Abstract
The largest and longest clinical trial of metformin for the prevention of diabetes is the Diabetes Prevention Program/Diabetes Prevention Program Outcomes Study (DPP/DPPOS). In this review, we summarise data from the DPP/DPPOS, focusing on metformin for diabetes prevention, as well as its long-term glycaemic and cardiometabolic effects and safety in people at high-risk of developing diabetes. The DPP (1996–2001) was a RCT of 3234 adults who, at baseline, were at high-risk of developing diabetes. Participants were assigned to masked placebo (n = 1082) or metformin (n = 1073) 850 mg twice daily, or intensive lifestyle intervention (n = 1079). The masked metformin/placebo intervention phase lasted 3.2 years, with primary outcome of diabetes incidence reported early, at 2.8 years, because of demonstrated efficacy. At the end of the DPP, all participants were offered lifestyle education and 88 % (n = 2776) of the surviving DPP cohort continued follow-up in the DPPOS. Participants originally assigned to metformin continued to receive metformin, unmasked. The DPP/DPPOS cohort has now been followed for over 15 years with prospective assessment of glycaemic, cardiometabolic, health economic, and safety outcomes. After an average follow-up of 2.8 years, metformin reduced the incidence of diabetes by 31 % compared with placebo, with a greater effect in those who were more obese, had a higher fasting glucose or a history of gestational diabetes. The DPPOS addressed the longer-term effects of metformin, showing a risk reduction of 18 % over 10 and 15 years post-randomisation. Metformin treatment for diabetes prevention was estimated to be cost-saving. At 15 years, lack of progression to diabetes was associated with a 28 % lower risk of microvascular complications across treatment arms, a reduction that was no different among treatment groups. Recent findings suggest metformin may reduce atherosclerosis development in men. Originally used for the treatment of type 2 diabetes, metformin, now proven to prevent or delay diabetes, may serve as an important tool in battling the growing diabetes epidemic. Long-term follow-up, currently underway in the DPP/DPPOS, is now evaluating metformin’s potential role, when started early in the spectrum of dysglycaemia, on later-stage comorbidities, including cardiovascular disease and cancer.
Keywords: Diabetes Prevention, DPP, DPPOS, Impaired glucose tolerance (IGT), Metformin, Prediabetes, Review
Introduction
The Diabetes Prevention Program (DPP; 1996–2001), an RCT to prevent or delay the onset of type 2 diabetes, was designed in the mid 1990s. Metformin was selected as one of the interventions, based on its mechanism of action and acceptable safety and tolerability profiles, with lifestyle intervention or placebo comprising the other treatment arms [1, 2]. The possibility of preventing or delaying diabetes in adults without diabetes but at high-risk had been hypothesised for decades. Small randomised clinical trials using type 2 diabetes treatment drugs (phenformin or tolbutamide) for diabetes prevention were performed in the 1960s/70s, but were inconclusive [3–5]. They were followed by larger clinical trials testing lifestyle interventions that proved to be effective [6, 7]. The DPP was the first major diabetes prevention trial using metformin [2].
The DPP/Diabetes Prevention Program Outcomes Study (DPPOS) represents the largest controlled clinical trial of metformin in a population at high-risk of developing diabetes, and also the longest trial of metformin for any indication. The effects of intensive lifestyle (ILS) intervention in the DPP and several other major trials, and the effects of other medications have been described elsewhere and are summarised in Table 1 [2, 8–10]. In this review, we focus on the effects of metformin on diabetes prevention, its long-term glycaemic and cardiometabolic effects, and its safety in the DPP/DPPOS.
Table 1.
Summary of select RCTs evaluating the prevention of progression to diabetes
| Study title (country of conduct, year of publication, n) | Risk eligibility criteria | Duration of follow-up | Intervention | Incidence/100 person-years (events) or cumulative incidence (%)at study end | Risk reduction in diabetes incidence compared with control/placebo |
|---|---|---|---|---|---|
| Da Qing Study [6] (China, 1997, n = 577) | IGT; age ≥ 25 years | 6.0 years | Diet | 10.0 | 31 % |
| Exercise | 8.3 | 46 % | |||
| Diet and exercise | 9.6 | 42 % | |||
| Control | 15.7 | – | |||
| Finnish Diabetes Prevention Study [7] (Finland, 2001, n = 522) | IGT; age 40–65 years; BMI > 25 kg/m2 | 3.2 years | Diet and activity | 3.2 | 58 % |
| Control | 7.8 | – | |||
| DPP [2] (USA, 2002, n = 3234) | IGT; FPG 5.3–6.9 mmol/l (<6.9 mmol/l for Native American ancestry); age ≥ 25 years; BMI ≥ 24 kg/m2 (≥ 22 kg/m2 in Asians) | 2.8 years | ILS | 4.8 | 58 % |
| Metformin 850 mg BID | 7.8 | 31 % | |||
| Placebo | 11.0 | – | |||
| STOP-NIDDM [41] (multiple countries, 2002, n = 1429) | IGT; FPG 5.6–7.7 mmol/l; age 40–70 years; BMI 25–40 kg/m2 | 3.3 years | Acarbose 100 mg TID | 10.1 | 25 % |
| Placebo | 12.1 | – | |||
| XENDOS [42] (Sweden, 2004, n = 3305) | BMI ≥ 30 kg/m2; age 30–60 years | 4.0 years | Orlistat 120 mg TID | 6.2 % | 37 % |
| Placebo | 9.0 % | – | |||
| Japanese IGT study [43] (Japan, 2005, n = 458) | Men with IGT | 4.0 years | Diet and exercise | 3.0 % | 67 % |
| Control | 9.3 % | – | |||
| Indian Diabetes Prevention Programme [44] (India, 2006, n = 531) | IGT; age 35–55 years | 30 months | Lifestyle modification | 39.3 % | 29 % |
| Metformin 250 mg BID | 40.5 % | 26 % | |||
| Lifestyle modification + metformin 250 mg BID | 39.5 % | 28 % | |||
| Control | 55.0 % | – | |||
| DREAM (rosiglitazone) [45] (multiple countries, 2006, n = 5269) | IFG and/or IGT; age ≥ 30 years | 3.0 years | Rosiglitazone 8 mg daily | 11.6 % | 60 %a |
| Placebo | 26.0 % | – | |||
| DREAM (ramipril) [46] (multiple countries, 2006, n = 5269) | IFG and/or IGT; age ≥ 30 years | 3.0 years | Ramipril, up to 15 mg per day | 18.1 % | Nonea |
| Placebo | 19.5 % | – | |||
| Voglibose Ph-3 [47] (Japan, 2009, n = 1780) | IGT; age 30–70 years; with additional risk factor for T2D | 48.1 weeks | Voglibose 0.2 mg TID | 3.6 % | 41 % |
| Placebo | 9.4 % | – | |||
| NAVIGATOR (valsartan) [48] (multiple countries, 2010, n = 9306) | IGT; FPG 5.3–< 7.0 mmol/l; with CVD/CVD risk | 5.0 years | Valsartan, up to 160 mg daily, and lifestyle modification instruction | 7.7 | 14 % |
| Placebo | 9.0 | – | |||
| NAVIGATOR (nateglinide) [49] (multiple countries, 2010, n = 9306) | IGT; FPG 5.3–< 7.0 mmol/l; with CVD/CVD risk | 5.0 years | Nateglinide, 60 mg before meals, TID | 8.6 | None |
| Placebo | 8.0 | – | |||
| CANOE [50] (Canada, 2010, n = 207) | IGT; age 30–75 years (18–75 for Native Canadian ancestry); with at least one risk factor for T2D | 3.9 years | Rosiglitazone + metformin (2 mg/500 mg, BID) | 13.6 % | 66 % |
| Placebo | 39.4% | – | |||
| ACT NOW [51] (USA, 2011, n = 602) | IGT; FPG 5.3–6.9 mmol/l; age ≥18 years; BMI ≥ 25 kg/m2; at least one risk factor T2D | 2.4 years | Pioglitazone 45 mg daily | 2.1 | 72 % |
| Placebo | 7.6 | – | |||
| SCALE Prediabetes [52] (multiple countries, 2017, n = 2254) | Prediabetesb; age ≥18 years; BMI ≥ 30 kg/m2 or ≥ 27 kg/m2 with comorbidities | 3.0 years | Liraglutide 3.0 mg | 3.0 % | 66 % |
| Placebo | 11.0 % | – |
Table includes RCTs studying progression to diabetes as a primary outcome with interventions that are currently available. Refer to original referenced studies for details on outcomes measured and reported.
Composite primary outcome of incident diabetes or death from any cause
Prediabetes was defined as fulfilment of at least one of the three ADA 2010 criteria: 5.7–6.4% HbA1c; FPG between 5.6–6.9 mmol/l; or 2-h post-challenge plasma glucose concentration between 7.8–11.0 mmol/l
ACT NOW, Actos Now for the prevention of diabetes; BID, twice daily; CANOE, CAnadian Normoglycemia Outcomes Evaluation; DREAM, Diabetes REduction Assessment with ramipril and rosiglitazone Medication; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; NAVIGATOR, Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research; SCALE, Satiety and Clinical Adiposity — Liraglutide Evidence in Nondiabetic and Diabetic Individuals; STOP-NIDDM, Study to Prevent Non-Insulin Dependent Diabetes Mellitus; T2D, type 2 diabetes; TID, thrice daily; XENDOS, XENical in the prevention of Diabetes in Obese Subjects
Overview of the DPP/DPPOS
Design of the DPP/DPPOS
The DPP enrolled 3234 participants aged 25 years or older who were at high-risk of developing diabetes, defined as impaired glucose tolerance, with elevated fasting plasma glucose (FPG) (5.3–6.9 mmol/l [≤6.9 mmol/l in Native Americans]) and a BMI of 24 kg/m2 or higher (≥ 22 kg/m2 in Asian-Americans). Participants were randomly assigned to placebo (n = 1082), metformin (n = 1073) titrated to 850 mg twice daily, or ILS intervention (n = 1079), which aimed for 7 % weight loss through a low-energy, low-fat diet (based on recommendations for health) and ≥ 150 min/week of moderate-intensity physical activity [2]. Diagnosis of diabetes was based on annual OGTTs or semi-annual FPG tests, using the ADA diagnostic criteria, with the diagnosis requiring confirmation with repeat testing [11]. Diagnosis of diabetes and FPG 7.8 mmol/l resulted in discontinuation of study medication and referral to the participant’s own physician for further treatment [2].
The DPP was stopped in 2001, 1 year ahead of schedule, owing to demonstrated efficacy of both metformin and the lifestyle intervention [2]. Given the demonstrated effects of ILS, all participants were offered a group-administered version of the lifestyle curriculum at the end of the DPP. Eighty-eight per cent (n = 2776) of eligible DPP participants continued follow-up in the DPPOS, in which placebo was discontinued, those previously assigned to metformin received metformin 850 mg twice daily (now unmasked) and lifestyle messages were intermittently reinforced. The study-provided metformin was discontinued if there were safety concerns, or if diabetes was diagnosed and HbA1c was ≥ 7 % (≥ 53 mmol/mol), hence requiring management by the participant’s physician [8]. Outcomes in the DPPOS from 2002–2013 centred on the long-term effects of the interventions on diabetes prevention, diabetes-associated microvascular complications [9] and cardiovascular disease (CVD) risk factors.
Participant characteristics
By intention, the DPP enrolled a heterogeneous population, with 45 % from racial or ethnic minorities, 20 % aged 60 years or older and 68 % women, including 350 women with a history of gestational diabetes (GDM). The mean age at randomisation was 51 years and mean BMI was 34 kg/m2. Mean FPG was 5.9 mmol/l and baseline HbA1c was 5.9 % (41 mmol/mol). Sex, ethnic distribution and risk factors for diabetes were similar among the randomised treatment arms [2].
Exposure to metformin
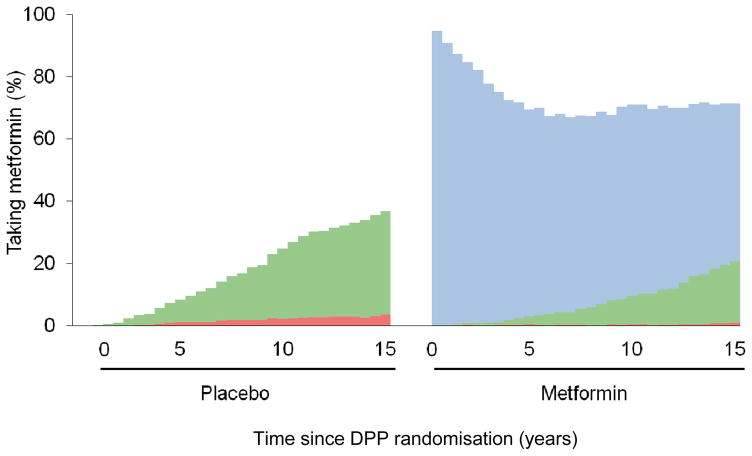
Integral to understanding the effects of metformin over time is the separation of study and non-study exposure, keeping in mind that DPP/DPPOS participants who developed diabetes were subsequently managed by their own physicians, often with non-study metformin. By 15 years after randomisation, 37% of the original placebo participants had been treated with metformin by their healthcare providers, the vast majority associated with diabetes diagnosis (Fig. 1). The mean exposure, including study- and non-study metformin, from 1996–2013, remained widely separated, at 10.7 vs 2.3 metformin-years in metformin vs placebo groups [9].
Fig. 1.
Exposure to metformin throughout the DPP/DPPOS. AUCs represent total metformin-years of exposure, including study-provided metformin (blue), non-study-provided metformin for diabetes treatment (green), and non-study-provided metformin prescribed to individuals without diabetes (red)
Throughout the DPP and DPPOS, pill counts and structured interviews were used to promote adherence [12]. During the DPP, adherence to metformin, defined as taking at least 80 % of assigned study pills, was 72 % (Fig. 1) [2]. An additional 10–15 % of participants took some metformin, albeit at less than 80 % of pills assigned. Adherence to metformin (at the > 80 % threshold) fell to an average of 49 % over the DPPOS (2002–2013) [9].
Results
Effects of metformin on diabetes prevention
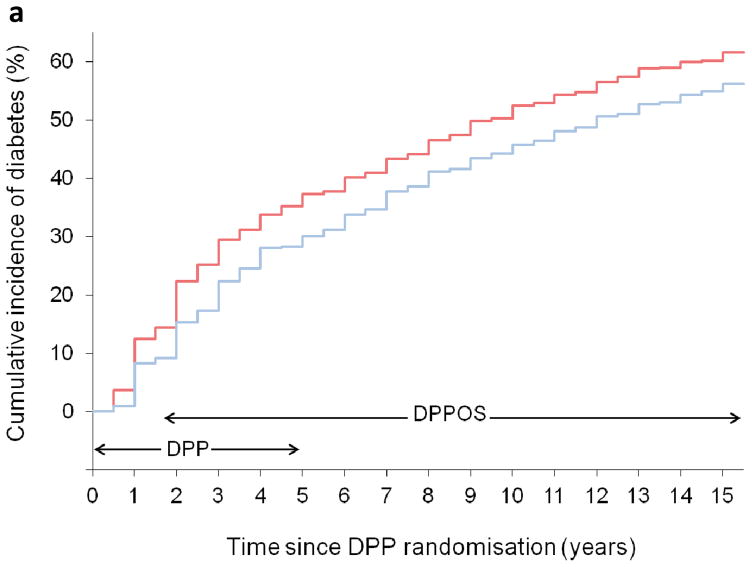
In 2002, the DPP published its primary findings from the masked-treatment phase, showing that the ILS and metformin groups had a respective 58 % and 31 % lower incidence of diabetes than the placebo group [2]. Subsequently, the DPPOS addressed the longer-term effects of metformin, showing a decline in risk reduction by 18% compared with placebo over 10 and 15 years post-randomisation (Fig. 2a) [8, 9]. Although the differences in incidence rates over the entire follow-up remained significant, the observed diabetes incidence rates during the DPPOS period (i.e. in the period after the DPP completed) were not significantly different between the formerly randomised groups. Diabetes incidence rates during the DPP were 7.8 cases per 100 person-years in the metformin group and 11.0 cases per 100 person-years in the placebo group [2], and these decreased in the DPPOS (2002–2008) to 4.9 cases per 100 person-years for metformin and 5.6 cases per 100 person-years for placebo [8], remaining stable thereafter. This reduced diabetes incidence approximates the 5 cases per 100 person-years rate observed in the lifestyle group during the DPP, which has remained nearly constant throughout the DPP/DPPOS [9]. The average genetic risk score, derived from 34 type 2 diabetes-associated genetic variants, declined over time among participants who remained without diabetes in the DPP/DPPOS, in both the metformin and placebo groups [13]. This suggests that the lower annual incidence rate of diabetes seen in the DPPOS was not entirely due to an effect of the lifestyle intervention offered during the transition to the DPPOS, but, in part, due to ‘exhaustion of susceptibles’, or that diabetes developed in the people who were most susceptible to diabetes during the DPP and that remaining participants in the DPPOS were less susceptible to diabetes [13].
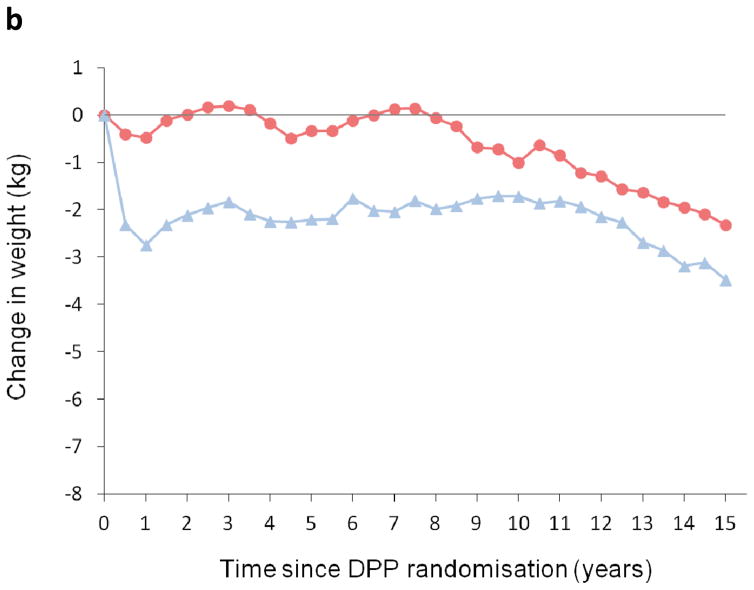
Fig. 2.
(a) Cumulative incidence of diabetes and (b) weight change over 15 years in the DPP/DPPOS, in metformin (blue line) and placebo (red line) groups. (a) Adapted from The Lancet Diabetes & Endocrinology 3:866–875 [9]; Diabetes Prevention Program Research Group, Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the Diabetes Prevention Program Outcomes Study, Copyright (2015), with permission from Elsevier
Effects of metformin on diabetes prevention/delay in subgroups of interest
The DPP was not powered to assess the significance of effects within subgroups. Nonetheless, examination of treatment effects in cohort subgroups revealed significant heterogeneity. For example, obese participants with a BMI ≥ 35 kg/m2 were more responsive to metformin than to placebo, with a 53 % risk reduction for diabetes and only a 3 % reduction in those with BMI 22 to <30 kg/m2. In addition, those with a higher fasting glucose (6.1–6.9 mmol/l) had a greater risk reduction with metformin (48 %) compared with those with a fasting glucose of 5.3–6.1 mmol/l (15 % risk reduction). Although not significant for heterogeneity, metformin appeared more effective in younger participants compared with placebo, reducing diabetes onset by 44 % in those 25–44 years old vs 11 % for those ≥ 60 years of age at study entry. Of note, no such differences were observed by sex, race/ethnicity, or tertiles of baseline 2-h plasma glucose (2-hPG) [2].
During the DPP, women with a history of GDM randomised to placebo had a 71 % higher risk of diabetes than parous women without such a history, despite similar FPG and 2-hPG values at baseline [14]. Significant heterogeneity was observed in response to metformin with a 50 % reduction in incidence of diabetes in women with a history of GDM compared with 14 % in parous women with no such history. Ten-year follow-up in the DPPOS confirmed these effects, demonstrating a sustained and relatively greater risk for diabetes in women with a history of GDM, which was reduced by 40 % with metformin [15].
Insights from the DPP/DPPOS on how metformin prevents or delays diabetes
Acute pharmacologic effect or amelioration of pathophysiology?
During the DPP, evaluations were carried out without interruption of study medication (placebo or metformin), except for withholding study medicine the morning of glycaemic testing. Thus, some (or all) of metformin’s effect could have been a transient pharmacological treatment effect (‘masking of diabetes’), rather than a true delay in the onset of diabetes. The DPP addressed this issue by retesting participants who had not developed diabetes, 1–2 weeks after stopping metformin. After this washout period, the incidence of diabetes was still reduced by 25 %, compared with the 31 % reduction seen in the primary analysis, suggesting a more durable effect of metformin treatment on glucose metabolism [16].
Explanation of metformin-induced effects
Some of metformin’s diabetes prevention effect is attributed to weight loss, which was durable over time in the DPP/DPPOS (Fig. 2b, Table 2) [2, 8, 9]. Weight loss with metformin explained 64 % of the drug’s beneficial effect on diabetes risk at the end of the DPP [17]. Favourable changes were also seen in other measures of adiposity (waist circumference, waist-to-hip ratio), and in fasting insulin and proinsulin [17]. No differences were seen in self-reported physical activity or diet, or insulin secretion measured by the insulinogenic index between the metformin and placebo groups. While no single covariate completely explained the beneficial effect of metformin vs placebo, the combination of weight, fasting insulin and proinsulin levels, and other metabolic factors explained 81 % of the beneficial outcomes with use of this drug [17]. Improvements in FPG and estimated insulin sensitivity with metformin may be owing to a combination of weight loss and other direct effects of the drug on the liver and, perhaps, other tissues.
Table 2.
Effect of metformin on diabetes risk and CVD risk factors at baseline and at the end of each phase of the DPP and DPPOS
| Characteristics | Baseline (1996–1999) | DPP (1996–2001) 3.2 years mean follow-upa |
DPPOS 1 (2002–2008) 10 years mean follow-up |
DPPOS 2 (2008–2013) 15 years mean follow-up |
||||
|---|---|---|---|---|---|---|---|---|
| Placebo n = 1082 | Metformin n = 1073 | Placebo n = 935 | Metformin n = 926 | Placebo n = 924 | Metformin n = 932 | Placebo n = 924 | Metformin n = 932 | |
| Anthropometrics | ||||||||
| Weight (kg) | 94.3 | 94.3 | 94.1 | 92.0* | 93.2 | 91.8* | 91.0 | 89.5 |
| BMI (kg/m2) | 34.2 | 33.9 | 33.9 | 33.2* | 33.6 | 33.1* | 33.1 | 32.3* |
| Diabetes | ||||||||
| Diabetes cases | 0 | 0 | 278 | 199* | 450 | 387* | 564 | 506* |
| Mean diabetes duration (years among cases) | – | – | 1.5 | 1.5 | 5.3 | 4.9* | 10.3 | 9.7 |
| FPG (mmol/l) | 5.9 | 5.9 | 6.2 | 5.9* | 6.5 | 6.2 | 6.8 | 6.5* |
| HbA1c (%) | 5.91 | 5.91 | 6.08 | 5.97* | 6.02 | 5.92 | 6.27 | 6.11* |
| HbA1c (mmol/mol) | 41 | 41 | 43 | 42* | 42 | 41 | 45 | 43* |
| CVD risk factors | ||||||||
| Systolic BP (mmHg) | 124 | 124 | 123 | 123 | 121 | 121 | 121 | 121 |
| Diastolic BP (mmHg) | 78 | 78 | 76 | 76 | 73 | 73 | 71 | 71 |
| LDL-c (mmol/l) | 3.2 | 3.2 | 3.2 | 3.2 | 2.7 | 2.7 | 2.5 | 2.5 |
| HDL-c (mmol/l) | 1.17 | 1.19 | 1.17 | 1.19 | 1.32 | 1.32 | 1.38 | 1.42 |
| Triacylglycerol (mmol/l) | 1.66 | 1.57 | 1.51 | 1.53 | 1.40 | 1.41 | 1.51 | 1.53 |
| CRP (nmol/l) | 33.52 | 31.8 | 32.76 | 28.38* | 24.95 | 20.48* | – | – |
| tPA (ng/ml) | 11.4 | 11.3 | 11.8 | 10.7* | – | – | – | – |
| Fibrinogen (μmol/l) | 386 | 380 | 387 | 383 | 457 | 445* | – | – |
| Coronary calcification (%)b | ||||||||
| Men | – | – | – | – | – | – | 84 | 75* |
| Women | – | – | – | – | – | – | 50 | 53 |
Data shown as means or n (%)
DPP intervention phase was 3.2 years with primary diabetes incidence analysis completed at 2.8 years owing to demonstrated efficacy.
Based on scan measured at DPPOS year 10, with 14 years of average follow-up
p < 0.05, metformin vs placebo
CRP, C-reactive protein; HDL-c, HDL-cholesterol; LDL-c, LDL-cholesterol; tPA, tissue plasminogen activator
Effects of metformin on blood glucose measures
The effects of the DPP interventions on FPG and HbA1c were examined in all participants, regardless of whether they had developed diabetes. During the DPP, metformin and ILS were similarly effective in restoring normal FPG values [2]. Despite metformin and ILS having similar effects on FPG, diabetes incidence was more significantly reduced by ILS than by metformin, reflecting the fact that most diabetes diagnoses in the DPP were triggered by the 2-hPG rather than FPG, and that ILS was more effective than metformin at restoring a normal 2-hPG. This latter observation was likely because, while both active interventions improved beta cell function, this effect was greater with ILS [18]. Consistent with metformin’s known ability to suppress hepatic glucose production during fasting [19, 20], its reduction of diabetes incidence compared with placebo was much greater in those entering the study with a FPG 6.1–6.9 mmol/l than in those with a FPG 5.3–6.1 mmol/l [2]. Metformin also lowered HbA1c relative to placebo, but to a lesser extent than did ILS [2].
After the DPP had been completed, an International Expert Committee and the ADA expanded the diagnostic criteria for diabetes to include HbA1c ≥ 6.5 % (≥ 48 mmol/mol) [21, 22]. Although HbA1c was measured during the DPP/DPPOS, eligibility and diabetes diagnoses were based on fasting and/or 2-hPG. Thus, a secondary analysis using HbA1c ≥ 6.5% (≥ 48 mmol/mol) as an alternative definition of diabetes was performed, excluding the 13 % of participants with HbA1c ≥6.5 % (≥48 mmol/mol) at study entry. Although ILS was more effective than metformin in reducing the incidence of diabetes defined by the FPG and OGTT criteria, the effect of metformin was no longer significantly different from ILS when diabetes was diagnosed based on HbA1c (44 % vs 49 % reduction in the DPP, 38 % vs 29 % reduction throughout DPP/DPPOS; metformin vs ILS) [23]. In summary, metformin was as effective as ILS in preventing diabetes by some measures (i.e. HbA1c), but not by 2-hPG, in the DPP/DPPOS population.
Metformin’s interaction with genetic factors
The DPP investigated several genetic variants previously associated with risk of type 2 diabetes or metformin action. For example, homozygosity for the major diabetes risk variant rs7903146 in the TCF7L2 gene was associated with an 81 % higher diabetes incidence in the placebo group that was reduced to a 62 % increased risk in the metformin group [24]. In addition, a genetic risk score predicted diabetes incidence in the DPP, but with no significant interaction between the score and treatment group. That is, the interventions were equally effective, regardless of genetic susceptibility [25]. There was, however, a nominal interaction with metformin (p = 0.006) with the variant rs8065082 in the metformin transporter gene SLC47A1, with the minor allele being associated with lower incidence of diabetes in the metformin arm (HR 0.78 [95% CI 0.64, 0.96]; p = 0.02) [26].
Effects of metformin on microvascular complications
At the end of the DPP, the only microvascular outcome assessed was microalbuminuria. There was no effect of treatment intervention on the percentage of participants with elevated albumin:creatinine ratio (ACR) levels, although those who developed diabetes had a 59 % increased risk of developing an elevated ACR (≥ 3.39 mg/mmol) [27]. One of the main goals in the longer-term follow-up of the DPPOS is to determine if treatments effective in preventing diabetes also affect the development of microvascular complications, specifically retinopathy, nephropathy and neuropathy. A composite of these microvascular outcomes at 15 years in the DPPOS was 28 % less frequent in those who did not progress to diabetes, but there was no difference between the original treatment arms [9]. The very small difference in HbA1c levels among the treatment groups, limited power, and early referral to care providers for treatment of hypertension and dyslipidaemia have been considered reasons for the lack of an effect of the active treatments on microvascular outcomes thus far, despite the reduction in diabetes incidence [9]. It is still possible that treatment effects may emerge with longer follow-up and longer diabetes duration in the cohort.
Effects of metformin on cardiovascular disease risk factors
In the DPP, metformin had favourable effects on several cardiovascular risk factors, including lipoprotein subfractions [28], C-reactive protein and tissue plasminogen activator [29]. It also reduced the incidence of the metabolic syndrome by 17 % compared with placebo [30]. No significant effects were seen with lipid levels or blood pressure [31] (Table 2). Over longer-term follow-up (10 years), no significant differences in traditional cardiovascular disease (CVD) risk factors have been noted between the metformin and placebo groups [32] (Table 2).
An average of 14 years after randomisation, subclinical atherosclerosis was assessed in 2029 participants using coronary artery calcium (CAC) measurements, according to the original randomisation group. There was a significant interaction between sex and the effects of metformin vs placebo on CAC presence (p = 0.01) and CAC severity (p = 0.08). Compared with placebo, metformin significantly lowered the presence and severity of CAC in men, with no effect in women. Of interest, no reduction in the prevalence of clinically significant plaque (Agatston score >100) was observed, suggesting the possibility that metformin affects smaller, more recently calcified plaques, rather than well-established plaques. There was no difference in CAC between ILS and placebo groups, suggesting a possible long-term differentiation between metformin and ILS [33]. Longer-term follow-up with ascertainment of CVD outcomes is underway.
Long-term safety and tolerability of metformin in the DPP/DPPOS
The long-term use of metformin within the context of a closely-monitored clinical trial has provided additional information on metformin safety and tolerability. Minor gastrointestinal symptoms were reported by 9.5 % of those randomised to metformin, compared with 1.1 % in the placebo group, but these were generally mild and tended to wane over time [34]. The risk of lactic acidosis with metformin use has recently been shown to be much lower than previously suspected [35] and there have been no reported cases of lactic acidosis in over 15,000 person-years of exposure to metformin in the DPP/DPPOS.
Metformin use has been associated with impaired intestinal absorption of vitamin B12 and increased risk of vitamin B12 deficiency. This risk was recognised in the design of the DPP and annual testing was performed to detect anaemia as a potential manifestation of low vitamin B12 levels. In addition, vitamin B12 levels were directly measured at two time points in the DPPOS. Biochemical vitamin B12 deficiency levels (<150 pmol/l) occurred more often in individuals in the metformin group than the placebo group at 5 years (4.3 % vs 2.3 %; p = 0.02); a similar pattern was observed but was not significant at 13 years (7.4 % vs 5.4 %; p = 0.12) [36]. Low or ‘borderline’ vitamin B12 (defined as levels <220 pmol/l) is accepted by some as evidence of inadequate vitamin B12 stores and was more common in those in the metformin group at 5 years vs placebo (19.1 % vs 9.5 %; p = 0.01) and 13 years (20.3 % vs 15.6 %; p = 0.02). In a multivariate model, years of metformin use, including metformin prescribed outside of the study, were associated with increased risk of vitamin B12 deficiency with the odds ratio for vitamin B12 < 150 pmol/l per year of metformin use being 1.13 (95% CI 1.06, 1.20). Anaemia prevalence was higher in the metformin group but, importantly, did not differ by vitamin B12 level, suggesting that haematological monitoring may not be sufficient to detect metformin-associated vitamin B12 deficiency [36]. Given these findings in this large cohort, current guidelines now recommend consideration of periodic measurement of vitamin B12 levels and supplementation as needed in patients treated with metformin [37].
Looking to the Future
The impact of prediabetes and diabetes worldwide is enormous, with 415 million adults currently having diabetes and a projected increase to 642 million by 2040 [38]. Both lifestyle intervention and metformin are effective in the prevention or delay of diabetes. Originally used for the treatment of type 2 diabetes, metformin, now proven to prevent or delay diabetes, may serve as an important additional tool in battling the growing diabetes epidemic. As detailed in this review, metformin had sustained benefit in preventing/delaying diabetes for at least 15 years. Further, while lifestyle intervention was uniformly effective across subgroups [2], the DPP identified significant benefit from metformin in those who were more obese, had a higher fasting glucose or a history of GDM, and a suggestion of greater effect than lifestyle intervention in those who were younger. Although not specific to treatment assignment, lack of progression to diabetes was associated with lower risk of microvascular complications [9] and, among men, metformin reduced atherosclerosis development [33]. Furthermore over 10 years, metformin treatment was estimated to be cost-saving, decreasing the cumulative costs of medical care received outside the DPP/DPPOS, compared with placebo [39]. Guidelines consistently recommend either lifestyle intervention or metformin therapy for the prevention of diabetes, with considerations for metformin in subgroups in which this drug was identified to have a relatively greater effect in the DPP [37, 40]. Given our current understanding of the beneficial effects of metformin to prevent or delay diabetes, a concerted global effort to translate this evidence may help redirect the continuing increase in the prevalence of type 2 diabetes.
The potential for additional benefits of metformin extends beyond diabetes prevention and represents the next phase of study for the DPPOS. As the largest and longest clinical trial of metformin treatment, uniquely in a population initially without diabetes, the DPP/DPPOS is now poised to evaluate whether starting metformin early in high-risk individuals impacts the development and risk for even later-stage comorbidities, notably CVD and cancer. Although decreasing the incidence of diabetes would be expected to decrease CVD risk, the effect of metformin and diabetes delay/prevention on CVD is unproven. In addition, based on experimental and epidemiological data, metformin has recently received attention as a potential anti-cancer agent. Prospective intervention studies with treatment of long duration and follow-up are needed to address these important questions. DPP/DPPOS, with over 15 years of randomised metformin experience, now aims to address this need.
In conclusion, the DPP/DPPOS clearly demonstrated a role for metformin in the prevention of diabetes. Looking to the future, understanding whether translation of these findings into routine clinical care improves current trends in the development of diabetes is of critical importance. The possibility that metformin can further impact additional complications of dysglycaemia that have not yet been investigated remains an exciting area of study.
Supplementary Material
Summary of metformin benefits in diabetes prevention.
Incidence of diabetes
31% reduction in diabetes incidence (DPP); longer-term (over 10 and 15 years) risk reduced by 18% (DPPOS). Heterogeneity in response, e.g. more effective in obesity, in those with high FBG, in women with history of GDM.
Metabolic factors
Metformin-induced weight loss explained 64 % of beneficial effects on diabetes risk (DPP); favourable changes in waist circumference, waist-to-hip ratio and fasting insulin/proinsulin.
Blood glucose
Restores normal FPG values; improves beta cell function; suppresses hepatic glucose production during fasting; lowers HbA1c.
Microvascular complications
No effects on microvascular complications have been observed; treatment effects may emerge with longer follow-up.
CVD risk factors
Favourable effects on lipoprotein subfractions, C-reactive protein and tissue plas-minogen activator (DPP); reduced incidence of the metabolic syndrome (17 %) vs placebo; no effect on lipids or BP. Longer-term follow-up (10 years), revealed no difference in traditional CVD risk factors vs placebo; 14 years after randomisation, significantly lowered presence and severity of CAC in men vs placebo (no effect in women), but no reduction in the prevalence of clinically significant plaque.
Acknowledgments
The Research Group gratefully acknowledges the commitment and dedication of the participants of the DPP and DPPOS. A complete list of Centers, investigators, and staff can be found in the electronic supplementary material (ESM). All members of the Steering Committee had input into the report’s contents. All authors in the writing group had access to all data.
Funding During the DPP and DPPOS, the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health provided funding to the clinical centers and the Coordinating Center for the design and conduct of the study, and collection, management, analysis, and interpretation of the data (U01 DK048489). The Southwestern American Indian Centers were supported directly by the NIDDK, including its Intramural Research Program, and the Indian Health Service. The General Clinical Research Center Program, National Center for Research Resources, and the Department of Veterans Affairs supported data collection at many of the clinical centers. The sponsor of the DPP/DPPOS study was represented on the Steering Committee and played a part in study design, how the study was done, and publication. Funding was also provided by the National Institute of Child Health and Human Development, the National Institute on Aging, the National Eye Institute, the National Heart Lung and Blood Institute, the Office of Research on Women’s Health, the National Center for Minority Health and Human Disease, the National Cancer Institute, the Centers for Disease Control and Prevention, and the American Diabetes Association. Bristol-Myers Squibb and Parke-Davis provided additional funding and material support during the DPP, Lipha (Merck-Sante) provided medication and LifeScan Inc. donated materials during the DPP and DPPOS. The opinions expressed are those of the investigators and do not necessarily reflect the views of the funding agencies. This material should not be interpreted as representing the viewpoint of the US Department of Health and Human Services, the National Institutes of Health, or the National Cancer Institute.
Abbreviations
- ACR
Albumin: creatinine ratio
- CAC
Coronary artery calcium
- CVD
Cardiovascular disease
- DPP
Diabetes Prevention Program
- DPPOS
Diabetes Prevention Program Outcomes Study
- FPG
Fasting plasma glucose
- GDM
Gestational diabetes
- 2-hPG
2-h plasma glucose
- ILS
Intensive lifestyle
DPPOS Research Group Investigators
-
Pennington Biomedical Research Center(Baton Rouge, LA)
George A. Bray, MD*
Kishore Gadde, MD*
Annie Chatellier, RN, CCRC**
Jennifer Arceneaux RN, BSN**
Amber Dragg RD, LDN**
Crystal Duncan, LPN
Frank L. Greenway, MD
Daniel Hsia, MD
Erma Levy, RD
Monica Lockett, LPN
Donna H. Ryan, MD
-
University of Chicago(Chicago, IL)
David Ehrmann, MD*
Margaret J. Matulik, RN, BSN**
Kirsten Czech, MS
Catherine DeSandre, BA
-
Jefferson Medical College(Philadelphia, PA)
Barry J. Goldstein, MD, PhD*
Kevin Furlong, DO*
Kellie A. Smith, RN, MSN**
Wendi Wildman, RN**
Constance Pepe, MS, RD
-
University of Miami(Miami, FL)
Ronald B. Goldberg, MD*
Jeanette Calles, MSEd**
Juliet Ojito, RN**
Sumaya Castillo-Florez, MPH
Hermes J. Florez, MD, PhD
Anna Giannella, RD, MS
Olga Lara
Beth Veciana
-
The University of Texas Health Science Center(San Antonio, TX)
Steven M. Haffner, MD, MPH*
Helen P. Hazuda, PhD*
Maria G. Montez, RN, MSHP, CDE**
Kathy Hattaway, RD, MS
Carlos Lorenzo, MD, PhD
Arlene Martinez, RN, BSN, CDE
Tatiana Walker, RD, MS, CDE
-
University of Colorado(Denver, CO)
Richard F. Hamman, MD, DrPH*
Dana Dabelea, MD, PhD*
Lisa Testaverde, MS**
Denise Anderson, RN, BSN
Alexis Bouffard, MA, RN, BSN
Tonya Jenkins, RD, CDE
Dione Lenz, RN, BSN, CDE
Leigh Perreault, MD
David W. Price, MD
Sheila C. Steinke, MS
-
Joslin Diabetes Center(Boston, MA)
Edward S. Horton, MD*
Catherine S. Poirier, RN, BSN**
Kati Swift, RN, BSN**
Enrique Caballero, MD
Barbara Fargnoli, RD
Ashley Guidi, BS
Mathew Guido, BA
Sharon D. Jackson, MS, RD, CDE
Lori Lambert, MS, RD, LD
Kathleen E. Lawton, RN
Sarah Ledbury, Med, RD
Jessica Sansoucy, BS
Jeanne Spellman, RD
-
VA Puget Sound Health Care System and University of Washington(Seattle, WA)
Steven E. Kahn, MB, ChB*
Brenda K. Montgomery, RN, BSN, CDE**
Wilfred Fujimoto, MD
Robert H. Knopp, MD
Edward W. Lipkin, MD
Ivy Morgan-Taggart
Anne Murillo, BS
Lonnese Taylor, RN, BS
April Thomas, RD, MPH, CDE
Elaine C. Tsai, MD, MPH
Dace Trence, MD
-
University of Tennessee(Memphis, TN)
Abbas E. Kitabchi, PhD, MD, FACP*
Samuel Dagogo-Jack, MD*
Mary E. Murphy, RN, MS, CDE, MBA**
Laura Taylor, RN, BSN, CDE**
Jennifer Dolgoff, RN, BSN**
Debra Clark, LPN
Uzoma Ibebuogu, MD
Helen Lambeth, RN, BSN
Harriet Ricks
Lily M.K. Rutledge, RN, BSN
Judith E. Soberman, MD
-
Northwestern University’s Feinberg School of Medicine(Chicago, IL)
Mark E. Molitch, MD*
Boyd E. Metzger, MD*
Mariana K. Johnson, MS, RN**
Mimi M. Giles, MS, RD
Diane Larsen, BS
Samsam C. Pen, BA
-
Massachusetts General Hospital(Boston, MA)
David M. Nathan, MD*
Mary Larkin, MSN*
Charles McKitrick, BSN**
Heather Turgeon, BSN**
Ellen Anderson, MS, RD
Laurie Bissett, MS, RD
Kristy Bondi, BS
Enrico Cagliero, MD
Kali D’Anna
Linda Delahanty, MS, RD
Jose C. Florez, MD, PhD
Valerie Goldman, MS, RD
Peter Lou, MD
Alexandra Poulos
Elyse Raymond, BS
Christine, Stevens
Beverly Tseng
-
University of California-San Diego(San Diego, CA)
Elizabeth Barrett-Connor, MD*
Mary Lou Carrion-Petersen, RN, BSN**
Lauren N. Claravall, BS
Jonalle M. Dowden, BS
Javiva Horne, RD
Diana Leos, RN, BSN
Sundar Mudaliar, MD
Jean Smith, RN
Simona Szerdi Janisch, BS
Karen Vejvoda, RN, BSN, CDE, CCRC
-
Columbia University(New York, NY)
F. Xavier Pi-Sunyer, MD*
Jane E. Lee, MS**
Sandra T. Foo, MD
Susan Hagamen, MS, RN, CDE
-
Indiana University(Indianapolis, IN)
David G. Marrero, PhD*
Kieren J. Mather, MD*
Susie M. Kelly, RN, CDE**
Paula Putenney, RN**
Marcia A. Jackson**
Gina McAtee**
Ronald T. Ackermann, MD
Carolyn M. Cantrell
Edwin S. Fineberg, MD
Angela Hadden (deceased)
Mario S. Kirkman
Erin O’Kelly Phillips
Paris J. Roach, MD
-
Medstar Health Research Institute(Washington, DC)
Robert E. Ratner, MD*
Vanita Aroda, MD*
Sue Shapiro, RN, BSN, CCRC**
Catherine Bavido-Arrage, MS, RD, LD
Peggy Gibbs
Gabriel Uwaifo, MD
Renee Wiggins, RD
-
University of Southern California/UCLA Research Center(Alhambra, CA)
Mohammed F. Saad, MD*
Karol Watson, MD*
Medhat Botrous, MD**
Sujata Jinagouda, MD**
Maria Budget
Claudia Conzues
Perpetua Magpuri
Kathy Ngo
Kathy Xapthalamous
-
Washington University(St. Louis, MO)
Neil H. White, MD, CDE*
Angela L. Brown, MD*
Samia Das, MS, MBA, RD, LD**
Prajakta Khare-Ranade, MSc, RDN, LD**
Tamara Stich, RN, MSN, CDE**
Ana Santiago, RN
Cormarie Wernimont, RD, LD
-
Johns Hopkins School of Medicine (Baltimore, MD)
Christopher D. Saudek, MD* (deceased)
Sherita Hill Golden, MD, MHS, FAHA*
Tracy Whittington, BS**
Frederick L. Brancati, MD, MHS (deceased)
Jeanne M. Clark, MD
Alicia Greene
Dawn Jiggetts
Henry Mosley
John Reusing
Richard R. Rubin, PhD (deceased)
Shawne Stephens
Evonne Utsey
-
University of New Mexico(Albuquerque, NM)
David S. Schade, MD*
Karwyn S. Adams, RN, MSN**
Claire Hemphill, RN, BSN**
Penny Hyde, RN, BSN**
Janene L. Canady, RN, CDE**
Kathleen Colleran, MD
Ysela Gonzales, RN, MSN
Doris A. Hernandez-McGinnis
Carolyn King
-
Albert Einstein College of Medicine(Bronx, NY)
Jill Crandall, MD*
Janet O. Brown, RN, MPH, MSN**
Gilda Trandafirescu, MD**
Elsie Adorno, BS
Helena Duffy, MS, C-ANP
Angela Goldstein, FNP-C, NPP, CSW
Jennifer Lukin, BA
Helen Martinez, RN, MSN, FNP-C
Dorothy Pompi, BA
Harry Shamoon, MD
Jonathan Scheindlin, MD
Elizabeth A. Walker, RN, DNSc, CDE
Judith Wylie-Rosett, EdD, RD
-
University of Pittsburgh(Pittsburgh, PA)
Trevor Orchard, MD*
Andrea Kriska, PhD*
Susan Jeffries, RN, MSN**
M. Kaye Kramer, BSN, MPH**
Marie Smith, RN, BSN**
Catherine Benchoff
Stephanie Guimond, BS
Jessica Pettigrew, CMA
Debra Rubinstein, MD
Linda Semler, MS, RD
Elizabeth Venditti, PhD
Valarie Weinzierl, MPH
-
University of Hawaii(Honolulu, HI)
Richard F. Arakaki, MD*
Narleen K. Baker-Ladao, BS**
Mae K. Isonaga, RD, MPH**
Nina E. Bermudez, MS
Marjorie K. Mau, MD
John S. Melish, MD
Robin E. Yamamoto, CDE, RD
-
Southwest American Indian Centers(Phoenix, AZ; Shiprock, NM; Zuni, NM)
William C. Knowler, MD, DrPH*
Norman Cooeyate**
Alvera Enote**
Mary A. Hoskin, RD, MS**
Camille Natewa**
Carol A. Percy, RN, MS**
Kelly J. Acton, MD, MPH
Vickie L. Andre, RN, FNP
Roz Barber
Shandiin Begay, MPH
Brian C. Bucca, OD, FAAO
Sherron Cook
Jeff Curtis, MD
Charlotte Dodge
Matthew S. Doughty, MD
Jason Kurland, MD
Justin Glass, MD
Martia Glass, MD
Robert L. Hanson, MD, MPH
Louise E. Ingraham, MS, RD, LN
Kathleen M. Kobus, RNC-ANP
Jonathan Krakoff, MD
Catherine Manus, LPN
Cherie McCabe
Sara Michaels, MD
Tina Morgan
Julie A. Nelson, RD
Christopher Piromalli, DO
Robert J. Roy
Sandra Sangster, RD
Miranda Smart
Darryl P. Tonemah, PhD
Rachel Williams, FNP
Charlton Wilson, MD
-
George Washington University Biostatistics Center (DPP Coordinating Center,Rockville, MD)
Sarah Fowler, PhD*
Marinella Temprosa, PhD*
Michael Larsen, PhD*
Tina Brenneman**
Hanna Sherif, MS**
Sharon L. Edelstein, ScM**
Solome Abebe, MS
Julie Bamdad, MS
Melanie Barkalow
Joel Bethepu
Tsedenia Bezabeh
Nicole Butler
Jackie Callaghan
Caitlin E. Carter
Costas Christophi, PhD
Gregory M. Dwyer
Mary Foulkes, PhD
Yuping Gao
Robert Gooding
Adrienne Gottlieb
Nisha Grover
Heather Hoffman, PhD
Ashley N. Hogan
Kathleen Jablonski, PhD
Richard Katz, MD
Preethy Kolinjivadi, MS
John M. Lachin, ScD
Yong Ma, PhD
Qing Pan, PhD
Susan Reamer
Alla Sapozhnikova
-
Lifestyle Resource Core
Elizabeth M. Venditti, PhD*
Andrea M. Kriska, PhD
Linda Semler, MS, RD, LDN
Valerie Weinzierl, MPH
-
Central Biochemistry Laboratory(Seattle, WA)
Santica Marcovina, PhD, ScD*
Greg Strylewicz, PhD**
John Albers, PhD
-
Epidemiological Cardiology Research Center- Epicare(Winston-Salem, NC)
Ronald J. Prineas, MD, PhD*
Zhuming Zhang, MD*
Elsayed Soliman, MD*
Teresa Alexander
Charles Campbell, MS
Yabing Li, MD
Sharon Hall
Julie Hu, MSc
Susan Hensley, BS
Lisa Keasler
Yabing Li, MD
Margaret Mills
Tonya Taylor
-
Fundus Photo Reading Center(Madison, WI)
Ronald Danis, MD*
Matthew Davis, MD*
Larry Hubbard*
Ryan Endres**
Deborah Elsas**
Samantha Johnson**
Vonnie Gama
Anne Goulding
-
Carotid Ultrasound
Gregory Evans
-
Neurocognitive Assessment Group
Jose A. Luchsinger, MD, MPH
Jennifer Manly, PhD
-
NIH/NIDDK(Bethesda, MD)
Judith Fradkin, MD
Sanford Garfield, PhD
-
Centers for Disease Control & Prevention(Atlanta, GA)
Edward Gregg, PhD
Ping Zhang, PhD
-
University of Michigan(Ann Arbor, MI)
William H. Herman, MD, MPH
Michael Brändle, MD, MS
Morton B. Brown, PhD
-
Nutrition Coding Center(Columbia, SC)
Elizabeth Mayer-Davis, PhD*
Robert R. Moran, PhD**
-
Quality of Well-Being Center(La Jolla, CA)
Ted Ganiats, MD*
Andrew J. Sarkin, PhD**
Naomi Katzir
Erik Groessl, PhD
-
Coronary Artery Calcification Reading Center
Matthew Budoff, MD
Chris Dailing
-
Dual Energy X-ray Absorptiometry Reading Center
Ann Schwartz, PhD
Caroline Navy
Lisa Palermo, MS
-
+Genetics Working Group
Ling Chen, MS1
Maegan Harden, BS2
Robert L. Hanson, MD, MPH3
William C. Knowler, MD, DrPH3
Toni I. Pollin, PhD4
Alan R. Shuldiner, MD4
Kathleen Jablonski, PhD5
Paul W. Franks, PhD, MPhil, MS6, 7, 8
Marie-France Hivert, MD1
Footnotes
denotes Principal Investigator
denotes Program Coordinator
Massachusetts General Hospital
Broad Institute
NIDDK
University of Maryland
Coordinating Center
Lund University, Sweden
Umeå University, Sweden
Harvard School of Public Health
NorthShore University HealthSystem
University of Chicago Pritzker School of Medicine
Trial registration: ClinicalTrials.gov NCT00038727 and NCT00004992
Data availability All DPP and DPPOS data are available in the NIDDK repository (www.niddkrepository.org/home/) and can be requested by any researcher.
Duality of interest The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement All authors were responsible for drafting the article and revising it critically for important intellectual content. All authors approved the version to be published.
References
- 1.Diabetes Prevention Program Research Group. The Diabetes Prevention Program. Design and methods for a clinical trial in the prevention of type 2 diabetes. Diabetes Care. 1999;22:623–634. doi: 10.2337/diacare.22.4.623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jarrett RJ, Keen H, Fuller JH, McCartney M. Worsening to diabetes in men with impaired glucose tolerance (“borderline diabetes”) Diabetologia. 1979;16:25–30. doi: 10.1007/BF00423146. [DOI] [PubMed] [Google Scholar]
- 4.Sartor G, Schersten B, Carlstrom S, Melander A, Norden A, Persson G. Ten-year follow-up of subjects with impaired glucose tolerance: prevention of diabetes by tolbutamide and diet regulation. Diabetes. 1980;29:41–49. doi: 10.2337/diab.29.1.41. [DOI] [PubMed] [Google Scholar]
- 5.Keen H, Jarrett RJ, McCartney P. The ten-year follow-up of the Bedford survey (1962–1972): glucose tolerance and diabetes. Diabetologia. 1982;22:73–78. doi: 10.1007/BF00254832. [DOI] [PubMed] [Google Scholar]
- 6.Pan XR, Li GW, Hu YH, et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study. Diabetes Care. 1997;20:537–544. doi: 10.2337/diacare.20.4.537. [DOI] [PubMed] [Google Scholar]
- 7.Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344:1343–1350. doi: 10.1056/NEJM200105033441801. [DOI] [PubMed] [Google Scholar]
- 8.Diabetes Prevention Program Research Group. Knowler WC, Fowler SE, et al. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet. 2009;374:1677–1686. doi: 10.1016/S0140-6736(09)61457-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the Diabetes Prevention Program Outcomes Study. Lancet Diabetes Endocrinol. 2015;3:866–875. doi: 10.1016/S2213-8587(15)00291-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aroda VR, Ratner RE. Interventional trials to prevent diabetes: Diabetes Prevention Program. In: LeRoith D, editor. Prevention of type 2 diabetes: from science to therapy. Springer; New York: 2012. pp. 143–166. [Google Scholar]
- 11.American Diabetes Association. Report of the Expert Committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 1997;20:1183–1197. doi: 10.2337/diacare.20.7.1183. [DOI] [PubMed] [Google Scholar]
- 12.Walker EA, Molitch M, Kramer MK, et al. Adherence to preventive medications: predictors and outcomes in the Diabetes Prevention Program. Diabetes Care. 2006;29:1997–2002. doi: 10.2337/dc06-0454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Diabetes Prevention Program Research Group. Hamman RF, Horton E, et al. Factors affecting the decline in incidence of diabetes in the Diabetes Prevention Program Outcomes Study (DPPOS) Diabetes. 2015;64:989–998. doi: 10.2337/db14-0333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ratner RE, Christophi CA, Metzger BE, et al. Prevention of diabetes in women with a history of gestational diabetes: effects of metformin and lifestyle interventions. J Clin Endocrinol Metab. 2008;93:4774–4779. doi: 10.1210/jc.2008-0772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aroda VR, Christophi CA, Edelstein SL, et al. The effect of lifestyle intervention and metformin on preventing or delaying diabetes among women with and without gestational diabetes: the Diabetes Prevention Program Outcomes Study 10-year follow-up. J Clin Endocrinol Metab. 2015;100:1646–1653. doi: 10.1210/jc.2014-3761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Diabetes Prevention Program Research Group. Effects of withdrawal from metformin on the development of diabetes in the Diabetes Prevention Program. Diabetes Care. 2003;26:977–980. doi: 10.2337/diacare.26.4.977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lachin JM, Christophi CA, Edelstein SL, et al. Factors associated with diabetes onset during metformin versus placebo therapy in the Diabetes Prevention Program. Diabetes. 2007;56:1153–1159. doi: 10.2337/db06-0918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kitabchi AE, Temprosa M, Knowler WC, et al. Role of insulin secretion and sensitivity in the evolution of type 2 diabetes in the Diabetes Prevention Program: effects of lifestyle intervention and metformin. Diabetes. 2005;54:2404–2414. doi: 10.2337/diabetes.54.8.2404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.DeFronzo RA. Pharmacologic therapy for type 2 diabetes mellitus. Ann Intern Med. 1999;131:281–303. doi: 10.7326/0003-4819-131-4-199908170-00008. [DOI] [PubMed] [Google Scholar]
- 20.Hundal RS, Krssak M, Dufour S, et al. Mechanism by which metformin reduces glucose production in type 2 diabetes. Diabetes. 2000;49:2063–2069. doi: 10.2337/diabetes.49.12.2063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.International Expert Committee. International Expert Committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care. 2009;32:1327–1334. doi: 10.2337/dc09-9033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2010;33(Suppl 1):S62–S69. doi: 10.2337/dc10-S062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Diabetes Prevention Program Research Group. HbA1c as a predictor of diabetes and as an outcome in the Diabetes Prevention Program: a randomized clinical trial. Diabetes Care. 2015;38:51–58. doi: 10.2337/dc14-0886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Florez JC, Jablonski KA, Bayley N, et al. TCF7L2 polymorphisms and progression to diabetes in the Diabetes Prevention Program. N Engl J Med. 2006;355:241–250. doi: 10.1056/NEJMoa062418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hivert MF, Jablonski KA, Perreault L, et al. Updated genetic score based on 34 confirmed type 2 diabetes loci is associated with diabetes incidence and regression to normoglycemia in the Diabetes Prevention Program. Diabetes. 2011;60:1340–1348. doi: 10.2337/db10-1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jablonski KA, McAteer JB, de Bakker PI, et al. Common variants in 40 genes assessed for diabetes incidence and response to metformin and lifestyle intervention in the Diabetes Prevention Program. Diabetes. 2010;59:2672–2681. doi: 10.2337/db10-0543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Diabetes Prevention Program Research Group. Changes in albumin excretion in the Diabetes Prevention Program. Diabetes Care. 2009;32:720–725. doi: 10.2337/dc08-1400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Goldberg R, Temprosa M, Otvos J, et al. Lifestyle and metformin treatment favorably influence lipoprotein subfraction distribution in the Diabetes Prevention Program. J Clin Endocrinol Metab. 2013;98:3989–3998. doi: 10.1210/jc.2013-1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Goldberg RB, Temprosa MG, Mather KJ, et al. Lifestyle and metformin interventions have a durable effect to lower CRP and tPA levels in the Diabetes Prevention Program except in those who develop diabetes. Diabetes Care. 2014;37:2253–2260. doi: 10.2337/dc13-2471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Orchard TJ, Temprosa M, Goldberg R, et al. The effect of metformin and intensive lifestyle intervention on the metabolic syndrome: the Diabetes Prevention Program randomized trial. Ann Intern Med. 2005;142:611–619. doi: 10.7326/0003-4819-142-8-200504190-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ratner R, Goldberg R, Haffner S, et al. Impact of intensive lifestyle and metformin therapy on cardiovascular disease risk factors in the Diabetes Prevention Program. Diabetes Care. 2005;28:888–894. doi: 10.2337/diacare.28.4.888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Diabetes Prevention Program Outcomes Study Research Group. Orchard TJ, Temprosa M, et al. Long-term effects of the Diabetes Prevention Program interventions on cardiovascular risk factors: a report from the DPP Outcomes Study. Diabet Med. 2013;30:46–55. doi: 10.1111/j.1464-5491.2012.03750.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goldberg RB, Aroda VR, Bluemke DA, et al. Effect of long-term metformin and lifestyle in the Diabetes Prevention Program and its outcome study on coronary artery calcium. Circulation. 2017 doi: 10.1161/CIRCULATIONAHA.116.025483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Diabetes Prevention Program Research Group. Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care. 2012;35:731–737. doi: 10.2337/dc11-1299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Inzucchi SE, Lipska KJ, Mayo H, Bailey CJ, McGuire DK. Metformin in patients with type 2 diabetes and kidney disease: a systematic review. JAMA. 2014;312:2668–2675. doi: 10.1001/jama.2014.15298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Aroda VR, Edelstein SL, Goldberg RB, et al. Long-term metformin use and vitamin B12 deficiency in the Diabetes Prevention Program Outcomes Study. J Clin Endocrinol Metab. 2016;101:1754–1761. doi: 10.1210/jc.2015-3754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.American Diabetes Association. Prevention or delay of type 2 diabetes. Diabetes Care. 2017;40:S44–S47. doi: 10.2337/dc17-S008. [DOI] [PubMed] [Google Scholar]
- 38.International Diabetes Federation. [Accessed 14 February 2017];IDF diabetes atlas. (7). 2016 www.diabetesatlas.org/
- 39.Diabetes Prevention Program Research Group. The 10-year cost-effectiveness of lifestyle intervention or metformin for diabetes prevention: an intent-to-treat analysis of the DPP/DPPOS. Diabetes Care. 2012;35:723–730. doi: 10.2337/dc11-1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hostalek U, Gwilt M, Hildemann S. Therapeutic use of metformin in prediabetes and diabetes prevention. Drugs. 2015;75:1071–1094. doi: 10.1007/s40265-015-0416-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chiasson JL, Josse RG, Gomis R, et al. Acarbose for prevention of type 2 diabetes mellitus: the STOP-NIDDM randomised trial. Lancet. 2002;359:2072–2077. doi: 10.1016/S0140-6736(02)08905-5. [DOI] [PubMed] [Google Scholar]
- 42.Torgerson JS, Hauptman J, Boldrin MN, Sjostrom L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care. 2004;27:155–161. doi: 10.2337/diacare.27.1.155. [DOI] [PubMed] [Google Scholar]
- 43.Kosaka K, Noda M, Kuzuya T. Prevention of type 2 diabetes by lifestyle intervention: a Japanese trial in IGT males. Diabetes Res Clin Pract. 2005;67:152–162. doi: 10.1016/j.diabres.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 44.Ramachandran A, Snehalatha C, Mary S, et al. The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1) Diabetologia. 2006;49:289–297. doi: 10.1007/s00125-005-0097-z. [DOI] [PubMed] [Google Scholar]
- 45.Dream Trial Investigators. Gerstein HC, Yusuf S, et al. Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: a randomised controlled trial. Lancet. 2006;368:1096–1105. doi: 10.1016/S0140-6736(06)69420-8. [DOI] [PubMed] [Google Scholar]
- 46.Dream Trial Investigators. Bosch J, Yusuf S, et al. Effect of ramipril on the incidence of diabetes. N Engl J Med. 2006;355:1551–1562. doi: 10.1056/NEJMoa065061. [DOI] [PubMed] [Google Scholar]
- 47.Kawamori R, Tajima N, Iwamoto Y, et al. Voglibose for prevention of type 2 diabetes mellitus: a randomised, double-blind trial in Japanese individuals with impaired glucose tolerance. Lancet. 2009;373:1607–1614. doi: 10.1016/S0140-6736(09)60222-1. [DOI] [PubMed] [Google Scholar]
- 48.Navigator Study Group. McMurray JJ, Holman RR, et al. Effect of valsartan on the incidence of diabetes and cardiovascular events. N Engl J Med. 2010;362:1477–1490. doi: 10.1056/NEJMoa1001121. [DOI] [PubMed] [Google Scholar]
- 49.Navigator Study Group. Holman RR, Haffner SM, et al. Effect of nateglinide on the incidence of diabetes and cardiovascular events. N Engl J Med. 2010;362:1463–1476. doi: 10.1056/NEJMoa1001122. [DOI] [PubMed] [Google Scholar]
- 50.Zinman B, Harris SB, Neuman J, et al. Low-dose combination therapy with rosiglitazone and metformin to prevent type 2 diabetes mellitus (CANOE trial): a double-blind randomised controlled study. Lancet. 2010;376:103–111. doi: 10.1016/S0140-6736(10)60746-5. [DOI] [PubMed] [Google Scholar]
- 51.DeFronzo RA, Tripathy D, Schwenke DC, et al. Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med. 2011;364:1104–1115. doi: 10.1056/NEJMoa1010949. [DOI] [PubMed] [Google Scholar]
- 52.le Roux CW, Astrup A, Fujioka K, et al. 3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes: a randomised, double-blind trial. Lancet. 2017;389:1399–1409. doi: 10.1016/S0140-6736(17)30069-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.