456. doi: 10.1002/art.33508. [DOI] [PubMed] [Google Scholar]
- 8.Kujala U M, Kettunen J, Paananen H et al. Knee osteoarthritis in former runners, soccer players, weight lifters, and shooters. Arthritis Rheum. 1995;38(04):539–546. doi: 10.1002/art.1780380413. [DOI] [PubMed] [Google Scholar]
- 9.Louboutin H, Debarge R, Richou J et al. Osteoarthritis in patients with anterior cruciate ligament rupture: a review of risk factors. Knee. 2009;16(04):239–244. doi: 10.1016/j.knee.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 10.Philbin E F, Ries M D, Groff G D, Sheesley K A, French T S, Pearson T A. Osteoarthritis as a determinant of an adverse coronary heart disease risk profile. J Cardiovasc Risk. 1996;3(06):529–533. [PubMed] [Google Scholar]
- 11.Sowers M R, Karvonen-Gutierrez C A. The evolving role of obesity in knee osteoarthritis. Curr Opin Rheumatol. 2010;22(05):533–537. doi: 10.1097/BOR.0b013e32833b4682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Widhalm H K, Marlovits S, Welsch G H et al. Obesity-related juvenile form of cartilage lesions: a new affliction in the knees of morbidly obese children and adolescents. Eur Radiol. 2012;22(03):672–681. doi: 10.1007/s00330-011-2281-5. [DOI] [PubMed] [Google Scholar]
- 13.Anandacoomarasamy A, March L. Current evidence for osteoarthritis treatments. Ther Adv Musculoskelet Dis. 2010;2(01):17–28. doi: 10.1177/1759720X09359889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang W, Moskowitz R W, Nuki G et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage. 2008;16(02):137–162. doi: 10.1016/j.joca.2007.12.013. [DOI] [PubMed] [Google Scholar]
- 15.Sofat N, Beith I, Anilkumar P G, Mitchell P. Recent clinical evidence for the treatment of osteoarthritis: what we have learned. Rev Recent Clin Trials. 2011;6(02):114–126. doi: 10.2174/157488711795177886. [DOI] [PubMed] [Google Scholar]
- 16.Altman R D, Akermark C, Beaulieu A D, Schnitzer T; Durolane International Study Group.Efficacy and safety of a single intra-articular injection of non-animal stabilized hyaluronic acid (NASHA) in patients with osteoarthritis of the knee Osteoarthritis Cartilage 20041208642–649. [DOI] [PubMed] [Google Scholar]
- 17.Piscitelli P, Iolascon G, Di Tanna G et al. Socioeconomic burden of total joint arthroplasty for symptomatic hip and knee osteoarthritis in the Italian population: a 5-year analysis based on hospitalization records. Arthritis Care Res (Hoboken) 2012;64(09):1320–1327. doi: 10.1002/acr.21706. [DOI] [PubMed] [Google Scholar]
- 18.Moreland L W. Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: mechanisms of action. Arthritis Res Ther. 2003;5(02):54–67. doi: 10.1186/ar623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ghosh P, Guidolin D. Potential mechanism of action of intra-articular hyaluronan therapy in osteoarthritis: are the effects molecular weight dependent? Semin Arthritis Rheum. 2002;32(01):10–37. doi: 10.1053/sarh.2002.33720. [DOI] [PubMed] [Google Scholar]
- 20.Brown G A. AAOS clinical practice guideline: treatment of osteoarthritis of the knee: evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013;21(09):577–579. doi: 10.5435/JAAOS-21-09-577. [DOI] [PubMed] [Google Scholar]
- 21.Nelson A E, Allen K D, Golightly Y M, Goode A P, Jordan J M. A systematic review of recommendations and guidelines for the management of osteoarthritis: The chronic osteoarthritis management initiative of the U.S. bone and joint initiative. Semin Arthritis Rheum. 2014;43(06):701–712. doi: 10.1016/j.semarthrit.2013.11.012. [DOI] [PubMed] [Google Scholar]
- 22.Fernandes L, Hagen K B, Bijlsma J W et al. EULAR recommendations for the non-pharmacological core management of hip and knee osteoarthritis. Ann Rheum Dis. 2013;72(07):1125–1135. doi: 10.1136/annrheumdis-2012-202745. [DOI] [PubMed] [Google Scholar]
- 23.Henrotin Y, Raman R, Richette P et al. Consensus statement on viscosupplementation with hyaluronic acid for the management of osteoarthritis. Semin Arthritis Rheum. 2015;45(02):140–149. doi: 10.1016/j.semarthrit.2015.04.011. [DOI] [PubMed] [Google Scholar]
- 24.Migliore A, Bizzi E, Herrero-Beaumont J, Petrella R J, Raman R, Chevalier X. The discrepancy between recommendations and clinical practice for viscosupplementation in osteoarthritis: mind the gap! Eur Rev Med Pharmacol Sci. 2015;19(07):1124–1129. [PubMed] [Google Scholar]
- 25.Bellamy N, Campbell J, Robinson V, Gee T, Bourne R, Wells G. Viscosupplementation for the treatment of osteoarthritis of the knee. Cochrane Database Syst Rev. 2006;(02):CD005321. doi: 10.1002/14651858.CD005321.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Finelli I, Chiessi E, Galesso D, Renier D, Paradossi G. A new viscosupplement based on partially hydrophobic hyaluronic acid: a comparative study. Biorheology. 2011;48(05):263–275. doi: 10.3233/BIR-2011-0596. [DOI] [PubMed] [Google Scholar]
- 27.Mainil-Varlet P, Schiavinato A, Ganster M M. Efficacy evaluation of a new hyaluronan derivative HYADD® 4-G to maintain cartilage integrity in a rabbit model of osteoarthritis. Cartilage. 2013;4(01):28–41. doi: 10.1177/1947603512455193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smith M M, Cake M A, Ghosh P, Schiavinato A, Read R A, Little C B. Significant synovial pathology in a meniscectomy model of osteoarthritis: modification by intra-articular hyaluronan therapy. Rheumatology (Oxford) 2008;47(08):1172–1178. doi: 10.1093/rheumatology/ken219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cake M, Read R, Edwards S et al. Changes in gait after bilateral meniscectomy in sheep: effect of two hyaluronan preparations. J Orthop Sci. 2008;13(06):514–523. doi: 10.1007/s00776-008-1279-6. [DOI] [PubMed] [Google Scholar]
- 30.Aletaha D, Neogi T, Silman A J et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62(09):2569–2581. doi: 10.1002/art.27584. [DOI] [PubMed] [Google Scholar]
- 31.Dowsey M M, Choong P FM. The utility of outcome measures in total knee replacement surgery. Int J Rheumatol. 2013;2013:506518. doi: 10.1155/2013/506518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lo G H, LaValley M, McAlindon T, Felson D T. Intra-articular hyaluronic acid in treatment of knee osteoarthritis: a meta-analysis. JAMA. 2003;290(23):3115–3121. doi: 10.1001/jama.290.23.3115. [DOI] [PubMed] [Google Scholar]
- 33.Paoloni M, Bernetti A, Belelli A et al. Appropriateness of clinical and organizational criteria for intra-articular injection therapies in osteoarthritis. A Delphi method consensus initiative among experts in Italy. Ann Ist Super Sanita. 2015;51(02):131–138. doi: 10.4415/ANN_15_02_11. [DOI] [PubMed] [Google Scholar]


