Abstract
Background
The US Centers for Disease Control and Prevention ensured adequate performance of the routine triglycerides methods used in Japan by a chromotropic acid reference measurement procedure used by the Centers for Disease Control and Prevention lipid standardization programme as a reference point. We examined standardized data to clarify the performance of routine triglycerides methods.
Methods
The two routine triglycerides methods were the fluorometric method of Kessler and Lederer and the enzymatic method. The methods were standardized using 495 Centers for Disease Control and Prevention reference pools with 98 different concentrations ranging between 0.37 and 5.15 mmol/L in 141 survey runs. The triglycerides criteria for laboratories which perform triglycerides analyses are used: accuracy, as bias ≤5% from the Centers for Disease Control and Prevention reference value and precision, as measured by CV, ≤5%.
Results
The correlation of the bias of both methods to the Centers for Disease Control and Prevention reference method was: y (%bias) = 0.516 × (Centers for Disease Control and Prevention reference value) −1.292 (n = 495, R2 = 0.018). Triglycerides bias at medical decision points of 1.13, 1.69 and 2.26 mmol/L was −0.71%, −0.42% and −0.13%, respectively. For the combined precision, the equation y (CV) = −0.398 × (triglycerides value) + 1.797 (n = 495, R2 = 0.081) was used. Precision was 1.35%, 1.12% and 0.90%, respectively. It was shown that triglycerides measurements at Osaka were stable for 36 years.
Conclusions
The epidemiologic laboratory in Japan met acceptable accuracy goals for 88.7% of all samples, and met acceptable precision goals for 97.8% of all samples measured through the Centers for Disease Control and Prevention lipid standardization programme and demonstrated stable results for an extended period of time.
Keywords: Centers for Disease Control and Prevention standardization, triglycerides, fluorometric method, enzymatic method, chromotropic acid reference measurement procedure, GC-IDMS
Introduction
The relationship between triglycerides (TGs), total cholesterol (TC) and cardiovascular diseases (CVD), including myocardial infarction and stroke, has been investigated in cohort studies since the early 1950s. Similar to TC, high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C), TG is an analyte that has been monitored by the international lipid standardization programme at the US Centers for Disease Control and Prevention (CDC). Accurate and precise measurements of TG are crucial in CVD risk management and are used for convenient and cost-effective estimation of LDL-C using Friedewald equation. Significantly elevated TG concentrations are clinically important due to their direct relationship to pancreatitis and their association with diabetes mellitus, and renal and liver diseases. However, it remains equivocal whether TG is an independent risk factor for CVD. Some of the reasons are variation in the molecular weights of TG that depend on the type of constituent fatty acids, TG blood levels being markedly affected by dietary intake and inverse correlation with HDL-C levels, which is one of the most confounding variables in assessing the role of hypertriglyceridemia in atherosclerosis. Previous studies reported a non-significant relationship between TG and the risk of CVD.1,2 A recent large-scale meta-analysis demonstrated that the relationship between TG and the risk of coronary artery disease and stroke became weak after adjusting for HDL-C and non-HDL-C levels.3 On the other hand, recent epidemiological studies found a positive relationship with CVD, with a growing number of studies suggesting non-fasting hypertriglyceridemia being a potential independent risk factor for CVD.4–8 To further investigate the relationship of TG levels and other lipids with CVD, a long-term dynamic cohort study of Japanese population, the Circulatory Risk in Communities Study (CIRCS), conducted cardiovascular risk surveys of approximately 11,000 middle-aged residents in four communities in Japan. Limited evidence was found for Asian population. The CIRCS investigators examined the relationship between TG concentrations and ischemic cardiovascular events in Japanese men and women4,7,9–11 using standardized TG data measured at the epidemiological laboratory in Osaka certified by the CDC Lipid Standardization Programme.
The aim of this paper is to examine the standardized data in order to clarify the performance (accuracy, reproducibility and stability for an extended period of time) of the routine TG measurements. Additionally, the Gas Chromatography-Isotope Dilution Mass Spectrometry (GC-IDMS) established at Osaka laboratory was used to study the relationship between net TG and TG (as total glycerides) values.
Materials and methods
Materials
CDC standardization pools for TG
Between the 1970s and 1990s, standardization pools for TG were prepared in the CDC lipid reference laboratory according to the directions proposed by Williams et al.12–14 Isolated serum samples resembled human serum in appearance, composition, and stability, and showed no evidence of interfering material in the cholesterol and TG methods tested. Since 2000, standardization materials for lipids have been prepared according to the Clinical Laboratory Standards Institute document C37-A.15 This guideline provides guidance for collecting and processing blood to manufacture frozen serum pools, and for performing quality assurance of the obtained serum.16
All survey pools were shipped frozen from the CDC to the participants in the lipid standardization programme and stored at −70°C for subsequent analysis. They were blinded to the participants. Every pool was analysed in duplicate. Between April 1975 and April 2011, 495 CDC pools from 98 different lots ranged from 0.37 to 5.15 mmol/L were analysed in 141 quarterly monitoring surveys. Each survey consisted of two to four different pools. CDC survey pools were analysed over the course of 36 years along with the serum samples collected in the CIRCS communities. TG measurements were conducted at the Osaka Medical Center for Cancer and Cardiovascular Diseases between April 1975 and June 2001, and at the Osaka Medical Center for Health Science and Promotion between July 2001 and March 2012.
Methods
Definition of net TG and TG (as total glycerides)
In this paper, TG (as total glycerides) is defined as the sum of monoglycerides, diglycerides, TGs and free glycerol. Net TG is the difference between total glycerides and free glycerol. Although some mono-and diglycerides are present in serum (~3%), results were calculated as TG.
Chromotoropic acid reference measurement procedure (CA RMP) for TG at the CDC
Since 1966, the CDC has been offering standardization assistance as a point of reference for the measurement of TG to epidemiological and lipid research laboratories. From 1966 to 2004, the semi-automated TG method using Technicon AutoAnalyzer II (AA II) was based on the methods of Carlson,17 Carlson and Wadström18 and the techniques of Van Handel and Zilversmit19 and Lofland.20 The procedure consisted of the following steps21: (a) serum lipids were extracted with methylene chloride, then treated with silicic acid to remove phospholipids and free glycerol. (b) An aliquot of the extract was saponified with alcoholic KOH to release glycerol. (c) The released glycerol was oxidized with sodium periodate to produce formaldehyde. (d) The produced formaldehyde reacted with chromotropic acid to form a chromogen, the absorbance of which was measured at 570 nm. The TG value obtained by CA RMP was net TG. For this method, the CDC used a standard composed of triolein/tripalmitin (2:1 w/w) in order to reflect the unsaturated/saturated TG in human serum; however, serum values are reported as triolein equivalents.21,22
Fluorometric method as a routine method for TG at Osaka laboratory
The fluorometric method for TG was used between April 1975 and August 1985 at Osaka laboratory through the WHO-CDC Cooperative Cholesterol-Triglycerides Standardization Programme. This semiautomated method was a modification of the procedure of Kessler and Lederer23,24 adapted to the AA II with a fluoronephelometer.25 Serum was extracted with isopropanol and treated with zeolite mixture to remove phospholipids, free glycerol, glucose, bilirubin and other substances that could interfere with the analysis. TG was hydrolysed with alkali and produced glycerol was oxidized to formaldehyde, which, when coupled with acetylacetone and ammonium ions, produced the fluorescent product, 3,5-diacetyl-4-dihydrolutidine. The TG value obtained by the fluorometric method was net TG. Calibration of the TG procedure was accomplished with primary standards of triolein in isopropanol.22
Enzymatic method as a routine method for TG at Osaka
The enzymatic method for TG was used between September 1985 and April 2011 through the CDC-NHLBI Lipid Standardization Programme. Since the mid-1970s, a fully automated enzymatic method has virtually replaced the earlier chemical methods that required solvent extraction of the specimen and treatment with adsorbents to remove phospholipids, free glycerol and other interfering substances. In the first enzymatic reaction of the TG method used in Japan, the free glycerol was eliminated by pretreatment of samples with the mixture of glycerol 3-phosphotransferase, glycerolkinase and glycerol-3-phosphate oxidase to release H2O and O2. Therefore, the TG value obtained by the enzymatic methods produced by Japanese reagent suppliers was net TG. The enzymatic serum values are reported as triolein equivalents.22,26
The relationship between TG (as total glycerides) and net TG by GC-IDMS
We have established the TG method using the GC-IDMS at Osaka.27 We have standardized our method by analysing the CDC reference pools for TG (as total glycerides, n = 64) and for net TG (n = 50) in quadruplicates in 16 independent analytical runs in the TG standardization programme28 of the Cholesterol Reference Method Laboratory Network (CRMLN). As application of the standardized GC-IDMS method at Osaka and in order to examine the relationship between TG (as total glycerides) and net TG, both analytes were measured in duplicate using fresh and non-diseased serum samples (n = 87).
TG performance goals for the clinical laboratory
Precision was evaluated in terms of the coefficient of variation (CV, %), and accuracy was evaluated in terms of deviations (%bias) from the CDC reference value. The US National Cholesterol Education Programme Working Group on lipoprotein measurement29 recommended that clinical laboratories perform TG analyses with the criteria for accuracy, bias ≤5% from the CDC reference point and for precision, as measured by CV, ≤5%.
Statistical analysis
We used the protocol of NCCLS guideline EP9-A from the Clinical and Laboratory Standards Institute for bias estimation30 and the STATA12 analysis program for all other calculations.31,32
Results
Correlation between CDC and Osaka
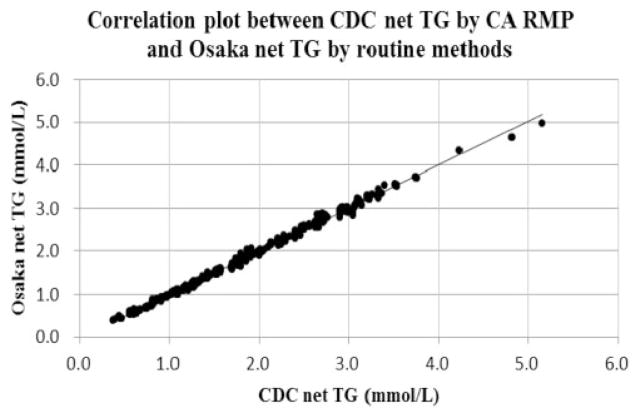
Standardized TG results of the fluorometric and enzymatic methods used at Osaka laboratory were combined. Their relationships with the corresponding CDC values were analysed. Figure 1 shows the correlation plots between CDC net TG by CA RMP and Osaka net TG by routine methods. These results indicated that TG concentrations at medical decision points of 1.13, 1.69 and 2.26 mmol/L at CDC corresponded to 1.12, 1.69 and 2.26 mmol/L at Osaka, respectively.
Figure 1.
Correlation plot between CDC net TG by CA RMP and Osaka net TG by routine methods.
CA RMP: chromotropic acid reference measurement procedure.
Note: X-axis indicates CDC net TG values (mmol/L) and y-axis indicates Osaka net TG (mmol/L).
The correlating equation was y = 1.008 × −0.017 (n = 495, R2 = 0.997). P-value and 95% CI for slope are <0.001 and (1.003, 1.013), respectively. P-value and 95% CI for intercept are <0.001 and (−0.027, −0.007), respectively.
Net TG: total glycerides–free glycerol; CI: confidence interval; CA RMP: chromotropic acid reference measurement procedure.
Accuracy and precision of both methods combined
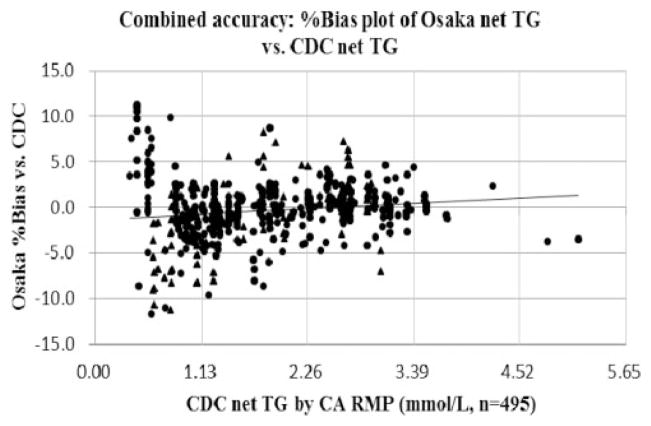
The combined accuracy of the fluorometric and the enzymatic methods was assessed. Figure 2 and Table 1 show the combined accuracy in the %bias plot of Osaka net TG vs. CDC net TG. The %bias vs. the CDC reference values of medical decision points for 1.13, 1.69 and 2.26 mmol/L were −0.71%, −0.42%, and −0.13% at Osaka, respectively. The Osaka laboratory met acceptable accuracy goals within ±5% of the CDC reference values for 88.7% of all samples measured. The maximum %bias at Osaka TG was +11.6% and the minimum was −11.7%. A slight constant and proportional bias was observed when comparing the bias of both methods combined against the CDC target values. The enzymatic TG value was on average 1.4% higher than the fluorometric TG value. The differences observed in accuracy between both methods were within acceptable ranges of TG analytical goals.
Figure 2.
Combined accuracy: %bias plot of Osaka net TG vs. CDC net TG.
Note: X-axis indicates CDC net TG by CA RMP (mmol/L) and y-axis indicates Osaka %bias vs. CDC.
The correlating equation was y (Osaka %bias) = 0.516 × (CDC) −1.292 (n = 495, R2 = 0.018).
Accuracy pass rate under the TG goals (%bias ≤5%) was 88.7% of all samples measured.
P-value and 95% CI for slope are <0.001 and (0.181, 0.851), respectively.
P-value and 95% CI for intercept are 0.003 and (−1.954, −0.630), respectively.
Black triangles (▲) indicate plots of the fluorometric method and black circles (●) indicate plots of the enzymatic method.
CA RMP: chromotropic acid reference measurement procedure; Net TG: Total glycerides – free glycerol; CI: confidence interval.
Table 1.
Accuracy and precision of net TG by Osaka routine methods vs. net TG by CDC CA RMP over time.
| Parameter | TG method | Number of samples | Pass rate, % | Slope, (95% CI), P | Intercept, (95% CI), P | R2 | Time period |
|---|---|---|---|---|---|---|---|
| Combined accuracy (as %bias vs. CDC) | Routine methods (combining fluorometric and enzymatic method) | 495 | 88.7 | 0.516 (0.181, 0.851) P ≤ 0.001 | −1.292 (−1.954, −0.630) P = 0.003 | 0.018 | April 1975 to April 2011 (36 years) |
| Combined precision (as CV, %) | Routine methods (combining fluorometric and enzymatic method) | 495 | 97.8 | −0.398 (−0.517, −0.280) P ≤ 0.001 | 1.798 (1.563, 2.032) P ≤ 0.001 | 0.081 | April 1975 to April 2011 (36 years) |
TG: triglycerides; Net TG: total glycerides-free glycerol; CDC: US Centers for Disease Control and Prevention; CA RMP: chromotoropic acid reference measurement procedure; CV: coefficient of variation; CI: confidence interval; CI: confidence interval; TG: total glycerides.
Note: Osaka routine methods for net TG include the fluorometric method used during April 1975 and August 1985, and the enzymatic method used during September 1985 and April 2011. For the calculation of slope and intercept, x-axis sets CDC reference value and y-axis sets routine methods combining fluorometric and enzymatic method at Osaka. TG accuracy criterion (%bias ≤5%) and TG precision criterion (CV ≤5%) were used for pass rate.
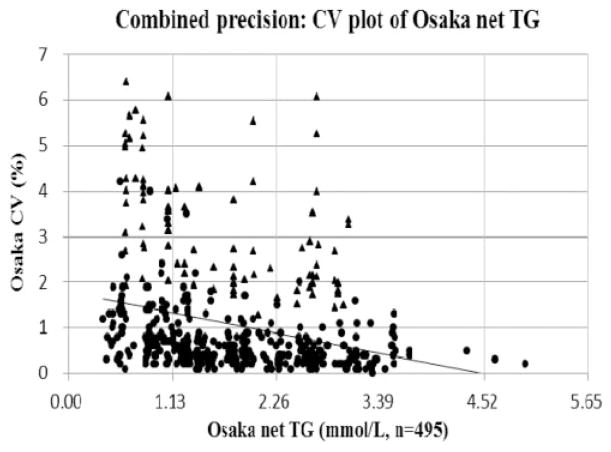
The combined precision of the fluorometric and the enzymatic methods was evaluated. Figure 3 and Table 1 show the combined precision in the CV plot of Osaka net TG. The CVs of medical decision points for 1.13, 1.69 and 2.26 mmol/L were 1.35%, 1.12% and 0.90% at Osaka, respectively. The Osaka laboratory met acceptable precision goals ≤5% at CV for 97.8% of all samples assayed. The maximum CV of Osaka TG measurement was 6.4%. The mean precision of TG measurements with the fluorometric method was larger than 3% but still within the precision requirement. Meanwhile, the mean CV of the enzymatic method was smaller than 1%. The CV of the semiautomated fluorometric method was four to five-fold larger, i.e. poorer precision, than that of the automated enzymatic method. These results indicated that the replacement of the enzymatic method contributed to improving reproducibility of the TG measurements.
Figure 3.
Combined precision: CV plot of Osaka net TG.
Note: X-axis indicates Osaka net TG (mmol/L) and y-axis indicates Osaka CV (%).
The correlating equation was y(Osaka CV) = −0.398 × (Osaka net TG) +1.797
(n = 495, R2 = 0.081). Precision pass rate under the TG goals (CV ≤5%) was 97.8% of all samples measured.
P-value and 95% CI for slope are<0.001 and (−0.517, −0.280), respectively.
P-value and 95% CI for intercept are <0.001 and (1.563, 2.032), respectively.
Black triangles (▲) indicate plots of the fluorometric method and black circles (●) indicate plots of the enzymatic method.
Net TG: total glycerides–free glycerol; CI: confidence interval.
%Bias plot of Osaka net TG by routine methods at each run over decades
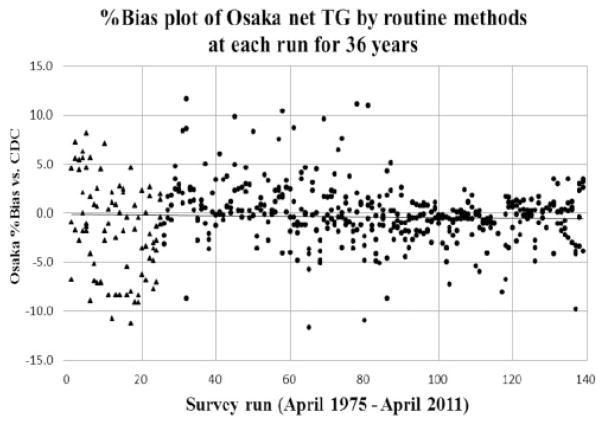
Figure 4 shows %bias plots of Osaka net TG by routine methods for each run from 141 surveys over 36 years. The x-axis indicated the number of survey runs, and the y-axis indicated the %bias at Osaka vs. the CDC reference value. Each survey run consisted of two to four CDC pools. No significant changes were observed when measurement of %bias was analysed over time.
Figure 4.
%Bias plot of Osaka net TG using routine methods at each run for 36 years.
Note: X-axis indicates each survey run from total 141 runs and y-axis indicates Osaka %bias vs. CDC reference value.
Black triangles (▲) indicate plots of the fluorometric method and black circles (●) indicate plots of the enzymatic method.
Net TG: total glycerides–free glycerol.
Relationship between net TG and TG (as total glycerides) by the GC-IDMS
In the assessment of the performance of the GC-IDMS at Osaka27 compared with CDC, the accuracy (%bias vs. CDC GC-IDMS) and precision (CV) of total glycerides concentration at 1.69 mmol/L (medical decision point) were −0.1% (n = 64) and 0.68%, respectively. And the accuracy and precision of the net TG concentration at 1.69 mmol/L were −1.8% (n = 50) and 0.75%, respectively. These results permit reliable quantification of net TG and total glycerides using Osaka GC-IDMS.
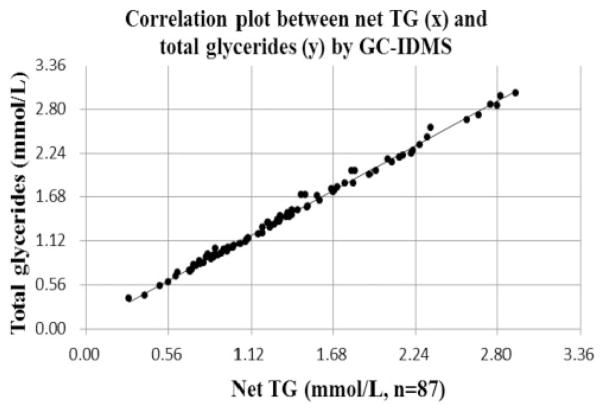
Based upon above mentioned performance between CDC and Osaka, Figure 5 shows that the relationship between total glycerides and net TG using fresh and non-diseased specimens (n = 87). In the estimation by the regression equation, net TG concentrations of medical decision points for 1.13, 1.69 and 2.26 mmol/L corresponded to 1.20, 1.77 and 2.35 mmol/L of total glycerides, respectively.
Figure 5.
Correlation plot between net TG (x) and total glycerides (y) by GC-IDMS.
Note: X-axis indicates net TG values (mmol/L) and y-axis indicates total glycerides values (mmol/L). The correlating equation was y = 1.020 × +4.057 (n = 87, R2 = 0.995). P-value and 95% CI for slope are <0.001 and (1.005, 1.036), respectively. P-value and 95% CI for intercept are <0.001 and (1.989, 6.125), respectively. GC-IDMS: gas chromatography-isotope dilution mass spectrometry; Total glycerides: the sum of monoglycerides, diglycerides, triglycerides and free glycerol; Net TG: total glycerides–free glycerol; CI: confidence interval.
Discussion
The reference method that is used to standardize TG measurements and to evaluate the performance of epidemiological and clinical laboratories underwent major changes over the past years. The semi-automated CA RMP was developed as an in-house procedure for the determination of TG in serum at CDC.21 The method utilizes manual extraction process that removes interfering substances such as phospholipids, protein and free glycerol, and allows for minimal retention of some monoglycerides and diglycerides. The method has been used for more than 40 years as the accuracy base for standardization and metrological traceability for TG testing.25 However, the CA RMP has several disadvantages including the requirement of very specific operational conditions, the use of obsolete and cumbersome instrumentation and toxic reagent chemical such as methylene chloride that complicate its use in routine clinical settings. Based on over seven years of our own experience, the method was very difficult to maintain due to complicated extraction and filtration steps designed to remove the silicic acid particles. It was very challenging to obtain reproducible data. Therefore, we were unable to establish the CA RMP as the secondary reference method.
To overcome these problems, the CA RMP was discontinued and replaced by a GC-IDMS established at the CDC.28 Edwards et al.28 reported that the CDC GC-IDMS has the specificity and ruggedness to accurately quantify total glycerides and demonstrates sufficiently acceptable agreement with the National Institute of Standards and Technology primary RMP for TG measurements. CDC GC-IDMS measures only total glycerides, while Osaka GC-IDMS operates as a secondary and practical reference method that has been established in 2011 to separately measure total glycerides and free glycerol.27 The total glycerides by Osaka GC-IDMS are currently standardized through the CRMLN standardization programme by the GC-IDMS RMP at CDC.28 Osaka laboratory is continuing to further verify the performance, selectivity and specificity for the GC-IDMS.
Several mass spectrometry methods for TG have been reported.28,33–37 To ensure new mass spectrometry as well as routine TG methods are accurate, CDC is implementing performance evaluations for TG reference and routine methods as part of its Lipid Standardization Programme. This will help to standardize TG measurements and to evaluate the performance of epidemiological and clinical laboratories and reagent manufacturers.
Free glycerol is physiologically present in blood and accounts for approximately 5% of total glycerides. TG concentrations are expressed as total glycerides in many Western countries and most regions of China. However, in Japan, they are presented as net TG. The mean difference observed between net TG and total glycerides in non-diseased samples (n = 87) measured with the GC-IDMS method at Osaka was approximately 0.08 mmol/L (Figure 5). TG applied to the Friedewald equation (LDL-C = TC – HDL-C – TG/5)38 is the net TG. This is due to the fact that Friedewald et al. used the net TG values assayed by the fluorometric method, not total glycerides values.23,24 Therefore, when total glycerides levels are 1.69 mmol/L (medical decision point), the application of the net TG values in the equation may result in an approximately 0.02 mmol/L higher estimation of LDL-C levels. This indicates the need to take this difference into account when comparing test results from Japan with those from Western countries. Among developed countries with a high prevalence of CVD, Japan is the only country where net TG measurement system is still adopted nationwide. A small number of assay kits for net TG are exported from Japan to several regions in China, Korea, Taiwan, Mongolia, Indonesia and Singapore. The majority of domestic suppliers of laboratory reagents in Japan market only net TG assay kits and laboratory reagents for total glycerides are available from only one supplier.
The clinical relevance of free glycerol in the blood remains to be defined. A considerable body of literature suggests a minimal need for measuring free glycerol.39,40 Several diseases (such as Hirschsprung’s disease and small bowel infarction) and heparin infusion have shown to increase free glycerol levels.39 However, more than 98% of our test samples are taken from individuals without these conditions. Therefore, measurement of net TG instead of total glycerides may not be clinically relevant for the majority of patients. It currently remains unclear whether free glycerol will impact clinical decision-making when measuring total glycerides in patients with these rare conditions. Although the concept of net TG measurements is more consistent with the definition of the analyte and with calculating LDL-C using the Friedewald equation, the measurement of total glycerides may not have a significant impact on clinical decision-making for the majority of patients. Further studies are needed to clarify the impact of using TG as compared to net TG on patient classification.
Conclusions
Routine TG methods used at the epidemiologic laboratory in Osaka were successfully standardized using metrological traceability to the CDC Lipid Standardization Programme. Accuracy and precision met the analytical performance goals set for the TG measurements and remained stable for 36 years with no changes in accuracy after switching to mass spectrometry-based methods. Accurate and reproducible TG measurements are important for clinical diagnosis of CVD and present a convenient and cost-effective tool for routinely estimating LDL-C using the Friedewald equation.
Acknowledgments
The authors profusely thank Dr. Katsuyuki Nakajima and Dr. Ikunosuke Sakurabayashi for their valuable comments and discussion, and Ms. Yukari Ichikawa for her excellent assistance in providing the references and manuscript.
Funding
This work was supported by ‘Health and Labor Science Research Grants, Comprehensive Research on Life-Style Related Diseases including Cardiovascular Diseases and Diabetes Mellitus. 015’ from the Ministry of Health, Labour, and Welfare of Japan.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MN.
Contributorship
All authors confirmed they have contributed to the intellectual content of this paper and have met the following three requirements: (a) significant contributions to the conception and design, acquisition of data or analysis and interpretation of data; (b) drafting or revising the article for intellectual content and (c) final approval of the published article.
References
- 1.Austin MA. Plasma triglyceride as a risk factor for coronary heart disease: the epidemiologic evidence and beyond. Am J Epidemiol. 1989;129:249–259. doi: 10.1093/oxfordjournals.aje.a115130. [DOI] [PubMed] [Google Scholar]
- 2.Miller M, Stone NJ, Ballantyne C, et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2011;123:2292–2333. doi: 10.1161/CIR.0b013e3182160726. [DOI] [PubMed] [Google Scholar]
- 3.Di Angelantonio E, Sarwar N, Perry P, et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA. 2009;302:1993–2000. doi: 10.1001/jama.2009.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Iso H, Naito Y, Sato S, et al. Serum triglycerides and risk of coronary heart disease among Japanese men and women. Am J Epidemiol. 2001;153:490–499. doi: 10.1093/aje/153.5.490. [DOI] [PubMed] [Google Scholar]
- 5.Nordestgaard BG, Benn M, Schnohr P, et al. Nonfasting triglycerides and risk of myocardial infarction ischemic heart disease and death in men and women. JAMA. 2007;298:299–308. doi: 10.1001/jama.298.3.299. [DOI] [PubMed] [Google Scholar]
- 6.Bansal S, Buring JE, Rifai N, et al. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA. 2007;298:309–316. doi: 10.1001/jama.298.3.309. [DOI] [PubMed] [Google Scholar]
- 7.Iso H, Imano H, Yamagishi K, et al. Fasting and non-fasting triglycerides and risk of ischemic cardiovascular disease in Japanese men and women: The Circulatory Risk in Communities Study (CIRCS) Atherosclerosis. 2014;237:361–368. doi: 10.1016/j.atherosclerosis.2014.08.028. [DOI] [PubMed] [Google Scholar]
- 8.Freiberg JJ, Tybjaerg-Hansen A, Jensen JS, et al. Nonfasting triglycerides and risk of ischemic stroke in the general population. JAMA. 2008;300:2142–2152. doi: 10.1001/jama.2008.621. [DOI] [PubMed] [Google Scholar]
- 9.Satoh H, Nishino T, Tomita K, et al. Fasting triglyceride is a significant risk factor for coronary artery disease in middle-aged Japanese men. Circ J. 2006;70:227–231. doi: 10.1253/circj.70.227. [DOI] [PubMed] [Google Scholar]
- 10.Iso H, Sato S, Kitamura A, et al. Metabolic syndrome and the risk of ischemic heart disease and stroke among Japanese men and women. Stroke. 2007;38:1744–1751. doi: 10.1161/STROKEAHA.106.469072. [DOI] [PubMed] [Google Scholar]
- 11.Noda H, Iso H, Saito I, et al. The impact of the metabolic syndrome and its components on the incidence of ischemic heart disease and stroke: the Japan Public Health Center-based Study. Hypertens Res. 2009;32:289–298. doi: 10.1038/hr.2009.14. [DOI] [PubMed] [Google Scholar]
- 12.Williams JH, Taylor L, Kuchmak M, et al. Preparation of hypercholesterolemic and/or hypertriglyceridemic sera for lipid determinations. Clin Chim Acta. 1970;28:247–253. doi: 10.1016/0009-8981(70)90087-2. [DOI] [PubMed] [Google Scholar]
- 13.Kuchmak M, Taylor L, Williams JH. Preparation of reference sera with desired levels of cholesterol and triglyceride. Clin Chim Acta. 1981;114:127–135. doi: 10.1016/0009-8981(81)90386-7. [DOI] [PubMed] [Google Scholar]
- 14.Bonderman DP, Proksch GJ, Bonderman PW. Addition of triglyceride to serum for use in quality control and reference. Clin Chem. 1976;22:1299–1301. [PubMed] [Google Scholar]
- 15.Preparation and validation of commutable frozen human serum pools as secondary reference materials for cholesterol measurement procedures; approved guideline. Clinical and Laboratory Standards Institute, Document C37-A, 1999.
- 16.Miller WG. Matrix effects in the measurement and standardization of lipids and lipoproteins. In: Rifai N, Warnick GR, Dominiczak MH, editors. Handbook of lipoprotein testing. 2. Washington, D.C: AACC Press; 2000. pp. 695–716. [Google Scholar]
- 17.Carlson LA. Determination of serum triglycerides. J Atheroscler Res. 1963;3:334–336. doi: 10.1016/s0368-1319(63)80012-5. [DOI] [PubMed] [Google Scholar]
- 18.Carlson LA, Wadström LB. Determination of glycerides in blood serum. Clin Chim Acta. 1959;4:197–205. doi: 10.1016/0009-8981(59)90130-5. [DOI] [PubMed] [Google Scholar]
- 19.Van Handel E, Zilversmit DB. Micromethod for the direct determination of serum triglycerides. J Lab Clin Med. 1957;50:152–157. [PubMed] [Google Scholar]
- 20.Lofland HB., Jr A semiautomated procedure for the determination of triglycerides in serum. Anal Biochem. 1964;9:393–400. doi: 10.1016/0003-2697(64)90199-x. [DOI] [PubMed] [Google Scholar]
- 21.CDC Lipid Reference Laboratory. CDC chromotropic acid method for the determination of triglycerides in serum. Atlanta, GA: CDC; 1993. [Google Scholar]
- 22.Klotzsch SG, McNamara JR. Triglyceride measurements: a review of methods and interferences. Clin Chem. 1990;36:1605–1613. [PubMed] [Google Scholar]
- 23.Kessler G, Lederer H. Fluorometric measurement of triglycerides. Automation in analytical chemistry, Technicon symposia; Tarrytown, NY. 17–19 October 1966; 1965. pp. 341–344. [Google Scholar]
- 24.Rush RL, Leon L, Turrell J. Advances in automated analysis, Technicon international congress 1970. Technicon; Tarrytown, NY: 1971. Automated simultaneous cholesterol and triglyceride determination on the AutoAnalyzer Instrument; pp. 503–507. [Google Scholar]
- 25.Manual of laboratory operations, lipid research clinics program, lipid and lipoprotein analysis revised, 1982.
- 26.Stein EA, Myers GL for the National Cholesterol Education Program Working Group on lipoprotein measurement. National Cholesterol Education Program Recommendations for triglyceride measurement: executive summary. Clin Chem. 1995;41:1421–1426. [PubMed] [Google Scholar]
- 27.Koyama I, Iso H, Kiyama M, et al. Establishment of practical procedure for measurement of total glycerides by Isotope Dilution/Gas Chromatography/Mass Spectrometry at the Osaka Medical Center for Health Science and Promotion (CRMLN lipid reference laboratory) Clin Chem. 2011;57(Suppl):A129. [Abstract]) [Google Scholar]
- 28.Edwards SH, Stribling SL, Pyatt SD, et al. Reference measurement procedure for total glycerides by isotope dilution GC-MS. Clin Chem. 2012;58:768–776. doi: 10.1373/clinchem.2011.177063. [DOI] [PubMed] [Google Scholar]
- 29.Recommendations on lipoprotein measurement, from the working group on lipoprotein measurement, NIH publication No. 95-3044, 1995.
- 30.Method comparison and bias estimation using patient samples: approved guideline. NCCLS EP9-A, Vol. 15 No. 17, 1995.
- 31.Westgard JO, Hunt MR. Use and interpretation of common statistical tests in method-comparison studies. Clin Chem. 1973;19:49–57. [PubMed] [Google Scholar]
- 32.Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;327:307–310. [PubMed] [Google Scholar]
- 33.Siekmann L. Reference methods for total cholesterol and total glycerol. Eur J Clin Chem Clin Biochem. 1991;29:277–279. doi: 10.1515/cclm.1991.29.4.277. [DOI] [PubMed] [Google Scholar]
- 34.Bernert JT, Jr, Bell CJ, McGuffey JE, et al. Determination of “free” glycerol in human serum reference materials by isotope-dilution gas chromatography-mass spectrometry. J Chromatogr. 1992;578:1–7. doi: 10.1016/0378-4347(92)80218-f. [DOI] [PubMed] [Google Scholar]
- 35.Ellerbe P, Sniegoski LT, Welch MJ. Isotope-dilution mass-spectrometry as a candidate definitive method for determining total glycerides and triglycerides in serum. Clin Chem. 1995;41:397–404. [PubMed] [Google Scholar]
- 36.Ackermans MT, Ruiter AF, Endert E. Determination of glycerol concentrations and glycerol isotopic enrichments in human plasma by gas chromatography/mass spectrometry. Anal Biochem. 1998;258:80–86. doi: 10.1006/abio.1997.2535. [DOI] [PubMed] [Google Scholar]
- 37.Mclntosh TS, Davis HM, Matthews DE. A liquid chromatography-mass spectrometry method to measure stable isotopic tracer enrichments of glycerol and glucose in human serum. Anal Biochem. 2002;300:163–169. doi: 10.1006/abio.2001.5455. [DOI] [PubMed] [Google Scholar]
- 38.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- 39.Jessen RH, Dass CJ, Eckfeldt JH. Do enzymatic analyses of serum triglycerides really need blanking for free glycerol? Clin Chem. 1990;36:1372–1375. [PubMed] [Google Scholar]
- 40.Cole TG. Glycerol blanking in triglyceride assays: Is it necessary? Clin Chem. 1990;36:1267–1268. [PubMed] [Google Scholar]