Abstract
The SMOOTHENED inhibitor vismodegib is FDA approved for advanced basal cell carcinoma (BCC), and shows promise in clinical trials for SONIC HEDGEHOG (SHH)-subgroup medulloblastoma (MB) patients. Clinical experience with BCC patients shows that continuous exposure to vismodegib is necessary to prevent tumor recurrence, suggesting the existence of a vismodegib-resistant reservoir of tumor-propagating cells. We isolated such tumor-propagating cells from a mouse model of SHH-subgroup MB and grew them as sphere cultures. These cultures were enriched for the MB progenitor marker SOX2 and formed tumors in vivo. Moreover, while their ability to self-renew was resistant to SHH inhibitors, as has been previously suggested, this self-renewal was instead WNT-dependent. We show here that loss of Trp53 activates canonical WNT signaling in these SOX2-enriched cultures. Importantly, a small molecule WNT inhibitor was able to reduce the propagation and growth of SHH-subgroup MB in vivo, in an on-target manner, leading to increased survival. Our results imply that the tumor-propagating cells driving the growth of bulk SHH-dependent MB are themselves WNT dependent. Further, our data suggest combination therapy with WNT and SHH inhibitors as a therapeutic strategy in patients with SHH-subgroup MB, in order to decrease the tumor recurrence commonly observed in patients treated with vismodegib.
INTRODUCTION
Medulloblastoma (MB) is the most common malignant brain tumor of childhood.1 While the bulk of patients respond to multimodal therapy (surgery, radiation and cytotoxic chemotherapy), treatment-induced morbidity leaves survivors with significant neurocognitive and endocrine disabilities.2 These patients thus require therapies that promise to be more effective and less toxic. The classification of MB into four molecular subgroups3,4 provided a template with which to begin identifying and testing such targeted therapies.5,6 The best characterized subgroup is driven by the SONIC HEDGEHOG (SHH) signaling pathway, most commonly via mutation and loss of the gene encoding the SHH receptor PATCHED (PTCH). The SHH inhibitor vismodegib targets the upstream signaling component SMOOTHENED (SMO) and is FDA-approved for advanced basal cell carcinoma,7 which is almost uniformly driven by SHH signaling.8,9 As similar SMO inhibitors effectively target SHH-dependent mouse models of MB, significantly reducing the bulk of such tumors,10 they are now being tested in SHH-subgroup MB patients.11–13
Tumors are heterogeneous, comprised of immune, stromal and tumor cells proper. Even the tumor cells themselves show significant variation in morphologies, proliferation rates and self-renewing capacity.14,15 This heterogeneity is thought to result from a hierarchical organization, in which a small population of relatively quiescent, long-lived, multipotent, propagating/initiating cells drives the production of a population of rapidly amplifying progenitor cells, which in turn can give rise to more differentiated cell types.16 Although the progenitor cells constitute the bulk of the tumor, they have limited self-renewing capacity, and are thus maintained by the smaller population of tumor-propagating cells. The tumor-propagating cells are relatively chemotherapy resistant, and are therefore thought to drive the tumor recurrence often observed in patients treated with chemotherapy.17 Similarly, while limited clinical experience with SMO inhibitors in clinical trials has shown that there is often a rapid initial response to vismodegib, this is frequently followed by aggressive recurrent MB growth.11,18 Additionally, it was noted that basal cell carcinoma patients often exhibited rapid recurrence upon vismodegib cessation, consistent with such tumors harboring a reservoir of vismodegib-resistant cells capable of driving recurrence.19–22
A number of tumor-propagating cells have been described for the SHH-subgroup of MB, with distinct properties, biomarkers, sensitivity to SMO inhibitors and abilities to be propagated as neurospheres ex vivo.23–28 Most recently, lineage tracing in a mouse model of SHH MB identified a small population of SOX2+ MB propagating cells (MPCs), and a population of SOX2− cells that constituted the bulk of the tumor proper.28,29 The SOX2+ cells were capable of efficient self-renewal but resistant to vismodegib, while the SOX2− cells exhibited a more limited capacity for self-renewal but were sensitive to vismodegib. Consistent with this observation, treatment of this mouse model of MB with vismodegib led to an enrichment of SOX2+ cells in the residual tumor.28 These SOX2+ cells may contribute to recurrence observed in patients treated with vismodegib. Here, we show that self-renewal of SOX2+ enriched sphere cultures (SCs) is dependent on canonical WNT activity, and that loss of Trp53 drives this WNT dependence. A small molecule WNT inhibitor also depleted this population of SOX2+ cells in SHH-subgroup MB in vivo, significantly reducing the ability of SHH tumors to propagate.
RESULTS
Characterization of MB sphere cultures
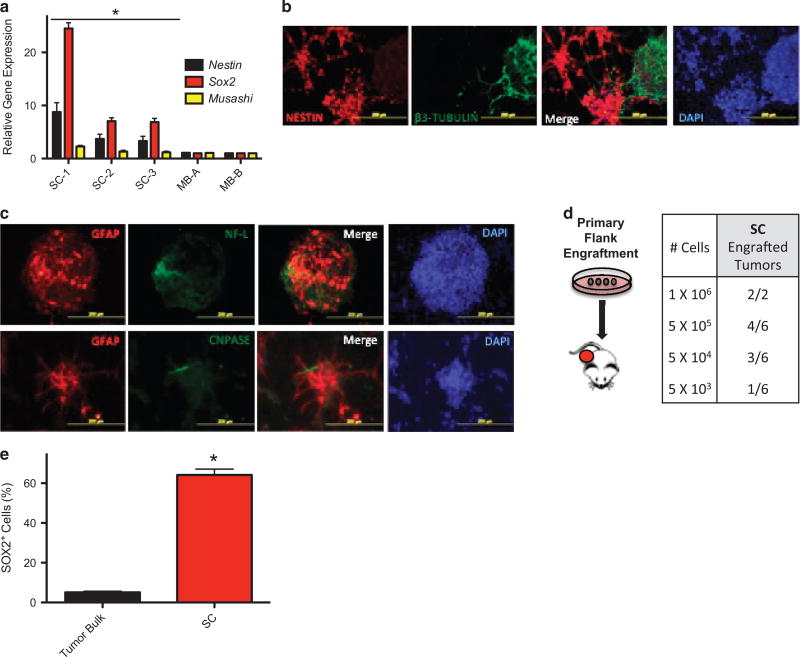
SHH-subgroup MBs harbor a small and phenotypically primitive population of MPCs that are enriched in cultured neurospheres ex vivo.25,27 We isolated and characterized the MPC-like characteristics of three independent Ptch1−/−-;Trp53−/− neurosphere cultures (SC 1–3). These cultures were enriched for neural stem cell markers, such as Nestin and Sox2, relative to bulk MB tissue (Figure 1a). They also displayed the cellular heterogeneity previously described for MPCs enriched cultures,25,30 as evidenced by the mutually exclusive staining of a neural stem cell marker (NESTIN) with that of a neuronal progenitor marker (β3-TUBULIN) (Figure 1b). The SCs could be induced to express biomarkers of three distinct neural lineages (Figure 1c), suggesting that they are also pluripotent. Consistent with them being enriched for tumor-propagating cells, as few as 5000 SC cells were able to form tumors when implanted into mice (Figure 1d). SOX2 has previously being described as a biomarker for such tumor-propagating cells.28,29 We therefore quantitated the percentage of SOX2+ cells in the SCs relative to that in primary MB tissue. SOX2+ cells were considerably more enriched in the SCs, relative to the approximate 5% of such cells observed in primary MB (Figure 1e).
Figure 1.
Medulloblastoma sphere cultures display stem cell properties. (a) The expression of the indicated progenitor cell biomarkers, from three independently derived Trp53-deficient MB sphere cultures (SC-1, 2 and 3) and two primary MBs (MB-A and MB-B), was determined and then normalized to that of MB-A. (b) SCs were immunostained for neuronal progenitor biomarkers (NESTIN and β3-TUBULIN). (c) The level of SC pluripotency was determined by examining the biomarkers GLIAL FRIBRILLARY ACIDIC PROTEIN (GFAP), NEUROFILAMENT-LARGE (NF-L) or CYCLIC-NUCLEOTIDE-PHOSPHODIESTERASE (CNPASE). Representative images of immunostained SC-2 are shown. (d) The indicated numbers of viable SC-2 cells were implanted subcutaneously into the flanks of immunocompromised mice. The frequency and number of tumor engraftments is shown. (e) The percentage of SOX2+ cells in MB flank tumors and SCs was determined by FACS analysis.
The self-renewal of MPCs is dependent on WNT activity
The MPC enriched SCs were exposed to the SMO inhibitors cyclopamine and vismodegib, the inactive cyclopamine analog tomatidine, or a vehicle control. Interestingly, even though the parental MB tissue responded to vismodegib in vivo (Supplementary Figure S1A), neither SMO inhibitor had significant effects on the cellular viability (Supplementary Figures S1B,C) or self-renewal (Supplementary Figures S1D,E) of the three SCs. However, they both attenuated SHH target gene expression (Supplementary Figures S1F,G,H) relative to either a vehicle control or cultures treated with tomatidine. These results suggest that SMO inhibitors are capable of attenuating SHH signaling, but that such attenuation has only minimal biological affects in these Trp53-deficient SCs.
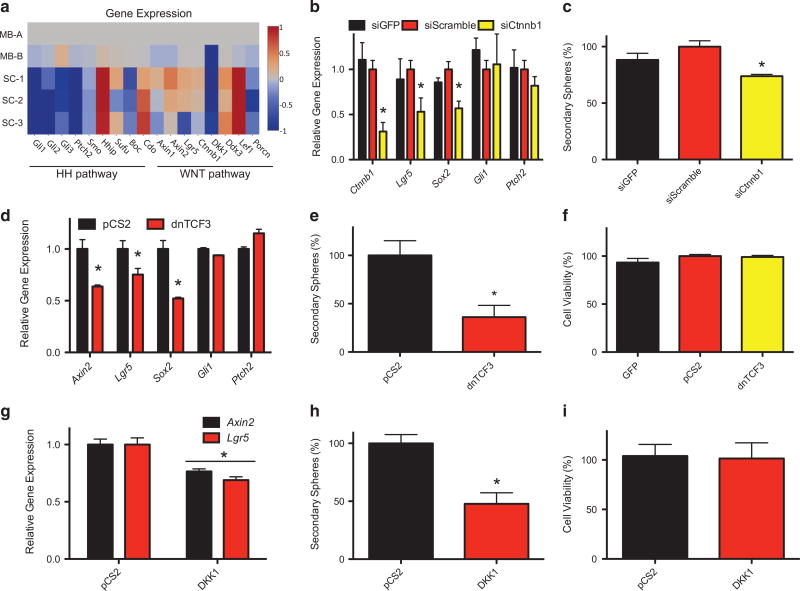
A WNT-driven gene expression profile was previously observed in Ptch1+/−;Trp53−/− SCs.27 Further, SOX2 expression, for which the SCs are enriched (Figure 1e), is WNT-dependent in many physiological settings.31 Thus, we hypothesized that the MB SCs we had isolated were WNT-dependent. Consistent with this hypothesis, SCs were enriched for WNT signaling biomarkers relative to primary MB (Figure 2a). To directly test whether SCs are WNT-dependent we transfected them with pools of siRNA targeting the pivotal WNT signaling component β-Catenin (Ctnnb1), green fluorescent protein (GFP), or a scrambled control siRNA. Relative to the control siRNAs, the Ctnnb1 siRNA reduced the expression of the WNT biomarker Lgr5 as well as Sox2, but had no effect on the SHH target genes Gli1 or Ptch2 (Figure 2b). Further, the Ctnnb1 siRNA attenuated the self-renewal capability of the SCs (Figure 2c), but had little effect on the overall viability of these cultures (Supplementary Figure S2A). Although WNT activation in MB is commonly linked to Ctnnb1 mutations,32,33 no Ctnnb1 mutations were identified in the SCs (Supplementary Figure S2B). As β-CATENIN also plays an essential role in E-CADHERIN signaling, distinct from its role in WNT signaling,34 we also utilized a dominant negative form of the WNT transcription factor TCF3 (dnTCF3) commonly used to definitively identify WNT-dependent biomarkers.35 Cultures expressing dnTCF3 exhibited significantly attenuated WNT target gene expression, including Sox2, but had no effect on the expression of two SHH target genes (Figure 2d). SCs expressing dnTCF3 had a reduced capacity for self-renewal (Figure 2e), but exhibited no change in viability (Figure 2f). To examine if the WNT activation we observe in SCs is ligand-driven we overexpressed a negative regulator of the canonical WNT pathway, DKK1, which antagonizes the LRP5/6 WNT co-receptor.36 Overexpression of DKK1 downregulated WNT target gene expression (Figure 2g), and reduced secondary sphere formation (Figure 2h) without affecting cell viability (Figure 2i). Combined, these results suggest that WNT signaling plays an important role in the self-renewal of the SCs, but a more minimal role in the immediate viability of these cultures. Consistent with this suggestion, two small-molecule activators of WNT signaling (GSK-3β inhibitors)37 significantly increased the self-renewal capacity of these SCs (Supplementary Figure S2C).
Figure 2.
The self-renewal of medulloblastoma sphere cultures is dependent on WNT activity. (a) A heat map representing the relative gene expression of SHH (Gli1, Gli2, Gli3, Ptch2, Smo, Hhip, Sufu, Boc, Cdo) and WNT pathway-related genes (Axin1, Axin2, Lgr5, Ctnnb1, Dkk1, Ddx3, Lef1, Porcn), as determined in the indicated Trp53-deficient SCs and Trp53-deficient MB tissues (MB-A and MB-B). The color scale was calculated using Log2 transformed gene expression data normalized to their expression in MB-A. (b) SC-2 were transfected with the indicated siRNA smart pools, and the expression of the indicated genes determined 72 h later. (c) The ability of SC-2 to form secondary spheres 72 h after transfection with the indicated siRNA smart pool was determined. (d) SC-2 were transfected with a plasmid expressing a dnTCF3 construct or a control plasmid (pCS2), and the expression of the indicated genes determined 48 h later. (e) The ability of SC-2 to form secondary spheres was determined 48 h after transfection with a dnTCF3 expression plasmid or control plasmid (pCS2). (f) SC-2 was transfected with a plasmid expressing a dnTCF3 construct or control plasmid (pCS2) and cell viability determined 5 days later using an MTT reduction assay. (g) SC-2 was transfected with a plasmid expressing DKK1 or control plasmid (pCS2) and the expression of WNT target genes determined. (h) The ability of SC-2 to form secondary spheres was determined 48 h after transfection with a plasmid expressing DKK1 or control plasmid (pCS2). (i) SC-2 was transfected with a plasmid expressing DKK1 or control plasmid (pCS2) and cell viability determined 5 days later using an MTT reduction assay. Results were normalized to that from pCS2 transfected cultures unless otherwise indicated. MTT, 3-(4,5-dimethyl-2-thiazolyl) 2,5-diphenyl-2H-tetrazolium bromide.
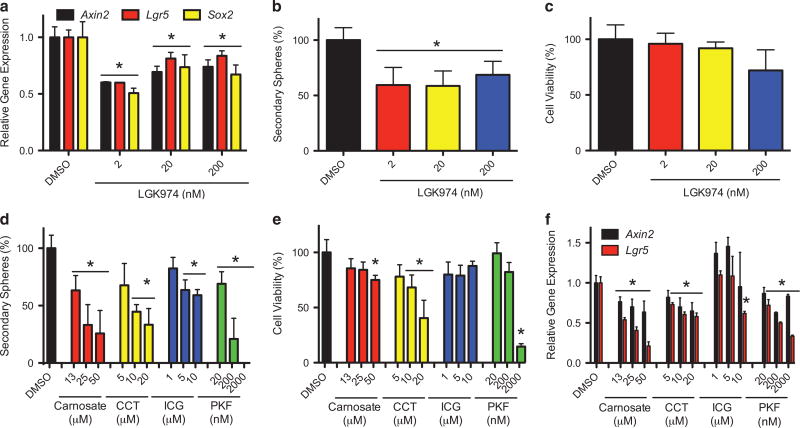
Small-molecule WNT inhibitors attenuate the self-renewal of MPCs To identify a small-molecule WNT inhibitor that could be used to attenuate MB growth in vivo, we screened a number of mechanistically distinct small-molecule WNT inhibitors for their ability to attenuate self-renewal ex vivo. As downregulation of WNT signaling by DKK1 overexpression reduced secondary sphere formation (Figure 2h), and DKK1 acts to suppress ligand-dependent WNT signaling,36 we tested a small-molecule that directly attenuates WNT ligand activity, the PORCUPINE inhibitor LGK974.38 Similar to DKK1 overexpression, PORCUPINE inhibition reduced WNT pathway activation (Figure 3a) and secondary sphere formation (Figure 3b), without affecting SC viability (Figure 3c). Consistent with this WNT activation being ligand driven, Wnt7a and Wnt7b were highly expressed in SCs relative to parental MB tissue (Supplementary Figure S3A).
Figure 3.
Small-molecule WNT inhibitors attenuate the self-renewal of medulloblastoma sphere cultures. (a) SC-2 were treated with the indicated concentrations of the PORCUPINE inhibitor LGK974, and the expression of the indicated genes determined 72 h later. (b) The ability of SC-2 to form secondary spheres was determined following incubation (72 h) with the indicated concentrations of LGK974. (c) SC-2 were incubated with the indicated concentrations of LGK974, and cell viability determined 5 days later using an MTT reduction assay. (d) The ability of SC-2 to form secondary spheres was determined following incubation (24 h) with the indicated concentrations of TCF/β-CATENIN inhibitors (carnosate, CCT-031374 hydrobromide [CCT], ICG-001 [ICG] or PKF-115-584 [PKF]). (e) SC-2 were incubated for 72 h with the indicated concentrations of TCF/β-CATENIN inhibitors, and cell viability determined 5 days later using an MTT reduction assay. (f) SC-2 were treated with the indicated TCF/β-CATENIN inhibitors for 24 h. The expression of the indicated genes was determined. Results were normalized to dimethyl sulfoxide control. MTT, 3-(4,5-dimethyl-2-thiazolyl) 2,5-diphenyl-2H-tetrazolium bromide.
As both Ctnnb1 knockdown (Figure 2c) and overexpression of dnTCF reduced secondary sphere formation (Figure 2e), we included in our screening antagonists of the TCF/β-CATENIN complex: carnosate,39 CCT-031374,40 ICG-00141 and PKF115-584.42 We first confirmed that this panel of TCF/β-CATENIN antagonists reduced WNT activity in a WNT-dependent reporter assay (Supplementary Figure S3B). We next treated SCs with these antagonists and observed a significant reduction in secondary sphere formation (Figure 3d), which occurred at lower doses than those that had effects on SC viability (Figure 3e). These WNT inhibitors also attenuated the expression of WNT target genes in SCs (Figure 3f), consistent with an on-target mechanism of action. We also verified that one of these inhibitors, carnosate, reduced the expression of Sox2 (Supplementary Figure S3C) and the numbers of SOX2+ cells in these cultures (Supplementary Figure S3D). Finally, we determined if carnosate affects either proliferation or survival of SOX+ cells. No changes in the expression of either proliferation proliferating cell nuclear antigen (PCNA) or apoptotic (CLEAVED CASPASE-3) biomarkers were observed in SOX2+ cells (Supplementary Figure S3E), consistent with minimal changes in the number of viable cells in culture after longer exposure to WNT inhibitor (Supplementary Figure S3F). Together, these results are consistent with WNT signaling controlling Sox2 expression, which is required for SCs to self-renew.
Loss of Trp53 results in WNT-dependent self-renewal
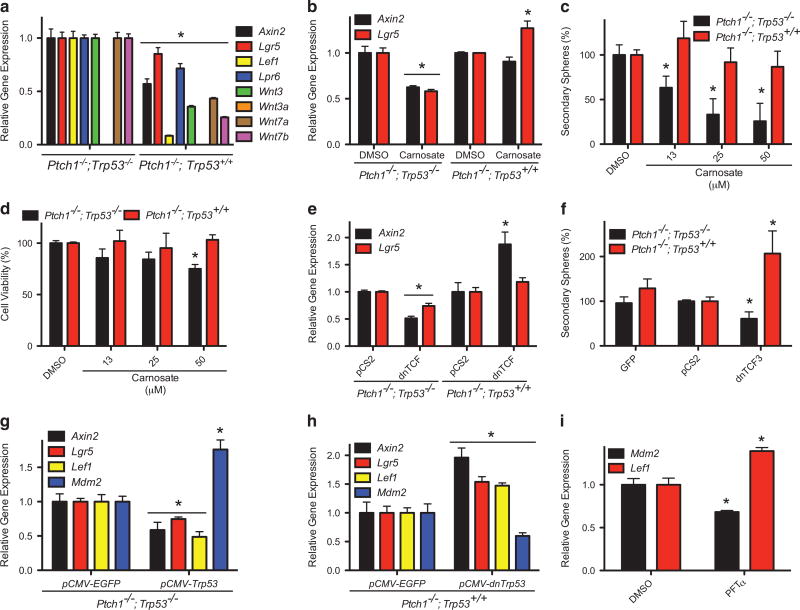
A number of groups have previously shown that WNT activation attenuates SHH-subgroup MB growth,43–45 observations inconsistent with our results. These previous results were obtained using SmoA1- or Ptch1-driven MB mouse models, or cells derived from such mice, which in all cases were in a wild type (WT) Trp53 background. Thus, we hypothesized that the WNT requirement for SC self-renewal that we observe results from loss of Trp53. To test this hypothesis, we compared the expression of WNT biomarkers in Ptch1−/− SCs expressing WT Trp53 or deficient for Trp53. We noted that expression of WNT biomarkers, including WNT ligands, was significantly reduced in the SCs expressing WT Trp53 (Figure 4a). SCs expressing WT Trp53 were unresponsive to small-molecule β-CATENIN/TCF inhibitors, as carnosate was unable to downregulate the expression of WNT target genes (Figure 4b), attenuate self-renewal (Figure 4c) or affect viability (Figure 4d). Further, overexpression of dnTCF3 did not attenuate WNT target gene expression (Figure 4e) or self-renewal (Figure 4f) in WT Trp53 SCs, but did in Trp53-deficient cultures. The cell viability of either SC was not reduced by dnTCF3 (Supplementary Figure S4A). To more directly address the effects of P53 on WNT signaling we attenuated P53 activity in SCs expressing WT Trp53 and expressed WT Trp53 in those cultures deficient in Trp53, and determined P53 and WNT activity. We observed increased expression of a P53 target gene (Mdm2) and decreased expression of WNT target genes upon transfection of Ptch1−/− SCs deficient in Trp53 with a plasmid expressing WT Trp53 (Figure 4g). Conversely, reducing P53 activity in Ptch1−/− SCs WT for Trp53, via expression of a dominant negative Trp53 construct or treatment with the small-molecule P53 inhibitor [Pifithrin-α (PFTα)], reduced Mdm2 expression but increased expression of WNT target genes (Figures 4h and i). Together, these results suggest that loss of Trp53 results in increased WNT activity in Ptch1 mutation-driven SCs.
Figure 4.
MPC enriched cultures expressing wild-type Trp53 are not WNT-dependent. (a) The expression of the indicated genes was determined in Ptch1−/−;Trp53−/− or Ptch1−/−;Trp53+/+ SCs, and normalized to the data from Ptch1−/−;Trp53−/− cultures. (b) SC cultures were treated with 25 µM carnosate for 24 h, and the expression of the indicated WNT target genes determined. (c) The ability of the SCs to form secondary spheres, following incubation (24 h) with the indicated concentrations of carnosate, was determined. (d) The indicated SCs were incubated for 3 days with the various concentrations of carnosate, and cell viability determined 5 days later using an MTT reduction assay. The results were normalized to those from the dimethyl sulfoxide control. (e) SCs were transfected with a plasmid expressing a dnTCF3 construct or a control plasmid (pCS2), and the expression of the indicated WNT target genes determined 48 h later. (f) The ability of SCs to form secondary spheres was determined 48 h after transfection with plasmids expressing dnTCF3, a control plasmid (pCS2) or GFP. Results were normalized to those from the pCS2 control. (g) Ptch1−/−;Trp53−/− SCs were transfected with plasmids expressing wild-type P53 (pCMV-Trp53), or an enhanced green fluorescent protein (EGFP) (pCMV-EGFP) control vector, and the expression of the indicated genes determined 72 h later. (h) Ptch1−/−; Trp53+/+ SCs were transfected with plasmids expressing dominant negative P53 (pCMV-dnTrp53) or EGFP (pCMV-EGFP), and the expression of the indicated genes determined 72 h later. The results were normalized to those from the EGFP expressing cultures. (i) Ptch1−/−;Trp53+/+ SCs were incubated with the P53/MDM2 inhibitor Pifithrin-α (PFT-α) for 24 h, and the expression of the indicated genes determined. The results were normalized to those from the dimethyl sulfoxide control. MTT, 3-(4,5-dimethyl-2-thiazolyl) 2,5-diphenyl-2H-tetrazolium bromide.
MB growth and propagation is WNT-dependent
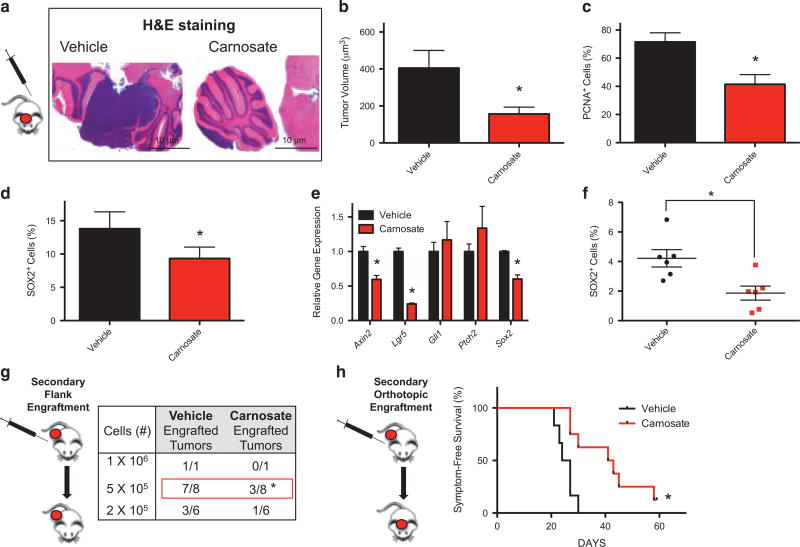
To examine the dependence of MB growth and initiation on WNT signaling in vivo, we implanted Trp53 mutant, Ptch1-driven MB tissue, which had never been cultured ex vivo, into the brains of immunocompromised mice and treated them with vehicle or carnosate. Carnosate was selected for these in vivo studies based on ex vivo efficacy and its blood brain barrier penetrance.46 These mice were treated for 20 days, or until they displayed behavior consistent with having MB (head tilt, hunched posture, ataxia, weight loss), at which time the mice were killed and their brains collected for further analyses. We noted that orthotopically grown MBs were significantly smaller in carnosate treated mice relative to vehicle treated mice (Figures 5a and b). These smaller residual tumors had decreased numbers of proliferating cells, as determined by PCNA immunostaining (Figure 5c, Supplementary Figure S5A), and decreased numbers of SOX2+ cells (Figure 5d, Supplementary Figure S5B). These results appear distinct from our ex vivo experiments in which carnosate did not affect culture growth (Figure 3e, Supplementary Figure S3F). We speculate that the effects we see on MB cell proliferation in vivo are indirect, resulting from loss of the MPCs that drive bulk tumor growth. Likely, similar effects were not observed ex vivo because of the significantly reduced time such cultures were exposed to carnosate.
Figure 5.
The propagation and growth of Trp53-deficient, SHH-subgroup medulloblastoma is WNT-dependent. Trp53 mutant, Ptch1-driven MB tissue, which had never been cultured ex vivo, was orthotopically implanted into the cerebellum of immunocompromised mice. These mice were then treated with carnosate or vehicle for 20 days, or until they developed MB symptoms. The mice were then killed and their brains collected. (a) Representative hematoxylin eosin staining of orthotopic MB tissue from a carnosate or vehicle-treated mouse is shown. (b) The volume of residual orthotopic tumors from these mice was calculated, and the mean volume and s.e.m. shown. (c) The residual orthotopic tumors from these mice were immunostained for the proliferation biomarker PCNA, and the numbers of PCNA+ cells quantified and normalized to total cell number per field (%). (d) The residual orthotopic tumors from these mice were immunostained for the MPC biomarker SOX2, and the numbers of SOX2+ cells quantified and normalized to total cell number per field (%). (e) MB tumor tissue was subcutaneously implanted into the flanks of immunocompromised mice, and treated daily with of carnosate, or vehicle control, for 8 days. The expression of the indicated genes in residual flank tumors was then determined. (f) The enrichment of SOX2+ cells in residual flank tissue, from carnosate or vehicle-treated mice, was determined by FACS analysis. (g) The indicated number of viable cells from residual MB tissue was transplanted into additional immunocompromised mice, and the frequency of tumor engraftment determined. (h) Equal numbers of viable cells from residual flank MB tissue were implanted into the cerebellum of additional immunocompromised mice, and MB symptom-free survival monitored for 60 days. Results were normalized to vehicle control.
To determine the WNT dependence of MPCs in vivo, we implanted MB tissue into the flanks of mice and treated them with carnosate or vehicle once tumor growth was palpable. Carnosate significantly reduced the expression of WNT target genes, but not SHH target genes, in these tumors (Figure 5e), and reduced the numbers of SOX2+ cells (Figure 5f, Supplementary Figure S5C). Consistent with carnosate treatment reducing the number of MPCs, MB tissue from the carnosate-treated primary tumors also displayed significantly reduced ability to initiate new tumors when similar numbers of viable donor cells were reimplanted into recipient mice (Figure 5g). We also reimplanted carnosate-treated MB tissue from donor mice orthotopically into naïve recipient mice, and monitored their MB symptom-free survival. Consistent with carnosate treatment reducing MB propagation, mice harboring carnosate-treated tumor tissue exhibited significantly increased MB symptom-free survival over those that received MB tissue from vehicle-treated donors (Figure 5h).
DISCUSSION
The WNT signaling pathway regulates multiple aspects of stem cell function. Indeed, WNT ligands play key roles in neuronal stem cell self-renewal,31 symmetric division,47 pluripotency48 and tumor initiation in cancer stem cells.31,49 We show here that the self-renewal of multipotent, SOX2+-enriched SCs, which ultimately drive the growth of Trp53-deficient SHH-subgroup MB, is dependent on WNT activity. This conclusion is based on inhibiting several components of the canonical WNT signaling pathway, using both molecular genetic and small-molecule inhibitors (Supplementary Figure S6). Further, we show that a small-molecule WNT inhibitor is able to reduce the number of SOX2+ cells, tumor initiation and tumor growth in vivo, increasing the overall survival of mice harboring Trp53-deficient SHH-subgroup MB.
Our Trp53-deficient SCs had reduced expression of SHH biomarkers relative to primary MB tissue, which is likely why they were resistant to SMO inhibitors.50 However, these cultures expressed both WNT ligands and biomarkers associated with an activated WNT signaling pathway. Consistent with MPC self-renewal being WNT ligand-dependent, a PORCUPINE inhibitor, which reduces WNT ligand activity,38 or overexpression of DKK1, which attenuates WNT co-receptor activation,36 prevented SCs self-renewal. The self-renewal of Trp53-deficient SCs also required canonical β-CATENIN/TCF activity, as knockdown of β-Catenin, overexpression of dnTCF3, or four distinct small-molecule inhibitors of this WNT-dependent transcriptional complex, inhibited self-renewal. Based on the sensitivity of these cultures to these mechanistically distinct small molecule and molecular genetic regulators of WNT signaling, we suggest that the MPC-enriched cultures elaborate a WNT ligand-dependent activation of canonical β-CATENIN/TCF signaling, which ultimately drives SC self-renewal.
It was previously suggested that WNT signaling attenuates the growth of SHH-subgroup MB.43–45 This work showed that the repressive effect of WNT activity on SHH-subgroup MB was indirect, via downregulation of SHH signaling. The major difference between these previous studies and our work is that we use a Trp53-deficient mouse model of MB. Consistent with this previous work, we show that SCs expressing WT Trp53 do not express high levels of WNT biomarkers, are not sensitive to WNT inhibition and do not self-renew in a WNT-dependent manner. However, we also show that reducing P53 activity in these WT Trp53 cultures increased WNT activity and that overexpression of Trp53 in Ptch1−/−;Trp53−/− SCs reduced WNT activity. Together, these results suggest that loss of Trp53 expression modulates the WNT dependence of Ptch1 mutant-driven MB. Similarly, it was previously suggested that P53 can repress canonical WNT signaling in several tumor types. In some tumors, such as neuroblastoma and breast cancer, this regulation was shown to occur via the transcriptional activation of specific micro-RNAs.51,52 While in colorectal cancer, loss of TRP53 activates WNT signaling via BONE MORPHOGENIC PROTEIN signaling.53
TRP53 status is the most important risk factor of SHH-subgroup MB patients.13,54,55 These patients, who tend to be children with germline TRP53 mutations, exhibit a significantly reduced 5-year survival rate.56 Further, clinical trials of vismodegib noted reduced progression-free survival of SHH-subgroup MB patients deficient in P53 activity.13 Our findings suggest a model in which loss of Trp53 results in the emergence of a vismodegib resistant, WNT-dependent population of MPCs. We further suggest that this population of WNT-dependent MPCs might underlie the poor outcome and relative vismodegib resistance of SHH-subgroup patients deficient in TRP53. Indeed, a similar model in which WNT-dependent basal cell carcinoma propagating cells drive bulk SHH-dependent basal cell carcinoma growth has been described.57,58 Although the translational relevance of our findings using mouse models of MB are not yet known, we speculate that a therapeutic approach using WNT inhibitors would improve the outcome of TRP53-deficient SHH-subgroup patients. Importantly, such patients would likely benefit most from the use of combination therapy that also includes conventional chemotherapy or one of the SMO inhibitors now in clinical trials. Such a therapeutic strategy would inhibit the growth of the bulk tumor by targeting the rapidly dividing MB cells as well as target the MPCs that are resistant to such agents.59
MATERIALS AND METHODS
MPC enrichment and assay
MB tissue was enzymatically and mechanically digested using the Papain Dissociation System (Worthington, Lakewood, NJ, USA).60,61 The resulting cell suspensions were grown ex vivo in Neurobasal serum-free medium containing 1% Glutamax, 2% B27, 1% N2, epidermal growth factor (25 ng/ml), fibroblast growth factor (25 ng/ml) and Pen-Strep (Invitrogen, Frederick, MD, USA) and allow to form spheres for up to 10 days. For analysis by immunocytochemistry, SCs were plated on poly-l-lysine-coated glass chamber slides (Millipore, Billerica, MA, USA) in the medium described above or in Neurobasal medium containing 2% fetal bovine serum for 6 days, essentially as previously described.62 All primary antibodies including: SOX2 #ab97959 (Abcam, Cambridge, UK), PCNA #2586, GFAP #12389, CNPASE #5664, β3-TUBULIN #5568, NESTIN #4760, NEUROFILAMENT-L #2837 (Cell Signaling, Danvers, MA, USA) were used according to their manufacturer’s instructions. For analyses by flow cytometry, cell suspensions from SCs or tumor tissues were incubated in Cytofix/Cytoperm solution (Becton & Dickinson, Franklin Lakes, NJ, USA) prior to staining with the corresponding fluorescent-conjugated antibody: SOX2 #656103, PCNA #307909 (BioLegend, San Diego, CA, USA), and CLEAVED CASPASE-3 #9669 (Cell Signaling). Fluorescence activated cell sorting (FACS) data were acquired using a BD CANTO II FACS (Becton & Dickinson) and analyzed using DiVa6 software.
Cell and molecular biology
Plasmids expressing a dominant-negative TCF3 construct (dnTCF3),63 DKK1 (Addgene, Cambridge, CA, USA), Trp53 (Takara, Mountain View, CA, USA), a dominant-negative P53 (dnTrp53) (Takara) or siRNA smart pools (Dharmacon, Lafayette, LA, USA) were transfected into SCs using Lipofectamine 2000 (Invitrogen). Cell viability was monitored by the reduction of 3-(4,5-dimethyl-2-thiazolyl) 2,5-diphenyl-2H-tetrazolium bromide to formazan,64 or by trypan blue exclusion when evaluating the number of viable cells prior to sphere formation assays, FACS analysis or engraftment in vivo. Secondary sphere formation was determined using equal numbers of viable cells, from treated primary SCs, to form spheres in 8 days. Total RNA was Trizol (Invitrogen) extracted and the expression of the indicated genes determined using quantitative real-time PCR (RT–qPCR) and Taqman probes (Invitrogen), followed by normalization to the expression of Gapdh.65 The efficacy of WNT antagonists was validated using a mycoplasma tested cell line and containing a TCF/LEF luciferase reporter gene, HEK 293 STF (ATCC, Manassas, VA, USA), treated with 10 mm LiCl and 10 mg/ml WNT3a, and normalized to protein concentration. For Ctnnb1 sequencing cDNA was prepared from total RNA extracted from SCs-1, 2, 3. The region of β-Catenin (Ctnnb1) encoding exon 3 was sequenced and compared to WT. Primer sequences used were Ctnnb1 Fw: 5′-CGC TGCGTGGACAATGGCTA-3′ and Ctnnb1 Rv: 5′-CGTGTGGCAAGTTCCGCGTC-3′. Sequence alignment was performed using Ape software.
Mouse work
All mouse work was conducted in accordance with protocols approved by the Institutional Animal Care and Use Committee at the University of Miami. Ptch1tm1Mps/J66 and B6.129S2-Trp53tm1Tyj/J67 mice (Jackson laboratory, Bar Harbor, ME, USA) were mated to generate a breeding colony. Spontaneous tumors Ptch1−/−;Trp53+/+ or Ptch1+/− ;Trp53+/− were expanded and maintained in 6-week-old male CD1-Foxn1nu mice as allografts (Charles River Laboratories, Worcester, MA, USA). For orthotopic studies, 100 000 viable cells were resuspended in 3 µl final volume and implanted into the cerebella of 6-week-old male CD1-Foxn1nu mice using the stereotaxic coordinates 2 mm posterior to lambda, 2 mm lateral to the middle line and 2 mm deep. Treatment was started 2 days after implantation. The same coordinates and final volume were used to implant 10 000 viable cells for symptom-free survival studies. For flank treatments, 1 000 000 viable cells were subcutaneously implanted into 6-week-old male CD1-Foxn1nu mice and treatment started once the tumors reached a size of approximately 200 mm3. Mouse tissues were fixed in formalin prior to be stained. For in vivo engraftment studies, the indicated numbers of viable cells from residual tumors were implanted into the flanks of 6-week-old male CD1-Foxn1nu mice. For all in vivo studies, 50 µl of 50 mg/kg vismodegib (Selleckchem, Houston, TX, USA) or 10 mg/kg carnosate (Enzo, Farmingdale, NY, USA) both dissolved in dimethyl sulfoxide and were administrated daily via intra-peritoneal injection.
Statistical analysis
For our ex vivo analyses, the results shown represent the mean of at least three independent experiments ± s.e.m. For in vivo analyses, the results shown represent the mean and s.e.m. of at least six mice. For IHC quantification, the results shown represent the mean and s.e.m. of at least six fields from three different tumors. Sample sizes were chosen based on similar well-characterized experiments to ensure adequate power to detect a pre-specified effect size. Mice were randomly distributed to different treatments and the investigator blinded to the assignment until final tumor analyses. For symptom-free survival analysis, those mice that did not show tumors as per hematoxylin eosin staining 60 days after experiment initiation were excluded from the analyses. Significance in multiple group comparisons was determined using a one-way analysis of variance followed by a post-hoc Student–Newman–Keuls analysis. Two sample analyses were determined using a one-tailed Student’s t-test. Data generated showed normal distribution with similar variances, and analysis was completed assuming equal variances. Statistical significance was reached when P < 0.05. The significance of tumor frequency was determined using a χ2 test, and statistically significance determined when α < 0.05. Survival significance was calculated using a Log-rank (Mantel–Cox) test and statistically significance reached when P < 0.05.
Supplementary Material
Acknowledgments
We would like to thank members of the Robbins, Capobianco and Pei laboratories for providing their insights during discussions regarding this manuscript. Also, we would like to acknowledge the skilled assistance of the Sylvester Cancer Center Flow Cytometry Core. Funding: Alex Lemonade Stand Foundation M1201547 (DJR), 1R21NS096502-01 (DJR), The University of Miami Women’s Cancer Association (DJR), B*Cured 2016 (DJR), Childhood Brain Tumor Foundation (JR-B), FICYT POST10- 27 (JR-B), funds from the Sylvester Cancer Center (DJR), NIH R01GM081635 (EL), and R01GM103926 (EL).
Footnotes
CONFLICT OF INTEREST
The authors declare no conflict of interest.
AUTHOR CONTRIBUTIONS
DJR and JR-B conceived and designed the experiments. JR-B performed the experiments. LP, CP and BL contributed to mouse work. JR-B and DJR analyzed the data. JL and EL contributed reagents/materials/analytic tools. VM, CR, DMN, PR, EL, WAW and AJC gave technical support and conceptual advice. NM provided pathological insights. DJR and JR-B wrote the paper.
Supplementary Information accompanies this paper on the Oncogene website (http://www.nature.com/onc)
References
- 1.Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114:97–109. doi: 10.1007/s00401-007-0243-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Crawford JR, MacDonald TJ, Packer RJ. Medulloblastoma in childhood: new biological advances. Lancet Neurol. 2007;6:1073–1085. doi: 10.1016/S1474-4422(07)70289-2. [DOI] [PubMed] [Google Scholar]
- 3.Gibson P, Tong Y, Robinson G, Thompson MC, Currle DS, Eden C, et al. Subtypes of medulloblastoma have distinct developmental origins. Nature. 2010;468:1095–1099. doi: 10.1038/nature09587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Northcott Pa, Dubuc AM, Pfister S, Taylor MD. Molecular subgroups of medulloblastoma. Expert Rev Neurother. 2012;12:871–884. doi: 10.1586/ern.12.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Remke M, Ramaswamy V, Taylor MD. Medulloblastoma molecular dissection: the way toward targeted therapy. Curr Opin Oncol. 2013;25:674–681. doi: 10.1097/CCO.0000000000000008. [DOI] [PubMed] [Google Scholar]
- 6.Olson JM. Therapeutic opportunities for medulloblastoma come of age. Cancer Cell. 2014;25:267–269. doi: 10.1016/j.ccr.2014.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Atwood SX, Whitson RJ, Oro AE. Advanced treatment for basal cell carcinomas. Cold Spring Harb Perspect Med. 2014;4:a013581. doi: 10.1101/cshperspect.a013581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johnson RL, Rothman aL, Xie J, Goodrich LV, Bare JW, Bonifas JM, et al. Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science. 1996;272:1668–1671. doi: 10.1126/science.272.5268.1668. [DOI] [PubMed] [Google Scholar]
- 9.Aszterbaum M, Rothman a, Johnson RL, Fisher M, Xie J, Bonifas JM, et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol. 1998;110:885–888. doi: 10.1046/j.1523-1747.1998.00222.x. [DOI] [PubMed] [Google Scholar]
- 10.Ng JMY, Curran T. The Hedgehog’s tale: developing strategies for targeting cancer. Nat Rev Cancer. 2011;11:493–501. doi: 10.1038/nrc3079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rudin CM, Hann CL, Laterra J, Yauch RL, Callahan Ca, Fu L, et al. Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. N Engl J Med. 2009;361:1173–1178. doi: 10.1056/NEJMoa0902903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.LoRusso PM, Rudin CM, Reddy JC, Tibes R, Weiss GJ, Borad MJ, et al. Phase I trial of hedgehog pathway inhibitor vismodegib (GDC-0449) in patients with refractory, locally advanced or metastatic solid tumors. Clin Cancer Res. 2011;17:2502–2511. doi: 10.1158/1078-0432.CCR-10-2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Robinson GW, Orr BA, Wu G, Gururangan S, Lin T, Qaddoumi I, et al. Vismodegib exerts targeted efficacy against recurrent sonic hedgehog-subgroup medulloblastoma: results from phase II Pediatric Brain Tumor Consortium Studies PBTC-025B and PBTC-032. J Clin Oncol. 2015;33:2646–2654. doi: 10.1200/JCO.2014.60.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Heppner GH. Tumor heterogeneity. Cancer Res. 1984;44:2259–2265. [PubMed] [Google Scholar]
- 15.Marusyk A, Polyak K. Tumor heterogeneity: causes and consequences. Biochim Biophys Acta. 2010;1805:105–117. doi: 10.1016/j.bbcan.2009.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Boman BM, Wicha MS. Cancer stem cells: a step toward the cure. J Clin Oncol. 2008;26:2795–2799. doi: 10.1200/JCO.2008.17.7436. [DOI] [PubMed] [Google Scholar]
- 17.Dean M, Fojo T, Bates S. Tumour stem cells and drug resistance. Nat Rev Cancer. 2005;5:275–284. doi: 10.1038/nrc1590. [DOI] [PubMed] [Google Scholar]
- 18.Yauch RL, Dijkgraaf GJ, Alicke B, Januario T, Ahn CP, Holcomb T, et al. Smoothened mutation confers resistance to a Hedgehog pathway inhibitor in medulloblastoma. Science. 2009;326:572–574. doi: 10.1126/science.1179386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wolfe CM, Green WH, Cognetta AB, Hatfield HK. Basal cell carcinoma rebound after cessation of vismodegib in a nevoid basal cell carcinoma syndrome patient. Dermatol Surg. 2012;38:1863–1866. doi: 10.1111/j.1524-4725.2012.02513.x. [DOI] [PubMed] [Google Scholar]
- 20.Sekulic A, Migden MR, Oro AE, Dirix L, Lewis KD, Hainsworth JD, et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med. 2012;366:2171–2179. doi: 10.1056/NEJMoa1113713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tang JY, Mackay-Wiggan JM, Aszterbaum M, Yauch RL, Lindgren J, Chang K, et al. Inhibiting the hedgehog pathway in patients with the basal-cell nevus syndrome. N Engl J Med. 2012;366:2180–2188. doi: 10.1056/NEJMoa1113538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Meani RE, Lim S-W, Chang ALS, Kelly JW. Emergence of chemoresistance in a metastatic basal cell carcinoma patient after complete response to hedgehog pathway inhibitor vismodegib (GDC-0449) Australas J Dermatol. 2014;55:218–221. doi: 10.1111/ajd.12196. [DOI] [PubMed] [Google Scholar]
- 23.Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63:5821–5828. [PubMed] [Google Scholar]
- 24.Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, et al. Identification of human brain tumour initiating cells. Nature. 2004;432:396–401. doi: 10.1038/nature03128. [DOI] [PubMed] [Google Scholar]
- 25.Ward RJ, Lee L, Graham K, Satkunendran T, Yoshikawa K, Ling E, et al. Multipotent CD15+ cancer stem cells in Patched-1—deficient mouse medulloblastoma. Cancer Res. 2009:4682–4690. doi: 10.1158/0008-5472.CAN-09-0342. [DOI] [PubMed] [Google Scholar]
- 26.Read T-A, Fogarty MP, Markant SL, McLendon RE, Wei Z, Ellison DW, et al. Identification of CD15 as a marker for tumor-propagating cells in a mouse model of medulloblastoma. Cancer Cell. 2009;15:135–147. doi: 10.1016/j.ccr.2008.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Corno D, Pala M, Cominelli M, Cipelletti B, Leto K, Croci L, et al. Gene signatures associated with mouse postnatal hindbrain neural stem cells and medulloblastoma cancer stem cells identify novel molecular mediators and predict human medulloblastoma molecular classification. Cancer Discov. 2012;2:554–568. doi: 10.1158/2159-8290.CD-11-0199. [DOI] [PubMed] [Google Scholar]
- 28.Vanner RJ, Remke M, Gallo M, Selvadurai HJ, Coutinho F, Lee L, et al. Quiescent sox2(+) cells drive hierarchical growth and relapse in sonic hedgehog subgroup medulloblastoma. Cancer Cell. 2014;26:33–47. doi: 10.1016/j.ccr.2014.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ahlfeld J, Favaro R, Pagella P, Kretzschmar HA, Nicolis S, Schüller U. Sox2 requirement in sonic hedgehog-associated medulloblastoma. Cancer Res. 2013;73:3796–3807. doi: 10.1158/0008-5472.CAN-13-0238. [DOI] [PubMed] [Google Scholar]
- 30.Huang X, Ketova T, Litingtung Y, Chiang C. Isolation, enrichment, and maintenance of medulloblastoma stem cells. J Vis Exp. 2010;43:2086. doi: 10.3791/2086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nusse R. Wnt signaling and stem cell control. Cell Res. 2008;18:523–527. doi: 10.1038/cr.2008.47. [DOI] [PubMed] [Google Scholar]
- 32.Koch A, Waha A, Tonn JC, Sörensen N, Berthold F, Wolter M, et al. Somatic mutations of WNT/wingless signaling pathway components in primitive neuroectodermal tumors. Int J Cancer. 2001;93:445–449. doi: 10.1002/ijc.1342. [DOI] [PubMed] [Google Scholar]
- 33.Thompson MC, Fuller C, Hogg TL, Dalton J, Finkelstein D, Lau CC, et al. Genomics identifies medulloblastoma subgroups that are enriched for specific genetic alterations. J Clin Oncol. 2006;24:1924–1931. doi: 10.1200/JCO.2005.04.4974. [DOI] [PubMed] [Google Scholar]
- 34.Kemler R. From cadherins to catenins: cytoplasmic protein interactions and regulation of cell adhesion. Trends Genet. 1993;9:317–321. doi: 10.1016/0168-9525(93)90250-l. [DOI] [PubMed] [Google Scholar]
- 35.Waterman ML. Lymphoid enhancer factor/T cell factor expression in colorectal cancer. Cancer Metastasis Rev. 2004;23:41–52. doi: 10.1023/a:1025858928620. [DOI] [PubMed] [Google Scholar]
- 36.Lewis SL, Khoo P-L, De Young RA, Steiner K, Wilcock C, Mukhopadhyay M, et al. Dkk1 and Wnt3 interact to control head morphogenesis in the mouse. Development. 2008;135:1791–1801. doi: 10.1242/dev.018853. [DOI] [PubMed] [Google Scholar]
- 37.Klein PS, Melton DA. A molecular mechanism for the effect of lithium on development. Proc Natl Acad Sci USA. 1996;93:8455–8459. doi: 10.1073/pnas.93.16.8455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liu J, Pan S, Hsieh MH, Ng N, Sun F, Wang T, et al. Targeting Wnt-driven cancer through the inhibition of Porcupine by LGK974. Proc Natl Acad Sci USA. 2013;110:20224–20229. doi: 10.1073/pnas.1314239110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.de la Roche M, Rutherford TJ, Gupta D, Veprintsev DB, Saxty B, Freund SM, et al. An intrinsically labile α-helix abutting the BCL9-binding site of β-catenin is required for its inhibition by carnosic acid. Nat Commun. 2012;3:680. doi: 10.1038/ncomms1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ewan K, Pajak B, Stubbs M, Todd H, Barbeau O, Quevedo C, et al. A useful approach to identify novel small-molecule inhibitors of Wnt-dependent transcription. Cancer Res. 2010;70:5963–5973. doi: 10.1158/0008-5472.CAN-10-1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Eguchi M, Nguyen C, Lee SC, Kahn M. ICG-001, a novel small molecule regulator of TCF/beta-catenin transcription. Med Chem. 2005;1:467–472. doi: 10.2174/1573406054864098. [DOI] [PubMed] [Google Scholar]
- 42.Gandhirajan RK, Staib PA, Minke K, Gehrke I, Plickert G, Schlösser A, et al. Small molecule inhibitors of Wnt/beta-catenin/lef-1 signaling induces apoptosis in chronic lymphocytic leukemia cells in vitro and in vivo. Neoplasia. 2010;12:326–335. doi: 10.1593/neo.91972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Anne SL, Govek E-E, Ayrault O, Kim JH, Zhu X, Murphy DA, et al. WNT3 inhibits cerebellar granule neuron progenitor proliferation and medulloblastoma formation via MAPK activation. PLoS ONE. 2013;8:e81769. doi: 10.1371/journal.pone.0081769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zinke J, Schneider FT, Harter PN, Thom S, Ziegler N, Toftgård R, et al. β-Catenin-Gli1 interaction regulates proliferation and tumor growth in medulloblastoma. Mol Cancer. 2015;14:17. doi: 10.1186/s12943-015-0294-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pöschl J, Bartels M, Ohli J, Bianchi E, Kuteykin-Teplyakov K, Grammel D, et al. Wnt/β-catenin signaling inhibits the Shh pathway and impairs tumor growth in Shh-dependent medulloblastoma. Acta Neuropathol. 2014;127:605–607. doi: 10.1007/s00401-014-1258-2. [DOI] [PubMed] [Google Scholar]
- 46.Azad N, Rasoolijazi H, Joghataie MT, Soleimani S. Neuroprotective effects of carnosic Acid in an experimental model of Alzheimer’s disease in rats. Cell J. 2011;13:39–44. [PMC free article] [PubMed] [Google Scholar]
- 47.Piccin D, Morshead CM. Wnt signaling regulates symmetry of division of neural stem cells in the adult brain and in response to injury. Stem Cells. 2011;29:528–538. doi: 10.1002/stem.589. [DOI] [PubMed] [Google Scholar]
- 48.Lange C, Mix E, Rateitschak K, Rolfs A. Wnt signal pathways and neural stem cell differentiation. Neurodegener Dis. 2006;3:76–86. doi: 10.1159/000092097. [DOI] [PubMed] [Google Scholar]
- 49.Fan X, Eberhart CG. Medulloblastoma stem cells. J Clin Oncol. 2008;26:2821–2827. doi: 10.1200/JCO.2007.15.2264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhao X, Ponomaryov T, Ornell KJ, Zhou P, Dabral SK, Pak E, et al. RAS/MAPK activation drives resistance to smo inhibition, metastasis, and tumor evolution in Shh pathway-dependent tumors. Cancer Res. 2015;75:3623–3635. doi: 10.1158/0008-5472.CAN-14-2999-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cha YH, Kim NH, Park C, Lee I, Kim HS, Yook JI. MiRNA-34 intrinsically links p53 tumor suppressor and Wnt signaling. Cell Cycle. 2012;11:1273–1281. doi: 10.4161/cc.19618. [DOI] [PubMed] [Google Scholar]
- 52.Kim NH, Kim HS, Kim N-G, Lee I, Choi H-S, Li X-Y, et al. p53 and miRNA-34 are Suppressors of Canonical Wnt Signaling. Sci Signal. 2011;4:ra71. doi: 10.1126/scisignal.2001744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Voorneveld PW, Kodach LL, Jacobs RJ, van Noesel CJM, Peppelenbosch MP, Korkmaz KS, et al. The BMP pathway either enhances or inhibits the Wnt pathway depending on the SMAD4 and p53 status in CRC. Br J Cancer. 2015;112:122–130. doi: 10.1038/bjc.2014.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhukova N, Ramaswamy V, Remke M, Pfaff E, Shih DJH, Martin DC, et al. Subgroup-specific prognostic implications of TP53 mutation in medulloblastoma. J Clin Oncol. 2013;31:2927–2935. doi: 10.1200/JCO.2012.48.5052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tabori U, Baskin B, Shago M, Alon N, Taylor MD, Ray PN, et al. Universal poor survival in children with medulloblastoma harboring somatic TP53 mutations. J Clin Oncol. 2010;28:1345–1350. doi: 10.1200/JCO.2009.23.5952. [DOI] [PubMed] [Google Scholar]
- 56.Kool M, Jones DTW, Jäger N, Northcott PA, Pugh TJ, Hovestadt V, et al. Genome sequencing of SHH medulloblastoma predicts genotype-related response to smoothened inhibition. Cancer Cell. 2014;25:393–405. doi: 10.1016/j.ccr.2014.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yang SH, Andl T, Grachtchouk V, Wang A, Liu J, Syu L-J, et al. Pathological responses to oncogenic Hedgehog signaling in skin are dependent on canonical Wnt/beta3-catenin signaling. Nat Genet. 2008;40:1130–1135. doi: 10.1038/ng.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Youssef KK, Lapouge G, Bouvrée K, Rorive S, Brohée S, Appelstein O, et al. Adult interfollicular tumour-initiating cells are reprogrammed into an embryonic hair follicle progenitor-like fate during basal cell carcinoma initiation. Nat Cell Biol. 2012;14:1282–1294. doi: 10.1038/ncb2628. [DOI] [PubMed] [Google Scholar]
- 59.Tam WL, Ng HH. Sox2: masterminding the root of cancer. Cancer Cell. 2014;26:3–5. doi: 10.1016/j.ccr.2014.06.024. [DOI] [PubMed] [Google Scholar]
- 60.Kenney AM, Rowitch DH. Sonic hedgehog promotes G1 cyclin expression and sustained cell cycle progression in mammalian neuronal precursors. Mol Cell Biol. 2000;20:9055–9067. doi: 10.1128/mcb.20.23.9055-9067.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lee HY, Greene LA, Mason CA, Manzini MC. Isolation and culture of post-natal mouse cerebellar granule neuron progenitor cells and neurons. J Vis Exp. 2009;23:990. doi: 10.3791/990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gritti A, Parati E, Cova L, Frolichsthal P, Galli R, Wanke E, et al. Multipotential stem cells from the adult mouse brain proliferate and self-renew in response to basic fibroblast growth factor. J Neurosci. 1996;16:1091–1100. doi: 10.1523/JNEUROSCI.16-03-01091.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kennedy MWL, Kao KR. Xrel3/XrelA attenuates β-catenin-mediated transcription during mesoderm formation in Xenopus embryos. Biochem J. 2011;435:247–257. doi: 10.1042/BJ20101801. [DOI] [PubMed] [Google Scholar]
- 64.Rodriguez-Blanco J, Martín V, Herrera F, García-Santos G, Antolín I, Rodriguez C. Intracellular signaling pathways involved in post-mitotic dopaminergic PC12 cell death induced by 6-hydroxydopamine. J Neurochem. 2008;107:127–140. doi: 10.1111/j.1471-4159.2008.05588.x. [DOI] [PubMed] [Google Scholar]
- 65.Fei DL, Li H, Kozul CD, Black KE, Singh S, Gosse JA, et al. Activation of Hedgehog signaling by the environmental toxicant arsenic may contribute to the etiology of arsenic-induced tumors. Cancer Res. 2010;70:1981–1988. doi: 10.1158/0008-5472.CAN-09-2898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Goodrich LV, Milenković L, Higgins KM, Scott MP. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science. 1997;277:1109–1113. doi: 10.1126/science.277.5329.1109. [DOI] [PubMed] [Google Scholar]
- 67.Wetmore C, Eberhart DE, Curran T. Loss of p53 but not ARF accelerates medulloblastoma in mice heterozygous for patched. Cancer Res. 2001;61:513–516. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.