Abstract
The purpose of this study is to evaluate the patterns of treatment and factors affecting outcomes in ovarian metastases of colorectal origins treated at our institution and to assess the response of ovarian metastases to chemotherapy. Survival in R0 and R+ resections and patients receiving only chemotherapy is also analyzed. This is a retrospective study of 25 patients registered between January 2012 and December 2015. Patient’s age, disease status, mode of presentation, disease spread, mode of treatment, response to chemotherapy, completeness of resection, histology, and outcomes were considered as variables for analysis. There were 21 synchronous presentations and 4 metachronous presentations. In synchronous presentations, only 2 had extra-abdominal disease. Of these patients, 15 underwent surgeries of various extents. The remaining 6 patients were treated with chemotherarpy initially. Only one of them could come up to surgery later. In R0 resections, disease recurred between 1 and 9 months (median 3.5 months). The recurrence was in peritoneum and ovaries. In operated cases, 12 of 15 patients received chemotherapy and 9 patients progressed on first line chemotherapy. In all 4 metachronous patients, the disease was in the peritoneum. No one underwent surgery for the recurrence due to the extensive nature of the disease. All received chemotherapy. Sixty-six percent ovarian metastases showed progression on chemotherapy. There was no significant difference in the median survival between patients treated with surgery plus chemotherapy (23 months) vs. those treated with chemotherapy alone (28 months). Age and presence of disease at other sites did not affect the outcomes. Non-signet ring cell histologies showed better outcomes compared to signet ring cell histology (p = 0.02). Synchronous presentation, R0 resections, and responsive disease showed better survival, however it was clinically not significant. Treatment of ovarian metastases of colorectal origins is varied but has consistently poor outcome. Non-signet histology was the only prognostic factor which showed better outcome. Survival was not different between patients treated with surgery+chemotherapy and chemotherapy alone but majority of ovarian metastases progressed on chemotherapy. Considering the poor response to chemotherapy and peritoneum being the most common site of disease, both in primary and recurrent setting, R0 resection should always be attempted after selecting the correct patients using PET scan, laparoscopy and standard exploratory protocols. Treatment should be tailored upon patient’s status and disease burden with an aim to do complete cytoreduction whenever possible. CRS+HIPEC (cytoreductive surgery + hyperthermic intraperitoneal chemotherapy) can be considered on case to case basis as even R0 resections tend to recur.
Keywords: Ovarian metastases, Krukenberg tumors, Colorectal cancer
Introduction
Ovarian metastasis from a distant primary site is not an uncommon finding, whereas Krukenberg tumors are rare, accounting for only one to 2% of all ovarian tumors [1]. The word “Krukenberg tumor” has been used loosely to include all types of ovarian metastasis, but not all ovarian metastases are Krukenberg tumors. Krukenberg tumors are defined as ovarian metastases from gastrointestinal tumors which are characterized by mucin-filled signet ring cells. Novak and Gray have given the criteria for an ovarian metastasis to be called as a Krukenberg tumor, i.e., (1) cancer in the ovary, (2) presence of mucin producing signet ring cells, and (3) ovarian stromal sarcomatoid proliferation [2].
Colon appears to be the most common site of primary resulting in ovarian metastasis [3–6]. The presence of ovarian metastasis suggests aggressive and disseminated disease with poor outcomes. Management strategy varies from systemic chemotherapy alone, systemic chemotherapy and surgical debulking to systemic chemotherapy with cytoreductive surgery (CRS), and hyperthermic intraperitoneal chemotherapy (HIPEC), in no particular order. But the optimal management remains controversial due to lack of robust data [7, 8].
It is debatable whether ovarian metastases from all origins can be treated in a similar manner due to the inherent differences in the biological behavior, natural history and clinical profile. In this study, we have evaluated the outcome of ovarian metastasis of colorectal/appendicular origin treated at our institution. The aim of the study was to evaluate
Treatment patterns and factors affecting outcomes
Response of ovarian metastasis to systemic chemotherapy
Survival in R0 and R+ resections Vs. systemic chemotherapy
Materials and Methods
It is a retrospective study. All patients who are diagnosed with ovarian metastases/Krukenberg tumors between January 2012 and December 2015 were identified from pathology database and the relevant data was obtained from electronic medical records (EMR). Diagnosis of ovarian metastasis was based on clinico-radiological, endoscopic findings and histopathology including immunohistochemistry (IHC) which included CK7, CK 20, CDX 2, and PAX8. Patients with no documented primary were presumed to be having CRC (colorectal cancer) primary based on IHC characteristics. Parameters studied included patient’s demography, mode of presentation, disease spread, mode of treatment, response to chemotherapy, completeness of resection and histology. Response to chemotherapy was measured using RECIST criteria 1.1.
Overall survival (OS) was calculated in months, which was the time between the date of diagnosis and date of last follow-up or death. Cancer related death was considered as the end point event. Patients who are alive at the end of the study period, who died due to non-cancer related causes and who are lost to follow-up were censored. Overall survival (OS) was calculated using Kaplan-Meier method. The log-rank test was used to compare survival curves for different patient subgroups. Statistical analysis was done using SPSS version 20.
Results
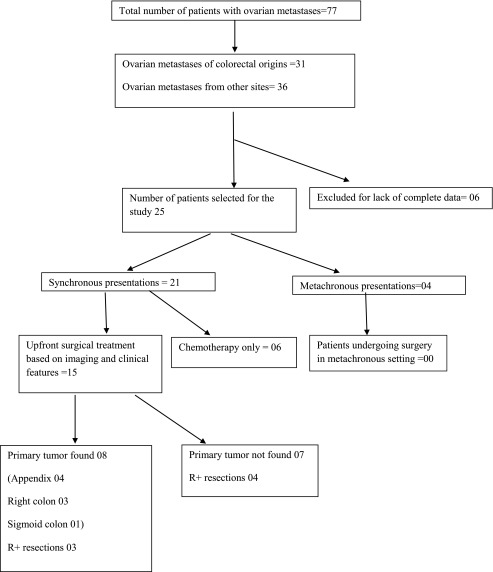
There were 77 patients with ovarian metastases, 31 were of colorectal origin and 36 were arising from other sites. Of the 31 cases from colorectal cancers (CRC), six were excluded for the lack of complete data. The remaining 25 were selected for this study. Patient characteristics and treatment details are given in Tables 1 and 2. Median age was 42 years (range 19–55 years). There were 21 synchronous and four metachronous presentations; 15 patients (60%) were premenopausal.
Table 1.
Patient selection and treatment characteristics
Table 2.
Details of surgeries performed
| Total number of patients | 25 |
| Synchronous presentations | 21 |
| Metachronous presentations | 04 |
| Surgical procedures: synchronous setting | |
| Unilateral adnexectomy | 02 |
| Unilateral adnexectomy+right hemicolectomy | 02 |
| Bilateral adnexectomy | 02 |
| Bilateral adnexectomy+appendectomy | 01 |
| Bilateral adnexectomy+right hemicolectomy | 01 |
| LSCS+bilateral adnexectomy | 01 |
| Sigmoid colectomy+removal of omental deposits+unilateral oophorectomy | 01 |
| Hysterectomy+bilateral salpingo oophorectomy+omentectomy | 02 |
| Hysterectomy+bilateral saplingo oophorectomy+omentectomy+appendectomy | 03 |
| Surgical procedure in metachronous setting: none | |
Synchronous Presentations
In 21 synchronous presentations, 14 had disease at other sites also, most commonly omental and peritoneal sites (eight patients each). The extra-abdominal disease was found in only two patients (one pleural effusion and one lung metastasis). Of 21 patients, 15 patients underwent upfront surgical treatment based on clinical and imaging characteristics, in which there were seven R+ resections (Table 1). Peritoneal metastasis was the most common reason for R+ resection (four patients) followed by liver metastasis (two patients) and abdominal wall invasion (one patient).
In eight patients, primary tumor was found and addressed (four appendectomies, three right hemicolectomies, and one sigmoid colectomy). In seven patients, the primary tumor was not found in spite of thorough preoperative and intraoperative examination. Appendectomy was not performed in any of these patients. These were treated as CRC based on IHC of the ovarian metastasis. (Table 1).
Six of 21 synchronous presentations were deemed unfit for upfront surgery due to the presence of extensive disease or poor performance status and received chemotherapy. Only one of these patients could later come up for surgery.
In five synchronous presentations, only the affected ovary was removed and disease recurred in the other ovary in four cases.
Fourteen of the synchronous cases were signet ring carcinomas, one was low-grade mucinous adenocarcinoma, and six were poorly differentiated adenocarcinomas. In R0 resections, disease recurred between 1 to 9 months (median 3.5 months). The recurrence was in peritoneum and remaining ovary.
Special Cases
Only one patient of rectal cancer in the cohort underwent CRS and HIPEC and is alive at the end of the study period (20 months post op). One patient underwent bilateral adnexectomy at the time of cesarean section. A 17-year-old girl underwent adnexectomy for ovarian mass which was found to be adenocarcinoma of colorectal origin on IHC. She was kept under observation. She is disease-free till now (24 months post op). In these last two patients, no primary tumor was found intraoperatively.
In operated cases, 12/15 patients received chemotherapy (six R0 patients, six R+ patients). Those who did not receive chemotherapy were the special cases mentioned above. Nine patients progressed on first-line chemotherapy and received second-line chemotherapy, and three received third-line chemotherapy.
Metachronous Presentations
There were four metachronous presentations. Two patients had received prior chemotherapy and two had received chemotherapy and radiation. In all four patients, the disease was in the peritoneum. No one underwent surgery for the recurrence due to the extensive nature of the disease. All received chemotherapy. Second-line chemotherapy was given to two patients with disease progression.
In all, 22 patients received chemotherapy (either upfront or after surgical intervention). CAPOX (capecitabine+oxaliplatin) was the most common chemotherapy regimen used (59%, 13/22). FOLFOX, CAPIRI, and FOLFIRI were the other regimen used. In three patients, combination of paclitaxel and carboplatin was used since the presentation mimicked primary ovarian malignancy. And 13 patients received second-line chemotherapy (59%).
Response Assessment
In 12 patients, at least one ovary was in situ. In these 12 cases, disease at other sites was in the form of ascites, mesenteric and peritoneal nodules, intra-abdominal nodes and omental deposits. Of the 12 patients, 66% showed progression on first-line chemotherapy. Eighty percent showed progression on second-line chemotherapy. One patient receiving third-line chemotherapy also showed progression on chemotherapy. There was documented disease progression in 68% patients (18/25).
Survival Analysis
The median duration of follow-up was 10 months (range 0–29 months). Overall survival (OS) is defined as the time period between date of diagnosis and date of death or last follow-up. Patients who are alive at the end of the study period are censored. There was no significant difference in the median survival between patients treated with surgery and chemotherapy (23 months) Vs. those treated with chemotherapy alone (28 months) (p = 0.376). Age and presence of disease at other sites (peritoneal or visceral) did not affect the outcomes in both categories. Patients with non-signet ring cell histologies had better outcomes compared to those with signet ring cell histolgy (p = 0.02), wherein signet ring patients had a median survival of just 12 months.
The synchronous presentation showed better survival compared to metachronous presentation (median OS 23.6 Vs. 12.2 months) and R0 resections had better survival compared to R+ resections (mean OS 22 Vs. 12 months) but both were not clinically significant. Responsive disease showed a trend toward better overall survival (p = 0.085). Stable disease had better outcomes compared to progressive disease but it was not clinically significant (Table 3).
Table 3.
Univariate analysis of prognostic factors
| Prognostic factor | Median OS in months | Mean OS in months | p value |
|---|---|---|---|
| Age | |||
| • <40 • >40 |
24 29 |
19.349 20.055 |
0.794 |
| Presence of disease at other sites | |||
| • Yes • No |
17.3 Not reached |
17.7 22.43 |
0.307 |
| Presentation | |||
| • Synchronous • Metachronous |
23.6 12.2 |
19.4 19.6 |
0.655 |
| Disease response | |||
| • Stable • Progression |
Not reached 12 months |
– | 0.085 |
| Histology | |||
| • Signet • Non-signet |
12 Not reached |
14 25 |
0.02 |
| Resection status | |||
| • R0 • R+ |
Not reached 17 |
22 12 |
0.357 |
Discussion
Ovarian metastases from colorectal adenocarcinoma occur in 3 to 4% of cases of CRCs [9].
Lymphatic permeation at the hilum, absence of surface deposits, and peritoneal seedlings indicate a retrograde spread to the ovaries rather than a transperitoneal spread in case of gastric malignancies [1] but in CRC contiguous spread through peritoneal cavity and retrograde lymphatic spread are cited as important reasons. The role of hematogenous spread to ovaries is not clear. Different types of tumor cells may spread via different routes [10, 11]. It is well recognized that adenocarcinomas composed of signet ring cells of various organs tend to metastasize to the ovaries much more commonly than adenocarcinomas of other histological types from the same sites [1]. In the present study, 64% (16/25) of patients had signet ring cell cancer.
In a large study consisting of more than 3000 CRC patients, synchronous presentation of peritoneal carcinomatosis was more common than metachronous presentation; 58% of the synchronous metastasis did not have systemic disease and 80% of synchronous metastasis were localized [12]. These results highlight the fact that there is a significant chance, in synchronous setting, of a radical cure. Overall, recurrences are limited to the peritoneum in 25% of patients with CRC [13], giving a possible chance of second surgery still aimed at R0 resection and cure. In the present study, 21 had the synchronous presentation, with only two patients having extra-abdominal disease. Of these, eight patients underwent R0 resection. Of the four patients with the metachronous presentation, no one underwent surgery.
Most of the literature about Krukenberg tumors focuses on epidemiological and clinical features, and there is a paucity of definitive treatment guidelines due to the rarity of the disease. Several authors have supported the beneficial effects of metastatectomy in this setting [14–17], but to what extent and the timing are not clear. Multiple prognostic factors have been cited by various studies. But most of the studies are done over a long period of time and are retrospective with small numbers (Table 4). R Jiang et al. used cytoreduction and IPC (intraperitoneal chemotherapy) in treating patients with Krukenberg tumors and PC (peritoneal carcinomatosis). Still, the overall survival was low at 17.8 months. R2 resection was a poor prognostic factor in the study but whether IPC helped improve the outcome is not clearly mentioned [8]. In Ayhan et al.’s [14] study, the high overall survival can be attributed to the inclusion of lymphomas and breast primaries (Table 4).
Table 4.
Comparisons of prognostic factors from other studies
| Author | Study period/type | No. of patients | Primary | Treatment modalities | Med OS (in months) | Poor prognostic factors |
|---|---|---|---|---|---|---|
| R Jiang [8] | 1997–2003 Retrospective |
54 | Multiple origins | Surgery+intraperitoneal chemotherapy | 17.8 | Gastric origin R2 surgery Lower KPS |
| Ayhan et al. [14] | 1982–2004 Retrospective |
154 | Multiple origins | Surgery+adjuvant therapy | 48 | Young age Premenopausal status Primary site(lymphoma>breast>colorectal>stomach+appendix>unknown primary) Suboptimal surgery Diffuse peritoneal involvement |
| Rayson et al. [16] | 1984–1998 Retrospective |
38 | Colorectal | Surgery+adjuvant therapy | 31 (pelvic-only disease with R0 resection) 14 (months in R+ and extrapelvic disease) 34.5 in age <50 17 in age >50 |
Extrapelvic disease R+ resection Age >50 |
| Fang wu et al. [7] | 1990–2010 Retrospective |
128 | Multiple origins | Surgery+adjuvant therapies | 16 | Synchronous metastasis Pelvic invasion Ascites No metastatectomy |
| Kim et al. [15] | 1994–2006 Retrospective |
34 | Stomach | Surgery+chemotherapy | 10.9 vs. 7.5 | Gross residual disease Extensive disease |
| Present study | 2012–2015 Retrospective |
25 | Colorectal | Surgery+chemotherapy | 23 | Signet ring histology |
The macroscopic residual disease has worse outcomes compared to complete excision/microscopic residual disease [8, 14]. This was also noted in the present study though it was not clinically significant. However, the reason for macroscopic residual disease and adjuvant treatments are not clearly mentioned in many of these studies. Rayson et al. [16] noted improved survival in complete excision and pelvic-only disease, stressing the need for aggressive surgery in selected patients and adequate staging to define goals of surgery. In the present study, R0 resection and synchronous presentations showed a trend toward better survival but not clinically significant, probably due to small sample size.
Forty-five percent CRC metastases are mistaken for ovarian primaries [6, 18]. This could be due to lack of adequate imaging and anticipation. Such a scenario leads to a high chance of incomplete resections. Reasons for incomplete resections have not been elucidated in literature. In the present series, the presence of peritoneal disease was the most common cause of incomplete resections. In a clinical scenario where one encounters an ovarian metastasis/Krukenberg tumor incidentally, a proper evaluation of peritoneal disease must be made. If patient’s condition and tumor burden permit, complete resection must be attempted.
Most of the ovarian metastases are associated with peritoneal surface metastasis. Sensitivity of CT scan in the diagnosis of PC has been reported as 70% for lesions measuring 2 cm in the greatest dimension and only 28% for lesions less than 5 mm in the greatest dimension [19, 20]. PET has been evaluated in the setting of peritoneal carcinomatosis. In one study of 55 patients, PET CT showed PPV of 78% for nonmucinous tumors and 55% for mucinous tumors [21]. However, low performance of PET in the setting of mucinous tumors should be kept in mind while using it in cases suspected to be Krukenberg tumors.
Laparoscopy is also a useful tool in the evaluation. In a study of 197 patients undergoing diagnostic laparoscopy for peritoneal carcinomatosis, full PCI (peritoneal carcinomatosis index) estimation was possible in all but one patient (99.49%) with under-staging of just 2% of patients [22]. Hence, laparoscopy should be considered whenever there is a reasonable doubt of an adnexal mass being an ovarian metastasis. In the present study, laparoscopy was not used as a diagnostic or staging tool.
There are instances where the primary tumor is not found in spite of extensive investigations. In such cases, it may be beneficial to remove the appendix even if it is macroscopically normal since it may harbor occult primary [23, 24]. In the present study, in all the seven cases of synchronous ovarian metastases where no primary was found, appendix was not removed in any of these patients. There is also a possibility of the presence of signet ring cells in primary ovarian cancer leading to the confusion of Krukenberg [1]. In the present study, any suspicion of ovarian metastasis of CRC origin was confirmed with IHC.
There are clinical scenarios where ovarian metastsis is encountered which is extremely limited without any disease at other sites in the peritoneal cavity. Removal of the other normal ovary in the setting of unilateral ovarian involvement is not supported by some. But the development of metastasis in the other ovary is a possibility and many times the patient’s general condition will not permit a second surgery. Hence, Yamaguchi et al. recommend prophylactic removal of the normal ovary in unilateral ovarian metastasis [25]. Banerjee et al. advocates prophylactic bilateral oophorectomy in postmenopausal age with a history of ovarian cancer and in left-sided CRCs. But the rarity of ovarian met in CRC would preclude it from becoming standard in all female CRCs [9]. In the present study, in five synchronous presentations, only the affected ovary was removed and disease recurred in the other ovary in four cases. Hence, we believe bilateral oophorectomy has to be done in all cases of unilateral ovarian metastasis.
CRS+HIPEC have become the standard for PC in CRC with median survival reaching 40 months in some studies, but most of this data comes from retrospective/phase2 studies from single centers [26]. The only RCT in this regard was done by Verwaal et al. comparing CRS+HIPEC+systemic therapy vs. systemic chemotherapy+/−palliative surgery in CRC. This study showed that CRS+HIPEC gave significant improvement in survival [27], compared to systemic therapy alone. Since ovarian lining and peritoneal lining are derived from the same embryological origins, the same results can be extrapolated to cases of ovarian metastases. And this has been confirmed in a study comparing PC patients with ovarian metastases undergoing CRS+HIPEC to PC patients without ovarian metastases undergoing CRS+HIPEC, wherein both groups showed similar survival rates [28]. Even the studies addressing CRS+HIPEC have stressed the need and significance of optimal cytoreduction to have a meaningful improvement in outcomes [29–31] while the role of HIPEC is still debated. However, one should not forget that only 5% of CRC with PC ever get CRS+HIPEC and it is not advised in poorly differentiated and signet ring varieties which form a considerable portion of ovarian metastasis of CRC origin. There may be a role for relook surgery with CRS+HIPEC in patients with ovarian metastases as they indicate a high risk for peritoneal relapse.
PET scan and diagnostic laparoscopy can be used effectively to select patients for optimal cytoreduction in cases of ovarian metastases. Standardization of exploratory laparotomy is also necessary to correctly assess the disease burden [27] as it has been shown that higher disease burden indicates a poor prognosis [27, 29, 30].
Verwaal et al.’s RCT had shown that systemic chemotherapy alone was inferior to CRS + HIPEC + systemic chemotherapy but there have been significant advances in chemotherapy for metastatic CRC and a recent retrospective study has shown that CRS with systemic chemo may give similar results to CRS + HIPEC [32] indicating the importance of systemic therapy. However, there are reports that have positively stated that ovarian metastasis is less responsive to chemotherapy [33, 34] even when the metastasis at other sites respond. In the present study, 10 patients had initial chemotherapy but only one could be up for surgery, and 66% ovarian metastases progressed on first-line chemotherapy and 80% progressed on second-line chemotherapy. In such a scenario, the decision to give initial chemotherapy should be based on the presence of metastasis at other sites, patient’s general condition to tolerate cytoreductive surgery and symptoms from the ovarian metastasis; while adjuvant chemotherapy may still have a role.
Shortcomings of the study
It is a retrospective analysis with considerable heterogeneity in patients and treatment patterns. The number is small so the results have to be considered with caution. Many factors which are showing better outcomes without clinical significance may become significant in larger sample sizes.
Conclusions
Limited conclusions can be drawn from this study. The treatment pattern of ovarian metastases from CRC is varied but the outcome is consistently poor. The survival does not seem to be significantly different between those treated by surgery + chemotherapy and those treated by chemotherapy alone, but most of the patient’s progress on chemotherapy. Non-signet ring cell histology was the only significant factor. R0 resection showed better outcomes but were clinically not significant. These results have to be interpreted with caution due to small sample size and heterogeneity of patients and treatment.
However, based on our findings and available evidence, we can safely recommend that in all cases of ovarian metastases from CRC, the goal should be R0 resection whenever possible as it definitely improves survival. The normal-looking ovary also has to be removed. PET scan, laparoscopy, and standardized exploration protocols will help in correct patient selection. Systemic chemotherapy can be offered up front in high disease burden and unfit patients though most are likely to progress. Postoperative chemotherapy may still have an important role though not proven prospectively. CRS + HIPEC can be explored on case to case basis as even R0 resection with systemic chemotherapy have shown to relapse. But no robust evidence for HIPEC is available at present.
The varied nature of the presentation and different treatment approaches by different specialties makes a well-designed prospective study a challenging prospect. The treatment needs to be tailored to the patient to gain maximum benefit with minimal adverse effects. The dearth of data in this scenario calls for maintenance of a registry.
References
- 1.Al-Agha OM, Nicastri AD. An in-depth look at Krukenberg tumor: an overview. Arch Pathol Lab Med. 2006;130(11):1725–1730. doi: 10.5858/2006-130-1725-AILAKT. [DOI] [PubMed] [Google Scholar]
- 2.Novak C, Gray LA. Krukenberg tumor of the ovary: clinical and pathological study of four cases. Surg Gynecol Obstet. 1938;66(1):157–167. [Google Scholar]
- 3.Mazur MT, Hsueh S, Gersell DJ. Metastases to the female genital tract. Analysis of 325 cases. Cancer. 1984;53:1978–1984. doi: 10.1002/1097-0142(19840501)53:9<1978::AID-CNCR2820530929>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 4.Taylor AE, Nicolson VM, Cunningham D. Ovarian metastases from primary gastrointestinal malignancies: the Royal Marsden Hospital experience and implications for adjuvant treatment. Br J Cancer. 1995;71:92–96. doi: 10.1038/bjc.1995.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moore RG, Chung M, Granai CO, Gajewski W, Steinhoff MM. Incidence of metastasis to the ovaries from nongenital tract primary tumors. Gynecol Oncol. 2004;93(1):87–91. doi: 10.1016/j.ygyno.2003.12.039. [DOI] [PubMed] [Google Scholar]
- 6.Ulbright TM, Roth LM, Stehman FB. Secondary ovarian neoplasia. A clinicopathologic study of 35 cases. Cancer. 1984;53(5):1164–1174. doi: 10.1002/1097-0142(19840301)53:5<1164::AID-CNCR2820530523>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 7.Wu F, Zhao X, Mi B, Feng LU, Yuan NA, Lei F, et al. Clinical characteristics and prognostic analysis of Krukenberg tumor. Mol Clin Oncol. 2015;3(6):1323–1328. doi: 10.3892/mco.2015.634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jiang R, Tang J, Cheng X, Zang RY. Surgical treatment for patients with different origins of Krukenberg tumors: outcomes and prognostic factors. Eur J Surg Oncol. 2009;35(1):92–97. doi: 10.1016/j.ejso.2008.05.006. [DOI] [PubMed] [Google Scholar]
- 9.Banerjee S, Kapur S, Moran BJ. The role of prophylactic oophorectomy in women undergoing surgery for colorectal cancer. Color Dis. 2005;7(3):214–217. doi: 10.1111/j.1463-1318.2005.00770.x. [DOI] [PubMed] [Google Scholar]
- 10.Bruls J, Simons M, Overbeek LI, Bulten J, Massuger LF, Nagtegaal ID. A national population-based study provides insight in the origin of malignancies metastatic to the ovary. Virchows Arch. 2015;467(1):79–86. doi: 10.1007/s00428-015-1771-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chang TC, Changchien CC, Tseng CW, Lai CH, Tseng CJ, Lin SE, et al. Retrograde lymphatic spread: a likely route for metastatic ovarian cancers of gastrointestinal origin. Gynecol Oncol. 1997;66(3):372–377. doi: 10.1006/gyno.1997.4793. [DOI] [PubMed] [Google Scholar]
- 12.Jayne DG, Fook S, Loi C, Seow-Choen F. Peritoneal carcinomatosis from colorectal cancer. Br J Surg. 2002;89(12):1545–1550. doi: 10.1046/j.1365-2168.2002.02274.x. [DOI] [PubMed] [Google Scholar]
- 13.Chu DZ, Lang NP, Thompson C, Osteen PK, Westbrook KC. Peritoneal carcinomatosis in nongynecologic malignancy. A prospective study of prognostic factors. Cancer. 1989;63(2):364–367. doi: 10.1002/1097-0142(19890115)63:2<364::AID-CNCR2820630228>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 14.Ayhan A, Guvenal T, Salman MC, Ozyuncu O, Sakinci M, Basaran M. The role of cytoreductive surgery in nongenital cancers metastatic to the ovaries. Gynecol Oncol. 2005;98(2):235–241. doi: 10.1016/j.ygyno.2005.05.028. [DOI] [PubMed] [Google Scholar]
- 15.Kim WY, Kim T-J, Kim SE, Lee J-W, Lee J-H, Kim B-G, et al. The role of cytoreductive surgery for non-genital tract metastatic tumors to the ovaries. Eur J Obstet Gynecol Reprod Biol. 2010;149(1):97–101. doi: 10.1016/j.ejogrb.2009.11.011. [DOI] [PubMed] [Google Scholar]
- 16.Rayson D, Bouttell E, Whiston F, Stitt L. Outcome after ovarian/adnexal metastectomy in metastatic colorectal carcinoma. J Surg Oncol. 2000;75(3):186–192. doi: 10.1002/1096-9098(200011)75:3<186::AID-JSO6>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 17.Cheong JH, Hyung WJ, Chen J, Kim J, Choi SH, Noh SH. Survival benefit of metastasectomy for Krukenberg tumors from gastric cancer. Gynecol Oncol. 2004;94(2):477–482. doi: 10.1016/j.ygyno.2004.05.007. [DOI] [PubMed] [Google Scholar]
- 18.Santesson L, Kottmeier HL General classification of ovarian tumours. In: Ovarian cancer [Internet]. Springer; 1968 [cited 2016 May 9]. p 1–8. Available from: http://link.springer.com/chapter/10.1007/978-3-642-87755-1_1
- 19.Jacquet P, Jelinek JS, Steves MA, Sugarbaker PH. Evaluation of computed tomography in patients with peritoneal carcinomatosis. Cancer. 1993;72(5):1631–1636. doi: 10.1002/1097-0142(19930901)72:5<1631::AID-CNCR2820720523>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 20.Goerg C, Schwerk WB. Peritoneal carcinomatosis with ascites. AJR Am J Roentgenol. 1991;156(6):1185–1187. doi: 10.2214/ajr.156.6.2028864. [DOI] [PubMed] [Google Scholar]
- 21.De Vos N, Goethals I, Ceelen W. Clinical value of (18)F-FDG- PET-CT in the preoperative staging of peritoneal carcinomatosis from colorectal origin. Acta Chir Belg. 2014;114(6):370–375. [PubMed] [Google Scholar]
- 22.Garofalo A, Valle M. Laparoscopy in the management of peritoneal carcinomatosis. Cancer J. 2009;15(3):190–195. doi: 10.1097/PPO.0b013e3181a58e93. [DOI] [PubMed] [Google Scholar]
- 23.Forsgren L, Molin K, Rieger A. Adenocarcinoma of the vermiform appendix. Acta Chir Scand. 1974;140(6):486–490. [PubMed] [Google Scholar]
- 24.Mandai M, Konishi I, Tsuruta Y, Suginami N, Kusakari T, Iwasaki T, et al. Krukenberg tumor from an occult appendiceal adenocarcinoid: a case report and review of the literature. Eur J Obstet Gynecol Reprod Biol. 2001;97(1):90–95. doi: 10.1016/S0301-2115(00)00503-0. [DOI] [PubMed] [Google Scholar]
- 25.Yamaguchi T, Takahashi H, Kagawa R, Takeda R, Sakata S, Yamamoto M, et al. The role of prophylactic bilateral oophorectomy at the time of initial diagnosis of a unilateral ovarian metastasis in cases with colorectal adenocarcinoma. Hepato-Gastroenterology. 2008;55(82–83):434–437. [PubMed] [Google Scholar]
- 26.Esquivel J, Elias D, Baratti D, Kusamura S, Deraco M. Consensus statement on the loco regional treatment of colorectal cancer with peritoneal dissemination. J Surg Oncol. 2008;98:263–267. doi: 10.1002/jso.21053. [DOI] [PubMed] [Google Scholar]
- 27.Verwaal VJ, van Ruth S, de Bree E, van Slooten GW, van Tinteren H, Boot H, et al. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J Clin Oncol. 2003;21:3737–3743. doi: 10.1200/JCO.2003.04.187. [DOI] [PubMed] [Google Scholar]
- 28.Bignell M, Mehta A, Alves S, Dayal S, Mohamed F, Cecil T, Moran B. Impact of ovarian Krukenberg tumours on survival in patients treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for peritoneal malignancy of appendiceal and colorectal cancer origin. Poster Abstracts Colorectal Dis. 2016;18:13–76. doi: 10.1111/codi.14057. [DOI] [PubMed] [Google Scholar]
- 29.Sugarbaker PH, Schellinx ME, Chang D, et al. Peritoneal carcinomatosis from adenocarcinoma of the colon. World J Surg. 1996;20:585–591. doi: 10.1007/s002689900091. [DOI] [PubMed] [Google Scholar]
- 30.Elias DM, Ouellet JF. Intraperitoneal chemohyperthermia: rationale, technique, indications, and results. Surg Oncol Clin North Am. 2001;10:915–933. [PubMed] [Google Scholar]
- 31.Verwaal VJ, Bruin S, Boot H, van Slooten G, van Tinteren H. 8-year follow-up of randomized trial: cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann Surg Oncol. 2008;15:2426–2432. doi: 10.1245/s10434-008-9966-2. [DOI] [PubMed] [Google Scholar]
- 32.Désolneux G, Mazière C, Vara J, et al. Cytoreductive surgery of colorectal peritoneal metastases: outcomes after complete cytoreductive surgery and systemic chemotherapy only. Trevino JG, ed. PLoS One. 2015;10(3):e0122816. doi: 10.1371/journal.pone.0122816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goéré D, Daveau C, Elias D, Boige V, Tomasic G, Bonnet S, et al. The differential response to chemotherapy of ovarian metastases from colorectal carcinoma. Eur J Surg Oncol. 2008;34(12):1335–1339. doi: 10.1016/j.ejso.2008.03.010. [DOI] [PubMed] [Google Scholar]
- 34.Sekine K, Hamaguchi T, Shoji H, Nakamura S, Miyamoto T, Takashima A, Honma Y, Iwasa S, Kato K, Takahashi K, Kato T, Kanemitsu Y, Boku N. Retrospective analysis of the effects of systemic chemotherapy to the ovarian metastases from colorectal cancer. J Clin Oncol. 2017;35(suppl 4S):abstract 731. doi: 10.1200/JCO.2017.35.4_suppl.731. [DOI] [Google Scholar]