Key Points
Question
Does a program to increase intensive care unit (ICU) admission rates among critically ill elderly patients have a beneficial effect on long-term outcomes?
Findings
In this cluster-randomized clinical trial of 3036 critically ill patients aged 75 years or older, a recommendation for systematic ICU admission led to a significantly higher ICU admission rate but had no significant effect on mortality at 6 months vs standard practice (adjusted relative risk, 1.05).
Meaning
A program to promote systematic ICU admission among critically ill elderly patients increased ICU use but did not reduce 6-month mortality.
Abstract
Importance
The high mortality rate in critically ill elderly patients has led to questioning of the beneficial effect of intensive care unit (ICU) admission and to a variable ICU use among this population.
Objective
To determine whether a recommendation for systematic ICU admission in critically ill elderly patients reduces 6-month mortality compared with usual practice.
Design, Setting, and Participants
Multicenter, cluster-randomized clinical trial of 3037 critically ill patients aged 75 years or older, free of cancer, with preserved functional status (Index of Independence in Activities of Daily Living ≥4) and nutritional status (absence of cachexia) who arrived at the emergency department of one of 24 hospitals in France between January 2012 and April 2015 and were followed up until November 2015.
Interventions
Centers were randomly assigned either to use a program to promote systematic ICU admission of patients (n=1519 participants) or to follow standard practice (n=1518 participants).
Main Outcomes and Measures
The primary outcome was death at 6 months. Secondary outcomes included ICU admission rate, in-hospital death, functional status, and quality of life (12-Item Short Form Health Survey, ranging from 0 to 100, with higher score representing better self-reported health) at 6 months.
Results
One patient withdrew consent, leaving 3036 patients included in the trial (median age, 85 [interquartile range, 81-89] years; 1361 [45%] men). Patients in the systematic strategy group had an increased risk of death at 6 months (45% vs 39%; relative risk [RR], 1.16; 95% CI, 1.07-1.26) despite an increased ICU admission rate (61% vs 34%; RR, 1.80; 95% CI, 1.66-1.95). After adjustments for baseline characteristics, patients in the systematic strategy group were more likely to be admitted to an ICU (RR, 1.68; 95% CI, 1.54-1.82) and had a higher risk of in-hospital death (RR, 1.18; 95% CI, 1.03-1.33) but had no significant increase in risk of death at 6 months (RR, 1.05; 95% CI, 0.96-1.14). Functional status and physical quality of life at 6 months were not significantly different between groups.
Conclusions and Relevance
Among critically ill elderly patients in France, a program to promote systematic ICU admission increased ICU use but did not reduce 6-month mortality. Additional research is needed to understand the decision to admit elderly patients to the ICU.
Trial Registration
clinicaltrials.gov Identifier: NCT01508819
This randomized trial compares the effect of a program to promote systematic intensive care unit (ICU) admission vs standard practice on 6-month mortality in critically ill elderly patients.
Introduction
The aging of the population leads to an increased demand for intensive care among elderly patients. These patients now account for 10% to 20% of all intensive care unit (ICU) admissions, and this trend continues to increase. In a context of expenditures control in health care, appropriate intensive care resource utilization is an important issue. Yet many physicians have doubts as to whether elderly patients benefit from ICU admission. Previous studies have focused on the benefit of intensive care for adult patients. However, elderly patients have an age-related diminution of physiological reserve, a higher prevalence of chronic diseases, and more common frailty. These age-related physiological and pathological changes make elderly patients more vulnerable to stress due to acute illnesses, putting them at high risk of death when critically ill, making the benefit of ICU admission uncertain in this population.
To date, there has been no randomized clinical trial of ICU triage in elderly patients. Observational studies have reported conflicting results: some studies suggested benefits from ICU admission while others did not. These uncertainties, coupled with the absence of triage guidelines adapted to elderly patients, lead to wide heterogeneity in clinical practices regarding ICU admission among elderly patients.
The Intensive Care for Elderly–CUB-Réa 2 (ICE-CUB 2) trial was a cluster-randomized clinical trial to determine whether a recommendation for systematic ICU admission in critically ill elderly patients reduces 6-month mortality compared with usual practice.
Methods
The trial was approved by the Comité de Protection des Personnes d’Ile-de-France 9. Participants or their surrogate decision makers were informed orally about the trial, and their nonopposition to trial participation was recorded in patient’ files. In accordance with French law, written informed consent was not required because both interventions are part of standard care. Patients who were eligible but incapable of receiving information and for whom a surrogate decision maker was not available might be included by deferred information. These participants were informed about study participation as soon as they were able to understand information. Information was collected during a 6-month follow-up period. Participants were informed that the authorization to use patient data could be withdrawn at any time. Details on steering committee members and conduct of the study are available in the eAppendix in Supplement 1.
The study protocol is available in Supplement 2. In brief, in this cluster-randomized clinical trial conducted in 24 hospitals in France from January 2012 through November 2015, academic and nonacademic hospitals with at least 1 emergency department and 1 ICU were randomly assigned either to the intervention or the control group.
The allocation schedule was independently established by a statistician at the clinical research unit using a computer-generated randomization list. Randomization was stratified by the median annual number of emergency department visits in participating centers (n=44 616), presence or absence of a geriatric ward, and geographical area (Paris area vs other regions in France). Each site allocation was kept secret by the clinical research unit until the training of the study personnel was completed and was revealed to the hospital primary investigators at the beginning of the inclusion period. Due to the study design, no allocation concealment was possible.
In hospitals assigned to the systematic strategy group (intervention), a program to promote systematic ICU admission was implemented. In this program, emergency department and ICU physicians were asked to systematically recommend an ICU admission for all included patients during the triage process. Other interventions to promote ICU admission included the following: a member of the steering committee visited each center and presented the trial protocol; when including a participant in the trial, the emergency department physician was required to systematically call the attending ICU physician; the ICU physician was required to systematically evaluate the patient at the bedside; and the emergency department and ICU physicians were required to jointly decide whether to admit the patient to the ICU with consideration of participant or surrogate decision-maker opinions about ICU admission. If no ICU bed was available in the hospital, the patient had to be transferred to an ICU located in another hospital. Monthly meetings were organized with the emergency department and ICU staff. Booklets and posters presenting the recommendations for ICU admission were used.
In hospitals assigned to the standard practice (control) group, there was no specific recommendation regarding the ICU triage process. In both groups, the final decision for admission was made by the physicians at the bedside.
Participant Selection
All patients aged 75 years or older who arrived in the emergency department were assessed for eligibility. Patients were included if they had one of the preestablished critical conditions listed in eTable 1 in Supplement 1, had preserved functional status as defined by an Index of Independence in Activities of Daily Living score of 4 or higher or not evaluable, had preserved nutritional status (defined as absence of cachexia, subjectively assessed by physicians at the bedside), and were free of active cancer. Exclusion criteria were an emergency department stay of more than 24 hours, a secondary referral to the emergency department, and refusal to participate. Patients in whom cancer was diagnosed after inclusion remained in the statistical analysis. Further information on inclusion and exclusion criteria and follow-up are available in the eAppendix in Supplement 1.
Outcome Measures
The primary outcome was overall mortality at 6 months. Secondary outcomes were ICU admission rate, in-hospital mortality, functional status at 6 months as assessed by the Index of Independence in Activities of Daily Living, and quality of life at 6 months as assessed by the 12-Item Short-Form Health Survey. The Index of Independence in Activities of Daily Living is based on an evaluation of the functional independence or dependence of patients in bathing, dressing, toileting, transferring, continence, and feeding and ranges from 0 (totally dependent) to 6 (independent). The 12-Item Short-Form Health Survey is a measure of health-related quality of life and includes a physical and mental component; the score ranges from 0 to 100, with higher scores representing better self-reported health. Other secondary outcomes were the characteristics of the triage process: opinions about ICU admission and involvement of physicians and participants in the decision process and the number of ICUs with all beds filled. Caregiver burden at 6 months was assessed in 2 centers and is not reported in this article. For patients discharged alive, information on outcomes were obtained through telephone calls, either directly from patients, from patients’ relatives or general practitioners, or from appropriate legal institutions. Data on patients who were lost to follow-up before 6 months were censored at the last follow-up assessment.
Statistical Analysis
The statistical analysis plan is available in Supplement 3. Assumptions based on sample size calculation were based on the ICE-CUB 1 study. Considering an estimated 32% 6-month mortality rate in the control group and an estimated intracluster correlation coefficient of 0.01, a sample size of 2802 was required to have 74% power to detect a 6% difference in mortality rates in a 2-sided test at a .05 level of significance. Twenty-four hospitals agreed to participate instead of 20 initially planned; to take into account cluster randomization with inflation dependent on intraclass correlation coefficient, the number of patients to be included was increased to 3000. The absolute reduction of 6% in mortality was established as a compromise resulting in a feasible sample size and a clinically relevant reduction in mortality. Recruitment was ultimately ended when the targeted sample size was achieved in each cluster.
Baseline characteristics of patients were analyzed as frequencies and percentages for categorical variables and as medians and interquartile ranges (IQRs) for continuous variables. Bivariable associations were evaluated using the t test for continuous variables and the χ2 or Fisher exact test for categorical variables as appropriate. Variables for all adjusted analyses were age, illness severity, initial clinical diagnosis, seniority of the emergency physician, time of ICU admission, baseline functional status, living place, and type of home support. Generalized estimating equation methods using robust sandwich estimators to estimate the variance-covariance matrix of the regression coefficient estimates were used to account for clustering. Binary outcomes (ICU admission rate, in-hospital death, death within 6 months, change in Index of Independence in Activities of Daily Living) were analyzed using logistic regression models. Absolute risk differences and relative risks (RRs) were derived from logistic regression models and 95% confidence intervals for these measures were obtained using nonparametric bootstrap methods with 1000 samples. The crude overall survival at 6 months was estimated by the Kaplan-Meier method and compared using a log-rank test. The 6-month survival adjusted for baseline characteristics was estimated using a Cox model. Adjusted survival curves were produced using an inverse probability-weighted Kaplan-Meier estimation. Significance was tested using a Cox regression model weighted by the same weights (inverse probability-weighted Cox). Health-related quality of life at 6 months was analyzed using linear regression models. We used multiple imputation for participants with missing data, using predictive mean matching for continuous variables, logistic regression for binary data, and polytomous regression for (unordered) categorical data. Ten imputations were drawn.
The analyses were performed on an intention-to-treat basis according to the prespecified analysis plan, without planned interim analysis. A post hoc exploratory analysis was performed to compare the characteristics of patients admitted to the ICU in each group. All analyses were performed at a 2-sided α=.05; a P<.05 was considered statistically significant. All analyses were performed by A.B. with R software, version 3.2.2 (R Foundation for Statistical Computing).
Results
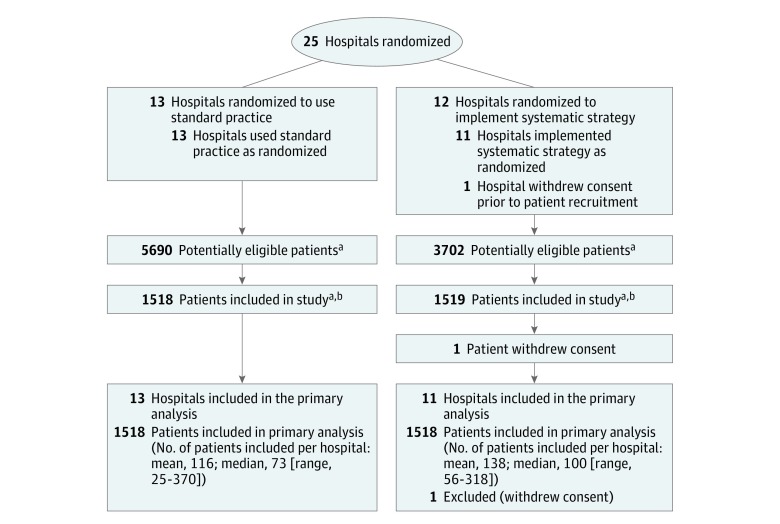
Between January 2012 and April 2015, a total of 3037 patients were enrolled in the trial, with 1519 patients in 11 hospitals randomized to the systematic strategy group and 1518 patients in 13 hospitals randomized to the standard practice group (Figure 1). The recruiting rate was higher in the systematic strategy group compared with the standard practice group. To achieve the total number of participants needed, the recruiting period was extended in the hospitals randomized to the standard practice group. The mean inclusion period was 22.5 months in the systematic strategy group and 28.5 months in the standard practice group. One patient subsequently withdrew consent, leaving a total of 3036 patients. There were 12 medical and 12 mixed (medical and surgical) ICUs.
Figure 1. Flow of Hospitals and Participants Through the ICE-CUB 2 Trial.
Cluster sizes are estimated according to the annual number of emergency department visits for all patients and for patients aged 75 years or older. There was no screening log in the emergency departments. The number of patients screened for inclusion in each hospital was estimated with the annual number of emergency department visits of patients aged 75 years or older, of which an estimated 8% (data from the ICE-CUB 1 study) were related to critical conditions, of which an estimated 33% (data from the ICE-CUB 1 study) fulfilled the inclusion criteria. Data are not available for the number of hospitals screened for eligibility or the number of or reasons for exclusion prior to randomization.
aInformation on number of patients not eligible or included is not available.
bParticipant recruitment ended when the target sample size was achieved.
Study Population
The baseline characteristics of the patients are presented in Table 1. The median age at inclusion was 85 years (IQR, 81-89 years); 45% (1361/3036) were men. The most frequent initial clinical diagnoses for hospital admission were septic shock (413/3036 [13.6%]), acute respiratory failure requiring noninvasive ventilation (347/3036 [11.4%]), severe pneumonia (250/3036 [8.2%]), and cardiac insufficiency requiring noninvasive ventilation (218/3036 [7.2%]) (a complete list is available in eTable 2 in Supplement 1). Patients in the systematic strategy group had a higher severity of illness at admission, as reflected by a higher Simplified Acute Physiology Score III score.
Table 1. Hospital and Participant Baseline Characteristicsa.
| Characteristics | Systematic Strategy | Standard Practice | Difference in Medians (95% CI) | P Value |
|---|---|---|---|---|
| Hospital characteristics | n=11 | n=13 | ||
| No. of emergency department visits among patients aged >75 y during the study period, mean (SD) | 12 746 (4402) | 16 580 (9468) | ||
| Location (Paris region), No. | 7 | 9 | ||
| Geriatric ward in the hospital, No. | 10 | 11 | ||
| Academic hospital, No. | 7 | 10 | ||
| Type of intensive care unit (medical), No. | 5 | 7 | ||
| Patient characteristics | n=1518 | n=1518 | ||
| Age, median (IQR), y | 85 (81-89) | 85 (81-89) | 0 (−1 to 1) | .60 |
| Male, No. (%) | 713 (47) | 648 (43) | ||
| Coexisting conditions, No./total No. (%) | ||||
| Ischemic heart disease or hypertension | 397/978 (41) | 456/1075 (42) | .40 | |
| Respiratory disorder | 296/978 (30) | 336/1074 (31) | .64 | |
| Congestive heart failure | 151/978 (15) | 119/1074 (11) | .004 | |
| Neurologic disorder | 112/979 (11) | 110/1075 (10) | .38 | |
| Cognitive impairment | 100/977 (10) | 153/1075 (14) | .006 | |
| Cirrhosis | 16/979 (2) | 16/1075 (1) | .79 | |
| SAPS 3 score at enrollment, median (IQR)b | 64 (57-71) | 59 (54-65) | 5 (4 to 6) | <.001 |
| Index of Independence in ADLs score, median (IQR)c | 6.0 (5.0-6.0) | 6.0 (5.5-6.0) | 0 (0 to 0) | .19 |
| Initial clinical diagnosis, No. (%)d | ||||
| Respiratory failure | 488 (32) | 491 (32) | <.001 | |
| Shock | 320 (21) | 238 (16) | ||
| Cardiac disorder | 177 (12) | 231 (15) | ||
| Coma | 187 (12) | 132 (9) | ||
| Gastrointestinal tract disorder | 57 (4) | 117 (8) | ||
| Acute kidney failure | 86 (6) | 61 (4) | ||
| Surgery | 26 (2) | 36 (2) | ||
| Multiple trauma without surgery | 10 (1) | 9 (1) | ||
| Other | 165 (11) | 202 (13) | ||
| Living situation, No./total No. (%) | ||||
| Home without assistance | 1070/1516 (71) | 968/1488 (65) | .01 | |
| Home with assistance | 255/1516 (17) | 307/1488 (21) | ||
| Nursing home | 108/1516 (7) | 98/1488 (7) | ||
| Long-term care facility | 72/1516 (5) | 97/1488 (7) | ||
| Hospital | 10/1516 (1) | 15/1488 (1) | ||
| Homeless | 1/1516 (0.1) | 3/1488 (0.2) | ||
| Home support, No./total No. (%) | ||||
| Living alone | 704/1443 (49) | 784/1415 (55) | <.001 | |
| Spouse/partner | 600/1443 (42) | 437/1415 (31) | ||
| Family | 139/1443 (10) | 194/1415 (14) |
Abbreviation: IQR, interquartile range.
A total of 3037 patients underwent randomization and 1 subsequently withdrew consent to use his data. Data are reported for available values. Because this is a cluster-randomized trial, difference in characteristics of clusters were not tested. 95% CIs for differences in medians were calculated using nonparametric bootstrap methods with 1000 samples. Participant characteristics reported in this table are known predictors of midterm outcome in critically ill elderly patients. Data for other participant characteristics are reported in the eTables in Supplement 2.
The Simplified Acute Physiology Score (SAPS) 3 ranges from 0 to 146, with higher scores indicating a more severe disease and a higher risk of death; data were available for 1469 patients in the systematic strategy group and 1383 patients in the standard practice group.
The Index of Independence in Activities of Daily Living (ADLs) is based on an evaluation of the functional independence or dependence of patients in bathing, dressing, toileting, transferring, continence, and feeding and ranges from 0 (totally dependent) to 6 (independent). Additional data on baseline Index of Independence in ADLs scores are available in eTable 7 in Supplement 2; data are available for 1330 patients in the systematic strategy group and 1200 patients in the standard practice group.
A detailed list of initial clinical diagnoses is available in eTable 2 in Supplement 2.
Characteristics of the Triage Process
In the systematic strategy group, physicians inquired more often about patients’ or surrogate decision makers’ opinions about ICU admission (750/1518 [49%] vs 359/1518 [24%]; difference in proportions, 25%; 95% CI, 22%-29%; P < .001), intensive care physicians were more often involved in the triage process (1474/1518 [97%] vs 938/1518 [62%]; difference in proportions, 35%; 95% CI, 33%-38%; P < .001) and were more favorable to an ICU admission (1111/1474 [75%] vs 623/938 [66%]; difference in proportions, 9%; 95% CI, 5%-13%; P < .001), and patients were more favorable to an ICU admission (414/470 [88%] vs 145/220 [66%]; difference in proportions, 22%; 95% CI, 15%-29%; P < .001) than in the standard practice group. More information about the triage process is available in Table 2 and eTable 3 in Supplement 1. Information on protocol violations is available in eTable 4 in Supplement 1.
Table 2. Characteristics of the Triage Process for ICU Admission.
| Characteristics | No./Total No. (%) | P Value | |
|---|---|---|---|
| Systematic Strategy (n = 1518) |
Standard Practice (n = 1518) |
||
| No empty ICU beds | 241/1276 (19) | 92/492 (19) | .98 |
| Physician sought participant’s opinion about ICU admissions | |||
| Yes | 470/1518 (31) | 220/1518 (14) | <.001 |
| No | 326/1518 (21) | 155/1518 (10) | |
| Patient unable to formulate opinion | 438/1518 (29) | 190/1518 (13) | |
| Not documented | 284/1518 (19) | 953/1518 (63) | |
| Participant opinion about ICU admission | |||
| Favorable | 414/470 (88) | 145/220 (66) | <.001 |
| Unfavorable | 21/470 (5) | 32/220 (15) | |
| No opinion | 35/470 (7) | 43/220 (20) | |
| Physician sought family’s opinion about ICU admission | |||
| Yes | 517/1518 (34) | 233/1518 (15) | <.001 |
| No | 337/1518 (22) | 155/1518 (10) | |
| Not present or could not be reached | 199/1518 (13) | 93/1518 (6) | |
| Not documented | 465/1518 (31) | 1037/1518 (68) | |
| Family’s opinion about ICU admissiona | |||
| Favorable | 421/514 (82) | 132/232 (57) | <.001 |
| Unfavorable | 69/514 (13) | 71/232 (30) | |
| No opinion | 24/514 (5) | 29/232 (13) | |
| Decision about admission destinationb | |||
| ICU in the same hospital | 856/1513 (57) | 458/1497 (31) | <.001 |
| ICU in another hospital | 75/1513 (5) | 58/1497 (4) | |
| Intermediate care or specialized unit | 242/1513 (16) | 319/1497 (21) | |
| Other ward | 104/1513 (7) | 369/1497 (25) | |
| Post–emergency department unit | 189/1513 (12) | 261/1497 (17) | |
| Geriatric unit | 17/1513 (1) | 24/1497 (2) | |
| Emergency department | 30/1513 (2) | 8/1497 (1) | |
Abbreviation: ICU, intensive care unit.
On 3 occasions in the systematic strategy group and 1 in the standard practice group, it was reported that physicians sought family opinions about ICU admission but the opinion was not reported.
Decision data may differ from actual admission destinations because some patients for whom a decision about ICU admission had been made were not subsequently admitted (eTable 5 in Supplement 2).
Primary Outcome
At 6 months, there were a total of 1273 deaths, 685 deaths in the systematic strategy group and 588 deaths in the standard practice group (Table 3). Patients in the systematic strategy group had an increased risk of death at 6 months (685/1518 [45%] vs 588/1518 [39%]; difference in proportions, 6% [95% CI, 3%-10%]; P < .001; RR, 1.16 [95% CI, 1.07-1.26]), but the difference did not remain significant after adjustments for baseline characteristics (RR, 1.05; 95% CI, 0.96-1.14). Crude and adjusted survival curves according to group assignment are presented in Figure 2.
Table 3. Primary and Secondary Outcomes.
| Outcomes | Systematic Strategy (n = 1518) |
Standard Practice (n = 1518) |
Difference in Mean or Rate (95% CI) | Relative Risk (95% CI) | Absolute Risk Difference, % (95% CI)a | P Valueb |
|---|---|---|---|---|---|---|
| Death at 6 mo (primary outcome), No. (%) | 685 (45) | 588 (39) | 6 (3 to 10) | 1.16 (1.07 to 1.26) | –6 (–9 to –3) | <.001 |
| Adjusted analysisc | 1.05 (0.96 to 1.14) | –2 (–6 to 2) | .28 | |||
| Intensive care unit admission rate, No. (%) | 932 (61) | 516 (34) | 27 (24 to 31) | 1.80 (1.66 to 1.95) | –27 (– 31 to –24) | <.001 |
| Adjusted analysisc | 1.68 (1.54 to 1.82) | –24 (–28 to –21) | <.001 | |||
| In-hospital mortality, No. (%) | 451 (30) | 326 (21) | 9 (5 to 11) | 1.39 (1.23 to 1.57) | –9 (–12 to –5) | <.001 |
| Adjusted analysisc | 1.18 (1.03 to 1.33) | –4 (–8 to –1) | .03 | |||
| Decrease in score in ≥1 domain of Index of Independence in ADLs, No./total No. (%)d | 463/680 (68) | 394/657 (60) | 8 (3 to 13) | 1.06 (0.99 to 1.13) | –4 (–8 to 0) | .08 |
| Adjusted analysisc | 1.02 (0.99 to 1.05) | –2 (–3 to 0) | .10 | |||
| SF-12 physical component score at 6 mo, mean (95% CI)e | 36.7 (35.9-37.5) | 36.2 (35.5-37.0) | 0.5 (–0.6 to 1.5) | 0.95 (–0.16 to 2.07)f | .09 | |
| Adjusted analysisc | 0.56 (–0.39 to 1.53)f | .24 | ||||
| SF-12 mental component score at 6 mo, mean (95% CI)e | 44.6 (44.1-45.1) | 43.7 (43.2-44.2) | 0.9 (0.1 to 1.6) | 1.05 (0.21 to 1.90)f | .02 | |
| Adjusted analysisc | 0.98 (0.15 to 1.81)f | .02 |
Absolute risk difference was calculated as probability of event in standard practice group minus probability of event in systematic strategy group.
P value for the coefficient associated with strategy equal to 0 in the logistic regression model and linear regression model as appropriate.
Analyses adjusted for the following baseline characteristics: age, illness severity, initial clinical diagnosis, seniority of the emergency department physician, time of intensive care unit admission, baseline functional status, living situation, and type of home support.
The Index of Independence in Activities of Daily Living (ADLs) is based on an evaluation of the functional independence or dependence of patients in bathing, dressing, toileting, transferring, continence, and feeding and ranges from 0 (totally dependent) to 6 (independent). The variable analyzed is the variation in at least 1 domain of the Index of Independence in ADLs. This is the only outcome with missing values; the estimated absolute risk difference is different than the observed difference in rates among groups.
The 12-Item Short Form Health Survey (SF-12) is a measure of health-related quality of life, includes physical and mental components, and ranges from 0 to 100, with higher scores representing better self-reported health.
Mean absolute increase in SF-12 score.
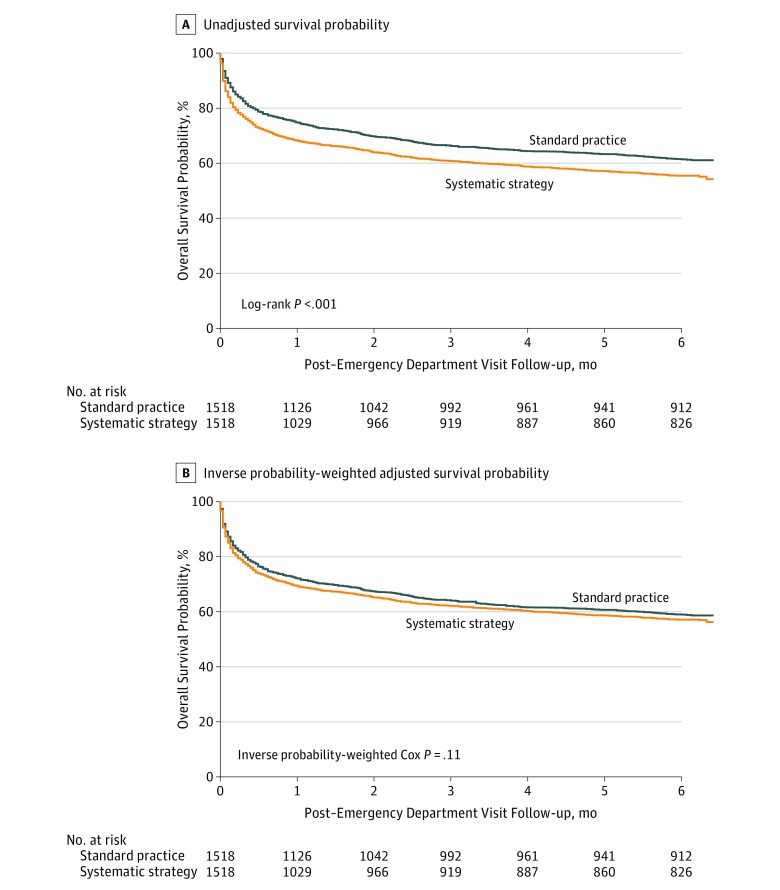
Figure 2. Unadjusted and Adjusted Probability of Survival.
Median duration of follow-up in alive patients was of 6 months (interquartile range, 6.0-6.1 months) in both the systematic strategy and the standard practice groups. Panel A shows Kaplan-Meier curves of the unadjusted probability of survival between the systematic strategy group and the standard practice group from emergency department visit to 6 months. Patients from the systematic strategy group had a lower 6-month survival rate vs patients in the standard practice group (Kaplan-Meier estimates, 55.7% [95% CI, 53.2%-58.2%] vs 61.8% [95% CI, 59.4%-64.3%]; P < .001; hazard ratio, 1.24; 95% CI, 1.02-1.51). Panel B shows Kaplan-Meier curves of the inverse probability-weighted adjusted probability of survival from emergency department visit to 6 months. The P value from the inverse probability-weighted Cox regression model is shown in the graph. After adjustments for baseline characteristics, survival rates at 6 months were not significantly different between groups (hazard ratio, 1.10; 95% CI, 0.93-1.31).
Secondary Outcomes
Intensive care unit admission rate was higher in the systematic strategy group (932/1518 [61%] vs 516/1518 [34%]; difference in proportions, 27% [95% CI, 24%-31%]; P < .001; RR, 1.80 [95% CI, 1.66-1.95]), and the difference remained significant after adjustments for baseline characteristics (RR, 1.68; 95% CI, 1.54-1.82) (Table 3). One patient for which a decision for ICU refusal had been made was finally admitted to the ICU (eTable 5 in Supplement 1).
A post hoc analysis was conducted to compare patients admitted to the ICU in each group (eTable 6 in Supplement 1). Patients admitted to the ICU in the systematic strategy group had a higher Simplified Acute Physiology Score III score (difference in medians, 3; 95% CI, 2-5; P < .001), more often underwent mechanical ventilation (374/884 [42%] vs 147/470 [31%]; difference in proportions, 11%; 95% CI, 6%-16%; P < .001), less often underwent noninvasive ventilation (251/884 [28%] vs 170/470 [36%]; difference in proportions, –8%; 95% CI, –13% to –3%; P < .001) and less often underwent fluid resuscitation (177/835 [21%] vs 151/469 [32%]; difference in proportions, –11%; 95% CI, –16% to –6%; P < .001) than patients admitted to the ICU in the standard practice group. The ICU and hospital length of stay were not significantly different between groups (difference in medians, respectively, of 1 [95% CI, –0.5 to 1] days and 1 [95% CI, –1 to 3] days). The overall mortality rate in the ICU was 23% (328/1448) and was not significantly different between groups (220/932 [24%] for systematic strategy vs 108/516 [21%] for standard practice; difference in proportions, 3%; 95% CI, –2% to 7%; P = .27).
The overall in-hospital mortality was 25.6% (777/3036). In-hospital mortality was higher in the systematic strategy group than in the standard practice group (451/1518 [30%] vs 326/1518 [21%]; difference in proportions, 9% [95% CI, 5%-11%]; P < .001; RR, 1.39 [95% CI, 1.23-1.57]), and the difference remained significant after adjustments for baseline characteristics (RR, 1.18; 95% CI, 1.03-1.33) (Table 3). Patients discharged alive from the hospital in the systematic strategy group had an increased length of hospital stay vs patients in the standard practice group (16.8 [SD, 16.2] days in the systematic strategy group and 13.6 [SD, 20.1] days in the standard practice group; difference in means, 3.2 days; 95% CI, 1.7-4.7 days; P < .001).
There was a greater decrease from baseline Index of Independence in Activities of Daily Living at 6 months in the systematic strategy group than in the standard practice group (median of the difference between follow-up and baseline scores, –0.5 [IQR, –2.0 to 0.0] vs –0.5 [IQR, –1.5 to 0.0]; P = .02; difference in means, –0.20; 95% CI, –0.37 to –0.03) (detailed baseline and follow-up data are reported in eTables 7 and 8 in Supplement 1). The probability of a decrease in at least 1 of the Index of Independence in Activities of Daily Living domains was not significantly different between groups after adjustments for baseline characteristics (RR, 1.02; 95% CI, 0.99-1.05) (Table 3). Self-reported physical quality of life at 6 months was not significantly different between groups (mean score, 36.7 vs 36.2; difference in means, 0.5 [95% CI, –0.6 to 1.5]). Self-reported mental quality of life at 6 months was higher in the systematic strategy group (mean score, 44.6 vs 43.7; difference in means, 0.9 [95% CI, 0.1-1.6]) (Table 3). Additional data at 6-month follow-up are available in eTable 9 in Supplement 1.
Discussion
In this trial, a program to promote systematic ICU admission of critically ill elderly patients led to a higher ICU admission rate and hospital mortality but after adjustment for baseline characteristics had no significant effect on mortality, functional status, or physical health-related quality of life at 6 months. The observed statistically significant difference in median mental health-related quality of life did not reach the minimum clinically important difference.
These results are consistent with the ICE-CUB 1 study, a prospective, multicenter observational study of 2646 critically ill patients aged 80 years or older, which suggested no mortality benefit at 6 months after ICU admission (50.6% vs 50.7%). Other observational studies reported controversial results: 2 studies suggested decreased mortality at 1 month in patients admitted vs not admitted to the ICU, whereas 1 study did not observe mortality benefit at 1 year. Limitations of these studies are their retrospective or observational design without randomization, their heterogeneity in age cutoff for inclusion and in adjustments for known prognostic factors, and the absence of consideration of the triage process by the emergency department physicians before the ICU referral.
The mortality rates were also consistent with available data in the literature. Recent observational data on critically ill elderly patients admitted to the ICU suggested in-hospital mortality rates ranging from 24% to 40%, 3-month mortality rates from 39% to 41%, 6-month mortality rates from 37% to 51%, and 1-year mortality rates from 44% to 68%. In the current trial, the selection of patients with preserved baseline functional and nutritional status and free of active cancer may explain the observed low rates of in-hospital (26%) and 6-month (42%) mortality compared with observational data. The higher in-hospital mortality rate observed in the systematic strategy group may be explained in part by the fact that a higher ICU admission rate in the intervention group might have precipitated early discussions about or more frequent withdrawals of life-sustaining therapies, possibly leading to a higher mortality rate in the systematic strategy group. However, the ICU and hospital lengths of stay and the number of patients with at least 1 organ in need of support were similar between the 2 groups.
To our knowledge, this is the first randomized clinical trial evaluating the effect of ICU triage on long-term mortality in an elderly population. Randomization by cluster (hospital) allowed evaluation of the effect of ICU triage without difficulties incurred by ethical considerations of randomization for ICU admission at an individual patient level. The intervention program led to different ICU admission rates between groups. Furthermore, senior physicians were more frequently involved in the triage decision-making process, and the decisions were frequently shared between physicians. However, despite a recommendation for systematic ICU admission in the intervention group, more than one-third of patients were not admitted to the ICU. This was expected, as physicians had the final decision for ICU admission and could refuse an ICU admission even if the hospital was randomized to the intervention group.
This trial has several strengths. First, the design takes into account the 2 steps of the triage process by both emergency department and intensive care physicians. This avoided potential bias related to variation in triage criteria for ICU admission proposal by emergency department physicians. Second, participants were patients without long-term adverse outcome factors as identified in the ICE-CUB 1 study, to avoid inclusion of patients with very high expected mortality. Third, the trial was pragmatic, which contributes to the generalizability of the results. Fourth, in addition to survival analyses, this trial assessed functional status and quality of life, which are important and relevant outcomes in the elderly population. Fifth, there were very few protocol violations, allowing for an intention-to-treat analysis, and few patients were lost to follow-up.
This study also has several limitations. First, the recruitment period in the standard practice cluster was longer that in the systematic strategy cluster, which could have introduced confounding due to secular changes. Second, because of the nature of the intervention, no blinding to group assignment was possible. However, the effect of the nonblinded design on 6-month mortality is considered low. Third, data on withdrawal of life-sustaining therapies were not collected. Fourth, the potential benefits of ICU admission might be confounded by recruitment of patients who were more severely ill in the systematic strategy group. However, results were adjusted for several baseline characteristics, including illness severity. Fifth, the benefits of increased ICU admission could accrue only to patients who would normally have received floor care but instead received ICU care. In this study, the question of whether benefit might accrue to patients on the margin was not addressed. However, because of the design of the study, variation in individual probability of ICU admission is likely to be small. Sixth, a qualitative approach to understand why patients were not admitted to the ICU was not conducted. Seventh, differences in the quality of care provided in the wards may affect the outcome. For example, ward admission as an alternative to the ICU might partially explain the negative results observed in the present study when patients were admitted to wards providing very good care. The cluster design and sample size should reduce this potential bias because hospitals with emergency departments and ICUs were randomized. Moreover, patients already in the hospital (secondary transfer) or admitted from another hospital were excluded to minimize the effect of previous quality of care on prognosis. Nonetheless, the hospitals that had lower ICU admission rates at baseline may in particular have developed higher-quality ward care; thus, a strategy to boost ICU admission rates may not have yielded the same gains in outcome in such centers as in other settings.
The findings of this study may be informative for health care delivery from a hospital perspective. When considering the increasing demand for intensive care among elderly patients, the importance of health care resource allocation and expenditure control, and the possible absence of long-term benefit of intensive care, systematic ICU admission of all critically ill elderly patients might not be warranted. However, this study should not be interpreted as suggesting that no elderly patient should be admitted to the ICU. Because of substantial uncertainty in outcomes among individual patients, there is a need to systematically and thoughtfully assess the potential benefits and harms of ICU admission for every elderly patient presenting with critical illness.
Conclusions
Among critically ill elderly patients in France, a program to promote systematic ICU admission increased ICU use but did not reduce 6-month mortality. Additional research is needed to understand the decision to admit elderly patients to the ICU.
eAppendix. Supplemental Methods
eTable 1. List of the Critical Conditions for Inclusion
eTable 2. Detailed List of Initial Clinical Diagnoses of Included Patients
eTable 3. Characteristics of the Triage Process
eTable 4. Number and Characteristics of Protocol Violations
eTable 5. Number of Decisions for ICU Admission to the ICU and Number of Patients Actually Admitted
eTable 6. Characteristics of Patients Admitted to the Intensive Care Unit
eTable 7. Baseline Index of Independence in ADL
eTable 8. Index of Independence in ADL Scale at 6 Months
eTable 9. Additional Data at 6-Month Follow-up
eReferences
Trial Protocol
Statistical Analysis Plan
Section Editor: Derek C. Angus, MD, MPH, Associate Editor, JAMA (angusdc@upmc.edu).
References
- 1.Flaatten H, de Lange DW, Artigas A, et al. The status of intensive care medicine research and a future agenda for very old patients in the ICU. Intensive Care Med. 2017;43(9):1319-1328. [DOI] [PubMed] [Google Scholar]
- 2.Chin-Yee N, D’Egidio G, Thavorn K, Heyland D, Kyeremanteng K. Cost analysis of the very elderly admitted to intensive care units. Crit Care. 2017;21(1):109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nguyen YL, Angus DC, Boumendil A, Guidet B. The challenge of admitting the very elderly to intensive care. Ann Intensive Care. 2011;1(1):29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Packham V, Hampshire P. Critical care admission for acute medical patients. Clin Med (Lond). 2015;15(4):388-391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bagshaw SM, Webb SA, Delaney A, et al. Very old patients admitted to intensive care in Australia and New Zealand: a multi-centre cohort analysis. Crit Care. 2009;13(2):R45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Boumendil A, Angus DC, Guitonneau AL, et al. ; ICE-CUB study group . Variability of intensive care admission decisions for the very elderly. PLoS One. 2012;7(4):e34387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heyland D, Cook D, Bagshaw SM, et al. ; Canadian Critical Care Trials Group; Canadian Researchers at the End of Life Network . The very elderly admitted to ICU: a quality finish? Crit Care Med. 2015;43(7):1352-1360. [DOI] [PubMed] [Google Scholar]
- 8.Sprung CL, Artigas A, Kesecioglu J, et al. The Eldicus prospective, observational study of triage decision making in European intensive care units, II: intensive care benefit for the elderly. Crit Care Med. 2012;40(1):132-138. [DOI] [PubMed] [Google Scholar]
- 9.Valley TS, Sjoding MW, Ryan AM, Iwashyna TJ, Cooke CR. Association of intensive care unit admission with mortality among older patients with pneumonia. JAMA. 2015;314(12):1272-1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fuchs L, Novack V, McLennan S, et al. Trends in severity of illness on ICU admission and mortality among the elderly. PLoS One. 2014;9(4):e93234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW. Studies of illness in the aged: the Index of ADL: a standardized measure of biological and psychosocial function. JAMA. 1963;185:914-919. [DOI] [PubMed] [Google Scholar]
- 12.Gandek B, Ware JE, Aaronson NK, et al. Cross-validation of item selection and scoring for the SF-12 Health Survey in nine countries: results from the IQOLA Project. J Clin Epidemiol. 1998;51(11):1171-1178. [DOI] [PubMed] [Google Scholar]
- 13.Parker SL, Mendenhall SK, Shau D, et al. Determination of minimum clinically important difference in pain, disability, and quality of life after extension of fusion for adjacent-segment disease. J Neurosurg Spine. 2012;16(1):61-67. [DOI] [PubMed] [Google Scholar]
- 14.Zarit SH, Reever KE, Bach-Peterson J. Relatives of the impaired elderly: correlates of feelings of burden. Gerontologist. 1980;20(6):649-655. [DOI] [PubMed] [Google Scholar]
- 15.Ospina-Tascón GA, Büchele GL, Vincent JL. Multicenter, randomized, controlled trials evaluating mortality in intensive care: doomed to fail? Crit Care Med. 2008;36(4):1311-1322. [DOI] [PubMed] [Google Scholar]
- 16.Austin PC. Absolute risk reductions, relative risks, relative risk reductions, and numbers needed to treat can be obtained from a logistic regression model. J Clin Epidemiol. 2010;63(1):2-6. [DOI] [PubMed] [Google Scholar]
- 17.Pavoni V, Gianesello L, Paparella L, Buoninsegni LT, Mori E, Gori G. Outcome and quality of life of elderly critically ill patients: an Italian prospective observational study. Arch Gerontol Geriatr. 2012;54(2):e193-e198. [DOI] [PubMed] [Google Scholar]
- 18.Moreno RP, Metnitz PG, Almeida E, et al. ; SAPS 3 Investigators . SAPS 3—from evaluation of the patient to evaluation of the intensive care unit, 2: development of a prognostic model for hospital mortality at ICU admission. Intensive Care Med. 2005;31(10):1345-1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boumendil A, Latouche A, Guidet B; ICE-CUB Study Group . On the benefit of intensive care for very old patients. Arch Intern Med. 2011;171(12):1116-1117. [DOI] [PubMed] [Google Scholar]
- 20.Leblanc G, Boumendil A, Guidet B. Ten things to know about critically ill elderly patients. Intensive Care Med. 2017;43(2):217-219. [DOI] [PubMed] [Google Scholar]
- 21.Andersen FH, Flaatten H, Klepstad P, Romild U, Kvåle R. Long-term survival and quality of life after intensive care for patients 80 years of age or older. Ann Intensive Care. 2015;5(1):53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Duke GJ, Barker A, Knott CI, Santamaria JD. Outcomes of older people receiving intensive care in Victoria. Med J Aust. 2014;200(6):323-326. [DOI] [PubMed] [Google Scholar]
- 23.Heyland DK, Garland A, Bagshaw SM, et al. Recovery after critical illness in patients aged 80 years or older: a multi-center prospective observational cohort study. Intensive Care Med. 2015;41(11):1911-1920. [DOI] [PubMed] [Google Scholar]
- 24.Zeng A, Song X, Dong J, et al. Mortality in relation to frailty in patients admitted to a specialized geriatric intensive care unit. J Gerontol A Biol Sci Med Sci. 2015;70(12):1586-1594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Le Maguet P, Roquilly A, Lasocki S, et al. Prevalence and impact of frailty on mortality in elderly ICU patients: a prospective, multicenter, observational study. Intensive Care Med. 2014;40(5):674-682. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eAppendix. Supplemental Methods
eTable 1. List of the Critical Conditions for Inclusion
eTable 2. Detailed List of Initial Clinical Diagnoses of Included Patients
eTable 3. Characteristics of the Triage Process
eTable 4. Number and Characteristics of Protocol Violations
eTable 5. Number of Decisions for ICU Admission to the ICU and Number of Patients Actually Admitted
eTable 6. Characteristics of Patients Admitted to the Intensive Care Unit
eTable 7. Baseline Index of Independence in ADL
eTable 8. Index of Independence in ADL Scale at 6 Months
eTable 9. Additional Data at 6-Month Follow-up
eReferences
Trial Protocol
Statistical Analysis Plan