Abstract
Aroma is important in assessing the quality of fresh fruit and their processed products, and could provide good indicators for the development of local cultivars in the mango industry. In this study, the volatile diversity of 25 mango cultivars from China, America, Thailand, India, Cuba, Indonesia, and the Philippines was investigated. The volatile compositions, their relative contents, and the intervarietal differences were detected with headspace solid phase microextraction tandem gas chromatography-mass spectrometer methods. The similarities were also evaluated with a cluster analysis and correlation analysis of the volatiles. The differences in mango volatiles in different districts are also discussed. Our results show significant differences in the volatile compositions and their relative contents among the individual cultivars and regions. In total, 127 volatiles were found in all the cultivars, belonging to various chemical classes. The highest and lowest qualitative abundances of volatiles were detected in ‘Zihua’ and ‘Mallika’ cultivars, respectively. Based on the cumulative occurrence of members of the classes of volatiles, the cultivars were grouped into monoterpenes (16 cultivars), proportion and balanced (eight cultivars), and nonterpene groups (one cultivars). Terpene hydrocarbons were the major volatiles in these cultivars, with terpinolene, 3-carene, caryophyllene and α-Pinene the dominant components depending on the cultivars. Monoterpenes, some of the primary volatile components, were the most abundant aroma compounds, whereas aldehydes were the least abundant in the mango pulp. β-Myrcene, a major terpene, accounted for 58.93% of the total flavor volatile compounds in ‘Xiaofei’ (Philippens). γ-Octanoic lactone was the only ester in the total flavor volatile compounds, with its highest concentration in ‘Guiya’ (China). Hexamethyl cyclotrisiloxane was the most abundant volatile compound in ‘Magovar’ (India), accounting for 46.66% of the total flavor volatiles. A typical aldehydic aroma 2,6-di-tert-butyl-4-sec-butylphenol, was detected in ‘Gleck’. A highly significant positive correlation was detected between Alc and K, Alk and Nt, O and L. Cultivars originating from America, Thailand, Cuba, India, Indonesia and the Philippines were more similar to each other than to those from China. This study provides a high-value dataset for use in development of health care products, diversified mango breeding, and local extension of mango cultivars.
Introduction
Aroma is an important attribute, reflecting fruit quality, ripeness, and consumer acceptance [1–4]. Aroma is conferred by a range of diverse low-molecular-weight, volatile,chemical compounds that occur widely in nature. It plays a key role in the quality profiles of fruit (including mango, apple, grape, etc.) and is an attribute considered in flavor and fragrance production not only in the food industry but also in the cosmetics industry. Monoterpenes, sesquiterpenes, esters, lactones, alcohols, aldehydes, ketones, volatile fatty acids, some degradation product of phenols, and some carotenoid contributes to the aromatic volatile profile of mango fruit [5–10]. Aroma is also important in assessing the quality of fresh mango and its processed products, and is a good index for screening mango germplasm resources for the development of local cultivars.
Mango (Mangifera indica L.), often called the “king of fruits”, is the most popular fruit throughout the world in terms of its production, marketing and consumption [11,12]. Among tropical fruits, it ranks second only to the banana in international trade [13]. Mango production throughout the world is nearly 42.66 million tones, and China is the second largest mango-producing country, producing slightly less than 4.62 million tonnes in 2013 [14]. Apart from the consumption of fresh mangoes, mango pulp and concentrated juice are widely traded in current international markets as a base material in the beverage industry and as a flavoring ingredient in the dairy industry and baby food formulations [14].
Aroma is specific to each mango cultivar [7]. ‘Alphonso’ is a unique mango cultivar with an abundance of both lactones and furanone in its ripe fruits [9,11,15]. δ-3-carene as the only compound that contributes the mango-like notes to western mango cultivars [8]. This observation is supported by a subsequent finding that non-Indian cultivars invariably contain δ-3-carene as their principle volatile component [9,11]. Sixty-one aromatic volatile compounds were identified in the ‘Kensington Pride’ mango by Lalel et al. [7], of which 35 had not been reported previously. Lalel et al. [16] also measured Glycosidically bound aromatic volatile compounds in the skin and pulp of the ‘Kensington Pride’ mango fruit at different stages of maturity, and found that only terpenes seem to contribute to the aroma of fresh ripe ‘Kensington Pride’ mangoes.
The composition and concentrations of aromatic volatiles in mango fruit are influenced by the cultivar [9,10]. Pandit et al.[9] analyzed 27 mango cultivars, detecting 84 volatile compounds, and the highest and lowest qualitative abundances of volatiles were detected in the ‘Alphonso’ and ‘Pairi’ cultivars, respectively. John et al. [17] found differences in the compositions and concentrations of terpenoid compounds in the sap of seven Indian mango varieties, including monoterpenes β-myrcene, trans-/cis-ocimene, and limonene. Based on a study that revealed significant differences in the fresh flavors and textures of six Thai mangoes, Ledeker et al. [18] suggested that manufacturers should select the appropriate cultivars for mango purées because heat treatment significantly changes the flavor and texture, Musharraf et al. [12] developed a quantitative method based on gas chromatography-triple quadrupole mass spectrometry for the analysis of the aroma component of mango sap in nine Pakistani cultivars, and identified seven terpenes, α-pinene, α-phellandrene, (+)-3-carene, sabinene, γ-terpinene, (-)-trans-caryophyllene, and α-humulene. When analyzing the aromas of 15 varieties of mango cultivated in Brazil, Andrade et al. [19] found that α-terpinolene was the principal constituent of the ‘Willard’, ‘Parrot’, ‘Bowen’ and ‘Kensington’ varieties; α-terpinolene and Δ3-carene mainly occurred in Sri Lanka varieties; Δ3-carene was the major contributor to the aroma of mango fruit grown in Venezuela and Myrcene was characteristic of the ‘Alphonso’ variety from India.
Although mango germplasm resource are relatively abundant throughout the world, even in China, mango breeding objectives largely focus on disease resistance, or fruit size, sweetness, or color [20], rather than on aroma quality. Currently, artificial breeding that has ignored the properties of fruit aroma, making mango cultivars lack aroma diversification and resource superiority has been not take full their advantage. The main constraint is that the aroma properties of mango germplasms are unclear. Therefore, we aimed at improving the database of mango germplasm resource to provide a basis for breeding unique-functional mango cultivars, to promote the classification of varieties. Twenty-five mango cultivars, either cultivated in China or indigenous cultivars from more than seven states or districts, were qualitatively and quantitatively analyzed with solid-phase microextraction-gas chromatography-tandem mass spectrometer (SPME-GC-MS). The volatile component profiles of all the mango cultivars are described and the cultivars were classified according to a cluster analysis based on their volatile species.
Materials and methods
Plant material, sampling, and chemicals
Twenty-five mango cultivars and their districts of origin are shown in Table 1, All were grafted on the 17-year-old rootstock Yuexi No.1. which grows in an orchard located in the Mango germplasm Nursery of the South Subtropical Crops Research Institute, Chinese Academy of Tropical Agricultural Sciences in Zhanjiang, Guangdong Province, China. For each cultivar, night trees of similar size and similar vigor were randomly selected for sampling. Of these, each three trees consisting of a replicate from which 15–20 mango fruits were sampled from the top, center, and bottom of the trees.
Table 1. Mango cultivars analyzed in this study and their origins.
| Origin | Cultivar |
|---|---|
| China | Zihua(1), Shixuan No.8(2), Renong No.1(3), Guire No.82(4), Tainong No.1(5), Hongmang No.8(6) |
| America | Haden (7), Tommy Atkins(8), Edward(9), Glenn(10) |
| Thailand | Hongmang No.11(11), Keawsaweuy(12), Choke Anand(13), Nam Dok Mai(14), Thai 506(15) |
| Cuba | Cuba No.1(16), Cuba No.3(17), Cubasankeli(18) |
| India | Mallika(19), Magovar(20), Saigon(21) |
| Indonesia & Philippines | Gleck(22), Baodaohuang(23), Guiya(24), Xiaofei(25) |
Note: Numbers in parentheses of following the cultivar names are the accession numbers.
All chemicals and reagents for aroma extraction and the GC/MS analysis were purchased from Thermo Fisher Scientific (USA), Alfa Aesar (USA) or Sigma-Aldrich Co. (St. Louis, MO, USA) and were of the highest available purity. The SPME instrument was from Supelco Company of Sigma-Aldrich (St. Louis, MO, USA). Water was purified with a Milli-Q deionization unit (Millipore, Bedford, MA, USA). The Trace GC Ultra-ISQ MS system was from Thermo Fisher Scientific, with a gas chromatograph (Agilent 6890N) coupled to a mass spectrometer (Agilent 5975). The GC column was a fused silica capillary column (Agilent DB-5 MS, 30 m × 0.25 mm ID × 0.25 μm film thickness).
Sample pretreatment and extraction
At the harvesting stage of maturity, fruit at about 80% full ripeness and of identical size were picked and transported immediately to the laboratory to accelerate their ripening. After about 3 days, the fruit were fully ripe and suitable for pulp collection. After the fruit were peeled and cut into small pieces, the pulp was frozen with liquid nitrogen and ground into powder. All the homogenized pulp powder were immediately stored at -40°C for subsequent analyses.
The powdered fresh pulp (8 g) was added to 25-mL vial, which was sealed with a polytetrafluoroethylene isolation membrane (Tianjin Jinteng Experiment Equipment Co., Ltd., China) to extract the aroma compounds with headspace SPME (HS-SPME). The SPME syringe (100 μm polydimethylsiloxane) was then inserted into the headspace of the vial and the volatiles were extracted at 25°C in a thermostatically controlled water bath for 40 min with continuous stirring.
GC/MS analysis and MS library matching
GC-MS analysis was performed as described by Musharraf et al. [12] and Wei et al. [21] with modifications. After extraction, the SPME fiber was placed into the GC-MS injector and maintained for 3 min for desorption. The GC-MS system consisted of a Trace™ GC Ultra gas chromatograph and a tandem ISQ mass spectrometer. Chromatographic conditions were: pressure of the carrier gas (helium) of 7.7 Psi at the initial oven temperature, with flow rate 1 mL·min-1. All samples were injected in the splitless mode. The injector temperature was 250°C; the initial oven temperature was 50°C for 1 min, which was increased to 140°C at a rate of 5°C min-1, and then to 250°C at a rate of 10°C min-1, where it was maintained for 10 min (total run time, 45 min). The mass spectrometer was operated in electron impact (EI) mode using parameters: EI mode at 70 eV, with a scan range of 35―335 m/z. The temperatures of the transfer line and of the ion source was set to 250 and 200°C, respectively. The sample (volume 1.0 μL) was injected in triplicate throughout the study. The Mass Hunter software (Agilent) was used for data acquisition and processing. The peaks of the aromas compounds were identified by their retention times, with references to standard compounds, and the mass spectra obtained were compared with those available in the Wiley and NIST08 libraries (Wiley RegistryTM, 8th Edition Mass Spectral Library, and the NIST 08 Mass Spectral Library [NIST/EPA/NIH] 2008 version), with an acceptance criterion of a score match > 70%. Signal were acquired and the analytes were quantified with Multiple Reaction Monitoring (MRM) to determine the precursor-product ion pairs of the transitions. The relative percentage contents of all the analytes were calculated by normalizing the peak areas.
Statistic analysis
The signal analysis and the automatic integration and generation of signal charts using the Analyst software were appropriate for the GC-MS equipment. For each component within a mango variety, the content was determined from three biology replicates and each replicate was analyzed with three technical repetitions. Microsoft Office Excel 2010 was used to calculate the target components, relative standard deviations, and bias.
In this study, all the components detected were first grouped into different compounds, according to the similarity of their chemical property. The relative contents of the compounds were subjected to a correlation analysis and a cluster analysis. The content of each compound in a replicate mango variety was generated with the formula: value-1 = Ʃ(mean scores of the three replicates of each component, including all components of the same compound from the same varieties). The value-1 data were used for a cluster analysis of the varieties, using the XLSTAT 2010 software, and to calculate Pearson’s correlation coefficients (correlation analysis) across compounds, with the SPASS (version 17.0) software. Similarly, value-2 = log 2 (Ʃ[value-1 of each variety from the same region]/number of varieties in the corresponding region) was used for the cluster analysis across regions with HemI 1.0.3.3 software.
Results and discussion
Scanning of volatile composition and proportion from 25 mango pulp
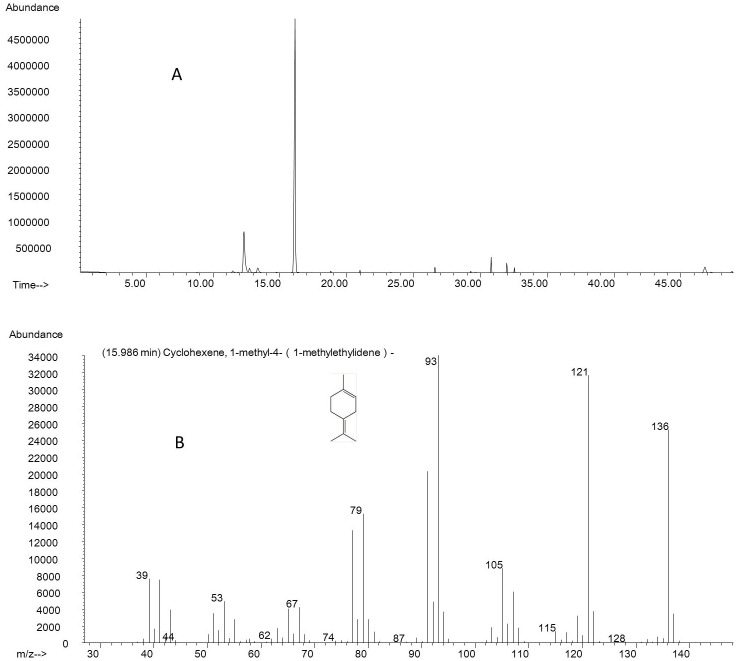
The aroma components of 25 mango pulps was determined independently with HS-SPME-GC-MS. Fig 1 (A & B) show a representative GC-MS chromatogram of a selected sample (‘Renong 1’) and the total ion mass spectrum of α-terpinolene, respectively. Altogether, 127 volatile components were detected in the mangoes in this study (Table 2). The compositions and proportions of volatiles in the mango fruit of individual cultivars are also shown in Table 2. These results suggest that mango pulp is very rich in volatile components. The Thai cultivar ‘Thai 506’ had the most volatile components (32 aroma components) ranking first among all the cultivars analyzed. In contrast, the Chinese cultivar ‘Shixuan No. 8’ and the Thai cultivars ‘Hongmang No. 11’ had the least volatile components, containing only two volatile components each, terpinolene (2.41%) and squalene (14.84%) in ‘Shixuan No. 8’ and 1-Methyl-4-(1-methylethylidene)-cyclohexene (1.12%) and 3-Carene (26.35%) in ‘Hongmang No. 11’.
Fig 1. Representative GC–MS chromatogram of volatiles from mango pulp.
A: Representative GC–MS chromatogram of a selected sample (‘Renong 1’); B: representative total ion mass spectrum of α-terpinolene (retention time, 15.986 min) from ‘Renong 1’.
Table 2. Compositions and relative contents of all volatiles from 25 mango cultivars (%).
Table in the annex.
| Code | Volatile component | Cultivar | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | ||
| 1 | α-Panasinsen | 0.0312 | 0.0798 | |||||||||||||||||||||||
| 2 | 4-α-Isopropenyl-2-carene | 1.6782 | ||||||||||||||||||||||||
| 3 | 4-Carene | 0.5576 | 1.6387 | 0.5476 | 81.2519 | |||||||||||||||||||||
| 4 | Epi-bicyclosesquiphellandrene | 0.0281 | ||||||||||||||||||||||||
| 5 | α-Caryophyllene | 0.7902 | 1.3301 | 0.8934 | 0.0906 | 0.8239 | 2.5755 | 2.1961 | 0.0797 | 7.1135 | 0.2779 | 0.1057 | 1.6404 | 0.4192 | 0.0981 | |||||||||||
| 6 | α-Cubebene | 0.2453 | 1.5866 | 0.2769 | 0.0347 | 0.026 | 0.0465 | 0.5382 | ||||||||||||||||||
| 7 | α-Pinene | 6.5207 | ||||||||||||||||||||||||
| 8 | β-Myrcene | 1.0777 | 0.5228 | 58.9252 | ||||||||||||||||||||||
| 9 | β-Panasinsene | 0.2599 | ||||||||||||||||||||||||
| 10 | β-Phellandrene | 0.6011 | 0.6324 | |||||||||||||||||||||||
| 11 | β-Pinene | 0.7436 | 0.7663 | 0.802 | 2.4686 | 0.0433 | 0.6671 | |||||||||||||||||||
| 12 | γ-Elemene | 1.2064 | ||||||||||||||||||||||||
| 13 | 1,2-Benzenedicarboxylic acid, bis(2-methylpropyl) ester | 0.0535 | ||||||||||||||||||||||||
| 14 | Diisooctyl phthalate | 0.7018 | ||||||||||||||||||||||||
| 15 | 1,2-Nonadiene | 0.717 | ||||||||||||||||||||||||
| 16 | 1,3,5,7,9-Pentaethylcyclopentasiloxane | 15.3875 | 14.3308 | |||||||||||||||||||||||
| 17 | 1,3,5,8-Undecatetraene | 0.1931 | 0.2697 | 0.1105 | ||||||||||||||||||||||
| 18 | 2,5,5-Trimethyl-1,3,6-heptatriene | 0.3421 | ||||||||||||||||||||||||
| 19 | (E)-3,7-Dimethyl-1,3,6-octatriene | 55.3169 | 5.5873 | 34.9883 | 15.4551 | |||||||||||||||||||||
| 20 | (Z)-3,7-Dimethyl-1,3,6-octatriene | 71.67 | 0.038 | 0.075 | 9.8259 | 0.1453 | ||||||||||||||||||||
| 21 | 1,7-Dimethyl-1,3,7-cyclodecatriene | 4.9807 | ||||||||||||||||||||||||
| 22 | 3,7-Dimethyl-1,3,7-octatriene | 1.7927 | ||||||||||||||||||||||||
| 23 | 1,3,8-p-Menthatriene | 0.1306 | 4.3817 | 0.0378 | 0.346 | |||||||||||||||||||||
| 24 | 1,3,5,5-Tetramethyl-1,3-cyclohexadiene | 0.7255 | ||||||||||||||||||||||||
| 25 | 1,5,5,6-Tetramethyl-1,3-cyclohexadiene | 2.0304 | ||||||||||||||||||||||||
| 26 | 1-Methyl-4-(1-methylethyl)-1,3-cyclohexadiene | 1.6878 | 2.2205 | 1.0879 | 2.9598 | 0.4117 | 1.4136 | 2.1669 | 2.0536 | |||||||||||||||||
| 27 | 3,6-Dimethyl-1-cyclohexene-1,2-dicarboxylic anhydride | 0.2495 | ||||||||||||||||||||||||
| 28 | 1,5,9,9-tetramethyl-1,4,7,-Cycloundecatriene | 0.3483 | 1.209 | |||||||||||||||||||||||
| 29 | 1-methyl-4-(1-methylethyl)-,1,4-Cyclohexadiene | 0.4054 | 0.1697 | 0.22 | 0.1143 | 0.2657 | 3.9026 | 0.1032 | 0.2589 | 0.1764 | ||||||||||||||||
| 30 | 1,5,5-Trimethyl-6-methylene-cyclohexene | 0.0432 | ||||||||||||||||||||||||
| 31 | 1-methyl-5-methylene-8-(1-methylethyl)-1,6-Cyclodecadiene | 0.8607 | 0.1277 | 3.5056 | 7.7999 | 0.098 | 1.0609 | 0.0035 | ||||||||||||||||||
| 32 | 3,4-dihydro-8-hydroxy-3-methyl-, (R)-,1H-2-Benzopyran-1-one | 0.0483 | ||||||||||||||||||||||||
| 33 | 1H-Cyclopenta[1,3]cyclopropa[1,2]benzene, octahydro-7-methyl-3-methylene-4-(1-methylethyl)-, [3aS-(3a.alpha.,3b.beta.,4.beta.,7.alpha.,7aS*)]-1H-Cyclopenta[1,3]cyclopropa[1,2]benzene | 0.0729 | 0.3677 | 0.0433 | ||||||||||||||||||||||
| 34 | 1a,2,3,4,4a,5,6,7b-octahydro-1,1,4,7-tetramethyl-, [1aR-(1a.alpha.,4.alpha.,4a.beta.,7b.alpha.)]-1H-Cycloprop[e]azulene | 0.0344 | 0.9584 | 0.0404 | 0.3654 | 0.3489 | 0.0913 | 0.4952 | 0.299 | |||||||||||||||||
| 35 | 1a,2,3,4,4a,5,6,7b-octahydro-1,1,4,7-tetramethyl-, [1aR-(1a.alpha.,4.alpha.,4a.beta.,7b.alpha.)]-1H-Cycloprop[e]azulene | 0.0551 | ||||||||||||||||||||||||
| 36 | Ledene | 0.175 | ||||||||||||||||||||||||
| 37 | decahydro-1,1,7-trimethyl-4-methylene-, [1aR-(1a.alpha.,4a.beta.,7.alpha.,7a.beta.,7b.alpha.)]-1H-Cycloprop[e]azulene | 0.0226 | 0.0247 | 0.2985 | 0.1251 | |||||||||||||||||||||
| 38 | 1a,2,3,3a,4,5,6,7b-octahydro-1,1,3a,7-tetramethyl-, [1aR-(1a.alpha.,3a.alpha.,7b.alpha.)]-1H-Cyclopropa[a]naphthalene | 0.2361 | ||||||||||||||||||||||||
| 39 | 1a,2,3,5,6,7,7a,7b-octahydro-1,1,7,7a-tetramethyl-, [1aR-(1a.alpha.,7.alpha.,7a.alpha.,7b.alpha.)]-1H-Cyclopropa[a]naphthalene | 0.2464 | 0.1262 | 0.0633 | 0.1703 | 0.0313 | ||||||||||||||||||||
| 40 | 1R-α-Pinene | 2.6515 | 0.167 | 1.6285 | 0.2793 | 0.2227 | 1.5891 | 3.4447 | 0.4983 | 0.2667 | 5.7238 | |||||||||||||||
| 41 | 1S-α-Pinene | 0.3832 | 2.7223 | 0.3547 | ||||||||||||||||||||||
| 42 | 5-butyldihydro-,2(3H)-Furanone | 1.0966 | 1.2702 | 0.0234 | 25.8522 | 0.3352 | ||||||||||||||||||||
| 43 | Thiophenone | 0.0162 | ||||||||||||||||||||||||
| 44 | 2,6-Dimethyl-2,4,6-Octatriene, | 36.9021 | 0.0357 | 0.1736 | ||||||||||||||||||||||
| 45 | 2,6,6-Trimethyl-2,4-cyclohrptadien-1-one | 3.0314 | ||||||||||||||||||||||||
| 46 | 2,6-Bis(1,1-dimethylethyl)-4-(1-oxopropyl)phenol | 0.0844 | ||||||||||||||||||||||||
| 47 | 3,7-dimethyl-, (Z)-,2,6-Octadien-1-ol | 0.1154 | ||||||||||||||||||||||||
| 48 | Crotonic acid cis-3-hexen-1-yl- ester | 0.1108 | 0.1025 | |||||||||||||||||||||||
| 49 | 1,3-dihydro-5-methyl-, 2H-Benzimidazol-2-one | 0.0146 | ||||||||||||||||||||||||
| 50 | tetrahydro-6-propyl-,2H-Pyran-2-one | 0.1407 | ||||||||||||||||||||||||
| 51 | 3-Methylnorcaran-2-one | 0.0459 | ||||||||||||||||||||||||
| 52 | 4-Methoxy-2,5-dimethyl-3(2H)-furanone | 1.162 | ||||||||||||||||||||||||
| 53 | 2,2,4,4,5,5,7,7-octamethyl-3,6-Dioxa-2,4,5,7-tetrasilaoctane | 9.4042 | ||||||||||||||||||||||||
| 54 | 3,6-Dioxa-2,4,5,7-tetrasilaoctane, 2,2,4,4,5,5,7,7-octamethyl-2,2,4,4,5,5,7,7-octamethyl-,3,6-Dioxa-2,4,5,7-tetrasilaoctane | 61.7749 | ||||||||||||||||||||||||
| 55 | 3-Carene | 15.8931 | 13.178 | 56.0521 | 18.5973 | 16.0272 | 26.3469 | 4.7682 | 0.606 | 66.5493 | 3.5464 | |||||||||||||||
| 56 | Leaf alcohol | 0.0662 | 0.4243 | |||||||||||||||||||||||
| 57 | di-TMS,3-Hydroxymandelic acid,ethyl ester | 6.8367 | ||||||||||||||||||||||||
| 58 | 4-Hydroxymandelic acid, ethyl ester | 4.625 | 7.2629 | |||||||||||||||||||||||
| 59 | 1,2,3,4,5,6,7,8-octahydro-1,4-dimethyl-7-(1-methylethenyl)-, [1S-(1.alpha.,4.alpha.,7.alpha.)]-Azulene | 0.3551 | 0.2305 | |||||||||||||||||||||||
| 60 | 1,2,3,5,6,7,8,8a-octahydro-1,4-dimethyl-7-(1-methylethenyl)-, [1S-(1.alpha.,7.alpha.,8a.beta.)]-Azulene | 1.3421 | 0.7405 | 0.562 | ||||||||||||||||||||||
| 61 | 2,5-bis[(trimethylsilyl)oxy]-Benzaldehyde | 0.3364 | ||||||||||||||||||||||||
| 62 | 1,2,3,4-Tetramethyl-benzene | 0.0232 | 0.2214 | |||||||||||||||||||||||
| 63 | 2-Methyl-m-phenylene diisocyanate | 0.0291 | 0.0038 | |||||||||||||||||||||||
| 64 | 1-ethyl-2,3-dimethyl-Benzene | 2.0041 | ||||||||||||||||||||||||
| 65 | 1-methyl-2-(1-methylethyl)-Benzene | 0.1692 | ||||||||||||||||||||||||
| 66 | M-Cymene | 0.1183 | ||||||||||||||||||||||||
| 67 | 1-methyl-4-(1-methylethenyl)-Benzene | 0.1546 | 3.9097 | 0.221 | ||||||||||||||||||||||
| 68 | 1-methyl-4-(1-methylethyl)-Benzene | 1.2242 | 0.0734 | |||||||||||||||||||||||
| 69 | 2,4-diisocyanato-1-methyl-Benzene | 0.0456 | 0.0405 | 0.0064 | ||||||||||||||||||||||
| 70 | 2-Ethyl-1,4-dimethyl—benzene | 0.1851 | ||||||||||||||||||||||||
| 71 | 2,3-dihydro-2-methyl-Benzofuran, | 0.1161 | ||||||||||||||||||||||||
| 72 | 5-methyl-2-trimethylsilyloxy-benzoic acid, trimethylsilyl ester | 10.8052 | ||||||||||||||||||||||||
| 73 | Thujone | 0.0387 | ||||||||||||||||||||||||
| 74 | 4-methyl-1-(1-methylethyl)-Bicyclo[3.1.0]hex-2-ene | 0.5373 | 1.924 | |||||||||||||||||||||||
| 75 | α-Pinene | 0.086 | ||||||||||||||||||||||||
| 76 | 2,6-dimethyl-6-(4-methyl-3-pentenyl)bicyclo[3.1.1]hept-2-ene | 0.0641 | ||||||||||||||||||||||||
| 77 | 3,6,6-trimethyl-Bicyclo[3.1.1]hept-2-ene | 2.629 | ||||||||||||||||||||||||
| 78 | 6,6-dimethyl-2-methylene-Bicyclo[3.1.1]heptane | 0.2901 | ||||||||||||||||||||||||
| 79 | 3,7,7-trimethyl-Bicyclo[4.1.0]hept-2-ene | 1.9211 | 3.132 | |||||||||||||||||||||||
| 80 | 3,7,7-trimethyl-(1S)-Bicyclo[4.1.0]hept-3-ene | 4.2718 | 11.149 | 5.4888 | 76.2961 | 0.3683 | 4.494 | 69.0401 | 4.3691 | 5.0789 | ||||||||||||||||
| 81 | 4,11,11-Trimethyl-8-methylene-Bicyclo[7.2.0]undec-4-ene | 0.0214 | ||||||||||||||||||||||||
| 82 | Butanoic acid,2-methylpropyl ester | 1.6604 | ||||||||||||||||||||||||
| 83 | (E)-Butanoic acid, 3-hexenyl ester | 0.4512 | ||||||||||||||||||||||||
| 84 | (Z)-Butanoic acid, 3-hexenyl ester | 0.9817 | 1.9066 | 1.7441 | 0.6321 | 0.5695 | 0.124 | 1.213 | ||||||||||||||||||
| 85 | Butanoic acid, hexyl ester | 0.0198 | ||||||||||||||||||||||||
| 86 | Butanoic acid, octyl ester | 0.0428 | ||||||||||||||||||||||||
| 87 | Camphene | 0.0898 | 0.8166 | 0.0109 | 0.0502 | |||||||||||||||||||||
| 88 | Caryophyllene | 1.3729 | 1.9911 | 1.6518 | 0.2664 | 0.7445 | 1.9939 | 5.0832 | 4.491 | 12.2009 | 0.1486 | 12.1097 | 0.678 | 0.3852 | 3.0453 | 2.1023 | 0.9352 | 0.2351 | ||||||||
| 89 | Copaene | 0.0626 | 1.6147 | |||||||||||||||||||||||
| 90 | 1-methyl-4-(1-methylethylidene)-Cyclohexene | 76.0088 | 2.4124 | 69.2617 | 0.7227 | 78.3034 | 34.4422 | 4.4233 | 1.1237 | 7.5131 | 6.4201 | 33.8182 | 5.8849 | 2.8273 | 63.003 | 1.0479 | 0.0115 | 81.5373 | 11.4756 | 0.029 | ||||||
| 91 | 1-methyl-4-(5-methyl-1-methylene-4-hexenyl)-Cyclohexene | 0.0462 | ||||||||||||||||||||||||
| 92 | 1-Methyl-5-(1-methylethenyl)-Cyclohexene, | 0.1947 | ||||||||||||||||||||||||
| 93 | 3-methyl-6-(1-methylethylidene)cyclohexene | 0.1718 | ||||||||||||||||||||||||
| 94 | 4-Methylene-1-(1-methylethyl)-cyclohexene | 1.9176 | 2.135 | 2.2519 | 1.8514 | 0.0739 | 1.4432 | 1.9632 | 1.9308 | |||||||||||||||||
| 95 | 3-isopropenyl-5,5-dimethyl-Cyclopentene | 3.8759 | ||||||||||||||||||||||||
| 96 | 4-ethenyl-1,5,5-trimethyl-Cyclopentene | 0.2091 | ||||||||||||||||||||||||
| 97 | 1-(2-Propenyl)-cyclopentene | 0.1364 | ||||||||||||||||||||||||
| 98 | octamethyl-Cyclotetrasiloxane | 1.0671 | ||||||||||||||||||||||||
| 99 | Hexamethyl-Cyclotrisiloxane | 0.9918 | 0.8073 | |||||||||||||||||||||||
| 100 | D-Limonene | 0.6041 | 0.3268 | |||||||||||||||||||||||
| 101 | Eicosane | 0.2245 | ||||||||||||||||||||||||
| 102 | 1,2-Bis(2-chloroethoxy)-ethane | 0.1557 | 0.0797 | |||||||||||||||||||||||
| 103 | 4'-Methylacetophenone | 0.0438 | ||||||||||||||||||||||||
| 104 | Heneicosane | 0.4635 | ||||||||||||||||||||||||
| 105 | Hexanoic acid,3-hexenyl ester | 0.0445 | 0.004 | |||||||||||||||||||||||
| 106 | Limonene | 0.2566 | 1.5074 | 0.9416 | ||||||||||||||||||||||
| 107 | Longifolene-(V4) | 0.0789 | 0.679 | |||||||||||||||||||||||
| 108 | 1,2,3,4,4a,5,6,8a-octahydro-4a,8-dimethyl-2-(1-methylethenyl)-, [2R-(2.alpha.,4a.alpha.,8a.beta.)]-Naphthalene | 0.2373 | 0.183 | 0.1038 | 5.3282 | 17.7823 | 0.3419 | |||||||||||||||||||
| 109 | 1,2,3,4,6,8alpha-Hexahydro-1-isopropyl-4,7-dimethylnaphthalene | 0.0674 | ||||||||||||||||||||||||
| 110 | Valencene | 0.2722 | 0.232 | |||||||||||||||||||||||
| 111 | 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1S-(1.alpha.,7.alpha.,8a.alpha.)]-Naphthalene | 0.0144 | ||||||||||||||||||||||||
| 112 | 1,2,3,5,6,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-(1S-cis)-Naphthalene | 0.0487 | 0.0252 | 0.1824 | 0.7626 | 0.1352 | 1.4689 | 1.5862 | ||||||||||||||||||
| 113 | 1,2,4a,5,6,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-Naphthalene | 0.0692 | 1.8182 | |||||||||||||||||||||||
| 114 | α-Muurolene | 0.1311 | 0.0471 | |||||||||||||||||||||||
| 115 | 1,2,4a,5,8,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-, [1S-(1.alpha.,4a.beta.,8a.alpha.)]-Naphthalene | |||||||||||||||||||||||||
| 116 | β-Cadinene | 0.3401 | 0.4812 | 0.1401 | 0.0059 | |||||||||||||||||||||
| 117 | decahydro-4a-methyl-1-methylene-7-(1-methylethenyl)-, [4aR-(4a.alpha.,7.alpha.,8a.beta.)]-Naphthalene | 3.0895 | 1.9284 | 2.7228 | 1.0316 | 1.2042 | 3.3372 | 3.9186 | 7.895 | |||||||||||||||||
| 118 | Decahydro-4a-methyl-1-methylene-7-(1-methylethenyl)-, [4aR-(4a.alpha.,7.alpha.,8a.beta.)]-Naphthalene | 7.499 | 0.3306 | |||||||||||||||||||||||
| 119 | γ-Selinene | 2.6824 | ||||||||||||||||||||||||
| 120 | Palmitic acid | 0.0282 | ||||||||||||||||||||||||
| 121 | 1-Nonanal | 0.0467 | ||||||||||||||||||||||||
| 122 | 2,6-Bis(1,1-dimethylethyl)-4-(1-methylpropyl)-phenol | 0.0088 | ||||||||||||||||||||||||
| 123 | 4,4'-(1-methylethylidene)bis-Phenol | 0.1716 | 12.1471 | 1.4004 | 2.7819 | 0.5711 | 0.1058 | |||||||||||||||||||
| 124 | Phthalic acid, isobutyl octyl ester | 1.28 | 2.9376 | |||||||||||||||||||||||
| 125 | 2-methyl-Propanoic acid, 3,7-dimethyl-2,6-octadienyl ester | 0.0497 | ||||||||||||||||||||||||
| 126 | 2-methyl-Propanoic acid,hexyl ester | 0.0267 | 0.201 | 0.0314 | 0.2107 | 0.036 | ||||||||||||||||||||
| 127 | p-Trimethylsilyloxyphenyl-bis(trimethylsilyloxy)ethane | 0.849 |
Note: Values correspond to relative volumetric amounts of individual components.
Different volatile compositions and proportions in the 25 mango pulps
The significant differences in the compositions and proportions of the volatile components in the 25 mango pulps are shown in Table 3. Monoterpenes, some of the primary volatile components, were the most abundant aroma constituent, whereas aldehydes were the least common volatile constituents in the mango pulps, ‘Tommy Atkins’ had the highest monoterpenes content of 97.48%, followed by ‘Renong No.1’, ‘Gleck’ and ‘Baodaohuang’, which all had content of more than 91%. The aldehydes content were lowest in these mango pulps (Table 3).
Table 3. Relative contents of various chemical classes in different mango cultivars.
| Cultivar code | Volatile compounds (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| M | S | Nt | L | Alc | Ald | K | Ac | Alk | P | O | |
| 1 | 87.92 | 4.46 | 1.12 | 1.15 | 0.25 | 0.20 | 0.21 | 0.98 | |||
| 2 | 2.41 | 14.84 | |||||||||
| 3 | 92.26 | 3.57 | 0.12 | 0.71 | 0.38 | 14.84 | |||||
| 4 | 85.43 | 6.59 | 1.27 | 1.16 | |||||||
| 5 | 89.32 | 2.93 | 2.31 | 0.16 | 0.16 | 0.13 | |||||
| 6 | 49.23 | 1.34 | 0.17 | 0.78 | 0.01 | 0.91 | 0.08 | ||||
| 7 | 81.11 | 10.32 | 0.99 | ||||||||
| 8 | 97.48 | 12.02 | |||||||||
| 9 | 18.60 | 1.68 | 6.84 | 12.15 | 15.39 | ||||||
| 10 | 17.82 | 71.18 | 0.85 | 4.98 | |||||||
| 11 | 27.48 | ||||||||||
| 12 | 69.53 | 12.74 | 1.21 | 2.11 | 1.4 | 0.37 | |||||
| 13 | 7.03 | 0.33 | 4.75 | 0.12 | |||||||
| 14 | 22.20 | 4.63 | |||||||||
| 15 | 61.33 | 1.60 | 13.72 | 2.53 | 0.05 | 1.07 | 4.38 | ||||
| 16 | 73.66 | 20.44 | 0.19 | 1.97 | |||||||
| 17 | 73.87 | 2.46 | 0.221 | 0.42 | 3.28 | 0.69 | 2.78 | ||||
| 18 | 72.65 | 6.09 | |||||||||
| 19 | 0.69 | ||||||||||
| 20 | 83.38 | 8.02 | 0.57 | 0.57 | |||||||
| 21 | 1.05 | 22.41 | |||||||||
| 22 | 91.21 | 1.41 | 0.02 | 0.15 | 0.01 | 0.04 | |||||
| 23 | 91.50 | 5.20 | 0.41 | 1.35 | 0.35 | ||||||
| 24 | 11.48 | 25.85 | .081 | 32.4 | |||||||
| 25 | 81.53 | 15.82 | 1.00 | 0.34 | 0.05 | 0.08 | 0.11 | 1.97 | |||
Note: M, S, Nt, L, Alc, Ald, K, Ac, O, Alk and P indicate monoterpene, sesquiterpen, non-terpene, lactones, alcohols, aldehydes, ketones, acids, others, alkanes, phenols, respectively. The same below. Values corresponds to the relative volumetric amounts, where volatile compounds = Ʃ(score of each component belonging to the same compound).
Correlation analysis of the volatiles
The highly significant positive correlations between volatiles alcohols (Alc) and ketones (K), alkanes (Alk) and non-terpene (Nt), O (others) and lactones (L), detected with a correlation analysis, are noteworthy. However, other pairs of volatile components showed negative correlations, such as Nt and monoterpene (M) or sesquiterpene (S), L and M, S, or Nt, etc., although these were not significant (Table 4).
Table 4. Correlation analysis of the volatiles.
| Indexes | M | S | Nt | L | Alc | K | Alk | P | O |
| M | 1 | 0.090 | -0.199 | -0.281 | 0.125 | 0.188 | 0.086 | -0.161 | -0.362 |
| S | —— | 1 | -0.206 | -0.274 | -0.168 | -0.112 | -0.189 | -0.181 | -0.279 |
| Nt | —— | —— | 1 | -0.074 | -0.075 | -0.069 | 0.521** | -0.046 | 0.035 |
| L | —— | —— | —— | 1 | -.061 | -0.090 | -0.075 | 0.167 | 0.797** |
| Alc | —— | —— | —— | —— | 1 | 0.795** | 0.264 | 0.109 | -0.157 |
| K | —— | —— | —— | —— | —— | 1 | 0.282 | 0.143 | -0.147 |
| Alk | —— | —— | —— | —— | —— | —— | 1 | -0.051 | -0.078 |
| P | —— | —— | —— | —— | —— | —— | —— | 1 | 0.266 |
| O | —— | —— | —— | —— | —— | —— | —— | —— | 1 |
Note:–means negative correlation;
** significant correlation (P<0.01);——none.
Cluster analysis of volatile components
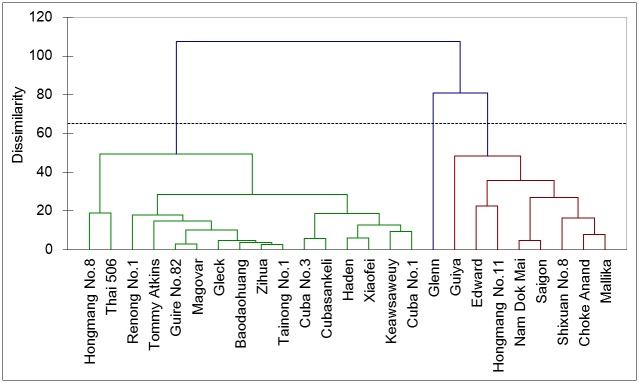
A cluster analysis was conducted based on the volatile composition and proportion data for the 25 cultivars, and the cluster dendrogram was presented in Fig 2. The results show that, at 50% dissimilarity, cultivar 6, 15, 3, 8, 4, 20, 22, 23, 1, 5, 17, 18, 7, 25, 12, and 16 were analogous in their volatile compositions and proportions, with diverse volatile compositions and containing mainly monoterpenes (these cultivars were grouped as the ‘monoterpenes’ group). Cultivars 24, 9, 11, 14, 21, 2, 13, and 19 clustered together, with lower volatile compositions and proportions (these cultivars were grouped in the ‘proportion and balanced’ group). Interestingly, cultivar 10 was classified alone based on its unique lack of terpene (this cultivar was independently classified in the ‘nonterpene’ group).
Fig 2. Cluster dendrogram of 25 mango cultivars.
Regional differences in mango volatiles
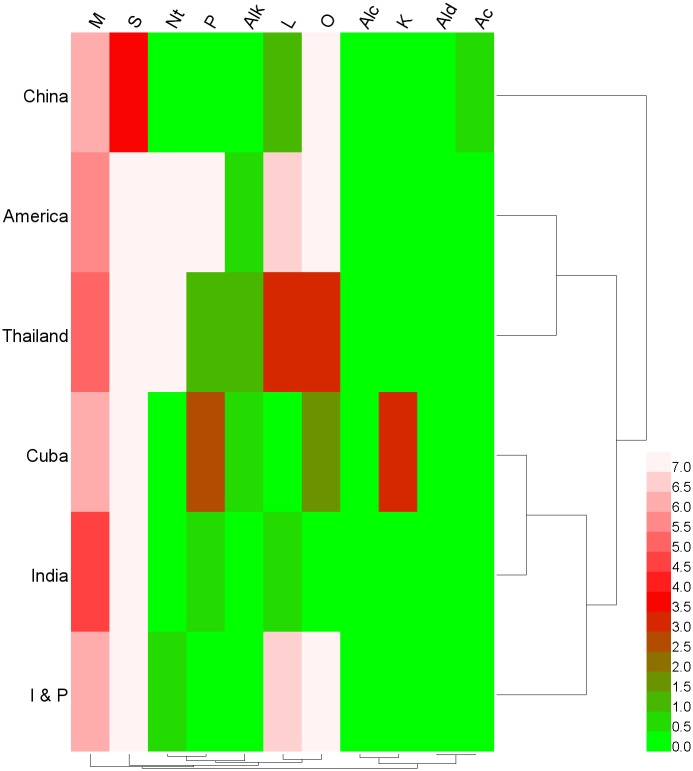
To clarigy the variations in the cultivars originating from seven distinct regions, a cluster analysis was performed base on their volatile contents. The results suggest that the cultivars originating from America, Thailand, Cuba, India, Indonesia and Philippines were more similar to one another than to those from China. The cultivars originating from America and Thailand, as well as Cuba and India, were especially similarity (Fig 3).
Fig 3. Cluster dendrogram of six regions.
“I & P”, Indonesia and the Philippines; M, monoterpene; S, sesquiterpene; Nt, nonterpene; P, phenols; Alk, alkanes; L, lactones; Alc, alcohols; K, ketones; Ald, aldehydes; Ac, acids and O, others.
Monoterpenes were again the most common volatiles, which is consistent with the results in Table 3. Individually, the cultivars originating from India contained the most monoterpene, followed by those originating from America and Thailand, whereas the cultivars originating from China, Cuba, Indonesia, and the Philippine contained the least monoterpenes (Fig 3). The cultivars originating from China had the highest contents of sesquiterpen, Whereas those originating from Cuba had the highest contents of both phenols and ketones, and those from Thailand had the most monoterpenes, lactones, and other volatiles.
Conclusion
The volatile components that confer the aroma of fruit are one of the most important indicators of quality. Our analysis of the volatile organic compounds in mango pulp allowed us to differentiate mango cultivars and indicated the complex synthetic mechanisms involved in the production of their aroma [22]. Altogether, 127 volatiles were detected in all the selected mango cultivars, which belonged to various chemical classes. The highest and lowest qualitative abundance of volatiles were observed in the ‘Zihua’ and ‘Mallika’ cultivars, respectively.
Previous research by Pino et al. indicated that terpenes are the primary aroma constituents of mangoes [10]. Andrade et al. divided mango cultivars into terpinolene cultivars, 3-carene cultivars, and myrcene cultivars, based on their different constituent terpenes [19]. Similar to previous studies, in the present study, we found that terpenes were the main aroma constituents of 16 mango cultivars, and included α-pinene, selinene, caryophyllene, careen, and terpinolene. However, we detected significant differences in terpenes compositions and proportions in the 25 cultivars: the terpenes contents of ‘Guire No. 82’, ‘Magovar’, ‘Gleck’, ‘Baodaohuang’, ‘Cuba No. 3’, and ‘Cubasankeli’ were much higher than those of ‘Shixuan No. 8’, ‘Choke Anand’, ‘Mallika’, and ‘Edward’ (Table 3). ‘Shixuan No. 8’ had only two major aroma components, terpinolene and squalene, and was the only cultivar to contain squalene, with a content of 43.91%. Liu et al. [23] reported that selinene, eremophilene, and aromadendrene were only detected in ‘JinHwang’; limonene were detected in ‘Irwin’; and α-caryophyllene were detected in ‘Keitt’. Interestingly, in this study, ‘Thai 506’ was the only cultivar to contain squalene, though the content was only 0.01%. Although a few β-squalene-, limonene-e, or α-caryophyllene-containing cultivars were found in this study, no cultivar contained aromadendrene. These large differences may be attributable to differences in the cultivation conditions, maturity levels, and postharvest storage treatments, as well as to the cultivars themselves. In these ways, differences in aroma and flavor are generated in mango fruit.
Aldehyde occurs at a low level, but plays a key role in mango flavor. This observation was supported by the earlier finding by Macleod et al. that the aldehydes content of mangoes was 0.03%-14.36% [24]. In the present study, only two cultivars contained aldehydes. However, Pino et al. [10] considered that the aldehydes in Cuban mango cultivars was associated with the sweet herbal flavor of their fruit. A typical aldehydic aroma, 2,6-di-tert-butyl-4-sec-butylphenol, was detected in ‘Gleck’ in the present study.
Aldehydes are easily broken down to alcohols and lactones [25]. Alcohols occur in small amounts in mango fruit and contribute slightly to their aroma and flavor [26,27]. In this study, only three cultivars contained alcohols. Lactones confer a full-bodied fruit aroma, and Andrade et al.[19] reported lactones to be the second most common aroma volatile in the mango. A study by Wilson et al. [28] suggested that lactones occur at low but still detectable level in mango fruit, and Pandit et al. [29] found eight lactones in 18 mango cultivars. γ-Octanoic lactone was the only ester detected among the total flavor volatile compounds in the present study, with its highest concentration in ‘Guiya’ (China).
The cluster analysis of regional difference was limited in this study by the partial and small number of specimens in the mini-core collection of mango germplasms. Therefore, further analysis should be conducted with more mango germplasms in the future. The highly significant positive correlation and other negative pairwise correlations among the volatile components suggest that metabolic interrelationships and the regulation of the volatiles in mango pulp warrant further research to assist the development and utilization of mango aromas.
Data Availability
All relevant data are within the manuscript.
Funding Statement
This research was supported by the the Special Fund for Agro-scientific Research in the Public Interest (201203092-3), the National Nature Science Foundation of Guangdong Province (2016A030307005) and Special Fund of Basic Scientific Research Project for State Level and Public Welfare-scientific Research Institutes (1630062015001, 1630062015007, 1630062016007). All the funders of these fundings belong to Chinese government. These fundings are public welfare research funds and had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Orchard J, Farrel G, Rees D (2012) “Chapter 6: Mango” appears in crop post-harvest: Science and Technology, Singh Z, Singh SP (Eds.), Blackwell Publishing Ltd, Oxford UK: 108–142. [Google Scholar]
- 2.Aragüez I, Valpuesta Fernández V (2013) Metabolic engineering of aroma components in fruits. Biotechnology journal 8: 1144–1158. doi: 10.1002/biot.201300113 [DOI] [PubMed] [Google Scholar]
- 3.El Hadi MAM, Zhang F-J, Wu F-F, Zhou C-H, Tao J (2013) Advances in fruit aroma volatile research. Molecules 18: 8200–8229. doi: 10.3390/molecules18078200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yufan Tang CZ, Songxiao Cao, Xiao Wang, Hongyan Qi (2015) The Effect of CmLOXs on the Production of Volatile Organic Compounds in Four Aroma Types of Melon (Cucumis melo). PLoS One 10: e0143567 doi: 10.1371/journal.pone.0143567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schwab W, Davidovich-Rikanati R, Lewinsohn E (2008) Biosynthesis of plant-derived flavor compounds. Plant J, 54(4): 712–732. doi: 10.1111/j.1365-313X.2008.03446.x [DOI] [PubMed] [Google Scholar]
- 6.Negri A, Allegra D, Simoni L, Rusconi F, Tonelli C, Espen L, Galbiati M (2015) Comparative analysis of fruit aroma patterns in the domesticated wild strawberries "Profumata di Tortona" (F. moschata) and "Regina delle Valli" (F. vesca). Front Plant Sci: 6: 56 doi: 10.3389/fpls.2015.00056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lalel HJD, Singh Z, Tan SC (2003) Aroma volatiles production during fruit ripening of ‘Kensington Pride’ mango. Postharvest Bio Tec 27(02): 323–336. [Google Scholar]
- 8.MacLeod AJ, Snyder CH (1985) Volatile components of two cultivars of mango from Florida. J Agr Food Chem 33(3): 380–384. [Google Scholar]
- 9.Pandit SS, Chidley HG, Kulkarni RS, Pujari KH, Giri AP, Gupta VS (2009) Cultivar relationships in mango based on fruit volatile profiles. Food Chem 114(1): 363–372. [Google Scholar]
- 10.Pino JA, Mesa JY, Marti MP, Marbot R (2005) Volatile Components from Mango (Mangifera indica L.) cultivars. J Agr Food Chem 53(6): 2213–2223. [DOI] [PubMed] [Google Scholar]
- 11.Dar Po MS, Chidley H, Deshpande A, Giri A, Gupta V (2015) “Chapter 19: Volatile Diversity of Mango Cultivars” appears in nutrient and nlavor content of mango (Mangifera indica L.) cultivars: An Appurtenance to the List of Staple Foods, Simmonds and Preedy (Eds.): 458, 461. [Google Scholar]
- 12.Musharraf SG, Uddin J, Siddiqui AJ, Akram MI (2016) Quantification of aroma constituents of mango sap from different pakistan mango cultivars using gas chromatography triple quadrupole mass spectrometry. Food Chem 96: 1355–1360. [DOI] [PubMed] [Google Scholar]
- 13.Masibo M, He Q (2009) Mango bioactive compounds and related nutraceutical properties—A review. Food Revi Int 25: 346–370. [Google Scholar]
- 14.FAOSTAT (2015) http://faostat3.fao.org/browse/Q/QC/ESD.
- 15.Idstein H, Schreier P (1985) Volatile constituents of alphonso mango (Mangifera indica), Phytochemistry 24(10): 2313–2316. [Google Scholar]
- 16.D.Lalel HJ, Singh Z, Tan SC (2003) Glycosidically-bound aroma volatile compounds in the skin and pulp of ‘Kensington Pride’ mango fruit at different stages of maturity. Postharvest Bio Tec 29(02): 205–218. [Google Scholar]
- 17.Johna KS, Raob LJM, Bhata SG, Raoa UJSP, Rao P (1999) Characterization of aroma components of sap from dierent Indian mango varieties. Phytochemistry 52(5): 891–894. [Google Scholar]
- 18.Ledeker CN, Suwonsichon S, Chambers DH, Adhikar IK (2014) Comparison of sensory attributes in fresh mangoes and heat-treated mango purées prepared from Thai cultivars. LWT—Food Sci Technol 56(1): 138–144. [Google Scholar]
- 19.Andrade EHA, Maia JGS, Graias MD, Zoghbi B (2000) Aroma volatile constituents of Brazilian varieties of mango fruit. J Food Compos Anal 13: 27–33. [Google Scholar]
- 20.Huang GD (2000) Research status and development of mango breeding in China. China Fruits: 47–49. [Google Scholar]
- 21.Wei CB, Liu SH, Liu YG, Lv LL, Yang WX, et al. (2011) Characteristic aroma compounds from different pineapple parts. Molecules 16: 5104–5112. doi: 10.3390/molecules16065104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.González-Mas MC, Rambla JL, Alamar MC, Gutiérrez A, Granell A (2011) Comparative analysis of the volatile fraction of fruit juice from different Citrus species. PLoS One 6: e22016 doi: 10.1371/journal.pone.0022016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu FX, Fu SF, Bi XF,Chen F, Liao XJ, Xiao SH, Wu JH (2013) Physico-chemical and antioxidant properties of four mango (Mangifera indica L.) Cultivars in China. Food Chem 138(1): 396–405. doi: 10.1016/j.foodchem.2012.09.111 [DOI] [PubMed] [Google Scholar]
- 24.Macleod AJ, Troconis NGD (1982) Volatile flavour components of mango fruit. Phytochemistry 21(10): 2523–2526. [Google Scholar]
- 25.Matsui K (2006) Green leaf volatiles: Hydroperoxide lyase pathway of oxylipin metabolism. Curr Opin Plant Biol 9(3): 274–280. doi: 10.1016/j.pbi.2006.03.002 [DOI] [PubMed] [Google Scholar]
- 26.Engel KH, Tressl R (1983) Studies on the volatile components of two mango varieties. J Agri Food Chem 31(4): 796–801. [Google Scholar]
- 27.Quijano CE, Salamanca G, Pino JA (2007) Aroma volatile constituents of colombian varieties of mango (Mangifera indica L.). Flavour Frag J 22(5): 401–406. [Google Scholar]
- 28.Wilson CWI, Shaw PE, Knight RJ (1990) Importance of some lactones and 2,5-dimethyl-4-hydroxy-3(2H)-furanone to mango (Mangifera indica L.) aroma. J Agri Food Chem 38(7): 1556–1559. [Google Scholar]
- 29.Pandit SS, Mitra S, Giri AP, Pujari KH, Patil BP, et al. (2007) Genetic diversity analysis of mango cultivars using inter simple sequence repeat markers. Current Sci India, 93(8): 1135–1145. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the manuscript.