Abstract
Henoch–Schönlein purpura is a systemic vasculitis characterized by IgA deposits, which target the skin, joints, and kidneys, among other organs. In children, prognosis is often good but little is known about biomarkers of pediatric nephritis. We hypothesized that biological markers, including cytokines, immunoglobulins, IgA-immune complexes, IgA glycosylation and neutrophil gelatinase-associated lipocalin (NGAL), may discriminate IgA vasculitis (IgAV) pediatric patients with renal involvement from those without renal involvement. Fifty children at the time of IgAV rash between 2010 and 2015 were prospectively enrolled and compared to 21 controls. All patients were assessed for clinical and biological parameters at the time of diagnosis, including the levels of cytokines, immunoglobulins, immune complexes, IgA glycosylation and NGAL in serum and urine. Among IgAV patients, 33 patients exhibited nephritis (IgAV-N) and 17 children were without nephritis (IgAV-woN). The serum level of galactose-deficient (Gd)-IgA1 (p<0.01) and the urinary concentrations of IgA, IgG, IgM, IL-6, IL-8, IL-10, IgA-IgG complexes and IgA-sCD89 complexes (p<0.001 for all) were higher in the IgAV-N patients than in the IgAV-woN patients. Among those markers, urinary IgA and IgM had the highest AUC (0.86 and 0.87 respectively, p<0.0001). This prospective cohort study furthers our understanding of the pathophysiology of IgAV. We identified biomarkers that are able to distinguish patients initially with or without nephritis. To conclude, serum Gd-IgA1 and urinary IgA, IgG, IgM, IL-6, IL-8, IL-10, and IgA-IgG and IgA-sCD89 complexes could identify IgAV pediatric patients with renal involvement at the time of diagnosis.
Introduction
In children, IgA vasculitis (IgAV) or Henoch-Schönlein purpura is a common systemic vasculitis affecting the small vessels of the skin and other organs, particularly the kidneys, joints and intestines [1]. Most of the cases are self-limiting but nephritis occurs in 20 to 54% of children leading to complications [2–5]. The natural history of nephritis ranges from persistent asymptomatic microscopic hematuria to progressive kidney failure, with an estimated long-term risk of end-stage renal failure of up to 15% in children [3]. Non-invasive biomarkers of nephritis are specifically required for children to avoid kidney biopsies.
IgAV is considered an inflammatory disease involving IgA immune complexes. The pathological mechanisms of organ-specific IgA deposition remain elusive. IgAV nephritis seems to share pathological mechanisms with IgA nephropathy. Correlations between immune parameters and organ injury have been explored suggesting a Th1/Th2 imbalance in Chinese children [6]. Moreover, the galactose deficiency of circulating IgA1 is associated to with nephritis as described in IgA nephropathy [7,8].
However, studies on IgAV pediatric cohorts are rare, particularly those exploring inflammatory mechanisms. Therefore, we assessed the levels of cytokines, immunoglobulins, IgA-immune complexes, IgA glycosylation and neutrophil gelatinase-associated lipocalin (NGAL) in blood and urine in a well characterized population of 50 children with IgAV to discriminate pediatric patients with renal involvement from those without renal involvement.
Materials and methods
Pediatric population
This cohort of children is part of a French national, multicenter, prospective study (HSPrognosis study) which was conducted between April 2010 and May 2015. Adult patients with IgAV (n = 85) were previously described [9]. The local ethical committee of Assistance Publique–Hôpitaux de Paris (AP-PH, approval number 10.649bis) approved this study, and written informed consent was obtained from the parents of all the included children before any study-related procedure was performed. Patients were enrolled at the time of skin rash appearance in the pediatric and emergency departments of the hospitals (most patients were recruited in Paris’ region except 2 from the South of France). Biological samples were concomitantly collected at the skin rash. Pediatric controls (healthy controls who underwent a systemic familial investigation and pediatric cases of a single kidney) were enrolled in a single center (R. Debré’s Hospital, Paris, France), and controls with hematuria or proteinuria >0.5 g/g were excluded. The inclusion criterion was a clinical diagnosis of IgAV (EULAR/PRINTO/PRES criteria [10]). The exclusion criteria were the following: (i) absence of skin lesions; (ii) treatment with immunosuppressive drugs or steroids during the 2 weeks prior to skin rash appearance; (iii) thrombocytopenia (platelet number <100,000/mm3); and (iv) absence of consent.
Renal involvement was defined as the presence of hematuria (determined by dipstick or a red cell count >5/mm3) and/or a proteinuria/creatininuria ratio ≥0.5 g/g and/or an eGFR <60 mL/min in children (determined using the Schwartz formula) [11].
A renal biopsy was performed if indicated by clinical examination and biological tests, because of hematuria and a ratio of proteinuria/creatininuria (PCR) greater than 0.5 g/g and/or renal failure. All renal biopsies were independently examined by two pathologists in blinded conditions. On immunofluorescence, the predominance of mesangial IgA among glomerular immune deposits was examined. At least ten glomeruli had to be present on the biopsy. We semi-quantitatively evaluated: (1) endocapillary lesions and their focal or diffuse distribution; (2) extracapillary proliferation, graded according to the number of glomeruli involved; and (3) interstitial fibrosis (area percentage). The proportions of glomeruli involved by crescents, fibrinoid necrosis, and global sclerosis were also recorded. All biopsies were then classified according to the ISKDC classification standard: Type I, minimal glomerular abnormalities; Type II, mesangial proliferation without crescents; Type III, focal segmental (IIIa) or diffuse (IIIb) mesangial proliferation with<50% crescents; Type IV, mesangial proliferation with 50–70% crescents; Type V, mesangial proliferation with>75% crescents; and Type VI, membrano-proliferative-like lesions.
Immunoglobulin, cytokine and NGAL measurements
The concentrations of IgA, IgM and IgG were determined in sera by nephelometry at the Immunology Department of Bichat Hospital. The levels of IgD and IgE were detected in sera, and all other immunoglobulin concentrations in urine samples were assessed by ELISA using Bethyl kits (Bethyl Laboratories, Montgomery, Texas, USA). Cytokine Binding Assay kits (BD Biosciences, Le Pont de Claix, France) were used to determine the concentrations of cytokines, including interleukin (IL)-1β, IL-6, IL-8, IL-10, IL-12p70 and tumor necrosis factor (TNF-α) in plasma and urine, and bead fluorescence was analyzed on a FACSCanto II cytometer (BD Biosciences, Le Pont de Claix, France). The NGAL concentration was determined using ELISA kits (Bioporto, Hellrup, Denmark).
ELISA for IgA1 glycosylation
IgA1 glycosylation was assessed by a lectin ELISA using Helix aspersa agglutinin (HAA). In brief, plates were coated with anti-IgA antibody (1/500 in PBS, Bethyl Laboratories Montgomery, Texas, USA) overnight at 4°C, and the blocking solution (PBS, bovine serum albumin 2%) was added and allowed to stand for 8 h at 4°C. Serum samples were diluted to 10 μg/mL for IgA and incubated overnight at 4°C. To remove sialic acids, neuraminidase was added and incubated for 3 h at 37°C, followed by the addition of biotinylated Helix aspersa agglutinin (HAA, Sigma-Aldrich, Saint-Louis, Missouri, USA) and an additional 3-h incubation at 37°C. The detection of HAA binding was performed using streptavidin coupled with alkaline phosphatase (1/10000, Jackson Immuno Research Laboratories, West Grove, PA, USA) and substrate (SIGMAFAST p-nitrophenyl phosphate tablets; Sigma-Aldrich, Saint-Louis, Missouri, USA). The plates were read at 405 nm using an Infinite M200 microplate reader (Tecan, Manndorf, Switzerland). The positive control (i.e., purified deglycosylated IgA1) corresponded to 100% binding.
IgA complex detection
For the IgA complex measurements, ELISA was performed as follows: coating with anti-IgA antibody (1/500 in PBS, Bethyl Laboratories, Montgomery, Texas, USA) and with A3 anti-CD89 (5 μg/mL, produced at laboratory) for IgA/IgG and IgA-sCD89, respectively, overnight at 4°C, followed by blocking for 8 h at 4°C. Polyethylene glycol-precipitated serum or urine samples were incubated overnight at 1/10. Revelation was performed using anti-IgG (1/5000, Southern Biotech, Birmingham, Alabama, USA) or anti-IgA (1/2000, BD Biosciences Le Pont de Claix, France) coupled with alkaline phosphatase. The positive control (i.e., a control serum with high levels of complexes) corresponded to 100% binding, and all samples were compared to this positive control.
CD89 expression on blood myeloid cells
Blood samples (50 μL) were labeled with antibodies (BD Biosciences) as follows: anti-CD11b coupled with Pacific Blue, anti-CD15 coupled with allophycocyanin (APC), anti-CD14 coupled with APC-H7, and anti-CD89 coupled with phycoerythrin (PE) or isotype-PE for 20 min at room temperature. Then, red blood cells were lysed using BD lysing solution (BD Biosciences, Le Pont de Claix, France) for 10 min and then washed twice with a PBS solution of 2% bovine serum albumin and 0.01% azide. The samples were analyzed using a FACSCanto II system (BD Biosciences, Le Pont de Claix, France) and FlowJo software.
Statistical analysis
Median (interquartile range IQR) values and frequencies are provided for the description of continuous and categorical variables, respectively. The Shapiro-Wilk test was used to test the normality of the data distribution. The mean values and proportions were compared using Mann-Whitney and Fisher’s exact tests, respectively. Aberrant values for biological variables were determined by a Dixon test and excluded. Spearman's rank correlation coefficient was used to assess the correlation between two continuous variables. For each biomarker, we constructed an ROC curve and calculated the Area Under the Curve (AUC) to evaluate its predictive accuracy to discriminate renal involvement and outcome. A non-parametric method was used to calculate the confidence interval (CI). All tests were two-sided, and values of p<0.05 were considered statistically significant. All statistical analyses were performed with GraphPad Prism version 7 (GraphPad Software).
Results
Characteristics of the pediatric cohort
Fifty children with IgAV and 21 pediatric controls (sex and age matched) were prospectively recruited to compose the study population. The demographic and baseline disease characteristics are presented in Table 1. The median age was 7 (6–9) years for children. Male gender represented 66.0% of the overall study population. Seventeen (34.0%) patients did not exhibit renal involvement (IgAV-woN group), and 33 (66.0%) exhibited nephritis (IgAV-N group) as defined by clinical parameters (hematuria and-/or proteinuria and/or eGFR<60 mL/min). For 23 children, the renal involvement was confirmed by a kidney biopsy. Nearly 93.9% of pediatric IgAV-N patients exhibited hematuria and 78.8% proteinuria over 1 g/g. Renal failure was less prevalent in children than in adults (3.0% of pediatric IgAV-N patients compared with 40.0% of adult IgAV-N patients (HSPrognosis study group, p<0.0001). The median proteinuria creatininuria ratio (PCR) was 2.5 (0.54–6.20) g/g and the median serum creatinine level 41.0 (34.5–57.0) μmol/L (for mg/dl conversion, divide by factor 88.4) for the pediatric IgAV-N group. The most frequent lesion in renal biopsies was diffuse mesangial proliferation with less than 50% crescent (class IIIb, 52.2%, Table 2). Only 3 patients had more than 50% crescents (two patients were class IV and one patient class V). None of the patients had global sclerosis and interstitial fibrosis was rare. After one year, 2 children were lost to follow-up. As described in other cohorts, the renal prognosis was good. No children exhibited de novo renal involvement or renal failure. The median proteinuria/creatininuria ratio only reached 0.12 (0.06–0.23) g/g for the IgAV-N group after one year. Only one (2.0%) patient maintained PCR >1 g/g, but the serum creatinine concentration was low (49.8 μmol/L). Concerning treatments, most of the patients received steroids (65.5%), 22 (45.8%) received Angiotensin Converting Enzyme inhibitors (ACEi) or Angiotensin Receptor Blockers (ARB), 15 (31.2%) patients received no treatment, 4 (8.3%) received cyclophosphamide and 1 (2.0%) rituximab. There was no correlation between histological classification and proteinuria after one year of follow-up (Table 2) or other clinical parameters.
Table 1. Patient characteristics.
| Controls | IgAV-woN | IgAV-N | |
|---|---|---|---|
| Number (percent) | 21 | 17 (34.0%) | 33 (66.0%) |
| Age (years): median (IQR) | 8 (7–11) | 6 (5–8) | 8 (7–10) a |
| Male gender: number (percent) | 13 (61.9%) | 12 (70.5%) | 20 (60.6%) |
| Skin involvement: number (percent) | 0 | 17 (100%) | 33 (100%) |
| Joint involvement: number (percent) | 0 | 12 (70.6%) | 26 (78.7%) |
| Gastrointestinal involvement: number (percent) | 0 | 7 (41.2%) | 20 (60.6%) |
| Renal involvement: number (percent) | 0 | 0 | 33 (100%) b,c |
| Hematuria: number (percent) | 0 | 0 | 31 (93.9%) b,c |
| PCR ≥ 0.5 g/g: number (percent) | 0 | 0 | 26 (78.8%) b,c |
| PCR (g/g): median (IQR) | 0.05 (0.04–0.07) | 0.05 (0.04–0.09) | 2.5 (0.54–6.20) a,d |
| PCR group | |||
| 0.5–1 g/g: number (percent) | 0 | 0 | 4 (12.1%) |
| 1–3 g/g: number (percent) | 0 | 0 | 9 (27.3%) a, d |
| ≥ 3 g/g: number (percent) | 0 | 0 | 13 (39.4%) a, d |
| eGFR (mL/min/1.73m2): median (IQR) | 137.0 (102.5–169.3) | 165.1 (139.3–206.2) | 152.1 (118.7–180.1) |
| eGFR group | |||
| ≥ 90 mL/min/1.73m2: number (percent) | 20 (95.2%) | 16 (94.1%) | 31 (93.9%) b |
| 60–89 mL/min/1.73m2: number (percent) | 1 (4.8%) | 0 | 1 (3.0%) b |
| 30–59 mL/min/1.73m2: number (percent) | 0 | 1 (5.9%) | 1 (3.0%) |
| 15–29 mL/min/1.73m2: number (percent) | 0 | 0 | 0 |
| < 15 mL/min/1.73m2: number (percent) | 0 | 0 | 0 |
| Renal failure (eGFR < 60): number (percent) | 0 | 0 | 1 (3.0%) d |
| Serum creatinine (μmol/L): median (IQR) | 41.0 (31.7–66.0) | 36.0 (29.0–43.8) | 41.0 (34.5–57.0) d |
eGFR was calculated the Schwartz formula for children.
Conversion factor for serum creatinine in mg/dL to μmol/L, x 88.4.
The Mann-Whitney U test was used for median comparisons and Fisher exact test for categorical comparisons:
a IgAV-woN vs IgAV-N, P<0.05
b controls vs IgAV-N, P<0.0001
c IgAV-woN vs IgAV-N, P<0.0001
d IgAV-woN vs IgAV-N, P<0.01.
Abbreviations: IgAV-woN = IgAV patients without nephritis. IgAV-N = IgAV patients with nephritis. IQR = Interquartile range. PCR = proteinuria creatininuria ratio. eGFR = estimated glomerular filtration rate.
Table 2. Histological class from renal biopsies.
| Histological Class | II | IIIa | IIIb | IV | V |
|---|---|---|---|---|---|
| Number (percent) | 5 (21.7%) | 3 (13.0%) | 12 (52.2%) | 2 (8.7%) | 1 (4.3%) |
| Age (years): median (IQR) | 9.0 (6.5–12.5) | 4.0 (4.0–8.0) | 8.0 (7.3–9.0) | 12.0 (10.0–14.0) | 7.0 |
| Male gender: number (percent) | 4 (80.0%) | 2 (66.7%) | 5 (41.6%) | 2 (100%) | 1 (100%) |
| PCR (g/g) at day 1: median (IQR) | 0.55 (0.29–2.48) | 3.12 (1.61–15.5) | 5.55 (2.68–8.85) | 2.35 (1.12–3.58) | 5.5 |
| PCR (g/g) after one year | 0.24 (0.06–0.50) | 0.22 (0.18–0.28) | 0.10 (0.07–0.22) | 1.61 (0.07–3.16) | 0.07 |
Abbreviations: IQR = interquartile range. PCR = proteinuria creatininuria ratio.
Immunoglobulins
The concentrations of immunoglobulins were measured in the serum and urine of children, collected at the time of the rash, before the onset of any treatment. The IgA and IgE serum concentrations of all IgAV patients were higher than those of the controls (p<0.0001), with no differences between IgAV-woN and IgAV-N patients (Table 2). The IgD, IgG and IgM serum concentrations and the Igλ/Igκ light-chain ratios were similar between IgAV patients and controls (Table 3).
Table 3. Biomarkers in the circulation and urine of pediatric patients and controls at the time of purpura rash (day 1).
| Controls | IgAV-woN | IgAV-N | |
|---|---|---|---|
| Immunoglobulins in serum | |||
| IgA (g/L) | 1.0±0.2 | 2.7±0.2 a | 2.4±0.2 b |
| IgD (mg/L) | 17.0±6.0 | 23.1±4.7 | 27.1±5.5 |
| IgE (μg/L) | 49.0±13.2 | 149.0±25.2 c | 164.0±27.0 b |
| IgG (g/L) | 9.0±0.4 | 10.5±0.7 | 8.4±0.7 |
| IgM (g/L) | 1.0±0.1 | 1.3±0.1 | 1.4±0.1 |
| Igλ/Igκ ratio | 0.4±0.1 | 0.5±0.0 | 0.5±0.0 |
| Immunoglobulins in urine | |||
| IgA/Cr (g/mmol) | 0.1±0.0 | 0.1±0.0 | 1.4±0.3 b, d |
| IgD/Cr (mg/mmol) | nd | nd | nd |
| IgE/Cr (μg/mmol) | nd | nd | nd |
| IgG/Cr (g/mmol) | 0.7±0.1 | 0.4±0.0 | 4.9±1.2 e |
| IgM/Cr (g/mmol) | 0.0±0.0 | 0.0±0.0 | 0.2±0.2 b, d |
| Igλ/Igκ ratio | 0.7±0.2 | 0.6±0.2 | 1.5±0.4 e |
| Cytokines in plasma | |||
| IL-1β (pg/mL) | 1.0±0.3 | 8.3±2.5 a, d | 1.9±0.4 |
| IL-6 (pg/mL) | 1.0±0.5 | 5.1±0.7 a, e | 3.6±0.6 b |
| IL-8 (pg/mL) | 10.0±2.6 | 95.8±25.4 a, e | 16.5±3.0 |
| IL-10 (pg/mL) | 3.0±0.7 | 1.7±0.8 | 1.6±0.3 |
| IL-12p70 (pg/mL) | 1.0±0.5 | 2.9±0.8 | 2.1±0.4 |
| TNF-α (pg/mL) | 1.0±0.2 | 1.2±0.3 | 1.1±0.4 |
| Cytokines in urine | |||
| IL-1β/Cr (μg/mmol) | 1.0±0.4 | 1.3±0.6 | 3.8±1.0 |
| IL-6/Cr (μg/mmol) | 0.0±0.1 | 0.6±0.2 | 4.5±1.1 b, e |
| IL-8/Cr (μg/mmol) | 2.0±0.6 | 1.6±0.5 | 10.9±2.4 b, e |
| IL-10/Cr (μg/mmol) | 0.1±0.1 | 0.2±0.1 | 0.8±0.1 e |
| IL-12p70/Cr (μg/mmol) | 0.0±0.0 | 0.0±0.0 | 0.5±0.3 |
| TNF-α/Cr (μg/mmol) | 0.0±0.0 | 0.0±0.0 | 0.3±0.2 |
| NGAL | |||
| NGAL in plasma (ng/ml) | 118.0±18.5 | 297.1±35.7 a | 240.3±30.3 b |
| NGAL/Cr in urine (μg/mmol) | 6.1±1.1 | 2.5±0.6 | 7.0±1.5 |
Values are represented as the mean ± sem. Mann-Whitney U test.
a controls vs IgAV-woN, P<0.0001
b controls vs IgAV-N, P<0.0001
c controls vs IgAV-woN, P<0.01
d IgAV-woN vs IgAV-N, P<0.0001
e IgAV-woN vs IgAV-N, P<0.01.
Abbreviations: Cr = creatinine, nd = not detected, NGAL = neutrophil gelatinase associated lipocalin
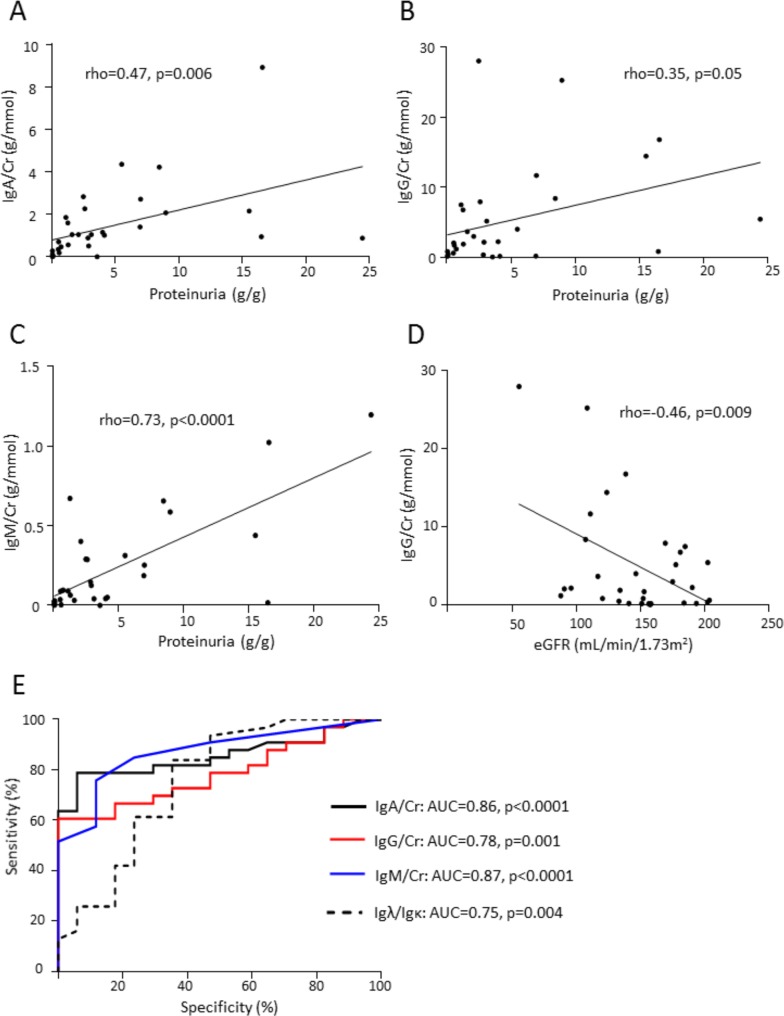
IgD and IgE were not detected by ELISA in urine. IgA, IgG and IgM concentrations were increased in IgAV-N patients compared with the controls (p<0.0001) and with IgAV-woN patients (p<0.0001) (Table 3). The Igλ/Igκ ratio in urine samples was also increased for IgA-N patients compared with the healthy controls and IgA-woN patients (p<0.0001; Table 3). IgA, IgG and IgM urine concentrations were positively correlated with proteinuria (rho = 0.47, p = 0.006; rho = 0.35, p = 0.05; rho = 0.73, p<0.0001, respectively, Fig 1A, 1B and 1C). Only urinary IgG was negatively correlated with eGFR (rho = -0.46, p = 0.009, Fig 1D). Moreover, IgA, IgG, and IgM levels and the Igλ/Igκ ratio could accurately identify patients with nephritis (AUC = 0.86, confidence interval (CI) = 0.75–0.96, p<0.0001; AUC = 0.78, CI = 0.66–0.90, p = 0.001; AUC = 0.87, CI = 0.77–0.97, p<0.0001; and AUC = 0.75, CI = 0.59–0.90, p = 0.004, respectively; Fig 1E).
Fig 1. Urinary immunoglobulins and correlation with clinical parameters.
Correlations between urinary IgA (A), IgG (B), IgM (C) and proteinuria in IgAV-N patients. (D) Correlations between urinary IgG and eGFR in IgAV-N patients. (E) Receiver operating characteristic (ROC) curves of the urinary concentrations of IgA, IgM, IgG and Igλ/Igκ ratio comparing the IgAV-woN and IgAV-N group.
IgA1 glycosylation and IgA complexes
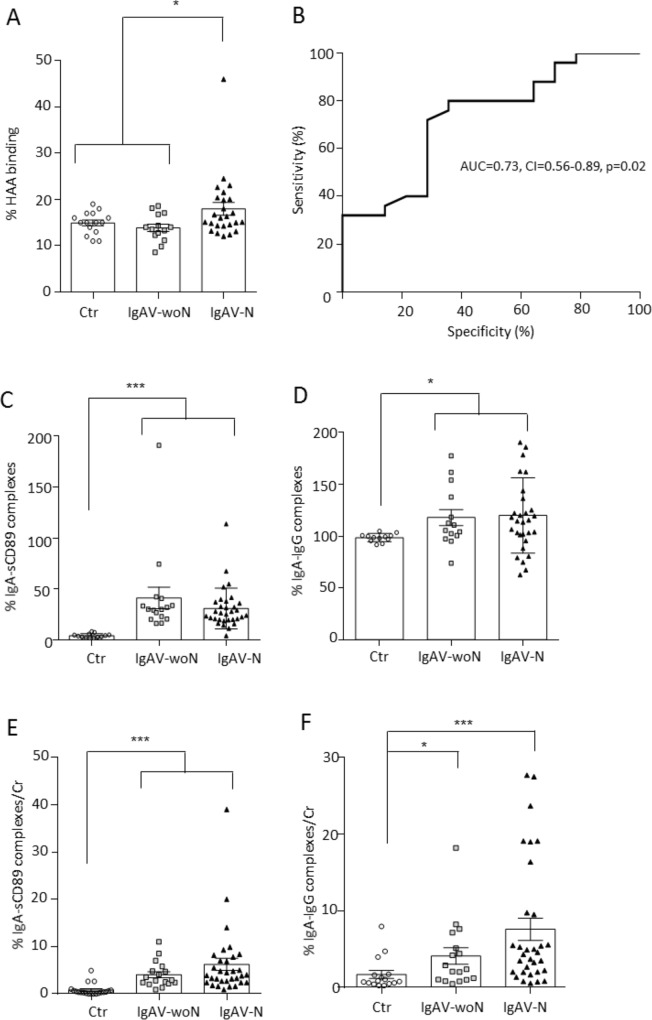
After the examination of the IgA quantities, we assessed the levels of IgA1 glycosylation in IgAV patients: the O-glycosylation of IgA1. Circulating galactose-deficient IgA1 (GD-IgA1) was more elevated in IgAV-N patients compared to controls and in IgAV-woN patients (Fig 2A), as previously described [7], and this feature could be used to identify patients with nephritis (AUC = 0.73, CI = 0.56–0.89, p = 0.02; Fig 2B). No correlations were found with proteinuria or eGFR.
Fig 2. Markers of IgA nephropathy in IgAV.
(A) Percentages of HAA binding on circulating IgA corresponding to Gd-IgA1 levels. (B) ROC curve of the serum Gd-IgA1 levels comparing the IgAV-woN and IgAV-N group. (C) Percentages of IgA-sCD89 complex level in patient sera. (D) Percentages of IgA-IgG complex level in patient sera. (E) Percentages of IgA-sCD89 complex level in patient urine. (F) Percentages of IgA-IgG complex level in patient urine.
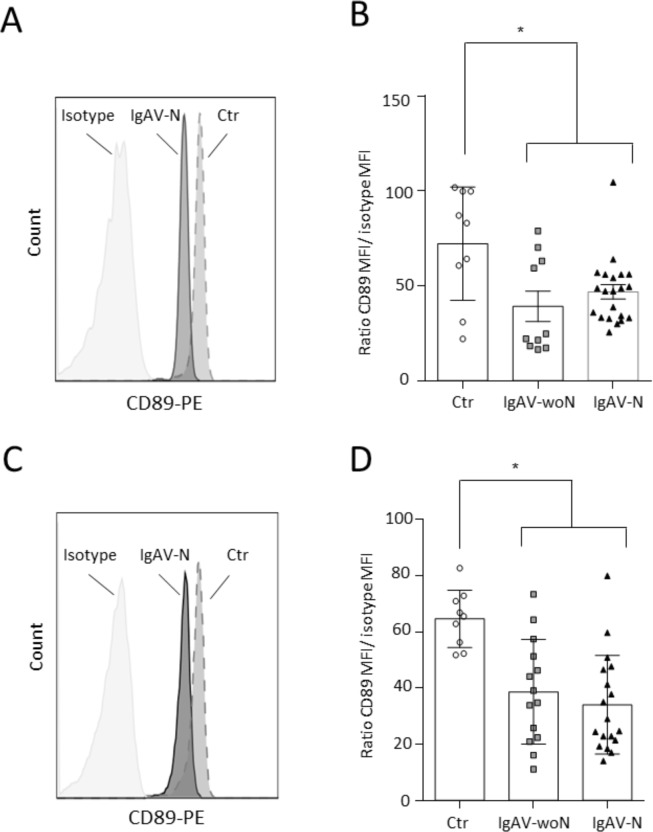
Other markers of IgA nephropathy [12] were examined in IgAV patients. The levels of circulating IgA complexes (IgA-sCD89 and IgA-IgG) were increased in all IgAV patients compared with the controls (p<0.0001; Fig 2C and 2D, respectively). In urine samples, these complex levels were also increased in all IgAV patients compared with the healthy controls and IgAV-woN patients (p<0.001 and p<0.01 respectively; Fig 2E and 2F). IgAV-N patients exhibited more excretion of IgA complexes compared to IgAV-woN, but this increase was not statistically significant. Since the increase of circulating sCD89 in IgAV patients could be due to an increase of CD89 shedding at the cell surface of immune cells, the expression of CD89 was assessed on fresh blood samples by flow cytometry. There was a significantly decreased expression of CD89 on the surface of circulating granulocytes (p = 0.007; Fig 3A and 3B) and monocytes (p = 0.0006; Fig 3C and 3D).
Fig 3. CD89 expression on blood granulocytes and monocytes.
(A) Representative histogram of CD89 expression on granulocytes. (B) Ratio of the CD89 mean fluorescence intensity (MFI) / isotype MFI on blood granulocytes. (C) Representative histogram of CD89 expression on monocytes. (D) Ratio of the CD89 mean fluorescence intensity (MFI) / isotype MFI on blood monocytes.
Cytokines
We then measured the concentrations of pro-inflammatory cytokines in plasma using CBA assays. IL-1β, IL-6 and IL-8 concentrations were higher in the IgAV group than in the control group (Table 2). The serum concentrations IL-12p70, TNF-α and the anti-inflammatory IL-10 were similar among IgAV patients and controls (Table 2).
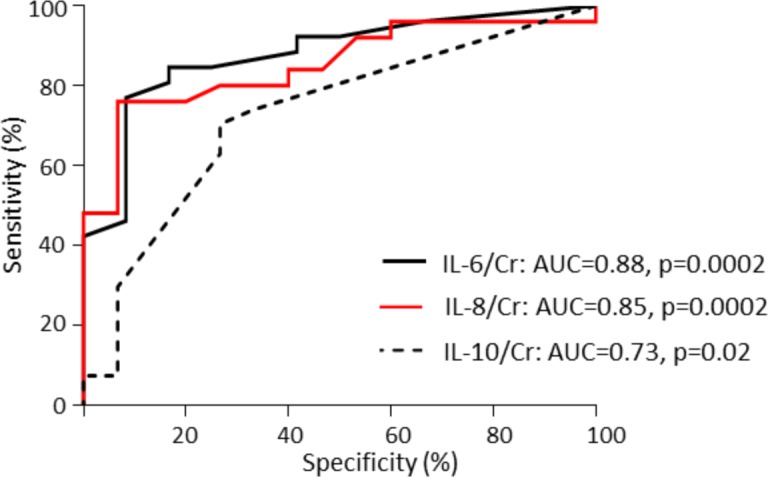
IgAV nephritis in children was associated with the urinary excretion of IL-6, IL-8 and IL-10. Their concentrations were significantly higher in IgAV-N patients relative to healthy controls and IgAV-woN patients (p<0.0001; Table 2). Moreover, IL-6, IL-8 and IL-10 concentrations could accurately identify patients with nephritis (IL-6: AUC = 0.88, CI = 0.76–0.99, p = 0.0002; IL-8: AUC = 0.85, CI = 0.73–0.97, p = 0.0002; IL-10: AUC = 0.73, CI = 0.56–0.89, p = 0.02; Fig 4).
Fig 4. ROC curves of urinary cytokines comparing the IgAV-woN and IgAV-N groups.
NGAL
The NGAL plasma concentration was increased in IgAV pediatric patients compared with the matched controls (p<0.0001, Table 2) with no difference between IgAV-woN and IgAV-N patients. In urine, NGAL was not a discriminating biomarker of nephritis for children (Table 2).
Discussion
In this prospective, multicenter study of a cohort of IgAV children, all of whom were included at the initial presentation of the disease, we described the clinical and biological features of the disease. In addition, we identified noninvasive biomarkers that could be used to safely diagnose IgAV nephritis in children. These results should be confirmed in a larger pediatric cohort and with a longer follow-up.
Immunoglobulins in IgAV
IgA deposition in the different organs led to the study of immunoglobulin production in IgAV. Previous studies showed increased IgA [6,13,14] and IgE [6,13,15] serum concentrations in IgAV children and one described an increase in IgD in 39 patients [16]. We confirmed these results for IgA and IgE, but not for IgD, in our pediatric cohort. These elevations did not accurately discriminate patients with nephritis from those without. Urinary IgA, IgM and IgG were more indicative of nephritis in our cohort. Moreover, other groups have studied the presence of immunoglobulins in organ deposits. IgM has been associated with IgA skin deposits in children [17] and nephritis in adults [18–20]. IgG deposition in the kidney was associated with the nephritis outcome in adult IgAV patients [20].
Hypogalactosylation of IgA1
The decrease of galactosylation in the hinge region of IgA1 (Gd-IgA1) molecules has been proposed to be the first pathological hit for kidney IgA1 deposition [7,21]. We confirmed in our pediatric IgAV cohort that the increase in serum Gd-IgA1 level was only associated with nephritis. Moreover, it has been described that in IgA-N, the recognition of this IgA1 hinge region neo-epitope, due to hypogalactosylation by IgA or IgG antibodies leads to the generation of immune complexes in the circulation (i.e., Gd-IgA1-IgA, Gd-IgA1-IgG) [22]. Here, we found that the level of IgA-IgG complexes was increased in the circulation of all pediatric patients, as previously described [23]. In contrast, in urine, these IgA complex levels were only associated with nephritis.
IgA receptors
The receptor FcαRI or CD89 binds to IgA followed by cleavage of the CD89 extracellular domain and the release of nephrotoxic IgA-sCD89 complexes. In IgA nephropathy (IgA-N), these immune complexes are trapped in the glomerular mesangium by the mesangial transferrin receptor, which serves as an IgA1-sCD89 receptor [24]. The same pathological phenomenon also seems to occur in IgAV. In children, the circulating IgA-sCD89 complex level was increased in all IgAV patients and was associated with the decreased expression of CD89 at the cell surface of circulating granulocytes and monocytes. IgAV-N patients exhibited a higher level of these complexes in their urine. Adult patients with progressive IgA nephropathy had lower serum levels of IgA-sCD89 complexes than did patients with a good prognosis [25], as did recurrent IgA-N patients after grafting compared to non-recurrent IgA-N patients [26].
Cytokines in IgAV
Concerning pro-inflammatory cytokines, we found elevated levels of IL-1β, IL-6 and IL-8 in the serum of all patients, with or without nephritis, compared to the controls. Urine IL-6, IL-8, and IL-10 concentrations distinguished IgAV with or without nephritis. Those cytokines have been implicated in IgA-N and IgAV with nephritis, particularly in mesangial cell activation, proliferation, crescent formation and glomerulosclerosis. Higher urine IL-6 levels at the time of IgA-N diagnosis increased the risk of progression in 59 patients followed for 8 years [27]. Increased serum IL-6 has been associated with the acute phase of IgAV [28]. More recently, serum IL-6 and IL-8 levels were found to be significantly increased in cases with renal involvement [29]. Circulating tumor necrosis factor receptor levels have been shown to be correlated with interstitial fibrosis and tubular atrophy in kidney biopsies, regardless of renal function; in addition, they were predictive of renal progression in 347 IgA-N cases confirmed by biopsy, indicating a role of the TNF pathway in this type of glomerulonephritis, which has been demonstrated in other chronic kidney diseases, such as diabetes or lupus nephritis [30]. Moreover, in small cohorts of IgAV patients, the serum level of TNFα was higher in IgAV-N than in IgAV-woN patients [31] and higher in active-phase IgAV than in the controls [13,32].
NGAL as a marker of kidney injury
Neutrophil gelatinase-associated lipocalin (NGAL) is a 25-kDa acute-phase protein that was originally purified from human neutrophils and is markedly induced in injured epithelial cells. When tubular damage occurs, NGAL mRNA expression is significantly upregulated in the distal nephron segment to promote cellular proliferation and differentiation [33,34]. The NGAL protein is released into the circulation, freely filtered by the glomerulus and reabsorbed in the proximal tubules. Thus, following tubular damage, both serum and urine NGAL expression levels are increased. Many studies have been published on the predictive value of NGAL in acute renal injury (e.g., cardiac surgery, contrast-induced nephropathy, kidney transplantation). In IgAV patients, urinary NGAL seems to distinguish adult patients with or without nephritis [35] similarly to children [36]. An increased serum NGAL concentration was present in both IgAV-N and IgAV-woN children in our study. There was a tendency for increased urinary NGAL in IgAV-N compared to IgAV-woN patients.
Conclusions
This prospective cohort study, with pediatric patients who were included at the onset of the disease, identified biomarkers that are able to segregate patients initially with or without nephritis. The serum levels of Gd-IgA1 and urinary levels of IgA, IgG, IgM, IL-6, IL-8, and IL-10 complexes showed an accurate predictive performance in the identification of IgAV patients with renal involvement at the time of diagnosis.
Acknowledgments
We are most grateful to the study participants, their parents and to the clinicians of the HSPrognostic Group. This study was financially supported by Assistance Publique–Hôpitaux de Paris (Département de la Recherche Clinique et du Développement),by INSERM (grant RCT 2010). and by Fondation pour la Recherche Médicale (FRM, grant number DMP20101120408).
The following members of the HSPrognostic group are listed alphabetically.
Zahir Amoura (Pitié Salpêtrière Hospital, Paris)
Vincent Audard (Henri Mondor’s Hospital, Créteil)
Sonia Azib (René Dubos’s Hospital, Pontoise)
Martine Bagot (Saint-Louis’s Hospital Paris)
Véronique Baudouin (Robert Debré’s Hospital, Paris)
Olivier Benveniste (Pitié Salpêtrière Hospital, Paris)
Maud Bezier (Saint-Louis’s Hospital Paris)
Coralie Bloch-Queyrat (Pitié Salpêtrière Hospital, Paris)
Nathalie Bocquet (Necker’s Hospital, Paris)
Jean-Jacques Boffa (Tenon’s Hospital, Paris)
Jean-David Bouaziz (Saint-Louis’s Hospital Paris)
Karim Bouchireb (Necker’s Hospital, Paris)
Emmanuelle Bourrat (Robert Debré’s Hospital, Paris)
Philippe Bouvier (La Dracenie Hospital, Draguignan)
Karine Brochard (Pediatric Hospital, Toulouse)
Guillaume Burda (Hospital of Bethune)
Guillaume Bussone (Cochin’s Hospital, Paris)
Valérie Caudwell (Sud Francilien Hospital, Evry)
Laure Champion (Bichat’s Hospital, Paris)
Jonathan M Chemouny (Bichat’s Hostpital, Paris)
Gérard Cheron (Necker’s Hospital, Paris)
Florence Cordoliani (Saint-Louis’s Hospital Paris)
Stéphane Decramer (Pediatric Hospital, Toulouse)
Laure Dehen (Saint-Louis’s Hospital Paris)
Laurène Dehoux (Robert Debré’s Hospital, Paris)
Michel Delahousse (Foch’s Hospital, Suresnes)
Mathilde de Menthon (Saint-Louis’s Hospital Paris)
Claire Demongeot (Saint-Louis’s Hospital Paris)
Antoine Dossier (Bichat’s Hospital, Paris)
Claire Dossier (Robert Debré’s Hospital, Paris)
Eric Daugas (Bichat’s Hospital, Paris)
Monique Dehoux (Bichat’s Hospital, Paris)
Olivier Fain (Jean Verdier’s Hospital, Bondy)
Dominique Farge (Saint-Louis’s Hospital Paris)
Marc Fila, (Robert Debré’s Hospital, Paris)
Charlotte Fite (Bichat’s Hospital, Paris)
Martin Flamant (Bichat’s Hospital, Paris)
Héloise Flament (Bichat’s Hospital, Paris
Antoine Froissart (Intercommunal Hospital of Créteil)
Elisa Funck-Brentano (Saint-Louis’s Hospital Paris)
Sophie Georgin-Lavialle (Georges Pompidou European Hospital, Paris)
Denis Glotz (Saint-Louis’s Hospital Paris)
Bertrand Godeau (Henri Mondor’s Hospital, Créteil)
Philippe Grimbert (Henri Mondor’s Hospital, Créteil)
Loïc Guillevin (Cochin’s Hospital, Paris)
Maya Halabi-Tawil (Saint-Louis’s Hospital Paris)
Isabelle Halphen (Necker’s Hospital, Paris)
Alexandre Karras (Georges Pompidou European Hospital, Paris)
Theresa Kwon (Robert Debré’s Hospital, Paris)
Céline Lebas (Jean Bernard Hospital, Valenciennes)
Nicolas Limal (Henri Mondor’s Hospital, Créteil)
Anne Maisin (Robert Debré’s Hospital, Paris)
Alexis Mathian (Pitié Salpêtrière Hospital, Paris)
Arsène Mekinian (Jean Verdier’s Hospital, Bondy)
Jean Christophe Mercier (Robert Debré’s Hospital, Paris)
Fabrice Mihout (Tenon’s Hospital, Paris)
Gentiane Monsel (Saint-Louis’s Hospital Paris)
Vincent Pestre (Cochin’s Hospital, Paris)
Maryam Piram (Hospital of Kremlin Bicêtre)
Thomas Quemeneur (Jean Bernard’s Hospital, Valenciennes)
Quentin Raimbourg (Bichat’s Hospital, Paris)
Agathe Raynaud-Simon (Bichat’s Hospital, Paris)
Philippe Rémy (Henri Mondor’s Hospital, Créteil)
Jacqueline Rivet (Saint-Louis’s Hospital Paris)
Alain Robert (La Dracenie Hospital, Draguignan)
Rémi Salomon (Necker’s Hospital, Paris)
Anne Saussine-Hickman (Saint-Louis’s Hospital Paris)
Alexandre Seidowsky (Amroise Paré’s Hospital, Boulogne)
Assia Smail (Robert Debré’s Hospital, Paris)
Stéphanie Tellier (Pediatric Hospital, Toulouse)
Eric Thervet (Georges Pompidou European Hospital, Paris)
Leila Tricot (foch’s Hospital, Suresnes)
Philippe Vanhille (Jean Bernard’s Hospital, Valenciennes)
David Verhelst (Henri Duffaut’s Hospital, Avignon)
Laurence Vrigneaud (Jean Bernard’s Hospital, Valenciennes)
Anne-Sophie Verhoeven (Bichat’s Hospital, Paris)
Jérôme Verine (Saint-Louis’s Hospital Paris)
Emmanuelle Vidal-Petiot (Bichat’s Hospital, Paris)
Manuelle Viguier (Saint-Louis’s Hospital Paris)
Nathalie Vittoz (Sud Francilien Hospital, Evry)
François Vrtovsnik (Bichat’s Hospital, Paris)
Data Availability
All relevant data are within the paper.
Funding Statement
This study was financially supported by Assistance Publique – Hôpitaux de Paris (Département de la Recherche Clinique et du Développement) and together by INSERM (grant RCT 2010). Fondation pour la Recherche Médicale (FRM) supported this study (grant number DMP20101120408). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Audemard-Verger A, Pillebout E, Guillevin L, Thervet E, Terrier B (2015) IgA vasculitis (Henoch-Shonlein purpura) in adults: Diagnostic and therapeutic aspects. Autoimmun Rev 14: 579–585. doi: 10.1016/j.autrev.2015.02.003 [DOI] [PubMed] [Google Scholar]
- 2.Coppo R, Mazzucco G, Cagnoli L, Lupo A, Schena FP (1997) Long-term prognosis of Henoch-Schonlein nephritis in adults and children. Italian Group of Renal Immunopathology Collaborative Study on Henoch-Schonlein purpura. Nephrol Dial Transplant 12: 2277–2283. [DOI] [PubMed] [Google Scholar]
- 3.Pillebout E, Thervet E, Hill G, Alberti C, Vanhille P,Nochi D (2002) Henoch-Schonlein Purpura in adults: outcome and prognostic factors. J Am Soc Nephrol 13: 1271–1278. [DOI] [PubMed] [Google Scholar]
- 4.Saulsbury FT (2007) Clinical update: Henoch-Schonlein purpura. Lancet 369: 976–978. doi: 10.1016/S0140-6736(07)60474-7 [DOI] [PubMed] [Google Scholar]
- 5.Davin JC, Coppo R (2014) Henoch-Schonlein purpura nephritis in children. Nat Rev Nephrol 10: 563–573. doi: 10.1038/nrneph.2014.126 [DOI] [PubMed] [Google Scholar]
- 6.Pan YX, Ye Q, Shao WX, Shang SQ, Mao JH, Zhang T, et al. (2014) Relationship between immune parameters and organ involvement in children with Henoch-Schonlein purpura. PLoS One 9: e115261 doi: 10.1371/journal.pone.0115261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Allen AC, Willis FR, Beattie TJ, Feehally J (1998) Abnormal IgA glycosylation in Henoch-Schonlein purpura restricted to patients with clinical nephritis. Nephrol Dial Transplant 13: 930–934. [DOI] [PubMed] [Google Scholar]
- 8.Lau KK, Wyatt RJ, Moldoveanu Z, Tomana M, Julian BA, Hogg RJ, et al. (2007) Serum levels of galactose-deficient IgA in children with IgA nephropathy and Henoch-Schonlein purpura. Pediatr Nephrol 22: 2067–2072. doi: 10.1007/s00467-007-0623-y [DOI] [PubMed] [Google Scholar]
- 9.Berthelot L Jamin A, Viglietti D, Chemouny JM, Ayari H, Housset P, et al. (2017) Value of biomarkers for predicting IgA vasculitis nephritis outcome in an adult prospective cohort. Nephrol Dial Transplant. doi: 10.1093/ndt/gfx300 [DOI] [PubMed] [Google Scholar]
- 10.Ozen S, Pistorio A, Iusan SM, Bakkaloglu A, Herlin T, Brik R, et al. (2010) EULAR/PRINTO/PRES criteria for Henoch-Schonlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part II: Final classification criteria. Ann Rheum Dis 69: 798–806. doi: 10.1136/ard.2009.116657 [DOI] [PubMed] [Google Scholar]
- 11.Schwartz GJ, Munoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, et al. (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20: 629–637. doi: 10.1681/ASN.2008030287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Robert T, Berthelot L, Cambier A, Rondeau E, Monteiro RC (2015) Molecular Insights into the Pathogenesis of IgA Nephropathy. Trends Mol Med 21: 762–775. doi: 10.1016/j.molmed.2015.10.003 [DOI] [PubMed] [Google Scholar]
- 13.Ye Q, Shang SQ, Liu AM, Zhang T, Shen HQ, Chen XJ, et al. (2015) 24h Urinary Protein Levels and Urine Protein/Creatinine Ratios Could Probably Forecast the Pathological Classification of HSPN. PLoS One 10: e0127767 doi: 10.1371/journal.pone.0127767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fretzayas A, Sionti I, Moustaki M, Nicolaidou P (2009) Clinical impact of altered immunoglobulin levels in Henoch-Schonlein purpura. Pediatr Int 51: 381–384. doi: 10.1111/j.1442-200X.2008.02762.x [DOI] [PubMed] [Google Scholar]
- 15.Davin JC, Pierard G, Dechenne C, Grossman D, Nagy J, Quacoe M, et al. (1994) Possible pathogenic role of IgE in Henoch-Schonlein purpura. Pediatr Nephrol 8: 169–171. [DOI] [PubMed] [Google Scholar]
- 16.Saulsbury FT (1998) Increased serum IgD concentrations in children with Henoch-Schonlein purpura. Br J Rheumatol 37: 570–572. [DOI] [PubMed] [Google Scholar]
- 17.Yuksel S, Caglar M, Evrengul H, Becerir T, Tepeli E, Ergin A, et al. (2015) Could serum pentraxin 3 levels and IgM deposition in skin biopsies predict subsequent renal involvement in children with Henoch-Schonlein purpura? Pediatr Nephrol 30: 969–974. doi: 10.1007/s00467-014-3026-x [DOI] [PubMed] [Google Scholar]
- 18.Belli AA, Dervis E (2014) The correlation between cutaneous IgM deposition and renal involvement in adult patients with Henoch-Schonlein purpura. Eur J Dermatol 24: 81–84. doi: 10.1684/ejd.2013.2243 [DOI] [PubMed] [Google Scholar]
- 19.Takeuchi S, Soma Y, Kawakami T (2010) IgM in lesional skin of adults with Henoch-Schonlein purpura is an indication of renal involvement. J Am Acad Dermatol 63: 1026–1029. doi: 10.1016/j.jaad.2009.11.690 [DOI] [PubMed] [Google Scholar]
- 20.Poterucha TJ, Wetter DA, Grande JP, Gibson LE, Camilleri MJ, Lohse CM, et al. (2014) A retrospective comparison of skin and renal direct immunofluorescence findings in patients with glomerulonephritis in adult Henoch-Schonlein purpura. J Cutan Pathol 41: 582–587. [DOI] [PubMed] [Google Scholar]
- 21.Kiryluk K, Moldoveanu Z, Sanders JT, Eison TM, Suzuki H, Julian BA, et al. (2011) Aberrant glycosylation of IgA1 is inherited in both pediatric IgA nephropathy and Henoch-Schonlein purpura nephritis. Kidney Int 80: 79–87. doi: 10.1038/ki.2011.16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mestecky J, Raska M, Julian BA, Gharavi AG, Renfrow MB, Moldoveanu Z, et al. (2013) IgA nephropathy: molecular mechanisms of the disease. Annu Rev Pathol 8: 217–240. doi: 10.1146/annurev-pathol-011110-130216 [DOI] [PubMed] [Google Scholar]
- 23.Blanco Quiros A, Blanco C, Alvarez J, Solis P, Conde F, Gomez S (1994) Anti-immunoglobulin antibodies in children with Schonlein-Henoch syndrome. Absence of serum anti-IgA antibodies. Eur J Pediatr 153: 103–106. [DOI] [PubMed] [Google Scholar]
- 24.Berthelot L, Papista C, Maciel TT, Biarnes-Pelicot M, Tissandie E, Wang PH, et al. (2012) Transglutaminase is essential for IgA nephropathy development acting through IgA receptors. J Exp Med 209: 793–806. doi: 10.1084/jem.20112005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vuong MT, Hahn-Zoric M, Gunnarsson I, van Kooten C,Wramner L, Saddighzadeh M, et al. (2010) Association of soluble CD89 levels with disease progression but not susceptibility in IgA nephropathy. Kidney Int 78:1281–1287. doi: 10.1038/ki.2010.314 [DOI] [PubMed] [Google Scholar]
- 26.Berthelot L, Robert T, Vuiblet V, Tabary T, Braconnier A, Drame M, et al. (2015) Recurrent IgA nephropathy is predicted by altered glycosylated IgA, autoantibodies and soluble CD89 complexes. Kidney Int 88: 815–822. doi: 10.1038/ki.2015.158 [DOI] [PubMed] [Google Scholar]
- 27.Harada K, Akai Y, Kurumatani N, Iwano M, Saito Y (2002) Prognostic value of urinary interleukin 6 in patients with IgA nephropathy: an 8-year follow-up study. Nephron 92: 824–826. doi: 10.1159/000065465 [DOI] [PubMed] [Google Scholar]
- 28.Lin CY, Yang YH, Lee CC, Huang CL, Wang LC, Chiang BL (2006) Thrombopoietin and interleukin-6 levels in Henoch-Schonlein purpura. J Microbiol Immunol Infect 39: 476–482. [PubMed] [Google Scholar]
- 29.Kimura S, Takeuchi S, Soma Y, Kawakami T (2013) Raised serum levels of interleukins 6 and 8 and antiphospholipid antibodies in an adult patient with Henoch-Schonlein purpura. Clin Exp Dermatol 38: 730–736. doi: 10.1111/ced.12089 [DOI] [PubMed] [Google Scholar]
- 30.Oh HJ, Ahn SV, Yoo DE, Kim SJ, Shin DH, Lee MJ, et al. (2012) Clinical outcomes, when matched at presentation, do not vary between adult-onset Henoch-Schonlein purpura nephritis and IgA nephropathy. Kidney Int 82: 1304–1312. doi: 10.1038/ki.2012.302 [DOI] [PubMed] [Google Scholar]
- 31.Besbas N, Saatci U, Ruacan S, Ozen S, Sungur A, Bakkaloglu A, et al. (1997) The role of cytokines in Henoch Schonlein purpura. Scand J Rheumatol 26: 456–460. [DOI] [PubMed] [Google Scholar]
- 32.Del Vecchio GC, Penza R, Altomare M, Piacente L, Aceto G, Lassandro G, et al. (2008) Cytokine pattern and endothelium damage markers in Henoch-Schonlein purpura. Immunopharmacol Immunotoxicol 30: 623–629. doi: 10.1080/08923970801973646 [DOI] [PubMed] [Google Scholar]
- 33.El Karoui K, Viau A, Dellis O, Bagattin A, Nguyen C, Baron W, et al. (2016) Endoplasmic reticulum stress drives proteinuria-induced kidney lesions via Lipocalin 2. Nat Commun 7: 10330 doi: 10.1038/ncomms10330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Viau A, El Karoui K, Laouari D, Burtin M, Nguyen C, Mori K, et al. (2010) Lipocalin 2 is essential for chronic kidney disease progression in mice and humans. J Clin Invest 120: 4065–4076. doi: 10.1172/JCI42004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen T, Lu YH, Wang WJ, Bian CY, Cheng XY, Su Y, et al. (2014) Elevated urinary levels of cystatin C and neutrophil gelatinase-associated lipocalin in Henoch-Schonlein purpura patients with renal involvement. PLoS One 9: e101026 doi: 10.1371/journal.pone.0101026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Du Y, Hou L, Guo J, Sun T, Wang X, eWu Y (2014) Renal neutrophil gelatinase-associated lipocalin and kidney injury molecule-1 expression in children with acute kidney injury and Henoch-Schonlein purpura nephritis. Exp Ther Med 7: 1130–1134. doi: 10.3892/etm.2014.1595 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.