Abstract
Background:
Biopolymer scaffolds have received great interest in academic and industrial environment because of their supreme characteristics like biological, mechanical, chemical, and cost saving in the biomedical science. There are various attempts for incorporation of biopolymers with cheap natural micro- or nanoparticles like lignin (Lig), alginate, and gums to prepare new materials with enhanced properties.
Methods:
In this work, the electrospinning (ELS) technique as a promising cost-effective method for producing polymeric scaffold fibers was used, which mimics extracellular matrix structure for soft tissue engineering applications. Nanocomposites of Lig and polycaprolactone (PCL) scaffold produced with ELS technique. Nanocomposite containings (0, 5, 10, and 15 wt.%) of Lig were prepared with addition of Lig powder into the PCL solution while stirring at the room temperature. The bioactivity, swelling properties, morphological and mechanical tests were conducted for all the samples to investigate the nanocomposite scaffold features.
Results:
The results showed that scaffold with 10 wt.% Lig have appropriate porosity, biodegradation, minimum fiber diameter, optimum pore size as well as enhanced tensile strength, and young modulus compared with pure PCL. Degradation test performed through immersion of samples in the phosphate-buffer saline showed that degradation of PCL nanocomposites could accelerate up to 10% due to the addition of Lig.
Conclusions:
Electrospun PCL-Lig scaffold enhanced the biological response of the cells with the mechanical signals. The prepared nanocomposite scaffold can choose for potential candidate in the biomedical science.
Keywords: Electrospinning, lignin, nanocomposite, polycaprolactone, scaffold, tissue engineering
Introduction
Synthetic biopolymer nanocomposite scaffolds applied for tissue engineering applications during last two decades.[1,2] Frequently, polymeric nanocomposite scaffolds contain porous shape that readily allows cell differentiation and vascularization used for bone and skin tissue substitute. Lignin (Lig) polymer is an abundant, biodegradable, natural biomaterial which received researcher's attention in biomedical field due to its unique mechanical, chemical, biological properties, low cost, and environmental concerns. Lig has proved antioxidant and antiviral effects in human body. The main problem with some polymers is their weak chemical stability and mechanical properties. Therefore, decreasing the crystallinity and hydrophobicity are two known approaches to improve biodegradability and increase the chemical stability.[2,3,4,5] Lig with its amorphous nature and functional groups (e.g., hydroxyl groups OH−) could guarantee better function for polycaprolactone (PCL) based scaffolds. Lig has not been significantly utilized in biomaterial field.[1,2,3] Electrospinning (ELS) is an inexpensive method of manufacturing tissue engineering scaffolds which has gained great interest in recent years for producing nano and submicron polymeric fibers.[2] PCL electrospun nanocomposites mimic extracellular matrix (ECM) and could act as a supportive scaffold for tissue engineering applications.[3,4] Nanocomposite polymer scaffold enhanced the biological response of the cells with the mechanical signals and show better cell attraction. PCL is a biodegradable semicrystalline synthetic polyester with proper mechanical properties which is capable of supporting a wide variety of cell types in tissue engineering application.[5] The hydrophilicity, porosity, mechanical, and biological behavior of the scaffold is the most important factors affecting cell adhesion and differentiation. The hydrophobic nature and low degradation rate of PCL have limited its usage in tissue engineering, whereas compounding with hydrophilic polymers could increase the mechanical and biological properties of the such polymers.[5,6] In the work conducted by Kim et al.,[7] PCL/alginate biocomposite showed improved hydrophilicity, cell attachment, proliferation, and degradation by adding alginate polymer. PCL electrospun scaffolds have been used widely for skin, bone, and cartilage tissue engineering.[8,9,10,11,12] Utilizing Lig in the polymeric matrix could take advantage of antioxidant, biocompatibility, and biodegradation properties also improves hydrophilicity in tissue engineering applications. Compounding different Lig types in the various polymeric matrix contributes different physical and mechanical properties due to polymer functional groups, molecular weight, and other properties.[9] Nitz et al. introduced that addition of up to 50 wt.%, Lig increased Ecoflex® commercial polyester from BASF (Ludwigshafen, Germany) and polyamide 11 young's modulus, whereas decreased BAK® commercial polyesteramide from BASF (Ludwigshafen, Germany) young's modulus. Increasing Lig content up to 30 wt.% leads to decrease yield stress of BAK® and Ecoflex®, whereas increase yield stress of the polyamide.[10] In this research, PCL/Lig nanocomposite scaffolds prepared by ELS technique and the Lig effects on the morphology, biological, and mechanical characteristics have been investigated. Generally, the present work confirmed that the PCL–Lig nanocomposite scaffolds had proper mechanical and morphological properties and proper cell inducement characteristic as assuring candidates for tissue engineering applications with better degradation and biological characteristic.
Materials and Methods
Materials Preparations
An amount of 0.1 g PCL (Mw: 80,000; Aldrich, Hamburg, Germany) dissolved in 1 cm3 of Dichloromethane (Merck, Darmstadt, Germany) to prepare 10% w/v solution as our control sample. Lig (Mn: 3000; Aldrich) at 5, 10, and 15 wt.% of PCL was added to the mixture. ELS was carried out in random (Random sample code: T0, T5, T10, and T15) and aligned (Aligned sample code: O0, O5, O10, and O15) orientations using a 1-ml syringe equipped with a 19-ga flat-tip needle connected to the positive high-voltage power supply as shown in Figure 1. The ELS technique setting is introduced in the following section. The flow rate was 1 ml h−1 and the collector speed for aligned samples was 25 rpm. All samples dried for 24 h under vacuum to let the solvent evaporate completely. Data were reported as mean ± SD of five samples.
Figure 1.
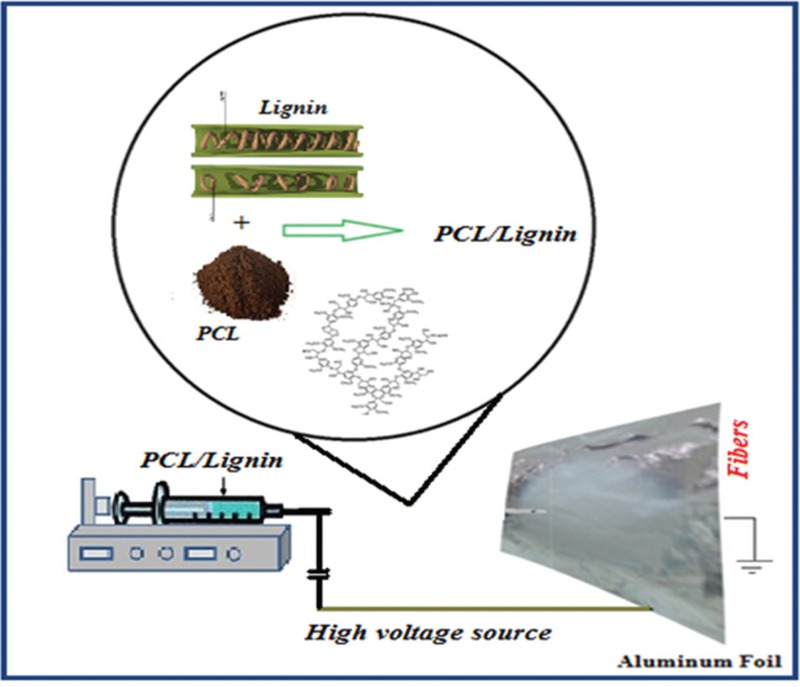
Schematic of electrospinning of PCL/Lig nanocomposite scaffold for tissue engineering
Composite Preparation
Four different polymer nanocomposite scaffold solution were prepared by addition of various ratios 100, 95:5, 90:10, and 85:15 of Lig in the PCL solution in 1 ml of dichloromethane. The solution was allowed to stir for 1 h in a sealed container and the obtained mixture was further sonicated for 20 sec.
Electrospinning Technique
The prepared nanocomposite mixture were separately obtained by dispersion of Lig into the PCL solution and then stirred. ELS technique is one of the most important approaches for developing continuous submicron fibers with diameters ranging from 20 to 50 nm according to the procedure conducted by Heydary et al.[16,17] The prepared solutions were applied into a 1-ml syringe of ELS as represented in schematic Figure 1. The condition for ELS were selected at room temperature (T = 25°C). The setting for ELS (feed rate) adjusted at 1 ml h−1. Then, the nanocomposite scaffolds produced on aluminum foil attached on the rotating drum. The collector distance from tip of needle set at 15 cm. The adequate high voltage was choosing at 15 with step of 5 kV. The optimum voltage observed to select was 18 kV. Also, the dimension of frame was 150 × 150 mm2 which was positioned at a constant distance of 15 cm from the needle. Finally, the prepared scaffolds dried at 50 to 60°C in the vacuum condition for removing the moisture and the residual solvent of scaffolds.
Biological Testing
Bioactivity and Biodegradation Test
To recognize the biological and biodegradation response of the nanocomposite scaffolds, the specimens are soaked in the phosphate-buffered saline (PBS). The SBF test reliability depends on the bioceramics and polymer type utilized. According to the Kokubo's test, the important requirement for any materials to mimic and bond with the host tissue can be recognized by the formation of apatite (bone-like or cauliflower) on its surface after the implantation to the living body.[16,17,18,19,20,21,22] The apatite formation on the surface of scaffold nanocomposite of polymer/bioglass revealed the ability to precipitate apatite (cauliflower) on the surface of biomaterials.
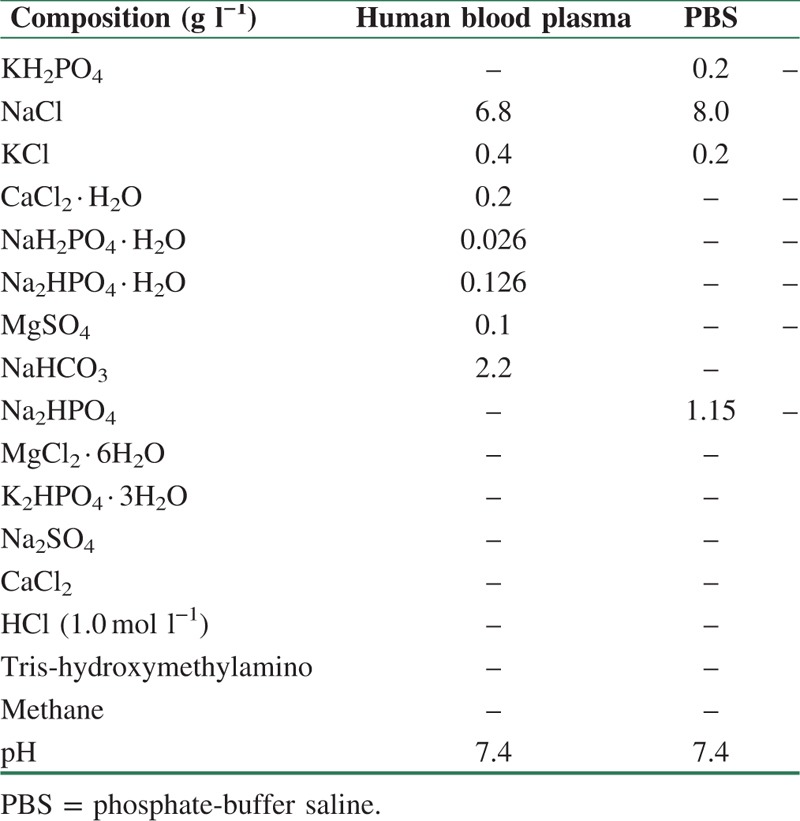
The degree of the weight loss of the nanocomposite samples was calculated by the use of Eq. (1). The test is carried out in the phosphate-buffered saline, PBS, (pH = 7.4) at 37°C for 2,4,6 and 8 weeks. The PBS solution was prepared by dissolving the NaCl (8 g), KCl (0.2 g), Na2HPO4 (1.44 g), and KH2PO4 (0.24 g) in 800 ml deionized water according to Table 1. Table 1 show the difference in elements of PBS, and blood solutions. The PBS ions concentration (pH) changes show the interaction and actions between the scaffolds and solutions. The pH of PBS prepared at room temperature has to be adjusted at 7.4. The nanocomposite scaffolds were inserted in a 20-ml falcon tube with 15-ml PBS solution. The produced samples were then placed in a water bath at 37 ± 1°C for 8 weeks. Weight change was calculated by this formula. 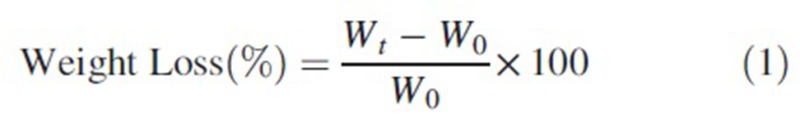
Table 1.
Comparison of nominal ion concentrations of phosphate-buffered saline (PBS), and human blood plasma
W0 is the initial dry sample weight and Wt is the dry sample weight after specific time.
Mechanical Properties
Tensile Testing
The PCL/Lig nanocomposite scaffold with various amounts of Lig nanoparticles were tested for mechanical analysis. The mechanical properties of scaffolds, including tensile strength and elongation at fracture point, were measured by tensile machine. The tensile test performed on the four samples for random and aligned specimens. Mechanical characteristics of samples were performed with the Instron (model. 5566, Buckinghamshire, UK) at room temperature using a 10 N load cell with 10 mm min−1 cross-head speed. All samples were prepared in 5 × 30 mm dimensions.
Porosity and Zeta potential Analysis
Porosity of the scaffolds was measured using “Matlab software” program. The average particle size and Zeta potential for Lig particles (model ZEN 3600; Malvern Instruments Ltd., Malvern, UK) at 25°C were measured.
Wettability Analysis
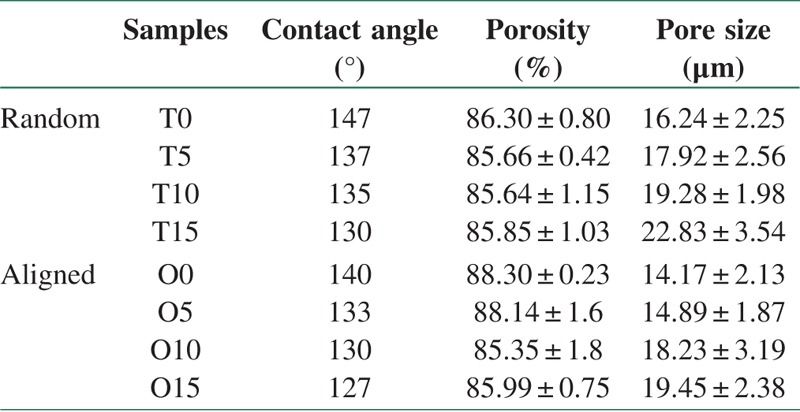
The wettability of scaffolds was assessed using a contact angle measuring machine (CA-X Contact Angle Meter; Kyowa Interface Science Co., Tokyo, Japan) and reported in Table 2.
Table 2.
Contact angle, porosity, pore size of PCL/lignin scaffolds
Cell Culture with MTT assay
Cell test and viability were defined using the 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) measure (Mannheim, Germany). This measurement is regarded to the cleavage of the yellow tetrazolium salt MTT by mitochondrial dehydrogenases in viable cells to prepare purple formazan crystals.[23,26] The cells on the surface of nanocomposite scaffolds were soaked with 0.5 mg ml−1 MTT for 3 h at room temperature (T = 37°C). All the four samples were tested for each incubation period, and each test was performed in triplicate. After 1, 7 and 14 days, samples were washed with PBS (Bio-Idea, Tehran, Iran) to eliminate unattached cells and fixed in 3.5% glutaraldehyde for 30 min at room temperature. After several dehydrations in ethanol (30 min in 40, 50, 60, 80 and 96% ethanol, subsequently), scanning electron microscopy (SEM) (Philips XL30, Amsterdam, The Netherlands) was used to study cells morphology on the surface according to the procedure described in reference.[26] The average value and standard deviation of optical density were calculated and reported. P < 0.05 was considered statistically significant in statistical analyses using analysis of variance (ANOVA).
Characterization of Materials
Morphology of Electrospun Nanofibers Analysis
The morphology and fiber diameter of electrospun fibers were examined using a Hitachi SEM (Cold Field Emission, Hitachi, model S4160, Japan), samples were coated with a thin layer of gold to produce a conductive surface. The surface of the samples was scanned at 26 kV at 100 and 1000 magnification. Diameter of fibers and pore size was assessed on SEM basis using Jimage software. A small section of aluminum foil which coated with electrospun nanofibers was cut and sputter coated with gold to prevent electron charging.
Fourier Transform Infrared Spectroscopy Analysis
The nanocomposite scaffolds were investigated with Fourier Transform Infrared Spectroscopy (FTIR) spectra. All spectrums were taken at room temperature on a spectrometer with spectral range of 4000–1000 cm−1 and 4.0 cm−1 resolutions. Spectra were performed for PCL, and PCL/Lig 5, 10 wt% scaffolds nanofiber. To evaluate the chemical composition changes of nanofibers the FTIR (Perkin-Elmer, London, UK) was used.
Results and Discussion
FTIR Result
Absorption peaks in the area 1036 cm−1 represents C–O stretch of the ester groups of PCL network. The asymmetric stretching vibration absorption peak in 1239 cm−1 is the group of COC. The absorption peak area of the bending 1350 to 1450 cm−1–CH2– group is asymmetric pattern. The 1723 cm−1 peaks appeared in the area of the carbonyl functional group C = O stretching vibration. Also, the presence peak 2960 relating to the C–H group stretch peak in the area is belongs to the CH3 groups. Absorption peaks at 2939 and 2862 cm−1 are related to the symmetric and asymmetric vibration status methylene (CH2) is also visible. On the other hand, the peak in the range of the fluctuations stretching 1293 cm−1 is dedicated to C–O and C–C in the main chain polymer and the crystalline phase is PCL as shown in Figure 2. A peak in the range of 1176 cm−1 visible is related to the amorphous phase structure of PCL. Lack of observed changes in reflect peaks is due to the lack of interaction between the two materials, respectively. So we can assume that dichloromethane solvent and Lig does not have any influence on the chemical nature of PCL. The vibration of PCL and the peak's name are presented in Figure 2.
Figure 2.
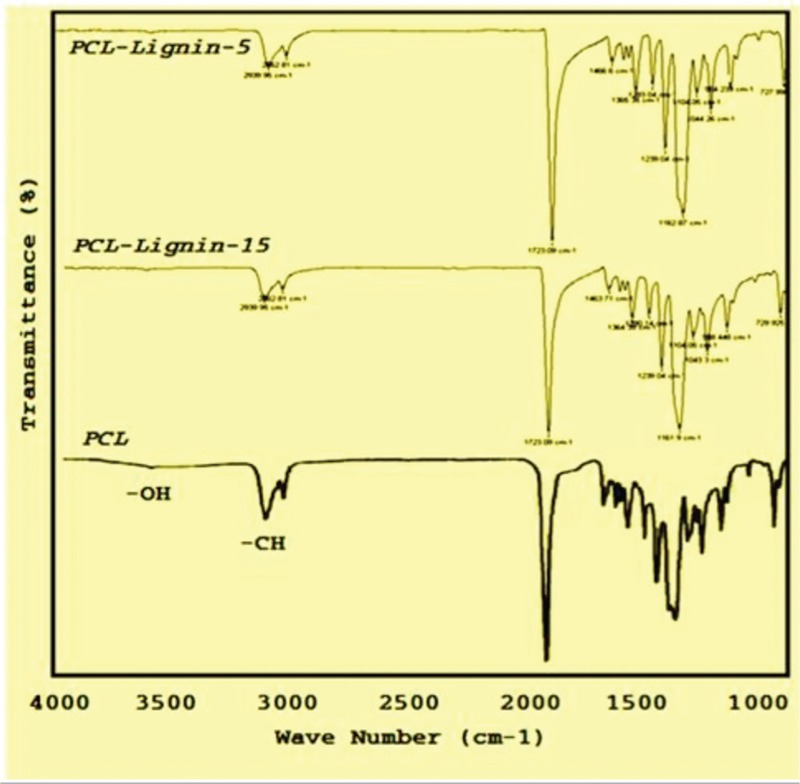
FTIR spectra for PCL and PCL/lignin with 5 and 15% lignin nanocomposite scaffold
Scanning Electron Microscopy Result
SEM results for aligned and random samples are depicted in Figures 3 and 4, respectively. Lig addition caused smoother fiber surface in all samples, whereas Lig particles appeared on the fiber surface like nodes which could have an importance role in cell attachment. The wettability, porosity, and pore size data are mentioned in Table 2.
Figure 3.

SEM images of aligned (a and e) O0, (b and f) O5, (c and g) O10, (d and h) O15 nanocomposite sample
Figure 4.

SEM images of random (a and e) T0, (b and f) T5, (c and g) T10, (d and h) T15 nanocomposite sample
As depicted in Figures 3 and 4, there are interconnected pores in all samples. Lig dispersion is homogenous in 5 and 10 wt.% Lig amount. In samples with 15 wt.% Lig content, due to high concentration and Lig agglomeration, greater viscosity leads to thicker diameter in both methods (Random and Aligned). Electrospun nanofibers with reinforced ultrafine nanostructures inside, could be ideal as substrate since it is capable of simulating the extracellular matrix. Decreasing the Lig nanoparticle concentration could prevent the agglomeration. Also, increasing the viscosity of polymer solution causes greater fiber diameter while the conductivity of solution increased the fiber diameter decreased.[5] This phenomenon is hindered in aligned samples nanocomposite scaffolds. Thinner fiber diameter in aligned samples contributed to the collector speed which stretches and forms more oriented fibers. The fiber diameter decreased from 5 to 10 wt.% Lig content due to lower molecular weight (Mw = 3000) of Lig compared with PCL (Mw = 80,000) which could decrease the total mixture viscosity, whereas in the higher loads of Lig, increased viscosity in electrospun solution caused thicker fiber diameter. Anionic nature of Lig neutralizes positive charge of power supply around the needle[11] to some extent, whereas the applied voltage was constant; therefore, the diameter has increased in 15 wt.% Lig content. Porosity in all samples was higher than 85% and pore size was more than 12 to 14 μm, which could guarantee appropriate cells infiltration.
The results of the statistical analysis of variance (ANOVA) showed that the average diameter of the fibers which had Lig improved. As can be seen, as the Lig percentage increases, the nanofibers diameter starts to decrease resulting in the increase in the mechanical and chemical stability of the samples.
Particle Size Analysis
Fiber diameter effects on the pore size and porosity which have important role in cell attachment, proliferation, and differentiation. Minimum pore size of 10 μm is needed for cell infiltration.[4] Increasing Lig content reduced contact angle (about 10%) and hydrophobic nature of the nanocomposite. The average particle size of Lig particles was 89.4 nm, and Zeta potential for Lig in deionized water was −32.6 mV which indicates suitable dispersion of particles in the solution. The particle size analysis (PSA) of Lig at 25°C has been done three times. The analysis of DLS for particles size is represented in Figure 5. It is seen that particles are in the nanosize range between 70 and 90 nm.
Figure 5.
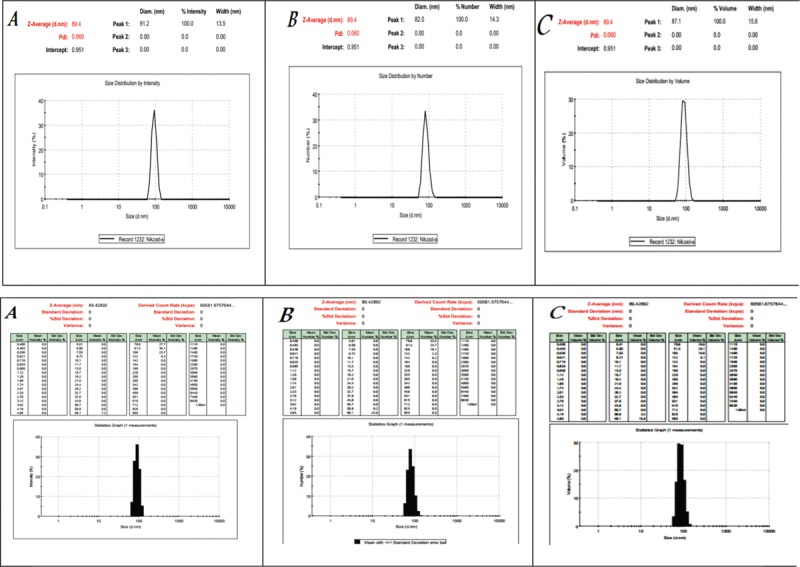
Particle size and zeta potential analysis of lignin powder as first row is size distribution and second row is zeta potential
The PSA of Lig powders at 25°C was repeated three times. In all the repetitions, the answers were similar and proved that Lig nanoparticles size were less than 89.4 nm.
Mechanical Analysis
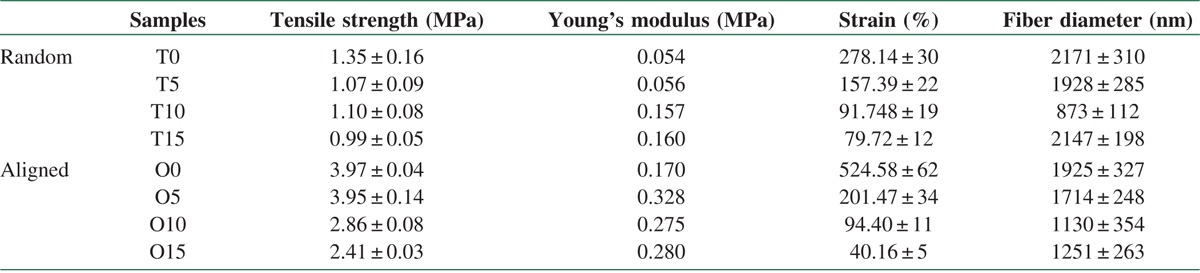
The result of mechanical test (Tensile strength test) of nanocomposite samples listed in the following Table 3. The tensile strength was (0.99–1.35 MPa) and Young's modulus was (0.054–0.160 MPa) in the Random samples. Although in the Aligned samples, the tensile strength (2.41–3.97 MPa) and Young's modulus (0.170–0.328 MPa) are in higher amount compare to the random samples. Pure PCL obtained maximum tensile strength and strain in both methods. Adding Lig leaded significant decrease in tensile strength for aligned samples (P ≤ 0.00035), whereas there was no significant change in tensile strength of random samples (P ≥ 0.05) due to their isotropic nature. Lig acts as stress concentration spots in PCL matrix and decreased homogeneity. Due to more chain orientation in aligned samples tensile strength is higher in comparison with random specimens. Lig is a rigid biopolymer and incorporation of Lig particles in PCL matrix results an increase in young's modulus in almost all samples in comparison with pure PCL. The O10 and O15 samples show less young's modulus in comparison with O5 samples due to retarded crystallinity during ELS process.[12,13,14] Elasticity shows a downward trend with increasing Lig content in all specimens due to rigidity of lower molecular Lig particles, cohesion loss, and nonhomogeneous matrix. Weak miscibility of Lig and PCL affects their blend properties.[13] Interfacial adhesion between Lig and PCL plays a key role in mechanical properties.[9] Pucciariello et al. in 2008 blended PCL with straw Lig through high-energy ball milling method, increasing Lig content up to 30% increased blend young's modulus while decreased tensile stress and elongation at break.[14] In another research, Lig in more compatible matrix (polyamide and polyesters) shows reinforcing effects and increase tensile strength and young's modulus.[15] Polymeric matrix are capable of being reinforced with nanoparticles. Heydary et al.[16,17] indicate that adding nanoclay (NC) to the Polyvinyl alcohol (PVA)/Gum can enhance the mechanical properties of PVA/Gum/NC blends, they have found that their nanocomposite samples shape clear homogeneous blends in comparison with those in PVA/Gum.[16]
Table 3.
Mechanical (tensile, young modulus, strain, and fiber diameter) properties of PCL/lignin nanocomposite scaffolds
Also, Heydary et al.[16,17] showed that the samples with 3% of NC powder have excellent bioactivity, biodegradation, and mechanical property compared to the samples with lower amount of NC powder. Furthermore, the blend nanofibers showed advancement in mechanical properties compared to the pure electrospun PVA. This is probably because of the more Young's modulus as well as tensile stress in the later samples. Antimicrobial activity of scaffolds is important in biomedical applications. In 2013, Rafienia et al.[23] mixed poly(ethylene glycol) (PEG), gentamicin sulphate (GS), and subsequently poly (vinylalcohol) for drug release applications (PEG/PVA/GS). The result indicates that hydrophilic properties of PVA play a key role in antimicrobial activity of the samples since hydrophilicity often reduces protein adsorption. Also, the data illustrate that cell adhesion is depend on the hydrophilicity of samples.[23,26] Therefore, PVA shows a potential ability as a barrier against bacterial adhesion in the PEG/PVA/GS.[23,24,25,26] Researchers investigated the PVA/alginate/hydroxyapatite with various content of alginate.[20,21,22,23,24,25] As it is seen in Figure 6a and b that the pure scaffolds PCL showed a higher tensile strength compared to the other samples. Adding nanoparticles leads to reduction in the tensile strength of directional fibers. The reduction in tensile strength seems to be points of stress concentration because of to the addition of Lig nanoparticles in polymer with nonelastic components. As the Lig nanoparticles are almost rigid bodies in composites. High tensile strength of pure scaffolds showed that thermoplastic polymers have ductile properties. The presence of large amounts of Lig is not a matter of tough polymer alloy polymer system and a reduction of tensile strength is causing of disturbance of Lig in the polymer matrix.
Figure 6.
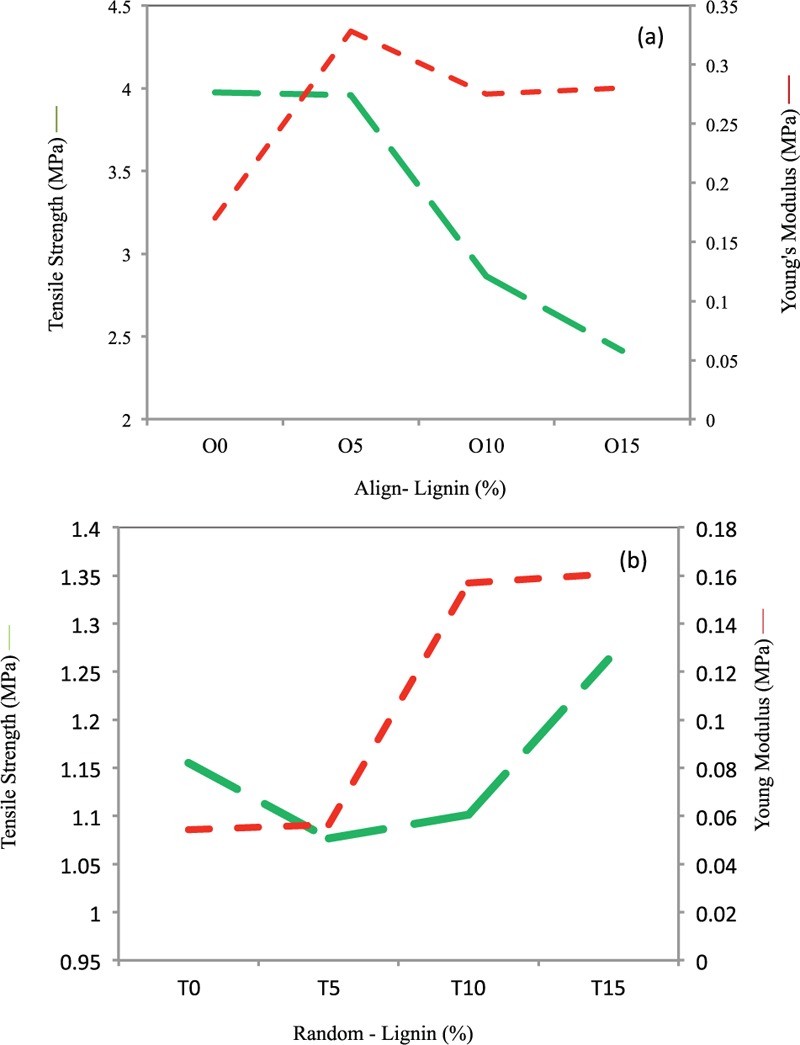
Mechanical properties (Tensile strength and Elastic Modulus) of PCL/Lignin nanocomposite scaffolds of (a) aligned and (b) random nanocomposite samples, SD = ±0.05
MTT Analysis
The PCL crystal size changed after mixing with Lig in the ELS procedure. The presence of Lig leads to a reduction in the crystallinity of PCL during the ELS process. Cell compatibility was also analyzed via MTT examine and SEM imaging as mentioned in other works.[26] The effect of crystallinity on materials properties is very important in the tissue engineering. The cell desire, adhesion, and proliferation are more in amorphous polymers. Though Lig has antioxidant characteristics, but in higher levels cause toxicity in the cell so it is important to find an optimum portion of Lig. Laboratory results from the MTT assay revealed that the biocompatibility of the ELS nanocomposite scaffold developed at rising percentages of Lig [Figures 7 and 8]. The investigation of in vitro and cells proliferation of fibroblast cells on electrospun scaffolds obtained by SEM analysis. Also, the images indicate that increasing Lig amounts influenced the strength properties of electrospun layers and improved the biocompatibility of PCL/Lig electrospun fibrous scaffolds. On the other hand, nanocomposite polymeric scaffold improved the biological response of the cells with the mechanical signals. This is the reason that associated to the coupled effects of improved matrix fabrication to the incorporated fibers along with more efficient force transduction through the fibers and ECM. Cells showed flattened morphology in intimate contact with coating after 14 days of culture as shown in Figures 7 and 8. Coated samples demonstrated appropriate bioactivity and biocompatibility. Moreover, in some particular conditions, cultured fibroblasts on the piezoelectric scaffolds have given that fancy to move, adhere, and secrete ECM; means can be bioactivity of samples leads to lower cell number and higher protein deposition, which requires a range of protein investigations for approval.[26]
Figure 7.

SEM images of sample after MTT test of sample containing 0, 10, and 15 wt.% Lig in the PCL scaffolds
Figure 8.
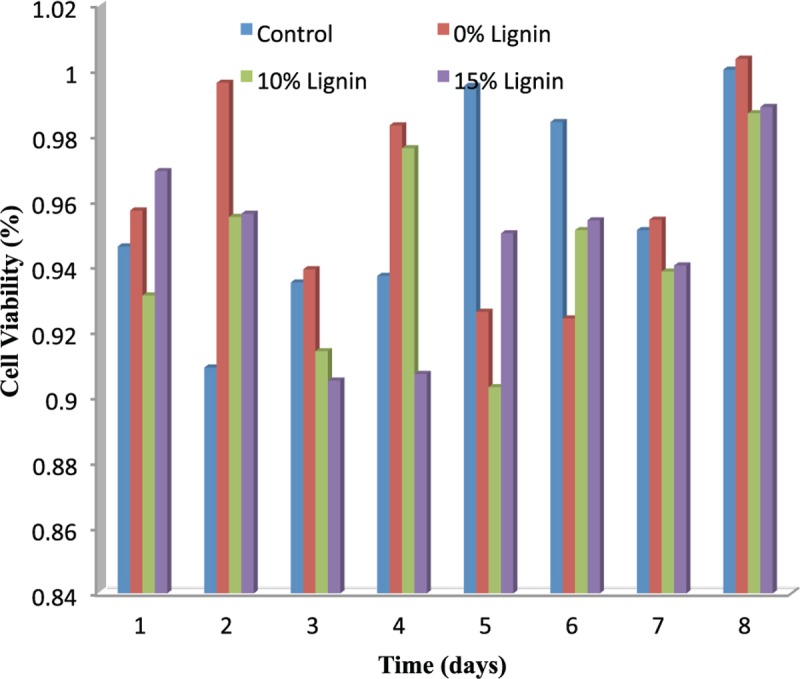
Cell viability and MTT test result of sample scaffolds nanaocomposites
MTT assay for cytotoxicity was performed on cell line and compared with the control group. Plot was prepared using data of metabolite isolate time v/s cell viability (absorbance of cells) [Figure 8]. The reason for the slight Lig cytotoxicity could be due to its known absorption capability, that is, it could slightly nonspecifically absorb some of the micronutrient constituents needed for sustaining tested peripheral blood mononuclear cells proliferation, but also could be due to Lig antioxidant activity, as shown in tests on human keratinocytes and mouse fibroblasts.[30,31,32,33,34] On the other hand, one can speculate that Lig structures became more condensed upon sintering, which could have led to formation of highly condensed aromatic structures, known for their toxicity. The result of MTT test showed that sample containing 15% Lig have better monotony and uniformity to the cells. As it is seen the sample containing 15% Lig has the highest cell viability compared to the sample with lower amount of Lig. As it is seen, the fibers made of two-component fibers are attached to each other with interconnected morphologically and uniform lattice. Also, one can say that nanoparticles are accumulating separately. Figure 9 showed the optical microscope image of the aligned sample 15% Lig that visible brown particles are represented.
Figure 9.

Optical microscope image of sample containing 15% lignin in scaffold
To overcome the above-mentioned drawbacks, the fabrication of an ideal synthetic scaffold has been devoted to soft tissue engineering, in which a three-dimensional porous scaffold made of ceramic like calcium phosphate and silicate bioceramic like (akermanite, diopside, baghdadite, and bredigite),[27,28,29,30] synthetic polymers [i.e., PVA, PCL, and Poly (lactic-co-glycolic) acid (PLGA)],[16,17] natural polymers (i.e., chitosan, gelatin, and cellulose),[31,32,33] or their composites[28,29,30,31,32,33] is used to mimic the natural bone behavior to some extent.
Degradation Analysis
Due to the fact that PCL degradation last more than 2 years, in our project samples did not show significant weight lost after 90 days. (Maximum weight lost was for samples with 15% Lig content and they lost 10% of their initial weight after 90 days). This result revealed that Lig addition to the PCL matrix could reduce degradation time.
Wettability Analysis
The wettability (contact angle result) of PCL and PCL/Lig nanocomposite scaffolds with milli-Q water were performed and represented in Table 2 and Figure 10. Pure PCL is hydrophobic in nature. Results of contact angle measured shows that Lig increased the hydrophilicity of the scaffolds to a certain extent (i.e., decrease in contact angle from 147 to 130), with increase in Lig content [Table 2]. Therefore, as it is shown in Figure 10 surface wettability of the scaffold is enhanced by Lig inclusion, which in turn can aid in cell attachment.
Figure 10.
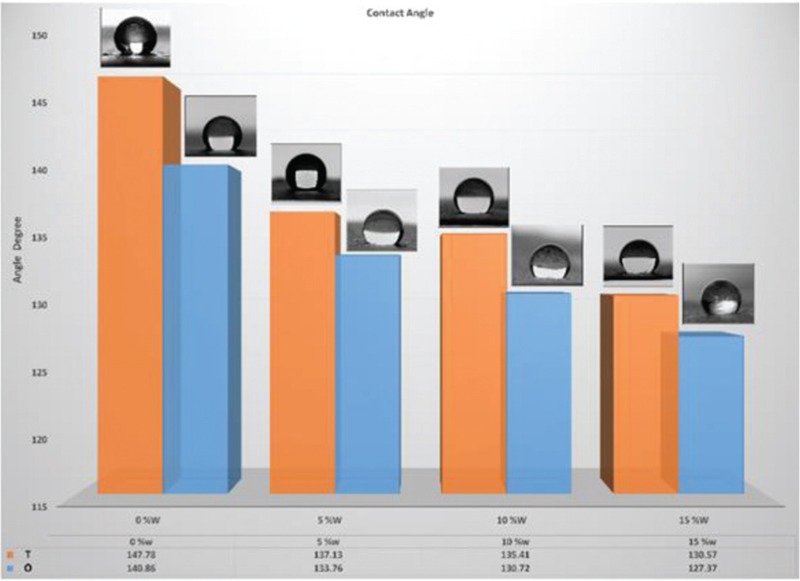
Wettability result (contact angle) of sample containing 0, 5, 10, and 15% lignin in PCL scaffold
Conclusion
In the current study, the novel polymer PCL and Lig fiber has been fabricated with improved biomedical properties. The PCL/Lig nanocomposite scaffolds containing various amount of weight fractions (0, 5, 10, and 15 wt.%) Lig, showed enhanced pore structures, which consisted of multilayered microsized struts. In addition the Young's modulus of PCL/Lig nanocomposite scaffolds was increased significantly compared with the pure PCL scaffold. Lig in the PCL nanocomposite scaffolds enhanced hydrophilicity and porosity properties. PCL/Lig ratio has effect on fiber diameter, contact angle, pore size, and mechanical properties of the scaffolds. Nanocomposite scaffolds containing 10% Lig (O10) due to its optimum mechanical, physical, and morphological properties has favorable conditions for the cell test. The general results suggest that PCL/Lig nanocomposite scaffolds may have potential for use in scaffolds for tissue regeneration.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Fernandes EM, Pires RA, Mano JF, Reis RL. Bionanocomposites from lignocellulosic resources: properties, applications and future trends for their use in the biomedical field. Prog Polym Sci. 2013;38:1415–41. [Google Scholar]
- 2.Yao ZC, Gao Y, Chang MW, Ahmad Z, Li JS. Regulating poly-caprolactone fiber characteristics in-situ during one-step coaxial electrospinning via enveloping liquids. Mater Lett. 2016;183:202–6. [Google Scholar]
- 3.Ramakrishna S. An Introduction to Electrospinning and Nanofibers. London: World Scientific; 2005. [Google Scholar]
- 4.Pham QP, Sharma U, Mikos AG. Electrospun poly(ε-caprolactone) microfiber and multilayer nanofiber/microfiber scaffolds: characterization of scaffolds and measurement of cellular infiltration. Biomacromolecules. 2006;7:2796–805. doi: 10.1021/bm060680j. [DOI] [PubMed] [Google Scholar]
- 5.Sultana N, Hassan MI, Lim MM. Composite Synthetic Scaffolds for Tissue Engineering and Regenerative Medicine. Cham: Springer; 2014. [Google Scholar]
- 6.Gupta D, Venugopal J, Prabhakaran MP, Dev VG, Low S, Choon AT, et al. Aligned and random nanofibrous substrate for the in vitro culture of Schwann cells for neural tissue engineering. Acta Biomater. 2009;5:2560–9. doi: 10.1016/j.actbio.2009.01.039. [DOI] [PubMed] [Google Scholar]
- 7.Kim YB, Kim GH. PCL/alginate composite scaffolds for hard tissue engineering: fabrication, characterization, and cellular activities. ACS Comb Sci. 2015;17:87–99. doi: 10.1021/co500033h. [DOI] [PubMed] [Google Scholar]
- 8.Pant HR, Baek WI, Nam KT, Seo YA, Oh HJ, Kim HY. Fabrication of polymeric microfibers containing rice-like oligomeric hydrogel nanoparticles on their surface: a novel strategy in the electrospinning process. Mater Lett. 2011;65:1441–4. [Google Scholar]
- 9.Wang K, Loo LS, Goh KL. A facile method for processing lignin reinforced chitosan biopolymer microfibres: optimising the fibre mechanical properties through lignin type and concentration. Mater Res Expr. 2016;3:035301. [Google Scholar]
- 10.Nitz H, Semke H, Mülhaupt R. Influence of lignin type on the mechanical properties of lignin based compounds. Macromol Mater Eng. 2001;286:737–43. [Google Scholar]
- 11.Schreiber M, Vivekanandhan S, Mohanty AK, Misra M. A study on the electrospinning behaviour and nanofibre morphology of anionically charged lignin. Adv Mat Lett. 2012;3:476. [Google Scholar]
- 12.Wool R, Sun XS. Bio-based Polymers and Composites. London: Academic Press; 2011. [Google Scholar]
- 13.Pucciariello R, D’Auria M, Villani V, Giammarino G, Gorrasi G, Shulga G. Lignin/poly(ε-caprolactone) blends with tuneable mechanical properties prepared by high energy ball-milling. J Polym Environ. 2010;18:326–34. [Google Scholar]
- 14.Pucciariello R, Bonini C, D’Auria M, Villani V, Giammarino G, Gorrasi G. Polymer blends of steam-explosion lignin and poly(ε-caprolactone) by high-energy ball milling. J Appl Polym Sci. 2008;109:309–13. [Google Scholar]
- 15.Pouteau C, Baumberger S, Cathala B, Dole P. Lignin-polymer blends: evaluation of compatibility by image analysis. C R Biol. 2004;327:935–43. doi: 10.1016/j.crvi.2004.08.008. [DOI] [PubMed] [Google Scholar]
- 16.Heydary HA, Karamian E, Poorazizi E, Heydaripour J, Khandan A. Electrospun of polymer/bioceramic nanocomposite as a new soft tissue for biomedical applications. J Asian Ceram Soc. 2015;3:417–25. [Google Scholar]
- 17.Heydary HA, Karamian E, Poorazizi E, Khandan A, Heydaripour J. A novel nano-fiber of Iranian gum tragacanth-polyvinyl alcohol/nanoclay composite for wound healing applications. Procedia Mater Sci. 2015;11:176–82. [Google Scholar]
- 18.Karamian E, Khandan A, Eslami M, Gheisari H, Rafiaei N. Vol. 829. Trans Tech Publications; 2014. Investigation of HA nanocrystallite size crystallographic characterizations in NHA, BHA and HA pure powders and their influence on biodegradation of HA. Advanced Materials Research; pp. 314–8. [Google Scholar]
- 19.Khandan A, Abdellahi M, Barenji RV, Ozada N, Karamian E. Introducing natural hydroxyapatite-diopside (NHA-Di) nano-bioceramic coating. Ceram Int. 2015;41:12355–63. [Google Scholar]
- 20.Khandan A, Ozada N, Karamian E. Novel microstructure mechanical activated nano composites for tissue engineering applications. J Bioeng Biomed Sci. 2015;5:1. [Google Scholar]
- 21.Karamian EB, Motamedi MR, Mirmohammadi K, Soltani PA, Khandan AM. Correlation between crystallographic parameters and biodegradation rate of natural hydroxyapatite in physiological solutions. Indian J Sci Res. 2014;4:092–9. [Google Scholar]
- 22.Karamian E, Abdellahi M, Khandan A, Abdellah S. Introducing the fluorine doped natural hydroxyapatite-titania nanobiocomposite ceramic. J Alloys Compounds. 2016;679:375–83. [Google Scholar]
- 23.Rafienia M, Zarinmehr B, Poursamar SA, Bonakdar S, Ghavami M, Janmaleki M. Coated urinary catheter by PEG/PVA/gentamicin with drug delivery capability against hospital infection. Iranian Polym J. 2013;22:75–83. [Google Scholar]
- 24.Kokubo T, Takadama H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials. 2006;27:2907–15. doi: 10.1016/j.biomaterials.2006.01.017. [DOI] [PubMed] [Google Scholar]
- 25.Kumar A, Negi YS, Choudhary V, Bhardwaj NK. Microstructural and mechanical properties of porous biocomposite scaffolds based on polyvinyl alcohol, nano-hydroxyapatite and cellulose nanocrystals. Cellulose. 2014;21:3409–26. [Google Scholar]
- 26.Rahmati S, Basiriani MB, Rafienia M, Yaghini J, Raeisi K. Synthesis and in vitro evaluation of electrodeposited Barium titanate coating on Ti6Al4V. J Med Signals Sens. 2016;6:106. [PMC free article] [PubMed] [Google Scholar]
- 27.Sharafabadi AK, Abdellahi M, Kazemi A, Khandan A, Ozada N. A novel and economical route for synthesizing akermanite (Ca 2 MgSi 2 O 7) nano-bioceramic. Mater Sci Eng C Mater Biol Appl. 2017;71:1072–8. doi: 10.1016/j.msec.2016.11.021. [DOI] [PubMed] [Google Scholar]
- 28.Kazemi A, Abdellahi M, Khajeh-Sharafabadi A, Khandan A, Ozada N. Study of in vitro bioactivity and mechanical properties of diopside nano-bioceramic synthesized by a facile method using eggshell as raw material. Mater Sci Eng C Mater Biol Appl. 2017;71:604–10. doi: 10.1016/j.msec.2016.10.044. [DOI] [PubMed] [Google Scholar]
- 29.Najafinezhad A, Abdellahi M, Ghayour H, Soheily A, Chami A, Khandan A. A comparative study on the synthesis mechanism, bioactivity and mechanical properties of three silicate bioceramics. Mater Sci Eng C Mater Biol Appl. 2017;72:259–67. doi: 10.1016/j.msec.2016.11.084. [DOI] [PubMed] [Google Scholar]
- 30.Khandan A, Abdellahi M, Ozada N, Ghayour H. Study of the bioactivity, wettability and hardness behaviour of the bovine hydroxyapatite-diopside bio-nanocomposite coating. J Taiwan Inst Chem Eng. 2016;60:538–46. [Google Scholar]
- 31.Karkeh-abadi F, Saber-Samandari S, Saber-Samandari S. The impact of functionalized CNT in the network of sodium alginate-based nanocomposite beads on the removal of Co(II) ions from aqueous solutions. J Hazard Mater. 2016;312:224–33. doi: 10.1016/j.jhazmat.2016.03.074. [DOI] [PubMed] [Google Scholar]
- 32.Saber-Samandari S, Saber-Samandari S, Kiyazar S, Aghazadeh J, Sadeghi A. In vitro evaluation for apatite-forming ability of cellulose-based nanocomposite scaffolds for bone tissue engineering. Int J Biol Macromol. 2016;86:434–42. doi: 10.1016/j.ijbiomac.2016.01.102. [DOI] [PubMed] [Google Scholar]
- 33.Saber-Samandari S, Saber-Samandari S. Biocompatible nanocomposite scaffolds based on copolymer-grafted chitosan for bone tissue engineering with drug delivery capability. Mater Sci Eng C Mater Biol Appl. 2017;75:721–32. doi: 10.1016/j.msec.2017.02.112. [DOI] [PubMed] [Google Scholar]
- 34.Erakovic S, Jankovic A, Tsui GC, Tang CY, Miskovic-Stankovic V, Stevanovic T. Novel bioactive antimicrobial lignin containing coatings on titanium obtained by electrophoretic deposition. Int J Mol Sci. 2014;15:12294–322. doi: 10.3390/ijms150712294. [DOI] [PMC free article] [PubMed] [Google Scholar]


