Abstract
A multitude of proteins are aberrantly expressed in cancer cells, including the oncogenic serine-threonine kinase CK2. In a previous report, we found increases in CK2 transcript expression that could explain the increased CK2 protein levels found in tumors from lung and bronchus, prostate, breast, colon and rectum, ovarian and pancreatic cancers. We also found that, contrary to the current notions about CK2, some CK2 transcripts were downregulated in several cancers. Here, we investigate all other cancers using Oncomine to determine whether they also display significant CK2 transcript dysregulation. As anticipated from our previous analysis, we found cancers with all CK2 transcripts upregulated (e.g. cervical), and cancers where there was a combination of upregulation and/or downregulation of the CK2 transcripts (e.g. sarcoma). Unexpectedly, we found some cancers with significant downregulation of all CK2 transcripts (e.g. testicular cancer). We also found that, in some cases, CK2 transcript levels were already dysregulated in benign lesions (e.g. Barrett’s esophagus). We also found that CK2 transcript upregulation correlated with lower patient survival in most cases where data was significant. However, there were two cancer types, glioblastoma and renal cell carcinoma, where CK2 transcript upregulation correlated with higher survival. Overall, these data show that the expression levels of CK2 genes is highly variable in cancers and can lead to different patient outcomes.
Introduction
In humans, there are two CK2 kinase genes, CSNK2A1 and CSNK2A2, that code for highly-conserved serine/threonine kinase proteins, CK2α and CK2α’, respectively. CK2α and CK2α’ differ in their C-terminal sequence [1–4]. They also differ in their expression pattern and the phenotype of knockout in mice. Thus, CK2α has higher levels and more widespread expression in mouse tissues than CK2α’ (mostly brain and testis) [5]. CK2α deficient mice die at mid-gestation while CK2α’ deficient mice are viable albeit males are sterile, suggesting that they have different functions that cannot be compensated by the other protein [5, 6]. CK2 kinases can function as monomeric kinases, and also within a tetrameric complex composed of two CK2 kinase proteins (CK2α and/or CK2α’) and two regulatory proteins with no enzymatic activity (CK2β, coded by CSNK2B). Within this tetrameric complex, CK2β alters CK2 kinase substrate specificity [7]. Additionally, an intronless CK2α pseudogene (CK2aP, coded by CSNK2A1P/CSNK2A3) [8] codes for a predicted non-coding RNA that is relevant in human cancer [9].
CK2 has been implicated in cancer in humans and mice [10–14]. CK2 proteins are upregulated in the human tumors tested so far, suggesting a role in cancer progression (Reviewed in [15, 16]). Indeed, overexpression of CK2α in mammary gland and the lymphoid compartment leads to tumors in mice [17–20]. In cancer, CK2 is proposed to regulate essential cellular processes such as cell growth [21], cell proliferation [22, 23],cell survival [24, 25], cell morphology [26, 27], cell transformation [12, 13] and angiogenesis [28]. Importantly, CK2 protein upregulation and/or nuclear localization correlate with cancer clinicopathology and prognosis in some cancers (e.g. gastric cancer [29, 30], and head and neck cancer [31–33]). These data suggest that CK2 is a target for cancer therapy and hence, several CK2 inhibitors are being tested in clinical trials (reviewed in [16, 34, 35]).
The importance of CK2 transcripts in cancer is also being investigated. The original view in the literature is that CK2 is predominantly regulated post-transcriptionally, however, recent studies strongly suggest that regulation at the transcriptional level is also important in some cancers ([9], and references within). Unpredictably, some cancers show underexpression of CK2 transcripts (e.g. CK2α’ in breast, ovarian, and pancreatic cancer)[9]. Importantly, recent studies show that CK2 transcripts could have a diagnostic value (e.g. CK2α and CK2α’ in renal cell carcinoma [36]; CK2β in invasive ductal and lobular breast carcinoma [9]). Furthermore, CK2 transcript levels could have a prognostic value in cancers (e.g. CK2α in squamous cell carcinoma of the lung [9, 37]). For the most part, high levels of CK2 transcript correlate with lower overall survival (e.g. breast and ovarian cancer [9], glioblastoma, kidney and liver cancer [38–40]). However, in lung adenocarcinoma, high levels of CK2α’ and CK2αP correlate with higher survival rates [9]. Overall, these data indicate the need to determine the extent to which CK2 genes could be significantly up- or down-regulated in other cancers not studied so far, and raise the question of whether in these other cancers CK2 gene expression levels correlated with overall patient survival. Therefore, using Oncomine, we analyzed the expression levels of CK2 transcripts in bladder, central nervous system (CNS), cervical, esophageal, gastric, head and neck, kidney, blood (leukemia, non-Hodgkin lymphoma, monoclonal gammopathies), liver, mesothelioma, parathyroid, sarcoma, skin, and testicular cancers. We also analyzed the correlation between CK2 gene expression and overall patient survival to determine whether it has prognostic value, in cancers where data was available.
Methods
Information on cancer types, prevalence and treatment
To retrieve this information, we used the NCI (http://www.cancer.gov), ACS (http://www.cancer.org) and CDC (http://www.cdc.gov) web sites.
Oncomine analysis
The transcript level of CK2 genes and pseudogene (CSNK2A1, CSNK2A2, CSNK2B, and CSNK2A1P/CSNK2A3) was analyzed using the Oncomine database (www.oncomine.org, July 2017, Compendia Bioscience, Ann Arbor, MI) [41]. This database contains different datasets, each containing the data from a single publication. We used datasets that compared cancer vs. normal patient specimens for the different cancer types. We used the default view, where all the expression data included is obtained before cancer treatment. Oncomine uses t-test statistics to compare the means of gene expression to determine whether a gene is significantly over or underexpressed in tumors compared to normal tissue. Each Oncomine search provides the p-value (probability that there is a true difference in gene expression), fold change (the difference between the mean values of the classes that are being compared), and rank (where genes are ranked by their P-value; the one’s at the top % have more significant P-values that other genes) for each gene. The data we obtained were represented in tables where #Samples represents the total number of samples in the dataset (controls and tumor samples), and Reference is the original publication of the data. To reduce false discovery rate, p < 0.01, fold change > +/- 1.5 were selected as thresholds. In the tables, we have included all the data that matched these thresholds. We have also included a few data that were significant but were below the 1.5-fold change threshold (up to approx. 1.3), as they indicate low variance. If a particular CK2 gene was not tested in the original publication, we have written it in the text and also noted in the final summary table as “-“. CK2 genes that were tested but found non-significant were not mentioned in the text, but noted as “n.s.” in the final summary table.
Kaplan-Meier analysis
The prognostic value of the expression of CK2 transcripts in gastric cancer was analyzed using the Kaplan-Meier Plotter (http://kmplot.com/analysis), a database that integrates gene expression and clinical outcomes [42]. At present, Kaplan-Meier Plotter contains information on survival outcomes for 1,065 gastric cancer patients in relation to the expression levels of the 22,277 genes in their database [43]. The best specific probes (JetSet probes) were selected for each CK2 transcript. Patients were split into two groups, high versus low expression levels of each CK2 gene (based on the median expression), and the overall survival rates of these two patient groups were compared. To reduce false discovery rate, we selected p < 0.01 as a threshold. Hazard ratio with 95% confidence intervals and log rank p value were also calculated.
The prognostic value of expression of the CK2 transcripts in other cancers was analyzed using data from the web sites of the University of California Santa Cruz (UCSC) Xena (https://genome-cancer.soe.ucsc.edu/proj/site/xena/heatmap/) and Cell Index (CellX) (http://54.149.52.246/cgi-bin/RPPA/cellx.cgi). These data repositories contain overall survival (OS) and recurrence free survival (RFS) information based upon data generated by the TCGA Research Network (http://cancergenome.nih.gov/). The data from these two databases were combined to make one complete data set with as many patient data as possible. We found information for the following cancers: cervical cancer, acute myeloid leukemia, bladder cancer, kidney papillary cell carcinoma, esophageal cancer, glioblastoma, head and neck cancer, kidney chromophobe, kidney clear cell carcinoma, liver cancer, large B-cell lymphoma, melanoma, mesothelioma, sarcoma, stomach cancer, and testicular cancer.
Kaplan-Meier survival curves and statistical analysis was performed for the CK2 transcripts for each cancer type by the Department of Medicine Biostatistics consultants, using SAS 9.3 (SAS institute, Cary, NC, USA). For each transcript and tumor type, the cancer patients were stratified into two groups: high and low transcript expression levels based on whether the expression of the gene was above or below the median expression of the gene. The Log-Rank test was used to compare the overall survival curves of the over-expression and under-expression patient groups, and a p-value obtained. For our analysis, alpha equal to or below 0.05 was chosen as significant. We used R Studio (R Studio, Boston, MA, USA) to represent the Kaplan Meier survival curves for the cancers with significant p-values in the Log-Rank test. We also included in the figures the hazard ratio (HR) and the confidence interval (CI) from the Cox proportional hazards model. The cancers for which none of the CK2 transcripts was significantly associated with patient overall survival were Leukemia (151 patients), Bladder (407 patients), Kidney (320 patients), Esophageal (184 patients), Kidney Chromophobe (65 patients), Lymphoma (47 patients), Melanoma (251 patients), Stomach (387 patients) and Testicular (134 patients).
Results and discussion: CK2 transcript expression levels and correlation with overall patient survival by cancer type
Here, the different cancer types studied are organized alphabetically expect for liver cancer, which is placed after all the blood cancers. To simplify the reading, we have chosen to use the terms CK2α, CK2α’, CK2β and CK2αP to refer to the genes CSNK2A1, CSNK2A2, CSNK2B, CSNK2A1P/CSNK2A3, respectively.
Bladder cancer
Transitional cell carcinomas (also known as urothelial carcinomas) make up about 90% of all bladder cancers, and originate in cells of the inner lining of the bladder. Transitional cell carcinomas are clinically subdivided into superficial (non-muscle invasive) and invasive tumors. Less common bladder cancer types include squamous cell carcinoma and adenocarcinoma. There is no standard or routine screening test for bladder cancer, leading to a low rate of early diagnosis. Treatments for bladder cancer include surgery, radiation therapy, chemotherapy, and biological therapy.
CK2 in bladder cancer
Oncomine analysis revealed significant overexpression of all three CK2 transcripts in both superficial and invasive types of bladder transitional cell carcinoma (Table 1). In line with our findings, Zhang et al. found overexpression of CK2α transcript in transitional cell carcinomas (subtype not specified) compared with adjacent normal tissue and also in transitional cell carcinoma cells lines compared with normal urinary epithelial cell lines [44]. There were no data in Oncomine for squamous cell carcinoma and adenocarcinoma, possibly due to their low frequency in the population.
Table 1. Analysis of changes in CK2 gene expression in bladder cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Superficial Bladder Carcinoma | ||||||
| CK2α | 2.26 10−14 | 2.044 | 5% | Sanchez-Carbayo Bladder 2 | 157 | [47] |
| 3.70 10−10 | 1.635 | 1% | Dyrskjot Bladder 3 | 60 | [48] | |
| CK2α’ | 4.17 10−6 | 1.775 | 9% | Dyrskjot Bladder 3 | 60 | [48] |
| 1.24 10−5 | 1.635 | 24% | Sanchez-Carbayo Bladder 2 | 157 | [47] | |
| CK2β | 2.65 10−17 | 2.874 | 3% | Sanchez-Carbayo Bladder 2 | 157 | [47] |
| 7.38 10−8 | 2.010 | 3% | Dyrskjot Bladder 3 | 60 | [48] | |
| Invasive Bladder Carcinoma | ||||||
| CK2α | 9.15 10−8 | 1.696 | 7% | Sanchez-Carbayo Bladder 2 | 157 | [47] |
| 2.33 10−5 | 1.583 | 7% | Dyrskjot Bladder 3 | 60 | [48] | |
| CK2α’ | 0.003 | 1.501 | 25% | Dyrskjot Bladder 3 | 60 | [48] |
| CK2β | 1.48 10−7 | 2.211 | 1% | Dyrskjot Bladder 3 | 60 | [48] |
| 0.001 | 1.420 | 21% | Sanchez-Carbayo Bladder 2 | 157 | [47] | |
Elevated levels of CK2α protein are also found in bladder cancer. Thus, Zhang et al. found increased levels of CK2α protein in bladder carcinomas (subtype not specified) compared with adjacent normal tissue, and in transitional cell carcinoma cells lines compared with normal urinary epithelial cell lines [44]. High staining of CK2α protein is detected in invasive bladder transitional cell carcinoma but not in low grade non-invasive tumors [45]. In addition, CK2α was increased in the lumen of exosomes of metastatic cells versus non-metastatic bladder cancer cells [46]. CK2α protein staining positively correlates with histological grade but not with tumor size, tumor stage or gender [44]. Kaplan–Meier analysis does not reveal an association between high CK2α staining and survival. We further reviewed the role of CK2 proteins in bladder cancer in [16]. All together, these data suggest that CK2α protein, but perhaps not the transcript, could be used as a diagnostic marker in bladder cancer.
Central nervous system (CNS) cancer
Overall (among children and adults), the most common CNS cancer type is glioma (33%), which is subclassified into astrocytomas (20% of total CNS cancer), oligodendrogliomas (2% of total CNS cancer), ependymomas (2% of total CNS cancer), and mixed gliomas. A high-grade (IV) astrocytoma is known as glioblastoma. However, among adults only, the most common CNS cancers is meningioma (33%). Other CNS tumor types include, but are not limited to, medulloblastomas, gangliogliomas, schwannomas, and craniopharyngiomas. While the rate of diagnosis at an early stage is relatively high, with 76.6% of patients diagnosed at the local stage, the 5-year survival for localized CNS cancer is only 36.3%. There are currently no screening tests for CNS cancers. Standard treatments involve watchful waiting, surgery, radiation therapy, chemotherapy, and targeted therapy.
CK2 in CNS cancer
Oncomine analysis revealed overexpression and underexpression of CK2α transcripts in astrocytoma and glioblastoma (Table 2). In agreement with our data, other publications also show under- and over-expression of CK2α transcripts. For example, Zheng et al. show CK2α transcripts mostly overexpressed but also underexpressed in glioblastomas from the TCGA [49]. They attribute increases in CK2α transcripts to gene dosage gains of CSNK2A1. Nitta et al. and Ladha et al. also find increased (mostly) and decreased CK2α transcripts in glioblastomas [38, 50]. Dubois et al. find heterogeneous increases in CK2α transcripts in glial brain tumors (oligodendrogliomas, astrocytomas and glioblastoma), but, in this case, CK2 transcript levels did not correlate with gene amplification, suggesting transcriptional mechanisms at play [51]. This heterogeneity in the levels of CK2α transcripts in these cancers may be resolved by increasing the sample size, to determine whether over- or under- expression correlate with any clinicopathological or demographic variables.
Table 2. Analysis of changes in CK2 gene expression in CNS cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Astrocytoma | ||||||
| CK2α | 0.001 | 1.352 | 8% | Shai Brain | 42 | [53] |
| 0.003 | -1.322 | 9% | Sun Brain | 180 | [58] | |
| CK2β | 3.12 10−4 | 1.518 | 5% | Shai Brain | 42 | [53] |
| Glioblastoma (Astrocytoma Grade IV) | ||||||
| CK2α | 2.02 10−6 | 1.370 | 15% | Sun Brain | 180 | [58] |
| 2.33 10−4 | 1.294 | 11% | Shai Brain | 42 | [53] | |
| 9.21 10−4 | 4.072 | 13% | Bredel Brain 2 | 54 | [59] | |
| 0.01 | -2.382 | 42% | Lee Brain | 101 | [52] | |
| CK2α’ | 2.59 10−4 | -2.132 | 23% | Lee Brain | 101 | [52] |
| 8.13 10−4 | -1.314 | 17% | Bredel Brain 2 | 54 | [59] | |
| CK2β | 1.52 10−7 | 1.440 | 2% | Shai Brain | 42 | [53] |
| 1.19 10−5 | -2.338 | 14% | Lee Brain | 101 | [52] | |
| Anaplastic Oligodendroglioma (grade III) | ||||||
| CK2α’ | 1.15 10−6 | 1.637 | 3% | French Brain | 33 | [60] |
| CK2β | 0.001 | 1.499 | 15% | French Brain | 33 | [60] |
| Anaplastic Oligoastrocytoma (oligodendroglioma + astrocytoma) | ||||||
| CK2α | 0.008 | 1.686 | 12% | French Brain | 33 | [60] |
| CK2α’ | 0.004 | -1.624 | 14% | Bredel Brain 2 | 54 | [59] |
| CK2β | 0.003 | 1.569 | 8% | French Brain | 33 | [60] |
Oncomine analysis showed overexpression of CK2α’ in oligodendrogliomas and astrocytomas, and over-and under-expression of CK2α’ transcripts in glioblastoma. In agreement with our data, Dubois et al. find elevated levels of CK2α’ transcripts in oligodendrogliomas, astrocytomas, and the majority of glioblastomas. They also found CK2α’ gene dosage loss in glioblastoma (1/18 samples) [51].
We found overexpression of CK2β transcripts in CNS cancers as detailed in Table 2, except in glioblastoma where there was conflicting data [52, 53]. We could not find information on demographic or clinicopathological characteristic that could further explain this discrepancy. Published data also show heterogeneous expression of CK2β transcripts. Thus, some studies find CK2β transcripts mostly unchanged [38] while others find CK2β transcripts mostly upregulated in glial brain tumors (oligodendrogliomas, astrocytomas and glioblastoma) [51]. Further studies will help determine to determine whether over- or under- expression correlate with any clinicopathological or demographic variables. Interestingly, the CK2β gene is deleted in a small percent of glioblastomas (7%, in Zheng et al., and 1/18 in Dubois et al.)[49] Dubois, 2016 #532], but gene dosage gains were also found in a small percent of glioblastomas (1/18 samples) [51]. If after further confirmation, if CK2α’ and/or CK2β are found to be downregulated in some subtypes of glioblastoma (grade IV astrocytoma), they could have diagnostic value for these subtypes and also to distinguishing high from low grade astrocytomas that have elevated levels of both transcripts. For other CNS cancer types, Oncomine analysis showed no significant findings for any of the CK2 transcripts. There was no data for CK2αP in astrocytoma.
Regarding CK2 proteins in CNS cancer, Dixit et al. (n = 5) find increased levels of CK2α protein in glioblastomas (5/5 samples), while Nitta et al. find increased (4/7 samples) and decreased (1/7 samples) levels CK2α protein [38, 54]. There was a correlation between increased levels of CK2α protein and transcript levels in glioblastomas [38]. In addition, CK2 kinase activity as also higher in most glial brain tumors (oligodendrogliomas, astrocytomas and glioblastoma) but did not correlate neither with transcript levels nor gene amplification [51]. These data suggest that translational regulatory mechanisms, post-translational modifications or different levels of CK2β protein (not measured in these studies) could play a role in the increased CK2 activity observed.
The increase in CK2 activity found in glial brain tumors does not correlate with tumor grade (II, III or IV), and, in the case of glioblastomas, with tumor subtype [51]. However, when CK2 subcellular localization is analyzed, there is a correlation between increased cytoplasmic staining of CK2α with increasing grades of malignancy (grades II, II and IV (glioblastoma)), suggesting that cytoplasmic CK2α protein level has diagnostic value [50]. In contrast, the same study find astrocytomas of all grades show a decrease in nuclear CK2α protein staining compared to control samples. However, other publications show increased levels of both nuclear and cytoplasmic CK2α in CNS cancer. For example, glioblastoma samples show higher staining of CK2α either in the cytoplasm or in both cytoplasm and nucleus, compared with normal brain tissue [51]; and CK2α protein is elevated in cytoplasm and nucleus in grade II, III and IV gliomas (no subtype information) [55]. As for CK2 transcripts, further studies are needed to determine whether over- or under- expression of CK2α proteins correlate with any clinicopathological or demographic variables.
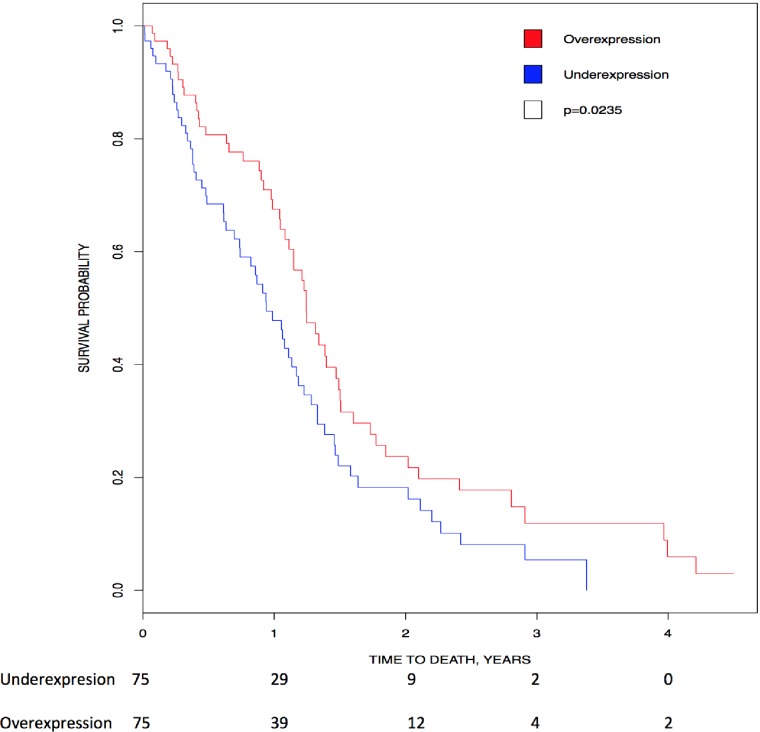
Unexpectedly, Kaplan-Meier analysis showed high expression of CK2α transcripts directly associated with higher overall survival in glioblastoma (p = 0.0235)(Fig 1). In contrast with our analysis, Nitta et al. show high expression of CK2α transcripts correlating with lower survival in mesenchymal glioblastoma, but not in the other types of glioblastomas (classical, neural and proneural) using the TCGA database [38]. In addition, using the Repository of Molecular Brain Neoplasia Data (Rembrandt) they found a trend where high CK2α transcript correlated with poor prognosis [38]. We do not have enough information to explain the difference between our analysis and that of Nitta et al. [38]. As for CK2α protein, there is no difference in patient survival between high staining of CK2α in the cytoplasm or in both cytoplasm and nucleus [51], therefore we may not be able to use CK2α protein upregulation as a prognostic factor in glioblastoma.
Fig 1. Correlation of CK2 gene expression to overall patient survival in glioblastoma.
Patients were stratified into above-median (red line) and below-median (blue line) expression for CK2α. Kaplan Meier analysis show that high levels of CK2α transcript correlated with higher survival (HR = 0.65, 95% CI [0.446, 0.947], p = 0.0235).
Mounting pre-clinical evidence suggests that CK2 inhibitors could be effective in glioblastoma [16]. An additional pre-clinical model show significantly improved survival of mice treated with a combination of TMZ (temozolomide, the standard chemotherapy for glioblastoma) and CK2 inhibitor CX-4945 when compared to TMZ alone (p <0.05) This effect occurs only when drugs are administered simultaneously every 6 days [56]. In addition, inhibition of CK2 with CX-4945 and TBB leads to decreased glioblastoma invasion in glioblastoma cell lines [57].
Cervical cancer
Cervical cancer is the 2nd leading cause of death among women worldwide, and most commonly caused by human papillomavirus (HPV) infection. Most cases of cervical cancer are preventable with the HPV vaccine. Despite well-established screening tests in place, only 46.9% of patients are diagnosed at the localized stage. Standard treatments involve surgery, radiation therapy, and chemotherapy.
CK2 in cervical cancer
Oncomine analysis revealed significant overexpression of all three CK2 genes in cervical cancer (Table 3). Oncomine had no data on CK2αP expression in cervical cancer.
Table 3. Analysis of changes in CK2 gene expression in cervical cancer.
P-values, fold change, rank and datasets are shown.
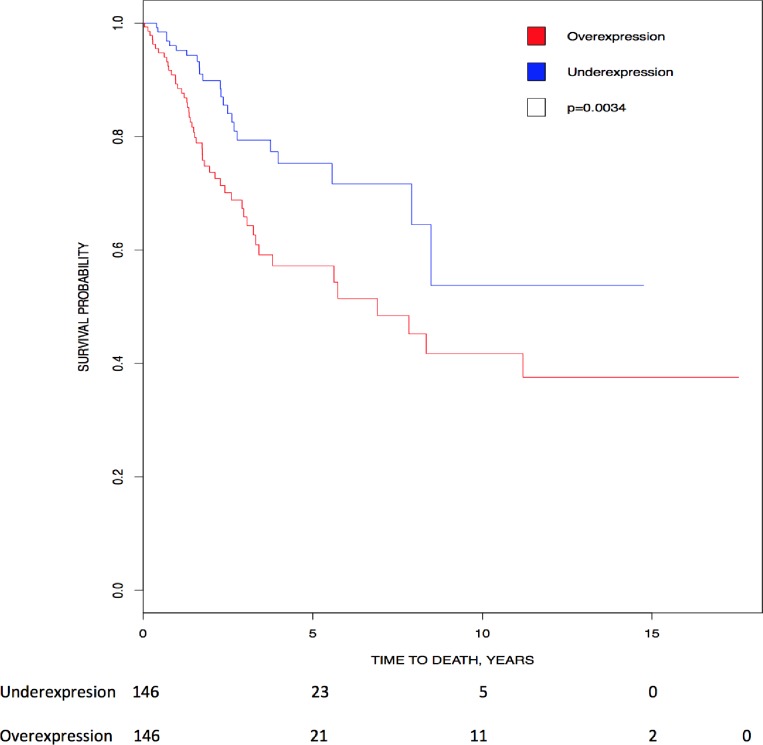
Kaplan-Meier analysis showed that higher expression of CK2αP transcripts directly correlated with lower survival, suggesting its prognostic value in cervical cancer (p = 0.0034)(Fig 2).
Fig 2. Correlation of CK2 gene expression to overall patient survival in cervical cancer.
Patients were stratified into above-median (red line) and below-median (blue line) expression of CK2αP. Kaplan Meier analysis show that high levels of CK2αP transcript correlated with lower survival (HR = 2.075, 95% CI [1.260, 3.418]; p = 0.0034).
Interestingly, CK2 activity is elevated in HPV-immortalized human keratinocytes and cervical and oral carcinoma cell lines compared to parental cell lines and normal cervical and tongue keratinocytes [61]. In HPV-immortalized cell lines, CK2 activity is highest during the G1 phase of the cell cycle [61]. During G1, CK2 seems to phosphorylate and activate the HPV type 18 E7 protein, a transcription factor that induces cellular proliferation by promoting S-phase entry [61, 62]. Based on these data, it is plausible that CK2 stimulates HPV-linked cervical cancer by promoting S-phase entry of the cell cycle. Importantly, CK2 inhibitors are being tested in cervical cancer in clinical trials (reviewed in [16]).
Esophageal cancer
The two most common types of esophageal cancer are adenocarcinoma (most common in USA) and squamous cell carcinoma (most common worldwide). Adenocarcinoma forms in glandular cells close to the stomach, and squamous cell carcinoma forms in flat cells of the upper and middle esophagus. Esophageal cancer is associated with aging, and alcohol and tobacco use (only squamous cell carcinoma), and affects more men than women across all racial and ethnic groups. Importantly, the risk for esophageal adenocarcinoma is increased in Barrett’s esophagus, a condition characterized by replacement of the esophageal tissue by tissue similar to that of intestinal lining that occurs in individuals with long-term gastroesophageal reflux disease. Procedures to detect or diagnose esophageal cancer include physical examination, upper endoscopy, chest x-ray, and a barium swallow test, but only 20.5% of patients are diagnosed at the local stage. Treatment options include surgery, radiation therapy, chemotherapy, chemoradiation therapy, laser therapy, and electrocoagulation.
CK2 in esophageal cancer
Oncomine analysis showed significant downregulation of all CK2 genes in Barrett’s esophagus and esophageal adenocarcinoma (Table 4). In esophageal adenocarcinoma, there was conflicting data for CK2α’ transcripts, where the Kim et al. study showed a downregulation while the Hao et al. study showed an upregulation [64, 65]. In both studies, age ranges are similar, and the patients were from the USA. The Kim et al. study included 90.7% male, average age 61.82, and 49.2% white patients; the Hao et al study included 94.1% male, average age 66.5±11.7, and no ethnicity was described. We do not have enough data to determine if clinicopathological characteristics (only Kim et al. study includes stages I to IV cancer) explain the discrepancy in the results.
Table 4. Analysis of changes in CK2 gene expression in esophageal cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Barrett’s Esophagus | ||||||
| CK2α | 5.27 10−10 | -2.356 | 6% | Kim Esophagus | 118 | [64] |
| CK2α’ | 1.36 10−4 | -1.660 | 23% | Kim Esophagus | 118 | [64] |
| CK2β | 2.53 10−6 | -1.935 | 15% | Kim Esophagus | 118 | [64] |
| 2.65 10−7 | -1.333 | 2% | Wang Esophagus | 52 | [67] | |
| CK2αP | 5.63 10−10 | -1.578 | 6% | Kim Esophagus | 118 | [64] |
| Esophageal Adenocarcinoma | ||||||
| CK2α | 2.86 10−9 | -1.805 | 12% | Kim Esophagus | 118 | [64] |
| 0.012 | -1.334 | 15% | Kimchi Esophagus | 24 | [68] | |
| CK2α’ | 1.37 10−6 | -1.591 | 19% | Kim Esophagus | 118 | [64] |
| 3.35 10−4 | 2.524 | 7% | Hao Esophagus | 48 | [65] | |
| CK2β | 4.9 10−9 | -2.042 | 13% | Kim Esophagus | 118 | [64] |
| CK2αP | 1.82 10−7 | -1.315 | 17% | Kim Esophagus | 118 | [64] |
In addition, Chen et al. find both overexpression (6/8) and underexpression (2/8) of CK2β transcripts also in esophageal carcinoma, compared with adjacent normal mucosal tissue [66]. They also find elevated CK2β staining in 86% of tumors levels while the rest have none, weak or moderate staining, and CK2β protein levels are elevated in ¾ samples. Furthermore, they find that CK2β transcript expression level correlated with cancer stage (I, II and III) therefore, CK2β transcripts could have diagnostic value [66].
Gastric cancer
The most common type of gastric cancer is adenocarcinoma, accounting for 95% of all gastric cancers, and develops from cells forming the mucosa (innermost lining) of the stomach. Other types can include carcinoid tumor and gastrointestinal stromal tumor. Gastric cancer is more common in men than women, and whites have the lowest rates of gastric cancer compared to all other racial/ethnic groups in the USA. A number of factors increase gastric cancer risk including Helicobacter pylori (H. pylori) infection of the stomach, smoking, family history of gastric cancer, high-salt diet or smoked foods, and low intake of fruits and vegetables. There is currently no standard or routine screening test for gastric cancer. Treatments include surgery, chemotherapy, radiation therapy, and chemoradiation.
CK2 in gastric cancer
Oncomine analysis revealed overexpression of CK2α transcripts in all types of gastric cancer found in Oncomine; some types also showed overexpression of CK2αP and CK2β transcripts (Table 5). CK2α’ transcripts showed a downregulation in diffuse gastric adenocarcinoma. However, there were conflicting findings for CK2α’ in gastric intestinal type adenocarcinoma, with the Cho et al. study showing decreased expression and the D’Errico et al. study showing overexpression [69, 70]. The studies have similar demographics except for country of origin: South Korea, 63.2% female, mean age 62 (32–83 years) versus Italy, 60% female, mean age 74 (61–89 years) respectively. We do not have enough data to determine if clinicopathological characteristics (only D’Errico et al. study included stage III cancer) explain the discrepancy in the results.
Table 5. Analysis of changes in CK2 gene expression in gastric cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Gastric Intestinal Type Adenocarcinoma | ||||||
| CK2α | 1.98 10−10 | 1.611 | 5% | Chen Gastric | 132 | [72] |
| 7.64 10−8 | 1.661 | 8% | DErrico Gastric | 69 | [69] | |
| CK2α’ | 5.68 10−5 | 1.473 | 20% | DErrico Gastric | 69 | [69] |
| 4.75 10−4 | -1.454 | 5% | Cho Gastric | 90 | [70] | |
| CK2β | 4.56 10−7 | 1.761 | 11% | DErrico Gastric | 69 | [69] |
| CK2αP | 1.34 10−5 | 1.346 | 16% | Chen Gastric | 132 | [72] |
| Diffuse Gastric Adenocarcinoma | ||||||
| CK2α | 6.9 10−5 | 1.540 | 5% | Chen Gastric | 132 | [72] |
| CK2α’ | 2.95 10−4 | -1.549 | 6% | Cho Gastric | 90 | [70] |
| Gastric Mixed Adenocarcinoma | ||||||
| CK2α | 0.011 | 2.136 | 19% | Chen Gastric | 132 | [72] |
| 0.012 | 1.449 | 17% | Cho Gastric | 90 | [70] | |
CK2α and CK2β protein levels are elevated in gastric carcinoma (2/2 samples) compared to non-tumor tissue [30]. As for subcellular localization, CK2α protein staining (nuclear and cytoplasmic) is elevated in gastric carcinoma and dysplastic lesions compared to normal mucosa [29], and CK2β staining is also elevated in gastric carcinoma (nuclear and cytoplasmic) [30].
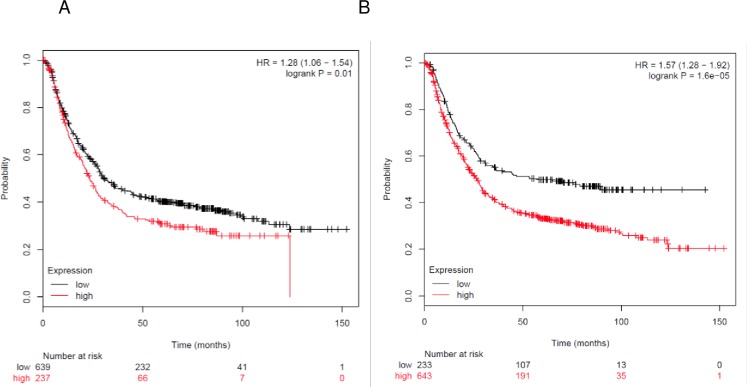
Kaplan-Meier Plotter analysis showed that higher expression of CK2α (p = 0.010) and CK2β (p = 1.6 10−5) transcripts directly correlated with lower survival in gastric cancer patients (Fig 3). In addition, published results show that overexpression of CK2α protein correlates with poor survival [29], and elevated levels of nuclear CK2β correlated with poor survival in gastric cancer [30]. Therefore, both CK2α and CK2β transcripts and proteins could be prognostic markers for gastric carcinoma.
Fig 3. Correlation of CK2 gene expression to overall patient survival in gastric cancer.
Patients were stratified into above-median (red line) and below-median (black line) expression of CK2. (A) High levels of CK2α transcript correlated with lower survival (HR = 1.28, 95% CI [1.06, 1.54]; p = 0.01). (B) High levels of CK2β transcript correlated with lower survival (HR = 1.57, 95% CI [1.28, 1.92]; p = 1.6 10−5).
CK2α protein was found also to be an independent diagnostic indicator for gastric carcinoma, as nuclear levels of CK2α were associated with higher tumor stage, presence of lymph node metastasis, presence of venous invasion and tumor invasion [29]. Additionally, elevated levels of nuclear CK2β correlated with depth of invasion [30]. These data show that increased CK2α and CK2β protein levels can be correlated with the invasive potential of gastric cancer cells. Indeed, CK2 inhibition led to a decreased migration and invasion of gastric epithelial cells and epithelial-to-mesenchymal transition phenotype, a hallmark for metastasis [29, 71]. At least two mechanisms are described to contribute to CK2α’s association with gastric carcinoma; through phosphorylation and activation of the protein “deleted in breast cancer 1” (DBC1) [29], and through facilitation of H. pylori’s effects on migration and invasion [71]. Overall, both CK2α and CK2β transcripts and proteins have prognostic value in gastric cancer, and CK2α and CK2β proteins have diagnostic value.
Head and neck cancer
Head and neck cancer encompasses cancers in the oral cavity, pharynx, larynx, paranasal sinuses and nasal cavity and salivary glands; 90% arise from squamous cells (e.g. tonsillar carcinoma, tongue carcinoma, floor of the mouth carcinoma, and oropharyngeal carcinoma). The two highest risk factors for head and neck cancer are alcohol and tobacco use. Other risk factors include HPV infection (especially HPV-16), Epstein-Barr virus infection (mainly nasopharyngeal cancer), chewing of paan, drinking yerba mate, eating salty foods, poor oral hygiene, exposure to radiation, and industrial exposures. Treatment options include surgery, chemotherapy, targeted therapy, and/or radiation therapy.
CK2 in head and neck cancer
Oncomine analysis showed that CK2α, CK2α’ and CK2β transcripts were significantly overexpressed in head and neck cancers (Table 6). In addition, Bian et al. using the TGCA database find genetic and transcript expression alterations in CK2α (21%), CK2α’ (11%) and CK2β (8%) in head and neck squamous cell carcinoma cases (HNSCC). Most are increases in CK2 transcript expression. However, in a small percentage of tumor samples, CK2 transcript levels decreased [73]. Intriguingly, the majority of these tumor samples have alterations (up or downregulation) in the levels of transcripts of only one of the CK2 genes. There was no data for CK2αP in any of the cancer types in Table 6. There were no studies in Oncomine for other types of head and neck cancer.
Table 6. Analysis of changes in CK2 gene expression in head and neck cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Floor of the Mouth Carcinoma | ||||||
| CK2α | 1.69 10−4 | 2.313 | 7% | Pyeon Multi-cancer | 84 | [63] |
| CK2α’ | 0.011 | 1.99 | 21% | Pyeon Multi-cancer | 84 | [63] |
| CK2β | 1.44 10−4 | 1.923 | 6% | Pyeon Multi-cancer | 84 | [63] |
| Oral Cavity Squamous Cell Carcinoma | ||||||
| CKα | 2.35 10−6 | 1.301 | 11% | Peng Head-Neck | 57 | [76] |
| Nasopharyngeal carcinoma | ||||||
| CK2α | 5.01 10−5 | 1.381 | 6% | Sengupta Head-Neck | 41 | [77] |
| Oropharyngeal Carcinoma | ||||||
| CK2α | 6.85 10−4 | 1.737 | 7% | Pyeon Multi-cancer | 84 | [63] |
| Tongue Carcinoma | ||||||
| CK2α | 1.31 10−6 | 1.735 | 2% | Pyeon Multi-cancer | 84 | [63] |
| CK2α’ | 1.74 10−6 | 1.836 | 3% | Pyeon Multi-cancer | 84 | [63] |
| CK2β | 5.75 10−4 | 1.689 | 14% | Pyeon Multi-cancer | 84 | [63] |
| 3.76 10−4 | 1.409 | 19% | Talbot Lung | 93 | [78] | |
| 0.003 | 1.605 | 23% | Estilo Head-Neck | 58 | [63] | |
| Tonsillar Carcinoma | ||||||
| CK2β | 0.006 | 1.514 | 7% | Pyeon Multi-cancer | 84 | [63] |
HNSCC sections show strong nuclear immunostaining of all three CK2 proteins [33, 74], correlating with elevated levels of CK2 activity in nuclear and chromatin fractions compared to cytosol fractions [31–33]. HNSCC cell lines also show increased expression levels of all three CK2 proteins [74].
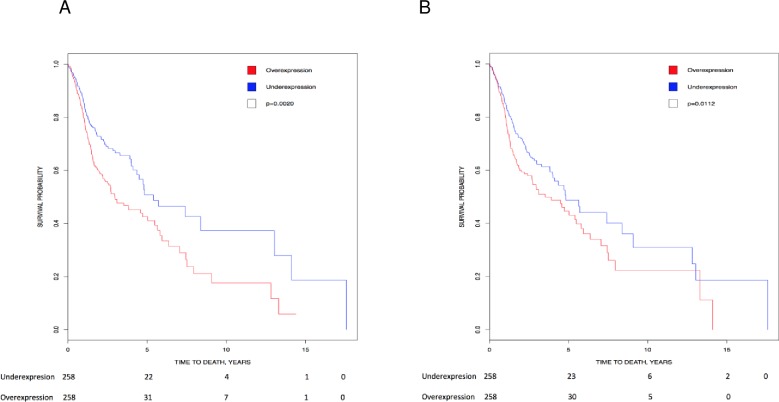
Kaplan-Meier analysis showed that higher expression of CK2α’ (p = 0.002) and CK2β (p = 0.0112) transcripts directly correlated with lower survival in head and neck cancer patients (Fig 4). In line with these data, there is high CK2 activity (nuclear, cytosolic and chromatin bound) in head and neck cancer [31–33], which correlates with disease status and decreased patient survival [31]. Based on the data above, levels of CK2 transcripts and protein activity could be a prognostic marker for HNSCC. Therefore, CK2 could be a target for cancer therapy. Indeed, pre-clinical evidence suggests that CK2 inhibitors could be effective in HNSCC (reviewed in [16]). In addition, a pre-clinical model of feline oral squamous cell carcinoma, thought to be a good model for human head and neck cancer, show promising results when CK2α and CK2α’ are targeted with RNAi [75]. CK2 targeting is associated with low toxicity (little adverse events, with weight loss and anorexia being the most common).
Fig 4. Correlation of CK2 gene expression to overall patient survival in head and neck cancer.
Patients were stratified into above-median (red line) and below-median (blue line) expression of CK2. (A) High levels of CK2α’ transcript correlated with lower survival (HR = 1.53, 95% CI [1.166, 2.009]; p = 0.002). (B) High levels of CK2β transcript correlated with lower survival (HR = 1.411, 95% CI [1.080, 1.843]; p = 0.0112).
Kidney cancer
Types of kidney cancer include renal cell carcinoma (80%) (subdivided into clear cell, papillary, and chromophobe), renal pelvis carcinoma, transitional cell carcinoma (7%), Wilms tumor (develops in children under 5 years of age), and renal sarcoma (including clear cell sarcoma of the kidney, a rare form that occurs in children typically between 1–4 years of age). Renal oncocytoma is a benign renal tumor. Incidence of kidney cancer has been increasing by an average of 1.6% per year over the past decade. Kidney cancer is twice as common in men as in women, and is also more prevalent among African Americans, American Indians, and Alaska natives. Factors contributing to kidney cancer include smoking, obesity, hypertension, and particular conditions that are inherited. There is currently no standard screening test for kidney cancer. However, individuals with increased risk due to inherited conditions can be screened for kidney cancer using computed tomography (CT) and magnetic resonance imaging (MRI). Treatments include surgery, radiation therapy, chemotherapy, biological therapy, and targeted therapy.
CK2 in kidney cancer
Data from Oncomine revealed a significant overexpression of all three CK2 genes in renal cancers as detailed in (Table 7). In contrast, Roelants et al. find a decrease in all CK2 transcript levels of 1.5–16 times by RT-qPCR in clear cell renal carcinoma [79]. This observation on decreased transcript levels is in conflict with our analysis and that of Rabjerg et al. [36, 39]. Rabjerg et al. show increased expression of CK2α, CK2α’ and CK2β transcripts in renal cell carcinoma by RT-qPCR in 97 patients (Rabjerg, Guerra et al. 2017)[39]. The Roelans et al. and Rabjerg et al. studies have similar demographics except for country of origin (Roelants et al: Grenoble, France, 53% males, 73% over 65 years, 33.3% pT3-T4, 66.7% Fuhrman grade III-IV, 20% metastasis,15 samples versus Rabjerg et al.: Denmark, 56% males, 49% over 65 years, 40% pT3-T4, 45% Fuhrman grade III-IV, 29% metastasis, 97 samples). The only difference between studies was the age of the patients. There was no data for CK2αP in any of the cancer types in Table 7.
Table 7. Analysis of changes in CK2 gene expression in kidney cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Chromophobe Renal Cell Carcinoma | ||||||
| CK2α | 1.64 10−5 | 2.212 | 1% | Yusenko Renal | 67 | [82] |
| CK2α’ | 0.002 | 1.517 | 13% | Jones Renal | 92 | [83] |
| 0.012 | 2.130 | 14% | Yusenko Renal | 67 | [82] | |
| Clear Cell Renal Cell Carcinoma | ||||||
| CK2α | 1.64 10−5 | 1.410 | 20% | Jones Renal | 92 | [83] |
| 1.11 10−4 | 1.444 | 6% | Yusenko Renal | 67 | [82] | |
| CK2β | 4.79 10−8 | 1.506 | 12% | Jones Renal | 92 | [83] |
| Clear Cell Sarcoma of the Kidney | ||||||
| CK2α | 9.5 10−5 | 1.569 | 3% | Cutcliffe Renal | 35 | [84] |
| Papillary Renal Cell Carcinoma | ||||||
| CK2α | 3.15 10−5 | 1.543 | 2% | Yusenko Renal | 67 | [82] |
| 5.5 10−4 | 1.360 | 26% | Jones Renal | 92 | [83] | |
| Renal Oncocytoma | ||||||
| CK2α | 4.76 10−5 | 1.993 | 2% | Yusenko Renal | 67 | [82] |
| 2.97 10−6 | 1.36 | 11% | Jones Renal | 92 | [83] | |
| CK2α’ | 1.11 10−7 | 1.505 | 7% | Jones Renal | 92 | [83] |
| 0.004 | 2.015 | 11% | Yusenko Renal | 67 | [82] | |
| CK2β | 0.005 | 1.424 | 12% | Yusenko Renal | 67 | [82] |
| Renal Pelvis Urothelial Carcinoma | ||||||
| CK2α | 9.74 10−4 | 1.364 | 19% | Jones Renal | 92 | [83] |
| CK2β | 9.79 10−4 | 1.448 | 19% | Jones Renal | 92 | [83] |
| Renal Wilms Tumor | ||||||
| CK2α | 5.36 10−5 | 2.199 | 1% | Yusenko Renal | 67 | [82] |
| 4.39 10−4 | 1.417 | 3% | Cutcliffe Renal | 35 | [84] | |
| CK2β | 0.002 | 1.500 | 5% | Cutcliffe Renal | 35 | [84] |
| 0.004 | 1.607 | 6% | Yusenko Renal | 67 | [82] | |
Despite finding decreased levels of all CK2 transcripts, Roelants et al. find increased CK2 activity, and increase in CK2α, CK2α’ and CK2β protein levels in renal cell carcinoma in 15 patients [79]. Similarly, other renal clear cell carcinoma studies show increase in CK2 activity [36, 80]. Timofeeva et al. find higher staining of CK2β protein in primary kidney tumors while expression of CK2α protein is unchanged [81]; and propose that CK2 activity may be upregulated in these kidney tumors, as increased expression of CK2β results in increased CK2 activity [81].
CK2 protein and transcript have diagnostic value in renal cell carcinoma. Thus, high nuclear CK2α staining correlates with late metastasis, but no other clinical variables [36]. Furthermore, high CK2α transcript expression correlated with high Fuhrman grade and tumor stage and, intriguingly, high CK2α’ transcript expression and low Fuhrman grade [36].
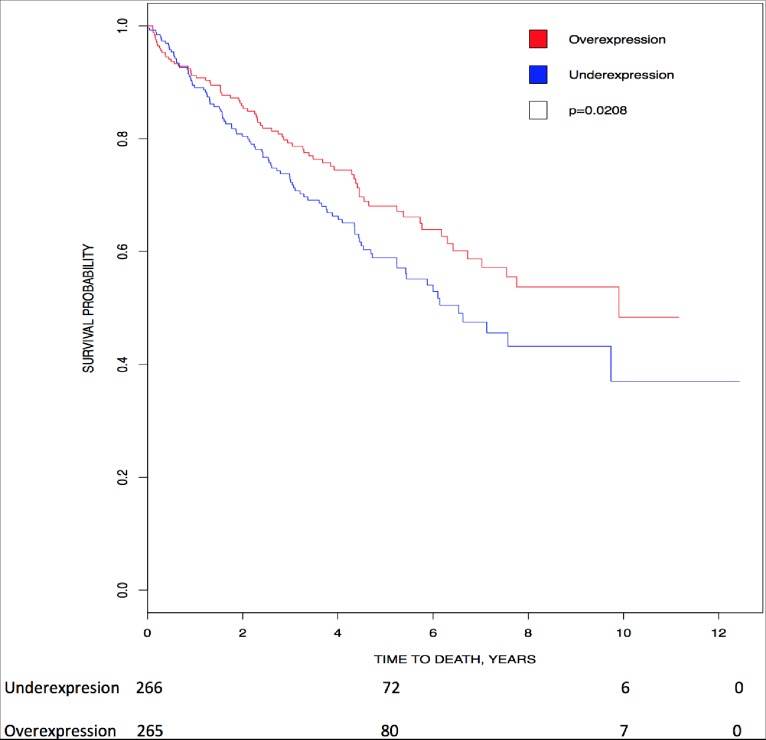
Unexpectedly, our Kaplan-Meier analysis showed that higher expression of CK2αP (p = 0.0208) directly correlated with higher survival in patients with renal clear cell carcinoma (Fig 5). In addition, Rabjerg et al. find that high CK2α transcript expression and high nuclear CK2α staining correlate with poor overall survival, disease specific survival and progression free survival in patients with renal cell carcinoma [36, 39]. In summary, CK2αP, and CK2α transcript and protein have a prognostic value in renal cell carcinoma; and nuclear CK2α protein, CK2α and CK2α’ transcripts have a diagnosis value.
Fig 5. Correlation of CK2 gene expression to overall patient survival in renal clear cell carcinoma.
Patients were stratified into above-median (red line) and below-median (blue line) expression of CK2αP. High levels of CK2αP transcript correlated with higher survival (HR = 0.702, 95% CI [0.520, 0.949]; p = 0.0208).
Leukemia
Leukemia is a cancer where the bone marrow produces abnormal white blood cells known as leukemia cells, and is one of the most common childhood cancers. Such cells are non-apoptotic, and their expansion can impede proper function of normal white blood cells, red blood cells, and platelets. Leukemia can be divided into four main types, which are acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL, most common in children), chronic lymphocytic leukemia (CLL), and chronic myeloid leukemia (CML). Hairy cell leukemia (HCL) is rare (2% of all leukemia cases). Risk factors include smoking, chemical exposure such as benzene, radiation exposure, history of chemotherapy or radiation therapy, inherited or genetic conditions, blood disorders, and a family history of leukemia. There is currently no standard screening test for leukemia. Treatments include watchful waiting, chemotherapy, targeted therapy, radiation therapy, and stem cell transplant.
CK2 in leukemia
CK2α’, CK2β and CK2αP transcripts were overexpressed in ALL. In T-cell ALL (T-ALL) and B-cell ALL (B-ALL) there were conflicting results for CK2α (Table 8). For B-ALL, the Coustan-Smith et al. (Finland) and the Maia et al. study (USA) studies showed significant downregulation, while the Andersson et al. study (Sweden) showed significant upregulation of the CK2α transcript [85–87]. In all three studies, leukemia samples were collected from children (Coustan-Smith et al. from patients age 1–18y; Maia et al. from patients age 0.1–17y; Andersson et al. did not specify children’s age range). The studies did not disclose patient demographics, including sex or ethnicity. The Coustan-Smith et al. and the Maia et al. studies both used age-matched controls while the Andersson et al. study used adult controls. This could potentially have confounded their results as there are innate differences in immunity between the adult and pediatric population [88]. Similarly, for T-ALL, the Coustan-Smith et al. study and Andersson et al. study showed opposing findings, with the Coustan-Smith et al. study showing significant downregulation while the Andersson et al. study showed significant upregulation of CK2α transcript expression. As stated above, these studies focused on the pediatric population, however Andersson et al. did not specify the age range. There was also no mention of patient demographics. Furthermore, the Andersson et al. study used adult controls. Together, these could have accounted for the differences in results between the two studies. Published studies find CK2α protein levels and CK2 activity are elevated in primary B-ALL [89] and T-ALL cells [90]. CK2α’ protein levels are elevated in primary B-ALL cells [89]. CK2β protein levels elevated in primary T-ALL cells [90].
Table 8. Analysis of changes in CK2 gene expression in leukemia.
P-values, fold change, rank and datasets are shown. (* = childhood leukemia).
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Pro-B Acute Lymphoblastic Leukemia | ||||||
| CK2α’ | 9.25 10−25 | 1.515 | 3% | Haferlach Leukemia | 2096 | [104] |
| B-Cell Acute Lymphoblastic Leukemia (B-ALL) | ||||||
| CK2α | 1.57 10−4 | -1.63 | 5% | Maia Leukemia* | 28 | [85] |
| 0.001 | -1.758 | 10% | Coustan-Smith Leukemia* | 288 | [86] | |
| 0.004 | 1.528 | 35% | Andersson Leukemia* | 127 | [87] | |
| CK2α’ | 4.54 10−26 | 1.421 | 5% | Haferlach Leukemia | 2096 | [104] |
| 8.31 10−29 | 1.389 | 6% | Haferlach Leukemia* | 2096 | [104] | |
| 2.65 10−7 | 2.189 | 10% | Andersson Leukemia* | 127 | [87] | |
| CK2β | 6.45 10−15 | 2.130 | 2% | Andersson Leukemia* | 127 | [87] |
| CK2αP | 0.004 | 1.528 | 35% | Andersson Leukemia* | 127 | [87] |
| T-Cell Acute Lymphoblastic Leukemia (T-ALL) | ||||||
| CK2α | 4.04 10−4 | 1.743 | 20% | Andersson Leukemia* | 127 | [87] |
| 0.004 | -1.399 | 14% | Coustan-Smith Leukemia* | 288 | [86] | |
| CK2α’ | 1.77 10−7 | 2.496 | 4% | Andersson Leukemia* | 127 | [87] |
| 1.35 10−27 | 1.576 | 5% | Haferlach Leukemia | 2096 | [104] | |
| CK2β | 1.44 10−8 | 2.654 | 2% | Andersson Leukemia* | 127 | [87] |
| CK2αP | 0.011 | 1.589 | 33% | Andersson Leukemia* | 127 | [87] |
| Acute Myeloid Leukemia (AML) | ||||||
| CK2α | 0.002 | 1.609 | 27% | Andersson Leukemia* | 127 | [87] |
| CK2α’ | 1.47 10−7 | 2.030 | 5% | Andersson Leukemia* | 127 | [87] |
| 1.05 10−20 | 1.417 | 5% | Haferlach Leukemia* | 2096 | [104] | |
| CK2β | 3.26 10−8 | 2.006 | 4% | Andersson Leukemia* | 127 | [87] |
| 2.96 10−4 | -1.928 | 5% | Stegmaier Leukemia* | 87 | [91] | |
| CK2αP | 0.002 | 1.609 | 27% | Andersson Leukemia* | 127 | [87] |
| Chronic Lymphocytic Leukemia (CLL) | ||||||
| CK2α | 1.34 10−6 | -2.228 | 5% | Rosenwald Multi-cancer | 102 | [105] |
| 5.89 10−34 | -1.453 | 7% | Haferlach Leukemia | 2096 | [104] | |
| 2.06 10−4 | -1.505 | 10% | Alizadeh Leukemia | 120 | [97] | |
| CK2α’ | 3.13 10−34 | 1.455 | 8% | Haferlach Leukemia | 2096 | [104] |
| 3.82 10−4 | -1.316 | 19% | Rosenwald Multi-cancer | 102 | [105] | |
| CK2β | 3.33 10−4 | 1.885 | 10% | Haslinger Leukemia | 111 | [98] |
| 2.81 10−6 | -1.391 | 8% | Basso Lymphoma | 336 | [100] | |
| 0.004 | -1.381 | 8% | Rosenwald Lymphoma | 293 | [101] | |
| Hairy Cell Leukemia (HCL) | ||||||
| CK2α | 2.48 10−4 | -1.308 | 16% | Basso Lymphoma | 336 | [100] |
| CK2α’ | 1.42 10−8 | -1.876 | 3% | Basso Lymphoma | 336 | [100] |
In AML, Oncomine analysis showed overexpression of CK2α, CK2α’ and CK2αP, and conflicting findings for CK2β. Thus, the Andersson et al. study (Sweden) assayed AML samples from pediatric patients, while the Stegmaier et al. study (USA) used adult patients [87, 91]. As above, the intrinsic differences between the pediatric and adult population may play a role in the discrepancy between the two studies for CK2β transcript expression levels. In this regard, there are differences in gene abnormalities between pediatric and adult patients in other cancers and diseases. For example, the frequency of prognostic markers among pediatric and adult B-ALL differ, with adult patients harboring more mutations and epigenetic modifications compared to pediatric patients [92]. Similarly, there are pediatric-adult differences in mature B-cell non-Hodgkin lymphoma and Celiac disease [93, 94]. These studies indicate that gene expression alterations may also be different among pediatric and adult AML patients, as we see here for CK2β. Similar to our findings, published studies also demonstrate CK2α transcript overexpression in AML cell lines compared to normal hematopoietic cells [95]. As for CK2 proteins, primary AML cells show both increased and decreased levels of CK2α protein and CK2 activity [95, 96]. High CK2α protein levels are a prognostic biomarker, as they correlated with low overall survival and decreased disease-free survival in AML patients [96]. However, high levels of CK2α protein do not correlate with clinical variables, including subtype [96].
CLL showed downregulation of CK2α, and conflicting findings for CK2α’ and CK2β. For CK2α’, the Haferlach et al. study found significant upregulation, while the Rosenwald et al. study showed significant downregulation. The Haferlach et al. study was a multicenter study conducted across seven countries in 11 different study centers, while the Rosenwald et al. study was a single center study conducted in the United States. However, demographic information in the two studies were not provided. Haferlach et al. used samples from patients who had not undergone prior treatment, while 12.1% of patients in the Rosenwald et al. study had received prior chemotherapy treatment. Additionally, the Haferlach et al. used bone marrow and/or peripheral blood samples, while the Rosenwald et al. used only peripheral blood samples. In the control group, Haferlach et al. used bone marrow samples from healthy individuals or individuals without leukemia (such individuals may have a preexisting blood disorder such as hemophilia). Rosenwald et al. used samples in their control group from a prior study where they collected cells form tonsils, adult apheresis products, or cord blood [97]. Together, these differences across the two studies could have led to the differences in results. For CK2β, the Haslinger et al (Germany) study had 62% male patients (30–87 years; median 62.5) [98]. 62% patients had Binet stage A, 33% had stage B, and 5 patients had stage C CLL (the stage that has the worst prognosis). Additionally, 47% of patients had VH-mutations [98], which has a more favorable prognosis and less aggressive CLL disease compared to patients with unmutated VH [99]. Rosenwald et al. (USA) and Basso et al. (USA) did not disclose patient demographic or clinicopathological information [100, 101]. Thus, we do not have enough data to explain the discrepancy in the results. In contrast to our transcript level results, primary CLL cells show increased CK2α and CK2β proteins and CK2 activity [102]. These data suggest that CK2 is regulated both transcriptionally and post-transcriptionally.
In HCL, CK2α’ was downregulated. Published data show that in Jurkat, a T-ALL cell line, CSNK2A1P has high transcript levels correlating with gene copy number [103].
There was no data for CML in Oncomine. Pre-clinical evidence suggests that CK2 inhibitors could be effective in treating leukemia (reviewed in [16]).
Lymphoma
Lymphoma, a cancer of the lymphocytes, is the leading blood cancer in the USA. Lymphoma can be subdivided into Hodgkin lymphoma (defined by the presence of Reed-Sternberg cells) and non-Hodgkin lymphoma (NHL). NHLs can derive from B-cells (85%), T-cells (<15%) or NK cells (rare). NHLs can be further subdivided into high grade (fast-growing) and low grade (slow-growing).
Types of B-cell NHLs include low-grade lymphomas (follicular lymphoma, mantle cell lymphoma) and high-grade lymphomas (diffuse large B-cell lymphoma (DLBCL), Burkitt lymphoma, and primary effusion lymphoma). DLBCL can be further subdivided into activated B cell-like DLBCL, which has the lowest survival of all NHL types, and germinal center B cell-like DLBCL. The most common high grade NHL is DLBCL; the most common low grade NHL is follicular lymphoma.
T-cell NHL can include anaplastic large cell lymphoma, angioimmunoblastic T-cell lymphoma, and unspecified peripheral T-cell lymphoma, which are all high-grade lymphomas. Adult T-cell leukemia/lymphoma (ATL) is usually a highly aggressive NHL.
Risk factors for lymphoma include HIV, Epstein-Barr virus, Helicobacter pylori, or human T-cell leukemia/lymphoma virus type 1 (HTLV-1) infection, having a weak immune system and age. Both high and low grade NHL are treated with chemotherapy, and, increasingly, biological (stem cell transplant) or targeted therapies.
CK2 in non-Hodgkin lymphoma
Oncomine analysis showed mixed regulation of all three CK2 transcripts in NHL (Table 9). There was only data for CK2αP in follicular lymphoma and DLBCL. The data in Oncomine for Hodgkin lymphoma was not-significant.
Table 9. Analysis of changes in CK2 gene expression in non-Hodgkin lymphoma.
P-values, fold change, rank and datasets are shown. (NOS = not otherwise specified).
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Follicular Lymphoma (low grade B-cell NHL) | ||||||
| CK2α | 3.21 10−7 | -1.421 | 17% | Compagno Lymphoma | 136 | [108] |
| CK2α’ | 8.51 10−4 | -2.263 | 3% | Storz Lymphoma | 27 | [109] |
| 0.004 | 1.341 | 13% | Brune Lymphoma | 67 | [110] | |
| 8.94 10−4 | -1.301 | 40% | Compagno Lymphoma | 136 | [108] | |
| CK2β | 4.37 10−8 | -1.493 | 13% | Compagno Lymphoma | 136 | [108] |
| Mantle Cell Lymphoma (low grade B-cell NHL) | ||||||
| CK2α | 7.63 10−4 | 1.400 | 11% | Basso Lymphoma | 336 | [100] |
| CK2β | 0.001 | 1.376 | 13% | Basso Lymphoma | 336 | [100] |
| Diffuse Large B Cell Lymphoma NOS (DLBCL-NOS) (high-grade B-cell NHL) | ||||||
| CK2α | 0.001 | 1.324 | 15% | Basso Lymphoma | 336 | [100] |
| 0.003 | 1.350 | 16% | Rosenwald Lymphoma | 293 | [101] | |
| 0.001 | -1.564 | 11% | Rosenwald Multi-cancer | 102 | [105] | |
| Activated B-Cell-like Diffuse Large B Cell Lymphoma (DLBCL) (high-grade B-cell NHL) | ||||||
| CK2α | 4.29 10−5 | 1.383 | 18% | Compagno Lymphoma | 136 | [108] |
| CK2β | 1.18 10−4 | 1.616 | 12% | Alizadeh Lymphoma | 120 | [97] |
| Germinal Center B-Cell-like Diffuse Large B Cell Lymphoma (DLBCL) (high-grade B-cell NHL) | ||||||
| CK2α | 0.003 | -1.380 | 26% | Compagno Lymphoma | 136 | [108] |
| CK2α’ | 1.15 10−5 | 1.322 | 8% | Alizadeh Lymphoma | 120 | [97] |
| CK2β | 0.008 | 1.530 | 23% | Alizadeh Lymphoma | 120 | [97] |
| Burkitt’s Lymphoma (high-grade B-cell NHL) | ||||||
| CK2α | 0.005 | 1.302 | 24% | Basso Lymphoma | 336 | [100] |
| CK2β | 2.44 10−4 | 2.331 | 9% | Brune Lymphoma | 67 | [110] |
| Primary Effusion Lymphoma (high-grade B-cell NHL) | ||||||
| CK2α’ | 0.001 | -1.853 | 11% | Basso Lymphoma | 336 | [100] |
| Anaplastic Large Cell Lymphoma (high-grade T-cell NHL) | ||||||
| CK2α | 2.78 10−6 | 1.844 | 5% | Piccaluga Lymphoma | 60 | [111] |
| 0.01 | 1.437 | 25% | Eckerle Lymphoma | 64 | [112] | |
| CK2α’ | 3.66 10−8 | 2.195 | 2% | Piccaluga Lymphoma | 60 | [111] |
| CK2β | 0.003 | 2.002 | 25% | Piccaluga Lymphoma | 60 | [111] |
| Angioimmunoblastic T-Cell Lymphoma (high-grade T-cell NHL) | ||||||
| CK2α | 4.57 10−4 | 1.940 | 15% | Piccaluga Lymphoma | 60 | [111] |
| CK2α’ | 0.004 | 1.858 | 25% | Piccaluga Lymphoma | 60 | [111] |
| CK2β | 0.006 | 1.549 | 26% | Piccaluga Lymphoma | 60 | [111] |
| Unspecified Peripheral T-Cell Lymphoma (high-grade T-cell NHL) | ||||||
| CK2α | 3.32 10−5 | 1.525 | 21% | Piccaluga Lymphoma | 60 | [111] |
| CK2α’ | 5.79 10−4 | 1.427 | 27% | Piccaluga Lymphoma | 60 | [111] |
| Chronic Adult T-cell Leukemia/Lymphoma (ATL or ATLL) | ||||||
| CK2α’ | 0.011 | -1.534 | 12% | Choi Leukemia | 47 | [113] |
| T-cell/Histiocyte-rich B-cell Lymphoma (T/HRBCL) | ||||||
| CK2α | 7.9 10−4 | 1.323 | 4% | Brune Lymphoma | 67 | [110] |
In follicular lymphoma, the Storz et al. and Compagno et al. studies show downregulation while the Brune et al. study show upregulation of CK2α’ transcript expression. Both the Storz et al. study and the Compagno et al. studies were conducted in the United States, while the Brune et al. study was conducted in Germany. Only the Storz et al. study provided demographic information, with patient ages ranging from 34 to 88. No other information was provided. All three studies used tonsil samples from healthy individuals. There is not enough information to speculate other reasons for the differences in results.
In DLBCL-NOS, the Basso et al. and 2002 Rosenwald et al. study show upregulation of CK2α transcript expression, while the 2001 Rosenwald et al. study showed downregulation. There is not enough information to speculate other reasons for the differences in results in these USA studies, as only the 2002 Rosenwald et al. study provided patient demographic information, with 55% of patients >60 years old (median age 63), 56% male, 37% had low risk DLBCL while 14% had high risk DLBCL, and most patients in this study (47%) had centroblastic monomorphic histologic subtype.
With respect to protein levels, CK2α and CK2β protein staining is stronger in follicular lymphoma, Burkitt’s lymphoma, and DLBCL, and in lymphoma cell lines, showing both nuclear and cytoplasmic staining [106]. However, the intensity of staining did not correlate with tumor grade, at least in follicular lymphoma [106]. In addition, CK2α and CK2β staining was mantle cell lymphoma tissue sections [107]. Interestingly, overexpression of CK2α in lymphocytes of transgenic mice results in T cell lymphomas, suggesting an oncogenic role for CK2α [17–19].
Monoclonal gammopathies
Myeloma is the 3rd most common blood cancer and is caused by abnormal plasma cells, called myeloma cells, that build up in the bone marrow and cause damage to the solid bone. Accumulation of myeloma cells leading to macroscopic lesions in multiple bone sites is termed “multiple myeloma”. In addition to affecting bones, myeloma can also damage other tissues and organs. Types of myeloma include smoldering myeloma (or asymptomatic myeloma), symptomatic myeloma, and plasma cell leukemia, a rare and aggressive form of myeloma with poor prognosis. Elevated levels of antibodies, known as M proteins, produced by myeloma cells in blood, urine, and organs leads to a condition called monoclonal gammopathy of undetermined significance (MGUS). While this condition can be asymptomatic, it can become associated with other diseases or progress to myeloma or other plasma cell malignancies (eg, lymphoma or amyloidosis). Risk factors for myeloma include older age, history of MGUS or having isolated plasmacytoma of the bone. Demografic (age, race), genetic and environmental factors (e.g. radiation, herbicides) play a role in the development of MGUS. There is currently no standard screening test for myeloma. Myeloma treatments include chemotherapy, targeted therapy, biological therapy, corticosteroid treatment, and stem cell transplant with high-dose chemotherapy. In certain instances, radiation or surgery to repair damaged bones may be needed.
CK2 in monoclonal gammopathies
Oncomine analysis showed significant overexpression of CK2α, CK2α’ and CK2β (Table 10). MGUS showed conflicting data for CK2α, where the Agnelli et al. study showed underexpression while the Zhan et al. study showed overexpression [114, 115]. The Zhan et al. study was conducted in the USA (45% females, 36% over 65 years, 84% white race) and the Agnelli et al. study was conducted in Italy (~40% females, median age 67). We cannot explain the discrepancy. There was no data for CK2αP in any of the types listed in Table 10.
Table 10. Analysis of changes in CK2 gene expression in monoclonal gammopathies.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Monoclonal Gammopathy of Undetermined Significance (MGUS) | ||||||
| CK2α | 1.18 10−6 | 1.594 | 4% | Zhan Myeloma 3 | 78 | [115] |
| 6.61 10−5 | -1.619 | 1% | Agnelli Myeloma 3 | 158 | [114] | |
| CK2α’ | 0.003 | 1.225 | 26% | Zhan Myeloma 3 | 78 | [115] |
| CK2β | 0.001 | 1.308 | 21% | Zhan Myeloma 3 | 78 | [115] |
| Smoldering Myeloma | ||||||
| CK2α | 3.36 10−9 | 1.763 | 1% | Zhan Myeloma 3 | 78 | [115] |
| CK2α’ | 0.002 | 1.378 | 32% | Zhan Myeloma 3 | 78 | [115] |
| CK2β | 1.73 10−4 | 1.779 | 22% | Zhan Myeloma 3 | 78 | [115] |
| Plasma Cell Leukemia | ||||||
| CK2α | 0.001 | 1.881 | 8% | Agnelli Myeloma 3 | 158 | [114] |
| CK2β | 6.4 10−4 | 1.461 | 7% | Agnelli Myeloma 3 | 158 | [114] |
Interestingly, multiple myeloma primary cells and cell lines had elevated levels of CK2α and CK2β proteins and CK2 activity [116], and CK2α and CK2β staining was darker in multiple myeloma tissue sections [107]. However, in MGUS, CK2α and CK2β staining was no different from normal hematopoietic cells [107]. Therefore, it is plausible that CK2 transcript levels are elevated in myeloma.
Liver cancer
Liver cancer most commonly begins in hepatocytes giving rise to hepatocellular carcinoma (also known as primary liver cancer). Other liver cancer types such as fibrolamellar carcinoma, hemangiosarcoma, and hepatoblastoma are rare. Hepatocellular carcinoma is most commonly caused by cirrhosis of the liver due to alcohol abuse, hepatitis B, hepatitis C, hemochromatosis, steatohepatitis, obesity, and diabetes. There is currently no standard screening test for liver cancer. Treatments include surgery, liver transplant, ablation, embolization, radiation therapy, chemotherapy, and targeted therapy.
CK2 in liver cancer
Oncomine analysis showed significant overexpression of CK2α and CK2β in hepatocellular carcinoma (Table 11). In agreement with our analysis, two other groups find CK2α transcripts are upregulated in hepatocellular carcinoma [40, 117]. CK2α transcript levels correlate with increasing tumor grade [40], and with elevated levels of CK2α protein in hepatocellular carcinoma samples [40, 117]. Therefore, increases in CK2α protein may be due to CK2α transcript upregulation. As for subcellular localization, CK2α staining is found in membrane and cytoplasm [117].
Table 11. Analysis of changes in CK2 gene expression in liver cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Hepatocellular Carcinoma | ||||||
| CK2α | 9.93 10−45 | 1.801 | 6% | Roessler Liver 2 | 445 | [119] |
| 1.94 10−5 | 1.774 | 9% | Roessler Liver | 43 | [119] | |
| CK2β | 1.69 10−50 | 1.630 | 4% | Roessler Liver 2 | 445 | [119] |
| 1.53 10−6 | 1.720 | 6% | Roessler Liver | 43 | [119] | |
| 1.07 10−8 | 1.417 | 8% | Chen Liver | 197 | [120] | |
| 0.006 | 1.371 | 20% | Wurmbach Liver | 75 | [119, 121] | |
Importantly, high CK2α protein staining correlates with histological grade, distant metastasis and tumor stage [117].
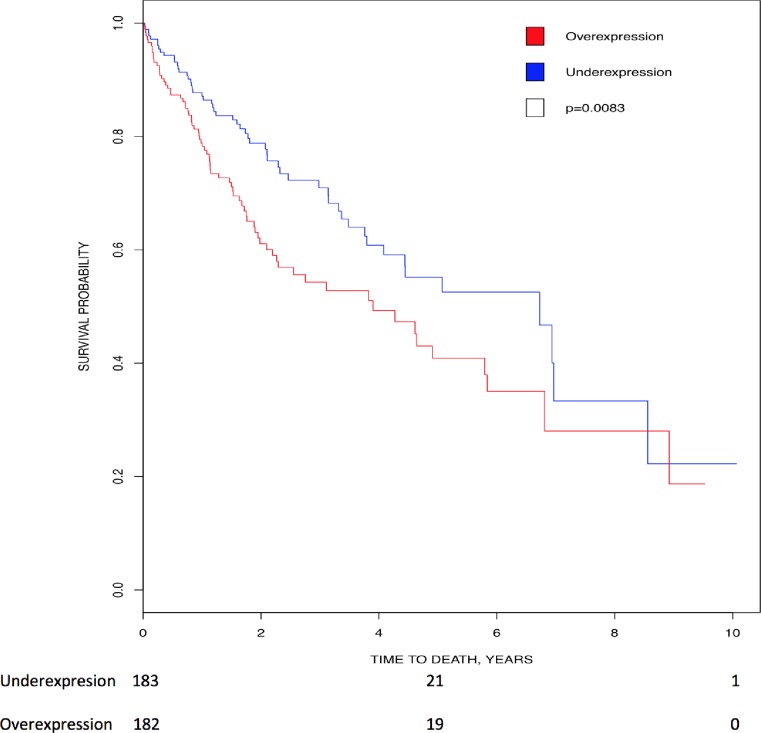
Kaplan-Meier analysis showed that higher expression of CK2α’ (p = 0.0083) directly correlated with lower survival in liver cancer (Fig 6). In addition, patients with elevated CK2α transcript [40] and protein staining [117] had a lower survival rate. These authors find that CK2α transcript overexpression is an independent prognostic factor (survival) [40, 117].
Fig 6. Correlation of CK2 gene expression to overall patient survival in liver cancer.
Patients were stratified into above-median (red line) and below-median (blue line) expression of CK2α’. High levels of CK2α’ transcript correlated with lower survival (HR = 1.592, 95% CI [1.123, 2.256]; p = 0.0083).
All together these data show that, CK2α and CK2α’ transcripts and CK2α protein have prognostic value, and CK2α transcript and protein have diagnostic value for hepatocellular carcinoma.
In addition to potential treatment of hepatic tumors with CK2 inhibitors, there is a potential role for CK2 inhibition in preventing steatohepatitis and subsequently associated hepatocellular carcinoma, as the CK2 phosphorylation site in Sirtuin 1 is important in the pathophysiology of obesity and hepatic steatosis [118].
Mesothelioma
Mesothelioma is a rare type of cancer that forms from cells of the mesothelium. Mesothelioma is subdivided based on location of origin into pleural (75%), peritoneal (10–20%), pericardial (1%) and testicular (< 1%). Mesothelioma is also classified according to their compromised cell type into epithelial (50–70%), sarcomatoid (10–20%), and biphasic (mixed epithelial and sarcomatoid; 20–35%). Patients with epithelial mesothelioma have the highest survival rate due to better response to treatment. Risk factors include exposure to asbestos, even for a short amount of time. Other risk factors include radiation exposure, intrapleural thorium dioxide, inhalation of other fibrous silicates, or inheriting a germline mutation on the BAP1 gene. Following diagnosis, only about 40% of patients survive past the first year. There is no standard screening test for mesothelioma, especially for those who were exposed to asbestos. Treatment options include surgery, radiation therapy, chemotherapy, immunotherapy, and/or intraoperative intraperitoneal chemotherapy.
CK2 in mesothelioma
Oncomine analysis showed significant overexpression of CK2α and CK2β transcripts in malignant pleural mesothelioma (Table 12). These findings were consistent with a recently published study, where they also find an overexpression of CK2α transcript, and elevated staining of CK2α protein in malignant pleural mesothelioma [122]. There was no data for CK2αP in mesothelioma.
Table 12. Analysis of changes in CK2 gene expression in mesothelioma.
P-values, fold change, rank and datasets are shown.
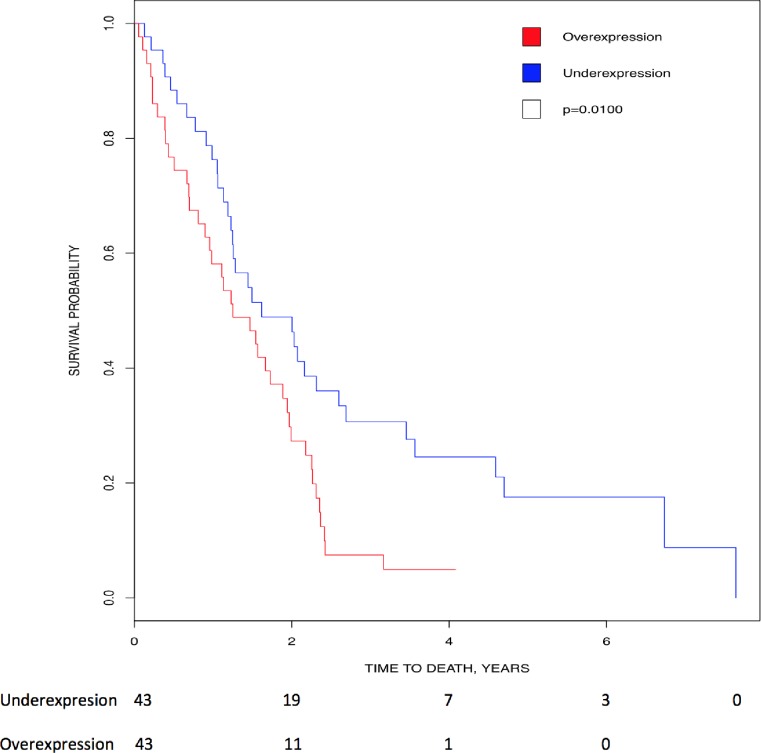
Kaplan-Meier analysis showed that higher expression of CK2αP (p = 0.01) directly correlated with lower survival in mesothelioma, suggesting its prognostic value in this cancer (Fig 7).
Fig 7. Correlation of CK2 gene expression to overall patient survival in mesothelioma.
Patients were stratified into above-median (red line) and below-median (blue line) expression of CK2αP. High levels of CK2αP transcript correlated with lower survival (HR = 1.873, 95% CI [1.153, 3.041]; p = 0.01).
Parathyroid cancer
Parathyroid cancer affects any of the four parathyroid glands. Both benign and malignant parathyroid tumors can lead to hyperparathyroidism, where excess parathyroid hormone (PTH) is produced leading to over-absorption of dietary calcium in the intestines, and movement of stored calcium in the bones into blood. These events ultimately lead to hypercalcemia, where amount of calcium in the blood rises above normal levels. Risk factors for parathyroid adenoma include rare inherited disorders such as familial isolated hyperparathyroidism and multiple endocrine neoplasia type 1 syndrome. Treatment options include minimally invasive surgery to remove the tumor itself or treating the hypercalcemia (gallium nitrate, calcitonin, IV fluids, or drugs that prevent breakdown and reabsorption of bones). Chemotherapy and radiation therapy are not very effective in preventing parathyroid cancer recurrence. Furthermore, radiation therapy can instead increase risks of developing parathyroid adenoma.
CK2 in parathyroid overgrowth
Oncomine analysis showed significant overexpression of CK2α transcript in three types of parathyroid overgrowth (hyperplasia and benign), as well as an overexpression of CK2α’ transcript in parathyroid hyperplasia (Table 13). There was no data for CK2αP in any of the types in Table 13.
Table 13. Analysis of changes in CK2 gene expression in parathyroid overgrowth.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Parathyroid Hyperplasia | ||||||
| CK2α | 0.005 | 1.629 | 9% | Morrison Parathyroid | 61 | [124] |
| CK2α’ | 0.008 | 1.506 | 13% | Morrison Parathyroid | 61 | [124] |
| Non-Familial Multiple Gland Neoplasia | ||||||
| CK2α | 0.003 | 1.678 | 9% | Morrison Parathyroid | 61 | [124] |
| Parathyroid Gland Adenoma | ||||||
| CK2α | 0.003 | 1.750 | 12% | Morrison Parathyroid | 61 | [124] |
Thyroid cancer
Thyroid cancer occurs in the thyroid gland, where thyroid follicular cells normally produce thyroid hormone which regulates bodily functions such as body temperature, weight, blood pressure, and heart rate. The thyroid gland also consists of C cells which secrete calcitonin, which is important in calcium homeostasis in the body. There are four types of thyroid cancer, including papillary, follicular, medullary, and anaplastic thyroid cancer. Papillary thyroid cancer is the most common among the four, while anaplastic thyroid cancer remains the most aggressive with the lowest cure rates. Risks for thyroid cancer include genetic conditions (eg, familial medullary thyroid cancer, multiple endocrine neoplasia type 2A syndrome, multiple endocrine neoplasia type 2B syndrome), and history of irradiation to the head and neck in childhood. There is no current standard screening test, other than neck palpation during annual primary care visits. Diagnosis is usually achieved using ultrasound of the neck and fine needle biopsy. Definitive treatment for thyroid cancer is achieved by thyroidectomy (surgical removal of the thyroid).
CK2 in thyroid cancer
Oncomine analysis showed significant underexpression of CK2α in papillary carcinoma (Table 14). There was no data for CK2αP.
Table 14. Analysis of changes in CK2 gene expression in thyroid cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Thyroid Gland Papillary Carcinoma | ||||||
| CK2α | 0.008 | -1.361 | 15% | He Thyroid | 18 | [126] |
Guo et al. found higher CK2α transcript levels and protein staining (in particular nuclear staining) in all four thyroid carcinoma subtypes (papillary, follicular, medullary, and anaplastic) [125]. Furthermore, higher nuclear CK2α protein staining was associated with lymph node metastasis and tumor stage and EMT markers, suggesting that CK2α staining can have diagnostic value [125]. This indicated a potential role for targeting CK2α for thyroid cancer treatment.
Sarcoma
Sarcoma develops from abnormal cells of mesenchymal origin. This can include muscle, bone, fat, and/or vascular tissues. There are more than 50 types of sarcoma, but the most common types include liposarcoma (adipose tissue), synovial sarcoma (synovial lining cells in joints), and leiomyosarcoma (smooth muscle). Rhabdomyosarcoma is the most common in children. Risk factors for sarcoma include inherited conditions such as retinoblastoma, Li-Faumeni syndrome, familial adenomatous polyposis, neurofibromatosis, Werner syndrome and tuberous sclerosis, and also chemical and/or radiation exposure. There is no current standard screening test for sarcomas, and they are usually diagnosed using imaging techniques such as CT and MRI scans. Additionally, removal of a tissue sample is needed to confirm the diagnosis. Treatment options include surgery, radiation therapy, and/or chemotherapy.
CK2 in sarcoma
Overall, Oncomine analysis revealed significant overexpression of CK2α, and mostly underexpression of CK2α’ in sarcoma. There was overexpression of CK2β in pleiomorphic liposarcoma and underexression in dedifferentiated liposarcoma. There were conflicting results for CK2α’ in leiomyosarcoma (Table 15). However, leiomyosarcoma samples in the Quade et al. study was obtained specifically from patients with leiomyosarcoma of the uterus. Detwiller et al. obtained samples from patients with leiomyosarcoma in any body region, although they did not specify. Control samples in the Quade et al. study were uterus samples from healthy individuals, while control samples in the Detwiller et al. study were soft tissue samples (not specified) from healthy individuals. Thus, these differences in sample source and type may have contributed to the different findings. There was no data for CK2αP in all types listed in Table 15.
Table 15. Analysis of changes in CK2 gene expression in sarcoma.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Dedifferentiated Liposarcoma | ||||||
| CK2α’ | 2.49 10−4 | -1.599 | 11% | Barretina Sarcoma | 158 | [128] |
| CK2β | 0.007 | -1.815 | 5% | Detwiller Sarcoma | 54 | [129] |
| Fibrosarcoma | ||||||
| CK2α | 7.85 10−4 | 1.975 | 7% | Detwiller Sarcoma | 54 | [129] |
| CK2α’ | 0.009 | -1.464 | 11% | Detwiller Sarcoma | 54 | [129] |
| Malignant fibrous histiocytoma (Pleomorphic Undifferentiated Sarcoma) | ||||||
| CK2α’ | 0.011 | -1.534 | 14% | Detwiller Sarcoma | 54 | [129] |
| Leiomyosarcoma | ||||||
| CK2α’ | 0.002 | 1.392 | 1% | Quade Uterus | 24 | [130] |
| 1.49 10−4 | -2.153 | 2% | Detwiller Sarcoma | 54 | [129] | |
| Myxofibrosarcoma | ||||||
| CK2α’ | 0.001 | -1.448 | 19% | Barretina Sarcoma | 158 | [128] |
| Pleomorphic Liposarcoma | ||||||
| CK2α | 0.003 | 1.422 | 6% | Detwiller Sarcoma | 54 | [129] |
| CK2α’ | 0.003 | -1.693 | 4% | Detwiller Sarcoma | 54 | [129] |
| 0.001 | -1.494 | 12% | Barretina Sarcoma | 158 | [128] | |
| CK2β | 5.13 10−5 | 1.334 | 11% | Barretina Sarcoma | 158 | [128] |
| Synovial Sarcoma | ||||||
| CK2α | 1.28 10−4 | 1.801 | 5% | Detwiller Sarcoma | 54 | [129] |
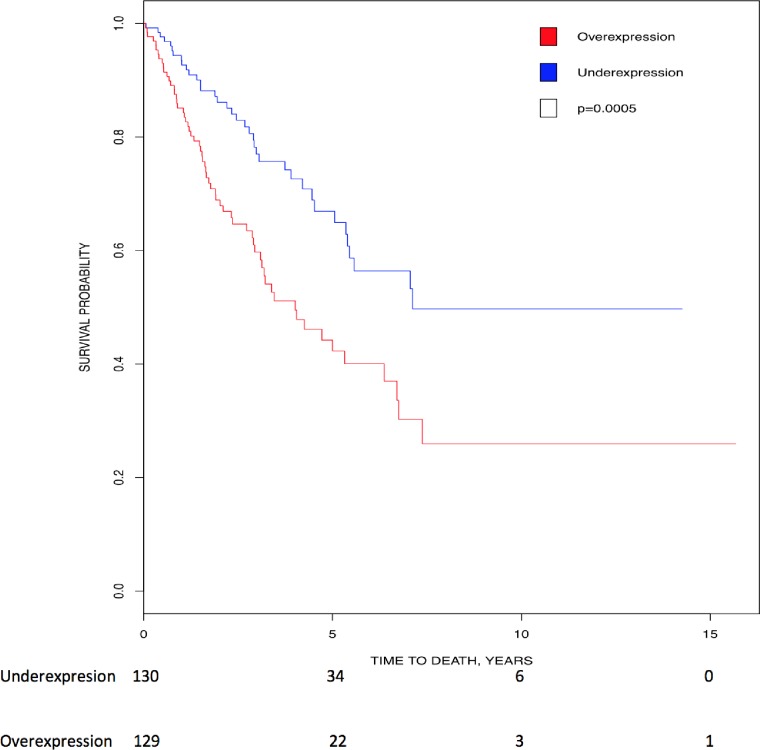
Kaplan-Meier analysis showed that higher expression of CK2α transcript (p = 0.0005) directly correlated with lower survival in sarcoma, suggesting its prognostic value in this cancer type (Fig 8).
Fig 8. Correlation of CK2 gene expression to overall patient survival in sarcoma.
Patients were stratified into above-median (red line) and below-median (blue line) expression of CK2α. High levels of CK2α transcript correlated with lower survival (HR = 2.038, 95% CI [1.353, 3.071]; p = 0.0005).
Takahashi et al. found overexpression of CK2α and CK2β protein in human osteosarcoma cell lines when compared to normal cells [127]. Furthermore, knockdown of either CK2α or CK2β with siRNA or incubation with CX-4945 led to prevention of osteosarcoma cell proliferation. However, CX-4945 did not inhibit cell proliferation of human mesenchymal stem cells, indicating a role for CK2 in osteosarcoma cell proliferation. In vivo results in xenograft mouse models found that CX-4945 inhibited osteosarcoma growth [127]. Together, these indicate a role for CK2 inhibition in osteosarcoma treatment.
Skin cancer
Skin cancer includes melanoma and non-melanoma skin cancer (basal cell carcinoma and squamous cell carcinoma). Melanoma is the most fatal form of skin cancer and arises from melanocytes. On the other hand, non-melanoma skin cancer usually responds well to treatment and is rarely metastatic. A risk factor for skin cancer is the presence of benign melanocytic skin nevus, more commonly known as moles and freckles. Other risk factors include fair skin, high exposure to natural (sun) or artificial UV light, history of blistering sunburns, and history (family or personal) of melanoma or atypical moles. The current standard screen for melanoma is a visual examination of the skin. Treatments include surgery, chemotherapy, targeted therapy, biological therapy, and radiation therapy.
CK2 in melanoma
Oncomine analysis revealed significant overexpression of all three CK2 genes as detailed in the Table below (Table 16). There was no data for CK2αP in any of the types listed in Table 16.
Table 16. Analysis of changes in CK2 gene expression in skin cancer.
P-values, fold change, rank and datasets are shown.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Benign Melanocytic Skin Nevus (moles and freckles) | ||||||
| CK2α | 0.002 | 5.016 | 10% | Talantov Melanoma | 70 | [133] |
| CK2α’ | 0.004 | 1.566 | 13% | Talantov Melanoma | 70 | [133] |
| CK2β | 8.28 10−4 | 1.797 | 8% | Talantov Melanoma | 70 | [133] |
| 1.02 10−5 | 1.754 | 2% | Haqq Melanoma | 37 | [134] | |
| Skin Basal Cell Carcinoma | ||||||
| CK2α | 5.15 10−4 | 1.453 | 6% | Riker Melanoma | 87 | [135] |
| Skin Squamous Cell Carcinoma | ||||||
| CK2α | 1.02 10−7 | 2.081 | 1% | Riker Melanoma | 87 | [135] |
| Melanoma | ||||||
| CK2α | 9.11 10−4 | 7.026 | 17% | Talantov Melanoma | 70 | [133] |
| CK2α’ | 1.91 10−4 | -2.533 | 3% | Riker Melanoma | 87 | [135] |
| CK2β | 9.30 10−5 | 2.478 | 10% | Talantov Melanoma | 70 | [133] |
| 0.004 | 2.198 | 14% | Haqq Melanoma | 37 | [134] | |
Interestingly, ninety percent of melanomas with CK2α transcript upregulation had mutations in the most prevalent genes mutated in melanoma: BRAF, NRAS and/or NF1 [131]. Moreover, these authors found increased CK2α protein expression in melanoma cell lines when compared with normal human melanocytes [131]. Intriguingly, CK2α staining and transcript levels are elevated in melanomas with NRAS Q61 mutations but not in melanomas with NRAS G12 mutations [132]. These suggest a potential for the use of CK2 inhibitors to treat this cancer. We reviewed the use of CK2 inhibitors in combination with current therapies for melanoma in [16].
Testicular cancer
90% of testicular cancer derive from germ cells, and the remaining 10% derive from supporting cells (Leydig cells, Sertoli cells). Germ cell-related testicular cancer can be subdivided equally into seminomas and non-seminomas. Non-seminoma testicular cancer is subdivided into teratomas, embryonal carcinomas (3–4%), mixed germ cell tumors, and yolk sac tumors. Typically, non-seminoma tumors grow faster, present with an earlier diagnosis age, and have a lower relative 5-year survival rate. In contrast, seminomas have a higher survival rate largely due to the fact that they are highly sensitive to radiation therapy and chemotherapy. Germ cell tumors can also occur outside of the gonads due to defects in embryonic development. There are many risk factors for testicular cancer, including cryptorchidism, Turner syndrome, androgen insensitivity syndrome, Klinefelter’s syndrome, low fertility, and family history. Treatment options for testicular cancer include surgery, radiation therapy, and chemotherapy.
CK2 in testicular cancer
Oncomine analysis revealed significant downregulation of all three CK2 genes and CK2αP in both seminomatous and non-seminomatous testicular cancer (Table 17). CK2αP was also downregulated in testicular seminoma. There was no data for CK2αP in any of the other types listed in Table 17.
Table 17. Analysis of changes in CK2 gene expression in testicular cancer.
P-values, fold change, rank and datasets are shown. Not Otherwise Specified was a category in Oncomine however, in the case of Seminoma and teratoma they are the same as the category above.
| Gene | p-value | Fold Change | Rank (Top %) | Dataset | #Samples | Reference |
|---|---|---|---|---|---|---|
| Testicular Seminoma | ||||||
| CK2α | 0.003 | -1.645 | 22% | Sperger Others | 74 | [142] |
| CK2α’ | 0.009 | -1.640 | 27% | Sperger Others | 74 | [142] |
| CK2β | 0.012 | 2.607 | 9% | Skotheim Testis | 30 | [143] |
| CK2αP | 0.003 | -1.645 | 22% | Sperger Others | 74 | [142] |
| Seminoma, Not Otherwise Specified | ||||||
| CK2α | 1.21 10−5 | -2.224 | 13% | Korkola Seminoma | 107 | [144] |
| CK2α’ | 8.56 10−11 | -8.257 | 1% | Korkola Seminoma | 107 | [144] |
| CK2β | 2.11 10−6 | -2.180 | 10% | Korkola Seminoma | 107 | [144] |
| Testicular Teratoma | ||||||
| CK2α’ | 0.006 | -4.093 | 7% | Skotheim Testis | 30 | [143] |
| CK2β | 0.009 | -2.471 | 9% | Skotheim Testis | 30 | [143] |
| Teratoma, Not Otherwise Specified | ||||||
| CK2α | 3.56 10−9 | -3.805 | 4% | Korkola Seminoma | 107 | [144] |
| CK2α’ | 5.12 10−12 | -9.860 | 1% | Korkola Seminoma | 107 | [144] |
| CK2β | 3.93 10−8 | -2.791 | 6% | Korkola Seminoma | 107 | [144] |
| Embryonal Carcinoma, Not Otherwise Specified | ||||||
| CK2α | 5.98 10−8 | -2.666 | 6% | Korkola Seminoma | 107 | [144] |
| CK2α’ | 6.42 10−12 | -9.596 | 1% | Korkola Seminoma | 107 | [144] |
| CK2β | 1.59 10−6 | -2.349 | 11% | Korkola Seminoma | 107 | [144] |
| Mixed Germ Cell Tumor, Not Otherwise Specified | ||||||
| CK2α | 1.25 10−7 | -2.302 | 10% | Korkola Seminoma | 107 | [144] |
| CK2α’ | 4.33 10−12 | -7.593 | 3% | Korkola Seminoma | 107 | [144] |
| CK2β | 6.47 10−7 | -2.377 | 12% | Korkola Seminoma | 107 | [144] |
| Yolk Sac Tumor, Not Otherwise Specified | ||||||
| CK2α | 0.014 | -1.524 | 30% | Korkola Seminoma | 107 | [144] |
| CK2α’ | 1.21 10−9 | -6.599 | 1% | Korkola Seminoma | 107 | [144] |
| CK2β | 9.08 10−5 | -2.123 | 15% | Korkola Seminoma | 107 | [144] |
To our knowledge, there is no data on the CK2 protein levels in testicular cancer, or on the mechanism by which CK2 dysregulation could be affecting testicular cancer. However, we have knowledge on CK2 levels in normal testicular tissue and in reproductive organ development. Specifically, CK2α’ and CK2β transcript, protein levels and CK2 activity are highest in mouse testicles compared to other mouse tissues [5, 136, 137]. In mice and Xenopus laevis, CK2 transcript levels are high in germ cells (reviewed in [137]). Importantly, CK2α’ is required for male gametogenesis, as knockout of CK2α’ mice are infertile and have oligospermia and globozoospermia (i.e. spermatozoa having round heads) [5]. In addition, the natural CK2 inhibitor Emodin causes hypospermatogenesis and apoptosis of mouse germ cells [138]. These phenotypes could be due in part to germ cell chromatin breakdown, a risk factor for testicular cancer [139]. Potentially, CK2 could be acting on two chromatin remodeling proteins, CK2 Target protein 2 (CKT2) and testis-specific protein, Y-encoded (TSPY) [140, 141]. Since CK2α’ is required for sperm differentiation in mice, downregulation of CK2α’ in testicular cancer could delay and/or prevent germ cell differentiation, and possibly divert sperm development towards tumor formation.
Conclusion
Our goal was to determine the extent to which CK2 genes could be significantly up- or down-regulated in cancers we did not study in our previous publication [9], and to determine whether the CK2 gene expression levels in these cancers correlated with overall patient survival. Here we show that there is dysregulation of CK2 transcripts in a number of these cancer types that was not reported previously, and that over- and under- expression of CK2 transcripts was cancer-type specific. In addition, the prognostic value of CK2 genes was also cancer-type specific as both over- and under-expression correlated with poor survival. In addition, we have compared our data to published results on CK2 gene and protein expression.
In Table 18, we summarized the analysis from Oncomine and of the literature. In many cases these results are robust, as several studies showed the same up- or down-regulation for the CK2 transcript (e.g. up to 4 studies for CK2α and CK2β increase in hepatocellular carcinoma) (Table 18). The fact that independent studies showed similar data gives strength to the conclusions reached. As we have previously found in other cancers [9], we found cancers with significant upregulation of all three CK2 genes, such as bladder cancers, and tumors with mixed up- and down- regulation of CK2 transcripts. In solid tumors with mixed up- and down-regulation, CK2α’ was almost always downregulated (e.g. sarcomas and melanoma), as it was in breast, ovarian and pancreatic cancers [9]. In addition, we found a third pattern of dysregulation of CK2 genes: significant downregulation of all three CK2 transcripts. Finding all three CK2 genes downregulated (e.g. Barrett’s esophagus and testicular cancer) was unexpected, particularly since CK2α/α’ downregulation leads to decreased proliferation (reviewed in [16, 22, 23]). This is a novel finding in CK2α/α’ biology, and requires further study to determine whether CK2α/α’ protein levels are equally decreased and, if so, which hallmark of cancer [145] could be affected by CK2α’ protein decrease. In contrast to the little data on CK2α’ downregulation, CK2β downregulation is already linked to cancer progression. In particular, to increased epithelial to mesenchymal transition [146]. Further investigation as to whether CK2 gene downregulation contributes to cancer development in these cancer types, including animal model studies, will address the relevance of this finding. One possibility in testicular cancer is that aberrant expression of CK2α’ and CK2β could cause a dysfunction in gametogenesis leading to subfertility; as subfertility has been linked to increased risk of developing testicular cancer [139].
Table 18. Expression of CK2 transcript and proteins in cancers.
This table summarizes some of our Oncomine data on upregulation (red arrow, ) or downregulation (dark blue arrow, ) of CK2 transcripts. Multiple arrows (e.g.: ) indicate the number of studies showing similar conclusions. We also included published data on upregulation (orange arrow, ) or downregulation (light blue arrow, ) of CK2 transcripts and protein levels. Each arrow indicates an independent study. (n.s.) Not significant; (-) No data; (ND) Not determined (*) Diagnostic potential; Abbreviations as defined in the text.
| Cancers | CK2 Transcripts | CK2 Proteins | ||||||
|---|---|---|---|---|---|---|---|---|
| α | α’ | β | αP | α | α’ | β | CK2 activity | |
| Bladder | ||||||||
| Superficial Bladder Carcinoma | n.s. | |||||||
| Invasive Bladder Carcinoma | n.s. | *[45] * [46] | ||||||
| Bladder Carcinoma (NOS) | [44] | ND | ND | ND | *[44] | |||
| CNS cancers | ||||||||
| Astrocytoma | [51] [50]/ [50] | n.s. [51] | [51] | - | [50] [50] | [51] | ||
| Glioblastoma (Astrocytoma Grade IV) | [49] [38] [51] [50]/ [38] [49] [50] | [51]/ | [51]/ | n.s. | *[50][38][55] [Dubois, 2016 #532][54]]/ [38] | [51] | ||
| Anaplastic Oligodendroglioma | n.s. [51] | [51] | [51] | n.s. | [51] | |||
| Anaplastic Oligoastrocytoma | n.s. | |||||||
| Cervical | ||||||||
| Cervical | - | |||||||
| Esophageal | ||||||||
| Barrett’s Esophagus | ||||||||
| Esophageal Adenocarcinoma | / | |||||||
| Esophageal Carcinoma | ND | ND | * [66]/[66] | ND | *[66]/[66] | |||
| Gastric | ||||||||
| Gastric Intestinal Type Adenocarcinoma | / | *[29] | *[30] | |||||
| Diffuse Gastric Adenocarcinoma | n.s. | n.s. | *[29] | *[30] | ||||
| Gastric Mixed Adenocarcinoma | n.s. | n.s. | n.s. | *[29] | ||||
| Head and Neck | ||||||||
| Floor of the Mouth Carcinoma | - | |||||||
| Oral Cavity Squamous Cell Carcinoma | n.s. | n.s. | - | |||||
| Nasopharyngeal Carcinoma | n.s. | n.s. | - | |||||
| Oropharyngeal Carcinoma | n.s. | n.s. | - | |||||
| Tongue Carcinoma | - | |||||||
| Tonsillar Carcinoma | n.s. | n.s. | - | |||||
| H&N squamous cell carcinoma | [73] | *[73] | *[73] | - | [74] [33] | [74] [33] | [74] | * [31] [32] [33] |
| Kidney | ||||||||
| Chromophobe Renal Cell Carcinoma | n.s. | - | ||||||
| Clear Cell Renal Cell Carcinoma | *[39] *[36][79] | n.s. [39] *[36]/ [79] | [36][79] | - | [79]* [36] n.s. [81] | [79]; | [79] [81] | [79] [80] [36] |
| Clear Cell Sarcoma of the Kidney | n.s. | n.s. | - | |||||
| Papillary Renal Cell Carcinoma | n.s. | n.s. | - | |||||
| Renal Oncocytoma | - | |||||||
| Renal Pelvis Urothelial Carcinoma | n.s. | - | ||||||
| Renal Wilms Tumor | n.s. | - | ||||||
| Leukemia | ||||||||
| Pro-B ALL | n.s. | n.s. | - | |||||
| ALL (B-Cell) | / | [89] | [89] | [89] [150] | ||||
| ALL (T-Cell) | / | [90] | [90] | [90] | ||||
| AML | / | [95, 96][95, 96] | [95, 96][95, 96] | |||||
| CLL | / | / | - | [102] | [102] | [102] | ||
| HCL | n.s. | - | ||||||
| NHL | ||||||||
| FL (B-cell, Low Grade) | n.s. | [106] | [106] | |||||
| Mantle (B-cell, Low Grade) | n.s. | - | [107] | [107] | ||||
| DLBCL-NOS (B-cell, High Grade) | n.s. | n.s. | n.s. | [106] | [106] | |||
| DLBCL-ABC (B-cell, High Grade) | n.s. | - | ||||||
| DLBCL-GCB (B-cell, High Grade) | - | [106] | [106] | |||||
| Burkitt’s Lymphoma (B-cell, High Grade) | n.s. | - | [106] | [106] | ||||
| PEL (B-cell, High Grade) | n.s. | n.s. | - | |||||
| ALCL (T-cell, High Grade) | - | |||||||
| AITL (T-cell, High Grade) | - | |||||||
| PTCL-U (T-cell NHL High Grade) | n.s. | - | ||||||
| ATL or ATLL | n.s. | n.s. | - | |||||
| T/HRBCL | n.s. | n.s. | - | |||||
| MGUS | / | - | n.s. [107] | n.s. [107] | ||||
| Smoldering Myeloma | - | |||||||
| Plasma Cell Leukemia | n.s. | - | ||||||
| Multiple Myeloma | n.s. | n.s. | n.s. | - | [107] [116] | n.s. [116] | [107] [116] | [116] |
| Liver | ||||||||
| Hepatocellular Carcinoma | *[40] [117] | n.s. | n.s. | [40] *[117] | ||||
| Mesothelioma | ||||||||
| Malignant Pleural Mesothelioma | [122] | n.s. | - | [122] | ||||
| Parathyroid | ||||||||
| Parathyroid hyperplasia | n.s. | - | ||||||
| Non-Familial Multiple Gland Neoplasia | n.s. | n.s. | - | |||||
| Parathyroid Gland Adenoma | n.s. | n.s. | - | |||||
| Thyroid | ||||||||
| Thyroid Carcinoma | [151] | n.s. | n.s. | - | [151] | |||
| Sarcoma | ||||||||
| Dedifferentiated Liposarcoma | n.s. | - | ||||||
| Fibrosarcoma | n.s. | - | ||||||
| Malignant fibrous histiocytoma | n.s. | n.s. | - | |||||
| Leiomyosarcoma | n.s. | / | n.s. | |||||
| Myxofibrosarcoma | n.s. | n.s. | ||||||
| Pleomorphic Liposarcoma | - | |||||||
| Synovial Sarcoma | n.s. | n.s. | ||||||
| Skin cancers | ||||||||
| Benign Melanocytic Skin Nevus | - | |||||||
| Basal Cell Carcinoma | n.s. | n.s. | - | |||||
| Squamous Cell Carcinoma | n.s. | n.s. | - | |||||
| Melanoma | [131] [132] | - | [131] [132] | . | ||||
| Testicular | ||||||||
| Testicular Seminoma | ||||||||
| Seminoma NOS | ||||||||
| Testicular Teratoma | n.s. | - | ||||||
| Teratoma NOS | - | |||||||
| Embryonal Carcinoma NOS | - | |||||||
| Mixed Germ Cell Tumor NOS | - | |||||||
| Yolk Sac Tumor NOS | - | |||||||
The Oncomine analysis led to additional insights. CK2αP was upregulated or downregulated following the prevalent pattern of up- or down-regulation of the other CK2 transcripts. Similarly, CK2αP follows the same pattern in lung, breast and colorectal cancers [9]. The importance of CK2αP deregulation in cancer is to be determined. We found altered expression of CK2 transcripts already in benign lesions (e.g. parathyroid overgrowth and benign Melanocytic Skin Nevus), and also in Barrett’s esophagus (that increases the risk of developing esophageal cancer). This is similar to what is previously found in benign colon adenomas [9]. This suggests a role in cancer initiation in these cancer types. In NHL, low grade (slow-growing) lymphomas (e.g. FL) had downregulation of CK2 transcripts while in high grade NHL (more aggressive) there was upregulation of CK2 transcripts. This suggest differential transactivation of the CK2 genes or differential stability of the transcripts may be playing a role in high grade NHL.
In Table 18 we also summarized the literature on CK2 protein levels in diverse cancers. In general, the increases in CK2 transcripts paralleled increases in CK2 protein expression levels, nuclear localization, and/or activity; with the exception of B-cell ALL, AML, CLL and Follicular lymphoma (Table 18). Therefore, we cannot directly infer the levels of CK2 proteins in human cancer from studies of transcript levels, particularly in blood cancers. These data support the notion that there are multiple layers of regulation of CK2 expression that need to be addressed in future studies.
A number of studies show that higher CK2 protein levels, nuclear localization, and/or activity have diagnostic value, that is, correlate with clinicopathological factors (mostly with higher tumor grade and tumor stage; and sometimes with metastasis and invasion) (Table 18, asterisks). Only one exception, were increase CK2α’ transcript correlated with low grade clear cell renal cell carcinoma Rabjerg, Guerra et al. 2017). The significance of this finding needs to be addressed. Interestingly, the diagnostic value of CK2 proteins is linked mostly to higher overall staining, nuclear staining or CK2 activity. However, in the case of astrocytomas, it is cytoplasmic CK2 staining that has a diagnostic value [50]. Importantly, in some cancers, increases in both CK2 transcript and protein expression have diagnostic value (e.g. Clear Cell Renal Cell Carcinoma [36]) (Table 18, asterisks). This suggests that both CK2 protein and transcript levels could be used as diagnostic biomarkers. Future in depth analyses will determine the extent to which CK2 transcript and protein levels correlate and have diagnostic value in these cancers. Ultimately, changes in the overall and relative levels of CK2 proteins and of the composition of the CK2 heterotetramer will affect diverse cellular functions (e.g. proliferation, epithelial to mesenchymal transition). These effects could be mediated through changes in the activation of CK2-dependent signaling pathways or expression of downstream genes as reviewed in [16].
In Table 19, we summarized the value of CK2 as a prognostic marker. In most cancers, overexpression of one or two CK2 gene transcripts led to lower survival (e.g. gastric cancer, sarcoma, head and neck and liver cancer) (Table 19). In addition, overexpression of the pseudogene CK2αP led to lower survival in cervical cancer and mesothelioma. However, similar to lung adenocarcinoma [9], overexpression of CK2αP in renal clear cell carcinoma led to increased survival. The significance of this finding is unclear, as we know little about the function of CK2αP. Importantly, there was a tight parallel between the prognostic value of CK2 transcripts and proteins, furthering the notion that abnormal levels of CK2 can be a useful prognostic marker (Table 18). Intriguingly, the levels of CK2α’ were not statistically significant in the Oncomine analysis while they were a prognostic marker in liver cancer. This lack of significance in Oncomine analysis may be due to a high standard deviation in CK2α’ transcript levels in liver cancer samples. This result indicates that the prognostic value of a CK2 transcript (and perhaps of CK2 protein) needs to be investigated independently of whether the average expression level is significantly different or not from control samples.
Table 19. Prognostic value of CK2 transcript and protein in cancer.
High CK2 transcript correlate with higher () or lower () patient survival. We also included published data on correlation of high CK2 transcripts and protein (indicated as (p)) levels with lower survival (light blue arrow, ). Each arrow indicates an independent study. (#) = increased CK2 activity correlates with decreased patient survival [31].
| High levels of Expression of: | CK2α | CK2α’ | CK2β | CK2αP |
|---|---|---|---|---|
| Glioblastoma | / [38] | n.s | n.s | n.s |
| Cervical Cancer | n.s | n.s | n.s | |
| Gastric Cancer | [29] | n.s | [30] | n.s |
| Head and Neck Cancer (#) | n.s | n.s | ||
| Renal Clear Cell Carcinoma | [36, 39] | n.s | n.s | |
| AML | [96] | - | - | - |
| Liver Cancer | [40] [117] | n.s | n.s | |
| Mesothelioma | n.s | n.s | n.s | |
| Sarcoma | n.s | n.s | n.s |
In this study, as we did for [9], we have followed an unbiased approach to analyze and report all the data available to date in the databases. This has resulted in unique studies which show that both CK2 over- and under- expression have significance in cancer. There are intrinsic limitations to the database analyses. For example, not all cancer types are included in the databases searched, and, in some cases, the number of patients studied is not large enough to find significant differences in transcript levels or in survival rates. There are also inherent factors (algorithms used in microarray analysis, reference genes used for normalization, the age of the samples, preservation, etc) which could limit the outcome of the analyses or contribute to the discrepancies between studies. Those discrepancies should be resolved when additional independent studies are performed. A broader limitation to our study is that Oncomine does not include information on CK2 gene mutations which might also affect overall patient survival.
There is a need for cancer biomarkers (diagnostic, prognostic and predictive), and for therapeutic targets and treatments to address cancer incidence and mortality in the world (Table 20). Our study advances the notion that CK2 transcript expression is a potential diagnostic and prognostic biomarker for specific types of cancer. However, a number of validation steps are needed before CK2 gene expression has an impact in clinical practice [147–149]. This includes whether CK2 transcripts and proteins could be used alone or in combination as diagnostic and prognostic factors. In addition, future studies could begin to address the predictive value of CK2 to select those patients who are more likely to benefit from a particular cancer treatment.
Table 20. Cancer statistics overview.
2016 USA statistics of the individual cancer types are analyzed and rank (USA and worldwide), incidence, mortality, 5-year survival rate (%), and estimated expenditure in billions (2014 data) are included. N/A indicates no data was available.
| Rank (USA) |
Rank (worldwide) |
Incidence | Mortality | 5-year survival rate (%) | Estimated cost (billion) | |
|---|---|---|---|---|---|---|
| Bladder | 5 | 9 | 76,960 (4.6%) | 16,390 (2.8%) | 77.5 | 4.1 |
| CNS | 16 | 17 | 23,770 (1.4%) | 16,050 (2.7%) | 33.8 | 4.9 |
| Cervical | 21 | 7 | 12,990 (0.8%) | 4,120 (0.7%) | 67.5 | 1.3 |
| Esophageal | 18 | 8 | 16,910 (1.0%) | 15,690 (2.6%) | 18.4 | 1.6 |
| Gastric | 15 | 5 | 26,370 (1.6%) | 10,730 (1.8%) | 30.4 | 1.8 |
| Head and neck | N/A | 15 | 48,330 (2.9%) | 9,570 (1.6%) | 64.0 | N/A |
| Kidney | 9 | 12 | 62,700 (3.7%) | 14,240 (2.4%) | 73.7 | 4.8 |
| Leukemia | 11 | 11 | 60,140 (3.6%) | 24,400 (4.1%) | 59.7 | 5.9 |
| Non-Hodgkin Lymphoma | 7 | 10 | 72,580 (4.3%) | 20,150 (3.4%) | 70.7 | 13.4 |
| Myeloma | 14 | 23 | 30,330 (1.8%) | 12,650 (2.1%) | 48.5 | N/A |
| Liver | 13 | 6 | 39,230 (2.3%) | 27,170 (4.6%) | 17.5 | N/A |
| Mesothelioma | N/A | N/A | 3,000 (0.02%) | N/A | N/A | N/A |
| Parathyroid | N/A | N/A | <100 | N/A | 89 | N/A |
| Thyroid | N/A | N/A | 56,870 (3.4%) | 2,010 (0.3%) | 98.2 | N/A |
| Sarcoma | N/A | 27 | 15,000 (1.0%) | 6,200 (1.0%) | 50 | N/A |
| Skin (Melanoma) | 6 | 19 | 76,380 (4.5%) | 10,130 (1.7%) | 91.5 | 2.9 |
| Testicular | 25 | 26 | 8,720 (0.5%) | 380 (0.1%) | 95.4 | N/A |
Acknowledgments
We thank the Dominguez lab for discussions. We thank Janice Weinberg and Na Wang, Biostatistics Consultants in the Department of Medicine, for their help with statistical analysis. We thank Kevan Hartshorn, Anurag Singh, Barbara Guerra and Sam Thiangalingam for their comments to the manuscript. This work was partially supported with funding from the National Institutes of General Medical Sciences (NIGMS); 1R01GM098367. M.C. was supported by a Barbur Kalique Scholarship through the Boston University School of Medicine Medical Student Summer Research Program (MSSRP); and M.L. was supported by a fellowship from the Boston University Undergraduate Research Opportunities Program (UROP). Data from WHO, NCI, CDC, ACS, Oncomine and Kaplan Meier Plotter are cited as per their requirements.
Data Availability
All relevant data are within the paper.
Funding Statement
This project was supported in part with funding from the National Institutes of General Medical Sciences; 1R01GM098367 [http://www.nigms.nih.gov/]. M.C. was supported by a Barbur Kalique Scholarship through the Boston University School of Medicine Medical Student Summer Research Program (MSSRP); and M.L. was supported by a fellowship from the Boston University Undergraduate Research Opportunities Program (UROP). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Litchfield DW, Bosc DG, Canton DA, Saulnier RB, Vilk G, Zhang C. Functional specialization of CK2 isoforms and characterization of isoform-specific binding partners. Mol Cell Biochem. 2001;227(1–2):21–9. Epub 2002/02/06. . [PubMed] [Google Scholar]
- 2.Maridor G, Park W, Krek W, Nigg EA. Casein kinase II. cDNA sequences, developmental expression, and tissue distribution of mRNAs for alpha, alpha', and beta subunits of the chicken enzyme. J Biol Chem. 1991;266(4):2362–8. Epub 1991/02/05. . [PubMed] [Google Scholar]
- 3.Lozeman FJ, Litchfield DW, Piening C, Takio K, Walsh KA, Krebs EG. Isolation and characterization of human cDNA clones encoding the alpha and the alpha' subunits of casein kinase II. Biochemistry. 1990;29(36):8436–47. Epub 1990/09/11. . [DOI] [PubMed] [Google Scholar]
- 4.Litchfield DW, Lozeman FJ, Piening C, Sommercorn J, Takio K, Walsh KA, et al. Subunit structure of casein kinase II from bovine testis. Demonstration that the alpha and alpha' subunits are distinct polypeptides. J Biol Chem. 1990;265(13):7638–44. Epub 1990/05/05. . [PubMed] [Google Scholar]
- 5.Xu X, Toselli PA, Russell LD, Seldin DC. Globozoospermia in mice lacking the casein kinase II alpha' catalytic subunit. Nature genetics. 1999;23(1):118–21. Epub 1999/09/02. doi: 10.1038/12729 . [DOI] [PubMed] [Google Scholar]
- 6.Lou DY, Dominguez I, Toselli P, Landesman-Bollag E, O'Brien C, Seldin DC. The alpha catalytic subunit of protein kinase CK2 is required for mouse embryonic development. Mol Cell Biol. 2008;28(1):131–9. doi: 10.1128/MCB.01119-07 ; PubMed Central PMCID: PMC2223292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bibby AC, Litchfield DW. The multiple personalities of the regulatory subunit of protein kinase CK2: CK2 dependent and CK2 independent roles reveal a secret identity for CK2beta. International journal of biological sciences. 2005;1(2):67–79. Epub 2005/06/14. ; PubMed Central PMCID: PMC1142214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wirkner U, Voss H, Lichter P, Weitz S, Ansorge W, Pyerin W. Human casein kinase II subunit alpha: sequence of a processed (pseudo)gene and its localization on chromosome 11. Biochim Biophys Acta. 1992;1131(2):220–2. . [DOI] [PubMed] [Google Scholar]
- 9.Ortega CE, Seidner Y, Dominguez I. Mining CK2 in cancer. PLoS One. 2014;9(12):e115609 doi: 10.1371/journal.pone.0115609 ; PubMed Central PMCID: PMC4277308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Trembley JH, Wu J, Unger GM, Kren BT, Ahmed K. CK2 Suppression of Apoptosis and Its Implication in Cancer Biology and Therapy. PInna LA, editor. Ames, Iowa: Wiley-Blackwell; 2013. 219–343 p. [Google Scholar]
- 11.Ruzzene M, Pinna LA. Addiction to protein kinase CK2: a common denominator of diverse cancer cells? Biochimica et biophysica acta. 2010;1804(3):499–504. Epub 2009/08/12. doi: 10.1016/j.bbapap.2009.07.018 . [DOI] [PubMed] [Google Scholar]
- 12.Seldin DC, Landesman-Bollag E, Farago M, Currier N, Lou D, Dominguez I. CK2 as a positive regulator of Wnt signalling and tumourigenesis. Mol Cell Biochem. 2005;274(1–2):63–7. . [DOI] [PubMed] [Google Scholar]
- 13.Dominguez I, Sonenshein GE, Seldin DC. Protein kinase CK2 in health and disease: CK2 and its role in Wnt and NF-kappaB signaling: linking development and cancer. Cellular and molecular life sciences: CMLS. 2009;66(11–12):1850–7. Epub 2009/04/24. doi: 10.1007/s00018-009-9153-z ; PubMed Central PMCID: PMC3905806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Guerra B, Issinger OG. Protein kinase CK2 in human diseases. Curr Med Chem. 2008;15(19):1870–86. Epub 2008/08/12. . [DOI] [PubMed] [Google Scholar]
- 15.Tawfic S, Yu S, Wang H, Faust R, Davis A, Ahmed K. Protein kinase CK2 signal in neoplasia. Histology and histopathology. 2001;16(2):573–82. Epub 2001/05/03. . [DOI] [PubMed] [Google Scholar]
- 16.Chua MM, Ortega CE, Sheikh A, Lee M, Abdul-Rassoul H, Hartshorn KL, et al. CK2 in Cancer: Cellular and Biochemical Mechanisms and Potential Therapeutic Target. Pharmaceuticals (Basel). 2017;10(1). Epub 2017/01/31. doi: 10.3390/ph10010018 ; PubMed Central PMCID: PMCPMC5374422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Seldin DC. New models of lymphoma in transgenic mice. Current opinion in immunology. 1995;7(5):665–73. Epub 1995/10/01. . [DOI] [PubMed] [Google Scholar]
- 18.Channavajhala P, Seldin DC. Functional interaction of protein kinase CK2 and c-Myc in lymphomagenesis. Oncogene. 2002;21(34):5280–8. Epub 2002/08/01. doi: 10.1038/sj.onc.1205640 . [DOI] [PubMed] [Google Scholar]
- 19.Seldin DC, Leder P. Casein kinase II alpha transgene-induced murine lymphoma: relation to theileriosis in cattle. Science. 1995;267(5199):894–7. . [DOI] [PubMed] [Google Scholar]
- 20.Landesman-Bollag E, Romieu-Mourez R, Song DH, Sonenshein GE, Cardiff RD, Seldin DC. Protein kinase CK2 in mammary gland tumorigenesis. Oncogene. 2001;20(25):3247–57. Epub 2001/06/26. doi: 10.1038/sj.onc.1204411 . [DOI] [PubMed] [Google Scholar]
- 21.Litchfield DW. Protein kinase CK2: structure, regulation and role in cellular decisions of life and death. The Biochemical journal. 2003;369(Pt 1):1–15. Epub 2002/10/25. doi: 10.1042/BJ20021469 ; PubMed Central PMCID: PMC1223072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pinna LA, Meggio F. Protein kinase CK2 ("casein kinase-2") and its implication in cell division and proliferation. Progress in cell cycle research. 1997;3:77–97. Epub 1997/01/01. . [DOI] [PubMed] [Google Scholar]
- 23.Ahmed K, Davis AT, Wang H, Faust RA, Yu S, Tawfic S. Significance of protein kinase CK2 nuclear signaling in neoplasia. J Cell Biochem Suppl. 2000;Suppl 35:130–5. . [DOI] [PubMed] [Google Scholar]
- 24.Ahmad KA, Wang G, Unger G, Slaton J, Ahmed K. Protein kinase CK2—a key suppressor of apoptosis. Advances in enzyme regulation. 2008;48:179–87. Epub 2008/05/22. doi: 10.1016/j.advenzreg.2008.04.002 ; PubMed Central PMCID: PMC2593134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ahmed K, Gerber DA, Cochet C. Joining the cell survival squad: an emerging role for protein kinase CK2. Trends in cell biology. 2002;12(5):226–30. Epub 2002/06/14. . [DOI] [PubMed] [Google Scholar]
- 26.Canton DA, Litchfield DW. The shape of things to come: an emerging role for protein kinase CK2 in the regulation of cell morphology and the cytoskeleton. Cellular signalling. 2006;18(3):267–75. Epub 2005/08/30. doi: 10.1016/j.cellsig.2005.07.008 . [DOI] [PubMed] [Google Scholar]
- 27.Filhol OD, A; Cochet C. Role of CK2 in the Control of Cell Plasticity in Breast Carcinoma Progression. P LA, editor. AMes, Iowa: Wiley-Blackwell; 2013. [Google Scholar]
- 28.Kramerov AA, Saghizadeh M, Caballero S, Shaw LC, Li Calzi S, Bretner M, et al. Inhibition of protein kinase CK2 suppresses angiogenesis and hematopoietic stem cell recruitment to retinal neovascularization sites. Mol Cell Biochem. 2008;316(1–2):177–86. Epub 2008/07/10. doi: 10.1007/s11010-008-9831-4 ; PubMed Central PMCID: PMC2913688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bae JS, Park SH, Kim KM, Kwon KS, Kim CY, Lee HK, et al. CK2alpha phosphorylates DBC1 and is involved in the progression of gastric carcinoma and predicts poor survival of gastric carcinoma patients. International journal of cancer Journal international du cancer. 2015;136(4):797–809. Epub 2014/06/26. doi: 10.1002/ijc.29043 . [DOI] [PubMed] [Google Scholar]
- 30.Lin KY, Fang CL, Chen Y, Li CF, Chen SH, Kuo CY, et al. Overexpression of nuclear protein kinase CK2 Beta subunit and prognosis in human gastric carcinoma. Annals of surgical oncology. 2010;17(6):1695–702. Epub 2010/01/21. doi: 10.1245/s10434-010-0911-9 . [DOI] [PubMed] [Google Scholar]
- 31.Gapany M, Faust RA, Tawfic S, Davis A, Adams GL, Ahmed K. Association of elevated protein kinase CK2 activity with aggressive behavior of squamous cell carcinoma of the head and neck. Molecular medicine. 1995;1(6):659–66. Epub 1995/09/01. ; PubMed Central PMCID: PMC2229971. [PMC free article] [PubMed] [Google Scholar]
- 32.Faust RA, Gapany M, Tristani P, Davis A, Adams GL, Ahmed K. Elevated protein kinase CK2 activity in chromatin of head and neck tumors: association with malignant transformation. Cancer letters. 1996;101(1):31–5. Epub 1996/03/19. . [DOI] [PubMed] [Google Scholar]
- 33.Faust RA, Niehans G, Gapany M, Hoistad D, Knapp D, Cherwitz D, et al. Subcellular immunolocalization of protein kinase CK2 in normal and carcinoma cells. Int J Biochem Cell Biol. 1999;31(9):941–9. Epub 1999/10/26. . [DOI] [PubMed] [Google Scholar]
- 34.Cozza G, Pinna LA. Casein kinases as potential therapeutic targets. Expert Opin Ther Targets. 2016;20(3):319–40. Epub 2015/11/14. doi: 10.1517/14728222.2016.1091883 . [DOI] [PubMed] [Google Scholar]
- 35.Trembley JH, Chen Z, Unger G, Slaton J, Kren BT, Van Waes C, et al. Emergence of protein kinase CK2 as a key target in cancer therapy. Biofactors. 2010;36(3):187–95. Epub 2010/06/10. doi: 10.1002/biof.96 ; PubMed Central PMCID: PMC2916697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rabjerg M, Guerra B, Olivan-Viguera A, Mikkelsen ML, Kohler R, Issinger OG, et al. Nuclear localization of the CK2alpha-subunit correlates with poor prognosis in clear cell renal cell carcinoma. Oncotarget. 2017;8(1):1613–27. Epub 2016/12/03. doi: 10.18632/oncotarget.13693 ; PubMed Central PMCID: PMC5352082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.O-charoenrat P, Rusch V, Talbot SG, Sarkaria I, Viale A, Socci N, et al. Casein kinase II alpha subunit and C1-inhibitor are independent predictors of outcome in patients with squamous cell carcinoma of the lung. Clinical cancer research: an official journal of the American Association for Cancer Research. 2004;10(17):5792–803. Epub 2004/09/10. doi: 10.1158/1078-0432.CCR-03-0317 . [DOI] [PubMed] [Google Scholar]
- 38.Nitta RT, Gholamin S, Feroze AH, Agarwal M, Cheshier SH, Mitra SS, et al. Casein kinase 2alpha regulates glioblastoma brain tumor-initiating cell growth through the beta-catenin pathway. Oncogene. 2015;34(28):3688–99. Epub 2014/09/23. doi: 10.1038/onc.2014.299 ; PubMed Central PMCID: PMC4369469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rabjerg M, Bjerregaard H, Halekoh U, Jensen BL, Walter S, Marcussen N. Molecular characterization of clear cell renal cell carcinoma identifies CSNK2A1, SPP1 and DEFB1 as promising novel prognostic markers. Apmis. 2016;124(5):372–83. Epub 2016/02/16. doi: 10.1111/apm.12519 . [DOI] [PubMed] [Google Scholar]
- 40.Kim HS, Chang YG, Bae HJ, Eun JW, Shen Q, Park SJ, et al. Oncogenic potential of CK2alpha and its regulatory role in EGF-induced HDAC2 expression in human liver cancer. The FEBS journal. 2014;281(3):851–61. Epub 2014/03/13. . [DOI] [PubMed] [Google Scholar]
- 41.Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia. 2004;6(1):1–6. ; PubMed Central PMCID: PMCPMC1635162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gyorffy B, Lanczky A, Szallasi Z. Implementing an online tool for genome-wide validation of survival-associated biomarkers in ovarian-cancer using microarray data from 1287 patients. Endocr Relat Cancer. 2012;19(2):197–208. doi: 10.1530/ERC-11-0329 . [DOI] [PubMed] [Google Scholar]
- 43.Szasz AM, Lanczky A, Nagy A, Forster S, Hark K, Green JE, et al. Cross-validation of survival associated biomarkers in gastric cancer using transcriptomic data of 1,065 patients. Oncotarget. 2016;7(31):49322–33. Epub 2016/07/08. doi: 10.18632/oncotarget.10337 ; PubMed Central PMCID: PMCPMC5226511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang X, Yang X, Yang C, Li P, Yuan W, Deng X, et al. Targeting protein kinase CK2 suppresses bladder cancer cell survival via the glucose metabolic pathway. Oncotarget. 2016;7(52):87361–72. Epub 2016/11/27. doi: 10.18632/oncotarget.13571 ; PubMed Central PMCID: PMCPMC5349994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shimada K, Anai S, Marco DA, Fujimoto K, Konishi N. Cyclooxygenase 2-dependent and independent activation of Akt through casein kinase 2alpha contributes to human bladder cancer cell survival. BMC urology. 2011;11:8 Epub 2011/05/20. doi: 10.1186/1471-2490-11-8 ; PubMed Central PMCID: PMC3111585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jeppesen DK, Nawrocki A, Jensen SG, Thorsen K, Whitehead B, Howard KA, et al. Quantitative proteomics of fractionated membrane and lumen exosome proteins from isogenic metastatic and nonmetastatic bladder cancer cells reveal differential expression of EMT factors. Proteomics. 2014;14(6):699–712. Epub 2014/01/01. doi: 10.1002/pmic.201300452 . [DOI] [PubMed] [Google Scholar]
- 47.Sanchez-Carbayo M, Socci ND, Lozano J, Saint F, Cordon-Cardo C. Defining molecular profiles of poor outcome in patients with invasive bladder cancer using oligonucleotide microarrays. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2006;24(5):778–89. Epub 2006/01/25. doi: 10.1200/JCO.2005.03.2375 . [DOI] [PubMed] [Google Scholar]
- 48.Dyrskjot L, Kruhoffer M, Thykjaer T, Marcussen N, Jensen JL, Moller K, et al. Gene expression in the urinary bladder: a common carcinoma in situ gene expression signature exists disregarding histopathological classification. Cancer Res. 2004;64(11):4040–8. Epub 2004/06/03. doi: 10.1158/0008-5472.CAN-03-3620 . [DOI] [PubMed] [Google Scholar]
- 49.Zheng Y, McFarland BC, Drygin D, Yu H, Bellis SL, Kim H, et al. Targeting protein kinase CK2 suppresses prosurvival signaling pathways and growth of glioblastoma. Clinical cancer research: an official journal of the American Association for Cancer Research. 2013;19(23):6484–94. Epub 2013/09/17. doi: 10.1158/1078-0432.CCR-13-0265 ; PubMed Central PMCID: PMC3932633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ladha J, Donakonda S, Agrawal S, Thota B, Srividya MR, Sridevi S, et al. Glioblastoma-specific protein interaction network identifies PP1A and CSK21 as connecting molecules between cell cycle-associated genes. Cancer Res. 2010;70(16):6437–47. Epub 2010/07/29. doi: 10.1158/0008-5472.CAN-10-0819 . [DOI] [PubMed] [Google Scholar]
- 51.Dubois N, Willems M, Nguyen-Khac MT, Kroonen J, Goffart N, Deprez M, et al. Constitutive activation of casein kinase 2 in glioblastomas: Absence of class restriction and broad therapeutic potential. Int J Oncol. 2016;48(6):2445–52. Epub 2016/04/22. doi: 10.3892/ijo.2016.3490 . [DOI] [PubMed] [Google Scholar]
- 52.Lee J, Kotliarova S, Kotliarov Y, Li A, Su Q, Donin NM, et al. Tumor stem cells derived from glioblastomas cultured in bFGF and EGF more closely mirror the phenotype and genotype of primary tumors than do serum-cultured cell lines. Cancer cell. 2006;9(5):391–403. Epub 2006/05/16. doi: 10.1016/j.ccr.2006.03.030 . [DOI] [PubMed] [Google Scholar]
- 53.Shai R, Shi T, Kremen TJ, Horvath S, Liau LM, Cloughesy TF, et al. Gene expression profiling identifies molecular subtypes of gliomas. Oncogene. 2003;22(31):4918–23. Epub 2003/08/02. doi: 10.1038/sj.onc.1206753 . [DOI] [PubMed] [Google Scholar]
- 54.Dixit D, Sharma V, Ghosh S, Mehta VS, Sen E. Inhibition of Casein kinase-2 induces p53-dependent cell cycle arrest and sensitizes glioblastoma cells to tumor necrosis factor (TNFalpha)-induced apoptosis through SIRT1 inhibition. Cell death & disease. 2012;3:e271 Epub 2012/02/10. doi: 10.1038/cddis.2012.10 ; PubMed Central PMCID: PMC3288342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mandal T, Bhowmik A, Chatterjee A, Chatterjee U, Chatterjee S, Ghosh MK. Reduced phosphorylation of Stat3 at Ser-727 mediated by casein kinase 2—protein phosphatase 2A enhances Stat3 Tyr-705 induced tumorigenic potential of glioma cells. Cellular signalling. 2014;26(8):1725–34. Epub 2014/04/15. doi: 10.1016/j.cellsig.2014.04.003 . [DOI] [PubMed] [Google Scholar]
- 56.Ferrer-Font L, Villamanan L, Arias-Ramos N, Vilardell J, Plana M, Ruzzene M, et al. Targeting Protein Kinase CK2: Evaluating CX-4945 Potential for GL261 Glioblastoma Therapy in Immunocompetent Mice. Pharmaceuticals (Basel). 2017;10(1). Epub 2017/02/18. doi: 10.3390/ph10010024 ; PubMed Central PMCID: PMC5374428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Pencheva N, de Gooijer MC, Vis DJ, Wessels LFA, Wurdinger T, van Tellingen O, et al. Identification of a Druggable Pathway Controlling Glioblastoma Invasiveness. Cell Rep. 2017;20(1):48–60. Epub 2017/07/07. doi: 10.1016/j.celrep.2017.06.036 . [DOI] [PubMed] [Google Scholar]
- 58.Sun L, Hui AM, Su Q, Vortmeyer A, Kotliarov Y, Pastorino S, et al. Neuronal and glioma-derived stem cell factor induces angiogenesis within the brain. Cancer cell. 2006;9(4):287–300. Epub 2006/04/18. doi: 10.1016/j.ccr.2006.03.003 . [DOI] [PubMed] [Google Scholar]
- 59.Bredel M, Bredel C, Juric D, Harsh GR, Vogel H, Recht LD, et al. Functional network analysis reveals extended gliomagenesis pathway maps and three novel MYC-interacting genes in human gliomas. Cancer Res. 2005;65(19):8679–89. Epub 2005/10/06. doi: 10.1158/0008-5472.CAN-05-1204 . [DOI] [PubMed] [Google Scholar]
- 60.French PJ, Swagemakers SM, Nagel JH, Kouwenhoven MC, Brouwer E, van der Spek P, et al. Gene expression profiles associated with treatment response in oligodendrogliomas. Cancer Res. 2005;65(24):11335–44. Epub 2005/12/17. doi: 10.1158/0008-5472.CAN-05-1886 . [DOI] [PubMed] [Google Scholar]
- 61.Tugizov S, Berline J, Herrera R, Penaranda ME, Nakagawa M, Palefsky J. Inhibition of human papillomavirus type 16 E7 phosphorylation by the S100 MRP-8/14 protein complex. Journal of virology. 2005;79(2):1099–112. Epub 2004/12/23. doi: 10.1128/JVI.79.2.1099-1112.2005 ; PubMed Central PMCID: PMC538578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Massimi P, Banks L. Differential phosphorylation of the HPV-16 E7 oncoprotein during the cell cycle. Virology. 2000;276(2):388–94. Epub 2000/10/21. doi: 10.1006/viro.2000.0514 . [DOI] [PubMed] [Google Scholar]
- 63.Pyeon D, Newton MA, Lambert PF, den Boon JA, Sengupta S, Marsit CJ, et al. Fundamental differences in cell cycle deregulation in human papillomavirus-positive and human papillomavirus-negative head/neck and cervical cancers. Cancer Res. 2007;67(10):4605–19. Epub 2007/05/19. doi: 10.1158/0008-5472.CAN-06-3619 ; PubMed Central PMCID: PMC2858285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kim SM, Park YY, Park ES, Cho JY, Izzo JG, Zhang D, et al. Prognostic biomarkers for esophageal adenocarcinoma identified by analysis of tumor transcriptome. PLoS One. 2010;5(11):e15074 Epub 2010/12/15. doi: 10.1371/journal.pone.0015074 ; PubMed Central PMCID: PMC2994829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hao Y, Triadafilopoulos G, Sahbaie P, Young HS, Omary MB, Lowe AW. Gene expression profiling reveals stromal genes expressed in common between Barrett's esophagus and adenocarcinoma. Gastroenterology. 2006;131(3):925–33. Epub 2006/09/06. doi: 10.1053/j.gastro.2006.04.026 ; PubMed Central PMCID: PMC2575112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chen B, Dong Z, Cao C, Wu D, Huang Z. [Effect of casein kinase 2beta in esophageal carcinoma and its clinical significance]. Nan Fang Yi Ke Da Xue Xue Bao. 2012;32(10):1491–4. Epub 2012/10/19. . [PubMed] [Google Scholar]
- 67.Wang S, Zhan M, Yin J, Abraham JM, Mori Y, Sato F, et al. Transcriptional profiling suggests that Barrett's metaplasia is an early intermediate stage in esophageal adenocarcinogenesis. Oncogene. 2006;25(23):3346–56. Epub 2006/02/02. doi: 10.1038/sj.onc.1209357 . [DOI] [PubMed] [Google Scholar]
- 68.Kimchi ET, Posner MC, Park JO, Darga TE, Kocherginsky M, Karrison T, et al. Progression of Barrett's metaplasia to adenocarcinoma is associated with the suppression of the transcriptional programs of epidermal differentiation. Cancer Res. 2005;65(8):3146–54. Epub 2005/04/19. doi: 10.1158/0008-5472.CAN-04-2490 . [DOI] [PubMed] [Google Scholar]
- 69.D'Errico M, de Rinaldis E, Blasi MF, Viti V, Falchetti M, Calcagnile A, et al. Genome-wide expression profile of sporadic gastric cancers with microsatellite instability. Eur J Cancer. 2009;45(3):461–9. Epub 2008/12/17. doi: 10.1016/j.ejca.2008.10.032 . [DOI] [PubMed] [Google Scholar]
- 70.Cho JY, Lim JY, Cheong JH, Park YY, Yoon SL, Kim SM, et al. Gene expression signature-based prognostic risk score in gastric cancer. Clinical cancer research: an official journal of the American Association for Cancer Research. 2011;17(7):1850–7. Epub 2011/03/31. doi: 10.1158/1078-0432.CCR-10-2180 ; PubMed Central PMCID: PMC3078023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lee YS, Lee do Y, Yu da Y, Kim S, Lee YC. Helicobacter pylori induces cell migration and invasion through casein kinase 2 in gastric epithelial cells. Helicobacter. 2014;19(6):465–75. doi: 10.1111/hel.12144 . [DOI] [PubMed] [Google Scholar]
- 72.Chen X, Leung SY, Yuen ST, Chu KM, Ji J, Li R, et al. Variation in gene expression patterns in human gastric cancers. Molecular biology of the cell. 2003;14(8):3208–15. Epub 2003/08/20. doi: 10.1091/mbc.E22912-0833 ; PubMed Central PMCID: PMC181561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bian Y, Han J, Kannabiran V, Mohan S, Cheng H, Friedman J, et al. MEK inhibitor PD-0325901 overcomes resistance to CK2 inhibitor CX-4945 and exhibits anti-tumor activity in head and neck cancer. International journal of biological sciences. 2015;11(4):411–22. Epub 2015/03/24. doi: 10.7150/ijbs.10745 ; PubMed Central PMCID: PMC4366640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Brown MS, Diallo OT, Hu M, Ehsanian R, Yang X, Arun P, et al. CK2 modulation of NF-kappaB, TP53, and the malignant phenotype in head and neck cancer by anti-CK2 oligonucleotides in vitro or in vivo via sub-50-nm nanocapsules. Clinical cancer research: an official journal of the American Association for Cancer Research. 2010;16(8):2295–307. Epub 2010/04/08. doi: 10.1158/1078-0432.CCR-09-3200 ; PubMed Central PMCID: PMC2861417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cannon CM, Trembley JH, Kren BT, Unger GM, O'Sullivan MG, Cornax I, et al. Therapeutic Targeting of Protein Kinase CK2 Gene Expression in Feline Oral Squamous Cell Carcinoma: A Naturally Occurring Large-Animal Model of Head and Neck Cancer. Hum Gene Ther Clin Dev. 2017;28(2):80–6. Epub 2017/03/25. doi: 10.1089/humc.2017.008 ; PubMed Central PMCID: PMCPMC5510041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Peng CH, Liao CT, Peng SC, Chen YJ, Cheng AJ, Juang JL, et al. A novel molecular signature identified by systems genetics approach predicts prognosis in oral squamous cell carcinoma. PLoS One. 2011;6(8):e23452 Epub 2011/08/20. doi: 10.1371/journal.pone.0023452 ; PubMed Central PMCID: PMCPMC3154947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Sengupta A, Banerjee D, Chandra S, Banerjee S. Gene therapy for BCR-ABL+ human CML with dual phosphorylation resistant p27Kip1 and stable RNA interference using an EBV vector. J Gene Med. 2006;8(10):1251–61. Epub 2006/09/05. doi: 10.1002/jgm.959 . [DOI] [PubMed] [Google Scholar]
- 78.Talbot SG, Estilo C, Maghami E, Sarkaria IS, Pham DK, Oc P, et al. Gene expression profiling allows distinction between primary and metastatic squamous cell carcinomas in the lung. Cancer Res. 2005;65(8):3063–71. Epub 2005/04/19. doi: 10.1158/0008-5472.CAN-04-1985 . [DOI] [PubMed] [Google Scholar]
- 79.Roelants C, Giacosa S, Duchemin-Pelletier E, McLeer-Florin A, Tisseyre C, Aubert C, et al. Dysregulated Expression of Protein Kinase CK2 in Renal Cancer. Pinna LA, Ahmed K, Issinger O-G, Szyszka R, editors: Springer International Publishing; 2015. 241–57 p. [Google Scholar]
- 80.Stalter G, Siemer S, Becht E, Ziegler M, Remberger K, Issinger OG. Asymmetric expression of protein kinase CK2 subunits in human kidney tumors. Biochemical and biophysical research communications. 1994;202(1):141–7. Epub 1994/07/15. doi: 10.1006/bbrc.1994.1904 . [DOI] [PubMed] [Google Scholar]
- 81.Timofeeva OA, Plisov S, Evseev AA, Peng S, Jose-Kampfner M, Lovvorn HN, et al. Serine-phosphorylated STAT1 is a prosurvival factor in Wilms' tumor pathogenesis. Oncogene. 2006;25(58):7555–64. Epub 2006/06/27. doi: 10.1038/sj.onc.1209742 . [DOI] [PubMed] [Google Scholar]
- 82.Yusenko MV, Kuiper RP, Boethe T, Ljungberg B, van Kessel AG, Kovacs G. High-resolution DNA copy number and gene expression analyses distinguish chromophobe renal cell carcinomas and renal oncocytomas. BMC cancer. 2009;9:152 Epub 2009/05/19. doi: 10.1186/1471-2407-9-152 ; PubMed Central PMCID: PMC2686725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Jones J, Otu H, Spentzos D, Kolia S, Inan M, Beecken WD, et al. Gene signatures of progression and metastasis in renal cell cancer. Clinical cancer research: an official journal of the American Association for Cancer Research. 2005;11(16):5730–9. Epub 2005/08/24. doi: 10.1158/1078-0432.CCR-04-2225 . [DOI] [PubMed] [Google Scholar]
- 84.Cutcliffe C, Kersey D, Huang CC, Zeng Y, Walterhouse D, Perlman EJ. Clear cell sarcoma of the kidney: up-regulation of neural markers with activation of the sonic hedgehog and Akt pathways. Clinical cancer research: an official journal of the American Association for Cancer Research. 2005;11(22):7986–94. Epub 2005/11/22. doi: 10.1158/1078-0432.CCR-05-1354 . [DOI] [PubMed] [Google Scholar]
- 85.Maia S, Haining WN, Ansen S, Xia Z, Armstrong SA, Seth NP, et al. Gene expression profiling identifies BAX-delta as a novel tumor antigen in acute lymphoblastic leukemia. Cancer Res. 2005;65(21):10050–8. Epub 2005/11/04. doi: 10.1158/0008-5472.CAN-05-1574 . [DOI] [PubMed] [Google Scholar]
- 86.Coustan-Smith E, Song G, Clark C, Key L, Liu P, Mehrpooya M, et al. New markers for minimal residual disease detection in acute lymphoblastic leukemia. Blood. 2011;117(23):6267–76. Epub 2011/04/14. doi: 10.1182/blood-2010-12-324004 ; PubMed Central PMCID: PMC3122946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Andersson A, Ritz C, Lindgren D, Eden P, Lassen C, Heldrup J, et al. Microarray-based classification of a consecutive series of 121 childhood acute leukemias: prediction of leukemic and genetic subtype as well as of minimal residual disease status. Leukemia. 2007;21(6):1198–203. Epub 2007/04/06. doi: 10.1038/sj.leu.2404688 . [DOI] [PubMed] [Google Scholar]
- 88.Jiang H, van de Ven C, Baxi L, Satwani P, Cairo MS. Differential gene expression signatures of adult peripheral blood vs cord blood monocyte-derived immature and mature dendritic cells. Exp Hematol. 2009;37(10):1201–15. Epub 2009/08/04. doi: 10.1016/j.exphem.2009.07.010 . [DOI] [PubMed] [Google Scholar]
- 89.Gomes AM, Soares MV, Ribeiro P, Caldas J, Povoa V, Martins LR, et al. Adult B-cell acute lymphoblastic leukemia cells display decreased PTEN activity and constitutive hyperactivation of PI3K/Akt pathway despite high PTEN protein levels. Haematologica. 2014;99(6):1062–8. doi: 10.3324/haematol.2013.096438 ; PubMed Central PMCID: PMCPMC4040910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Silva A, Yunes JA, Cardoso BA, Martins LR, Jotta PY, Abecasis M, et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability. The Journal of clinical investigation. 2008;118(11):3762–74. Epub 2008/10/03. doi: 10.1172/JCI34616 ; PubMed Central PMCID: PMC2556239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Stegmaier K, Ross KN, Colavito SA, O'Malley S, Stockwell BR, Golub TR. Gene expression-based high-throughput screening(GE-HTS) and application to leukemia differentiation. Nature genetics. 2004;36(3):257–63. Epub 2004/02/11. doi: 10.1038/ng1305 . [DOI] [PubMed] [Google Scholar]
- 92.Liu YF, Wang BY, Zhang WN, Huang JY, Li BS, Zhang M, et al. Genomic Profiling of Adult and Pediatric B-cell Acute Lymphoblastic Leukemia. EBioMedicine. 2016;8:173–83. Epub 2016/07/20. doi: 10.1016/j.ebiom.2016.04.038 ; PubMed Central PMCID: PMC4919728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Deffenbacher KE, Iqbal J, Sanger W, Shen Y, Lachel C, Liu Z, et al. Molecular distinctions between pediatric and adult mature B-cell non-Hodgkin lymphomas identified through genomic profiling. Blood. 2012;119(16):3757–66. Epub 2012/03/01. doi: 10.1182/blood-2011-05-349662 ; PubMed Central PMCID: PMC3335381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Pascual V, Medrano LM, Lopez-Palacios N, Bodas A, Dema B, Fernandez-Arquero M, et al. Different Gene Expression Signatures in Children and Adults with Celiac Disease. PLoS One. 2016;11(2):e0146276 Epub 2016/02/10. doi: 10.1371/journal.pone.0146276 ; PubMed Central PMCID: PMC4747499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Quotti Tubi L, Gurrieri C., Brancalion A., Bonaldi L., Bertorelle R., Manni S., Piazza F. Inhibition of protein kinase CK2 with the clinical-grade small ATP-competitive compound CX-4945 or by RNA interference unveils its role in acute myeloid leukemia cell survival, p53-dependent apoptosis and daunorubicin-induced cytotoxicity. Journal of Hematology & Oncology. 2013;(6):78 http://doi.org/10.1186/1756-8722-6-78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kim JS, Eom J. I., Cheong J.-W., Choi A. J., Lee J. K., Yang W. I., & Min Y. H. Protein kinase CK2alpha as an unfavorable prognostic marker and novel therapeutic target in acute myeloid leukemia. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research. 2007;13(3):1019–28. http://doi.org/10.1158/1078-0432.CCR-06-1602 [DOI] [PubMed] [Google Scholar]
- 97.Alizadeh AA, Eisen MB, Davis RE, Ma C, Lossos IS, Rosenwald A, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000;403(6769):503–11. Epub 2000/02/17. doi: 10.1038/35000501 . [DOI] [PubMed] [Google Scholar]
- 98.Haslinger C, Schweifer N, Stilgenbauer S, Dohner H, Lichter P, Kraut N, et al. Microarray gene expression profiling of B-cell chronic lymphocytic leukemia subgroups defined by genomic aberrations and VH mutation status. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2004;22(19):3937–49. Epub 2004/10/02. doi: 10.1200/JCO.2004.12.133 . [DOI] [PubMed] [Google Scholar]
- 99.Bilous N, Abramenko I, Kryachok I, Bazyka D, Chumak A, Bebeshko V. Significance of VH genes mutation status for prognosis of CLL patients. Exp Oncol. 2005;27(4):325–9. Epub 2006/01/13. . [PubMed] [Google Scholar]
- 100.Basso K, Margolin AA, Stolovitzky G, Klein U, Dalla-Favera R, Califano A. Reverse engineering of regulatory networks in human B cells. Nature genetics. 2005;37(4):382–90. Epub 2005/03/22. doi: 10.1038/ng1532 . [DOI] [PubMed] [Google Scholar]
- 101.Rosenwald A, Wright G, Chan WC, Connors JM, Campo E, Fisher RI, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. The New England journal of medicine. 2002;346(25):1937–47. Epub 2002/06/21. doi: 10.1056/NEJMoa012914 . [DOI] [PubMed] [Google Scholar]
- 102.Martins LR, Lucio P, Silva MC, Anderes KL, Gameiro P, Silva MG, et al. Targeting CK2 overexpression and hyperactivation as a novel therapeutic tool in chronic lymphocytic leukemia. Blood. 2010;116(15):2724–31. doi: 10.1182/blood-2010-04-277947 . [DOI] [PubMed] [Google Scholar]
- 103.Hung MS, Lin YC, Mao JH, Kim IJ, Xu Z, Yang CT, et al. Functional polymorphism of the CK2alpha intronless gene plays oncogenic roles in lung cancer. PLoS One. 2010;5(7):e11418 doi: 10.1371/journal.pone.0011418 ; PubMed Central PMCID: PMCPMC2896393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Haferlach T, Kohlmann A, Wieczorek L, Basso G, Kronnie GT, Bene MC, et al. Clinical utility of microarray-based gene expression profiling in the diagnosis and subclassification of leukemia: report from the International Microarray Innovations in Leukemia Study Group. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2010;28(15):2529–37. Epub 2010/04/22. doi: 10.1200/JCO.2009.23.4732 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Rosenwald A, Alizadeh AA, Widhopf G, Simon R, Davis RE, Yu X, et al. Relation of gene expression phenotype to immunoglobulin mutation genotype in B cell chronic lymphocytic leukemia. J Exp Med. 2001;194(11):1639–47. ; PubMed Central PMCID: PMCPMC2193523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Pizzi M, Piazza F, Agostinelli C, Fuligni F, Benvenuti P, Mandato E, et al. Protein kinase CK2 is widely expressed in follicular, Burkitt and diffuse large B-cell lymphomas and propels malignant B-cell growth. Oncotarget. 2015;6(9):6544–52. Epub 2015/03/20. doi: 10.18632/oncotarget.3446 ; PubMed Central PMCID: PMC4466633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Manni S, Brancalion A, Mandato E, Tubi LQ, Colpo A, Pizzi M, et al. Protein kinase CK2 inhibition down modulates the NF-kappaB and STAT3 survival pathways, enhances the cellular proteotoxic stress and synergistically boosts the cytotoxic effect of bortezomib on multiple myeloma and mantle cell lymphoma cells. PLoS One. 2013;8(9):e75280 doi: 10.1371/journal.pone.0075280 ; PubMed Central PMCID: PMCPMC3785505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Compagno M, Lim WK, Grunn A, Nandula SV, Brahmachary M, Shen Q, et al. Mutations of multiple genes cause deregulation of NF-kappaB in diffuse large B-cell lymphoma. Nature. 2009;459(7247):717–21. Epub 2009/05/05. doi: 10.1038/nature07968 ; PubMed Central PMCID: PMC2973325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Storz MN, van de Rijn M, Kim YH, Mraz-Gernhard S, Hoppe RT, Kohler S. Gene expression profiles of cutaneous B cell lymphoma. The Journal of investigative dermatology. 2003;120(5):865–70. Epub 2003/04/26. doi: 10.1046/j.1523-1747.2003.12142.x . [DOI] [PubMed] [Google Scholar]
- 110.Brune V, Tiacci E, Pfeil I, Doring C, Eckerle S, van Noesel CJ, et al. Origin and pathogenesis of nodular lymphocyte-predominant Hodgkin lymphoma as revealed by global gene expression analysis. The Journal of experimental medicine. 2008;205(10):2251–68. Epub 2008/09/17. doi: 10.1084/jem.20080809 ; PubMed Central PMCID: PMC2556780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Piccaluga PP, Agostinelli C, Califano A, Rossi M, Basso K, Zupo S, et al. Gene expression analysis of peripheral T cell lymphoma, unspecified, reveals distinct profiles and new potential therapeutic targets. The Journal of clinical investigation. 2007;117(3):823–34. Epub 2007/02/17. doi: 10.1172/JCI26833 ; PubMed Central PMCID: PMC1794115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Eckerle S, Brune V, Doring C, Tiacci E, Bohle V, Sundstrom C, et al. Gene expression profiling of isolated tumour cells from anaplastic large cell lymphomas: insights into its cellular origin, pathogenesis and relation to Hodgkin lymphoma. Leukemia. 2009;23(11):2129–38. Epub 2009/08/07. doi: 10.1038/leu.2009.161 . [DOI] [PubMed] [Google Scholar]
- 113.Choi YL, Tsukasaki K, O'Neill MC, Yamada Y, Onimaru Y, Matsumoto K, et al. A genomic analysis of adult T-cell leukemia. Oncogene. 2007;26(8):1245–55. Epub 2006/08/16. doi: 10.1038/sj.onc.1209898 . [DOI] [PubMed] [Google Scholar]
- 114.Agnelli L, Mosca L, Fabris S, Lionetti M, Andronache A, Kwee I, et al. A SNP microarray and FISH-based procedure to detect allelic imbalances in multiple myeloma: an integrated genomics approach reveals a wide gene dosage effect. Genes, chromosomes & cancer. 2009;48(7):603–14. Epub 2009/04/28. doi: 10.1002/gcc.20668 . [DOI] [PubMed] [Google Scholar]
- 115.Zhan F, Barlogie B, Arzoumanian V, Huang Y, Williams DR, Hollmig K, et al. Gene-expression signature of benign monoclonal gammopathy evident in multiple myeloma is linked to good prognosis. Blood. 2007;109(4):1692–700. Epub 2006/10/07. doi: 10.1182/blood-2006-07-037077 ; PubMed Central PMCID: PMC1794073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Piazza FA, Ruzzene M, Gurrieri C, Montini B, Bonanni L, Chioetto G, et al. Multiple myeloma cell survival relies on high activity of protein kinase CK2. Blood. 2006;108(5):1698–707. Epub 2006/05/11. doi: 10.1182/blood-2005-11-013672 . [DOI] [PubMed] [Google Scholar]
- 117.Zhang HX, Jiang SS, Zhang XF, Zhou ZQ, Pan QZ, Chen CL, et al. Protein kinase CK2alpha catalytic subunit is overexpressed and serves as an unfavorable prognostic marker in primary hepatocellular carcinoma. Oncotarget. 2015;6(33):34800–17. Epub 2015/10/03. doi: 10.18632/oncotarget.5470 ; PubMed Central PMCID: PMC4741491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Choi SE, Kwon S, Seok S, Xiao Z, Lee KW, Kang Y, et al. Obesity-Linked Phosphorylation of SIRT1 by Casein Kinase 2 Inhibits Its Nuclear Localization and Promotes Fatty Liver. Mol Cell Biol. 2017;37(15). Epub 2017/05/24. doi: 10.1128/MCB.00006-17 ; PubMed Central PMCID: PMC5514454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Roessler S, Jia HL, Budhu A, Forgues M, Ye QH, Lee JS, et al. A unique metastasis gene signature enables prediction of tumor relapse in early-stage hepatocellular carcinoma patients. Cancer Res. 2010;70(24):10202–12. Epub 2010/12/17. doi: 10.1158/0008-5472.CAN-10-2607 ; PubMed Central PMCID: PMC3064515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Chen X, Cheung ST, So S, Fan ST, Barry C, Higgins J, et al. Gene expression patterns in human liver cancers. Molecular biology of the cell. 2002;13(6):1929–39. Epub 2002/06/12. doi: 10.1091/mbc.02-02-0023. ; PubMed Central PMCID: PMC117615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Wurmbach E, Chen YB, Khitrov G, Zhang W, Roayaie S, Schwartz M, et al. Genome-wide molecular profiles of HCV-induced dysplasia and hepatocellular carcinoma. Hepatology. 2007;45(4):938–47. Epub 2007/03/30. doi: 10.1002/hep.21622 . [DOI] [PubMed] [Google Scholar]
- 122.Zhang S, Yang YL, Wang Y, You B, Dai Y, Chan G, et al. CK2alpha, over-expressed in human malignant pleural mesothelioma, regulates the Hedgehog signaling pathway in mesothelioma cells. Journal of experimental & clinical cancer research: CR. 2014;33:93 Epub 2014/11/26. doi: 10.1186/s13046-014-0093-6 ; PubMed Central PMCID: PMC4254219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Gordon GJ, Rockwell GN, Jensen RV, Rheinwald JG, Glickman JN, Aronson JP, et al. Identification of novel candidate oncogenes and tumor suppressors in malignant pleural mesothelioma using large-scale transcriptional profiling. The American journal of pathology. 2005;166(6):1827–40. Epub 2005/05/28. doi: 10.1016/S0002-9440(10)62492-3 ; PubMed Central PMCID: PMC1363736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Morrison C, Farrar W, Kneile J, Williams N, Liu-Stratton Y, Bakaletz A, et al. Molecular classification of parathyroid neoplasia by gene expression profiling. The American journal of pathology. 2004;165(2):565–76. Epub 2004/07/28. doi: 10.1016/S0002-9440(10)63321-4 ; PubMed Central PMCID: PMC1618556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Guo C, Yu S, Davis AT, Wang H, Green JE, Ahmed K. A potential role of nuclear matrix-associated protein kinase CK2 in protection against drug-induced apoptosis in cancer cells. J Biol Chem. 2001;276(8):5992–9. Epub 2000/11/09. doi: 10.1074/jbc.M004862200 . [DOI] [PubMed] [Google Scholar]
- 126.He H, Jazdzewski K, Li W, Liyanarachchi S, Nagy R, Volinia S, et al. The role of microRNA genes in papillary thyroid carcinoma. Proc Natl Acad Sci U S A. 2005;102(52):19075–80. Epub 2005/12/21. doi: 10.1073/pnas.0509603102 ; PubMed Central PMCID: PMCPMC1323209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Takahashi K, Setoguchi T, Tsuru A, Saitoh Y, Nagano S, Ishidou Y, et al. Inhibition of casein kinase 2 prevents growth of human osteosarcoma. Oncol Rep. 2017;37(2):1141–7. Epub 2016/12/14. doi: 10.3892/or.2016.5310 . [DOI] [PubMed] [Google Scholar]
- 128.Barretina J, Taylor BS, Banerji S, Ramos AH, Lagos-Quintana M, Decarolis PL, et al. Subtype-specific genomic alterations define new targets for soft-tissue sarcoma therapy. Nature genetics. 2010;42(8):715–21. Epub 2010/07/06. doi: 10.1038/ng.619 ; PubMed Central PMCID: PMC2911503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Detwiller KY, Fernando NT, Segal NH, Ryeom SW, D'Amore PA, Yoon SS. Analysis of hypoxia-related gene expression in sarcomas and effect of hypoxia on RNA interference of vascular endothelial cell growth factor A. Cancer Res. 2005;65(13):5881–9. Epub 2005/07/05. doi: 10.1158/0008-5472.CAN-04-4078 . [DOI] [PubMed] [Google Scholar]
- 130.Quade BJ, Wang TY, Sornberger K, Dal Cin P, Mutter GL, Morton CC. Molecular pathogenesis of uterine smooth muscle tumors from transcriptional profiling. Genes Chromosomes Cancer. 2004;40(2):97–108. Epub 2004/04/22. doi: 10.1002/gcc.20018 . [DOI] [PubMed] [Google Scholar]
- 131.Zhou B, Ritt DA, Morrison DK, Der CJ, Cox AD. Protein Kinase CK2alpha Maintains Extracellular Signal-regulated Kinase (ERK) Activity in a CK2alpha Kinase-independent Manner to Promote Resistance to Inhibitors of RAF and MEK but Not ERK in BRAF Mutant Melanoma. J Biol Chem. 2016;291(34):17804–15. doi: 10.1074/jbc.M115.712885 ; PubMed Central PMCID: PMCPMC5016172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Posch C, Sanlorenzo M, Vujic I, Oses-Prieto JA, Cholewa BD, Kim ST, et al. Phosphoproteomic Analyses of NRAS(G12) and NRAS(Q61) Mutant Melanocytes Reveal Increased CK2alpha Kinase Levels in NRAS(Q61) Mutant Cells. J Invest Dermatol. 2016;136(10):2041–8. Epub 2016/06/03. doi: 10.1016/j.jid.2016.05.098 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Talantov D, Mazumder A, Yu JX, Briggs T, Jiang Y, Backus J, et al. Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin Cancer Res. 2005;11(20):7234–42. doi: 10.1158/1078-0432.CCR-05-0683 . [DOI] [PubMed] [Google Scholar]
- 134.Haqq C, Nosrati M, Sudilovsky D, Crothers J, Khodabakhsh D, Pulliam BL, et al. The gene expression signatures of melanoma progression. Proc Natl Acad Sci U S A. 2005;102(17):6092–7. Epub 2005/04/19. doi: 10.1073/pnas.0501564102 ; PubMed Central PMCID: PMC1087936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Riker AI, Enkemann SA, Fodstad O, Liu S, Ren S, Morris C, et al. The gene expression profiles of primary and metastatic melanoma yields a transition point of tumor progression and metastasis. BMC medical genomics. 2008;1:13 Epub 2008/04/30. doi: 10.1186/1755-8794-1-13 ; PubMed Central PMCID: PMC2408576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Guerra B, Siemer S, Boldyreff B, Issinger OG. Protein kinase CK2: evidence for a protein kinase CK2beta subunit fraction, devoid of the catalytic CK2alpha subunit, in mouse brain and testicles. FEBS Lett. 1999;462(3):353–7. Epub 2000/01/06. . [DOI] [PubMed] [Google Scholar]
- 137.Macias Alvarez LR-C, J.; Dominguez, I. CK2 in embryonic development. The Wiley-IUBMB Series on Biochemistry and Molecular Biology: Protein Kinase CK2. 2013.
- 138.Oshida K, Hirakata M, Maeda A, Miyoshi T, Miyamoto Y. Toxicological effect of emodin in mouse testicular gene expression profile. J Appl Toxicol. 2011;31(8):790–800. doi: 10.1002/jat.1637 . [DOI] [PubMed] [Google Scholar]
- 139.Peng X, Zeng X, Peng S, Deng D, Zhang J. The association risk of male subfertility and testicular cancer: a systematic review. PLoS One. 2009;4(5):e5591 Epub 2009/05/15. doi: 10.1371/journal.pone.0005591 ; PubMed Central PMCID: PMC2680046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Bai X, Silvius D, Chan ED, Escalier D, Xu SX. Identification and characterization of a novel testis-specific gene CKT2, which encodes a substrate for protein kinase CK2. Nucleic acids research. 2009;37(8):2699–711. Epub 2009/03/11. doi: 10.1093/nar/gkp094 ; PubMed Central PMCID: PMC2677865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Krick R, Aschrafi A, Hasgun D, Arnemann J. CK2-dependent C-terminal phosphorylation at T300 directs the nuclear transport of TSPY protein. Biochem Biophys Res Commun. 2006;341(2):343–50. doi: 10.1016/j.bbrc.2005.12.190 . [DOI] [PubMed] [Google Scholar]
- 142.Sperger JM, Chen X, Draper JS, Antosiewicz JE, Chon CH, Jones SB, et al. Gene expression patterns in human embryonic stem cells and human pluripotent germ cell tumors. Proc Natl Acad Sci U S A. 2003;100(23):13350–5. Epub 2003/11/05. doi: 10.1073/pnas.2235735100 ; PubMed Central PMCID: PMC263817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Skotheim RI, Lind GE, Monni O, Nesland JM, Abeler VM, Fossa SD, et al. Differentiation of human embryonal carcinomas in vitro and in vivo reveals expression profiles relevant to normal development. Cancer Res. 2005;65(13):5588–98. Epub 2005/07/05. doi: 10.1158/0008-5472.CAN-05-0153 . [DOI] [PubMed] [Google Scholar]
- 144.Korkola JE, Houldsworth J, Chadalavada RS, Olshen AB, Dobrzynski D, Reuter VE, et al. Down-regulation of stem cell genes, including those in a 200-kb gene cluster at 12p13.31, is associated with in vivo differentiation of human male germ cell tumors. Cancer Res. 2006;66(2):820–7. Epub 2006/01/21. doi: 10.1158/0008-5472.CAN-05-2445 . [DOI] [PubMed] [Google Scholar]
- 145.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74. Epub 2011/03/08. doi: 10.1016/j.cell.2011.02.013 . [DOI] [PubMed] [Google Scholar]
- 146.Deshiere A, Duchemin-Pelletier E, Spreux E, Ciais D, Combes F, Vandenbrouck Y, et al. Unbalanced expression of CK2 kinase subunits is sufficient to drive epithelial-to-mesenchymal transition by Snail1 induction. Oncogene. 2013;32(11):1373–83. Epub 2012/05/09. doi: 10.1038/onc.2012.165 . [DOI] [PubMed] [Google Scholar]
- 147.Ludwig JA, Weinstein JN. Biomarkers in cancer staging, prognosis and treatment selection. Nat Rev Cancer. 2005;5(11):845–56. Epub 2005/10/22. doi: 10.1038/nrc1739 . [DOI] [PubMed] [Google Scholar]
- 148.Michiels S, Ternes N, Rotolo F. Statistical controversies in clinical research: prognostic gene signatures are not (yet) useful in clinical practice. Ann Oncol. 2016;27(12):2160–7. Epub 2016/09/17. doi: 10.1093/annonc/mdw307 ; PubMed Central PMCID: PMC5178139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Goossens N, Nakagawa S, Sun X, Hoshida Y. Cancer biomarker discovery and validation. Transl Cancer Res. 2015;4(3):256–69. Epub 2015/07/28. doi: 10.3978/j.issn.2218-676X.2015.06.04 ; PubMed Central PMCID: PMC4511498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Song C, Gowda C, Pan X, Ding Y, Tong Y, Tan BH, et al. Targeting casein kinase II restores Ikaros tumor suppressor activity and demonstrates therapeutic efficacy in high-risk leukemia. Blood. 2015;126(15):1813–22. doi: 10.1182/blood-2015-06-651505 ; PubMed Central PMCID: PMCPMC4600018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Guo M, Liu C, Qi FJ, Zhang XM, Ren LL, Liu YM, et al. Elevated expression of nuclear protein kinase CK2alpha as a poor prognosis indicator in lymph node cancerous metastases of human thyroid cancers. Asian Pac J Cancer Prev. 2014;15(17):7425–32. Epub 2014/09/18. . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.