Abstract
Purpose
The aim of our study was to analyse paediatric sight-impairment trends in Northern Ireland (NI) over a 28-year period to better understand the impact which changes in health-care provision may be having on childhood blindness and to enable us to assess our progress towards achieving the World Health Organisation (WHO) aims.
Methods
A database of Certificates of Visual Impairment completed for NI children aged <16 years was used to determine the cause of sight impairment from 1984 to 2011. Causes were classified into preventable or treatable conditions and analysed for trends.
Results
Five hundred and ninety-eight children were registered as having impaired vision over the 28-year period. A total of 22% had preventable or treatable conditions. Optic atrophy was the most common cause responsible for 16% of registrations followed by albinism (12%), cerebral visual impairment (11%), congenital cataract (8%), retinopathy of prematurity (ROP) (8%), and congenital motor nystagmus (7%). The incident rate (per million population aged <16 years) for registerable vision loss due to congenital cataract decreased from 5.89 (CI 2.82–10.83) in 1984–1987 to 2.63 (CI 0.72–6.74) in 2008–2011. For ROP, the incident rate peaked during 2000–2003 at 8.87 (CI 4.85–14.88). Thereafter, there was a statistically significant reduction in incident rate to 1.98 (CI 0.41–5.77) in 2008–2011 (P=0.008).
Conclusions
Sight-impairment registrations due to preventable or treatable causes have decreased over the past 28 years. This is likely due to better surgical techniques and improved refractive care for conditions such as congenital cataract and ongoing research and treatment protocols for conditions such as ROP. Future advances in this area may help to further reduce the burden of childhood sight impairment and improve quality of life for these patients.
Introduction
Preventable sight impairment has been a major focus of the World Health Organisation (WHO) for many years. In 1999, WHO published its initiative for eliminating avoidable blindness, VISION 2020—The Right to Sight.1 Its vision is a world in which no one is needlessly blind and those with unavoidable vision loss can achieve their full potential.1 Childhood blindness is one of the priority eye diseases highlighted in this document. An estimated worldwide prevalence of 1.4 million blind children confers significant social, economic, and emotional impact on society.2 Major focus is on the preventable or treatable causes that are felt to account for 40% of childhood blindness.3 The main avoidable causes of visual impairment worldwide are corneal scarring, cataract, glaucoma, retinopathy of prematurity (ROP), and refractive errors.3 Early treatment of these conditions is vital to prevent amblyopia and ensure normal visual development.4
Worldwide, there have been several breakthroughs since the publication of VISION 2020 in relation to childhood blindness. The United Nations Children’s Fund (UNICEF) estimates significant reduction in Vitamin A deficiency-related visual impairment due to the implementation of the Vitamin A global Initiative.3 The Measles initiative has reduced measles-related deaths by 28% from 1999 to 2004 and an associated reduction in the incidence of corneal scarring is expected.3
WHO outlined objectives for 2020 in relation to childhood blindness that include provision of services to treat children with cataract, glaucoma, ROP, and corneal scarring.3 It also states that optical services to address refractive errors and provision of low vision services should be made available.
The aim of our study was to analyse paediatric sight-impairment trends in Northern Ireland (NI) over a 28-year period to give us a better understanding of the impact which changes in health-care provision may have had on childhood sight loss and to enable us to assess our progress towards achieving the WHO aims for VISION 2020.
Materials and methods
A database with information gathered from Certificates of Visual Impairment completed for NI residents from 1984 is held by the hospital optometric department of the Belfast Health and Social Care Trust (BHSCT). The Certificate of Visual Impairment form for NI is available for viewing on the Royal College of Ophthalmologists website.5
As the data available after 2011 was incomplete, due to issues with lack of staff to maintain the database, data were extracted for all patients aged <16 years who were registered as either sight impaired (SI) or severely sight impaired (SSI) between 1984 and 2011 inclusive. This information was then entered into and analysed using Microsoft Excel 2007 (Microsoft Corporation, Redmond, WA, USA). Of note, data were not available for 1989 or 1998, again due to staffing issues impacting on database maintenance.
Full approval was obtained from the BHSCT Standards, Quality and Audit department and data were managed in accordance with BHSCT current guidance on data protection.
We adhered to the definitions for SI and SSI as set out by the Department of Health, Certificate of Visual Impairment: Explanatory notes for Consultant Ophthalmologists and Hospital Eye Clinic Staff issued in September 2007.6 This document defines severe sight impairment as Snellen visual acuity <3/60, or visual acuity between 3/60 and 6/60 with a very contracted field of vision, or visual acuity >6/60 but contracted field of vision especially inferiorly. In our paediatric population, equivalent visual acuity measurements using other optotypes were valid, or if visual acuity could not be measured formally, clinical features of visual defects (eg, not fixing or following to light) were included but classified as SI. The 2007 Department of Health document defines sight impairment as Snellen visual acuity between 3/60 and 6/60 with full field, visual acuity up to 6/24 with moderate field contraction, opacities in the media or aphakia, or visual acuity of 6/18 or better with marked field contraction.6 We appreciate that these definitions do not directly correlate with the WHO 2016 ICD-10 definitions of mild, moderate, and severe visual impairment and blindness.7
However, as we analysed data from Certificates of Visual Impairment, we felt it was appropriate to present this data using the Department of Health Certificate of Visual Impairment definitions of sight impairment and severe sight impairment.
Based on the information on the cause of reduced vision in each patient, we classified aetiology using a dual taxonomy described by Rahi et al.8 The causes were classified in two ways: by individual disorders, grouped by the anatomical site affected, and by timing of specific causal factors leading to vision loss.
We further classified the individual disorders causing sight loss into preventable or non-preventable and treatable or non-treatable based on current understanding with a view to identifying the proportion of avoidable vision loss in our study population.
Looking at the SI and SSI registrations as a whole, the six leading causes of reduced vision were identified for our NI population. For every 4-year period between 1984 and 2011, we identified the number of registrations for each condition and estimated the annual incident rates per million population aged <16 years. In the periods which included 1 year of missing data (1988–1991 and 1996–1999), incident rates were computed using the available information (3 years in each case). 95% confidence intervals were computed for the incidence rates assuming a Poisson distribution model. In comparisons between periods, statistical significance of differences in incidence rates was assessed using Fisher’s exact test. Poisson regression model analysis was used to check for trends in incidence rates over the seven 4-year periods. Differences and trends in incidence rates with a P-value of <0.05 were considered statistically significant. Analysis was performed using MATLAB 9.1 (R2016b) (Mathworks, Natick, MA, USA).
Results
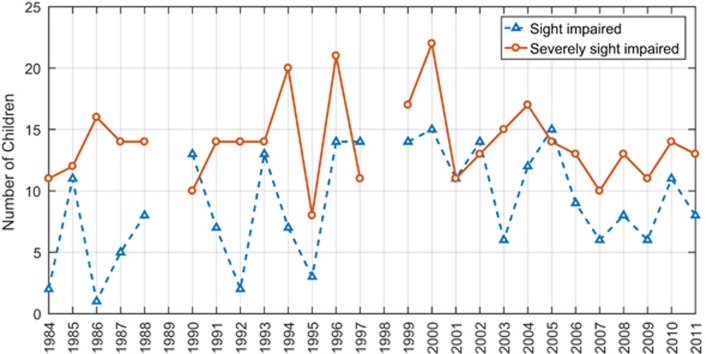
Five hundred and ninety-eight children were registered as having impaired vision over the 28-year period (Figure 1). Two hundred and thirty-four (39%) were registered as SI and 364 (61%) were registered as SSI. In all, 53% were males and 26% had associated systemic conditions (Table 1). The mean age at registration was 7.4 years (SD±4.6 years) with a range of 2 months to 16 years. Three of these patients had no cause of sight impairment documented on their registration forms and were therefore excluded from further analysis.
Figure 1.
The number of children registered as SSI and SI per year. (Note: Data not collected for 1989 and 1998).
Table 1. Demographic details, associations and timing of insult for severely sight-impaired and sight-impaired children.
| Severely sight impaired (n=364) | Sight impaired (n=234) | Overall (n=598) | |
|---|---|---|---|
| Gender | |||
| Male | 181 | 134 | 315 |
| Female | 181 | 98 | 279 |
| Unknown | 2 | 2 | 4 |
| Age, years | |||
| <1 | 20 | 1 | 21 |
| 1–4 | 121 | 48 | 169 |
| 5–15 | 212 | 176 | 388 |
| Unknown | 11 | 9 | 20 |
| Positive family history of condition | 39 (11%) | 32 (14%) | 71 (12%) |
| Known associated systemic/syndromic condition | 125 (34%) | 28 (12%) | 153 (26%) |
| Time of insult | |||
| Prenatal | 222 (61%) | 187 (80%) | 409 (68%) |
| Perinatal/neonatal | 110 (30%) | 19 (8%) | 129 (21%) |
| Childhood | 30 (8%) | 27 (11%) | 57 (10%) |
| Unknown | 2 (1%) | 1 (1%) | 3 (1%) |
The timing of the insult was prenatal in 69% (of which 77% were hereditary/congenital), perinatal/neonatal in 22%, and occurred during childhood in 9% (Table 2). Three hundred and fifty-seven cases (60%) were neither preventable nor treatable. Of the remaining 238 cases, 132 (22%) were considered either preventable or treatable and 104 (18%) were considered to be possibly preventable or treatable (Table 3).
Table 2. Causal factors categorised into time of insult.
| Severely sight impaired (n=362) | Sight impaired (n=233) | Overall (n=595) | |
|---|---|---|---|
| Prenatal | n=222 | n=187 | |
| Hereditary/congenital | 153 | 163 | 316 |
| Infection | 4 | 2 | 6 |
| Other | 65 | 22 | 87 |
| Perinatal/neonatal | n=110 | n=19 | |
| Hypoxia/ischaemia | 50 | 3 | 53 |
| Infection | 2 | 0 | 2 |
| Hydrocephalus | 10 | 5 | 15 |
| Other | 48 | 11 | 59 |
| Childhood | n=30 | n=27 | |
| Tumour | 12 | 5 | 17 |
| Infection | 2 | 0 | 2 |
| Refractive | 4 | 12 | 16 |
| Accidental injury | 1 | 1 | 2 |
| Systemic disorders | 6 | 4 | 10 |
| Other | 5 | 5 | 10 |
Table 3. Potentially preventable or treatable causes.
| Number of children (n=595) | |
|---|---|
| Preventable | Total=50 (8%) |
| Retinopathy of prematurity | 48 |
| Prenatal Rubella | 2 (cataracts) |
| Possibly preventable | Total=44 (7%) |
| Autosomal-dominant conditions | |
| Aniridia | 2 |
| Retinal dystrophy | 11 |
| Primary optic atrophy | 9 |
| Congenital glaucoma | 2 |
| Congenital cataract | 11 |
| Retinoblastoma | 2 |
| Other | 5 |
| Trauma | 2 |
| Treatable | Total=82 (14%) |
| Refractive error | 17 |
| Uveitis | 3 |
| Glaucoma | 15 |
| Ectopia lentis | 7 |
| Cataract | 32 |
| Retinal detachment | 2 |
| Retinoblastoma | 4 |
| Meningitis | 1 |
| Infection | 1 |
| Possibly treatable | Total=62 (10%) |
| Hydrocephalus | 21 |
| Tumour | 18 |
| Toxoplasmosis | 5 |
| Other | 18 |
| Not treatable or preventable | Total=357 (60%) |
Anatomically, the majority (37%) of registrations for impaired vision were due to retinal disorders (Supplementary Table 4). Only 31% of these 218 patients with retinal pathology had potentially preventable/treatable causes, such as ROP or autosomal-dominant retinal dystrophy.
Overall, the most common cause of vision loss was optic atrophy (16%); of these 39% were primary optic atrophy and 61% had secondary optic atrophy. The subsequent five main causes in order after optic atrophy were, albinism (12%), cerebral visual impairment (CVI; 11%), congenital cataract (8%), ROP (8%), and congenital motor nystagmus (CMN; 7%).
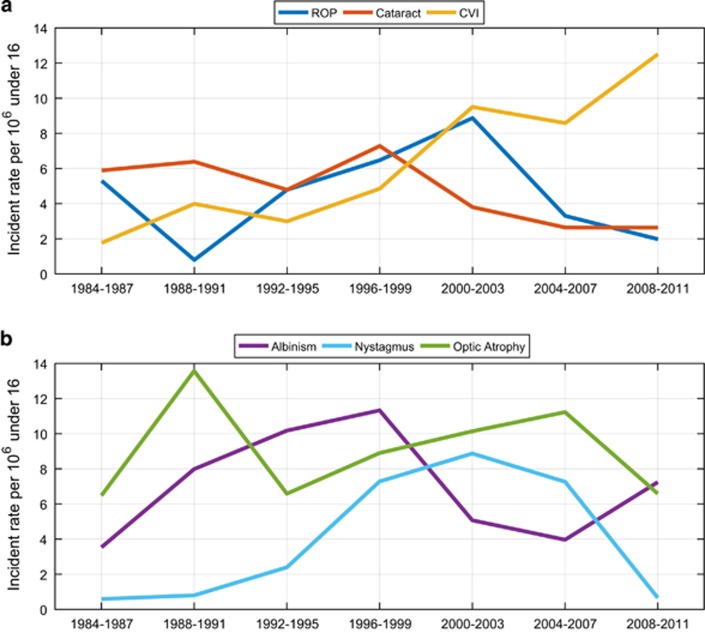
When we analyse the incident rates for the six leading causes of all registrations (SI and SSI) in NI for each 4-year period from 1984 to 2011, we can identify some interesting trends in both the preventable/treatable conditions and also the non-preventable/non-treatable conditions (Figure 2).
Figure 2.
Incident rates (per million population aged <16 years) for the six leading causes of paediatric sight impairment in Northern Ireland for each 4-year period from 1984 to 2011. (a) ROP, Cataract & CVI; (b) Albinism, Congenital Nystagmus & Optic Atrophy.
Incident rates for optic atrophy and albinism fluctuate over the years, but there was no significant change between 1984–1987 and 2008–2011 for either condition (P for trend=0.88 and 0.94, respectively). This reflects the fact that visual loss due to either optic atrophy or albinism can neither be prevented nor treated.
CVI as a cause for registration shows a general upward trend over the past 28 years. The incident rate increased from 1.77 per million (CI 0.36–5.16) in 1984–1987 to 12.51 per million (CI 7.53–19.54) in 2008–2011. This sevenfold increase was statistically significant (P for trend <0.0001).
CMN also shows a significant upward trend with an incident rate of 0.59 per million (CI 0.01–3.28) in 1984–1987 increasing to 8.87 per million (CI 4.85–14.88) in 2000–2003 (P for difference=0.0003); at which point, it dramatically reduces to 0.66 per million (CI 0.02–3.67) in 2008–2011 (P for difference=0.0006).
The two main preventable/treatable causes of paediatric vision loss that show interesting trends over the past 28 years are congenital cataract and ROP, which together are responsible for 16% of reported sight impairment and severe sight impairment.
The incident rate for congenital cataract decreased from 5.89 per million (CI 2.82–10.83) in 1984–1987 to 2.63 per million (CI 0.72–6.74) in 2008–2011. This twofold decrease in registered vision loss due to congenital cataract failed to reach statistical significance (P for trend=0.053).
The incident rate for ROP reduced from 5.30 per million (CI 2.42–10.06) in 1984–1987 to 0.80 per million (CI 0.02–4.45) in 1988–1991 (P for difference=0.03) but peaked again during 2000–2003 with an incident rate of 8.87 per million (CI 4.85–14.88). Thereafter, there was a statistically significant reduction in incident rate to 1.98 per million (CI 0.41–5.77) in 2008–2011 (P for difference=0.008).
Discussion
The NI Statistics and Research Agency reported a paediatric population (aged <16 years) of 379 323 following the 2011 NI Census.9 We identified 21 new cases of children registered with impaired sight in 2011, giving an annual incidence of 0.55 per 10 000 (CI 0.34–0.84). Of these 21 children, 13 were registered as SSI giving an incidence of blind registrations of 0.34 per 10 000 (CI 0.18–0.58) in 2011 in children aged <16 years that is comparable to reported annual incidence of new blind registrations of 0.41 per 10 000 in 2011 in children aged <17 years in a similar study conducted in England by Mitry et al.10
A 1976 NI population study by Bryars and Archer11 identified genetic disease and birth hypoxia as the leading causes of significant sight impairment. This finding has been replicated in the current study with 56% of SSI children having perinatal hypoxia or hereditary/congenital causal factors recorded on their registration forms.
Avoidable causes of blindness have been given priority by the WHO organisation and ROP is recognised as one of those conditions. ROP contributed to 8% of all registrations in our overall study population but there were significant fluctuations over time. Our study data extends back to 1984 and the first significant change in incident rate for vision loss secondary to ROP occurred between 1984–1987 and 1988–1991 when there was a 6.6-fold reduction in incident rate (CI 0.92–291.06, P for difference=0.03). This decline coincided with the publication of the results of the CRYO-ROP trial in early 1988. This large multicentre trial demonstrated that those neonates with threshold ROP treated with cryotherapy had a significantly lower rate of adverse outcome compared with those untreated (21.8 vs 43%).12 This study clearly impacted on the visual outcome of neonates with ROP as demonstrated by our findings. As the percentage of preterm babies surviving annually continued to increase and the survival rate for those born at extremely low gestational age (<26 weeks) continued to improve with advancing neonatal care, the rate of sight impairment attributable to ROP in NI steadily increased after 1988–1991.13, 14 With the hope of further lowering the rate of adverse outcomes, the National Eye Institute put together a collaborative group to study earlier treatment for ROP (ETROP), the results of which were published in 2003.15 The ETROP study was pivotal in highlighting the importance of early ablative therapy in improving visual outcomes for patients with high-risk ROP. Our study demonstrates coinciding positive effects on childhood vision with the incident rate for vision loss due to ROP falling from its highest level of 8.87 per million during 2000–2003 to 1.98 per million in 2008–2011. The adoption of the ETROP recommendations for earlier treatment of ROP was also reflected in the reported increase in ROP treatments in NI with the percentage of ‘at-risk’ infants surviving to 42 weeks gestational age requiring ROP treatment increasing from 1% in 2000 to 5.8% in 2011.14
The most common treatable cause of vision loss in our population was cataract (8% of all registrations), which also demonstrated an interesting trend with incident rates decreasing by 50% over the 28-year period. At the start of our study, congenital cataracts were responsible for 14% of the 72 registrations during 1984–1987, which gradually declined to 5% of the 84 registrations during 2008–2011 (Figure 2). This is comparable to the findings of a large study performed by the British Childhood Visual Impairment Study Group, which also reported 5% of severe visual impairment to be caused by cataract in 2000.8
Although the incidence of congenital cataract is relatively low in developed countries such as the United Kingdom, it remains the leading cause of surgically correctable blindness worldwide.16 In the United Kingdom, during the 1960s and 1970s, UK child health surveillance programmes began to develop that recognised the importance of vision screening in children. By the late 1980s, these programmes were being rationalised and more robust screening programmes for preschool children were established.17 Newborn screening should now occur within the first 72 h after birth and between 6 and 8 weeks of age to identify any abnormality of the red reflex.18 This early detection of cataract is vital in providing timely treatment for what is an extremely amblyogenic condition. Although it did not reach statistical significance, the observed downward trend in vision loss due to cataract may possibly be explained by this early detection in addition to improved surgical techniques and refractive aftercare.16
CVI as a cause for registerable visual loss demonstrated a sevenfold increase in incident rate between 1984–1987 and 2008–2011. This is now one of the main causes of impaired vision in our population, and although it is not treatable or reversible, it should be a focus of study and investment to reduce the impact on patient’s quality of life. This increase in CVI may be explained by the improvement in neonatal care for the premature or disabled child, resulting in increased survival rates. The proportion of premature neonates has not significantly increased over the past 28 years.19 However, the survival rate of these preterm infants has greatly improved owing to the improved neonatal care. In the United Kingdom and Wales, neonatal mortality rate fell by 60% from 7.7 deaths per 1000 live births in 1980 to 3.1 deaths per 1000 live births in 2009.20 This along with increased awareness and reporting are possible explanations for the significant increase in reported cases of CVI.
Another interesting trend that may reflect the effect of improved diagnostic investigations is the observed changes in incident rates in CMN and albinism over the past decade. In 2004–2007, the incident rate for registration due to CMN was 7.26 per million (CI 3.63–13.00), which decreased to 0.66 per million (CI 0.02–3.67) in 2008–2011 (P for difference=0.003). This decrease coincided with an increase in the incident rate for registrations due to albinism from 3.96 per million (CI 1.45–8.62) in 2004–2007 to 7.24 per million (CI 3.62–12.96) in 2008–2011 (P for difference=0.17).
Foveal hypoplasia has long been recognised as an associated feature of albinism, even in mild cases, but is not evident in patients with CMN.21 With improved availability of optical coherence tomography, foveal hypoplasia is now a much more easily identified feature. In turn, children who were once diagnosed as having CMN may be now more correctly diagnosed as having ocular albinism.
Our patient cohort is population based. According to migration statistics, immigration in NI is greater than emigration making NI an ideal population for epidemiological studies. Our study, similar to other published studies on paediatric sight impairment, analysed data from registration databases that are largely non-compulsory and therefore may not have captured all children with significant sight impairment. A further limitation of our study was the lack of data collection for 1989 and 1998 due to staffing issues. We also recognise that the concept of childhood sight impairment has changed over the past three decades and ‘blind’ registers do not comprehensively capture all children with complex visual needs.22
The data are useful, however, as a minimum estimate and can provide useful indication of trends. This study demonstrates the positive impact improvements in health care and disease prevention can have on the quality of life of our paediatric population. In line with the aims of VISION 2020, the implementation of effective screening and treatment protocols for preventable and treatable conditions has resulted in a decreasing trend in some of our leading causes of paediatric sight impairment. Future advances in therapies for genetic ophthalmic disorders may help to further reduce the burden of childhood sight impairment due to currently non-treatable and non-preventable conditions, such as albinism and hereditary optic atrophy.

Acknowledgments
Special thanks to Ms Pat Hart, Consultant Ophthalmologist and Rosaleen McCann, Hospital Optometrist for facilitating access to the Belfast Health & Social Care Trust Certificate of Visual Impairment Database.
Footnotes
Supplementary Information accompanies this paper on Eye website (http://www.nature.com/eye)
The authors declare no conflict of interest.
Supplementary Material
References
- World Health Organization with collaboration of the Task Force of Partnership Committee of Nongovernmental Development OrganizationsGlobal Initiative for the Elimination of Avoidable Blindness. World Health Organization: Geneva, Switzerland, 1998. (unpublished document WHO/PBL/97.51). [Google Scholar]
- World Health Organisation and International Agency for the Prevention of BlindnessPreventing Blindness in Children: Report of a WHO/IAPB Scientific Meeting. World Health Organization: Geneva, Switzerland, 2000. (unpublished document WHO/PBL/00.77). [Google Scholar]
- World Health Organisation. VISION 2020—the Right to Sight. Global initiative for the elimination of avoidable blindness. Action Plan 2006–2011, World Health Organisation: WHO Press: Geneva, Switzerland, 2007.
- Gilbert C, Foster A. Childhood blindness in the context of VISION 2020—the right to sight. Bull World Health Organ 2001; 79: 227–232. [PMC free article] [PubMed] [Google Scholar]
- Certificate of visual impairment form Northern Ireland. Published December 2014. Royal college of Ophthalmologists https://www.rcophth.ac.uk/publications/.
- Department of Health. Certificate of Visual Impairment: Explanatory notes for Consultant Ophthalmologists and Hospital Eye Clinic Staff. http://webarchive.nationalarchives.gov.uk/20130107105354/http://www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/documents/digitalasset/dh_078294.pdf.
- World Health Organisation International Statistical Classification of Diseases and Related Health Problems 10th Revision (ICD-10) 2016. http://apps.who.int/classifications/icd10/browse/2016/en#/H54.0.
- Rahi J, et Cable N. Severe visual impairment and blindness in children in the UK. British Childhood Visual Impairment Study Group (BCVISG). Lancet 2003; 362: 1359–1365. [DOI] [PubMed] [Google Scholar]
- Northern Ireland Statistics and Research Agency. Northern Ireland Census 2015. https://www.nisra.gov.uk/publications/2011-census-table-lookups.
- Mitry D, Bunce C, Wormald R et al. Childhood visual impairment in England: a rising trend. Arch Dis Child 2013; 98: 378–380. [DOI] [PubMed] [Google Scholar]
- Bryars J, Archer D. Aetiological survey of visually handicapped children in Northern Ireland. Trans Ophthamol Soc UK 1977; 97(1): 26–29. [PubMed] [Google Scholar]
- Cryotherapy for Retinopathy of Prematurity Cooperative Group. Multicenter trial of cryotherapy for retinopathy of prematurity: preliminary results. Pediatrics 1988; 81(5): 697–706. [PubMed] [Google Scholar]
- Centre for Maternal and Child Enquiries. Perinatal and Maternal Mortality 2009: Feedback Report 2010. Public Health Agency Northern ireland. Available at: http://www.publichealth.hscni.net/sites/default/files/Perinatal%20%20Maternal%20Mortality%202009%20Northern%20Ireland.pdf.
- McCall E, Craig S. Neonatal Care in Northern Ireland. Neonatal Intensive Care Outcomes Research & Evaluation (NICORE) 2009. Published July 2012, NICORE, Printed CDS, Belfast, ISBN: 978-1-909131-00-2.
- Good W. Final results of the Early Treatment for Retinopathy of Prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc 2004; 102: 233–250. [PMC free article] [PubMed] [Google Scholar]
- Alagaratnam J, Sharma TK, Lim CS, Fleck BW. A survey of visual impairment in children attending the Royal Blind School, Edinburgh using the WHO childhood visual impairment database. Eye 2002; 16: 557–561. [DOI] [PubMed] [Google Scholar]
- Stewart Brown SL, Haslum MN, Howlett B. Preschool vision screening: a service in need of rationalisation. Arch Dis Child 1988; 63: 356–359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis A, Elliman D. Newborn and Infant Physical Examination. UK National Screening Committee 2008. Public Health England. Available at: https://www.gov.uk/government/publications/newborn-and-infant-physical-examination-screening-standards.
- NHS Scotland. Births in Scottish Hospitals, August 2013. A National Statistics Publication for Scotland. Available at: http://www.isdscotland.org/Health-Topics/Maternity-and-Births/Publications/2014-08-26/2014-08-26-Births-Report.pdf.
- Office for National Statstics. Childhood, Infant and Perinatal Mortality in England and Wales. Office for National Statistics: Newport, UK 2011. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/bulletins/childhoodinfantandperinatalmortalityinenglandandwales/2013-02-27.
- Meyer C, Lapolice D, Freedman S. Foveal hypoplasia in oculocutaneous albinism demonstrated by optical coherence tomography. Am J Ophthalmol 2002; 133(3): 409–410. [DOI] [PubMed] [Google Scholar]
- Flanagan N, Jackson A, Hill AE. Visual impairment in childhood: insights from a community-based survey. Child Care Health Dev 2003; 29(6): 493–499. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.