Abstract
Introduction
Mucopolysaccharidosis Type II (MPS II; Hunter syndrome) is an X- linked lysosomal storage disorder caused by a deficiency of iduronate-2-sulfatase (IDS). IDS deficiency leads to primary accumulation of dermatan sulfate (DS) and heparan sulfate (HS). MPS II is both multi-systemic and progressive. Phenotypes are classified as either attenuated or severe (based on absence or presence of central nervous system impairment, respectively).
Areas covered
Current treatments available are intravenous enzyme replacement therapy (ERT), hematopoietic stem cell transplantation (HSCT), anti-inflammatory treatment, and palliative care with symptomatic surgeries. Clinical trials are being conducted for intrathecal ERT and gene therapy is under pre-clinical investigation. Treatment approaches differ based on age, clinical severity, prognosis, availability and feasibility of therapy, and health insurance.
This review provides a historical account of MPS II treatment as well as treatment development with insights into benefits and/or limitations of each specific treatment.
Expert opinion
Conventional ERT and HSCT coupled with surgical intervention and palliative therapy are currently the treatment options available to MPS II patients. Intrathecal ERT and gene therapy are currently under investigation as future therapies. These investigative treatments are critical to address the limitations in treatment of the central nervous system (CNS).
Keywords: MPS II, enzyme replacement therapy, hematopoietic stem cell transplantation, gene therapy, anti-inflammatory therapy
1. Overview
1.1. Historical aspect
Mucopolysaccharidosis Type II (MPS II; Hunter Syndrome) was first described in 1917 by Dr. Charles Hunter who examined two brothers (8 and 10 years old) with short stature, large heads, umbilical hernias, and joint stiffness [1]. The younger brother experienced central nervous system involvement (CNS) while the older brother did not. Both cases died at ages 11 and 16, respectively, due to respiratory and cardiovascular complications [1].
In 1952, clinical features associated with a general accumulation of mucopolysaccharides or glycosaminoglycans (GAGs), including defects of collagenous tissues- cartilage, tendons fasciae and heart valves, were recorded [2]. In 1962, Teller et al. reported an elevation of GAGs in the urine of MPS children, and found that differentiation of MPS types was possible based on accumulation of different GAG subclasses [3]. In 1970, cross-correctional studies elucidated differences in enzyme deficiencies between MPS II and MPS I (Hurler) syndromes, though both MPS I and MPS II lead to accumulation of dermatan sulfate (DS) and heparan sulfate (HS) [4]. In 1973, Bach et al. identified sulfoiduronate sulfatase as the deficient enzyme in MPS II, causing the accumulation of dermatan DS and HS [5], and from 1982 to 1991, the first ten hematopoietic stem cell transplantations (HSCT) were conducted on MPS II patients [7]. The cDNA of IDS was cloned in 1990 [8], and the first clinical trial of enzyme replacement therapy (ERT) for patients with MPS II was conducted in 2001 [9]. Phase III clinical trials for ERT in MPS II patients were completed in 2006 after which it gained approval for patient treatment [10].
1.2. Biochemical aspect
MPS II is an X-linked disorder caused by the deficiency of iduronate-2-sulfatase (IDS) due to mutations in the IDS gene (Xq28) [11, 12]. Typically, males are affected, however, females can be affected [11–19], due to skewed X-chromosome inactivation [19, 20] or chromosomal rearrangement [17,18].
1.3. Clinical phenotype
MPS II is a progressive disease involving multiple systems and presenting with a wide spectrum of clinical symptoms. The primary accumulation of DS and HS leads to impairment of several processes including cell trafficking, endocytosis, autophagy, ion homeostasis, and nutrient sensing [21]. DS accumulation results in enhanced cytotoxicity via nitric oxide (NO) release [22] and induction of the inflammatory response [23].
Accumulation of HS results in accumulation of secondary molecules such as gangliosides (GM2 and GM3) in the brain, causing microglial activation [24–29], which activates the inflammatory response pathway in the brain [23].
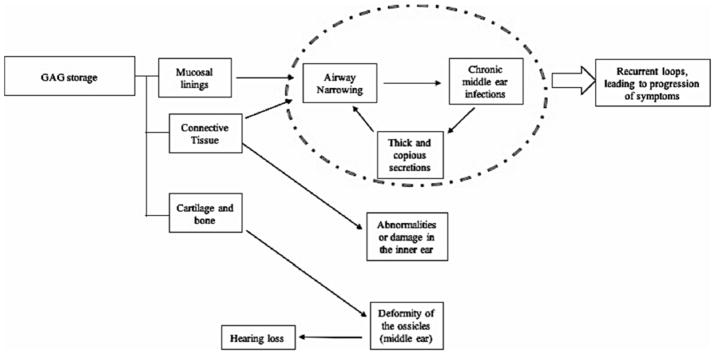
While genotype variations can cause some of the clinical heterogeneity (extensive range of signs and symptoms) a specific mutation does not necessarily coordinate with the observed phenotype [30]. Patients are currently categorized, in the most general terms, as having either attenuated (no CNS impairment) or severe (CNS impairment) [25,18, 31,] The severe phenotype of MPS II is more prevalent (60%) [31]. Typically, hydrocephalus develops before any behavioral changes indicative of CNS involvement [33,34]. This is followed by behavioral difficulties and hyperactivity at around 3.5 years of age, although CNS manifestation can occur much later. Attention difficulties, particularly in school aged children are prevalent, potentially due to abnormal corpus callosum volumes [37–39]. Scholastically, children with severe MPS II develop normally until around 3 to 4 years of age when cognition begins to decline [35,36,32]. Language skills are typically slow to develop due to the cognitive decline and hearing loss that frequently occur in conjunction with other symptoms. Mild to moderate conductive deafness results from frequent ear infections, as well as inner and middle ear abnormalities including deformity of the ossicles (Fig 1) [40]. Frequent epileptic seizure is another common consequence of CNS involvement.
Figure 1.
Pathophysiology of hearing loss in MPS II (Reused with permission from [161] permitted by Japanese MPS Society)
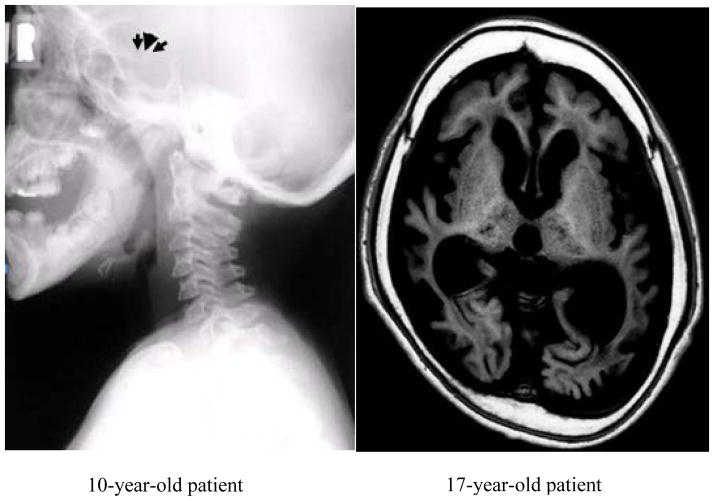
There is evidence of both gray and white matter being involved in behavioral deficits in MPS II patients. White matter abnormalities (WMA) consist of diffuse or focal areas prolonged in T1 (longitudinal relaxation time) and T2 (transverse relaxation time) relaxation times. Periventricular white matter exhibits more frequent abnormalities [34]. Gray matter signal intensity abnormalities have been found in the basal ganglia [41, 34]. Brain magnetic resonance imaging (MRI) scans have revealed brain atrophy typical in patients with seizures [42]. MPS II patients with CNS involvement often display a “honeycomb-like appearance” in the brain which describes the patchy areas of increased and decreased signals in the T1 and T2 regions (Figure 2). This imbalanced distribution of white and gray matter is a potential cause for the epileptic seizures seen in MPS II patients [43, 34, 44].
Figure 2.
Images of skull X-ray and brain MRI from a 10-year-old patient and 17-year-old MPS II patients, respectively. The left pane (a 10-year-old patient with severe phenotype): Scaphocephalic form, partial sutural synostosis, and enlarged omega- or ‘J-shaped’ sella turcica. Right panel (a 17-year-old patient with severe phenotype): T1 image shows severe brain atrophy, cystic or cribriform lesions, white matter signal changes, and ventricular enlargement.
Patients with attenuated phenotypes do not have CNS involvement, allowing normal intellectual development. Nevertheless, long-term follow-up examination in some patients with an attenuated phenotype shows a slow progression of CNS involvement and/or retinal degeneration in cases where patients might have previously been considered to have no CNS involvement [37]. For this reason, prognosis and diagnosis of clinical severity should be made carefully in favor of a more robust and comprehensive outlook.
Dental Abnormalities
Patients with MPS II often show dental abnormalities. Teeth are typically abnormal in number, shape and form including malocclusion and enamel defects (both milk and definite teeth), anterior open bite, undershot jaw, and other orthodontic problems. Patients typically need to be followed by an oral specialist before symptoms begin. Patients are more prone to developing cysts and abscesses due to increased rates of oral infection [45–47].
1.3.1 Ear, nose, and throat
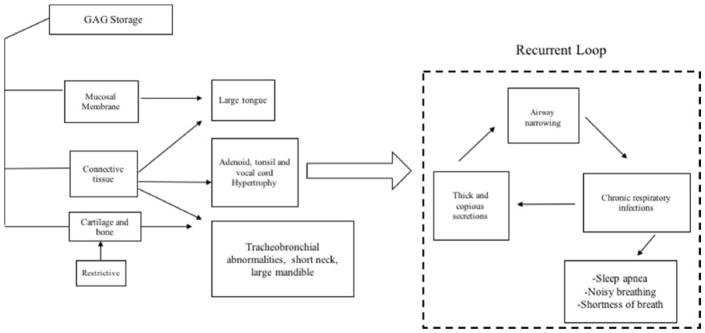
Patients often have ear, nose, and throat (ENT) problems including noisy breathing, recurrent otitis, sinusitis, and upper respiratory infections with atypical mucosal discharge [48–50]. These disturbances are caused by an enlarged and swollen tongue, redundant trachea, and thickened and copious mucosal secretions in addition to an increased inflammatory immune response. Breathing difficulties, often accompanied by sleep apnea, are caused by engorged tonsils and adenoids, narrowed trachea, and thickened vocal chords (Fig. 2) [48,51].
Frequent respiratory infections typically lead to limited endurance including intolerance of activity and decreased activity of daily living (ADL), which is a standard measure of independence in completing routine activities. Superficial, loud, and rapid breathing are typical results. The restrictive airway disease is exacerbated by bone and joint pathology including an abnormally shaped ribcage and thoracolumbar deformities (Fig. 3). Progressive deposition of GAGs in the soft tissue of the throat and trachea leads to a narrow and difficult airway [52]. These breathing abnormalities often occur in conjunction with cardiovascular disease [49,52].
Figure 3.
Pathophysiology of the airway in MPS II (Reused from [161] with permission from Japanese MPS Society)
1.3.2 Cardiovascular involvement
The MPS II outcome survey revealed that valvular heart disease (63%), followed by left ventricular hypertrophy (48%) and elevated blood pressure (25%), are common cardiovascular abnormalities [37,28,53]. Valvular thickening leading to insufficient cardiac activity accounts for most of the observed cardiovascular effects [54,53].
General findings upon examination are fatigue, cardiac murmur, cardiomyopathy, and aortic or mitral regurgitation [53]. A recent report evaluating the cause of death for 129 deceased MPS II patients showed that airway involvement was the primary cause of death (65%) followed by cardiac involvement (16%) [55]. These rates do not take into consideration deaths in which cardiac involvement is listed as a secondary cause of death suggesting that that actual role cardiac disease plays in patient mortality rates is even greater than reported [55,56].
1.3.3 Gastrointestinal tract
Gastrointestinal involvement often leads to discomfort and a decreased quality of life. The protuberance of the abdomen is often one of the first noticeable symptoms for MPS II [57] (See supplemental data). Progressive and degenerative hepatosplenomegaly is seen due to excessive storage of GAGs. Hepatosplenomegaly causes increased intra-abdominal pressure, leading to inguinal and umbilical hernias (Fig. 3). Patients are often plagued by chromic watery stool that does not appear to be associated with malabsorption. In older patients, loss of bowel movement and loss of muscle strength can lead to constipation.
1.3.4 Skeletal, joint and muscle disease
Patients often present with coarse facial features as one of the first recognizable symptoms. Skeletal involvement typically occurs earlier in the more severe form and is characterized by dysostosis multiplex [58]. Short stature, present in most types of MPS, is thought to be a result of the failure of endochondral ossification in growth plates which are secondary to GAG storage. The delay in ossification centers results in many of the characteristic features associated with the skeletal manifestations of this disease including thoracic kyphosis, scoliosis, and odontoid hypoplasia, with the latter leading to atlantoaxial instability [59,60].
Inflammatory events affect chondrocytes, osteoblasts, and their extracellular matrix; often resulting in severe arthroplasty. Although there is GAG storage in and potentially around all joints, the hip is particularly susceptible [23]. Hip inflammation and erosive damage are painful and significantly decrease mobility and quality of life. Decreased mobility exacerbates problems seen in other organs.
Myopathy caused by GAG storage in muscle cells and around tendons leads to muscular dysplasia in MPS II patients. The destruction of the articular and epiphyseal cartilage layers of deformed bones leads to rigidity of joints and increased pain [61, 58]. These effects culminate in restriction and contraction of the hips, knees, toes, shoulders, wrists, and elbows (see supplemental data). In the spine, the skeletal and joint manifestations result in ovoid contour and the humpback characteristic of this disorder (see supplemental data). This results in sagittal deformity and instability, axial deformity and instability, and spinal cord and root compression. As the disease progresses, these factors reduce mobility. Cervical myelopathy often leads to bowel and bladder dysfunction, decreased endurance and eventually quadriparesis. If the compression becomes substantial, these orthopedic deformities can result in death [37].
The skeletal manifestations also affect hand motion. Claw hand deformities (Fig. 5) develop due to joint stiffness and contractures. The inability to hold and grip leads to a significant loss of mobility and thus a loss of independence. This is in part caused by ossification delay. GAG accumulation in and around tendons and nerve sheaths leads to carpal tunnel syndrome which causes further pain [25].
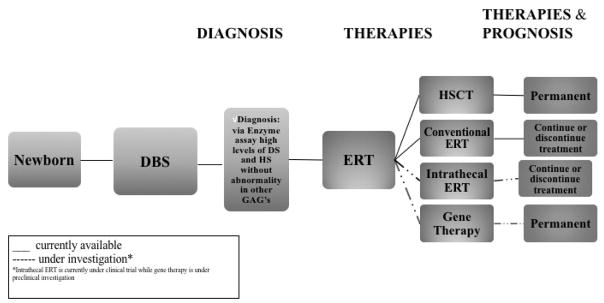
Figure 5.
Progression of disease from diagnosis via DBS to therapies both currently available and under investigation.
1.4. Diagnosis
1.4.1. Clinical
The initial signs and symptoms of MPS II are typically physical rather than psychological or developmental. Coarse facial features are often the first and most indicative appearance of MPS and occur in both phenotypes of MPS II, to varying degrees of severity. Other early clinical manifestations include bone deformity, abnormal gait, and difficulty of joint movement. The average age at onset of symptoms in untreated children has been reported as 1.5 years old for severe phenotype and 4.3 years old for attenuated phenotype [62]. Some patients with an attenuated form of MPS II might not show symptoms until later in childhood or even early adolescence. Patients with attenuated MPS II are most frequently diagnosed between the ages of 4 and 8 [37] Frequent ear nose and throat infections as well as snoring, difficulty sleeping, and hearing loss are also often present from early onset [63].
Some patients might present early cognitive dysfunction including difficulty sleeping, hyperactivity, pervasive chewing, seizures, and inappropriate bowel and bladder training [38]. Often these inappropriate behaviors become more identifiable after the initial diagnosis. Due to the heterogeneity of signs and symptoms, more than 60% of patients are seen by at least three physicians to obtain an accurate diagnosis [64].
1.4.2. Biochemical and Molecular findings
The gold standard in diagnosis of MPS II is the measurement of IDS [4]. Enzyme activity can be measured in a variety of samples: cultured fibroblasts, leukocytes, plasma/serum, dried blood spots (DBS), and chorionic villi for prenatal analysis [65–73]. Wang’s group used a novel approach with the addition of a synthetic substrate for the enzyme to a buffer rehydrated with 2 disks from a DBS. The enzyme-generated product was then measured by tandem mass spectrometry (MS/MS) [68,74]. To exclude multiple sulfatase deficiency, other lysosomal enzymes should be measured [75,76].
Although there is no clear correlation between residual levels of enzyme and disease severity, typically patients with the severe form have very low or undetectable levels of IDS [77–81].
Correlations between genotype-phenotype are challenging due to variation between patients and a large number of mutations [87,88].
1.5. GAGs as a biomarker
Urinary GAGs have been used as biomarkers for several forms of MPS and as a surrogate marker for IDS activity in a clinical trial [9, 89]. Oguma et al. first developed a method to assay specific GAGs including DS, HS, and KS using liquid chromatography tandem mass spectrometry (LC/MS/MS) [90, 94]. Screening of MPS positive patients using LC/MS/MS showed significant elevation of plasma HS and DS in MPS I, II, III and IV patients as opposed to the controls [91]. Auray-Blais et al. developed DS and HS quantification in urine by ultra-pressure liquid chromatography (UPLC) tandem mass spectrometry (MS/MS) post methanolysis [92].
Measurement of urinary GAGs can be limited due to small changes in levels over time, variations in anthropometry, and renal status [82,83,80,84]. The exact correlation between urinary GAG and clinical severity or therapeutic efficacy remains unclear [77–80] and sometimes attenuated forms, and less severe disease cannot be detected resulting in false negatives [ 81,77,86,84,].
Shimada et al. have also established a GAG assay by high-throughput mass spectrometry (HT/MS/MS) with RapidFire system (Agilent Technologies) allowing discrimination of phenotypes and measurement of treatment efficacy in ERT and HSCT treated patients [93].
1.6 Other Biomarkers
Heparin cofactor II thrombin complex (HCII-T) is elevated in MPS II and decreases post-ERT treatment [94,80,95]. Secondary elevation of keratan sulfate (KS) has been reported in MPS II suggesting its use as a biomarker [96,97,98,93,92]. Alternative biomarkers including pro-inflammatory cytokines and the ratio of dermatan to chondroitin sulfate [99,100, 101] are under investigation.
2.7 Incidence
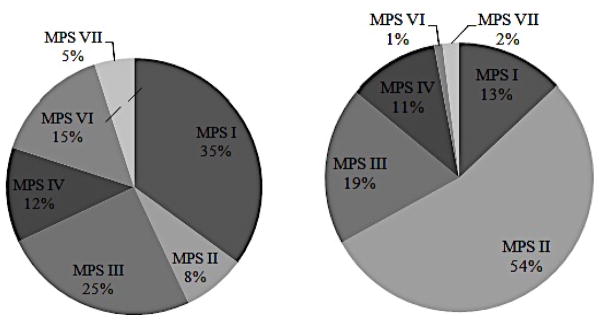
Globally, MPS II occurs in 1: 100,000 to 1: 170,000 male births. MPS II is the most prevalent MPS in East Asian regions, accounting for 54% of MPS patients in Japan (Fig 4), 52% in Taiwan [102], 50% in South Korea [103], and 47.4% in Eastern China [104]. Incidence is lower in European regions [105], examples include accounting for only 7.6% of diagnosed MPS patients in the Netherlands between 1970 and 1996 [106], and 7.6 % in Portugal between 1982 and 2001 [107] (Fig 4).
Figure 4.
Incidence of MPS II in Japan (left) and incidence of MPS in Portugal (right) (Reused from [161] permitted by the Japanese MPS Society)
2. Treatment
2.1. Anti-inflammatory therapy
Several treatments are available that target the consequences of IDS deficiency without correcting the enzyme deficiency directly. Osteoarthritis is very common in MPS II patients. It results from the accumulated GAGs in bone, cartilage, the extracellular matrix (ECM) and inducing proinflammatory factors that stimulate cartilage degradation [108]. Suppression of metabolic inflammation with anti-inflammatory agents can help patients manage this discomfort, thereby improving their quality of life [109]. Pentosan polysulfate (PPS), an anti-inflammatory drug approved by FDA for interstitial cystitis, is currently under investigation. The exact mechanism by which PPS decreases GAG levels and stimulates chondrocyte formation remains under investigation [111].
2.2. ERT (conventional and intrathecal)
Conventional ERT for MPS II is administered by weekly intravenous (IV) infusions of recombinant IDS. A review of the efficacy of IV-ERT on patients, who were given at least one year of ERT, were between the ages of 2–24 years old, and were classified as severely affected showed that 50 out of 66 patients experienced at least one type of somatic improvement including reduction in the frequency of respiratory infections, a reduction in the coarseness of facial features, and/or an improved range of motion in joints [112]. These somatic improvements result in a better quality of life for some patients [29, 113]. However, conventional ERT does not alter the course of neurological decline [37,33, 112,114].
Conventional ERT has been approved in many countries, including the United States, Canada, EU, and Japan, as a treatment option for patients who have a confirmed diagnosis of MPS II. Treatment before six years old provides the positive effects of somatic improvement [28,115]. This method of treatment has been available since 2006, although guidelines for use vary between nations. In Australia, for example, only patients with an attenuated phenotype are eligible to receive ERT whereas in the UK all patients with MPS II are eligible to receive ERT regardless of phenotype [113]. In Latin America, there are approved guidelines stating that ERT should be offered to all patients older than five years of age with an attenuated phenotype, despite the current data suggesting that ERT is more effective when started earlier [61]. When The Hunter Syndrome European Expert Council suggests that all MPS II patients are eligible for ERT unless patients are at a disease terminal stage. ERT should be continued unless ineffective after 6 months of clinical observation [40].
When Elaprase® is denied to patients with the cognitive decline, it is primarily because recombinant enzyme does not cross the blood-brain barrier (BBB) when administered intravenously and consequently provides a little impact on cognitive impairment and retinal degeneration.
Intrathecal injections (IT) of enzyme were proposed to overcome this barrier [117]. Infusion of IT-ERT was administered directly into the cerebrospinal fluid (CSF) of patients using an indwelling intrathecal drug delivery device (IDDD). In 16 males with neurodegeneration, patients were randomly divided into four treatment groups to receive monthly infusions (0, 1, 10, or 39 mg idursulfase-IT) (clinical trial No: NCT00920647) [118]. IT-ERT appeared to slow the progression of cognitive decline. Neurodegeneration did continue for all patients, but progression was slower and less severe in treated patients. However, due to safety concerns, only older patients (average age 8.2 with neurological impairment already present) were included in the study, limiting interpretation of the results. GAG buildup in CSF was reduced by 80–90%, but more than 80% of the patients showed serious adverse effects related to the IDDD [117]. The difficulties experienced by patients as a result of IDDD malfunction fell into two main categories; device breakage and catheter migration from the spinal canal. These severe side effects were unrelated to use of enzyme that was well tolerated [117]. As it is impossible to reverse neuronal decline, younger patients may benefit more from IT- ERT before clinical signs appear. A clinical trial for younger patients (3 to 18 years old) who tolerated at least 4 months of IV-ERT previously is in progress to test the effectiveness of 12 monthly doses of IT- ERT on neurodevelopmental status (Clinical Trail No. NCT02055118 and NCT02262338). It is not yet clear whether IT-ERT will be an appropriate strategy to treat CNS involvement in patients with MPS II.
2.3. Hematopoietic stem cell transplantation (HSCT)
In hematopoietic stem cell transplantation (HSCT) healthy donor cells are obtained from bone marrow (BM), cord blood (CB), or peripheral blood stem cells (PBSC), and transplanted into patients to provide cross correction of enzyme in deficient tissues [119–123, 138]. The first patient with MPS II to undergo treatment with HSCT was treated in 1982. Before 2000, HSCT had high mortality rates due to poor donor selection, rigorous conditioning and high risk of, graft-versus-host disease (GVHD) [124–128, 31,35,32].. Nevertheless, improvements in donor-selection and regimens have decreased mortality rates of HSCT for MPS to less than 5% [130].
The efficacy of HSCT on visceral organs is well documented. These effects can include normalization of hepatosplenomegaly, improvement of the thickening of the aortic valve and an increase in elasticity of the joints allowing for increased independence and mobility for the patient. HSCT has shown better improvement in the quality of life measured by activity of daily living (ADL) when compared to conventional ERT [132,36,32,123]. Knowledge of the effects of HSCT on bone growth is somewhat limited. A comparative study, which examined the difference of HSCT, conventional ERT and combined ERT and HSCT on long bone growth found that HSCT and ERT were equally effective in restoring growth of MPS II patients [37]. Although this study included some patients between the ages of 4 and 12, it is expected that HSCT will have a greater efficacy if given before the age of 2. It would be prudent to conduct a similar study with younger patients, if newborn screening allows younger patients to be identified. HSCT should be a one-time procedure, and consequently cost considerably less than ERT.
Recently it has been found that donor cells cross the blood-brain barrier (BBB) [131]. Tanaka et al. demonstrated that when HSCT is carried out before developmental delay becomes apparent, it is effective in certain aspects of CNS manifestation as measured by both intellectual and imaging analyses. They also found that speech deterioration was significantly less severe in patients who underwent HSCT versus the untreated group. While other studies indicated that HSCT does not have a clear impact on neuropsychological outcomes, the positive comparative results seen in this study indicate that HSCT could have some effect on CNS manifestations if conducted at early disease stages, and HSCT is likely to have similar or superior effects on somatic symptoms when compared to ERT.
While the strong preconditioning regimen prior to HSCT causes severe side effects including increased susceptibility to infection, particularly in immunocompromised patients, a recent murine study suggests that the establishment of a milder conditioning regimen could be as effective while limiting the risk of side effects for the patients. In this study, MPS II mice were pre-treated with anti-c kit antibody 2 (ACK2) followed by low lose irradiation. 59% donor chimerism was achieved 16 weeks after transplantation in the peripheral blood of the mice [135]. This study did not show any effect of the treatment on CNS symptoms; however, it did suggest a potential new pre-conditioning treatment for patients with attenuated MPS II who are unable to tolerate the harsh preconditioning treatments that are currently necessary before HSCT. With the decreased risk of HSCT for patients, comparative studies involving both severe and attenuated patients, should be conducted. This is especially important for patients who can be treated before clinical symptoms of MPS II are apparent.
2.4. Gene Therapy
Since MPS II is caused by a genetic defect in only one gene, the IDS gene, gene therapy is an emerging treatment that could potentially offer a permanent solution for patients. Gene therapy aims to deliver the defective gene to host cells by a specific vector; ex vivo, IV, IT, or intracisternal injections. Since treatment of CNS manifestations remains unsolved for current treatment, many strategies of gene therapy are currently being investigated on the use of CNS-targeted vectors to decrease CNS manifestations of the disease [136,137]. Motas et al. used adeno-associated virus 9 (AAV9-Ids) delivery injected directly to the CSF through intracisternal injections in 2-month-old MPS II mice [138]. IDS activity was significantly increased throughout the CNS with full reversal of CNS and somatic pathology post-treatment. The improvement was also seen in behavioral deficits and survival rates [138].
Another study investigated the use of AAV-9 by CSF injections in MPS II mice and found a dose-dependent resolution of brain storage lesions and decreased GAG storage in the visceral organs. Behavioral testing showed some improvement although the limited effect was seen in fear conditioning and attention span [139].. Mice in this study were treated when they were between 2 and 3-months old when clinical symptoms had developed. The purpose of this later treatment was to determine if neurological and somatic manifestations could be reversed. Although reversal of CNS pathology is less likely in humans, delivery of AAV vectors directly into the CSF is a potential treatment option to prevent further CNS pathology for MPS II patients [139].
Lentiviral vectors in ex vivo gene therapy have been used as a clinical option for several genetic diseases [140–143] and have been shown to be effective for correcting some aspects of neuronal defects in mouse models of MPS I and IIIA [144,140,145]. Lentiviral ex vivo HSC gene therapy improves biochemical abnormalities in tissues, rescues autophagic flux retardation, reduces neurofunctional impairments in the CNS, and decreases deterioration of the neuronal function of MPS II mice [140]. While there are risks, this therapy shows stronger efficacy against neurological defects in MPS II than other treatment regimens [149].
In the future, genome editing may be of value for correcting IDS deficiency in MPS II. Although integration efficiency of this technique is often low, this may be sufficient for MPS II where expression of even very low levels of IDS will lead to a dramatic clinical improvement.
In summary, gene therapy is expected to be superior to both ERT and HSCT due to greater efficacy in the treatment of CNS symptoms. Gene therapy would be a potential one-time treatment offering greater potential than either conventional or intrathecal ERT and may also avoid some of the most serious complications of HSCT. More pre-clinical studies and clinical trials are needed to elucidate safety and long-term effects of this treatment approach.
2.5 Substrate reduction therapy
Another treatment option is substrate reduction therapy (SRT). This methodology aims to reduce GAG synthesis which allows for correction of the imbalance between the formation and breakdown of GAGs [151–153]. Genistein (4′ 5,7 trihydroxy isoflavone) is a soy isoflavone which has been investigated in the context of MPS II as capable of decreasing the expression of genes coding for one or more enzymes involved in GAG synthesis [152–157].
Genistein was shown first to reduce GAG accumulation in the fibroblasts of MPS II patients [152]. Experimental data on the effects of Genistein in an MPS II mouse model showed decreased storage of GAGs in several peripheral tissues with some signs of reduction in brain storage [153]. The complete mechanism for the observed effects was originally thought to be due to the inhibition of the epithelial growth factor (EGF) receptor (EGFR or ErB1), which inhibits trypsin specific protein kinase activity. EGF is required for GAG synthesis so without this the synthesis is significantly decreased [152–154].
Recent studies suggest that Genistein modulates cell cycle in addition to modifying GAG metabolism. Its effect on cell proliferation was similar in fibroblasts from MPS II patients and healthy individuals, although the MPS II fibroblasts had significantly higher proportion of cells in G0/G1 [157].
Previous data has suggested that high does genistein is safe in patients [155]. Genistein was also associated with an improvement in connective tissue elasticity and range of motion in joints of MPS II patients [154]. There is not yet conclusive data to indicate that this treatment can cross the BBB sufficiently to improve neurological symptoms. However, it was shown to cross the blood-brain barrier in an MPS II mice model, with histochemical evidence of GAG reduction at the choroid plexus of the ventricular region [153].
3.6 Surgical interventions, palliative care
The two currently approved treatments for MPS II (ERT and HSCT) have limited efficacy. Therefore, MPS II patients need supportive treatment to manage symptoms. It is necessary for each patient to have a team of specialists who can treat the different symptoms, particularly in regards to the CNS and skeletal manifestations. A comprehensive assessment of the patient’s condition should be completed at least once a year. Continuous follow-up of disease progression should improve both physical and psychological well-being of the patient and the patient’s caregiver.
Specialists must address the orthopedic problems that affect the patient. Orthopedic problems affect patients of both phenotypes and can dramatically reduce the patient’s independence and quality of life. Many of these secondary problems result from the inflammatory responses previously described. The hip joints are particularly susceptible to erosive dysplasia which can render a patient wheelchair bound because walking causes severe pain. The characteristic “claw hand” deformity often seen in patients results from the metaphyseal deformities and thickened joint capsules with inflammation causing carpal tunnel syndrome. These symptoms should be managed with steroids, physical therapy, and surgical release of the transverse carpal ligament if symptoms continue to worsen.
Cardiac problems must be managed by regular consultation with a cardiac specialist. Patients with MPS II often require valve replacement due to valve regurgitation or stenosis [54]. Regular echocardiograms should be given to MPS II patients to monitor for cardiac arrhythmia due to cardiomyopathy. Some success has been seen in physical therapy slowing the decline of exercise-induced endurance in patients.
Respiratory problems in patients with MPS II can lead to upper airway complications (Fig. 2). Chronic upper respiratory tract infections from very early childhood and increased mucosal secretion make more complications for the MPS II patient. The difficulty of breathing leads to sleep apnea which must be treated in severe cases with supplemental oxygen to avoid hypercapnia. Children will often have tonsillectomy and removal of the adenoids to decrease airway obstruction [158]. Recurrent ear infections often lead to infection of the middle ear and subsequent hearing loss. Many patients, therefore, require ear tube placement which will add an otolaryngologist to their team of specialists.
The mechanism that causes the watery stool in many patients with MPS II is not fully understood [9,69]. This diarrhea can be chronic and/or episodic and severely impacts the quality of life for patients and caretakers. Anti-motility drugs as well-planned diet control should be used to remedy this. Abdominal and inguinal hernias, classic manifestations seen in most MPS types, including MPS II, are fixed with surgery although this surgery is a temporary solution and recurrence typically occurs [37].
3. Activity of daily living
Learning disability and psychological well-being of patients must be closely and carefully monitored. It is critical that patients, particularly patients with a severe phenotype, receive a stimulating and enriching environment at an early age, as early enrichment helps with skill retention and delay of the disease progression. For patients with the attenuated phenotype who do not have involvement of the CNS, physical difficulties in school result from a lack of stamina. Pain when sitting and deafness cause learning in a traditional school environment to be very challenging.
Recent development of the MPS II specific questionnaire to measure the activity of daily living (ADL) as described by both the patient and their caregiver(s) has made significant progress in understanding the psychology of MPS II patients (37,64,32]. Attenuated patients, in particular, tend to suffer from depression and withdraw with the onset of adolescence. Caretakers report difficulty in some MPS II patients with hyperactivity which can be controlled via behavioral therapy and classroom accommodations. The educational personnel involved with the MPS II patients should be trained in the manifestations of this disorder so that they are aware of behaviors that are expected. Therapy for the patient can also help with the depression and withdrawal that result from the disorder. Psychological support should also be given to the families of the patient from the onset of diagnosis.
4. Conclusions
MPS II is typically detected in young children once they begin displaying characteristic somatic symptoms including course facial features, inguinal and umbilical hernias, delayed bone growth and missed milestones, both physical and mental. While physical abnormalities are typically the first sign or symptoms detected, over half of the cases with MPS II have substantial CNS involvement leading to progressive mental retardation. Current therapies include both conventional ERT as well as HSCT. Conventional ERT and HSCT improve somatic symptoms as well as ADL. IDS from ERT does not cross the BBB so does not affect CNS symptoms while HSCT has limited effect on CNS symptoms and no effect if performed after cognitive decline has begun. Both therapies were less effective against skeletal symptoms if performed after symptoms were detected.
Intrathecal ERT, currently under investigation, is expected to have a greater effect on the CNS involvement of the disease, but recent studies showed severe side effects occur due to the IDDD device. The breadth and efficacy of the initial data are also limited, as safety concerns allowed only older patients to be included in the first clinical trial. Safety of intrathecal enzyme delivery for MPS II and other conditions needs to be improved substantially before this technique can be considered as a treatment.
Gene therapy shows great promise as a possible one-time treatment to solve CNS, skeletal and somatic manifestations of the disease. The best route of administration and most effective vectors to minimize side effects are currently under investigation.
Early treatment is critical for any of these options as all treatments are less effective as the disease progresses. Figure 5 shows the scheme from diagnosis via DBS through treatment. Both current treatment options and those under investigation are shown.
As no therapy currently available completely corrects disease manifestations, orthopedic surgical interventions and supportive treatments are needed. Surgical interventions predominately address mobility, airway and cardiac malfunctions.
The team of specialists necessary for treatment of this disease includes members in the fields of orthopedics, anesthesiology, gastroenterology, genetics, auditory function, orthodontic specialist, ENT specialist, neurologist, speech therapist, ophthalmologist, pulmonologist, and cardiologists. Geneticists, in conjunction with the patient’s medical team offer guidance in treatment options and provide genetic counseling for patients and their families. Psychologists can provide support for patients and families.
Regardless of what treatment option is chosen, the key to a successful outcome is early treatment which can only be provided if early diagnosis is made. Establishment of affordable and efficient newborn screenings will likely lead to early diagnosis and early treatment [18, 74, 159].
5. Expert Opinion
MPS II is an X-linked disease caused by enzyme deficiency due to mutations in the IDS gene. Treatments currently available include ERT and HSCT coupled with surgical intervention and palliative care. However, both treatments are limited by age of start and disease status (e.g. neurological and heart impairment) at the time of treatment. ERT does not provide continuous relief from symptoms, and must be administered at intervals throughout the patient’s life span. This continuous administration can cause both financial burden and negative impact on the quality of life for patients and caregivers. Furthermore, if immune reactions such as antibody formation occur during treatment, the efficacy will be compromised, and severe side effects can occur. Intrathecal ERT under clinical trial involves intrathecal injections of the deficient enzyme directly into patient’s cerebrospinal fluid using an IDD device. While this method has been shown to improve some CNS symptoms, the indwelling device needs to be improved for patient safety. As with conventional ERT, IT-ERT will be also required continuous administration throughout a patient’s life.
Gene therapy is scheduled for clinical trial and seems to have the most promise as an effective and permeant solution if it proves to be as safe and effective as seen in animal models. The recent evidence from animal trials suggests that it is possible to reverse CNS and somatic pathology from MPS II through intrascisternal injection. If this technique can be performed safely, it appears to have the greatest potential for treatment of CNS symptoms of MPS II. As treatment for CNS manifestations remains the most elusive task for MPS II research, this potential cannot be understated. Gene therapy offers the potential for a greater quality of life for both caretaker and patient as this method is self-sustaining and does not require indefinite applications. Research into gene therapy also offers potential financial incentives for its use in patient treatment. ERT is known to be an incredibly expensive treatment, and expense that follows the patient through adulthood. In many cases, the high cost of ERT prevents patients from seeking treatment. Insurance companies and governmental programs are hesitant to pay in many instances due to the high per patient yearly cost.
It is critical to note that beneficial effects of gene therapy have not yet been elucidated in human patients. While mouse models have shown dramatic improvement, reduced risk when compared to other treatments, and complete reversal of symptoms in many cases, it has not been established whether this will be the case in humans. It is also necessary for further longitudinal studies to examine the safety and long-term effects of gene therapy. As this treatment option is in its infancy, no studies, have yet proven the safety of this particular treatment. While the potential for gene therapy is great, it should also be understood that is likely to have the greatest impact in reliving or preventing the symptoms in very young patients. Gene therapy is unlikely to completely reverse damage already caused in older children and adult patients. Therefore, this therapy must be investigated in conjunction with continued efforts to establish affordable and effective newborn screening to allow for the earliest diagnosis and treatment.
Article highlights.
MPS II is an X-linked recessive disorder manifesting in multi-systemic manifestations present in the CNS, skeletal, and somatic systems.
Current treatments include conventional ERT and HSCT coupled with palliative care.
Current treatments are limited in their efficacy on CNS impairment which decreases a patient’s quality of life.
Intrathecal ERT is currently under clinical investigation and has shown evidence of some efficacy in the CNS although serious side effects related to the indwelling device are reported.
Gene therapy is expected to provide self-sustaining IDS levels with an evidence of reversal of disease manifestations in mouse models
Any treatment proposed will increase in efficacy if administered to patients before clinical symptoms of the disease are present, suggesting importance of newborn screening
Acknowledgments
Funding
This work was supported by the Japanese MPS Society. R Mason and S Tomatsu were supported by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of NIH under grant number P30GM114736. S Tomatsu was supported by National Institutes of Health grant 1R01HD065767. F Kubaski was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico, from Brazil (CNPq).
Footnotes
Declaration of Interest
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
References
Papers of special note have been highlighted as either of interest (*) or of considerable interest (**) to readers.
- 1.Hunter C. A rare disease in two brothers. Proc R Soc Med. 1917;10:104–106. doi: 10.1177/003591571701001833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brante G. Gargoylism–A mucopolysaccharidosis. Scandinavian journal of clinical and laboratory investigation. 1952;4(1):43–46. doi: 10.3109/00365515209060631. [DOI] [PubMed] [Google Scholar]
- 3.Teller WM, Burke EC, Rosevear JW, et al. Urinary excretion of acid mucopolysaccharides in normal children and patients with gargoylism. The Journal of Laboratory and Clinical Medicine. 1962;59(1):95–101. [PubMed] [Google Scholar]
- 4.Neufeld EF, Fratantoni JC. Inborn errors of mucopolysaccharide metabolism. Science. 1970;169(3941):141–146. doi: 10.1126/science.169.3941.141. [DOI] [PubMed] [Google Scholar]
- 5.Bach G, Eisenberg F, Jr, Cantz M, et al. The defect in the Hunter Syndrome: deficiency of sulfoiduronate sulfatase. Proceedings of the National Academy of Sciences. 1973;70(7):2134–2138. doi: 10.1073/pnas.70.7.2134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hugh-Jones K, Hobs JR, Chambers D, et al. Bone marrow transplantation in mucopolysaccharidoses. In: Barranger JA, Brady RO, editors. Molecular basis of lysosomal storage disorders. Orlando: Academic Press Inc; 1984. pp. 411–425. [Google Scholar]
- 7.Vellodi A, Young E, Cooper A, et al. Long-term follow-up following bone marrow transplantation for Hunter disease. JIMD. 1999;22:638–648. doi: 10.1023/a:1005525931994. [DOI] [PubMed] [Google Scholar]
- 8.Wilson PJ, Morris CP, Anson DS, et al. Hunter Syndrome: isolation of an iduronate-2-sulfatase cDNA clone and analysis of patient DNA. Proceedings of the National Academy of Sciences. 1990;87(21):8531–8535. doi: 10.1073/pnas.87.21.8531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muenzer J, Wraith JE, Beck M, et al. A phase II/III clinical study of enzyme replacement therapy with idursulfase in mucopolysaccharidosis II (Hunter Syndrome) Genet Med. 2006;8:465–73. doi: 10.1097/01.gim.0000232477.37660.fb. [DOI] [PubMed] [Google Scholar]
- 10.Clarke JT, Willard HF, Teshima I, et al. Hunter disease (mucopolysaccharidosis type II) in a karyotipically normal girl. Clin Genet. 1990;37:355–62. doi: 10.1111/j.1399-0004.1990.tb03519.x. [DOI] [PubMed] [Google Scholar]
- 11.Clarke JT, Wilson PJ, Morris CP, et al. Characterization of a deletion at Xq27–q28 associated with unbalanced inactivation of the nonmutant X chromosome. American journal of human genetics. 1992;51(2):316–22. [PMC free article] [PubMed] [Google Scholar]
- 12.Neufeld EF, Liebaers I, Epstein CJ, et al. The Hunter Syndrome in females: is there an autosomal recessive form of iduronate sulfatase deficiency? American journal of human genetics. 1977;29(5):455. [PMC free article] [PubMed] [Google Scholar]
- 13.Winchester B, Young E, Geddes S, et al. Female twin with Hunter disease due to nonrandom inactivation of the of the X-chromosome: A consequence of twinning. Am J Med Genet. 1992;44:834–838. doi: 10.1002/ajmg.1320440625. [DOI] [PubMed] [Google Scholar]
- 14.Sukegawa K, Matsuzaki T, Fukuda S, et al. Brother/sister siblings affected with Hunter disease: evidence for skewed X chromosome inactivation. Clinical genetics. 1998;53(2):96–101. doi: 10.1111/j.1399-0004.1998.tb02654.x. [DOI] [PubMed] [Google Scholar]
- 15.Sukegawa K, Song XQ, Masumo M, et al. Hunter disease in a girl caused by R468Q mutation in the iduronate-2-sulfatase gene and skewed inactivation of the X chromosome carrying the normal allele. Hum Mutat. 1997;10:361–7. doi: 10.1002/(SICI)1098-1004(1997)10:5<361::AID-HUMU5>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 16.Martin R, Beck M, Eng C, et al. Recognition and diagnosis of Mucopolysaccharidosis II (Hunter Syndrome) Pediatrics. 2008;121:377–86. doi: 10.1542/peds.2007-1350. [DOI] [PubMed] [Google Scholar]
- 17.Tuschl K, Gal A, Paschke E, et al. Mucopolysaccharidosis type II in females: case report and review of literature. Pediatric neurology. 2005;32(24):270–272. doi: 10.1016/j.pediatrneurol.2004.10.009. [DOI] [PubMed] [Google Scholar]
- 18.Kloska A, Jakobkiewicz-Banecka, Tylki-Szymanska A, et al. Female Hunter syndrome caused by a single mutation and familial XCl skewing: Implications for other X-linked disorders. Clinical Genetics. 2011;80(5):459–465. doi: 10.1111/j.1399-0004.2010.01574.x. [DOI] [PubMed] [Google Scholar]
- 19*.Tomatsu S, Fujii T, Fukushi M, et al. Newborn screening and diagnosis of mucopolysaccharidoses. Mol Genet Metab. 2013;110:45–53. doi: 10.1016/j.ymgme.2013.06.007. Discusses the application and necessity of newborn screening and how cutoff values might be established for the three enzymes. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cudry S, Tigaud I, Froissart R, et al. MPS II in females: molecular basis of two different cases. Journal of medical genetics. 2000;37(10):e29–e29. doi: 10.1136/jmg.37.10.e29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Broadhead DM, Kirk JM, Burt AJ, et al. Full expression of Hunter’s disease in a female with an X chromosome deletion leading to non-random inactivation. Clin Genet. 1986;30:392–8. doi: 10.1111/j.1399-0004.1986.tb01896.x. [DOI] [PubMed] [Google Scholar]
- 22.Xu H, Ren D. Lysosomal physiology. Annu Rev Physiol. 2015;77:57–80. doi: 10.1146/annurev-physiol-021014-071649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wrenshall LE, Stevens RB, Cerra FB, et al. Modulation of macrophage and B cell function by glycosaminoglycans. Leukoc Biol. 1999;66:391–400. doi: 10.1002/jlb.66.3.391. [DOI] [PubMed] [Google Scholar]
- 24.Li JP, Vlodavsky I. Heparin, heparan sulfate and heparanase in inflammatory reactions. Thromb Haemost. 2009;102:823–8. doi: 10.1160/TH09-02-0091. [DOI] [PubMed] [Google Scholar]
- 25.Constantopoulos G, Dekaban AS. Neurochemistry of the mucopolysaccharidoses: brain lipids and lysosomal enzymes in patients with four types of mucopolysaccharidosis and in normal controls. J Neurochem. 1978;30:965–973. doi: 10.1111/j.1471-4159.1978.tb12388.x. [DOI] [PubMed] [Google Scholar]
- 26.Wraith JE. The mucopolysaccharidoses: a clinical review and guide to management. Arch Dis Child. 1995;72(3):263–267. doi: 10.1136/adc.72.3.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Krivit W, Sung JH, Shapiro EG, et al. Microglia-the effector cell for reconstitution of the central-nervous-system following bone-marrow transplantation for lysosomal and peroxisomal storage diseases. Cell. 1995;4:385–92. doi: 10.1177/096368979500400409. [DOI] [PubMed] [Google Scholar]
- 28.Daniele A, Tomanin R, Villani GR, et al. Uptake of recombinant iduronate-2-sulfatase into neuronal and glial cells in vitro. Biochim Biophys Acta. 2002;1588:203–9. doi: 10.1016/s0925-4439(02)00166-7. [DOI] [PubMed] [Google Scholar]
- 29.Muenzer J, Beck M, Giugliani R, et al. Idursulfase treatment of Hunter Syndrome in children younger than 6 years: results from the Hunter Outcome Survey. Genetics in Medicine. 2011;13(2):102–109. doi: 10.1097/GIM.0b013e318206786f. [DOI] [PubMed] [Google Scholar]
- 30.Archer LD, Langford-Smith KJ, Bigger BW, et al. Mucopolysaccharide diseases: a complex interplay between neuroinflammation, microglial activation and adaptive immunity. JIMD. 2014;37:1–12. doi: 10.1007/s10545-013-9613-3. [DOI] [PubMed] [Google Scholar]
- 31.Froissart R, Silva IM, Guffon N, et al. Mucopolysaccharidosis type II–genotype/phenotype aspects. Acta Paediatrica. 2002;91(s439):82–87. doi: 10.1111/j.1651-2227.2002.tb03116.x. [DOI] [PubMed] [Google Scholar]
- 32.Annibali R, Caponi L, Morganti A, et al. Hunter Syndrome (Mucopolysaccharidosis type II), severe phenotype: long term follow-up on patients undergone to hematopoietic stem cell transplantation. Minerva pediatrica. 2013;65(5):487–496. [PubMed] [Google Scholar]
- 33.Tanjuakio J, Suzuki Y, Patel P, et al. Activities of daily living in patients with Hunter Syndrome: impact of enzyme replacement therapy and hematopoietic stem cell transplantation. Molecular genetics and metabolism. 2015;114(2):161–169. doi: 10.1016/j.ymgme.2014.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Holt J, Poe MD, Escolar ML, et al. Early clinical markers of central nervous system involvement in mucopolysaccharidosis type II. The Journal of pediatrics. 2011;159(2):320–326. doi: 10.1016/j.jpeds.2011.03.019. [DOI] [PubMed] [Google Scholar]
- 35.Zafeiriou DI, Batzios SP. Brain and spinal MR imaging findings in mucopolysaccharidoses: a review. American Journal of Neuroradiology. 2013;34(1):5–13. doi: 10.3174/ajnr.A2832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Patel P, Suzuki Y, Maeda M, et al. Growth charts for patients with Hunter Syndrome. Molecular genetics and metabolism reports. 2014;1:5–18. doi: 10.1016/j.ymgmr.2013.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Patel P, Suzuki Y, Tanaka A, et al. Impact of enzyme replacement therapy and hematopoietic stem cell therapy on growth in patients with Hunter Syndrome. Molecular genetics and metabolism reports. 2014;1:184–196. doi: 10.1016/j.ymgmr.2014.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wraith JE, Scarpa M, Beck M, et al. Mucopolysaccharidosis type II (Hunter Syndrome): a clinical review and recommendations for treatment in the era of enzyme replacement therapy. European journal of pediatrics. 2008;167(3):267–277. doi: 10.1007/s00431-007-0635-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Muenzer J, Beck M, Eng CM, et al. Multidisciplinary management of Hunter Syndrome. Pediatrics. 2009;124(6):e1228–e1239. doi: 10.1542/peds.2008-0999. [DOI] [PubMed] [Google Scholar]
- 40.Scarpa M, Zsuzsanna A, Beck M, et al. Mucopolysaccharidosis type II: European recommendations for the diagnosis and multidisciplinary management of a rare disease. Orphanet J Rare Dis. 2011;6:1–18. doi: 10.1186/1750-1172-6-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Peck JE. Hearing loss in Hunter Syndrome– mucopolysaccharidosis II. Ear Hear. 1984;5:243–6. doi: 10.1097/00003446-198407000-00009. [DOI] [PubMed] [Google Scholar]
- 42.Taccone A, Tortori DP, Marzoli A, et al. Mucopolysaccharidosis: thickening of dura matter at the craniocervical junction and other CT/MRI findings. Pediatric Radiology. 1993;23(5):349–352. doi: 10.1007/BF02011954. [DOI] [PubMed] [Google Scholar]
- 43.Vedolin L, Schwartz IV, Komlos M, et al. Brain MRI in mucopolysaccharidosis: Effect of aging and correlation with biochemical findings. Neurology. 2007;69(9):917–924. doi: 10.1212/01.wnl.0000269782.80107.fe. [DOI] [PubMed] [Google Scholar]
- 44.Shinomiya N, Nagayama T, Fujioka, et al. MRI in the mild type of mucopolysaccharidosis II (Hunter syndrome) Neuroradiology. 1996;38(5):483–485. doi: 10.1007/BF00607284. [DOI] [PubMed] [Google Scholar]
- 45*.Tsivgoulis G, Papathanasiou MA, Krogias C, et al. Neuroimaging findings in Hunter disease. Journal of the neurological sciences. 2014;342(1–2):200–201. doi: 10.1016/j.jns.2014.04.029. Confirmatory measures which through MRI can show the extent of the multisystem involvement of MPS II through neuroimaging. [DOI] [PubMed] [Google Scholar]
- 46.Lustmann J, Bimstein E, Yatziv S. Dentigerous cysts and radiolucent lesions of the jaw associated with Hunter’s Syndrome. Journal of oral surgery (American Dental Association: 1965) 1975;33(9):679. [PubMed] [Google Scholar]
- 47.Oussoren E, Brands MMMG, Ruijter GJG, et al. Bone, joint and tooth development in mucopolysaccharidoses: relevance to therapeutic options. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease. 2011;1812(11):1542–1556. doi: 10.1016/j.bbadis.2011.07.013. [DOI] [PubMed] [Google Scholar]
- 48.Gajula P, Ramalingam K, Bhadrashetty D. A rare case of mucopolysaccharidosis: Hunter Syndrome. Journal of Natural Science, Biology and Medicine. 2012;3(1):97–100. doi: 10.4103/0976-9668.95984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Brama I, Gay I, Feinmesser R, et al. Upper airway obstruction in Hunter Syndrome. Int J Pediatr Otorhinolaryngol. 1986;11:229–35. doi: 10.1016/s0165-5876(86)80034-9. [DOI] [PubMed] [Google Scholar]
- 50.Sasaki CT, Ruiz R, Gaito R, Jr, et al. Hunter Syndrome: a study in airway obstruction. Laryngoscope. 1987;97:280–5. [PubMed] [Google Scholar]
- 51.Yoskovitch A, Tewfik TL, Brouillette RT, Schloss MD, Der Kaloustian VM. Acute airway obstruction in Hunter Syndrome Int. J Pediatr Otorhinolaryngol. 1998;44:273–8. doi: 10.1016/s0165-5876(98)00063-9. [DOI] [PubMed] [Google Scholar]
- 52.Leighton SE, Papsin B, Vellodi A, et al. Disordered breathing during sleep in patients with mucopolysaccharidoses. Int J Pediatr Otorhinolaryngol. 2001;58:127–38. doi: 10.1016/s0165-5876(01)00417-7. [DOI] [PubMed] [Google Scholar]
- 53.Gaitini L, Fradis M, Vaida S, et al. Failure to control the airway in a patient with Hunter’s syndrome. J Laryngol Otol. 1998;112:380–2. doi: 10.1017/s0022215100140526. [DOI] [PubMed] [Google Scholar]
- 54.Braulin EA, Harmatz PR, Scarpa M, et al. Cardiac disease in patients with mucopolysaccharidosis. Journal of Inherited Metabolic disease. 2011;34(6):1183–1197. doi: 10.1007/s10545-011-9359-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rigante D, Segni G. Cardiac structural involvement in mucopolysaccharidoses. Cardiology. 2002;98:18–20. doi: 10.1159/000064674. [DOI] [PubMed] [Google Scholar]
- 56.Burton BK, Giugliani R. Diagnosing Hunter Syndrome in pediatric practice: practical considerations and common pitfalls. European journal of pediatrics. 2012;171(4):631–639. doi: 10.1007/s00431-012-1703-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jones SA, Almassy Z, Beck M, et al. Mortality and cause of death in mucopolysaccharidosis type II—a historical review based on data from the Hunter Outcome Survey (HOS) Journal of inherited metabolic disease. 2009;32(4):534–543. doi: 10.1007/s10545-009-1119-7. [DOI] [PubMed] [Google Scholar]
- 58.Elsner B. Ultrastructure of the rectal wall in Hunter’s syndrome. Gastroenterology. 1970;58(6):856–862. [PubMed] [Google Scholar]
- 59.Simonaro CM, D’Angelo M, He X, et al. Mechanism of glycosaminoglycan-mediated bone and joint disease: implications for the mucopolysaccharidoses and other connective tissues diseases. Am J Pathol. 2008;172:112–22. doi: 10.2353/ajpath.2008.070564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Samet JD, Rusinak, Grant T. Case 174: Hunter Syndrome. Radiology. 2011;261(1):321–324. [Google Scholar]
- 61.Giugliani R, Villarreal MLS, Valdez CAA, et al. Guidelines for diagnosis and treatment of Hunter Syndrome for clinicians in Latin America. Genetics and molecular biology. 2014;37(2):315–329. doi: 10.1590/s1415-47572014000300003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kiselyov K, Jennings JJ, Rbaibi Y, et al. Autophagy, mitochondria and cell death in lysosomal storage diseases. Autophagy. 2007;3:259–262. doi: 10.4161/auto.3906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Alcalde-Martin C, Muro-Tudelilla JM, Cancho-Candela R, et al. First experience of enzyme replacement therapy with idrsulfase in Spanish patients with Hunter syndrome under 5 years of age: case observations from the Hunter Outcome Survey (HOS) doi: 10.1016/j.ejmg.2010.07.013. [DOI] [PubMed] [Google Scholar]
- 64.Kamin W. Diagnosis and management of respiratory involvement in Hunter Syndrome. Acta Pædiatrica. 2008;97(s457):57–60. doi: 10.1111/j.1651-2227.2008.00650.x. [DOI] [PubMed] [Google Scholar]
- 65.Guffon N, Heron B, Chabrol B, et al. Diagnosis, quality of life, and treatment of patients with Hunter Syndrome in the French healthcare system: a retrospective observational study. Orphanet journal of rare diseases. 2015;10(1):1. doi: 10.1186/s13023-015-0259-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Voznyi YV, Keulemans JL, van Diggelen OP, et al. A fluorimetric enzyme assay for the diagnosis of MPS II (Hunter disease) JIMD. 2001;24:675–80. doi: 10.1023/a:1012763026526. [DOI] [PubMed] [Google Scholar]
- 67.Civallero G, Michelin K, de Mari J, et al. Twelve different enzyme assays on dried-blood filter paper samples for detection of patients with selected inherited lysosomal storage diseases. Clinica Chimica cta. 2006;372(1):98–102. doi: 10.1016/j.cca.2006.03.029. [DOI] [PubMed] [Google Scholar]
- 68.Dean CJ, Bockmann MR, Hopwood JJ, Brooks, et al. Detection of mucopolysaccharidosis type II by measurement of iduronate-2-sulfatase in dried blood spots and plasma samples. Clin Chem. 2006;52(6):643–649. doi: 10.1373/clinchem.2005.061838. [DOI] [PubMed] [Google Scholar]
- 69.Wang D, Wood T, Sadilek M, et al. Tandem mass spectrometry for the direct assay of enzymes in dried blood spots. Clin Chem. 2007;53:137–40. doi: 10.1373/clinchem.2006.077263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Martin R, Beck M, Eng C, et al. Recognition and diagnosis of mucopolysaccharidosis II (Hunter Syndrome) Pediatrics. 2008;121(2):e377–e386. doi: 10.1542/peds.2007-1350. [DOI] [PubMed] [Google Scholar]
- 71.Sista R, Eckhardt AE, Wang T, et al. Rapid, single-step assay for Hunter Syndrome in dried blood spots using digital microfluidics. Clin Chem Acta. 2011;412:1985–97. doi: 10.1016/j.cca.2011.06.015. [DOI] [PubMed] [Google Scholar]
- 72.Tolun AA, Graham C, Shi Q, et al. A novel fluorimetric enzyme analysis method for Hunter Syndrome using dried blood spots. Mol Genet Metabol. 2012;105:519–21. doi: 10.1016/j.ymgme.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 73.Giugliani R, Brusius-Facchin, de Sounza ACM, et al. Diagnosis and therapy options in mcuopolysaccharidosis II (Hunter Syndrome) Expert Opinion on Orphan drugs. 2016;3:141–50. [Google Scholar]
- 74.Verma J, Thomas DC, Kasper DC, et al. Inherited Metabolic Disorders: Efficacy of Enzyme Assays on Dried Blood Spots for the Diagnosis of Lysosomal Storage Disorders. JIMD. 2016:1–13. doi: 10.1007/8904_2016_548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kumar AB, Masi S, Ghomaschi F, et al. Tandem Mass Spectrometry Has a Larger Analytical Range than Fluorescence Assays of Lysosomal Enzymes: Application to Newborn Screening and Diagnosis of Mucopolysaccharidoses Types II, IVA, and VI. Clinical chemistry. 2015;61(11):1363–1371. doi: 10.1373/clinchem.2015.242560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fratantoni JC, Hall CW, Neufeld EF. The defect in Hurler’s and Hunter’s syndrome: Faulty degradation of mucopolysaccharide. Proc Natl Acad Sci USA. 1968a;60:699–706. doi: 10.1073/pnas.60.2.699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Fratantoni JC, Hall C, Neufeld E. Hurler and Hunter Syndrome: mutual correction of the defect in cultured fibroblasts. Science. 1968;162(3853):570–572. doi: 10.1126/science.162.3853.570. [DOI] [PubMed] [Google Scholar]
- 78.Chuang CK, Lin SP, Chung SF. Diagnostic Screening for Mucopolysaccharidoses by the Dimethylmethylene Blue Method and Two-Dimensional Electrophoresis. Chin Med J (Tai pei ) 2001;64:15–22. [PubMed] [Google Scholar]
- 79.Swiedler S, Beck M, Bajboui M, et al. Threshold effect of urinary glycosaminoglycans and the walk test as indicators of disease progression in a survey of subjects with Mucopolysaccharidosis VI (Maroteaux–Lamy syndrome) American Journal of Medical Genetics Part A. 2005;134(2):144–150. doi: 10.1002/ajmg.a.30579. [DOI] [PubMed] [Google Scholar]
- 80.Schwartz IV, Riberiro MG, Mota JG, et al. A clinical study of 77 patients with mucopolysaccharidosis type II. Acta Paediatrica. 2007;96(s455):63–70. doi: 10.1111/j.1651-2227.2007.00212.x. [DOI] [PubMed] [Google Scholar]
- 81.Clarke LA, Hemmelgarn H, Colobong K, et al. Longitudinal observations of serum heparin cofactor II-thrombin complex in treated Mucopolysaccharidosis I and II patients. Journal of inherited metabolic disease. 2012;35(2):355–362. doi: 10.1007/s10545-011-9369-6. [DOI] [PubMed] [Google Scholar]
- 82.De Jong JG, Hasselman JJ, van Landeghem AA, et al. The spot test is not a reliable screening procedure for mucopolysaccharidoses. Clinical chemistry. 1991;37(4):572–575. [PubMed] [Google Scholar]
- 83.Gallegos-Arreola MP, Machorro-Lazo MV, Flores-Martínez SE, et al. Urinary glycosaminoglycan excretion in healthy subjects and in patients with mucopolysaccharidoses. Archives of medical research. 2000;31(5):505–510. doi: 10.1016/s0188-4409(00)00104-1. [DOI] [PubMed] [Google Scholar]
- 84.Mahalingam K, Janani S, Priya S, et al. Diagnosis of mucopolysaccharidoses: how to avoid false positives and false negatives. The Indian Journal of Pediatrics. 2004;71(1):29–32. doi: 10.1007/BF02725652. [DOI] [PubMed] [Google Scholar]
- 85.Müller-Lutz A, Schleich C, Pentang G, et al. Age-dependency of glycosaminoglycan content in lumbar discs: A 3t gagcEST study. Journal of Magnetic Resonance Imaging. 2015;42(6):1517–1523. doi: 10.1002/jmri.24945. [DOI] [PubMed] [Google Scholar]
- 86.Chih-Kuang C, Shuan-Pei L, Shyue-Jye L, et al. MPS screening methods, the berry spot and acid turbidity tests, cause a high incidence of false-negative results in sanfilippo and morquio syndromes. Journal of clinical laboratory analysis. 2002;16(5):253–258. doi: 10.1002/jcla.10051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mabe P, Valiente A, Soto V, et al. Evaluation of reliability for urine mucopolysaccharidosis screening by dimethylmethylene blue and Berry spot tests. Clinica chimica acta. 2004;345(1):135–140. doi: 10.1016/j.cccn.2004.03.015. [DOI] [PubMed] [Google Scholar]
- 88.Froissart R, Silva IM, Guffon N, et al. Mucopolysaccharidosis type II–genotype/phenotype aspects. Acta Paediatrica. 2002;91(s439):82–87. doi: 10.1111/j.1651-2227.2002.tb03116.x. [DOI] [PubMed] [Google Scholar]
- 89.Giugliani R. Mucopolysacccharidoses: from understanding to treatment, a century of discoveries. Genetics and molecular biology. 2012;35(4):924–931. doi: 10.1590/s1415-47572012000600006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.de Ruijter J, Ru MH, Wagemans T, et al. Heparan sulfate and dermatan sulfate derived disaccharides are sensitive markers for newborn screening for mucopolysaccharidoses type I, II and III. Mol Genet Metab. 2012;107:705–10. doi: 10.1016/j.ymgme.2012.09.024. [DOI] [PubMed] [Google Scholar]
- 91.Oguma T. Analytical method for the determination of disaccharides derived from keratan, heparan, and dermatan sulfates in human serum and plasma by high-performance liquid chromatography/turbo ionspray ionization tandem mass spectrometry. Analytical biochemistry. 2007;368(1):79–86. doi: 10.1016/j.ab.2007.05.016. [DOI] [PubMed] [Google Scholar]
- 92.Tomatsu S, Montano AM, Oguma T, et al. Validation of disaccharide compositions derived from dermatan sulfate and heparan sulfate in mucopolysaccharidoses and mucolipidoses II and III by tandem mass spectrometry. Mol Genet Metab. 2010;99(2):124–131. doi: 10.1016/j.ymgme.2009.10.001. [DOI] [PubMed] [Google Scholar]
- 93.Auray-Blais C, Bhérer P, Gagnon R, et al. Efficient analysis of urinary glycosaminoglycans by LC-MS/MS in mucopolysaccharidoses type I, II and VI. Molecular genetics and metabolism. 2011;102(1):49–56. doi: 10.1016/j.ymgme.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 94.Shimada T, Kelly J, LaMarr WA, et al. Novel heparan sulfate assay by using automated high-throughput mass spectrometry: Application to monitoring and screening for mucopolysaccharidoses. Molecular genetics and metabolism. 2008;113(1):92–99. doi: 10.1016/j.ymgme.2014.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Randall DR, Colobong KE, Hemmelgarn H, et al. Heparin cofactor II–thrombin complex: a biomarker of MPS disease. Molecular genetics and metabolism. 2008;94(4):456–461. doi: 10.1016/j.ymgme.2008.05.001. [DOI] [PubMed] [Google Scholar]
- 96.Pandey SK, Singh A, Dubey AP, et al. Heparin co-factor II Thrombin complex as a biomarker for mucopolysaccharidosis: Indian experience. Indian pediatrics. 2014;51(2):128–130. doi: 10.1007/s13312-014-0344-9. [DOI] [PubMed] [Google Scholar]
- 97.Tomatsu S, Gutierrez MA, Ishimaru T, et al. Heparan sulfate levels in mucopolysaccharidoses and mucolipidoses. Journal of inherited metabolic disease. 2005;28(5):743–757. doi: 10.1007/s10545-005-0069-y. [DOI] [PubMed] [Google Scholar]
- 98.Tomatsu S, Montano AM, Oguma T, et al. Dermatan sulfate and heparin sulfate as a biomarker for mucopolysaccharidosis I. J Inherit, Metab Dis. 2010;33:141–150. doi: 10.1007/s10545-009-9036-3. [DOI] [PubMed] [Google Scholar]
- 99.Oguma T. Analytical method for determination of disaccharides derived from keratan sulfates in human serum and plasma by high-performance liquid chromatography/turbo-ionspray ionization tandem mass spectrometry. Biomedical Chromatography. 2007;21(4):356–362. doi: 10.1002/bmc.760. [DOI] [PubMed] [Google Scholar]
- 100*.Hetmańczyk K, Bednarska-Makaruk M, Kierus K, et al. Monitoring of dipeptidyl peptidase-IV (DPP-IV) activity in patients with mucopolysaccharidoses types I and II on enzyme replacement therapy—Results of a pilot study. Clinical biochemistry. 2016;49(6):458–462. doi: 10.1016/j.clinbiochem.2015.11.011. Recent pilot study examining MPS I and MPS II patients who were treated with ERT over a period of 19 months. This study clearly demonstrates the necessity of early treatment for patients and establishes that DPP-IV activity is not a useful marker of treatment efficacy, which must be further determined in the case of ERT particularly in the case of patients with severe central nervous system disease manifestations. [DOI] [PubMed] [Google Scholar]
- 101*.Lum SH, Jones SA, Ghosh A, et al. Hematopoietic stem cell transplant for the mucopolysaccharidoses. Expert Opinion on Orphan Drugs. 2016;4(4):379–393. Gives a concise description of where HSCT is currently at in terms of efficacy for MPS II. [Google Scholar]
- 102.Parini G, Naia S, Dall’Amico R, et al. Simple spectrophotometric quantification of urinary excretion of glycosaminoglycan sulfates. Clinical chemistry. 1986;32(11):2073–2076. [PubMed] [Google Scholar]
- 103.Lin HY, Lin SP, Chuang CK, et al. Incidence of the mucopolysaccharidoses in Taiwan 1984–2004. Am J Med Genet A. 2009;149A:960–4. doi: 10.1002/ajmg.a.32781. [DOI] [PubMed] [Google Scholar]
- 104.Cho SY, Sohn YB, Jin DK. An overview of Korean patients with mucopolysaccharidosis and collaboration through the Asia Pacific MPS Network. Intractable & rare diseases research. 2014;3(3):79–86. doi: 10.5582/irdr.2014.01013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chen X, Qiu W, Ye J, et al. Demographic characteristics and distribution of lysosomal storage disorder subtypes in Eastern China. Journal of human genetics. 2016;61(4):345–349. doi: 10.1038/jhg.2015.155. [DOI] [PubMed] [Google Scholar]
- 106.Baehner F, Schmiedeskamp C, Krummenauer F, et al. Cumulative incidence rates of the mucopolysaccharidoses in Germany. Journal of inherited metabolic disease. 2005;28(6):1011–1017. doi: 10.1007/s10545-005-0112-z. [DOI] [PubMed] [Google Scholar]
- 107.Poorthuis BJ, Wevers RA, Kleijer WJ, et al. The frequency of lysosomal storage diseases in the Netherlands. Human Genetics. 1999;105(1–2):151–156. doi: 10.1007/s004399900075. [DOI] [PubMed] [Google Scholar]
- 108.Pinto LLC, Vieira TA, Giugliani, et al. Expression of the disease on female carriers of X-linked lysosomal disorders: a brief review. Orphanet journal of rare diseases. 2010;5(1):1. doi: 10.1186/1750-1172-5-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Clarke LA. Pathogenesis of skeletal and connective tissue involvement in the mucopolysaccharidoses: glycosaminoglycan storage is merely the instigator. Rheumatology. 2011:v13–v18. doi: 10.1093/rheumatology/ker395. [DOI] [PubMed] [Google Scholar]
- 110.Polgreen LE, Vehe RK, Rudser K, et al. Elevated TNF-α is associated with pain and physical disability in mucopolysaccharidosis types I, II, and VI. Molecular genetics and metabolism. 2016;117(4):427–430. doi: 10.1016/j.ymgme.2016.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Simonaro CM, Tomatsu S, Sikora T, et al. Pentosan polysulfate: oral versus subcutaneous injection in mucopolysaccharidosis type I dogs. PloS one. 2016;11(4):e0153136. doi: 10.1371/journal.pone.0153136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Frohbergh M, Ge Y, Meng F, et al. Dose Responsive effects of subcutaneous pentosan polysulfate injection in mucopolysaccharidosis Type VI rats and comparison to oral treatment. doi: 10.1371/journal.pone.0100882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Muenzer J, Bodamer O, Burton B, et al. The role of enzyme replacement therapy in severe Hunter Syndrome—an expert panel consensus. European journal of pediatrics. 2012;171(2):181–188. doi: 10.1007/s00431-011-1606-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.da Silva EMK, Strufaldi MW, Andriolo RB, et al. The Cochrane Library. 2011. Enzyme replacement therapy with idursulfase for mucopolysaccharidosis type II (Hunter Syndrome) [DOI] [PubMed] [Google Scholar]
- 115.Parini R, Rigbold M, Tedesco L, et al. Enzymatic replacement therapy for Hunter disease: Up to 9 years’ experience with 17 patients. 2015;3:65–74. doi: 10.1016/j.ymgmr.2015.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Noh H, Lee JI. Current and potential therapeutic strategies for mucopolysaccharidoses. J Clin Pharm Ther. 2014;39:215–24. doi: 10.1111/jcpt.12136. [DOI] [PubMed] [Google Scholar]
- 117.Ekdahl CT, Claasen JH, Bonde S, et al. Inflammation is detrimental for neurogenesis in adult brain. Proc Natl Acad Sci USA. 2003;100:13632–37. doi: 10.1073/pnas.2234031100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118*.Muenzer J, Hendriksz CJ, Fan Z, et al. A phase I/II study of intrathecal idursulfase-IT in children with severe mucopolysaccharidosis II. Genetics in Medicine. 2015;18(1):73–81. doi: 10.1038/gim.2015.36. Gives a summary of the clinical trial for pioneer intrathecal ERT which has the potential for greater efficacy across the blood brain barrier. [DOI] [PubMed] [Google Scholar]
- 119.Public summary of opinion on orphan designation Iduronate-2-sulfatase for the treatment of mucopolysaccharidosis, type II (Hunter Syndrome) n.d Retrieved September 08, 2016, from http://www.ema.europa.eu/docs/en_GB/document_library/Orphan_designation/2010/10/WC500097957.pdf.
- 120.Warkentin PI, Dixon MS, Schafer I, et al. Bone marrow transplantation in Hunter Syndrome: a preliminary report. Birth defects. 1986;22:31–9. [PubMed] [Google Scholar]
- 121.Mullen CA, Thompson JN, Richard LA, et al. Unrelated umbilical cord blood transplantation in infancy for mucopolysaccharidosis type IIB (Hunter Syndrome) complicated by autoimmune hemolytic anemia. Bone marrow transplantation. 2000;25:1093–9. doi: 10.1038/sj.bmt.1702397. [DOI] [PubMed] [Google Scholar]
- 122.Sands MS, Davidson BL. Gene therapy for lysosomal storage diseases. Mol Ther. 2006;13:839–49. doi: 10.1016/j.ymthe.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 123.Prasad VK, Kurtzberg J. Cord blood and bone marrow transplantation in inherited metabolic diseases: scientific basis, current status and future directions. Br J Haematol. 2010;148:356–372. doi: 10.1111/j.1365-2141.2009.07974.x. [DOI] [PubMed] [Google Scholar]
- 124.Kubaski F, Guha A, Suzuki Y, et al. Hematopoietic stem cell transplantation and ERT for Hunter Syndrome. Molecular Genetics and Metabolism. 2016;117(2):S69. doi: 10.1016/j.ymgme.2015.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Bergstrom SK, Quinn JJ, Greenstein R, et al. Long term follow-up of a patient transplanted for Hunter’s disease type IIB: a case report and literature review. Bone Marrow Transplant. 1994;14(4):653–658. [PubMed] [Google Scholar]
- 126.Imaizumi M, Gushi K, Kurobane I, et al. Long-term effects of bone marrow transplantation for inborn errors of metabolism: a study of four patients with lysosomal storage diseases. Acta Paediatrica Japonica. 1994;36:30–6. doi: 10.1111/j.1442-200x.1994.tb03125.x. [DOI] [PubMed] [Google Scholar]
- 127.Tokimasa S, Ohta H, Takizawa S, et al. Umbilical cord-blood transplantations from unrelated donors in patients with inherited metabolic diseases: Single- institute experience. Pediatr Transplant. 2008;12(6):672–676. doi: 10.1111/j.1399-3046.2007.00876.x. [DOI] [PubMed] [Google Scholar]
- 128.Guffon N, Bertrand Y, Forest I, et al. Bone marrow transplantation in children with Hunter Syndrome: outcome after 7 to 17 years. J Pediatric. 2009;154:733–737. doi: 10.1016/j.jpeds.2008.11.041. [DOI] [PubMed] [Google Scholar]
- 129.Tanaka A, Okuyama T, Suzuki Y, et al. Long-term efficacy of hematopoietic stem cell transplantation on brain involvement in patients with mucopolysaccharidosis type II: a nationwide survey in Japan. Molecular genetics and metabolism. 2012;107(3):513–520. doi: 10.1016/j.ymgme.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 130.Kutzberg J. Early HSCT corrects the skeleton in MPS. Blood. 2015;125:1518–19. doi: 10.1182/blood-2014-11-606681. [DOI] [PubMed] [Google Scholar]
- 131.Aldenhoven M, Jones SA, Bonney D, et al. Hematopoietic cell transplantation for mucopolysaccharidosis patients is safe and effective: results after implementation of international guidelines. Bio Blood Bone Marrow Transplant. 2015;21:1106–9. doi: 10.1016/j.bbmt.2015.02.011. [DOI] [PubMed] [Google Scholar]
- 132.Araya K, Sakai N, Mohri I, et al. Localized donor cells in brain of a Hunter I disease patient after cord blood stem cell transplantation. Mol Genet Metab. 2009;98:255–263. doi: 10.1016/j.ymgme.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 133.Ito K, Ochiai T, Suzuki H, et al. The effect of hematopoietic stem cell transplant on papules with ‘pebbly ‘appearance in Hunter’s Syndrome. British Journal of Dermatology. 2004;151(1):207–211. doi: 10.1111/j.1365-2133.2004.05944.x. [DOI] [PubMed] [Google Scholar]
- 134.Belinson S, Rothenberg BM, Chopra R, et al. Future Research Needs for Hematopoietic Stem-Cell Transplantation in the Pediatric Population: Identification of future research needs from comparative effectiveness review No. 48 [Internet] Rockville (MD): Agency for Healthcare Research and Quality (US); 2012. Feb, (Future Research Needs Papers, No. 10.) [PubMed] [Google Scholar]
- 135.Yokoi T, Yokoi K, Akiyama K, et al. Non-Myeloablative preconditioning with ACK2 (anti-c-kit antibody) is efficient in bone marrow transplantation for murine models of mucopolysaccharidosis type II. Molecular Genetics and Metabolism. 2016;119(3):232–238. doi: 10.1016/j.ymgme.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 136*.Yokoi K, Akiyama K, Kaneshiro E, et al. Effect of donor chimerism to reduce the level of glycosaminoglycans following bone marrow transplantation in a murine model of mucopolysaccharidosis type II. Journal of Inherited Metabolic Diseases. 2015;38(2):333–340. doi: 10.1007/s10545-014-9800-x. Discusses the difficulties faced in bone marrow transplant which is critical to overcome to make this treatment most effective and decrease risk. [DOI] [PubMed] [Google Scholar]
- 137.Hong Y, Yu SS, Kim JM, et al. Construction of a high efficiency retroviral vector for gene therapy of Hunter’s Syndrome. J Gene Med. 2003;5:18–29. doi: 10.1002/jgm.316. [DOI] [PubMed] [Google Scholar]
- 138.Jung SC, Park ES, Choi EN, et al. Characterization of a novel mucopolysaccharidosis type II mouse model and recombinant AAV2/8 vector-mediated gene therapy. Mol Cells. 2010;30:13–18. doi: 10.1007/s10059-010-0083-2. [DOI] [PubMed] [Google Scholar]
- 139**.Motas S, Haurigot V, Garcia M, et al. CNS-directed gene therapy for the treatment of neurologic and somatic mucopolysaccharidosis type II (Hunter Syndrome) JCI Insight. 2016;1(9) doi: 10.1172/jci.insight.86696. A pioneer report in gene therapy approach for treatment in MPS II in a mouse model. The procedure was accomplished through direct delivery of vector to CNS and positive results provide strong proof in CNS treatment including behavioral normalization in addition to prolonged survival, giving clear implication for future use in clinical translation of this approach. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140**.Hinderer C, Katz N, Louboutin JP, et al. Delivery of an adeno-associated virus vector into CSF attenuates central nervous system disease in mucopolysaccharidosis type II mice. Human Gene Therapy. 2016;11:906–915. doi: 10.1089/hum.2016.101. Using adeno associated virus serotype 9 vector provides potential treatment for CNS treatment of MPS II though injection into the cerebrospinal fluid. Was found to significantly treat brain lesions and long term memory suggesting long term, positive effects which could translate into clinical treatment. [DOI] [PubMed] [Google Scholar]
- 141.Wakabayashi T, Shimada Y, Akiyama K, et al. Hematopoietic stem cell gene therapy corrects neuropathic phenotype in murine model of mucopolysaccharidosis type II. Human gene therapy. 2015;26(6):357–366. doi: 10.1089/hum.2014.158. [DOI] [PubMed] [Google Scholar]
- 142.Yadak R, Torres-Torronteras J, Bogaerts E, et al. OP45–3024: Efficient lentiviral vector-mediated hematopoietic stem cell gene therapy in MNGIE mice. European Journal of Pediatric Neurology. 2015;19:S15. [Google Scholar]
- 143.Eichler FS, Cox TM, Crombez E, et al. Metachromatic Leukodystrophy: An Assessment of Disease Burden. Journal of Child Neurology. 2016;31(13):1457–1463. doi: 10.1177/0883073816656401. [DOI] [PubMed] [Google Scholar]
- 144.Meneghini V, Frati G, Sala D, et al. Generation of Human Induced Pluripotent Stem Cell-Derived Bona-Fide Neural Stem Cells for Ex Vivo Gene Therapy of Metachromatic Leukodystrophy. Stem Cells Translational Medicine. 2016;5(11):1–17. doi: 10.5966/sctm.2015-0414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.McIntyre C, Roberts ALD, Ranieri E, et al. Lentiviral-mediated gene therapy for murine mucopolysaccharidosis type IIIA. Molecular genetics and metabolism. 2008;93(4):411–418. doi: 10.1016/j.ymgme.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 146.Derrick-Roberts AL, Panir K, Pyragius CE, et al. Reversal of established bone pathology in MPS VII mice following lentiviral-mediated gene therapy. Molecular Genetics and Metabolism. 2016;119(3):249–257. doi: 10.1016/j.ymgme.2016.09.003. [DOI] [PubMed] [Google Scholar]
- 147.Connolly JB. Lentiviruses in gene therapy clinical research. Gene therapy. 2002;9(24) doi: 10.1038/sj.gt.3301893. [DOI] [PubMed] [Google Scholar]
- 148.Cardone M, Polito VA, Pepe S, et al. Correction of Hunter Syndrome in the MPSII mouse model by AAV2/8 mediated gene delivery. Hum Mol Genet. 2006;15:1225–36. doi: 10.1093/hmg/ddl038. [DOI] [PubMed] [Google Scholar]
- 149.Christianson HC, Svensson KJ, van Kuppevelt TH, et al. Cancer cell exosomes depend on cell-surface heparan sulfate proteoglycans for their internalization and functional activity. PNAS. 2013;110:17380–85. doi: 10.1073/pnas.1304266110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Biffi A. Gene therapy for lysosomal storage disorders: a good start. Human Mol Genet. 2016;25:R65–R75. doi: 10.1093/hmg/ddv457. [DOI] [PubMed] [Google Scholar]
- 151.Cox TM. Substrate reduction therapy for lysosomal storage diseases. Acta Paediatrica. 2005;04(s447):69–75. doi: 10.1111/j.1651-2227.2005.tb02116.x. [DOI] [PubMed] [Google Scholar]
- 152.Piotrowska E, Jakobkiewicz-Banecka J, Baranska S, et al. Genistein-mediated inhibition of glycosaminoglycan synthesis as a basis for gene expression targeted isoflavone therapy for mucopolysaccharidoses. European Journal of Human Genetics. 2006;14:846–852. doi: 10.1038/sj.ejhg.5201623. [DOI] [PubMed] [Google Scholar]
- 153.Friso A, Tomanin R, Salvalaio M, et al. Genistein reduces glycosaminoglycan levels in a mouse model of mucopolysaccharidosis type II. British Journal of Pharmacology. 2010;159(5):1082–1091. doi: 10.1111/j.1476-5381.2009.00565.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Marucha J, Tylki-Szymanska A, Jakobkiewicz-Banecka J, et al. Improvement in the range of joint motion in seven patients with mucopolysaccharidosis type II during experimental gene expression-targeted isoflavone therapy (GET IT) American Journal of Medical Genetics. 2011;155(9):2257–2262. doi: 10.1002/ajmg.a.34146. [DOI] [PubMed] [Google Scholar]
- 155.Kim K, Dodsworth C, Paras A, et al. High dose genistein aglycone therapy is safe in patients with mucopolysaccharidoses involving the central nervous system. Molecular Genetics and Metabolism. 2013;109(4):382–385. doi: 10.1016/j.ymgme.2013.06.012. [DOI] [PubMed] [Google Scholar]
- 156.Mostcot M, Jakobkiewicz-Banecka J, Kloska A, et al. Modulation of expression of genes involved in glycosaminoglycan metabolism and lysosome biogenesis by flavonoids. Scientific Reports. 2015;5:1–13. doi: 10.1038/srep09378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157*.Moskot M, Gabig-Ciminska M, Jakobkiewicz-Banecka J, et al. Cell Cycle is disturbed in mucopolysaccharidosis type II fibroblasts and can be improved by genistein. Gene. 2016;585(1):100–103. doi: 10.1016/j.gene.2016.03.029. Provides novel approach for mechanism through which the harmful effects of GAG accumulation in MPS II can be mediated. [DOI] [PubMed] [Google Scholar]
- 158.Sharma R, Anguela XM, Doyon Y, et al. In vivo genome editing of the albumin locus as a platform for protein replacement therapy. Blood. 2015;126(15):1777–1784. doi: 10.1182/blood-2014-12-615492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Moores C, Rogers JG, McKenzie IM, et al. Anesthesia for children with mucopolysaccharidoses. Anaesth Intensive Care. 1996;24:459–63. doi: 10.1177/0310057X9602400408. [DOI] [PubMed] [Google Scholar]
- 160*.Kubaski F, Mason RW, Nakatomi A, et al. Newborn screening for mucopolysaccharidoses: A pilot study of measurement of glycosaminoglycans by tandem mass spectrometry. Journal of Inherited Metabolic Disease. 2017;40:151–158. doi: 10.1007/s10545-016-9981-6. Pilot study which describes realistic cutoff values for screening infant dried blood spot samples. Such studies are critical in the development of screenings and subsequent implementation and early treatment. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Orii T, Suzuki Y, Tomatsu S. Educational CD for Hunter syndrome Mucopolysaccharidosis type II - Hunter disease. 2005:10, 11, 55, 57, 74, 82, 72. [Google Scholar]