Abstract
This systematic review aims to assess the efficacy of titanium (Ti) implant surfaces with or without strontium (Sr) incorporation on osseointegration in animal experimental studies. An electronic search was conducted using databases of PubMed and EMBASE up to November 2016 to identify studies focusing on osseointegration of strontium-modified titanium implants following PRISMA criteria. The primary outcome was the percentage of bone-to-implant contact (BIC) around the implants with or without strontium-modified surface. Of the 1320 studies, 17 studies fulfilling the inclusion criteria were finally included. A random effect meta-analysis was conducted based on BIC in 17 studies, and the results demonstrated considerable heterogeneity (I² = 79%). A sensitivity analysis found that three studies using the same surface modification method were the major source of the heterogeneity. Therefore, exploratory subgroup analysis was performed. Subgroup one including 14 studies showed a standard mean differences (SMD) of 1.42 (95% CI, 1.13–1.71) with no heterogeneity (I² = 0.0%), while subgroup two including the other three studies showed a SMD of 9.49.95% CI, 7.51–11.47) with low heterogeneity (I² = 0.1%). Sr-modified implants in both subgroups showed significantly higher BIC than unmodified implants (P < 0.01). The results showed a statistically significant effect of Sr-modified titanium implant surfaces on osseointegration and bone apposition in animal models.
Introduction
The long-term success of endosseous implant mainly depends on osseointegration, which is defined as a direct contact between living bone and implant in histological sections. Albrektsson et al. defined six factors as pre-requisites for the establishment of osseointegration, implant material, implant design, implant surface, status of bone, surgical technique and the implant loading condition1.
Nowadays, titanium and its alloys have been widely applied for fabricating endosseous implant devices such as artificial knees, hip prosthesis and dental implants owing to its excellent biocompatibility, bio-inertness and adequate mechanical properties2. The evolution of clinical protocols have not only shortened treatment time but also expanded indications for implant therapy with significant progress of titanium surfaces. Physical, chemical, biological and topographical modifications have been proposed to accelerate bone healing and promote bone formation attempting to reach a rapid, long-living implant anchorage3–5. Currently, various studies have demonstrated surface modification with inorganic metal elements such as magnesium (Mg), zinc (Zn), strontium (Sr) incorporation could achieve rapid osseointegration and promote new bone formation6–8.
Strontium(Sr) aroused great attention clinically since strontium ranelate (SrRan) had been proved to have significant effect on reducing the risk of fracture in osteoporosis patients9,10. Sr, an essential trace element in human body, has been reported to enhance the osseointegration in vitro and in vivo. In vitro studies have found that Sr ion stimulates osteogenic differentiation of mesenchymal stem cells (MSCs) by activating Wnt signaling11,12. Sr exerts an inhibitory effect on osteoclast activity and differentiation through the activation of RANK/RANKL pathway and expression of osteoprotegerin (OPG)13,14. Moreover, the mechanism is believed to depend on the results of angiogenesis and osteogenesis that Sr facilitates osteogenic differentiation of BMSCs as well as promotes the angiogenic growth factors secretion, which can result in blood vessel formation15,16. In vivo studies showed that Sr could promote osseointegration both in Sr-loaded implants and via oral administration of SrRan8,17. In addition, the beneficial effect on osteogenesis was also observed in Sr-enriched applications such as CaSiO3 ceramics18, bioactive glasses19 and bone cement20.
However, due to the high cost of animal studies, most of the sample sizes in previous studies were limited. Moreover, the methods of adding Sr into implant surfaces varied. Meanwhile, limited high-quality evidence for effect of Sr-modified implant on bone apposition was available. Thus, a systematic review is highly in demand for evaluating the effect of Sr-modified titanium implants surface on enhancing osseointegration.
Therefore, the aim of present review was to systematically analyze the scientific literature reporting the efficacy of treating titanium surfaces with Sr on osseointegration of implants in animal experimental models. A parameter frequently used to quantify osseointegaration is bone-implant contact rate (BIC). It is defined as the ratio between the linear measurement of the surface of implant in direct contact with bone and the total length of the implant profile. As a crucial parameter of histomorphometry, BIC was selected as the primary outcome in the present review. In addition, the null hypothesis was no significant difference of osseointegration could be found between Sr-modified implants and unmodified implants.
Results
Study selection
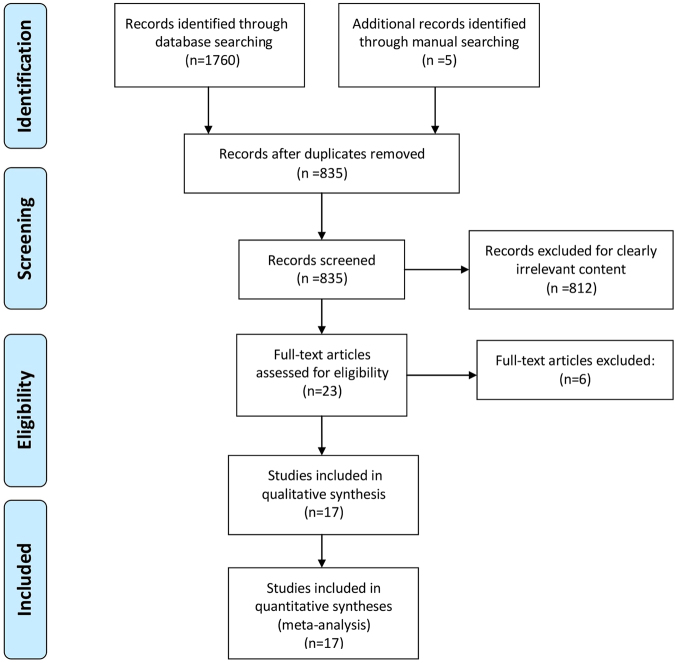
Electronic search showed a total number of 1760 titles, of which 835 titles and abstracts were retrieved for possible inclusion after automatic duplication removed. After manual searching bibliographies of the selected studies, 5 studies were added to full-text evaluation. 23 articles were selected for full text evaluation. Three studies were excluded due to no Sr-only experimental group21–23. Another three studies were excluded because BIC was not reported8,24,25. The final 17 studies26–42 were included in this systematic review. The search pathway was showed in Fig. 1. An overview on details about experimental details per study was given in Table 1.
Figure 1.
Search flowchart.
Table 1.
Characteristics of included studies.
| Author | Animal model | Control Groups | Strontium Incorporation Method | Follow-up | Analysis Methods | Outcome |
|---|---|---|---|---|---|---|
| Offermanns et al.26 | 30 OVX rats | Smooth-Ti | magnetron sputtering process | 6, 12 w | Histomorphometry | BIC BA |
| Zhang et al.26 | 6 beagle dogs | MAO-Ti | micro-arc oxidation | 6 w | Histomorphometry | BIC BA |
| Fan et al.28 | 16 male New Zealand white rabbits | SLA Ti | HTP with 0.02 mol/l Sr(OH)2·8 H2O solution | 3, 6 w | RTT Histomorphometry | RTV BIC BA |
| Dang et al.28 | 28 female rats | NT-40 cpTi | HTP with 0.02 M Sr(OH)2 60 mL solution. 200 °C for 1 or 3 h | 12 w | pull-out test Micro-CT Histomorphometry | BIC BV/TV, Tb.N Tb.Th, Tb.Sp, Tb.P.F maximal push-out force |
| Tao et al. 2016a | 50 female OVX rats | HA-Ti | electrochemical deposition | 12 w | pull-out test Micro-CT Histomorphometry | BIC BA BV/TV,Tb.N,Conn.D Tb.Th,Tb.Sp maximal push-out force |
| Tao et al. 2016b | 40 female OVX rats | HA-Ti | electrochemical deposition | 12 w | pull-out test Micro-CT Histomorphometry | BIC BA BV/TV, Tb.N, Conn.D Tb.Th, Tb.Sp maximal push-out force |
| Zhang et al.39 | 36 female OVX rats | HA-SLA | electrochemical deposition | 4, 8, 12 w | Histomorphometry | BIC BA |
| Li et al.35 | 40 female rats | Smooth-Ti | magnetron sputtering | 12 w | pull-out test Micro-CT Histomorphometry | BIC BV/TV, Tb.N, Tb.Th, Tb.Sp, maximal push-out force |
| Offermanns et al.40 | 30 female rats | Smooth-Ti | magnetron sputtering process | 4 w | Histomorphometry | BIC BA |
| Andersen et al.33 | 20 female rats | Smooth-Ti | magnetron co-sputtering | 4 w | Histomorphometry | BIC BA |
| Zhang et al.36 | 12 adult beagle dogs | HT-Ti–6Al–4 V | plasma spray techniques | 12 w | pull-out test Micro-CT Histomorphometry | BIC BA BV/TV, Tb.N, Tb.Th maximal push-out force |
| Park et al.6 | 10 male New Zealand white rabbits | SLActive Ti | HTP with mixed solution of SrO and NaOH dissolved in deionized water 180 °C for 2 h | 2 w | Histomorphometry RFA | BIC BA ISQ |
| Yan et al.11 | 30 adult rabbits | HA-Ti | micro-arc oxidation | 4, 12 w | pull-out test Micro-CT Histomorphometry | BIC BV/TV, Tb.N, Tb.Th, Tb.Sp, maximal push-out force |
| M. Ballo et al. 2012 | 20 male rats | HA-Ti | biomimetic process | 1, 4 w | Histomorphometry | BIC BA |
| FU et al.42 | 10 New Zealand White rabbits | HA-Ti | electrochemical deposition | 1, 4, 8 w | Histomorphometry | BIC BA |
| Li et al.31 | 20 female OVX rats | HA-Ti | Sol-gel dip coating with Sr(NO3)2 solution | 12 w | pull-out test Micro-CT Histomorphometry | BIC BA BV/TV, Tb.N, Conn.D Tb.Th, Tb.Sp, Tb.P.F maximal push-out force |
| Park et al.37 | 7 New Zealand White rabbits | Ti–6Al–4 V | Hydrothermal treatment | 4 w | RTV Histomorphometry | BIC BA RTV |
| Implants (n) | Implant Dimensions, D × L (mm) | Location of Implant Placement | Implant Shape | Sr-modified Implant Surface Characteristics | ||
| Ti (60) | 1.6 × 5 | Tibia | Cylindrical | Ti-Sr-O layer 2000 nm micro-/nano-structures | ||
| Ti alloy (12) | NR | Mandible | NR | Ti-Sr-O layer 1 μm micro-/nano-structures | ||
| cp Ti (64) | 4 × 8 | Tibia/ Femur | Screw | Ti-Sr-O layer micro-/nano-structures | ||
| cp Ti (112) | 3 × 6 | Tibia/ Femur | Screw Cylindrical | Sr-loaded nanotubes | ||
| Ti (80) | 1.2 × 15 | Femur | NR | Sr-HA coatings micro-/nano-size | ||
| Ti (20) | 1 × 20 | Femur | NR | Sr-HA coatings micro-/nano-size | ||
| SLA Ti (72) | 2 × 6 | Tibia | NR | Sr-HA coatings micro-/nano-size | ||
| Ti implants (80) | 1.5 × 3 | Femur | Screw | Ti-Sr-O layer 20–40 nm Sr-loaded nano-textured | ||
| Ti (60) | 1.1 × 5 | Femur | NR | Ti-Sr-O layer 1200 nm micro-/nano-structures | ||
| Ti (40) | 1.1 × 6 | Femur | Rod | Ti-Sr-O layer 1000 nm micro-/nano-structures | ||
| Ti–6Al–4 V (48) | 3 × 10 | Femur | Rod | Sr-HT coatings nano/micron hierarchical structure | ||
| SLA Ti (20) | 3.3 × 10 | Femur | Screw | Ti-Sr-O layer micro-/nano-structures | ||
| cp Ti (120) | 3.75 × 6 | Femur | Rod | Sr-HA coatings 32 μm micro-/nano-size | ||
| cp Ti (80) | 2 × 2.3 | Tibia | Rod | Sr-HA coatings micro-/nano-size | ||
| Ti (20) | 4.1 × 8 | Femur | Screw | Sr-HA coatings | ||
| Ti (50) | 1 × 12 | Tibia | Rod | Sr-HA coatings 898 ± 102 nm micro-/nano-size | ||
| Ti–6Al–4 V (64) | 2.4 × 8 | Tibia/ Femur | Screw | Ti-Sr-O layer 50 nm nanostructure | ||
OVX: ovariectomized; Micro-CT: microcomputed tomography; BIC: bone to implant contact; BA: bone area; MAO: micro-arc oxidation; RTT: removal torque test; RTV: removal torque value; cpTi: commercially pure titanium; BV/TV: bone volume/total volume; Tb.Sp: trabecular spacing; Tb.N: trabecular number; Tb.Th: trabecular thickness; Conn.D: the mean connective density; Tb.P.F: trabecular pattern factor; NT: nanotube; HTP: hydrothermal process; Sr: strontium; HA: hydroxyapatite; HT: hardystonite; SLA: sandblasted acid-etched
cp Ti: Commercially pure titanium; SLA: sandblasted acid-etched; HA: hydroxyapatites; HT: hardystonite; NR: not reported.
Risk of bias and quality assessment of included studies
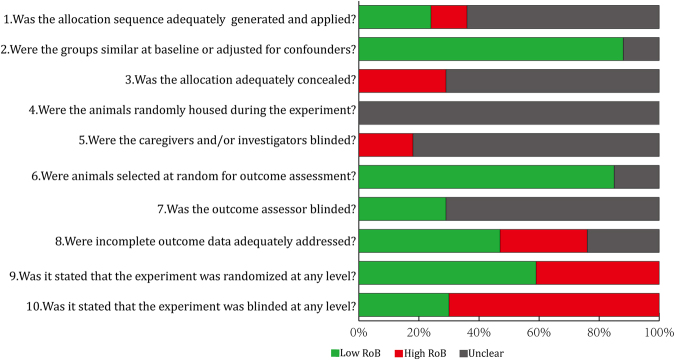
The results of the risk of bias evaluation of included studies were shown in Fig. 2. For items 9 and 10, 60% of included studies reported the experiment was randomized at some level, while 29% reported blinding at any level during the study.
Figure 2.
Risk of bias (RoB) measured using the Systematic Review Centre for Laboratory animal Experimentation (SYRCLE) RoB tool, averaged per item.
The ARRIVE criteria of included studies was shown in Table 2. The mean score of all studies was 17.2 (±1.98) out of a maximum of 24. All studies reported adequate information concerning title, abstract, introduction, ethical statement, species, surgical procedure, outcome evaluation, statistical analysis and results. Information regarding experimental animals housing conditions and study limitation were generally inadequate. Animals were randomly allocated to different treatment groups in nine studies (53%). Moreover, five studies reported blinding of assessors to test groups (29%). The 3Rs (in the results section) was not reported in any of studies.
Table 2.
Checklist of ARRIVE criteria reported by the included studies.
| No | ARRIVE criteria | Offer-manns et al. (2016) | Zhang et al. (2016) | Fan et al. (2016) | Dang et al. (2016) | Tao et al. (2016a) | Tao et al. (2016b) | Zhang et al. (2015) | Li et al. (2015) | Offer-manns et al. (2015) | Andersen et al. (2013) | Zhang et al. (2013) | Park et al. (2012) | Yan et al. (2012) | M.Ballo et al. (2012) | FU et al. (2012) | Li et al. (2010) | Park et al. (2010) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Title | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Abstract | ||||||||||||||||||
| 2 | Species | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 3 | Key finding | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Introduction | ||||||||||||||||||
| 4 | Background information | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 5 | Reasons for animal model | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 6 | Hypothesis | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 |
| Methods | ||||||||||||||||||
| 7 | Ethical statement | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 8 | Randomization of animal | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 |
| 9 | Blinding of assessor | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 |
| 10 | Anaesthesia | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 11 | Antibiotics | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 1 |
| 12 | Analgesia | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 13 | Surgical procedure | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 14 | Reporting species | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 15 | Housing conditions | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 16 | Implant randomization | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 1 |
| 17 | Statistical methods | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Results | ||||||||||||||||||
| 18 | Results reported | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 19 | Standard error/confidence interval | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Discussion | ||||||||||||||||||
| 20 | Interpretation | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 21 | Study limitations | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 1 |
| 22 | 3 Rs reported | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 23 | Relevance to humans | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 |
| 24 | Funding | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 |
| Total score | 18 | 16 | 18 | 18 | 18 | 19 | 18 | 18 | 18 | 15 | 18 | 13 | 18 | 16 | 13 | 21 | 17 | |
Bone to implant contact
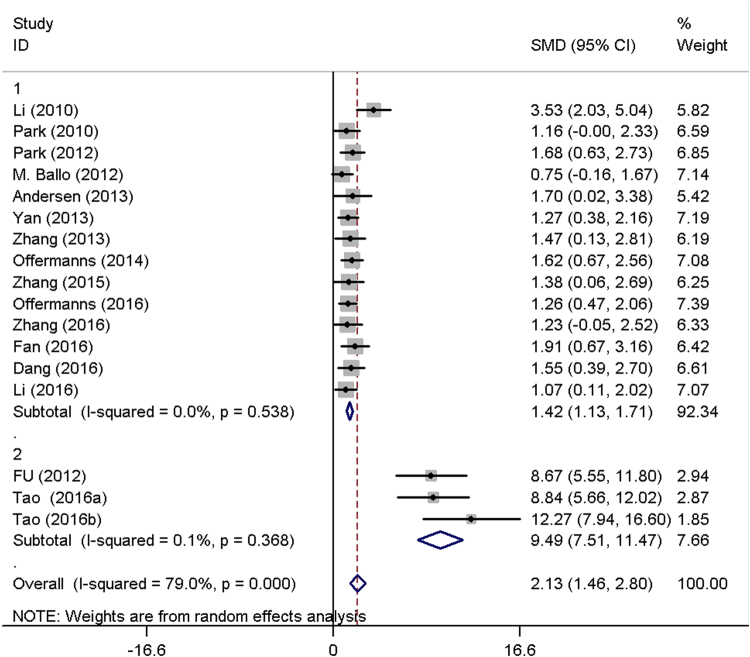
All included studies measured the effect on BIC around implants with or without Sr-enriched surface. A random effect meta-analysis was conducted based on BIC in 17 studies, and the overall results demonstrated considerable heterogeneity (I² = 79%). A sensitivity analysis found that three studies using the same surface modification method were the major source of the heterogeneity. Therefore, exploratory subgroup analysis was performed. The subgroup1 including 14 studies showed a standard mean differences (SMD) of 1.42 (95% CI, 1.13–1.71) with no heterogeneity (I² = 0.0%), while subgroup2 including the other three studies showed a SMD of 9.49 (95% CI, 7.51–11.47) with low heterogeneity (I² = 0.1%) (Fig. 3). The Sr-modified implants in both subgroups showed significantly higher BIC than unmodified implants (P < 0.01). However, high publication bias was found in the present study. (Begg, p = 0.039; Egger, p = 0.000) (Fig. 4.).
Figure 3.
Forest plots of bone to implant contact (BIC).
Figure 4.
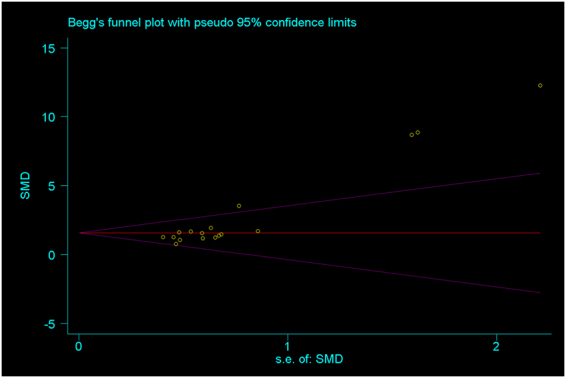
Begg’s funnel plot of included studies.
Bone area ratio
14 of 17 studies26–28,30–34,37–42 calculated the percentage of new bone area (BA) of peri-implants. No meta-analysis could be conducted due to the considerable heterogeneity. 10 studies reported that significant higher bone area could be observed around strontium modified implants than those implants without strontium incorporation. However, no significant difference in bone area was reported in other four studies28,30,37,42.
Micro-CT evaluation and biomechanical test
Micro-computed tomography (micro-CT) evaluation was performed in seven of included studies29,31,32,34–36. In quantitative assessment, four studies31,32,34,38 showed Sr-enhanced implants demonstrated stronger effect on all micro-CT parameters including bone volume per total volume (BV/TV), 3D bone distribution such as trabecular number (Tb.N), thickness (Tb.Th), and/or spacing (Tb.Sp) and/or the connective density (Conn.D) than control implants, while other three studies found no statistically significant changes in some parameters.
Nine of included studies28,29,31,32,34–38 reported the results of biomechanical test including pull-out test or removal torque testing. All studies revealed a significant increase in implant fixation, with removal torque value, the maximal push-out force and/or the ultimate shear strength markedly raised compared to control groups.
Discussion
The primary finding of this meta-analysis was that Sr-modified implant surfaces significantly increased the percentage of BIC. Therefore, the null hypothesis should be rejected.
To our knowledge, this is the first systematic review to assess the effect of Sr-modified implants on enhancing osseointegration and bone apposition in animal experimental studies. To quantify the potential effect of Sr-containing surfaces on peri-implant bone apposition, a meta-analysis of BIC was performed. Overall, the results of subgroup meta-analysis revealed that titanium implants with strontium incorporation demonstrated significantly better BIC than unmodified implants in small (rats, rabbits) and large (dogs) animals. The result verified the expectation in vitro studies that the Sr-containing titanium surface is expected to shorten bone healing period and enhance implant osseointegration43,44. Similarly, this result was in agreement with studies for appraising the efficiency of Sr-modified magnesium (Mg) based implant45,46. In these studies, Sr-enriched implant showed significantly higher percentage of BIC than that of pure Mg or Mg alloy based implant.
It is worth mentioning that subgroup two reported Sr-modified surfaces had significant effect on BIC and the difference between implants with and without strontium incorporation was more significant than that in subgroup one. The identical electrochemical deposition process was applied to incorporate strontium-substituted hydroxyapatite (Sr-HA) into surface in these three studies indicating the heterogeneity may be caused by method of surface modification. It has been reported that an electrochemical process can produce a homogeneous 2- to approximately 3 μm HA coating and nano-hydroxyapatite (nano-HA) on the metallic substrate surface47. Yang et al.48 reported that significant superiority of osseointegration and bone apposition was found when electrochemical deposition method was used. However, it is difficult to determine the best surface modification methods due to the limited information.
New bone area plays an important role in evaluating the osteoconductive property of biomaterials. The new mineralized bone tissue area inside all the implant threads was measured to evaluate the percentage of bone area. Several studies have demonstrated that Sr-containing biomaterials could increase new bone apposition. Studies for evaluating Sr-incorporated bioactive glass scaffolds and bone cement in impaired bone found that materials were covered by more new bone than unmodified groups49,50. In this review, ten studies using rat animal model reported significantly higher BA in Sr-modified implants than unmodified implants, while the other four studies used rabbit animal model reporting no significant difference in BA. The difference may be attributed to the different animal model, which implies different dynamics of bone formation especially in early healing intervals51. Therefore, additional preclinical and clinical studies should be performed to assess the effect of Sr-modified implant on bone apposition and osseointegration.
The 3-D micro-CT image clearly provides the information of bone–implant interface and trabecular microstructure of peri-implant bone tissue from both qualitative and quantitative perspectives. Seven included studies29,31,32,34–36,38 performed CT evaluation and four of them showed significantly improved BV/TV, Tb.N, Tb.Th, Tb.Sp and/or Conn.D. Other three studies found the parameters of CT evaluation were partially improved by Sr-modified titanium surface. Possible reasons that cause the disparity could be the different surface topography and Sr concentration of titanium implant surfaces. Furthermore, biomechanical testing further demonstrated a significant increase in implant fixation. It was apparent to be verified by the improved trabecular bone microarchitecture together with increased BIC and BA around the implant.
All the included studies reported that the improved implant osseointegration and bone apposition was attributed to the released Sr ions and modified surface topography. Actually, the exact mechanism regarding bone remodeling effect of Sr has not been clearly understood. Recently, it has been suggested that the possible mechanism of Sr relies on the calcium-sensing receptor (CaR) which is expressed in several types of pre-osteoblastic cells and bone marrow stromal cells52. Through a CaR-mediated mechanism, Sr is reported to increase bone apposition by promoting pre-osteoblastic cell proliferation and differentiation, reducing osteoclast differentiation, enhancing matrix mineralization52,53. In addition, the surface topography changed by strontium incorporation process also contributed to bone-implant integration. Three of included studies revealed that micro/nanoscale topography enhanced new bone apposition and osseointegration of Sr-modified implants or had a synergistic effect with released Sr ions28,29,35. Hence, in-depth investigations are required to isolate the pure and independent effect of strontium or surface topography on improving bone-implant integration.
The present systematic review had several limitations. Firstly, the follow-up of included studies ranged from 1 week to 12 weeks, so it remained unclear whether the osseointegration of Sr-incorporated Ti implants would be a stable anchorage, which could contribute to their long-term survival. Secondly, high publication bias (Begg, p = 0.039; Egger, p = 0.000) was found in Begg’s and Egger’s test. Therefore, the results need to be interpreted with caution. Thirdly, none of the implants included in the present study were loaded. Thus, future studies should evaluate the effect of Sr-incorporated Ti implants under loading conditions.
Conclusion
Based on available evidence so far, it can be concluded that Sr-modified titanium implants could enhance osseointegration and new bone formation of peri-implant area in animal models. Nonetheless, future clinical investigations are needed to verify the safety and effectiveness of Sr-modified implants.
Methods
PICO
According to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines54, a specific question was identified based on the Participants, Interventions, Control, Outcomes (PICO) principle. The focused question was, “Does incorporating strontium into titanium implant surfaces influence the osseointegration?”
(P) Participants: Subjects received endosseous implantation.
(I) Interventions: Implants with strontium incorporation.
(C) Control: Implants without strontium incorporation.
(O) Outcome: BIC, BA and results from biomechanical test and micro-CT evaluation.
Search strategy and study selection
Databases of PubMed and EMBASE were searched up to November 2016 for relevant articles published, using search terms “strontium”, in combination with “osseointegration”, “bone apposition”, “osteogenic”, “osteogenesis”, “new bone formation”, “bone to implant contact” and “bone regeneration”. Additionally, bibliographies of the selected studies and relevant review articles were also scrutinized for cross-references. Titles and abstracts of searches were initially screened by two authors (SJY, LY). Uncertainty in the determination of eligibility was resolved by discussion. Two authors reviewed full-text articles independently and final inclusion was based on the inclusion criteria.
Inclusion and exclusion criteria
The inclusion criteria for the study selection were:
Studies regarding titanium implants modified with strontium;
Studies reporting the percentage of bone-to-implant contact of Sr-modified and unmodified implants;
Studies with a minimum of 3 implants/group;
The exclusion criteria for the study selection were:
In vitro studies;
Studies assessing the combined effect of Sr and other inorganic elements (e.g. Ag, phosphate) modified surface without strontium-only test group.
Risk of bias and quality assessment
The risk of bias (RoB) of included studies was assessed using the SYRCLE RoB tool for animal studies55. The tool, which aims to assess methodological quality, was adapted to appraise bias in animal studies. RoB was evaluated by providing a response of “high”, “low” or “unclear” in each of the 10 items. As reported in a previous review, a modified RoB tool was used in which items 9 and 10 were adjusted to include information on whether the experiment was randomized or blinded at any level (Fig. 2.).
Reporting quality of the included studies was assessed based on a modified ARRIVE guidelines in which a checklist of 24 items was included56. Each item was judged as “0” (not reported) or “1” (reported). The total score of each of included studies was also recorded (Table 2).
Data extraction
Two independent reviewers (SJY, LY) extracted data from the full-texts of selected articles. General information, animal parameters (total number, species), methods of strontium incorporation, evaluation time points, analysis methods and outcomes and implant parameters (total number, material, length, diameter, shape, location and surface characteristics of test and control implants) were retrieved. The primary outcome and secondary outcomes were extracted (Tables 3 and 4). If data were only expressed graphically, numerical values were requested from the authors, and if a response was not received, digital ruler software was used to measure graphical data (ImageJ, National Institutes of Health, Bethesda, MD).
Table 3.
Micro-CT values of included studies.
| Parameters | Dang et al. (2016) | Tao et al. (2016) | Tao et al. (2016) | Li et al. (2015) | Zhang et al. (2013) | Yan et al. (2013) | Li et al. (2010) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | T | C | T | C | T | C | T | C | T | C | T | C | T | |
| BV/TV (%) | 16.7 ± 0.8 | 15.9 ± 0.2 | 23.8 ± 1.2 | 40.2 ± 2.4* | 23.8 ± 1.2 | 28.6 ± 1.2* | 36.4 ± 3.6 | 39.8 ± 5.3* | 27.0 ± 6.2 | 43.2 ± 6.4** | 38.6 ± 2.9 | 42.4 ± 3.3** | 24.7 ± 4.9 | 42.9 ± 6.7* |
| Tb.N(mm-1) | 4.1 ± 0.1 | 4.3 ± 0.0 | 226.2 ± 7.3 | 503.9 ± 15.4* | 226.2 ± 7.3 | 382.2 ± 13.8* | 3.3 ± 0.2 | 3.8 ± 0.2 | 2.1 ± 0.4 | 3.7 ± 0.6** | 2.8 ± 0.3 | 3.2. ± 0.2 | 2.3 ± 0.4 | 4.9 ± 0.7** |
| Tb.Th (μm) | 0.044 ± 0.0 | 0.046 ± 0.0 | 77.6 ± 3.7 | 116.3 ± 4.1* | 77.6 ± 3.7 | 90.4 ± 3.5* | 108.2 ± 19.4 | 132.5 ± 21.1 | 163.1 ± 26.9 | 225.9 ± 34.3** | 140.0 ± 10.4 | 148.3 ± 22.7 | 80.2 ± 9.7 | 102.3 ± 12.6* |
| Tb.Sp (μm) | 0.21 ± 0.01 | 0.19 ± 0.0 | 428.98 ± 25.1 | 291.59 ± 10.9* | 428.98 ± 25.1 | 364.84 ± 13.8* | 276.6 ± 52.8 | 200.1 ± 51.2 | NR | 220.1 ± 32.4 | 183.9 ± 27.3** | 425.8 ± 50.2 | 342.1 ± 42.4* | |
| Conn.D(mm-3) | NR | 25.19 ± 1.1 | 42.97 ± 2.4* | 25.19 ± 1.1 | 29.03 ± 1.1* | NR | NR | NR | 25.9 ± 3.8 | 40.3 ± 6.7* | ||||
| Tb.P.F(mm-1) | 13.5 ± 0.3 | 11.2 ± 0.5** | NR | NR | NR | NR | NR | NR | ||||||
| Number of implants/group | 4 | 9 | 10 | 10 | 6 | 12 | 10 | |||||||
C: control group; T: test group; BV/TV: bone volume/total volume; Tb.Sp: trabecular spacing; Tb.N: trabecular number; Tb.Th: trabecular thickness; Conn.D: the mean connective density; Tb.P.F: trabecular pattern factor; NR: not reported; Data were expressed as mean ± SD; *p Value < 0.05; **p Value < 0.01.
Table 4.
Bone-to-Implant Contact (BIC) Values.
| Author | Year | Number of implants per group | BIC | BA | Biomechanical test | |||
|---|---|---|---|---|---|---|---|---|
| control | test | control | test | control | test | |||
| Offermanns et al.# | 2016 | 15 | 65.2 ± 10.4 | 78.2 ± 9.6* | 23.8 ± 4.2 | 44.6 ± 9.4* | NR | |
| Zhang et al. | 2016 | 6 | 49.6 ± 6.5 | 58.7 ± 7.1** | 29.5 ± 8.6 | 60.8 ± 10.3** | NR | |
| Fan et al. | 2016 | 8 | 64 ± 5.9 | 77.1 ± 7.0* | 6.3 ± 6.5 | 0.8 ± 1.7 | ★41.1 ± 8.2 | ★56.8 ± 18.6* |
| Dang et al. | 2016 | 8 | 59.5 ± 3.2 | 63.2 ± 0.1* | NR | 10.8 ± 1.2 | 24.6 ± 2.4** | |
| Tao et al. | 2016a | 10 | 17.2 ± 2.0 | 37.1 ± 2.3* | 18.3 ± 0.9 | 33.7 ± 2.1* | 121.2 ± 11.4 | 168.9 ± 22.2* |
| Tao et al. | 2016b | 10 | 34.6 ± 0.7 | 62.5 ± 3.0* | 31.3 ± 0.8 | 47.32 ± 2.3* | 126.3 ± 12.3 | 219.5 ± 21.8* |
| Zhang et al. | 2015 | 6 | 51 ± 6.6 | 58.6 ± 2.9* | 28.6 ± 6.2 | 44.5 ± 9.7* | NR | |
| Li et al. | 2015 | 10 | 39.7 ± 6.0 | 46.1 ± 5.5* | NR | 108.9 ± 46.5 | 136.9 ± 21.7* | |
| Offermanns et al.# | 2015 | 12 | 27.8 ± 1.7 | 45.6 ± 2.2** | 17.8 ± 2.2 | 24.4 ± 7.8* | NR | |
| Andersen et al.# | 2013 | 5 | 0 ± 1.2 | 18 ± 14.3** | 0 ± 5.1 | 22 ± 7.3* | NR | |
| Zhang et al. | 2013 | 6 | 37 ± 8.7 | 51.2 ± 9.1* | 40.6 ± 5.7 | 55.2 ± 9.8* | 229.08 ± 59.0 | 388.84 ± 100.5** |
| Park et al. | 2012 | 10 | 75.4 ± 5.4 | 84.6 ± 5.1** | 57.3 ± 9.4 | 60.9 ± 10 | NR | |
| Yan et al.# | 2012 | 12 | 58.8 ± 4.7 | 65.1 ± 4.9* | NR | 119.23 ± 3.9 | 142.31 ± 9.6* | |
| M. Ballo et al. | 2012 | 10 | 26.4 ± 10.7 | 35.7 ± 12.9* | 25.7 ± 2.9 | 32.9 ± 9.3* | NR | |
| FU et al. | 2012 | 10 | 72.2 ± 12.8 | 87.7 ± 2.8* | 9.3 ± 0.5 | 9.8 ± 1.5 | NR | |
| Park et al. | 2010 | 7 | 47.3 ± 10.4 | 60.1 ± 10.2* | 46.7 ± 10.7 | 46.6 ± 6.5 | ★1.6 ± 0.9 | ★2.6 ± 1.2* |
| Li et al. | 2010 | 10 | 42.5 ± 4.1 | 63 ± 6.7* | 25.2 ± 2.2 | 42.73 ± 4.3* | 38.7 ± 5.3 | 80.2 ± 9.9** |
Values are shown as mean ± SD; #values are presented as median value ± interquartile range;
BIC, BA values in %; pull-out test values in N; ★removal torque testing values in Ncm;
*p Value < 0.05; **p Value < 0.01; NR: not reported.
Statistical analysis
The primary and secondary outcomes were present in descriptive statistics. The standardized mean differences, together with 95% confidence intervals, were analyzed using random-effect model. Heterogeneity was tested using the I 2 statistic to describe the proportion of total variation. Values of 25, 50 and 75% were regarded as low, moderate and considerable heterogeneity, respectively57. When the value was >50%, qualitative analysis was conducted. A forest plot was generated, and heterogeneity was calculated by use of the statistical software package STATA (v11.0; StataCorp, College Station, TX). A p value < 0.05 was considered to indicate statistical significance, unless specified otherwise.
Potential publication bias was assessed using Begg’s funnel plots and Egger’s test at the p < 0.10 level of significance.
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (81500892).
Author Contributions
H.C.L. contributed the concept and study design. J.Y.S. and Y.L. screened the literature, selected studies for exclusion and inclusion, performed data extraction and meta-analysis and drafted the manuscript. Y.X.G. contributed part of figures of this manuscript and S.C.Q. contributed the statistical consultation. In addition, X.M.Z. critically reviewed the manuscript. All authors reviewed and approved the final manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Junyu Shi and Yuan Li contributed equally to this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Albrektsson T, Branemark PI, Hansson HA, Lindstrom J. Osseointegrated titanium implants. Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta orthopaedica Scandinavica. 1981;52:155–170. doi: 10.3109/17453678108991776. [DOI] [PubMed] [Google Scholar]
- 2.Annunziata M, Guida L. The Effect of Titanium Surface Modifications on Dental Implant Osseointegration. Frontiers of oral biology. 2015;17:62–77. doi: 10.1159/000381694. [DOI] [PubMed] [Google Scholar]
- 3.Kloss FR, et al. The role of oxygen termination of nanocrystalline diamond on immobilisation of BMP-2 and subsequent bone formation. Biomaterials. 2008;29:2433–2442. doi: 10.1016/j.biomaterials.2008.01.036. [DOI] [PubMed] [Google Scholar]
- 4.Schwarz F, et al. Potential of chemically modified hydrophilic surface characteristics to support tissue integration of titanium dental implants. Journal of biomedical materials research. Part B, Applied biomaterials. 2009;88:544–557. doi: 10.1002/jbm.b.31233. [DOI] [PubMed] [Google Scholar]
- 5.Yazici H, et al. Biological response on a titanium implant-grade surface functionalized with modular peptides. Acta biomaterialia. 2013;9:5341–5352. doi: 10.1016/j.actbio.2012.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Park JW, An CH, Jeong SH, Suh JY. Osseointegration of commercial microstructured titanium implants incorporating magnesium: a histomorphometric study in rabbit cancellous bone. Clinical oral implants research. 2012;23:294–300. doi: 10.1111/j.1600-0501.2010.02144.x. [DOI] [PubMed] [Google Scholar]
- 7.Li X, et al. Effect of zinc ions on improving implant fixation in osteoporotic bone. Connective tissue research. 2013;54:290–296. doi: 10.3109/03008207.2013.813495. [DOI] [PubMed] [Google Scholar]
- 8.Liang Y, et al. Strontium coating by electrochemical deposition improves implant osseointegration in osteopenic models. Experimental and therapeutic medicine. 2015;9:172–176. doi: 10.3892/etm.2014.2038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Deeks ED, Dhillon S. Strontium ranelate: a review of its use in the treatment of postmenopausal osteoporosis. Drugs. 2010;70:733. doi: 10.2165/10481900-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 10.Reginster JY, Deroisy R, Jupsin I. Strontium ranelate: a new paradigm in the treatment of osteoporosis. Drugs of Today. 2004;39:89. doi: 10.1358/dot.2003.39.2.799416. [DOI] [PubMed] [Google Scholar]
- 11.Yang F, et al. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating Wnt/catenin signaling. Stem cells (Dayton, Ohio) 2011;29:981–991. doi: 10.1002/stem.646. [DOI] [PubMed] [Google Scholar]
- 12.Saidak Z, Marie PJ. Strontium signaling: molecular mechanisms and therapeutic implications in osteoporosis. Pharmacology & therapeutics. 2012;136:216–226. doi: 10.1016/j.pharmthera.2012.07.009. [DOI] [PubMed] [Google Scholar]
- 13.Peng S, et al. The cross-talk between osteoclasts and osteoblasts in response to strontium treatment: involvement of osteoprotegerin. Bone. 2011;49:1290–1298. doi: 10.1016/j.bone.2011.08.031. [DOI] [PubMed] [Google Scholar]
- 14.Yamaguchi M, Weitzmann MN. The intact strontium ranelate complex stimulates osteoblastogenesis and suppresses osteoclastogenesis by antagonizing NF-kappaB activation. Molecular and cellular biochemistry. 2012;359:399–407. doi: 10.1007/s11010-011-1034-8. [DOI] [PubMed] [Google Scholar]
- 15.Lin K, et al. Enhanced osteoporotic bone regeneration by strontium-substituted calcium silicate bioactive ceramics. Biomaterials. 2013;34:10028–10042. doi: 10.1016/j.biomaterials.2013.09.056. [DOI] [PubMed] [Google Scholar]
- 16.Bose S, Fielding G, Tarafder S, Bandyopadhyay A. Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics. Trends in biotechnology. 2013;31:594–605. doi: 10.1016/j.tibtech.2013.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li Y, et al. Strontium ranelate treatment enhances hydroxyapatite-coated titanium screws fixation in osteoporotic rats. Journal of Orthopaedic Research Official Publication of the Orthopaedic Research Society. 2010;28:578–582. doi: 10.1002/jor.21169. [DOI] [PubMed] [Google Scholar]
- 18.Wu C, Ramaswamy Y, Kwik D, Zreiqat H. The effect of strontium incorporation into CaSiO3 ceramics on their physical and biological properties. Biomaterials. 2007;28:3171–3181. doi: 10.1016/j.biomaterials.2007.04.002. [DOI] [PubMed] [Google Scholar]
- 19.Santocildes-Romero ME, et al. The osteogenic response of mesenchymal stromal cells to strontium-substituted bioactive glasses. Journal of tissue engineering and regenerative medicine. 2015;9:619–631. doi: 10.1002/term.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schumacher M, Lode A, Helth A, Gelinsky M. A novel strontium(II)-modified calcium phosphate bone cement stimulates human-bone-marrow-derived mesenchymal stem cell proliferation and osteogenic differentiation in vitro. Acta biomaterialia. 2013;9:9547–9557. doi: 10.1016/j.actbio.2013.07.027. [DOI] [PubMed] [Google Scholar]
- 21.Cheng H, et al. Strontium (Sr) and silver (Ag) loaded nanotubular structures with combined osteoinductive and antimicrobial activities. Acta biomaterialia. 2016;31:388–400. doi: 10.1016/j.actbio.2015.11.046. [DOI] [PubMed] [Google Scholar]
- 22.Park JW. Increased bone apposition on a titanium oxide surface incorporating phosphate and strontium. Clinical oral implants research. 2011;22:230–234. doi: 10.1111/j.1600-0501.2010.01974.x. [DOI] [PubMed] [Google Scholar]
- 23.Newman SD, et al. Enhanced osseous implant fixation with strontium-substituted bioactive glass coating. Tissue engineering. Part A. 2014;20:1850–1857. doi: 10.1089/ten.tea.2013.0304. [DOI] [PubMed] [Google Scholar]
- 24.Lovati AB, et al. In vivoevaluation of bone deposition in macroporous titanium implants loaded with mesenchymal stem cells and strontium-enriched hydrogel. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2015;103:448–456. doi: 10.1002/jbm.b.33228. [DOI] [PubMed] [Google Scholar]
- 25.Liu P, et al. Entangled titanium fibre balls combined with nano strontium hydroxyapatite in repairing bone defects. Medical principles and practice: international journal of the Kuwait University, Health Science Centre. 2014;23:264–270. doi: 10.1159/000359951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Offermanns V, et al. Bone regenerating effect of surface-functionalized titanium implants with sustained-release characteristics of strontium in ovariectomized rats. International journal of nanomedicine. 2016;11:2431–2442. doi: 10.2147/IJN.S101673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang W, et al. A strontium-incorporated nanoporous titanium implant surface for rapid osseointegration. Nanoscale. 2016;8:5291–5301. doi: 10.1039/C5NR08580B. [DOI] [PubMed] [Google Scholar]
- 28.Fan YP, et al. Positive effect of strontium-oxide layer on the osseointegration of moderately rough titanium surface in non-osteoporotic rabbits. Clinical oral implants research. 2017;28:911–919. doi: 10.1111/clr.12897. [DOI] [PubMed] [Google Scholar]
- 29.Dang Y, et al. In vivo osseointegration of Ti implants with a strontium-containing nanotubular coating. International journal of nanomedicine. 2016;11:1003–1011. doi: 10.2147/IJN.S102552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Park JW, Kwon TG, Suh JY. The relative effect of surface strontium chemistry and super-hydrophilicity on the early osseointegration of moderately rough titanium surface in the rabbit femur. Clinical oral implants research. 2013;24:706–709. doi: 10.1111/j.1600-0501.2012.02444.x. [DOI] [PubMed] [Google Scholar]
- 31.Li Y, et al. The effect of strontium-substituted hydroxyapatite coating on implant fixation in ovariectomized rats. Biomaterials. 2010;31:9006–9014. doi: 10.1016/j.biomaterials.2010.07.112. [DOI] [PubMed] [Google Scholar]
- 32.Tao ZS, et al. A comparative study of zinc, magnesium, strontium-incorporated hydroxyapatite-coated titanium implants for osseointegration of osteopenic rats. Materials science & engineering. C, Materials for biological applications. 2016;62:226–232. doi: 10.1016/j.msec.2016.01.034. [DOI] [PubMed] [Google Scholar]
- 33.Andersen OZ, et al. Accelerated bone ingrowth by local delivery of strontium from surface functionalized titanium implants. Biomaterials. 2013;34:5883–5890. doi: 10.1016/j.biomaterials.2013.04.031. [DOI] [PubMed] [Google Scholar]
- 34.Tao ZS, et al. A comparative study of strontium-substituted hydroxyapatite coating on implant’s osseointegration for osteopenic rats. Medical & biological engineering & computing. 2016;54:1959–1968. doi: 10.1007/s11517-016-1494-9. [DOI] [PubMed] [Google Scholar]
- 35.Li Y, et al. Effects of a micro/nano rough strontium-loaded surface on osseointegration. International journal of nanomedicine. 2015;10:4549–4563. doi: 10.2147/IJN.S84398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yan J, Sun JF, Chu PK, Han Y, Zhang YM. Bone integration capability of a series of strontium-containing hydroxyapatite coatings formed by micro-arc oxidation. Journal of biomedical materials research. Part A. 2013;101:2465–2480. doi: 10.1002/jbm.a.34548. [DOI] [PubMed] [Google Scholar]
- 37.Park JW, et al. Osteoblast response and osseointegration of a Ti-6Al-4V alloy implant incorporating strontium. Acta biomaterialia. 2010;6:2843–2851. doi: 10.1016/j.actbio.2010.01.017. [DOI] [PubMed] [Google Scholar]
- 38.Zhang W, et al. The synergistic effect of hierarchical micro/nano-topography and bioactive ions for enhanced osseointegration. Biomaterials. 2013;34:3184–3195. doi: 10.1016/j.biomaterials.2013.01.008. [DOI] [PubMed] [Google Scholar]
- 39.Zhang, J., Liu, L., Zhao, S., Wang, H. & Yang, G. Characterization and In Vivo Evaluation of Trace Element-Loaded Implant Surfaces in Ovariectomized Rats. The International journal of oral & maxillofacial implants, doi:10.11607/jomi.3906 (2015). [DOI] [PubMed]
- 40.Offermanns V, et al. Enhanced osseointegration of endosseous implants by predictable sustained release properties of strontium. Journal of biomedical materials research. Part B, Applied biomaterials. 2015;103:1099–1106. doi: 10.1002/jbm.b.33279. [DOI] [PubMed] [Google Scholar]
- 41.Ballo AM, et al. Bone tissue reactions to biomimetic ion-substituted apatite surfaces on titanium implants. Journal of the Royal Society, Interface. 2012;9:1615–1624. doi: 10.1098/rsif.2011.0808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fu DL, Jiang QH, He FM, Yang GL, Liu L. Fluorescence microscopic analysis of bone osseointegration of strontium-substituted hydroxyapatite implants. Journal of Zhejiang University. Science. B. 2012;13:364–371. doi: 10.1631/jzus.B1100381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yang HW. Osteogenesis of bone marrow mesenchymal stem cells on strontium-substituted nano-hydroxyapatite coated roughened titanium surfaces. Journal of Tongji University. 2015;8:257–264. [PMC free article] [PubMed] [Google Scholar]
- 44.Ni GX, Yao ZP, Huang GT, Liu WG, Lu WW. The effect of strontium incorporation in hydroxyapatite on osteoblasts in vitro. Journal of Materials Science: Materials in Medicine. 2011;22:961–967. doi: 10.1007/s10856-011-4264-0. [DOI] [PubMed] [Google Scholar]
- 45.Mushahary D, et al. Zirconium, calcium, and strontium contents in magnesium based biodegradable alloys modulate the efficiency of implant-induced osseointegration. International journal of nanomedicine. 2013;8:2887–2902. doi: 10.2147/IJN.S47378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tie D, et al. An in vivo study on the metabolism and osteogenic activity of bioabsorbable Mg-1Sr alloy. Acta biomaterialia. 2016;29:455–467. doi: 10.1016/j.actbio.2015.11.014. [DOI] [PubMed] [Google Scholar]
- 47.Ye W, Wang XX. Morphologies of Hydroxyapatite Crystal Deposited on Titanium Surface with Electrochemical Technique. Key Engineering Materials. 2007;330-332:601–604. doi: 10.4028/www.scientific.net/KEM.330-332.601. [DOI] [Google Scholar]
- 48.Yang GL, He FM, Hu JA, Wang XX, Zhao SF. Effects of biomimetically and electrochemically deposited nano-hydroxyapatite coatings on osseointegration of porous titanium implants. Oral surgery, oral medicine, oral pathology, oral radiology, and endodontics. 2009;107:782–789. doi: 10.1016/j.tripleo.2008.12.023. [DOI] [PubMed] [Google Scholar]
- 49.Wu CC, Kuo CL, Fan FY, Yang KC. Strontium-impregnated bioabsorbable composite for osteoporotic fracture fixation. Journal of biomedical materials research. Part A. 2015;103:3355–3363. doi: 10.1002/jbm.a.35471. [DOI] [PubMed] [Google Scholar]
- 50.Wei L, et al. A comparative study of Sr-incorporated mesoporous bioactive glass scaffolds for regeneration of osteopenic bone defects. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2014;25:2089–2096. doi: 10.1007/s00198-014-2735-0. [DOI] [PubMed] [Google Scholar]
- 51.Pearce AI, Richards RG, Milz S, Schneider E, Pearce SG. Animal models for implant biomaterial research in bone: a review. European cells & materials. 2007;13:1–10. doi: 10.22203/eCM.v013a01. [DOI] [PubMed] [Google Scholar]
- 52.Chattopadhyay N, Quinn SJ, Kifor O, Ye C, Brown EM. The calcium-sensing receptor (CaR) is involved in strontium ranelate-induced osteoblast proliferation. Biochem Pharmacol. 2007;74:438–447. doi: 10.1016/j.bcp.2007.04.020. [DOI] [PubMed] [Google Scholar]
- 53.Brown EM. Is the calcium receptor a molecular target for the actions of strontium on bone? Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. 2003;14(Suppl 3):S25–34. doi: 10.1007/s00198-002-1343-6. [DOI] [PubMed] [Google Scholar]
- 54.Vrabel M. Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Oncology nursing forum. 2015;42:552–554. doi: 10.1188/15.ONF.552-554. [DOI] [PubMed] [Google Scholar]
- 55.Hooijmans CR, et al. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14:43. doi: 10.1186/1471-2288-14-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Stadlinger B, Pourmand P, Locher MC, Schulz MC. Systematic review of animal models for the study of implant integration, assessing the influence of material, surface and design. Journal of clinical periodontology. 2012;39(Suppl 12):28–36. doi: 10.1111/j.1600-051X.2011.01835.x. [DOI] [PubMed] [Google Scholar]
- 57.Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ (Clinical research ed.)327, 557-560, doi:10.1136/bmj.327.7414.557 (2003). [DOI] [PMC free article] [PubMed]