Abstract
Background
Inflammatory cytokines have been demonstrated to be involved in developing insulin resistance and type-2 diabetes (T2D). Natural antibodies in the circulation have protective effects on common diseases in humans. The present study was thus designed to test the hypothesis that natural antibodies against inflammatory cytokines could be associated with T2D.
Methods
An enzyme-linked immunosorbent assay (ELISA) was developed in-house to detect plasma IgG against peptide antigens derived from interleukin 1α (IL1α), IL1β, IL6, IL8 and tumor necrosis factor-α (TNF-α) in 200 patients with T2D and 220 control subjects.
Results
Binary regression showed that compared with control subjects, T2D patients had a decreased level of plasma anti-IL6 IgG (adjusted r 2=0.034, p=0.0001), anti-IL8 IgG (adjusted r 2=0.021, p=0.002) and anti-TNF-α IgG (adjusted r 2=0.017, p=0.003). Female patients mainly contributed to decreased levels of anti-IL6 IgG (adjusted r 2=0.065, p=0.0008) and anti-IL8 IgG (adjusted r 2=0.056, p=0.003), while male patients mainly contributed to decreased anti-TNF-α IgG levels (adjusted r 2=0.024, p=0.005). ROC curve analysis revealed a sensitivity of 16.5% against specificity of 95.5% for anti-IL6 IgG assay and a sensitivity of 19.5% against specificity of 95.9% for anti-IL8 IgG assay. Glycated hemoglobin levels measured after 6-month glucose-lowering treatment appeared to be inversely correlated with plasma anti-IL1α IgG (r=-0.477, df=17, p=0.039) and anti-IL6 IgG (r=-0.519, df=17, p=0.023) although such correlation failed to survive the Bonferroni correction.
Conclusions
Deficiency of natural IgG against inflammatory cytokines is likely to be a risk factor for T2D development and detection of such antibodies may be useful for personalized treatment of the disease.
Electronic supplementary material
The online version of this article (10.1186/s12950-017-0171-6) contains supplementary material, which is available to authorized users.
Keywords: Natural antibodies, IgG antibody, Inflammatory cytokines, Type-2 diabetes, ELISA
Background
Type-2 diabetes (T2D) is a metabolic disorder that results from interaction between genetic predisposition and environmental components [1]. The prevalence of T2D is rising globally, accounting for 85%–95% of all forms of diabetes in developed countries and a higher percentage in developing countries [2]. China has become a global epicenter of diabetes; a national study from June 2007 through May 2008 demonstrated that the prevalence of diabetes was 9.7% among Chinese adults [3]. Obesity is a major risk factor for T2D and obesity-induced chronic inflammation is likely to play a key role in the pathogenesis of insulin resistance that leads to the development of T2D [4, 5]. Multiple inflammatory inputs contribute to metabolic dysfunction [6]. The World Health Organization (WHO) published an epidemiological report in 2014, indicating that more than 1.9 billion adult people (18 years and older) in the world were overweight, 600 millions of whom were obese (http://www.who.int/mediacentre/factsheets/fs311/en/index.html). It is worth noting that approximately one third of the population is overweight or obese, but only ~5% develops T2D. There must be something in our body to regulate susceptibility to T2D in obese people.
Inflammatory cytokines, such as interleukin 1 (IL1), IL6 and tumor necrosis factor alpha (TNF-α) have been confirmed to be involved in developing insulin resistance [7]. Circulating IL8 was also found to be elevated in patients with T2D and associated with obesity-related parameters [8, 9]. Anti-inflammatory system could reduce susceptibility to insulin resistance and T2D. It has been indicated that the blockage of inflammatory cytokines has T2D-protective characteristics [4, 10]. Monoclonal antibodies targeting inflammatory cytokines have been used to treat coronary artery disease [11], rheumatoid arthritis and other systemic autoimmune diseases [12, 13]. Natural antibodies are defined as immunoglobulins that are spontaneously and constitutively secreted by the B1 type of lymphocytes in the absence of external antigen stimulation or immunization [14, 15]. Natural antibodies play an important role not only in eliminating pathogens invaded but also in maintaining homeostasis of the immune system by prevention of autoimmune and inflammatory reactions [16–19]. Possibly, the content of natural antibodies is in inverse proportion to inflammatory cytokines, and a decrease in natural antibody levels may lead to increased activities of inflammatory cytokines [20–22]. From an immunological point of view, the effects of inflammatory cytokines on our body may depend on circulating levels of their corresponding natural antibodies [15]. Accordingly, the present work was undertaken to examine if circulating levels of natural antibodies against inflammatory cytokines were associated with T2D and the therapeutic effects of glucose-lowering treatment on the disease in a Chinese population.
Methods
Subjects
A total of 200 patients aged 48.2±7.2 years, who were diagnosed as having T2D, and 220 healthy controls aged 44.2±8.6 years, were recruited by Dalang Hospital of Dongguan, China for this study. These 200 patients had a disease duration of 1.9±2.4 years on average, ranging from 0 to 9.4 years and they all fulfilled the diagnostic criteria for diabetes published by the WHO, 2006. (http://www.who.int/diabetes/publications/diagnosis_diabetes2006/en/). Those patients who developed clinical complications or suffered from autoimmune disease, malignant disease and mental illness, were excluded from this study. Glycated hemoglobin (HbA1c) levels in blood were taken from clinical records if available, which were measured before glucose-lowering treatment and in 3 and 6 months of post-treatment. Control subjects did not have history of diabetes, autoimmune disease, malignant illness and mental disorders. There were 54 smokers (27.0%) in the patient group and 60 smokers (27.3%) in the control group. All the subjects were of Chinese Han origin, and all gave written informed consent to participate in this study. This work was approved by an Institutional Review Board and conformed to the requirements of the Declaration of Helsinki.
Antibody testing
Five linear peptide antigens that are respectively derived from IL1α, IL1ß, IL6, IL8 and TNF-α, were designed according to the computational prediction of human leukocyte antigen (HLA) class II epitopes [23, 24], and their amino acid sequences are given in Table 1. An enzyme-linked immunosorbent assay (ELISA) was developed in-house based on a recent publication [25]. Briefly, solid-phase synthetic peptides were dissolved in 67 % acetic acid to obtain a concentration of 5mg/ml as stock solution. Maleimide-activated plates (Thermo Scientific, Shanghai, China) were coated according to the Manufacturer’s instruction. The antigen-coated plates were washed twice with 200μl Wash Buffer that was phosphate-buffered saline (PBS) (P4417, Sigma-Aldrich, Shanghai, China) containing 0.05% Tween-20; 50μl plasma sample diluted 1:200 in Assay Buffer that was PBS containing 0.5% bovine serum albumin (BSA) was then added to each sample well; 50μl Assay Buffer was added to each negative control (NC) well and 50μl positive control (PC) sample was added to each PC well. Following incubation at room temperature for 1.5 hours, the plate was washed three times with 200μl Wash Buffer and 50μl peroxidase-conjugated goat anti-human IgG antibody (ab98567, Abcam, Guangzhou, China) diluted 1:50000 in Assay Buffer was added to each well. After incubation at room temperature for 1 hour, color development was initiated by adding 50μl Stabilized Chromogen (SB02, Life Technologies, Guangzhou, China) and terminated after 20 min by adding 25μl Stop Solution (SS04, Life Technologies). The measurement of optical density (OD) was completed on a microplate reader within 10 min at 450nm with a reference wavelength of 620nm. All the samples were tested in duplicate and the specific binding ratio (SBR) was used to represent the relative levels of plasma IgG antibodies. Calculation of SBR is as follows: SBR= (OD Sample – OD NC) / (OD PC – OD NC)
Table 1.
Sequence of peptide antigens used for in-house ELISA
| Antigen | Sequence |
|---|---|
| IL1α | LLFFWETHGTKNYFTSVAHPNLFIATKQDYWVCLAGGP |
| IL1β | LNCTLRDSQQKSLVMSGPYELKALHLQGQDMEQQVVF |
| IL6 | TCLVKIITGLL EFEVYLEYLQNRFESSEEQARAVQM |
| IL8 | ELRCQCIKTYSKPFHPKFIKELRVIESGPH |
| TNF-α | LIYSQVLFKGQGCPSTHVLLTHTISRIAVSYQTKVNLLS |
To minimize an intra-assay deviation, the ratio of the difference between duplicated OD values of each sample to their sum was used to assess the precision for the in-house ELISA antibody test. If the ratio was found to be >10%, the test of this sample was treated as being invalid and was not used for data analysis.
Data analysis
The coefficient of variation (CV) was used to represent an inter-assay deviation estimated using pooled plasma, namely quality control (QC) sample, which was randomly collected from >20 healthy subjects and tested on every 96-well plate. Binary regression analysis was applied to examine the differences in circulating IgG levels between T2D patients and control subjects, with adjustment for age in either male or female samples, and for gender and age in combined samples. Pearson correlation analysis was performed to examine the correlation between plasma IgG levels and the duration of T2D or HbA1c levels in blood. Receiver operating characteristic (ROC) curve analysis was applied to estimate the areas under the ROC curve (AUC) with calculation of ELISA sensitivity against a specificity of ≥95% as well as 95% confident interval (CI). Because five antigens were tested in this study, the p-value of 0.01 was considered to be statistically significant based on the Bonferroni correction.
Results
The CV estimated based on SBR from the QC sample was 16.2% for anti-IL1α IgG assay, 9.5% for anti-IL1β IgG assay, 8.6% for anti-IL6 IgG assay, 13.9% for anti-IL8 IgG assay and 11.7% for anti-TNF-α IgG assay (Table 2), suggesting that the in-house ELISA developed with linear peptide antigens was highly reproducible.
Table 2.
Inter-assay deviation between ELISA-testing plates
| Antigen | Number of plates | Mean ± SDa | CV (%) |
|---|---|---|---|
| IL1α | 20 | 1.70±0.28 | 16.2 |
| IL1β | 23 | 1.12+0.11 | 9.5 |
| IL6 | 20 | 1.38+0.12 | 8.6 |
| IL8 | 20 | 1.41+0.17 | 11.7 |
| TNF-α | 23 | 1.33+0.19 | 13.9 |
a SD standard deviation
Binary regression analysis showed that compared with control subjects, T2D patients had a decreased level of circulating IgG against peptide antigens derived from IL6 (adjusted r 2=0.034, p=0.0001), IL8 (adjusted r 2=0.021, p=0.002) and TNF-α (adjusted r 2=0.017, p=0.003). Female patients mainly contributed to decreased levels of anti-IL6 IgG (adjusted r 2=0.065, p=0.0008) and IL8 IgG (adjusted r 2=0.056, p=0.003), while male patients mainly contributed to decreased anti-TNF-α IgG levels (adjusted r 2=0.024, p=0.005). Circulating IgG against peptide antigens derived from IL1α and IL1β did not show a significant change in patients with T2D (Table 3).
Table 3.
Binary regression analysis of circulating IgG against inflammatory cytokines in T2D
| Antigens | Patient (n) | Control (n) | Adjusted r 2 | p a |
|---|---|---|---|---|
| IL1α | ||||
| Male | 1.15±0.42 (124) | 1.21±0.39 (131) | -0.002 | 0.228 |
| Female | 1.20±0.43 (76) | 1.32±0.39 (89) | 0.009 | 0.083 |
| Combined | 1.17±0.42 (200) | 1.26±0.39 (220) | 0.012 | 0.04 |
| IL1β | ||||
| Male | 0.70±0.28 (124) | 0.77±0.30 (131) | 0.019 | 0.04 |
| Female | 0.78±0.31 (76) | 0.80±0.33 (89) | -0.01 | 0.788 |
| Combined | 0.73±0.29 (200) | 0.78±0.31 (220) | 0.011 | 0.07 |
| IL6 | ||||
| Male | 1.13±0.21 (124) | 1.18±0.19 (131) | 0.01 | 0.038 |
| Female | 1.11±0.23 (76) | 1.24±0.22 (89) | 0.065 | 0.0008 |
| Combined | 1.12±0.22 (200) | 1.20±0.21 (220) | 0.034 | 0.0001 |
| IL8 | ||||
| Male | 0.97±0.35 (124) | 1.02±0.29 (131) | 0.003 | 0.134 |
| Female | 0.94±0.33 (76) | 1.12±0.35 (89) | 0.056 | 0.003 |
| Combined | 0.96±0.34 (200) | 1.06±0.32 (220) | 0.021 | 0.002 |
| TNF-α | ||||
| Male | 0.77±0.40 (124) | 0.95±0.60 (131) | 0.024 | 0.005 |
| Female | 0.85±0.48 (76) | 0.95±0.45 (89) | -0.002 | 0.226 |
| Combined | 0.80±0.43 (200) | 0.95±0.54 (220) | 0.017 | 0.003 |
Data were expressed as mean±SD. aAdjusted for age in male and female samples, and for gender and age in combined samples.
As shown in Table 4, none of plasma IgG antibodies for these five inflammatory cytokines was significantly correlated with HbA1c levels measured either before glucose-lowering treatments or after 3-month treatment. However, HbA1c levels measured after 6-month glucose-lowering treatment appeared to be inversely correlated with anti-I IL1α IgG (r=-0.477, df=17, p=0.039) and anti-IL6 IgG (r=-0.519, df=17, p=0.023), although such significant correlations failed to survive the Bonferroni correction (p>0.01).
Table 4.
Correlation between plasma IgG and HbA1c levels in T2D patients before and after glucose-lowering treatment
| Antigen | Before treatment | 3-month treatment | 6-month treatment | |||
|---|---|---|---|---|---|---|
| r | p a | r | p b | r | p c | |
| IL1α | -0.024 | 0.762 | -0.005 | 0.978 | -0.477 | 0.039 |
| IL1β | -0.011 | 0.888 | -0.030 | 0.875 | -0.278 | 0.250 |
| IL6 | -0.052 | 0.517 | -0.191 | 0.304 | -0.519 | 0.023 |
| IL8 | -0.089 | 0.266 | -0.239 | 0.196 | -0.400 | 0.090 |
| TNF-α | 0.069 | 0.384 | -0.129 | 0.489 | -0.137 | 0.576 |
adf=157; bdf=29; cdf=17
ROC curve analysis revealed that anti-IL6 IgG assay had an AUC of 0.601 (95% CI 0.55-0.66) with sensitivity of 16.5% against a specificity of 95.5%, and anti-IL8 IgG assay had an AUC of 0.593 (95% CI 0.54-0.65) with sensitivity of 19.5% against a specificity of 95.9%; all other three IgG tests had a sensitivity of less than 15% against the specificity of 95% (Fig. 1). Pearson correlation analysis failed to show a significant correlation between the duration of T2D and the levels of circulating IgG against inflammatory cytokines (Fig. 2).
Fig. 1.
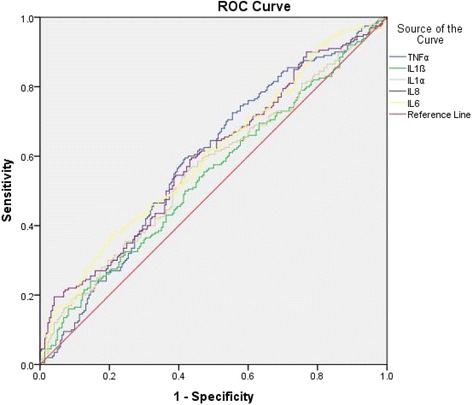
ROC curve analysis of circulating IgG against inflammatory cytokines in T2D. Anti-IL1α IgG had an AUC of 0.564 (95% CI 0.509-0.619) with sensitivity of 13.0% against a specificity of 95.0%; anti-IL1β IgG had an AUC of 0.544 (95% CI 0.489-0.599) with sensitivity of 10.0% against a specificity of 95.0%; anti-IL6 IgG had an AUC of 0.601 ( 95% CI 0.547-0.655) with sensitivity of 16.5% against a specificity of 95.5%; anti-IL8 IgG had an AUC of 0.593 (95%CI 0.538-0.647) with sensitivity of 19.5% against a specificity of 95.9%; anti-TNF-α IgG had an AUC of 0.589 (95%CI 0.534-0.643) with sensitivity of 4.5% against a specificity of 95.0%
Fig. 2.
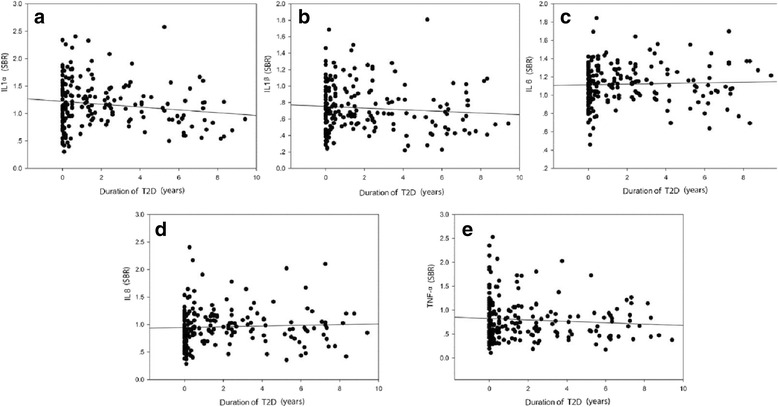
Correlation between the duration of T2D and the levels of circulating IgG against inflammatory cytokines a. IL1α IgG : r = -0.148, df =199, p= 0.037; b. IL1β IgG: r = -0.085, df = 199, p= 0.234; c. IL6 IgG: r = 0.04, df =199, p= 0.576; d. IL8 IgG: r = 0.047, df = 199, p= 0.508; e. TNF-α IgG: r = -0.083, df = 199, p= 0.242
Discussion
Natural antibodies are present in both animals and humans; they are thought to comprise the bulk of resting IgM, along with portions of isotype-switched IgA and IgG [26, 27]. Natural antibodies also tend to be autoreactive and perform a second beneficial function in housekeeping and homeostatic activity for elimination of dying cells and noxious molecular species [16, 28]. It is believed that natural antibodies are related to the most common, distressing and burdensome diseases, majority of which is associated with aging [15]. In this study, we found that deficiency of natural antibodies against IL6, IL8 and TNF-α was associated with T2D (Table 3) although their levels in the circulation may not be correlated with the duration of T2D (Fig. 2). ROC curve analysis revealed a sensitivity of >15% against the specificity of >95% for both the anti-IL6 assay and the anti-IL8 IgG assay. The sensitivity may represent a clinical subgroup that has undergone an inflammatory process in patients with T2D. Interestingly, HbA1c levels in blood are inversely correlated with plasma anti-IL1α and anti-IL6 IgG levels (Table 4). These observations raise the possibility that natural antibodies against inflammatory cytokines may be useful biomarkers for the development of personalized treatment of T2D on the one hand, and on the other hand, they can also serve as a key component in the body to bridge the gap between T2D and obesity, and counteract the onset of obesity-related diseases.
Gender differences in insulin resistance and T2D have been observed in several studies [29–32]. Women with T2D generally have poor glycemic control and are less likely to reach the goals for HbA1c, as compared with men [30]. Diversities in biology, culture, lifestyle, environment and socioeconomic status impact the differences in risk, pathophysiology and complications of T2D between male and female patients [32]. In this study, we found that there was a gender difference in circulating IgG antibodies against inflammatory cytokines (Table 3); decreased levels of anti-IL6 and anti-IL8 IgG antibodies were more likely to occur in female than male patients, whereas deficiency of anti-TNF-α IgG was more likely to be observed in male than female patients. The gender differences in natural IgG antibodies against inflammatory cytokines provide a clue to the insight into the pathological mechanism behind T2D development in humans. To our knowledge, this is the first report on a decrease in circulating IgG antibodies against inflammatory cytokines. However, this is a piece of preliminary work and the initial finding needs further replication in a large sample size and also in subpopulations.
Conclusions
Deficiency of natural IgG against inflammatory cytokines is likely to be a risk factor for T2D development and detection of such antibodies may be useful for personalized treatment of the disease.
Acknowledgements
We thank all the patients and control subjects for their participation in this study.
Funding
This work was supported by Hailanshen Biomedical Technology Ltd, Shenzhen, China.
Availability of data and materials
A spreadsheet with detailed data is available as Additional file 1.
Abbreviations
- AUC
Areas under the ROC curve
- BSA
Bovine serum albumin
- CI
Confident interval
- CV
Coefficients of Variation
- ELISA
Enzyme-linked immunosorbent assay
- HbA1c
Glycated hemoglobin
- HLA
Human leukocyte antigen
- IgA
Immunoglobulin A
- IgG
Immunoglobulin G
- IgM
Immunoglobulin M
- IL1α
Interleukin 1α
- IL1β
Interleukin 1β,
- IL6
Interleukin 16
- IL8
Interleukin 8
- NC
Negative control
- OD
Optical density
- PBS
Phosphate -buffered saline
- PC
Positive control
- QC
Quality control
- ROC
Receiver operating characteristic curve
- SBR
Specific binding ratio
- T2D
Type-2 diabetes
- TNF-α
Tumor necrosis factor-α
- WHO
World Health Organization
Additional file
Supplementary information. (XLSX 195 kb)
Authors’ contributions
CW and QC mainly carried out laboratory work and data analysi; ZH, CX and ZX identified patients with diabetes and collected clinical data; MQ and WJ conceived of this study, supervised laboratory work and drafted manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
This work was approved by the Institutional Review Board of the Second Hospital of Jilin University, Changchun, China, (IRB#: SHJU2017-101), and performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent for publication
Not applicable.
Competing interests
All authors declared that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Footnotes
Electronic supplementary material
The online version of this article (10.1186/s12950-017-0171-6) contains supplementary material, which is available to authorized users.
Contributor Information
Weiyi Cai, Email: loveweiyi@126.com.
Cailing Qiu, Email: youling85@126.com.
Hongyu Zhang, Email: yidadoudou@126.com.
Xiangyun Chen, Email: 373899251@qq.com.
Xuan Zhang, Email: zhangxuankj@163.com.
Qingyong Meng, Phone: +86-769-22896231, Email: qingyong_meng@outlook.com.
Jun Wei, Phone: +44 1463279563, Email: jun.wei@uhi.ac.uk.
References
- 1.Stumvoll M, Goldstein BJ, van Haeften TW. Type 2 diabetes: Principles of pathogenesis and therapy. Lancet. 2005;365:1333–1346. doi: 10.1016/S0140-6736(05)61032-X. [DOI] [PubMed] [Google Scholar]
- 2.Ramachandran A, Ma RC, Snehalatha C. Diabetes in Asia. Lancet. 2010;375:408–418. doi: 10.1016/S0140-6736(09)60937-5. [DOI] [PubMed] [Google Scholar]
- 3.Yang W, Lu J, Weng J, Jia W, Ji L, Shan Z, Ji L, Xiao J, Shan Z, Liu J, Tian H, Ji Q, Zhu D, Ge J, Lin L, Chen L, Guo X, Zhao Z, Li Q, Zhou Z, Shan G, He J. China National Diabetes and Metabolic Disorders Study Group: Prevalence of diabetes among men and women in China. N Engl J Med. 2010;362:1090–1101. doi: 10.1056/NEJMoa0908292. [DOI] [PubMed] [Google Scholar]
- 4.de Luca C, Olefsky JM. Inflammation and insulin resistance. FEBS Lett. 2008;582:97–105. doi: 10.1016/j.febslet.2007.11.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mushtaq G, Khan JA, Kumosani TA, Kamal MA. Alzheimer's disease and type 2 diabetes via chronic inflammatory mechanisms. Saudi J Biol Sci. 2015;22:4–13. doi: 10.1016/j.sjbs.2014.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lumeng CN, Saltiel AR. Inflammatory links between obesity and metabolic disease. J Clin Invest. 2011;121:2111–2117. doi: 10.1172/JCI57132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bastard JP, Maachi M, Lagathu C, Kim MJ, Caron M, Vidal H, Capeau J, Feve B. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw. 2006;17:4–12. [PubMed] [Google Scholar]
- 8.Abou-Shousha S, Abd El-Megeed MH, Sultan HK. Interleukin-8, ferritin and soluble transferrin receptors in type II diabetes mellitus. Egypt J Immunol. 2006;13:19–25. [PubMed] [Google Scholar]
- 9.Kim CS, Park HS, Kawada T, Kim JH, Lim D, Hubbard NE, Kwon BS, Erickson KL, Yu R. Circulating levels of MCP-1 and IL-8 are elevated in human obese subjects and associated with obesity-related parameters. Int J Obes (Lond) 2006;30:1347–1355. doi: 10.1038/sj.ijo.0803259. [DOI] [PubMed] [Google Scholar]
- 10.Herder C, Carstensen M. OuwensDM: Anti-inflammatory cytokines and risk of type 2 diabetes. Diabetes Obes Metab. 2013;15(suppl 3):39–50. doi: 10.1111/dom.12155. [DOI] [PubMed] [Google Scholar]
- 11.Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ, CANTOS Trial Group Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N Engl J Med. 2017;377:1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 12.Murdaca G, Spanò F, Puppo F. Long-term treatment of rheumatoid arthritis with adalimumab. Open Access Rheumatol. 2013;5:43–49. doi: 10.2147/OARRR.S32582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ogata A, Tanaka T. Tocilizumab for the treatment of rheumatoid arthritis and other systemic autoimmune diseases: current perspectives and future directions. Int J Rheumatol. 2012;2012:946048. doi: 10.1155/2012/946048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schwartz-Albiez R, Monteriro RC, Rodriguez M, Binder CJ, Shoenfeld Y. Natural antibodies, intravenous immunoglobulin and their role in autoimmunity, cancer and inflammation. Clin Exp Immunol. 2009;158(Suppl 1):43–50. doi: 10.1111/j.1365-2249.2009.04026.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rothstein TL. Natural Antibodies as Rheostats for Susceptibility to Chronic Diseases in the Aged. Front Immunol. 2016;7:127. doi: 10.3389/fimmu.2016.00127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shaw PX, Hörkkö S, Chang MK, Curtiss LK, Palinski W, Silverman GJ, Witztum JL. Natural antibodies with the T15 idiotype may act in atherosclerosis, apoptotic clearance, and protective immunity. J Clin Invest. 2000;105:1731–1740. doi: 10.1172/JCI8472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Silverman GJ. Protective natural autoantibodies to apoptotic cells: evidence of convergent selection of recurrent innate-like clones. Ann N Y Acad Sci. 2015;1362:164–175. doi: 10.1111/nyas.12788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vincent T, Plawecki M, Goulabchand R, Guilpain P, Eliaou JF. Mergining clinical phenotypes associated with anti-cytokine autoantibodies. Autoimmun Rev. 2015;14:528–535. doi: 10.1016/j.autrev.2015.01.015. [DOI] [PubMed] [Google Scholar]
- 19.Born WK, Huang Y, Zeng W, Torres RM, O'Brien RL. A Special Connection between γδ T Cells and Natural Antibodies? Arch Immunol Ther Exp (Warsz) 2016;64:455–462. doi: 10.1007/s00005-016-0403-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pasquier B, Launay P, Kanamaru Y, Moura IC, Pfirsch S, Ruffié C, Hénin D, Benhamou M, Pretolani M, Blank U, Monteiro RC. Identification of FcαRI as an inhibitory receptor that controls inflammation. Immunity. 2005;22:31–42. doi: 10.1016/j.immuni.2004.11.017. [DOI] [PubMed] [Google Scholar]
- 21.Tsimikas S, Brilakis ES, Lennon RJ, Miller ER, Witztum JL, McConnell JP, Kornman KS, Berger PB. Relationship of IgG and IgM autoantibodies to oxidized low density lipoprotein with coronary artery disease and cardiovascular events. J Lipid Res. 2007;48:425–433. doi: 10.1194/jlr.M600361-JLR200. [DOI] [PubMed] [Google Scholar]
- 22.Soto Y, Conde H, Aroche R, Brito V, Luaces P, Nasiff A, Obregón A, Vázquez López AM. Autoantibodies to oxidized low density lipoprotein in relation with coronary artery disease. Hum Antibodies. 2009;18:109–117. doi: 10.3233/HAB-2009-0202. [DOI] [PubMed] [Google Scholar]
- 23.Söllner J, Heinzel A, Summer G, Fechete R, Stipkovits L, Szathmary S, Mayer B. Concept and application of a computational vaccinology workflow. Immunome Res. 2010;6(suppl 2):S7. doi: 10.1186/1745-7580-6-S2-S7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang P, Sidney J, Kim Y, Sette A, Lund O, Nielsen M, Peters B. Peptide binding predictions for HLA DR, DP and DQ molecules. BMC Bioinformatics. 2010;11:568. doi: 10.1186/1471-2105-11-568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hallford P, Clair DS, Halley L, Mustard C, Wei J. A study of type-1 diabetes associated autoantibodies in schizophrenia. Schizophr Res. 2016;176:186–190. doi: 10.1016/j.schres.2016.07.020. [DOI] [PubMed] [Google Scholar]
- 26.Panda S, Ding JL. Natural antibodies bridge innate and adaptive immunity. J Immunol. 2015;194:13–20. doi: 10.4049/jimmunol.1400844. [DOI] [PubMed] [Google Scholar]
- 27.Montecino-Rodriguez E, Leathers H, Dorshkind K. Identification of a B-1 B cell-specified progenitor. Nat Immunol. 2006;7:293–301. doi: 10.1038/ni1301. [DOI] [PubMed] [Google Scholar]
- 28.Rothstein TL, Griffin DO, Holodick NE, Quach TD, Kaku H. Human B-1 cells take the stage. Ann N Y Acad Sci. 2013;1285:97–114. doi: 10.1111/nyas.12137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kautzky-Willer A, Handisurya A. Metabolic diseases and associated complications: sex and gender matter. Eur J Clin Invest. 2009;39:631–648. doi: 10.1111/j.1365-2362.2009.02161.x. [DOI] [PubMed] [Google Scholar]
- 30.Arnetz L, Ekberg NR, Alvarsson M. Sex differences in type 2 diabetes: focus on disease course and outcomes. Diabetes Metab Syndr Obes. 2014;7:409–420. doi: 10.2147/DMSO.S51301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Goedecke JH, George C, Veras K, Peer N, Lombard C, Victor H, Steyn K, Levitt NS. Sex differences in insulin sensitivity and insulin response with increasing agein black South African men and women. Diabetes Res Clin Pract. 2016;122:207–214. doi: 10.1016/j.diabres.2016.11.005. [DOI] [PubMed] [Google Scholar]
- 32.Kautzky-Willer A, Harreiter J, Pacini G. Sex and Gender Differences in Risk, Pathophysiology and Complications of Type 2 Diabetes Mellitus. Endocr Rev. 2016;37:278–316. doi: 10.1210/er.2015-1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
A spreadsheet with detailed data is available as Additional file 1.


