Dear Editor:
Atopic dermatitis (AD) is a chronic relapsing inflammatory skin disorder with severe itching and relapsing eczematous lesions1. The main principles of AD management are epidermal barrier repair, identification and elimination of trigger factors, anti-inflammatory therapy with topical steroid or calcineurin inhibitors, and phototherapy or systemic immunosuppressants in severe cases2,3,4. With regard to phototherapy, ultraviolet (UV) A1 (in the acute phase) and narrowband UVB (in the chronic phase) have been reported as the most suitable phototherapy modalities for AD treatment4. However, these conventional phototherapies have limitations, especially in pediatrics, because of a potentially increased cumulative risk of skin cancer5.
Recently, the therapeutic effects of low-level laser therapy (LLLT) with a light-emitting diode (LED) have been demonstrated. LEDs cover a wide spectrum ranging from UV to visible to near infra-red (NIR) bandwidth (247 to 1,300 nm)6. The energy level of an LED is low, and it is much more economical and safe than conventional laser sources7. In addition, it allows production of an efficient wavelength combination optimal for a variety of purposes, and can be prepared in all sizes for the treatment of small or large areas7. Therefore, LED phototherapy has become a treatment for various dermatological conditions, particularly because of its healing and anti-inflammatory properties6.
Despite the potential effects of LED or LLLT in AD treatment, there have been only two experimental trials8,9. The effects of LED phototherapy on AD as it correlates with wavelength remain largely unproven. Thus, this study aimed to investigate the therapeutic effects of LED irradiation with variable wavelengths and its immunomodulatory effects on AD-like skin lesions in a NC/Nga mouse model.
As an animal model of AD, four-week-old NC/Nga male mice, purchased from SLC Japan (Tokyo, Japan), were divided into six groups (n=5 per each group). Animal care, handling and experimental procedures were performed in accordance with a protocol approved by the Animal Care and Use Committee of the Catholic University of Korea (CUMC-2015-0124-01). Induction of AD using 2,4-dinitrochlorobenzene (DNCB) was performed as previously described10. After induction, the ear skin and back skin of mice was irradiated by an LED device, with 10 J/cm2 twice a week for 2 weeks (on days 1, 4, 8, and 11). The LED irradiation device (Korea Electronics Technology Institute, Seongnam, Korea) used in this study emitted 415 (4.23 mW/cm2), 525 (3.85 mW/cm2), 660 (2.42 mW/cm2), and 830 nm (24.72 mW/cm2), respectively. The results were evaluated on the last day of the experiment (day 11). Ear thickness was measured using a dial caliper (KoriSeiki MFG, Tokyo, Japan). Ear skin samples were obtained on day 11, fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned at a thickness of 5 µm. Tissue sections were stained with hematoxylin and eosin (H&E) and toluidine blue for counting the number of mast cells in five high power fields (×400). Interleukin (IL)-4 and interferon (IFN)-γ in the ear skin and serum, and serum immunoglobulin (Ig)E levels were measured by a quantitative reverse transcription-polymerase chain reaction and enzyme-linked immunosorbent assay, respectively, as previously described10. Statistical analysis was performed using the Kruskal-Wallis test followed by the Mann-Whitney test with Bonferroni correction. All data were expressed in box-and-whisker plots that represented the median, the lower and upper quartiles, and the minimum to the maximum value. A p-value less than 0.05 was considered significant.
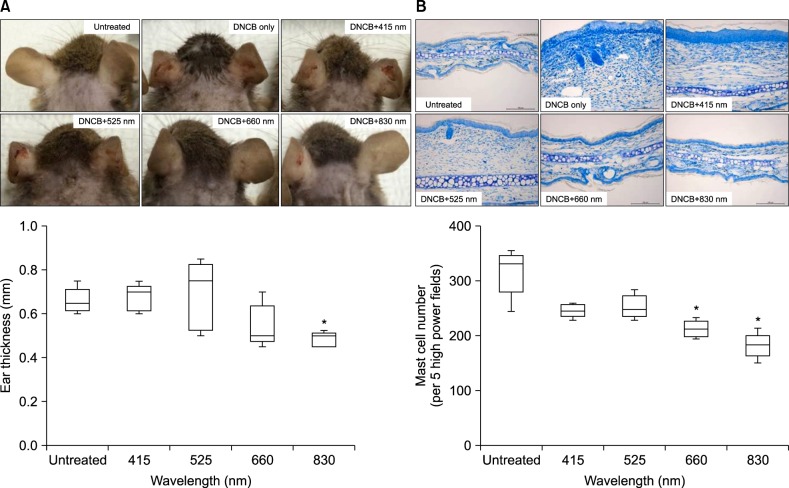
The representative clinical features of NC/Nga mice are shown in Fig. 1A. Compared with the non-irradiated group (DNCB only), the ear thickness of the 830-nm-irradiated group was significantly decreased (p<0.05), and that of the 660-nm-irradiated group was also decreased, although there was no significant difference (Fig. 1A). In ear skin, epidermal hyperplasia and inflammatory cell infiltrations, including eosinophils and mast cells in the dermis, were commonly decreased in all LED-irradiated groups, compared with the non-irradiated group (data not shown), and the number of mast cells was significantly decreased in the 660-nm and 830-nm-irradiated groups (p<0.05) (Fig. 1B).
Fig. 1. Clinical and histologic features of DNCB-induced AD-like lesions in NC/Nga mice. (A) Representative images of the ear lesions and ear thickness taken from each group on day 11. (B) Histological features of ear skin (toluidine blue stain, ×200) and number of mast cells in 5 randomly chosen visual fields at ×400 magnification. Data are expressed in box-and-whisker plots that represent the median, the lower and upper quartile and the minimum to the maximum values. DNCB: 2,4-dinitrochlorobenzene, AD: atopic dermatitis. *p<0.05 compared with the non-irradiated group (DNCB only).
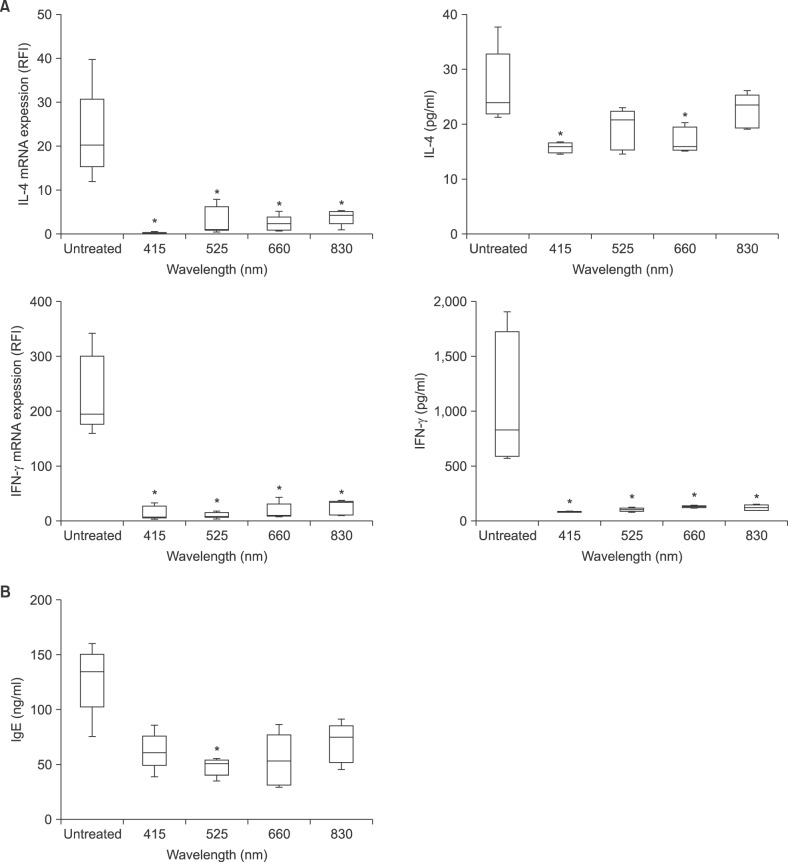
The mRNA expression of IL-4 and IFN-r in ear skin was significantly decreased in all LED-irradiated groups compared with the non-irradiated group (p<0.05). Compared with the non-irradiated group, the protein production of IFN-r in serum was also significantly decreased in all LED-irradiated groups (p<0.05), but there was no difference in serum IL-4 levels (Fig. 2A). Total serum IgE levels also showed decreased production in all LED-irradiated groups, compared with the non-irradiated group, although this difference was not significant, except in the 525-nm-irradiated group (Fig. 2B).
Fig. 2. Effects of light-emitting diode (LED) irradiation on IL-4, IFN-γ, and IgE production in DNCB-induced AD-like lesions of NC/Nga mice. (A) The expression of IL-4 and IFN-γ mRNA in ear skin lesions and serum IL-4 and IFN-γ levels, and (B) the serum IgE levels. Data are expressed in Box-and-Whisker plots that represent the median, the lower and upper quartile, and the minimum to the maximum values. IL: interleukin, IFN: interferon, IgE: immunoglobulin E, DNCB: 2,4-dinitrochlorobenzene, AD: atopic dermatitis. *p<0.05 compared with the non-irradiated group (DNCB only).
In this study, we analyzed the therapeutic effect of LED phototherapy with variable wavelengths on AD-like skin lesions induced by DNCB in NC/Nga mice. Clinical and histological analysis showed LED phototherapy with longer wavelengths (red and NIR) had significant therapeutic effects on AD-like skin lesions. In previous studies, LED phototherapy with exclusively NIR wavelengths (830 and 850 nm) also led to improvement in AD8,9.
The suppression of both IL-4 (Th2 subset) and IFN-r (Th1 subset) mRNA expression in this study implies that LED phototherapy might have therapeutic effects on both acute and chronic AD. Moreover, the decrease of IL-4 and IFN-γ production in serum suggested that LED phototherapy might exert systemic as well as local immunomodulatory effects on AD. These findings were also supported by decreases in serum IgE levels in all LED-irradiated groups. To elucidate the therapeutic mechanisms of LED phototherapy with variable wavelengths, further studies are needed.
In conclusion, our study demonstrates that LED phototherapy with longer wavelengths can improve AD-like skin lesions in NC/Nga mice, probably through regulation of both Th1 and Th2 responses. Thus, LED phototherapy with red and NIR wavelengths could be a potential phototherapeutic modality for AD management.
ACKNOWLEDGMENT
This work was supported by the Industrial Fundamental Technology Development Program (No. 10048898) funded by the Ministry of Trade, Industry & Energy (Korea).
Footnotes
CONFLICTS OF INTEREST: The authors have nothing to disclose.
References
- 1.Cui HS, Ahn IS, Byun YS, Yang YS, Kim JH, Chung BY, et al. Dietary pattern and nutrient intake of korean children with atopic dermatitis. Ann Dermatol. 2014;26:570–575. doi: 10.5021/ad.2014.26.5.570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schmitt J, von Kobyletzki L, Svensson A, Apfelbacher C. Efficacy and tolerability of proactive treatment with topical corticosteroids and calcineurin inhibitors for atopic eczema: systematic review and meta-analysis of randomized controlled trials. Br J Dermatol. 2011;164:415–428. doi: 10.1111/j.1365-2133.2010.10030.x. [DOI] [PubMed] [Google Scholar]
- 3.von Kobyletzki G, Pieck C, Hoffmann K, Freitag M, Altmeyer P. Medium-dose UVA1 cold-light phototherapy in the treatment of severe atopic dermatitis. J Am Acad Dermatol. 1999;41:931–937. doi: 10.1016/s0190-9622(99)70249-5. [DOI] [PubMed] [Google Scholar]
- 4.Gambichler T, Othlinghaus N, Tomi NS, Holland-Letz T, Boms S, Skrygan M, et al. Medium-dose ultraviolet (UV) A1 vs. narrowband UVB phototherapy in atopic eczema: a randomized crossover study. Br J Dermatol. 2009;160:652–658. doi: 10.1111/j.1365-2133.2008.08984.x. [DOI] [PubMed] [Google Scholar]
- 5.Pavlovsky M, Baum S, Shpiro D, Pavlovsky L, Pavlotsky F. Narrow band UVB: is it effective and safe for paediatric psoriasis and atopic dermatitis? J Eur Acad Dermatol Venereol. 2011;25:727–729. doi: 10.1111/j.1468-3083.2010.03832.x. [DOI] [PubMed] [Google Scholar]
- 6.Barolet D. Light-emitting diodes (LEDs) in dermatology. Semin Cutan Med Surg. 2008;27:227–238. doi: 10.1016/j.sder.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 7.Yeh NG, Wu CH, Cheng TC. Light-emitting diodes-Their potential in biomedical applications. Renew Sustain Energy Rev. 2010;14:2161–2166. [Google Scholar]
- 8.Morita H, Kohno J, Tanaka S, Kitano Y, Sagami S. Clinical application of GaAlAs 830 nm diode laser for atopic dermatitis. Laser Ther. 1993;5:75–78. [Google Scholar]
- 9.Cheong KA, Kim CH, Choi Y, Park CD, Lee AY. Irradiation of light emitting diode at 850nm inhibits T cell-induced cytokine expression. J Dermatol Sci. 2012;65:27–37. doi: 10.1016/j.jdermsci.2011.10.005. [DOI] [PubMed] [Google Scholar]
- 10.Jung KE, Lee YJ, Ryu YH, Kim JE, Kim HS, Kim BJ, et al. Effects of topically applied rapamycin and mycophenolic acid on TNCB-induced atopic dermatitis-like skin lesions in NC/Nga mice. Int Immunopharmacol. 2015;26:432–438. doi: 10.1016/j.intimp.2015.03.007. [DOI] [PubMed] [Google Scholar]