Abstract
It is known that transient receptor potential ankyrin 1 (TRPA1) channels, expressed by nociceptors, contribute to neuropathic pain. Here we show that TRPA1 is also expressed in Schwann cells. We found that in mice with partial sciatic nerve ligation, TRPA1 silencing in nociceptors attenuated mechanical allodynia, without affecting macrophage infiltration and oxidative stress, whereas TRPA1 silencing in Schwann cells reduced both allodynia and neuroinflammation. Activation of Schwann cell TRPA1 evoked NADPH oxidase 1 (NOX1)-dependent H2O2 release, and silencing or blocking Schwann cell NOX1 attenuated nerve injury-induced macrophage infiltration, oxidative stress and allodynia. Furthermore, the NOX2-dependent oxidative burst, produced by macrophages recruited to the perineural space activated the TRPA1–NOX1 pathway in Schwann cells, but not TRPA1 in nociceptors. Schwann cell TRPA1 generates a spatially constrained gradient of oxidative stress, which maintains macrophage infiltration to the injured nerve, and sends paracrine signals to activate TRPA1 of ensheathed nociceptors to sustain mechanical allodynia.
Following peripheral nerve injury, influx of immune cells to the site may contribute to the development of chronic pain. Here the authors show that TRPA1 is expressed on Schwann cells and contributes to immune cell influx in a mouse model of neuropathic pain.
Introduction
Neuropathic pain, which is defined as pain caused by a lesion or disease of the somatosensory nervous system1, encompasses a large variety of conditions2. Lesions of the peripheral nervous system can cause lifelong neuropathic pain. Following peripheral nerve injury, local infiltration of inflammatory cells, a hallmark of Wallerian degeneration, occurs3–5, and is associated with the development of neuropathic pain. Although the infiltration of macrophages into the damaged nerve trunk is known to induce mechanical allodynia in mice with sciatic nerve injury6–9, the precise pathway by which inflammatory cells cause persistent allodynia is only partially defined. A series of mediators have been reported to contribute to macrophage infiltration in the damaged nerve10. Notably, inhibition of the chemokine (C–C motif) ligand 2 (CCL2) has been shown to attenuate neuroinflammation and allodynia7,8,11. Oxidative stress contributes to neuropathic pain, since antioxidants attenuate mechanical hypersensitivity in mouse models, including chronic constriction of the sciatic nerve12 and spinal nerve ligation13.
The transient receptor potential ankyrin 1 (TRPA1) channel is highly expressed by a subpopulation of primary sensory neurons14,15 that contain and release the proinflammatory neuropeptides substance P (SP) and calcitonin gene-related peptide (CGRP)15. TRPA1 is activated by a series of exogenous agents, including allyl isothiocyanate (AITC)16,17, and is typically sensitive to the redox state of the milieu18. Notably, a series of reactive oxygen, nitrogen or carbonyl species, including hydrogen peroxide (H2O2), activate TRPA1, resulting in nociceptor stimulation or sensitization19–24. TRPA1 has been shown to mediate mechanical hypersensitivity in different models of inflammatory and neuropathic pain, including those evoked by peripheral nerve injury25–29.
Recent findings in mice with trigeminal nerve injury (constriction of the infraorbital nerve, CION) show that macrophages, recruited by a CCL2-dependent process, increase H2O2 levels within the site of nerve injury30. The resulting oxidative stress and the ensuing increases in reactive carbonyl species were proposed to mediate prolonged mechanical allodynia by gating TRPA1 in trigeminal nerve fibers30. Thus, TRPA1, expressed by primary sensory neurons, appears to be the target of the macrophage-dependent oxidative burst required to promote neuropathic pain. Here, we surprisingly found that pharmacological blockade or genetic deletion of TRPA1 not only induced the expected inhibition of mechanical allodynia, but also suppressed macrophage infiltration and H2O2 generation in the injured nerve. The current study was undertaken to identify the cellular and molecular mechanisms responsible for this TRPA1-mediated macrophage infiltration and generation of oxidative stress.
By using pharmacological and genetic approaches to disrupt TRPA1, including conditional deletion in Schwann cells, we found that Schwann cells that ensheath the injured sciatic nerve axons express TRPA1. Macrophages, which are recruited by CCL2, generate a NADPH oxidase-2 (NOX2)-dependent oxidative burst that targets Schwann cell TRPA1. TRPA1, via NOX1, produces sustained oxidative stress that maintains, in a spatially confined manner, macrophage infiltration into the injured nerve, and which activates TRPA1 on nociceptor nerve fibers to produce allodynia.
Results
TRPA1 mediates neuroinflammation
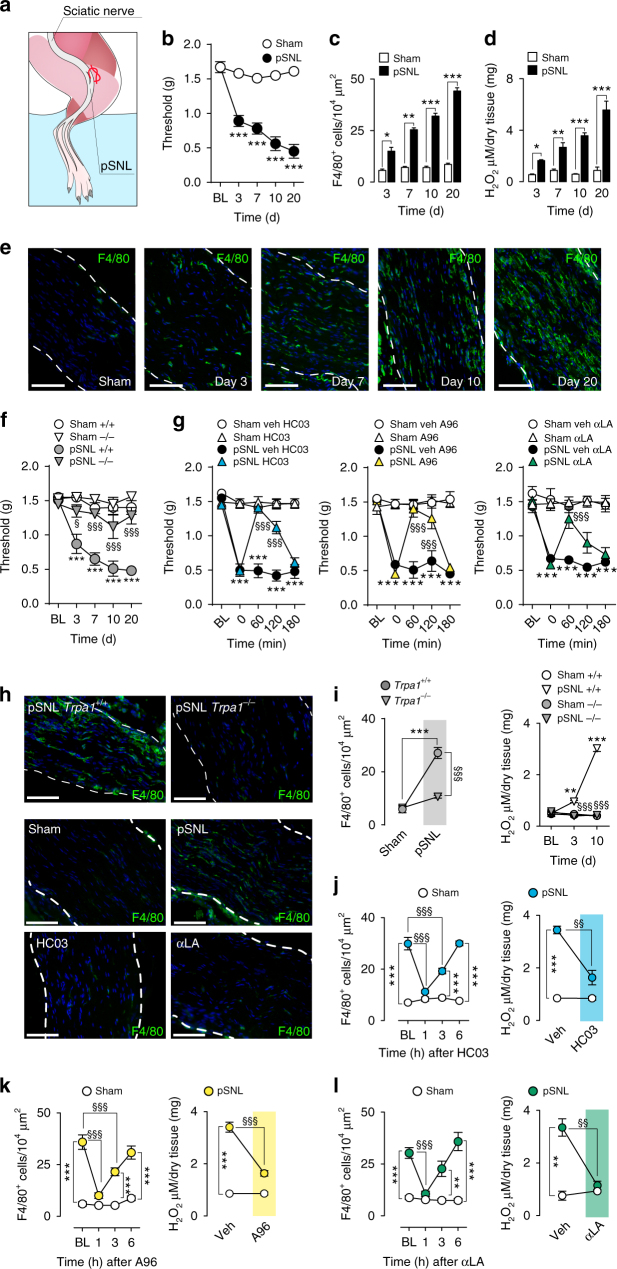
In C57BL/6 mice pSNL, but not sham surgery (Fig. 1a), induced prolonged (3–20 days) mechanical allodynia (Fig. 1b) accompanied by macrophage (F4/80+ cells) recruitment (Fig. 1c, e and Supplementary Fig. 1) and oxidative stress (H2O2) generation (Fig. 1d) within the injured nerve. Trpa1 (Fig. 1f), but not Trpv1 or Trpv4 (Supplementary Fig. 2a), deletion prevented mechanical allodynia. Trpa1, but not Trpv1 or Trpv4, deletion also attenuated cold allodynia, but this response was not further investigated in the present study (Supplementary Fig. 2b). Heat hyperalgesia was unaffected by Trpa1, Trpv1, and Trpv4 deletion (Supplementary Fig. 2c). As previously reported28,30,31 in similar models, at day 10 after pSNL (all measurements were at 10 days unless otherwise specified), TRPA1 antagonists (HC-030031, A-967079) and antioxidants (α-lipoic acid (αLA) and phenyl-N-tert-butylnitrone (PBN)) (Fig. 1g and Supplementary Fig. 3a) reversed mechanical allodynia. Treatments for 3 days with the monocyte-depleting agent clodronate32 or an anti-CCL2 antibody (CCL2-Ab)30,33 attenuated allodynia, macrophage infiltration and H2O2 generation (Supplementary Fig. 4a–c), confirming the proalgesic role of these cells.
Fig. 1.
TRPA1 mediates pSNL-evoked allodynia and neuroinflammation. a Drawing representing the pSNL surgery in mice. b–e Time-dependent (3–20 days, d) mechanical allodynia (b), number and representative images of macrophages (F4/80+ cells) (c, e) and H2O2 content (d) in the sciatic nerve trunk induced by pSNL in C57BL/6 compared to sham mice (n = 6, *P < 0.005, **P < 0.01 ***P < 0.001 pSNL vs. Sham; two-way ANOVA followed by Bonferroni post hoc analyses and unpaired two-tailed Student’s t-test). f Time-dependent (3–20 d) mechanical allodynia in sham/pSNL Trpa1 +/+/Trpa1 −/− mice (n = 8, ***P < 0.001 pSNL+/+ vs. Sham+/+; n = 6, § P < 0.05 and §§§ P < 0.001 pSNL−/− vs. pSNL+/+; two-way ANOVA followed by Bonferroni post hoc analyses). g Mechanical allodynia (at day 10 after surgery) in sham/pSNL mice after HC-030031 (HC03, 100 mg kg−1, i.p.), A-967079 (A96, 100 mg/kg, i.p.) and α-lipoic acid (αLA, 100 mg kg−1, i.p.) or respective vehicles (veh, 4% DMSO and 4% tween 80 in isotonic saline) (n = 6, ***P < 0.001 pSNL veh vs. Sham veh; §§§ P < 0.001 pSNL-HC03, A96 or αLA vs. pSNL-veh; two-way ANOVA followed by Bonferroni post hoc analyses). h–l Representative images, number of F4/80+ cells, and H2O2 content in the sciatic nerve of sham/pSNL Trpa1 +/+/Trpa1 −/− and C57BL/6 mice, before (BL) and 1–6 h after HC03, A96, αLA (all, 100 mg kg−1, i.p.) or respective vehicles (veh, 4% DMSO and 4% tween 80 in isotonic saline) (n = 6, **P < 0.01 and ***P < 0.001 pSNL Trpa1 +/+ vs. Sham-Trpa1 +/+ and pSNL veh vs. Sham veh; §§ P < 0.01 and §§§ P < 0.001 pSNL Trpa1 −/− vs. pSNL-Trpa1 +/+ and pSNL HC03, A96 or αLA vs. pSNL-veh; two-way and one-way ANOVA followed by Bonferroni post hoc analyses). (Scale bars: 50 μm; (e, h) dashed lines, perineurium). Data are represented as mean ± s.e.m
Other inflammatory cells, which are recruited to sites of nerve injury, may also contribute to mechanical allodynia9,34. To explore their role in the delayed phase of mechanical allodynia, the number of neutrophils and T lymphocytes was evaluated in the nerve trunk at day 10 after surgery. Although both neutrophils (Ly6g+ cells) and T lymphocytes (CD8+ cells) were increased by pSNL (Supplementary Fig. 4d), treatment with clodronate, which markedly attenuated both the infiltrating macrophages and allodynia, did not affect the number of neutrophils or T lymphocytes (Supplementary Fig. 4d). In agreement with a previous report34, these data exclude the contribution of neutrophils and T cells to mechanical allodynia assessed 10 days after pSNL.
The hypothesis that oxidative stress produced by infiltrating macrophages targets neuronal TRPA1 to signal neuropathic pain30 implies that the channel inhibition reduces allodynia but does not affect neuroinflammation. Surprisingly, Trpa1 deletion prevented infiltration of F4/80+ cells and H2O2 generation in the injured sciatic nerve (Fig. 1h, i). TRPA1 antagonists (Fig. 1h, j, k) and antioxidants (Fig. 1h, l and Supplementary Fig. 3b) also transiently reversed macrophage infiltration and H2O2 production. Thus, the TRPA1-oxidative stress pathway mediates both neuropathic pain and neuroinflammation in the injured nerve.
CCL2 induces neuroinflammation via TRPA1
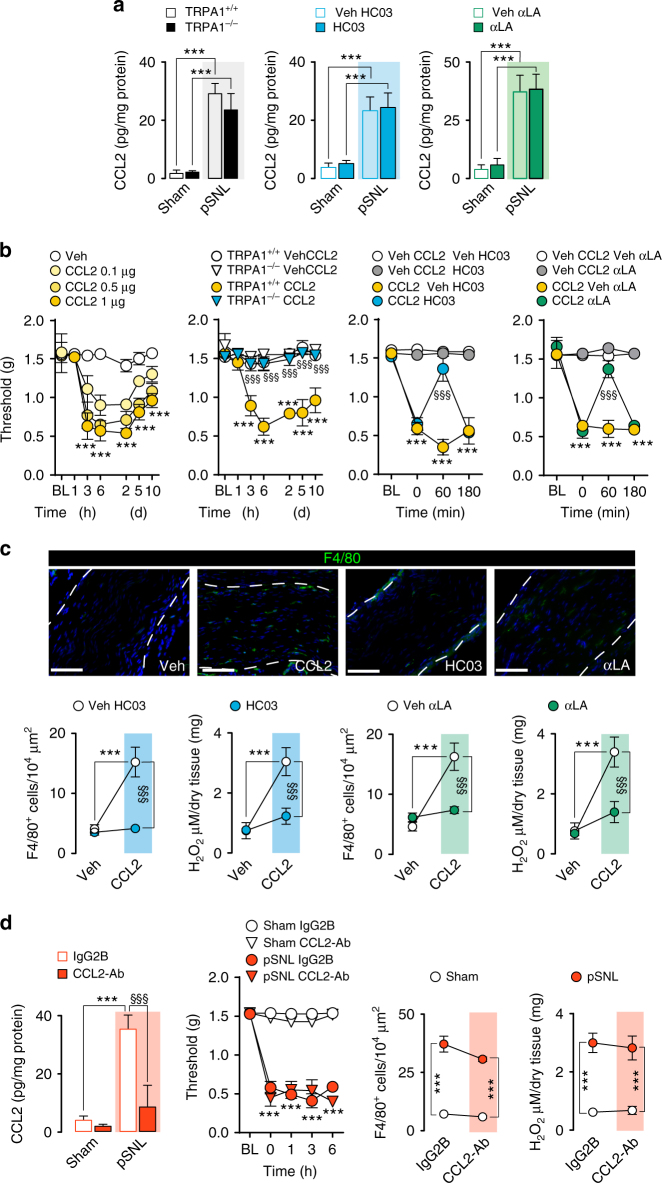
One possible explanation may be that TRPA1 mediates the release of the monocyte chemoattractant, CCL2, generated by injured nerves8. However, as neither TRPA1 deletion or antagonism nor antioxidants affected CCL2 increases in ligated sciatic nerves (Fig. 2a), the chemokine should originate from a TRPA1-oxidative stress-independent pathway. As previously shown8,35, local perineural CCL2 administration induced mechanical allodynia, as well as producing F4/80+ cell infiltration and H2O2 generation (Fig. 2b, c). TRPA1 deletion or antagonism and antioxidants prevented or reversed the effects of CCL2 (Fig. 2b, c). Pretreatment with clodronate, which depletes circulating monocytes and thereby inhibits their neural accumulation30, prevented mechanical allodynia evoked by CCL2 (Supplementary Fig. 4e). Furthermore, in mice with pSNL clodronate treatment depleted macrophages and attenuated mechanical allodynia (Supplementary Fig. 4a), but did not affect the increased CCL2 levels within the ligated nerve trunk (Supplementary Fig. 4f). Together, the present findings support the view that oxidative stress and TRPA1 induce neuroinflammation downstream from CCL2. There was a distinct temporal difference between the effects of CCL2-Ab and TRPA1 antagonists/antioxidants on pSNL-induced neuroinflammation and allodynia. One-hour after HC-030031, A-967079, αLA or PBN, pSNL-induced F4/80+ cell infiltration, H2O2 formation and allodynia were all prominently inhibited (Fig. 1g, h, j–l and Supplementary Fig. 3b), whereas a high dose of the CCL2-Ab, which at 1 h already attenuated neural CCL2 levels, was completely ineffective (over 6 h) in reducing mechanical allodynia (Fig. 2d). Successful inhibition of pain and inflammation required the administration of a lower CCL2-Ab dose for 3 consecutive days that, as expected30,33, also reduced CCL2 levels in the nerve trunk (Supplementary Fig. 4b). Thus, while TRPA1-antagonism/antioxidants rapidly (within 1 h) reversed neuroinflammation, CCL2-blockade required a much longer time (3 days) to produce the same inhibitory effects30,33.
Fig. 2.
TRPA1 mediates CCL2-evoked allodynia and neuroinflammation. a CCL2 levels in sciatic nerves (at day 10 after surgery) of sham/pSNL Trpa1 +/+/Trpa1 −/− and C57BL/6 mice after HC-030031 (HC03, 100 mg kg−1, i.p.), α-lipoic acid (αLA, 100 mg kg−1, i.p.) or respective vehicles (veh, 4% DMSO and 4% tween 80 in isotonic saline) (n = 6, ***P < 0.001 pSNL-Trpa1 +/+ vs. sham-Trpa1 +/+ and pSNL-veh vs. sham-veh; one-way ANOVA followed by Bonferroni post hoc analyses). b Mechanical allodynia induced by perineural CCL2 (0.1–1 µg) or vehicle (veh, isotonic saline) in C57BL/6 mice (n = 4, ***P < 0.001 veh vs. CCL2 (1 µg), two-way ANOVA followed by Bonferroni post hoc analyses) and CCL2 (1 µg) in Trpa1 +/+/Trpa1 −/− and after HC03, αLA (both, 100 mg/kg, i.p.) or respective vehicles (veh, 4% DMSO and 4% tween 80 in isotonic saline) in C57BL/6 mice (n = 4, ***P < 0.001 Trpa1 +/+ CCL2 vs. Trpa1 +/+veh; CCL2 veh HC03, αLA vs. veh CCL2; §§§ P < 0.001 Trpa1 −/− CCL2 vs. Trpa1 +/+ CCL2 and CCL2 HC03, αLA vs. CCL2 veh HC03, αLA; two-way ANOVA followed by Bonferroni post hoc analyses). c Representative images, F4/80+ cell number and H2O2 content in sciatic nerves of mice treated with perineural CCL2 (1 µg) after HC03, αLA (both, 100 mg kg−1, i.p.) or respective vehicles (veh, 4% DMSO and 4% tween 80 in isotonic saline) (n = 5, ***P < 0.001 CCL2 vs. veh HC03, αLA; §§§ P < 0.001 CCL2 HC03, αLA vs. CCL2 veh HC03, αLA; one-way ANOVA followed by Bonferroni post hoc analyses) (Scale bars: 50 μm, dashed lines indicate perineurium). d CCL2 levels, mechanical allodynia, F4/80+ cell number and H2O2 content in sciatic nerves (at day 10 after surgery) of sham/pSNL C57BL/6 mice after an anti-CCL2 antibody (CCL2-Ab) or IgG2B control (120 µg 200 µl−1, i.p., single administration) (n = 6, ***P < 0.001 pSNL-CCL2-Ab vs. sham-CCL2-Ab; §§§ P < 0.001 pSNL-CCL2-Ab vs. pSNL-IgG2B; one-way ANOVA followed by Bonferroni post hoc analyses). Data are represented as mean ± s.e.m
Schwann cells express TRPA1 and release H2O2
Nerve fibers, macrophages and Schwann cells within the injured nerve trunk could potentially mediate TRPA1-dependent oxidative stress. By targeting TRPV1, resiniferatoxin (RTX) defunctionalizes TRPV1+/TRPA1+ neurons36–38. RTX abolished nociceptive responses to TRPV1 (capsaicin) and AITC (Supplementary Fig. 5a) and reversed pSNL-evoked allodynia, but did not affect F4/80+ cell infiltration and H2O2 generation (Supplementary Fig. 5b). Thus, TRPV1+/TRPA1+ nerve fibers mediate neuropathic pain but not neuroinflammation. Naïve or lipopolysaccharide-activated mouse peritoneal macrophages in culture neither expressed TRPA1 mRNA (RT-qPCR), TRPA1 protein (immunocytochemistry) nor responded to AITC (Ca2+-signaling) (Supplementary Fig. 5c–e). Thus, macrophages infiltrating the injured nerve cannot generate TRPA1-dependent oxidative stress.
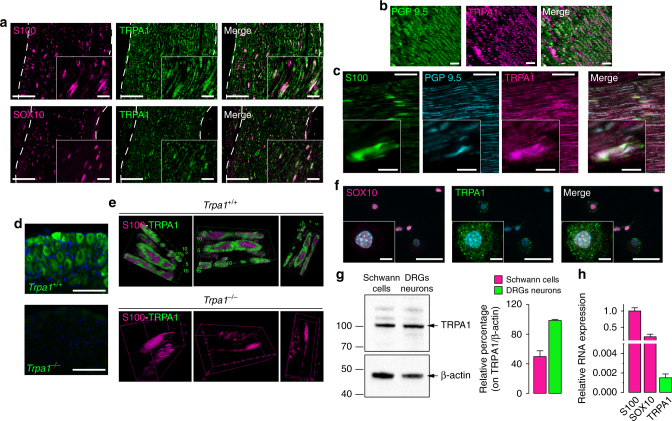
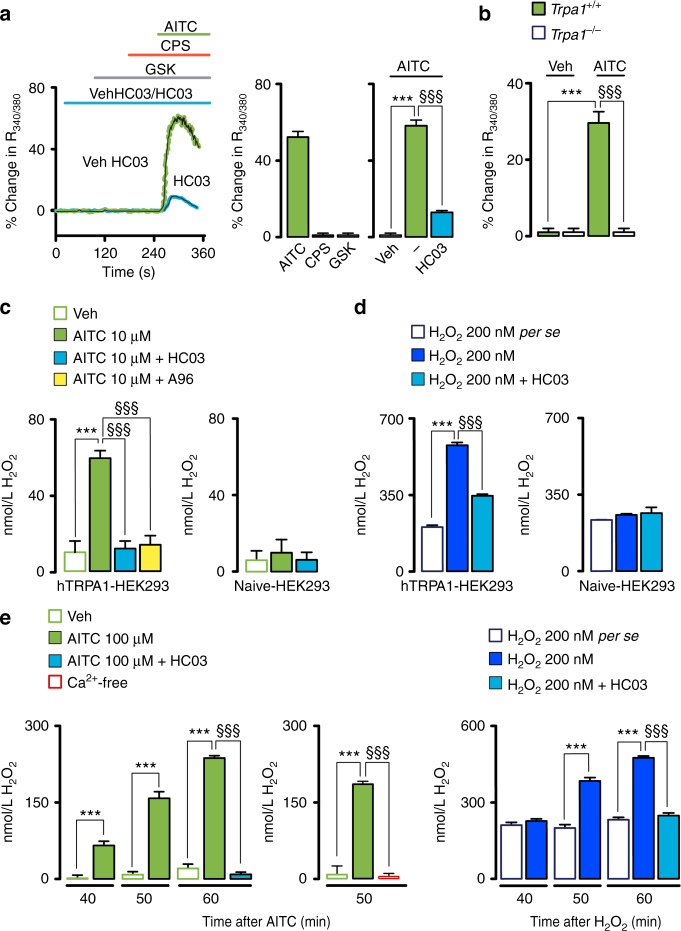
Schwann cells ensheath nerve fibers, including C-fiber nociceptors, and represent 90% of the nucleated cells of the nerve trunk39. We localized immunoreactive TRPA1 to PGP9.5+ nerve fibers and to S-100+ or SOX10+ Schwann cells in the sciatic nerve trunk from C57BL/6 mice (Fig. 3a–c). TRPA1 immunoreactivity was not detected in dorsal root ganglia (DRG, L4-L6) (Fig. 3d) or in S-100+ cells in the sciatic nerve trunk (Fig. 3e) from Trpa1 −/− mice, which confirms antibody selectivity. Expression of TRPA1 (protein and mRNA) in cultured mouse Schwann cells was confirmed by immunofluorescence, western blotting and RT-qPCR (Fig. 3f–h and Supplementary Fig. 6a). Furthermore, AITC-induced intracellular Ca2+ response in cultured Schwann cells from wild type mice, which was attenuated by HC-030031 (Fig. 4a). In contrast, capsaicin or a TRPV4 agonist (GSK1016790A) failed to produce any Ca2+ response (Fig. 4a). Importantly, Schwann cells from Trpa1 +/+ mice, but not from Trpa1 −/− mice, responded to AITC (Fig. 4b). The ability of TRPA1 to promote oxidative stress was explored by measuring H2O2 generation. Both AITC and H2O2, which has been shown to gate TRPA120, stimulated H2O2 release from HEK293 cells expressing the human TRPA1 (hTRPA1-HEK293 cells), but not from untransfected HEK293 cells (Fig. 4c, d). AITC or H2O2 also produced a time- and Ca2+-dependent H2O2 generation in Schwann cells, which was prevented by HC-030031 (Fig. 4e). Thus, Schwann cells generate H2O2 in response to TRPA1 activation.
Fig. 3.
Schwann cells express TRPA1. a Double immunofluorescence staining of TRPA1 and S-100 and SOX10 (two specific markers for detecting Schwann cells), in sciatic nerve from C57BL/6 mice (Scale bars: 50 μm and inset 20 μm). b 3D confocal images of TRPA1 and PGP9.5 staining in Schwann cells from sciatic nerve trunks of C57BL/6 mice (Scale bars: 20 μm). c Triple immunofluorescence staining of S-100, PGP9.5 and TRPA1 in sciatic nerve trunks from C57BL/6 mice (Scale bars: 20 μm and inset 10 μm). d TRPA1 staining in DRGs neurons from Trpa1 +/+ and Trpa1 −/− mice (Scale bars: 50 μm). e 3D confocal image reconstructions of TRPA1 and S100 in Schwann cells from sciatic nerve trunks of Trpa1 +/+ and Trpa1 −/− mice. f TRPA1 and SOX-10 immunoreactivity in cultured C57BL/6 mouse Schwann cells (Scale bars: 50 μm and inset 10 µm). g Representative blot and TRPA1 protein content in cultured Schwann cells and DRGs neurons taken from C57BL/6 mice. Equally loaded protein was checked by expression of β-actin (n = 4 independent experiments). h TRPA1 mRNA relative expression in cultured C57BL/6 mouse Schwann cells (n = 3 replicates from two independent experiments). Data are represented as mean ± s.e.m
Fig. 4.
Schwann cells expressing TRPA1 release H2O2. a Ca2+ responses to AITC (1 mM) in cultured Schwann cells with HC-030031 (HC03, 30µM) or its vehicle (veh, 0.3% DMSO) and to TRPV1- (capsaicin, CPS, 0.5 µM) or TRPV4- (GSK1016790A, GSK, 50 nM) agonists (n = 25 cells from 3 independent experiments, ***P < 0.001 AITC vs. veh; §§§ P < 0.001 HC03 vs. AITC; one-way ANOVA followed by Bonferroni post hoc analyses). b AITC (1 mM)-evoked calcium response in Schwann cells from Trpa1 +/+, but not from Trpa1 −/− mice (n = 25 cells from 3 independent experiments, ***P < 0.001 Trpa1 +/+ AITC vs. Trpa1 +/+ veh; §§§ P < 0.001 Trpa1 −/− AITC vs. Trpa1 +/+ AITC; one-way ANOVA followed by Bonferroni post hoc analyses). c, d H2O2 release from hTRPA1-HEK293 or untransfected (naïve-HEK293) cells induced by AITC (10 µM) or H2O2 (200 nM) and effect of HC03 (30 µM), A-967079 (A96, 30 µM) or respective vehicles (veh, 0.3% DMSO) (n = 8 replicates from three independent experiments, ***P < 0.001 AITC, H2O2 vs. veh; §§§ P < 0.001 AITC, H2O2 + HC03/A96 vs. AITC, H2O2; one-way ANOVA followed by Bonferroni post hoc analyses; H2O2 200 nM per se represents the value of H2O2 over the time, not in presence of cells). e H2O2 release from cultured mouse Schwann cells evoked by AITC (100 µM) or H2O2 (200 nM) and effect of HC03 (30 µM) and Ca2+-free medium (Ca2+-free) (n = 8 replicates from three independent experiments, ***P < 0.001, veh-AITC/H2O2 vs. AITC/H2O2; §§§ P < 0.001 HC03 vs. AITC, H2O2; one-way ANOVA followed by Bonferroni post hoc analyses; H2O2 200 nM per se represents the value of H2O2 without cells). Data are represented as mean ± s.e.m
Schwann cells generate oxidative stress via TRPA1 and NOX1
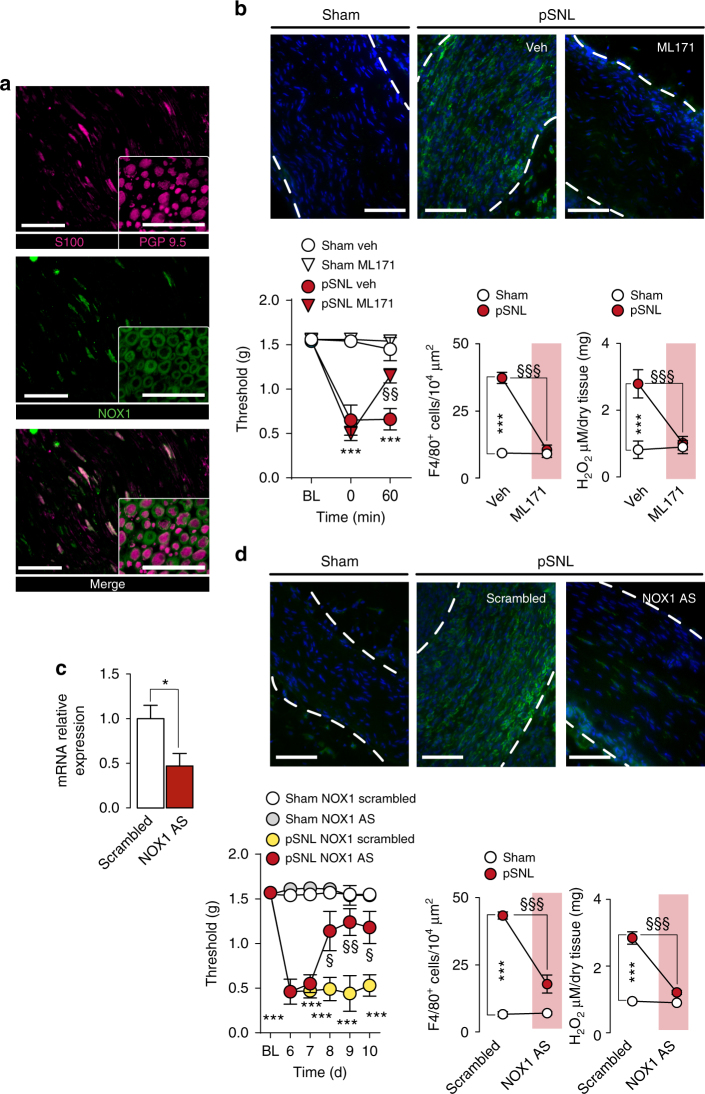
Different NOXs play a key role in oxidative stress40. While we observed that macrophages contain exclusively NOX2 (mRNA and protein) (Supplementary Fig. 5c, f, g), cultured Schwann cells expressed NOX1, NOX2 and NOX4 mRNAs (Supplementary Fig. 6a). Yet, only NOX1 protein was detected by immunofluorescence (Fig. 5a and Supplementary Fig. 6b). Functional data corroborated the molecular/morphological findings as the selective NOX2-inhibitor (Gp91ds-tat peptide)41, but not the NOX1/NOX4 inhibitor (GKT13783)42, attenuated phorbol myristate acetate-stimulated H2O2 release from cultured peritoneal macrophages (Supplementary Fig. 5h). However, GKT13783, but not gp91ds-tat peptide, inhibited AITC-induced H2O2 release from cultured Schwann cells (Supplementary Fig. 5i), indicating a major role for NOX1. 1 h after the administration of 2-acetylphenothiazine (ML171, selective NOX1-inhibitor)43 or GKT13783, pSNL-induced allodynia and neuroinflammation were attenuated (Fig. 5b and Supplementary Fig. 6c), whereas gp91ds-tat peptide was ineffective (Supplementary Fig. 6d). Perineural administration of antisense oligonucleotides (AS-ODN) for NOX1, NOX2 or NOX4 effectively knocked down respective mRNA expression (Fig. 5c and Supplementary Fig. 6e, g). However, only NOX1 AS-ODN attenuated pSNL-induced allodynia, macrophage infiltration and H2O2 generation (Fig. 5d and Supplementary Fig. 6f, h).
Fig. 5.
NOX1 blockade inhibited pSNL-evoked allodynia and neuroinflammation. a Images of S100/PGP9.5 and NOX1 immunofluorescence in sciatic nerves (Scale bars: 50 μm and inset 20 μm). b Mechanical allodynia, representative images and F4/80+ cell number, and H2O2 content (at day 10 after surgery) in sham/pSNL mice after NOX1-inhibitor (ML171, 60 mg kg−1, i.p) or vehicle (veh, 4% DMSO and 4% tween 80 in isotonic saline) (n = 6, ***P < 0.001 pSNL-veh vs. sham-veh; §§ P < 0.01 and §§§ P < 0.001 pSNL-ML171 vs. pSNL-veh; two-way ANOVA followed by Bonferroni post hoc analyses) (Scale bars: 50 μm; dashed lines, perineurium). c NOX1 mRNA relative expression in sciatic nerve after perineural NOX1 antisense oligonucleotides (AS-ODN) or scrambled-ODN (both, 10 nmol 10 µl−1) (n = 3 replicates from two independent experiments, *P < 0.05 AS-ODN vs. scrambled-ODN; unpaired two-tailed Student’s t-test). d Mechanical allodynia, representative images and F4/80+ cell number, and H2O2 content (at day 10 after surgery) in sham/pSNL mice after NOX1 AS-ODN or scrambled-ODN (both, 10 nmol 10 µl−1) (n = 6, ***P < 0.001 pSNL-NOX1-scrambled vs. sham-NOX1scrambled; § P < 0.05, §§ P < 0.01 and §§§ P < 0.001 pSNL-NOX1 AS-ODN vs. pSNL-NOX1scrambled; two-way ANOVA followed by Bonferroni post hoc analyses) (Scale bars: 50 μm; dashed lines, perineurium). Data are represented as mean ± s.e.m
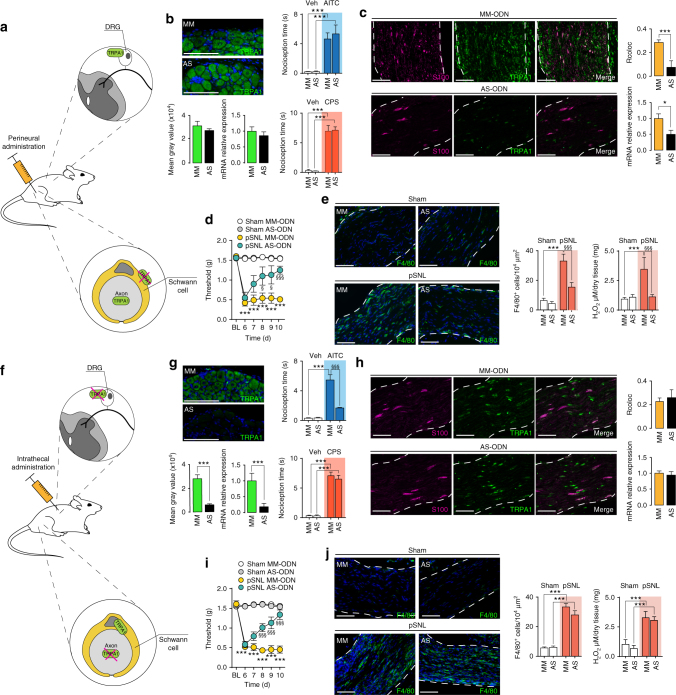
To define the contribution of the TRPA1/NOX1 pathway of Schwann cells to neuroinflammation and neuropathic pain evoked by pSNL, antisense TRPA1 oligonucleotide (TRPA1 AS-ODN) or their mismatched (TRPA1 MM-ODN) analogs were administered to pSNL mice by perineural or intrathecal route (Fig. 6a, f). Perineural treatment with TRPA1 AS-ODN did not affect expression of TRPA1 mRNA and immunoreactivity in DRGs (L4-L6), or acute nociceptive responses to perineural AITC or capsaicin (Fig. 6b). However, it markedly reduced TRPA1 mRNA and the TRPA1/S-100 overlap (Fig. 6c) in injured sciatic nerves, and inhibited pSNL-evoked allodynia, neural F4/80+ cell infiltration, and H2O2 generation (Fig. 6d, e). Thus, Schwann cell TRPA1 silencing preserved TRPA1-mediated acute nociceptive signaling, but disrupted pSNL-evoked neuroinflammation and allodynia. Intrathecal administration of TRPA1 AS-ODN (Fig. 6f) down-regulated TRPA1 mRNA and immunoreactivity44 in DRGs (L4-L6), suppressed the acute nociception by perineural AITC, but not capsaicin, and inhibited pSNL-evoked allodynia (Fig. 6g, i). However, TRPA1 mRNA, the TRPA1/S-100 overlap, and pSNL-evoked F4/80+ cell infiltration and H2O2 generation in injured nerves were unaffected (Fig. 6h, j).
Fig. 6.
Oxidative stress from Schwann cell TRPA1 recruits macrophages and signal pain in C57BL/6 mice. a, f Schematic representation of perineural/intrathecal injection of TRPA1 antisense/mismatch oligonucleotides (AS/MM-ODN). b, g TRPA1 immunofluorescence (mean gray value) and TRPA1 mRNA relative expression in DRGs and acute nociception after perineural AITC (20 nmol 10 µl−1) or capsaicin (CPS, 1 nmol 10 µl−1) following perineural (10 nmol 10 µl−1) (b) or intrathecal (5 nmol 5 µl−1) (g) TRPA1 AS/MM-ODN treatment (once/day for 4 consecutive days) in C57BL/6 (n = 6, ***P < 0.001 MM/AS AITC, CPS vs. MM/AS veh; §§§ P < 0.001 AS AITC vs. MM AITC; one-way ANOVA followed by Bonferroni post hoc analyses, Scale bars: 20 µm). c, h Representative images (Scale bars: 50 μm; dashed lines, perineurium), (j) colocalization value (Rcoloc) of S100/TRPA1 and mRNA-TRPA1 expression in sciatic nerve after perineural (c) and intrathecal (h) AS/MM-ODN (n = 6, *P < 0.05; ***P < 0.001 AS vs MM; unpaired two-tailed Student’s t-test). d, i Mechanical allodynia, and (e, j) representative images, F4/80+-cells, and H2O2-content (at day 10 after surgery) in sham/pSNL mice after perineural (d, e) and intratechal (i, j) AS/MM-ODN (n = 8, ***P < 0.001 pSNL-MM-ODN vs. sham-MM-ODN; § P < 0.05 and §§§ P < 0.001 pSNL-AS-ODN vs. pSNL-MM-ODN; (d, i) two-way ANOVA followed by Bonferroni post hoc analyses and (e, j) one-way ANOVA followed by Bonferroni post hoc analyses) (Scale bars: 50 μm; dashed lines, perineurium). Data are represented as mean ± s.e.m
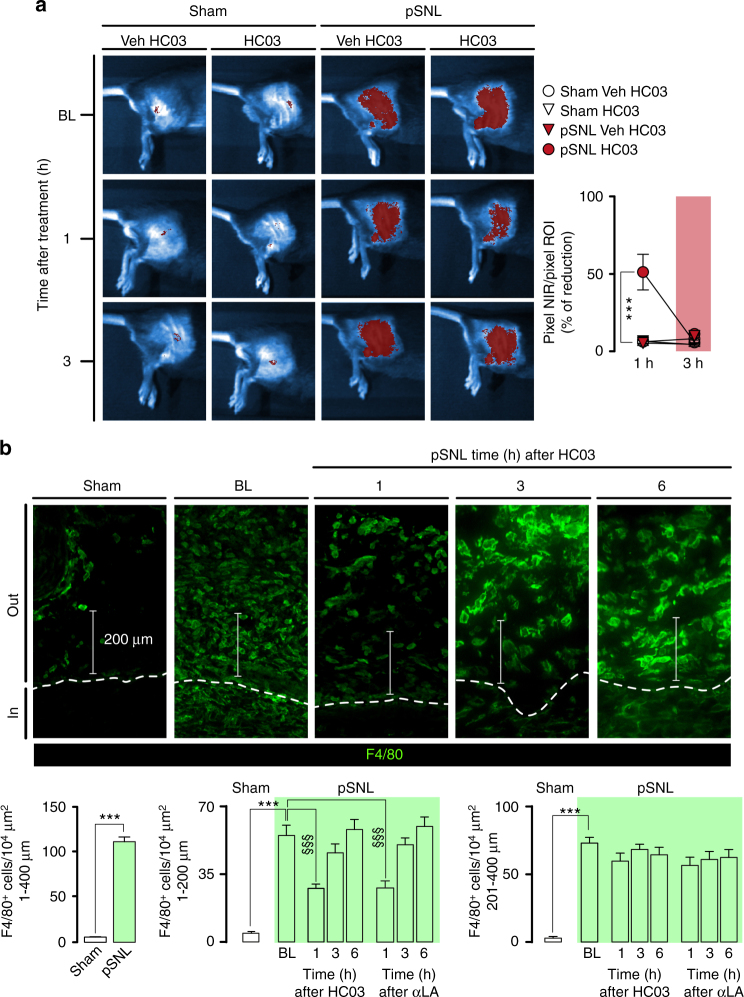
HC-030031 reduced the number of fluorescent macrophages accumulated at the site of pSNL by ~50% (Fig. 7a), assessed by in vivo imaging45. Increased F4/80+ cells were found in the tissue surrounding the injured nerve trunk (Fig. 7b). HC-030031 or αLA reduced F4/80+ cells in the pSNL-injured nerve trunk and neighboring tissue, but not in perineural tissue distant from the injury site (Fig. 7b). The Schwann cell-mediated TRPA1/NOX1 pathway regulates the final stages of macrophage migration from the circulation into the injured nerve trunk, in a manner dependent on the H2O2 concentration gradient.
Fig. 7.
TRPA1 blockade and antioxidant reduced the number of fluorescent macrophages accumulated at the site of pSNL. a In vivo imaging and quantitative data (NIR area/total ROI) of NIR labeled macrophages (at day 10 after surgery) in sham/pSNL mice at baseline (BL), 1 and 3 h after HC-030031 (HC03, 100 mg kg−1, i.p.) (n = 4, ***P < 0.001 pSNL HC03 vs. pSNL Veh HC03; two-way ANOVA followed by Bonferroni post hoc analyses). b Representative images and F4/80+-cell number surrounding the injured nerve trunk (at day 10 after surgery) of sham/pSNL-mice at BL and 1, 3 and 6 h after HC03 or alpha-lipoic acid (αLA) (both, 100 mg kg−1, i.p.). (n = 4, ***P < 0.001 pSNL vs sham; §§§ P < 0.001 pSNL HC03, αLA vs. pSNL veh; one-way ANOVA followed by Bonferroni post hoc analyses) (Scale bars, 200 µm; inside (in) and outside (out) sciatic nerves). Data are represented as mean ± s.e.m
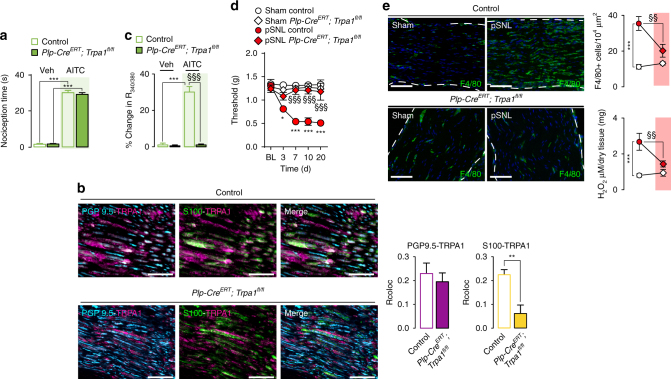
To provide further support for the involvement of Schwann cell TRPA1 in orchestrating neuroinflammation and ensuing neuropathic pain in the pSNL model, we selectively deleted TRPA1 from Schwann cells. We crossed a floxed TRPA1 mouse (TRPA1fl/fl) with a Plp1-Cre/ERT mouse in which Cre recombinase is expressed in Schwann cells/oligodendrocytes (Plp1-Cre ERT ;Trpa1 fl/fl mice). Plp1-Cre ERT ;Trpa1 fl/fl mice were treated with tamoxifen to induce TRPA1 deletion in Schwann cells. In Plp1-Cre ERT ;Trpa1 fl/fl mice, the ability of intraplantar injection of AITC to evoke acute nociception was unaffected (Fig. 8a). In nerve trunks from Plp1-Cre ERT ;Trpa1 fl/fl mice, immunoreactive TRPA1 was detected in PGP9.5+ nerve fibers, indicating preservation of TRPA1 expression in sensory nerve fibers (Fig. 8b). In contrast, immunoreactive TRPA1 was markedly down-regulated in S100+ cells, but not in PGP9.5+ nerve fibers, confirming effective channel deletion in Schwann cells (Fig. 8b). Functional confirmation of the selective conditional Trpa1 gene knock-out was obtained by the failure of AITC to increase [Ca2+]i in Schwann cells from Plp1-Cre ERT ;Trpa1 fl/fl mice (Fig. 8c). In Plp1-Cre ERT ;Trpa1 fl/fl mice, mechanical allodynia (Fig. 8d) and macrophage recruitment and oxidative stress (H2O2 generation) in the injured nerve (Fig. 8e) were markedly attenuated. Thus, tissue-selective deletion shows that Schwann cell TRPA1 promotes macrophage infiltration and oxidative stress in injured nerve trunks, whereas nociceptor TRPA1 does not contribute to neuroinflammation.
Fig. 8.
Plp1-Cre/ERT–mediated Trpa1 deletion from Schwann cells prevented the partial sciatic nerve ligation (pSNL)-evoked allodynia and neuroinflammation. a Acute nociception response induced by intraplantar AITC (10 nmol 20 µl−1) or vehicle (veh, 0.5% DMSO) in Plp1-Cre ERT ;Trpa1 fl/fl and control mice (n = 6, ***P < 0.001 AITC vs. veh; one-way ANOVA followed by Bonferroni post hoc analyses). b Triple immunofluorescence staining and colocalization value (Rcoloc) of TRPA1, PGP9.5, and S100 (Scale bars: 20 µm) in sciatic nerve trunks obtained from Plp1-Cre ERT ;Trpa1 fl/fl (n = 5) and control mice (n = 4), (**P < 0.01; Plp1-Cre ERT ;Trpa1 fl/fl vs. control; unpaired two-tailed Student’s t-test). c Ca2+ responses to AITC (1 mM) or vehicle (veh, 1% DMSO) in cultured Schwann cells from sciatic nerve trunks of Plp1-Cre ERT ;Trpa1 fl/fl and control mice (n = 25 cells/two independent experiments, ***P < 0.001 AITC vs. veh; §§§ P < 0.001 AITC Plp1-Cre ERT ;Trpa1 fl/fl vs. AITC control; one-way ANOVA followed by Bonferroni post hoc analyses). d Mechanical allodynia, and (e) representative images, F4/80+ cells, and H2O2 content (at day 10 after surgery) in sham/pSNL Plp1-Cre ERT ;Trpa1 fl/fl and control mice (n = 8, *P < 0.05 and ***P < 0.001 pSNL control vs. sham control; §§ P < 0.01 and §§§ P < 0.001 pSNL Plp1-Cre ERT ;Trpa1 fl/fl vs. pSNL control; (d) two-way ANOVA followed by Bonferroni post hoc analyses and (e) one-way ANOVA followed by Bonferroni post hoc analyses) (Scale bars: 50 μm). Data are represented as mean ± s.e.m
Discussion
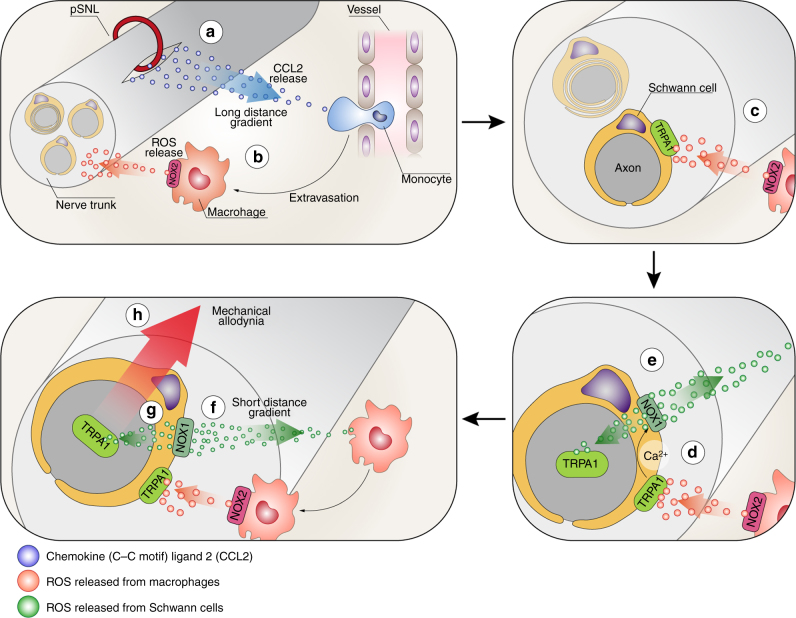
We report the discovery of a role for TRPA1 in Schwann cells in neuroinflammation and ensuing neuropathic pain. Previous studies have implicated TRPA1 in primary sensory neurons as a mediator of mechanical allodynia14,15,46. The established capacity of TRPA1 to sense oxidative stress15,18,19,22, and recent data obtained in a model of neuropathic pain caused by trigeminal nerve injury30, led to the hypothesis that mechanical allodynia is sustained by the oxidative burden generated by infiltrating macrophages that continuously target TRPA1 in nerve fibers. Our present results support the view that nociceptor TRPA1 is the ultimate peripheral target to signal pSNL-evoked allodynia to the brain. However, our findings demonstrate that Schwann cell TRPA1, rather than neuronal TRPA1, orchestrates the neuroinflammation and oxidative stress that sustain neuropathic pain (Fig. 9).
Fig. 9.
Cellular and molecular events contributing to TRPA1-mediated mechanical allodynia and neuroinflammation in a neuropathic pain model. Partial sciatic nerve ligation (pSNL) by releasing CCL2 (a) promotes the extravasation of hematogenous monocytes (b) that, via their rapid NOX2-dependent oxidative burst (red dots) target the TRPA1 channel localized in Schwann cells (c). TRPA1 activation in Schwann cells evokes a Ca2+-dependent, NOX1-mediated (d) prolonged H2O2 (green dots) generation (e) with a dual function. The outward H2O2 release (f) produces a space-scaled gradient that determines the final macrophage influx to the injured nerve trunk, whereas the inward H2O2 release (g) targets nociceptor TRPA1 to produce mechanical allodynia (h). ROS reactive oxygen species
Diverse lines of evidence support the hypothesis that Schwann cell TRPA1 is necessary and sufficient to mediate neuroinflammation and neuropathic pain. TRPA1 blockade, achieved with chemically unrelated antagonists, markedly decreased macrophage accumulation and the oxidative burden in the injured nerve. Studies of Trpa1 −/− mice confirmed the findings obtained with pharmacological antagonists. Although in the present model of neuropathic pain both approaches unequivocally demonstrated the key role of TRPA1, they could not discriminate between the specific contribution of neuronal vs. non-neuronal channels. TRPA1 is expressed by peptidergic primary sensory neurons that, by releasing SP and CGRP, promote neurogenic inflammation47–49. Stimulants of neurogenic inflammation, including the prototypic TRPV1 agonist capsaicin, evoke a transient and moderate inflammatory response, which is chemokine/cytokine-independent and is characterized by CGRP-mediated arteriole vasodilatation and SP-mediated plasma protein and leukocyte extravasation from postcapillary venules15,50. Although neurogenic inflammation neither induces the infiltration of inflammatory cells after nerve injury nor mediate neuropathic pain, it does contribute to migraine attacks51,52. In contrast, neuroinflammation is a localized and persistent inflammatory process that is confined to the injured nerve and neighboring tissues. The hallmarks of neuroinflammation encompasses chronic infiltration of leukocytes, activation of glial cells, and increased production of inflammatory mediators, including a series of cytokines and chemokines and neuropathic pain5,53. Experiments with RTX, which defunctionalizes TRPV1/TRPA1-expressing neurons and abrogate their sensory and proinflammatory efferent functions36,54,55, exclude the possibility that TRPA1-dependent neurogenic inflammation contributes to pSNL-evoked neuroinflammation. RTX attenuated mechanical allodynia, but not macrophage number or H2O2 levels, which suggests that TRPA1 present in TRPV1+ peptidergic neurons may signal allodynia, but does not promote the neuroinflammatory component.
We observed that the site-specific (perineural vs. intrathecal) administration of TRPA1 AS-ODN efficiently disrupted TRPA1 expressed in nociceptors or Schwann cells, respectively, as demonstrated by behavioral and molecular studies. A reduced expression of the nociceptor TRPA1 was associated with attenuation of pain, whereas diminished expression of Schwann cell TRPA1 inhibited both pain and neuroinflammation. These findings support the hypothesis that non-neuronal TRPA1 channels exert a key role in inflammatory cell recruitment and oxidative stress generation. Confirmation of this proposal was derived from experiments with Plp1-Cre ERT ;Trpa1 fl/fl mice, which exhibited selective depletion of Trpa1 in Schwann cells and markedly attenuated neuroinflammation and mechanical allodynia. This localization of TRPA1 in Schwann cells represents a plausible explanation for the widely-reported efficacy of TRPA1 antagonists in different models of neuropathic pain produced by nerve injury25,27,28,30, where neuroinflammation is the underlying mechanism of the ongoing pain condition.
The CCL2 receptor (CCR2) is expressed by primary sensory neurons56,57, and CCL2 has been shown to increase TRPV1 expression58 and to sensitize TRPA1 and TRPV159 in nociceptors. CCL2 is upregulated during neuronal injury, and may activate its cognate receptor CCR2 on TRPV1-positive nociceptors58. The CCL2 system has been reported to augment nociceptor sensitivity by increasing TRPV1 expression58 and TRPA1 and TRPV1 function59. The present findings, showing that CCL2 rapidly increases neuronal hypersensitivity, support the view that this chemokine may directly stimulate primary sensory neurons, thereby enhancing mechanical allodynia under short-lived experimental conditions59,60. However, as indicated by studies with macrophage depletion, CCL2 requires the contribution of infiltrated macrophages within the injured nerve trunk to sustain the allodynia in a prolonged model of neuropathic pain, such as the pSNL in mice. Neutrophils and lymphocytes have been reported to accumulate, although at a minor extent compared to macrophages, at sites of nerve damage, where they may contribute to the initial9, but not delayed phase34, of neuropathic pain. Their role in mechanical allodynia at day 10 after surgery is further excluded by the present observation that clodronate attenuated allodynia and macrophage infiltration, whereas the influx of neutrophils and lymphocytes was unchanged.
Our results reveal distinct kinetics of macrophage accumulation by CCL2 and the TRPA1/oxidative stress pathways. Despite its ability to suppress perineural CCL2 levels within 1 h, a high dose of the CCL2-Ab failed to affect macrophage number, oxidative stress, and allodynia over 6 h. In contrast, a prolonged, 3-day antibody treatment was required for successful inhibition of neuroinflammation and pain30. The persistent temporal frame necessary for CCL2 inhibition to attenuate neuroinflammation and pain is, therefore, markedly different from the very short time-period (1–3 h) required by TRPA1 antagonists or antioxidants to produce the same inhibitory responses. Oxidative burst has been reported to exert a chemoattractant activity toward macrophages61, which is limited by time and spatial constrains. Leukocyte-induced H2O2 release is a rapid event, lasting a few seconds62, and is spatially confined to a range that does not exceed a few hundred µm63 (Fig. 7b). Our data, including those obtained by genetic or pharmacological manipulation of NOXs, are consistent with previous observations. Macrophages express solely NOX240, while Schwann cells, which potentially express mRNAs for NOX1, NOX2, and NOX4, apparently express only the NOX1 protein. Since NOX1, but not NOX2 or NOX4, inhibitors or AS-ODNs attenuated neuroinflammation and allodynia, it is possible to propose that Schwann cell TRPA1 activates intracellular pathways, including Ca2+ transients, resulting in NOX1-dependent release of oxidant molecules. Furthermore, the prominent role of NOX1, but not of NOX2, in generating allodynia excludes phagocyte-derived oxidative burst in the final activation of nociceptor TRPA1.
The most parsimonious explanation of the present results is that oxidative stress generated by Schwann cell TRPA1/NOX1 has bidirectional effects. The inwardly released H2O2 targets TRPA1 on adjacent nociceptor nerve fibers in a paracrine fashion to sustain allodynia. The outwardly released H2O2 promotes the final part (about 200 µm) of the journey of macrophages, which, deriving from the blood stream, slowly accumulate into the perineural space following the CCL2 gradient. Thereafter, following the Schwann cell-derived oxidative stress gradient, macrophages rapidly pass across the perineurium to enter the damaged nerve trunk (Fig. 9). TRPA1 has been identified in oligodendrocytes, with possible detrimental roles in ischemia and neurodegeneration64. Herein, we extend this observation to Schwann cells, the peripheral analogs of oligodendrocytes, which, via TRPA1, orchestrate neuroinflammation and ensuing neuropathic pain. Amelioration of neuropathic pain by currently developed TRPA1 antagonists may derive from their ability to attenuate macrophage-dependent neuroinflammation.
Methods
Animals and drugs
In vivo experiments and tissue collection were carried out according to the European Union (EU) guidelines for animal care procedures and the Italian legislation (DLgs 26/2014) application of the EU Directive 2010/63/EU. Studies were conducted under University of Florence research permits #204/2012-B and #194/2015-PR. C57BL/6 mice (male, 20–25 g, 5–6 weeks; Envigo, Milan, Italy), littermate wild type (Trpa1 +/+) and TRPA1-deficient (Trpa1 −/−) mice (25–30 g, 5–8 weeks), generated by heterozygotes on a C57BL/6 background (B6.129P-Trpa1 tm1Kykw/J; Jackson Laboratories, Bar Harbor, ME, USA)65, wild type (Trpv4 +/+) and TRPV4-deficient (Trpv4 −/−) mice (25–30 g, 5–8 weeks), generated by heterozygotes on a C57BL/6 background66 and TRPV1-deficient mice (Trpv1 −/−; B6.129 × 1-Trpv1 tm1Jul/J) backcrossed with C57BL/6 mice (Trpv1 +/+) for at least 10 generations (Jackson Laboratories, Bar Harbor, ME, USA; 25–30 g, 5–8 weeks), were used. B6.Cg-Tg(Plp1-CreERT)3Pop/J mice (Plp1-Cre ERT, Stock No: 005975), expressing a tamoxifen-inducible Cre in myelinating cells (Plp1, proteolipid protein myelin 1)67, and 129S-Trpa1tm2Kykw/J mice (floxed TRPA1, Trpa1 fl/fl, Stock No: 008650), which possess loxP sites on either side of the S5/S6 transmembrane domains of the Trpa1 gene, were obtained from Jackson Laboratories (Bar Harbor, ME, USA). To generate mice in which the Trpa1 gene was conditionally silenced in Schwann cells/oligodendrocytes homozygous Trpa1 fl/fl mice were crossed with hemizygous Plp1-Cre ERT mice.
The progeny was genotyped by standard PCR for Cre ERT and Trpa1 alleles (PCR protocol ID 005975; PCR protocol ID 008650, Jackson Laboratories, Bar Harbor, ME, USA). Both positive or negative mice to Cre ERT and homozygous for floxed Trpa1 (Plp1-Cre ERT ;Trpa1 fl/fl and Plp1-Cre ERT− ;Trpa1 fl/fl, respectively) were treated with intraperitoneal (i.p.) 4-hydroxytamoxifen (1 mg 100 µl−1 in corn oil, once a day, for 5 consecutive days)67 resulting in Cre-mediated ablation of Trpa1 in PLP-expressing Schwann cells/oligodendrocytes. Successful Cre-driven deletion of TRPA1 mRNA was confirmed by RT-qPCR. Mice negative to CreERT (Plp1-Cre ERT− ;Trpa1 fl/fl) were used as control. Mice were allocated to pSNL/sham surgery or underwent to functional experiments (AITC challenge) 15 days after the last tamoxifen injection. Tissues or cells used to assess the level of TRPA1 expression were collected also 15 days after the last tamoxifen injection.
Animals were housed in a temperature- and humidity-controlled vivarium (12 h dark/light cycle, free access to food and water, 10 animals per cage). Behavioral experiments were performed, after 1 h of animal acclimation, in a quiet, temperature-controlled room (20–22 °C) between 9 a.m. and 5 p.m. The sample sizes chosen for animal groups were adequately powered to observe the effects based on both our past experience in similar experimental settings and data published by others. Some animals were excluded because of failure to reach the training criteria or mortality. Exclusions for training were based on scores established before starting experiments and routinely used. Animals were randomized to vehicle(s) or treatment(s) administration. The assessors (scientists who performed in vitro and in vivo tests), were blinded to the identity (genetic background or allocation to treatment group) of the animals. Identity of the animals was unmasked to assessors only after data collection. Every effort has been made to minimize the discomfort and pain of the animals in each phase of the study. Animals were euthanized with inhaled CO2 plus 10–50% O2. HC-030031 (2-(1,3-Dimethyl-2,6-dioxo-1,2,3,6-tetrahydro-7H-purin-7-yl)-N-(4-isopropylphenyl) acetamide) was synthesized as previously described68. If not otherwise indicated, reagents were obtained from Sigma-Aldrich (Milan, Italy).
Partial ligation of the sciatic nerve
Partial ligation of the sciatic nerve (pSNL) was performed in C57BL/6, in Trpa1 +/+ or Trpa1 −/−, in Trpv1 +/+ or Trpv1 −/−, in Trpv4 +/+ or Trpv4 −/− and in Plp1-Cre ERT ;Trpa1 fl/fl or control mice as previously described69. Briefly, mice were anesthetized with i.p. injection of a mixture of ketamine (90 mg kg−1) and xylazine (3 mg kg−1), the right sciatic nerve was exposed, and a partial ligation was made by tying one-third to one half of the dorsal portion of the sciatic nerve. In sham-operated mice, used as controls, the right sciatic nerve was exposed, but not ligated. Mice were monitored, adequately rehydrated, and maintained in a controlled temperature (37 °C) until fully recovered from anesthesia.
Experimental design
C57BL/6, Trpa1 +/+ or Trpa1 −/−, Trpv1 +/+ or Trpv1 −/−, Trpv4 +/+, or Trpv4 −/− and Plp1-Cre ERT ;Trpa1 fl/fl or control mice were randomly allocated to pSNL or sham surgery. Ten days after surgery, C57BL/6 mice were randomly allocated to the treatment group with i.p. administration of TRPA1 selective antagonists, HC-030031 (HC03) or A-967079 [(1E,3E)-1-(4-Fluorophenyl)-2-methyl-1-penten-3-one oxime] (A96) and antioxidants, α-lipoic acid (αLA) or phenyl-N-tert-butylnitrone (PBN) (all 100 mg kg−1), the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX) inhibitors, GKT137831 [2-(2-chlorophenyl)-4-[3-(dimethylamino)phenyl]-5-methyl-1H-pyrazolo[4,3-c]pyridine-3,6(2H,5H)-dione] (GKT, 60 mg kg−1, NOX1/4 selective), 2-acetylphenothiazine (ML171, 60 mg kg−1, NOX1 selective), gp91ds-tat peptide (10 mg kg−1, NOX2 selective) or vehicles (all 4% dimethyl sulfoxide, DMSO, 4% tween 80 in isotonic saline, NaCl 0.9%; PBN was dissolved in NaCl 0.9%).
In different experiments, at days 6, 7, 8 and 9 after surgery, pSNL or sham C57BL/6 mice were randomly allocated to the groups receiving perineural (p.n.) treatment (10 nmol 10 μl−1) of TRPA1, NOX1, NOX2 and NOX4 antisense (AS), mismatch (MM) oligonucleotide (ODN) or scrambled control. TRPA1 AS-ODN sequence was 5′-TATCGCTCCACATTGCTAC-3′, TRPA1 MM-ODN sequence was 5′-ATTCGCCTCACATTGTCAC-3′44, NOX1 AS-ODN sequence was 5′-TTAACCAGCCAGTTTCCCATTG-3′, NOX2 AS-ODN sequence was 5′-TTCACAGCCCAGTTCCCCATG T-3′, NOX4 AS-ODN sequence was 5′-TTGGCCAGCCAG CTCCTCCA-3′, NOX1 and NOX2 scrambled p47phox sequence control was 5′-TGAGGCTCC GTCCGCTGGAGCG-3′, NOX4 scrambled sequence was 5′-CGTCACGCTCAGCTCACCGT-3′70,71. Perineural injections were performed as previously reported11,72. Briefly, compounds were injected into the region surrounding the sciatic nerve at high thigh level of right hind limbs without skin incision in a volume of 10 μl using a microsyringe fitted with a 30-gauge needle. Other mice were treated with intratechal (i.t.) treatment (5 nmol 5 μl−1) of TRPA1 AS/MM-ODN44. Phosphorothioate-modified ODNs, which is reported to be more effective in the rate of uptake and more specific in binding the target sequence, generally increasing ODNs potency73 were used for both perineural and intrathecal treatments. Some mice treated with TRPA1 AS/MM-ODN (p.n. or i.t.) were tested for the acute nociceptive response to AITC (20 nmol 10 μl−1, p.n.) or CPS (1 nmol 10 μl−1, p.n.).
pSNL/sham C57BL/6 mice were treated (i.p.) with an antibody directed to the chemokine (C–C motif) ligand 2 (CCL2) (CCL2-Ab) (R&D system, Minneapolis, USA) with two different protocols. In the first, the CCL2-Ab or vehicle (IgG2B Isotype Control, R&D system, Minneapolis, USA) were given at day 7, 8 and 9 (40 µg 200 µl−1, i.p.) after surgery. In the second, one single triple-dose (120 µg 200 µl−1, i.p.) or vehicle were given at day 10 after surgery. To transiently deplete the monocyte/macrophage population, pSNL or sham C57BL/6 mice received liposome-encapsulated clodronate (LCL, 5 mg ml−1 i.p., ClodronateLiposomes.com Amsterdam, The Netherlands) or vehicle (liposome-encapsulated phosphate buffer saline) at day 7, 8 and 9 after surgery.
C57BL/6 mice received subcutaneous (s.c.) resiniferatoxin (RTX, 50 μg kg−1) or its vehicle (10% ethanol and 10% tween 80 in isotonic saline)55. pSNL or sham surgery was carried out five days after RTX treatment when the eye wiping test responses to both capsaicin (CPS, 1 nmol 5 µl−1) and AITC (20 nmol 5 µl−1) were abolished (Supplementary Fig. 4A). In different experiments, perineural (p.n.) CCL2 was given to Trpa1 +/+ or Trpa1 −/ mice and C57BL/6 mice (0.1, 0.5 and 1 µg 10 µl−1), which after 2 h received i.p. HC03 or αLA (both 100 mg kg−1) or vehicles. Some C57BL/6 mice were treated with LCL (5 mg ml−1 i.p.) or vehicle for 3 consecutive days and then CCL2 (1 µg 10 µl−1, p.n.) was administered.
Assessment of pain-like behaviors
Mechanical allodynia, cold hypersensitivity and thermal hyperalgesia were assessed before (baseline) and 3, 7, 10, 20 days after pSNL or sham surgery in C57BL/6, Trpa1 +/+, Trpa1 −/−, Trpv1 +/+, Trpv1 −/−, Trpv4 +/+, Trpv4 −/−, Plp1-Cre ERT ;Trpa1 fl/fl, and control mice. In C57BL/6 mice, mechanical allodynia was assessed before and after the various pharmacological interventions at different time points. To measure nociceptive responses, mice were placed in a Plexiglas chamber immediately after p.n. injection of AITC or CPS, or intraplantar injection of AITC (10 nmol 20 µl−1) and the total time spent licking and lifting (nociception time, s) the injected right hind limb and paw was recorded for 5 min. Eye wiping was assayed after ocular instillation of CPS or AITC or vehicles (2 and 4% DMSO, respectively)47. Mechanical threshold was measured by the up-and-down paradigm74,75. Cold hypersensitivity was assessed by measuring the acute nociceptive response to acetone-evoked evaporative cooling24.
Thermal hyperalgesia was measured by exposing the mid plantar surface of the hind paw to a beam of radiant heat through a transparent surface, using a plantar analgesimeter for paw stimulation (Ugo Basile, Comerio, Italy). Paw withdrawal latency was recorded as the time from onset of the thermal stimulus to the withdrawal response. In each paw mean withdrawal latency of three measures was calculated. The interval between trials on the same paw was at least 5 min. The cutoff latency was set at 20 s to avoid tissue damage in case of failure to remove the paw.
CCL2 enzyme-linked immunosorbent assay
The CCL2 content in the sciatic nerve (ipsilateral to the surgery) was measured in Trpa1 +/+, Trpa1 −/− and C57BL/6 mice 10 days after pSNL/sham surgery and 60 min after treatment (C57BL/6 mice) with HC03, αLA, or their vehicles and CCL2-Ab or IgG2B control (single and triple administration) or LCL, by using a mouse CCL2/MCP-1 quantikine ELISA Kit (R&D system, Minneapolis, USA). Samples were homogenized at 4 °C in PBS containing a protease inhibitor cocktail tablet (Roche Diagnostics, Mannheim, Germany). The homogenate was then centrifuged (10,000 × g, 20 min, 4 °C); supernatants were collected and assayed according to the manufacturer’s instructions. The concentration of CCL2 was expressed in pg/mg of total protein content76.
Measurement of H2O2 content in tissue
The H2O2 content of the sciatic nerve (ipsilateral to the surgery) was firstly determined in C57BL/6 mice at day 3, 7, 10 and 20 after pSNL/sham surgery. Then, all the measurements were performed at day 10 after pSNL/sham surgery and 60 min after treatment with HC03, A96, αLA, GKT, PBN, ML171, gp91ds-tat peptide, or anti-CCL2 antibody. In Trpa1 +/+, Trpa1 −/−, Plp1-Cre ERT ;Trpa1 fl/fl, control, and C57BL/6 mice pretreated with RTX or treated with TRPA1, NOX1, NOX2, NOX4 AS/MM-ODN, anti-CCL2 antibody or LCL, H2O2 content was assessed 10 days after pSNL/sham surgery. H2O2 was determined by using the Amplex Red® assay (Invitrogen, Milan, Italy). Briefly, sciatic nerves were rapidly removed and placed into modified Krebs/HEPES buffer (composition in mmol l−1: 99.01 NaCl, 4.69 KCl, 2.50 CaCl2, 1.20 MgSO4, 1.03 KH2PO4, 25.0 NaHCO3, 20.0 Na-HEPES, and 5.6 glucose [pH 7.4]). Samples were minced and incubated with Amplex red (100 µM) and HRP (1 U ml−1) (60 min, 37 °C) in modified Krebs/HEPES buffer protected from light77. Fluorescence excitation and emission were at 540 and 590 nm, respectively. H2O2 production was calculated using H2O2 standard and expressed as µmol l−1 of mg of dry tissue.
Cell culture
HEK293 cells stably transfected with the cDNA for human TRPA1 (hTRPA1-HEK293, kindly donated by A.H. Morice, University of Hull, Hull, UK) and naive untransfected HEK293 cells (American Type Culture Collection, Manassas, VA, USA; ATCC® CRL-1573™), were cultured as previously described78. For all cell lines, the cells were used when received without further authentication. Schwann cells were isolated from sciatic nerves of C57BL/6, Trpa1 +/+, Trpa1 −/−, Plp1-Cre ERT ;Trpa1 fl/fl, or control mice. Briefly, the epineurium was removed, and nerve explants were divided into 1 mm segments and dissociated enzymatically using collagenase (0.05%) and hyaluronidase (0.1%) in HBSS (2 h, 37 °C). Cells were collected by centrifugation (800×rpm, 10 min, RT) and the pellet was resuspended and cultured in DMEM containing: 10% FCS, 2 mM l-glutamine, 100 U ml−1 penicillin/100 mg ml−1 streptomycin or 50 mg ml−1 gentamycin. Three days later, cytosine arabinoside (10 mM) was added to remove fibroblasts. To enhance Schwann cell proliferation, forskolin (2 µM) was added to the culturing medium79.
To obtain cultured peritoneal macrophages, C57BL/6 mice were i.p. injected with thioglycolate (3%, 1 ml). After 3 days, cells were harvested from sacrificed animals by peritoneal lavage for a total of 10 ml PBS and centrifuged (400 × g, 10 min, 4 °C). Cells were cultured in DMEM supplemented with 10% FBS. After incubation at 37 °C for 24 h, non-adherent cells were removed by repeated washing80. Before each experiment, cells were tested with specific kits for cells mycoplasma contamination based on PCR (EMK090020, N-GARDE kit, Euroclone, Milan, Italy).
Measurement of H2O2 released from cells
H2O2 was determined by using the Amplex Red assay (Invitrogen, Milan, Italy). hTRPA1-HEK293 or naive untransfected HEK293, Schwann cells or peritoneal macrophages were plated in 96-well clear bottom black (5 × 105 cells well−1) and maintained in 5% CO2 and 95% O2 (24 h, 37 °C). The cultured medium was replaced with Krebs-Ringer phosphate (KRP, composition in mmol l−1: 2 CaCl2; 5.4 KCl; 0.4 MgSO4; 135 NaCl; 10 D-glucose; 10 HEPES [pH 7.4]) added with HC03, A96 (both 30 µM) or vehicle (0.3% DMSO) for 10 min at RT. Peritoneal macrophages were incubated with GKT (100 nM) or gp91ds-tat (0.1–100 nM) for 20 min. hTRPA1-HEK293, naive HEK293 or Schwann cells were stimulated with AITC (10 and 100 µM, respectively), H2O2 (200 nM) or their vehicle (0.01% DMSO or KRP, respectively), peritoneal macrophages were stimulated with phorbol myristate acetate (PMA, 20 nM) or vehicle (0.00001% DMSO diluted in KRP) added with Amplex red (50 µM) and HRP (1 U ml−1), and maintained for 30 min at RT protected from light. Some experiments in Schwann cells were performed in Ca2+-free KRP containing EDTA (1 mM). Signal was detected 60 min (hTRPA1-HEK293/naïve-HEK293) or 40, 50, and 60 min (Schwann cells) after exposure to the stimulus. H2O2 release was calculated using H2O2 standards and expressed as nmol l−1.
Calcium imaging
Schwann cells and macrophages were plated on glass coated (poly-l-lysine, 8.3 µM) coverslips and intracellular calcium response was measured as previously reported81. Schwann cells were challenged with the selective TRPA1 agonist, AITC (1 mM), and the selective TRPV1 and TRPV4 agonists, CPS (0.5 µM) and GSK1016790A (GSK, 50 nM), respectively. Results are expressed as % increase in Ratio340/380 over baseline normalized to the maximum effect induced by ionomycin (5 µM) added at the end of each experiment (% change in R340/380). Macrophages were stimulated with fresh medium containing 100 ng ml−1 LPS, then incubated at 37 °C for 6, 12, 18, 24, 36 and 48 h, before being challenged with AITC (1 mM) and ionomycin (5 µM). Results are expressed as Ratio340/380.
Immunofluorescence and confocal microscopy
Anesthetized mice were transcardially perfused with PBS, followed by 4% paraformaldehyde. The sciatic nerves (ipsilateral to the surgery) or dorsal root ganglia (DRGs, L4-L6) were removed, postfixed for 24 h, and paraffin embedded or cryoprotected overnight at 4 °C in 30% sucrose until cryosectioning. Cryosections (10 µm) were stained with hematoxylin and eosin (H&E) for histological examination or incubated with the following primary antibodies: F4/80 (MA516624, rat monoclonal (Cl:A3-1), 1:50, Thermo Fisher Scientific, Rockford, USA), CD8 (ab22378, rat monoclonal (YTS169.4), 1:200, Abcam, Cambridge, UK) and Ly6G (ab25377, rat monoclonal (RB6-8C5), 1:200, Abcam, Cambridge, UK) (1 h, RT), diluted in fresh blocking solution (PBS, pH 7.4, 10% normal goat serum, NGS). Formalin fixed paraffin-embedded sections (5 µm) were incubated with the following primary antibodies: protein gene product 9.5 (PGP9.5, ab8189, mouse monoclonal [13C4/I3C4], 1:600, Abcam, Cambridge, UK), TRPA1 (ab58844, rabbit polyclonal, 1:400, Abcam, Cambridge, UK), S100 (ab14849, mouse monoclonal (4B3), 1:300, Abcam, Cambridge, UK), SOX10 (ab216020, mouse monoclonal (SOX10/1074), 1:300, Abcam, Cambridge, UK), 4- HNE (ab48506, mouse monoclonal (HNEJ-2), 1:40, Abcam, Cambridge, UK) or NOX1 (ab131088, rabbit polyclonal, 1:250, Abcam, Cambridge, UK) (1 h, RT) diluted in antibody diluent (Roche Diagnostics, Mannheim, Germany). Sections were then incubated with fluorescent secondary antibodies: polyclonal Alexa Fluor 488, polyclonal Alexa Fluor 594, polyclonal Alexa Fluor 546, and polyclonal Alexa Fluor 647 (1:600, Invitrogen, Milan, Italy) (2 h, RT, protected from light). Sections were coverslipped using a water-based mounting medium with 4′6′-diamidino-2-phenylindole (DAPI) (Abcam, Cambridge, UK). The analysis of negative controls (non-immune serum) was simultaneously performed to exclude the presence of non-specific immunofluorescent staining, cross-immunostaining, or fluorescence bleed-through. Tissues were visualized and digital images were captured using an Olympus BX51 or confocal scan a LEICA TCS SP5. High power 3D renderings of the images were obtained using ImageJ 3D viewer.
Direct counting of F4/80+ cells was performed in 104 μm2 boxes in the sciatic nerve (inside the nerve trunk) in: Trpa1 +/+, Trpa1 −/−, Plp1-Cre ERT ;Trpa1 fl/fl, and control mice, 10 days after pSNL/sham surgery, and in pSNL/sham C57BL/6 mice at day 10 after surgery following treatment with RTX, CCL2-Ab, LCL and TRPA1, NOX1, NOX2, NOX4 AS/MM-ODN and at different time points after administration of A96, αLA, GKT, PBN, ML171, gp91ds-tat peptide or CCL2-Ab. In some samples, direct counting of F4/80+ cells was performed in pSNL/sham C57BL/6 mice in 104 μm2 boxes outside the sciatic nerve trunk at two different distances (~0–200 µm and ~ 200–400 µm from the epineurium) before and after HC03 or αLA. Direct counting of CD8+ and Ly6G+ cells was performed in 104 μm2 boxes in the sciatic nerve (inside the nerve trunk) in pSNL/sham C57BL/6 mice at day 10 after surgery. The counting was performed by an operator blinded to drug treatment and timing.
TRPA1 staining in DRG was evaluated as the fluorescence intensity measured by an image processing software (ImageJ 1.32J, National Institutes of Health, Bethesda, USA). The Pearson correlation (Rcoloc) value for TRPA1 and S100 in the colocalization studies were calculated using the colocalization Plugin of the ImageJ software82. Schwann cells were grown on glass coated (poly-l-lysine, 8.3 µM) coverslips and cultured for 2–3 days before being used for staining. Cells were then fixed in ice-cold methanol/acetone (5 min at −20 °C), washed with PBS and blocked with NGS (10%) (1 h, RT). The cells were then incubated with the primary antibodies (TRPA1, ab58844, rabbit polyclonal, 1:400; S100, ab14849, mouse monoclonal (4B3), 1:300, SOX10, ab216020, mouse monoclonal (SOX10/1074), 1:300, Abcam, Cambridge, UK) (1 h, RT). Cells were then incubated with fluorescent secondary antibodies (1:600, polyclonal Alexa Fluor 488, and polyclonal Alexa Fluor 594, Invitrogen, Milan, Italy) (2 h, RT) and mounted using water-based mounting medium DAPI (Abcam, Cambridge, UK). Cells were visualized and digital images were captured using an Olympus BX51.
Real-time PCR
RNA was extracted from cultured Schwann cells or peritoneal macrophages obtained from C57BL/6 mice, and from the sciatic nerve or L4-L6 DRGs (ipsilateral to the surgery) of pSNL C57BL/6 mice after TRPA1, NOX1 and NOX4 scrambled/AS/MM-ODN (i.t. or p.n.) To avoid the confounding contribution of NOX2 mRNA from invading macrophages, for this analysis RNA was extracted from the sciatic nerve (ipsilateral to the surgery) of sham C57BL/6 mice. RNA was also extracted from sciatic nerve of Plp1-Cre ERT ;Trpa1 fl/fl and control. The standard Trizol extraction method was used. RNA concentration and purity was assessed spectrophotometrically by measuring the absorbance at 260 and 280 nm. The RNA (100 ng) was reverse-transcribed using the iScript cDNA Synthesis kit (Bio-Rad, Hercules, USA) according to the manufacturer’s protocol. For relative quantification of mRNA, real-time PCR was performed on Rotor Gene Q (Qiagen, Hilden, GE). The sets of primers-probes were as follows: 18S-FW (forward): 5′-CGCGGTTCTATTTTGTTGGT-3′, 18S-RE (reverse): 5′-AGTCGGCATCGTTTATGGTC-3′ (NCBI Ref Seq: NR_003278.3); TRPA1-FW: 5′-CAGGATGCTACGGTTTTTTCATTACT-3′, TRPA1-RE: 5′-GCATGTGTCAATGTTTGGTACTTCT-3′ (NCBI Ref Seq: NM_177781.4); S100-FW: 5′-TGGATGAAAACGGAGATGGGG-3′, S100-RE: 5′-ACAGACTGTGCTCAACTGGT-3′ (NCBI Ref Seq: NM_011309); SOX10-FW: 5′-AGATCCAGTTCCGTGTCAATAA-3′, SOX10-RE:5′GCGAGAAGAAGGCTAGGTG-3′ (NCBI Ref Seq: U70441.1); NOX1-FW: 5′-CACTCACCAATGCCCAGGAT-3′, NOX1-RE: 5′-TGGAAGCAAAGGGAGTGACC-3′ (NCBI Ref Seq: NM_172203.2); NOX2-FW: 5′-GAGGTTGGTTCGGTTTTGGC-3′, NOX2-RE: 5′-CAGGAGCAGAGGTCAGTGTG-3′ (NCBI Ref Seq: NM_007807.5); NOX4-FW: 5′-TGTTGGGCCTAGGATTGTGT-3′, NOX4-RE: 5′-TCCTGCTAGGGACCTTCTGT-3′ (NCBI Ref Seq: NM_015760.5); F4/80-FW: 5′-CCCAGCTTATGCCACCTGCA-3′, F4/80-RE: 5′-TCCAGGCCCTGGAACATTGG-3′ (NCBI Ref Seq: NC_000083.6); Floxed TRPA1-FW: 5′-GGGCAGCTTATTGCCTTCAC-3′, Floxed TRPA1-RE: 5′-TTGCGTAAGTACCAGAGTGGC-3′ (NCBI Ref Seq: NM_177781.4)
The chosen reference gene was the 18S. The SsoAdvanced™ Universal SYBR Green Supermix (Bio-Rad, Hercules, USA) was used for amplification, and the cycling conditions were the following: samples were heated to 95 °C for 1 min followed by 40 cycles of 95 °C for 10 s, and 65 °C for 20 s. PCR reaction was carried out in triplicate. Relative expression of TRPA1 mRNA was calculated using the 2−Δ(ΔCT) comparative method, with each gene normalized against the internal endogenous reference 18S gene for the same sample.
Protein extraction and western immunoblot assay
Cultured Schwann cells and DRGs (L4–L6) neurons were homogenized in a lysis buffer containing (mM): 50 Tris, 150 NaCl, 2 EGTA, 100 NaF, 1 Na3VO4, 1% Nonidet P40 (pH 7.5) and complete protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany). Lysates were centrifuged at 14,000 × g at 4 °C for 45 min. Protein concentration in supernatants was determined using DC protein assay (Bio-Rad, Milan, Italy). Samples with equal amounts of proteins (20 µg) were then separated by NuPAGE 4–12% Bis–Tris gel electrophoresis (Life Technologies, Carlsbad, USA), and the resolved proteins were transferred to a polyvinylidenedifluoride membrane (Merck Millipore Billerica, USA). Membranes were incubated with 5% dry milk in Tris buffer containing 0.1% Tween 20 (TBST; 20 mM Tris at pH 7.5, 150 mM NaCl) for 1 h at RT, and incubated with TRPA1 (NB110-40763, rabbit polyclonal, 1:200, Novus Biologicals, Littleton, USA) or β-actin (mouse monoclonal primary antibody, 1:6000, Thermo Scientific, Rockford, USA) antibodies, at 4 °C overnight. Membranes were then probed with goat anti-mouse or donkey anti-rabbit IgG conjugated with horseradish peroxidase (HRPO) (Bethyl Laboratories Inc., Cambridge, UK) for 1 h at RT. Finally, membranes were washed three times with TBST, and bound antibodies were detected using chemiluminescence reagents (ECL, Pierce, Thermo Scientific, Rockford, USA). The density of specific bands was measured using an image processing program (ImageJ 1.32J, National Institutes of Health, Bethesda, USA) and normalized to β-actin24. The uncropped scan of the blot is reported in the Supplementary Fig. 7.
Live animal imaging
Macrophage localization in vivo was obtained by NIR imaging of the fluorescent label macrophage mice by using PhotonImager (Biospace Laboratory, Paris, France)83. Mouse thioglycollate-elicited peritoneal macrophages were harvested (up to 250 × 106 cells per ml) and incubated for 15 min at RT with VivoTrack 680 (PerkinElmer, Inc., Waltham, USA), dissolved in sterile PBS, washed, centrifuged (400 × g, 10 min) and diluted to a final concentration of 5 × 106 cells 40 μl−1. Retro-orbital vein injection (40 μl) of labeled macrophages was performed in pSNL/sham C57BL/6 mice at day 9 after surgery. Twenty-four h later, anesthetized mice were put inside the pre-heated chamber. NIR imaging was performed before and 1 and 3 h after HC03 (100 mg kg−1, i.p.) administration. Images were acquired with Photo Acquisition software and processed with M3 Vision software (Biospace Laboratory, Paris, France). The NIR pixel area was measured by ImageJ 1.32J from a region of the same size over the sciatic nerve, identifying an area of interest (ROI) around the fluorescent signal evident in the pSNL. That ROI perimeter was then reported to other images derived from different experimental settings. Data were expressed as NIR pixel area/ROI pixel area.
Statistical analysis
Statistical analysis was performed by the unpaired two-tailed Student’s t-test for comparisons between two groups and the one-way or two-way ANOVA followed by the post hoc Bonferroni’s test for comparisons of multiple groups. Sample sizes for experiments were determined based on previous studies, to ensure appropriate statistical power. P < 0.05 was considered statistically significant (GraphPad Prism version 5.00, La Jolla, USA).
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
Electronic supplementary material
Acknowledgements
We thank A.H. Morice (University of Hull, UK) for the hTRPA1-HEK293 cells and D. Preti (University of Ferrara, Italy) for providing HC-030031. We also thank Mary K. Lokken for her expert English revision. This work was supported by grants from Istituto Toscano Tumori (ITT), grant 2014 and Regione Toscana, grant Nutraceuticals 2014, ‘POFCADT’ (to P. Geppetti).
Author contributions
F.D.L., R.N., R.P., N.W.B. and P.G. designed experiments and interpreted results. F.D.L., R.N., S.M., M.C.G., I.M.M., D.R.D.I., J.F., S.L.P., S.B., G.T. and D.S.M.d.A. performed experiments. F.D.L., R.N., R.P., N.W.B. and P.G. wrote the manuscript.
Competing interests
The authors declare no competing financial interests.
Footnotes
Francesco De Logu and Romina Nassini contributed equally to this work.
Electronic supplementary material
Supplementary Information accompanies this paper at doi:10.1038/s41467-017-01739-2.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Jensen TS, et al. A new definition of neuropathic pain. Pain. 2011;152:2204–2205. doi: 10.1016/j.pain.2011.06.017. [DOI] [PubMed] [Google Scholar]
- 2.van Hecke O, Austin SK, Khan RA, Smith BH, Torrance N. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain. 2014;155:654–662. doi: 10.1016/j.pain.2013.11.013. [DOI] [PubMed] [Google Scholar]
- 3.Gaudet AD, Popovich PG, Ramer MS. Wallerian degeneration: gaining perspective on inflammatory events after peripheral nerve injury. J. Neuroinflammation. 2011;8:110. doi: 10.1186/1742-2094-8-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ramer MS, French GD, Bisby MA. Wallerian degeneration is required for both neuropathic pain and sympathetic sprouting into the DRG. Pain. 1997;72:71–78. doi: 10.1016/S0304-3959(97)00019-5. [DOI] [PubMed] [Google Scholar]
- 5.von Hehn CA, Baron R, Woolf CJ. Deconstructing the neuropathic pain phenotype to reveal neural mechanisms. Neuron. 2012;73:638–652. doi: 10.1016/j.neuron.2012.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Komori T, Morikawa Y, Inada T, Hisaoka T, Senba E. Site-specific subtypes of macrophages recruited after peripheral nerve injury. Neuroreport. 2011;22:911–917. doi: 10.1097/WNR.0b013e32834cd76a. [DOI] [PubMed] [Google Scholar]
- 7.Abbadie C, et al. Impaired neuropathic pain responses in mice lacking the chemokine receptor CCR2. Proc. Natl Acad. Sci. USA. 2003;100:7947–7952. doi: 10.1073/pnas.1331358100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Van Steenwinckel J, et al. Stromal cell-derived CCL2 drives neuropathic pain states through myeloid cell infiltration in injured nerve. Brain Behav. Immun. 2014;45:198–210. doi: 10.1016/j.bbi.2014.10.016. [DOI] [PubMed] [Google Scholar]
- 9.Kim CF, Moalem-Taylor G. Detailed characterization of neuro-immune responses following neuropathic injury in mice. Brain Res. 2011;1405:95–108. doi: 10.1016/j.brainres.2011.06.022. [DOI] [PubMed] [Google Scholar]
- 10.White FA, Jung H, Miller RJ. Chemokines and the pathophysiology of neuropathic pain. Proc. Natl Acad. Sci. USA. 2007;104:20151–20158. doi: 10.1073/pnas.0709250104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kiguchi N, Kobayashi Y, Saika F, Kishioka S. Epigenetic upregulation of CCL2 and CCL3 via histone modifications in infiltrating macrophages after peripheral nerve injury. Cytokine. 2013;64:666–672. doi: 10.1016/j.cyto.2013.09.019. [DOI] [PubMed] [Google Scholar]
- 12.Khalil Z, Liu T, Helme RD. Free radicals contribute to the reduction in peripheral vascular responses and the maintenance of thermal hyperalgesia in rats with chronic constriction injury. Pain. 1999;79:31–37. doi: 10.1016/S0304-3959(98)00143-2. [DOI] [PubMed] [Google Scholar]
- 13.Kim HK, et al. Reactive oxygen species (ROS) play an important role in a rat model of neuropathic pain. Pain. 2004;111:116–124. doi: 10.1016/j.pain.2004.06.008. [DOI] [PubMed] [Google Scholar]
- 14.Story GM, et al. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell. 2003;112:819–829. doi: 10.1016/S0092-8674(03)00158-2. [DOI] [PubMed] [Google Scholar]
- 15.Nassini R, Materazzi S, Benemei S, Geppetti P. The TRPA1 channel in inflammatory and neuropathic pain and migraine. Rev. Physiol. Biochem. Pharmacol. 2014;167:1–43. doi: 10.1007/112_2014_18. [DOI] [PubMed] [Google Scholar]
- 16.Jordt SE, et al. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature. 2004;427:260–265. doi: 10.1038/nature02282. [DOI] [PubMed] [Google Scholar]
- 17.Bautista DM, et al. Pungent products from garlic activate the sensory ion channel TRPA1. Proc. Natl Acad. Sci. USA. 2005;102:12248–12252. doi: 10.1073/pnas.0505356102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mori Y, et al. Redox-sensitive transient receptor potential channels in oxygen sensing and adaptation. Pflugers Arch. 2016;468:85–97. doi: 10.1007/s00424-015-1716-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Andersson DA, Gentry C, Moss S, Bevan S. Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. J. Neurosci. 2008;28:2485–2494. doi: 10.1523/JNEUROSCI.5369-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sawada Y, Hosokawa H, Matsumura K, Kobayashi S. Activation of transient receptor potential ankyrin 1 by hydrogen peroxide. Eur. J. Neurosci. 2008;27:1131–1142. doi: 10.1111/j.1460-9568.2008.06093.x. [DOI] [PubMed] [Google Scholar]
- 21.Taylor-Clark TE, Ghatta S, Bettner W, Undem BJ. Nitrooleic acid, an endogenous product of nitrative stress, activates nociceptive sensory nerves via the direct activation of TRPA1. Mol. Pharmacol. 2009;75:820–829. doi: 10.1124/mol.108.054445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Trevisani M, et al. 4-Hydroxynonenal, an endogenous aldehyde, causes pain and neurogenic inflammation through activation of the irritant receptor TRPA1. Proc. Natl Acad. Sci. USA. 2007;104:13519–13524. doi: 10.1073/pnas.0705923104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bautista DM, et al. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell. 2006;124:1269–1282. doi: 10.1016/j.cell.2006.02.023. [DOI] [PubMed] [Google Scholar]
- 24.Trevisan G, et al. Novel therapeutic strategy to prevent chemotherapy-induced persistent sensory neuropathy by TRPA1 blockade. Cancer Res. 2013;73:3120–3131. doi: 10.1158/0008-5472.CAN-12-4370. [DOI] [PubMed] [Google Scholar]
- 25.Obata K, et al. TRPA1 induced in sensory neurons contributes to cold hyperalgesia after inflammation and nerve injury. J. Clin. Invest. 2005;115:2393–2401. doi: 10.1172/JCI25437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Katsura H, et al. Antisense knock down of TRPA1, but not TRPM8, alleviates cold hyperalgesia after spinal nerve ligation in rats. Exp. Neurol. 2006;200:112–123. doi: 10.1016/j.expneurol.2006.01.031. [DOI] [PubMed] [Google Scholar]
- 27.Caspani O, Zurborg S, Labuz D, Heppenstall PA. The contribution of TRPM8 and TRPA1 channels to cold allodynia and neuropathic pain. PLoS ONE. 2009;4:e7383. doi: 10.1371/journal.pone.0007383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Eid SR, et al. HC-030031, a TRPA1 selective antagonist, attenuates inflammatory- and neuropathy-induced mechanical hypersensitivity. Mol. Pain. 2008;4:48. doi: 10.1186/1744-8069-4-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McNamara CR, et al. TRPA1 mediates formalin-induced pain. Proc. Natl Acad. Sci. USA. 2007;104:13525–13530. doi: 10.1073/pnas.0705924104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Trevisan G, et al. TRPA1 mediates trigeminal neuropathic pain in mice downstream of monocytes/macrophages and oxidative stress. Brain. 2016;139:1361–1377. doi: 10.1093/brain/aww038. [DOI] [PubMed] [Google Scholar]
- 31.Kim HK, et al. Reactive oxygen species (ROS) play an important role in a rat model of neuropathic pain. Pain. 2004;111:116–124. doi: 10.1016/j.pain.2004.06.008. [DOI] [PubMed] [Google Scholar]
- 32.Liu T, van Rooijen N, Tracey DJ. Depletion of macrophages reduces axonal degeneration and hyperalgesia following nerve injury. Pain. 2000;86:25–32. doi: 10.1016/S0304-3959(99)00306-1. [DOI] [PubMed] [Google Scholar]
- 33.Zhu X, Fujita M, Snyder LA, Okada H. Systemic delivery of neutralizing antibody targeting CCL2 for glioma therapy. J. Neurooncol. 2011;104:83–92. doi: 10.1007/s11060-010-0473-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Perkins NM, Tracey DJ. Hyperalgesia due to nerve injury: role of neutrophils. Neuroscience. 2000;101:745–757. doi: 10.1016/S0306-4522(00)00396-1. [DOI] [PubMed] [Google Scholar]
- 35.Hackel D, et al. The connection of monocytes and reactive oxygen species in pain. PLoS ONE. 2013;8:2013. doi: 10.1371/journal.pone.0063564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Szolcsanyi J, Szallasi A, Szallasi Z, Joo F, Blumberg PM. Resiniferatoxin: an ultrapotent selective modulator of capsaicin-sensitive primary afferent neurons. J. Pharmacol. Exp. Ther. 1990;255:923–928. [PubMed] [Google Scholar]
- 37.Pecze L, et al. Resiniferatoxin mediated ablation of TRPV1+neurons removes TRPA1 as well. Can. J. Neurol. Sci. 2009;36:234–241. doi: 10.1017/S0317167100006600. [DOI] [PubMed] [Google Scholar]
- 38.Nassini R, et al. Transient receptor potential ankyrin 1 channel localized to non-neuronal airway cells promotes non-neurogenic inflammation. PLoS ONE. 2012;7:14. doi: 10.1371/journal.pone.0042454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Campana WM. Schwann cells: activated peripheral glia and their role in neuropathic pain. Brain Behav. Immun. 2007;21:522–527. doi: 10.1016/j.bbi.2006.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bedard K, Krause KH. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol. Rev. 2007;87:245–313. doi: 10.1152/physrev.00044.2005. [DOI] [PubMed] [Google Scholar]
- 41.Rey FE, Cifuentes ME, Kiarash A, Quinn MT, Pagano PJ. Novel competitive inhibitor of NAD(P)H oxidase assembly attenuates vascular O(2)(−) and systolic blood pressure in mice. Circ. Res. 2001;89:408–414. doi: 10.1161/hh1701.096037. [DOI] [PubMed] [Google Scholar]
- 42.Gorin Y, et al. Targeting NADPH oxidase with a novel dual Nox1/Nox4 inhibitor attenuates renal pathology in type 1 diabetes. Am. J. Physiol. Renal. Physiol. 2015;308:4. doi: 10.1152/ajprenal.00396.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gianni D, et al. A novel and specific NADPH oxidase-1 (Nox1) small-molecule inhibitor blocks the formation of functional invadopodia in human colon cancer cells. ACS Chem. Biol. 2010;5:981–993. doi: 10.1021/cb100219n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bonet IJ, Fischer L, Parada CA, Tambeli CH. The role of transient receptor potential A 1 (TRPA1) in the development and maintenance of carrageenan-induced hyperalgesia. Neuropharmacology. 2013;65:206–212. doi: 10.1016/j.neuropharm.2012.09.020. [DOI] [PubMed] [Google Scholar]
- 45.Vasudeva K, et al. Imaging neuroinflammation in vivo in a neuropathic pain rat model with near-infrared fluorescence and (1)(9)F magnetic resonance. PLoS ONE. 2014;9:2014. doi: 10.1371/journal.pone.0090589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nilius B, Szallasi A. Transient receptor potential channels as drug targets: from the science of basic research to the art of medicine. Pharmacol. Rev. 2014;66:676–814. doi: 10.1124/pr.113.008268. [DOI] [PubMed] [Google Scholar]
- 47.Nassini R, et al. The ‘headache tree’ via umbellulone and TRPA1 activates the trigeminovascular system. Brain. 2012;135:376–390. doi: 10.1093/brain/awr272. [DOI] [PubMed] [Google Scholar]
- 48.Huang D, Li S, Dhaka A, Story GM, Cao YQ. Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura. Mol. Pain. 2012;8:66. doi: 10.1186/1744-8069-8-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Owsianik G, D’Hoedt D, Voets T, Nilius B. Structure-function relationship of the TRP channel superfamily. Rev. Physiol. Biochem. Pharmacol. 2006;156:61–90. [PubMed] [Google Scholar]
- 50.Kodji X, Aubdool AA, Brain SD. Evidence for physiological and pathological roles for sensory nerves in the microvasculature and skin. Curr. Res. Transl. Med. 2016;64:195–201. doi: 10.1016/j.retram.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 51.Edvinsson L. CGRP receptor antagonists and antibodies against CGRP and its receptor in migraine treatment. Br. J. Clin. Pharmacol. 2015;80:193–199. doi: 10.1111/bcp.12618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Benemei S, Fusi C, Trevisan G, Geppetti P. The TRPA1 channel in migraine mechanism and treatment. Br. J. Pharmacol. 2014;171:2552–2567. doi: 10.1111/bph.12512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ji RR, Chamessian A, Zhang YQ. Pain regulation by non-neuronal cells and inflammation. Science. 2016;354:572–577. doi: 10.1126/science.aaf8924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Szallasi A, Joo F, Blumberg PM. Duration of desensitization and ultrastructural changes in dorsal root ganglia in rats treated with resiniferatoxin, an ultrapotent capsaicin analog. Brain Res. 1989;503:68–72. doi: 10.1016/0006-8993(89)91705-8. [DOI] [PubMed] [Google Scholar]
- 55.Pecze L, et al. Resiniferatoxin mediated ablation of TRPV1+neurons removes TRPA1 as well. Can. J. Neurol. Sci. 2009;36:234–241. doi: 10.1017/S0317167100006600. [DOI] [PubMed] [Google Scholar]
- 56.Wang CH, Zou LJ, Zhang YL, Jiao YF, Sun JH. The excitatory effects of the chemokine CCL2 on DRG somata are greater after an injury of the ganglion than after an injury of the spinal or peripheral nerve. Neurosci. Lett. 2010;475:48–52. doi: 10.1016/j.neulet.2010.03.044. [DOI] [PubMed] [Google Scholar]
- 57.Sun JH, Yang B, Donnelly DF, Ma C, LaMotte RH. MCP-1 enhances excitability of nociceptive neurons in chronically compressed dorsal root ganglia. J. Neurophysiol. 2006;96:2189–2199. doi: 10.1152/jn.00222.2006. [DOI] [PubMed] [Google Scholar]
- 58.Kao DJ, et al. CC chemokine ligand 2 upregulates the current density and expression of TRPV1 channels and Nav1.8 sodium channels in dorsal root ganglion neurons. J. Neuroinflammation. 2012;9:189. doi: 10.1186/1742-2094-9-189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jung H, Toth PT, White FA, Miller RJ. Monocyte chemoattractant protein-1 functions as a neuromodulator in dorsal root ganglia neurons. J. Neurochem. 2008;104:254–263. doi: 10.1111/j.1471-4159.2007.04969.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.White FA, et al. Excitatory monocyte chemoattractant protein-1 signaling is up-regulated in sensory neurons after chronic compression of the dorsal root ganglion. Proc. Natl Acad. Sci. USA. 2005;102:14092–14097. doi: 10.1073/pnas.0503496102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tauzin S, Starnes TW, Becker FB, Lam PY, Huttenlocher A. Redox and Src family kinase signaling control leukocyte wound attraction and neutrophil reverse migration. J. Cell Biol. 2014;207:589–598. doi: 10.1083/jcb.201408090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang Y, et al. Nanoelectrodes for determination of reactive oxygen and nitrogen species inside murine macrophages. Proc. Natl Acad. Sci. USA. 2012;109:11534–11539. doi: 10.1073/pnas.1201552109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Niethammer P, Grabher C, Look AT, Mitchison TJ. A tissue-scale gradient of hydrogen peroxide mediates rapid wound detection in zebrafish. Nature. 2009;459:996–999. doi: 10.1038/nature08119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hamilton NB, Kolodziejczyk K, Kougioumtzidou E, Attwell D. Proton-gated Ca(2+)-permeable TRP channels damage myelin in conditions mimicking ischaemia. Nature. 2016;529:523–527. doi: 10.1038/nature16519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kwan KY, et al. TRPA1 contributes to cold, mechanical, and chemical nociception but is not essential for hair-cell transduction. Neuron. 2006;50:277–289. doi: 10.1016/j.neuron.2006.03.042. [DOI] [PubMed] [Google Scholar]
- 66.Liedtke W, Friedman JM. Abnormal osmotic regulation in trpv4-/- mice. Proc. Natl Acad. Sci. USA. 2003;100:13698–13703. doi: 10.1073/pnas.1735416100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Doerflinger NH, Macklin WB, Popko B. Inducible site-specific recombination in myelinating cells. Genesis. 2003;35:63–72. doi: 10.1002/gene.10154. [DOI] [PubMed] [Google Scholar]
- 68.Andre E, et al. Cigarette smoke-induced neurogenic inflammation is mediated by alpha,beta-unsaturated aldehydes and the TRPA1 receptor in rodents. J. Clin. Invest. 2008;118:2574–2582. doi: 10.1172/JCI34886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Malmberg AB, Basbaum AI. Partial sciatic nerve injury in the mouse as a model of neuropathic pain: behavioral and neuroanatomical correlates. Pain. 1998;76:215–222. doi: 10.1016/S0304-3959(98)00045-1. [DOI] [PubMed] [Google Scholar]
- 70.Lai CF, et al. TNFR1-activated reactive oxidative species signals up-regulate osteogenic Msx2 programs in aortic myofibroblasts. Endocrinology. 2012;153:3897–3910. doi: 10.1210/en.2012-1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ellmark SH, Dusting GJ, Fui MN, Guzzo-Pernell N, Drummond GR. The contribution of Nox4 to NADPH oxidase activity in mouse vascular smooth muscle. Cardiovasc. Res. 2005;65:495–504. doi: 10.1016/j.cardiores.2004.10.026. [DOI] [PubMed] [Google Scholar]
- 72.Ma W, Quirion R. Increased calcitonin gene-related peptide in neuroma and invading macrophages is involved in the up-regulation of interleukin-6 and thermal hyperalgesia in a rat model of mononeuropathy. J. Neurochem. 2006;98:180–192. doi: 10.1111/j.1471-4159.2006.03856.x. [DOI] [PubMed] [Google Scholar]
- 73.Brown DA, et al. Effect of phosphorothioate modification of oligodeoxynucleotides on specific protein binding. J. Biol. Chem. 1994;269:26801–26805. [PubMed] [Google Scholar]
- 74.Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods. 1994;53:55–63. doi: 10.1016/0165-0270(94)90144-9. [DOI] [PubMed] [Google Scholar]
- 75.Dixon WJ. Efficient analysis of experimental observations. Annu. Rev. Pharmacol. Toxicol. 1980;20:441–462. doi: 10.1146/annurev.pa.20.040180.002301. [DOI] [PubMed] [Google Scholar]
- 76.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 77.Landmesser U, et al. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J. Clin. Invest. 2003;111:1201–1209. doi: 10.1172/JCI200314172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sadofsky LR, et al. Unique responses are observed in transient receptor potential ankyrin 1 and vanilloid 1 (TRPA1 and TRPV1) co-expressing cells. Cells. 2014;3:616–626. doi: 10.3390/cells3020616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tao Y. Isolation and culture of Schwann cells. Methods Mol. Biol. 2013;1018:93–104. doi: 10.1007/978-1-62703-444-9_9. [DOI] [PubMed] [Google Scholar]
- 80.Zhang, X., Goncalves, R. & Mosser, D. M. The isolation and characterization of murine macrophages. Curr. Protoc. Immunol.14, 1–18 (2008). [DOI] [PMC free article] [PubMed]
- 81.Fusi C, et al. Steroidal and non-steroidal third-generation aromatase inhibitors induce pain-like symptoms via TRPA1. Nat. Commun. 2014;5:5736. doi: 10.1038/ncomms6736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Mahdessian H, et al. TM6SF2 is a regulator of liver fat metabolism influencing triglyceride secretion and hepatic lipid droplet content. Proc. Natl Acad. Sci. USA. 2014;111:8913–8918. doi: 10.1073/pnas.1323785111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Eisenblatter M, et al. In vivo optical imaging of cellular inflammatory response in granuloma formation using fluorescence-labeled macrophages. J. Nucl. Med. 2009;50:1676–1682. doi: 10.2967/jnumed.108.060707. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the corresponding author on reasonable request.