Abstract
Objective(s):
Tuberculosis (TB) has still remained a global health issue. One third of the world’s population is infected with tuberculosis and the current BCG vaccine has low efficiency; hence, it is necessary to develop a new vaccine against TB. The aim of the current study was to evaluate the efficiency of a novel DNA vaccine encoding Mtb32C-HBHA antigen in inducing specific immune responses against Mycobacterium tuberculosis.
Materials and Methods:
A DNA plasmid vaccine expressing Mtb32C-HBHA fusion protein was constructed and its ability in protein expression was examined by RT-PCR and Western blot methods. Female BALB/c mice were vaccinated with 100 μg of purified recombinant vector in an attempt to assess its immunogenicity and protective efficacy. Further, the cytokines, IFN-γ, IL-12, IL-4, IL-10, and TGF-β were assessed.
Results:
The levels of all the studied cytokines were significantly increased (P<0.05) compared with the control group. IFN-γ production in the group receiving DNA vaccine plus BCG was increased compared with those receiving only DNA vaccine or BCG (P<0.001).
Conclusion:
The immunogenicity of the new chimeric DNA vaccine was confirmed alone and in combination with BCG. Based on the results of the current study, the constructed DNA vaccine induced the expression of Mtb32C-HBHA fusion protein efficiently in vitro. Furthermore, high levels of the specific cytokines were induced in mice. By using this DNA vaccine as a booster after BCG, higher amounts of IFN-γ will be produced.
Keywords: BCG, DNA, Mycobacterium –tuberculosis, PCR, Plasmid
Introduction
Tuberculosis (TB) is a life threatening disease caused by Mycobacterium tuberculosis and has remained a major global public health problem (1). About 9 million new cases of infection with TB and 2 million deaths due to TB are reported each year (2). Despite the relatively high efficiency of BCG vaccine, its effectiveness in the adult pulmonary system and population of endemic regions remains controversial. This necessitates the development of a new vaccine against TB with higher efficiency than the currently available BCG (3).
Recently, different vaccines such as M. tuberculosis mutants, recombinant BCG expressing various immunogenic antigens, subunit vaccines based on the highly immunogenic M. tuberculosis antigens, and DNA vaccine have been introduced (4-6). DNA vaccine is a simple way to introduce different antigens to the immune system, which is capable of inducing both cellular and humoral immune responses (7). Previous studies have shown that Mtb32C and HBHA are highly immunogenic antigens. HBHA is a mycobacterial adhesin that binds to proteoglycan molecules on the surface of the lung epithelial cells (8, 9). HBHA participates in extrapulmonary dissemination of M. tuberculosis from lung to spleen. Previous studies have indicated that immunization with HBHA can induce a protection equal to that of BCG vaccination and can be used as a booster (10-12). Mtb32C is conserved in BCG and M. tuberculosis, and has specific epitopes (GAPINSATAM). Among MHC-1 restricted mycobacterial epitopes, Mtb32C can enhance CD8+ T-cell-dependent protective immunity and induce higher IFN-γ production (13, 14). In fact, IFN-γ has a key role in triggering cellular responses against intracellular pathogens such as M. tuberculosis. Consequently, it can be used as a specific antigen in IFN-γ production.
Prime-boost strategy is generally considered to be a promising way to enhance the efficacy of BCG (15). Using this strategy, the designated vaccine containing highly immunogenic mycobacterial antigens is administered to BCG pre-treated animals. This vaccination regimen induces higher levels of protection against TB compared with BCG alone. In the current study, a DNA vaccine based on mtb32C and hbha genes was constructed, and its ability to stimulate the immune system alone and as a booster was evaluated in an animal model.
Materials and Methods
Pathogen-free female BALB/c mice between 6 and 8 weeks of age were obtained from Razi Vaccine and Serum Research Institute (Mashhad, Iran). All mice were kept under SPF conditions and maintained in accordance with the Ethical Policies of Mashhad University of Medical Sciences.
Plasmid construction
Gene isolation, cloning, fusion, and plasmid construction were performed previously (16, 17). Briefly, Mtb32C and HBHA genes were isolated from M. tuberculosis H37Rv genome and were cloned into pCDNA3.1 (+). Restriction enzyme analysis and DNA sequencing confirmed the accuracy of the cloning procedure.
Transfection, RT-PCR, and Western blot
To confirm the in vitro expression of Mtb32C-HBHA fusion genes, the plasmid was transfected into HeLa cells (American Type Culture Collection Manassas, VA, USA) using lipofectamine transfection reagent according to the manufacturer’s instructions (Invitrogen, USA). Then, 72 hr after transfection, the cells were treated with 0.5 ml trypsin (Invitrogen, USA) and were incubated for 10 min. Cell suspension was harvested and total RNA was extracted using RNX-Plus (SinaClon, Iran), as described previously (16, 17). Purified RNA was used for cDNA synthesis using cDNA synthesis kit (Pars Tous, Iran) and was amplified by PCR. To detect the presence of His-Tag marker in chimeric Mtb32C-HBHA protein (a marker of constructed rather than natural protein), Western blot method was performed using mouse anti-His Tag antibody as the primary antibody and peroxidase conjugated rabbit anti-mouse IgG as the secondary antibody (AbD SeroTec, USA).
Vaccination
A total of 40 female BALB/c mice were randomly divided into these groups (10 mice in each group): control (pCDNA3.1+ vector), vaccine (pcDNA3.1+-Mtb32C-HBHA), BCG and BCG prime plus booster vaccine (pcDNA3.1+-Mtb32C-HBHA). The BCG and BCG prime plus booster vaccine groups were firstly immunized subcutaneously with BCG (5×105 CFU/PBS) (Pasteur institute, Iran) and then boosted three times intramuscularly with 100 μg designated vaccine at two-week intervals. The control group and vaccine groups were immunized three times intramuscularly at two-week intervals with 100 μg pCDNA3.1 (+) and recombinant vectors, respectively. Four weeks after the last immunization, all animals were sacrificed and their spleens were excised for further analysis (18).
Cytokine assay
Four weeks after the third vaccination, the spleen lymphocyte was extracted from all vaccinated and negative control mice. The 3×105 viable cells were cultured in 96-well microplates with RPMI 1640 medium (Invitrogen, USA) supplemented with 10% fetal calf serum (FCS), penicillin, and streptomycin in a total volume of 300 μl.
Splenocytes extracted from BCG vaccinated mice were cultured with different concentrations (0.5-5 mg/ml) of heat-killed M. tuberculosis H37Rv (MTB antigen) and 104 105 CFU of live BCG bacterium (BCG antigen). The plates were incubated at 37 °C in a humidified incubator with 5% CO2 for 72 hr. After the stimulation, the supernatants were harvested and IFN-γ release assay was performed using ELISA kits (eBioscience, SanDiego, CA) according to the manufacturer’s instructions. The maximum production of IFN-γ was seen in 0.5 mg/ml of MTB antigen and 2×104 CFU of BCG antigen in PBS. Based on these results, all splenocytes extracted from all groups were stimulated with these antigen concentrations (19, 20).
The freshly isolated splenocytes were incubated with MTB and BCG antigens for three days. At the end of day three, cell supernatants (after centrifugation) were used for subsequent analysis.
The concentrations of IFN-γ, interleukin-4 (IL-4), IL-12, TGF-β, and IL-10 in the culture supernatants were evaluated by an ELISA kit (eBioscience, SanDiego, CA) following the manufacturer’s recommendations. Assay sensitivity levels for IFN-γ, IL-12p70, IL-4, and IL-10 were 15 pg/ml, 15 pg/ml, 4 pg/ml, 30 pg/ml, and 8 pg/ml, respectively. The data were expressed as mean± standard deviation for each group of mice (eBioscience, SanDiego, CA).
Statistical analysis
The normality of the acquired data was assessed by Kolmogorov-Smirnov test. All data were analyzed by SPSS19.0 software using two-way ANOVA, and P<0.05 was considered statistically significant.
Results
RT-PCR and Western blot to assess the expression of DNA construct
Expression of the constructed DNA vaccine was first confirmed by RT-PCR to detect mRNA production, and was followed by Western blot to detect the recombinant Mtb32C-HBHA protein. The constructed Mtb32C-HBHA fusion protein was successfully expressed in the eukaryotic system. The amplified fragment using Mtb32C and HBHA specific primers demonstrated that recombinant vector could be transcribed in HeLa cell line. To assess the ability of vector in producing recombinant protein in cell culture system, Western blot technique and staining with specific antibodies and ECL detection reagent were used (abCam, UK) (Figures 1 and 2).
Figure 1.
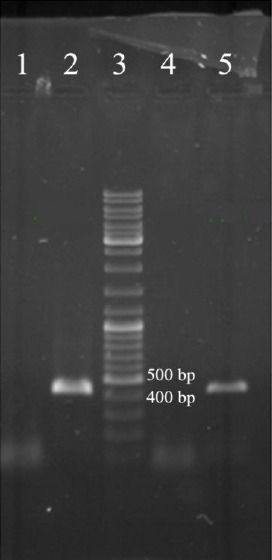
RT-PCR results on cDNA from transfected HeLa cells (lane numbers 2 and 5) and non-transfected cells (lane numbers 1 and 4); lane 3: 1 kb DNA size marker (Fermentas, Germany)
Figure 2.
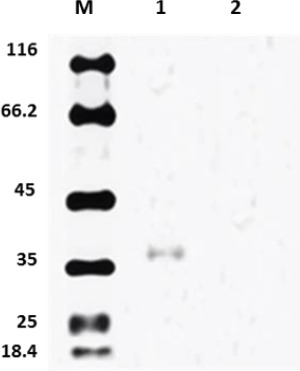
Western blot analysis of cell lysate from HeLa cells transfected with pCDNA3.1-Mtb32C-HBHA construct (lane 1) and untransfected cells (lane 2); lane M: protein size marker (Fermentas Company, Germany)
Cytokine assay
Four weeks after the last immunization, the mice were sacrificed, and specific cytokine production (IFNγ, IL-10, TGF-β, IL-12 and IL-4) was evaluated by ELISA in the supernatant of the splenocytes cultures after in vitro stimulation with MTB (0.5 mg/ml) and BCG antigens (2×104 CFU) for 72 hr. All mice vaccinated with Mtb32C-HBHA as a DNA vaccine, BCG and BCG-prime boosted with Mtb32C-HBHA (in the absence of adjuvant) produced high levels of IFNγ, IL-10, TGF-β, IL-12, and IL-4 compared to the control group of mice vaccinated only with pCDNA3.1 vector (P<0.05).
Significant differences (at significance level of P<0.001) were observed in all vaccinated groups (BCG, DNA vaccine, and BCG-vaccine) in the four examined cytokines in comparison with those in the control group.
As shown in Figure 3 and Table 1, significant differences were found between BCG-vaccine and vaccine and control groups at the significance level P<0.001, while the difference between BCG and control group was at P<0.05. The BCG-vaccinated mice boosted with Mtb32C-HBHA had a higher level of IFN-γ than other groups. Splenocytes from BCG-primed mice boosted with Mtb32C-HBHA produced substantially higher IFN-γ (265.73±119 pg/ml) than BCG-vaccinated mice (85.24±54 pg/ml), Mtb32C-HBHA vaccinated mice (144.78±37 pg/ml), and control mice (25.57±13 pg/ml). Significant differences between BCG-vaccine group and BCG and vaccine groups were at P<0.01, while the difference between BCG and vaccine group was at P<0.05. Also, IFN-γ cytokine was increased in all groups except the control group. The mice vaccinated with BCG plus Mtb32C-HBHA induced the highest levels of IFN-γ responses in the spleen. The combination of Mtb32C-HBHA and BCG significantly increased the expression of IFN-γ compared with BCG or Mtb32C-HBHA alone.
Figure 3.

The immune response to Mtb32C-HBHA after the last vaccination. The mice were divided into four groups and were immunized with BCG alone (BCG group), BCG/Mtb32C-HBHA (vaccine +BCG group), Mtb32C-HBHA alone (vaccine group), and empty vector (control group)
The immune responses for IFN-γ (B), IL-10(B), IL-12(A), IL-4 (A), and TGF-β (A) after stimulation were quantified by ELISA. The results are expressed as mean±standard deviation for the mice per group, tested in triplicates. The two-way ANOVA test was used to analyze the results, and significant difference was set as P<0.05
Table 1.
Spleen cytokine profile in different groups of BALB/c mice
| Cytokine Vaccination groups | IL-12p70 | IL-4 | IL-10 | IFN-γ | TGF-β |
|---|---|---|---|---|---|
| Control | 6.54±2.18 | 3.64±.93 | 289.88±76.54 | 25.57±13 | 3.06±1.18 |
| Vaccine | 31.98±7.45 | 9.65±2.8 | 783.84 ±394 | 144.78±37 | 9.93±4.18 |
| BCG | 24.78±6.79 | 8.53±4.2 | 825.64±.454 | 85.24±54 | 8.53±1.88 |
| BCG-vaccine | 28.84 ±7.5 | 8.58±2.8 | 1142.43±.577 | 265.73±119 | 8.22±1.41 |
Four weeks after the third Mtb32C-HBHA injection, ten mice from each group were sacrificed and their spleen cells were cultured in the presence of MTB and BCG antigens. Levels of cytokines were measured in a 72 hr culture supernatant. Values are expressed in pg/ml and represented as mean± standard deviation. The mice were primed with BCG and then boosted with 100 μg Mtb32C- HBHA (BCG–HBHA) subcutaneously
The amount of IL-12 in the group receiving Mtb32C-HBHA vaccine (31.98±7.45) was higher than that of BCG (24.78±6.79), BCG-vaccine (28.84±7.54), and control (6.54±2.18) groups. Significant differences were observed only between BCG and vaccine group at P<0.01.
In the case of IL-4 and TGF-β, no statistically signi-ficant differences were seen between BCG, BCG-vaccine and vaccine groups (P>0.05), but there were significant differences between control group and other groups. IL-4 production levels of BCG (8.53±4.2), BCG-vaccine (8.58±2.8) and vaccine (9.65±2.8) groups were higher than that of the control (3.64±.93) group.
As shown in Figure 3 and Table 1, splenocytes from the mice receiving BCG (8.53±1.88), BCG-vaccine (8.22±1.41) and vaccine (9.93±4.18) showed higher TGF-β production than control (3.06±1.18) group and the group receiving just pCDNA3.1(+) vector.
The amount of IL-10 was significantly higher in BCG-vaccine group (1142.43±.577) than in BCG (825.64± .454), vaccine (783.84±394), and control (289.88± 67.54) groups. Furthermore, there were no significant differences between BCG and vaccine groups (P>0.05), but significant differences were observed between BCG-vaccine group and other groups, including BCG and vaccine groups at P<0.01.
Discussion
The goals of WHO regarding TB infection are 50% reduction in TB mortality by 2015 and reduction of the incidence of new cases to less than one in a million people by 2050. These objectives are expected to be achieved to provide at least partially new drugs and more effective vaccines (21, 22). Producing more effective vaccines than BCG is a critical step in global TB control. Nevertheless, replacing BCG vaccine with other vaccines is not rational because BCG is more effective against severe forms of childhood TB. DNA vaccine has been considered an alternative candidate for inducing an immune response against TB. In animal models, DNA vaccine has provided satisfactory results, whereas DNA vaccines in human have comparatively lower immunogenicity. This defect can be resolved in the prime-boost strategy (4, 23). In this study, the constructed DNA vaccine was used alone and in combination with BCG in prime-boost strategy. Rouanet et al. showed that HBHA antibody titers were low during BCG vaccination. This indicates that despite the presence of hbha gene in BCG strain, it cannot be expressed. Therefore, the efficacy of BCG vaccine can be elevated by adding HBHA protein (24).
Administration of pCDNA-Mtb32C-HBHA vaccine showed that it was safe and well tolerated. Other studies have provided evidence that HBHA can be used as a booster vaccine, and methylation in the C-terminal domain of mycobacterial HBHA plays an important role in T-cell stimulation (11, 25). This study showed that Mtb32C–HBHA without post-translational modifications is able to induce high levels of IFN-γ as a cellular immune response marker (13). Indeed, Mtb32C could compensate this deficiency and could enhance HBHA immunogenicity (14). Previous studies have shown that HBHA in the presence of a specific Th1 adjuvant is able to induce strong cellular immune responses against M. tuberculosis, identical to that of BCG (32-34). In the present study, the ability of pCDNA3.1-Mtb32C-HBHA vector to stimulate immune responses was evaluated in the absence of adjuvant. Mtb32C-HBHA fusion protein without C-terminal methylation and in the absence of an adjuvant could stimulate strong cellular immune responses in BALB/c mice. Mtb32C-HBHA induced a robust Thl response that is characterized by elevated levels of IL-12, IFN-γ, and TGF-β in contrast to low levels of IL-4. The findings of this research indicated that pCDNA-Mtb32C-HBHA construct could be used in heterologous prime boost regimen because in this strategy other cytokines which are necessary for development, magnitude and maintenance of T cells such as IL-12 and TGF-β were produced. This result is also consistent with the results of the study performed by Guerrero et al. in which high levels of IL-10 were observed (32, 33). Another study concluded that the absence of methylation pattern in HBHA affects T-reg cells function, but others suggested that after induction, Th1 cells are the main source of IL-10 production. Cellular immune response has a critical role against intracellular pathogens, including M. tuberculosis. Protection against TB is dependent on cellular immunity, so IFN-γ and IL-12 as intracellular pathogens have a pivotal role in protection against M. tuberculosis (26-28). Consistent with the results of previous studies, boosting Mtb32C-HBHA vaccination following BCG priming exhibited significant increase in IFN-γ and IL-12 levels compared to other groups. Previous studies have reported that elevation of TGF-β in the presence of IL-10 and IL-12 stimulates T-reg and memory T cells, respectively. Also in our study, all these cytokines were elevated compared to control group (29). Spleen lymphocytes from all groups, which were stimulated by both antigens, produced significant amounts of IL12p70. High production of IL12p70 was observed in the group receiving only the constructed vaccine and in the group immunized by BCG- Mtb32C-HBHA vaccine in the prime-boost strategy. The highest rate of IL-12 was observed in the vaccine group, which indicated the designated vaccine can efficiently be expressed in the host cells and has the ability to promote antigen-presenting cells (APCs). Elevation of IL-12 along with TGF-β can trigger responses characterizing the effectors-memory T cells (30, 31). This study showed that administration of BCG in combination with DNA vaccine induced stronger Th1 immune response because IFN-γ was produced at greater levels than IL-4 by spleen lymphocyte upon in vitro stimulation. The Th1-type immune response has a key role in host response to M. tuberculosis infection. The obtained results strongly support this idea that vaccination with pCDNA3.1-Mtb32C-HBHA plasmid and use of prime-boost regimen enhance the immune response, which is turned toward Th1 type. In Prime-boost strategy, employing DNA vaccines has been shown to elicit stronger and more diverse cellular immune responses than BCG vaccine alone.
Conclusion
It was shown that IFN-γ production in the mice primed with BCG and then boosted with DNA vaccine was more remarkable than those vaccinated only with BCG or vaccine. Indeed, this construct can induce not only high levels of IFN-γ but also other cytokines such as IL-12 and TGF-β. The profile of cytokine production by spleen cells showed that the vaccine is so strong that can stimulate the immune system. These findings indicate that prime-boost vaccination scheme is an efficient vaccination model against TB.
Acknowledgment
The results described in this paper were part of a PhD student thesis (Thesis No.454-A). This study was financially supported by Mashhad University of Medical Sciences, Mashhad, Iran (Grant No. 910016).
References
- 1.Brewer TF. Preventing tuberculosis with bacillus Calmette-Guerin vaccine: a meta-analysis of the literature. Clin Infect Dis. 2000;3:S64–67. doi: 10.1086/314072. [DOI] [PubMed] [Google Scholar]
- 2.Dara M, Dadu A, Kremer K, Zaleskis R, Kluge HH. Epidemiology of tuberculosis in WHO European Region and public health response. Eur Spine J. 2013;22:549–555. doi: 10.1007/s00586-012-2339-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Colditz GA, Brewer TF, Berkey CS, Wilson ME, Burdick E, Fineberg HV, et al. Efficacy of BCG vaccine in the prevention of tuberculosis. Meta-analysis of the published literature. JAMA. 1994;271:698–702. [PubMed] [Google Scholar]
- 4.Delogu G, Fadda G. The quest for a new vaccine against tuberculosis. J Infect Dev Ctries. 2009;3:5–15. doi: 10.3855/jidc.99. [DOI] [PubMed] [Google Scholar]
- 5.Dietrich G, Viret JF, Hess J. Mycobacterium bovis BCG-based vaccines against tuberculosis: novel developments. Vaccine. 2003;21:667–670. doi: 10.1016/s0264-410x(02)00577-7. [DOI] [PubMed] [Google Scholar]
- 6.Parida SK, Kaufmann SH. Novel tuberculosis vaccines on the horizon. Curr Opin Immunol. 2010;22:374–384. doi: 10.1016/j.coi.2010.04.006. [DOI] [PubMed] [Google Scholar]
- 7.Ghanem A, Healey R, Adly FG. Current trends in separation of plasmid DNA vaccines: a review. Anal Chim Acta. 2013;760:1–15. doi: 10.1016/j.aca.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 8.Pethe K, Aumercier M, Fort E, Gatot C, Locht C, Menozzi FD. Characterization of the heparin-binding site of the mycobacterial heparin-binding hemagglutinin adhesin. J Biol Chem. 2000;275:14273–14280. doi: 10.1074/jbc.275.19.14273. [DOI] [PubMed] [Google Scholar]
- 9.Vidal Pessolani MC, Marques MA, Reddy VM, Locht C, Menozzi FD. Systemic dissemination in tuberculosis and leprosy: do mycobacterial adhesins play a role?Microbes Infect. 2003;5:677–684. doi: 10.1016/s1286-4579(03)00098-4. [DOI] [PubMed] [Google Scholar]
- 10.Krishnan N, Robertson BD, Thwaites G. The mechanisms and consequences of the extra-pulmonary dissemination of Mycobacterium tuberculosis. Tuberculosis (Edinb) 2010;90:361–366. doi: 10.1016/j.tube.2010.08.005. [DOI] [PubMed] [Google Scholar]
- 11.Guerrero GG, Debrie AS, Locht C. Boosting with mycobacterial heparin-binding haemagglutinin enhances protection of Mycobacterium bovis BCG-vaccinated newborn mice against M. tuberculosis. Vaccine. 2010;28:4340–4347. doi: 10.1016/j.vaccine.2010.04.062. [DOI] [PubMed] [Google Scholar]
- 12.Locht C, Hougardy JM, Rouanet C, Place S, Mascart F. Heparin-binding hemagglutinin, from an extrapulmonary dissemination factor to a powerful diagnostic and protective antigen against tuberculosis. Tuberculosis (Edinb) 2006;86:303–309. doi: 10.1016/j.tube.2006.01.016. [DOI] [PubMed] [Google Scholar]
- 13.Irwin SM, Izzo AA, Dow SW, Skeiky YA, Reed SG, Alderson MR, et al. Tracking antigen-specific CD8 T lymphocytes in the lungs of mice vaccinated with the Mtb72F polyprotein. Infect Immun. 2005;73:5809–5816. doi: 10.1128/IAI.73.9.5809-5816.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Skeiky YA, Alderson MR, Ovendale PJ, Guderian JA, Brandt L, Dillon DC, et al. Differential immune responses and protective efficacy induced by components of a tuberculosis polyprotein vaccine, Mtb72F, delivered as naked DNA or recombinant protein. J Immunol. 2004;172:7618–7628. doi: 10.4049/jimmunol.172.12.7618. [DOI] [PubMed] [Google Scholar]
- 15.Romano M, D'Souza S, Adnet PY, Laali R, Jurion F, Palfliet K, et al. Priming but not boosting with plasmid DNA encoding mycolyl-transferase Ag85A from Mycobacterium tuberculosis increases the survival time of Mycobacterium bovis BCG vaccinated mice against low dose intravenous challenge with M. tuberculosis H37Rv. Vaccine. 2006;24:3353–3364. doi: 10.1016/j.vaccine.2005.12.066. [DOI] [PubMed] [Google Scholar]
- 16.Teimourpour R, Sadeghian A, Meshkat Z, Esmaelizad M, Sankian M, Jabbari AR. Construction of a DNA vaccine encoding Mtb32C and HBHA genes of mycobacterium tuberculosis. Jundishapur J Microbiol. 2015;8:e21556. doi: 10.5812/jjm.21556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Teimourpour R, Zare H, Rajabnia R, Yahyapoor Y, Meshkat Z. Evaluation of the eukaryotic expression of mtb32C-hbha fusion gene of Mycobacterium tuberculosis in Hepatocarcinoma cell line. Iran J Microbiol. 2016;8:132–138. [PMC free article] [PubMed] [Google Scholar]
- 18.Meshkat Z, Teimourpour A, Rashidian S, Arzanlou M, Teimourpour R. Immunogenicity of a DNA Vaccine Encoding Ag85a-Tb10. 4 antigens from mycobacterium tuberculosis. Iran J Immunol. 2016;13:289. doi: 10.1002/jsfa.4218. [DOI] [PubMed] [Google Scholar]
- 19.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- 20.Meshkat Z, Mirshahabi H, Meshkat M, Kheirandish M, Hassan ZM. Strong immune responses induced by a DNA vaccine containing HPV16 truncated E7 C-terminal linked to HSP70 gene. Iran J Immunol. 2011;8:65. [PubMed] [Google Scholar]
- 21.Ottenhoff TH, Kaufmann SH. Vaccines against tuberculosis: where are we and where do We need to go?PLoS Pathogens. 2012;8:e1002607. doi: 10.1371/journal.ppat.1002607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Martin C. Tuberculosis vaccines: past, present and future. Curr Opin Pulm Med. 2006;12:186–191. doi: 10.1097/01.mcp.0000219267.27439.1b. [DOI] [PubMed] [Google Scholar]
- 23.Feng CG PU, Demangel C, Spratt JM, Malin AS, Britton WJ. Priming by DNA immunization augments protective efficacy of Mycobacteriumbovis Bacille Calmette-Guerin against tuberculosis. Infect Immun. 2001;69:4174–4176. doi: 10.1128/IAI.69.6.4174-4176.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rouanet C, Lecher S, Locht C. Subcutaneous boosting with heparin binding haemagglutinin increases BCG-induced protection against tuberculosis. Microbes Infect. 2009;11:995–1001. doi: 10.1016/j.micinf.2009.07.005. [DOI] [PubMed] [Google Scholar]
- 25.Guerrero GG, Locht C. Recombinant HBHA boosting effect on BCG-induced immunity against Mycobacterium tuberculosis infection. Clin Dev Immunol. 2011;2011:730702. doi: 10.1155/2011/730702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Barker LF, Brennan MJ, Rosenstein PK, Sadoff JC. Tuberculosis vaccine research: the impact of immunology. Curr Opin Immunol. 2009;21:331–338. doi: 10.1016/j.coi.2009.05.017. [DOI] [PubMed] [Google Scholar]
- 27.Cavalcanti YV, Brelaz MC, Neves JK, Ferraz JC, Pereira VR. Role of TNF-Alpha, IFN-Gamma, and IL-10 in the Development of Pulmonary Tuberculosis. Pulm Med. 2012;2012:10. doi: 10.1155/2012/745483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Redford P, Murray P, O'Garra A. The role of IL-10 in immune regulation during M. tuberculosis infection. Mucosal Immunol. 2011;4:261–270. doi: 10.1038/mi.2011.7. [DOI] [PubMed] [Google Scholar]
- 29.Jung YJ, Ryan L, LaCourse R, North RJ. Increased interleukin-10 expression is not responsible for failure of T helper 1 immunity to resolve airborne Mycobacterium tuberculosis infection in mice. Immunology. 2003;109:295–299. doi: 10.1046/j.1365-2567.2003.01645.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wyndham-Thomas C, Corbiere V, Dirix V, Smits K, Domont F, Libin M, et al. Key role of effector memory CD4+T lymphocytes in a short-incubation heparin-binding hemagglutinin gamma interferon release assay for the detection of latent tuberculosis. Clin Vaccine Immunol. 2014;21:321–328. doi: 10.1128/CVI.00651-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shafiani S DC, Ertelt JM, Moguche AO, Siddiqui I, Smigiel KS, et al. Pathogen-specific Treg cells expand early during mycobacterium tuberculosis infection but are later eliminated in response to Interleukin-12. J Exp Med. 2013;38:1261–1270. doi: 10.1016/j.immuni.2013.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Guerrero GG, Locht C. Recombinant HBHA boosting effect on BCG-induced immunity against Mycobacterium tuberculosis infection. Clin Dev Immunol. 2011;2011:730702. doi: 10.1155/2011/730702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Guerrero GG, Feunou FP, Locht C. The coiled-coil N-terminal domain of the heparin-binding haemagglutinin is required for the humoral and cellular immune responses in mice. Mol Immunol. 2008;46:116–24. doi: 10.1016/j.molimm.2008.07.009. [DOI] [PubMed] [Google Scholar]
- 34.Lebrun P, Raze D, Fritzinger B, Wieruszeski J-M, Biet F, Dose A, et al. Differential contribution of the repeats to heparin binding of HBHA, a major adhesin of Mycobacterium tuberculosis. PLoS One. 2012;7:e32421. doi: 10.1371/journal.pone.0032421. [DOI] [PMC free article] [PubMed] [Google Scholar]


