Table 4. FDA benefit‐risk assessment.
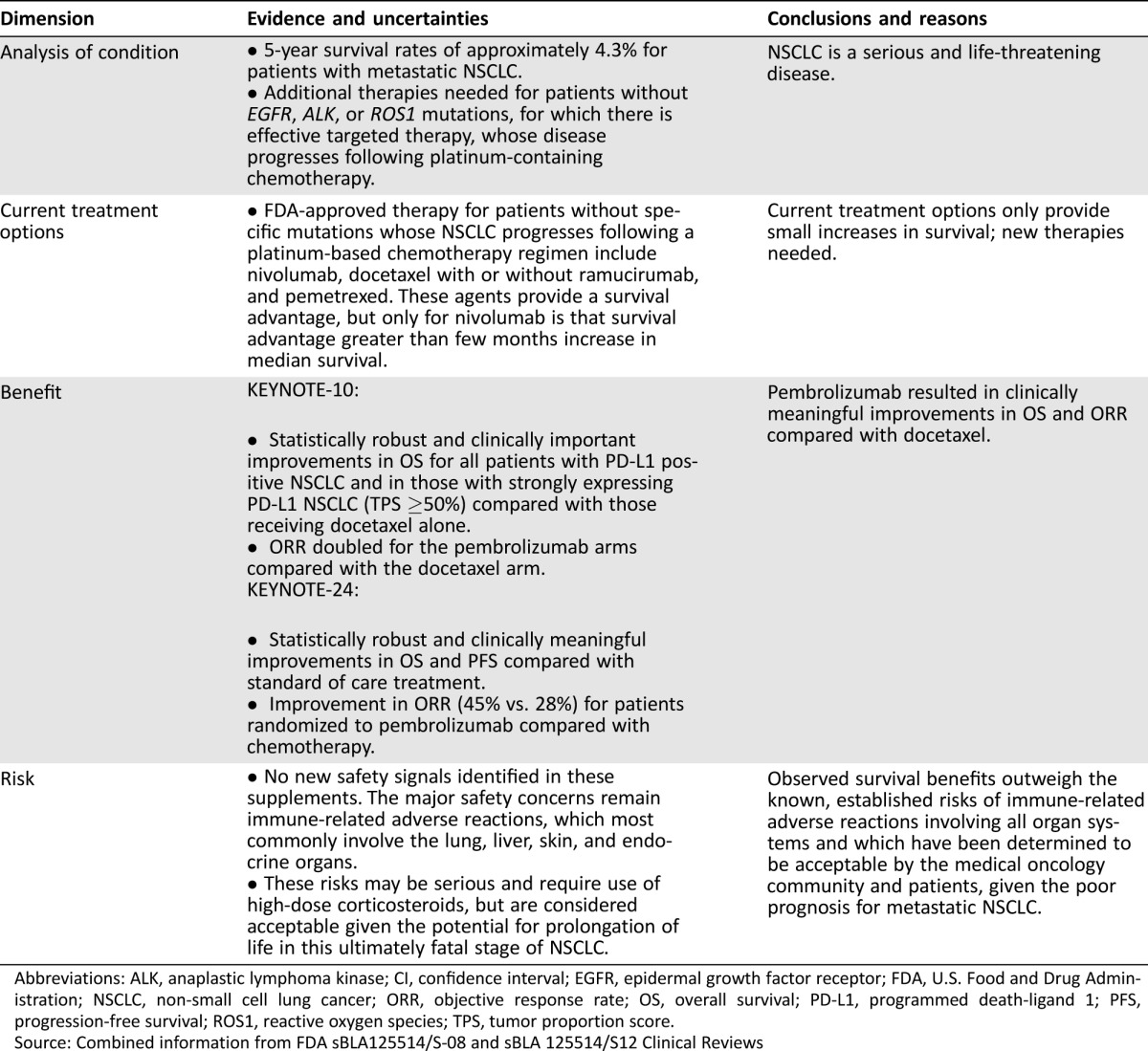
Abbreviations: ALK, anaplastic lymphoma kinase; CI, confidence interval; EGFR, epidermal growth factor receptor; FDA, U.S. Food and Drug Administration; NSCLC, non‐small cell lung cancer; ORR, objective response rate; OS, overall survival; PD‐L1, programmed death‐ligand 1; PFS, progression‐free survival; ROS1, reactive oxygen species; TPS, tumor proportion score.
Source: Combined information from FDA sBLA125514/S‐08 and sBLA 125514/S12 Clinical Reviews
