Abstract
Background
Peripheral artery disease (PAD) is associated with increased cardiovascular mortality, and PAD risk factors overlap with those for aortic stenosis (AS). The prevalence of and outcomes associated with PAD in a population undergoing transcatheter aortic valve replacement (TAVR) is unknown.
Methods and Results
Using the STS/TVT Registry linked to Medicare claims data, we identified patients ≥ 65 years old undergoing TAVR from 2011-2015. We calculated hazard ratios (HR) for 1-year adverse outcomes, including mortality, readmission, and bleeding, for patients with PAD compared with those without, adjusting for baseline characteristics and post-procedure medications. Analyses were performed separately by access site (transfemoral [TF] and non-TF). Of 19,660 patients undergoing TF TAVR, 4,810 (24.5%) had PAD; 3,730 (47.9%) of 7,780 patients undergoing non-TF TAVR had PAD. In both groups, patients with PAD were significantly more likely to have coronary and carotid artery disease. At 1-year follow-up, patients with PAD undergoing TAVR via TF access had a higher incidence of death (16.8 vs. 14.4%; adjusted HR 1.14, p = 0.01), readmission (45.5 vs, 42.1%; HR 1.11, p < 0.001), and bleeding (23.1 vs. 19.7%; HR 1.18, p < 0.001) compared with patients without PAD. Patients with PAD undergoing TAVR via non-TF access did not have significantly higher rates of 1-year mortality or readmission compared with patients without PAD.
Conclusions
PAD is common among patients undergoing commercial TAVR via TF and non-TF access. Among patients undergoing TF TAVR, PAD is associated with a higher incidence of 1-year adverse outcomes compared with absence of PAD.
Keywords: peripheral vascular disease, transcutaneous aortic valve implantation, outcomes research, readmission, bleeding
Introduction
Peripheral artery disease (PAD) affects > 200 million people worldwide, including > 50 million people in Europe and North America, with symptoms ranging from none to critical limb ischemia.1 Its prevalence is known to increase in those over the age of 50, especially in those with other vascular risk factors, including diabetes, hyperlipidemia, smoking, and chronic renal insufficiency.2 These risk factors overlap with those of aortic stenosis.3
Given the shared risk factors, PAD is a frequent comorbidity in patients referred for transcatheter aortic valve replacement (TAVR).4 In clinical trials, the prevalence of PAD in patients undergoing TAVR ranged from 27.8% in the PARTNER B (prohibitive risk) trial to 41.3% in the CoreValve U.S. study.5, 6 However, the population studied in clinical trials may differ from the population undergoing commercial TAVR in important ways; commercial TAVR patients are more often male and have lower operative risk than patients enrolled in pivotal trials, for example.7, 8 Moreover, PAD has been associated with increased cardiovascular mortality and stroke risk in the general population and in patients undergoing coronary artery bypass grafting,9-11 and may therefore represent an important risk factor for stroke and mortality among patients undergoing TAVR. Among patients with acute coronary syndromes and those undergoing percutaneous coronary intervention, patients with PAD have higher rates of major procedural and non-procedural bleeding than those without PAD,12-16 but it is not known whether this extends to patients undergoing TAVR, which has a high baseline rate of vascular access complications due to the need for large-bore vascular access.17
We therefore analyzed the Society of Thoracic Surgeons/American College of Cardiology (STS/ACC) Transcatheter Valve Therapy (TVT) database to address the following aims: 1) Describe the prevalence of PAD among patients undergoing commercial TAVR and 2) Examine the relationship between baseline PAD and cardiovascular outcomes. We analyzed patients undergoing TAVR via transfemoral (TF) and non-TF access separately to avoid confounding related to access route selection.
Methods
The STS/ACC TVT registry has been described previously.18 Briefly, it was launched in December 2011 following FDA approval of the Sapien transcatheter heart valve, and was designed to be responsive to Centers for Medicare/Medicaid Services (CMS) requirements that all TAVR centers participate in a national registry. It collects detailed data on demographics, comorbidities, functional status, quality of life, hemodynamics, procedural details, and in-hospital outcomes from patients undergoing TAVR at all commercial centers in the U.S. The National Cardiovascular Data Registry and the Duke Clinical Research Institute conduct data quality checks, including data quality feedback reports to participating centers.
To facilitate collection of long-term outcomes, the STS/ACC TVT registry has previously been linked to CMS claims data provided by CMS.7 Linkage was performed by CMS using direct patient identifiers.
Patient population
Between November 2011 and September 2015, 48,160 TAVR procedures were performed at 395 centers (Figure 1). We excluded patients undergoing additional transcatheter valve procedures in addition to TAVR (n = 101), procedures with missing access site (n = 340), procedures performed for aortic insufficiency or failed bioprosthetic valves (n = 1,135), valve-in-valve procedures (n = 1,996), procedures for which PAD status was not captured on the data collection form (n = 62), patients < 65 years old (n = 1,583) or without Medicare (n = 3,651), patients for whom CMS linkage could not be performed (n = 8,318), patients who did not have at least 1 year of fee-for-service Medicare coverage prior to the procedure date (n = 3,356), and repeat admission for TAVR (n = 178). Non-TF access included patients undergoing TAVR via transapical, transaortic, transaxillary, transsubclavian, transiliac, transseptal, and transcarotid approaches. One year fee-for-service coverage prior to the procedure date was necessary to identify patients with revascularization procedures performed to facilitate access. For all analyses, patients that underwent TF access were analyzed separately from those that underwent non-TF access, and no formal comparisons were performed between the two populations because of marked differences in patient characteristics that resulted in the clinical decision to select one or the other access approach.
Figure 1. CONSORT diagram.
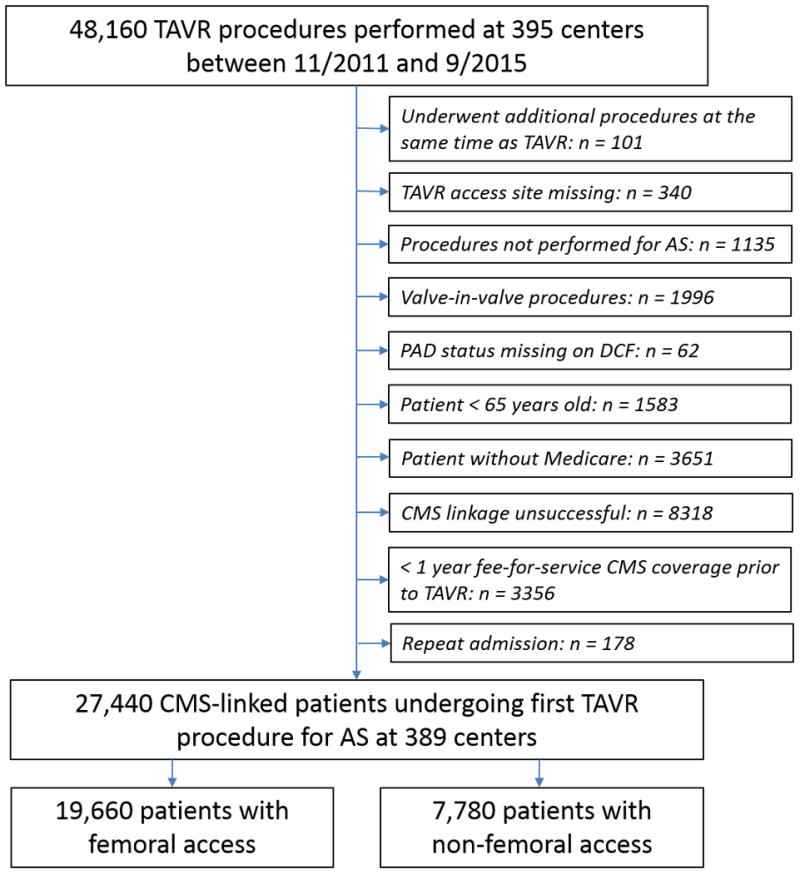
Flow chart depicting derivation of the study population from the Transcatheter Valve Therapies (TVT) registry. TAVR = transcatheter aortic valve replacement; AS = aortic stenosis; PAD = peripheral arterial disease; DCF = data collection form; CMS = Centers for Medicare and Medicaid Services
Definitions and Outcomes
According to the STS/TVT definition, PAD includes patients with aortic aneurysm with or without repair, claudication (either on exertion or at rest), positive non-invasive test (including ABI ≤ 0.9, or > 50% diameter stenosis in any peripheral artery on ultrasound, MRI, computed tomography, or angiographic imaging), prior amputation for arterial vascular insufficiency, and/or prior vascular reconstruction (excluding dialysis fistula creation and vein stripping), peripheral bypass surgery, or percutaneous peripheral vascular intervention. PAD thus includes disease of the upper and lower extremity arteries, renal arteries, mesenteric arteries, and abdominal aortic systems, but excludes disease in the carotid and cerebrovascular arteries. The TVT registry's data collection form indicates if PAD is present or absent; we used this as our indicator of PAD status. The registry does not indicate which criteria were used to make the diagnosis of PAD, or when the diagnosis of PAD was made.
To provide more detailed information on peripheral arterial procedures performed to facilitate access for TAVR, patients undergoing revascularization were identified by CPT and ICD-9 codes (Supplemental Appendix 1). Revascularization procedures performed within 90 days of TF TAVR were defined, conservatively so as to be maximally sensitive, as procedures performed to facilitate vascular access.
In-hospital outcomes were all-cause mortality, stroke, myocardial infarction (MI), bleeding, and major vascular complications. We also report rates of device success, as determined by VARC-2 criteria, and procedure completion.19, 20 One-year outcomes were all-cause mortality, all-cause readmission, stroke, myocardial infarction (MI). The stroke, MI, and bleeding outcomes included both events occurring during the index TAVR hospitalization and events leading to hospital readmission during follow-up. In-hospital outcomes were collected on the registry's data collection form; readmission events were identified from Medicare in-hospital claims data using ICD-9 codes in Supplemental Appendix 2.
Statistical Analysis
Patients undergoing TF and non-TF TAVR were grouped according to whether they had PAD as identified on the TVT data collection form. Descriptive statistics are reported as median (Q1, Q3) for continuous variables and frequency (%) for categorical variables. For continuous variables, differences between groups were compared using the Wilcoxon rank-sum test. For categorical variables, differences between groups were assessed using the Pearson Chi-square test.
To determine the association between PAD and long-term outcomes, we plotted unadjusted cumulative incidence curves for death, stroke, MI, and bleeding. Unadjusted mortality was compared for patients with and without PAD using Cox proportional hazards; for unadjusted comparisons of the incidence of readmission, stroke, MI, and bleeding by PAD status, Fine and Gray sub-distribution hazards were used to account for the competing risk of mortality.21 We subsequently used Cox proportional hazards to compare the risk-adjusted 1-year hazard ratio of all-cause death for patients with and without baseline PAD; we used Fine and Gray's proportional subdistribution hazards model, treating death as a competing risk, to compare the risk-adjusted 1-year hazards ratios of readmission, stroke, MI, and bleeding for patients with and without PAD. Covariates for risk adjustment included all variables comprising the STS/TVT TAVR in-hospital mortality risk score as well as other variables selected by expert opinon,22, 23 including demographic variables, cardiac risk factors, pre-existing cardiovascular disease, catheterization or echocardiographic-derived hemodynamic parameters, and discharge antiplatelet or anticoagulant therapy (Full list of covariates in Supplemental Appendix 3).
We performed landmark analyses for 1 year outcomes from 30 days to 1 year to determine if results were driven mainly by early procedural complications (< 30 days) or a late prognostic association (30 days to 1 year).
To further examine whether results were driven by early procedural complications or later prognostic associations, we also determined unadjusted and risk-adjusted in-hospital incidence of death, stroke, MI, bleeding, and major vascular complications, along with associated odds ratios, for patients with and without PAD. Covariates for adjustment were the same as for the one-year model, but excluded discharge antiplatelet or anticoagulant therapy.
Rates of missing data were < 2% for all variables except for carotid disease (17% in the TF cohort, 14% in the non-TF cohort), AV peak gradient (5%, 5%), AV peak velocity (8%, 9%), aspirin prescription at discharge (4%, 8%), P2Y12 inhibitor prescription at discharge (4%, 8%). Missing values were imputed using multiple imputation techniques. Specifically, missing values for categorical variables were imputed using the fully conditional method, with the discriminant function allowing all continuous and categorical variables to be predictors for imputation. Continuous variables were imputed using the predictive mean matching method, which generates imputed variables consistent with observed values. A total of five data sets were created in the imputation phase. These datasets were analyzed separately, and estimates from each imputed dataset were pooled into a single set of statistics. All statistical analyses were performed using SAS software (version 9.4, SAS Institute, Cary, NC). The Duke Clinical Research Institute conducted all analyses. The Duke University Medical Center Institutional Review Board granted a waiver of informed consent and authorization for this study.
Results
The final patient population comprised 27,440 patients undergoing TAVR at 389 centers – 19,660 via TF access and 7,780 via non-TF access (Figure 1). Characteristics for patients who could and could not be linked to CMS data are presented in Supplemental Table 1. Of 19,660 patients undergoing TAVR via TF access, 4,810 (24.5%) had PAD; of 7,780 patients undergoing TAVR via non-TF access, 3,730 (47.9%) had PAD. The majority of non-TF patients underwent apical access regardless of PAD status, 67.4% of patients with PAD and 69.0% of patients without (Table 1).
Table 1. Baseline and procedural characteristics by PAD status.
| Transfemoral access (n = 19,660) | Non-transfemoral access (n = 7780) | |||||
|---|---|---|---|---|---|---|
| Variable | No PAD (n = 14,850) |
PAD (n = 4810) |
p-value | No PAD (n = 4050) |
PAD (n = 3730) |
p-value |
| Baseline characteristics | ||||||
| Age | 85 (80, 88) | 84 (78, 88) | < 0.001 | 84 (79, 88) | 82 (77, 87) | < 0.001 |
| Male sex | 7485 (50.4) | 2981 (62.0) | < 0.001 | 1504 (37.1) | 1936 (51.9) | < 0.001 |
| Body Mass Index (kg/m2) | 27 (24,31) | 27 (24,31) | 0.84 | 26 (23, 30) | 26 (23, 30) | 0.94 |
| Hypertension | 13,061 (88.0) | 4474 (93.0) | < 0.001 | 3579 (88.4) | 3528 (94.6) | < 0.001 |
| Diabetes Mellitus | 5076 (34.2) | 1939 (40.3) | < 0.001 | 1300 (32.1) | 1428 (38.3) | < 0.001 |
| Current/Recent Smoker | 393 (2.7) | 249 (5.2) | < 0.001 | 226 (5.6) | 364 (9.8) | < 0.001 |
| Moderate to severe chronic lung disease | 3576 (24.1) | 1468 (30.5) | < 0.001 | 1085 (26.8) | 1299 (34.8) | < 0.001 |
| Prior aortic valve balloon valvuloplasty | 1600 (10.8) | 554 (11.5) | 0.15 | 523 (12.9) | 504 (13.5) | 0.38 |
| Carotid artery disease | 1861 (12.5) | 1300 (27.0) | < 0.001 | 646 (16.0) | 1389 (37.2) | < 0.001 |
| Prior stroke or TIA | 2507 (16.9) | 1144 (23.8) | < 0.001 | 720 (17.8) | 872 (23.4) | < 0.001 |
| Coronary artery disease | 7458 (50.2) | 3151 (65.5) | < 0.001 | 2216 (54.7) | 2640 (70.8) | < 0.001 |
| Prior CABG | 3436 (23.1) | 1779 (37.0) | < 0.001 | 1086 (26.8) | 1583 (42.4) | < 0.001 |
| Prior PCI | 4844 (32.6) | 2021 (42.0) | < 0.001 | 1391 (34.4) | 1611 (43.2) | < 0.001 |
| Chronic kidney disease* | 7252 (48.8) | 2543 (52.9) | < 0.001 | 2066 (51.0) | 2048 (54.9) | < 0.001 |
| Currently on dialysis | 485 (3.3) | 239 (5.0) | < 0.001 | 136 (3.4) | 183 (4.9) | < 0.001 |
| NYHA class III or IV HF within 2 weeks | 11,872 (80.0) | 4021 (83.6) | < 0.001 | 3220 (79.5) | 3066 (82.2) | 0.008 |
| Porcelain aorta | 555 (3.7) | 257 (5.3) | < 0.001 | 308 (7.6) | 451 (12.1) | < 0.001 |
| Atrial fibrillation/flutter | 6307 (42.5) | 2095 (43.6) | 0.20 | 1663 (41.1) | 1562 (41.9) | 0.47 |
| LVEF (%) | 58 (48, 65) | 56 (45, 63) | < 0.001 | 58 (50, 65) | 56 (45, 63) | < 0.001 |
| AV Area (cm2) | 0.68 (0.53, 0.80) | 0.69 (0.55, 0.80) | 0.03 | 0.65 (0.50, 0.78) | 0.66 (0.52, 0.80) | 0.001 |
| AV Peak Gradient (mmHg) | 70 (59, 85) | 69 (56, 82) | < 0.001 | 70 (58, 85) | 69 (56, 81) | < 0.001 |
| AV Peak Velocity (m/s) | 4.2 (3.8, 4.6) | 4.1 (3.8, 4.5) | < 0.001 | 4.2 (3.8, 4.6) | 4.1 (3.8, 4.5) | < 0.001 |
| Moderate or severe aortic insufficiency | 2668 (18.0) | 867 (18.0) | 0.94 | 806 (19.9) | 757 (20.3) | 0.67 |
| Moderate or severe mitral insufficiency | 4439 (36.1) | 1452 (35.2) | 0.27 | 1231 (37.2) | 1125 (35.0) | 0.07 |
| Procedural characteristics | ||||||
| Procedure status | 0.10 | 0.02 | ||||
| Urgent | 1198 (8.1) | 430 (8.9) | 393 (9.7) | 353 (9.5) | ||
| Elective | 13,611 (91.6) | 4366 (90.8) | 3647 (90.1) | 3372 (90.4) | ||
| Mechanical assist device in place | 80 (0.5) | 30 (0.6) | 0.56 | 42 (1.1) | 45 (1.2) | 0.81 |
| Cardiopulmonary bypass used | 228 (1.5) | 78 (1.6) | 0.67 | 205 (5.1) | 169 (4.5) | 0.27 |
| Type of anesthesia | 0.03 | 0.11 | ||||
| Moderate sedation | 1573 (10.6) | 441 (9.2) | 14 (0.4) | 22 (0.6) | ||
| General anesthesia | 13,177 (88.7) | 4341 (90.3) | 4021 (99.3) | 3690 (98.9) | ||
| Other | 70 (0.5) | 19 (0.4) | 7 (0.2) | 13 (0.4) | ||
| Sheath access site | -- | < 0.001 | ||||
| Transfemoral | 14,850 (100) | 4810 (100) | -- | -- | ||
| Transapical | -- | -- | 2796 (69.0) | 2512 (67.4) | ||
| Transaortic | -- | -- | 881 (21.8) | 873 (23.4) | ||
| Transsubclavian/Transaxillary | -- | -- | 155 (3.8) | 198 (5.3) | ||
| Other‡ | -- | -- | 92 (2.3) | 75 (2.0) | ||
| Sheath access method | < 0.001 | 0.03 | ||||
| Percutaneous | 9174 (61.8) | 2935 (61.0) | 51 (1.3) | 56 (1.5) | ||
| Cut-down | 5641 (38.0) | 1841 (38.3) | 430 (10.6) | 361 (9.7) | ||
| Mini-thoracotomy | -- | -- | 2811 (69.4) | 2668 (71.5) | ||
| Mini-sternotomy | -- | -- | 691 (17.1) | 606 (16.3) | ||
| Other | 19 (0.1) | 28 (0.6) | 61 (1.5) | 32 (0.9) | ||
| Sheath size | 18 (18, 22) | 18 (18, 22) | < 0.001 | 24 (22, 26) | 24 (22, 26) | 0.01 |
| Valve type | < 0.001 | < 0.001 | ||||
| Self expanding | 3816 (25.7) | 1523 (31.7) | 242 (6.0) | 338 (9.1) | ||
| Balloon expandable | 10,833 (73.0) | 3185 (66.2) | 3768 (93.0) | 3362 (90.1) | ||
| Smallest valve size | < 0.001 | < 0.001 | ||||
| 23 mm | 4208 (28.3) | 1063 (22.1) | 1732 (42.8) | 1289 (34.6) | ||
| 26 mm | 5979 (40.3) | 1894 (39.4) | 1901 (46.9) | 1861 (49.9) | ||
| 29 mm | 3182 (21.4) | 1184 (24.6) | 310 (7.7) | 446 (12.0) | ||
| 31 mm | 1243 (8.4) | 560 (11.6) | 68 (1.7) | 103 (2.8) | ||
| Discharge Antithrombotic therapy | ||||||
| Antithrombotic strategy | 0.03 | 0.29 | ||||
| None | 770 (5.2) | 298 (6.2) | 403 (10.0) | 363 (9.7) | ||
| Anticoagulants alone | 507 (3.4) | 145 (3.0) | 123 (3.0) | 88 (2.4) | ||
| Antiplatelets alone | 9825 (66.2) | 3141 (65.3) | 2494 (61.6) | 2305 (61.8) | ||
| Both | 3735 (25.2) | 1219 (25.4) | 1027 (25.4) | 971 (26.0) | ||
| Antiplatelet therapy | ||||||
| Aspirin | 12,448 (83.8) | 4005 (83.2) | 0.43 | 3286 (81.1) | 3048 (81.7) | 0.32 |
| P2Y12 inhibitor | 9335 (62.9) | 3054 (63.5) | 0.07 | 2260 (55.8) | 2146 (57.5) | 0.09 |
Creatinine Clearance < 60 mL/kg/m2 or on dialysis;
transiliac, transseptal, transcarotid; TIA, transient ischemic attack; CABG, coronary artery bypass graft surgery; PCI, percutaneous coronary intervention; NYHA, New York Heart Association; LVEF, left ventricular ejection fraction; AV, aortic valve
In both access groups, the cohort with PAD was younger, more likely to be male, and had a significantly greater prevalence of hypertension, diabetes mellitus, coronary artery and cerebrovascular disease, prior stroke, and heart failure within the 2 weeks prior to TAVR. In both the TF and non-TF access groups, patients with PAD more frequently were treated with larger sized valves. There were no clinically relevant differences in discharge antithrombotic therapy between patients with and without PAD, regardless of access site.
Overall, among patients with PAD undergoing TF TAVR, peripheral vascular interventions in the 90 days prior to TAVR were performed in 381 patients (7.9%).
Outcomes in patients undergoing TF TAVR
Among patients with PAD undergoing TAVR via TF access, 807 (16.8%) died over 1-year follow-up, compared with 2,143 (14.4%) without PAD (unadjusted HR 1.22; 95% CI 1.12-1.33; p < 0.001). Compared with patients without PAD, patients with PAD also had higher unadjusted rates of hospital readmission, MI, and major bleeding over 1-year follow-up (p < 0.001 for all comparisons). Risk of stroke was not significantly greater in patients with PAD than in those without (p = 0.31) (Figure 2). After adjustment for baseline characteristics, the associations between PAD and 1-year outcomes were attenuated, but PAD remained significantly associated with a higher rate of all-cause mortality, readmission, and bleeding (adjusted HR for mortality, PAD vs. no PAD, 1.14; 95% CI 1.03-1.25; p = 0.008) (Table 2).
Figure 2. Cumulative incidence of outcomes over time among patients undergoing TAVR via femoral access, stratified by PAD status.
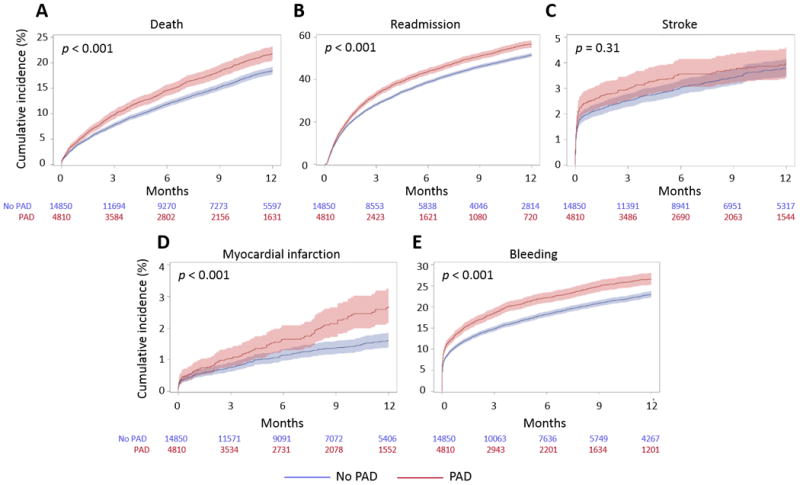
Kaplan-Meier curve depicts unadjusted all-cause mortality (A); cumulative incidence curves depict unadjusted readmission (B), stroke (C), myocardial infarction (D), and major bleeding (E).
Table 2. 1-year outcomes by PAD status.
| Transfemoral access (n = 19,689) | ||||||
|---|---|---|---|---|---|---|
| Unadjusted | Risk-adjusted | |||||
| Outcome | No PAD (n = 14,865) |
PAD (n = 4824) |
HR (95% CI) | p-value | HR (95% CI) | p-value |
| Death | 2143 (14.4) | 807 (16.8) | 1.22 (1.12-1.33) | < 0.001 | 1.14 (1.03-1.25) | 0.008 |
| Readmission | 6256 (42.1) | 2188 (45.5) | 1.16 (1.11-1.22) | < 0.001 | 1.11 (1.06-1.17) | < 0.001 |
| Myocardial infarction | 192 (1.3) | 96 (2.0) | 1.59 (1.27-1.99) | < 0.001 | 1.24 (0.98-1.56) | 0.07 |
| Stroke | 490 (3.3) | 171 (3.5) | 1.09 (0.92-1.30) | 0.31 | 1.07 (0.90-1.27) | 0.43 |
| Any bleed | 2927 (19.7) | 1109 (23.1) | 1.22 (1.13-1.31) | < 0.001 | 1.18 (1.09-1.27) | < 0.001 |
| Non-transfemoral access (n = 7780) | ||||||
| Unadjusted | Risk-adjusted | |||||
| Outcome |
No PAD (n = 4050) |
PAD (n = 3730) |
HR (95% CI) | p-value | HR (95% CI) | p-value |
| Death | 986 (24.4) | 933 (25.0) | 1.07 (0.98-1.17) | 0.13 | 1.03 (0.93-1.14) | 0.61 |
| Readmission | 2118 (52.3) | 1972 (52.9) | 1.05 (0.98-1.12) | 0.14 | 1.03 (0.97-1.10) | 0.33 |
| Myocardial infarction | 92 (2.3) | 99 (2.7) | 1.22(0.90-1.64) | 0.20 | 1.05 (0.77-1.42) | 0.76 |
| Stroke | 170 (4.2) | 169 (4.5) | 1.10 (0.89-1.36) | 0.37 | 1.15 (0.91-1.45) | 0.24 |
| Any bleed | 968 (23.9) | 1019 (27.3) | 1.20(1.08-1.32) | < 0.001 | 1.22 (1.11-1.36) | < 0.001 |
For mortality and readmission, the excess hazard associated with PAD was evident in the first 30 days and from 30 days to 1 year on unadjusted and risk-adjusted landmark analyses (Supplemental Figure S1). By contrast, for stroke and bleeding, the excess hazard was seen almost entirely in the peri-procedural time period. Unadjusted and risk-adjusted in-hospital rates of bleeding and stroke were significantly greater for patients with PAD than those without PAD (Table 3). In unadjusted and risk-adjusted landmark analyses, patients with PAD had a significantly higher rate of bleeding than those without PAD in the 30 days following the procedure (p < 0.001) and from 30 days to 1 year (p = 0.009), but the association between PAD and bleeding was strongest in the first 30 days. Patients with PAD had a numerically higher incidence of stroke in the 30 days following the procedure that was of borderline statistical significance (p = 0.08), but not from 30 days to 1 year.
Table 3. In-hospital outcomes by PAD status.
| Transfemoral access (n = 19,689) | ||||||
|---|---|---|---|---|---|---|
| Unadjusted | Risk-adjusted | |||||
| Outcome | No PAD (n = 14,865) |
PAD (n = 4824) |
OR (95% CI) | p-value | OR (95% CI) | p-value |
| Death | 395 (2.7) | 164 (3.4) | 1.30 (1.08-1.58) | 0.006 | 1.30 (1.08-1.57) | 0.006 |
| Myocardial infarction | 48 (0.3) | 21 (0.4) | 1.28 (0.78-2.08) | 0.32 | 1.36 (0.83-2.21) | 0.22 |
| Stroke | 257 (1.7) | 109 (2.3) | 1.32 (1.06-1.63) | 0.01 | 1.29 (1.03-1.61) | 0.03 |
| Any bleed | 1241 (8.4) | 516 (10.8) | 1.30 (1.15-1.47) | < 0.001 | 1.33 (1.17-1.51) | < 0.001 |
| Significant vascular complication | 1203 (8.1) | 492 (10.3) | 1.27 (1.12-1.45) | < 0.001 | 1.39 (1.21-1.59) | < 0.001 |
| Device success | 13,995 (94.2) | 4494 (93.4) | 0.94 (0.79-1.12) | 0.50 | -- | -- |
| Procedure completed | 14,661 (98.7) | 4714 (98.0) | 0.60 (0.47-0.78) | < 0.001 | -- | -- |
| Non-transfemoral access (n = 7780) | ||||||
| Unadjusted | Risk-adjusted | |||||
| Outcome |
No PAD (n = 4050) |
PAD (n = 3730) |
OR (95% CI) | p-value | OR (95% CI) | p-value |
| Death | 275 (6.8) | 250 (6.7) | 0.99 (0.83-1.18) | 0.88 | 0.99 (0.82-1.20) | 0.93 |
| Myocardial infarction | 27 (0.7) | 27 (0.7) | 1.01 (0.55-1.87) | 0.97 | 1.04 (0.55-1.96) | 0.92 |
| Stroke | 101 (2.5) | 96 (2.6) | 1.04 (0.78-1.38) | 0.81 | 1.14 (0.83-1.56) | 0.42 |
| Any bleed | 410 (10.1) | 485 (13.0) | 1.14 (0.99-1.30) | 0.06 | 1.18 (1.01-1.37) | 0.04 |
| Significant vascular complication | 141 (3.5) | 136 (3.7) | 1.04 (0.80-1.35) | 0.78 | 1.14 (0.87-1.49) | 0.34 |
| Device success | 3795 (93.7) | 3464 (92.9) | 0.91 (0.75-1.11) | 0.67 | -- | -- |
| Procedure completed | 4017 (99.2) | 3704 (99.3) | 1.20 (0.72-2.00) | 0.49 | -- | -- |
Significant vascular complications were more common in patients with PAD compared with those without, on both unadjusted and risk-adjusted analyses (10.3 vs. 8.1%; p < 0.001; Table 3). The rate of device success was > 93% in patients with and without PAD without significant differences between groups; the TAVR procedure was completed in > 98% of patients in both cohorts.
Outcomes in patients undergoing non-TF TAVR
Among patients with PAD undergoing TAVR via non-TF access, 933 (25.0%) died over 1-year follow-up, compared with 986 (24.4%) without PAD (unadjusted HR 1.07; 95% CI, 0.98-1.17; p = 0.13) (Figure 3). After adjustment for baseline risk, the association between PAD and 1-year mortality remained non-significant (HR 1.03; 95% CI 0.93-1.04; p = 0.61) (Table 2). In contrast to patients undergoing TAVR via TF access, patients with PAD did not have significantly higher rates of all-cause readmission or readmission for MI on adjusted and unadjusted analyses, though they did have a higher rate of bleeding (adjusted HR for bleeding, PAD vs. no PAD, 1.22, 95% CI 1.11-1.36; p < 0.001).
Figure 3. Cumulative incidence of outcomes over time among patients undergoing TAVR via non-femoral access, stratified by PAD status.
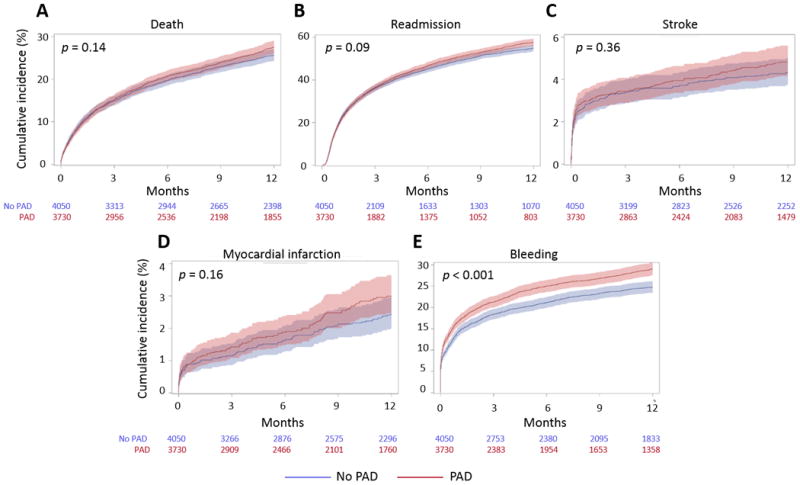
Kaplan-Meier curve depicts unadjusted all-cause mortality (A); cumulative incidence curves depict unadjusted readmission (B), stroke (C), myocardial infarction (D), and major bleeding (E).
Patients with PAD undergoing TAVR did not have higher unadjusted or risk-adjusted in-hospital rates of death, stroke, MI, or bleeding than those without PAD (Table 3). In unadjusted landmark analyses covering 30 days to 1 year post-TAVR, patients with PAD did have higher unadjusted rates of mortality and readmission, but these differences were attenuated and non-significant after risk adjustment (Supplemental Figure S2).
Discussion
In this analysis of the STS/ACC TVT registry, we found that PAD is present in nearly 1 in 4 patients undergoing commercial TAVR via TF access and nearly half of patients undergoing commercial TAVR via non-TF access. Compared with patients without PAD, patients with PAD were more likely to have coronary artery and cerebrovascular disease. In both adjusted and unadjusted analyses, patients with PAD undergoing TAVR via TF access were at higher risk for 1-year death, readmission, bleeding, and MI, but patients with PAD undergoing TAVR via non-TF access were only at increased risk of bleeding.
Prior to this study, incidence rates of PAD in patients undergoing TAVR have been reported in pivotal clinical trials evaluating the safety and efficacy of TAVR. Among all patients undergoing TAVR, PAD rates have ranged from 27.8 to 42.3%.6, 8, 24 For patients that were candidates for TF access, PAD rates ranged from 28.1 to 34.9%; PAD rates in patients requiring non-TF access were 42.7 to 60.2%. The lower prevalence of PAD in this commercial cohort is consistent with the fact that patients undergoing commercial TAVR are at lower risk of mortality than those enrolled in pivotal clinical trials. The definition of PAD used in the various sources may also play a role.19 In our study and others, the prevalence of PAD in patients undergoing TAVR is higher than that in the general population due both to overlapping risk factors and routine arterial imaging of these patients prior to TAVR.
One single center study, which included patients undergoing TAVR via TF or non-TF access, found that PAD was associated with a 25% increased risk of 2-year mortality, but this association was not statistically significant (HR 1.25; 95% CI 0.94-1.64).25 However, in that study, the prevalence of PAD among patients undergoing non-TF TAVR was double that of the patients undergoing TF TAVR. In a propensity-matched analysis of TF versus non-TF access from the PARTNER 1 trial, 95% of the matched cohort had PAD, confounding efforts to determine an association between PAD and TAVR outcomes.26 These studies underscore the principal difficulty of studying the effect of PAD on TAVR outcomes – PAD is strongly correlated with non-TF access for TAVR, which has a strong association, across multiple studies, with inferior outcomes.22, 27, 28
The scale of the TVT registry and the design of our study enabled us to tease apart the effects of PAD and access route on outcomes in patients undergoing TAVR. In a cohort of patients undergoing TF TAVR, PAD was associated with higher risk of death, readmission, MI, and bleeding. For all four of these outcomes, landmark analyses showed an association with PAD both from 0-30 days after the procedure and from 30-365 days, suggesting that these associations are mediated both by procedural factors and PAD, itself. In the cohort of patients undergoing non-TF access, PAD had no significant effect on unadjusted or risk-adjusted mortality, stroke, or MI, though it was associated with increased bleeding risk. Though we did not perform direct comparisons of outcomes for TF versus non-TF TAVR due to the risk of considerable confounding by unmeasured differences between the groups, and the likelihood that PAD severity differs between patients undergoing TF and non-TF access, patients with and without PAD undergoing TAVR via non-TF access had a numerically higher risk of death, readmission, MI, stroke, and bleeding than their counterparts undergoing TF TAVR, consistent with previously published reports.26, 29 Altogether, these data indicate that PAD is associated with worse outcomes in patients undergoing TAVR via TF access but that the effect of PAD on mortality and all-cause readmission among patients undergoing TAVR via non-TF access may be overwhelmed by the increased morbidity associated with more invasive access or other uncaptured non-PAD comorbidities that make patients with and without PAD physiologically less distinct in the non-TF group than in the TF group. The prevalence of PAD in the non-TF group (48%) is also lower than might be expected, which raises concerns that systematic underreporting of PAD in the registry may further limit the ability to detect a difference in outcomes between patients with and without PAD in this group. However, the TVT Registry is systematically audited for data accuracy, and the prevalence of PAD in our non-TF cohort is consistent with the prevalence of PAD in the non-TF cohorts in pivotal TAVR clinical trials (43-60%)6, 8, 24 and other “real-world” sources (22-78%)25, 30, 31 making systematic underreporting of PAD in the non-TF cohort unlikely. The perhaps unexpectedly low incidence of PAD in patients undergoing non-TF TAVR may be explained by the need for large bore vascular access with early generation devices. It is likely that many of the patients who underwent non-TF TAVR without PAD had femoral and iliac arteries too small to allow for the placement of large access sheaths but did not have > 50% obstruction on non-invasive imaging or PAD symptoms.32 With nearly 8000 patients undergoing non-TF TAVR, the lack of association identified between PAD and outcomes in these patients is unlikely to be due to lack of statistical power. Importantly, as we did not perform any comparisons by access site due to the strong likelihood of confounding, our analysis does not support any conclusions related to access site choice in patients with or without PAD.
Bleeding, which occurred in > 10% of patients undergoing TAVR in this commercial cohort, has important implications for post-TAVR costs and prognosis. In an analysis of patients undergoing TAVR in the PARTNER I randomized clinical trial, the 7.5% of patients with a major bleeding complication in hospital had hospital costs $43,374 higher and length of stay 7.7 days longer than patients without complications.33 In the same clinical trial, longer-term bleeding complications were strongly associated with a 1-year mortality.34 In our commercial cohort, patients with PAD were at higher risk of bleeding, both before and after adjustment for other comorbidities and concomitant antithrombotic therapy, whether they underwent TF or non-TF access. In-hospital outcomes, landmark analyses and visual inspection of cumulative incidence curves demonstrated that, among patients with PAD undergoing TF TAVR, much of the increased bleeding risk was concentrated in the peri-procedural period. In addition, the incidence of significant vascular complications, many of which likely manifested as bleeding, was higher in patients with PAD compared with those without. Similarly, though there was no long-term difference in the incidence of stroke between patients with and without PAD, patients with PAD had a higher risk of stroke, another important driver of hospital costs and length of stay, in the periprocedural period. Technological improvements designed to improve TAVR safety, both related to vascular access and cerebral embolic protection,35 may be particularly helpful for patients with PAD. It is possible that vascular complication rates in current practice are already lower than we report, due to reductions in sheath size over the past several years.
We also found extremely high rates (∼ 50%) of all-cause readmission among patients undergoing TAVR, which has been previously reported,29, 36 but the association between PAD and readmission had not been reported. Costs associated with readmission following TAVR account for 12-18% of total spending related to the episode of care.36-38 Though the majority of post-TAVR readmissions are for non-cardiac reasons,36 patients with PAD may derive particular benefit from strategies to reduce readmissions, especially if, as has been proposed, TAVR is included in CMS bundled payment models.
Several limitations must be acknowledged. The principal limitation of our study is its observational design, which prevents us from drawing firm conclusions regarding the causative nature of the association between PAD and outcomes in patients undergoing TAVR. However, regardless of whether the observed associations are causal, the adjusted and unadjusted association of PAD with worse outcomes is nevertheless important for patient counseling and physician decision-making. Our analyses of long term outcomes and the frequency with which patients undergo peripheral revascularization to facilitate TAVR or treat TAVR complications are further limited by their reliance on administrative data. Because we analyzed data from the TVT registry linked to CMS claims data, our cohort necessarily only includes patients ≥ 65 years old enrolled in fee-for-service Medicare. Though the cohort of patients we excluded due to lack of fee-for-service Medicare service were broadly similar to the patients we ultimately studied, it is possible that differences between these groups do exist. Lastly, though the registry defines PAD using standardized criteria, it does not indicate which criteria were used to make the diagnosis for individual patients
Conclusion
Nearly 1 in 4 patients undergoing TAVR via TF access, and nearly half of patients undergoing TAVR via non-TF access, have PAD. Compared with patients without PAD, patients with PAD undergoing TF TAVR have a higher incidence of death, readmission, MI, and bleeding over 1-year follow-up; however, among patients undergoing non-TF TAVR, patients with PAD do not have a greater risk of 1-year death or readmission than patients without PAD. Further work is necessary to reduce adverse outcomes among patients with PAD undergoing TAVR.
Supplementary Material
What is Known.
Risk factors for peripheral artery disease (PAD) and aortic stenosis (AS) overlap, and PAD was a frequent comorbidity in patients undergoing transcutaneous aortic valve replacement (TAVR) in pivotal clinical trials
PAD is associated with worse outcomes in the general population, as well as in patients undergoing coronary artery bypass grafting and percutaneous coronary intervention
What the Study Adds.
In a registry that collects data from every U.S. TAVR center, nearly 25% of patients undergoing transfemoral (TF) TAVR and 50% of patients undergoing non-TF TAVR had PAD
Compared with patients without PAD, patients with PAD undergoing TF TAVR had a higher incidence of death, readmission, and bleeding over 1-year follow-up
Patients with PAD undergoing non-TF TAVR did not have significantly higher rates of 1-year death or readmission compared with patients without PAD.
Acknowledgments
Sources of Funding: The Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry is an initiative of the Society of Thoracic Surgeons and the American College of Cardiology Foundation. This research was supported by the American College of Cardiology Foundation's National Cardiovascular Data Registry and the Society of Thoracic Surgeons.
Disclosures: Dr. Fanaroff was supported during the conduct of this study by NIH grant 5T32HL069749-13 and American Heart Association grant 17FTF33661087. Dr. Cohen has received research grants from Edwards Lifesciences, Metronic, Boston Scientific, and Abbott Vascular, and has served as a consultant for Edwards Lifesciences and Medtronic. Dr. Harrison reports research grants to his institution from Edwards Lifesciences, Medtronic, and Abbott Vascular for structural heart disease research. Dr. Mack has served as the co-principal investigator the PARTNER-3 clinical trial, sponsored by Edwards Lifesciences. Dr. Vemulapalli has received research grants from the American College of Cardiology, the Society of Thoracic Surgeons, the Patient Centered Outcomes Research Institute, and Abbott Vascular, and has served as a consultant for Novella. All other authors report no relationships relevant to the content of this paper to disclose.
Footnotes
Clinical Trial Registration Information: URL: http://clinicaltrials.gov. Unique identifier: NCT01737528
References
- 1.Fowkes FGR, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, Norman PE, Sampson UK, Williams LJ, Mensah GA. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet. 2013;382:1329–1340. doi: 10.1016/S0140-6736(13)61249-0. [DOI] [PubMed] [Google Scholar]
- 2.Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, de Ferranti S, Despres JP, Fullerton HJ, Howard VJ, Huffman MD, Judd SE, Kissela BM, Lackland DT, Lichtman JH, Lisabeth LD, Liu S, Mackey RH, Matchar DB, McGuire DK, Mohler ER, 3rd, Moy CS, Muntner P, Mussolino ME, Nasir K, Neumar RW, Nichol G, Palaniappan L, Pandey DK, Reeves MJ, Rodriguez CJ, Sorlie PD, Stein J, Towfighi A, Turan TN, Virani SS, Willey JZ, Woo D, Yeh RW, Turner MB. Heart disease and stroke statistics-2015 update: a report from the american heart association. Circulation. 2015;131:e29–e322. doi: 10.1161/CIR.0000000000000152. [DOI] [PubMed] [Google Scholar]
- 3.Eveborn GW, Schirmer H, Heggelund G, Lunde P, Rasmussen K. The evolving epidemiology of valvular aortic stenosis. the Tromsø study. Heart. 2013;99:396–400. doi: 10.1136/heartjnl-2012-302265. [DOI] [PubMed] [Google Scholar]
- 4.Fusini L, Mirea O, Tamborini G, Muratori M, Gripari P, Cefalù C, Ali SG, Maffessanti F, Andreini D, Pontone G. Incidence and severity of atherosclerotic cardiovascular artery disease in patients undergoing TAVI. Int J Cardiovasc Imaging. 2015;31:975–985. doi: 10.1007/s10554-015-0651-9. [DOI] [PubMed] [Google Scholar]
- 5.Leon MB, Smith CR, Mack M, Miller DC, Moses JW, Svensson LG, Tuzcu EM, Webb JG, Fontana GP, Makkar RR. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363:1597–1607. doi: 10.1056/NEJMoa1008232. [DOI] [PubMed] [Google Scholar]
- 6.Adams DH, Popma JJ, Reardon MJ, Yakubov SJ, Coselli JS, Deeb GM, Gleason TG, Buchbinder M, Hermiller J, Jr, Kleiman NS. Transcatheter aortic-valve replacement with a self-expanding prosthesis. N Engl J Med. 2014;370:1790–1798. doi: 10.1056/NEJMoa1400590. [DOI] [PubMed] [Google Scholar]
- 7.Grover FL, Vemulapalli S, Carroll JD, Edwards FH, Mack MJ, Thourani VH, Brindis RG, Shahian DM, Ruiz CE, Jacobs JP. 2016 annual report of the society of thoracic surgeons/american college of cardiology transcatheter valve therapy registry. Ann Thorac Surg. 2017;103:1021–1035. doi: 10.1016/j.athoracsur.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 8.Smith CR, Leon MB, Mack MJ, Miller DC, Moses JW, Svensson LG, Tuzcu EM, Webb JG, Fontana GP, Makkar RR. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364:2187–2198. doi: 10.1056/NEJMoa1103510. [DOI] [PubMed] [Google Scholar]
- 9.Ankle Brachial Index Collaboration. Ankle brachial index combined with Framingham Risk Score to predict cardiovascular events and mortality: a meta-analysis. JAMA. 2008;300:197. doi: 10.1001/jama.300.2.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Banerjee A, Fowkes FG, Rothwell PM. Associations Between Peripheral Artery Disease and Ischemic Stroke Implications for Primary and Secondary Prevention. Stroke. 2010;41:2102–2107. doi: 10.1161/STROKEAHA.110.582627. [DOI] [PubMed] [Google Scholar]
- 11.Popovic B, Arnould MA, Selton-Suty C, Latarche C, Angioi M, Carteaux JP, Villemot JP, Aliot E. Comparison of two-year outcomes in patients undergoing isolated coronary artery bypass grafting with and without peripheral artery disease. Am J Cardiol. 2009;104:1377–1382. doi: 10.1016/j.amjcard.2009.07.001. [DOI] [PubMed] [Google Scholar]
- 12.Bonaca MP, Bhatt DL, Storey RF, Steg PG, Cohen M, Kuder J, Goodrich E, Nicolau JC, Parkhomenko A, López-Sendón J. Ticagrelor for prevention of ischemic events after myocardial infarction in patients with peripheral artery disease. J Am Coll Cardiol. 2016;67:2719–2728. doi: 10.1016/j.jacc.2016.03.524. [DOI] [PubMed] [Google Scholar]
- 13.Dencker D, Pedersen F, Engstrøm T, Køber L, Højberg S, Nielsen MB, Schroeder TV, Lönn L. Major femoral vascular access complications after coronary diagnostic and interventional procedures: A Danish register study. Int J Cardiol. 2016;202:604–608. doi: 10.1016/j.ijcard.2015.09.018. [DOI] [PubMed] [Google Scholar]
- 14.Jones WS, Tricoci P, Huang Z, Moliterno DJ, Harrington RA, Sinnaeve PR, Strony J, Van de Werf F, White HD, Held C. Vorapaxar in patients with peripheral artery disease and acute coronary syndrome: insights from Thrombin Receptor Antagonist for Clinical Event Reduction in Acute Coronary Syndrome (TRACER) Am Heart J. 2014;168:588–596. doi: 10.1016/j.ahj.2014.06.017. [DOI] [PubMed] [Google Scholar]
- 15.Patel MR, Becker RC, Wojdyla DM, Emanuelsson H, Hiatt WR, Horrow J, Husted S, Mahaffey KW, Steg PG, Storey RF. Cardiovascular events in acute coronary syndrome patients with peripheral arterial disease treated with ticagrelor compared with clopidogrel: Data from the PLATO Trial. Eur J Prev Cardiol. 2014 doi: 10.1177/2047487314533215. 2047487314533215. [DOI] [PubMed] [Google Scholar]
- 16.Saw J, Bhatt DL, Moliterno DJ, Brener SJ, Steinhubl SR, Lincoff AM, Tcheng JE, Harrington RA, Simoons M, Hu T. The influence of peripheral arterial disease on outcomes: a pooled analysis of mortality in eight large randomized percutaneous coronary intervention trials. J Am Coll Cardiol. 2006;48:1567–1572. doi: 10.1016/j.jacc.2006.03.067. [DOI] [PubMed] [Google Scholar]
- 17.Généreux P, Webb JG, Svensson LG, Kodali SK, Satler LF, Fearon WF, Davidson CJ, Eisenhauer AC, Makkar RR, Bergman GW. Vascular complications after transcatheter aortic valve replacement: insights from the PARTNER (Placement of AoRTic TraNscathetER Valve) trial. J Am Coll Cardiol. 2012;60:1043–1052. doi: 10.1016/j.jacc.2012.07.003. [DOI] [PubMed] [Google Scholar]
- 18.Mack MJ, Brennan JM, Brindis R, Carroll J, Edwards F, Grover F, Shahian D, Tuzcu EM, Peterson ED, Rumsfeld JS. Outcomes following transcatheter aortic valve replacement in the United States. JAMA. 2013;310:2069–2077. doi: 10.1001/jama.2013.282043. [DOI] [PubMed] [Google Scholar]
- 19.Holmes DR, Nishimura RA, Grover FL, Brindis RG, Carroll JD, Edwards FH, Peterson ED, Rumsfeld JS, Shahian DM, Thourani VH. Annual outcomes with transcatheter valve therapy: from the STS/ACC TVT Registry. Ann Thorac Surg. 2016;101:789–800. doi: 10.1016/j.athoracsur.2015.10.049. [DOI] [PubMed] [Google Scholar]
- 20.Kappetein AP, Head SJ, Généreux P, Piazza N, Van Mieghem NM, Blackstone EH, Brott TG, Cohen DJ, Cutlip DE, van Es GA. Updated standardized endpoint definitions for transcatheter aortic valve implantation: the Valve Academic Research Consortium-2 consensus document. Eur Heart J. 2012;33:2403–2418. doi: 10.1093/eurheartj/ehs255. [DOI] [PubMed] [Google Scholar]
- 21.Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509. [Google Scholar]
- 22.Edwards FH, Cohen DJ, O'Brien SM, Peterson ED, Mack MJ, Shahian DM, Grover FL, Tuzcu EM, Thourani VH, Carroll J. Development and validation of a risk prediction model for in-hospital mortality after transcatheter aortic valve replacement. JAMA Cardiol. 2016;1:46–52. doi: 10.1001/jamacardio.2015.0326. [DOI] [PubMed] [Google Scholar]
- 23.O'Brien SM, Cohen DJ, Rumsfeld JS, Brennan JM, Shahian DM, Dai D, Holmes DR, Hakim RB, Thourani VH, Peterson ED. Variation in Hospital Risk–Adjusted Mortality Rates Following Transcatheter Aortic Valve Replacement in the United States. Circ Cardiovasc Qual Outcomes. 2016 doi: 10.1161/CIRCOUTCOMES.116.002756. 2016:CIRCOUTCOMES. 116.002756. [DOI] [PubMed] [Google Scholar]
- 24.Leon MB, Smith CR, Mack MJ, Makkar RR, Svensson LG, Kodali SK, Thourani VH, Tuzcu EM, Miller DC, Herrmann HC. Transcatheter or surgical aortic-valve replacement in intermediate-risk patients. N Engl J Med. 2016;374:1609–1620. doi: 10.1056/NEJMoa1514616. [DOI] [PubMed] [Google Scholar]
- 25.Schymik G, Würth A, Bramlage P, Herbinger T, Heimeshoff M, Pilz L, Schymik JS, Wondraschek R, Süselbeck T, Gerhardus J. Long-term results of transapical versus transfemoral TAVI in a real world population of 1000 patients with severe symptomatic aortic stenosis. Circ Cardiovasc Interv. 2015;8:e000761. doi: 10.1161/CIRCINTERVENTIONS.113.000761. [DOI] [PubMed] [Google Scholar]
- 26.Blackstone EH, Suri RM, Rajeswaran J, Babaliaros V, Douglas PS, Fearon WF, Miller DC, Hahn RT, Kapadia S, Kirtane AJ. Propensity-Matched Comparisons of Clinical Outcomes After Transapical or Transfemoral Transcatheter Aortic Valve Replacement A Placement of Aortic Transcatheter Valves (PARTNER)-I Trial Substudy. Circulation. 2015;131:1989–2000. doi: 10.1161/CIRCULATIONAHA.114.012525. [DOI] [PubMed] [Google Scholar]
- 27.van der Boon RM, Marcheix B, Tchetche D, Chieffo A, Van Mieghem NM, Dumonteil N, Vahdat O, Maisano F, Serruys PW, Kappetein AP. Transapical versus transfemoral aortic valve implantation: a multicenter collaborative study. Ann Thorac Surg. 2014;97:22–28. doi: 10.1016/j.athoracsur.2013.09.088. [DOI] [PubMed] [Google Scholar]
- 28.Blackman DJ, Baxter PD, Gale CP, Moat NE, Maccarthy PA, HILDICK-SMITH D, Trivedi U, Cunningham D, De Belder MA, Ludman PF. Do outcomes from transcatheter aortic valve implantation vary according to access route and valve type? The UK TAVI Registry. J Interv Cardiol. 2014;27:86–95. doi: 10.1111/joic.12084. [DOI] [PubMed] [Google Scholar]
- 29.Holmes DR, Brennan JM, Rumsfeld JS, Dai D, O'Brien SM, Vemulapalli S, Edwards FH, Carroll J, Shahian D, Grover F. Clinical outcomes at 1 year following transcatheter aortic valve replacement. JAMA. 2015;313:1019–1028. doi: 10.1001/jama.2015.1474. [DOI] [PubMed] [Google Scholar]
- 30.Koifman E, Magalhaes M, Kiramijyan S, Escarcega RO, Didier R, Torguson R, Ben-Dor I, Corso P, Shults C, Satler L. Impact of transfemoral versus transapical access on mortality among patients with severe aortic stenosis undergoing transcatheter aortic valve replacement. Cardiovasc Revasc Med. 2016;17:318–321. doi: 10.1016/j.carrev.2016.05.002. [DOI] [PubMed] [Google Scholar]
- 31.Murashita T, Greason KL, Pochettino A, Sandhu GS, Nkomo VT, Bresnahan JF, Reeder GS, Holmes DR, Rihal CS, Mathew V. Clinical outcomes after transapical and transfemoral transcatheter aortic valve insertion: An evolving experience. Ann Thorac Surg. 2016;102:56–61. doi: 10.1016/j.athoracsur.2015.11.073. [DOI] [PubMed] [Google Scholar]
- 32.Basir MB, Velez C, Fuller B, Wyman J, Paone G, Wang DD, Guerrero M, Greenbaum A, O'neill W. Rates of vascular access use in transcatheter aortic valve replacement: a look into the next generation. Catheter Cardiovasc Interv. 2016;87 doi: 10.1002/ccd.26116. [DOI] [PubMed] [Google Scholar]
- 33.Arnold SV, Lei Y, Reynolds MR, Magnuson EA, Suri RM, Tuzcu EM, Petersen JL, Douglas PS, Svensson LG, Gada H. Costs of Periprocedural Complications in Patients Treated With Transcatheter Aortic Valve Replacement Results From the Placement of Aortic Transcatheter Valve Trial. Circ Cardiovasc Interv. 2014;7:829–836. doi: 10.1161/CIRCINTERVENTIONS.114.001395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Généreux P, Cohen DJ, Mack M, Rodes-Cabau J, Yadav M, Xu K, Parvataneni R, Hahn R, Kodali SK, Webb JG. Incidence, predictors, and prognostic impact of late bleeding complications after transcatheter aortic valve replacement. J Am Coll Cardiol. 2014;64:2605–2615. doi: 10.1016/j.jacc.2014.08.052. [DOI] [PubMed] [Google Scholar]
- 35.Giustino G, Mehran R, Veltkamp R, Faggioni M, Baber U, Dangas GD. Neurological outcomes with embolic protection devices in patients undergoing transcatheter aortic valve replacement: a systematic review and meta-analysis of randomized controlled trials. JACC Cardiovasc Interv. 2016;9:2124–2133. doi: 10.1016/j.jcin.2016.07.024. [DOI] [PubMed] [Google Scholar]
- 36.Kolte D, Khera S, Sardar MR, Gheewala N, Gupta T, Chatterjee S, Goldsweig A, Aronow WS, Fonarow GC, Bhatt DL. Thirty-Day Readmissions After Transcatheter Aortic Valve Replacement in the United States. Circ Cardiovasc Interv. 2017;10:e004472. doi: 10.1161/CIRCINTERVENTIONS.116.004472. [DOI] [PubMed] [Google Scholar]
- 37.Reynolds MR, Magnuson EA, Lei Y, Wang K, Vilain K, Li H, Walczak J, Pinto DS, Thourani VH, Svensson LG. Cost-effectiveness of transcatheter aortic valve replacement compared with surgical aortic valve replacement in high-risk patients with severe aortic stenosis: results of the PARTNER (Placement of Aortic Transcatheter Valves) trial (Cohort A) J Am Coll Cardiol. 2012;60:2683–2692. doi: 10.1016/j.jacc.2012.09.018. [DOI] [PubMed] [Google Scholar]
- 38.Reynolds MR, Lei Y, Wang K, Chinnakondepalli K, Vilain KA, Magnuson EA, Galper BZ, Meduri CU, Arnold SV, Baron SJ. Cost-effectiveness of transcatheter aortic valve replacement with a self-expanding prosthesis versus surgical aortic valve replacement. J Am Coll Cardiol. 2016;67:29–38. doi: 10.1016/j.jacc.2015.10.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


