Abstract
Purpose
There are limited long-term data on patients treated with image guided intensity modulated radiation therapy (IG-IMRT) for prostate cancer recurrence or high-risk disease features after radical prostatectomy. We report single-institution results for patients treated with IG-IMRT and identify variables associated with outcome.
Methods and materials
This is a retrospective chart review consisting of 313 consecutive patients who were treated with adjuvant or salvage IG-IMRT from 2004 to 2013. Cox proportional hazards analysis was used to identify factors related to survival and toxicity. Toxicity was graded using the Common Terminology Criteria for Adverse Events Version 4.0.
Results
The median follow-up was 55 months (range, 6-131 months). The median pre-radiation therapy (RT) prostate-specific antigen (PSA) was 0.3 ng/mL (range, <0.01-55.4). The vast majority of patients (87%) received elective pelvic nodal irradiation (median dose: 45 Gy). Androgen deprivation therapy (ADT) was given to 39% of patients for a median of 9 months. Five-year biochemical progression-free survival and distant metastasis-free survival were 59% (95% confidence interval, 53%-66%) and 89% (95% confidence interval, 85%-93%), respectively. On multivariate analysis, higher pre-RT PSA (>0.2 ng/mL), biopsy Gleason score (≥7 [4+3]), and duration of ADT (>6 months) were significantly associated (P < .05) with biochemical progression-free survival. Actuarial late grade 3 genitourinary and gastrointestinal toxicities at 5 years were 10% and 2%, respectively.
Conclusion
Our results suggest that lower pre-RT PSA level and longer duration of ADT are associated with improved biochemical control. The incidence of late grade 3 gastrointestinal toxicity was low, but late grade 3 genitourinary toxicity was higher than anticipated.
Summary.
There are limited long-term data on patients treated with image guided intensity modulated radiation therapy (IG-IMRT) for prostate cancer recurrence after radical prostatectomy. We report the largest series of patients treated with adjuvant or salvage IG-IMRT to identify factors related to survival and toxicity. Our results suggest that initiation of IG-IMRT at lower prostate-specific antigen levels is associated with superior biochemical progression-free survival and that late grade 3 genitourinary toxicity may be higher than anticipated.
Alt-text: Unlabelled box
Introduction
Approximately one-third of men with prostate cancer treated with radical prostatectomy (RP) experience biochemical failure, with increased rates in patients with high-risk features.1 Three randomized studies have shown the benefit of adjuvant radiation therapy (RT) on biochemical disease-free survival in men with pathologic T3 disease or positive margins,2, 3, 4 with one study showing a metastasis-free and overall survival benefit.2
The rates of biochemical failure remain high after postoperative radiation, ranging from 39% to 44% at 10 years with adjuvant treatment and up to 60% at 5 years for salvage treatment.5 Factors associated with recurrence after adjuvant and salvage RT are not well understood, and the ideal timing for starting radiation is controversial.
Although published phase 3 studies used conventional2, 3 or 3-dimensional conformal radiation,4 the predominant technique is now intensity modulated radiation therapy (IMRT). As the use of IMRT for intact prostate cancer has become standard practice, there has been an increase in acceptance of IMRT in the postprostatectomy setting as well.6 Several retrospective postprostatectomy IMRT series have shown low rates of toxicity7, 8, 9 and rates of biochemical progression-free survival (bPFS) comparable to non-IMRT studies,7, 8, 10 but long-term data are limited.
In 2004, our institution began treating postprostatectomy patients with helical tomotherapy (HT). The aim of this study is to report outcomes from this experience and to identify significant factors associated with survival and toxicity.
Methods and materials
This single-institution retrospective study was approved by the institutional review board. Charts were reviewed for patients who were treated with IG-IMRT between November 2004 and December 2013. Eligible patients were treated after RP with HT. Patients were excluded from the study if they had metastatic disease, pathologic T4 disease, positive pelvic lymph nodes, or less than 6 months of follow-up.
Treatment characteristics
The majority of patients received radiation to the prostate bed and pelvic lymph nodes (87%). The median doses to the lymph nodes and prostate bed were 45 Gy and 67 Gy (range, 59.4-77.4 Gy), respectively. All patients were treated in 1.8 to 2 Gy daily fractions. Patients were instructed to have a full bladder and empty rectum for simulation and treatments. Additional details on simulation and treatment planning have been previously described.11 Daily megavoltage computed tomography images were acquired before every fraction. Patients were seen weekly during RT for toxicity evaluation.
Toxicity grading
Acute (within 90 days from the start of RT) and late (>90 days from the start of RT) toxicity were retrospectively graded using the Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0.12 CTCAE was chosen because it includes detailed descriptions for scoring toxicity, and Version 4.0 was used because it was the most current version at the time of chart review. Any new toxicity or exacerbation of existing baseline toxicity was included. Toxicities that were scored by the treating physicians using the Radiation Therapy Oncology Group (RTOG) criteria or older versions of CTCAE were regraded at the time of chart review on the basis of the description of the event.
Endpoint definitions and statistical analysis
Adjuvant treatment was defined as starting RT within 16 weeks of prostatectomy. Patients who started radiation more than 16 weeks after prostatectomy were considered salvage. This definition was chosen because it was used in 2 of the randomized adjuvant trials.2, 3 Pre-RT prostate-specific antigen (PSA) level was not considered in categorizing patients as adjuvant or salvage.
PSA doubling time (PSADT) was calculated using all detectable PSA values and was determined by regressing the natural log of PSA against time from the first evaluable post-RP PSA. PSA failure was defined as the date of the first of 2 PSA values that exceeded 0.2 or a single value that exceeded 0.4 after postradiation PSA nadir. If PSA never decreased after RT, time to PSA failure was zero. Distant failure was defined as disease relapse outside of the pelvis. Times to event were measured from the first day of radiation. bPFS was defined as the time to PSA failure. For the distant metastasis-free survival (DMFS) analysis, patients were censored at the date of last follow-up or death. For the bPFS analysis, patients were censored at the date of last PSA.
Kaplan-Meier product limit survival was used for survival estimates.13 Cox proportional hazards analysis was used to identify baseline and treatment factors that were related to survival and toxicity outcomes.14 P-values of < .05 were considered statistically significant. Statistical analyses were done using SPSS Version 17.0 (SPSS Inc, Chicago, IL).
The decision to administer androgen deprivation therapy (ADT) and the duration of therapy was based on physician discretion. To identify which patients were more likely to receive ADT, logistic regression was used to evaluate factors associated with the use of ADT.
The following variables were considered in the models for bPFS and DMFS: age, race, pre-surgery PSA, biopsy Gleason score, pathologic Gleason score, pathologic T-stage, National Comprehensive Cancer Network risk category, margin status, whether PSA became undetectable after surgery, maximum preradiation PSA, PSADT, adjuvant versus salvage treatment, duration of ADT, radiation field (pelvis vs prostate fossa), radiation dose, and number of lymph nodes removed. The same variables were used in the model for late toxicity, in addition to pre-RT genitourinary (GU) function and time interval between RT and surgery. Logistic regression was used to evaluate the association between baseline GU/gastrointestinal (GI) function and acute GU/GI toxicity.
Multivariate analysis was performed using forward likelihood ratio (LR) with an entry criterion of 0.2 ng/mL and a removal criterion of 0.3 ng/mL. PSADT was not significant at this level; therefore, it was removed because of the large number of patients for whom PSADT was not calculable. Variables that were significant at the 0.2 and 0.3 ng/mL level were then entered using forward LR with an entry criterion of 0.05 ng/mL and a removal criterion of 0.1 ng/mL. The variables that were significant at this level were then entered alone using forward LR to confirm that they all remained in the model.
Results
The analysis included 313 consecutive patients with localized prostate cancer treated with either adjuvant (14%) or salvage (86%) HT from 2004 to 2013. Patient and treatment characteristics are detailed in Table 1. The median follow-up was 55 months (range, 6-131 months). The median pre-RT PSA was 0.3 ng/mL (range, <0.01-55.4 ng/mL). A large majority of patients (88%) had pre-RT PSA ≤1 ng/mL. Of the 38 patients with PSA >1 ng/mL, 37 had restaging bone scans that were negative for metastatic disease. One patient refused a bone scan because it had been done at the time of his diagnosis (7 months earlier).
Table 1.
Patient and treatment characteristics
| Characteristic | n = 313 |
| Age, median (range), y | 63 (39-81) |
| Biopsy Gleason score, n (%) | |
| ≤6 | 84 (27) |
| 7: 3 + 4 | 85 (27) |
| 7: 4 + 3 | 58 (19) |
| 8 | 54 (17) |
| 9-10 | 25 (8) |
| Unknown | 7 (2) |
| Presurgery PSA, n (%) | |
| <10 ng/mL | 212 (68) |
| 10-20 ng/mL | 63 (20) |
| >20 ng/mL | 32 (10) |
| Missing | 6 (2) |
| Type of surgery, n (%) | |
| Robotic | 278 (89) |
| Laparoscopic | 25 (8) |
| Open | 5 (2) |
| Unknown | 5 (2) |
| Pathologic Gleason score, n (%) | |
| 6 | 22 (7) |
| 7: 3 + 4 | 97 (31) |
| 7: 4 + 3 | 116 (37) |
| 8 | 24 (8) |
| 9 | 45 (14) |
| Unknown | 9 (3) |
| Pathologic tumor stage, n (%) | |
| T2 | 153 (49) |
| T2a | 9 (3) |
| T2b | 3 (1) |
| T2c | 141 (45) |
| T3 | 156 (50) |
| T3a | 80 (26) |
| T3b | 76 (24) |
| Unknown | 4 (1) |
| No. of lymph nodes removed, n (%) | |
| 0 | 95 (30) |
| 1-4 | 88 (28) |
| 5-10 | 91 (29) |
| >10 | 36 (12) |
| Unknown | 3 (1) |
| Margins, n (%) | |
| Positive | 165 (53) |
| Negative | 147 (47) |
| Unknown | 1 (<1) |
| Seminal vesicle invasion, n (%) | |
| Positive | 76 (24) |
| Negative | 236 (75) |
| Unknown | 1 (<1) |
| Extracapsular extension, n (%) | |
| Present | 136 (43) |
| Absent | 169 (54) |
| Unknown | 8 (3) |
| PSA undetectable, n (%) | |
| Yes | 206 (66) |
| No | 107 (34) |
| NCCN risk category, n (%) | |
| Low | 1 (<1) |
| Intermediate | 120 (38) |
| High | 182 (58) |
| Unknown | 10 (3) |
| Duration of ADT, n (%) | |
| None | 191 (61) |
| ≤6 mo | 53 (17) |
| >6-12 mo | 27 (9) |
| >12 mo | 41 (13) |
| Unknown | 1 (<1) |
| Radiation therapy field, n (%) | |
| Pelvis | 273 (87) |
| Prostate bed | 40 (13) |
| Intent, n (%) | |
| Adjuvant | 45 (14) |
| Salvage | 268 (86) |
| Radiation therapy dose, n (%) | |
| ≤66 Gy | 89 (28) |
| 66.4-69.8 Gy | 132 (42) |
| ≥70 Gy | 92 (29) |
| Pre–radiation therapy PSA, n (%) | |
| ≤0.2 | 100 (32) |
| 0.21-0.499 (32) | 99 (32) |
| 0.41-176 (24) | 76 (24) |
| 1.01-4 | 27 (9) |
| >4 | 11 (4) |
| PSA doubling time, n (%) | |
| Undetectable PSA | 38 (12) |
| ≤10 mo | 172 (55) |
| >10 mo | 74 (24) |
| Single PSA value | 29 (9) |
ADT, androgen deprivation therapy; NCCN, National Comprehensive Cancer Network; PSA, prostate-specific antigen.
Thirty-nine percent of patients received ADT for a median of 9 months (range, 3 months to continuous). On logistic regression, the following variables were associated with receiving ADT: pathologic stage T3a (odds ratio [OR] = 2.5;95% confidence interval [CI], 1.3-4.8; P = .007) or T3b disease (OR = 8.1; 95% CI, 3.8-17.3; P < .001), pre-RT PSA >1 ng/mL (OR = 2.9; 95% CI, 1.0-7.9; P = .045), and PSADT ≤10 months (OR = 3.0; 95% CI, 1.4-6.1; P = .003).
Biochemical progression-free survival
Five-year bPFS was 59% (95% CI, 53%-66%). On univariate analysis, the following variables were significantly associated with lower bPFS: higher pre-RT PSA, negative margins, PSADT ≤10 months, and never reaching undetectable PSA after surgery (Table 2). The final multivariate model included 305 patients who had complete data available (Table 2). Higher pre-RT PSA (>0.2 ng/mL), higher biopsy Gleason score (Gleason 7 [4 + 3]-9 vs Gleason 6), duration of ADT, and margin status were significantly associated (P < .05) with bPFS in the model. Positive margins were an independent predictor for higher bPFS (hazard ratio [HR] = 0.54; 95% CI, 0.36-0.82; P < .01). Pre-RT PSA of 0.2-1 (HR = 2.2; 95% CI, 1.4-3.7; P < .01) and >1 (HR = 9.7; 95% CI, 5.1-18.2; P < .001) significantly predicted for lower bPFS compared with PSA ≤0.2 ng/mL.
Table 2.
Multivariate analysis for bPFS and DMFS
| bPFS | Univariate | Multivariate | ||
|---|---|---|---|---|
| Variable | HR (95% CI) | P-value | HR (95% CI) | P-value |
| Pre-RT PSA | <.001 | <.001 | ||
| ≤0.2 | Refa | Refa | ||
| 0.2-1 | 2.3 (1.4-3.7) | <.01 | 2.2 (1.4-3.7) | <.01 |
| >1 | 5.1 (2.8-9.2) | <.001 | 9.7 (5.1-18.2) | <.001 |
| ADT duration | NS | <.05 | ||
| None | Refa | Refa | ||
| ≤6 mo | 0.90 (0.53-1.5) | NS | 0.75 (0.43-1.3) | NS |
| >6-12 mo | 0.80 (0.41-1.5) | NS | 0.39 (0.19-0.79) | <.01 |
| >12 mo | 0.60 (0.33-1.1) | NS | 0.49 (0.26-0.90) | <.05 |
| Margins | <.01 | <.01 | ||
| Negative | Refa | Refa | ||
| Positive | 0.57 (0.39-0.83) | <.01 | 0.54 (0.36-0.82) | <.01 |
| Biopsy Gleason | .05 | <.01 | ||
| ≤6 | Refa | Refa | ||
| 7 (3 + 4) | 1.0 (0.59-1.8) | NS | 1.5 (0.9-2.7) | NS |
| 7 (4 + 3) | 1.7 (0.96-2.9) | NS | 1.9 (1.1-3.4) | <.05 |
| 8 | 1.7 (0.94-2.9) | NS | 2.6 (1.5-4.7) | <.01 |
| 9-10 | 2.2 (1.1-4.3) | <.05 | 4.1 (2.0-8.3) | <.001 |
| PSA undetectable | <.01 | NS | NS | |
| Yes | Refa | |||
| No | 1.7 (1.2-2.5) | <.01 | ||
| PSA doubling time | <.001 | NS | NS | |
| >10 months | Refa | |||
| ≤10 months | 1.7 (1.04-2.8) | <.05 | ||
| Undetectable | 0.44 (0.2-1.0) | NS | ||
| DMFS | Univariate | Multivariate | ||
|---|---|---|---|---|
| Variable | HR (95% CI) | P-value | HR (95% CI) | P-value |
| Pre-RT PSA | <.001 | <.001 | ||
| ≤0.2 | Refa | Refa | ||
| 0.2-1 | 1.7 (0.7-4.1) | NS | 2.2 (0.90-5.4) | NS |
| >1 | 7.3 (2.9-18.6) | <.001 | 8.1 (3.2-20.9) | <.001 |
| Biopsy Gleason | <.001 | <.001 | ||
| ≤6 | Refa | Refa | ||
| 7 (3 + 4) | 1.2 (0.37-3.9) | NS | 1.6 (0.47-5.2) | NS |
| 7 (4 + 3) | 2.2 (0.7-7.0) | NS | 2.4 (0.76-7.6) | NS |
| 8 | 3.3 (1.1-10.0) | <.05 | 4.3 (1.4-13.1) | <.05 |
| 9-10 | 9.3 (3.1-27.4) | <.001 | 9.6 (3.2-29.1) | <.001 |
| PSA undetectable | <.05 | NS | NS | |
| Yes | Refa | |||
| No | 2.1 (1.1-4.0) | <.05 | ||
| Pathologic Gleason | <.01 | NS | NS | |
| ≤6 | Refa | |||
| 7 (3 + 4) | 0.9 (0.1-8.0) | NS | ||
| 7 (4 + 3) | 2.2 (0.3-17.1) | NS | ||
| 8 | 1.8 (0.2-19.4) | NS | ||
| 9 | 7.2 (0.9-55.3) | NS | ||
ADT, androgen deprivation therapy; bPFS, biochemical progression-free survival; CI, confidence interval; DMFS, distant metastasis-free survival; HR, hazard ratio; NS, not shown; PSA, prostate-specific antigen; RT PSA, radiation therapy prostate-specific antigen.
Ref is the reference category for the univariate/multivariate analysis.
The 5-year bPFS for pre-RT PSA ≤0.2, 0.21-1, and >1.0 ng/mL were 73% (95% CI, 63%-83%), 57% (95% CI, 48%-65%), and 37% (95% CI, 20%-54%), respectively (Fig 1, P < .001). When narrower pre-RT PSA ranges were used (≤0.2, 0.21-0.4, 0.41-1, and >1.0 ng/mL), there was no significant difference in the 5-year bPFS for pre-RT PSA 0.21-0.4 (55%; 95% CI, 42%-67%) compared with 0.41-1 ng/mL (59%; 95% CI, 46%-71%, P = .96). More patients in the pre-RT PSA 0.4-1 ng/mL group received ADT, but this was not significant (P = .18). Of the 38 patients with pre-RT PSA >1 ng/mL, 11 patients had very high PSA values of >4 (range, 4.1-55.4 ng/mL). Four of these 11 patients have not yet experienced PSA failure.
Figure 1.
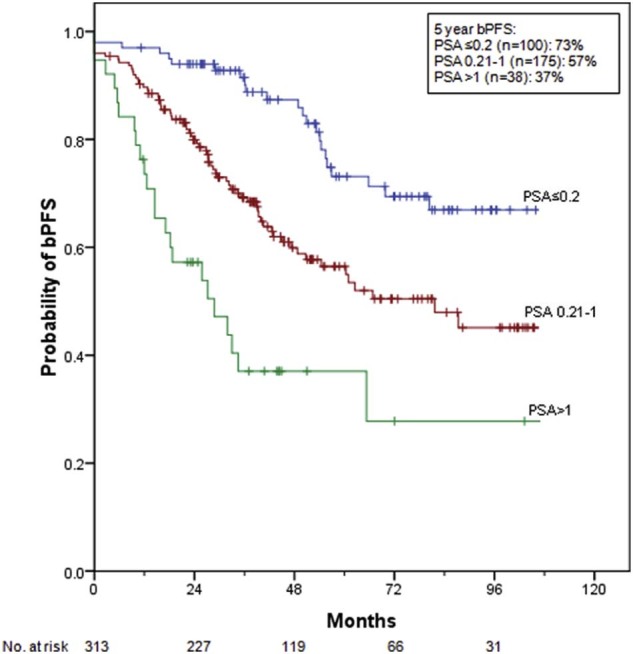
Probability of biochemical progression-free survival by pre–radiation therapy prostate-specific antigen level.
A separate multivariate analysis (n = 191) was conducted among patients who received radiation without ADT to address concerns regarding the impact that ADT may have had on other competing variables. The same variables remained significant in the model for bPFS: pre-RT PSA, biopsy Gleason score, and margin status.
Distant metastasis-free survival
Five-year DMFS was 89% (95% CI, 85%-93%). On univariate analysis, the following variables were significantly associated with DMFS: pre-RT PSA, biopsy Gleason score, pathologic Gleason score, and never reaching undetectable PSA after prostatectomy (Table 2). Pre-RT PSA and biopsy Gleason score were significant predictors of DMFS on multivariate analysis (Table 2). In the multivariate model for patients who received RT alone (n = 191), pre-RT PSA was the only factor to reach significance.
Acute genitourinary toxicity
Toxicity data are presented in Table 3. Grade 2 GU toxicity was observed in 25% of patients, but no acute grade 3 or higher GU toxicities were noted. Patients with grade 2 GU symptoms at baseline were more likely to develop another grade 2 GU toxicity during RT compared with patients with no symptoms at baseline (OR = 3.1; 95% CI, 1.6-5.9; P = .001).
Table 3.
Crude incidence of acute and late toxicity
| Grade | Genitourinary | Gastrointestinal | ||
|---|---|---|---|---|
| Acute (%) | Late (%) | Acute (%) | Late (%) | |
| 0 | 102 (33) | 105 (34) | 85 (27) | 221 (71) |
| 1 | 134 (43) | 61 (19) | 123 (39) | 36 (12) |
| 2 | 77 (25) | 116 (37) | 103 (33) | 49 (16) |
| 3 | 0 (0) | 31 (10) | 2 (<1) | 7 (2) |
There were no grade 4 or 5 acute or late toxicities.
Acute gastrointestinal toxicity
Grade 2 GI toxicity was observed in 33% of patients. There were 2 episodes of hemorrhoidal bleeding that required transfusion, which were designated as grade 3. Thefirst patient had a history of hemorrhoids with mild episodes of bleeding and the second had a history of hemorrhoids with moderate bleeding requiring hemorrhoidectomy. No acute grade 4 or higher GI toxicities were noted. Patients with grade 1 to 2 GI symptoms at baseline were more likely to develop grade 2 or 3 GI toxicity during RT compared with patients with no symptoms at baseline (OR = 4.0; 95% CI, 2.0-8.0; P < .001).
Late toxicity
Late toxicity data are also presented in Table 3. Actuarial late grade 3 GU and GI toxicities at 5 years were 10% (95% CI, 6.0%-14.2%) and 2% (95% CI, 0.5%-3.5%), respectively. The most common late grade 3 GU toxicities were incontinence requiring surgical intervention or Cunningham clamp (5%), stricture requiring surgical dilation (3%), and hematuria (3%). Of the 17 patients (5%) who developed grade 3 incontinence, 14 (82%) had at least grade 2 incontinence before RT. The most common surgery for incontinence was placement of an artificial urinary sphincter (3%). Of the 10 patients who developed a grade 3 stricture after radiation, 2 (20%) had a history of stricture before radiation. On multivariate analysis, only baseline urinary function was associated with higher likelihood of late grade 3 GU toxicity (baseline grade 2 GU toxicity vs grade 0 GU toxicity; HR = 4.8; 95% CI, 1.6-13.8; P = .004; Suppl Table 1; Suppl Fig S1). Patients who developed grade 3 GU toxicity had shorter mean intervals between surgery and RT compared with those who did not (25 vs 15 months), which approached significance (t test, P = .06). Interval from surgery to RT was not significant when included as a categorical variable (<12, 12-24, and >24 months) on multivariate analysis. Multivariate analysis for late grade 3 GI toxicity was not included because of the small number of events.
Discussion
To our knowledge, this is the largest outcomes report of patients with prostate cancer who received IG-IMRT after prostatectomy. Comparing outcomes between series is challenging because bPFS varies widely in the literature depending on how PSA failure is defined, whether adjuvant or salvage patients are included, and the radiation technique employed. In the 3 randomized trials of observation versus adjuvant RT (in which IMRT was not used) after surgery, the adjuvant arms had a 5-year bPFS of 60% to 74%.2, 3, 4 Stephenson et al reported a 6-year progression-free probability of 32% in a large series of patients who received salvage RT.5 In the current series, more than 80% of patients were treated with salvage intent, yielding a 5-year bPFS of 59%. A summary of published IMRT series is included in Table 4, with 5-year bPFS varying widely from 40% to 93%.
Table 4.
Prostate IMRT studies reporting bPFS
| Study | N | IMRT | Adjuvant/Salvage Therapy | FU | Median PSA | Fossa only | Median RT Dose | ADT | PSA Failure Definition | 5-year bPFS |
bPFS multivariate Analysis | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Significant | NS | |||||||||||
| Berlin et al. 20157 | 68 | 68 | Adjuvant (22%) + Salvage (78%) | 71 | 0.23 | 99% | 66 | 0% | Two consecutive rises above 0.2 ng/mL (failure dated at first PSA rise) | 73% | NR | NR |
| Goenka et al. 201115 | 285 | 176 | Salvage | 53 | 0.24 | 94% | NR, 94% ≥ 70 | 30% | >0.2 ng/mL followed by an increase, continued increase after RT or initiation of ADT after RT | 40% | LVSI Margins Pre-RT PSA GS ADT SVI |
ECE, LN + , PSA nadir, preoperative PSA, time from RP to PSA ≥0.2, interval to RT, RT dose, radiographic failure |
| Ost et al. 200916 | 104 | 104 | Adjuvant | 36 | NR, 86% < 0.2 | 100% | 74 | 65% | Increase >0.2 ng/mL above the lowest postoperative value | 93% | None | Preoperative PSA, postoperative PSA, PNI, GS, margins, SVI, ECE, ADT |
| Mishra et al. 201510 | 186 | 186 | Adjuvant (40%) + Salvage (60%) | 88 | 0.2 | 78% | 66.6 | 22% | >0.4 ng/mL with a subsequent confirmation, 3 documented increases ≥6 weeks apart or initiation of ADT | Adjuvant: 84% Salvage: 55% |
SVI | Adjuvant vs salvage, ECE, interval to RT, margins, GS, field, ADT, age, RT field |
| De Meerleer et al. 20088; Ost et al. 201117 | 136 | 136 | Salvage | 60 | 0.7 | 100% | 76 | 71% | Single PSA >0.2 ng/mL after post-RT nadir or continued rise | 56% | PNI pre-RT PSA ADT |
Preoperative PSA, PS ADT, GS, margins, SVI, ECE, LN + , RT dose |
| Current Study | 313 | 313 | Adjuvant (14%) + Salvage (86%) | 55 | 0.3 | 13% | 67 | 39% | Two values >0.2 or one value >0.4 ng/mL after post-RT PSA nadir | 59% | Pre-RT PSA ADT duration Margins Bx GS |
Age, race, path GS, NCCN risk, pT stage, undetectable PSA, preoperative PSA, PS ADT, adjuvant/salvage, RT field, RT dose, LN removed |
ADT, androgen deprivation therapy; bPFS, biochemical progression-free survival; Bx, biopsy; ECE, extracapsular extension; FU, follow up; GS, Gleason score; IMRT, intensity modulated radiation therapy; LN, lymph nodes; LVSI, lymphovascular space invasion; NCCN, National Comprehensive Cancer Network; NR, not reported; NS, not shown; PNI, perineural invasion; PS, prostate-specific; PSA; prostate-specific antigen; RP, radical prostatectomy; RT, radiation therapy; SVI, seminal vesicle invasion.
Our analysis found positive margins to be significantly associated with improved bPFS. In the ARO 96-024 and EORTC 229113 trials, patients with positive margins were found to experience a greater benefit from RT. Patients with positive surgical margins may have a higher probability of disease recurrence in the prostate bed; therefore, they would be expected to derive a higher benefit from RT compared with patients who have negative margins. Conclusions with regard to the interaction of margin status with treatment effect in the current series are limited because all patients received RT.
Biopsy Gleason score was significantly related to bPFS on multivariate analysis, but surgery Gleason score was not. Many patients with a biopsy Gleason score of 6 (n = 58) or 8 (n = 25) were reclassified as Gleason 7 at the time of surgery. As a result, there was much less variability in surgery Gleason score (70% with score 7), which may have hindered this variable from being significant on multivariate analysis. Although it is intuitive that the surgery Gleason score represents the “true” Gleason score, other series have reported the prognostic value of biopsy Gleason score even when the surgery Gleason score is also considered.18, 19, 20 The finding that biopsy Gleason score was significant but pathologic Gleason score was not is an unanticipated finding that may be related to the retrospective nature of the analysis.
Pre-RT PSA was also found to be a significant independent predictor of bPFS and DMFS. Our results are consistent with several reports that indicate that higher bPFS is more likely in patients treated with PSA ≤0.2.21, 22 Abugharib et al21 recently reported on a large cohort of patients who received salvage RT and found that presalvage RT PSA strongly predicted for biochemical relapse-free survival, freedom from salvage ADT, DMFS, and prostate cancer–specific survival. King et al22 reported a decreasing likelihood of bPSF with every 0.1 incremental increase in pre-RT PSA over 0.2 ng/mL. We found high rates of control with pre-RT PSA ≤0.2, which worsened with increasing PSA. However, it must be considered that a proportion of patients with pre-RT PSA ≤0.2 ng/mL may have continued to have biochemical control without RT, particularly those with undetectable PSA. Patients who received RT with pre-RT PSA >1 ng/mL fared poorly, with a 37% probability of biochemical control at 5 years. However, in contrast to King et al22 and Abugharib et al,21 we were unable to observe a deleterious effect of rising PSA in the range of 0.2 to 1 ng/mL. Patients with PSA between 0.41 and 1 ng/mL (n = 76) had bPFS similar to that of patients with PSA of 0.21 to 0.4 ng/mL (n = 99). King et al. also described a dose response with a 2% interval in improvement in 5-year biochemical recurrence-free survival per Gray.22 In our model, radiation dose was not a statistically significant predictor of bPFS (P > .05).
ADT was associated in our series with a decreased risk of biochemical recurrence, despite being used in higher risk patients. This is in agreement with the recently published results of RTOG 9601, which showed significantly improved bPFS and overall survival in patients who received bicalutamide during and after salvage RT compared with RT alone.23 In RTOG 96-01, no benefit to bicalutamide was seen in men with detectable pre-RT PSA <0.7. We examined this subgroup of patients in the current series (n = 165) and also did not observe a significant benefit of ADT on bPFS or DMFS. RTOG 96-01 also found an increased benefit of ADT in patients with positive surgical margins, which was not observed on our analysis. The Groupe d'Étude des Tumeurs Urogénitales and Association Française d'Urologie 16 trial also showed significantly improved progression free survival at 5 years with the addition of short-term goserelin to salvage RT.24 The benefit of ADT has been reported in other IMRT series as well.15, 16, 17
The positive effect of ADT in a retrospective report should be interpreted with caution because ADT, which routinely results in immediate PSA suppression, can cause skewing of the bPFS endpoint, with the effect being more pronounced with longer ADT duration. There was no significant difference between the median follow-up for patients who received ADT and for those who did not (P = .33). With a median follow-up of 55 months and a median ADT length of 9 months, the spurious effect of ADT delaying biochemical failure is likely minimal in our series.
PSADT was not significantly associated with increased risk of PSA failure on multivariate analysis. Stephenson et al analyzed 1540 patients who received salvage RT and found PSADT ≤10 months to be associated with an increased risk of PSA progression.5 PSADT ≥6 months was reported by Trock et al25 to be associated with improved 10-year prostate cancer–specific survival. Other IMRT series (Table 4) have not examined PSADT or have not found it to be significantly associated with bPFS.
The timing of radiation (adjuvant vs salvage) did not significantly affect bPFS or DMFS in our series, which may be due to a small proportion of patients who received adjuvant therapy (14%). This is in agreement with recent data from Abugharib et al,21 who showed that pre-RT PSA level predicted for bPFS and DMFS, but time from surgery to radiation did not. Their data and our findings support the notion that that the time interval between prostatectomy and radiation may not have as meaningful a prognostic value; therefore, traditional definitions of “adjuvant” or “salvage” on the basis of timing alone need to be revisited.
The benefit of pelvic nodal irradiation is controversial, and we found no improvement in bPFS or DMFS in the current series. The vast majority of patients received pelvic RT (87%), which was the practice at our institution because it was the treatment used in the adjuvant versus salvage trials.2, 3, 4 However, pelvic nodal irradiation is used less commonly in more modern trials. Pelvic nodal irradiation was not allowed in RTOG 96-0123 and was administered to only 16% of patients in GETUG-AFU-16.24
The rates of grade 3 GU toxicity in the current study are higher than those in other published IMRT series (Table 5). Incontinence comprised the majority (55%) of grade 3 GU toxicity. Prostatectomy series without radiation report 6% to 7% of patients as requiring surgical intervention for urinary incontinence.27, 28, 29 In general, toxicity is expected to be higher in patients treated with both surgery and radiation compared with either modality alone.28 It can also be difficult to discern whether toxicity is solely attributable to RT or represents a worsening of a previously existing postoperative toxicity. This is further complicated by the relatively short surgery-to-RT intervals (mean, 15 months) in patients who developed grade 3 GU toxicity compared with those who did not (mean, 25 months). Other IMRT series reporting a multivariate analysis for GU toxicity did not explore the effect of surgery-to-RT interval on toxicity.9, 15
Table 5.
Prostate IMRT studies reporting late toxicity
| Study | N | IMRT | FU | 5-year bPFS |
Toxicity Scoring | GI Toxicity | GU toxicity | Incontinence | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Acute | Late | Acute | Late | |||||||
| Berlin et al. 20157 | 68 | 68 | 71 | 73% | CTCAE v3.0 | G2: 16% G3: 0% |
G2: 12% G3: 0% |
G2: 19% G3: 0% |
G2: 13% G3: 0% |
G3: 0% |
| Goenka et al. 201115 | 285 | 176 | 53 | 40% | RTOG+CTCAE v 3.0 | G2: 8% G3: 0 |
≥G2: 2% G3: 1.4%a |
≥G2: 13% G3: NR |
≥G2: 17% G3: NR |
G3: 6%a |
| Nath et al. 20109 | 50 | 50 | 24 | NR | CTCAE v3.0 | G2: 8% G3: 0% |
G2: 2% G3: 0% |
G2: 14% G3: 0% |
G2: 16% G3: 2% |
NR |
| Ost et al. 200916 | 104 | 104 | 36 | 93% | In-house toxicity score | G2: 22% G3: 0% |
G2: 7% G3: 0% |
G2: 26% G3: 8% |
G2: 22% G3: 4% Stricture: 6% |
G3: 2% |
| De Meerleer et al. 2008,8 Ost et al. 201117 | 136 | 136 | 60 | 56% | In-house toxicity score | G2: 15% G3: 0% |
G2: 13% G3: < 1% |
G2: 28% G3: 3% |
G2: 31% G3: 3% Stricture: 6% |
G3: 1% |
| Fonteyne et al. 2015 (combined 16 and 17)26 | 232 | 232 | 60 | NR | In-house toxicity score | NR | G2: 11% G3: 1% |
NR | G2: 26% G3: 6% |
G2: 2% |
| Current Study | 313 | 313 | 55 | 59% | CTCAE v4.0 | G2: 33% G3: < 1% |
G2: 16% G3: 2% |
G2: 25% G3: 0% |
G2: 37% G3: 10% |
G3: 5% |
bPFS, biochemical progression-free survival; CTCAE, Common Terminology Criteria for Adverse Events; FU, follow up; G, grade; GI, gastrointestinal; GU, genitourinary; IMRT, intensity modulated radiation therapy; NR, not reported; RTOG, Radiation Technology Oncology Group.
Rates include both patients were treated with IMRT and those who were not.
Rates of grade 3 GU toxicity overall were similar or improved compared with large 3-dimensional conformal RT series with a longer follow-up, ranging from 7% to 12%.24, 30, 31 Goenka et al15 reported very similar rates (5%) of grade 3 urinary incontinence in a large series of patients treated with IMRT and 3-dimensional conformal RT. They found no improvement in incontinence in patients treated with IMRT. It is possible that IMRT does not significantly alter urinary incontinence rates because contouring guidelines for IMRT include the urethral anastomosis as well as a significant portion of the bladder.32 Similarly, this area would be included independent of whether pelvic lymph nodes were treated. This may explain our finding that field size (pelvic RT vs prostate fossa) was not significantly associated with grade 3 GU toxicity on multivariate analysis.
Baseline urinary function in our population was found to be highly predictive of the likelihood of grade 3 toxicity. Twenty percent of patients who had grade 2 GU toxicity at baseline developed grade 3 late GU toxicity, compared with 4% of patients with grade 0 GU toxicity at baseline (P < .001). Goenka et al15 reported a similar increased risk of grade 3 incontinence in patients with baseline grade 2 incontinence compared with baseline grade 0 to 1 incontinence (13.9% vs 1%-4%, P = .04).
The increased GU toxicity in our series raises the question of whether dosimetric differences in HT increase toxicity. Multiple papers comparing the dosimetric qualities of HT with other types of IMRT for the treatment of prostate cancer have reached different conclusions but have not shown HT to be inferior in terms of normal-tissue sparing.33, 34, 35, 36 Because all patients who were treated for prostate cancer at our institution during this timeframe were treated with HT, there is no internal comparison group. A detailed dosimetric analysis may help to identify factors associated with toxicity, but such analysis is beyond the scope of the current paper.
Incidence of late grade 3 GI toxicity was low, which is in agreement with similar IMRT studies (Table 5). Fonteyne et al26 reported a 1% late grade 3 GI toxicity with median of 5 years of follow-up for 232 patients who were treated with IMRT postoperatively, which is comparable with our finding of 2%. However, many IMRT series with lower patient numbers report 0% late grade 3 GI toxicity. Our study includes a higher proportion of patients who were treated with pelvic RT (87%) compared with other IMRT series (Table 4), which report lower rates of toxicity. It is possible that pelvic irradiation increased the dose to normal tissue and resulted in increased toxicity, but this was not found to be significant on univariate or multivariate analysis.
Although we did not find an association between radiation dose or field with late toxicity, a more detailed dosimetric analysis is currently in process. Parameters such as consistency of bladder filling, planning bladder volume, or urethral dose may be associated with grade 3 toxicity.
This study is subject to limitations that are known to be associated with a retrospective analysis. Other limitations of this study are as follows. A specific follow-up protocol was not used as rigorously as in a clinical trial. Some patients were followed up by outside providers or were lost to follow-up. A specific hypothesis was not tested. Stage and Gleason score migration may have occurred over the course of the study. Our inclusion of patients receiving ADT may limit the interpretation of our bPFS outcome. Patients who had a higher pathologic stage, pre-RT PSA >1, and shorter PSADT were more likely to receive ADT. Although risk group was not significant on multivariate analysis, ADT still may be a confounding factor in our analysis. In the ADT group, the majority of patients received short-term ADT; therefore, the impact on the overall results of this analysis is expected to be minimal. Finally, toxicity data were physician reported. Patient-reported toxicity and quality of life data were not available.
Strengths of this analysis include higher patient numbers than previously published series, patients treated uniformly with HT, and follow-up length comparable to smaller series. Close follow-up allowed for detailed reporting of grade 3 toxicity data.
Conclusion
We report the largest series to our knowledge of patients who were treated with postprostatectomy IMRT. A longer follow-up and prospective studies are needed to further identify factors that are associated with outcomes and toxicity.
Footnotes
Sources of support: None to disclose.
Conflicts of interest: None to disclose.
Supplementary material related to this article can be found online at doi:10.1016/j.adro.2017.08.004.
Supplementary data
The following is the supplementary data to this article:
Actuarial Grade 3 GU Toxicity by pre-RT GU symptoms.
Multivariate analysis for Late Grade 3 GU Toxicity.
References
- 1.Stephenson A.J., Scardino P.T., Eastham J.A. Postoperative nomogram predicting the 10-year probability of prostate cancer recurrence after radical prostatectomy. J Clin Oncol. 2005;23:7005–7012. doi: 10.1200/JCO.2005.01.867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Thompson I.M., Tangen C.M., Paradelo J. Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: Long-term followup of a randomized clinical trial. J Urol. 2009;181:956–962. doi: 10.1016/j.juro.2008.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bolla M., van Poppel H., Tombal B. Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: Long-term results of a randomised controlled trial (EORTC trial 22911) Lancet. 2012;380:2018–2027. doi: 10.1016/S0140-6736(12)61253-7. [DOI] [PubMed] [Google Scholar]
- 4.Wiegel T., Bartkowiak D., Bottke D. Adjuvant radiotherapy versus wait-and-see after radical prostatectomy: 10-year follow-up of the ARO 96-02/AUO AP 09/95 trial. Eur Urol. 2014;66:243–250. doi: 10.1016/j.eururo.2014.03.011. [DOI] [PubMed] [Google Scholar]
- 5.Stephenson A.J., Scardino P.T., Kattan M.W. Predicting the outcome of salvage radiation therapy for recurrent prostate cancer after radical prostatectomy. J Clin Oncol. 2007;25:2035–2041. doi: 10.1200/JCO.2006.08.9607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goldin G.H., Sheets N.C., Meyer A.M. Comparative effectiveness of intensity-modulated radiotherapy and conventional conformal radiotherapy in the treatment of prostate cancer after radical prostatectomy. JAMA Intern Med. 2013;173:1136–1143. doi: 10.1001/jamainternmed.2013.1020. [DOI] [PubMed] [Google Scholar]
- 7.Berlin A., Cho E., Kong V. Phase 2 trial of guideline-based postoperative image guided intensity modulated radiation therapy for prostate cancer: Toxicity, biochemical, and patient-reported health-related quality-of-life outcomes. Pract Radiat Oncol. 2015;5:e473–e482. doi: 10.1016/j.prro.2015.02.015. [DOI] [PubMed] [Google Scholar]
- 8.De Meerleer G., Fonteyne V., Meersschout S. Salvage intensity-modulated radiotherapy for rising PSA after radical prostatectomy. Radiother Oncol. 2008;89:205–213. doi: 10.1016/j.radonc.2008.07.027. [DOI] [PubMed] [Google Scholar]
- 9.Nath S.K., Sandhu A.P., Rose B.S. Toxicity analysis of postoperative image-guided intensity-modulated radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2010;78:435–441. doi: 10.1016/j.ijrobp.2009.08.023. [DOI] [PubMed] [Google Scholar]
- 10.Mishra M.V., Scher E.D., Andrel J. Adjuvant versus salvage radiation therapy for prostate cancer patients with adverse pathologic features: Comparative analysis of long-term outcomes. Am J Clin Oncol. 2015;38:55–60. doi: 10.1097/COC.0b013e318287bb6b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cheng J.C., Schultheiss T.E., Nguyen K.H., Wong J.Y. Acute toxicity in definitive versus postprostatectomy image-guided radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2008;71:351–357. doi: 10.1016/j.ijrobp.2007.09.043. [DOI] [PubMed] [Google Scholar]
- 12.National Institute of Health, Department of Health and Human Services . 2009. National Cancer Institute Common Terminology Criteria for Adverse Events v4.0. NIH publication # 09-7473. May 29. [Google Scholar]
- 13.Kaplan E.L., Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:471–481. [Google Scholar]
- 14.Cox D.R. Regression model and life tables. J R Stat Soc Ser. 1972;B34:187–220. [Google Scholar]
- 15.Goenka A., Magsanoc J.M., Pei X. Improved toxicity profile following high-dose postprostatectomy salvage radiation therapy with intensity-modulated radiation therapy. Eur Urol. 2011;60:1142–1148. doi: 10.1016/j.eururo.2011.08.006. [DOI] [PubMed] [Google Scholar]
- 16.Ost P., Fonteyne V., Villeirs G. Adjuvant high-dose intensity-modulated radiotherapy after radical prostatectomy for prostate cancer: Clinical results in 104 patients. Eur Urol. 2009;56:669–675. doi: 10.1016/j.eururo.2009.05.041. [DOI] [PubMed] [Google Scholar]
- 17.Ost P., Lumen N., Goessaert A.S. High-dose salvage intensity-modulated radiotherapy with or without androgen deprivation after radical prostatectomy for rising or persisting prostate-specific antigen: 5-year results. Eur Urol. 2011;60:842–849. doi: 10.1016/j.eururo.2011.04.021. [DOI] [PubMed] [Google Scholar]
- 18.Fitzsimons N.J., Presti J.C., Jr, Kane C.J. Is biopsy Gleason score independently associated with biochemical progression following radical prostatectomy after adjusting for pathological Gleason score? J Urol. 2006;176:2453–2458. doi: 10.1016/j.juro.2006.08.014. [DOI] [PubMed] [Google Scholar]
- 19.Müntener M., Epstein J.I., Hernandez D.J. Prognostic significance of Gleason score discrepancies between needle biopsy and radical prostatectomy. Eur Urol. 2008;53:767–775. doi: 10.1016/j.eururo.2007.11.016. [DOI] [PubMed] [Google Scholar]
- 20.D'Amico A.V., Whittington R., Malkowicz S.B. A multivariate analysis of clinical and pathological factors that predict for prostate specific antigen failure after radical prostatectomy for prostate cancer. J Urol. 1995;154:131–138. [PubMed] [Google Scholar]
- 21.Abugharib A., Jackson W.C., Tumati V. Very early salvage radiotherapy improves distant metastasis-free survival. J Urol. 2017;197:662–668. doi: 10.1016/j.juro.2016.08.106. [DOI] [PubMed] [Google Scholar]
- 22.King C.R. The timing of salvage radiotherapy after radical prostatectomy: A systematic review. Int J Radiat Oncol Biol Phys. 2012;84:104–111. doi: 10.1016/j.ijrobp.2011.10.069. [DOI] [PubMed] [Google Scholar]
- 23.Shipley W.U., Seiferheld W., Lukka H.R. Radiation with or without antiandrogen therapy in recurrent prostate cancer. N Engl J Med. 2017;376:417–428. doi: 10.1056/NEJMoa1607529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carrie C., Hasbini A., de Laroche G. Salvage radiotherapy with or without short-term hormone therapy for rising prostate-specific antigen concentration after radical prostatectomy (GETUG-AFU 16): A randomised, multicentre, open-label phase 3 trial. Lancet Oncol. 2016;17:747–756. doi: 10.1016/S1470-2045(16)00111-X. [DOI] [PubMed] [Google Scholar]
- 25.Trock B.J., Han M., Freedland S.J. Prostate cancer-specific survival following salvage radiotherapy vs observation in men with biochemical recurrence after radical prostatectomy. JAMA. 2008;299:2760–2769. doi: 10.1001/jama.299.23.2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fonteyne V., Sadeghi S., Ost P. Impact of changing rectal dose volume parameters over time on late rectal and urinary toxicity after high-dose intensity-modulated radiotherapy for prostate cancer: A 10-years single centre experience. Acta Oncol. 2015;54:854–861. doi: 10.3109/0284186X.2014.974826. [DOI] [PubMed] [Google Scholar]
- 27.Kim P.H., Pinheiro L.C., Atoria C.L. Trends in the use of incontinence procedures after radical prostatectomy: A population based analysis. J Urol. 2013;189:602–608. doi: 10.1016/j.juro.2012.08.246. [DOI] [PubMed] [Google Scholar]
- 28.Jarosek S.L., Virnig B.A., Chu H. Propensity-weighted long-term risk of urinary adverse events after prostate cancer surgery, radiation, or both. Eur Urol. 2015;67:273–280. doi: 10.1016/j.eururo.2014.08.061. [DOI] [PubMed] [Google Scholar]
- 29.Bianco F.J., Jr, Riedel E.R., Begg C.B. Variations among high volume surgeons in the rate of complications after radical prostatectomy: Further evidence that technique matters. J Urol. 2005;173:2099–2103. doi: 10.1097/01.ju.0000158163.21079.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cortés-González J.R., Castellanos E., Sandberg K. Early salvage radiation therapy combined with short-term hormonal therapy in recurrent prostate cancer after radical prostatectomy: Single-institution 4-year data on outcome, toxicity, health-related quality of life and co-morbidities from 184 consecutive patients treated with 70 Gy. Int J Oncol. 2013;42:109–117. doi: 10.3892/ijo.2012.1694. [DOI] [PubMed] [Google Scholar]
- 31.Cozzarini C., Fiorino C., Da Pozzo L.F. Clinical factors predicting late severe urinary toxicity after postoperative radiotherapy for prostate carcinoma: A single-institute analysis of 742 patients. Int J Radiat Oncol Biol Phys. 2012;82:191–199. doi: 10.1016/j.ijrobp.2010.09.027. [DOI] [PubMed] [Google Scholar]
- 32.Michalski J.M., Lawton C., El Naqa I. Development of RTOG consensus guidelines for the definition of the clinical target volume for postoperative conformal radiation therapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2010;76:361–368. doi: 10.1016/j.ijrobp.2009.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pasquier D., Cavillon F., Lacornerie T. A dosimetric comparison of tomotherapy and volumetric modulated arc therapy in the treatment of high-risk prostate cancer with pelvic nodal radiation therapy. Int J Radiat Oncol Biol Phys. 2013;85:549–554. doi: 10.1016/j.ijrobp.2012.03.046. [DOI] [PubMed] [Google Scholar]
- 34.Wolff D., Stieler F., Welzel G. Volumetric modulated arc therapy (VMAT) vs. serial tomotherapy, step-and-shoot IMRT and 3D-conformal RT for treatment of prostate cancer. Radiother Oncol. 2009;93:226–233. doi: 10.1016/j.radonc.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 35.Tsai C.L., Wu J.K., Chao H.L. Treatment and dosimetric advantages between VMAT, IMRT, and helical tomotherapy in prostate cancer. Med Dosim. 2011;36:264–271. doi: 10.1016/j.meddos.2010.05.001. [DOI] [PubMed] [Google Scholar]
- 36.Kinhikar R.A., Pawar A.B., Mahantshetty U. Rapid Arc, helical tomotherapy, sliding window intensity modulated radiotherapy and three-dimensional conformal radiation for localized prostate cancer: A dosimetric comparison. J Cancer Res Ther. 2014;10:575–582. doi: 10.4103/0973-1482.138200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Actuarial Grade 3 GU Toxicity by pre-RT GU symptoms.
Multivariate analysis for Late Grade 3 GU Toxicity.


