Abstract
Introduction
The impact of time to treatment on clinical outcome is an established precept in infectious disease but is not established in peritoneal dialysis–related peritonitis (PDRP).
Methods
In a prospective multicenter study of PDRP, symptom-to-contact time (SC), contact-to-treatment time (CT), defined as the time from health care presentation to initial antibiotic, and symptom-to-treatment time (ST) were determined.
Results
One hundred sixteen patients had 159 episodes of PDRP. Median SC for all episodes was 5.0 hours (first to third quartile [Q1–Q3]: 1.3–13.9); CT, 2.3 hours (Q1–Q3: 1.2–4.0); and ST, 9.0 hours (Q1–Q3: 4.7–25.3). Thirty-eight (23.9%) patient episodes (28 catheter removals and 10 deaths) met the primary composite outcome of PD failure at 30 days (PD-fail). The risk of PD-fail increased by 5.5% for each hour of delay of administration of antibiotics (odds ratio [OR] for CT: 1.055; 95% confidence interval [CI]: 1.005–1.109; P = 0.032). Neither SC (OR: 1.00; 95% CI: 0.99–1.01; P = 0.74) nor ST (OR: 1.00; 95% CI: 0.99–1.01; P = 0.48) was associated with PD-fail. In a multivariable analysis, only CT for presentation to a hospital-based facility compared with a community facility (OR: 1.068; 95% CI: 1.013–1.126; P = 0.015) and female sex (OR: 2.4; 95% CI: 1.1–5.4; P = 0.027) were independently associated with PD-fail. Each hour of delay in administering antibacterial therapy from the time of presentation to a hospital facility increased the risk of PD failure or death by 6.8%.
Discussion
Strategies targeted to expedited antibiotic treatment should be implemented to improve outcomes from PDRP.
Keywords: antibiotics, outcomes, peritoneal dialysis, peritonitis
In Australia peritoneal dialysis (PD) is the initial choice of renal replacement therapy for 19% of all dialysis patients; however, only 8% of patients remain on PD for more than 5 years, principally due to technique failure associated with peritonitis.1, 2 Observational studies have variably identified multiple risk factors for PD peritonitis, including residual renal function, female sex, indigenous or ethnic origin, lower socioeconomic status, older age, continuous ambulatory PD versus automated PD, obesity, malnutrition, coronary artery disease, chronic lung disease, and hypertension.3, 4 Prior episodes of peritonitis,5 exit-site infection,6, 7 nasal carriage of Staphylococcus aureus,8 and organism virulence9 have also been implicated, and recent peritonitis or antimicrobial therapy has been shown to increase the risk of fungal peritonitis.10 Despite defined strategies to mitigate PDRP risk,11 the treatment of PDRP is dependent on patient recognition of symptoms and presentation to a health facility for administration of antibiotics. Even with treatment, a significant proportion of episodes of peritonitis result in hospitalization, catheter loss, or patient death.12 Although it is implicit in international guidelines that treatment should be timely,13 no study has identified the relationship between the timing of administration of antibiotics and the subsequent chance of recovery, cure, hospitalization, or death. Guidelines and studies in other infectious and vascular diseases recommend rapid therapeutic interventions because the outcomes from intervention are time dependent.14, 15, 16, 17, 18, 19, 20, 21 We hypothesized that there was a relationship between the time of antibiotic administration after presentation with peritonitis and patient episode outcomes.
Materials and Methods
Study Population
All episodes of PDRP in adults treated between August 2012 and July 2014 in Western Australia were prospectively enrolled as part of a clinical audit approved by the Royal Perth Hospital ethics committee. In 2012 there were 1144 patients receiving dialysis, of whom 320 received PD. At the time of study commencement in 2012, the Western Australia peritonitis rate was 1:21 patient-months, and the Australian average was 1:28 patient-months.1 In Western Australia all patients receiving outpatient PD are trained and managed by a publicly funded private provider, Fresenius Medical Care®, and supervised by 3 tertiary hospitals located in Perth, the capital city. A single statewide PDRP protocol is used and requires the initial i.p. administration of 2 g vancomycin and 200 mg gentamicin for all suspected PDRP episodes pending culture confirmation, with subsequent treatment duration and adjustments according to International Society for Peritoneal Dialysis (ISPD) guidelines. PD exit-site care protocols include bacterial wash, dressing, and intranasal mupirocin administered 5 days per month. Following recognition of symptoms of possible PDRP, patients are instructed to contact the 24-hour Fresenius Medical Care® service that advises on management, which may involve treatment at 1 of 2 Fresenius Medical Care® ambulant home care facilities located in metropolitan Perth, a metropolitan hospital, or a non-metropolitan health facility according to time of contact (in working hours vs. out of working hours) and patient location. Patients may also present directly to hospital facilities when symptoms are vague or unrecognized as peritonitis or diagnostic uncertainty persists after phone contact.
Data Collection
Patient information regarding symptom presentation and onset at the time of initial presentation was recorded using a standardized data collection form. Data were confirmed by access to emergency department records, hospital admission notes, laboratory data, electronic databases, and direct communication with patients for confirmation of symptom onset and timing. Data were collected by nursing staff and then collated and checked by clinicians (KM, PH) for accuracy and to resolve any ambiguity. The PDRP episode data included the following time points: (i) symptom onset, defined as the time the patient identified an abnormality of 1 or more of pain, fever, or cloudy bags; (ii) initial contact with the medical services, defined as the time recorded by the institution receiving the patient; and (iii) time of treatment, defined as the time of commencement of initial antibiotic administration. Episodes were excluded from the final analysis if data were missing for 2 or more of these time variables.
Definitions
After treatment for presumed PDRP, peritonitis was retrospectively confirmed using the ISPD definition,13 as the presence of any 2 of the following: (i) abdominal pain or cloudy bag; (ii) white cell count ≥100 in the PD effluent with neutrophil count ≥50% or a neutrophil count >50% if the dwell time was <2 hours; (iii) a positive PD effluent culture. Metropolitan was defined as <100 km from Perth, non-metropolitan as >100 and up to 2400 km from Perth. An abnormal exit site was defined clinically at the time of presentation by the treating team. Treatment was defined as the administration of any antimicrobial therapy, which was then reconciled as consistent with the Western Australia PDRP protocol. The treating health facility was coded as the patient’s initial point of presentation and administration of antibiotic therapy, even if subsequently they re-presented or were admitted to other health facilities. The following three main time variables were derived: (i) symptom-to-contact time (SC), defined as the time from recognition of the first patient-described symptom to the initial time the health provider was contacted; (ii) contact-to-treatment time (CT), defined as the time between the first health provider contact to the initial introduction of antimicrobial therapy; and (iii) symptom-to-treatment time (ST), the sum of SC and CT.
Outcome Measures
The outcome of each PDRP episode was determined at 30 days as (i) resolution of peritonitis (PD-cure), (ii) Tenckhoff catheter removal, or (iii) death. The composite primary outcome of PD failure (PD-fail) was defined as either catheter removal or death at 30 days.
Statistical Analysis
Data are summarized using proportions, means, SDs, medians, and 25th (Q1) and 75th (Q3) quartiles as appropriate. Logistic regressions were performed to investigate associations between SC and CT and the outcome PD-fail. Linearity of the relationship between outcomes and SC or CT was initially investigated using fractional polynomials. Where evidence of non-linearity was detected, the time variable was divided into quintiles and the proportion in each quintile with the outcome plotted. This illustrated points at which the proportions changed and then remained altered, thus identifying a potential cut point in the continuous variable. If a cut point was observed this was favored over fractional polynomials for simplicity of interpretation. CT was therefore also investigated as a dichotomous variable with a cut point of 4 hours, as this period is associated with Australian Hospital Emergency Department decision and admission rules and corresponded to a potential cut point identified. Two episodes with particularly large and unlikely values of CT (>120 hours) were removed from the analysis when CT was investigated as a continuous variable but were included in the upper category of the dichotomous variable.
Associations between outcomes and patient and episode characteristics were also explored. To determine whether the relationship between delays and outcomes was independent of these factors, all variables significant or close to significance (P < 0.1) in univariate analysis were included in a multivariable model. The final model was obtained following a manual backward stepwise elimination process where models were examined for indications of influence attributable to the removed non-significant variable. Potential modification of SC, ST, or CT effects on PD-fail by gender or institution type was investigated using an interaction term.
Associations between the type of health facility to which initial presentation was made and patient characteristics were investigated using Wilcoxon rank sum and χ2 tests, while associations with episode characteristics were explored using multinomial logistic regression.
Due to potential correlations between multiple episodes for the same patient, robust variance estimates for all regression models were obtained by applying a per-person cluster adjustment. Analyses were performed using Stata 13 (StataCorp LP, College Station, TX), and significance was set at P < 0.05.
Results
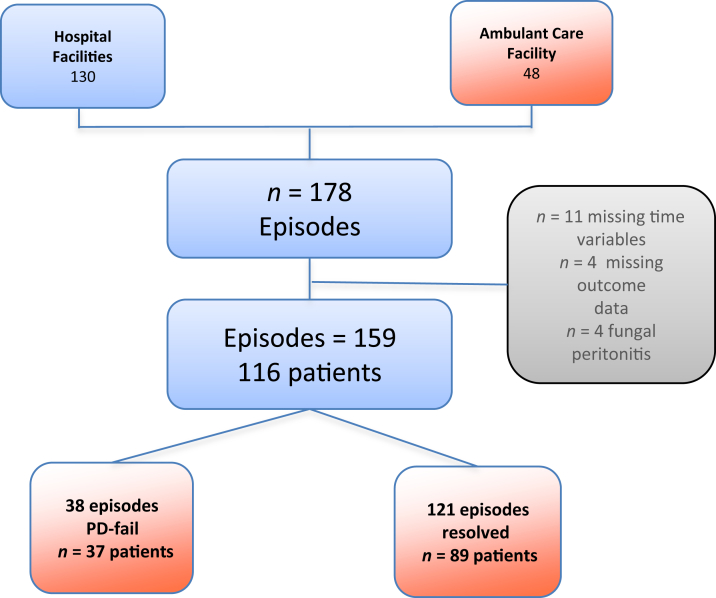
During the 24-month period there were 178 treated episodes of presumed PDRP in 122 patients (Figure 1). A total of 19 episodes and 6 patients were eliminated from analysis due to incomplete or missing time variables (n = 11) or outcome data (n = 4) or due to fungal peritonitis (n = 4), leaving 159 episodes in 116 patients, of whom 73% had single episodes and the remainder had 2 episodes (19%) or ≥3 episodes (8%). Six episodes were classified as repeat (same organism, n = 1) or recurrent (different organism, n = 5).13 Approximately one-third of patients entered the study receiving automated PD, and during the study only 3 patients changed between automated PD and continuous ambulatory PD. Baseline data of the 116 patients is shown in Table 1. A cloudy bag was the most commonly identified presenting symptom (91%), while 72% had abdominal pain and 63.5% had both. The ISPD guidelines for diagnosis of peritonitis were met by 89% of patient episodes treated for PDRP. Causative organisms by the primary outcome are shown in Table 2. Gram-positive cocci (50%) and culture negative (30%) were the most common causative groups. There was no statistical association between the distribution of bacterial organisms and PD-fail (P = 0.72).
Figure 1.
Flow diagram of episodes, presentation, follow-up, and outcomes. PD, peritoneal dialysis.
Table 1.
Demographics and baseline characteristics of the whole study population
| Variable | All patients (n = 116) n (%) |
|---|---|
| Age (yr)a | 62.5 (14.8) |
| Female | 45 (38.8) |
| Caucasian | 83 (71.5) |
| Aboriginal | 14 (12.0) |
| Diabetes mellitus | 58 (50.0) |
| Metropolitan resident | 87 (75.0) |
| Time on peritoneal dialysis (mo)b | 13.2 (4.9–29.8) |
| Prior peritonitisc | 24 (20.1) |
| Automated peritoneal dialysis | 37 (33.3) |
| Exit site abnormald | 14 (12.2) |
Mean (SD).
Median (first to third quartile).
Prior peritonitis at first presentation from 157 episodes.
The findings were coded as exit site normal or abnormal.
Table 2.
Distribution of microorganism by composite outcome
| Organism | Total episodes n (%) |
PD-cure n (%) |
PD-fail n (%) |
|---|---|---|---|
| Other Gram positive | 58 (36.9) | 47 (39.5) | 11 (28.9) |
| Staphylococcus aureus | 19 (12.1) | 14 (11.8) | 5 (13.2) |
| Gram negative | 31 (19.7) | 22 (18.5) | 9 (23.7) |
| Culture negative | 47 (29.9) | 35 (29.4) | 12 (31.6) |
| Mixed organism | 2 (1.3) | 1 (0.8) | 1 (2.6) |
Culture results were available for 157 of 159 episodes.
Thirty-eight (23.9%) episodes led to the primary composite end point of death (n = 10, 6.3%) or PD catheter removal (n = 28, 17.6%) at 30 days. Deaths were attributed to cardiovascular events (n = 2), biliary complications (n = 1), sepsis (n = 6), and unknown (n = 1). Two of the patients who died had tube removal prior to death. Characteristics of patient episodes are compared according to the primary end point (PD-fail) in Table 3, and for each component of the composite outcome. Compared with patient episodes associated with resolution of PDRP (PD-cure), there were a higher number of females with PD-fail but no significant association between PD-fail and age, time on PD, automated PD versus continuous ambulatory PD, presentation in or outside of standard working hours, diabetes mellitus, abnormal exit site, aboriginality, geographic location or presentation, or symptoms at presentation. Patients who died were older and more likely diabetic and female.
Table 3.
Demographic and clinical variables according to the composite outcome of PD-fail (death or catheter loss) at 30 days
| Variable | PD removed n = 28 (17.6%) |
Death n = 10 (6.3%) |
PD-fail n = 38 (23.9%) |
PD-cure n = 121 (76.1%) |
OR | 95% CI | P value |
|---|---|---|---|---|---|---|---|
| Age (yr) (SD) | 56.1 (17.6) | 74.1 (12.0) | 60.8 (18.0) | 63.1 (14.0) | 0.99 | 0.96–1.02 | 0.48 |
| Female | 15 (53.6) | 7 (70.0) | 22 (57.9) | 39 (32.2) | 2.89 | 1.34–6.23 | 0.007 |
| Aboriginal | 4 (14.3) | 0 (0) | 4 (10.5) | 14 (11.6) | 0.89 | 0.27–2.96 | 0.86 |
| Diabetes mellitus | 14 (50.0) | 8 (80.0) | 22 (57.9) | 63 (52.0) | 1.26 | 0.59–2.73 | 0.55 |
| Metropolitana | 20 (71.4) | 9 (90.0) | 29 (76.3) | 91 (75.2) | 1.06 | 0.43–2.62 | 0.9 |
| Time on PD (mo) | 10.8 (4.9–15.1) | 18.5 (5.3–42.1) | 11.6 (5.3–29.0) | 20.7 (8.0–32.7) | 0.99 | 0.96–1.01 | 0.37 |
| Prior peritonitisb | 6 (21.4) | 5 (50.0) | 11 (28.9) | 55 (45.5) | 0.49 | 0.22–1.08 | 0.07 |
| Cloudy effluent | 23 (82.1) | 8 (80.0) | 31 (81.6) | 113 (93.4) | 0.31 | 0.104–0.93 | 0.04 |
| Abdominal pain | 23 (82.1) | 6 (60.0) | 29 (76.3) | 84 (69.4) | 1.41 | 0.61–3.27 | 0.41 |
| Fever | 8 (28.6) | 4 (40.0) | 12 (31.6) | 27 (22.3) | 1.60 | 0.72–3.56 | 0.24 |
| Exit site abnormalc | 3 (10.7) | 1 (10.0) | 4 (10.5) | 15 (12.9) | 0.79 | 0.25–2.48 | 0.69 |
| CAPDd | 16 (64.0) | 6 (66.7) | 22 (64.7) | 78 (65.0) | 0.99 | 0.43–2.28 | 0.98 |
| APDd | 9 (36.0) | 3 (33.3) | 12 (35.3) | 42 (35) | |||
| ISPD peritonitis defined | 27 (96.4) | 8 (80.0) | 35 (92.1) | 106 (89.0) | 1.43 | 0.38–5.44 | 0.59 |
| Initial presentation to a hospital-based facility | 20 (71.4) | 10 (100.0) | 30 (78.9) | 83 (68.6) | 1.71 | 0.73–3.99 | 0.21 |
| 1st contact: 8 AM–6 PM | 18 (64.3) | 7 (70.0) | 25 (65.8) | 82 (67.8) | 0.91 | 0.43–1.93 | 0.81 |
| SC (h) | 7.2 (3.5–25.3) | 4.5 (2.5–6.5) | 5.6 (2.9–18.8) | 5.0 (1.0–13.8) | 1.002 | 0.99–1.01 | 0.73 |
| ST (h) | 12.4 (7.5–32.5) | 16.3 (5–30.3) | 13.6 (7–31) | 8.0 (4–20.8) | 1.003 | 0.99–1.01 | 0.48 |
| CT (h) | 3.6 (1.4–5.9) | 2.8 (2–19.5) | 3.4 (1.6–6.4) | 2.0 (1.00–3.6) | 1.055 | 1.005–1.11 | 0.032 |
| CT >4 h | 12 (43) | 4 (40) | 16 (42.1) | 27 (22.7) | 2.47 | 1.14–5.38 | 0.02 |
OR by logistic regression on composite outcome, data as mean (SD) or median and first to third quartile or n (%).
APD, automated peritoneal dialysis; CAPD, continuous ambulatory peritoneal dialysis; CI, confidence interval; CT, contact-to-treatment time; ISPD, International Society for Peritoneal Dialysis; OR, odds ratio; PD, peritoneal dialysis; SC, symptom-to-contact time; ST, symptom-to-treatment time.
Less than 100 km from Perth city.
Prior peritonitis episode defined as treated peritonitis >4 weeks before presentation.
Recorded for 154 episodes as exit site normal or abnormal.
Data missing for 5 episodes; OR for CAPD compared with APD shown.
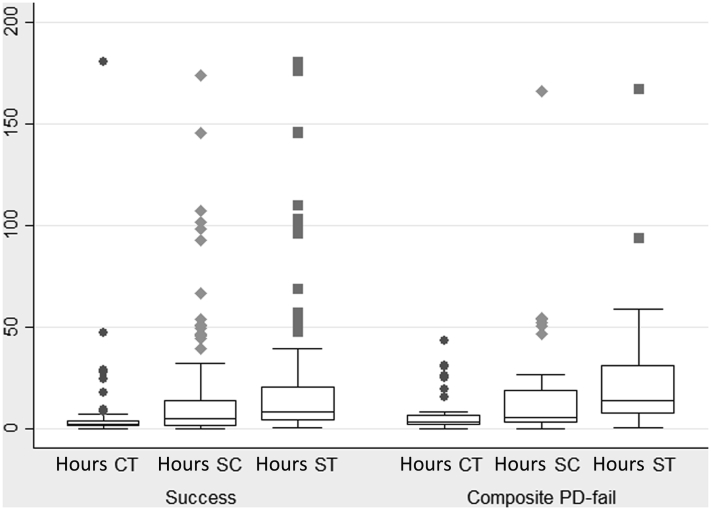
The median SC was 5.0 hours (Q1–Q3: 1.3–13.9) in all patients, 5.6 hours (2.9–18.8) in the PD-fail group, and 5.0 hours (1.0–13.8) in the PD-cure group, with no association between SC and PD-fail. The median CT was 2.3 hours (1.2–4.0) in all patients, 3.4 hours (1.6–6.3) in the PD-fail group, and 2.0 hours (1.0–3.6) in the PD-cure group. The median ST was 9.0 hours (4.7–25.3) in all patients, 13.6 hours (7–31) in the PD-fail group, and 8.0 hours (4.0–20.8) in the PD-cure group. Each hour of delay in the initiation of antimicrobial therapy was associated with an increased risk of catheter loss and death by 5.5% (OR: 1.055; 95% CI: 1.005–1.109; P = 0.032). In the PD-fail group, 42.1% of episodes compared with 22.7% in the PD-cure group received treatment ≥4 hours after initial contact. A CT of ≥4 hours increased the risk of PD-fail or death 2.5-fold (OR: 2.47; 95% CI: 1.14–5.4; P = 0.014). There was no association between presentation within or outside of working hours and PD-fail. The distribution of treatment times by the composite outcome is illustrated in Figure 2. The median SC in patients who presented to hospitals compared with ambulant facilities was 4.6 and 7.8 hours, respectively (P = 0.48), and the CT was 2.8 and 1.7 hours, respectively (P = 0.036). The median CT in patients presenting to hospitals compared with ambulant facilities according to the composite outcome was 2.5 and 1.5 hours, respectively, for PD-cure, and 4.0 and 2.5 hours, respectively, for PD-fail.
Figure 2.
Distribution of time variables (box-and-whisker plots) by composite outcome. CT, contact-to-treatment time; SC, symptom-to-contact time; ST, symptom-to-treatment time.
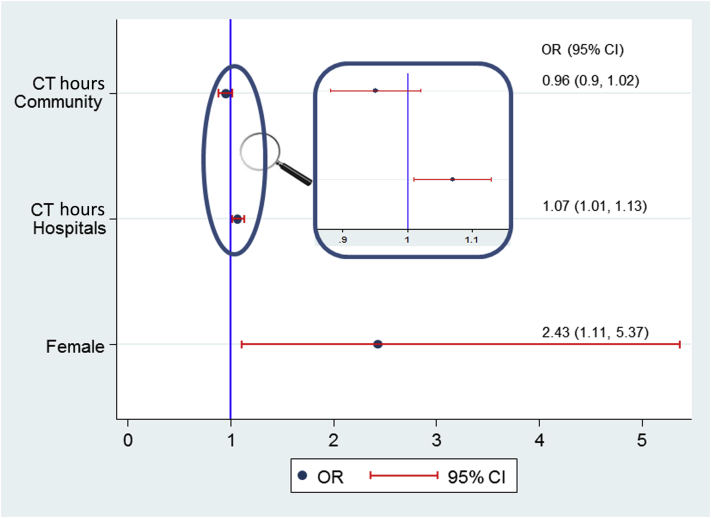
In a multivariable logistic regression, a significant interaction between CT (but not SC or ST) and health facility at presentation was detected (P = 0.013), indicating that the odds of PD-fail due to treatment delay was dependent on the initial site of presentation. No association between CT and PD-fail was detected for presentation to an ambulant care facility (OR: 0.96; 95% CI: 0.90–1.02; P = 0.22); however, at a hospital-based facility each additional hour of treatment delay was found to increase the odds of PD-fail by 6.8% (OR: 1.068; 95% CI: 1.01–1.13; P = 0.015) (Figure 3). Female sex continued to be independently associated with a 2.4-fold higher odds of PD-fail compared to males after adjusting for CT and health facility effects. Excluding episodes that did not meet the ISPD criteria for peritonitis did not significantly alter the primary multivariable analysis results (CT in hospitals OR: 1.063; 95% CI: 1.003–1.13; P = 0.04; and female sex OR: 2.5; 95% CI: 1.09–5.06; P = 0.031). In a separate analysis examining the association of gender with other variables including age, diabetes, cloudy bag, effluent white cell count, previous episodes, abdominal pain, and CT, there were no significant associations found to potentially account for the higher female PD-fail outcome (data not shown).
Figure 3.
Multivariable model for predictors of the composite outcome PD-fail. CI, confidence interval; CT, contact-to-treatment time; OR, odds ratio.
Discussion
This study demonstrated that in PDRP episodes presenting to hospitals, every hour of antibiotic-treatment delay from the initial contact with a hospital facility increases the odds of the composite outcome of catheter loss and death. The time from symptom onset to presentation to a health facility for antibiotic treatment, although numerically greater than that from presentation to treatment, was not associated with the composite outcome. In contrast with other studies, diabetes, age, aboriginality, organism type, automated PD versus continuous ambulatory PD, abnormal exit site, time on PD, and prior peritonitis episodes were not associated with the outcome. Although ISPD guidelines implicitly recognize that antibiotic treatment is a priority based on expert opinion, there is no evidence to support a relationship between the timing of administration of antibiotics and PD-related outcomes. Many studies in medicine support antibacterial therapy during the “golden hour” or “antibiotics as soon as possible,” including meningitis,21 Pseudomonas17 and staphylococcal18 bacteremia, septic shock, and pneumonia.15, 16, 19, 20 The principle is also well recognized in acute vascular syndromes such as stroke22 and myocardial infarction.23 The timely administration of antimicrobial therapy in hypotensive patients with sepsis was associated with increased survival to hospital discharge, and there was a linear relationship between timing of antibiotic administration and survival.24 Houck et al.16 similarly demonstrated that a >4-hour delay in administration of antibiotics after an intensive care unit admission increased mortality in community-acquired pneumonia. The finding of a similar relationship between treatment delay and outcomes in PD peritonitis in this study is consistent with established infectious disease evidence and, to our knowledge, the first to document such an association.
The high rate of patient death or catheter loss at 30 days observed in this cohort is consistent with outcomes reported in Australia1 and other international studies.25, 26, 27 The 3-step process of symptom recognition, health-provider contact, and administration of antibiotic allows multiple potential barriers to timely treatment. The adverse effect of delayed antibiotic administration detected only in patients treated in hospitals may, in part, be due to self-selection of the better patient to an ambulant care facility whereas sicker patients are more likely to be referred or present directly to hospitals. Patients attending an ambulant care facility had earlier treatment times than those presenting to hospitals in both the PD-fail and PD-cure groups. Ambulant care services in this study only manage patients receiving dialysis and are more likely to be familiar with the presenting symptoms and signs and rapidly assess and deliver care without awaiting unnecessary tests, additional clinical review, or medical approval. In contrast, presentation to hospitals with differing triage priorities and variable familiarity with PDRP may impose delays pending additional tests and medical review, despite the existence of established treatment protocols. However, no association between presentation within or outside working hours and outcome was found. We did not show the timing from onset of symptoms, or remoteness from a health facility, to be associated with an adverse outcome. Patient delay in recognizing symptoms and their significance, or recalling their onset accurately, is suggested by the wide spread of symptom time prior to hospital contact and may account for the absence of any association between symptom duration and outcome.
Females had twice the odds of catheter loss or death. This is consistent with some27, 28 but not other studies.5, 29 We could not identify any additional gender-specific differences within our study that might explain this finding. It is likely that variability and bias in patient selection, inclusion of different variables, and different study populations contribute to the reported differences in outcome by gender.
We included patients who were presumptively treated for PDRP and used the diagnostic criteria of the ISPD guidelines for verification. The majority met these definitions, indicating a high degree of clinically appropriate treatment. The failure to fully confirm diagnostic criteria in all cases may have been due to inadequate sample collection, prior antibiotic exposure, and laboratory technical aspects. Variability in the pathogenicity of causative microorganisms is a recognized factor associated with varying risk of technique failure and mortality. Gram-positive peritonitis has often shown better resolution rates compared with other bacterial infection, and non-Pseudomonas Gram-negative infections have better outcomes over Pseudomonas infections.17, 27, 30, 31 The rate of culture-negative episodes was slightly higher in our study compared with other reports, although consistent with contemporary Australian data.2 We speculate that varying laboratory collection standards and timing of collection of samples across a large geographical provider base might influence these results. In our study the distribution of bacteria and culture-negative peritonitis was not different between patients who recovered or failed treatment, suggesting that the timing of the administration of appropriate antibiotics is a better predictor of outcome than the type of infection. However, small sample size may have restricted the opportunity to discern an effect of specific organisms and response to treatment. Despite registry data suggesting that remoteness and aboriginal race4 are associated with inferior PDRP outcomes, this was not confirmed in our study, perhaps because of the smaller patient numbers in our study, or because the timing of administration of antibiotics is unaccounted for as a confounder in registry data analysis.
This study has several strengths and limitations. We provided a “real world” evaluation of treatment of PDRP in a diverse population managed by multiple health providers across a vast geographic area. All the patients were treated according to a standardized local protocol, and we obtained follow-up data on all included episodes and patients. Data were collected from consecutive episodes with prospective recruitment and retrospective data confirmation. We included a small number of episodes that did not meet the ISPD guidelines for peritonitis; however, we show that exclusion of these patients does not alter the conclusions of this study. In our study, antibiotic treatment was administered at a wide range of geographically dispersed health care providers, which may not be typical of other health care providers or health networks.
In conclusion, we have demonstrated, for the first time to our knowledge, an independent graded and significant association between contact with a medical facility with presumed PDRP, and the time of the initial administration of antimicrobial therapy, and the risk of technique failure and mortality. This increased risk was confined to those presenting via hospitals when compared with ambulant care dialysis facilities, potentially implicating health care system impediments to timely care. These data suggest the requirement of new research, exploring strategies to expedite the timely delivery of antibiotics to improve the current PDRP outcomes and reduce the risk of death or catheter loss.
Disclosure
All the authors declared no competing interests.
Acknowledgments
The authors are grateful for the invaluable assistance provided by the nurses in all the hospital PD units: Denise Fallon, Toni Bennett, Nicky Warnecke, Viki Link, Alison Farmer, and Vivean Perreau. We are also grateful to Angelina Villarba, Adriana Viola, and Gerrie Vandepeer at Fresenius Medical Services for their involvement.
References
- 1.Brown F, Gulyani A, McDonald S, Hurst K. Peritoneal Dialysis, ANZDATA Registry Report 2012. Australia and New Zealand Dialysis and Transplant registry; 2012. Chapter 6: 1–32.
- 2.Boudville N, Dent H, McDonald Stephen, et al. Peritoneal Dialysis, ANZDATA Registry Report 2013. Australia and New Zealand Dialysis and Transplant Registry; 2013. Chapter 6: 1–31.
- 3.Kolesnyk I., Dekker F.W., Boeschoten E.W., Krediet R.T. Time-dependent reasons for peritoneal dialysis technique failure and mortality. Perit Dial Int. 2010;30:170–177. doi: 10.3747/pdi.2008.00277. [DOI] [PubMed] [Google Scholar]
- 4.Lim W.H., Johnson D.W., McDonald S.P. Higher rate and earlier peritonitis in Aboriginal patients compared to non-Aboriginal patients with end-stage renal failure maintained on peritoneal dialysis in Australia: analysis of ANZDATA. Nephrology. 2005;10:192–197. doi: 10.1111/j.1440-1797.2005.00374.x. [DOI] [PubMed] [Google Scholar]
- 5.Nessim S.J., Bargman J.M., Austin P.C. Predictors of peritonitis in patients on peritoneal dialysis: results of a large, prospective Canadian database. Clin J Am Soc Nephrol. 2009;4:1195–1200. doi: 10.2215/CJN.00910209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lloyd A., Tangri N., Shafer L.A. The risk of peritonitis after an exit site infection: a time-matched, case-control study. Nephrol Dial Transplant. 2013;28:1915–1921. doi: 10.1093/ndt/gft002. [DOI] [PubMed] [Google Scholar]
- 7.van Diepen A.T.N., Tomlinson G.A., Jassal S.V. The association between exit site infection and subsequent peritonitis among peritoneal dialysis patients. Clin J Am Soc Nephrol. 2012;7:1266–1271. doi: 10.2215/CJN.00980112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wanten G.J., van Oost P., Schneeberger P.M., Koolen M.I. Nasal carriage and peritonitis by Staphylococcus aureus in patients on continuous ambulatory peritoneal dialysis: a prospective study. Perit Dial Int. 1996;16:352–356. [PubMed] [Google Scholar]
- 9.Cho Y., Badve S.V., Hawley C.M. Seasonal variation in peritoneal dialysis-associated peritonitis: a multi-centre registry study. Nephrol Dial Transplant. 2012;27:2028–2036. doi: 10.1093/ndt/gfr582. [DOI] [PubMed] [Google Scholar]
- 10.Miles R., Hawley C.M., McDonald S.P. Predictors and outcomes of fungal peritonitis in peritoneal dialysis patients. Kidney Int. 2009;76:622–628. doi: 10.1038/ki.2009.202. [DOI] [PubMed] [Google Scholar]
- 11.Jose M.D., Johnson D.W., Mudge D.W. Peritoneal dialysis practice in Australia and New Zealand: a call to action. Nephrology. 2011;16:19–29. doi: 10.1111/j.1440-1797.2010.01390.x. [DOI] [PubMed] [Google Scholar]
- 12.Cho Y., Johnson D.W. Peritoneal dialysis-related peritonitis: towards improving evidence, practices, and outcomes. Am J Kidney Dis. 2014;64:278–289. doi: 10.1053/j.ajkd.2014.02.025. [DOI] [PubMed] [Google Scholar]
- 13.Li P.K., Szeto C.C., Piraino B. Peritoneal dialysis-related infections recommendations: 2010 update. Perit Dial Int. 2010;30:393–423. doi: 10.3747/pdi.2010.00049. [DOI] [PubMed] [Google Scholar]
- 14.Tunkel A.R., Hartman B.J., Kaplan S.L. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39:1267–1284. doi: 10.1086/425368. [DOI] [PubMed] [Google Scholar]
- 15.Gacouin A., Le Tulzo Y., Lavoue S. Severe pneumonia due to Legionella pneumophila: prognostic factors, impact of delayed appropriate antimicrobial therapy. Intensive Care Med. 2002;28:686–691. doi: 10.1007/s00134-002-1304-8. [DOI] [PubMed] [Google Scholar]
- 16.Houck P.M., Bratzler D.W., Nsa W. Timing of antibiotic administration and outcomes for Medicare patients hospitalized with community-acquired pneumonia. Arch Intern Med. 2004;164:637–644. doi: 10.1001/archinte.164.6.637. [DOI] [PubMed] [Google Scholar]
- 17.Kang C.I., Kim S.H., Kim H.B. Pseudomonas aeruginosa bacteremia: risk factors for mortality and influence of delayed receipt of effective antimicrobial therapy on clinical outcome. Clin Infect Dis. 2003;37:745–751. doi: 10.1086/377200. [DOI] [PubMed] [Google Scholar]
- 18.Lodise T.P., McKinnon P.S., Swiderski L., Rybak M.J. Outcomes analysis of delayed antibiotic treatment for hospital-acquired Staphylococcus aureus bacteremia. Clin Infect Dis. 2003;36:1418–1423. doi: 10.1086/375057. [DOI] [PubMed] [Google Scholar]
- 19.McGarvey R.N., Harper J.J. Pneumonia mortality reduction and quality improvement in a community hospital. QRB Qual Rev Bull. 1993;19:124–130. doi: 10.1016/s0097-5990(16)30605-4. [DOI] [PubMed] [Google Scholar]
- 20.Meehan T.P., Fine M.J., Krumholz H.M. Quality of care, process, and outcomes in elderly patients with pneumonia. JAMA. 1997;278:2080–2084. [PubMed] [Google Scholar]
- 21.Miner J.R., Heegaard W., Mapes A., Biros M. Presentation, time to antibiotics, and mortality of patients with bacterial meningitis at an urban county medical center. J Emerg Med. 2001;21:387–392. doi: 10.1016/s0736-4679(01)00407-3. [DOI] [PubMed] [Google Scholar]
- 22.Saver J.L., Fonarow G.C., Smith E.E. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA. 2013;309:2480–2488. doi: 10.1001/jama.2013.6959. [DOI] [PubMed] [Google Scholar]
- 23.Herlitz J., Wireklintsundstrom B., Bang A. Early identification and delay to treatment in myocardial infarction and stroke: differences and similarities. Scand J Trauma Resusc Emerg Med. 2010;18:48. doi: 10.1186/1757-7241-18-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kumar A., Roberts D., Wood K.E. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34:1589–1596. doi: 10.1097/01.CCM.0000217961.75225.E9. [DOI] [PubMed] [Google Scholar]
- 25.Perl J., Wald R., Bargman J.M. Changes in patient and technique survival over time among incident peritoneal dialysis patients in Canada. Clin J Am Soc Nephrol. 2012;7:1145–1154. doi: 10.2215/CJN.01480212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sanchez A.R., Madonia C., Rascon-Pacheco R.A. Improved patient/technique survival and peritonitis rates in patients treated with automated peritoneal dialysis when compared to continuous ambulatory peritoneal dialysis in a Mexican PD center. Kidney Int Suppl. 2008;(108):S76–S80. doi: 10.1038/sj.ki.5002606. [DOI] [PubMed] [Google Scholar]
- 27.Perez Fontan M., Rodriguez-Carmona A., Garcia-Naveiro R. Peritonitis-related mortality in patients undergoing chronic peritoneal dialysis. Perit Dial Int. 2005;25:274–284. [PubMed] [Google Scholar]
- 28.Ros S., Remon C., Qureshi A.R. Increased risk of fatal infections in women starting peritoneal dialysis. Perit Dial Int. 2013;33:487–494. doi: 10.3747/pdi.2012.00243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jager K.J., Merkus M.P., Dekker F.W. Mortality and technique failure in patients starting chronic peritoneal dialysis: results of The Netherlands Cooperative Study on the Adequacy of Dialysis. NECOSAD Study Group. Kidney Int. 1999;55:1476–1485. doi: 10.1046/j.1523-1755.1999.00353.x. [DOI] [PubMed] [Google Scholar]
- 30.Siva B., Hawley C.M., McDonald S. Pseudomonas peritonitis in Australia: predictors, treatment and outcomes in 191 cases. Clin J Am Soc Nephrol. 2009;4:957–964. doi: 10.2215/CJN.00010109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Barretti P., Montelli A.C., Batalha J.E.N. The role of virulence factors in the outcome of staphylococcal peritonitis in CAPD patients. BMC Infect Dis. 2009;9 doi: 10.1186/1471-2334-9-212. 212–212. [DOI] [PMC free article] [PubMed] [Google Scholar]