Abstract
BACKGROUND/OBJECTIVES
This study aimed to investigate the association of dietary patterns with overweight risk and all-cause mortality in pediatric cancer patients.
SUBJECTS/METHODS
Prospective cohort study was undertaken; 83 cancer patients admitted to the pediatric cancer ward at a university hospital in Seoul were included and followed for obesity and death over 24 months. Food consumption data were collected from patients using validated meal order sheets for breakfast, lunch, and dinner at the pediatric cancer ward over 3 days. Using principal component analysis, three dietary patterns were derived from 29 food groups.
RESULTS
Eighteen deaths occurred among the patient cohort during the follow-up period. The “spicy & fried meat and fish” dietary pattern was positively associated with overweight risk at both baseline [odds ratio (OR) = 4.396, 95% confidence interval (CI) = 1.111-17.385, P for trend = 0.023] and after 6 months (OR = 4.088, 95% CI = 1.122-14.896, P for trend = 0.025) as well as all-cause mortality (hazard ratios = 5.124, 95% CI = 1.080-24.320, P for trend = 0.042), when comparing the highest and lowest tertiles after adjusting for covariates. The “fish, egg, meat, and fruits & vegetables” dietary pattern was associated with lower overweight risk after 24 months (OR = 0.157, 95% CI = 0.046-0.982, P for trend = 0.084).
CONCLUSION
The results imply that dietary patterns might be associated with weight gain and premature death among pediatric cancer patients.
Keywords: Child, neoplasms, obesity, diet, mortality
INTRODUCTION
Continued technological advancements in cancer treatment have led to significant improvements in the prognostic outcomes and survival rates of many types of childhood malignancies [1]. According to data from Statistics Korea, the relative 5-year survival rates of pediatric cancer patients have increased from 41.2% (1993 to 1995) to 68.1% (2008 to 2012), with treatment rates reported to be as high as 80%. Despite this success, childhood cancer survivors face numerous long-term complications, including obesity, endocrine disorders (e.g., thyroid disease, disorders affecting regulation of pubertal growth), cardiac disease, osteonecrosis, pulmonary disorders, neurological and neurosensory disorders, and secondary cancers [2,3]. Over 60% of pediatric cancer survivors have been reported to suffer from one or more of these complications, depending on the diagnostic category and study population [4,5]. Consequently, long-term monitoring has become an important part of the overall healthcare strategy for pediatric cancer survivors.
Obesity is recognized as a major long-term complication affecting the health status and mortality of pediatric cancer patients, and long-term survivors of pediatric cancer are more likely to be obese than children of the same age and sex in the reference population [6]. Overweight or obesity has known to be associated with increased incidence of treatment-related mortality [7,8] and relapse risk [9] as well as reduction of overall quality of life of patients due to physical impairment and negative psychological impact [10]. However, the mechanism(s) contributing to obesity in pediatric cancer patients have not been clearly identified. Various factors such as hypothalamic-pituitary axis damage from cancer treatments, adverse effects of chemotherapy, growth hormone deficiencies, steroid use, biological characteristics at diagnosis, inactivity, and diet have been considered as potential causes of obesity in pediatric cancer patients [11]. Among them, diet is one of the main factors that can be modified through lifestyle interventions in order to prevent obesity and premature death in pediatric cancer patients.
Previous studies have reported that pediatric cancer patients are more likely to exhibit unhealthy dietary behaviors, including low intakes of fruits and vegetables [12,13,14,15] and whole grains [12,15] as well as high intake of energy from fat [12,15,16], sweets [12,15], sodium [12], and refined carbohydrates [15]. However, most studies have focused on the relationship between individual nutrients or foods and obesity among children with cancer. Until now, the association between overall diet quality and obesity or mortality has not yet been evaluated.
A dietary pattern approach can be useful for assessing associations between overall diet quality and health outcomes, as it takes into account the complex interactions between nutrients and other dietary components [17]. To the best of our knowledge, very few reported studies have focused on the association of dietary patterns with obesity and mortality in pediatric cancer patients. Therefore, we examined associations between dietary patterns and overweight risk and all-cause mortality using data from pediatric cancer patient cohorts.
SUBJECTS AND METHODS
Participants
Of 130 children admitted to the pediatric cancer ward between October 28, 2013 and January 31, 2014, 83 who were diagnosed with cancer in accordance with the International Classification of Childhood Cancer (Third Edition) were selected as study subjects. Thirty-five patients prescribed to receive a low microbial diet for less than 3 consecutive days were excluded. Six patients lacking anthropometric data at diagnosis due to transfer from other hospitals as well as six patients hospitalized for peripheral blood stem cell transplantation were excluded. Information on general characteristics and disease status of subjects, including gender, age, age at diagnosis, diagnosis of cancer, steroid treatment, duration of disease, and absolute neutrophil counts (ANCs), was obtained through the electronic medical chart. In instances where patients were hospitalized more than once during the study period, data from the first hospitalization were used. All procedures were approved by the Catholic Medical Center Institutional Review Board (No. KC15RISI0145).
Medical examination and death ascertainment
Height and weight were measured by trained nursing staffs following standard procedures in order to assess overweight status at admission and follow-ups: after 6 months and 24 months. According to the recommendations from the Nutrition Committee of the Children's Oncology Group, a patient was defined as being overweight if the percentage of ideal body weight (PIBW) was ≥ 110% [18]. Immunocompromised patients were defined as those with ANCs < 500, in accordance with the American Cancer Society (2010). Deaths were confirmed by a review of medical records up to January 31, 2016.
Analysis of dietary patterns
Dietary patterns were analyzed using data from the meal order sheets for breakfast, lunch, and dinner at the pediatric cancer ward. The meals available on the order form consisted of both Korean and Western type food items, with menu rotations in 10-day cycles. Each patient could select a maximum of six food items every day for breakfast (of 23, 26.1%), lunch (of 19, 31.6%), and dinner (of 16, 37.5%). We collected data on all food items ordered over a 3-day period. The validity of food selection frequency from the meal order forms was examined by comparison with food intake using the 3-day 24-hour recall data. The mean of pearson correlation coefficients between the two methods was 0.42, ranging from 0.00 (cookies) to 0.91 (other grains).
For dietary pattern analysis, 220 food items consumed by the subjects were categorized into 29 groups, based on the food groups and items defined by Shin et al. [19] and the Korean National Health and Nutrition Examination Survey (KNHANES). Foods that could not be consumed on a low microbial diet (e.g., carbonated drinks, hamburgers, chocolate, ice cream, gum, candy, seeds and nuts, sandwiches, and simple sugars) were excluded as nobody selected. Foods that tended to be preferred by patients during chemotherapy, such as fried meat, processed meat, potatoes, and seasoned spicy meat, etc. [20,21], were divided into greater detail to reflect their dietary characteristics. In addition, the “grains” and “chicken” that children eat frequently were divided into “white rice” and “other grains” as well as “fried chicken”, “seasoned spicy chicken”, and “chicken”, respectively.
Statistical analysis
All analyses were performed using Statistical Package for the Social Sciences for Windows Version 12.0, 2004, (SPSS Inc., Chicago, IL, USA). Statistical significance was defined by a P-value below 0.05. Comparisons between variables were conducted using linear regression analyses and the Mantel-Haenszel chi-square test. All statistical tests were two-sided.
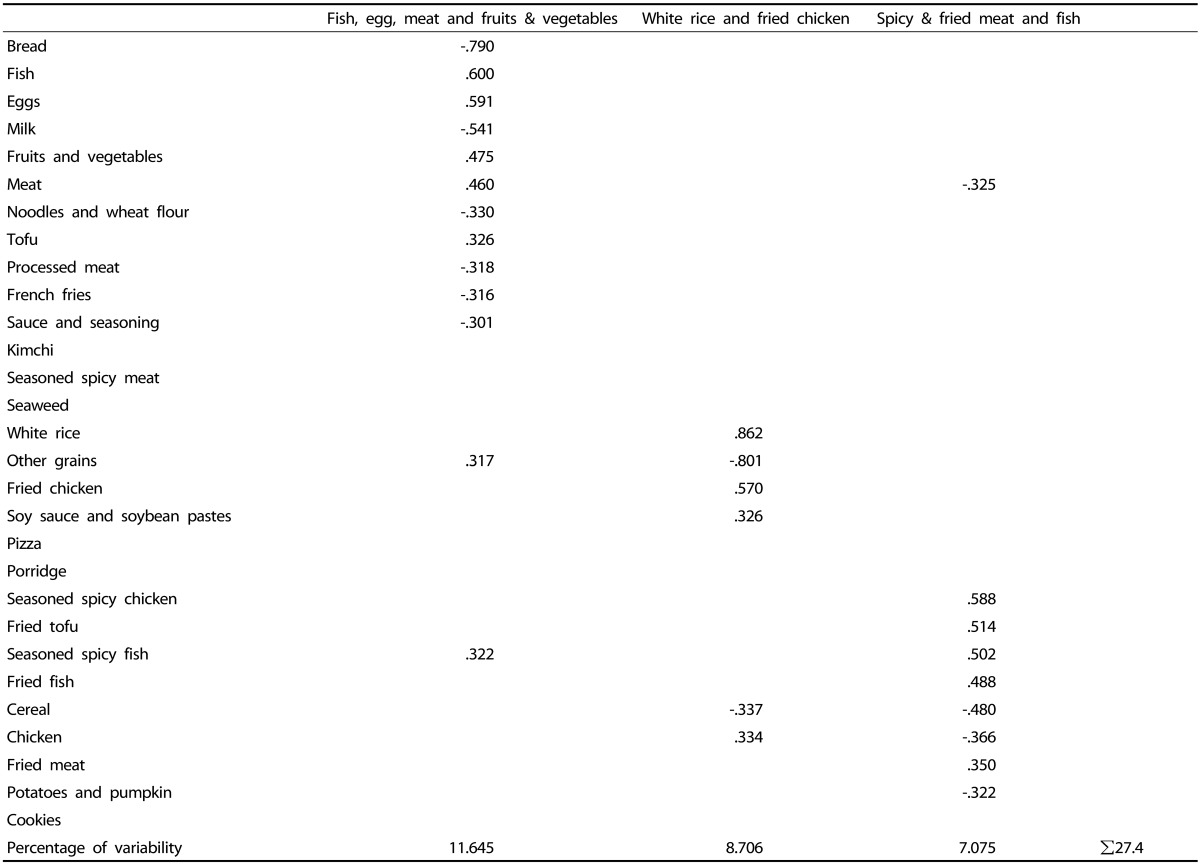
To derive dietary patterns, principal component analysis was performed, and three distinct dietary patterns were identified through varimax rotation. The number of factors was selected from eigenvalues > 1.25 with identification of a break point in the scree plots [22]. Foods or food groups with an absolute factor loading > 0.30 are presented in Table 1 participants were divided into three groups based on the tertile of each dietary pattern score, with the lowest tertile defined as the reference category. Relationships between dietary patterns and overweight were analyzed using logistic regression analysis, adjusted for covariates. The final model (Model 2) was controlled for sociodemographic characteristics such as age (years, continuous) and gender (men, women), and treatment effects such as diagnostic categories [Acute lymphoblastic leukemia (ALL), Peripheral blood stem cell transplantation (PBSCT), and Others] and duration of disease (day, continuous). The results were presented as odds ratios (OR) with 95% confidence intervals (CI). The Cox proportional hazard model was used to examine the association between dietary pattern and all-cause mortality, adjusted for age at diagnosis (years, continuous), gender (men, women), diagnostic categories (ALL, PBSCT, and Others), and duration of disease (day, continuous), and the results are presented as hazard ratios (HR) with 95% CI. In the survival analysis, age at diagnosis was used as a confounding variable instead of age. To test for linear trends across tertiles of dietary pattern scores, we created a continuous variable using the median value of each tertile of total dietary pattern scores.
Table 1. Factor loading matrix1) for the three dietary patterns2).
1)The correlation among the food groups and each factor (dietary patterns). For simplicity, factor loading values less than 0.30 are not listed in the matrix.
2)Three dietary patterns were derived by principal component analysis after entering the 29 food groups into the FACTOR PROCEDURE.
RESULTS
Dietary patterns of subjects
Factor analysis revealed three distinct dietary patterns: “fish, egg, meat, and fruits & vegetables”, “white rice and fried chicken”, and “spicy & fried meat and fish” based on the factor loadings (Table 1). The three dietary patterns explained 27.4% of the variance in total food intake (11.7%, 8.7%, and 7.1%, respectively). The “fish, egg, meat, and fruits & vegetables” pattern featured high consumption of fish, eggs, fruits & vegetables, meat, tofu, seasoned spicy fish, and other grains. The “white rice and fried chicken” pattern was highly correlated with white rice, fried chicken, soy sauce & soybean pastes, and chicken. The “spicy & fried meat and fish” pattern showed a high loading for seasoned spicy chicken, fried tofu, seasoned spicy fish, fried fish, and fried meat.
Baseline characteristics of subjects according to the dietary patterns
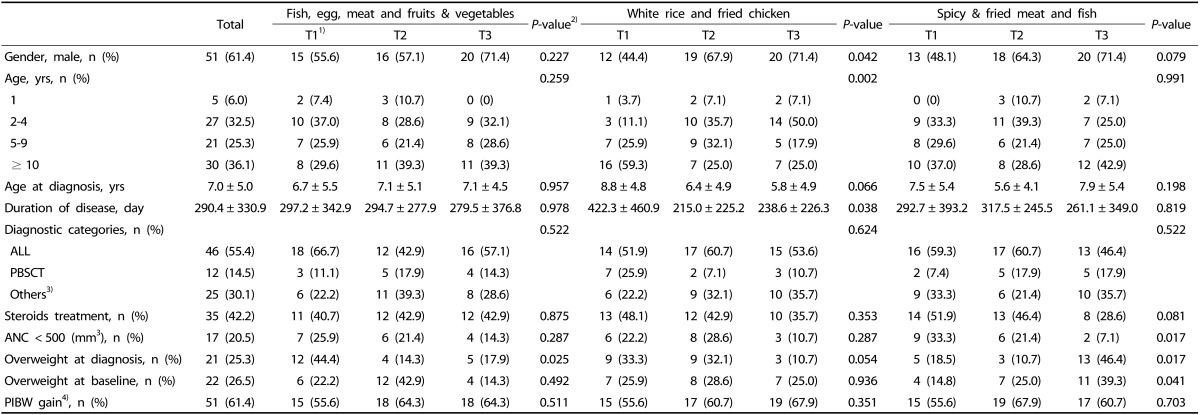
The baseline characteristics of the study participants are shown in Table 2. The mean age was 7.6 years, and the percentage of boys was 61.4%. The disease with the highest number of cases was ALL (n = 46, 55.4%), followed by other pediatric cancers (n = 25, 30.1%) and PBSCT (n = 12, 14.5%). In all, 35 children (42.2%) were receiving steroid treatment at the study baseline. The percentage of children that were overweight at the study baseline was 26.5% (n = 22). The number of patients in whom PIBW had increased from the time of diagnosis to the study baseline was 51 (61.4%).
Table 2. Subjects' baseline characteristics by tertiles of dietary pattern scores.
ALL, acute lymphoblastic leukemia; PBSCT, peripheral blood stem cell transplantation; ANC, absolute neutrophil count; PIBW, percentage of ideal body weight; T, tertile.
Data are presented as n (%) or mean ± standard deviations.
1)Tertiles of dietary pattern scores.
2)P for trend was calculated from a linear regression analysis for continuous variables and Mantel-Haenszel χ2 for categorical variables.
3)Others: acute myeloblastic leukemia, neuroblastoma, aplastic anemia, wilms tumor, relapsed lymphoma, sarcoma, osteosarcoma, etc.
4)PIBW gain from the diagnostic point.
Children with high scores for the “fish, egg, meat, and fruits & vegetables” pattern were less likely to be overweight at diagnosis (P = 0.025). Higher scores for the “white rice and fried chicken” pattern were observed in boys (P = 0.042), younger children (P = 0.002), and those with a shorter duration of disease (P = 0.038). Children with high scores for the “spicy & fried meat and fish” pattern were more likely to be overweight at diagnosis (P = 0.017) and baseline (P = 0.041) as well as have normal ANCs (P = 0.017).
Association between dietary pattern and overweight
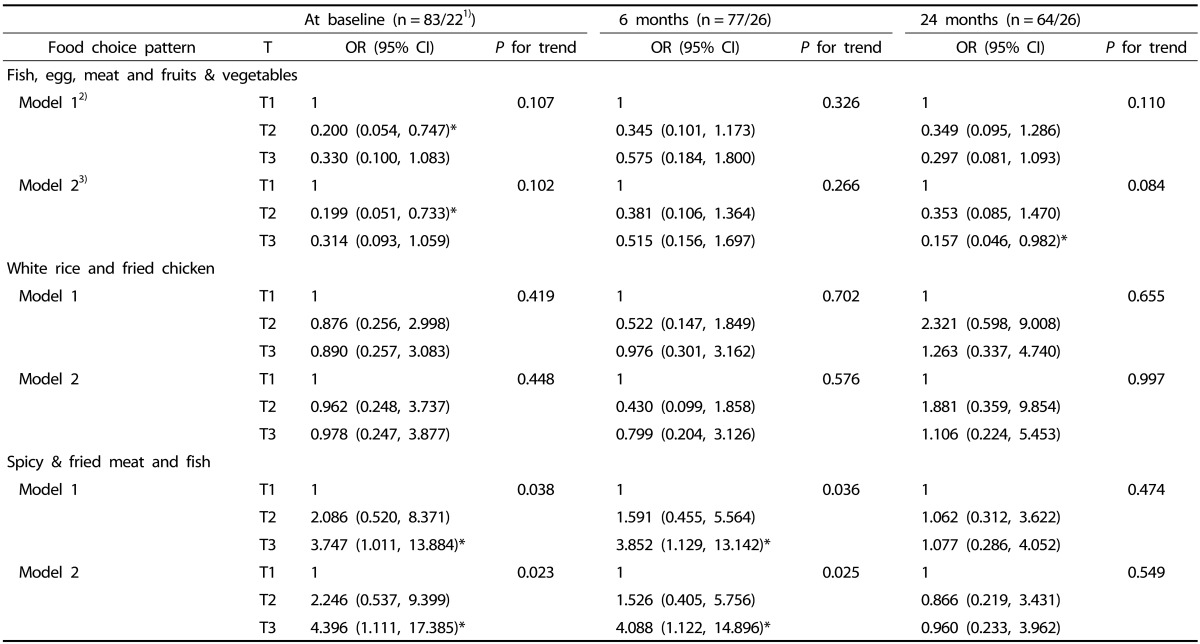
The OR and 95% CI of overweight according to the tertile of each dietary pattern are presented in Table 3. Children in the highest tertile of the “spicy & fried meat and fish (T3)” pattern score were more likely to be overweight at both baseline (OR = 4.396, 95% CI = 1.111-17.385, P for trend = 0.023) and after 6 months (OR = 4.088, 95% CI = 1.122-14.896, P for trend = 0.025), as compared to the lowest tertile of the pattern score, after adjusting for age, gender, diagnostic categories, and duration of disease. Children in the highest tertile of the “fish, egg, meat, and fruits & vegetables (T3)” pattern score showed lower risk of overweight at 24 months (OR = 0.157, 95% CI = 0.046-0.982, P for trend = 0.084), as compared to the lowest tertile of the pattern score, after adjusting for covariates. The “white rice and fried chicken” pattern, however, did not show any association with overweight in pediatric cancer patients.
Table 3. Odds ratios (95% CI) for overweight risk according to the tertiles (T) of dietary pattern scores at baselines and follow-ups.
OR, odds ratio; CI, confidence interval; T, tertile.
1)n = total patients/overweight patients
2)Model 1 adjusted for age (years, continuous).
3)Model 2 adjusted for age (years, continuous), gender (men, women), diagnostic categories (acute lymphoblastic leukemia, peripheral blood stem cell transplantation, and others), and duration of disease (day, continuous).
*P < 0.05
Association between dietary pattern and all-cause mortality
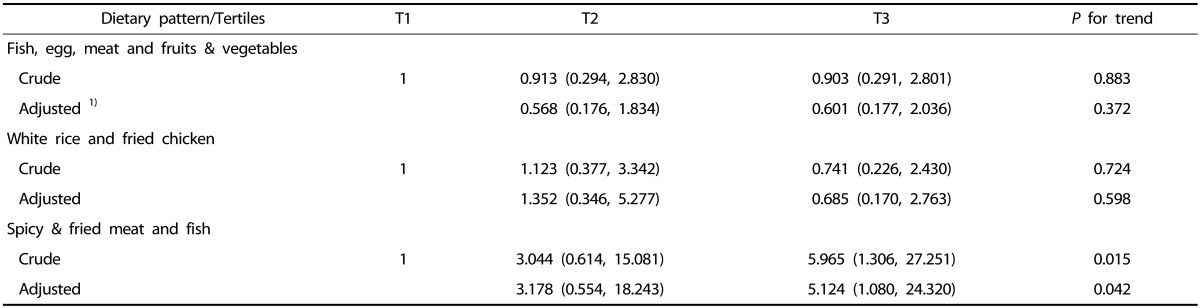
During the study period, 18 patients died while one patient was transferred to another facility. After adjusting for potential confounders, pediatric cancer patients in the highest tertile of the “spicy & fried meat and fish (T3)” pattern score showed increased risk of all-cause mortality (HR = 5.124, 95% CI = 1.080-24.320, P for trend = 0.042), as compared to the lowest tertile of the pattern score. No associations were observed between the other patterns and mortality (Table 4).
Table 4. Hazard ratios (HR) and 95% confidence intervals (95% CIs) for all-cause mortality by tertiles (T) of dietary pattern scores.
1)Cox proportional hazards regression model adjusted for age at diagnosis (years, continuous), gender (men, women), diagnostic categories (acute lymphoblastic leukemia, peripheral blood stem cell transplantation, and Others), and duration of disease (day, continuous).
DISCUSSION
We identified three distinct dietary patterns among pediatric cancer patients. The “spicy & fried meat and fish” dietary pattern was positively associated with overweight risk and all-cause mortality, whereas the “fish, egg, meat, and fruits & vegetables” dietary pattern was associated with decreased overweight risk.
In the present study, the three identified dietary patterns explained 27.4% of the total variance in food intake, which is similar to a previous study (28.4%) by Shin et al. [19] on four types of dietary patterns (“traditional Korean,” “fast food”, “milk and cereals”, and “snacks”) consisting of similar food items and groups as the present study. Since total variance can differ according to the number of food items/dietary patterns, direct comparisons between studies are difficult [23,24]. However, most studies have used a threshold eigenvalue of 1.0 in order to identify two to three dietary patterns that explain 21.7-30.5% of the total variance [19,25,26]. When these dietary patterns were compared with previous results from the same country, the “fish, egg, meat, and fruits & vegetables” pattern exhibited a balanced dietary composition centered on rice and kimchi with fish, legumes, and seaweed [19]. Although most previous studies conducted in Korea have reported a “traditional Korean” dietary pattern, high consumption of rice and kimchi as the major dietary group for adolescents [27,28,29] as well as adults [30], our analysis showed that the “fish, egg, meat, and fruits & vegetables” pattern explained the highest in the variance of the total food intake. This may be due to the influence of parents, who are more concerned about their children's health. A review of healthy children found that the family strongly influences childhood eating practices [31], however, these issues have not yet been examined among childhood cancer survivors. Therefore, further research is needed, specifically with regard to the effect of mothers on the dietary patterns of Korean pediatric cancer patients.
As studies analyzing dietary patterns in pediatric cancer patients are rare, it is difficult to compare these findings with previously published results. In the present study, the “spicy & fried meat and fish” dietary pattern was shown to be similar to Western style diets characterized by high intakes of energy-dense and fat-rich foods such as red and processed meat, high-fat dairy products, and snacks [32,33]. The Western dietary pattern in both adults and children is known to be positively associated with being overweight. A recent systematic review suggested that dietary patterns high in energy-dense, high-fat, and low-fiber foods predispose young healthy individuals becoming overweight and obese in later life [34]. Regarding children with cancer, several studies reported that long-term survivors of childhood cancer do not adhere to cancer prevention guidelines, as they consume more sweets and fat as well as less fiber than dietary recommendations [12,13,15,16]. Although data are scarce and inconsistant regarding the impact of spicy foods on obesity, a recent population-based epidemiological study in China suggested that spicy food intake is a potential risk factor for obesity, especially for central obesity in men [35]. Sun et al. speculated that spicy foods increase the palatability of meals and consequently may increase carbohydrate intake. Additionally, hot pepper consumption has been shown to increase the desire to eat sweet foods, which reduce the sensation of heat and relieve pain on the tongue. Therefore, consumption of spicy foods may contribute to obesity. Further studies are needed to identify whether or not there is a causal mechanism between spicy food consumption and obesity.
In our study, the “fish, egg, meat, and fruits & vegetables” dietary pattern was associated with lower overweight risk at the end of the study, in agreement with previous studies [36]. The “fish, egg, meat, and fruits & vegetables” dietary pattern, which is characterized by high intakes of fruits, vegetables, fish, and tofu, is similar to the Mediterranean diet. It is well known that the Mediterranean dietary pattern is inversely associated with risk of obesity [22]. In a study on 117 adult survivors of ALL, Tonorezos et al. [37] noted that the Mediterranean dietary pattern was associated with low body mass index, low visceral fat, low subcutaneous fat, and a small waist size.
In the present study, the proportion of patients whose PIBW increased was 59.4%, and the proportion of patients who were overweight increased from 26.5% at baseline to 40.6% at the end of the study. Although the percentage (26.5%) of patients that were overweight at baseline was relatively low compared to other studies reporting percentages of 38.2% [38] and 40% [39], the percentage (40.6%) of overweight patients at the end of the study was similar to those in other studies. It should be noted, however, that the percentage of overweight patients can vary according to the study group due to varying definitions of obesity and the population used. For example, in 1,291 Korean adolescents from the data of the 2005 KNHANES, 13.5% and 8.2% of 10 to 19-year-olds were overweight and obese, respectively [27], and 17.4% of 12 to 19-year-olds in the 2004 US National Health and Nutrition Examination Survey were obese [40]. Moreover, most previous studies have reported that significant weight gain often occurs in children with cancer between diagnosis and the end of treatment [13,39,41,42]. We also observed that among those patients who were overweight at baseline, 62% (n = 10) remained overweight by the end of the study. To prevent obesity, early lifestyle interventions targeting weight control at the point of diagnosis are needed for pediatric cancer patients.
In recent years, there has been growing evidence that dietary factors after cancer diagnosis are related to mortality, especially for adult colorectal, breast, and prostate cancer patients [43,44,45,46]. However, the influence of dietary intake on mortality in survivors of childhood cancer is largely unknown. Adult colon cancer survivors in the highest quintile of a Western dietary pattern score after diagnosis showed a 2.3-fold increase in risk of all-cause mortality, as compared to patients in the lowest quintile (HR = 2.32, 95% CI = 1.36-3.96, P for trend < 0.001) [43]. Similarly, Western dietary patterns have been reported to be positively associated with all-cause mortality in adult prostate cancer patients [45]. In this regard, the positive association between the “spicy & fried meat and fish” dietary pattern and mortality in our study is consistent with previous studies. Conversely, we observed no association between the “fish, egg, meat, and fruits & vegetables” dietary pattern and all-cause mortality, unlike previous study results. In a meta-analysis of 16 prospective cohort studies on the non-cancer population, higher consumption of fruits and vegetables, similar to the “fish, egg, meat, and fruits & vegetables” dietary pattern in our study, was significantly associated with lower risk of all-cause mortality [47]. A separate meta-analysis of 117 cohort studies enrolling adult cancer survivors confirmed these findings by showing that higher intake of vegetables and fish was inversely associated with overall mortality [48]. Taken together, our findings could be used to guide dietary interventions aimed at improving long-term health outcomes among childhood cancer survivors.
This study is significant in that it is the first to examine the association between dietary factors of obesity and all-cause mortality through a dietary pattern analysis of pediatric cancer patients. However, there are several limitations. First, dietary patterns identified with food intake in the hospital may not accurately represent children's usual dietary intakes. Second, we analyzed dietary patterns by the food choice pattern, not actual food intake. Therefore, the results need to be confirmed using adequately quantified dietary intakes. Third, we did not analyze relative risk of mortality according to the interaction between dietary pattern and obesity. Finally, our sample size may not be sufficient to assess the association of dietary pattern with obesity and mortality. However, this study is the first cohort study to reveal associations between specific dietary patterns and overweight risk and all-cause mortality in pediatric cancer patients. Our results convey important messages for the clinical care of pediatric cancer patients in relation to obesity prevention.
In summary, the “spicy & fried meat and fish” dietary pattern was positively associated with overweight risk and all-cause mortality, whereas the “fish, egg, meat, and fruits & vegetables” dietary pattern was associated with lower overweight risk in pediatric cancer patients. Additional comprehensive studies are needed to fully elucidate the associations between dietary pattern as well as overweight risk and premature death in children with cancer.
ACKNOWLEDGMENTS
We would like to extend our sincerest gratitude to the nutritionists working at the Catholic University of Korea, Seoul St. Mary's Hospital.
Footnotes
This work was carried out with the support of ‘Research Program for Agricultural Science and Technology Development’, National Academy of Agricultural Science, Rural Development Administration (Project No. PJ011637022017).
CONFLICT OF INTEREST: The authors declare no potential conflicts of interests.
References
- 1.Jung KW, Won YJ, Kong HJ, Oh CM, Cho H, Lee DH, Lee KH. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2012. Cancer Res Treat. 2015;47:127–141. doi: 10.4143/crt.2015.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Haddy TB, Mosher RB, Reaman GH. Late effects in long-term survivors after treatment for childhood acute leukemia. Clin Pediatr (Phila) 2009;48:601–608. doi: 10.1177/0009922809332680. [DOI] [PubMed] [Google Scholar]
- 3.Diller L, Chow EJ, Gurney JG, Hudson MM, Kadin-Lottick NS, Kawashima TI, Leisenring WM, Meacham LR, Mertens AC, Mulrooney DA, Oeffinger KC, Packer RJ, Robison LL, Sklar CA. Chronic disease in the Childhood Cancer Survivor Study cohort: a review of published findings. J Clin Oncol. 2009;27:2339–2355. doi: 10.1200/JCO.2008.21.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Oeffinger KC, Mertens AC, Sklar CA, Kawashima T, Hudson MM, Meadows AT, Friedman DL, Marina N, Hobbie W, Kadan-Lottick NS, Schwartz CL, Leisenring W, Robison LL Childhood Cancer Survivor Study. Chronic health conditions in adult survivors of childhood cancer. N Engl J Med. 2006;355:1572–1582. doi: 10.1056/NEJMsa060185. [DOI] [PubMed] [Google Scholar]
- 5.Essig S, Li Q, Chen Y, Hitzler J, Leisenring W, Greenberg M, Sklar C, Hudson MM, Armstrong GT, Krull KR, Neglia JP, Oeffinger KC, Robison LL, Kuehni CE, Yasui Y, Nathan PC. Risk of late effects of treatment in children newly diagnosed with standard-risk acute lymphoblastic leukaemia: a report from the Childhood Cancer Survivor Study cohort. Lancet Oncol. 2014;15:841–851. doi: 10.1016/S1470-2045(14)70265-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhang FF, Kelly MJ, Saltzman E, Must A, Roberts SB, Parsons SK. Obesity in pediatric ALL survivors: a meta-analysis. Pediatrics. 2014;133:e704–e715. doi: 10.1542/peds.2013-3332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lange BJ, Gerbing RB, Feusner J, Skolnik J, Sacks N, Smith FO, Alonzo TA. Mortality in overweight and underweight children with acute myeloid leukemia. JAMA. 2005;293:203–211. doi: 10.1001/jama.293.2.203. [DOI] [PubMed] [Google Scholar]
- 8.Rogers PC, Meacham LR, Oeffinger KC, Henry DW, Lange BJ. Obesity in pediatric oncology. Pediatr Blood Cancer. 2005;45:881–891. doi: 10.1002/pbc.20451. [DOI] [PubMed] [Google Scholar]
- 9.Gelelete CB, Pereira SH, Azevedo AM, Thiago LS, Mundim M, Land MG, Costa ES. Overweight as a prognostic factor in children with acute lymphoblastic leukemia. Obesity (Silver Spring) 2011;19:1908–1911. doi: 10.1038/oby.2011.195. [DOI] [PubMed] [Google Scholar]
- 10.Kanellopoulos A, Hamre HM, Dahl AA, Fosså SD, Ruud E. Factors associated with poor quality of life in survivors of childhood acute lymphoblastic leukemia and lymphoma. Pediatr Blood Cancer. 2013;60:849–855. doi: 10.1002/pbc.24375. [DOI] [PubMed] [Google Scholar]
- 11.Iughetti L, Bruzzi P, Predieri B, Paolucci P. Obesity in patients with acute lymphoblastic leukemia in childhood. Ital J Pediatr. 2012;38:4. doi: 10.1186/1824-7288-38-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Robien K, Ness KK, Klesges LM, Baker KS, Gurney JG. Poor adherence to dietary guidelines among adult survivors of childhood acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 2008;30:815–822. doi: 10.1097/MPH.0b013e31817e4ad9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fuemmeler BF, Pendzich MK, Clark K, Lovelady C, Rosoff P, Blatt J, Demark-Wahnefried W. Diet, physical activity, and body composition changes during the first year of treatment for childhood acute leukemia and lymphoma. J Pediatr Hematol Oncol. 2013;35:437–443. doi: 10.1097/MPH.0b013e318279cd3e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang FF, Saltzman E, Kelly MJ, Liu S, Must A, Parsons SK, Roberts SB. Comparison of childhood cancer survivors' nutritional intake with US dietary guidelines. Pediatr Blood Cancer. 2015;62:1461–1467. doi: 10.1002/pbc.25521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tylavsky FA, Smith K, Surprise H, Garland S, Yan X, McCammon E, Hudson MM, Pui CH, Kaste SC. Nutritional intake of long-term survivors of childhood acute lymphoblastic leukemia: evidence for bone health interventional opportunities. Pediatr Blood Cancer. 2010;55:1362–1369. doi: 10.1002/pbc.22737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Badr H, Paxton RJ, Ater JL, Urbauer D, Demark-Wahnefried W. Health behaviors and weight status of childhood cancer survivors and their parents: similarities and opportunities for joint interventions. J Am Diet Assoc. 2011;111:1917–1923. doi: 10.1016/j.jada.2011.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hu FB. Dietary pattern analysis: a new direction in nutritional epidemiology. Curr Opin Lipidol. 2002;13:3–9. doi: 10.1097/00041433-200202000-00002. [DOI] [PubMed] [Google Scholar]
- 18.Rogers PC, Melnick SJ, Ladas EJ, Halton J, Baillargeon J, Sacks N Children's Oncology Group (COG) Nutrition Committee. Children's Oncology Group (COG) Nutrition Committee. Pediatr Blood Cancer. 2008;50:447–450. doi: 10.1002/pbc.21414. [DOI] [PubMed] [Google Scholar]
- 19.Shin S, Hong K, Kang SW, Joung H. A milk and cereal dietary pattern is associated with a reduced likelihood of having a low bone mineral density of the lumbar spine in Korean adolescents. Nutr Res. 2013;33:59–66. doi: 10.1016/j.nutres.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 20.Shams-White M, Kelly MJ, Gilhooly C, Liu S, Must A, Parsons SK, Saltzman E, Zhang FF. Food craving and obesity in survivors of pediatric ALL and lymphoma. Appetite. 2016;96:1–6. doi: 10.1016/j.appet.2015.08.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Okkels SL, Bredie WL, Klausen TW, Beck AM. An investigation into between-meal food desires among hospitalised haematological cancer patients. Clin Nutr. 2016;35:440–445. doi: 10.1016/j.clnu.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 22.Kim JO, Mueller CW. Factor Analysis: Statistical Methods and Practical Issues. Newbury Park (CA): Sage; 1978. [Google Scholar]
- 23.McCann SE, Marshall JR, Brasure JR, Graham S, Freudenheim JL. Analysis of patterns of food intake in nutritional epidemiology: food classification in principal components analysis and the subsequent impact on estimates for endometrial cancer. Public Health Nutr. 2001;4:989–997. doi: 10.1079/phn2001168. [DOI] [PubMed] [Google Scholar]
- 24.Nurliyana AR, Mohd Nasir MT, Zalilah MS, Rohani A. Dietary patterns and cognitive ability among 12- to 13 year-old adolescents in Selangor, Malaysia. Public Health Nutr. 2015;18:303–312. doi: 10.1017/S1368980014000068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McNaughton SA, Ball K, Mishra GD, Crawford DA. Dietary patterns of adolescents and risk of obesity and hypertension. J Nutr. 2008;138:364–370. doi: 10.1093/jn/138.2.364. [DOI] [PubMed] [Google Scholar]
- 26.Noh HY, Song YJ, Lee JE, Joung H, Park MK, Li SJ, Paik HY. Dietary patterns are associated with physical growth among school girls aged 9-11 years. Nutr Res Pract. 2011;5:569–577. doi: 10.4162/nrp.2011.5.6.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Song Y, Park MJ, Paik HY, Joung H. Secular trends in dietary patterns and obesity-related risk factors in Korean adolescents aged 10-19 years. Int J Obes (Lond) 2010;34:48–56. doi: 10.1038/ijo.2009.203. [DOI] [PubMed] [Google Scholar]
- 28.Kim JA, Kim SM, Lee JS, Oh HJ, Han JH, Song Y, Joung H, Park HS. Dietary patterns and the metabolic syndrome in Korean adolescents: 2001 Korean National Health and Nutrition Survey. Diabetes Care. 2007;30:1904–1905. doi: 10.2337/dc06-2591. [DOI] [PubMed] [Google Scholar]
- 29.Joung H, Hong S, Song Y, Ahn BC, Park MJ. Dietary patterns and metabolic syndrome risk factors among adolescents. Korean J Pediatr. 2012;55:128–135. doi: 10.3345/kjp.2012.55.4.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Song Y, Joung H. A traditional Korean dietary pattern and metabolic syndrome abnormalities. Nutr Metab Cardiovasc Dis. 2012;22:456–462. doi: 10.1016/j.numecd.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 31.Patrick H, Nicklas TA. A review of family and social determinants of children's eating patterns and diet quality. J Am Coll Nutr. 2005;24:83–92. doi: 10.1080/07315724.2005.10719448. [DOI] [PubMed] [Google Scholar]
- 32.Pérez-Escamilla R, Obbagy JE, Altman JM, Essery EV, McGrane MM, Wong YP, Spahn JM, Williams CL. Dietary energy density and body weight in adults and children: a systematic review. J Acad Nutr Diet. 2012;112:671–684. doi: 10.1016/j.jand.2012.01.020. [DOI] [PubMed] [Google Scholar]
- 33.Naja F, Hwalla N, Itani L, Karam S, Sibai AM, Nasreddine L. A western dietary pattern is associated with overweight and obesity in a national sample of Lebanese adolescents (13-19 years): a cross-sectional study. Br J Nutr. 2015;114:1909–1919. doi: 10.1017/S0007114515003657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ambrosini GL. Childhood dietary patterns and later obesity: a review of the evidence. Proc Nutr Soc. 2014;73:137–146. doi: 10.1017/S0029665113003765. [DOI] [PubMed] [Google Scholar]
- 35.Sun D, Lv J, Chen W, Li S, Guo Y, Bian Z, Yu C, Zhou H, Tan Y, Chen J, Chen Z, Li L China Kadoorie Biobank collaborative group. Spicy food consumption is associated with adiposity measures among half a million Chinese people: the China Kadoorie Biobank study. BMC Public Health. 2014;14:1293. doi: 10.1186/1471-2458-14-1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lee J, Jeon JY, Meyerhardt JA. Diet and lifestyle in survivors of colorectal cancer. Hematol Oncol Clin North Am. 2015;29:1–27. doi: 10.1016/j.hoc.2014.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tonorezos ES, Robien K, Eshelman-Kent D, Moskowitz CS, Church TS, Ross R, Oeffinger KC. Contribution of diet and physical activity to metabolic parameters among survivors of childhood leukemia. Cancer Causes Control. 2013;24:313–321. doi: 10.1007/s10552-012-0116-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chow EJ, Pihoker C, Hunt K, Wilkinson K, Friedman DL. Obesity and hypertension among children after treatment for acute lymphoblastic leukemia. Cancer. 2007;110:2313–2320. doi: 10.1002/cncr.23050. [DOI] [PubMed] [Google Scholar]
- 39.Collins L, Zarzabal LA, Nayiager T, Pollock BH, Barr RD. Growth in children with acute lymphoblastic leukemia during treatment. J Pediatr Hematol Oncol. 2010;32:e304–e307. doi: 10.1097/MPH.0b013e3181ece2bb. [DOI] [PubMed] [Google Scholar]
- 40.Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999-2004. JAMA. 2006;295:1549–1555. doi: 10.1001/jama.295.13.1549. [DOI] [PubMed] [Google Scholar]
- 41.Withycombe JS, Smith LM, Meza JL, Merkle C, Faulkner MS, Ritter L, Seibel NL, Moore K. Weight change during childhood acute lymphoblastic leukemia induction therapy predicts obesity: a report from the Children's Oncology Group. Pediatr Blood Cancer. 2015;62:434–439. doi: 10.1002/pbc.25316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Withycombe JS, Post-White JE, Meza JL, Hawks RG, Smith LM, Sacks N, Seibel NL. Weight patterns in children with higher risk ALL: a report from the Children's Oncology Group (COG) for CCG 1961. Pediatr Blood Cancer. 2009;53:1249–1254. doi: 10.1002/pbc.22237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Meyerhardt JA, Niedzwiecki D, Hollis D, Saltz LB, Hu FB, Mayer RJ, Nelson H, Whittom R, Hantel A, Thomas J, Fuchs CS. Association of dietary patterns with cancer recurrence and survival in patients with stage III colon cancer. JAMA. 2007;298:754–764. doi: 10.1001/jama.298.7.754. [DOI] [PubMed] [Google Scholar]
- 44.Ollberding NJ, Aschebrook-Kilfoy B, Caces DB, Smith SM, Weisenburger DD, Chiu BC. Dietary intake of fruits and vegetables and overall survival in non-Hodgkin lymphoma. Leuk Lymphoma. 2013;54:2613–2619. doi: 10.3109/10428194.2013.784968. [DOI] [PubMed] [Google Scholar]
- 45.Yang M, Kenfield SA, Van Blarigan EL, Batista JL, Sesso HD, Ma J, Stampfer MJ, Chavarro JE. Dietary patterns after prostate cancer diagnosis in relation to disease-specific and total mortality. Cancer Prev Res (Phila) 2015;8:545–551. doi: 10.1158/1940-6207.CAPR-14-0442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vrieling A, Buck K, Seibold P, Heinz J, Obi N, Flesch-Janys D, Chang-Claude J. Dietary patterns and survival in German postmenopausal breast cancer survivors. Br J Cancer. 2013;108:188–192. doi: 10.1038/bjc.2012.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang X, Ouyang Y, Liu J, Zhu M, Zhao G, Bao W, Hu FB. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: systematic review and dose-response meta-analysis of prospective cohort studies. BMJ. 2014;349:g4490. doi: 10.1136/bmj.g4490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schwedhelm C, Boeing H, Hoffmann G, Aleksandrova K, Schwingshackl L. Effect of diet on mortality and cancer recurrence among cancer survivors: a systematic review and meta-analysis of cohort studies. Nutr Rev. 2016;74:737–748. doi: 10.1093/nutrit/nuw045. [DOI] [PMC free article] [PubMed] [Google Scholar]