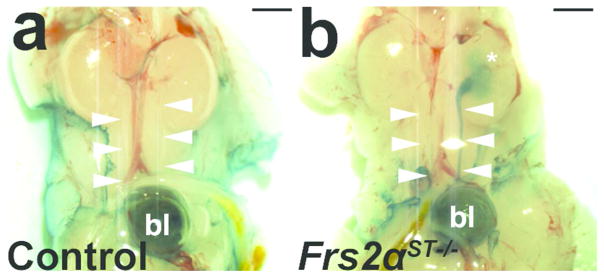Abstract
Background
Fibroblast growth factor receptor 2 (Fgfr2) deletion from murine peri-Wolffian duct stroma (ST) results in aberrant ureteric bud induction, abnormal ureteral insertion into the bladder, and high rates of vesicoureteral reflux (VUR). It is unclear which receptor docking protein(s) is/are responsible for Fgfr2 actions in these tissues. We investigated whether the docking protein, fibroblast receptor substrate 2α (Frs2α), had roles in peri-Wolffian duct stroma similar to Fgfr2.
Methods
We conditionally deleted Frs2α in per-Wolffian duct stroma with a Tbx18cre mouse line (Frs2αST−/−). We assessed for ureteric induction defects and alterations in downstream targets mediating defects. We performed euthanized cystograms and assessed ureter-bladder junctions via three dimensional (3D) reconstructions.
Results
Embryonic day (E) 11.5 Frs2αST−/− embryos had many displaced ureteric bud induction sites versus controls. E11.0 Frs2αST−/− embryos had decreased Bmp4 expression and signaling, which can cause abnormal ureteric bud induction. Postnatal day 1 (P1) and P30 Frs2αST−/− mice had higher VUR rates and grades versus controls. Mutant refluxing ureters inserted improperly into the bladder and had shortened intravesicular tunnels versus controls:
Conclusions
Frs2αST−/− have aberrant ureteric induction sites, improper ureteral insertion, shortened intravesicular lengths and VUR. Induction site defects appear secondary to reduced Bmp4 expression, similar to Fgfr2 mutants.
Introduction
Vesicoureteral reflux (VUR), retrograde flow of urine from bladder into kidneys, is present in ~1% of children in North America (1). While VUR appears to follow an autosomal dominant inheritance pattern, no single gene defect has yet been shown to cause the majority of reflux cases in patients. VUR can be associated with urinary tract infections, congenital renal and lower urinary tract malformations, and reflux nephropathy. Reflux nephropathy, characterized by severe renal scarring, is a common cause of end-stage renal disease in children and adults (2).
Aberrant ureteric bud induction has been identified as a cause of vesicoureteral reflux (1). In early stages of renal development, the ureteric bud is induced to evaginate from the caudal mesonephric (Wolffian) duct by growth factors from the nearby metanephric mesenchyme (3). In the 1970s, Drs. Mackie and Stephens hypothesized that improper ureteric bud induction site(s) from the Wolffian duct led to an abnormal position of ureteral insertion into the bladder, resulting in vesicoureteral reflux (4). Additional studies have shown that reflux through abnormally-positioned ureters is associated with shortened intravesicular ureteral tunnels and defects in bladder and ureter muscle (1). Under normal circumstances, the ureteric bud induction site is constrained to its proper position by repressive signaling from the stroma lying between the Wolffian duct and metanephric mesenchyme (5). One critical repressive factor from the stroma is bone morphogenetic protein 4 (Bmp4), as even loss of one Bmp4 allele leads to ureteric induction defects in mice (5).
Fibroblast growth factor receptor (Fgfr) signaling appears to be critical for constraining ureteric bud induction and preventing reflux in mouse models. While global deletion of Fgfr1 and/or Fgfr2 leads to early embryonic lethality, conditional knockout approaches have given insights into how the receptors regulate ureteric induction. Pax3cre-mediated deletion of Fgfr1 and Fgfr2 in both the metanephric mesenchyme and peri-mesonephric duct stroma led to aberrant ureteric induction into a largely absent metanephric mesenchyme (6). Pax3cre mediated deletion of Fgfr2 alone did not result in renal aplasia, but still often resulted in ureteric bud induction site abnormalities (too high or too low) with subsequent high rates of VUR (7, 8). Furthermore, Tbx18cre-driven loss of Fgfr2 in the stroma but not the metanephric mesenchyme recapitulated the aberrant ureteral insertion and high rate VUR, revealing that the receptor was acting through the stroma alone to constrain the ureteric induction site (9). In both Pax3cre and Tbx18cre models, loss of Fgfr2 was associated with depressed Bmp4 expression (9, 10).
Fgfr signaling is mediated via docking adapter proteins, including Fgf receptor substrate 2α (Frs2α) among others. Frs2α binds constitutively to the receptor juxtamembrane region and becomes phosphorylated upon receptor stimulation leading to activation of Erk, Akt and alternative forms of protein kinase C (11). While Frs2α appears to drive most of the Fgfr signaling in metanephric mesenchyme (specifically nephron progenitors) (12), Frs2α does not appear to interact with Fgfr2 in ureteric epithelium (13). Thus, the role of Frs2α in mediating the effects of Fgfr2 signaling in peri-mesonephric duct stroma is unclear. To answer this question, we used a Tbx18cre line to conditionally delete Frs2α and examined the effects on ureteric bud induction, ureter insertion into the bladder, and rates of VUR.
Materials and Methods
Mice
Mixed genetic background Tbx18cre; Frs2αfl/fl mice (Frs2αST−/−) with deletion of Frs2α in peri-Wolffian duct stroma (ST) were produced by breeding Tbx18creTg/+ mice (a gift from Dr. Feng Chen (14)) that drive cre expression in the peri-Wolffian duct stroma with Frs2αfl/fl mice (a gift from Dr. Fen Wang). For embryonic studies, the day that vaginal plug was identified was deemed as E0.5. All experiments were carried out with approval of the University of Pittsburgh Institutional Animal Care and Use committee.
Genotyping
For PCR genotyping, DNA was extracted from tail clippings or embryonic tissues as described (9). Table 1 lists the primers and fragment sizes for various genes.
Table 1.
Primers used for Genotyping
| Gene | Primer Pairs | Size (bp) |
|---|---|---|
|
| ||
| TBX18CRE | 5′-CCA TCC AAC AGC ACC TGG GCC AGC TCA ACA-3′ | 500 (wt) |
| 5′-CCA CCA TCG GTG CGG GAG ATG TCC TTC ACT-3′ | 250 (mt) | |
|
| ||
| Frs2α | 5′-GAG TGT GCT GTG ATT GGA AGG CAG-3′ | 224 (wt) |
| 5′-GGC ACG AGT GTC TGC AGA CAC ATG-3′ | 319 (mt) | |
|
| ||
| CAG | 5′-AAG GGA GCT GCA GTG GAG TA-3′ | 297 (wt) |
| 5′-CCG AAA ATC TGT GGG AAG TC-3′ | ||
| 5′-GGC ATT AAA GCA GCG TAT CC-3′ | 196 (CAG-positive) | |
| 5′-CTG TTC CTG TAC GGC ATG G-3′ | ||
Cystograms
To assess for reflux, euthanized cystograms were performed in P1 and P30 Frs2αST−/− and littermate control pups as described (9). Briefly, mice were euthanized and abdominal walls were reflected to expose the bladder. A 30-gauge needle was inserted into the bladder of P1 and 25-gauge needle inserted into the bladder of P30 mice. Then, bladders were gravity filled with 1% methylene blue dye in a 50 ml syringe raised 30 cm at 5-second intervals starting at a height of 0 cm to a maximum height of 150 cm. Refluxing dye from the bladder toward the kidney (if any) was graded on a I–V scale according to the standard international classification (15). At the completion of the cystogram, all kidneys were photographed and kidney long axis was assessed via Image J software (National Institutes of Health, Bethesda, MD).
Post cystogram 3D reconstructions
Following P1 cystograms, lower bodies from control and Frs2αST−/− mice were fixed in 4% PFA/PBS overnight at 4°C, dehydrated and paraffin embedded. Tissues were serially sectioned at 10 μm form the junction of the ureters and bladder to the bladder neck, and then stained with hematoxylin and eosin (H&E). Tissues were reconstructed using Stereo Investigator, insertion angles calculated and intravesicular tunnel lengths determined using Neurolucida (MBF Biosciences, Williston, VT) as previously described (9).
P30 bladder histology and immunofluorescence
6μm paraffin embedded tissues were stained with H&E. Sections were also immunostained as described (13) using primary antibodies against αSMA (1:250; Sigma Aldrich, St. Louis, MO), and secondary antibodies, donkey anti-mouse 594 Rabbit (1:500; Molecular Probes, Grand Island, NY); and DAPI (1:10,000).
In situ hybridization
Whole-mount in situ hybridization (WISH) was performed on E11.5 control and Frs2αST−/− tissues as described (9). Briefly, DNA fragment templates for Bmp4 (Gene Accession number: NM_007554), Frs2α (NM_177798) and Ret (NM_001080780) were ligated into plasmids using pGEM-T Easy Vector system (Promega, Madison, WI). Plasmids were introduced into competent cells (Promega), after which cells were cultured on Blue/White Select Ampicillin treated agarose plates overnight at 37 C. White colonies were selected and underwent DNA isolation using QIAprep Spin Miniprep Kit (Qiagen). Following isolation plasmid samples were digested using restriction enzyme (EcoRI), sequenced, and linearized using PCR primers designed for M13 sites. Linearized samples were used to generate digoxigenin UTP-labeled antisense RNA probes using DIG RNA Labeling kit as per manufactures instructions (Roche).
For in situ hybridization, 4% paraformaldehyde/phosphate buffered saline (PFA/PBS) fixed tissues were washed in PBS-Tween (PBST), treated with 6% H202 in PBST, followed by 10ug/ml Proteinase K in PBST. Samples were re-fixed in 4% PFA/PBS, washed in PBST and incubated overnight at 58C in hybridization solution containing DIG RNA probe (1:200). Following day, samples were washed blocked in 1% Blocking solution (Roche) in Maleic acid buffer with tween (MABT), and incubated overnight at 4C with 1:2000 anti-DIG antibody (Roche). Samples were subsequently washed in MABT, and developed in BM purple + 0.1% Tween at 4 C with color change observed every 8 hours.
Western Blot Analysis
Protein samples (30μg each) from six whole E11.5 control and Frs2αST−/− urogenital ridges were resolved in 8% SDS-Tris gels (Cat#161-0732, Bio-Rad, Hercules, CA). Proteins were electro-transferred to nitrocellulose membranes (Cat#162-0115, Bio-Rad), blocked with 5% non-fat dry milk in TBST and incubated overnight at 4°C with primary antibodies (pSmad 1/5/8 1:1000; Cat#9511, Cell Signaling and β-Actin: 1:1000, Cat#8457, Cell Signaling, Beverly, MA). Membranes were washed with TBST, and probed with HRP-conjugated secondary antibodies (1:2000; Anti-Rabbit HRP Cat#7076, Cell Signaling). Bound antibodies were visualized using the Amersham ECL Prime Western Blotting Detection kit (Cat#RPN2232GE, Lifesciences, Pittsburgh, PA) according to the manufacturer’s instructions. Blots were imaged using a Fujifilm LAS-3000 (Fujifilm Medical Systems, Stamford, CT) and densitometry analysis was performed using Image J Software.
Common nephric duct length
To determine the length of the common nephric duct, (the caudal portion of the Wolffian duct between the ureteric bud and the urogenital sinus; CND), six E11.5 Frs2αST−/− and control embryos were immersion fixed overnight in 4% PFA/PBS and embedded in paraffin. Tissues along the Wolffian duct were serially sectioned at 4 μm from the cloaca to the base of the ureteric bud and stained with hematoxylin and eosin (H&E). 3D images were rendered using Stereo Investigator and common nephric duct lengths (from the base of the ureteric bud to the cloaca) were determined using Neurolucida software (MBF Biosciences, Williston, VT) as previously described (9). Data are represented as mean ± standard deviation.
Common nephric duct cell proliferation and apoptosis
To examine proliferation and apoptosis in the CND, E12.5 Frs2αST−/− and littermate control embryos were fixed in 4%PFA/PBS, embedded in paraffin and serially sectioned at 5 μm along the CND. For proliferation analysis, sections were labeled with antibodies against phospho-histone H3 (pH3: 1:200; Sigma Aldrich) and E-Cadherin (Ecad: 1:250; Sigma Aldrich) to visualize proliferating cells and the CND epithelium respectively. Phospho-histone H3 was detected with goat anti-rabbit Alex Fluor 594 and E-Cadherin was visualized with donkey anti-rat Alex Fluor 488 (1:500; Molecular Probes). To identify apoptotic cells, Terminal deoxynucleotidyl transferase mediated dUTP Nick End Labeling (TUNEL) was performed via ApopTag Fluorescein Direct in Situ Apoptosis Detection Kit (Cat#S7160 EMD Millipore, Darmstadt Germany) according to the manufacturer’s instructions. Quantification of apoptosis and proliferation along the CND (number of cells per 100 CND cells) was determined (n=3 per genotype) using Image J Software.
Statistics
One-way ANOVA followed by Bonferroni’s post-hoc analysis, student’s T-tests, or Fisher’s exact test analyses were conducted where appropriate, using GraphPad Prism™ 5 (GraphPad Software Inc, La Jolla, CA).
Results
The Tbx18cre line efficiently deletes Frs2α expression peri-Wolffian duct stroma
To determine the role of Frs2α in peri-Wolffian duct stroma, we generated Tbx18cre; Frs2αfl/fl mice (Frs2αST−/−) with deletion of Frs2α in peri-Wolffian duct stroma (ST). As shown in (9), breeding a CAG reporter mouse to the Tbx18cre line reveals significant cre recombination throughout the Wolffian duct stroma at E10.5. Before examining the defects in these mice, we determined the efficiency of Frs2α deletion in Frs2αST−/− mice, by performing whole mount in situ hybridization in control and Tbx18cre; Frs2αFl/Fl E10.5 embryos. Whole-mount in situ hybridization illustrated a significant reduction in Frs2α mRNA expression in mutant peri-Wolffian duct stroma at E10.5 when compared with age-matched controls (Figure 1a, b).
Figure 1. Frs2α expression is significantly reduced in E10.5 Frs2αST−/− peri-Wolffian duct stromal cells.
(a–b) Representative whole-mount in situ hybridization shows dramatically reduced peri-Wolffian duct Frs2α mRNA expression (purple, white arrowheads) in Frs2αST−/− embryos (b) at E10.5 when compared with age-matched controls (a). Importantly, intersomite Frs2α mRNA expression (black arrowheads) is comparable between both genotypes. (Insets in a and b represent higher magnifications of the peri-Wolffian duct regions in the embryos. (a–b) Scale bar = 500μm, Inset scale bar = 150μm.
Frs2αST−/− embryos have abnormal ureteric bud induction
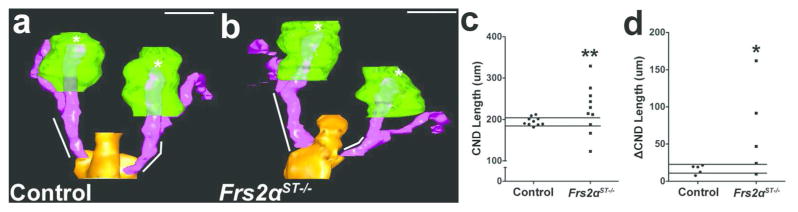
Given that fibroblast growth factor receptors utilize different docking proteins to transmit downstream signaling, we determined whether Tbx18cre mediated deletion of Frs2α in the peri-Wolffian duct stroma affected ureteric induction similar to Tbx18cre; Fgfr2fl/fl mutants (8, 9). Thus, we measured common nephric duct (CND) length (Wolffian duct segment between the cloaca to the base of the ureteric bud) in E11.5 control and Frs2αST−/− embryos. 3D reconstructions revealed similar CND lengths on each side of controls, but frequently divergent lengths on each side of the mutants (Figure 2a, b). While mean CND lengths were comparable between both genotypes (Control 195.84 ± 9.90 μm; Frs2αST−/−: 223.96 ± 57.98 μm, p = 0.8), Frs2αST−/− embryos had a wide distribution of CND lengths, whereas controls had CND lengths tightly clustered around their mean (Figure 2c). Following analysis of five paired sets of control and Frs2αST−/− embryos, we observed 70% (7/10) of control CND lengths were within 1 SD of the mean control CND length mean value, whereas, only 10% (1/10) of Frs2αST−/− CND lengths were within 1 SD of the mean control CND length (Figure 2c). As suggested by the 3D reconstruction images (Figure1a, b), intra-embryo variation in CND length is also significantly greater in Frs2αST−/− embryos when compared with controls (Figure 2d). Thus, similar to Tbx18cre; Fgfr2fl/fl mice, Tbx18cre; Frs2αfl/fl embryos have significant ureteric bud induction defects characterized by a virtual randomization of inductions sites along the Wolffian duct.
Figure 2. E11.5 Frs2αST−/− embryos exhibit aberrant ureteric bud induction.
(a–b) 3D reconstructions of E11.5 control and Frs2αST−/− urogenital tissues reveal common nephric duct lengths (white lines) of greater variability in Frs2αST−/− embryos (b) versus controls (a). (c) Graph illustrating that most control CND lengths are within 1 SD of the control mean length (lines), whereas Frs2αST−/− CND lengths are frequently > 1 SD on either side of the control mean length. (d) Graph illustrating that control intra-embryonic CND lengths are almost all within 1SD of the control mean length (lines), whereas almost all Frs2αST−/− intra-embryonic CND lengths are > 1 SD on either side of the control mean length. (n) = 5 embryos per genotype. Fisher’s Exact Test = *p<0.05. **p<0.01. Green – Metanephric mesenchyme; Orange – Cloaca; Pink – Wolffian Duct and ureteric bud; Asterisks – ureteric bud tips. Scale bar = 200μm.
CND proliferation and apoptosis is similar in both genotypes
We then assessed whether intrinsic defects in the common nephric duct (i.e. aberrant proliferation and/or apoptosis) could be responsible for the induction defects and altered CND lengths in the Frs2αST−/− mutants. Phospho-histone H3 staining of control and Frs2αST−/− CND indicates comparable mean numbers of proliferating cells in both genotypes (Control: 3.77 ± 0.50 × 10−2 cells per 100 CND cells; Frs2αST−/−: 4.24 ± 1.11 × 10−2 cells per 100 CND cells, p = 0.54) (Supplemental Figure S1, online). In addition, TUNEL staining of control and Frs2αST−/− CNDs illustrated similar levels of apoptosis (Control: 26.06 ± 16.73 × 10−2 cells per 100 CND cells; Frs2αST−/−: 32.75 ± 8.11 × 10−2 cells per 100 CND cells. p = 0.57) in both genotypes (Supplemental Figure S1, online). Thus, changes in CND cell proliferation or apoptosis do not lead to changes in CND length observed in Frs2αST−/− embryos; this suggests that defects in signals from the adjacent peri-Wolffian duct stroma likely drive the induction defects.
Frs2αST−/− have reduced Bmp4 expression in peri-Wolffian duct stroma
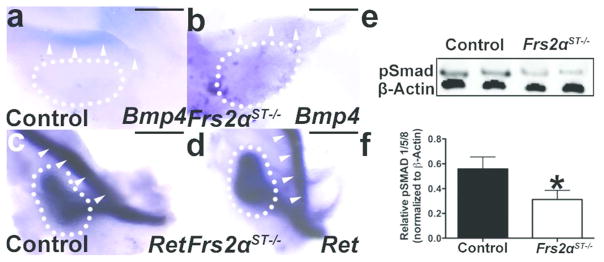
We then determined whether the aberrant ureteric bud induction observed in Frs2αST−/− mice was due associated with a decrease in Bmp4 expression, similar to Fgfr2 mutants. Whole mount in situ hybridization on the urogenital ridges revealed significantly reduced Bmp4 mRNA expression in Frs2αST−/− peri-Wolffian stroma (Figure 3a, b). Furthermore, expression of Ret, a marker of Wolffian duct and ureteric epithelium, appeared unaltered in mutants versus controls, making it unlikely that alterations in Ret expression were responsible for the induction defects (Figure 3c, d). Western blot analysis revealed about a 43% decrease in phosphorylated Smad 1/5/8 expression in E11.5 Frs2αST−/− urogenital ridges versus controls (Figure 3e, f), strongly suggesting that the loss of mutant Bmp4 mRNA led to a decrease in canonical Bmp4 signaling. Thus the loss of Frs2α in peri-Wolffian duct stroma appears to cause reduced Bmp4 expression and canonical Bmp4 signaling, which may explain the aberrant ureteric bud induction in Frs2αST−/− embryos.
Figure 3. Bmp4 expression and canonical signaling is reduced in E11.5 Frs2αST−/− peri-Wolffian duct stroma.
(a–b) Representative whole-mount in situ hybridization in urogenital ridges reveals that compared to the control (a), Bmp4 expression is virtually absent adjacent to the Wolffian duct (arrowheads) in Frs2αST−/− embryos (b, arrowheads). (c–d) In contrast to Bmp4, in situ hybridization reveals similar Ret expression in Wolffian ducts (arrowheads) of the control (c) and mutant (d). (e) Representative Western blot analysis for pSmad 1/5/8 (pSmad) expression in control and Frs2αST−/− urogenital ridges suggests reduced canonical Bmp signaling in Frs2αST−/− tissues. (f) Ratios of pSMAD 1/5/8 and β-actin loading control bands quantified by densitometry confirms a significant reduction in canonical Bmp4 signaling. Dotted lines encircle the metanephros. (n) = 3. Values = Mean + SD. Student’s Test = *p<0.05. Scale bar = 200μm.
Frs2αST−/− mice have high rates of vesicoureteral reflux at birth
To determine whether the ureteric induction defects in Frs2αST−/− mice correlate with increased susceptibility for VUR, we performed cystograms in P1 control and Frs2αST−/− dissected urinary tracts (Figure 4). P1 Frs2αST−/− mice exhibit significantly higher rates of VUR (48.3%) when compared to age-matched controls (6.9%; p<0.001) (Table 2). The higher rates of reflux held true when comparing female or male mutants to their gender-matched controls (Table 2). While most reflux (mutant or control) was unilateral, all of the P1 controls had low grade (I – II) reflux, while 31% of refluxing mutants had high grade (III–IV) VUR (Table 2). Thus, P1 mutants have significantly higher rates and grades of reflux compared with their littermate controls.
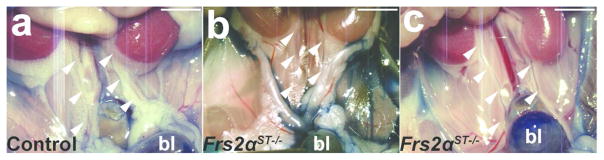
Figure 4. Cystograms in P1 Frs2αST−/− and control mice.
(a) Representative image of a control cystogram revealing methylene blue dye in the bladder (bl), no dye present in the ureters (arrowheads). (b) Image of a Frs2αST−/− cystogram illustrating dye present in the bladder that tracked up the left ureter into the left renal pelvis (*). Scale bar = 500μm.
Table 2.
Incidence and severity of VUR in P1 control and Frs2αST−/− mice
| Genotype | Incidence of Reflux | Reflux Laterality | Reflux Grade | |||
|---|---|---|---|---|---|---|
| Total | Male | Female | Unilateral | I–II | III–IV | |
| Control | 4/58 (7%) | 2/34 (6%) | 2/24 (8%) | 4/4 (100%) | 4/4 (100%) | NA |
| Frs2αST −/− | 15/31 (48%)* | 7/15 (47%) | 8/16 (50%) | 14/15 (93%) | 11/16 (69 %) | 5/16 (31%) |
p < 0.001 vs control
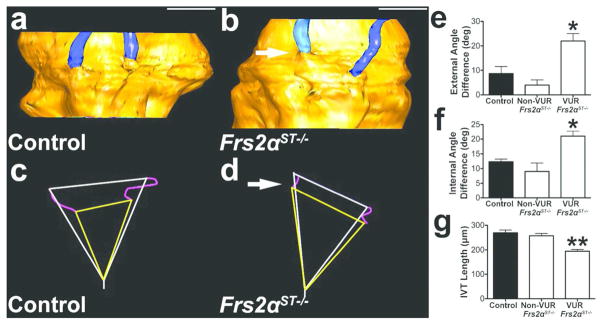
Location and length of ureteral insertion into the bladder are key components determining susceptibility for VUR and aberrant ureteric induction is associated with abnormal ureter insertion into the bladder. To investigate ureteral insertion we reconstructed ureter-bladder junctions in control, non-refluxing Frs2αST−/−, and refluxing (unilateral) Frs2αST−/− mice (Figure 5). In control and non-refluxing mutants, the ureters appeared to insert at a similar location on the bladder, whereas the mutants with unilateral reflux had dissimilar insertion sites; the ureters with reflux inserted either high and lateral or low and medial to the non-refluxing ureter (Figure 5a, b and not shown). Triangles generated from the (internal or external) insertion sites of the ureters and the bladder neck generally were close to isosceles triangles in controls and non-refluxing mutants, whereas they were distorted in mutants with unilateral VUR (Figure 5c, d, and not shown). Measurements of either the external or internal angles at the site of ureter insertion revealed a much larger difference in mutants with unilateral reflux than either control or non-refluxing Frs2αST−/− mice (Figure 5e, f). Moreover, mean intravesicular tunnel (IVT) lengths (i.e. the distance between the external and internal ureter insertion points) of refluxing Frs2αST−/− ureters were significantly shorter than control and non-refluxing Frs2αST−/− mean IVT lengths (Figure 5g). Individual H&E stained sections used for 3D reconstructions show that the intravesicular tunnels for the refluxing mutant ureters have a linear, straight shape, whereas the non-refluxing mutant ureters and control ureters appear to have a more curvilinear trajectory (consistent with smaller tunnel lengths within refluxing mutant ureters) (Supplemental Figure S2, online). These data indicate that Frs2αST−/− mice have an increased susceptibility and severity for VUR due to improper ureteral insertion and shortened intravesicular tunnel length resulting from ureteric induction defects.
Figure 5. Refluxing ureters in P1 Frs2αST−/− mice have improper ureteral insertion and shortened intravesicular tunnel lengths.
(a–b) Representative 3D reconstruction reveal similar bladder insertion points of both control ureters (a, blue) into the bladder (yellow), but a high and lateral insertion of the right Frs2αST−/− ureter (arrow) into the bladder. (c–d) Triangles connecting the external ureter (white) and internal (yellow) insertion points and the bladder neck (from panels a and b) illustrate similar insertion site angles in the control, indicating relatively parallel levels of insertion (c); however, the high and lateral refluxing Frs2αST−/− ureter (arrow) leads to triangles with very different insertion point angles (d). (e–f) Graphs confirm that compared to control and non-refluxing Frs2αST−/− mice, Frs2αST−/− mice with unilateral reflux have much larger differences in external (e) and internal (f) insertion site angles. (g) Graph revealing that mean intravesicular tunnel length (IVT) (visualized as purple lines in panels c and d) is significantly shorter in refluxing Frs2αST−/− ureters versus control and non-refluxing Frs2αST−/− ureters. (n) = 3. Values = Mean + SD. One-way ANOVA = *p<0.05, **p<0.01. Scale bar = 500μm.
VUR does not resolve with age in Frs2αST−/− mice
While primary VUR spontaneously resolves in most patients, approximately 33% have persistent reflux (16). To determine the natural history of VUR in Frs2αST−/− mice, we performed cystograms in young adult (P30) mice (Figure 6). At P30, 14% of control mice exhibit VUR, while 82% of Frs2αST−/− mice have reflux. (Table 2; p<0.01). While every control had unilateral VUR, 22% of the mutants had bilateral VUR (Table 3). Lastly, while none of the controls had high grade reflux, 18.0% of the mutants exhibited high grade VUR (Table 3). These data illustrate that high rates of VUR in Frs2αST−/− do not dissipate with age. Finally, while the Tbx18cre line is also likely deleting Frs2α in ureter and bladder mesenchyme, H&E stains of mutant ureters at P1 appear normal (see Supplemental Figure S2, online) and H&E stains and anti-aSMA stains of mutant bladders at P30 appear normal (see Supplemental Figure S3, online).
Figure 6. Cystograms in P30 Frs2αST−/− and control mice.
(a) Representative control showing dye in the bladder (bl), but not in either ureter (arrowheads). (b–c) Representative Frs2αST−/− mice with left, unilateral (B) and bilateral (c) reflux from the bladders (bl) into the ureters (arrowheads). Scale bar = 5mm.
Table 3.
Incidence and severity of VUR in P30 control and Frs2αST−/− mice
| Genotype | Incidence of Reflux | Reflux Laterality | Reflux Grade | |||
|---|---|---|---|---|---|---|
| Total | Male | Female | Unilateral | I–II | III–IV | |
| Control | 2/14 (14%) | 2/7 (29%) | 0/7 (0%) | 2/2 (100%) | 2/2 (100%) | NA |
| Frs2αST −/− | 9/11 (82%)* | 6/7 (86%) | 3/4 (75%) | 7/9 (78%) | 9/11 (82 %) | 2/11 (18%) |
p < 0.01 vs control
Discussion
Previous studies have examined the role of Fgfr2 in ureteric induction defects and risks for reflux. Using a transgenic Pax3cre mouse line to delete Fgfr2 in both the peri-Wolffian duct stroma and the metanephric mesenchyme, we observed ureteric induction defects and subsequent VUR as well as other anomalies such as duplex collecting systems and renal hypoplasia (7, 8). To resolve whether the effects were from the receptor actions in the peri-Wolffian duct stroma or the metanephric mesenchyme, we previously deleted Fgfr2 with a Tbx18cre mouse line that drives cre expression in the peri-Wolffian duct stroma and not in the metanephric mesenchyme. Results from this study demonstrated that Fgfr2 signaling in the peri-Wolffian duct stroma regulated the ureteric bud induction site, preventing VUR and other urinary tract anomalies (9). To elucidate which receptor signaling docking protein mediates the effects of Fgfr2 in the stroma, we utilized Tbx18 cre line to conditionally delete Frs2α.
Similar to the Tbx18cre; Fgfr2fl/fl line, Frs2αST−/− mice have ureteric bud induction defects, as evidenced by the extreme variability of mutant common nephric duct lengths (Wolffian duct segment between the ureteric bud base and the cloaca) compared to littermate controls. Apoptosis and proliferation were unaltered in Frs2αST−/− common nephric ducts versus control littermates suggesting the differences between the genotypes is not because of persistence of or premature loss of the CND. Moreover, the previous Tbx18cre; Fgfr2fl/fl study suggested that the molecular mechanism for ureteric induction defects is due to decreased Bmp4 expression and signaling in the peri-Wolffian duct stroma adjacent, as has been shown in Bmp4 heterozygous mice (9). Accordingly, we found an ~40% reduction in Bmp4 mRNA and pSmad 1/5/8 protein expression in Frs2αST−/− urogenital ridges compared to controls. While a one allele loss of Bmp4 has previously been shown to cause ureteric induction defects, these data are the first to suggest that this loss leads to a defect in canonical Bmp4 signaling (5). Taken together, these data strongly suggest that peri-Wolffian signaling through Fgfr2 is mediated mostly by Frs2α, which in turn drives expression and canonical signaling of Bmp4 to constrain the ureteric bud induction site..
The Frs2αST−/− ureteric bud induction defects led to aberrant ureter insertion into the bladder and high rates of VUR, as would be predicted by Drs. Mackie and Stephens studies in humans with VUR (4, 17). 3D reconstructions revealed that refluxing P1 mutant ureters inserted either high and lateral or low and medial into the bladder compared to the non-reflexing ureter. In addition, mean refluxing Frs2αST−/− intravesicular lengths were shortened in comparison with non-refluxing ureters, which is also a known risk for reflux in humans (4). Based on data from Tbx18cre; Fgfr2fl/fl and Tbx18cre; Frs2αfl/fl mice, the threshold intravesicular length for reflux appears ~ 200 μm or less (9). While Frs2α is also likely deleted in postnatal ureter and bladder mesenchymal derivatives (muscle and lamina propria), these structures appear normal in mutants (Supplemental Figures S2 and S3, online) and not likely reasons for VUR in the mutants. While we cannot exclude that the mutant ureters have peristalsis defects (which could be contributing to reflux in live mice), our cystogram assays are on euthanized mouse urinary tracts, none of which are undergoing peristalsis.
Notably, both control and Frs2αST−/− mice exhibited reflux during analysis, however the reflux observed in control mice was similar to that observed in P1 controls of other recent studies indicating a similar degree of VUR susceptibility in our controls as compared with previously published findings (8, 9). In comparing the current study to our previous studies, we observed some differences in P1 reflux rates between Fgfr2ST−/− mice and Frs2αST−/− mice (9). While control mice in both studies exhibit comparable rates of reflux, approximately 75% of P1 Fgfr2ST−/− mice had reflux compared to ~50% in Frs2αST−/− mice in this study (9). One explanation for the differences is that downstream docking proteins outside of Frs2α (such as Plc or Crk2) may have minor roles in mediating effects of Fgfr2 signaling in peri-Wolffian duct stroma. Other explanations include operator differences between the studies or some drift in the background strain between the Fgfr2 mutants in the previous study and the Frs2α mutants in the present study. Other studies from our laboratory using Six2cre lines to manipulate Fgfr and/or Frs2α expression in nephron progenitors show a slight rescue of nephron progenitor depletion in the latter mutants versus the former (12). In contrast, in the ureteric epithelium, Fgfr2 and Frs2α appear to act independently to regulate ureteric morphogenesis (13). Taken with the current study, Frs2α does appear to be the major docking protein for Fgfrs in metanephric and peri-Wolffian duct mesenchyme, but not in ureteric epithelium. It is not clear if this pattern extends to other developing mesenchymal and epithelial tissues.
Finally, unlike our previous studies, we examined the rates of VUR in Frs2αST−/− mice with age. Interestingly, we noted persistent, if not higher rates of VUR in mutants aged to P30. One explanation may be that we used different needle sizes for the P1 versus P30 (30 gauge versus 25 gauge, respectively); we and other investigators performing cystograms in mice have found that 25g needles fill the P1 bladders too quickly and generate very high pressures, whereas, 30g needles are too small to adequately fill the P30 bladders in a timely fashion (18, 19). In support of the “needle” hypothesis is that P30 control rates of reflux appear to be higher than at P1. Another potential explanation for the persistence/worsening of reflux in P30 mutants may be the effects of Tbx18cre-driven loss of Frs2α in bladder mesenchyme, which are yet to be determined. Thus the apparently higher rates of reflux in P30 Frs2αST−/− mice may be secondary to bladder defects. Given that the Frs2αST−/− mice have progressive limb defects, we have been unable to perform many assays to interrogate bladder function (such as void stain on paper). Regardless of the cause, the apparently persistent reflux in the Frs2α mutants raises interesting clinical questions, including whether perturbations/mutations in the Fgfr/Frs2α family may be linked to patients that have persistent reflux.
Supplementary Material
Acknowledgments
Financial Support: The study was supported by NIH grants R01 DK104374 (CMB) and P30 DK079307 (Tom Kleyman, PD; CMB co-I).
Footnotes
Disclosure: None of the authors have any financial ties with any portions of the study or any other conflicts of interest with the study.
References
- 1.Murawski IJ, Gupta IR. Gene discovery and vesicoureteric reflux. Pediatr Nephrol. 2008;23:1021–1027. doi: 10.1007/s00467-007-0704-y. [DOI] [PubMed] [Google Scholar]
- 2.Fillion ML, Watt CL, Gupta IR. Vesicoureteric reflux and reflux nephropathy: from mouse models to childhood disease. Pediatr Nephrol. 2014;29:757–766. doi: 10.1007/s00467-014-2761-3. [DOI] [PubMed] [Google Scholar]
- 3.Dressler GR. The cellular basis of kidney development. Annu Rev Cell Dev Biol. 2006;22:509–529. doi: 10.1146/annurev.cellbio.22.010305.104340. [DOI] [PubMed] [Google Scholar]
- 4.Mackie GG, Awang H, Stephens FD. The ureteric orifice: the embryologic key to radiologic status of duplex kidneys. J Pediatr Surg. 1975;10:473–481. doi: 10.1016/0022-3468(75)90187-6. [DOI] [PubMed] [Google Scholar]
- 5.Miyazaki Y, Oshima K, Fogo A, Hogan BL, Ichikawa I. Bone morphogenetic protein 4 regulates the budding site and elongation of the mouse ureter. J Clin Invest. 2000;105:863–873. doi: 10.1172/JCI8256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Poladia DP, Kish K, Kutay B, et al. Role of fibroblast growth factor receptors 1 and 2 in the metanephric mesenchyme. Dev Biol. 2006;291:325–339. doi: 10.1016/j.ydbio.2005.12.034. [DOI] [PubMed] [Google Scholar]
- 7.Hains D, Sims-Lucas S, Kish K, Saha M, McHugh K, Bates CM. Role of fibroblast growth factor receptor 2 in kidney mesenchyme. Pediatr Res. 2008;64:592–598. doi: 10.1203/PDR.0b013e318187cc12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hains DS, Sims-Lucas S, Carpenter A, et al. High incidence of vesicoureteral reflux in mice with Fgfr2 deletion in kidney mesenchyma. J Urol. 2010;183:2077–2084. doi: 10.1016/j.juro.2009.12.095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Walker KA, Sims-Lucas S, Di Giovanni VE, et al. Deletion of fibroblast growth factor receptor 2 from the peri-wolffian duct stroma leads to ureteric induction abnormalities and vesicoureteral reflux. PloS one. 2013;8:e56062. doi: 10.1371/journal.pone.0056062. [DOI] [PMC free article] [PubMed] [Google Scholar] [Research Misconduct Found]
- 10.Sims-Lucas S, Di Giovanni V, Schaefer C, Cusack B, Eswarakumar VP, Bates CM. Ureteric Morphogenesis Requires Fgfr1 and Fgfr2/Frs2alpha Signaling in the Metanephric Mesenchyme. J Am Soc Nephrol 2012. 2012;23(4):607–17. doi: 10.1681/ASN.2011020165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ong SH, Guy GR, Hadari YR, et al. FRS2 proteins recruit intracellular signaling pathways by binding to diverse targets on fibroblast growth factor and nerve growth factor receptors. Mol Cell Biol. 2000;20:979–989. doi: 10.1128/mcb.20.3.979-989.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Di Giovanni V, Walker KA, Bushnell D, et al. Fibroblast growth factor receptor-Frs2alpha signaling is critical for nephron progenitors. Dev Biol. 2015;400:82–93. doi: 10.1016/j.ydbio.2015.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sims-Lucas S, Cusack B, Eswarakumar VP, Zhang J, Wang F, Bates CM. Independent roles of Fgfr2 and Frs2alpha in ureteric epithelium. Development. 2011;138:1275–1280. doi: 10.1242/dev.062158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang Y, Tripathi P, Guo Q, Coussens M, Ma L, Chen F. Cre/lox recombination in the lower urinary tract. Genesis. 2009;47:409–413. doi: 10.1002/dvg.20515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lebowitz RL, Olbing H, Parkkulainen KV, Smellie JM, Tamminen-Mobius TE. International system of radiographic grading of vesicoureteric reflux. International Reflux Study in Children. Pediatr radiol. 1985;15:105–109. doi: 10.1007/BF02388714. [DOI] [PubMed] [Google Scholar]
- 16.Peters CA, Skoog SJ, Arant BS, Jr, et al. Summary of the AUA Guideline on Management of Primary Vesicoureteral Reflux in Children. J Urol. 2010;184:1134–1144. doi: 10.1016/j.juro.2010.05.065. [DOI] [PubMed] [Google Scholar]
- 17.Mackie GG, Stephens FD. Duplex kidneys: a correlation of renal dysplasia with position of the ureteral orifice. J Urol. 1975;114:274–280. doi: 10.1016/s0022-5347(17)67007-1. [DOI] [PubMed] [Google Scholar]
- 18.Boualia SK, Gaitan Y, Murawski I, Nadon R, Gupta IR, Bouchard M. Vesicoureteral reflux and other urinary tract malformations in mice compound heterozygous for Pax2 and Emx2. PloS one. 2011;6:e21529. doi: 10.1371/journal.pone.0021529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Murawski IJ, Watt CL, Gupta IR. Vesico-ureteric reflux: using mouse models to understand a common congenital urinary tract defect. Pediatr Nephrol. 2011;26(9):1513–22. doi: 10.1007/s00467-011-1821-1. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.