Abstract
Objective
To evaluate the oncologic outcomes of adjuvant chemotherapy (CT) alone after radical surgery.
Methods
We retrospectively reviewed the medical records of patients who underwent type C radical hysterectomy and pelvic lymphadenectomy with or without paraaortic lymphadenectomy for stage IB–IIA cervical cancer from March 2006 to December 2014 at 2 academic medical centers in Korea. Adjuvant CT alone for patients who met the Sedlis criteria or had lymph node (LN) involvement. Chemotherapeutic regimens consisted of either platinum alone or platinum-based combination.
Results
A total of 101 consecutive patients were included in the analysis. The median age was 46.0 years (range, 23–73). Seventy-four patients (73.3%) were clinically staged as IB1; 23 and 4 patients as IB2 and IIA, respectively. The median number of retrieved pelvic and paraaortic LNs (PALNs) were 55.0 (range, 16–101) and 10.0 (range, 2–30), respectively. LN involvement was observed in 25 patients (24.8%). After the median follow-up of 65 months, 14 patients (13.9%) developed disease recurrence. In all patients, the estimated 3-year disease-free survival (DFS) rate and 5-year overall survival (OS) rate were 90.7% and 90.6%, respectively. In patients who met the Sedlis criteria, the 3-year DFS and 5-year OS rates were 94.6% and 90.6%, respectively. Patients with LN metastasis showed a 3-year DFS rate of 79.4% and a 5-year OS rate of 90.6%.
Conclusion
Adjuvant CT alone could be reasonable option for patients with surgical-pathologic risk factors after radical surgery in stage IB–IIA cervical cancer.
Keywords: Adjuvant Chemotherapy, Drug Therapy, Uterine Cervical Neoplasms
INTRODUCTION
In Korea, there are about 3,600 new cases of cervical cancer each year, and about two-third of them are diagnosed as early-stage disease [1]. Radical hysterectomy (RH) with pelvic lymphadenectomy has been widely accepted as the preferred treatment in patients with early-stage cervical cancer (stage IA2–IIA) in Korea [2].
Even after curative resection for early-stage cervical cancer, disease recurrence occurs in approximately 20%–30% of patients [3,4]. Postoperative pathologic examinations reveal risk factors for recurrence. High risk factors for recurrence are lymph node (LN) involvement and/or microscopic parametrial invasion, and intermediate-risk factors are tumor size, depth of stromal invasion (DOI), and/or lymphovascular space invasion (LVSI) [5,6]. Radiation therapy (RT) or concurrent chemoradiation therapy (CCRT) has been recommended as the standard adjuvant therapy to reduce recurrence in patients with these risk factors [7]. However, adjuvant RT or CCRT could be associated with an increased risk of certain complications and a negative impact on the quality of life [8]. In addition, survival benefit from adjuvant RT is currently unclear in patients with intermediate-risk factors [9].
Platinum is the most active agent in treating cervical cancer, and platinum-based chemotherapy (CT) is widely used to treat recurrent or metastatic disease [10,11]. Several studies have reported that platinum-based CT alone has an equivalent effect of RT or CCRT, with the benefit of fewer long-term complications in adjuvant setting for patients with risk factors [12,13,14,15]. In this study, we evaluated the oncologic outcomes of adjuvant CT alone in 101 consecutive patients with surgical-pathologic risk factors after radical surgery in stage IB–IIA cervical cancer.
MATERIALS AND METHODS
We reviewed the cervical cancer surgery database and medical records at 2 academic medical centers in Korea (Gachon University Gil Medical Center and Kyung Hee University Hospital at Gangdong) to identify patients who underwent radical surgery for stage IB–IIA cervical cancer and received adjuvant CT alone between March 2006 and December 2014. Patients were excluded if they had microscopic involvement of the parametrium or positive surgical margin, received neoadjuvant therapy, had incomplete clinicopathologic data, or received RT or CCRT. This retrospective study was approved by the local institutional review board of each institution. The stage of each patient was determined by the 2009 International Federation of Gynecology and Obstetrics (FIGO) classification.
Radical surgery consisted of type C hysterectomy and pelvic lymphadenectomy with or without paraaortic lymphadenectomy. We tried to completely remove all common iliac (including presacral), external iliac, internal iliac, and obturator LNs. And, if intraoperative pelvic LN was suspicious or positive, paraaortic lymphadenectomy up to inferior mesenteric artery was done.
Based on the pathologic assessment after radical surgery, adjuvant CT was given to patients who met the Sedlis criteria that defines intermediate-risk disease or had LN involvement. Sedlis criteria included one of the following: 1) positive LVSI plus a deep third stromal invasion and any tumor size; 2) positive LVSI plus a middle third stromal invasion and a tumor size of ≥2 cm; 3) positive LVSI plus a superficial third stromal invasion and a tumor size of ≥5 cm; and 4) no LVSI, but deep or middle third stromal invasion and a tumor size of ≥4 cm. Adjuvant CT, using either the platinum monotherapy or platinum-based combination, began within 4 weeks after radical surgery. Platinum monotherapy consisted of cisplatin (Platinol®; Bristol-Myers Squibb Company, Princeton, NJ, USA) 50 mg/m2 on day 1 every 3 weeks. A combination regimen was paclitaxel (175 mg/m2 on day 1, Taxol®; Bristol-Myers Squibb Company) combined with either cisplatin (50 mg/m2 on day 1; Bristol-Myers Squibb Company) or carboplatin (AUC 5 on day 1, Paraplatin®; Bristol-Myers Squibb Company) every 3 weeks.
Categorical variables are presented as frequencies and percentages, and continuous variables are expressed as the median with range. Overall survival (OS) and disease-free survival (DFS) were estimated by the Kaplan-Meier method. DFS was defined from the date of surgery to physical or radiologic evidence of disease recurrence, or at the last patient visit. OS was defined as from the date of surgery to death, or to the date of last visit. Disease-specific survival (DSS) was defined as the time interval from date of surgery to the date of cervical cancer related death or to the date of last visit. Patients who died of causes other than cervical cancer were censored at the time of their death for DSS analysis. All analyses were performed using SPSS for Windows version 18.0 (SPSS Inc., Chicago, IL, USA). All end points were last updated in July 2016.
RESULTS
1. Patient characteristics
During the period, radical surgery was performed in 591 patients. Of them, a total of 101 consecutive patients were included in the study. The median age was 46.0 (range, 23–73) years; the median body mass index was 23.0 (range, 17.5–31.6); the median parity was 2.0 (range, 0–5); and 34 women (33.7%) were in menopausal status. Seventy-four patients (73.3%) were staged as IB1, 23 patients (22.8%) as IB2, and 4 patients (3.9%) as IIA. The histologic cell type was squamous in 75 women (74.3%), adenocarcinoma in 15 (14.9%), adenosquamous in 6 (5.9%), and others in 5 (4.9%). The median diameter of tumor was 4.0 cm (range, 1–7.3). The median number of retrieved pelvic LNs was 55.0 (range, 16–101); and the median number of positive pelvic LN was 2 (range, 1–33). Fifty patients underwent both pelvic and paraaortic lymphadenectomy. The median number of retrieved paraaortic LN (PALN) was 10.0 (range, 2–30); and the median number of positive PALN was 2.0 (range, 1–17). The median safety margin of vagina was 1.5 cm (range, 0.4–4.5). A single platinum CT was used in 47 patients (47%), and platinum-based combination CT was used in 54 patients (53%). Thirty-one patients (31%) received 3 cycles of CT, and 70 patients (69%) received 6 cycles of CT (Table 1). The patients (n=23) with LN metastasis received 6 cycles of platinum-based combination and the remaining patients (n=2) received 3 cycles of platinum-based combination. The patients with Sedlis criteria received 3 cycles (n=29) of platinum-based combination or 6 cycles (n=47) of platinum monotherapy.
Table 1. Patient's characteristics (n=101).
| Characteristics | Value | |
|---|---|---|
| Age (yr) | 46.0 (23–73) | |
| Body mass index | 23.0 (17.5–31.6) | |
| Parity | 2.0 (0–5) | |
| Menopause | ||
| Yes | 34 (33.7) | |
| No | 67 (66.3) | |
| Clinical stage | ||
| IB1 | 74 (73.3) | |
| IB2 | 23 (22.8) | |
| IIA | 4 (3.9) | |
| Histology | ||
| Squamous | 75 (74.3) | |
| Adenocarcinoma | 15 (14.9) | |
| Adenosquamous | 6 (5.9) | |
| Others | 5 (4.9) | |
| Pathologic large tumor diameter (cm) | 4.0 (1–8) | |
| No. of retrieved pelvic LN | 55.0 (16–101) | |
| No. of pelvic LN metastasis | 2.0 (1–33) | |
| No. of patients undergoing pelvic and paraaortic lymph dissection | 50 | |
| No. of retrieved PALN | 10.0 (2–30) | |
| No. of PALN metastasis | 2.0 (1–17) | |
| Safety margin of vagina (cm) | 1.5 (0.4–4.5) | |
| CT regimen | ||
| Single platinum | 47 (47) | |
| Paclitaxel and platinum | 54 (53) | |
| Cycles of CT | ||
| 3 cycles | 31 (31) | |
| 6 cycles | 70 (69) | |
Values are presented as median (range) or number of patients (%).
CT, chemotherapy; LN, lymph node; PALN, paraaortic lymph node.
There were 76 patients with a combination of 3 risk factors without LN metastasis: 29 (38.2%) with positive LVSI, deep 1/3 DOI, and tumor with any size; 27 (35.6%) with positive LVSI, middle 1/3 DOI, and ≥2 cm of tumor size; 18 (23.7%) with negative LVSI, deep or middle 1/3 DOI, and ≥4 cm of tumor size; 2 (1.5%) with positive LVSI, superficial 1/3 DOI, and ≥5 cm of tumor size (Table 2). Twenty-five patients had 1 or more LN metastases: 21 with only pelvic LN metastasis, and 4 with pelvic and PALN metastasis.
Table 2. Combination of the 3 risk factors without positive LN (n=76).
| LVSI | DOI | Tumor size (cm) | No. (%) |
|---|---|---|---|
| Positive | Deep 1/3 | Any | 29 (38.2) |
| Positive | Middle 1/3 | ≥2 | 27 (35.6) |
| Negative | Deep or middle 1/3 | ≥4 | 18 (23.7) |
| Positive | Superficial 1/3 | ≥5 | 2 (1.5) |
DOI, depth of stromal invasion; LN, lymph node; LVSI, lymphovascular space invasion.
2. Treatment outcomes
Fourteen (13.9%) of the 101 patients suffered from recurrence. Nine of those patients had a combination of the 3 risk factors without LN metastasis; of which 5 patients had recurrences in the loco-regional site and 4 patients in the distant sites. Moreover, of these 9 patients after salvage treatment (ST), 3 had no evidence of the disease, 4 patients are alive with the disease, and 2 patients died with the disease. Of the 5 patients with LN metastasis, 2 patients had recurrences in the loco-regional site and 3 patients in distant sites; after ST, 1 patient had no evidence of the disease, 1 patient is alive with the disease, and 3 patients died with the disease (Table 3). The median follow-up time was 65 months (range, 19–108). Comparison of the single with combination regimen in the patients with Sedlis criteria revealed no significant differences in 3-year DFS (p=0.11), 5-year OS (p=0.25), and the 5-year DSS (p=0.25). Although 61.8% of patients with intermediate-risk factors received a single agent platinum, the recurrence rates were lower in patients with intermediate-risk factors than in patients with high-risk factors (11.8% vs. 20.0%, p=0.326). Additional studies are necessary to establish the optimal regimen for adjuvant CT.
Table 3. Disease relapse pattern and ST.
| Case No. | Age | Stage | TS (cm) | DOI | LVSI | LN metastasis | RS | ST | PS |
|---|---|---|---|---|---|---|---|---|---|
| Patients with combination of the 3 risk factors without LN metastasis | |||||||||
| Case 1 | 52 | IB1 | 1.5 | Deep | Yes | No | Vulva | CCRT | NED |
| Case 2 | 40 | IB1 | 4.9 | Middle | Yes | No | Pelvis | Surgery | NED |
| Case 3 | 42 | IB2 | 4.5 | Middle | Yes | No | Pelvis | CCRT | NED |
| Case 4 | 65 | IB1 | 2.9 | Deep | Yes | No | Vagina | RT | AWD |
| Case 5 | 44 | IB2 | 4.5 | Deep | Yes | No | Brain | RT | AWD |
| Case 6 | 52 | IB1 | 2.0 | Middle | Yes | No | Lung | CT | AWD |
| Case 7 | 40 | IB1 | 3.2 | Middle | Yes | No | Vagina | RT | AWD |
| Case 8 | 49 | IB1 | 5.5 | Deep | Yes | No | Lung | CT | Dead |
| Case 9 | 72 | IB2 | 3.7 | Deep | No | No | Lung | CT | Dead |
| Patients with LN metastasis irrespective the 3 risk factors | |||||||||
| Case 10 | 37 | IB1 | 2.9 | Deep | Yes | Yes | Vagina | RT | NED |
| Case 11 | 40 | IB1 | 2.0 | Deep | Yes | Yes | Vagina | CCRT | AWD |
| Case 12 | 66 | IB1 | 4.0 | Deep | Yes | Yes | Lung | RT | Dead |
| Case 13 | 49 | IB2 | 4.5 | Deep | Yes | Yes | Lung | CT | Dead |
| Case 14 | 50 | IB2 | 3.0 | Deep | Yes | Yes | PALN | CT | Dead |
AWD, alive with disease; CCRT, concurrent chemoradiation therapy; CT, chemotherapy; DOI, depth of stromal invasion; LN, lymph node; LVSI, lymphovascular space invasion; NED, no evidence of disease; PALN, paraaortic lymph node; PS, present status; RS, recurrence site; RT, radiation therapy; ST, salvage treatment; TS, pathologic large tumor size.
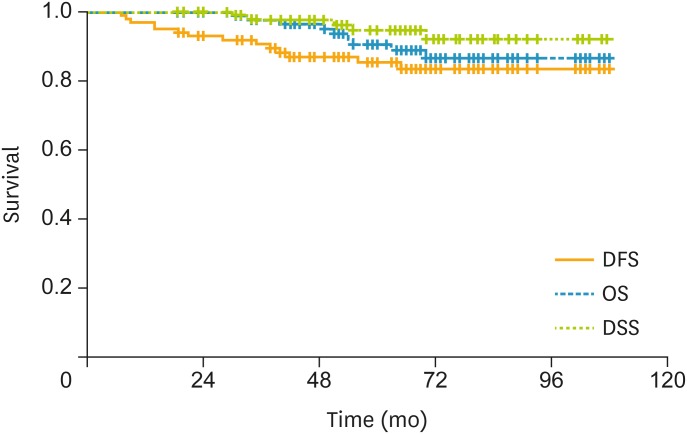
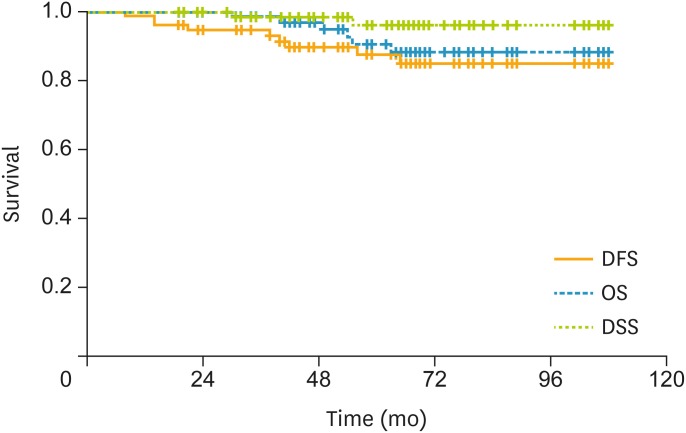
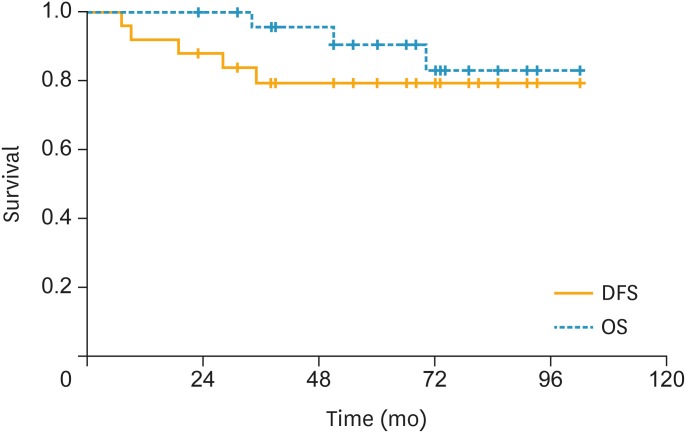
In all patients, the estimated 3-year DFS rate was 90.7%; the estimated 5-year OS rate was 90.6%; and the 5-year DSS rate was 94.7% (Fig. 1). In patients with a combination of 3 risk factors, the 3-year DFS rate was 94.6%; the 5-year OS rate was 90.6%; and the 5-year DSS rate was 96.2% (Fig. 2). In patients with LN metastasis, the 3-year DFS rate was 79.4%; the 5-year OS rate was 90.6%; and the 5-year DSS rate was 90.6% (Fig. 3). The causes of death unrelated to cervical cancer were car accident, tongue cancer, myocardial infarction, and brain stroke.
Fig. 1.
In the all patients, the 3-year DFS rate was 90.7%, the 5-year OS rate was 90.6%, and the 5-year DSS rate was 94.7%.
DFS, disease-free survival; DSS, disease-specific survival; OS, overall survival.
Fig. 2.
In the patients with combination of 3 risk factors, the 3-year DFS rate was 94.6%, the 5-year OS rate was 90.6%, and the 5-year DSS rate was 96.2%.
DFS, disease-free survival; DSS, disease-specific survival; OS, overall survival.
Fig. 3.
In the patients with LN metastasis, the 3-year DFS rate was 79.4%, the 5-year OS rate was 90.6%, and the 5-year DSS rate was 90.6%.
DFS, disease-free survival; DSS, disease-specific survival; LN, lymph node; OS, overall survival.
There were no treatment-related deaths and treatment-related morbidity was as follows; grade 3–4 hematologic toxicity occurring in 18 patients (25 episodes), infected lymphocele requiring drain 2 patients, hydronephrosis 2 patients, herniation of abdominal 1 patient, and lymphedema of lower limb 15 patients.
DISCUSSION
This retrospective study included one of the largest cohorts of patients who received adjuvant CT alone. With the median follow-up duration of 65 months, the 3-year DFS and 5-year OS rates were 90.7% and 90.6%, respectively. Our results were better than those of historical controls, who had received surgery-only in randomized phase III trials (5-year DFS rates, 72.1%–74.0%; 5-year OS rates, 78.6%–84.0%) [5,16], and comparable to the results from previous studies evaluating adjuvant CT alone (5-year DFS rates, 84.5%–93.3%; 5-year OS rates, 86.5%–94.9%) [13,17,18,19]. We additionally analyzed survival data of 59 patients having relative risk of recurrence >120 and no LN metastasis based on Gynecologic Oncology Group (GOG) scoring system [20]. These patients had relatively high risk of recurrence on Sedlis criteria. The 3-year DFS (94.9%), 5-year OS (90.9%), and 5-year DSS (95.2%) were not different from all patients. Thus, adjuvant CT seems to be effective even in relatively higher risk patients in a combination of 3 risk factors.
Loco-regional recurrence is a predominant pattern of failure in radically resected early-stage cervical cancer [4]. To control pelvic recurrence, many investigators have been focused on whether RT is beneficial in the adjuvant setting. Although pelvic RT alone after radical resection significantly reduces the risk of local recurrence, it is not associated with survival benefit [9]. On the other hand, the addition of CT to pelvic RT was associated with significant improvement of survival in early-stage, high-risk patients undergoing radical resection for cervical cancer [6]. However, a previously reported randomized phase III trial failed to show the superiority of CCRT over CT alone as adjuvant therapy in early-stage cervical cancer patients at high-risk for recurrence, which resulted in the early closure of the trial [15]. There is a major concern that adjuvant CT alone may be ineffective to prevent local recurrence. However, a large retrospective study showed that adjuvant CT alone may be effective in reducing both loco-regional and distal recurrences; the CT alone group exhibited a significantly lower pelvic recurrence and distant metastasis rates than the RT alone group. Thus, CT could be considered the effective tool of controlling locoregional and distant micrometastasis [18,19,21].
Adjuvant RT or CCRT is associated with an increased incidence of complications, including bowel obstruction, urinary disturbance, and lower-limb lymph edema [8,12,18]. In addition, it was reported that women treated with adjuvant RT had worse quality of life and sexual dysfunction than those treated with surgery alone or surgery followed by CT alone [18,22]. Given that cervical cancer mostly affects younger women, and since most patients with early-stage disease have good prognosis, sexual health and quality of life are very important issues for cervical cancer survivors [23]. In our study, 66.3% of patients were in the premenopausal state at the time of diagnosis, and treatment-related morbidity was acceptable.
In addition to adverse events, CT alone as adjuvant therapy has additional advantages. First, it can reduce hospital visits or costs. In our hospital, the average cost is as follows: adjuvant CCRT $6,006, adjuvant RT $5,450, platinum only per cycle $112, and paclitaxel and platinum per cycle $578. Second, if the patient experiences loco-regional recurrence, RT or CCRT are still available with curative potential [15]. In our study, 4 of 7 patients with pelvic recurrences are alive without evidence of disease after subsequent local treatment. Therefore, if CT alone as adjuvant therapy is at least as effective as RT or CCRT, it is reasonable to use CT alone after radical resection. Third, if RT facilities are unavailable, adjuvant CT only after radical surgery could be used. In contrary to cervical cancer, CT alone is accepted as one of standard adjuvant therapies after microscopically margin-negative resection in other solid cancers (e.g., gastric cancer and non-small cell lung cancer) [24,25].
In previous several studies, various combination regimens, such as paclitaxel/cisplatin, 5-fluorouriacil/cisplatin, bleomycin/vincrisitine/mitomycin/cisplatin, etoposide/cisplatin, were explored for adjuvant CT [12,13,17,18,19]. To date, optimal chemotherapeutic agents and treatment cycles have not yet been determined for cervical cancer in adjuvant setting. The combination of paclitaxel and platinum could be appropriate as adjuvant CT, based on the results achieved in phase III trials for advanced or recurrent cervical cancer [8,26]. However, our study suggested that single agent platinum could be used for patients with intermediate-risk disease. In conclusion, our results show that CT alone may be an effective adjuvant option for stage IB–IIA cervical cancer patients with surgical-pathologic risk factors. Further large prospective randomized trials that make a comparison between CT and RT or CCRT are necessary to confirm our findings and establish an optimal adjuvant CT regimen.
Footnotes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
Presentation: We presented this topic at SGO 48th Annual Meeting on Women's Cancer in a Scientific Plenary on Monday, March 13, 2017.
- Conceptualization: L.K.B., L.J.M.
- Data curation: L.K.B., L.J.M.
- Formal analysis: L.K.B., L.J.M.
- Investigation: L.K.B., K.Y.S., L.J.M.
- Methodology: L.K.B., L.J.M.
- Project administration: L.J.M.
- Resources: L.K.B., L.J.M.
- Software: L.K.B.
- Supervision: L.J.M.
- Validation: L.K.B.
- Visualization: L.K.B.
- Writing - original draft: L.K.B.
- Writing - review & editing: L.K.B., K.Y.S., L.J.M.
References
- 1.Oh CM, Won YJ, Jung KW, Kong HJ, Cho H, Lee JK, et al. Cancer Statistics in Korea: incidence, mortality, survival, and prevalence in 2013. Cancer Res Treat. 2016;48:436–450. doi: 10.4143/crt.2016.089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ryu HS, Kang SB, Kim KT, Chang KH, Kim JW, Kim JH. Efficacy of different types of treatment in FIGO stage IB2 cervical cancer in Korea: results of a multicenter retrospective Korean study (KGOG-1005) Int J Gynecol Cancer. 2007;17:132–136. doi: 10.1111/j.1525-1438.2007.00803.x. [DOI] [PubMed] [Google Scholar]
- 3.Friedlander M, Grogan M U.S. Preventative Services Task Force. Guidelines for the treatment of recurrent and metastatic cervical cancer. Oncologist. 2002;7:342–347. [PubMed] [Google Scholar]
- 4.Rotman M, Sedlis A, Piedmonte MR, Bundy B, Lentz SS, Muderspach LI, et al. A phase III randomized trial of postoperative pelvic irradiation in Stage IB cervical carcinoma with poor prognostic features: follow-up of a gynecologic oncology group study. Int J Radiat Oncol Biol Phys. 2006;65:169–176. doi: 10.1016/j.ijrobp.2005.10.019. [DOI] [PubMed] [Google Scholar]
- 5.Sedlis A, Bundy BN, Rotman MZ, Lentz SS, Muderspach LI, Zaino RJ. A randomized trial of pelvic radiation therapy versus no further therapy in selected patients with stage IB carcinoma of the cervix after radical hysterectomy and pelvic lymphadenectomy: a Gynecologic Oncology Group Study. Gynecol Oncol. 1999;73:177–183. doi: 10.1006/gyno.1999.5387. [DOI] [PubMed] [Google Scholar]
- 6.Peters WA, 3rd, Liu PY, Barrett RJ, 2nd, Stock RJ, Monk BJ, Berek JS, et al. Concurrent chemotherapy and pelvic radiation therapy compared with pelvic radiation therapy alone as adjuvant therapy after radical surgery in high-risk early-stage cancer of the cervix. J Clin Oncol. 2000;18:1606–1613. doi: 10.1200/JCO.2000.18.8.1606. [DOI] [PubMed] [Google Scholar]
- 7.Korean Society of Gynecologic Oncology. Practice guideline for uterine cervical cancer [Internet] Seoul: Korean Society of Gynecologic Oncology; 2015. [cited year month day]. Available from: http://www.sgo.or.kr/community/pdf/guide_01_2016_01.pdf. [Google Scholar]
- 8.Asano H, Todo Y, Watari H. Adjuvant chemotherapy for early-stage cervical cancer. Chin J Cancer Res. 2016;28:228–234. doi: 10.21147/j.issn.1000-9604.2016.02.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rogers L, Siu SS, Luesley D, Bryant A, Dickinson HO. Radiotherapy and chemoradiation after surgery for early cervical cancer. Cochrane Database Syst Rev. 2012:CD007583. doi: 10.1002/14651858.CD007583.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lorusso D, Petrelli F, Coinu A, Raspagliesi F, Barni S. A systematic review comparing cisplatin and carboplatin plus paclitaxel-based chemotherapy for recurrent or metastatic cervical cancer. Gynecol Oncol. 2014;133:117–123. doi: 10.1016/j.ygyno.2014.01.042. [DOI] [PubMed] [Google Scholar]
- 11.Scatchard K, Forrest JL, Flubacher M, Cornes P, Williams C. Chemotherapy for metastatic and recurrent cervical cancer. Cochrane Database Syst Rev. 2012;10:CD006469. doi: 10.1002/14651858.CD006469.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hosaka M, Watari H, Kato T, Odagiri T, Konno Y, Endo D, et al. Clinical efficacy of paclitaxel/cisplatin as an adjuvant chemotherapy for patients with cervical cancer who underwent radical hysterectomy and systematic lymphadenectomy. J Surg Oncol. 2012;105:612–616. doi: 10.1002/jso.22136. [DOI] [PubMed] [Google Scholar]
- 13.Lee KB, Lee JM, Ki KD, Lee SK, Park CY, Ha SY. Comparison of adjuvant chemotherapy and radiation in patients with intermediate risk factors after radical surgery in FIGO stage IB-IIA cervical cancer. Int J Gynecol Cancer. 2008;18:1027–1031. doi: 10.1111/j.1525-1438.2007.01136.x. [DOI] [PubMed] [Google Scholar]
- 14.Matsuo K, Shimada M, Aoki Y, Sakamoto M, Takeshima N, Fujiwara H, et al. Comparison of adjuvant therapy for node-positive clinical stage IB-IIB cervical cancer: systemic chemotherapy versus pelvic irradiation. Int J Cancer. 2017;141:1042–1051. doi: 10.1002/ijc.30793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Curtin JP, Hoskins WJ, Venkatraman ES, Almadrones L, Podratz KC, Long H, et al. Adjuvant chemotherapy versus chemotherapy plus pelvic irradiation for high-risk cervical cancer patients after radical hysterectomy and pelvic lymphadenectomy (RH-PLND): a randomized phase III trial. Gynecol Oncol. 1996;61:3–10. doi: 10.1006/gyno.1996.0087. [DOI] [PubMed] [Google Scholar]
- 16.Landoni F, Maneo A, Colombo A, Placa F, Milani R, Perego P, et al. Randomised study of radical surgery versus radiotherapy for stage Ib-IIa cervical cancer. Lancet. 1997;350:535–540. doi: 10.1016/S0140-6736(97)02250-2. [DOI] [PubMed] [Google Scholar]
- 17.Lee TY, Jeung YJ, Lee CJ, Kim HY, Kim SH, Kim WG. Promising treatment results of adjuvant chemotherapy following radical hysterectomy for intermediate risk stage 1B cervical cancer. Obstet Gynecol Sci. 2013;56:15–21. doi: 10.5468/OGS.2013.56.1.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li S, Hu T, Chen Y, Zhou H, Li X, Cheng X, et al. Adjuvant chemotherapy, a valuable alternative option in selected patients with cervical cancer. PLoS One. 2013;8:e73837. doi: 10.1371/journal.pone.0073837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Takeshima N, Umayahara K, Fujiwara K, Hirai Y, Takizawa K, Hasumi K. Treatment results of adjuvant chemotherapy after radical hysterectomy for intermediate- and high-risk stage IB-IIA cervical cancer. Gynecol Oncol. 2006;103:618–622. doi: 10.1016/j.ygyno.2006.04.019. [DOI] [PubMed] [Google Scholar]
- 20.Delgado G, Bundy B, Zaino R, Sevin BU, Creasman WT, Major F. Prospective surgical-pathological study of disease-free interval in patients with stage IB squamous cell carcinoma of the cervix: a Gynecologic Oncology Group study. Gynecol Oncol. 1990;38:352–357. doi: 10.1016/0090-8258(90)90072-s. [DOI] [PubMed] [Google Scholar]
- 21.Argiris A, Jayaram P, Pichardo D. Revisiting induction chemotherapy for head and neck cancer. References and reviews. Oncology (Williston Park) 2005;19:932–934. [PubMed] [Google Scholar]
- 22.Greimel ER, Winter R, Kapp KS, Haas J. Quality of life and sexual functioning after cervical cancer treatment: a long-term follow-up study. Psychooncology. 2009;18:476–482. doi: 10.1002/pon.1426. [DOI] [PubMed] [Google Scholar]
- 23.Vistad I, Fosså SD, Dahl AA. A critical review of patient-rated quality of life studies of long-term survivors of cervical cancer. Gynecol Oncol. 2006;102:563–572. doi: 10.1016/j.ygyno.2006.03.050. [DOI] [PubMed] [Google Scholar]
- 24.Arriagada R, Dunant A, Pignon JP, Bergman B, Chabowski M, Grunenwald D, et al. Long-term results of the international adjuvant lung cancer trial evaluating adjuvant Cisplatin-based chemotherapy in resected lung cancer. J Clin Oncol. 2010;28:35–42. doi: 10.1200/JCO.2009.23.2272. [DOI] [PubMed] [Google Scholar]
- 25.Noh SH, Park SR, Yang HK, Chung HC, Chung IJ, Kim SW, et al. Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol. 2014;15:1389–1396. doi: 10.1016/S1470-2045(14)70473-5. [DOI] [PubMed] [Google Scholar]
- 26.Monk BJ, Sill MW, McMeekin DS, Cohn DE, Ramondetta LM, Boardman CH, et al. Phase III trial of four cisplatin-containing doublet combinations in stage IVB, recurrent, or persistent cervical carcinoma: a Gynecologic Oncology Group study. J Clin Oncol. 2009;27:4649–4655. doi: 10.1200/JCO.2009.21.8909. [DOI] [PMC free article] [PubMed] [Google Scholar]