Abstract
A comprehensive understanding of the transport and fate of per- and poly-fluoroalkyl substances (PFAS) in the subsurface is critical for accurate risk assessments and design of effective remedial actions. A multi-process retention model is proposed to account for potential additional retardation processes for PFAS transport in source zones. These include partitioning to the soil atmosphere, adsorption at air-water interfaces, partitioning to trapped organic liquids (NAPL), and adsorption at NAPL-water interfaces. An initial assessment of the relative magnitudes and significance of these retention processes was conducted for two PFAS of primary concern, perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), and an example precursor (fluorotelomer alcohol, FTOH). The illustrative evaluation was conducted using measured porous-medium properties representative of a sandy vadose-zone soil. Data collected from the literature were used to determine measured or estimated values for the relevant distribution coefficients, which were in turn used to calculate retardation factors for the model system. The results showed that adsorption at the air-water interface was a primary source of retention for both PFOA and PFOS, contributing approximately 50%, of total retention for the conditions employed. Adsorption to NAPL-water interfaces and partitioning to bulk NAPL were also shown to be significant sources of retention. NAPL partitioning was the predominant source of retention for FTOH, contributing ~98%, of total retardation. These results indicate that these additional processes may be, in some cases, significant sources of retention for subsurface transport of PFAS. The specific magnitudes and significance of the individual retention processes will depend upon the properties and conditions of the specific system of interest (e.g., PFAS constituent and concentration, porous medium, aqueous chemistry, fluid saturations, co-contaminants). In cases wherein these additional retention processes are significant, retardation of PFAS in source areas would likely be greater than what is typically estimated based on the standard assumption of solid-phase adsorption as the sole retention mechanism. This has significant ramifications for accurate determination of the migration potential and magnitude of mass flux to groundwater, as well as for calculations of contaminant mass residing in source zones. Both of which have critical implications for human-health risk assessments.
Keywords: Perfluoroalkyl, PFOS, PFOA, retardation, partitioning, air-water interface, NAPL-water interface
Graphical Abstract

1. Introduction
The use of per- and poly-fluoroalkyl substances (PFAS) in numerous industrial, commercial, and military applications has resulted in their widespread distribution in the environment (cf., EPA 2009a, Rayne and Forest, 2009, Ahrens, 2011, Krafft and Riess, 2015a, Cousins et al., 2016). For example, it has been demonstrated that PFAS contamination of soil and groundwater exists at many Department of Defense fire-fighting training sites and other areas (Moody and Field, 2000, Moody et al., 2003, Ahrens, 2011, Backe et al., 2013, Anderson et al., 2016). While many of the PFAS-contaminated sites are associated with military facilities, groundwater contamination is also observed for non-military sites such as manufacturing plants and airports (EPA, 2009a, Rayne and Forest, 2009, Cousins et al., 2016). Perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) are two primary PFAS of concern. A recent tabulation of their occurrences in groundwater shows measured concentrations ranging up to >2000 and 300 µg/L for PFOS and PFOA, respectively (Cousins et al., 2016).
There is increased scrutiny and concern regarding the bioaccumulation and toxicity potential of PFAS, particularly for PFOS and PFOA (EPA, 2009, 2014). In 2009, the US EPA established provisional health advisory values of 0.4 µg/L for PFOA and 0.2 µg/L for PFOS (EPA, 2009). In 2016, a lifetime health advisory of 0.07 µg/L was issued for the combined total of PFOA and PFOA for long-term exposure through drinking water (EPA, 2016). The observed concentrations of PFOS/PFOA in groundwater noted above are orders of magnitude greater than US EPA’s drinking water health advisory value. Hence, the occurrence of PFAS in groundwater and the resultant potential ramifications for human exposure are of significant interest. For example, a recent survey was conducted to determine the occurrence of select PFAS in drinking water for all 4064 public water supplies that serve >10000 individuals in the US. The results showed that drinking water supplies for 6 million U.S. residents exceed US EPA’s lifetime health advisory for PFOS/PFOA (Hu et al., 2016). The preceding information indicates that there is potential significant risk posed to water resources and human health by PFAS-contaminated sites.
The risk posed by PFAS soil and groundwater contamination, as well as the effective remediation of such contamination, is mediated by the transport and fate behavior of PFAS in the subsurface. PFOA, PFOS, and related PFAS are typically highly recalcitrant owing to their strong C–F bonds. This, in addition to high aqueous solubilities and relatively low retardation for select compounds, can result in the generation of large, persistent groundwater contaminant plumes. Another complicating factor is that PFAS contamination generally consists of numerous compounds of varied physicochemical properties, some of which can transform to PFOA/PFOS (often noted as precursor compounds). Remediation of this complex system is highly challenging, and most available technologies have been shown to be ineffective, as noted in a recent review (Merino et al., 2016).
The high potential health risks associated with PFAS-contaminated sites, and the difficulty and costliness of their remediation, makes it imperative that risk assessments are as accurate as possible to support effective decision-making. Accurate risk assessments require a comprehensive and definitive understanding of the subsurface transport and fate behavior of PFAS, including accurate determination of migration potential from sources to groundwater. Rates of migration are governed in part by the retention and attenuation capacities of the system, which are a function of site conditions and inherent physicochemical properties of the compounds. Primary retention-attenuation processes in general include diffusive flux into lower-permeability units, phase partitioning (e.g., sorption), and transformation reactions.
PFOA, PFOS, and select other PFAS of concern are highly recalcitrant, as noted above. Therefore, transformation-related attenuation processes are likely to be of minimal impact under many natural conditions. However, transformation processes have been shown to be important for PFAS precursors (e.g., Houtz et al., 2013, Anderson et al., 2016). The impact of diffusive mass transfer on solute transport and plume attenuation/persistence in heterogeneous systems is well established. Sorption of PFAS by the solid phase of geologic media is one phase-partitioning retention process that may have significant impact on PFAS transport and attenuation. The sorption behavior of PFAS in geomedia has been investigated over the past decade, and is being considered in conceptual models of transport and fate, as will be further discussed in a following section.
The processes discussed in the preceding paragraph are the standard factors typically considered in conceptual and mathematical models of subsurface contaminant transport and fate. Indeed, these factors are predominant for solute transport within groundwater contaminant plumes. However, there are additional retention/attenuation processes that may influence the transport and fate of PFAS in source zones. These include (i) adsorption to air-water interfaces in the vadose zone, (ii) partitioning to the soil atmosphere in the vadose zone, (iii) partitioning (absorption) to nonaqueous-phase liquids (NAPL) trapped in source zones (both vadose and saturated zones), and (iv) adsorption to NAPL-water interfaces in NAPL-contaminated source zones. Recent in-depth assessments of PFAS occurrence and fate at field sites have demonstrated that vadose-zone sources are a primary subsurface reservoir of PFAS, serving as long-term (decades) contaminant sources to groundwater (Shin et al., 2011, Xiao et al., 2015, Weber et al.,2017). In addition, it is likely that PFAS co-occur with NAPLs at certain types of sites, as has been observed for fire training areas (e.g., Moody et al., 2003, McGuire et al., 2014). Hence, it is critical to examine the potential contribution of these additional retention processes to PFAS transport and fate. This is relevant for example for accurate characterization and modeling of PFAS migration potential from sources to groundwater in support of robust risk assessments, and selection and design of cost-effective remediation systems.
These additional processes have to date received minimal to no investigation for PFAS transport in the subsurface. The objective of this work is to provide an initial assessment of the potential impact of air-water interfacial adsorption, air-water partitioning, NAPL-water interfacial adsorption, and NAPL-water partitioning on PFAS retention and transport. Physicochemical properties of the two primary PFAS of concern, PFOA and PFOS, are reviewed to assess their distribution potential. A comprehensive conceptual model is presented for characterizing retention of PFAS in geomedia systems. Data reported in the literature are used to determine phase-distribution coefficients, which are then employed to calculate retardation factors. These results are used to characterize potential magnitudes of retention for the respective processes and to evaluate their relative significance to overall retardation for a model representative system.
2. Evaluation of Retention Processes
2a. Solid-phase Sorption
Research investigating PFAS adsorption by solids has become a focus over the past decade, with investigations examining water treatment applications (as reviewed in Du et al., 2014 and Merino et al., 2016) and subsurface porous media--- soils and sediments (Liu and Lee, 2005, 2007, Higgins and Luthy, 2006, 2007, Johnson et al., 2007, Carmosini and Lee, 2008, Chen et al., 2009, Pan et al., 2009, Ferrey et al., 2012, Guelfo and Higgins, 2013, Zhao et al., 2014, Milinovic et al., 2015, Zhang et al., 2015, Hellsing et al., 2016). Adsorption of PFAS by solids is complex due to the nature of the molecular structure of PFAS. For example, the sorption of PFAS such as the telomers can be relatively large, due primarily to organic-carbon partitioning associated with hydrophobic interactions (e.g., Liu and Lee, 2005, Goss et al., 2006). Conversely, PFOS and PFOA in their anionic forms have relatively high aqueous solubilities and generally have lower sorption potential. However, the sorption of ionizable PFAS can be influenced by electrostatic interactions, based on the properties of the functional group(s) of the PFAS (Higgins and Luthy, 2007, Johnson et al., 2007, Ferrey et al., 2012, Du et al., 2014, Zhao et al., 2014, Hellsing et al., 2016, Merino et al., 2016). In such cases, zwitterionic and especially cationic species would likely have increased retention.
Sorption of constituents by subsurface porous media is typically more complex than adsorption by water-treatment solids because the media used for water treatment are generally comprised of homogeneous materials designed to behave ideally. Conversely, subsurface media are typically physically and geochemically heterogeneous. As would be anticipated, research has shown that PFAS sorption by subsurface media is greatly affected by the geochemical properties of the solid, particularly with respect to specific components present. Critical factors investigated for PFAS include the magnitude and nature of organic carbon, magnitude and type of metal oxides, and clay mineralogy (Higgins and Luthy, 2006, Liu and Lee, 2007, Ferrey et al., 2012, Milinovic et al., 2015, Zhang et al., 2015). In addition, it is well known that the adsorption of surfactants is sensitive to water chemistry. This has been demonstrated for PFAS, wherein research has shown that factors such as ionic strength, salt composition, pH, and presence of co-contaminants affect solid-phase adsorption (Higgins and Luthy, 2006, Chen et al., 2009, Pan et al., 2009, Guelfo and Higgins, 2013, Zhao et al., 2014).
The discussion above clearly shows that the sorption of PFAS is sensitive to physicochemical properties of the compound, physical and geochemical properties of the porous media, and water chemistry properties. Thus, an accurate evaluation of PFAS transport and retention requires thorough investigation of the impact of these factors. Research conducted over the past decade has significantly advanced our understanding of the sorption behavior of PFAS in subsurface systems. While more remains to be done, research into the impact of solid-phase sorption for PFAS is quite advanced compared to the status for the retention processes discussed in the following subsections. Thus, the following processes are the focus of the present work.
2b. Air-water Partitioning
Partitioning to the soil atmosphere can serve as a source of retention during aqueous-phase transport of volatile organic compounds in vadose-zone systems. For example, Kim et al. (1998) demonstrated with a series of column experiments that such partitioning contributed approximately 15–66%, of total retardation for transport of several aromatic compounds. The functioning of this retention process can differ in some circumstances from the standard retardation behavior. First, the soil atmosphere may be subject to advective flux due to for example soil-venting operations, landfill-gas generation, and barometric-pressure changes. In such cases, vapor-phase advective transport of PFAS would need to be explicitly considered. In addition, gas-phase diffusion coefficients are several orders of magnitude greater than aqueous values. Hence, gas-phase diffusive flux may be a significant transport mechanism depending on extant conditions. For situations wherein gas-phase advective flux is absent and gas-phase diffusive flux is insignificant compared to aqueous advective flux, partitioning to the soil atmosphere can be treated as a standard retention process.
Air-water partition coefficients (dimensionless Henry’s constant, H) vary greatly among the different PFAS. Measured and calculated H values for PFOA and PFOS are in the 10−3 range (Ding and Peijnenburg, 2013). These values are typically determined for the protonated form, and are thus influenced by dissociation. The reported pKa values for PFOA are generally less than 3 (Ding and Peijnenburg, 2013), and is likely close to 0 (Goss, 2008), which means that it will occur in its dissociated form under most environmental conditions. Hence, the effective H values for PFOA and PFOS are likely to be smaller than reported under environmentally relevant pHs. Given their very small H values, partitioning to the soil atmosphere in the vadose zone is unlikely to be a significant retention process for these two compounds. Conversely, H values for other PFAS, such as fluorotelomer alcohols (FTOHs) and fluorotelomer olefins are quite large, ranging approximately 0.05–4 and 100–10,000, respectively (Goss et al., 2006, Ding and Peijnenburg, 2013). Thus, air-water partitioning is anticipated to be a significant process influencing the transport behavior of these compounds. Indeed, atmospheric transport of the fluorotelomers has been identified as the probable cause of their observed widespread distributions in the environment (e.g., Ellis et al., 2003, Ahrens, 2011, Ding and Peijnenburg, 2013).
The retardation factor (R) for aqueous-phase transport of solute undergoing partitioning to soil atmosphere is given by (e.g., Kim et al., 1998):
| (1) |
where H is the air-water partition (Henry’s) coefficient (−), θ,a is volumetric air content (−), and θ,w is volumetric water content (−).
2c. Adsorption at the Air-Water Interface
One retention factor that may be of significance for vadose-zone systems is adsorption at the air-water interface. Interfacial adsorption is distinguished from air-water partitioning wherein the former represents accumulation of constituents at the fluid-fluid interface and whereas the latter represents transfer of constituents into the bulk fluid. For aqueous-phase constituent transport, air-water interfaces provide an additional domain for retention and retardation for conditions wherein the interface is immobile or moves at a velocity lower than that of the bulk aqueous phase. While this factor is often ignored for characterizing the transport and fate of organic contaminants in water-unsaturated systems, it has been demonstrated to be important in some cases. For example, Brusseau and colleagues demonstrated for a bench-scale system that adsorption at the air-water interface contributed 29–73%, of total trichloroethene (TCE) retardation for gas-phase transport, depending on porous-medium type (Brusseau et al., 1997). They later determined that air-water interface adsorption contributed ~24%, of total TCE retardation for an intermediate-scale field experiment (Costanza-Robinson et al., 2013). Kim et al. (1998, 2001) demonstrated similarly that adsorption at the air-water interface contributed significantly to total retardation of various organic compounds for both gas-phase and aqueous-phase transport. Several studies have investigated the impact of air-water interfacial adsorption on retention of surfactants during transport in unsaturated porous media, primarily in terms of the use of surfactants as tracers for interfacial partitioning tracer tests (e.g., Karkare and Fort, 1996, Kim et al., 1997, Anwar et al., 2000, Schaefer et al., 2000, Brusseau et al., 2007, 2015).
PFAS contamination is likely to be present in the vadose zone at many sites as a result of waste disposal, spillage, and specified applications (e.g., fire training). PFOS, PFOA, and related PFAS of concern are surfactants, and by their nature will accumulate at air-water interfaces. Thus, it is anticipated that adsorption to air-water interfaces may be a significant retention and attenuation mechanism for these PFAS in vadose-zone sources. However, this process has not yet been investigated for the transport and fate of PFAS in the subsurface environment.
Fluorinated surfactants are typically more surface active than the analogous standard hydrocarbon surfactants (e.g., Krafft and Riess, 2015). This is related to the unique nature of the “tail” portion of the compound. The tail of fluorinated surfactants is hydrophobic due to its nonpolar, fully saturated nature, just as are the tails of standard hydrocarbon surfactants. However, unlike hydrocarbon surfactants, the tails of fluorinated surfactants are also lipophobic due to the low polarizability of the C–F bond. This unique property results in significantly greater reductions in surface tension caused by fluorinated surfactants. For example, PFOA reduces surface tension to ~22 mN/m at the critical micelle concentration (CMC) of ~12,400 mg/L (Downes et al., 1995, López-Fontan et al., 2005, Vecitis et al., 2008), compared to 45 mN/m for facial areas of 80 and 20 cm−1, respectively, are used based on values measured for a sandy subsoil at the corresponding fluid saturations (Brusseau et al., 2007, 2008, 2015).
Research results from investigations related to chemical applications of perfluorinated surfactants demonstrate that PFOS, PFOA, and related compounds are very strongly adsorbed at the air-water interface. For example, Psillakis et al. (2009) measured enrichment factors for perfluoro-n-alkyl carboxylate F(CF2)nCO2− and sulfonate F(CF2)nSO3− surfactant adsorption at the air-water interface. The enrichment factors measured for PFOS and PFOA were 158 and 63, respectively, demonstrating very large preferential adsorption at the interface. Others have reported similar measurements of strong air-water interface adsorption, measured in terms of surface excess, for PFOS and PFOA (Downes et al., 1995; Vecitis et al., 2008; Lunkenheimer et al., 2015). Recently, it has been hypothesized that adsorption to the air-water interfaces of air bubbles trapped on the surfaces of carbonaceous water-treatment sorbents is a primary source of the retention they afford (Meng et al., 2014). These results indicate that adsorption at air-water interfaces has the potential to be a significant retention mechanism for PFAS transport in vadose-zone systems.
The R for aqueous-phase transport of solute undergoing adsorption at the air-water interface is given as (e.g., Kim et al., 1998; Brusseau et al., 2007):
| (2) |
where Kai is the air-water interface adsorption coefficient (cm3/cm2), Aai is the specific air-water interfacial area (cm2/cm3), and θw is volumetric water content (−). Inspection of equation 2 shows that the magnitude of air-water interface adsorption depends upon the value of Kai and the amount of air-water interface present (Aai).
The value of Kai is determined by molecular properties of the compound, and is influenced by water chemistry properties (e.g., ionic strength, pH, T) (Costanza and Brusseau, 2000). Prior research for PFAS has shown that surface activity (and air-water interface adsorption potential) is influenced by compound chain length (Psillakis et al., 2009; Baba et al., 2013; Lunkenheimer et al., 2015), water chemistry properties such as ionic composition (Downes et al., 1995), and the presence of co-occurring PFAS compounds (Vecitis et al., 2008).
Regarding the amount of interface present, air-water interface is comprised of two components, capillary interface associated with contact between bulk fluid phases (e.g., terminal meniscii, pendular rings, wedges) and film interface associated with air in contact with water films solvating the solid surfaces. The results of experimental, theoretical, and modeling investigations have shown that the magnitude of air-water interface depends upon the amount of water and air present. Specifically, total (capillary + film) air-water interfacial area increases continuously as water content decreases, i.e., as air content increases (e.g., Cary, 1994; Kim et al., 1997, 1999; Or and Tuller, 1999; Schaefer et al., 2000; Oostrom et al., 2001; Costanza and Brusseau, 2002; Dalla et al., 2002; Peng and Brussseau, 2005; Brusseau et al., 2006, 2007). As water content approaches very small values close to zero (equivalent to a few molecular layers of adsorbed water) and the porous medium is almost fully saturated with air, the total interfacial area asymptotically approaches the specific solid surface area of the porous medium. Research has also demonstrated that fluid-fluid interfacial area is a function of soil properties such as grain size, grain-size distribution, and surface roughness (Anwar et al., 2000; Cho and Annable, 2005; Dobson et al., 2006; Brusseau et al., 2008, 2009, 2010; Peng and Brusseau, 2005).
2d. NAPL-water Partitioning
Another potential source of retardation of organic compounds is retention by organic liquids in NAPL source zones. This is of potential significance for PFAS given that they often occur in the presence of NAPL contamination at mixed-waste sites. For example, a primary source of PFAS contamination is the prior use of aqueous film forming foams for fire suppression. In some cases, sites that served as designated fire training areas were also used as disposal sites for solvent and oil wastes.
Initial investigations of the impact of solute partitioning to NAPL (e.g., absorption or “NAPL partitioning”) on aqueous-phase transport of organic solutes were reported by Bouchard et al. (1990), Boyd and Sun (1990), and Brusseau (1990). Brusseau (1992) developed a model that accounted explicitly for NAPL partitioning and solid-phase adsorption, and demonstrated that the transport of organic contaminants can be greatly influenced by NAPL partitioning. For example, simulations showed that the retardation factor for a TCE-like solute increased from 2 for the case of only solid-phase adsorption to 17 for a case including partitioning to NAPL, with a NAPL saturation of 1%. The R increased to 152 when NAPL saturation was increased to 10%. Studies conducted for the partitioning tracer test method of measuring NAPL saturations have demonstrated the impact of NAPL partitioning on field-scale solute transport (Nelson and Brusseau, 1996; Annable et al., 1998a; Cain et al., 2000; Brusseau et al., 2003).
The R for aqueous-phase transport of solute undergoing partitioning into an immobile NAPL phase is given as (e.g., Brusseau 1990):
| (3) |
where Knw is the NAPL-water partition coefficient (−), θn is volumetric NAPL content (−), and θw is volumetric water content (−). Treating NAPL as a retention phase for the transport of solute is predicated on the condition that the NAPL is immobile. This is likely the case for most old sites, with the exception of those with specific remedial operations that may promote significant NAPL movement.
Minimal research has been conducted to examine this factor for PFAS retention or transport in subsurface systems. Log Kow values of 1.92 and 2.45 have been measured for PFOA and PFOS, respectively, at circumneutral pH (Jing et al., 2009). Given these magnitudes, it is anticipated that partitioning to NAPL may be a relevant retention process for PFAS. In one prior study, the impact of oil added to soil on PFOS retention was examined with batch experiments (Chen et al., 2009). The presence of the oil was observed to significantly increase sorption. Log oil-water partition coefficients were estimated to be greater than 4. The impact of trichloroethene NAPL on retention of a PFAS mixture was investigated using batch methods (Guelfo and Higgins, 2013). The results showed complex impacts, wherein sorption was reduced, not changed, or enhanced in the presence of TCE, depending upon the specific PFAS, its concentration, and the specific soil. In both of these studies, the samples were prepared by thoroughly mixing the NAPL with the soil. This approach most likely produces NAPL distributions that are not representative of typical subsurface conditions (e.g., generation of NAPL films coating a significant fraction of the grain surfaces). This distribution could cause interference with solid-phase adsorption via site blockage (as hypothesized in Guelfo and Higgins, 2013), and would produce maximal NAPL-water interface wherein some of the enhanced sorption may be due to adsorption at such interfaces. Conversely, residual trichloroethene NAPL saturation was emplaced using standard methods in the column study reported by McKenzie et al. (2016), wherein the NAPL would be distributed as discrete blobs and ganglia distributed nonuniformly throughout the soil pack. With such distribution, the fraction of soil-grain surface contacted by NAPL would be relatively small, and thus there would likely be minimal direct impact on solid-phase adsorption via surface blockage. This does not discount the potential for indirect effects due to the presence of high aqueous concentrations of trichloroethene. For this latter study, the presence of the residual NAPL was observed to measurably increase overall retardation for most of the PFAS. In these studies, the retention was attributed primarily to NAPL partitioning, while the potential for adsorption to the NAPL-water interface was also noted. However, the specific, discrete impacts of partitioning versus interfacial adsorption were not quantitatively differentiated. Log Knw values ranging from approximately 0 to 0.5 were reported for PFOA and PFOS for the two trichloroethene NAPL studies. These are much smaller than the reported log Kow values and the PFOS log Knw values reported for retention by crude oil (Chen et al., 2009). This may perhaps be due in part to the impact of co-solute effects for the trichloroethene studies, which employed a suite of PFAS. The magnitudes of reported Kow values, along with the results of the few initial studies, indicate that NAPL partitioning may be a significant retention process for select PFAS.
2e. Adsorption at the NAPL-Water Interface
The adsorption of constituents at the NAPL-water interface is similar to adsorption at the air-water interface, and is governed by the same factors as discussed above for the latter process. Adsorption of surfactants to NAPL-water interfaces has been measured for a variety of porous media (Saripalli et al., 1998; Cho and Annable, 2005; Dobson et al., 2006; Brusseau et al., 2008, 2009, 2010; Narter and Brusseau, 2010; McDonald et al., 2016; Zhong et al., 2016). These data have been used to determine the impact of soil properties such as grain size on interfacial adsorption (Cho and Annable, 2005; Dobson et al., 2006; Brusseau et al., 2009, 2010).
The R for aqueous-phase transport of solute undergoing adsorption at the NAPL-water interface is given as (e.g., Brusseau et al., 2008):
| (4) |
where Kni is the NAPL-water interface adsorption coefficient (cm3/cm2), Ani is the specific NAPL-water interfacial area (cm2/cm3), and θw is volumetric water content (−). As discussed in subsection 2c, it is expected that surfactant type PFAS will accumulate at fluid-fluid interfaces, including NAPL-water interfaces. Thus, NAPL-water interface adsorption may be important for transport of PFAS in the source zones of NAPL-contaminated sites. It should also be noted that contaminants present in the vapor phase can adsorb at air-NAPL interfaces. Given the focus herein on PFOA and PFOS, this process is not considered for the analysis. However, it may be relevant for other PFAS.
3. Comprehensive Retention Model
Current published conceptual and mathematical models of PFAS transport and fate in the subsurface focus on solid-phase adsorption as the sole source of retention. For example, all three of the recent detailed assessments of field-scale PFAS transport and fate discussed above employed this assumption (Shin et al., 2011; Xiao et al., 2015; Weber et al., 2017). In addition, this assumption is present in the most recent comprehensive reports on PFAS management (CONCAWE, 2016; CRCCARE, 2017) and in the PFAS section of the Environmental Restoration Wiki (SERDP, 2017). However, the research discussed above examining the impact of adsorption at air-water and NAPL-water interfaces, in conjunction with the strong surfactant properties of some PFAS, indicates that we may anticipate significant retention of PFAS by these processes. In addition, partitioning to NAPL and air phases may be relevant for select PFAS. Thus, it is likely that retardation and transport of PFAS in source zones can be, under certain conditions, influenced by multiple retention processes. In such cases, retardation of PFAS in source areas may be significantly greater than what is typically estimated based on the standard assumption of solid-phase adsorption as the sole retention mechanism. A comprehensive conceptual model is proposed for PFAS retention and retardation to address this situation. It includes the following retention processes: (i) solid-phase adsorption, (ii) adsorption at the air-water interface, (iii) partitioning to the soil atmosphere, (iv) adsorption at the NAPL-water interface, and (v) partitioning to NAPL. To our knowledge, this comprehensive, multi-process retention approach has not been employed to date for PFAS.
The retardation factor for aqueous-phase transport of solute undergoing retention by all of the aforementioned processes is given as:
| (5) |
where H is the air-water partition (Henry’s) coefficient (−), Kd is the solid-phase adsorption coefficient (cm3/g), Knw is the NAPL-water partition coefficient (−), Kai is the air-water interface adsorption coefficient (cm3/cm2), Kni is the NAPL-water interface adsorption coefficient (cm3/cm2), Aai is the specific air-water interfacial area (cm2/cm3), Ani is the specific NAPL-water interfacial area (cm2/cm3), ρb is porous-medium bulk density (g/cm3), θn is volumetric NAPL content (−), θa is volumetric air content (−), and θw is volumetric water content (−). By phase balance, θw + θa + θn = n, where n is porosity, and Sw + Sa + Sn = 1, where Sw, Sa, and Sn are fluid saturations of water, air, and NAPL, respectively (and Si = θ i/n).
The conceptual approach employed has been validated for each of the individual retention processes for a range of constituents in prior work (as noted in section 2), albeit not for PFAS. Thus, while the approach has not been tested specifically for PFAS, there is general confidence in its use to represent phase distributions in subsurface systems. The conceptual model is focused solely on retention and retardation, not transport. Thus, factors such as rate-limited mass transfer and spatial variability of system properties and conditions are not considered. Application of the approach implicitly considers the system to be under pseudo-equilibrium conditions. As discussed above, PFAS distribution coefficients have been shown to be influenced by system conditions. The impact of system conditions on retention and retardation can be accounted for by incorporating distribution coefficients determined for the specific relevant conditions. Note that the model is simplified by assuming that gas-phase transport of compounds in the soil atmosphere is relatively insignificant compared to advective transport in the aqueous phase, such that partitioning to soil atmosphere can be treated as a retention process. In addition, NAPL is assumed to be essentially immobile, and distributed in such a manner that its impact on solid surfaces (e.g., blocking solid-phase adsorption) is minimal. Hence, NAPL is assumed to act as a retention phase for aqueous solute.
4. Parameterization for Illustrative Retardation Factor Calculations
Measured and estimated values for the relevant distribution coefficients will be used to calculate retardation factors for a representative porous medium. The measured porous-medium properties represent a sandy vadose-zone subsoil composed primarily of quartz with moderately low organic-carbon content. The porous-medium related parameter values used for the calculations are presented in Table 1. A water saturation of 78% is selected for the initial analysis to represent a moderately wet system wherein aqueous-phase advective transport is predominant. The impact of different water saturations will also be examined. A NAPL saturation of 2% is used, representative of a mean value averaged across the entire source zone. This value is representative of measured field-scale values (e.g., Nelson and Brusseau, 1996). The air saturation is 20% given the designated water and NAPL saturations. Air-water and NAPL-water interfacial areas of 80 and 20 cm−1, respectively, are used based on values measured for a sandy subsoil at the corresponding fluid saturations (Brusseau et al., 2007, 2008, 2015).
Table 1.
Properties of representative porous medium.
| Property | Value |
|---|---|
| Bulk density (g/cm3) | 1.5 |
| Porosity | 0.333 |
| Fraction of soil organic carbon | 0.002 |
| Water saturation | 0.78 |
| Air saturation | 0.2 |
| NAPL saturation | 0.02 |
| Volumetric water content | 0.26 |
| Volumetric air content | 0.067 |
| Volumetric NAPL content | 0.0067 |
| Air-water interfacial area (cm−1) | 80 |
| NAPL-water interfacial area (cm−1) | 20 |
The two primary PFAS of most concern will be examined, PFOS and PFOA. In addition, 8:2 fluorotelomer alcohol (FTOH) will be included as a representative precursor of PFOA, given that it can undergo transformation to PFOA (e.g., CONCAWE, 2016). While FTOH has not been reported in published reports of sampled groundwater, it and other select PFAS may transport from facilities via atmospheric processes with subsequent deposition to land (e.g., Ellis et al., 2003; Ahrens, 2011; Shin et al., 2011; Ding and Peijnenburg, 2013). Given FTOH’s precursor status and potential for widespread surface distribution, it is relevant to consider its potential transport behavior in soil, as noted by Liu and Lee (2005). It also serves to illustrate the significant differences in transport behavior that exists among the numerous PFAS.
Distribution coefficient values used for the calculations are presented in Table 2. Reported Kow values are used as a surrogate for Knw. This is a reasonable first approximation given that both are measures of liquid-liquid partitioning. As noted above, relatively low TCE-water Knw values were reported for PFOA and PFOS for a PFAS mixture. The impact of these lower values will be discussed below. Measured or calculated H values are taken from the literature. Solid-phase sorption is simplified for this assessment by employing the assumption that sorption is governed by hydrophobic interactions, and is correlated to organic carbon. In this case, Kd is calculated as focKoc, where foc is the fraction of soil organic carbon and Koc is the OC-normalized sorption coefficient. Representative Koc values are taken from the literature. This simplification is reasonable for the representative porous medium used herein, which has minimal clay-mineral and metal-oxide content. Sorption interactions with clay and oxides would need to be considered for more heterogeneous media. Nonlinear adsorption for PFOA and PFOS is accounted for by employing a Freundlich power term of 0.8, based on values reported in the literature.
Table 2.
Distribution coefficients.
| Coefficient | Value | Source |
|---|---|---|
| PFOA | ||
| Koc | 108 | mean of values reported by ref a and ba |
| H | 0.002 | compiled value from Ding and Peijnenburg (2013) |
| Knw [Kow] | 83 | Jing et al. (2009) |
| Kai, Kni (cm)b | 0.0234 | analysis of Vecitis et al. (2008) data |
| PFOS | ||
| Koc | 568 | mean of values reported by ref a and ba |
| H | 0.002 | compiled value from Ding and Peijnenburg (2013) |
| Knw [Kow] | 282 | Jing et al. (2009) |
| Kai, Kni (cm)b | 0.0755 | analysis of Vecitis et al. (2008) data |
| FTOH | ||
| Koc | 13490 | Liu and Lee (2005) |
| H | 3.8 | Goss et al. (2006) |
| Knw [Kow] | 380189 | Carmosini and Lee (2008) |
| Kai, Kni (cm)b | 0.0005 | analysis of Li et al. (2006) data |
Ref a: Higgins and Luthy, 2006, ref b: Milinovic et al., 2015
Calculated for an aqueous PFAS concentration of 0.1 mg/L.
To our knowledge, Kai and Kni values have not yet been reported for PFAS. They can be determined by analyzing measured surface tension or interfacial tension data, which is the standard method for measuring Kai and Kni. The surface excess Γ (mol/cm2) is related to aqueous phase concentration (C) using the Gibbs equation (e.g., Adamson 1982):
| (6) |
where Ki represents the interfacial adsorption coefficient (in our case Kai or Kni), γ is the interfacial tension (dyn/cm), C represents the aqueous phase concentration (mol/cm3), and R is the gas constant (erg/mol °K). In the absence of excess electrolyte, a correction factor of ½ is applied to equation 6. Surface tension data have been reported for PFOA by several investigators (Downes et al., 1995; López et al., 2005; Vecitis et al., 2008; Lunkenheimer et al., 2015), and the results are consistent among the studies. Data reported by Vecitis et al. (2008) are used to calculate Kai values for PFOS and PFOA as these were the sole data available for PFOS. The data were re-plotted and regressed to determine Kai (Figure 1). Note that these data were measured for pure water. Thus, the resultant Kai values may vary somewhat from what would be measured for an aqueous solution containing electrolytes (e.g., groundwater). Interface adsorption data for fluorotelomers is very scarce, with the only data reported for 8:2 FTOH (Li et al., 2006; Rontu and Vaida, 2007). Conversion of the interface uptake coefficient reported by Li et al. (2006) results in a Kni value of approximately 0.0005 cm. This is in the range of values reported for various polar organic compounds (Kim et al., 1998; Costanza and Brusseau, 2000). In the absence of NAPL-water interfacial tension data for PFOA and PFOS, the Kni value will be set equal to Kai. This is a reasonable first approximation, supported by the similarity of Kni and Kai values measured for a common surfactant used as an interfacial partitioning tracer, SDBS (Brusseau et al., 2007,2008, 2010). However, it is possible that Kni and Kai values for PFAS may exhibit greater disparity given their unique physicochemical properties.
Figure 1.
Surface tension measurements for PFOA and PFOS over relevant concentration range. PFOA regression: γ=−0.276*Ln(C)+74.2, r2=0.972; PFOS regression: γ=−0.737*Ln(C)+74.3, r2=0.89. Raw data from Vecitis et al. (2008).
5. Assessing the Contribution of Retention Processes to Retardation
5a. Results
Individual process-specific retardation factor (R) values are calculated and reported in Table 3 for each PFAS, along with the total R for the combined retention from all processes. The fractions of total retention associated with each retention process are also reported. For both PFOA and PFOS, the retardation factors for sorption are of sufficient magnitude to have a significant impact on migration potential. This is consistent with prior analyses reported in the literature. Retention associated with partitioning to the soil atmosphere is negligible, as would be anticipated given the small H values. Partitioning to bulk NAPL contributes similar magnitudes of retention to that of solid-phase sorption. Finally, retention associated with adsorption at the air-water and NAPL-water interfaces is observed to be quite large for both PFOA and PFOS. Specifically, interfacial adsorption comprises approximately 65% of total retention for both compounds. Additionally, air-water interface adsorption alone accounts for approximately 50% of total retention. These results illustrate the significance of interfacial adsorption on PFOA and PFOS retention for this example system.
Table 3.
Process-specific and Total Retardation Factors
| Retention Processa | PFOA | PFOS | FTOH |
|---|---|---|---|
| Sorption | 3.0 (15)b | 11.4 (22) | 157 (2) |
| Air-water Partitioning | 1.0 (0) | 1.0 (0) | 2.0 (0) |
| Air-water Interfacial Adsorption | 8.2 (55) | 24.4 (50) | 1.2 (0) |
| NAPL-water Partitioning | 3.1 (16) | 8.2 (15) | 9749 (98) |
| NAPL-water Interfacial Adsorption | 2.8 (14) | 6.9 (12) | 1.0 (0) |
| Total Retardation Factor | 14.1 | 47.9 | 9906 |
| Rtotal/Rsorption | 4.7 | 4.2 | 63 |
Retardation factors for individual retention processes are calculated using the relevant process-specific equations. An R value of 1 denotes no retention.
Percentage of total retention associated with each retention process (rounded), calculated as (Ri-1)/(Rt-1)*100, where Ri and Rt are the process-specific and total retardation factors, respectively.
Quite different results are obtained for 8:2 FTOH. Adsorption to fluid-fluid interfaces is minimal, which is expected given the much lower surface activity of FTOH compared to PFOS and PFOA. Partitioning to the soil atmosphere is relatively small. However, this could be significant under conditions wherein vapor-phase transport is relevant. The retardation factor for sorption is relatively large, signifying small migration potential. By far the largest source of retention is partitioning to bulk NAPL, contributing ~98% of total retention. Comparison of the results between PFOA/PFOS and FTOH serves to illustrate the significant differences in retention behavior that may be anticipated for the various PFAS as a function of their specific physicochemical properties.
5b. Condition Dependency of the Retention Processes
The illustrative application presented above was developed using a single set of system variables for generally idealized conditions. As discussed in the prior sections, retention by the various processes is influenced by numerous factors. Hence, it is important to discuss the condition dependency of the retardation factors reported in Table 3. Factors that may influence the values include aqueous PFAS concentration, porous-medium conditions such as fluid saturation, and water-chemistry properties (ionic strength, salt composition, co-contaminants). In addition, some of these may change with time or space.
Air-water partitioning and NAPL-water partitioning are linear mass-transfer processes under normal conditions. Therefore, the magnitudes of the associated distribution coefficients reported in Table 2 are independent of PFAS concentration. Thus, the calculated R values apply irrespective of the PFAS concentration present. For this assessment, sorption was treated as a nonlinear process for PFOA and PFOS, and thus the associated R values are a function of concentration. Fluid-fluid interface adsorption coefficients are strongly nonlinear, as seen through examination and application of equation 6. The Kai and Kni values used for the analysis presented in Table 3 are based on an aqueous PFAS concentration of 0.1 mg/L. This represents an approximate median groundwater concentration for ranges of values reported in the literature (cf., Cousins et al., 2016). Lower concentrations would result in larger Kai and Kni values, leading to larger contributions of interface adsorption to retention, and vice versa. The effect of aqueous PFOS concentration on Kai values and resultant R values for air-water interface adsorption and solid-phase adsorption is illustrated in Table 4. The magnitude of the R for air-water interfacial adsorption is observed to differ by a factor of ~200 over a concentration range from 0.01 to 10 mg/L. The contribution of solid-phase adsorption is observed to be greater than that of air-water interfacial adsorption at aqueous PFOS concentrations near and greater than 1 mg/L.
Table 4.
Impact of aqueous PFOS concentration on Kai and R values for air-water interfacial adsorption (Raw), solid-phase adsorption (Rs), and the sum of the two (Rt).
| Concentration (mg/L) |
Kai (cm) | Raw | Rs | Rt |
|---|---|---|---|---|
| 10 | 0.0007 | 1.2 | 5.1 | 5.4 |
| 1 | 0.0076 | 3.3 | 7.6 | 9.9 |
| 0.1 | 0.0755 | 24.3 | 11.4 | 34.7 |
| 0.01 | 0.755 | 234 | 17.5 | 250 |
Kai values were calculated using equation 6 and the regression data from Figure 2.
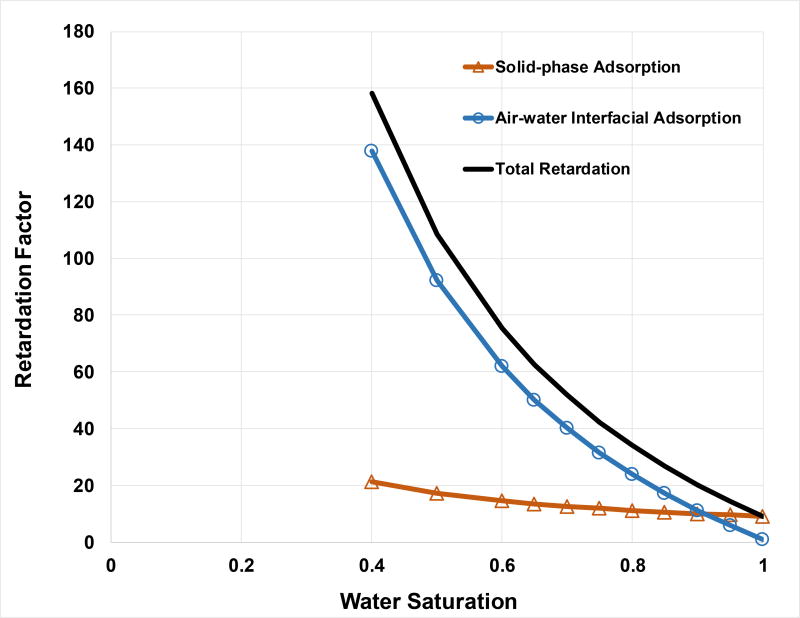
For a given porous medium, bulk density, porosity, and foc are presumed to be constants for most conditions (although site activities may induce changes). Conversely, fluid saturations are condition dependent and can vary over a wide range. Changes in fluid saturation control the magnitudes of the fluid volumes and fluid-fluid interfacial areas, which affects the associated magnitudes of PFAS retention. Thus, the magnitude of the calculated R values will depend upon fluid saturation. This is illustrated in Figure 2, where the R for air-water interface adsorption is observed to increase to 140 as water saturation decreases to 0.4. For this analysis, Aai was assumed to be a linear function of Sw. However, total Aai is actually a nonlinear function of Sw, particularly at lower Sw (e.g., Or and Tuller, 1999; Kim et al., 1999; Peng and Brusseau, 2005; Brusseau et al., 2007). Hence, the increase in R for air-water interface adsorption may in fact be greater than illustrated, especially for Sw lower than 0.4.
Figure 2.
Impact of water saturation on PFOS retardation factors for solid-phase adsorption, air-water interface adsorption, and the sum of the two (total).
The calculations presented above were produced for an idealized system treating each PFAS as the sole constituent of concern in solution. However, it is likely that many PFAS constituents will be present in solution at most sites. In addition, other contaminants such as solvents, fuel constituents, salts, non-PFAS surfactants, and metals may also be present. The presence of co-contaminants is likely to have impacts on the magnitudes of PFAS partitioning to the various phases. For example, competitive and/or cooperative sorption effects may influence solid-phase adsorption. This is illustrated by the results presented by Guelfo and Higgins (2013), who examined the impact of co-contaminants on the sorption of several PFAS. As noted above, the interfacial activity of PFAS has been shown to be influenced by water chemistry properties such as ionic composition (Downes et al., 1995) and the presence of co-occurring PFAS compounds (Vecitis et al., 2008). In addition, the presence of co-contaminants can affect surface wettability, which can influence fluid configuration and distribution, and thereby alter interfacial adsorption. The preceding demonstrates that it is critical to assess the potential impact of co-contaminants on the various retention processes.
The system conditions may change with time, which may influence retention. For example, water saturations may change relatively rapidly with a precipitation-infiltration-redistribution event. Changes in Sw would lead to changes in the magnitudes of air-water interfacial area, which would result in changes in the magnitude of air-water interfacial adsorption. This in turn would cause redistribution of PFAS among the phases. Natural or remediation-induced changes to system conditions may occur, thereby altering retention. For example, site remediation activities may induce changes in the composition and/or physical properties of the NAPL, which can alter its configuration and distribution, thereby influencing retention. Site perturbations may also affect properties of the PFAS mixture, the porous medium, and water chemistry, all of which may influence retention.
Accounting for the impacts of the complexities discussed above is feasible, albeit likely difficult in many cases. The impact of water-chemistry properties and the presence of co-contaminants would ideally be addressed by measuring distribution coefficients for the specific system of interest (i.e., using site groundwater, with all relevant constituents present). The impact of changes in water or NAPL saturation on magnitudes of interfacial area can be accounted for using the correlations that are available in the literature relating interfacial area to porous-medium properties (texture) and fluid saturation.
An alternative or supplemental approach for characterizing multi-process retention under specific site conditions would be to conduct tracer tests using a suite of conservative and partitioning tracers (Brusseau et al., 1997). For example, to delineate NAPL partitioning versus NAPL-water interfacial adsorption, one would use a tracer that partitions to bulk NAPL (with minimal interfacial adsorption) and another tracer that adsorbs at the interface (with minimal bulk partitioning) (e.g., Annable et al., 1998b; Brusseau et al. 2003). Similarly, specific tracers can be selected to characterize solid-phase adsorption, air-water partitioning, and air-water interfacial adsorption (e.g., Nelson and Brusseau, 1996; Annable et al., 1998a; Cain et al., 2000; Brusseau et al., 2003; Costanza-Robinson et al., 2013).
6. Conclusion
Currently, solid-phase adsorption is the sole retention process considered for published conceptual and mathematical models of PFAS transport and fate in the subsurface. However, specific physicochemical properties of PFAS impart a potential propensity to partition to other retention phases. A comprehensive conceptual model is proposed for PFAS retention and retardation to account for these additional retention processes. It includes: (i) solid-phase adsorption, (ii) adsorption at the air-water interface, (iii) partitioning to the soil atmosphere, (iv) adsorption at the NAPL-water interface, and (v) absorption by NAPL. An assessment of the relative magnitudes and significance of these retention processes was conducted for three representative PFAS for a representative, idealized system.
The results showed that adsorption to air-water and NAPL-water interface was a primary source of retention for both PFOA and PFOS, contributing approximately 65% of total retention for the example system. Additionally, air-water interface adsorption alone accounted for ~50% of total retention. This assessment represents a first-order analysis of these factors, given the use of a single system and simplified conditions. Clearly, the specific magnitudes of R reported apply solely to the specific system investigated. The magnitudes and relative significance of the various retention processes will depend upon conditions of each specified system of interest. However, the parameter values employed were obtained primarily from measured data sets, providing confidence in the general validity of the overall results indicating the potential significance of interfacial adsorption. For example, the values used for the Kai were determined from measured surface-tension data, which is the standard method for its determination. The values used for Aai were obtained from reported measured data from prior works. Hence, both parameters used to determine the magnitude of air-water interfacial adsorption originate from measured data.
While simplified conditions were employed in the analysis, the potential impacts of more complex system conditions were discussed. These factors can be accounted for by measuring the distribution coefficients under conditions representative of the particular subsurface environment of concern. In addition, partitioning tracer tests serve as a means by which to characterize multiprocess retention under site-specific field conditions.
The results of this initial assessment indicate that we may anticipate significant retention of PFAS by these additional processes under certain conditions. For example, the total R values for PFOS and PFOA, which account for all potential sources of retention, were approximately 4–10 times larger than the R values for solid-phase adsorption alone. As discussed above, several recent studies have indicated that PFAS present in the vadose zone is a primary, long-term source of groundwater contamination. Thus, the results of the present study suggest specifically that air-water interface adsorption should be considered when assessing PFAS transport and fate in the vadose zone. In addition, the potential for NAPL-water partitioning and NAPL-water interfacial adsorption interactions should be considered for sites with NAPL present.
In cases wherein one or more of the additional retention processes are relevant, retardation of PFAS in source areas may be significantly greater than what is typically estimated based on the assumption of solid-phase adsorption as the sole retention mechanism. This has significant ramifications for example for accurate determination of the migration potential and magnitude of mass flux to groundwater. In addition, calculations of contaminant mass residing in source zones would also be influenced by the existence of these additional retention processes, with the potential for significant underestimation. Both of these elements have critical implications for risk assessment and remedial-action decision-making. Based on these results, additional investigation of the impact of these retention processes on PFAS transport is warranted.
Highlights.
A comprehensive model for PFAS retention in porous media is proposed
Adsorption at the air-water interface contributes greatly to PFOA/PFOS retention
Adsorption at the NAPL-water interface and NAPL absorption are also significant
Acknowledgments
This work was supported by the NIEHS Superfund Research Program (grant# P42 ES04940). Stephen Vinson is thanked for drafting the Graphical Abstract. I thank the reviewers for their constructive comments.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adamson AW. Physical chemistry of surfaces. 4. J. Wiley; New York: 1982. p. 664. [Google Scholar]
- Ahrens L. Polyfluoroalkyl compounds in the aquatic environment: a review of their occurrence and fate. J. Environ. Monit. 2011;13:20–31. doi: 10.1039/c0em00373e. [DOI] [PubMed] [Google Scholar]
- Anderson RH, Long GC, Porter RC, Anderson JK. Occurrence of select perfluoroalkyl substances at U.S. Air Force aqueous film-forming foam release sites other than fire-training areas: Field-validation of critical fate and transport properties. Chemo. 2016;150:678–685. doi: 10.1016/j.chemosphere.2016.01.014. [DOI] [PubMed] [Google Scholar]
- Annable M, Rao PSC, Hatfield K, Graham W, Wood LJ. Use of partitioning tracers for measuring residual NAPL: Results from a field-scale test. Environ. Eng. 1998a;124:498–503. [Google Scholar]
- Annable MD, Jawitz JW, Rao PSC, Dai DP, Kim H, Wood AL. Field evaluation of interfacial and partitioning tracers for characterization of effective NAPL-water contact areas. Ground Water. 1998b;36(3):495–502. [Google Scholar]
- Anwar A, Bettahar M, Matsubayashi UJ. A method for determining air-water interfacial area in variably saturated porous media. J. Contam. Hydrol. 2000;43:129–146. [Google Scholar]
- Backe WJ, Day TC, Field JA. Zwitterionic, cationic, anionic fluorinated chemicals in AFFF formulations and groundwater from U.S. military bases by non-aqueous large-volume injection HPLC-MS/MS. Environmental Science & Technology. 2013;47:5226–5234. doi: 10.1021/es3034999. [DOI] [PubMed] [Google Scholar]
- Bouchard DC, Mravik SC, Smith GB. Benzene and naphthalene sorption on soil contaminated with high molecular weight residual hydrocarbons from unleaded gasoline, Chemosphere. 1990;21(8):975–989. [Google Scholar]
- Boyd SA, Sun S. Residual petroleum and polychlorinatedbiphenyl oils as sorptive phases for organic contaminants in soils. Environ. Sci. Technol. 1990;24(1):142–144. [Google Scholar]
- Brusseau ML. Micro-scale mass transfer processes and field-scale transport of organic solutes, paper presented at International Conference on Transport and Mass Exchange Processes in Sand and Gravel Aquifers. Proc. published by Atomic Energy Can. Ltd; Chalk River, Canada: 1990. [Google Scholar]
- Brusseau ML. Rate-limited mass transfer and transport of organic solutes in porous media that contain immobile immiscible organic liquid. Water Resources Research. 1992;28:33–45. [Google Scholar]
- Brusseau ML, Popovicova J, Silva J. Characterizing gas-water interfacial and bulk-water partitioning for transport of gas-phase contaminants in unsaturated porous media. Environmental Science and Technology. 1997;31:1645–1649. [Google Scholar]
- Brusseau ML, Nelson NT, Costanza-Robinson MS. Partitioning tracer tests for characterizing immiscible-fluid saturations and interfacial areas in the vadose zone. Vadose Zone Journal. 2003;2:138–147. [Google Scholar]
- Brusseau ML, Peng S, Schnaar G, Costanza-Robinson MS. Relationships among air-water interfacial area, capillary pressure, and water saturation for a sandy porous medium. Water Resour Res. 2006;42:W03501. doi: 10.1029/2005WR004058. [DOI] [Google Scholar]
- Brusseau ML, Peng S, Schnaar G, Murao A. Measuring air-water interfacial areas with x-ray microtomography and interfacial partitioning tracer tests. Environmental Science & Technology. 2007;41:1956–1961. doi: 10.1021/es061474m. [DOI] [PubMed] [Google Scholar]
- Brusseau ML, Janousek H, Murao A, Schnaar G. Synchrotron x-ray microtomography and interfacial partitioning tracer test measurements of NAPL-water interfacial areas. Water Resources Research. 2008;44:W01411. doi: 10.1029/2006WR005517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brusseau ML, Narter M, Schnaar S, Marble J. Measurement and estimation of organic-liquid/water interfacial areas for several natural porous media. Environmental Science & Technology. 2009;43:3619–3625. doi: 10.1021/es8020827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brusseau ML, Narter N, Janousek H. Interfacial partitioning tracer test measurements of organic-liquid/water interfacial areas: application to soils and the influence of surface roughness. Environmental Science & Technology. 2010;44:7596–7600. doi: 10.1021/es101312n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brusseau ML, El Ouni A, Araujo JB, Zhong H. Novel methods for measuring air-water interfacial area in unsaturated porous media. Chemo. 2015;127:208–213. doi: 10.1016/j.chemosphere.2015.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cain RB, Johnson GR, McCray JE, Blanford WJ, Brusseau ML. Partitioning Tracer Tests for Evaluating Remediation Performance. Ground Water. 2000;38:752–761. [Google Scholar]
- Carmosini N, Lee LS. Partitioning of fluorotelomer alcohols to octanol and different sources of dissolved organic carbon. Environ. Sci. Technol. 2008;42(17):6559–6565. doi: 10.1021/es800263t. [DOI] [PubMed] [Google Scholar]
- Cary JW. Estimating the surface area of fluid phase interfaces in porous media. J. Contam. Hydrol. 1994;15:243–248. [Google Scholar]
- Chen H, Chen S, Quan X, Zhao YZ, Zhao HM. Sorption of perfluorooctane sulfonate (PFOS) on oil and oil-derived black carbon: Influence of solution pH and [Ca2+] Chemo. 2009;77:1406–1411. doi: 10.1016/j.chemosphere.2009.09.008. [DOI] [PubMed] [Google Scholar]
- Cho J, Annable MD. Characterization of pore scale NAPL morphology in homogeneous sands as a function of grain size and NAPL dissolution. Chemo. 2005;61:899–908. doi: 10.1016/j.chemosphere.2005.04.042. [DOI] [PubMed] [Google Scholar]
- CONCAWE. Environmental fate and effects of poly and perfluoroalkyl substances (PFAS) Network for Industrially Contaminated Land in Europe, CONCAWE: 2017. [Google Scholar]
- Costanza M, Brusseau ML. Influence of adsorption at the air-water interface on the transport of volatile contaminants in unsaturated porous media. Environmental Science and Technology. 2000;34:1–11. [Google Scholar]
- Costanza-Robinson MS, Brusseau ML. Air-water interfacial areas in unsaturated soils: Evaluation of interfacial domains. Water Resour. Res. 2002;38:131–137. [Google Scholar]
- Costanza-Robinson MS, Carlson TD, Brusseau ML. Vapor-phase transport of trichloroethene in an intermediate-scale vadose-zone system: retention processes and tracer-based prediction. Journal of Contaminant Hydrology. 2013;145:182–189. doi: 10.1016/j.jconhyd.2012.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cousins IT, Vestergren R, Wang Z, Scheringer M, McLachlan MS. The precautionary principle and chemicals management: The example of perfluoroalkyl acids in groundwater. Environ. Inter. 2016;94:331–340. doi: 10.1016/j.envint.2016.04.044. [DOI] [PubMed] [Google Scholar]
- CRCCARE. Assessment, management and remediation guidance for perfluorooctanesulfonate (PFOS) and perfluorooctanoic acid (PFOA) - Part 5: management and remediation of PFOS and PFOA, CRC CARE Technical Report no. 38. CRC for Contamination Assessment and Remediation of the Environment; Newcastle, Australia: 2017. [Google Scholar]
- Dalla E, Hilpert M, Miller CT. Computation of the interfacial area for two fluid porous medium systems. J. Contam. Hydrol. 2002;56:25–48. doi: 10.1016/s0169-7722(01)00202-9. [DOI] [PubMed] [Google Scholar]
- Ding G, Peijnenburg WJGM. Physicochemical Properties and Aquatic Toxicity of Poly- and Perfluorinated Compounds. Critical Reviews in Environmental Science and Technology. 2013;43(6):598–678. [Google Scholar]
- Dobson R, Schroth MH, Oostrom M, Zeyer J. Determination of NAPL-water interfacial areas in well-characterized porous media. Environ. Sci. Technol. 2006;40:815–822. doi: 10.1021/es050037p. [DOI] [PubMed] [Google Scholar]
- Downes N, Ottewill GA, Ottewill RH. An investigation of the behavior of ammonium perfluoro-octanoate at the air/water interface in the absence and presence of salts. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 1995;102:203–211. [Google Scholar]
- Du Z, Deng S, Bei Y, Huang Q, Wang B, Huang J, Yu G. Adsorption behavior and mechanism of perfluorinated compounds on various adsorbents—A review. J. Hazard. Mater. 2014;274:443. doi: 10.1016/j.jhazmat.2014.04.038. [DOI] [PubMed] [Google Scholar]
- Ellis DA, Martin JW, Mabury SA, Hurley MD, Andersen MPS, Wallington TJ. Atmospheric lifetime of fluorotelomer alcohols. Environ. Sci. Technol. 2003;37:3816–3820. doi: 10.1021/es034136j. [DOI] [PubMed] [Google Scholar]
- Environmental Protection Agency (USEPA) Long-Chain Perfluorinated Chemicals (PFCs) Action Plan. U.S. Environmental Protection Agency; Washington, DC: 2009a. [December 30, 2009]. https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/long-chain-perfluorinated-chemicals-pfcs-action-plan. [Google Scholar]
- Environmental Protection Agency (USEPA) Provisional Health Advisories for Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS) U.S. Environmental Protection Agency; Washington, DC: 2009b. [January 8, 2009]. [Google Scholar]
- Environmental Protection Agency (USEPA) Emerging Contaminants Perflurooctane sulfonate (PFOS) and Perfluorooctanic acid (PFOA) U.S. Environmental Protection Agency; Washington, DC: 2014. pp. 1–10. EPA 505-F-14001. [Google Scholar]
- Environmental Protection Agency (USEPA) FACT SHEET PFOA & PFOS Drinking Water Health Advisories. U.S. Environmental Protection Agency; Washington, DC: 2016. November 2016 EPA 800-F-16-003. [Google Scholar]
- Ferrey ML, Wilson JT, Adair C, Su C, Fine DD, Liu X, Washington JW. Behavior and fate of PFOA and PFOS in sandy aquifer sediment. Groundwater Monit. Remed. 2012;32(4):63–71. [Google Scholar]
- Goss KU. The pKa values of PFOA and other highly fluorinated carboxylic acids. Environmental Science and Technology. 2008;42:456–458. doi: 10.1021/es702192c. [DOI] [PubMed] [Google Scholar]
- Goss K, Bronner G, Harner T, Hertel M, Schmidt TC. The partition behavior of fluorotelomer alcohols and olefins. Environmental Science and Technology. 2006;40:3572–3577. doi: 10.1021/es060004p. [DOI] [PubMed] [Google Scholar]
- Guelfo JL, Higgins CP. Subsurface transport potential of perfluoroalkyl acids at aqueous film-forming foam (AFFF)-impacted sites. Environ. Sci. Technol. 2013;47:4164–4171. doi: 10.1021/es3048043. [DOI] [PubMed] [Google Scholar]
- Hellsing MS, Josefsson S, Hughes AV, Ahrens L. Sorption of perfluoroalkyl substances to two types of minerals. Chemosphere. 2016;159:385–391. doi: 10.1016/j.chemosphere.2016.06.016. [DOI] [PubMed] [Google Scholar]
- Higgins CP, Luthy RG. Sorption of perfluorinated surfactants on sediments. Environ. Sci. Technol. 2006;40:7251. doi: 10.1021/es061000n. [DOI] [PubMed] [Google Scholar]
- Higgins CP, Luthy RG. Modeling sorption of anionic surfactants onto sediment materials: An a priori approach for perfluoroalkyl surfactants and linear alkylbenzene sulfonates. Environ. Sci. Technol. 2007;41:3254–3261. doi: 10.1021/es062449j. [DOI] [PubMed] [Google Scholar]
- Higgins CP, Luthy RG. Correction to: modeling sorption of anionic surfactants onto sediment materials: An a priori approach for perfluoroalkyl surfactants and linear alkylbenzene sulfonates. Environ. Sci. Technol. 2007;41:6316. doi: 10.1021/es062449j. [DOI] [PubMed] [Google Scholar]
- Houtz EF, Higgins CP, Field JA, Sedlak DL. Persistence of perfluoroalkyl acid precursors in AFFF-impacted groundwater and soil. Environ. Sci. Technol. 2013;47(15) doi: 10.1021/es4018877. [DOI] [PubMed] [Google Scholar]
- Hu XC, Andrews DQ, Lindstrom AB, Bruton TA, Schaider LA, Grandjean P, Lohmann R, Carignan CC, Blum A, Balan SA, Higgins CP, Sunderland EM. Detection of poly- and perfluoroalkyl substances (PFAS) in U.S. drinking water linked to industrial sites, military fire training areas and wastewater treatment plants. Environ. Sci. Technol. Lett. 2016;3:344–350. doi: 10.1021/acs.estlett.6b00260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jing P, Rodgers PJ, Amemiya S. High lipophilicity of perfluoroalkyl carboxylate and sulfonate: Implications for their membrane permeability. Journal of the American Chemical Society. 2009;131:2290–2296. doi: 10.1021/ja807961s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson RL, Anschutz AJ, Smolen JM, Simcik MF, Penn RL. The adsorption of perfluorooctane sulfonate onto sand, clay, and iron oxide surfaces. Journal of Chemical & Engineering Data. 2007;52:1165–1170. [Google Scholar]
- Karkare MV, Fort T. Determination of the air-water interfacial area in wet “unsaturated” porous media. Langmuir. 1996;12:2041–4044. [Google Scholar]
- Kim H, Rao PSC, Annable MD. Determination of effective air–water interfacial area in partially saturated porous media using surfactant adsorption. Water Resour. Res. 1997;33(12):2705–2711. [Google Scholar]
- Kim H, Rao PSC, Annable MD. Influence of air-water interfacial adsorption and gas-phase partitioning on the transport of organic chemicals in unsaturated porous media. Env. Sci. Technol. 1998;32:1253–1259. doi: 10.1021/es001965l. [DOI] [PubMed] [Google Scholar]
- Kim H, Rao PSC, Annable MD. Gaseous tracer technique for estimating air-water interfacial areas and interface mobility. Soil Sci. Soc. Am. J. 1999;63:1554–1560. [Google Scholar]
- Kim H, Annable MD, Rao PSC. Gaseous transport of volatile organic chemicals in unsaturated porous media: effect of water-partitioning and air-water interfacial adsorption. Env. Sci. Technol. 2001;35:4457–4462. doi: 10.1021/es001965l. [DOI] [PubMed] [Google Scholar]
- Krafft MP, Riess JG. Per- and polyfluorinated substances (PFAS): environmental challenges. Curr. Opin. Colloid Interface Sci. 2015a;20:192–212. [Google Scholar]
- Krafft MP, Riess JR. Selected physicochemical aspects of poly and perfluoroalkylated substances relevant to performances, environment and sustainability—part one. Chemosphere. 2015b;129:4–19. doi: 10.1016/j.chemosphere.2014.08.039. [DOI] [PubMed] [Google Scholar]
- Li Y, Demerjian KL, Williams LR, Worsnop DR, Kolb CE, Davidovits PJ. Phys. Chem. A. 2006;110:6814–6820. doi: 10.1021/jp0563134. [DOI] [PubMed] [Google Scholar]
- Liu JX, Lee LS. Solubility and sorption by soils of 8:2 fluorotelomer alcohol in water and cosolvent systems. Environ. Sci. Technol. 2005;39:7535–7540. doi: 10.1021/es051125c. 2005. [DOI] [PubMed] [Google Scholar]
- Liu J, Lee LS. Effect of fluorotelomer alcohol chain length on aqueous solubility and sorption by soils. Environ. Sci. Technol. 2007;41(15):5357–5362. doi: 10.1021/es070228n. [DOI] [PubMed] [Google Scholar]
- López-Fontán JL, Sarmiento F, Schulz PC. The aggregation of sodium perfluorooctanoate in water. Colloid and Polymer Science. 2005;283:862–871. [Google Scholar]
- Lunkenheimer K, Prescher D, Hirte R, Geggel K. Adsorption properties of surface chemically pure sodium perfluoro-n-alkanoates at the air/water interface: counterion effects within homologous series of 1:1 ionic surfactants. Langmuir. 2015;31:970–981. doi: 10.1021/la503450k. [DOI] [PubMed] [Google Scholar]
- McDonald K, Carroll KC, Brusseau ML. Comparison of fluid-fluid interfacial areas measured with X-ray microtomography and interfacial partitioning tracer tests for the same samples. Water Resour Res. 2016;52:5393–5399. doi: 10.1002/2016WR018775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGuire ME, Schaefer C, Richards T, Backe WJ, Field JA, Houtz E, Sedlak DL, Guelfo JL, Wunsch A, Higgins CP. Evidence of remediation-induced alteration of subsurface poly- and perfluoroalkyl substance distribution at a former firefighter training area. Environ. Sci. Technol. 2014;48(12):6644–6652. doi: 10.1021/es5006187. [DOI] [PubMed] [Google Scholar]
- McKenzie ER, Siegrist RL, McCray JE, Higgins CP. The influence of a non-aqueous phase liquid (NAPL) and chemical oxidant application on perfluoroalkyl acid (PFAA) fate and transport. Water Res. 2016;92:199–207. doi: 10.1016/j.watres.2016.01.025. [DOI] [PubMed] [Google Scholar]
- Meng P, Deng SB, Deng S, Lu X, Du Z, Wang B, Huang J, Wang Y, Yu G, Xing B. Environ. Sci. Technol. 2014;48:13785–13792. doi: 10.1021/es504108u. [DOI] [PubMed] [Google Scholar]
- Merino N, Qu Y, Deeb RA, Hawley EL, Hoffmann MR, Mahendra S. Degradation and Removal Methods for Perfluoroalkyl and Polyfluoroalkyl Substances in Water. Environmental Engineering Science. 2016;33:615–648. [Google Scholar]
- Milinovic J, Lacorte S, Vidal M, Rigol A. Sorption behaviour of perfluoroalkyl substances in soils. Sci. Total Environ. 2015;511:63–71. doi: 10.1016/j.scitotenv.2014.12.017. [DOI] [PubMed] [Google Scholar]
- Moody CA, Field JA. Perfluorinated surfactants and the environmental implications of their use in fire-fighting foams. Environ. Sci. Technol. 2000;34:3864–3870. [Google Scholar]
- Moody CA, Hebert GN, Strauss SH, Field JA. Occurrence and persistence of perfluorooctanesulfonate and other perfluorinated surfactants in groundwater at a fire-training area at Wurtsmith air force base, Michigan, USA. J. Environ. Monit. 2003;5:341–345. doi: 10.1039/b212497a. [DOI] [PubMed] [Google Scholar]
- Narter M, Brusseau ML. Comparison of interfacial partitioning tracer test and high-resolution microtomography measurements of fluid-fluid interfacial areas for an ideal porous medium. Water Resour Res. 2010;46:W08602. doi: 10.1029/2009WR008375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson NT, Brusseau ML. Field study of the partitioning tracer method for detection of dense nonaqueous phase liquid in a trichloroethene-contaminated aquifer. Environmental Science and Technology. 1996;30:2859–2863. [Google Scholar]
- Oostrom M, White MD, Brusseau ML. Theoretical estimation of free and entrapped nonwetting-wetting fluid interfacial areas in porous media. Adv. Water Resour. 2001;24:887–898. [Google Scholar]
- Or D, Tuller M. Liquid retention and interfacial area in variably saturated porous media: Upscaling from single-pore to sample-scale model. Water Resour. Res. 1999;35:3591–3605. [Google Scholar]
- Pan G, Jia CX, Zhao DY, You C, Chen H, Jiang GB. Effect of cationic and anionic surfactants on the sorption and desorption of perfluorooctane sulfonate (PFOS) on natural sediments. Environ. Pollut. 2009;157:325–330. doi: 10.1016/j.envpol.2008.06.035. [DOI] [PubMed] [Google Scholar]
- Peng S, Brusseau ML. Impact of soil texture on air-water interfacial areas in unsaturated sandy porous media. Water Resources Research. 2005;41:W03021. doi: 10.1029/2004WR003233. [DOI] [Google Scholar]
- Psillakis E, Cheng J, Hoffmann MR, Colussi AJ. Enrichment factors of perfluoroalkyl oxoanions at the air/water interface. The Journal of Physical Chemistry A Letters. 2009;113:8826–8829. doi: 10.1021/jp902795m. [DOI] [PubMed] [Google Scholar]
- Rayne S, Forest K. Perfluoroalkyl sulfonic and carboxylic acids: A critical review of physicochemical properties, levels and patterns in waters and wastewaters, and treatment methods. J. Environ. Sci. Health Part A. 2009;44:1145–1199. doi: 10.1080/10934520903139811. [DOI] [PubMed] [Google Scholar]
- Rontu N, Vaida V. Surface Partitioning and stability of pure and mixed films of 8-2 fluorotelomer alcohol at the air-water interface. J. Phys. Chem. C. 2007;111:11612–11618. [Google Scholar]
- Saripalli KP, Rao PSC, Annable MD. Determination of specific NAPL-water interfacial areas of residual NAPLs in porous media using the interfacial tracers technique. J. Contam. Hydrol. 1998;30:375–391. [Google Scholar]
- Schaefer CE, DiCarlo DA, Blunt MJ. Experimental measurement of air-water interfacial area during gravity drainage and secondary imbibition in porous media. Water Resour. Res. 2000;36:885–890. [Google Scholar]
- SERDP Environmental Restoration Wiki. [Last accessed July 2017]; http://www.environmentalrestoration.wiki/index.php?title=Perfluoroalkyl_and_Polyfluoroalkyl_Substances_(PFASs)
- Shin HM, Vieira VM, Ryan PB, Detwiler R, Sanders B, Steenland K, Bartell SM. Environmental fate and transport modeling for perfluorooctanoic acid emitted from the Washington works facility in West Virginia. Environ. Sci. Technol. 2011;45(4):1435–1442. doi: 10.1021/es102769t. [DOI] [PubMed] [Google Scholar]
- Vecitis CD, Park H, Cheng J, Mader BT, Hoffmann MR. Enhancement of perfluorooctanoate (PFOA) and perfluorooctanesulfonate (PFOS) activity at acoustic cavitation bubble interfaces. Journal of Physical Chemistry. 2008;112:16850–16857. [Google Scholar]
- Weber AK, Barber LB, LeBlanc DR, Sunderland EM, Vecitis CD. Geochemical and hydrologic factors controlling subsurface transport of poly- and perfluoroalkyl substances, Cape Cod, Massachusetts. Environ. Sci. Technol. 2017;51:4269–4279. doi: 10.1021/acs.est.6b05573. 2017. [DOI] [PubMed] [Google Scholar]
- Xiao F, Simcik MF, Halbach TR, Gulliver JS. Perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) in soils and groundwater of a U.S. metropolitan area: Migration and implications for human exposure. Water Res. 2015;72:64–74. doi: 10.1016/j.watres.2014.09.052. [DOI] [PubMed] [Google Scholar]
- Zhang R, Yan W, Jing C. Experimental and molecular dynamic simulation study of perfluorooctane sulfonate adsorption on soil and sediment components. J. Environ. Sci. 2015;29:131–138. doi: 10.1016/j.jes.2014.11.001. [DOI] [PubMed] [Google Scholar]
- Zhao L, Bian J, Zhang Y, Zhu L, Liu Z. Comparison of the sorption behaviors and mechanisms of perfluorosulfonates and perfluorocarboxylic acids on three kinds of clay minerals. Chemosphere. 2014;114:51–60. doi: 10.1016/j.chemosphere.2014.03.098. [DOI] [PubMed] [Google Scholar]
- Zhong H, El Ouni A, Lin D, Wang B, Brusseau ML. The two-phase flow IPTT method for measurement of nonwetting-wetting liquid interfacial areas at higher nonwetting saturations in natural porous media. Water Resour. Res. 2016;52:5506–5515. doi: 10.1002/2016WR018783. [DOI] [PMC free article] [PubMed] [Google Scholar]