Abstract
Introduction
Quantitative CT (QCT) imaging-based metrics have quantified disease alterations in asthma and chronic obstructive pulmonary disease (COPD), respectively. We seek to characterise the similarity and disparity between these groups using QCT-derived airway and parenchymal metrics.
Methods
Asthma and COPD subjects (former-smoker status) were selected with a criterion of post-bronchodilator FEV1 <80%. Healthy non-smokers were included as a control group. Inspiratory and expiratory QCT images of 75 asthmatic, 215 COPD and 94 healthy subjects were evaluated. We compared three segmental variables: airway circularity, normalised wall thickness and normalised hydraulic diameter, indicating heterogeneous airway shape, wall thickening and luminal narrowing, respectively. Using an image registration, we also computed six lobar variables including per cent functional small-airway disease, per cent emphysema, tissue fraction at inspiration, fractional-air-volume change, Jacobian and functional metric characterising anisotropic deformation.
Results
Compared with healthy subjects, both asthma and COPD subjects demonstrated a decreased airway circularity especially in large and upper lobar airways, and a decreased normalised hydraulic diameter in segmental airways. Besides, COPD subjects had more severe emphysema and small-airway disease, as well as smaller regional tissue fraction and lung deformation, compared with asthmatic subjects. The difference of emphysema, small-airway disease and tissue fraction between asthma and COPD was more prominent in upper and middle lobes.
Conclusions
Patients with asthma and COPD, with a persistent FEV1 <80%, demonstrated similar alterations in airway geometry compared with controls, but different degrees of alterations in parenchymal regions. Density-based metrics measured at upper and middle lobes were found to be discriminant variables between patients with asthma and COPD.
Keywords: emphysema, functional small airway disease, image registration, quantitative computed tomography, airway luminal narrowing
Key messages.
We demonstrate similarity and disparity of structure and function between patients with asthma and chronic obstructive pulmonary disease (COPD) with chronic functional alteration using sensitively measured imaging metrics.
Image-derived functional alterations of asthmatics were regionally prominent in lower lobes, while those of patients with COPD were found in whole lungs.
Introduction
Asthma is a disease with functional airway reversibility through the aid of inhaled corticosteroids and bronchodilator, whereas chronic obstructive pulmonary disease (COPD) is a disease with persistent airflow limitation.1–3 According to recent reports,4 5 around 15%–45% of patients with COPD may have asthma–COPD overlap, so-called ACO. In previous studies,6 7 some asthmatic patients were of neutrophilic dominance with chronic airway functional alteration, while some patients with COPD were of eosinophilic dominance with airway reversibility.8 9 Thus, objective differentiation of the two populations is essential for proper treatments. Meanwhile, two National Institutes of Health (NIH)-supported multicentre studies, SubPopulations and InteRmediate Outcome Measures In COPD Study (SPIROMICS)10 and Severe Asthma Research Program (SARP),11 have been established to identify genetic, environmental and clinical phenotypes for COPD and asthma, respectively. SARP excluded patients with a smoking history, while SPIROMICS, except healthy subjects, excluded never-smokers. By the study designs, the recruited subjects are less likely to be ACO. With these populations, we pursue to investigate the imaging-based similarity and disparity between two respiratory diseases.
Quantitative CT (QCT) imaging has successfully identified unique structural and functional phenotypes for asthma and COPD, respectively. For instance, Aysola et al 12 found that asthmatics had an increase of wall area percentage depending on severity, and Busacker et al 13 found an increase of air trapping in severe asthmatics. Using the same imaging datasets from SARP, Choi et al 14 15 demonstrated that existing imaging-based metrics were problematic due to intersubject and intersite variability, resulting in inaccurate estimation. To address these issues, they developed a new air-trapping measure and new normalisation schemes for luminal hydraulic diameter and wall thickness.14 15 These studies demonstrated that on average severe asthmatics were characterised by airway narrowing, wall thickening and air trapping. In addition, with an image registration technique, they demonstrated regional alterations of deformational metrics in severe asthmatics.16 The sensitive imaging-based metrics derived from multiple studies were then integrated to identify clinically meaningful subgroups of asthma.17
In patients with COPD, structural and functional alterations have been assessed by QCT imaging-based variables including luminal diameter,18 wall thickness,19 air trapping (or functional small-airway disease) and emphysema. Existing assessment of air trapping in COPD was also problematic because air trapping at expiration contains some portion of emphysema at inspiration. Therefore, Galban et al 20 employed an image registration technique to dissociate the portion of emphysema from air trapping, allowing for characterisation of three subgroups, that is, emphysema-dominant, functional small-airway disease-dominant and normal groups. In addition to the CT density mapping, image registration provided local deformational metrics including air-volume change, the determinant of Jacobian (Jacobian; a measure of volume change), anisotropic deformation index (ADI; a measure of the magnitude of directional preference in volume change), slab rod index (a measure of the nature of directional preference in volume change) and more.21 Bodduluri et al 22 used the image registration metrics to perform a supervised learning for the purpose of distinguishing patients with COPD from non-COPD subjects. Further, Smith et al 19 compared wall thicknesses of patients with COPD and non-COPD subjects, and found thinner airway walls in patients with COPD than non-COPD subjects when wall thickness is compared at the same location.
QCT imaging-based variables have been employed in a variety of studies to identify local or global alterations in airway dimension and lobar function. Although there are several studies to compare airway structure or lung function between asthma and COPD,23–25 the numbers of subjects under investigation were limited. Therefore, this study aims to investigate the similarity and disparity between large cohorts of asthma and COPD subjects acquired from SARP and SPIROMICS, especially those with overlapping clinical symptoms of chronic functional alterations measured by post-bronchodilator FEV1 <80%. In order to assess structure and function at global and lobar levels, we employed multiscale imaging-based variables,14–17 including local airway structural variables at inspiration scan, and lobar/global functional variables at expiration scan. We further employed image registration metrics including Jacobian, ADI and functional small-airway disease percentage (fSAD%) and emphysematous lung percentage (Emph%).
Methods
Inclusion criteria and QCT imaging data
We employed QCT imaging data from SARP and SPIROMICS. Both populations used a similar imaging protocol which has been described by Sieren et al 26 and were approved by the respective institutional review boards. We selected subjects who demonstrated post-bronchodilator FEV1 %predicted values <80% to focus on subjects with airflow obstruction. In SARP data, none of the asthmatics were current smokers as one of the inclusion criteria was ‘no smoking within the past five years and <5 pack-years of smoking’.27 SPIROMICS, except for healthy controls, excluded non-smokers, and in this study only former smokers were included using the criterion of ‘not currently smoking as of 1 month ago’.10 The numbers of asthmatic and COPD subjects meeting these criteria were 75 and 215. In addition, 94 healthy subjects without smoking history including 51 SARP and 43 SPIROMICS subjects were used as a control group (table 1). There are more imaging data in SARP and SPIROMICS, but the imaging data from eight SPIROMICS and three SARP centres were only used in this study (table 2). The imaging data from SARP were previously employed to identify imaging-based asthma clusters.17 All of the imaging centres acquired inspiratory and expiratory QCT scans for each subject. While both SPIROMICS and SARP obtained inspiration scans at TLC, expiration scans were obtained at functional residual capacity (FRC) for SARP and residual volume (RV) for SPIROMICS. Thus, caution must be taken in interpreting results associated with expiration scan and volumetric difference. For instance, fSAD% and Jacobian may increase and decrease respectively by using FRC rather than RV scans.
Table 1.
Demographic, pre-bronchodilator and post-bronchodilator pulmonary function tests, blood inflammatory information among healthy subjects, patients with asthma and patients with chronic obstructive pulmonary disease (COPD)
| Healthy subjects (n=94§) | Patients with asthma (n=75) | Patients with COPD (n=215) | |
| Demography | |||
| Age (years) | 50.7 (17.2) | 48.7 (11.2) | 67.1 (7.2)†‡ |
| Female sex (%) | 59 | 47 | 41 |
| Body mass index (kg/m2) | 27.7 (5.5) | 33.5 (7.5)* | 27.9 (4.9)† |
| Race (white/African-American/other) (%) | 65/20/15 | 76/17/7 | 87/7/6‡ |
| Pre-bronchodilator | |||
| FEV1, %predicted | 98 (12) | 53 (14)* | 44 (18)†‡ |
| FVC, %predicted | 98 (10) | 69 (13)* | 76 (17) †‡ |
| FEV1/FVC | 79 (6) | 61 (11)* | 43 (13)†‡ |
| Post-bronchodilator | |||
| FEV1, %predicted | 103 (11) | 65 (11)* | 51 (19)†‡ |
| FVC, %predicted | 99 (10) | 80 (14)* | 85 (18)‡ |
| FEV1/FVC | 82 (6) | 65 (11)* | 45 (13)†‡ |
| Blood tests | |||
| Samples (n) | 93 | 73 | 213 |
| Total white blood cells (N/μL) | 5894 (1717) | 7902 (2962)* | 7247 (1999)‡ |
| Neutrophils (%) | 58.1 (7.8) | 59.2 (12.4) | 63.3 (9.7)‡ |
| Lymphocytes (%) | 31.7 (7.4) | 28.2 (10.5) | 24.6 (8.3)‡ |
| Eosinophils (%) | 2.3 (1.6) | 3.9 (3.7)* | 2.9 (1.8)‡ |
*P<0.01 for healthy versus asthmatic subjects.
†P<0.01 for asthmatic versus COPD subjects.
‡P<0.01 for COPD subjects versus healthy subjects.
§Healthy datasets contain 51 Severe Asthma Research Program and 43 SubPopulations and InteRmediate Outcome Measures In COPD Study subjects. Benjamini–Hochberg post-hoc tests were performed.
FEV1, Forced expiratory volume in one second; FVC, Forced vital capacity.
Table 2.
The number of quantitative CT imaging datasets according to imaging centres: each dataset contains one inspiration and one expiration scan
| Project | Imaging centre (abbreviation) | Number of datasets |
| SubPopulations and InteRmediate Outcome Measures In COPD Study (eight sites) | 284 | |
| Columbia University (CU) | 61 | |
| Johns Hopkins University (JH) | 24 | |
| University of California at Los Angeles (LA, LV) | 36, 15 (two sites) |
|
| University of Michigan (MU) | 39 | |
| University of California at San Francisco (SF) | 31 | |
| University of Utah (UT) | 59 | |
| Wake Forest University (WF) | 19 | |
| Severe Asthma Research Program (three sites) | 100 | |
| University of Pittsburgh (PIT) | 44 | |
| University of Wisconsin (WIS) | 38 | |
| Washington University in Saint Louis (WSL) | 18 | |
An expanded set of multiscale imaging-based variables
We employed a total of 67 imaging-based metrics, where 55 of them were multiscale imaging-based metrics developed for the asthma cluster analysis.17 Using Apollo (VIDA Diagnostics),28 we derived three local structural variables: airway circularity (Cr), wall thickness (WT) and hydraulic diameter (D h) extracted at total lung capacity (TLC) scans. WT and D h were normalised with the tracheal WT and D h predicted from healthy subjects, denoted as WT* and D h*.14 Cr, WT* and D h* indicate heterogeneous airway shape, airway wall thickening and airway luminal narrowing, respectively. A detailed description of airway-structural imaging metrics can be found in ref.14 Furthermore, for the comparison with patients with COPD, we used the parametric response map20 to subtract the emphysematous lung regions from regions labelled as air trapped on expiratory scans using an image registration technique,29 being denoted as fSAD%. We also added two more variables: Emph% and tissue fraction at TLC (βtissue). Rather than the CT density-threshold method, we used a fraction-threshold method to determine fSAD% (air fraction, βair >90%) and Emph% (βair >98.5%), eliminating intersite variability.15 Next, βtissue was computed by the portion of tissue volume in a voxel to evaluate an alteration of local tissue. Furthermore, we computed fractional lobar air-volume change (ΔVair f), Jacobian and ADI via image registration.16 17 ΔVair f, Jacobian and ADI indicated preferential lobar air-volume change (a measure of regional ventilation), local expansion (a measure of regional volume change) and non-uniform stretch (a measure of the magnitude of direction preference in volume change), respectively, in parenchymal regions. A detailed description of functional imaging metrics is available in refs.16 17
Statistical analysis
Kruskal-Wallis and χ2 test30 were performed to compare differences of continuous and categorical variables between healthy, asthmatic and COPD subjects. We performed a total of 87 comparison tests, and a P value of 0.01 was taken as the significant level in all tests. This controls the false discovery rate of multiple comparisons at 1%, as estimated by using the method of Benjamin and Hochberg.31
Results
Demography, PFTs and blood tests
Table 1 shows demography, pre-bronchodilator and post-bronchodilator pulmonary function test (PFT) and blood test results among three populations, that is, healthy subjects, patients with asthma and patients with COPD. Ages of patients with COPD were greater than those of healthy and asthmatic subjects, and the portion of female sex was similar between asthma (47%) and COPD (41%). Asthmatic patients were more obese than healthy subjects and patients with COPD, whereas there was a lower portion of African-Americans in COPD. Compared with healthy subjects, both asthmatic patients and patients with COPD had lower FEV1 and FVC %predicted values according to their inclusion criteria with FEV1 %predicted value <80%. Pre-bronchodilator FEV1 %predicted values of patients with COPD were lower than those of asthma, while FVC % predicted values of patients with COPD were greater than those of asthma, leading to a further reduction of FEV1/FVC in patients with COPD. Thus, at pre-bronchodilator, flow restriction and obstruction were dominant for asthma and COPD, respectively. At post-bronchodilator, the difference between FEV1 %predicted values for asthma and COPD were more significant, whereas FVC %predicted values were close for both populations due to the functional reversibility of asthma. We further compared blood inflammatory biomarkers. Both patients with asthma and COPD demonstrated greater numbers of white blood cell counts than those of healthy subjects. The percentages of neutrophils and lymphocytes increased and decreased in patients with COPD, respectively, compared with those of healthy and asthmatic subjects. The percentages of eosinophils in both asthma and COPD were elevated, relative to that of healthy subjects.
Central airway-structural characteristics
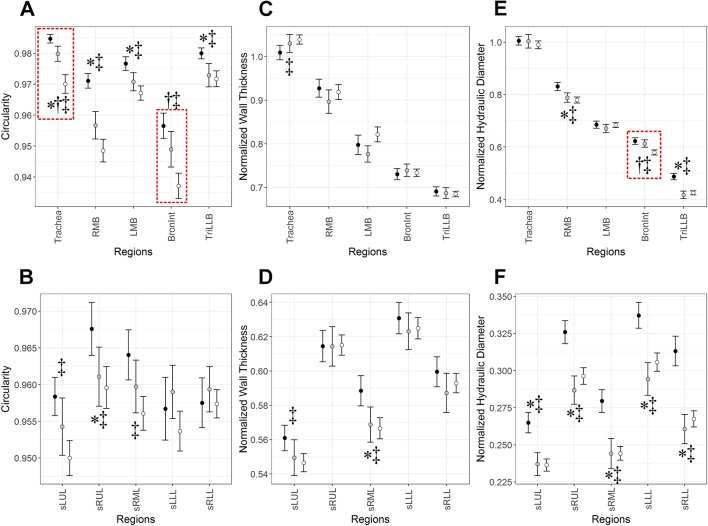
Figure 1 shows regional difference of Cr, WT* and D h* among healthy, asthmatic and COPD subjects. For a better presentation, we divided 10 local branches into two regions (top vs bottom columns), that is, five central airways (trachea, right main bronchus (RMB), left main bronchus (LMB), right intermediate bronchus (BronInt) and trifurcation of left lower lobe (TriLLB)), and five segmental airways (subgrouped left upper lobe (sLUL), subgrouped right upper lobe (sRUL), subgrouped right middle lobe (sRML), subgrouped left lower lobe (sLLL) and subgrouped right lower lobe (sRLL)). Cr significantly decreased in both asthmatic patients and patients with COPD compared with healthy subjects especially in central airways, where COPD decreased more than asthma in trachea and BronInt (figure 1A). In segmental airways, Cr of healthy subjects versus patients with COPD was only different in upper and middle lobes (see sLUL, sRUL and sRML in figure 1B). WT* of COPD subjects increased only in trachea compared with healthy subjects (figure 1C), while it decreased in sLUL and sRML of patients with COPD (figure 1D). Other than these three regions, there was no noticeable difference of WT* among three populations. There was no difference of D h* in trachea and LMB, whereas D h* of patients with asthma and COPD significantly decreased in other regions except these two regions (figure 1E, F). The reduced quantities of D h* in segmental airways between asthma and COPD were similar.
Figure 1.
(A, B) Airway circularity (Cr), (C, D) normalised wall thickness (WT*) and (E, F) normalised hydraulic diameter (D h*) in 10 local regions among healthy, asthmatic and chronic obstructive pulmonary disease (COPD) subjects. Values are presented as mean (±CI). *P<0.01 for healthy versus asthmatic subjects. †P<0.01 with red-dashed box for asthmatic versus COPD subjects. ‡P<0.01 for COPD subjects versus healthy subjects. BronInt, right intermediate bronchus; LMB, left main bronchus; RMB, right main bronchus; sLLL, subgrouped left lower lobe; sLUL, subgrouped left upper lobe; sRLL, subgrouped right lower lobe; sRML, subgrouped right middle lobe; sRUL, subgrouped right upper lobe; TriLLB, trifurcation of left lower lobe.
Parenchymal functional characteristics
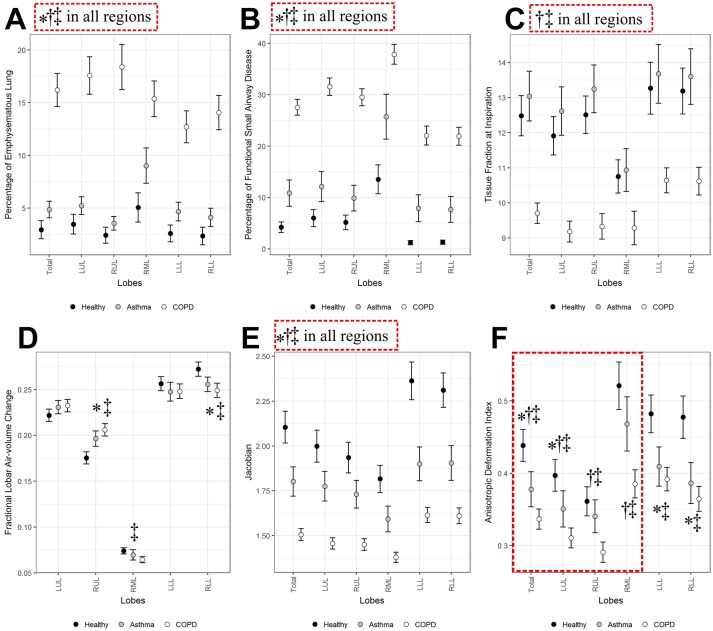
We also compared parenchymal/global functional metrics including Emph%, fSAD%, βtissue, ΔV air f, Jacobian and ADI (figure 2A–F). The former four variables were associated with CT density-based assessment, whereas the latter two variables were associated with mechanical strains estimated by image registration. As expected, Emph% of COPD subjects significantly increased compared with those of healthy and asthma subjects, and asthma subjects also showed a significant elevation of Emph% (figure 2A). fSAD% significantly increased in patients with COPD and asthma, especially in patients with COPD (figure 2B). Relative to healthy subjects, a new variable βtissue at inspiration decreased in entire lungs in patients with COPD (figure 2C), possibly due to tissue destruction by emphysema. Unlike Emph%, βtissue was similar between healthy and asthmatic subjects, whereas it decreased only in patients with COPD. ΔV air f behaved similarly in both patients with asthma and COPD, such that ΔV air f increased in RUL and it decreased in right lower lobe (RLL) (figure 2D). Thus the relative contribution of air-volume change was shifted from lower and middle lobes to upper lobes. Jacobian, indicating local volume change, decreased in both disease groups but more in patients with COPD (figure 2E). ADI, indicating preferential deformation, decreased more in upper lobes of COPD subjects, whereas it decreased similarly in lower lobes of patients with asthma and COPD (figure 2F). This may indicate difference of upper lobar characteristics in deforming lungs between patients with asthma and COPD.
Figure 2.
(A) Percentage of emphysematous lung (Emph%), (B) functional small airway disease (fSAD%), (C) tissue fraction at inspiration (βtissue), (D) fractional lobar air-volume change (ΔV air f), (E) the determinant of Jacobian (Jacobian) and (F) anisotropic deformation index (ADI) among healthy, asthmatic and chronic obstructive pulmonary disease (COPD) subjects. Values are presented as mean (±CI). *P<0.01 for healthy versus asthmatic subjects. †P<0.01 with red-dashed box for asthmatic versus COPD subjects. ‡P<0.01 for COPD subjects versus healthy subjects. LLL, left lower lobe; LUL, left upper lobe; RLL, right lower lobe; RML, right middle lobe; RUL, right upper lobe.
Discussion
Using QCT imaging-based variables, we have investigated the structural and functional characteristics at segmental and lobar/global scales between two airway obstructive diseases, for example, asthma and COPD. These subjects were clinically diagnosed with either asthma or COPD by NIH-sponsored multicentre studies of SARP and SPIROMICS. In this study, we compared currently non-smoking patients with asthma and former smoker patients with COPD with similar clinically overlapping characteristics of reduced lung function measured by post-bronchodilator FEV1 <80%. Asthma is characterised by airway narrowing, wall thickening and air trapping (or functional small-airway disease) and reduced lung deformation.12–16 COPD is characterised by airway narrowing, air trapping, increased emphysematous lung percentage and reduced lung deformation.18–20 22 As multicentre studies, SARP and SPIROMICS contributed to identify disease phenotypes by collecting subject demographics, biospecimens, QCTs and other data for patients with asthma and COPD. The present study has been able to take unique advantage of these multicentre studies with similar imaging protocols and similar image analysis, allowing for a comparison of the QCT data serving to define asthma and COPD imaging phenotypes. A better understanding of distinct structural and functional features between the two populations may help to develop personalised treatment intervention and improve the classification of asthma and COPD.
Regarding airway structure, Cr, WT* and D h* demonstrated different characteristics from observed features in healthy subjects. Cr, indicating the heterogeneous distribution of airway shape, was mostly altered in large airways and segmental bronchi in upper and middle lobes. COPD demonstrated the greater degree of these alterations relative to asthma (figure 2A, B). WT* seems to be clearly increased only in the trachea of patients with COPD compared with healthy patients and patients with asthma. The alterations of Cr and WT* in larger airways (or lower generations) may indicate that airway remodelling due to chronic inflammation or chronic bronchitis was predominantly caused in large airways rather than small airways. Unlike Cr and WT*, D h*, indicating airway narrowing, was significantly decreased in both asthma and COPD to a similar degree compared with healthy subjects. We have previously reported that among airway structural variables D h* was the most significantly correlated with PFT-based FEV1 and FEV1/FVC.14 Since we studied patients with lower FEV1, a smaller D h* was expected. With regard to regional phenotype, as shown in figure 2, airway structural alterations estimated by Cr, WT* and D h* were located randomly, implying that each metric demonstrates different phenotype.
Compared with airway structural variables, parenchymal functional variables provided much clearer differences between asthma and COPD. Although asthma demonstrated an increase of Emph%, which might be the result of unmeasurable environmental exposures.32 As would be expected, we found a significantly greater elevation in Emph% in COPD. Although fSAD% is an independent variable from Emph%, it exhibited a similar pattern with Emph%, such that patients with COPD had a greater elevation of fSAD% than asthma. While D h* had similar values between asthma and COPD, fSAD%, indicating small-airway disease, had significantly different values between asthma and COPD. This may indicate that small airway disease in patients with COPD is more prominent than asthmatic subjects. In other words, asthma reversibility is an effect predominantly localised to the peripheral airways as demonstrated by Verbanck et al.33 34 In this study, we also introduced a new variable βtissue to evaluate the state of tissue alteration.35 A decrease of βtissue is possibly related to tissue destruction due to emphysema. Thus, βtissue can serve as a metric used to differentiate between asthma, COPD and healthy subjects in addition to Emph% and fSAD%.
Using an image registration technique, we derived ΔV air f, Jacobian and ADI. ΔV air f, indicating regional distribution of air-volume change, seemed to increase in RUL and decrease in RLL consistently in asthma and COPD subjects. The similar finding has been reported in our previous study with 30 severe asthmatics.16 17 Jacobian, indicating the degree of local deformation, was significantly reduced in both asthma and COPD, with a more severe reduction in COPD. ADI seemed to have different characteristics from Jacobian in that it decreased in lower lobes for both asthma and COPD. On the other hand, it seemed to be relatively normal in RUL and RML in asthmatics, unlike patients with COPD. Based on ΔV air f and ADI, functional alterations of asthmatics were observed more in lower lobes, whereas those of COPD were observed in entire lungs.
When comparing multicentre data, it is necessary to use standardised imaging protocols. Since this study is designed retrospectively, there were protocol differences. The most critical difference between SARP and SPIROMICS is about lung volume when obtaining expiratory scans. SARP took expiratory scans at FRC, whereas SPIROMICS did this at RV. Thus COPD subjects obtained from SPIROMICS should have larger Jacobian and smaller fSAD% because RV is smaller lung volume than FRC. Nevertheless, the results showed that COPD subjects had significantly smaller Jacobian and larger fSAD% than asthmatic subjects which likely go beyond differences that would be expected from imaging at RV versus FRC. Therefore, the actual difference between asthma and COPD should be greater than that reported here if the same imaging protocol were used. It also should be noted that obesity in asthmatics may reduce expiratory reserve volume, making FRC closer to RV.36 Furthermore, due to the disease features, asthma is likely to be early onset, whereas COPD is likely to be late onset. Asthma is also associated with obesity, whereas COPD is with loss of weight. These disease features prevent from matching their demographics. Another issue may be raised due to intersubject variability including sex, age and height. We previously found that sex is the most important metric to determine airway size including hydraulic diameter and wall thickness; therefore, we developed new normalisation schemes to control the intersubject variability. Furthermore, there was no bias of samples due to the female sex between asthma and COPD (table 1).
In conclusion, QCT imaging-based variables were found to be effective in differentiating asthmatics from healthy subjects as well as COPD subjects from asthma subjects. Both asthma and COPD had a decrease of airway luminal circularity particularly in larger airways and a decrease of hydraulic diameter in segmental airways to the same degree. Functional metrics, especially density-based metrics, obtained at lobar/global regions were found to be significantly different between the two disease populations. Compared with asthma, COPD has significantly more lung emphysema, more small-airway disease, with reduced tissue fraction and regional lung deformation. Regionally, functional alterations of asthmatics were prominent in lower lobes, while those of patients with COPD were found in whole lungs. Based on these functional and regional features, density-based metrics including tissue fraction, fSAD% and Emph% measured at parenchymal levels were found to be important in differentiating patients with asthma and COPD.
Acknowledgments
The authors thank the SPIROMICS participants and participating physicians, investigators and staff for making this research possible. More information about the study and how to access SPIROMICS data is at https://www.spiromics.org. The authors acknowledge the following current and former investigators of the SPIROMICS sites and reading centers: Neil EAlexis, PhD; Wayne H Anderson, PhD; Igor Barjaktarevic, MD, PhD; RGraham Barr, MD, DrPH; Eugene R Bleecker, MD; Richard C Boucher, MD;Russell P Bowler, MD, PhD; Elizabeth E Carretta, MPH; Stephanie AChristenson, MD; Alejandro P Comellas, MD; Christopher B Cooper, MD, PhD; David J Couper, PhD; Gerard J Criner, MD; Ronald G Crystal, MD; Jeffrey L Curtis, MD; Claire M Doerschuk, MD; Mark T Dransfield, MD;Christine M Freeman, PhD; MeiLan K Han, MD, MS; Nadia N Hansel, MD, MPH; Annette T Hastie, PhD; Eric A Hoffman, PhD; Robert J Kaner, MD;Richard E Kanner, MD; Eric C Kleerup, MD; Jerry A Krishnan, MD, PhD;Lisa M LaVange, PhD; Stephen C Lazarus, MD; Fernando J Martinez, MD, MS; Deborah A Meyers, PhD; Wendy C Moore, MD; John D Newell Jr, MD;Laura Paulin, MD, MHS; Stephen Peters, MD, PhD; Cheryl Pirozzi, MD; Elizabeth C Oelsner, MD, MPH; Wanda K O’Neal, PhD; Victor E Ortega, MD, PhD; Robert Paine, III, MD; Nirupama Putcha, MD, MHS; Sanjeev Raman, MBBS, MD; Stephen I. Rennard, MD; Donald P Tashkin, MD; JMichael Wells, MD; Robert A Wise, MD; and Prescott G Woodruff, MD, MPH. The project officers from the Lung Division of the National Heart, Lung, and Blood Institute were Lisa Postow, PhD, andThomas Croxton, PhD, MD.
Footnotes
Contributors: Conception and design: SC. Acquisition of data: SC, BH, EAH, SEW, MC, SBF, SBF, NNJ, MLS, RGB., MKH, ERB, CBC, DC, NH, REK, EAK, EACK, FJM, WKO, PGW and C-LL. Analysis and interpretation of data and final approval of the version to be published: all authors. Drafting the article or revising it critically for important intellectual content: SC, JC, EAH, APC, MLS, REK, WKO and C-LL.
Funding: This study was supported by the NIH grants: U01 HL114494, HL209152; R01HL094315, HL112986, HL69174, HL064368, HL091762, HL069116; S10RR022421; U10 HL109257, HL109168; UL1 RR024153 (CTSI), UL1 TR000448, UL1 TR000427 (CTSA), and by BasicScience Research Program through the National Research Foundation ofKorea (NRF) funded by the Ministry of Education(NRF-2017R1D1A1B03034157). SPIROMICS was supported bycontracts from the NIH/NHLBI (HHSN268200900013C, HHSN268200900014C, HHSN268200900015C, HHSN268200900016C, HHSN268200900017C, HHSN268200900018C, HHSN268200900019C, HHSN268200900020C), and supplemented by contributions made through the Foundation for the NIH and the COPD Foundation from AstraZeneca/MedImmune; Bayer; Bellerophon Therapeutics; Boehringer-Ingelheim Pharmaceuticals, Inc; Chiesi Farmaceutici S.p.A; Forest Research Institute, Inc; GlaxoSmithKline; Grifols Therapeutics, Inc; Ikaria, Inc; Nycomed GmbH; Takeda Pharmaceutical Company; Novartis Pharmaceuticals Corporation; ProterixBio; Regeneron Pharmaceuticals, Inc; Sanofi; and Sunovion.
Competing interests: EAH is a shareholder in VIDA diagnostics, a company that is commercialising lung image analysis software derived by the University of Iowa lung imaging group. He is also a member of the Siemens CT advisory board. SBF receives grant funding from GE Healthcare.
Patient consent: Obtained.
Ethics approval: The imaging protocols for acquiring CT images were approved by the institutional review boards of the respective institutions.
Provenance and peer review: Not commissioned; internally peer reviewed.
Data sharing statement: No additional data are available.
References
- 1.Barrecheguren M, Esquinas C, Miravitlles M. The asthma-chronic obstructive pulmonary disease overlap syndrome (ACOS): opportunities and challenges. Curr Opin Pulm Med 2015;21:74–9. doi:10.1097/MCP.0000000000000118 [DOI] [PubMed] [Google Scholar]
- 2.Celli BR, MacNee W. ATS/ERS Task Force. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur Respir J 2004;23:932–46. doi:10.1183/09031936.04.00014304 [DOI] [PubMed] [Google Scholar]
- 3.Postma DS, Rabe KF. The asthma-COPD overlap syndrome. N Engl J Med 2015;373:1241–9. doi:10.1056/NEJMra1411863 [DOI] [PubMed] [Google Scholar]
- 4.de Marco R, Pesce G, Marcon A, et al. . The coexistence of asthma and Chronic Obstructive Pulmonary Disease (COPD): prevalence and risk factors in young, middle-aged and elderly people from the general population. PLoS One 2013;8:e62985 doi:10.1371/journal.pone.0062985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gibson PG, Simpson JL. The overlap syndrome of asthma and COPD: what are its features and how important is it? Thorax 2009;64:728–35. doi:10.1136/thx.2008.108027 [DOI] [PubMed] [Google Scholar]
- 6.Moore WC, Meyers DA, Wenzel SE, et al. . Identification of asthma phenotypes using cluster analysis in the severe asthma research program. Am J Respir Crit Care Med 2010;181:315–23. doi:10.1164/rccm.200906-0896OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moore WC, Hastie AT, Li X, et al. . Sputum neutrophil counts are associated with more severe asthma phenotypes using cluster analysis. J Allergy Clin Immunol 2014;133:1557–63. doi:10.1016/j.jaci.2013.10.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Papi A, Romagnoli M, Baraldo S, et al. . Partial reversibility of airflow limitation and increased exhaled NO and sputum eosinophilia in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2000;162:1773–7. doi:10.1164/ajrccm.162.5.9910112 [DOI] [PubMed] [Google Scholar]
- 9.Chanez P, Vignola AM, O’Shaugnessy T, et al. . Corticosteroid reversibility in COPD is related to features of asthma. Am J Respir Crit Care Med 1997;155:1529–34. doi:10.1164/ajrccm.155.5.9154853 [DOI] [PubMed] [Google Scholar]
- 10.Couper D, LaVange LM, Han M, et al. . Design of the Subpopulations and Intermediate Outcomes in COPD Study (SPIROMICS). Thorax 2014;69:492–5. doi:10.1136/thoraxjnl-2013-203897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wenzel SE, Busse WW. National Heart, Lung, and Blood Institute’s Severe Asthma Research Program. Severe asthma: lessons from the severe asthma research program. J Allergy Clin Immunol 2007;119:14–21. doi:10.1016/j.jaci.2006.10.025 [DOI] [PubMed] [Google Scholar]
- 12.Aysola RS, Hoffman EA, Gierada D, et al. . Airway remodeling measured by multidetector CT is increased in severe asthma and correlates with pathology. Chest 2008;134:1183–91. doi:10.1378/chest.07-2779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Busacker A, Newell JD, Keefe T, et al. . A multivariate analysis of risk factors for the air-trapping asthmatic phenotype as measured by quantitative CT analysis. Chest 2009;135:48–56. doi:10.1378/chest.08-0049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Choi S, Hoffman EA, Wenzel SE, et al. . Quantitative assessment of multiscale structural and functional alterations in asthmatic populations. J Appl Physiol 2015;118:1286–98. doi:10.1152/japplphysiol.01094.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Choi S, Hoffman EA, Wenzel SE, et al. . Improved CT-based estimate of pulmonary gas trapping accounting for scanner and lung-volume variations in a multicenter asthmatic study. J Appl Physiol 2014;117:593–603. doi:10.1152/japplphysiol.00280.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Choi S, Hoffman EA, Wenzel SE, et al. . Registration-based assessment of regional lung function via volumetric CT images of normal subjects vs. severe asthmatics. J Appl Physiol 2013;115:730–42. doi:10.1152/japplphysiol.00113.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Choi S, Hoffman EA, Wenzel SE, et al. . Quantitative computed tomographic imaging-based clustering differentiates asthmatic subgroups with distinctive clinical phenotypes. J Allergy Clin Immunol 2017;140:690–700. doi:10.1016/j.jaci.2016.11.053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mair G, Maclay J, Miller JJ, et al. . Airway dimensions in COPD: relationships with clinical variables. Respir Med 2010;104:1683–90. doi:10.1016/j.rmed.2010.04.021 [DOI] [PubMed] [Google Scholar]
- 19.Smith BM, Hoffman EA, Rabinowitz D, et al. . Comparison of spatially matched airways reveals thinner airway walls in COPD. The Multi-Ethnic Study of Atherosclerosis (MESA) COPD study and the Subpopulations and Intermediate Outcomes in COPD Study (SPIROMICS). Thorax 2014;69:987–96. doi:10.1136/thoraxjnl-2014-205160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Galbán CJ, Han MK, Boes JL, et al. . Computed tomography-based biomarker provides unique signature for diagnosis of COPD phenotypes and disease progression. Nat Med 2012;18:1711–5. doi:10.1038/nm.2971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Amelon R, Cao K, Ding K, et al. . Three-dimensional characterization of regional lung deformation. J Biomech 2011;44:2489–95. doi:10.1016/j.jbiomech.2011.06.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bodduluri S, Newell JD, Hoffman EA, et al. . Registration-based lung mechanical analysis of chronic obstructive pulmonary disease (COPD) using a supervised machine learning framework. Acad Radiol 2013;20:527–36. doi:10.1016/j.acra.2013.01.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Oguma T, Hirai T, Fukui M, et al. . Longitudinal shape irregularity of airway lumen assessed by CT in patients with bronchial asthma and COPD. Thorax 2015;70:719–24. doi:10.1136/thoraxjnl-2014-206651 [DOI] [PubMed] [Google Scholar]
- 24.Kosciuch J, Krenke R, Gorska K, et al. . Airway dimensions in asthma and COPD in high resolution computed tomography: can we see the difference? Respir Care 2013;58:1335–42. doi:10.4187/respcare.02175 [DOI] [PubMed] [Google Scholar]
- 25.Kurashima K, Hoshi T, Takayanagi N, et al. . Airway dimensions and pulmonary function in chronic obstructive pulmonary disease and bronchial asthma. Respirology 2012;17:79–86. doi:10.1111/j.1440-1843.2011.02052.x [DOI] [PubMed] [Google Scholar]
- 26.Sieren JP, Newell JD, Barr RG, et al. . SPIROMICS protocol for multicenter quantitative computed tomography to phenotype the lungs. Am J Respir Crit Care Med 2016;194:794–806. doi:10.1164/rccm.201506-1208PP [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Moore WC, Bleecker ER, Curran-Everett D, et al. . Characterization of the severe asthma phenotype by the national Heart, Lung, and Blood institute ’s severe asthma research program. J Allergy Clin Immunol 2007;119:405–13. doi:10.1016/j.jaci.2006.11.639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tschirren J, Hoffman EA, McLennan G, et al. . Segmentation and quantitative analysis of intrathoracic airway trees from computed tomography images. Proc Am Thorac Soc 2005;2:484–7. doi:10.1513/pats.200507-078DS [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yin Y, Hoffman EA, Lin CL. Mass preserving nonrigid registration of CT lung images using cubic B-spline. Med Phys 2009;36:4213–22. doi:10.1118/1.3193526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yates F. Contingency tables involving small numbers and the χ2 test. J R Stat Soc 1934;1:217–35. doi:10.2307/2983604 [Google Scholar]
- 31.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc B Met 1995;57:289–300. [Google Scholar]
- 32.Paganin F, Séneterre E, Chanez P, et al. . Computed tomography of the lungs in asthma: influence of disease severity and etiology. Am J Respir Crit Care Med 1996;153:110–4. doi:10.1164/ajrccm.153.1.8542102 [DOI] [PubMed] [Google Scholar]
- 33.Verbanck S, Schuermans D, Van Muylem A, et al. . Conductive and acinar lung-zone contributions to ventilation inhomogeneity in COPD. Am J Respir Crit Care Med 1998;157:1573–7. doi:10.1164/ajrccm.157.5.9710042 [DOI] [PubMed] [Google Scholar]
- 34.Verbanck S, Schuermans D, Noppen M, et al. . Evidence of acinar airway involvement in asthma. Am J Respir Crit Care Med 1999;159:1545–50. doi:10.1164/ajrccm.159.5.9809017 [DOI] [PubMed] [Google Scholar]
- 35.Choi J, Hoffman EA, Lin CL, et al. . Quantitative computed tomography determined regional lung mechanics in normal nonsmokers, normal smokers and metastatic sarcoma subjects. PLoS One 2017;12:e0179812 doi:10.1371/journal.pone.0179812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jones RL, Nzekwu MM. The effects of body mass index on lung volumes. Chest 2006;130:827–33. doi:10.1378/chest.130.3.827 [DOI] [PubMed] [Google Scholar]