Abstract
Wnt3a is a major regulator of bone metabolism however, very few of its target genes are known in bone. Wnt3a preferentially signals through transmembrane receptors Frizzled and co-receptors Lrp5/6 to activate the canonical signaling pathway. Previous studies have shown that the canonical Wnt co-receptors Lrp5 and Lrp6 also play an essential role in normal postnatal bone homeostasis, yet, very little is known about specific contributions by these co-receptors in Wnt3a-dependent signaling. We used high-throughput sequencing technology to identify target genes regulated by Wnt3a in osteoblasts and to elucidate the role of Lrp5 and Lrp6 in mediating Wnt3a signaling. Our study identified 782 genes regulated by Wnt3a in primary calvarial osteoblasts. Wnt3a up-regulated the expression of several key regulators of osteoblast proliferation/ early stages of differentiation while inhibiting genes expressed in later stages of osteoblastogenesis. We also found that Lrp6 is the key mediator of Wnt3a signaling in osteoblasts and Lrp5 played a less significant role in mediating Wnt3a signaling.
Introduction
Wnt proteins constitute a family of 19 highly conserved secreted signaling proteins that have various roles in development and disease. Wnts activate either β-catenin dependent canonical signaling pathway or β-catenin independent non-canonical pathways to perform their diverse functions [1]. Several studies have identified Wnt signaling pathway as a key regulator of bone development and homeostasis. Canonical Wnt signaling has been shown to promote osteoblast differentiation, enhance osteoblast proliferation, maturation and mineralization, and inhibit osteoblast apoptosis [2]. Canonical Wnt signaling has also been shown to inhibit osteoclast differentiation [3]. Several Wnt ligands including Wnt1, Wnt3a and Wnt10b preferentially activate canonical Wnt signaling [4]. Previous studies have shown that these Wnts promote bone formation [5–9]. However, very little is known about the molecular mechanism by which canonical Wnts regulate osteoblastogenesis and bone formation or target genes regulated by these Wnts during this process.
Wnt proteins activate canonical signaling by binding to transmembrane receptors Frizzled and co-receptors Lrp5/6 which trigger a cascade of intracellular events that facilitate the translocation of β-catenin to the nucleus where it interacts with Tcf/Lef family of transcription factors and activates transcription of Wnt-responsive genes [10]. Previous studies have suggested that Wnt co-receptors Lrp5 and Lrp6 have both overlapping and non-redundant functions in the skeleton [11, 12]. Mice lacking Lrp6 display severe developmental defects and die shortly after birth [13] whereas Lrp5 knockout mice have no developmental defects but acquire a low bone mass phenotype postnatally [14]. In humans, mutations in Lrp5 result in high bone mass (HBM) or low bone mass (LBM) depending on the nature of the mutation [15–17]. Mice lacking either Lrp5 or Lrp6 in mature osteoblasts displayed LBM and mice lacking both Lrp5 and Lrp6 in osteoblasts developed severe osteopenia [11]. Also, mechanical loading induced bone formation was significantly reduced in Lrp5 knockout mice whereas mice harboring HBM-causing Lrp5 mutations exhibited increased bone formation in response to loading [18, 19]. Wnt-Lrp5 signaling has also been shown to regulate fatty acid metabolism in the osteoblast [12]. These findings suggest that both Lrp5 and Lrp6 are essential for normal postnatal bone homeostasis [12, 14]. However, very little is known about their specific roles in mediating canonical Wnt signaling in osteoblasts and the compendium of target genes regulated through these co-receptors.
To understand how Wnt3a signaling regulates gene expression and to identify the roles of Lrp5 and Lrp6 in mediating Wnt3a signaling in osteoblasts, neonatal calvarial osteoblasts isolated from C57Bl6 (WT) and osteoblasts lacking either Lrp5, Lrp6 or, both Lrp5 and 6 were treated with Wnt3a for 24 hours (h) and gene expression changes were quantified by RNA sequencing (RNA-seq). We found that Wnt3a up-regulated the expression of several key regulators of osteoblast proliferation/early stages of differentiation while inhibiting genes highly expressed in later stages of osteoblastogenesis. We also found that Lrp6 is the key mediator of Wnt3a signaling in osteoblasts and loss of Lrp5 had minimal effect on Wnt3a signaling.
Materials and methods
Generation of knockout animals
All animal experimental procedures were completed in accordance with guidelines under the institutional animal care and use committees at Lawrence Livermore National Laboratory under an approved protocol by the IACUC committee, and conform to the NIH guide for the care and use of Laboratory animals. 3–5 days old pups from timed matings were genotyped by PCR as previously described [20]. Lrp5 global knockout mice have been previously described and will be referred to herein as Lrp5KO [21]. To generate Lrp6 and dual Lrp5/6 deficient osteoblasts, two previously described conditional alleles for Lrp5 (Lrp5flox/flox) and Lrp6 (Lrp6flox/flox) [11] were crossed to a tamoxifen inducible ubiquitous Cre recombinase transgenic strain of mice (UBC-Cre-ERT2; Jackson Laboratories, Bar Harbor, ME, USA) to generate Lrp6flox/flox;UBC-Cre-ERT2 and Lrp5flox/flox;Lrp6flox/flox;UBC-Cre-ERT2 mice; calvarial osteoblasts isolated from these pups (5–6 day old) and treated with hydroxytamoxifen (TMX), in vitro, will be referred to in this manuscript as Lrp6KO OBs and Lrp5/6KO OBs, respectively. Lrp6KO OBs and Lrp5/6KO OBs were compared to TMX treated Lrp6flox/flox and Lrp5flox/flox;Lrp6flox/flox OBs, respectively, which will be referred to in this manuscript as controls.
Quantitative Real-time PCR
Total RNA was purified using RNeasy Mini Kit (QIAGEN Inc, Germantown, MD, USA) according to manufacturer’s protocol. Superscript III First-Strand Synthesis System (Invitrogen, Waltham, MA USA) was used with oligodT primers for reverse transcription according to manufacturer’s protocol. The qPCR was then performed with SYBR Select Master Mix (Applied Biosystems, Waltham, MA USA) using Applied Biosystems 7900HT Fast Real-Time PCR System with the following cycling conditions: 50°C for 2 min for SYBR then 95°C for 2 min, followed by 40 cycles of 95°C for 3 s and 30 s at 60°C. Data was normalized to control gene Gapdh and fold changes were calculated using the comparative Ct method [22]. Primers used for qPCR are given in S1 Table.
Osteoblast isolation and culture
Osteoblasts were isolated from calvaria of neonates (5–6 days old) by serial digestion in Collagenase 1 as previously described [23]. Osteoblast enriched fractions were centrifuged, washed and plated in 12 well plates at 2.6X10^5 cells/well to ensure high cell number and subconfluency at the time of RNA collection. These cells were cultured in DMEM/F12 supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin at 37°C. Following 24h incubation the media was changed to remove digestion debris and non-viable cells and these cultures were treated with 100 ng/ml recombinant Wnt3a (R&D systems, Minneapolis, MN, USA). The RNA was isolated from these cultures after incubating the cells for another 24h. A qPCR analysis was conducted on known Wnt target genes Axin2, Lef1, Igfbp2 and Ibh and the Wnt3a activity was confirmed (S1 Fig). For Lrp5/6KOand Lrp6KOcells, following isolation of osteoblasts, cells were plated at 1x10^5 cells/well, cultured for 24h in DMEM+FBS+P/S media, followed by 48h treatment with DMEM+FBS+P/S with 1 uM hydroxytamoxifen (Sigma-Aldrich, St. Louis, MO, USA) prior to 24h Wnt3a treatment.
RNA-isolation and sequencing
Total RNA was purified using RNeasy Mini Kit (QIAGEN Inc, Germantown, MD, USA) according to manufacturer’s protocol. RNA integrity was assessed using a bioanalyzer (Agilent Technologies, Santa Clara, CA, USA). Poly(A)+-enriched cDNA libraries were generated using the Illumina TruSeq Sample Preparation kit (Illumina Inc, Hayward, CA, USA) and checked for quality and quantity using the bioanalyzer and qPCR. The sequencing was performed using Illumina (Illumina Inc, Hayward, CA, USA) HiSeq 2000 (35-bp single-end reads) or NextSeq 550 (75-bp single-end reads). At least 3 replicates were generated for each experimental condition.
RNA-seq data analysis
RNA sequence data quality was checked using FastQC (version 0.11.5) software. Trimmomatic (version 0. 30) [24] was used for data pre-processing. Using Trimmomatic, the reads were scanned with a 4-base wide sliding window, cutting when the average quality (Phread score) per base drops below 15. Reads with length <25 bases after preprocessing were discarded. Sequence reads were aligned to the mouse reference genome (mm10) using TopHat (version 2.0.11) [25, 26]. More than 90% of the reads were mapped to the mouse genome generating at least 15 million uniquely mapped reads/library. After read mapping, ‘featureCounts’ from Rsubread package (version 1.22.2) [27] was used to perform summarization of reads mapped to RefSeq genes and gene-wise read counts were generated. Genes were filtered from downstream analysis if they did not have a count per million mapped reads (CPM) value of at least 2 in at least five libraries. Subsequently, ‘TMM’ normalization was performed using the calcNormFactors function in edgeR (version 3.14.0) [28]. Differentially expressed genes were identified using ‘limma’ (version 3.28.12) after ‘voom’ [29] normalization. Experimental batch effects were adjusted for by including experimental batch as a covariate in the statistical model. A gene is called significantly differentially expressed when its false discovery rate adjusted p-value (FDR) is less than 0.05 and fold change is greater than 1.5.
BEDTools (version 2.26.0) [30] was used to count the number of RNA-seq reads mapped to deleted exons (exon2 in both Lrp5 and Lrp6) in Lrp6KO OBs, Lrp5/6KO OBs and corresponding control samples. Cre-recombinase induced deletion was confirmed in all Lrp6KO and Lrp5/6KO RNA-seq samples by comparing the number of reads (in CPM) mapped to Lrp5 exon2 and Lrp6 exon2 in conditional knockout samples to corresponding control samples.
We also analyzed a publicly available RNA-seq dataset (GEO: GSE54461) profiling gene expression changes during differentiation of FACS sorted primary calvarial cells expressing Cyan Fluorescent protein (CFP) driven by a Col3.6 promoter to mature osteoblasts, and used this data to understand how the expression of Wnt3a targets change during differentiation of pre-osteoblasts to mature osteoblast. After aligning the RNA-seq reads to mouse genome (mm10) with TopHat [25, 26], gene expression values were calculated using Cuffnorm from Cufflinks (version 2.2.1) [31]. For Wnt3a targets, the expression values from all stages of differentiation were obtained from this dataset and clustered using hierarchical clustering to get clusters of genes with similar temporal expression profiles. Heatmaps were generated using heatmap.2 function in ‘gplots’ R package.
Functional annotation
Gene ontology (GO) and pathway enrichment analysis was performed using functional annotation tool ToppGene [32] and enriched ‘biological processes’ and ‘pathways’ associated with differentially expressed genes were identified (FDR less than 0.05). ToppGene was also used to identify genes associated with bone phenotypes.
Results
Identification of Wnt3a-regulated transcriptome
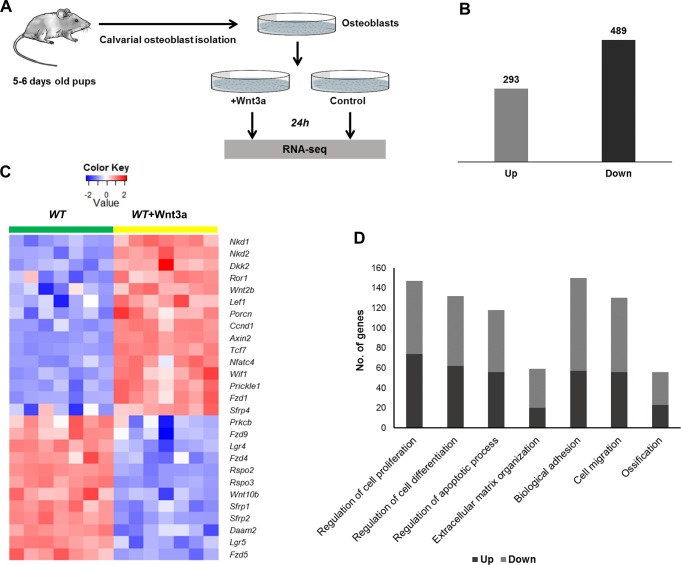
To investigate how Wnt3a regulates osteoblastic gene expression, neonatal calvarial osteoblasts from C57Bl/6 mice (WT OBs) were treated with 100 ng/ml recombinant Wnt3a for 24h and the gene expression changes were profiled using RNA-seq (Fig 1A). By comparing gene expression data from Wnt3a treated WT OBs to sham treated WT OBs we identified 293 up- and 489 down-regulated genes (Fig 1B, Table 1 and S2 Table). Several previously known canonical Wnt targets including Axin2, Igfbp2, Cyr61, Lef1 and Tnfrsf19 were identified as differentially expressed in osteoblasts in response to Wnt3a treatment. We also identified 33 growth factors, 15 transcription factors and 88 genes with receptor activity as Wnt3a targets in osteoblasts (S2 Table).
Fig 1. Identification of Wnt3a regulated transcriptome in osteoblasts.
A) Experimental setup. Osteoblasts were isolated from calvaria of 5–6 days old mice and treated with Wnt3a recombinant protein. RNA was isolated from Wnt3a treated and sham treated cultures 24h post treatment, and sequenced using Illumina high-throughput sequencing technology. B) Genes up- and down-regulated by Wnt3a in osteoblasts. C) Heatmap shows the expression of differentially regulated Wnt pathway members in sham treated WT OBs (WT) and Wnt3a treated WT OBs (WT+Wnt3a). D) Enriched biological processes associated with up- and down-regulated genes. Figure shows the number of up- and down-regulated genes associated with relevant biological processes.
Table 1. Top 25 up- and down-regulated genes with highest log2 fold changes.
| Up-regulated genes | Down-regulated genes | ||
|---|---|---|---|
| Gene | Fold change | Gene | Fold change |
| Bex1 | 4.388 | Ucma | -4.600 |
| Slc13a4 | 4.128 | Stk32b | -3.975 |
| Fgfbp1 | 3.782 | Fmo1 | -3.538 |
| Sema4f | 2.975 | Angpt1 | -3.035 |
| Sstr2 | 2.844 | Gprin3 | -2.987 |
| Nell2 | 2.826 | Lect1 | -2.863 |
| Ndnf | 2.799 | Hgf | -2.763 |
| Axin2 | 2.741 | Scara5 | -2.671 |
| Ntf3 | 2.468 | Sfrp2 | -2.612 |
| Tmem100 | 2.261 | Rspo2 | -2.491 |
| Grik3 | 2.221 | Matn4 | -2.387 |
| Igfbp2 | 2.187 | Rspo3 | -2.377 |
| C430002N11Rik | 2.139 | Serpinb1a | -2.331 |
| Aqp5 | 2.092 | Entpd3 | -2.230 |
| Colec10 | 2.070 | Cxcl12 | -2.165 |
| Syt13 | 2.059 | Hp | -2.148 |
| Tnfsf15 | 2.024 | Dio3 | -2.141 |
| Hhip | 2.012 | Smoc1 | -2.136 |
| Dio2 | 1.971 | Comp | -2.127 |
| Nkd2 | 1.958 | Palmd | -2.087 |
| Slc1a2 | 1.926 | Dner | -2.062 |
| Bmper | 1.925 | Pparg | -2.059 |
| Tgfb2 | 1.923 | Chrdl1 | -1.985 |
| Grem1 | 1.888 | Sox9 | -1.932 |
| Dpp4 | 1.883 | Fzd9 | -1.919 |
Wnt3a altered the expression of 27 known genes from the Wnt signaling pathway: Porcn, Fzd1, Lef1, Tcf7 and Axin2 were up- while Sfrp1, Sfrp2, Fzd4, Fzd9, Rspo2, Rspo3 and Lgr4 were down-regulated in response to Wnt3a treatment (Fig 1C, Table 2). Wnt3a also regulated the expression of several members of TGF-β signaling pathway and MAPK signaling, two other key regulators of bone development and metabolism [33, 34] (Table 2, S3 Table).
Table 2. Key signaling pathways regulated by Wnt3a.
| Signaling pathways | Genes regulated by Wnt3a |
|---|---|
| Wnt signaling |
Up:
Axin2, Fzd1, Tcf7, Sfrp4, Wnt2b, Nfatc4, Dkk2, Porcn, Ccnd1, Lef1, Prickle1, Wif1, Nkd1, Nkd2, Ror1* Down: Sfrp2, Fzd9, Fzd4, Fzd5, Sfrp1, Wnt10b, Prkcb, Daam2, Rspo2*, Rspo3*, Lgr4*, Lgr5* |
| TGF-β signaling |
Up:
Tgfb2, Tgfb3, Bmp2, Bmp4, Bmp7, Inhba, Inhbb, Bmpr1b, Nog Down: Dcn, Id4, Fst |
| MAPK signaling |
Up:
Cacna1c, Cacnb2, Dusp9, Fgfr1, Cacnb4, Cacna1g, Rras2, Dusp1, Fgf13, Fgf18, Tgfb3, Fgf21, Pdgfa, Ngf, Gadd45g, Tgfb2, Ntf3 Down: Cacna1d, Fgf7, Map2k6, Fgfr3, Pdgfra, Prkcb, Egfr |
*Genes obtained from http://web.stanford.edu/group/nusselab/cgi-bin/wnt/. Remaining genes were identified by functional annotation tool ToppGene.
GO analysis of differentially expressed genes showed enrichment for genes associated with several GO categories, including ‘regulation of cell proliferation’, ‘regulation of cell differentiation’, ‘extracellular matrix organization’, ‘biological adhesion’, ‘regulation of apoptotic process’, ‘cell migration’ and ‘ossification’ (Fig 1D, S4 Table). ‘Regulation of cell proliferation’ was one of the most significantly enriched terms for up-regulated genes with 74 genes including Bmp2, Bmp4, Tgfb2, Pdgfa, Gdnf, and Hbegf in that category (S4 Table). ‘Biological adhesion’ and ‘extracellular matrix organization’ were among the most significantly enriched GO terms associated with down-regulated genes (S4 Table). Wnt3a up-regulated 23 and down-regulated 33 genes associated with GO category ‘ossification’ (Table 3). We also identified several genes associated with bone phenotypes in mice as Wnt3a targets in osteoblasts. Forty-eight genes including Bmp2, Fosl2 and Cthrc1 with known bone phenotypes were up-regulated while 53 genes with bone phenotypes including Spp1, Fzd9 and Sfrp1 were down-regulated in response to Wnt3a treatment (Table 4).
Table 3. Wnt3a targets associated with GO term ‘ossification’.
| Up-regulated genes | Down-regulated genes | ||||
|---|---|---|---|---|---|
| Adrb2 | Cthrc1 | Lef1 | Chrdl1 | Gpm6b | Ostn |
| Axin2 | Cyr61 | Noct | Col11a2 | Hgf | Penk |
| Bmp2 | Enpp1 | Nog | Col13a1 | Id4 | Ptn |
| Bmp3 | Fgf18 | Ptch1 | Col2a1 | Igf1 | Rassf2 |
| Tgfb2 | Bmp4 | Ptgs2 | Dhrs3 | Igsf10 | Rspo2 |
| Tgfb3 | Bmp7 | Grem1 | Dlx5 | Jag1 | S1pr1 |
| Rras2 | Fgfr1 | Bmpr1b | Sox9 | Vcan | Egfr |
| Stc1 | Fzd1 | Spp1 | Wnt10b | Fgfr3 | |
| Srgn | Sfrp1 | Gdf10 | |||
| Ucma | Sfrp2 | Smoc1 | |||
| Kazald1 | Lgr4 | Mmp13 | |||
Table 4. Wnt3a target genes associated with bone phenotypes in mice.
| Up-regulated genes | Down-regulated genes | ||||||
|---|---|---|---|---|---|---|---|
| Enpp1 | Dkk2 | Errfi1 | Tgfb2 | Sfrp2 | Meox2 | Fam46a | Col2a1 |
| Rictor | Cdo1 | Ptch1 | Dio2 | Rspo2 | Spp1 | Id4 | Hivep3 |
| Sfrp4 | Prrx2 | Nov | Axin2 | Rspo3 | Cd74 | Sfrp1 | Mamld1 |
| Rb1 | Osbpl3 | Ptgs2 | Jak2 | Comp | Dhrs3 | Chrna7 | Irak3 |
| Itgb1 | Sema3f | Slc20a1 | Adrb2 | Pparg | Spns2 | Pdgfra | Ank1 |
| Sgms1 | Wbscr17 | Lif | Prickle1 | Sox9 | Fgfr3 | Vdr | Egfr |
| Bmp4 | Bmp2 | Hck | Fgf18 | Fzd9 | Itgb3 | Rassf2 | Tlr4 |
| Cthrc1 | Bmpr1b | Tcf7 | Bmp7 | Col14a1 | Clstn3 | Gnao1 | Daam2 |
| Pxylp1 | Nog | Cnn1 | Grem1 | Penk | Dcn | Fmod | Xylt1 |
| Mllt3 | Galnt3 | Grem2 | Fosl2 | C3 | Dlx5 | Ebf1 | Fgf7 |
| Fgfr1 | Bmp3 | Itga8 | Smim3 | Mmp13 | Tlr2 | Plagl1 | Ednrb |
| Efemp1 | Lepr | Tceal5 | Col7a1 | Cdkn1c | Epas1 | Igf1 | |
| Col9a2 | Pappa2 | Epyc | |||||
| Aldh3b1 | Stat1 | Npr3 | |||||
Wnt3a activates genes associated with early stages of osteogenesis and inhibits genes associated with late stage osteogenesis
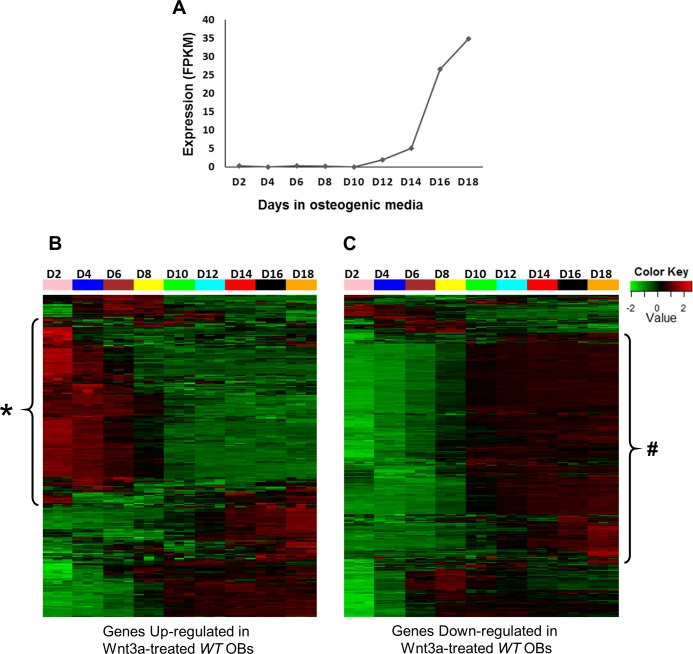
Temporal gene expression profiling across various stages of osteoblast differentiation can highlight cohort of transcripts with distinct roles during osteogenesis. To understand the temporal expression patterns of Wnt3a targets (identified above) during osteoblast differentiation, we analyzed the expression levels of these genes during the differentiation of purified pre-osteoblasts to mature osteoblasts capable of matrix mineralization (Fig 2A), by curating a publicly available dataset (GEO: GSE54461). This dataset includes RNA-seq from 2, 4, 6, 8, 10, 12, 14, 16 and 18 days post differentiation of pre-osteoblasts cultured in an osteoblast differentiation cocktail. Expression values of Wnt3a targets from all stages of osteoblast differentiation were obtained from this dataset and clustered using hierarchical clustering to get clusters of genes with similar temporal expression profiles. We determined that a large number (>62%) of genes up-regulated by Wnt3a were expressed at high levels during the early stages (2–8 days) of osteogenic differentiation (Fig 2B, S5 Table) whereas most of the genes down-regulated by Wnt3a were highly expressed in mature osteoblasts only (Fig 2C, S5 Table). Wnt3a also up-regulated several genes with higher expression in mature osteoblasts than immature osteoblasts including BMP/ TGF-β signaling pathway genes Bmp2, Bmp3, Bmp4, Bmp7 and Tgfb2, FGF signaling pathway genes such as Fgf13, Fgf18 and Fgfr1 and, Wnt signaling pathway members including Porcn, Axin2, Fzd1, Tcf7, Sfrp4, Nkd1 and Prickle1 (Fig 2B, S5 Table). Several of these genes have been shown to regulate early stages of osteoblast differentiation as well as osteoblast maturation and mineralization [2, 35–38]. This data suggest that Wnt3a treatment promotes the expression of genes involved in osteoblast proliferation or early stages of differentiation while inhibiting genes involved in the later stages of osteoblastogenesis.
Fig 2. Expression profiles of Wnt3a targets during the differentiation of pre-osteoblast to mature osteoblasts.
A) Expression profile of osteocalcin, a mature osteoblast marker. Osteocalcin expression starts around day 10. B) Expression profiles of genes up-regulated by Wnt3a. A large number of genes up-regulated by Wnt3a were highly expressed in early stage (D2-D8) osteoblasts (highlighted with *). C) Expression profiles of genes down-regulated by Wnt3a. Most of the genes down-regulated by Wnt3a were highly expressed in mature (D10-D18) osteoblasts (highlighted with #).
Ablation of Lrp5 and Lrp6 has different effects on osteoblastic gene expression
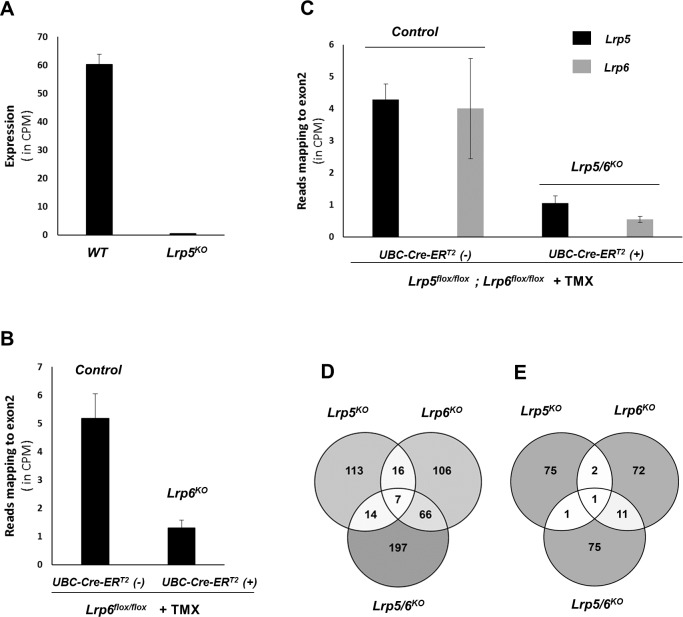
To investigate the roles of Wnt co-receptors Lrp5 and Lrp6 in regulating gene expression in osteoblasts we generated Lrp5 deficient (Lrp5KO), Lrp6 deficient (Lrp6KO) and dual Lrp5/6 deficient (Lrp5/6KO) osteoblasts. Lrp5KO OBs were isolated from mice lacking Lrp5 globally. Lrp6KO OBs and Lrp5/6KO OBs were generated via in vitro deletion of these genes in primary osteoblasts purified from floxed mice expressing UBC-Cre-ERT2 transgene. Lrp5KO OB RNA-seq samples showed 99.3% reduction in Lrp5 expression compared to WT OBs (Fig 3A). To account for the effects tamoxifen treatment (TMX) may have on gene expression, Lrp6KOand Lrp5/6KO OBs were compared to TMX treated osteoblasts isolated from floxed littermates lacking the UBC-Cre-ERT2 transgene (controls). We found the expression of Lrp6 to be reduced by ~75% in Lrp6KO OB samples (Fig 3B), while Lrp5 was ~76% and Lrp6 was ~86% reduced in Lrp5/6KO OBs (Fig 3C).
Fig 3. Effect of Lrp5 and Lrp6 ablation in osteoblasts.
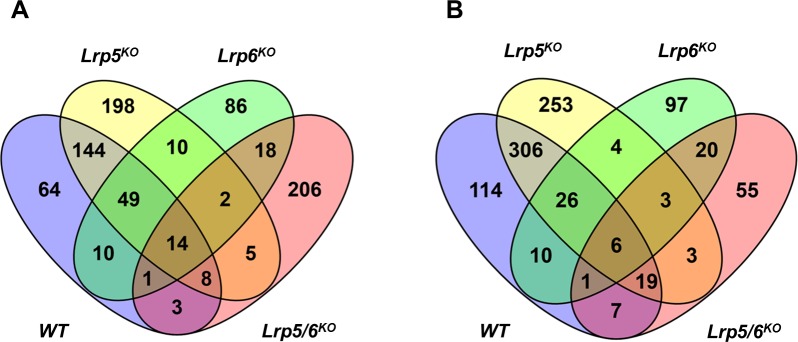
A) Expression of Lrp5 in WT and Lrp5KO OBs. Expression values are given in counts per million (CPM) mapped reads. B) Expression of Lrp6 in controls (Lrp6fl/fl+TMX) and Lrp6KO OBs (Lrp6fl/fl;UBC-Cre-ERT2+TMX). Expression values indicate the number of RNA-seq reads mapped to exon2 (deleted exon) in Lrp6 (in CPM). C) Expression of Lrp5 and Lrp6 in controls (Lrp5fl/fl;Lrp6fl/fl+TMX) and Lrp5/6KO OBs (Lrp5fl/fl;Lrp6fl/fl; UBC-Cre-ERT2+TMX). Expression values indicate the number of RNA-seq reads mapped to exon2 in Lrp5 and Lrp6 (in CPM). D) Venn diagram showing overlap between genes up-regulated in Lrp5KO OBs, Lrp6KO OBs and Lrp5/6KO OBs compared to respective controls. E) Venn diagram showing overlap between genes down-regulated in Lrp5KO OBs, Lrp6KO OBs and Lrp5/6KO OBs compared to respective controls.
By comparing gene expression in Lrp5KO OBs to WT OBs we identified 150 genes up- and 79 genes down-regulated in Lrp5KO OBs (S6 Table). Next, we identified the top 50 enriched biological processes associated with differentially expressed genes. Thirty-nine genes associated with GO term ‘defense response’ including Ccl7, Cxcl5, Tnfaip6 and Ntrk2, 28 genes associated with ‘cell migration’ including Igfbp3, Ednrb, Dcn and Hgf, 37 genes associated with ‘regulation of response to stress’ including Xdh, Hgf, Casp4 and Serpine2, and 27 genes associated with ‘regulation of cell proliferation’ including Cxcl12, Rbp4, Fabp4 and Aldh3a1 were up-regulated in Lrp5KO OBs (S7 Table). Down-regulated genes did not show enrichment for any biological processes. Key down-regulated genes include muscle genes Actg2, Cnn1 and Lmod1, and Pdpn, a gene induced during osteoblast to osteocyte transition. Only 9 genes induced by Wnt3a in WT OB (identified above) including Sfn, Ahr, Ankrd1and Ahrr showed low expression in Lrp5KO OBs relative to WT OBs while 25 genes down-regulated by Wnt3a in WT OBs including Cxcl12, Dcn, Mmp13, Hgf and Mt2 showed increased expression in Lrp5KO OBs (S6 Table).
By comparing Lrp6KO to controls we identified 195 genes up- and 86 genes down-regulated in Lrp6KO(S6 Table). Genes up-regulated in Lrp6KOalso showed enrichment for GO term ‘defense response’ with 41 genes including Ntrk2, Il33 and Mecom in that category (S8 Table). Seventeen genes associated with ‘skeletal development’ including Col2a1, Comp, Col9a1and Frzb were also up-regulated in Lrp6KO. Other enriched GO categories include ‘cellular response to type I interferon (12 genes)’, ‘extracellular matrix organization (17 genes)’, ‘locomotion (34 genes)’, ‘apoptotic process (35)’ and ‘response to biotic stimulus (28 genes)’. Top 50 GO terms associated with down-regulated genes included ‘muscle cell differentiation (23 genes)’ and ‘actin cytoskeleton organization (14 genes)’. Five genes induced by Wnt3a in WT OB including Grem1, Ndnf, and Dynap were down-regulated and 70 genes down-regulated by Wnt3a in WT OBs including Comp, Fzd9, Mmp13 and F13a1 were up-regulated in Lrp6KO OBs compared to controls (S6 Table).
Two hundred and eighty-four genes were up- and 88 genes were down-regulated in Lrp5/6KO OBs compared to controls (S6 Table). Twenty-one genes induced by Wnt3a in WT OBs including Axin2, Ndnf, Nkd1 and Tnfrsf19 showed down-regulation in Lrp5/6KO OBs while 84 genes down-regulated by Wnt3a in WT OBs were found to be up-regulated in Lrp5/6KO OBs compared to controls (S6 Table).
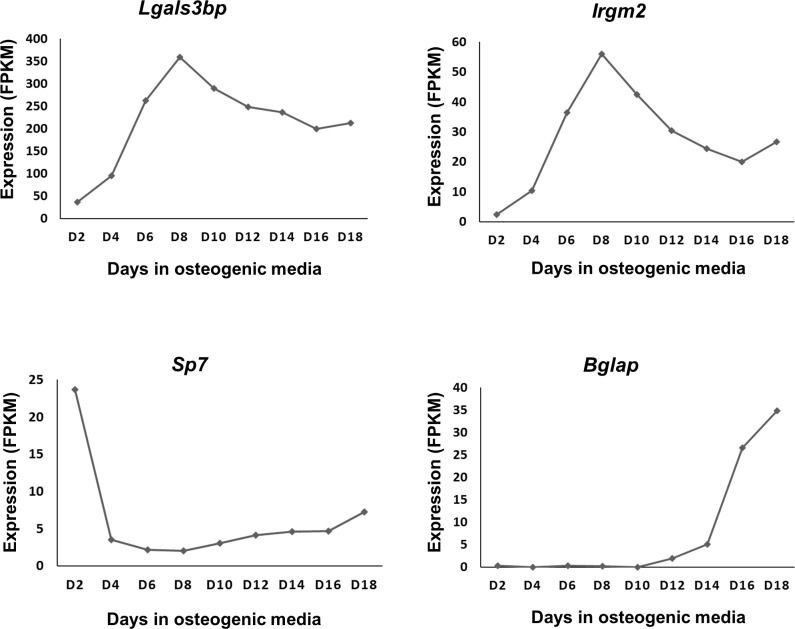
Of the 229 genes differentially expressed in Lrp5KO OBs vs. WT OBs 26 (23 up; 3 down) and 23 (21 up; 2 down) genes overlapped with genes differentially expressed in Lrp6KOand Lrp5/6KO OBs, respectively (Fig 3D and 3E, S6 Table). Eighty-five genes (73 up; 12 down) differentially expressed in Lrp6KO vs. controls overlapped with genes differentially expressed in Lrp5/6KO OBs relative to controls (Fig 3D and 3E, S6 Table). Eight genes (7 up; 1 down) including Pou3f4, Ntrk2, Siglec1, Irgm2, Lgals3bp (up) and Col15a1 (down) were differentially expressed in all three receptor knockouts compared to respective controls (Fig 3D and 3E, S6 Table). Two of these up-regulated genes, Irgm2 and Lgals3bp were also among the genes down-regulated by Wnt3a in WT OBs and, both genes exhibited very similar expression patterns during osteoblast differentiation with highest expression at day 8 (Fig 4).
Fig 4. Expression profiles of Lgals3bp and Irgm2 during osteoblast differentiation.
Expression profiles of Lgals3bp and Irgm2 compared to pre-osteoblast marker Sp7 and mature osteoblast marker Bglap (Osteocalcin). Expression values were obtained from the RNA-seq dataset GSE54461. Both Lgals3bp and Irgm2 were robustly expressed on osteoblasts and showed highest expression at day 8 (D8).
Ablation of Lrp6 but, not Lrp5, significantly impaired Wnt3a signaling in osteoblasts
To investigate the role of Lrp5 and Lrp6 in mediating Wnt3a signaling in osteoblasts we quantified the gene expression changes in Wnt3a treated Lrp5KO (Lrp5KO+Wnt3a), Lrp6KO(Lrp6KO+Wnt3a) and Lrp5/6KO(Lrp5/6KO+Wnt3a) OBs compared to respective sham treated controls. We identified 1050 (430 up; 620 down) differentially expressed genes in Lrp5KO+Wnt3a compared to sham treated WT OBs (Lrp5KO+Wnt3a vs. WT OBs; S9 Table). This included genes regulated by Wnt3a independent of Lrp5 and genes that may not be Wnt3a dependent but, changed as a result of loss of Lrp5. Four hundred and fifty-five out of the 1050 genes differentially expressed in Lrp5KO+Wnt3a vs. WT OBs were not significantly differentially expressed in Lrp5KO+Wnt3a vs. Lrp5KO OBs (Fig 5A and 5B), suggesting that their expression is Lrp5 dependent, and Wnt3a treatment may not have a significant impact on the expression of some of these genes. Only 88 of these 455 genes were identified as Wnt3a targets in WT OBs (identified above as differentially expressed in WT OBs +Wnt3a vs. WT OBs), suggesting that the remaining genes may not be Wnt3a dependent. It is possible that these genes are regulated by other Wnt ligands such as Wnt1 and Wnt10b via Lrp5 or a complex interplay between Wnts and other signaling pathways and, loss of Lrp5 resulted in up- or down-regulation of these genes.
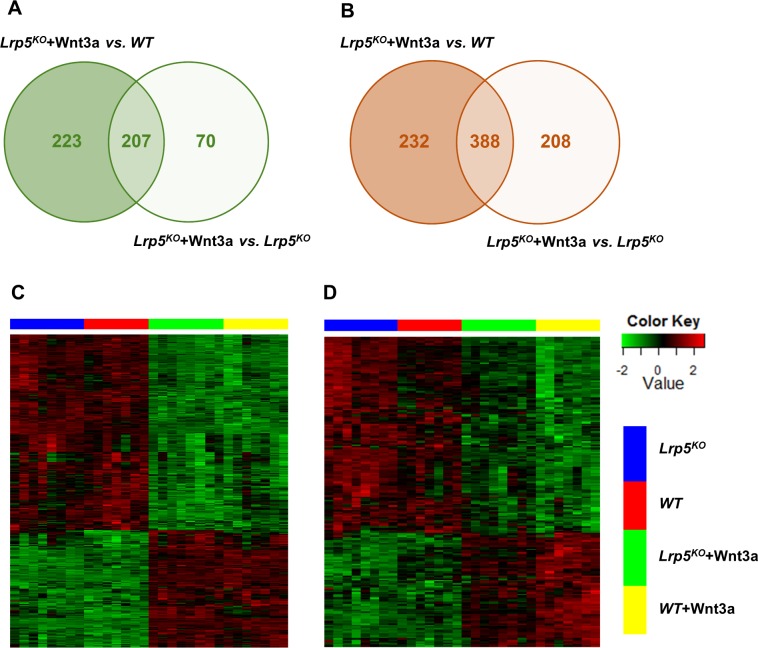
Fig 5. Wnt3a regulated genes in Lrp5KO osteoblasts.
A) Venn diagram showing overlap between genes up-regulated in Wnt3a treated Lrp5KO OBs (Lrp5KO+Wnt3a) compared to sham treated WT OBs (WT) and sham treated Lrp5KO OBs (Lrp5KO). B) Venn diagram showing overlap between genes down-regulated in Wnt3a treated Lrp5KO OBs (Lrp5KO+Wnt3a) compared to sham treated WT OBs (WT) and sham treated Lrp5KO OBs (Lrp5KO). C) Heatmap showing genes up- or down-regulated by Wnt3a in both WT OBs (WT +Wnt3a) and Lrp5KO OBs (Lrp5KO+Wnt3a) compared to WT (WT) controls. D) Heatmap showing genes up- or down-regulated by Wnt3a in WT OBs (WT +Wnt3a) but, not in Lrp5KO OBs (Lrp5KO+Wnt3a) compared to WT (WT) controls. These genes either had a fold change < 1.5 or FDR corrected p-value > 0.05.
About 73% (572/782) of the Wnt3a targets identified in WT OBs (identified above as differentially expressed in WT OBs +Wnt3a vs. WT OBs) were also significantly differentially expressed in Lrp5KO OBs in response to Wnt3a treatment (Lrp5KO+Wnt3a vs. WT OBs), suggesting that Wnt3a signaling is minimally affected by the loss of Lrp5 in osteoblasts (Figs 5C & 6, S9 Table). Majority of the remaining Wnt3a targets also showed up- or down-regulation in Lrp5KO +Wnt3a vs. WT OBs although the changes were below the significance threshold used in this study (Fig 5D), suggesting that Lrp5 is required for the optimal Wnt3a-mediated activation or repression of these genes.
Fig 6. Overlap between Wnt3a targets identified in WT, Lrp5KO, Lrp6KO and Lrp5/6KO osteoblasts.
A) Overlap between genes up-regulated by Wnt3a. B) Overlap between genes down-regulated by Wnt3a.
By comparing Lrp6KO+Wnt3a to sham treated controls, we identified 357 (190 up, 167 down) differentially expressed genes (S9 Table). Two hundred and fifty-five of these genes were not differentially expressed in Lrp6KO+Wnt3a vs. Lrp6KOsuggesting that their expression is Lrp6 dependent and may not directly depend on Wnt3a. Only ~15% (117/782) of the Wnt3a targets identified in WT OBs (identified above as differentially expressed in WT OBs +Wnt3a vs. WT OBs) were found to be differentially expressed between Wnt3a treated Lrp6KO OBs and sham treated controls, suggesting that the lack of Lrp6 significantly impaired Wnt3a signaling in osteoblasts (Fig 6, S9 Table).
Three hundred and seventy-one (257 up; 114 down) genes were differentially expressed between Lrp5/6KO+Wnt3a and sham treated controls (S9 Table). However, only ~8% (59/782) of the Wnt3a target genes including Inhbb, Sema4f and Nell2 were found to be significantly differentially expressed in Wnt3a treated Lrp5/6KO OBs compared to sham treated controls (Fig 6, S9 Table). This suggests that Wnt3a-dependent signaling is greatly impaired in osteoblasts lacking both Lrp5 and Lrp6.
Discussion
Several studies have shown that Wnt3a plays a key role in skeletal development and bone metabolism [8, 39]. Although numerous Wnt3a target genes have been identified in different cell types including pluripotent mesenchymal cell line C3H10T1/2 [40] and stromal cell line ST2 [41], very little is known about the target genes regulated by Wnt3a in primary osteoblasts. This study provides the first account of Wnt3a regulated transcriptome in primary osteoblasts.
We identified 782 Wnt3a targets in osteoblasts including 101 genes with bone phenotypes in mice, highlighting the importance of Wnt3a signaling in regulating skeletal development and bone metabolism. Previous studies have shown that Wnt signaling induces the expression of the osteoblast marker alkaline phosphatase (Alp) [42]. Although Alp and other osteoblast markers such as Sp7 and Runx2 were expressed in the primary murine osteoblast used in this study, we did not observe any significant changes in the expression of these genes in response to treatment with Wnt3a for 24 hours. However, culturing these Wnt3a stimulated cells for a longer period or treating these cells with recombinant Wnt3a for a longer duration might lead to an elevated expression of Alp and other osteoblast markers in these cells.
Our study identified several members of Wnt signaling pathway as Wnt3a targets, suggesting a feedback regulatory mechanism mediated by Wnt3a in osteoblasts. Wnt3a also regulated the expression of members of other signaling pathways including BMP/ TGF-β signaling and MAPK signaling, several growth factors including Pdgfa, Hbegf, Ngf and Ntf3, and transcription factors including Hdac9, Ankrd1, Vdr, Pparg, Sox9 and Nfatc4. It is reasonable to speculate that Wnt3a indirectly regulated the expression of some of the genes identified in this study through the activation of other pathways and transcription factors. The expression of Pparg, a key regulator of adipogenesis, was >4-fold down in Wnt3a treated osteoblasts. Reduced Pparg expression is associated with increased bone formation and, canonical Wnt signaling has been shown to down-regulate Pparg [41, 43]. Previous studies have also shown that loss of canonical Wnt signaling causes cell fate shift of pre-osteoblasts from osteoblasts to adipocytes [44]. Our data suggest that Wnt3a may repress the differentiation of pre-osteoblasts to adipocytes by suppressing Pparg expression. Wnt3a also down-regulated Sox9, a transcription factor that regulates chondrocyte differentiation [45]. In mice, overexpression of Sox9 in osteoblasts from a 2.3-kb Col1a1 promoter resulted in dwarfism and osteopenia, with a significant reduction in bone volume, osteoblasts number and bone formation rate [45]. Bone marrow stromal cells isolated from Sox9 transgenic mice also displayed enhanced adipocyte differentiation and decreased osteoblast differentiation in vitro [45]. This data suggest that Sox9 is a negative regulator of osteoblast differentiation and bone formation. Down-regulation of Sox9 in osteoblasts by Wnt3a may also contribute to enhanced osteogenesis and bone formation.
Few in vitro studies have suggested that Wnt3a promotes osteoblast differentiation [41, 46, 47]. In contrast, several studies have reported that Wnt3a promotes osteoblast proliferation and suppresses osteoblasts differentiation [8, 48]. Examination of the temporal expression patterns of Wnt3a targets during osteoblastogenesis revealed that a large number (>62%) of genes up-regulated by Wnt3a are generally highly expressed during the early stages of osteogenic differentiation and the majority of the genes down-regulated by Wnt3a are highly expressed in mature osteoblasts. This data suggests that Wnt3a promotes osteoblast proliferation or early stages of osteoblast differentiation and inhibits osteoblast maturation/mineralization. This is in line with previous findings by Boland et al. [8] and Caverzasio et al. [48] that Wnt3a enhances proliferation of MSCs, pre-osteoblast cell lines and mouse primary osteoblasts [8]. Boland et al. have also shown that Wnt3a treatment inhibited mineralization of MSCs that had been osteogenically differentiated for 12 days prior to Wnt3a exposure and this effect was reversible [8]. Our study identified several regulators of early stage osteogenesis induced by Wnt3a including Pdgfa [49], Cyr61 [50] and Tgfb3 [51] and regulators of osteoblast maturation/mineralization repressed by Wnt3a including Vdr [52] and Rspo2 [53]. Wnt3a also up-regulated a number of genes with higher expression in mature osteoblasts than early stage osteoblasts including several members of BMP/TGF-β signaling pathway (Bmp2, Bmp3, Bmp7, Tgfb2 etc.), FGF signaling pathway (Fgf13, Fgf18, Fgfr1 etc.) and Wnt signaling pathway (Porcn, Axin2, Fzd1, Tcf7, Sfrp4, Nkd1, Prickle1 etc.). Many of these genes have been shown to regulate both early and late stages of osteoblast differentiation [2, 35–38]. This suggests that some of the Wnt3a activated genes identified in this study may play a role in both early and late stages of osteogenesis.
Lrp5 and Lrp6 are key mediators of β-catenin dependent Wnt signaling. However, very little is known about their specific roles in regulating gene expression in osteoblasts. We found very little overlap between genes differentially regulated in the absence of Lrp5 and Lrp6 in osteoblasts suggesting that these two receptors have non-redundant functions in regulating osteoblastic gene expression. We found that the expression of several known canonical Wnt targets were not dramatically altered in osteoblasts lacking either Lrp5 or Lrp6; however, osteoblasts lacking both Lrp5 and Lrp6 showed down-regulation of several known canonical Wnt targets including Axin2, Nkd1 and Tnfrsf19. Two genes up-regulated in sham treated Lrp5KO OBs, Lrp6KO OBs and Lrp5/6KO OBs compared to respective sham treated controls, Irgm2 and Lgals3bp, were also found to be down-regulated by Wnt3a in WT OBs and, both genes had very similar expression profiles during osteoblast differentiation with highest expression at day 8 (Fig 4). Irgm2 is a GTPase and Lgals3bp is involved in cell–cell and cell–matrix interactions. However, their functions in bone is not known. It is likely that these genes play a significant role in regulating osteoblastogenesis.
We also found that a large number of genes suppressed by Wnt3a in WT OBs were up-regulated in sham treated Lrp5KO, Lrp6KO and Lrp5/6KO OBs compared to respective sham treated controls. However, only few genes up-regulated by Wnt3a were down-regulated in osteoblasts lacking these receptors. The total number of genes suppressed by Wnt3a in WT OBs were also significantly higher than the number of Wnt3a activated genes (489 down vs. 293 up). This suggests that transcriptional repression is a major mechanism by which Wnt3a preforms its functions in osteoblasts. This is consistent with a recent report by Karner et al. that, in stromal cell line ST2, Wnt3a activated significantly fewer genes compared to the number of genes it inhibited [41]. They also showed that Wnt3a inhibited gene expression by suppressing histone acetylation possibly in an Lrp5/6 dependent but, β-catenin independent manner. Further studies are required to understand the exact mechanism by which canonical Wnt signaling suppress gene expression in osteoblasts, and how these outcomes are interconnected with other molecular pathways.
Our study also showed that that Wnt3a regulated >73% of its target genes independent of Lrp5 whereas lack of Lrp6 significantly impaired the ability of Wnt3a to regulate target gene expression. This suggests that Lrp6 is the key mediator of Wnt3a signaling in osteoblasts. However, global genetic deletion of Lrp5 greatly impairs bone metabolism, suggesting that Lrp5 deficiency does play a significant role in bone. Our data suggests that Lrp5 may mediate its signaling via other Wnt ligands, and such candidates include Wnt1 and Wnt10b; alternatively, Lrp5 may have a context dependent function in bone. ~8% (59/782) of the Wnt3a target genes including Inhbb, Sema4f and Nell2 were also differentially expressed in Wnt3a treated Lrp5/6KO OBs compared to sham treated controls. It is likely that these genes are activated via non-canonical Wnt pathways as Wnt3a has previously been shown to activate non-canonical pathways [54].
The analysis of gene expression changes is a powerful approach for elucidating the molecular mechanisms by which signaling pathways regulate biological processes such as bone metabolism. However, the current study is limited in its examination of the role of Wnt3a signaling in neonatal calvarial osteoblasts 24h post treatment. Future studies could include osteoblasts isolated from adult mice, osteoblasts from different skeletal locations and osteoblasts from various stages of differentiation treated with Wnts for varying duration to get a more detailed picture of canonical Wnt signaling in osteoblasts. Also, a large number of Wnt targets identified in this study have not been previously characterized in the context of bone metabolism. Further studies are required to determine their impact on osteoblast function and bone metabolism. Overall, the data presented herein will further our understanding of the role of the canonical Wnt signaling pathway in the regulation of osteoblast differentiation and function and in addition, this study will enhance current knowledge of the Wnt signaling pathway itself.
Supporting information
A qPCR analysis showed that Wnt3a up-regulated known canonical Wnt target genes Axin2, Lef1, Igfbp2 and down-regulated Ibh, a gene suppressed by canonical Wnt signaling.
(TIF)
(PDF)
Fold changes are given in log2 scale.
(XLSX)
(PDF)
(PDF)
(XLSX)
Fold changes are given in log2 scale. Empty cells represent no significant change in expression.
(XLSX)
(PDF)
(PDF)
Fold changes are given in log2 scale. Empty cells represent no significant change in expression.
(XLSX)
Acknowledgments
This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under Contract DE-AC52-07NA27344.
Data Availability
All relevant data are within the paper and its Supporting Information files. RNA-seq data has been deposited in NCBI and is available at https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE103492.
Funding Statement
This work was supported by LLNL-LDRD-13-ERD-042.
References
- 1.Niehrs C. The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol. 2012;13(12):767–79. doi: 10.1038/nrm3470 . [DOI] [PubMed] [Google Scholar]
- 2.Khosla S, Westendorf JJ, Oursler MJ. Building bone to reverse osteoporosis and repair fractures. J Clin Invest. 2008;118(2):421–8. doi: 10.1172/JCI33612 ; PubMed Central PMCID: PMCPMC2214701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Weivoda MM, Ruan M, Hachfeld CM, Pederson L, Howe A, Davey RA, et al. Wnt Signaling Inhibits Osteoclast Differentiation by Activating Canonical and Noncanonical cAMP/PKA Pathways. J Bone Miner Res. 2016;31(1):65–75. doi: 10.1002/jbmr.2599 ; PubMed Central PMCID: PMCPMC4758681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Krishnan V, Bryant HU, Macdougald OA. Regulation of bone mass by Wnt signaling. J Clin Invest. 2006;116(5):1202–9. doi: 10.1172/JCI28551 ; PubMed Central PMCID: PMCPMC1451219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Keupp K, Beleggia F, Kayserili H, Barnes AM, Steiner M, Semler O, et al. Mutations in WNT1 cause different forms of bone fragility. Am J Hum Genet. 2013;92(4):565–74. doi: 10.1016/j.ajhg.2013.02.010 ; PubMed Central PMCID: PMCPMC3617378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kook SH, Heo JS, Lee JC. Crucial roles of canonical Runx2-dependent pathway on Wnt1-induced osteoblastic differentiation of human periodontal ligament fibroblasts. Mol Cell Biochem. 2015;402(1–2):213–23. doi: 10.1007/s11010-015-2329-y . [DOI] [PubMed] [Google Scholar]
- 7.Bennett CN, Ouyang H, Ma YL, Zeng Q, Gerin I, Sousa KM, et al. Wnt10b increases postnatal bone formation by enhancing osteoblast differentiation. J Bone Miner Res. 2007;22(12):1924–32. doi: 10.1359/jbmr.070810 . [DOI] [PubMed] [Google Scholar]
- 8.Boland GM, Perkins G, Hall DJ, Tuan RS. Wnt 3a promotes proliferation and suppresses osteogenic differentiation of adult human mesenchymal stem cells. J Cell Biochem. 2004;93(6):1210–30. doi: 10.1002/jcb.20284 . [DOI] [PubMed] [Google Scholar]
- 9.Minear S, Leucht P, Jiang J, Liu B, Zeng A, Fuerer C, et al. Wnt proteins promote bone regeneration. Sci Transl Med. 2010;2(29):29ra30 doi: 10.1126/scitranslmed.3000231 . [DOI] [PubMed] [Google Scholar]
- 10.Sebastian A, Loots GG. Transcriptional control of Sost in bone. Bone. 2017;96:76–84. doi: 10.1016/j.bone.2016.10.009 . [DOI] [PubMed] [Google Scholar]
- 11.Riddle RC, Diegel CR, Leslie JM, Van Koevering KK, Faugere MC, Clemens TL, et al. Lrp5 and Lrp6 exert overlapping functions in osteoblasts during postnatal bone acquisition. PLoS One. 2013;8(5):e63323 doi: 10.1371/journal.pone.0063323 ; PubMed Central PMCID: PMCPMC3651091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Frey JL, Li Z, Ellis JM, Zhang Q, Farber CR, Aja S, et al. Wnt-Lrp5 signaling regulates fatty acid metabolism in the osteoblast. Mol Cell Biol. 2015;35(11):1979–91. doi: 10.1128/MCB.01343-14 ; PubMed Central PMCID: PMCPMC4420919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pinson KI, Brennan J, Monkley S, Avery BJ, Skarnes WC. An LDL-receptor-related protein mediates Wnt signalling in mice. Nature. 2000;407(6803):535–8. doi: 10.1038/35035124 . [DOI] [PubMed] [Google Scholar]
- 14.Kato M, Patel MS, Levasseur R, Lobov I, Chang BH, Glass DA 2nd, et al. Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J Cell Biol. 2002;157(2):303–14. doi: 10.1083/jcb.200201089 ; PubMed Central PMCID: PMCPMC2199263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gong Y, Slee RB, Fukai N, Rawadi G, Roman-Roman S, Reginato AM, et al. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell. 2001;107(4):513–23. . [DOI] [PubMed] [Google Scholar]
- 16.Boyden LM, Mao J, Belsky J, Mitzner L, Farhi A, Mitnick MA, et al. High bone density due to a mutation in LDL-receptor-related protein 5. N Engl J Med. 2002;346(20):1513–21. doi: 10.1056/NEJMoa013444 . [DOI] [PubMed] [Google Scholar]
- 17.Niziolek PJ, Farmer TL, Cui Y, Turner CH, Warman ML, Robling AG. High-bone-mass-producing mutations in the Wnt signaling pathway result in distinct skeletal phenotypes. Bone. 2011;49(5):1010–9. doi: 10.1016/j.bone.2011.07.034 ; PubMed Central PMCID: PMCPMC3412139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sawakami K, Robling AG, Ai M, Pitner ND, Liu D, Warden SJ, et al. The Wnt co-receptor LRP5 is essential for skeletal mechanotransduction but not for the anabolic bone response to parathyroid hormone treatment. J Biol Chem. 2006;281(33):23698–711. doi: 10.1074/jbc.M601000200 . [DOI] [PubMed] [Google Scholar]
- 19.Niziolek PJ, Warman ML, Robling AG. Mechanotransduction in bone tissue: The A214V and G171V mutations in Lrp5 enhance load-induced osteogenesis in a surface-selective manner. Bone. 2012;51(3):459–65. doi: 10.1016/j.bone.2012.05.023 ; PubMed Central PMCID: PMCPMC3784262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Castilla-Cortazar I, Guerra L, Puche JE, Munoz U, Barhoum R, Escudero E, et al. An experimental model of partial insulin-like growth factor-1 deficiency in mice. J Physiol Biochem. 2014;70(1):129–39. doi: 10.1007/s13105-013-0287-y . [DOI] [PubMed] [Google Scholar]
- 21.Holmen SL, Giambernardi TA, Zylstra CR, Buckner-Berghuis BD, Resau JH, Hess JF, et al. Decreased BMD and limb deformities in mice carrying mutations in both Lrp5 and Lrp6. J Bone Miner Res. 2004;19(12):2033–40. doi: 10.1359/JBMR.040907 . [DOI] [PubMed] [Google Scholar]
- 22.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–8. doi: 10.1006/meth.2001.1262 . [DOI] [PubMed] [Google Scholar]
- 23.Hudson BD, Hum NR, Thomas CB, Kohlgruber A, Sebastian A, Collette NM, et al. SOST Inhibits Prostate Cancer Invasion. PLoS One. 2015;10(11):e0142058 doi: 10.1371/journal.pone.0142058 ; PubMed Central PMCID: PMCPMC4636315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30(15):2114–20. doi: 10.1093/bioinformatics/btu170 ; PubMed Central PMCID: PMCPMC4103590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Trapnell C, Pachter L, Salzberg SL. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics. 2009;25(9):1105–11. doi: 10.1093/bioinformatics/btp120 ; PubMed Central PMCID: PMCPMC2672628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim D, Pertea G, Trapnell C, Pimentel H, Kelley R, Salzberg SL. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 2013;14(4):R36 doi: 10.1186/gb-2013-14-4-r36 ; PubMed Central PMCID: PMCPMC4053844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liao Y, Smyth GK, Shi W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics. 2014;30(7):923–30. doi: 10.1093/bioinformatics/btt656 . [DOI] [PubMed] [Google Scholar]
- 28.Robinson MD, McCarthy DJ, Smyth GK. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010;26(1):139–40. doi: 10.1093/bioinformatics/btp616 ; PubMed Central PMCID: PMCPMC2796818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Law CW, Chen Y, Shi W, Smyth GK. voom: Precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol. 2014;15(2):R29 doi: 10.1186/gb-2014-15-2-r29 ; PubMed Central PMCID: PMCPMC4053721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Quinlan AR, Hall IM. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics. 2010;26(6):841–2. doi: 10.1093/bioinformatics/btq033 ; PubMed Central PMCID: PMCPMC2832824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc. 2012;7(3):562–78. doi: 10.1038/nprot.2012.016 ; PubMed Central PMCID: PMCPMC3334321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen J, Bardes EE, Aronow BJ, Jegga AG. ToppGene Suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res. 2009;37(Web Server issue):W305–11. doi: 10.1093/nar/gkp427 ; PubMed Central PMCID: PMCPMC2703978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen G, Deng C, Li YP. TGF-beta and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci. 2012;8(2):272–88. doi: 10.7150/ijbs.2929 ; PubMed Central PMCID: PMCPMC3269610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chandra A, Lan S, Zhu J, Siclari VA, Qin L. Epidermal growth factor receptor (EGFR) signaling promotes proliferation and survival in osteoprogenitors by increasing early growth response 2 (EGR2) expression. J Biol Chem. 2013;288(28):20488–98. doi: 10.1074/jbc.M112.447250 ; PubMed Central PMCID: PMCPMC3711314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wu M, Chen G, Li YP. TGF-beta and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016;4:16009 doi: 10.1038/boneres.2016.9 ; PubMed Central PMCID: PMCPMC4985055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jacob AL, Smith C, Partanen J, Ornitz DM. Fibroblast growth factor receptor 1 signaling in the osteo-chondrogenic cell lineage regulates sequential steps of osteoblast maturation. Dev Biol. 2006;296(2):315–28. doi: 10.1016/j.ydbio.2006.05.031 ; PubMed Central PMCID: PMCPMC2077084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yu S, Yerges-Armstrong LM, Chu Y, Zmuda JM, Zhang Y. E2F1 effects on osteoblast differentiation and mineralization are mediated through up-regulation of frizzled-1. Bone. 2013;56(2):234–41. doi: 10.1016/j.bone.2013.06.019 ; PubMed Central PMCID: PMCPMC3758927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ohbayashi N, Shibayama M, Kurotaki Y, Imanishi M, Fujimori T, Itoh N, et al. FGF18 is required for normal cell proliferation and differentiation during osteogenesis and chondrogenesis. Genes Dev. 2002;16(7):870–9. doi: 10.1101/gad.965702 ; PubMed Central PMCID: PMCPMC186331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhou H, Mak W, Kalak R, Street J, Fong-Yee C, Zheng Y, et al. Glucocorticoid-dependent Wnt signaling by mature osteoblasts is a key regulator of cranial skeletal development in mice. Development. 2009;136(3):427–36. doi: 10.1242/dev.027706 . [DOI] [PubMed] [Google Scholar]
- 40.Jackson A, Vayssiere B, Garcia T, Newell W, Baron R, Roman-Roman S, et al. Gene array analysis of Wnt-regulated genes in C3H10T1/2 cells. Bone. 2005;36(4):585–98. doi: 10.1016/j.bone.2005.01.007 . [DOI] [PubMed] [Google Scholar]
- 41.Karner CM, Esen E, Chen J, Hsu FF, Turk J, Long F. Wnt Protein Signaling Reduces Nuclear Acetyl-CoA Levels to Suppress Gene Expression during Osteoblast Differentiation. J Biol Chem. 2016;291(25):13028–39. doi: 10.1074/jbc.M115.708578 ; PubMed Central PMCID: PMCPMC4933220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rawadi G, Vayssiere B, Dunn F, Baron R, Roman-Roman S. BMP-2 controls alkaline phosphatase expression and osteoblast mineralization by a Wnt autocrine loop. J Bone Miner Res. 2003;18(10):1842–53. doi: 10.1359/jbmr.2003.18.10.1842 . [DOI] [PubMed] [Google Scholar]
- 43.Bennett CN, Longo KA, Wright WS, Suva LJ, Lane TF, Hankenson KD, et al. Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc Natl Acad Sci U S A. 2005;102(9):3324–9. doi: 10.1073/pnas.0408742102 ; PubMed Central PMCID: PMCPMC552924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Song L, Liu M, Ono N, Bringhurst FR, Kronenberg HM, Guo J. Loss of wnt/beta-catenin signaling causes cell fate shift of preosteoblasts from osteoblasts to adipocytes. J Bone Miner Res. 2012;27(11):2344–58. doi: 10.1002/jbmr.1694 ; PubMed Central PMCID: PMCPMC3474875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Liang B, Cotter MM, Chen D, Hernandez CJ, Zhou G. Ectopic expression of SOX9 in osteoblasts alters bone mechanical properties. Calcif Tissue Int. 2012;90(2):76–89. doi: 10.1007/s00223-011-9550-9 ; PubMed Central PMCID: PMCPMC3272153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jullien N, Maudinet A, Leloutre B, Ringe J, Haupl T, Marie PJ. Downregulation of ErbB3 by Wnt3a contributes to wnt-induced osteoblast differentiation in mesenchymal cells. J Cell Biochem. 2012;113(6):2047–56. doi: 10.1002/jcb.24076 . [DOI] [PubMed] [Google Scholar]
- 47.Shin HR, Islam R, Yoon WJ, Lee T, Cho YD, Bae HS, et al. Pin1-mediated Modification Prolongs the Nuclear Retention of beta-Catenin in Wnt3a-induced Osteoblast Differentiation. J Biol Chem. 2016;291(11):5555–65. doi: 10.1074/jbc.M115.698563 ; PubMed Central PMCID: PMCPMC4786698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Caverzasio J, Biver E, Thouverey C. Predominant role of PDGF receptor transactivation in Wnt3a-induced osteoblastic cell proliferation. J Bone Miner Res. 2013;28(2):260–70. doi: 10.1002/jbmr.1748 . [DOI] [PubMed] [Google Scholar]
- 49.Wu Y, Zhang Y, Yin Q, Xia H, Wang J. Plateletderived growth factor promotes osteoblast proliferation by activating Gproteincoupled receptor kinase interactor1. Mol Med Rep. 2014;10(3):1349–54. doi: 10.3892/mmr.2014.2374 . [DOI] [PubMed] [Google Scholar]
- 50.Si W, Kang Q, Luu HH, Park JK, Luo Q, Song WX, et al. CCN1/Cyr61 is regulated by the canonical Wnt signal and plays an important role in Wnt3A-induced osteoblast differentiation of mesenchymal stem cells. Mol Cell Biol. 2006;26(8):2955–64. doi: 10.1128/MCB.26.8.2955-2964.2006 ; PubMed Central PMCID: PMCPMC1446962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.de Gorter DJ, van Dinther M, Korchynskyi O, ten Dijke P. Biphasic effects of transforming growth factor beta on bone morphogenetic protein-induced osteoblast differentiation. J Bone Miner Res. 2011;26(6):1178–87. doi: 10.1002/jbmr.313 . [DOI] [PubMed] [Google Scholar]
- 52.van Driel M, van Leeuwen JP. Vitamin D endocrine system and osteoblasts. Bonekey Rep. 2014;3:493 doi: 10.1038/bonekey.2013.227 ; PubMed Central PMCID: PMCPMC3944124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Friedman MS, Oyserman SM, Hankenson KD. Wnt11 promotes osteoblast maturation and mineralization through R-spondin 2. J Biol Chem. 2009;284(21):14117–25. doi: 10.1074/jbc.M808337200 ; PubMed Central PMCID: PMCPMC2682860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Qiu W, Chen L, Kassem M. Activation of non-canonical Wnt/JNK pathway by Wnt3a is associated with differentiation fate determination of human bone marrow stromal (mesenchymal) stem cells. Biochem Biophys Res Commun. 2011;413(1):98–104. doi: 10.1016/j.bbrc.2011.08.061 . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
A qPCR analysis showed that Wnt3a up-regulated known canonical Wnt target genes Axin2, Lef1, Igfbp2 and down-regulated Ibh, a gene suppressed by canonical Wnt signaling.
(TIF)
(PDF)
Fold changes are given in log2 scale.
(XLSX)
(PDF)
(PDF)
(XLSX)
Fold changes are given in log2 scale. Empty cells represent no significant change in expression.
(XLSX)
(PDF)
(PDF)
Fold changes are given in log2 scale. Empty cells represent no significant change in expression.
(XLSX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files. RNA-seq data has been deposited in NCBI and is available at https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE103492.