Abstract
Background:
Some studies have suggested using epidural analgesia after cancer surgery to reduce metastasis. This article examines the relationship between regional anesthesia (RA) and cancer metastasis in an array of cancers.
Methods:
We conducted a review of the literature using PubMed and included 67,577 patients across 28 studies in a metaanalysis, evaluating the hazard ratios (HRs) of overall survival, recurrence-free survival, and biochemical recurrence-free survival.
Results:
We found no benefit to RA as it relates to cancer. The HR was 0.92 for overall survival, 1.06 for recurrence-free survival, and 1.05 for biochemical recurrence-free survival. Despite the overall analysis showing no benefit, we found some benefit when we evaluated only the randomized trials. However, we found no significant benefit of RA when we evaluated the cancers (gastrointestinal, prostate, breast, and ovarian) individually.
Conclusion:
This metaanalysis shows that RA has no overall survival, recurrence-free survival, or biochemical recurrence-free survival benefit. However, some individual studies have shown significant benefit in terms of cancer recurrence. Further, RA reduces the use of opioids, which has led to some secondary benefits. Further studies are needed to establish the benefits of RA as it relates to cancer.
Keywords: Analgesics–opioid, angiogenesis inducing agents, morphine, neoplasm metastasis
INTRODUCTION
Cancer-related pain is a severe and debilitating problem affecting millions of patients. This pain can often be unbearable, and healthcare providers are often compelled to increase such patients' doses of opioid analgesia. However, research in animal models has shown that opioids are immunosuppressive1-3 through mediating inflammation and modulating angiogenesis,4-8 and the stresses associated with surgical procedures can also impair immunity. Therefore, alternative multimodal pain management techniques are required to reduce the negative impact of opioids and to attempt to minimize surgical stress. Regional anesthesia (RA) is becoming a popular choice for pain management for many healthcare providers, and some evidence suggests that RA may play a role in inhibiting cancer progression. Various theories have been proposed to explain how RA may inhibit cancer progression: inhibition of neuroendocrine stress by the sympathetic block,9 effects of local anesthetics on inflammation of cancer cell proliferation,10-12 and reduction of opioid consumption and its immunosuppressive3,13 and proangiogenic effects.6
Tumor Metastasis
Cancer occurs at sites of injury and inflammation. These inflammatory mediators play a key role in cancer formation and progression. Cancer forms in the settings of DNA damage and alteration in the cell environment. These initial changes have been termed initiation, and these changes often persist until another cell injury leads to promotion. Promotion can be caused by inflammation or other injury that causes an imbalance of the tumor's metastatic potential and antimetastatic potential.14-16 Promotion leads to the recruitment of inflammatory cells, release of chemical mediators, damage to the surrounding tissue, and eventually failure of apoptosis, leading to rapid cellular proliferation.17 Initially, tumors are only weakly antigenic, but they continue to mutate over time and become more antigenic.17
Several theories explain how cellular proliferation and growth occur, but one that is often cited is “seed and soil.”18 Tumor nutrition is initially met with diffusion, but with time, angiogenic factors are secreted, allowing neovascularization to occur. Neovascularization often occurs in response to injury and inflammation. An evolving tumor cannot progress beyond a 2-mm diameter size without angiogenesis occurring to meet its increasing metabolic requirements.17 The mediators of this process include vascular endothelial growth factor (VEGF), matrix metalloproteinases (MMPs), tumor necrosis factor-α (TNF-α), transforming growth factor-β (TGF-β), and prostaglandin E2 (PGE2).18 VEGF stimulates signaling pathways that lead to proliferation and migration of endothelial cells, increase vascular permeability, and—if tumor cells express tyrosine kinase VEGF receptors—lead to de-differentiation and tumor spread in an autocrine manner.18,19 MMPs lead to degradation and remodeling of the extracellular matrix.18,20 PGE2 is important in phagocyte-mediated immunity and in limiting the potential harmful activation of cytotoxic cells.18 PGE2 has been shown to play an important role in cancer by inhibiting apoptosis, stimulating angiogenesis, enhancing invasion,18 and enhancing migration and invasion via activation of the epidermal growth factor receptor (EGFR).21 Of note, mutations of EGFR have been linked to several cancers.21 In the postoperative period, PGE2, which is also produced by tissue injury and postoperative wound healing, may mediate metastatic progression.22 The cytokines TNF-α and TGF-β are involved in systemic inflammation and function in the regulation of immune cells. When the inflammation cascade is activated, cancer cells spread by entering the lymphatic system and, finally, the general circulation.
Cyclooxygenase (COX) plays an important role in the formation of prostaglandins. Concentrations of COX-2 are upregulated in various cancers including breast, prostate, and gastrointestinal (GI) cancers.23 One of the main products of COX-2 is PGE2. Nonsteroidal antiinflammatory drugs (NSAIDs) are inhibitors of COX-1 and COX-2. Thus, animal and human studies have shown the benefit of NSAIDs in the prevention of cancer.24 Melamed et al found that administration of indomethacin reduced the increase in lung metastasis caused by surgery in rats inoculated with mammary adenocarcinoma.24 Similar findings were reported by Farooqui et al in breast cancer: increasing the level of PGE2 promoted survival in animal models.25 Farooqui et al also identified the benefit of improved pain control, which has also been shown to decrease the inflammatory response and the potential for metastasis.25
Surgery and the Proangiogenic Response
Surgery often removes the bulk of the tumor cells, but individual tumor cells are occasionally left at the margins of the cancer, often referred to as micrometastasis.26 The immunosuppressive effects of anesthesia are additive to those of surgery.17 Many cancer cells are dormant for long periods of time, and surgery provides the opportunity for growth. Surgical stress has an effect on MMPs, the proteolytic enzymes that facilitate the penetration of the extracellular matrix and the basement membrane during metastasis.20 Surgery plays 4 key roles in promoting metastasis: (1) management and disruption of tumor-releasing tumor cells into circulation,27 (2) decreased circulation of antiangiogenic factors (angiostatin and endostatin),28,29 (3) increased local and systemic release of growth factors after surgery,30,31 and (4) postoperative immunosuppression.32 When the primary tumor is removed, the tumor milieu and homeostasis within the body seem to be altered.17,26 As a result, the balance between inducers and inhibitors can be altered, leading to additional activation of circulating tumor cells and metastasis.26,33-38 Endostatin given in vivo, using a spontaneous metastasis model, is associated with a reduction in distant metastasis.39 Open rather than laparoscopic surgeries worsen the homeostatic milieu because of increased inflammatory reactions.32,40 The effects of surgery on antiangiogenic factors (ie, VEGF) have been shown in in vivo models of breast cancer following mastectomy6 and in animal models of ovarian cancer.37 If the surgery is complicated by blood transfusions or hypothermia, recurrence may be higher.17 Immunosuppressed patients are also increasingly susceptible to cancer recurrence compared to patients with an intact immune system.41 Patients with sarcoma; melanoma; myeloma; or skin, bladder, or kidney tumors have higher recurrence rates of metastasis if they are on immunosuppressive therapies.41
Experimental and clinical studies have shown that surgery inhibits T-cell, B-cell, and natural killer (NK) cell function for several days after a surgical insult.42 In addition, the production of cytokines that favor cell-mediated immunity—such as interleukin (IL)-2, IL-12, and interferon (IFN)-gamma—decreases, and the production of cytokines that interfere with cell-mediated immunity—such as IL-10—increases.17 Peak immunosuppression is thought to occur on postoperative day 3 and to provide an opportunity for the micrometastasis to grow.17 A decrease in NK cell numbers is associated with increased susceptibility to cancer or metastases after oncologic surgery.17 Additionally, there is a linear correlation between NK activity and metastatic activity.35,43
Further, the type of analgesia used plays a key role in determining immunity.35 General anesthesia (GA) is thought to suppress the immune system.44 Anesthetics may inhibit cell-mediated immunity45 or produce an alteration in the balance between the proinflammatory and antiinflammatory cytokines.46 In particular, NK cytotoxicity has been shown to be suppressed by various anesthetics. All volatile anesthetics reduce NK cell activity.47-50 Fentanyl seems to have a greater suppressive effect on NK cell activity compared to ketamine and clonidine.35 Beilin et al evaluated a group of 40 patients undergoing major surgery who were randomized to either a high-dose fentanyl regimen that included midazolam and isoflurane as necessary or a low-dose fentanyl regimen with anesthetic maintenance using nitrous oxide or isoflurane.13 In vitro NK activity was suppressed in all patients receiving fentanyl, but high-dose fentanyl therapy was associated with a slower rate of recovery of NK cell activity compared to low-dose fentanyl.13
Melamed et al compared the effects of propofol, halothane, ketamine, and thiopental on NK cell activity and metastatic spread of tumor cells in rats.50 They found that all of these agents, except propofol, reduce NK cell activity.50 Further, propofol also reduces inflammatory cytokines.50 All anesthetics except propofol increased lung metastases and tumor retention.50 Ketamine had the greatest effect on metastasis, increasing the frequency of metastasis almost 2.5-fold.50 This increase was reduced with use of a β-receptor antagonist (nadolol), a prostaglandin synthesis inhibitor (indomethacin), or both.50 Further, administration of a β-receptor agonist, prostaglandin, or both promoted metastasis of tumor cells.50 Similar conclusions were found by Sloan et al.51 In animal models, the stress-induced neuroendocrine activation from surgery had a negligible effect on the growth of the primary tumor but induced a 30-fold increase in metastasis to distant tissues.51 In these animal studies, the effect was theorized to be via β-adrenergic signaling and inhibited via the β-antagonist propranolol.51
While use of anesthetics and analgesics may suppress NK cell activity, acute pain may also suppress NK cell activity.52,53 Of note, perioperative psychological stress and anxiety impact the neuroendocrine stress response, exerting a significant effect on the microenvironment of the tumor or the micrometastasis.54,55
Role of Regional Anesthesia
RA may reduce the stress associated with surgery, reduce pain, and lead to improved neuroendocrine function and cytokine-mediated stress response. The addition of intraoperative epidural analgesia reduces the levels of cortisol, β-endorphin, and epinephrine.56 RA may inhibit neuroendocrine stress via sympathetic block.9 Bar-Yosef et al demonstrated that RA leads to reduced metastatic burden in rats inoculated with metastatic cells (MADB106) postlaparotomy.9 Their study compared anesthetized rats that underwent laparoscopic intervention to rats that did not under 3 different anesthetic regimens and found no significant difference in the number of lung metastases between the anesthetic regimens but did find a significant difference between the groups with or without surgical intervention.9 Volatile anesthetics suppress the immune system and negatively impact cancer spread. They have been shown to increase concentrations of VEGF and MMPs, known stimulators of angiogenesis, and to increase cancer cell migration in vitro.20 Further, volatile anesthetics have been shown to upregulate hypoxia-inducible factors. These factors are thought to be protective of ischemia-reperfusion injury, but they have been shown to be influential in angiogenesis and cell migration.57 In the Bar-Yosef et al study, surgery with halothane increased the number of metastases 2-fold compared to the control group.9 The addition of RA, in particular spinal anesthesia, reversed this effect.9
Cytotoxic T cells (CTCs) play an important role in the development of cancer. Patients with high CTC counts, in opposition to primary localized lung cancer, have been shown to have complete remission at 5 years, while patients with low CTC counts were less likely to survive.58 Further, tumor infiltration by CTCs has been associated with a positive prognosis in colorectal cancer.59 Ahlers et al showed that epidural analgesia in abdominal surgery was associated not only with a higher number of T-helper (Th) cells but also with a higher number of lymphocytes and preserved IFN-gamma concentrations.60 Clinically, the higher number of Th cells led to decreased liver metastasis.60 Le Cras et al showed that the ratio of Th1 to Th2 cells was higher in patients who had prostate surgery with spinal anesthesia vs GA.61
RA influences the expression of several cytokines perioperatively, including increasing IL-4 and decreasing IL-10.62 IL-4 increases the expression of Th1. Ahlers et al reached a similar conclusion and found that the ratio of Th1 to Th2 cells was increased in patients who received epidural analgesia.60 In contrast, administration of fentanyl or morphine is associated with increased plasma concentrations of IL-10, suggesting a predominant antiinflammatory and immunosuppressive profile.63,64 IL-10 reduces expression of Th1 and the presence of NK cells.65 This effect can be additive to the effect of surgery.17 Gupta et al demonstrated that morphine, a VEGF activator, stimulates endothelial cell proliferation via a mitogen-activated protein kinase.6 Further, morphine inhibits apoptosis and promotes cell-cycle progression in endothelial cells.6 Similarly, Singleton et al found that opioids induce VEGF receptor activation, resulting in endothelial cell migration that is a step for angiogenesis.66 VEGF receptor activation was inhibited by methylnaltrexone, a peripherally acting opioid antagonist.66
METHODS
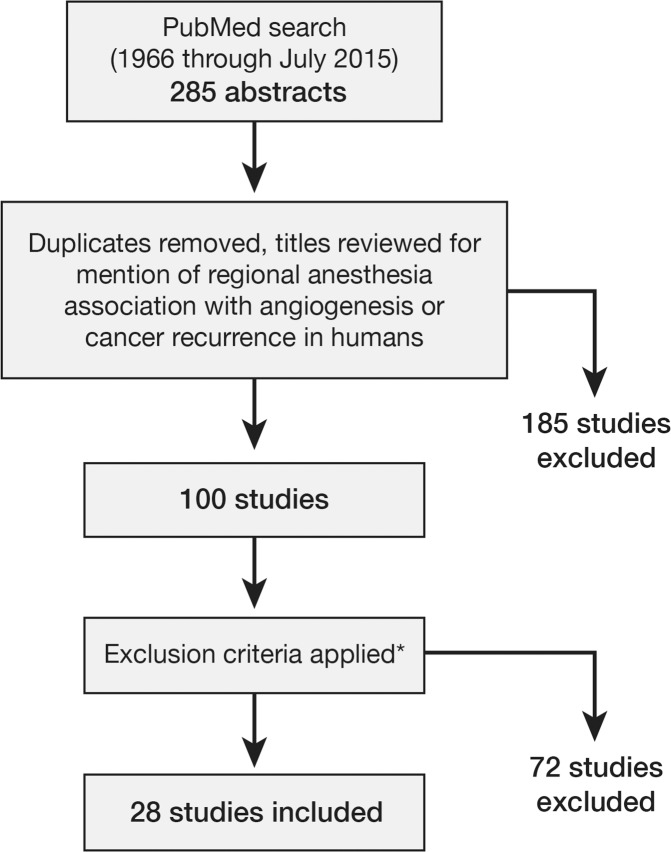
In an attempt to identify the areas in which RA may have a proven benefit on cancer progression, we performed a metaanalysis. We reviewed the literature, searching PubMed for “regional anesthesia and cancer angiogenesis” and “regional anesthesia and cancer recurrence.” After duplicates were removed, the search yielded 285 abstracts for initial review, 100 of which discussed RA association with angiogenesis or cancer recurrence in humans. We excluded reviews, metaanalyses, editorials, opinion pieces, and articles that exclusively discussed intraoperative analgesia, did not provide a comparison between GA and RA, were written in languages other than English, or were unavailable as complete articles. If our author team knew of pertinent literature that did not include specific details for inclusion in the review, the corresponding authors were contacted for additional information. We also reviewed the references from the included articles to ensure no article had been overlooked. The primary factor for inclusion in this metaanalysis was a comparison between RA and GA. Twenty-eight articles met this inclusion criterion. Figure 1 illustrates the literature search methodology. The primary outcomes were overall survival, recurrence-free survival, and biochemical recurrence-free survival compared via hazard ratios (HRs).
Figure 1.
Literature search methodology. *Reviews, metaanalyses, editorials, opinion pieces, articles that exclusively discussed intraoperative analgesia, articles that did not compare general anesthesia and regional anesthesia, non–English language articles, and papers that were not available as complete articles were excluded.
HR is a measure of how often a particular event happens in one group (treatment group) compared to how often it occurs in the control group. HR provides opportunities for articles to be evaluated in a uniform fashion. Weighted HRs were obtained by averaging the HRs from each of the individual articles. The ratios were weighted to highlight the effects of sample size in the analysis. Some of the observational studies were large and had the potential to skew the analysis significantly. An HR >1 denotes increased risk, an HR <1 denotes decreased risk, and an HR equal to 1 denotes no change in risk. Both observational studies and randomized controlled trials (RCTs) were evaluated. Results from both observational studies and RCTs were analyzed individually and together to provide a comprehensive analysis. Further, some of the studies provided both recurrence-free intervals and mortality rates that were analyzed individually because they evaluated different metrics.
RESULTS
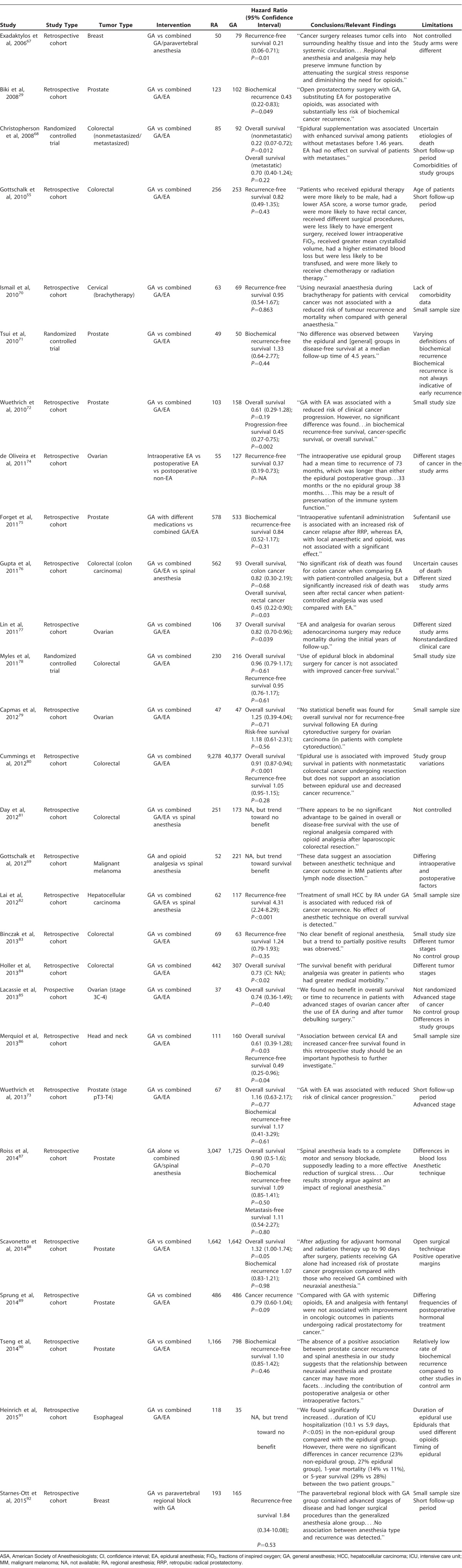
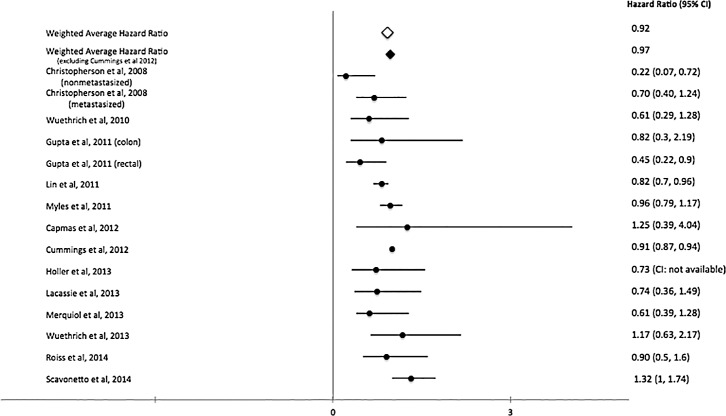
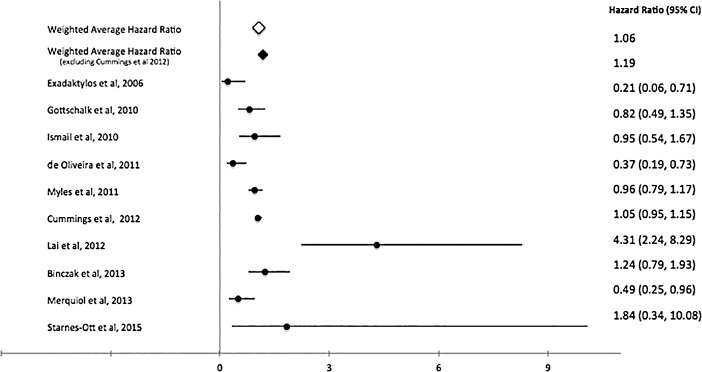
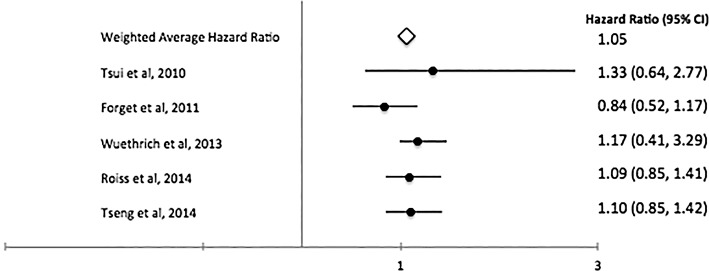
A total of 28 studies that evaluate the role of RA in cancer angiogenesis (Table 1) met our inclusion criteria.29,55,67-92 Most studies were retrospective or observational, but we identified 3 randomized controlled trials.68,71,78 The number of patients placed into the analysis from all the studies was 67,577. The pooled weighted HR for overall survival was 0.92 (Figure 2), while the weighted recurrence-free survival was 1.06 (Figure 3), and the weighted HR for biochemical recurrence-free survival was 1.05 (Figure 4). Despite no significant overall survival benefit shown with the pooled averages, we found a slight survival benefit when evaluating just the RCTs, which had weighted HRs of 0.83 and 0.88 for overall survival and recurrence-free survival, respectively. On the aggregate, no survival benefit was seen in GI, prostate, breast, and ovarian cancers (Table 2). The HRs for overall survival and recurrence-free survival in GI cancers, especially colorectal, were 0.9168,76,78,80,84 and 1.05,55,78,80,82,83 respectively. However, excluding the large observational study (Cummings et al80), we found that use of RA had limited benefit in overall survival in GI cancers (HR=0.86). When this same study was excluded in the overall analysis, the HRs for overall survival and recurrence-free survival were 0.9768,72,73,76-79,84-88 and 1.19,55,67,70,74,78,82,83,86,87,92 respectively. Further, the HRs for overall survival and biochemical recurrence-free survival in prostate cancer were 1.0672,73,87,88 and 1.05,71,73,75,87,90 respectively, and the HR for overall survival in ovarian cancer was 0.94.77,79,85
Table 1.
Summary of Available Human Trials Examining Cancer Recurrence and Regional Anesthesia
Figure 2.
Pooled and individual study hazard ratios for overall survival.68,72,73,76-80,84-88
Figure 3.
Pooled and individual study hazard ratios for recurrence-free survival. 55,67,70,74,78,80,82,83,86,92
Figure 4.
Pooled and individual study hazard ratios for biochemical recurrence-free survival.71,73,75,87,90
Table 2.
Pooled Weighted Hazard Ratios for All Cancers and by Cancer Type

DISCUSSION
For decades, opioids have been the analgesia of choice intraoperatively and postoperatively for patients with pain associated with cancer-related surgery. In addition to a potential mortality benefit, the benefits of reduced opioid usage include reducing the length of stay at hospitals and reducing side effects of opioid consumption such as respiratory depression or constipation.93 Despite some trials showing favorability, when the cancers are evaluated macroscopically (overall survival, recurrence-free survival, and biochemical recurrence-free survival), no benefit is seen.29,55,67-92
Gastrointestinal Cancers
The results from evaluating the use of RA in colorectal cancers are mixed. Key factors in the benefits of RA include stage/type of colorectal cancer, age, timing of epidural, and American Society of Anesthesiology physical status classification (ASA class).55,68,76,84 Gupta et al found that patients with rectal cancer had improved overall survival, but this benefit was not seen in patients with colon cancer.76 Further, Christopherson et al conducted a small RCT that found improved overall survival in patients with epidurals up to 1.46 years after surgery if they had colorectal cancer without metastasis.68 In the Surveillance, Epidemiology, and End Results (SEER) study, Cummings et al studied 49,655 patients who underwent surgery for colon cancer and found that those receiving epidural analgesia vs GA had no difference in recurrence-free survival but had a significant benefit in overall survival.80 The RA group had an improved 5-year overall survival (61% vs 55%).80 However, the authors found no difference in 4-year disease recurrence.80 Holler et al found that patients with ASA class III-IV had a significant difference in 5-year overall survival associated with RA compared to GA.84 However, this difference was not found in patients with ASA class I and II.84
Other studies have shown no benefit to RA. In an RCT of 446 patients, Myles et al found that the use of epidural block in abdominal surgery for colorectal cancer was not associated with improved cancer-free survival or 5-year mortality rate.78 The small sample size of the study made it challenging to find subtle differences between the groups; however, larger differences could still be found. Gottschalk et al reported similar conclusions.55 However, they found a lower risk of recurrence in the epidural group for patients aged >64 years.55 In a group of 424 patients, Day et al found no difference in overall survival when comparing RA with GA.81 Further, the length of stay was longer for patients in the epidural group at 5 days compared with 3 days for the spinal and patient-controlled analgesia group.81
In a retrospective study of 132 patients with a 17-year follow-up, Binczak et al found no statistical difference in recurrence-free survival between patients receiving bupivacaine thoracic epidural analgesia or fentanyl followed by continuous subcutaneous morphine.83 However, the long follow-up may have minimized the impact of anesthesia technique.83 While Heinrich et al did not find any direct benefits of RA for the management of esophageal cancer, they found that the reduced opioid use associated with RA led to fewer days in the intensive care unit and fewer days on mechanical ventilation, as well as reduced risk of reintubation, fewer days of antibiotics, and lower risk of perioperative anemia.91 Despite these benefits, there was no difference in cancer recurrence, tumor spreading, or overall survival in a multivariate Cox analysis associated with epidural analgesia.91
We found only 1 study that suggested a worse outcome with use of RA compared to GA. In a retrospective study, Lai et al compared the use of GA vs epidural anesthesia in patients undergoing radiofrequency ablation to treat hepatocellular carcinoma.82 Their analysis suggested that treatment of hepatocellular carcinoma by radiofrequency ablation under GA is associated with reduced risk of cancer recurrence, but the authors found no effect of anesthetic technique on overall survival.82 However, this study is different from all the above studies in that it was evaluating hepatocellular carcinoma, which is pathologically different from colorectal cancer.82
Prostate Cancer
Survival outcomes in patients with prostate cancer are not clear because many patients live for extended periods after diagnosis. As a result, many studies involving prostate cancer use biochemical recurrence as the endpoint, and those that use overall survival should use dual study arms to compare outcomes accurately. Biochemical recurrence is defined as either an increase of prostate antigen from its postoperative nadir or >0.2 ng/mL.73 Biochemical recurrence is not a perfect endpoint because it does not translate into cancer-specific survival.94 In addition, many studies focus on cancers in the advanced stages to help discern the survival benefit. No randomized trials are available, and the identified studies are mostly observational and retrospective.
We identified 3 studies that showed benefits in different markers of clinical significance. Biki et al found that in patients undergoing open prostatectomy surgery with GA, substitution of postoperative opioids with epidural analgesia was associated with a 57% reduction in biochemical cancer recurrence (95% confidence interval [CI] 17%-78%).29 In a small observational study of 261 patients with approximately 50% having invasive disease, Wuethrich et al found that epidural analgesia resulted in better clinical progression-free survival but only found a small difference in biochemical recurrence-free survival and no difference in overall survival.72 However, the study was underpowered to detect small changes, and the P values were high (P=0.19 for overall survival). Of note, patients in the GA group were given ketorolac every 8 hours, which may have confounded results.72 COX inhibitors have been shown to induce apoptosis in prostate cancer cell lines.23 In a matched study, Scavonetto et al found benefit in the use of epidural analgesia via decreased systemic progression of the cancer and improved overall survival.88 Further, although not statistically significant on a multivariate analysis, prostate cancer death was also reduced with RA.88
We identified 6 studies that showed no benefit for RA in prostate cancer surgery. Tsui et al found no difference between epidural and control groups in disease-free survival at a median follow-up time of 4.5 years in their secondary analysis of patients undergoing radical prostatectomy.71 In another study, Wuethrich et al found no difference in biochemical recurrence-free, local and distance recurrence-free, and overall survival in patients with invasive prostate cancer undergoing radical prostatectomy with combined GA and epidural analgesia or GA alone.73 However, Wuethrich et al found a reduced risk of clinical cancer progression.73 As in the Tsui et al study,71 patients in the GA group in the Wuethrich et al study received ketorolac, which may have confounded the results.73 In a study of 1,111 patients undergoing radical prostatectomy, Forget et al found no significant association between epidural analgesia and risk of cancer relapse.75 Furthermore, the authors found an increased risk associated with the use of intravenous sufentanil with an HR of 7.78 (95% CI 5.79-9.78). However, the follow-up time in this study was fairly short, approximately 3 years, and patients often received multimodal analgesia, which made individual evaluation challenging.75 Another large study (4,772 patients) by Roiss et al compared patients undergoing radical prostatectomy with either GA alone or GA with spinal anesthesia and found no difference in overall survival or biochemical recurrence-free survival.87 However, this study used propensity-scoring matching because of differences in prostate specific antigens, tumor grades, and histology.87 Similarly, Sprung et al found no benefit with the use of epidural analgesia.89 Sprung et al performed neuraxial analgesia without utilizing volatile anesthetics. Volatile anesthetics have been thought to affect cancer recurrence because of their inhibition of NK cells. Despite this theoretical association, no differences in outcomes were seen. Tseng et al used spinal anesthesia to look for potential cancer or non–malignancy-associated benefits for radical prostatectomy.90 The authors found no benefit in biochemical recurrence after a 4- to 5-year follow-up period.90
Breast Cancer
Exadaktylos et al performed one of the first studies evaluating the benefit of neuraxial anesthesia.67 In a retrospective analysis of 129 patients with breast cancer who underwent mastectomy, the researchers found that paravertebral anesthesia and analgesia for breast cancer surgery reduced the risk of recurrence or metastasis during the initial 3 years of follow-up compared to GA alone.67 The authors found no significant differences between the 2 study arms. The recurrence-free survival rate was 94% (95% CI 87%-100%) in patients who received paravertebral analgesia compared to 82% (95% CI 74%-91%) in GA patients at 24 months.67 At 36 months, this difference became more pronounced with a recurrence-free survival of 94% (95% CI 87%-100%) in patients who received paravertebral analgesia and 77% (95% CI 68%-87%) in patients who did not receive paravertebral analgesia.67 However, in another retrospective study, Starnes-Ott and colleagues found that in a group of 358 patients, anesthetic choice did not result in a significant difference in recurrence-free survival at the 28-month mark.92 Despite the similar outcome, the paravertebral block group included patients with more advanced stages of cancer, more invasive treatments, longer surgery times, and decreased body mass index (BMI) compared to the GA group.92 BMI has been associated with increased risk of recurrence and death from cancer.95 These differences may confound the results. Schnabel et al published a metaanalysis of RCTs analyzing efficacy and safety of paravertebral blocks for breast cancer surgery.96 They concluded that a reduced need for postoperative morphine among the group of patients undergoing surgery with paravertebral block correlated with a lower recurrence of breast cancer.96
Because of benefits in terms of overall survival, a large multicenter, international trial (NCT00418457) is underway in patients with stage I-III breast cancer undergoing mastectomy with or without axillary dissection.97 While this trial may take years to complete, some of the initial data based on tissue samples have shown promise. Early clinical results from Wu et al show an analgesic benefit in patients undergoing breast cancer surgery.98 RA compared with GA resulted in a greater percentage decrease in postoperative compared to preoperative concentrations of IL-1B (proinflammatory), an increase in the concentrations of IL-10 (antiinflammatory), and an attenuation in MMPs involved in tumor migration and metastasis.20 Deegan et al showed that paravertebral anesthesia alters some of the proinflammatory cytokines involved in regulating perioperative cancer immunity.99 In addition, the authors found reduced proliferation of the cancer cell line associated with the use of RA.99 In a study by Desmond et al, excised breast cancer specimens from the RA group demonstrated increased infiltration of NK and Th cells compared to the GA group.100 Further, RA has been shown to lead to a smaller increase in VEGF-C compared to GA.101 VEGF-C has been shown to promote angiogenesis and can be overexpressed in breast cancer.101
Ovarian Cancer
We identified 4 retrospective studies that compared the effects of RA and GA in patients with ovarian cancer. Lin et al77 and de Oliveira et al74 found a benefit from the intraoperative use of epidural anesthesia compared to GA. Lin et al, in a sample of 143 patients, found that 3-year and 5-year overall survival rates were 79% and 61% in the epidural group compared to 58% and 49% in the GA group, respectively.77 After adjusting for various factors such as carcinoma antigen 125 (CA-125) concentration, histology, residual tumor, and lymphatic metastasis, GA was associated with an HR of 1.214 (95% CI 1.075-1.431, P=0.043).77 De Oliveira et al, in a sample of 182 patients, found that the group with intraoperative and postoperative epidural use had a significantly greater time to recurrence compared with the GA group.74 Further, the intraoperative epidural group also had an increased mean time to death compared with the GA and postoperative epidural group (mean time 96 months in the intraoperative epidural group vs 71 months and 70 months in the postoperative nonepidural group and in the postoperative epidural group, respectively).74
Other studies have found no benefit for cancer recurrence. In a group of 94 patients with advanced ovarian cancer, Capmas et al found no improvement in overall survival or recurrence-free survival with the use of postoperative RA.79 Further, roughly 44% of patients in the Capmas et al study received blood transfusions, which has been shown to increase the risk of recurrence.79 Lacassie et al evaluated a group of 80 patients matched using propensity scoring and also found no benefit in overall survival or time to recurrence.85
Other Cancers
RA has also been studied in other cancers including malignant melanoma, cervical cancer, and laryngeal cancer. In a retrospective review of 4,329 patients, Schlagenhauff et al found that RA is associated with longer survival in surgeries associated with malignant melanoma compared to GA.102 Further, Gottschalk et al found a nonsignificant trend toward longer overall survival in patients undergoing spinal anesthesia (96 vs 70 months, P=0.087).69 Ismail et al found no benefit in the use of epidural analgesia in patients undergoing brachytherapy for cervical cancer.70 In contrast to open surgery, brachytherapy involves less tissue manipulation and shorter procedure duration, factors that can affect cancer recurrence.70
Merquiol et al, evaluating a group of 271 patients undergoing surgery for laryngeal and hypopharyngeal cancer, found that combined GA and epidural anesthesia with postoperative epidural analgesia resulted in significantly improved cancer-free survival and overall survival.86
LIMITATIONS
This study has multiple limitations, the most important being that many of the studies included are retrospective. While some of these studies used several factors to match patients in the study arms, some factors may still be unaccounted for. In studies that evaluate cancers in advanced stages, the mortality rate is high at baseline. Consequently, defining overall survival and measuring recurrence-free survival or biochemical recurrence-free survival in these populations can be difficult. Patients with advanced-stage cancer have a high likelihood of dying from diseases that are secondary to the cancer but not directly attributable to the cancer, limiting the ability to calculate survival benefits. In studies that use recurrence as the primary outcome, different criteria are often used to define recurrence. Many types of recurrence exist, and comparing the different types can be challenging. Some types of recurrence are not associated with overall survival, diminishing its prognostic value. Further, many studies evaluate patients during a period of time that is too short to reach significant conclusions. Numerous studies lacked significant power to draw strong conclusions. Some studies included in this analysis were performed on particular populations, so translating some of these findings to the general population may be challenging. Finally, in some studies, patients received multimodal analgesia, which made evaluating the analgesia techniques individually especially challenging.
CONCLUSION
RA has been shown to have no overall benefit in overall survival, recurrence-free survival, and biochemical recurrence-free survival. However, numerous individual studies have shown some benefit, and results have been controversial. Different mechanisms have been proposed to explain this benefit but none has been proven. Thus, more work is needed to critically evaluate the role of RA in a prospective, randomized fashion. Clinical trials are underway across the world to evaluate the impact of RA. RA has the potential to alter the way cancer pain is managed and could significantly impact morbidity and mortality.
ACKNOWLEDGMENTS
The authors have no financial or proprietary interest in the subject matter of this article.
This article meets the Accreditation Council for Graduate Medical Education and the American Board of Medical Specialties Maintenance of Certification competencies for Patient Care, Medical Knowledge, and Practice-Based Learning and Improvement.
REFERENCES
- 1. Roy S, Wang J, Kelschenbach J, Koodie L, Martin J. . Modulation of immune function by morphine: implications for susceptibility to infection. J Neuroimmune Pharmacol. 2006. March; 1 1: 77- 89. [DOI] [PubMed] [Google Scholar]
- 2. Vaghari BA, Ahmed OI, Wu CL. . Regional anesthesia-analgesia: relationship to cancer recurrence and infection. Anesthesiol Clin. 2014. December; 32 4: 841- 851. 10.1016/j.anclin.2014.08.004. [DOI] [PubMed] [Google Scholar]
- 3. Sacerdote P, Bianchi M, Gaspani L, et al. The effects of tramadol and morphine on immune responses and pain after surgery in cancer patients. Anesth Analg. 2000. June; 90 6: 1411- 1414. [DOI] [PubMed] [Google Scholar]
- 4. Fimiani C, Mattocks D, Cavani F, et al. Morphine and anandamide stimulate intracellular calcium transients in human arterial endothelial cells: coupling to nitric oxide release. Cell Signal. 1999. March; 11 3: 189- 193. [DOI] [PubMed] [Google Scholar]
- 5. Grivennikov SI, Greten FR, Immunity Karin M., . inflammation, and cancer. Cell. 2010. March 19; 140 6: 883- 899. 10.1016/j.cell.2010.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Gupta K, Kshirsagar S, Chang L, et al. Morphine stimulates angiogenesis by activating proangiogenic and survival-promoting signaling and promotes breast tumor growth. Cancer Res. 2002. August 1; 62 15: 4491- 4498. [PubMed] [Google Scholar]
- 7. Hsiao PN, Chang MC, Cheng WF, et al. Morphine induces apoptosis of human endothelial cells through nitric oxide and reactive oxygen species pathways. Toxicology. 2009. February 4; 256 1-2: 83- 91. 10.1016/j.tox.2008.11.015. [DOI] [PubMed] [Google Scholar]
- 8. Stefano GB, Hartman A, Bilfinger TV, et al. Presence of the mu3 opiate receptor in endothelial cells: coupling to nitric oxide production and vasodilation. J Biol Chem. 1995. December 22; 270 51: 30290- 30293. [DOI] [PubMed] [Google Scholar]
- 9. Bar-Yosef S, Melamed R, Page GG, Shakhar G, Shakhar K, Ben-Eliyahu S. . Attenuation of the tumor-promoting effect of surgery by spinal blockade in rats. Anesthesiology. 2001. June; 94 6: 1066- 1073. [DOI] [PubMed] [Google Scholar]
- 10. Lahat A, Ben-Horin S, Lang A, Fudim E, Picard O, Chowers Y. . Lidocaine down-regulates nuclear factor-kappaB signalling and inhibits cytokine production and T cell proliferation. Clin Exp Immunol. 2008. May; 152 2: 320- 327. 10.1111/j.1365-2249.2008.03636.x. Erratum in: Clin Exp Immunol . 2008 Aug;153(2):307. Horin, SB [corrected to Ben-Horin, S]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Krog J, Hokland M, Ahlburg P, Parner E, Tønnesen E. . Lipid solubility- and concentration-dependent attenuation of in vitro natural killer cell cytotoxicity by local anesthetics. Acta Anaesthesiol Scand. 2002. August; 46 7: 875- 881. [DOI] [PubMed] [Google Scholar]
- 12. Martinsson T. . Ropivacaine inhibits serum-induced proliferation of colon adenocarcinoma cells in vitro. J Pharmacol Exp Ther. 1999. February; 288 2: 660- 664. [PubMed] [Google Scholar]
- 13. Beilin B, Shavit Y, Hart J, et al. Effects of anesthesia based on large versus small doses of fentanyl on natural killer cell cytotoxicity in the perioperative period. Anesth Analg. 1996. March; 82 3: 492- 497. [DOI] [PubMed] [Google Scholar]
- 14. Coussens LM, Werb Z. . Inflammation and cancer. Nature. 2002; 420 6917: 860- 867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Almog N, Ma L, Raychowdhury R, et al. Transcriptional switch of dormant tumors to fast-growing angiogenic phenotype. Cancer Res. 2009; 69 3: 836- 844. [DOI] [PubMed] [Google Scholar]
- 16. Aguirre-Ghiso JA. . The problem of cancer dormancy: understanding the basic mechanisms and identifying therapeutic opportunities. Cell Cycle. 2006; 5 16: 1740- 1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Snyder GL, Greenberg S. . Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br J Anaesth. 2010. August; 105 2: 106- 115. 10.1093/bja/aeq164. [DOI] [PubMed] [Google Scholar]
- 18. Langley RR, Fidler IJ. . The seed and soil hypothesis revisited—the role of tumor-stroma interactions in metastasis to different organs. Int J Cancer. 2011. June 1; 128 11: 2527- 2535. 10.1002/ijc.26031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Yamamizu K, Hamada Y, Narita M. . κ Opioid receptor ligands regulate angiogenesis in development and in tumours. Br J Pharmacol. 2015. January; 172 2: 268- 276. 10.1111/bph.12573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Deegan CA, Murray D, Doran P, et al. Anesthetic technique and the cytokine and matrix metalloproteinase response to primary breast cancer surgery. Reg Anesth Pain Med. 2010. Nov-Dec; 35 6: 490- 495. 10.1097/AAP.0b013e3181ef4d05. [DOI] [PubMed] [Google Scholar]
- 21. Mammoto T, Higashiyama S, Mukai M, et al. Infiltration anesthetic lidocaine inhibits cancer cell invasion by modulating ectodomain shedding of heparin-binding epidermal growth factor-like growth factor (HB-EGF). J Cell Physiol. 2002. September; 192 3: 351- 358. [DOI] [PubMed] [Google Scholar]
- 22. Ash SA, Buggy DJ. . Does regional anaesthesia and analgesia or opioid analgesia influence recurrence after primary cancer surgery? An update of available evidence. Best Pract Res Clin Anaesthesiol. 2013. December; 27 4: 441- 456. 10.1016/j.bpa.2013.10.005. [DOI] [PubMed] [Google Scholar]
- 23. Cha YI, DuBois RN. . NSAIDs and cancer prevention: targets downstream of COX-2. Annu Rev Med. 2007; 58: 239- 252. [DOI] [PubMed] [Google Scholar]
- 24. Melamed R, Rosenne E, Shakhar K, Schwartz Y, Abudarham N, Ben-Eliyahu S. . Marginating pulmonary-NK activity and resistance to experimental tumor metastasis: suppression by surgery and the prophylactic use of a beta-adrenergic antagonist and a prostaglandin synthesis inhibitor. Brain Behav Immun. 2005. March; 19 2: 114- 126. [DOI] [PubMed] [Google Scholar]
- 25. Farooqui M, Li Y, Rogers T, et al. COX-2 inhibitor celecoxib prevents chronic morphine-induced promotion of angiogenesis, tumour growth, metastasis and mortality, without compromising analgesia. Br J Cancer. 2007. December 3; 97 11: 1523- 1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Demicheli R, Terenziani M, Valagussa P, Moliterni A, Zambetti M, Bonadonna G. . Local recurrences following mastectomy: support for the concept of tumor dormancy. J Natl Cancer Inst. 1994. January 5; 86 1: 45- 48. [DOI] [PubMed] [Google Scholar]
- 27. Yamaguchi K, Takagi Y, Aoki S, Futamura M, Saji S. . Significant detection of circulating cancer cells in the blood by reverse transcriptase-polymerase chain reaction during colorectal cancer resection. Ann Surg. 2000; 232 1: 58- 65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Holmgren L, O'Reilly MS, Folkman J. . Dormancy of micrometastases: balanced proliferation and apoptosis in the presence of angiogenesis suppression. Nat Med. 1995; 1 2: 149- 153. [DOI] [PubMed] [Google Scholar]
- 29. Biki B, Mascha E, Moriarty DC, Fitzpatrick JM, Sessler DI, Buggy DJ. . Anesthetic technique for radical prostatectomy surgery affects cancer recurrence: a retrospective analysis. Anesthesiology. 2008. August; 109 2: 180- 187. 10.1097/ALN.0b013e31817f5b73. [DOI] [PubMed] [Google Scholar]
- 30. Ikeda M, Furukawa H, Imamura H, et al. Surgery for gastric cancer increases plasma levels of vascular endothelial growth factor and von Willebrand factor. Gastric Cancer. 2002; 5 3: 137- 141. [DOI] [PubMed] [Google Scholar]
- 31. Maniwa Y, Okada M, Ishii N, Kiyooka K. . Vascular endothelial growth factor increased by pulmonary surgery accelerates the growth of micrometastases in metastatic lung cancer. Chest. 1998; 114 6: 1668- 1675. [DOI] [PubMed] [Google Scholar]
- 32. Vittimberga FJ Jr, Foley DP, Meyers WC, Callery MP. . Laparoscopic surgery and the systemic immune response. Ann Surg. 1998. March; 227 3: 326- 334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Tsuchiya Y, Sawada S, Yoshioka I, et al. Increased surgical stress promotes tumor metastasis. Surgery. 2003. May; 133 5: 547- 555. [DOI] [PubMed] [Google Scholar]
- 34. Demicheli R, Biganzoli E, Boracchi P, Greco M, Retsky MW. . Recurrence dynamics does not depend on the recurrence site. BCR. 2008; 10 5: R83 10.1186/bcr2152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Forget P, Collet V, Lavand'homme P, De Kock M. . Does analgesia and condition influence immunity after surgery? Effects of fentanyl, ketamine and clonidine on natural killer activity at different ages. Eur J Anaesthesiol. 2010. March; 27 3: 233- 240. 10.1097/EJA.0b013e32832d540e. [DOI] [PubMed] [Google Scholar]
- 36. Retsky M. . New concepts in breast cancer emerge from analyzing clinical data using numerical algorithms. Int J Environ Res Public Health. 2009. January; 6 1: 329- 348. 10.3390/ijerph6010347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Lee JW, Shahzad MM, Lin YG, et al. Surgical stress promotes tumor growth in ovarian carcinoma. Clin Cancer Res. 2009. April 15; 15 8: 2695- 2702. 10.1158/1078-0432.CCR-08-2966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Thaker PH, Han LY, Kamat AA, et al. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat Med. 2006. August; 12 8: 939- 944. [DOI] [PubMed] [Google Scholar]
- 39. Wang HL, Ning T, Li M, et al. Effect of endostatin on preventing postoperative progression of distant metastasis in a murine lung cancer model. Tumori. 2011. Nov-Dec; 97 6: 787- 793. 10.1700/1018.11097. [DOI] [PubMed] [Google Scholar]
- 40. Lacy AM, García-Valdecasas JC, Delgado S, et al. Laparoscopy-assisted colectomy versus open colectomy for treatment of non-metastatic colon cancer: a randomised trial. Lancet. 2002. June 29; 359 9325: 2224- 2229. [DOI] [PubMed] [Google Scholar]
- 41. Penn I. . The effect of immunosuppression on pre-existing cancers. Transplantation. 1993. April; 55 4: 742- 747. [DOI] [PubMed] [Google Scholar]
- 42. Angele MK, Faist E. . Clinical review: immunodepression in the surgical patient and increased susceptibility to infection. Crit Care. 2002. August; 6 4: 298- 305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Ben-Eliyahu S, Page GG, Yirmiya R, Shakhar G. . Evidence that stress and surgical interventions promote tumor development by suppressing natural killer cell activity. Int J Cancer. 1999. March 15; 80 6: 880- 888. [DOI] [PubMed] [Google Scholar]
- 44. Tavare AN, Perry NJ, Benzonana LL, Takata M, Ma D. . Cancer recurrence after surgery: direct and indirect effects of anesthetic agents. Int J Cancer. 2012. March 15; 130 6: 1237- 1250. 10.1002/ijc.26448. [DOI] [PubMed] [Google Scholar]
- 45. Galley HF, DiMatteo MA, Webster NR. . Immunomodulation by anaesthetic, sedative and analgesic agents: does it matter? Intensive Care Med. 2000. March; 26 3: 267- 274. [DOI] [PubMed] [Google Scholar]
- 46. Kelbel I, Weiss M. . Anaesthetics and immune function. Curr Opin Anaesthesiol. 2001. December; 14 6: 685- 691. [DOI] [PubMed] [Google Scholar]
- 47. Cassinello F, Prieto I, del Olmo M, Rivas S, Strichartz GR. . Cancer surgery: how may anesthesia influence outcome? J Clin Anesth. 2015. May; 27 3: 262- 272. 10.1016/j.jclinane.2015.02.007. [DOI] [PubMed] [Google Scholar]
- 48. Kushida A, Inada T, Shingu K. . Enhancement of antitumor immunity after propofol treatment in mice. Immunopharmacol Immunotoxicol. 2007; 29 3-4: 477- 486. [DOI] [PubMed] [Google Scholar]
- 49. Ke JJ, Zhan J, Feng XB, Wu Y, Rao Y, Wang YL. . A comparison of the effect of total intravenous anaesthesia with propofol and remifentanil and inhalational anaesthesia with isoflurane on the release of pro- and anti-inflammatory cytokines in patients undergoing open cholecystectomy. Anaesth Intensive Care. 2008. January; 36 1: 74- 78. [DOI] [PubMed] [Google Scholar]
- 50. Melamed R, Bar-Yosef S, Shakhar G, Shakhar K, Ben-Eliyahu S. . Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: mediating mechanisms and prophylactic measures. Anesth Analg. 2003. November; 97 5: 1331- 1339. [DOI] [PubMed] [Google Scholar]
- 51. Sloan EK, Priceman SJ, Cox BF, et al. The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res. 2010. September 15; 70 18: 7042- 7052. 10.1158/0008-5472.CAN-10-0522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Sacerdote P, Manfredi B, Bianchi M, Panerai AE. . Intermittent but not continuous inescapable footshock stress affects immune responses and immunocyte beta-endorphin concentrations in the rat. Brain Behav Immun. 1994. September; 8 3: 251- 260. [DOI] [PubMed] [Google Scholar]
- 53. Shavit Y, Martin FC, Yirmiya R, et al. Effects of a single administration of morphine or footshock stress on natural killer cell cytotoxicity. Brain Behav Immun. 1987. December; 1 4: 318- 328. [DOI] [PubMed] [Google Scholar]
- 54. Sood AK, Bhatty R, Kamat AA, et al. Stress hormone-mediated invasion of ovarian cancer cells. Clin Cancer Res. 2006. January 15; 12 2: 369- 375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Gottschalk A, Ford JG, Regelin CC, et al. Association between epidural analgesia and cancer recurrence after colorectal cancer surgery. Anesthesiology. 2010. July; 113 1: 27- 34. 10.1097/ALN.0b013e3181de6d0d. [DOI] [PubMed] [Google Scholar]
- 56. Forget P, De Kock M. . Could anaesthesia, analgesia and sympathetic modulation affect neoplasic recurrence after surgery? A systematic review centred over the modulation of natural killer cells activity [in French]. Ann Fr Anesth Reanim. 2009. September; 28 9: 751- 768. 10.1016/j.annfar.2009.07.078. [DOI] [PubMed] [Google Scholar]
- 57. Zhao H, Iwasaki M, Yang J, Savage S, Ma D. . Hypoxia-inducible factor-1: a possible link between inhalational anesthetics and tumor progression? Acta Anaesthesiol Taiwan. 2014. June; 52 2: 70- 76. 10.1016/j.aat.2014.05.008. [DOI] [PubMed] [Google Scholar]
- 58. Uchida A, Kariya Y, Okamoto N, Sugie K, Fujimoto T, Yagita M. . Prediction of postoperative clinical course by autologous tumor-killing activity in lung cancer patients. J Natl Cancer Inst. 1990. November 7; 82 21: 1697- 1701. [DOI] [PubMed] [Google Scholar]
- 59. Pagès F, Berger A, Camus M, et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med. 2005. December 22; 353 25: 2654- 2666. [DOI] [PubMed] [Google Scholar]
- 60. Ahlers O, Nachtigall I, Lenze J, et al. Intraoperative thoracic epidural anaesthesia attenuates stress-induced immunosuppression in patients undergoing major abdominal surgery. Br J Anaesth. 2008. December; 101 6: 781- 787. 10.1093/bja/aen287. [DOI] [PubMed] [Google Scholar]
- 61. Le Cras AE, Galley HF, Webster NR. . Spinal but not general anesthesia increases the ratio of T helper 1 to T helper 2 cell subsets in patients undergoing transurethral resection of the prostate. Anesth Analg. 1998. December; 87 6: 1421- 1425. [DOI] [PubMed] [Google Scholar]
- 62. Moselli NM, Baricocchi E, Ribero D, Sottile A, Suita L, Debernardi F. . Intraoperative epidural analgesia prevents the early proinflammatory response to surgical trauma: results from a prospective randomized clinical trial of intraoperative epidural versus general analgesia. Ann Surg Oncol. 2011. October; 18 10: 2722- 2731. 10.1245/s10434-011-1700-9. [DOI] [PubMed] [Google Scholar]
- 63. Kim MH, Hahm TS. . Plasma levels of interleukin-6 and interleukin-10 are affected by ketorolac as an adjunct to patient-controlled morphine after abdominal hysterectomy. Clin J Pain. 2001. March; 17 1: 72- 77. [DOI] [PubMed] [Google Scholar]
- 64. Yardeni IZ, Beilin B, Mayburd E, Alcalay Y, Bessler H. . Relationship between fentanyl dosage and immune function in the postoperative period. J Opioid Manag. 2008. Jan-Feb; 4 1: 27- 33. [DOI] [PubMed] [Google Scholar]
- 65. Buckley A, McQuaid S, Johnson P, Buggy DJ. . Effect of anaesthetic technique on the natural killer cell anti-tumour activity of serum from women undergoing breast cancer surgery: a pilot study. Br J Anaesth. 2014. July; 113 Suppl 1:i56-i62. 10.1093/bja/aeu200. [DOI] [PubMed]
- 66. Singleton PA, Lingen MW, Fekete MJ, Garcia JG, Moss J. . Methylnaltrexone inhibits opiate and VEGF-induced angiogenesis: role of receptor transactivation. Microvasc Res. 2006. Jul-Sep; 72 1-2: 3- 11. [DOI] [PubMed] [Google Scholar]
- 67. Exadaktylos AK, Buggy DJ, Moriarty DC, Mascha E, Sessler DI. . Can anesthetic technique for primary breast cancer surgery affect recurrence or metastasis? Anesthesiology. 2006. October; 105 4: 660- 664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Christopherson R, James KE, Tableman M, Marshall P, Johnson FE. . Long-term survival after colon cancer surgery: a variation associated with choice of anesthesia. Anesth Analg. 2008. July; 107 1: 325- 332. 10.1213/ane.0b013e3181770f55. [DOI] [PubMed] [Google Scholar]
- 69. Gottschalk A, Brodner G, Van Aken HK, Ellger B, Althaus S, Schulze HJ. . Can regional anaesthesia for lymph-node dissection improve the prognosis in malignant melanoma? Br J Anaesth. 2012. August; 109 2: 253- 259. 10.1093/bja/aes176. [DOI] [PubMed] [Google Scholar]
- 70. Ismail H, Ho KM, Narayan K, Kondalsamy-Chennakesavan S. . Effect of neuraxial anaesthesia on tumour progression in cervical cancer patients treated with brachytherapy: a retrospective cohort study. Br J Anaesth. 2010. August; 105 2: 145- 149. 10.1093/bja/aeq156. [DOI] [PubMed] [Google Scholar]
- 71. Tsui BC, Rashiq S, Schopflocher D, et al. Epidural anesthesia and cancer recurrence rates after radical prostatectomy. Can J Anaesth. 2010. February; 57 2: 107- 112. 10.1007/s12630-009-9214-7. [DOI] [PubMed] [Google Scholar]
- 72. Wuethrich PY, Hsu Schmitz SF, Kessler TM, et al. Potential influence of the anesthetic technique used during open radical prostatectomy on prostate cancer-related outcome: a retrospective study. Anesthesiology. 2010. September; 113 3: 570- 576. 10.1097/ALN.0b013e3181e4f6ec. [DOI] [PubMed] [Google Scholar]
- 73. Wuethrich PY, Thalmann GN, Studer UE, Burkhard FC. . Epidural analgesia during open radical prostatectomy does not improve long-term cancer-related outcome: a retrospective study in patients with advanced prostate cancer. PLoS One. 2013. August 19; 8 8: e72873 doi: 10.1371/journal.pone.0072873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. de Oliveira GS Jr, Ahmad S, Schink JC, Singh DK, Fitzgerald PC, McCarthy RJ. . Intraoperative neuraxial anesthesia but not postoperative neuraxial analgesia is associated with increased relapse-free survival in ovarian cancer patients after primary cytoreductive surgery. Reg Anesth Pain Med. 2011. May-Jun; 36 3: 271- 277. 10.1097/AAP.0b013e318217aada. [DOI] [PubMed] [Google Scholar]
- 75. Forget P, Tombal B, Scholtès JL, et al. Do intraoperative analgesics influence oncological outcomes after radical prostatectomy for prostate cancer? Eur J Anaesthesiol. 2011. December; 28 12: 830- 835. 10.1097/EJA.0b013e32834b7d9a. [DOI] [PubMed] [Google Scholar]
- 76. Gupta A, Björnsson A, Fredriksson M, Hallböök O, Eintrei C. . Reduction in mortality after epidural anaesthesia and analgesia in patients undergoing rectal but not colonic cancer surgery: a retrospective analysis of data from 655 patients in central Sweden. Br J Anaesth. 2011. August; 107 2: 164- 170. 10.1093/bja/aer100. [DOI] [PubMed] [Google Scholar]
- 77. Lin L, Liu C, Tan H, Ouyang H, Zhang Y, Zeng W. . Anaesthetic technique may affect prognosis for ovarian serous adenocarcinoma: a retrospective analysis. Br J Anaesth. 2011. June; 106 6: 814- 822. 10.1093/bja/aer055. [DOI] [PubMed] [Google Scholar]
- 78. Myles PS, Peyton P, Silbert B, Hunt J, Rigg JR, Sessler DI;. ANZCA Trials Group Investigators. Perioperative epidural analgesia for major abdominal surgery for cancer and recurrence-free survival: randomised trial. BMJ. 2011. Mar 29;342:d1491. 10.1136/bmj.d1491. [DOI] [PubMed]
- 79. Capmas P, Billard V, Gouy S, et al. Impact of epidural analgesia on survival in patients undergoing complete cytoreductive surgery for ovarian cancer. Anticancer Res. 2012. April; 32 4: 1537- 1442. [PubMed] [Google Scholar]
- 80. Cummings KC 3rd, Xu F, Cummings LC, Cooper GS. . A comparison of epidural analgesia and traditional pain management effects on survival and cancer recurrence after colectomy: a population-based study. Anesthesiology. 2012. April; 116 4: 797- 806. 10.1097/ALN.0b013e31824674f6. [DOI] [PubMed] [Google Scholar]
- 81. Day A, Smith R, Jourdan I, Fawcett W, Scott M, Rockall T. . Retrospective analysis of the effect of postoperative analgesia on survival in patients after laparoscopic resection of colorectal cancer. Br J Anaesth. 2012. August; 109 2: 185- 190. 10.1093/bja/aes106. [DOI] [PubMed] [Google Scholar]
- 82. Lai R, Peng Z, Chen D, et al. The effects of anesthetic technique on cancer recurrence in percutaneous radiofrequency ablation of small hepatocellular carcinoma. Anesth Analg. 2012. February; 114 2: 290- 296. 10.1213/ANE.0b013e318239c2e3. Erratum in: Anesth Analg . 2013 Jan;116(1):266. [DOI] [PubMed] [Google Scholar]
- 83. Binczak M, Tournay E, Billard V, Rey A, Jayr C. . Major abdominal surgery for cancer: does epidural analgesia have a long-term effect on recurrence-free and overall survival? Ann Fr Anesth Reanim. 2013. May; 32 5: e81- e88. 10.1016/j.annfar.2013.02.027. [DOI] [PubMed] [Google Scholar]
- 84. Holler JP, Ahlbrandt J, Burkhardt E, et al. Peridural analgesia may affect long-term survival in patients with colorectal cancer after surgery (PACO-RAS-Study): an analysis of a cancer registry. Ann Surg. 2013. December; 258 6: 989- 993. 10.1097/SLA.0b013e3182915f61. [DOI] [PubMed] [Google Scholar]
- 85. Lacassie HJ, Cartagena J, Brañes J, Assel M, Echevarría GC. . The relationship between neuraxial anesthesia and advanced ovarian cancer-related outcomes in the Chilean population. Anesth Analg. 2013. September; 117 3: 653- 660. 10.1213/ANE.0b013e3182a07046. [DOI] [PubMed] [Google Scholar]
- 86. Merquiol F, Montelimard AS, Nourissat A, Molliex S, Zufferey PJ. . Cervical epidural anesthesia is associated with increased cancer-free survival in laryngeal and hypopharyngeal cancer surgery: a retrospective propensity-matched analysis. Reg Anesth Pain Med. 2013. Sep-Oct; 38 5: 398- 402. 10.1097/AAP.0b013e31829cc3fb. [DOI] [PubMed] [Google Scholar]
- 87. Roiss M, Schiffmann J, Tennstedt P, et al. Oncological long-term outcome of 4772 patients with prostate cancer undergoing radical prostatectomy: does the anaesthetic technique matter? Eur J Surg Oncol. 2014. December; 40 12: 1686- 1692. 10.1016/j.ejso.2014.02.223. [DOI] [PubMed] [Google Scholar]
- 88. Scavonetto F, Yeoh TY, Umbreit EC, et al. Association between neuraxial analgesia, cancer progression, and mortality after radical prostatectomy: a large, retrospective matched cohort study. Br J Anaesth. 2014. July; 113 Suppl 1:i95-i102. 10.1093/bja/aet467. [DOI] [PMC free article] [PubMed]
- 89. Sprung J, Scavonetto F, Yeoh TY, et al. Outcomes after radical prostatectomy for cancer: a comparison between general anesthesia and epidural anesthesia with fentanyl analgesia: a matched cohort study. Anesth Analg. 2014. October; 119 4: 859- 866. 10.1213/ANE.0000000000000320. [DOI] [PubMed] [Google Scholar]
- 90. Tseng KS, Kulkarni S, Humphreys EB, et al. Spinal anesthesia does not impact prostate cancer recurrence in a cohort of men undergoing radical prostatectomy: an observational study. Reg Anesth Pain Med. 2014. Jul-Aug; 39 4: 284- 288. 10.1097/AAP.0000000000000108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Heinrich S, Janitz K, Merkel S, Klein P, Schmidt J. . Short- and long term effects of epidural analgesia on morbidity and mortality of esophageal cancer surgery. Langenbecks Arch Surg. 2015. January; 400 1: 19- 26. 10.1007/s00423-014-1248-9. [DOI] [PubMed] [Google Scholar]
- 92. Starnes-Ott K, Goravanchi F, Meininger JC. . Anesthetic choices and breast cancer recurrence: a retrospective pilot study of patient, disease, and treatment factors. Crit Care Nurs Q. 2015. Apr-Jun; 38 2: 200- 210. 10.1097/CNQ.0000000000000062. [DOI] [PubMed] [Google Scholar]
- 93. Grandhi RK, Lee S, Abd-Elsayed A. . Does opioid use cause angiogenesis and metastasis? Pain Med. 2017. January 1; 18 1: 140– 151. 10.1093/pm/pnw132. [DOI] [PubMed] [Google Scholar]
- 94. Jhaveri FM, Zippe CD, Klein EA, Kupelian PA. . Biochemical failure does not predict overall survival after radical prostatectomy for localized prostate cancer: 10-year results. Urology. 1999. November; 54 5: 884- 890. [DOI] [PubMed] [Google Scholar]
- 95. Vitolins MZ, Kimmick GG, Case LD. . BMI influences prognosis following surgery and adjuvant chemotherapy for lymph node positive breast cancer. Breast J. 2008. Jul-Aug; 14 4: 357- 365. 10.1111/j.1524-4741.2008.00598.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Schnabel A, Reichl SU, Kranke P, Pogatzki-Zahn EM, Zahn PK. . Efficacy and safety of paravertebral blocks in breast surgery: a meta-analysis of randomized controlled trials. Br J Anaesth. 2010. December; 105 6: 842- 852. 10.1093/bja/aeq265. Erratum in: Br J Anaesth . 2013 Sep;111(3):522. [DOI] [PubMed] [Google Scholar]
- 97. Sessler DI, Ben-Eliyahu S, Mascha EJ, Parat MO, Buggy DJ. . Can regional analgesia reduce the risk of recurrence after breast cancer? Methodology of a multicenter randomized trial. Contemp Clin Trials. 2008. July; 29 4: 517- 526. 10.1016/j.cct.2008.01.002. [DOI] [PubMed] [Google Scholar]
- 98. Wu J, Buggy D, Fleischmann E, et al. Thoracic paravertebral regional anesthesia improves analgesia after breast cancer surgery: a randomized controlled multicentre clinical trial. Can J Anaesth. 2015. March; 62 3: 241- 251. 10.1007/s12630-014-0285-8. [DOI] [PubMed] [Google Scholar]
- 99. Deegan CA, Murray D, Doran P, Ecimovic P, Moriarty DC, Buggy DJ. . Effect of anaesthetic technique on oestrogen receptor-negative breast cancer cell function in vitro. Br J Anaesth. 2009. November; 103 5: 685- 690. 10.1093/bja/aep261. Erratum in: Br J Anaesth . 2010 Apr;104(4):516. [DOI] [PubMed] [Google Scholar]
- 100. Desmond F, McCormack J, Mulligan N, Stokes M, Buggy DJ. . Effect of anaesthetic technique on immune cell infiltration in breast cancer: a follow-up pilot analysis of a prospective, randomised, investigator-masked study. Anticancer Res. 2015. March; 35 3: 1311- 1319. [PubMed] [Google Scholar]
- 101. Looney M, Doran P, Buggy DJ. . Effect of anesthetic technique on serum vascular endothelial growth factor C and transforming growth factor β in women undergoing anesthesia and surgery for breast cancer. Anesthesiology. 2010. November; 113 5: 1118- 1125. 10.1097/ALN.0b013e3181f79a69. [DOI] [PubMed] [Google Scholar]
- 102. Schlagenhauff B, Ellwanger U, Breuninger H, Stroebel W, Rassner G, Garbe C. . Prognostic impact of the type of anaesthesia used during the excision of primary cutaneous melanoma. Melanoma Res. 2000. April; 10 2: 165- 169. [PubMed] [Google Scholar]