Abstract
Mitochondrial fission and selective mitochondrial autophagy (mitophagy) form an essential axis of mitochondrial quality control that plays a critical role in the development of cardiac ischemia-reperfusion (IR) injury. However, the precise upstream molecular mechanism of fission/mitophagy remains unclear. Dual-specificity protein phosphatase1 (DUSP1) regulates cardiac metabolism, but its physiological contribution in the reperfused heart, particularly its influence on mitochondrial homeostasis, is unknown. Here, we demonstrated that cardiac DUSP1 was downregulated following acute cardiac IR injury. In vivo, compared to wild-type mice, DUSP1 transgenic mice (DUSP1TG mice) demonstrated a smaller infarcted area and the improved myocardial function. In vitro, the IR-induced DUSP1 deficiency promoted the activation of JNK which upregulated the expression of the mitochondrial fission factor (Mff). A higher expression level of Mff was associated with elevated mitochondrial fission and mitochondrial apoptosis. Additionally, the loss of DUSP1 also amplified the Bnip3 phosphorylated activation via JNK, leading to the activation of mitophagy. Increased mitophagy overtly consumed mitochondrial mass resulting into the mitochondrial metabolism disorder. However, the reintroduction of DUSP1 blunted Mff/Bnip3 activation and therefore alleviated the fatal mitochondrial fission/mitophagy by inactivating the JNK pathway, providing a survival advantage to myocardial tissue following IR stress. The results of our study suggest that DUSP1 and its downstream JNK pathway are therapeutic targets for conferring protection against IR injury by repressing Mff-mediated mitochondrial fission and Bnip3-required mitophagy.
Keywords: Cardiac IR injury, Mitochondrial fission, Mitophagy, Mff, Bnip3, DUSP1, JNK
Graphical abstract

Highlights
-
•
IR injury induces DUSP1 downregulation.
-
•
Loss of DUSP1 leads to an increase in JNK phosphorylation.
-
•
JNK activates Mff and Bnip3, contributing to the fatal mitochondrial fission and mitophagy, respectively.
-
•
Fission and mitophagy induces cell damage via trigging caspase9-related apoptosis and mitochondrial energy disorder.
1. Introduction
Despite the successful revascularization of the epicardial occluded vessels, reperfusion failure in cardiomyocytes at the microvascular bed post-ischemia is referred to as cardiac ischemia/reperfusion (IR) injury [1]. IR injury is a phenomenon in which the cellular damage in an organ that is initiated during hypoxia or anoxia becomes exacerbated when oxygen is redelivered to the tissue [2]. Despite its profound clinical importance, bedside therapies to suppress IR remain limited largely due to the complex mechanisms that contribute to IR. The previous studies [3] have suggested that mitochondrial homeostasis is the target of cardiac IR injury. On the one hand, mitochondrial fission factor (Mff)-induced mitochondrial fission causes an overproduction of reactive oxygen species (ROS) that induces cardiolipin oxidation; on the other hand, it triggers hexokinase 2 (HK2) disassociation that opens mitochondrial permeability transition pore (mPTP). These alterations lead to cytochrome c (cyt-c) release from the mitochondria into the cytoplasm, which initiates caspase9-dependent apoptosis. These results lay the foundation for describing the detailed molecular mechanisms of IR injury initiation and regulation. However, the signals upstream of Mff upregulation are not completely understood.
In addition to mitochondrial fission, mitophagy is another specific process underlying the degradation of dysfunctional or damaged mitochondria, which maintains a healthy mitochondrial population and mitochondrial quality [4]. A considerable number of studies have suggested that mitophagy is regulated through two pathways including receptor-mediated mitophagy and Parkin-dependent mitophagy [5]. The receptor-related factors included BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (Bnip3) and FUN14 domain containing 1 (FUNDC1) [6]. Bnip3 is the mitochondrial outer membrane proteins that are required for the autophagic elimination of mitochondria during erythroid cell maturation [7]. FUNDC1 is also a mitochondrial outer membrane protein that binds LC3 to induce mitophagy upon hypoxia treatment [8]. The Parkin-related mitophagy mainly relies on PINK1 stabilization and accumulation on the mitochondrial outer membrane upon mitochondrial depolarization [9]. Our team has performed significant research concerning the role of mitophagy in cardiac IR injury [8], [9], [10], [11]. However, different receptors may have various influences on cell fate. In the cardiac reperfusion injury, excessive Parkin-involved mitophagy exacerbates cell death [9]. In contrast, FUNDC1-related mitophagy provides beneficial effects on cell survival under hypoxic stimulation [10]. However, the role of Bnip3 and its involvement in mitophagy under IR injury remain unclear. Bnip3 is reported to be activated under ischemia stimulation and promotes cellular death in the heart [12], [13]. Meanwhile, recent study confirms that activation of Bnip3-related mitophagy would impair the liver cancer metastasis through blunting mitochondrial energy metabolism [11]. In the current study, we explore the role of Bnip3-mediated mitophagy in the development of cardiac IR injury.
Dual-specificity protein phosphatase (DUSP), which is also known as mitogen-activated protein kinase (MAPK) phosphatase, is a threonine-tyrosine dual-specificity phosphatase that dephosphorylates and inactivates MAPKs, extracellular signal-regulated kinase (ERK), p38, and c-Jun N-terminal kinase (JNK) in a context-dependent manner [14], [15]. DUSP1, which is an anti-apoptotic phosphatase, is expressed in a wide variety of tissues, and its highest levels are observed in the heart, lungs and liver [16]. However, its precise role in cardiac IR injury remains unknown. In breast cancer [17], DUSP1 is inducible in radiation-treated breast cancer cells and is correlated with the expression of human epidermal growth factor receptor 2, which is the pro-survival signaling for cancer cells [18]. In contrast, an inhibitor of DUSP1 is considered an adjuvant and/or neo-adjuvant therapy for breast cancer via enhancing cell apoptosis. Thus, we speculated DUSP1 might send protective signals to the reperfused heart. Notably, ample evidence has uncovered the downstream effectors of DUSP1 such as JNK and ERK are the regulator of mitochondrial fission and mitophagy [19], [20], and accordingly, we hypothesized that DUSP1 may handle fission/mitophagy in IR injury via JNK. Thus, the goal of this study was to define the contribution of DUSP1 in the control of acute cardiac IR injury, particularly its influence on fission and mitophagy.
2. Methods
2.1. Animal models of cardiac IR injury
All animal procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals, which was published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996) and were approved by the University of Wyoming Animal Care and Use Committee. The DUSP1 transgenic mice (DUSP1TG), with an FVB/N background, were generated according to the methods described in previous studies [21], [22]. Briefly, the pBSII-12.4kbVill-DUSP1 plasmids were obtained from OriGene. Then, the pBSII-12.4kbVill- DUSP1 plasmids were digested with EcoRI and KpnI to obtain villin-DUSP1 DNA fragments. Then the transgene was purified and injected into mouse blastocysts to generate transgenic mice. The wild-type (WT) mice and DUSP1TG mice (8 weeks old) were used to induce an IR injury model according to our previous studies [9]. Briefly, the mice were anesthetized with isoflurane, and the heart was exposed via a left thoracotomy. Then, a 7-0 silk suture was tied around the left anterior descending coronary artery (LAD) with a slipknot for 2 h to induce ischemia, followed by 0–24 h of reperfusion. Upon the completion of the reperfusion period, the hearts were stained with 2% Evans Blue and 1% 2,3,5-triphenyltetrazolium chloride. The infarct size was expressed as a percentage of the risk zone (n = 6/group). After the IR injury, the content of lactate dehydrogenase (LDH), Troponin T and creatine kinase-MB (CK-MB) in the blood was evaluated via ELISA assays as our previous study described [8].
2.2. Echocardiogram and electron microscopy
Echocardiography was performed in all mice 6 h after the reperfusion according to our previous studies [8]. An echocardiogram (14.0 MHz, Sequoia C512; Acuson, Germany) was used to detect both 2-dimensional and M-mode images.
Electron microscopy was conducted as previously described. The whole heart was immediately fixed at 4 °C with 2% glutaraldehyde in a 0.1 mol/L sodium cacodylate buffer and postfixed for 1 h on ice with 1% osmium tetroxide after IR injury. After making the slices, the samples were stained with lead citrate and uranyl acetate and observed under a Hitachi H600 Electron Microscope (Hitachi, Japan) [23]. At least 30 cells in a minimum of 5 randomly selected fields were observed.
2.3. H9C2 cells culture and HR injury induction
The HR injury model was mimicked in vitro by 2 h of hypoxia and 6 h of reoxygenation according to our previous study. The H9C2 cells were used in vitro. The pDC316-mCMV-DUSP1 adenovirus plasmid (Ad-DUSP1) and control adenovirus plasmid (Ad-ctrl) were purchased from Vigene Biosciences. The Ad-DUSP1 and Ad-ctrl were used to infect the H9C2 cells. To activate mitochondrial fission, FCCP (5 μM, Selleck Chemicals) was used 2 h before treatment. To inhibit the mitophagy, siRNA against Bnip3 was transfected to H9C2 cells to block mitophagy activity. To suppress and activate the JNK pathway, SP600125 (SP, 10 μM, Selleck Chemicals) and Anisomycin (Ani, 10 μM, Selleck Chemicals) were used 2 h before treatment, respectively.
2.4. Mitochondrial fission analysis and mitochondrial membrane potential measurements
Mitochondrial fission was measured according to our previous study [3]. After the mitochondria were labeled with TOM20, a single-cell image was obtained under a confocal microscope, and the image was analyzed using ImageJ 1.47 version software. The JC-1 Kit (Beyotime, China) was used to measure the change in the mitochondrial membrane potential (ΔΨm) [24]. The images were captured under a fluorescence microscope (OLYMPUS DX51; Olympus, Tokyo, Japan) and were analyzed with Image-Pro Plus 6.0 (Media Cybernetics, Rockville, MD) to obtain the mean densities of the regions of interest, which were normalized to those in the control group.
2.5. Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay, mPTP opening detection and ATP measurements
The TUNEL assay was used to detect cellular death following the IR injury. For the quantification, the numbers of TUNEL-positive cells in the infarcted areas or cardiomyocytes were calculated by counting at least 5 random separate fields as the ratio of the experimental samples to the control samples. The opening of the mPTP was observed as a rapid dissipation of tetramethylrhodamine ethyl ester fluorescence according to our previous studies. The cellular ATP levels were measured using a firefly luciferase-based ATP assay kit (Beyotime) based on a fluorescence technique (Genmed Scientifics Inc.) as previously described [25].
2.6. Mitochondrial oxygen consumption rate (OCR) assay
The mitochondrial oxygen consumption rate (OCR) was evaluated with an XFe96 extracellular flux analyzer (Agilent Technologies, CA, USA). Cells were seeded at 40,000 cells/well on 96-well XFe96 cell culture microplates and cultured for 48 h. For respiration assays, cells were incubated in a CO2-free environment for 1 h, and OCR was measured every 3 min for the next 90 min [26]. First, OCR was quantified in basal conditions (20 mM glucose), then with 1 μM oligomycin (ATP Synthase inhibitor), next with 0.125 μM FCCP (mitochondrial respiration uncoupler), and finally with 1 μM Rotenone/Antimycin A (Complex I and III inhibitors, respectively).
2.7. Cell shortening/relengthening assay and cellular viability detection
Mechanical properties of cardiomyocytes were assessed using a SoftEdge MyoCam system (IonOptix, Milton, MA) as our previously reported [27]. Cell shortening and relengthening were assessed using the following indices: resting cell length, peak shortening (PS), time-to-PS (TPS), time-to-90% relengthening (TR90), and maximal velocity of shortening/relengthening (±dL/dt).
Cellular viability was detected via caspase3 and caspase9 activity as well as MMT assay. Caspase9 and caspase3 activities were determined with caspase assay kits (Beyotime, China), which detect the production of the chromophore p-nitroanilide after its cleavage from the peptide substrate DEVD-p-nitroanilide and LEHD- p-nitroanilide. The MMT assay was conducted as our previously reported [28].
2.8. Western blotting analysis
To detect the protein expression, immunoblotting was carried out as described previously. Firstly, samples were washed under PBS for three times at room temperature, and then lysed in RIPA buffer containing 1 mM phenylmethylsulfonyl fluoride (PMSF). Subsequently, samples were centrifuged for about 10 min at 4 °C, to obtain the supernatant [29]. Then, proteins concentrations were detected via BCA Protein Quantification Kit [30]. About 50 mg protein for each sample was loaded and transferred to PVDF membrane. Primary antibodies used for immunoblotting were as follows: DUSP1 (1:1000, Abcam, #ab195261), pro-caspase3 (1:1000, Cell Signaling Technology, #9662), cleaved caspase3 (1:1000, Cell Signaling Technology, #9664), Bax (1:2000, Cell Signaling Technology, #5023), caspase9 (1:1000, Abcam, #ab32539), survivin (1:1000, Cell Signaling Technology, #2808), c-IAP1 (1:1000, Cell Signaling Technology, #7065), LC3II (1:1000, Cell Signaling Technology, #3868), p62 (1:1000, Abcam, #ab56416), Beclin1 (1:1000, Cell Signaling Technology, #3495), Atg5 (1:1000, Cell Signaling Technology, #12994), Mff (1:1000, Cell Signaling Technology, #86668), Bnip3 (1:1000, Cell Signaling Technology, #44060), JNK (1:1000, Cell Signaling Technology, #2656). The Bnip3 (ser17) (1:500) polyclonal antibodies were produced by immunizing rabbits with synthesized and purified phosphorylated and nonphosphorylated peptides from Bnip3 (Abgent, SuZhou, China) according to our previous study.
2.9. Immunohistochemistry and immunofluorescence staining
After treatments, samples were firstly washed with PBS for three times, and then fixed with 4% paraformaldehyde for 30 min at room temperature. Subsequently, samples were incubated with the primary antibody at 4 °C overnight [31]. Then, PBS was used to wash samples for three times, and then samples were stained with fluorescent second antibody at 37 °C for 30 min. DAPI was used for nuclear staining. The pictures were acquired using a fluorescence microscopy (Olympus Corporation, Tokyo, Japan) with standard excitation filters. The primary antibodies used in the present study were as follows: DUSP1 (Abcam, #ab195261), LAMP1 (Abcam, #ab24170), Bnip3 (Cell Signaling Technology, #44060) and p-JNK (Cell Signaling Technology, #2656).
2.10. qPCR and small interfering RNA assay
Reverse transcription was performed using the High Capacity cDNA Reverse Transcription Kits (Invitrogen, USA) with random oligo(dT) priming according to the manufacturer's protocols. PCR was performed using a sequence detection system (ABI PRISM 7500, Applied Biosystems, Foster City, CA) with SYBR Green (Bio-Rad, Hercules, CA) as the fluorescent marker according to our previous studies [32].
For the gene silencing, Bnip3 siRNA (Santa Cruz Biotechnology, USA) was used to knockdown the expression of Bnip3. For the RNAi knockdown, the cells were seeded in plates containing a medium without antibiotics for 24 h before the transfection [33]. The siRNAs were transfected into the cells by Lipofectamine 2000 transfection reagent (Invitrogen, U.S.A.) in serum-free Opti-MEM (Invitrogen, U.S.A.), according to our previous study [34].
2.11. Chromatin immunoprecipitation
Chromatin immunoprecipitation was performed according to our previous study [11]. Formaldehyde cross-linked chromatin fragments were immunoprecipitated with either anti-p-JNK antibody or anti-histone H3 antibody overnight at 4 °C. Control samples were treated in the same way except that no antibody was present. To reverse cross-links, eluates and input DNA were incubated overnight at 65 °C. DNA was purified using the Qiagen PCR clean-up kit (Qiagen, Valencia, CA, USA). PCRs were performed with primers flanking the Mff promoter. PCR products were resolved by electrophoresis in agarose gel and visualized with ethidium bromide.
2.12. Statistical analysis
Data analysis was conducted using SPSS 19.0 statistical software (SPSS, Chicago, IL, USA). Measurement data were presented as mean ± standard error of mean (SEM). The one-way analysis of variance (ANOVA) was conducted among multi-groups. P < 0.05 indicated statistical significance.
3. Results
3.1. IR injury downregulated DUSP1
First, we observed the changes in DUSP1 expression before and after the IR injury. Compared with the no-infarcted zone, the expression of DUSP1 was significantly reduced in the infarct area (Fig. 1A-B), suggesting that the IR injury downregulated the content of DUSP1 in the heart. In addition, the expression of DUSP1 was lowest after 6 h reperfusion. Therefore, reperfusion for 6 h was used in the following experiments. To visualize this phenotype change, an immunohistochemical analysis was performed. The DUSP1 protein expression in the infarcted area was lower than that in the non-infarcted zone (Fig. 1C-D). Furthermore, to explore the consequences of the DUSP1 downregulation in cardiac IR injury, DUSP1TG mice were subjected to an IR injury (2-h ischemia and 6-h reperfusion), and the degree of infarction was quantified, demonstrating a significant reduction in infarction area compared with that in the WT group (Fig. 1E-F). These data suggested that DUSP1 provided protective effects against the IR injury. The cellular death was the primary consequence of the IR stimulation, the TUNEL assay was used to evaluate the cellular damage under DUSP1 deficiency. The DUSP1TG mice had fewer TUNEL-positive cells than the mice in the WT group (Fig. 1G-H). Furthermore, we also detected the cellular damage after 24-h reperfusion via caspase3 activity and caspase3 expression. After 24-h reperfusion, we found that the casaspe3 activity and expression were lower in DUSP1TG mice when compared to the WT mice (Supplemental Fig. A-C).
Fig. 1.
Downregulation of DUSP1 at the infarcted area. WT mice and DUSP1TG mice underwent the 2-h ischemia and 0–24-h reperfusion (IR injury, n = 6/group). In vitro, 2-h of hypoxia and 6-h of reoxygenation (HR) was used to mimic the IR injury. Meanwhile, the gain-of-function assay about DUSP1 was also conducted via adenovirus vector overexpression of DUSP1 (Ad-DUSP1) or control adenovirus transfection (Ad-ctrl) in H9C2 cells. A. DUSP1 expression in the infarcted area or non-infarcted area. B. Quantitative analysis of the relative expression of DUSP1. C-D. The immunohistochemistry of DUSP1 in heart underwent 2-h ischemia and 6-h reperfusion. E-F. Representative images of heart sections with TTC and Evans Blue staining of the infarcted area. Bar graph indicates the infarct size. G-H. TUNEL assay for cellular apoptosis analysis. I-J. The control adenovirus (Ad-ctrl) and DUSP1-overexpression adenovirus (Ad-DUSP1) were transfected into H9C2 cells. The transfection efficiency was confirmed by western blots. K-L. Caspase3 activity and MTT assay were used to detect the cellular viability and apoptosis. #P < 0.05 vs the sham group or control group; *P < 0.05 vs IR+WT group or HR+Ad-ctrl group.
To gain more solid evidence, we used H9C2 cells to mimic the IR injury in vitro via hypoxia reoxygenation (HR) model. After 2-h hypoxia and 6-h reoxygenation, the DUSP1 expression was significantly reduced when compared to the control group (Fig. 1I-J). Subsequently, the gain-of function assay about DUSP1 was carried out via adenovirus vector transfection (Ad-DUSP1). The transfection efficiency was confirmed via western blots (Fig. 1I-J). Meanwhile, the cellular viability after transfection was determined by caspase3 activity and MTT assay (Fig. 1K-L). Overexpression of DUSP1 had no influence on the cellular viability. However, regaining of DUSP1 reduced the HR-induced cellular death (Fig. 1K-L). These data indicated that reintroduction of DUSP1 was able to prevent the IR injury via inhibiting cellular apoptosis.
3.2. DUSP1 deficiency contributed to the cardiac dysfunction
In addition to the cell death, we further detected markers of cardiac injury, including LDH, Troponin T and CK-MB, which are indicators of the extent of IR injury in the clinic. Compared with the WT mice, the DUSP1TG mice generated less LDH, Troponin T and CK-MB (Supplemental Fig. D-F). Moreover, we also observed changes in the cardiac function via echocardiogram. Compared to the sham groups, the left ventricular ejection fraction (LVEF), left ventricular diastolic dimension (LVDd) and left ventricular fractional shorting (LVFS) had obviously declined after the IR injury (Supplemental Fig. G-I). However, the cardiac dysfunction was improved in the DUSP1TG mice. To elucidate the protective role of DUSP1 in cardiac structure, electron microscopy (EM) was used. Compared with the sham groups, more mitochondrial damage, myocyte dissolution, muscular fiber twisting and Z line disappearance were observed in response to the IR injury. However, the reintroduction of DUSP1 improved the myocardial structure (Supplemental Fig. J).
To provide more direct evidence for the cardio-protective action of DUSP1, we observed the cardiomyocytes properties in WT and the DUSP1TG mice as our previously reported [27]. Neither IR injury nor DUSP1 overexpression overtly affected the resting cell length in cardiomyocytes (Supplemental Fig. K). However, cardiomyocytes from WT mice exposed to IR injury displayed significantly depressed PS and ± dL/dt as well as prolonged TPS and TR90 (Supplemental Fig. L-P). In contrast, overexpression of DUSP did not affect these mechanical parameters tested; it apparently reduced or abrogated IR-induced mechanical changes. These data indicated that DUSP1 sustained the cardiac function in the setting of cardiac IR injury.
3.3. DUSP1 deficiency induced mitochondrial damage and cellular apoptosis via mitochondrial fission
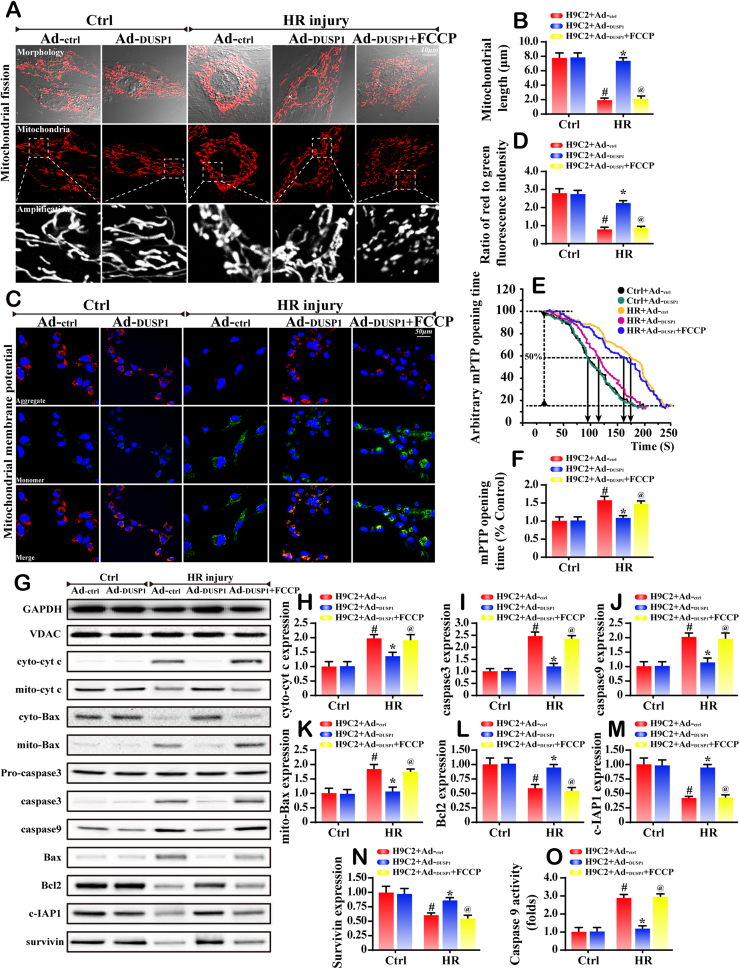
To explain the protective effects of DUSP1 on IR injury, we focused on mitochondrial fission, which is the target of IR injury. After HR injury, many balloon- or bulb-like structures were observed at the base of the mitochondrial tubules in the Ad-ctrl group but not in the Ad-DUSP1 group (Fig. 2A). Notably, FCCP (10 μM), an activator of fission, was used to recall the fission in cells transfected with Ad-DUSP1. To quantify the fission, the mitochondrial length was recorded (Fig. 2B) and these results were in accordance with the above findings.
Fig. 2.
DUSP1 influenced mitochondrial fission. A. Tom20 was used to observe the mitochondrial fission or mitochondrial debris. The bottom picture under each micrograph shows an amplification of mitochondrial fragments. B. The quantification of mitochondrial fission via mitochondrial length. FCCP, the fission activator, was used to re-activate fission in DUSP1-overexpressed cells. C-D. The change in the membrane potential (ΔΨm) via JC-1 staining. E-F. The mPTP opening time was recorded and the mPTP opening rate was evaluated with DUSP1 overexpression or not. G-N. Western blots was used to evaluate the change of mitochondrial apoptosis-related proteins. O. Caspase9 activity was measured to reflect the mitochondrial apoptosis. #P < 0.05 vs the control group; *P < 0.05 vs HR+Ad-ctrl group; @P < 0.05 vs HR+Ad-DUSP1 group.
To explain the role of fission in cellular damage under IR injury, we focused on the mitochondria-dependent cellular apoptosis. Firstly, mitochondrial electrochemical gradient (ΔΨm), which was measured by JC-1, showed that HR injury impaired the ΔΨm (Fig. 2C-D). However, DUSP1 overexpression reversed the stability of ΔΨm. Additionally, the mPTP opening rate was increased after HR injury but was decreased to normal levels after DUSP1 overexpression (Fig. 2E-F). As a consequence of the dissipated ΔΨm and long-lasting mPTP opening, the pro-apoptotic factor cyt-c is released from the mitochondria into the cytoplasm via western blots (Fig. 2G-H). However, regaining of DUSP1 limited the cyt-c leakage. Other markers related to the mitochondrial apoptosis such as caspase3, caspase9 and mito-Bax were increased in HR-treated cells but reduced in cells transfected with Ad-DUSP1 (Fig. 2G-O). In contrast, the anti-apoptotic proteins such as Bcl2, c-IAP1 and survivin were reduced after HR attack but returned to the normal levels after DUSP1 overexpression (Fig. 2G-O). Notably, if application of fission activator FCCP in DUSP1-overexpressed cells, the protective role of DUSP1 in ΔΨm, mPTP opening and mitochondrial apoptosis disappeared. These data identified DUSP1 as the defender of mitochondrial homeostasis via repression of mitochondrial fission.
3.4. DUSP1 regulated Mff-related mitochondrial fission via JNK
To explore the mechanism by which DUSP1 regulated mitochondrial fission, we focused on Drp1 and its receptors (Mff, Fis1, Mid49 and Mid51). Drp1 shuttled on the mitochondrial surface is dependent on its receptor. Interestingly, IR mainly increased Mff receptors rather than the other receptors, and the reintroduction of DUSP1 decreased the expression of Mff but not of mitochondrial fission 1 (Fis1), mitochondrial dynamics proteins of 49 kDa or 51 kDa (Mid49 and Mid51) (Fig. 3A-B).
Fig. 3.
DUSP1 handled fission via JNK/Mff pathways. A-B. The changes of mitochondrial fission receptors. Only Mff was increased in response to HR injury and was also handled by DUSP1. C-E. DUSP1 inactivated Mff via JNK. SP600125 (SP) is the inhibitor of JNK and it was used to block the HR-mediated JNK phosphorylation. Anisomycin (Ani) is the activator of JNK, which was used to re-activate the JNK activity in DUSP1-overepxressed cells. F. The co-localization of Drp1 and mitochondria. The pictures under each micrograph represent the amplification of the white square. More Drp1 was located on fragmented mitochondria, while the reintroduction of DUSP1 reduced the Drp1 migration on mitochondria. G. The qPCR assay was used to analyze the change of Mff expression. H-I. Chromatin-anti-p-JNK immunoprecipitation (ChIP) assay displayed that p-JNK interacted with the Mff promoter under HR injury, but it did not in DUSP1-overexpression or JNK inhibitor-treated cells. Immunoglobulin-G (IgG) was used as negative control, and anti-histone-3 (α-H3) as a positive control; input demonstrated equal amplification of DNA in non-immunoprecipitated samples; IP corresponds to the amplified DNA band in the immunoprecipitated samples. #P < 0.05 vs the control group; *P < 0.05 vs HR+Ad-ctrl group; @P < 0.05 vs HR+Ad-DUSP1 group.
DUSP1 is a dual-specificity phosphatase that specifically targets JNK and renders JNK inactive, we reasoned that the regulatory effect of DUSP1 on Mff may be relied on JNK activity. Inhibition of JNK via SP600125 (SP) reversed the upregulation of Mff under the IR stimulation (Fig. 3C-E). In contrast, reactivation of JNK in DUSP1-overexpressed cells via Anisomycin (Ani), the Mff expression was re-elevated (Fig. 3C-E). Furthermore, to observe the translocation of Drp1 from cytoplasm to mitochondria, immunofluorescence was used. As shown in Fig. 3F, in the control cells, little Drp1 was located on the mitochondria. After HR injury, mitochondria divided into several fragmentations which were labeled by Drp1. This tendency was inhibited by SP or DUSP1 overexpression. Notably, activation of JNK re-induced mitochondria debris which were marked by Drp1. These data indicated that DUSP1 handled Mff-related fission via JNK activity.
To further explore how DUSP1/JNK modified the Mff content, we first examined Mff gene transcription. As shown in Fig. 3G, Mff transcription was increased in response to the HR injury. However, the reintroduction of DUSP1 or the inhibition of JNK markedly reduced the Mff transcription. If activation of JNK in DUSP1-overexpressed cells, the Mff transcription was re-elevated. These data hinted that JNK may contribute to Mff gene transcription. Through chromatin-anti-p-JNK immunoprecipitation (ChIP) assay, we demonstrated that p-JNK structurally bound to Mff promoter (Fig. 3H-I). In response to HR injury, p-JNK could interact with Mff promoter (Fig. 3H-I). Whereas SP inhibited this tendency (Fig. 3H-I), similar to the results obtained from DUSP1-overexpressed cells. In contrast, JNK activator Ani re-caused the interaction between p-JNK and Mff promoter in DUSP1-overexpressed cells. Collectively, these data suggested that loss of DUSP1 under IR injury led to an increase in Mff-related mitochondrial fission that occurred at least partially through JNK-mediated Mff transcription.
3.5. DUSP1 deficiency promoted excessive mitophagy leading to mitochondrial energy disorder
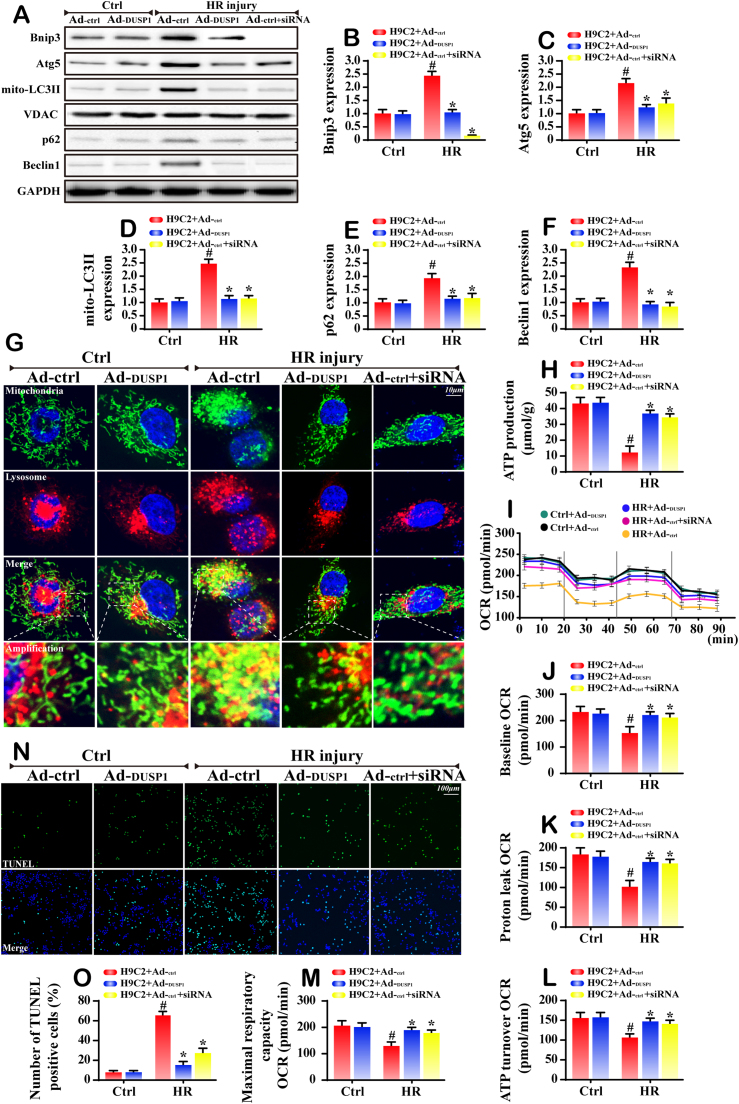
In addition to mitochondrial fission, mitophagy is another mechanism that preserves mitochondrial homeostasis. Fission is reportedly accompanied by mitophagy, which aggravates mitochondrial injury facilitating cellular death via excessive self-consumption. As shown in Fig. 4A-F, we noticed that IR robustly increased the markers related to mitophagy including mito-LC3II, Atg5, Beclin1 and p62. However, these changes were reversed by the overexpression of DUSP1. The mitophagy was activated by its receptor (Fundc1, Bnip3 and Parkin). For Bnip3-required mitophagy, few references exist describing its role in cardiac IR injury, although several studies have hinted that Bnip3 promotes the cellular apoptosis. Based on these, we explored the role of Bnip3-related mitophagy in cardiac IR injury. In the present study, Bnip3 expression was upregulated in HR-treated cells but reduced to normal levels in DUSP1-overexpressed cells (Fig. 4A-B). Notably, once knockdown Bnip3 expression via siRNA, the markers of mitophagy were all decreased. Therefore, these data indicated that IR injury activated the mitophagy via Bnip3. Furthermore, we used the immunofluorescence of mitochondria and lysosome to observe the mitophagy. After loss of Bnip3, the overlap of mitochondria and lysosome was inhibited, comparable to the results obtained from cells transfected with Ad-DUSP1 (Fig. 4G).
Fig. 4.
DUSP1 overexpression was involved in mitophagy inhibition via Bnip3. A-F. Western blots was used to analyze the proteins related to mitophagy. The siRNA against Bnip3 was used to knockdown the expression of Bnip3, which was considered as the negative control group. G. The immunofluorescence assay of mitochondria and lysosome. In the amplified panel, the yellow fluorescence is indicative of mitophagy. H. The ATP production was measured with DUSP1 overepxression or Bnip3 silence. I-M. The OCR assay was used to obverse the mitochondrial respiratory function. Data are shown as the means ± SEM. O-N. TUNEL assay was used to explore the role of Bnip3-related mitophagy in cell death. #P < 0.05 vs the control group; *P < 0.05 vs HR+Ad-ctrl group.
The central role of mitochondria is to generate the energy and fuels the cellular metabolism, we detected the change of mitochondrial metabolism in Bnip3-related mitophagy. HR injury reduced the ATP production and this change was recused by DUSP1 overexpression or Bnip3 knockdown (Fig. 4H). Furthermore, the oxygen consumption rate (OCR) assay was carried out to observe mitochondrial respiratory function. The OCR was decreased after HR injury (Fig. 4I). However, DUSP1 overexpression or Bnip3 knockdown, could restore the OCR. Moreover, compared with control cells, basal respiration rates were significantly decreased with HR treatment but increased after DUSP1 overexpression or Bnip3 deletion (Fig. 4J). Furthermore, proton leaking, maximal respiratory capacity and ATP turnover were significantly reduced in HR-treated cells compared with the control group (Fig. 4K-M). However, DUSP1 overexpression or Bnip3 ablation improved the mitochondrial respiratory parameters. Furthermore, TUNEL assay illustrated that the excessive Bnip3-related mitophagy was associated with more cellular apoptosis in response to HR stimulation. Loss of Bnip3 or overexpression of DUSP1 could reverse the cellular viability (Fig. 4N-O). Altogether, these data illustrated that DUSP1 maintained mitochondrial energy balance via limiting Bnip3-related mitophagy.
3.6. Bnip3-related mitophagy was activated by JNK via post-transcriptional phosphorylation
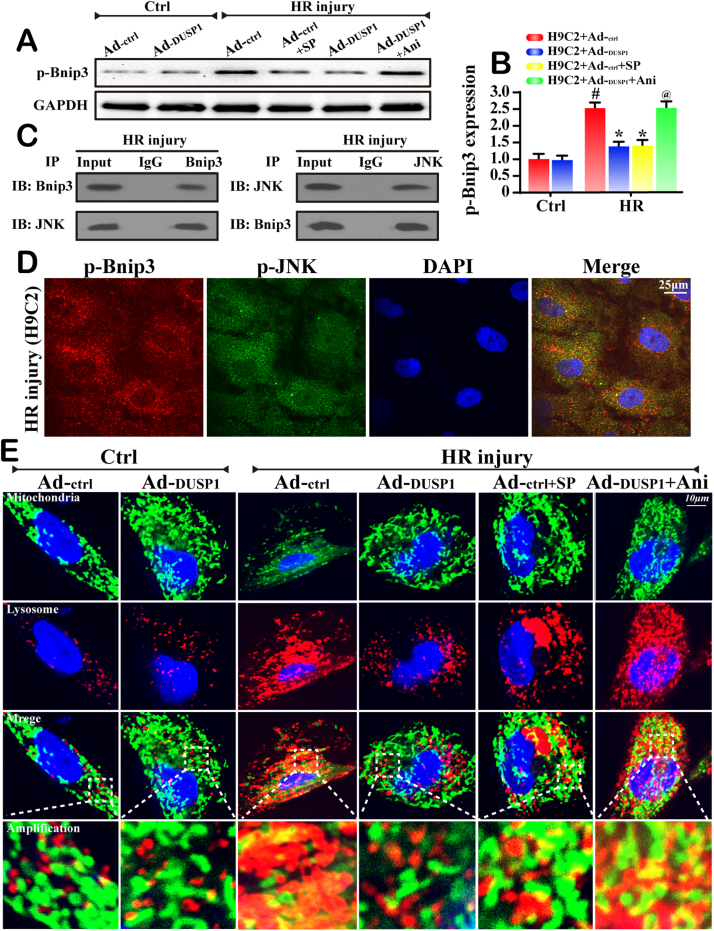
Bnip3-mediated mitophagy was activated via post-transcriptional phosphorylation modification at Ser17 and the phosphorylated Bnip3 has an increased capacity of Bnip3 to interact with LC3. In the current study, we found that HR injury promoted the Bnip3 phosphorylation at Ser17 site (Fig. 5A-B). However, the phenotypic change was recused by DUSP1 overexpression. Furthermore, to demonstrate whether JNK was also involved in the Bnip3 modification, JNK's inhibitor and agonist were used respectively. Under HR injury, SP treatment reduced the p-Bnip3 expression (Fig. 5A-B), similar to the results obtained from DUSP1-overexpressed cells. However, activation of JNK pathway in DUSP1-overexpressed cells, the p-Bnip3 content was elevated (Fig. 5A-B). These data indicated that JNK was also involved in the DUSP1-regulated Bnip3 modification.
Fig. 5.
DUSP1 inactivated Bnip3 via inhibiting JNK-mediated Bnip3 phosphorylation. A-B. Bnip3 phosphorylation was analyzed via western blots. SP600125 (SP) is the inhibitor of JNK and it was used to block the HR-mediated JNK phosphorylation. Anisomycin (Ani) is the activator of JNK, which was used to reactivate the JNK activity in DUSP1-overepxressed cells. C. Cell lysates were immunoprecipitated with anti-Bnip3 or anti-JNK antibodies, followed by immunoblotting with anti-JNK or anti-Bnip3 antibody. IgG was used as a control. D. The cytoplasmic localization of JNK-Bnip3 interaction has also been observed. The confocal microscopy showed approximately 80% colocalization of JNK and Bnip3 in the cytoplasm of H9C2 cells. E. The co-immunofluorescence of mitochondria and lysosomes. DUSP1 alleviated the overlap of mitochondria and lysosomes. #P < 0.05 vs the control group; *P < 0.05 vs HR+Ad-ctrl group; @P < 0.05 vs HR+Ad-DUSP1 group.
To study the mechanism by which JNK activated Bnip3 phosphorylation, we examined the possibility whether JNK interacted with Bnip3. Surprisingly, coimmunopre-cipitation (co-IP) assay showed a constitutive interaction between endogenous p-JNK and Bnip3 in H9C2 cells under HR injury (Fig. 5C). Considering that Bnip3 often resides in the cytoplasm We therefore guessed that the p-JNK-Bnip3 interaction took places in the cytoplasm. Indeed, confocal microscopy showed approximately 80% co-localization of p-JNK and Bnip3 in the cytoplasm of H9C2 cells (Fig. 5D). These results suggested that p-JNK can interact with Bnip3 in the cytoplasm.
Next, through co-staining of mitochondria and lysosome, we further demonstrated that JNK inhibition facilitated the disassociation between mitochondria and lysosome in the context of HR injury (Fig. 5E). In contrast, activation of JNK re-caused the fusion between mitochondria and lysosome in DUSP1-overexpressed cells (Fig. 5E). This data confirmed our hypothesis that JNK functioned upstream of Bnip3-mediated mitophagy.
4. Discussion
Ample evidence highlights the involvement of DUSP1 in the pathophysiological process of cancer [35], [36]. However, little is known regarding the role of DUSP1 in acute cardiac IR injury. In this study, we found that (1) IR injury greatly caused DUSP1 downregulation, which was instrumental for scar expansion, cardiac dysfunction, and cellular death; (2) the loss of DUSP1 robustly led to an increase in JNK phosphorylation; (3) activated JNK increased the expression of Mff, which initiated fatal mitochondrial fission; (4) excessive fission unfortunately launched the caspase9-related mitochondrial apoptosis pathway, resulting into the extensive cell death; (5) meanwhile, JNK also phosphorylated the Bnip3 and elevated the mitophagy activity; (6) mitophagy overtly consumed mitochondrial mass impairing the mitochondrial energy production (Fig. 6). To the best of our knowledge, this is the first study to describe the role of the DUSP1/JNK pathways as a mechanism responsible for cardiac IR injury via the mediation of Mff-required fission and Bnip3-related mitophagy.
Fig. 6.
IR injury caused DUSP1 downregulation, leading to an increase in JNK phosphorylation. Activated JNK increased the expression of Mff, which initiated fatal mitochondrial fission. Excessive fission induced the pro-apoptotic cyt-c leakage from mitochondria into cytoplasm, launching the caspase9 related mitochondrial apoptosis. Meanwhile, JNK also phosphorylated Bnip3 and elevated mitophagy activity. Extensive mitophagy overtly consumed mitochondrial mass impairing the mitochondrial energy production. However, recovery of DUSP1 activity could cut off the excessive fission and mitophagy, providing pro-survival advantages for the heart in the context of IR injury.
The DUSP protein family comprises 11 members and three classes (Class I: DUSP1, 2, 4, and 5 are found in the nucleus; Class II: DUSP6, 7, and 9 are found in the cytoplasm; and class III: DUSP8, 10, and 16 are found both in the nucleus and cytoplasm) [37] that can dephosphorylate both the serine/threonine and tyrosine residues of their substrates, particularly JNK [38]. Although it has been over a decade since the discovery of DUSP1, its precise functions have not been determined to date. In human prostate tumors, DUSP1 expression increases during the early stages of carcinogenesis and progressively decreases with higher histologic grades and metastasis [39], suggesting that DUSP1 is inversely related to apoptosis. Paradoxically, in breast cancer, DUSP1 overexpression increases cancer apoptosis, and DUSP1 downregulation is significantly correlated with an advanced tumor stage, positive recurrence and poor survival [40]. Considerable disagreements exist with regard to the functional effects of DUSP1 signaling because both pro- and anti-apoptotic regulatory roles have been reported. One possibility is that the opposite functions in cellular survival are cell-context dependent [41]. In the present study, we uncovered that DUSP1 was downregulated in the context of cardiac IR injury and that reintroduction of DUSP1 protected the heart tissue against an acute IR injury, supporting the pro-survival effects of DUSP1 on cardiomyocytes. In fact, the diversity of DUSP1 in cellular survival is reminiscent of the different effects of MAPKs in cardiac IR injury. ERK is activated by various growth factors, controls cell growth and promotes cellular survival in IR injury [42]. In contrast, p38 and JNK are activated by stress conditions, leading to the cellular apoptosis via multiple mechanisms [43]. Several studies have argued that the intensity and balance of MAPK activities are important determinants of the cell fate. However, considering the protective role played by DUSP1 in IR injury, we proposed that the pro-apoptosis functions exerted by MAPKs could occupy a dominant position under IR injury stimulation.
Mitochondrial fission and fragmentation are commonly observed in cells with IR injury [44], [45]. During mitochondrial fission, Drp1 is a critical executor because GFP-tagged Drp1 has been demonstrated to assemble into a ring structure, and the ring constricts along the mitochondrial tubules [46]. Remarkably, Drp1 recruitment on mitochondria requires its corresponding receptors that are located on the mitochondrial outer membrane. Accumulating evidence indicates that four proteins are involved in fission: Fis1, Mff, MiD49 and MiD51 [47], [48]. Our previous studies have described that Mff was increased in reaction to IR injury, which was involved in Drp-required mitochondrial fission [3]. However, the exact mechanisms underlying the Mff-upregulation process remain unclear. The work presented here identifies that DUSP1 is the regulatory system of Mff-required mitochondrial fragmentation. IR injury downregulated the content of DUSP1, leading to the phosphorylated activation of the JNK pathways. Phosphorylated JNK could translocate into the nucleus to promote Mff expression. Thus, DUSP1 likely lies upstream of Mff-required mitochondrial fission in the infarcted heart.
The mitochondrial quantity is maintained via mitochondrial dynamics and/or the selective clearance of damaged organelles [49]. Mitochondrial autophagy (or mitophagy) is thus considered the core mechanism of both mitochondrial quality and quantity control [50]. Increasing evidence indicates the key role of mitophagy receptors, such as Atg32 in yeast, and Bnip3 or FUNDC1 in mammals, in the clearance of mitochondria [5]. Bnip3 has been found to play a critical role in cardiomyocyte cell death following ischemic and non-ischemic injuries [51], [52]. Bnip3 can also interact with LC3 family proteins via its LIR motifs facing the cytosol, thereby mediating mitophagy [53]. However, the detailed role of Bnip3-mediated mitophagy in cardiac IR injury is not completely understood. In the present study, IR injury increased the content of Bnip3, followed by an increase in the numbers of mitochondria swallowed by lysosomes, indicating elevated levels of mitophagy. These changes were accompanied by mitochondrial energy imbalance, suggesting the contributory effects of Bnip3-required mitophagy in cardiac IR injury. These data were consistent with previous studies that restoring Bnip3 had a biphasic effect [54], first elevating the mitophagy levels but later leading to cell death. Notably, Bnip3-mediated cell death is actually independent of Apaf-1, caspase activation and the nuclear translocation of apoptosis-inducing factors [55], [56], although Bnip3 was first classified as a pro-apoptotic factor in cardiomyocytes under hypoxia [57]. Several studies argued that this type of death is necrosis-like cell death [56], while our data identified that the Bnip3-induced cellular death was associated with mitophagy. Notably, we illustrated that excessive Bnip3-mediated mitophagy promoted the ATP shortage, which is the key feature of necrosis. Accordingly, more data are needed to further identify the types of cell deaths mediated by Bnip3-induced mitophagy. Based on our data, we argued that the Bnip3-induced mitophagy could increase cell death via the excessive elimination of mitochondria, leading to an ATP/energy production disorder due to a robust shortage of mitochondria.
Ample evidences have confirmed that mitochondrial fission could promote the mitophagy via inducing mitochondria division into several small debris [58], [59]. Further, some studies have pointed out that Drp1 is the direct regulator of mitophagy [60]. However, there's no consensus yet about the role of fission-related mitophagy in cell fate because different mitophagy receptors regulate the mitophagy function. For example, Parkin-involved mitophagy exacerbates cell death [9] whereas FUNDC1-related mitophagy provides beneficial effects on cell survival under IR injury [10]. Accordingly, we think that mitophagy activity can be definitively modulated by fission. However, mitophagy function is regulated by its receptors. In the present study, we identified that DUSP1 could both handle the mitochondrial fission and mitophagy, suggesting the multiple actions of DUSP1 on mitochondrial homeostasis. Furthermore, we found that the decrease in DUSP1 occurred at the early stage of reperfusion. This change may be attributed to the decrease in DUSP1 transcription or the increase in DUSP1 proteolytic degradation. Considering that IR injury happens in a short time, we guess that protein degradation may be responsible for the DUSP1 down-regulation. However, more researches are needed to support our notion.
In conclusion, IR injury was associated with degraded DUSP1 activity, leading to the activation of JNK. Increased JNK enhanced Mff and Bnip3 activation, which caused excessive mitochondrial fission and mitophagy, promoting cellular death under IR injury. The reintroduction of DUSP1 could blunt the JNK pathways and subsequently cut off the Mff-mediated fission and Bnip3-required mitophagy, finally preventing myocardial IR injury
Conflict of interest statement
The authors declared that they have no conflicts of interest.
Finding
This study was financially supported by grants from the National Natural Science Foundation of China (No. 81030002, 81270186, 81441008, and 81102079) and the National Key Research and Development Program of China (2016YFC1300304). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnotes
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.redox.2017.11.004.
Appendix A. Supplementary material
Fig. S1.
Reintroduction of DUSP1 improved cardiac function. A. After 24-h reperfusion, the casapse3 activity in infarcted myocardium was detected. B-C. The western blots of caspase3 in infarcted myocardium after 24-h reperfusion. C-E. The content of LDH, Troponin T, and CK-MB, which are markers of cardiac damage. G-I. Cardiac function is evaluated through echocardiography. Quantitative analysis of the data derived from echocardiography. J. EM was used to observe the ultrastructural changes after IR injury. Compared to the WT mice, the myofibrils in the DUSP1TG mice were regularly arranged. In addition, the Z lines were straight. Most mitochondria had normal structures. K-P. The cardiomyocytes contractile properties in WT and DUSP1TG in the context of IR injury. +dL/dt is the maximal velocity of shortening. –dL/dt is the maximal velocity of relengthening. TPS is the time-to-peak shortening. TR90 is the time-to-90% relengthening. #P < 0.05 vs the sham group; *P < 0.05 vs IR+WT group. FS indicates fractional shortening; LVDd, left ventricular diastolic dimension; LVEF, left ventricular ejection fraction.
References
- 1.Chen W.R., Chen Y.D., Tian F., Yang N., Cheng L.Q., Hu S.Y., Wang J., Yang J.J., Wang S.F., Gu X.F. Effects of liraglutide on reperfusion injury in patients with ST-segment-elevation myocardial infarction. Circ. Cardiovasc Imaging. 2016;9(12) doi: 10.1161/CIRCIMAGING.116.005146. [DOI] [PubMed] [Google Scholar]
- 2.Chen W.R., Hu S.Y., Chen Y.D., Zhang Y., Qian G., Wang J., Yang J.J., Wang Z.F., Tian F., Ning Q.X. Effects of liraglutide on left ventricular function in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Am. Heart J. 2015;170(5):845–854. doi: 10.1016/j.ahj.2015.07.014. [DOI] [PubMed] [Google Scholar]
- 3.Zhou H., Hu S., Jin Q., Shi C., Zhang Y., Zhu P., Ma Q., Tian F., Chen Y. Mff-dependent mitochondrial fission contributes to the pathogenesis of cardiac microvasculature ischemia/reperfusion injury via induction of mROS-mediated cardiolipin oxidation and HK2/VDAC1 disassociation-involved mPTP opening. J. Am. Heart Assoc. 2017;6(3) doi: 10.1161/JAHA.116.005328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ni H.M., Williams J.A., Ding W.X. Mitochondrial dynamics and mitochondrial quality control. Redox Biol. 2015;4:6–13. doi: 10.1016/j.redox.2014.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bravo-San Pedro J.M., Kroemer G., Galluzzi L. Autophagy and mitophagy in cardiovascular disease. Circ. Res. 2017;120(11):1812–1824. doi: 10.1161/CIRCRESAHA.117.311082. [DOI] [PubMed] [Google Scholar]
- 6.Georgakopoulos N.D., Wells G., Campanella M. The pharmacological regulation of cellular mitophagy. Nat. Chem. Biol. 2017;13(2):136–146. doi: 10.1038/nchembio.2287. [DOI] [PubMed] [Google Scholar]
- 7.Ney P.A. Mitochondrial autophagy: origins, significance, and role of BNIP3 and NIX. Biochim. Biophys. Acta. 2015;1853(10 Pt B):2775–2783. doi: 10.1016/j.bbamcr.2015.02.022. [DOI] [PubMed] [Google Scholar]
- 8.Zhou H., Li D., Zhu P., Hu S., Hu N., Ma S., Zhang Y., Han T., Ren J., Cao F., Chen Y. Melatonin suppresses platelet activation and function against cardiac ischemia/reperfusion injury via PPARgamma/FUNDC1/mitophagy pathways. J. Pineal Res. 2017 doi: 10.1111/jpi.12438. [DOI] [PubMed] [Google Scholar]
- 9.Zhou H., Zhang Y., Hu S., Shi C., Zhu P., Ma Q., Jin Q., Cao F., Tian F., Chen Y. Melatonin protects cardiac microvasculature against ischemia/reperfusion injury via suppression of mitochondrial fission-VDAC1-HK2-mPTP-mitophagy axis. J. Pineal Res. 2017;63(1) doi: 10.1111/jpi.12413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhou H., Zhu P., Guo J., Hu N., Wang S., Li D., Hu S., Ren J., Cao F., Chen Y. Ripk3 induces mitochondrial apoptosis via inhibition of FUNDC1 mitophagy in cardiac IR injury. Redox Biol. 2017;13:498–507. doi: 10.1016/j.redox.2017.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shi C., Cai Y., Li Y., Li Y., Hu N., Ma S., Hu S., Zhu P., Wang W., Zhou H. Yap promotes hepatocellular carcinoma metastasis and mobilization via governing cofilin/F-actin/lamellipodium axis by regulation of JNK/Bnip3/SERCA/CaMKII pathways. Redox Biol. 2017;14:59–71. doi: 10.1016/j.redox.2017.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang Z., Han Z., Ye B., Dai Z., Shan P., Lu Z., Dai K., Wang C., Huang W. Berberine alleviates cardiac ischemia/reperfusion injury by inhibiting excessive autophagy in cardiomyocytes. Eur. J. Pharmacol. 2015;762:1–10. doi: 10.1016/j.ejphar.2015.05.028. [DOI] [PubMed] [Google Scholar]
- 13.Barteczek P., Li L., Ernst A.S., Bohler L.I., Marti H.H., Kunze R. Neuronal HIF-1alpha and HIF-2alpha deficiency improves neuronal survival and sensorimotor function in the early acute phase after ischemic stroke. J. Cereb. Blood Flow. Metab. 2017;37(1):291–306. doi: 10.1177/0271678X15624933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sabathie M., De Coninck L., Fabre P., Michel G. Use of diarginine alpha-ketoglutarate following abdominal surgery. Apropos of 30 cases. Sem. Hop. Ther. 1975;51(9):457–458. [PubMed] [Google Scholar]
- 15.Johnson G.L., Lapadat R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science. 2002;298(5600):1911–1912. doi: 10.1126/science.1072682. [DOI] [PubMed] [Google Scholar]
- 16.Kwak S.P., Hakes D.J., Martell K.J., Dixon J.E. Isolation and characterization of a human dual specificity protein-tyrosine phosphatase gene. J. Biol. Chem. 1994;269(5):3596–3604. [PubMed] [Google Scholar]
- 17.Candas D., Li J.J. MKP1 mediates resistance to therapy in HER2-positive breast tumors. Mol. Cell Oncol. 2015;2(4):e997518. doi: 10.1080/23723556.2014.997518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang Z., Xu J., Zhou J.Y., Liu Y., Wu G.S. Mitogen-activated protein kinase phosphatase-1 is required for cisplatin resistance. Cancer Res. 2006;66(17):8870–8877. doi: 10.1158/0008-5472.CAN-06-1280. [DOI] [PubMed] [Google Scholar]
- 19.Li L., Gao G., Shankar J., Joshi B., Foster L.J., Nabi I.R. p38 MAP kinase-dependent phosphorylation of the Gp78 E3 ubiquitin ligase controls ER-mitochondria association and mitochondria motility. Mol. Biol. Cell. 2015;26(21):3828–3840. doi: 10.1091/mbc.E15-02-0120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wainstein E., Seger R. The dynamic subcellular localization of ERK: mechanisms of translocation and role in various organelles. Curr. Opin. Cell Biol. 2016;39:15–20. doi: 10.1016/j.ceb.2016.01.007. [DOI] [PubMed] [Google Scholar]
- 21.Braz J.C., Bueno O.F., Liang Q., Wilkins B.J., Dai Y.S., Parsons S., Braunwart J., Glascock B.J., Klevitsky R., Kimball T.F., Hewett T.E., Molkentin J.D. Targeted inhibition of p38 MAPK promotes hypertrophic cardiomyopathy through upregulation of calcineurin-NFAT signaling. J. Clin. Investig. 2003;111(10):1475–1486. doi: 10.1172/JCI17295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bueno O.F., De Windt L.J., Lim H.W., Tymitz K.M., Witt S.A., Kimball T.R., Molkentin J.D. The dual-specificity phosphatase MKP-1 limits the cardiac hypertrophic response in vitro and in vivo. Circ. Res. 2001;88(1):88–96. doi: 10.1161/01.res.88.1.88. [DOI] [PubMed] [Google Scholar]
- 23.Winiarska K., Dzik J.M., Labudda M., Focht D., Sierakowski B., Owczarek A., Komorowski L., Bielecki W. Melatonin nephroprotective action in Zucker diabetic fatty rats involves its inhibitory effect on NADPH oxidase. J. Pineal Res. 2016;60(1):109–117. doi: 10.1111/jpi.12296. [DOI] [PubMed] [Google Scholar]
- 24.Das N., Mandala A., Naaz S., Giri S., Jain M., Bandyopadhyay D., Reiter R.J., Roy S.S. Melatonin protects against lipid-induced mitochondrial dysfunction in hepatocytes and inhibits stellate cell activation during hepatic fibrosis in mice. J. Pineal Res. 2017;62(4) doi: 10.1111/jpi.12404. [DOI] [PubMed] [Google Scholar]
- 25.Chen W., Zou P., Zhao Z., Chen X., Fan X., Vinothkumar R., Cui R., Wu F., Zhang Q., Liang G., Ji J. Synergistic antitumor activity of rapamycin and EF24 via increasing ROS for the treatment of gastric cancer. Redox Biol. 2016;10:78–89. doi: 10.1016/j.redox.2016.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kakimoto P.A., Kowaltowski A.J. Effects of high fat diets on rodent liver bioenergetics and oxidative imbalance. Redox Biol. 2016;8:216–225. doi: 10.1016/j.redox.2016.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hu N., Han X., Lane E.K., Gao F., Zhang Y., Ren J. Cardiac-specific overexpression of metallothionein rescues against cigarette smoking exposure-induced myocardial contractile and mitochondrial damage. PLoS One. 2013;8(2):e57151. doi: 10.1371/journal.pone.0057151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhu H., Jin Q., Li Y., Ma Q., Wang J., Li D., Zhou H., Chen Y. Melatonin protected cardiac microvascular endothelial cells against oxidative stress injury via suppression of IP3R-[Ca2+]c/VDAC-[Ca2+]m axis by activation of MAPK/ERK signaling pathway. Cell Stress Chaperones. 2017 doi: 10.1007/s12192-017-0827-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.King A.L., Mantena S.K., Andringa K.K., Millender-Swain T., Dunham-Snary K.J., Oliva C.R., Griguer C.E., Bailey S.M. The methyl donor S-adenosylmethionine prevents liver hypoxia and dysregulation of mitochondrial bioenergetic function in a rat model of alcohol-induced fatty liver disease. Redox Biol. 2016;9:188–197. doi: 10.1016/j.redox.2016.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mailloux R.J., Treberg J.R. Protein S-glutathionlyation links energy metabolism to redox signaling in mitochondria. Redox Biol. 2016;8:110–118. doi: 10.1016/j.redox.2015.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pariente R., Pariente J.A., Rodriguez A.B., Espino J. Melatonin sensitizes human cervical cancer HeLa cells to cisplatin-induced cytotoxicity and apoptosis: effects on oxidative stress and DNA fragmentation. J. Pineal Res. 2016;60(1):55–64. doi: 10.1111/jpi.12288. [DOI] [PubMed] [Google Scholar]
- 32.Zhou H., Yang J., Xin T., Zhang T., Hu S., Zhou S., Chen G., Chen Y. Exendin-4 enhances the migration of adipose-derived stem cells to neonatal rat ventricular cardiomyocyte-derived conditioned medium via the phosphoinositide 3-kinase/Akt-stromal cell-derived factor-1alpha/CXC chemokine receptor 4 pathway. Mol. Med. Rep. 2015;11(6):4063–4072. doi: 10.3892/mmr.2015.3243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mao L., Dauchy R.T., Blask D.E., Dauchy E.M., Slakey L.M., Brimer S., Yuan L., Xiang S., Hauch A., Smith K., Frasch T., Belancio V.P., Wren M.A., Hill S.M. Melatonin suppression of aerobic glycolysis (Warburg effect), survival signalling and metastasis in human leiomyosarcoma. J. Pineal Res. 2016;60(2):167–177. doi: 10.1111/jpi.12298. [DOI] [PubMed] [Google Scholar]
- 34.Zhou H., Yang J., Xin T., Li D., Guo J., Hu S., Zhou S., Zhang T., Zhang Y., Han T., Chen Y. Exendin-4 protects adipose-derived mesenchymal stem cells from apoptosis induced by hydrogen peroxide through the PI3K/Akt-Sfrp2 pathways. Free Radic. Biol. Med. 2014;77:363–375. doi: 10.1016/j.freeradbiomed.2014.09.033. [DOI] [PubMed] [Google Scholar]
- 35.Kesarwani M., Kincaid Z., Gomaa A., Huber E., Rohrabaugh S., Siddiqui Z., Bouso M.F., Latif T., Xu M., Komurov K., Mulloy J.C., Cancelas J.A., Grimes H.L., Azam M. Targeting c-FOS and DUSP1 abrogates intrinsic resistance to tyrosine-kinase inhibitor therapy in BCR-ABL-induced leukemia. Nat. Med. 2017;23(4):472–482. doi: 10.1038/nm.4310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lopes L.J.S., Tesser-Gamba F., Petrilli A.S., De Seixas Alves M.T., Garcia-Filho R.J., Toledo S.R.C. MAPK pathways regulation by DUSP1 in the development of osteosarcoma: potential markers and therapeutic targets. Mol. Carcinog. 2017;56(6):1630–1641. doi: 10.1002/mc.22619. [DOI] [PubMed] [Google Scholar]
- 37.Shen J., Zhang Y., Yu H., Shen B., Liang Y., Jin R., Liu X., Shi L., Cai X. Role of DUSP1/MKP1 in tumorigenesis, tumor progression and therapy. Cancer Med. 2016;5(8):2061–2068. doi: 10.1002/cam4.772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wancket L.M., Frazier W.J., Liu Y. Mitogen-activated protein kinase phosphatase (MKP)-1 in immunology, physiology, and disease. Life Sci. 2012;90(7–8):237–248. doi: 10.1016/j.lfs.2011.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Magi-Galluzzi C., Mishra R., Fiorentino M., Montironi R., Yao H., Capodieci P., Wishnow K., Kaplan I., Stork P.J., Loda M. Mitogen-activated protein kinase phosphatase 1 is overexpressed in prostate cancers and is inversely related to apoptosis. Lab Investig. 1997;76(1):37–51. [PubMed] [Google Scholar]
- 40.Hou M.F., Chang C.W., Chen F.M., Wang S.N., Yang S.F., Chen P.H., Su J.H., Yeh Y.T. Decreased total MKP-1 protein levels predict poor prognosis in breast cancer. World J. Surg. 2012;36(8):1922–1932. doi: 10.1007/s00268-012-1608-y. [DOI] [PubMed] [Google Scholar]
- 41.Boutros T., Chevet E., Metrakos P. Mitogen-activated protein (MAP) kinase/MAP kinase phosphatase regulation: roles in cell growth, death, and cancer. Pharmacol. Rev. 2008;60(3):261–310. doi: 10.1124/pr.107.00106. [DOI] [PubMed] [Google Scholar]
- 42.Feng Y., Madungwe N.B., Da Cruz Junho C.V., Bopassa J.C. Activation of G protein-coupled estrogen receptor 1 at the onset of reperfusion protects the myocardium against ischemia/reperfusion injury by reducing mitochondrial dysfunction and mitophagy. Br. J. Pharmacol. 2017 doi: 10.1111/bph.14033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang X., Wang Y., Shen W., Ma S., Chen W., Qi R. Rosa rugosa flavonoids alleviate myocardial ischemia reperfusion injury in mice by suppressing JNK and p38 MAPK. Microcirculation. 2017 doi: 10.1111/micc.12385. [DOI] [PubMed] [Google Scholar]
- 44.Nduhirabandi F., Lamont K., Albertyn Z., Opie L.H., Lecour S. Role of toll-like receptor 4 in melatonin-induced cardioprotection. J. Pineal Res. 2016;60(1):39–47. doi: 10.1111/jpi.12286. [DOI] [PubMed] [Google Scholar]
- 45.Ursini F., Maiorino M., Forman H.J. Redox homeostasis: the golden mean of healthy living. Redox Biol. 2016;8:205–215. doi: 10.1016/j.redox.2016.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Doi K., Noiri E. Mitochondrial dysfunction in cardiorenal syndrome. Antioxid. Redox Signal. 2016;25(4):200–207. doi: 10.1089/ars.2016.6654. [DOI] [PubMed] [Google Scholar]
- 47.Atkins K., Dasgupta A., Chen K.H., Mewburn J., Archer S.L. The role of Drp1 adaptor proteins MiD49 and MiD51 in mitochondrial fission: implications for human disease. Clin. Sci. (Lond.) 2016;130(21):1861–1874. doi: 10.1042/CS20160030. [DOI] [PubMed] [Google Scholar]
- 48.Marin-Garcia J., Akhmedov A.T. Mitochondrial dynamics and cell death in heart failure. Heart Fail Rev. 2016;21(2):123–136. doi: 10.1007/s10741-016-9530-2. [DOI] [PubMed] [Google Scholar]
- 49.Xiao L., Xu X., Zhang F., Wang M., Xu Y., Tang D., Wang J., Qin Y., Liu Y., Tang C., He L., Greka A., Zhou Z., Liu F., Dong Z., Sun L. The mitochondria-targeted antioxidant MitoQ ameliorated tubular injury mediated by mitophagy in diabetic kidney disease via Nrf2/PINK1. Redox Biol. 2017;11:297–311. doi: 10.1016/j.redox.2016.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Onphachanh X., Lee H.J., Lim J.R., Jung Y.H., Kim J.S., Chae C.W., Lee S.J., Gabr A.A., Han H.J. Enhancement of high glucose-induced PINK1 expression by melatonin stimulates neuronal cell survival: involvement of MT2 /Akt/NF-kappaB pathway. J. Pineal Res. 2017;63(2) doi: 10.1111/jpi.12427. [DOI] [PubMed] [Google Scholar]
- 51.Chinnadurai G., Vijayalingam S., Gibson S.B. BNIP3 subfamily BH3-only proteins: mitochondrial stress sensors in normal and pathological functions. Oncogene. 2008;27(Suppl 1):S114–S127. doi: 10.1038/onc.2009.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Regula K.M., Ens K., Kirshenbaum L.A. Inducible expression of BNIP3 provokes mitochondrial defects and hypoxia-mediated cell death of ventricular myocytes. Circ. Res. 2002;91(3):226–231. doi: 10.1161/01.res.0000029232.42227.16. [DOI] [PubMed] [Google Scholar]
- 53.Rikka S., Quinsay M.N., Thomas R.L., Kubli D.A., Zhang X., Murphy A.N., Gustafsson A.B. Bnip3 impairs mitochondrial bioenergetics and stimulates mitochondrial turnover. Cell Death Differ. 2011;18(4):721–731. doi: 10.1038/cdd.2010.146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Moulis M.F., Millet A.M., Daloyau M., Miquel M.C., Ronsin B., Wissinger B., Arnaune-Pelloquin L., Belenguer P. OPA1 haploinsufficiency induces a BNIP3-dependent decrease in mitophagy in neurons: relevance to dominant optic atrophy. J. Neurochem. 2017;140(3):485–494. doi: 10.1111/jnc.13894. [DOI] [PubMed] [Google Scholar]
- 55.Hamacher-Brady A., Brady N.R., Logue S.E., Sayen M.R., Jinno M., Kirshenbaum L.A., Gottlieb R.A., Gustafsson A.B. Response to myocardial ischemia/reperfusion injury involves Bnip3 and autophagy. Cell Death Differ. 2007;14(1):146–157. doi: 10.1038/sj.cdd.4401936. [DOI] [PubMed] [Google Scholar]
- 56.Vande Velde C., Cizeau J., Dubik D., Alimonti J., Brown T., Israels S., Hakem R., Greenberg A.H. BNIP3 and genetic control of necrosis-like cell death through the mitochondrial permeability transition pore. Mol. Cell Biol. 2000;20(15):5454–5468. doi: 10.1128/mcb.20.15.5454-5468.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bellot G., Garcia-Medina R., Gounon P., Chiche J., Roux D., Pouyssegur J., Mazure N.M. Hypoxia-induced autophagy is mediated through hypoxia-inducible factor induction of BNIP3 and BNIP3L via their BH3 domains. Mol. Cell Biol. 2009;29(10):2570–2581. doi: 10.1128/MCB.00166-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Anzell A.R., Maizy R., Przyklenk K., Sanderson T.H. Mitochondrial quality control and disease: insights into ischemia-reperfusion injury. Mol. Neurobiol. 2017 doi: 10.1007/s12035-017-0503-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wu Q., Luo C.L., Tao L.Y. Dynamin-related protein 1 (Drp1) mediating mitophagy contributes to the pathophysiology of nervous system diseases and brain injury. Histol. Histopathol. 2017;32(6):551–559. doi: 10.14670/HH-11-841. [DOI] [PubMed] [Google Scholar]
- 60.Kageyama Y., Hoshijima M., Seo K., Bedja D., Sysa-Shah P., Andrabi S.A., Chen W., Hoke A., Dawson V.L., Dawson T.M., Gabrielson K., Kass D.A., Iijima M., Sesaki H. Parkin-independent mitophagy requires Drp1 and maintains the integrity of mammalian heart and brain. EMBO J. 2014;33(23):2798–2813. doi: 10.15252/embj.201488658. [DOI] [PMC free article] [PubMed] [Google Scholar]