Abstract
Purpose
To determine the clinical relevance of vitreous biomarkers in patients with uveitis.
Design
Multicentre, prospective, observational study.
Setting
Uveitis outpatient clinics of two academic medical centres in Japan.
Patient population
This study included 234 eyes of 191 patients with various uveitis aetiologies: definitive sarcoidosis (61 eyes of 46 patients), suspected sarcoidosis (60 eyes of 45 patients), intraocular tumour (34 eyes of 27 patients), viral infection (20 eyes of 18 patients), non-sarcoidosis (16 eyes of 16 patients) and unknown aetiology (43 eyes of 39 patients).
Observation procedure
Vitreous samples (taken by pars planta vitrectomy) were analysed with flow cytometry, cytology and multiplex PCR analysis.
Main outcome measures
The primary outcome measures were the diagnostic values of various biomarkers (T cells, B cells and pathogen DNA) in vitreous samples. The secondary outcome was visual acuity after vitrectomy.
Results
Sarcoidosis showed higher CD4/CD8 or CD4+ measurements than other aetiologies (p<0.01). In samples with viral infection, pathogen DNA was detected, and CD8+ counts were higher than the other aetiologies (p<0.01). Eyes with tumour had higher CD19+ (p<0.05). Non-sarcoidosis had lower CD4/CD8 than sarcoidosis, higher CD8+ than sarcoidosis and lower CD19+ than tumour (p<0.01). Unknown uveitis had lower CD4/CD8 than sarcoidosis (p<0.01), and higher CD4/CD8 than non-sarcoidosis, viral infection or tumour (p<0.001). Visual acuity improved after vitrectomy (p<0.001).
Conclusions
Uveitis aetiologies had distinct vitreous biomarker profiles, especially of infiltrating lymphocytes. Analyses of CD4/CD8 ratio, T-lymphocyte and B-lymphocyte subset, and pathogen DNA in vitreous samples have good safety profiles and high diagnostic value for uveitis classification.
Trial registration number
UMIN000004980; Pre-results.
Keywords: uveitis, vitreous sample, cd4/cd8 ratio, flowcytometry analysis
Strengths and limitations of this study.
Both flow cytometric and cytological analyses of the vitreous fluid are useful adjuncts to standard diagnostic methods for uveitis and intraocular tumours.
Detecting the DNA of pathogens in vitreous specimens with multiplex PCR analysis is a beneficial method for diagnosing infectious uveitis.
Combined cell and pathogen DNA analyses have the potential to become the new gold standard for diagnosing uveitis.
As this study only examined a limited number of patients with viral, tuberculosis and other types of infections, a further investigation will need to collect and analyse additional vitreous samples from patients with these types of uveitis.
The system described here is unsuitable for use in patients with high-grade endophthalmitis or in patients with uveitis with few cells infiltrating the vitreous.
Introduction
In Japan, uveitis is considered to consist of several immune-mediated diseases, including sarcoidosis, Behçet’s disease and Vogt-Koyanagi-Harada disease.1 Infectious diseases are also recognised as being able to cause uveitis. However, in order to determine the cause of uveitis, it is important that both a detailed medical history and a thorough physical examination are conducted. Although one of the best ways to diagnose uveitis is to observe the disease course, in many cases the diagnoses are made without clinical examinations or evaluations of the laboratory data from patient specimens. As a result, this can lead to incorrect diagnoses. In fact, approximately 30% of all of the uveitis cases found in the Japanese population have been diagnosed as idiopathic uveitis.1
Various types of immune cells have been found in the vitreous fluid of patients with uveitis. Thus, the type of uveitis diagnosed will depend on the specific populations found in the vitreous fluid of the patient. A previous study reported finding a high number of CD4+ and CD8+ cells in the vitreous cell population in eyes with acute retinal necrosis (ARN) induced by herpes simplex virus type 1 (HSV-1) or varicella zoster virus (VZV). Flow cytometry analysis has also been reported to be useful in diagnosing primary intraocular lymphoma (PIOL)2 and other types of uveitis, especially Vogt-Koyanagi-Harada disease.3 4 In our previous study, we demonstrated that vitreous analysis was beneficial for diagnosing ocular sarcoidosis, as the sensitivity and specificity of the vitreal CD4/CD8 ratio with cut-off points of 3.5 were 100% and 96.3%, respectively.4 Based on these previous findings, we believe that vitreous specimens may be one of the most important and informative factors for diagnosing uveitis.
The purpose of this study was to examine the different types of the known uveitis and then attempt to determine whether there was variation in the cell populations and pathogen DNA in the eyes of these patients, thereby enabling the creation of a more precise classification of unknown uveitis.
Methods
Design and patients
All invasive and therapeutic procedures were conducted in accordance with the tenets set forth in the Declaration of Helsinki. This study was registered with the University Hospital Medical Information Network Clinical Trial Registry. The purpose of the research, and the invasive and therapeutic protocols were explained in detail to all of the patients. In all cases, informed consent was obtained prior to the patients taking part in the study. This multicentre study was prospectively approved by the institutional review boards of both Tohoku University Graduate School of Medicine and the Kyoto Prefectural University of Medicine.
Patients in this prospective observational study who visited the Uveitis Outpatient Clinic at Tohoku University Hospital or the University Hospital at Kyoto Prefectural University of Medicine and met all of the inclusion criteria were invited to participate. All subjects were uveitis patients with visual disturbances due to the prolonged presence of vitreous opacities, cystoid macular oedemas or epiretinal membranes that were refractory to conventional steroid therapy, such as oral, topical or sub-Tenon’s injections. All patients agreed to undergo pars plana vitrectomy and take part in the study. Patients were excluded from the uveitis group if they had any prior history of intraocular surgery, especially vitreous surgery.
Categorisation of patients
Subjects in the study were divided into six groups. The categories included patients who (1) were diagnosed with sarcoidosis, (2) were suspected of having sarcoidosis, (3) had an intraocular tumour, (4) had a viral infection, (5) had non-sarcoidosis uveitis or (6) had uveitis of an unknown aetiology (table 1).
Table 1.
Patients details
| Disease (cases) | Number (case/eye) | Age (mean±SD) | Gender (M:F) |
| D-S | |||
| Skin biopsy: 16 | |||
| Lung biopsy: 15 | 46/61 | 67.0±8.6 | 6:40 |
| Clinical manifestation: 15 | |||
| S-S | 45/60 | 67.6±6.5 | 5:40 |
| Tumour/PIOL | |||
| DLBCL: 23 | |||
| T-LGL leukaemia: 2 | 27/34 | 67.4±14.5 | 13:14 |
| ATL: 1 | |||
| NK: 1 | |||
| Viral infection | |||
| VZV: 4 | |||
| HSV-1: 2 | |||
| HSV-2: 2 | 18/20 | 55.4±16.8* | 11:7 |
| CMV: 7 | |||
| HTLV-1: 3 | |||
| Non-sarcoidosis | |||
| Endophthalmitis: 10 | |||
| Behçet: 2 | 16/16 | 66.8±9.5 | 9:7 |
| Toxocara: 2 | |||
| Toxoplasma: 2 | |||
| Unknown | 39/43 | 61.6±2.9 | 14:25 |
| 191/234 | *V: D-S, V: S-S (p<0.05) | 47:118 |
All patients underwent vitreous analysis.
ATL, adult T-cell lymphoma; CMV, cytomegalovirus; DLBCL, diffuse large B-cell lymphoma; D-S, definitive sarcoidosis; HTLV-1, human T-cell leukemia virus; HSV, herpes simplex virus; NK, natural killer cell tumour; S-S, suspected sarcoidosis; PIOL, primary intraocular lymphoma; T-LGL, T-cell large granular lymphocyte leukemia; VZV, varicella zoster virus.
The sarcoidosis group was defined according to the international criteria for the diagnosis of ocular sarcoidosis established by the International Workshop on Ocular Sarcoidosis.5 Patients in the intraocular tumour group included those with diffuse, large B-cell lymphomas, adult T-cell leukaemia, natural killer cell lymphoma and T-cell large granular lymphocyte leukaemia. The viral infection group consisted of patients with HSV-1, HSV-2, VZV, cytomegalovirus (CMV) and human T-cell leukaemia virus type 1 (HTLV-1). The non-sarcoidosis group consisted of patients with Toxoplasma, Toxocara, fungal infection, bacterial infection and Behçet’s disease. Patients were classified as having uveitis of unknown aetiology when they failed to satisfy the criteria for any of the above disease classifications after several types of clinical examination.
Sample collection (vitrectomy) procedures
A vitreous specimen was obtained from each patient at the start of the conventional 25-gauge pars plana vitrectomy. The procedure used a CV-24000 (NIDEK Co., Aichi, Japan) and an Accurus (Alcon Laboratories, Fort Worth, Texas, USA) or Constellation (Alcon) vitrectomy system. Our current experimental protocol was based on a method that we have previously reported.4 After all of the obtained samples had been promptly stored at 4°C, they were brought to the clinical laboratory of the university hospital for cell analysis (including flow cytometry).
Cell analysis of vitreous samples
Using a previously reported method,4 the vitreous samples were filtered with a 70 µm cell strainer (BD Falcon Cell Strainer; BD Biosciences, Bedford, Massachusetts, USA). All of the samples contained at least 100 lymphocytes, specifically the CD45+/SS low cells in each tube. All samples were then washed and resuspended with phosphate-buffered saline (Nissui, Tokyo, Japan) containing 2% bovine serum albumin (Nacalai Tesque, Kyoto, Japan) and 0.1% sodium azide (Nacalai Tesque) at a final volume of 0.5 mL. For the flow cytometric analysis, 0.1 mL of each sample was incubated with one of the previously described monoclonal antibody mixtures.4 Lymphocyte subsets of the vitreous samples were examined with an Epics XL-MCL Flow Cytometer (Beckman Coulter, Brea, California, USA). Moreover, when we suspected a patient of having lymphoma, we submitted their information to a registration, examination, analysis and description (READ) system that had been previously established to diagnose lymphoma within our hospital.6
Real-time PCR analysis of vitreous samples
Genomic DNA of the human herpes virus in the vitreous was measured using two previously described independent PCR assays.7–9 These assays were also designed to measure other ocular pathogens.9 PCR was performed using a LightCycler (Roche Diagnostics, Schweiz AG, Rotkreuz, Switzerland). Primers and probes for the pathogens and PCR conditions have been previously described.7 When more than 50 copies/tube (5000 copies/mL) were detected, the value of the viral copy number in the sample was considered significant. In the PIOL group, the READ system detected the immunoglobulin heavy chain (IgH) gene rearrangement of the B-cell lymphoma in the vitreous fluid.
Statistical analysis
Statistical comparisons between each disease group for the CD4/CD8 ratios, CD4+, CD8+ and CD19+ populations were performed by a Kruskal-Wallis test with Dunn’s multiple comparisons test. A p value of <0.05 was considered statistically significant, and effect size: d (ESd) was calculated: Absolute difference in mean value (Mean Group A−Mean Group B) /((SD Group A+SD Group B)/2) (see online supplementary file CI). The performance of the CD4/CD8 ratio, CD8+ and CD19+ populations and their cut-off points were assessed using the receiver operator curve (ROC) space. The comparison of the visual acuity preoperation and postoperation was performed with a Wilcoxon matched-pair signed-rank test. Statistical significance was set at p<0.05. All analyses were performed using Prism software V.5.0.1 (Graph Pad Software).
bmjopen-2016-014549supp001.pdf (48.1KB, pdf)
Results
Comparison of flow cytometric analysis of vitreous fluid in all cases
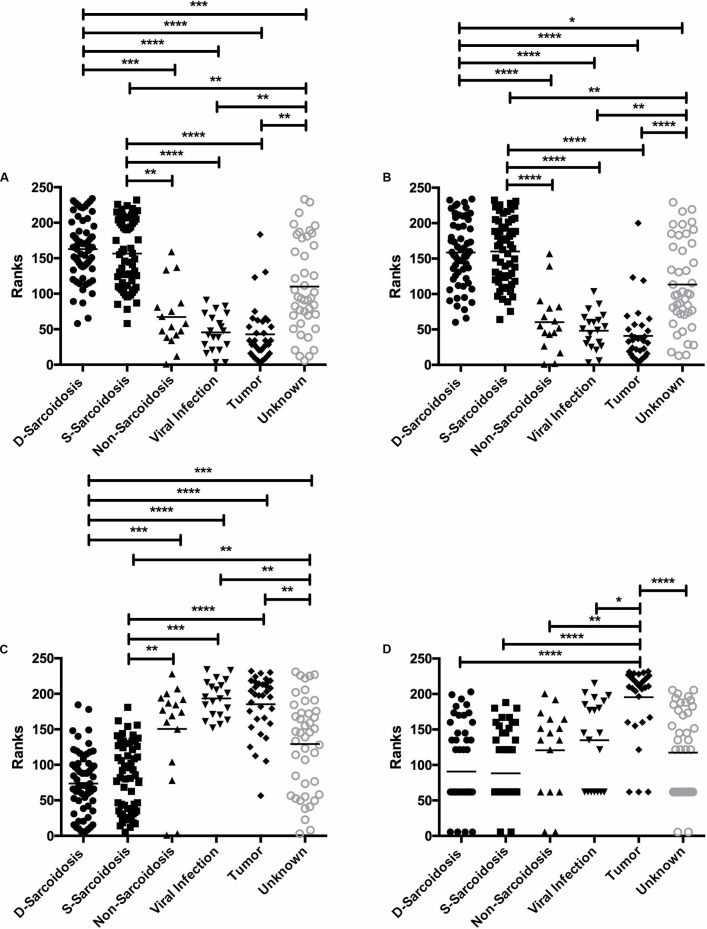
Figure 1A and table 2 show the CD4/CD8 lymphocyte ratios in vitreous samples from the patient population. The vitreous samples of eyes with definitive sarcoidosis had higher CD4/CD8 lymphocyte ratios compared with vitreous samples of non-sarcoidosis (p<0.001; ESd=0.5), viral infection (p<0.001; ESd=0.6), tumour (p<0.001; ESd=0.6) and unknown aetiology (p<0.01, ESd=0.04) (figure 1A). Similarly, the vitreous samples of eyes with suspected sarcoidosis had higher CD4/CD8 lymphocyte ratios compared with vitreous samples of non-sarcoidosis (p<0.001; ESd=1.1), viral infection (p<0.001; ESd=1.3), tumour (p<0.001; ESd=1.2) and unknown aetiology (p<0.01; ESd:0.1). However, we did not find any significant difference of CD4/CD8 ratio between the definitive and suspected sarcoidosis groups (p>0.999; ESd=0.2).
Figure 1.
Ranks: Kruskal-Wallis test of the (A) CD4/CD8 ratio, (B) CD4, (C) CD8 and (D) CD19 in the vitreous samples from several types of uveitis. Significant differences in each group are indicated by the asterisks **** or *** p<0.001, **p<0.01, *p<0.05. D, definitive sarcoidosis; S, suspected sarcoidosis.
Table 2.
CD4/CD8 lymphocyte ratios in vitreous samples
| n | Mean | SD | Difference in average value | Effect size: d | p Value | |
| D-sarcoidosis | 61 | 38.12 | 124.90 | 16.99 | 0.22 | >0.9999 |
| S-sarcoidosis | 60 | 21.13 | 29.94 | |||
| D-sarcoidosis | 61 | 38.12 | 124.90 | 34.86 | 0.54 | <0.0001 |
| Non-sarcoidosis | 16 | 3.26 | 3.84 | |||
| D-sarcoidosis | 61 | 38.12 | 124.90 | 36.64 | 0.58 | <0.0001 |
| Viral infection | 20 | 1.48 | 1.07 | |||
| D-sarcoidosis | 61 | 38.12 | 124.90 | 36.25 | 0.57 | <0.0001 |
| Tumour | 34 | 1.87 | 3.37 | |||
| D-sarcoidosis | 61 | 38.12 | 124.90 | 5.85 | 0.04 | 0.0014 |
| Unknown | 43 | 32.27 | 146.80 | |||
| S-sarcoidosis | 60 | 21.13 | 29.94 | 17.87 | 1.06 | <0.0001 |
| Non-sarcoidosis | 16 | 3.26 | 3.84 | |||
| S-sarcoidosis | 60 | 21.13 | 29.94 | 19.65 | 1.27 | <0.0001 |
| Viral infection | 20 | 1.48 | 1.07 | |||
| S-sarcoidosis | 60 | 21.13 | 29.94 | 19.26 | 1.16 | <0.0001 |
| Tumour | 34 | 1.87 | 3.37 | |||
| S-sarcoidosis | 60 | 21.13 | 29.94 | 11.14 | 0.13 | 0.0086 |
| Unknown | 43 | 32.27 | 146.80 | |||
| Non-sarcoidosis | 16 | 3.26 | 3.84 | 1.78 | 0.73 | >0.9999 |
| Viral infection | 20 | 1.48 | 1.07 | |||
| Non-sarcoidosis | 16 | 3.26 | 3.84 | 1.39 | 0.39 | >0.9999 |
| Tumour | 34 | 1.87 | 3.37 | |||
| Non-sarcoidosis | 16 | 3.26 | 3.84 | 29.01 | 0.39 | 0.4573 |
| Unknown | 43 | 32.27 | 146.80 | |||
| Viral infection | 20 | 1.48 | 1.07 | 0.39 | 0.18 | >0.9999 |
| Tumour | 34 | 1.87 | 3.37 | |||
| Viral infection | 20 | 1.48 | 1.07 | 30.79 | 0.42 | 0.0063 |
| Unknown | 43 | 32.27 | 146.80 | |||
| Tumour | 34 | 1.87 | 3.37 | 30.40 | 0.40 | 0.0002 |
| Unknown | 43 | 32.27 | 146.80 |
Figure 1B and table 3 show the CD4+ lymphocyte populations in vitreous samples from the patient population. Vitreous samples of definitive sarcoidosis had higher CD4+ lymphocyte populations than samples from patients with non-sarcoidosis (p<0.001; ESd=2.1), viral infection (p<0.001; ESd=2.9), tumour (p<0.001, ESd=3.2) and unknown aetiology (p<0.05; ESd=1.0). Similarly, suspected sarcoidosis also showed higher vitreous CD4+ lymphocyte populations compared with non-sarcoidosis (p<0.001; ESd=2.2), viral infection (p<0.001, ESd=3.0), tumour (p<0.001, ESd=3.3) and unknown aetiology (p<0.01; ESd=1.0). However, we could not find any significant difference of CD4+ population between definitive and suspected sarcoidosis groups (p>0.999; ESd=0.1).
Table 3.
CD4+ lymphocyte ratios in vitreous samples
| n | Mean | SD | Difference in average value | Effect size: d | p Value | |
| D-sarcoidosis | 61 | 87.27 | 8.28 | 0.44 | 0.06 | >0.9999 |
| S-sarcoidosis | 60 | 87.71 | 7.37 | |||
| D-sarcoidosis | 61 | 87.27 | 8.28 | 35.74 | 2.08 | <0.0001 |
| Non-sarcoidosis | 16 | 51.53 | 26.15 | |||
| D-sarcoidosis | 61 | 87.27 | 8.28 | 39.07 | 2.85 | <0.0001 |
| Viral infection | 20 | 48.20 | 19.16 | |||
| D-sarcoidosis | 61 | 87.27 | 8.28 | 47.68 | 3.15 | <0.0001 |
| Tumour | 34 | 39.59 | 22.03 | |||
| D-sarcoidosis | 61 | 87.27 | 8.28 | 14.07 | 0.96 | 0.0122 |
| Unknown | 43 | 73.20 | 21.02 | |||
| S-sarcoidosis | 60 | 87.71 | 7.37 | 36.18 | 2.16 | <0.0001 |
| Non-sarcoidosis | 16 | 51.53 | 26.15 | |||
| S-sarcoidosis | 60 | 87.71 | 7.37 | 39.51 | 2.98 | <0.0001 |
| Viral infection | 20 | 48.20 | 19.16 | |||
| S-sarcoidosis | 60 | 87.71 | 7.37 | 48.12 | 3.27 | <0.0001 |
| Tumour | 34 | 39.59 | 22.03 | |||
| S-sarcoidosis | 60 | 87.71 | 7.37 | 14.51 | 1.02 | 0.0083 |
| Unknown | 43 | 73.20 | 21.02 | |||
| Non-sarcoidosis | 16 | 51.53 | 26.15 | 3.33 | 0.15 | >0.9999 |
| Viral infection | 20 | 48.20 | 19.16 | |||
| Non-sarcoidosis | 16 | 51.53 | 26.15 | 11.94 | 0.50 | >0.9999 |
| Tumour | 34 | 39.59 | 22.03 | |||
| Non-sarcoidosis | 16 | 51.53 | 26.15 | 21.67 | 0.92 | 0.111 |
| Unknown | 43 | 73.20 | 21.02 | |||
| Viral infection | 20 | 48.20 | 19.16 | 8.61 | 0.42 | >0.9999 |
| Tumour | 34 | 39.59 | 22.03 | |||
| Viral infection | 20 | 48.20 | 19.16 | 25.00 | 1.24 | 0.0056 |
| Unknown | 43 | 73.20 | 21.02 | |||
| Tumour | 34 | 39.59 | 22.03 | 33.61 | 1.56 | <0.0001 |
| Unknown | 43 | 73.20 | 21.02 |
Figure 1C and table 4 show the CD8+ lymphocyte populations in vitreous samples from the patient population. Vitreous samples from patients with uveitis with viral infections had higher CD8+ lymphocyte populations than samples from patients with definitive sarcoidosis (p<0.01; ESd=2.9), suspected sarcoidosis (p<0.01; ESd=2.8) and uveitis of unknown aetiology (p<0.01; ESd=1.1). Similarly, vitreous samples from patients with intraocular tumour had higher CD8+ lymphocyte populations than samples from patients with definitive sarcoidosis (p<0.01; ESd=2.6), suspected sarcoidosis (p<0.01; ESd d=2.4) and uveitis of unknown aetiology (p<0.01; ESd=0.9). However, we did not find any significant differences of CD8+ population between the tumour and non-sarcoidosis patients (p>0.999; ESd=0.6), or between the viral infection and non-sarcoidosis patients (p=0.88; ESd=0.8).
Table 4.
CD8 lymphocyte ratios in vitreous samples
| n | Mean | SD | Difference in average value | Effect size: d | p Value | |
| D-sarcoidosis | 61 | 7.81 | 6.50 | 1.25 | 0.19 | >0.9999 |
| S-sarcoidosis | 60 | 9.06 | 6.83 | |||
| D-sarcoidosis | 61 | 7.81 | 6.50 | 21.03 | 1.67 | 0.0008 |
| Non-sarcoidosis | 16 | 28.84 | 18.69 | |||
| D-sarcoidosis | 61 | 7.81 | 6.50 | 35.37 | 2.89 | <0.0001 |
| Viral infection | 20 | 43.18 | 17.97 | |||
| D-sarcoidosis | 61 | 7.81 | 6.50 | 32.47 | 2.56 | <0.0001 |
| Tumour | 34 | 40.28 | 18.87 | |||
| D-sarcoidosis | 61 | 7.81 | 6.50 | 14.84 | 1.12 | 0.0006 |
| Unknown | 43 | 22.65 | 20.01 | |||
| S-sarcoidosis | 60 | 9.06 | 6.83 | 19.78 | 1.55 | 0.0039 |
| Non-sarcoidosis | 16 | 28.84 | 18.69 | |||
| S-sarcoidosis | 60 | 9.06 | 6.83 | 34.12 | 2.75 | <0.0001 |
| Viral infection | 20 | 43.18 | 17.97 | |||
| S-sarcoidosis | 60 | 9.06 | 6.83 | 31.22 | 2.43 | <0.0001 |
| Tumour | 34 | 40.28 | 18.87 | |||
| S-sarcoidosis | 60 | 9.06 | 6.83 | 13.59 | 1.01 | 0.0055 |
| Unknown | 43 | 22.65 | 20.01 | |||
| Non-sarcoidosis | 16 | 28.84 | 18.69 | 14.34 | 0.78 | 0.8786 |
| Viral infection | 20 | 43.18 | 17.97 | |||
| Non-sarcoidosis | 16 | 28.84 | 18.69 | 11.44 | 0.61 | >0.9999 |
| Tumour | 34 | 40.28 | 18.87 | |||
| Non-sarcoidosis | 16 | 28.84 | 18.69 | 6.19 | 0.32 | >0.9999 |
| Unknown | 43 | 22.65 | 20.01 | |||
| Viral infection | 20 | 43.18 | 17.97 | 2.90 | 0.16 | >0.9999 |
| Tumour | 34 | 40.28 | 18.87 | |||
| Viral infection | 20 | 43.18 | 17.97 | 20.53 | 1.08 | 0.0068 |
| Unknown | 43 | 22.65 | 20.01 | |||
| Tumour | 34 | 40.28 | 18.87 | 17.63 | 0.91 | 0.0045 |
| Unknown | 43 | 22.65 | 20.01 |
Figure 1D and table 5 show CD19+ lymphocyte populations of vitreous samples in the patient population. The vitreous samples from patients with intraocular tumour had higher CD19+ lymphocyte populations than samples from patients with definitive sarcoidosis (p<0.001; ESd=1.9), suspected sarcoidosis (p<0.001; ESd=1.9), viral infection (p<0.05; ESd=1.6), non-sarcoidosis (p<0.001; ESd=1.9) and uveitis of unknown aetiology (p<0.001; ESd=1.8).
Table 5.
CD19 lymphocyte ratios in vitreous samples
| n | Mean | SD | Difference in average value | Effect size: d | p Value | |
| D-sarcoidosis | 61 | 0.42 | 0.82 | 0.16 | 0.26 | >0.9999 |
| S-sarcoidosis | 60 | 0.27 | 0.37 | |||
| D-sarcoidosis | 61 | 0.42 | 0.82 | 0.28 | 0.31 | >0.9999 |
| Non-sarcoidosis | 16 | 0.70 | 0.95 | |||
| D-sarcoidosis | 61 | 0.42 | 0.82 | 1.42 | 0.63 | 0.1123 |
| Viral infection | 20 | 1.84 | 3.69 | |||
| D-sarcoidosis | 61 | 0.42 | 0.82 | 29.06 | 1.89 | <0.0001 |
| Tumour | 34 | 29.48 | 29.92 | |||
| D-sarcoidosis | 61 | 0.42 | 0.82 | 0.54 | 0.46 | 0.6018 |
| Unknown | 43 | 0.96 | 1.53 | |||
| S-sarcoidosis | 60 | 0.27 | 0.37 | 0.43 | 0.65 | >0.9999 |
| Non-sarcoidosis | 16 | 0.70 | 0.95 | |||
| S-sarcoidosis | 60 | 0.27 | 0.37 | 1.57 | 0.77 | 0.0717 |
| Viral infection | 20 | 1.84 | 3.69 | |||
| S-sarcoidosis | 60 | 0.27 | 0.37 | 29.21 | 1.93 | <0.0001 |
| Tumour | 34 | 29.48 | 29.92 | |||
| S-sarcoidosis | 60 | 0.27 | 0.37 | 0.69 | 0.73 | 0.3762 |
| Unknown | 43 | 0.96 | 1.53 | |||
| Non-sarcoidosis | 16 | 0.70 | 0.95 | 1.14 | 0.49 | >0.9999 |
| Viral infection | 20 | 1.84 | 3.69 | |||
| Non-sarcoidosis | 16 | 0.70 | 0.95 | 28.78 | 1.86 | 0.0017 |
| Tumour | 34 | 29.48 | 29.92 | |||
| Non-sarcoidosis | 16 | 0.70 | 0.95 | 0.26 | 0.21 | >0.9999 |
| Unknown | 43 | 0.96 | 1.53 | |||
| Viral infection | 20 | 1.84 | 3.69 | 27.64 | 1.64 | 0.0113 |
| Tumour | 34 | 29.48 | 29.92 | |||
| Viral infection | 20 | 1.84 | 3.69 | 0.88 | 0.34 | >0.9999 |
| Unknown | 43 | 0.96 | 1.53 | |||
| Tumour | 34 | 29.48 | 29.92 | 28.52 | 1.81 | <0.0001 |
| Unknown | 43 | 0.96 | 1.53 |
Results of the multiplex PCR analysis of the vitreous samples
In the viral infection group, multiplex PCR detected VZV DNA in four patients, while HSV-2 infection was found in two patients, HSV-1 infection was found in two patients, CMV infection was found in seven patients, HTLV-1 infection was found in three patients and bacterial 16S rRNA was found in ten patients. Toxoplasma DNA was detected in two patients (table). Moreover, although PCR analysis did not detect Toxocara DNA, Toxocara serology10 using vitreous samples and peripheral blood determined two positive results. In patients with PIOL, IgH gene rearrangement was detected.
Sensitivity and specificity
We calculated the sensitivity and specificity of the CD4/CD8 ratio of the vitreous in order to detect each type of sarcoidosis, including both definitive sarcoidosis and suspected sarcoidosis, and then compared the findings with the other types of uveitis, including intraocular tumour, viral infection, non-sarcoidosis and uveitis of an unknown aetiology. The ROC space was used to assess the performance of the CD4/CD8 ratio and its cut-off point. When the CD4/CD8 ratio was more than 4.60 in the patients with sarcoidosis, the specificity was 76.99% and the sensitivity was 92.56% (area under the curve (AUC): 0.87) (figure 2A). We also calculated the sensitivity and specificity of the CD8+ population of the vitreous in order to detect viral infection and compare the findings with the other types of uveitis, including sarcoidosis, tumour, non-sarcoidosis and immune-mediated uveitis. The performance of the CD8+ population and its cut-off point were assessed using the ROC space. When the CD8+ population was more than 28.25 in the patients with viral infection, the specificity was 77.10% and the sensitivity was 75.00% (AUC: 0.86) (figure 2b). Moreover, when the CD19+ population was more than 3.10 in the patients with PIOL and tumour, the specificity was 95.43% and the sensitivity was 74.29% (AUC: 0.90) (figure 2c).
Figure 2.
Performance of the (A) CD4/CD8 ratios, (B) CD8+ and (C) CD19+ in the vitreous samples for the purpose of discriminating sarcoidosis and other uveitis groups in (A) receiver operator curve space; (B) performance of the CD8+ population in vitreous samples for the purpose of discriminating between the viral infection group and the other uveitis groups in the receiver operator curve space; (C) performance of CD19+ population in the vitreous samples for the purpose of discriminating between the tumour group and other uveitis groups in the receiver operator curve space. Comparisons of the diagnostic performance of the vitreal CD4/CD8 ratio, CD8+ population and CD19+ population were performed by plotting their performance results in the receiver operator curve space.
Clinical course of patients
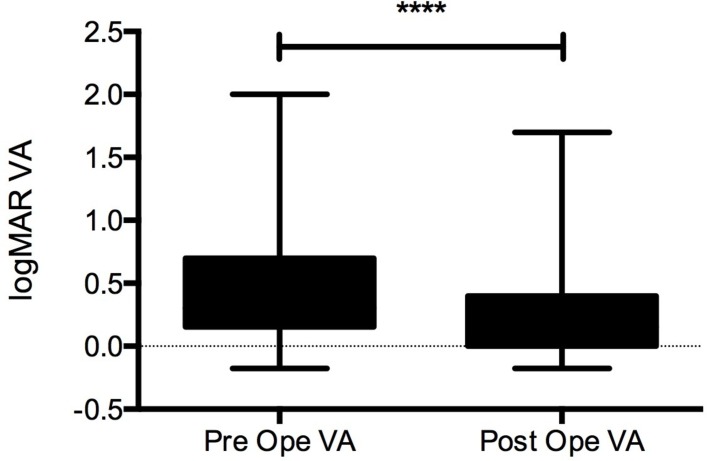
Visual acuity in most of the patients in this study improved between preoperation (0.46±0.03) and postoperation (0.27±0.02, p<0.0001) (figure 3). After the vitrectomy, 22 patients gained visual acuity including in the 5 patients with definitive sarcoidosis, 3 patients with suspected sarcoidosis, 3 patients with tumour/PIOL, 4 patients with viral infection, 1 patient with bacterial infection and in the 6 patients with uveitis of unknown aetiology. These patients had severe macular oedema, retinal vessel occlusion and retinal necrosis. Treatment for adverse events such as recurrent retinal detachment was only performed in four patients (one patient with ARN, one patient with Toxoplasma infection, one patient with endophthalmitis and one patient with tumour).
Figure 3.
Quantitative analysis of the preoperative and postoperative VA. Significant differences were observed (****p<0.001). VA, visual acuity.
Discussion
The present report describes unique immunological features of the vitreous T-lymphocyte and B-lymphocyte populations and provides information on the detectible types of microbial DNA that are found in patients with various types of uveitis. Since the cells that infiltrate the ocular fluid reflect the patient’s pathological condition, the extensive analysis described in this report will help to support the development of new diagnostic tools that can be used to identify underlying disease that causes uveitis. Moreover, determination of the characteristics of the cells and detectible types of microbial DNA found in the vitreous fluid in patients with uveitis may be able to reveal new biomarkers, thereby improving our ability to classify these diseases. Furthermore, with some exceptions, the clinical course was favourable in almost all of the patients involved in this study. In addition, there were no severe adverse events associated with any of the surgical procedures.
Davis et al examined flow cytometry in two previous studies and found that the procedure could be used to quantify the percentages and ratios of vitreous cell types. As a result, these findings made it possible to differentiate between intraocular lymphoma and immunologically mediated uveitis.2 3 Our study results demonstrated that there was a high CD4/CD8 T-cell ratio in the vitreous cavity of patients with sarcoidosis. It was reported that higher CD4/CD8 ratio in bronchoalveolar lavage fluid strongly implicate sarcoidosis, and the T-cell population is one of the critical factors for diagnosing sarcoidosis. Our previous data indicated that patients with sarcoidosis who received bronchoalveolar lavage fluid and vitrectomy during their clinical course had the same T-cell profiles (high CD4/CD8 ratio) in both samples. Moreover, Goto et al reported that non-caseous granuloma existed in epiretinal membrane tissues of patients with sarcoidosis.11 From those points of view, the pathological lesions in both lung and ocular tissue may have the same aetiology. Thus, high CD4/CD8 T-cell ratios may be a potential biomarker that can be used during the diagnosis of sarcoidosis. In fact, in our previous study, we found that a diagnosis of ocular sarcoidosis based on a CD4/CD8 T-cell ratio of vitreous-infiltrating lymphocytes greater than 3.5 exhibited a sensitivity of 100% and a specificity of 96.3%.4
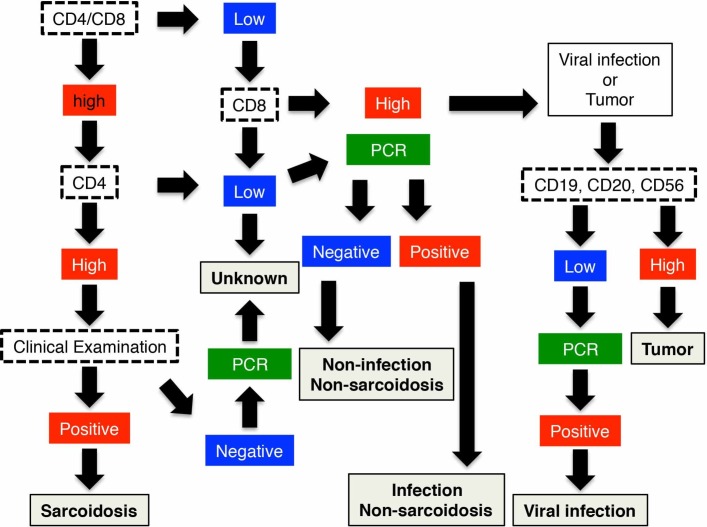
Due to the large number of examinations performed in the current study, this led to our further observation that there was a characteristic T-cell population in the vitreous fluid of the viral infection group. In fact, with the exception of the tumour group, the CD8+ cell population was significantly higher in the viral infection group compared with all of the other uveitis patient groups. A previous study has also reported finding a robust CD8+-cell population in humans during the acute phase of a viral infection.12 Conventional wisdom has been that the CD8+-cell response plays a major role in the antiviral immunity.12 During the proliferation period, there is recruitment of more CD8+ cells. Subsequently, large quantities of inflammatory cytokines, such as tumour necrosis factor (TNF)-alpha and interferon gamma, are produced by these activated CD8+ cells. Thus, the CD8+ cells play a crucial role in viral infections. However, our finding that there was a high number of CD8 T cells in the vitreous body in the tumour group was unexpected. A previous experimental study reported that biological treatments for tumours created a memory system for peripheral lymphoma.13 Thus, this may explain the reason why there was a high CD8 T-cell population in the vitreous body in the tumour group. When differentiating between viral infection and tumour, the population of CD19+ or other tumour cells in the vitreous fluid may be a useful marker of lymphoma.6 14 15 In fact, the cytological analysis in our previous study revealed that there were characteristic tumour cells, such as flower cells, found in patients with acute T-cell leukaemia.15 Multiplex PCR analysis has also been shown to be an effective and useful tool for diagnosing infectious uveitis.9 Therefore, when evaluating patient vitreous samples, the combination of cell analysis and multiplex PCR examination is considered to be one of the most powerful diagnostic tools currently available for diagnosing infectious uveitis (figure 4).
Figure 4.
Flow chart for the diagnosis and categorisation of uveitis based on the vitreous samples examined by flow cytometry analysis, PCR analysis and cytokine detection.
Cells that infiltrate the vitreous during the immunological reaction process, such as macrophages, have unique roles. In bacterial or fungal infectious endophthalmitis (ie, excluding viral infections), the majority of the cells that infiltrate the vitreous fluid are monocytes and macrophages (data not shown). Since the presence of severe vitreous opacity can make it dangerous to collect samples from patients with high-grade endophthalmitis, we only collected vitreous samples from patients with low-grade or middle-grade endophthalmitis. Even so, we found that T-cell and B-cell infiltration into the vitreous fluid was lower in patients with endophthalmitis than with non-infectious uveitis. However, these results do not constitute a definitive finding on the infiltration of the T-cell population in endophthalmitis, as an endophthalmitis diagnosis requires that PCR or culture examinations be performed.
Behçet’s disease is known to be a monocyte-related and macrophage-related disease,16 and is classified as a non-granulomatous uveitis. Thus, this suggests that the infiltrating cell population in the vitreous should exhibit the same pattern as fungal or bacterial infections, and should have a different pattern from other types of infectious granulomatous uveitis, such as toxoplasmosis. However, the patients with Behçet’s disease in our current study did not exhibit any positive results for infection during the PCR or culture examinations. Patients with Behçet’s disease usually do not receive any surgical treatment, as these treatments can induce additional severe inflammation.17–20 Furthermore, since the number of patients with Behçet’s disease was very limited as compared with the large numbers of patients with sarcoidosis, lymphoma and viral infection, it was necessary to conclude that the non-sarcoidosis groups in the present paper had a lower number of diagnostic diseases. Further investigations of vitreous samples from this patient subgroup will need to be undertaken.
Uveitis of an unknown aetiology also exhibited unique characteristics in this study. Based on our flow chart for diagnosing uveitis/tumour (figure 4) in patients with uveitis of unknown aetiology, we observed that many of our cases (21/35, or 60%) had a high CD4/CD8 ratio (>3.5). During the surgical procedure in these patients, we also observed that the retinal disease lesions appeared to be similar to those found in sarcoidosis. However, these patients did not exhibit any of the systemic lesions that are required for a definitive diagnosis of ocular sarcoidosis. In fact, Kataoka et al reported that ocular symptoms are the most frequently seen symptoms in Japanese patients during initial evaluations of sarcoidosis.21 Moreover, ocular sarcoidosis is the primary type of uveitis found in Japanese patients.1 In our previous study, we reported finding similar CD4/CD8 ratios between the vitreous and the bronchoalveolar lavage fluid in patients with sarcoidosis.22 Therefore, these findings suggest that the CD4/CD8 ratio in the vitreous fluid has predictive value for diagnosing sarcoidosis. If a patient is suspected of having sarcoidosis based on vitreous sample examination, it is then necessary to perform further clinical observations and treatments.
In these patients, however, it is important to be able to distinguish between tuberculosis and sarcoidosis, as patients with pulmonary tuberculosis exhibit the same CD4/CD8 ratio as that found in sarcoidosis. Moreover, patients with sarcoidosis and tuberculosis present with similar ocular clinical symptoms. These similar features include vasculitis, retinal exudate and granuloma formation. However, our present study did not examine any patients with tuberculosis uveitis, as medication is generally sufficient for treating these patients,23 and they are not required to undergo any type of surgical treatment. If in the future we are able to collect sufficient vitreous samples from patients with tuberculosis, we are hoping to be able to measure CD4/CD8 ratios and perform PCR examinations in these patients. The PCR examinations of the vitreous samples from the patients with sarcoidosis in our current study did not reveal any tuberculosis DNA. Moreover, based on our present findings, we speculate that a high CD8+ population (more than 28.25) would be expected in patients with viral infection. In fact, some of our patients who had uveitis of unknown aetiology (14/35, or 40%) exhibited a high CD8+ population in the vitreous. Thus, it is our belief that the cases of uveitis of unknown aetiology included in our current study might very well have been classifiable as ocular sarcoidosis or viral infectious uveitis. Thus, once we have been able to collect enough vitreous samples from patients with this type of uveitis, we will need to perform further investigations in order to clarify this supposition.
In conclusion, the results of our current study were consistent with the findings of our previous investigation.4 Our results showed that samples that were obtained through diagnostic vitrectomy in patients with uveitis and then evaluated by flow cytometric lymphocyte analysis, cytology and PCR examination were useful adjuncts to the standard diagnostic procedures. Vitreous cell examination has the potential to become a useful research tool for investigating immune-mediated and infectious uveitis. However, secure vitrectomy and a subsequent analysis of the cell population in subjects with questionable systemic diagnoses remains a topic for future investigation.
Supplementary Material
Acknowledgments
The authors would like to thank Dr Wendy Chao and Timothy Hilts for editing and critically reading this manuscript. The authors would also like to thank Kazuhito Yoneda and Kentaro Kojima for their helpful sample collections.
Footnotes
Contributors: KM contributed to the design, planning and conduction of the study, performing data analysis, and writing and reviewing of the manuscript. TI contributed to the design and planning of the study, performing the data analysis, and writing and reviewing of the manuscript. SS contributed to the PCR data analysis and to the writing and reviewing of the manuscript. RI contributed to the design and the planning of the process for diagnosing lymphoma and to the reviewing of the manuscript. KN contributed to the planning of the process for sample collection and the reviewing of the manuscript. SK, MM and TN contributed to the writing and reviewing of the manuscript.
Competing interests: None declared.
Patient consent: Obtained.
Ethics approval: This multicentre study was approved prospectively by the Institutional Review Boards of both Tohoku University Graduate School of Medicine and the Kyoto Prefectural University of Medicine.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: Extra data can be accessed via the Dryad data repository at http://datadryad.org/with the doi:10.5061/dryad.597ch.
References
- 1. Ohguro N, Sonoda KH, Takeuchi M, et al. . The 2009 prospective multi-center epidemiologic survey of uveitis in Japan. Jpn J Ophthalmol 2012;56:432–5. 10.1007/s10384-012-0158-z [DOI] [PubMed] [Google Scholar]
- 2. Davis JL, Viciana AL, Ruiz P. Diagnosis of intraocular lymphoma by flow cytometry. Am J Ophthalmol 1997;124:362–72. 10.1016/S0002-9394(14)70828-1 [DOI] [PubMed] [Google Scholar]
- 3. Davis JL, Miller DM, Ruiz P. Diagnostic testing of vitrectomy specimens. Am J Ophthalmol 2005;140:822–9. 10.1016/j.ajo.2005.05.032 [DOI] [PubMed] [Google Scholar]
- 4. Kojima K, Maruyama K, Inaba T, et al. . The CD4/CD8 ratio in vitreous fluid is of high diagnostic value in sarcoidosis. Ophthalmology 2012;119:2386–92. 10.1016/j.ophtha.2012.05.033 [DOI] [PubMed] [Google Scholar]
- 5. Herbort CP, Rao NA, Mochizuki M. Members of Scientific Committee of First International Workshop on Ocular Sarcoidosis. International criteria for the diagnosis of ocular sarcoidosis: results of the first International Workshop On Ocular Sarcoidosis (IWOS). Ocul Immunol Inflamm 2009;17:160–9. 10.1080/09273940902818861 [DOI] [PubMed] [Google Scholar]
- 6. Maruyama K, Kunikata H, Sugita S, et al. . First case of primary intraocular natural killer t-cell lymphoma. BMC Ophthalmol 2015;15:169 10.1186/s12886-015-0158-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sugita S, Shimizu N, Watanabe K, et al. . Use of multiplex PCR and real-time PCR to detect human herpes virus genome in ocular fluids of patients with uveitis. Br J Ophthalmol 2008;92:928–32. 10.1136/bjo.2007.133967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Miyanaga M, Sugita S, Shimizu N, et al. . A significant association of viral loads with corneal endothelial cell damage in cytomegalovirus anterior uveitis. Br J Ophthalmol 2010;94:336–40. 10.1136/bjo.2008.156422 [DOI] [PubMed] [Google Scholar]
- 9. Sugita S, Ogawa M, Shimizu N, et al. . Use of a comprehensive polymerase chain reaction system for diagnosis of ocular infectious diseases. Ophthalmology 2013;120:1761–8. 10.1016/j.ophtha.2013.02.020 [DOI] [PubMed] [Google Scholar]
- 10. Hayashi E, Tuda J, Imada M, et al. . The high prevalence of asymptomatic Toxocara infection among school children in Manado, Indonesia. Southeast Asian J Trop Med Public Health 2005;36:1399–406. [PubMed] [Google Scholar]
- 11. Goto H, Usui Y, Umazume A, et al. . Propionibacterium acnes as a possible pathogen of granuloma in patients with ocular sarcoidosis. Br J Ophthalmol 2017:bjophthalmol-2016-309248 10.1136/bjophthalmol-2016-309248 [DOI] [PubMed] [Google Scholar]
- 12. Sant AJ, McMichael A. Revealing the role of CD4(+) T cells in viral immunity. J Exp Med 2012;209:1391–5. 10.1084/jem.20121517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ben Abdelwahed R, Donnou S, Ouakrim H, et al. . Preclinical study of Ublituximab, a Glycoengineered anti-human CD20 antibody, in murine models of primary cerebral and intraocular B-cell lymphomas. Invest Ophthalmol Vis Sci 2013;54:3657–65. 10.1167/iovs.12-10316 [DOI] [PubMed] [Google Scholar]
- 14. Missotten T, Tielemans D, Bromberg JE, et al. . Multicolor flowcytometric immunophenotyping is a valuable tool for detection of intraocular lymphoma. Ophthalmology 2013;120:991–6. 10.1016/j.ophtha.2012.11.007 [DOI] [PubMed] [Google Scholar]
- 15. Maruyama K, Nagata K, Kojima K, et al. . Intraocular Invasion of Adult T-Cell Leukemia Cells without Systemic Symptoms after Cataract Surgery. Case Rep Ophthalmol 2013;4:252–6. 10.1159/000355486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sahin S, Lawrence R, Direskeneli H, et al. . Monocyte activity in Behçet’s disease. Br J Rheumatol 1996;35:424–9. [DOI] [PubMed] [Google Scholar]
- 17. Krause L, Altenburg A, Bechrakis NE, et al. . Intraocular surgery under systemic interferon-alpha therapy in ocular Adamantiades-Behçet’s disease. Graefes Arch Clin Exp Ophthalmol 2007;245:1617–21. 10.1007/s00417-007-0577-6 [DOI] [PubMed] [Google Scholar]
- 18. Ozdemir O, Erkam N, Bakkaloglu A. Results of pars plana vitrectomy in Behçet’s disease. Ann Ophthalmol 1988;20:38. [PubMed] [Google Scholar]
- 19. Ozertürk Y, Bardak Y, Durmuş M. Vitreoretinal surgery in Behçet’s disease with severe ocular complications. Acta Ophthalmol Scand 2001;79:192–6. 10.1034/j.1600-0420.2001.079002192.x [DOI] [PubMed] [Google Scholar]
- 20. Matsuo T, Takahashi M, Inoue Y, et al. . Ocular attacks after phacoemulsification and intraocular lens implantation in patients with Behçet disease. Ophthalmologica 2001;215:179–82. 10.1159/000050854 [DOI] [PubMed] [Google Scholar]
- 21. Kataoka M. [Clinical features and diagnostic approach of sarcoidosis]. Nihon Rinsho 2002;60:1734–40. [PubMed] [Google Scholar]
- 22. Maruyama K, Inaba T, Tamada T, et al. . Vitreous lavage fluid and bronchoalveolar lavage fluid have equal diagnostic value in sarcoidosis. Medicine 2016;95:e5531 10.1097/MD.0000000000005531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Ng KK, Nisbet M, Damato EM, et al. . Presumed tuberculous uveitis in non-endemic country for tuberculosis: case series from a New Zealand tertiary uveitis clinic. Clin Exp Ophthalmol 2017;45 10.1111/ceo.12881 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2016-014549supp001.pdf (48.1KB, pdf)