Abstract
Objective
To assess the efficacy and cost-effectiveness of modulated electrohyperthermia (mEHT) concurrent to dose-dense temozolomide (ddTMZ) 21/28 days regimen versus ddTMZ 21/28 days alone in patients with recurrent glioblastoma (GBM).
Design
A cohort of 54 patients with recurrent GBM treated with ddTMZ+mEHT in 2000–2005 was systematically retrospectively compared with five pooled ddTMZ 21/28 days cohorts (114 patients) enrolled in 2008–2013.
Results
The ddTMZ+mEHT cohort had a not significantly improved mean survival time (mST) versus the comparator (p=0.531) after a significantly less mean number of cycles (1.56 vs 3.98, p<0.001). Effect-to-treatment analysis (ETA) suggests that mEHT significantly enhances the efficacy of the ddTMZ 21/28 days regimen (p=0.011), with significantly less toxicity (no grade III–IV toxicity vs 45%–92%, p<0.0001). An estimated maximal attainable median survival time is 10.10 months (9.10–11.10). Cost-effectiveness analysis suggests that, unlike ddTMZ 21/28 days alone, ddTMZ+mEHT is cost-effective versus the applicable cost-effectiveness thresholds €US$25 000–50 000/quality-adjusted life year (QALY). Budget impact analysis suggests a significant saving of €8 577 947/$11 201 761 with 29.1–38.5 QALY gained per 1000 patients per year. Cost-benefit analysis suggests that mEHT is profitable and will generate revenues between €3 124 574 and $6 458 400, with a total economic effect (saving+revenues) of €5 700 034 to $8 237 432 per mEHT device over an 8-year period.
Conclusions
Our ETA suggests that mEHT significantly improves survival of patients receiving the ddTMZ 21/28 days regimen. Economic evaluation suggests that ddTMZ+mEHT is cost-effective, budget-saving and profitable. After confirmation of the results, mEHT could be recommended for the treatment of recurrent GBM as a cost-effective enhancer of ddTMZ regimens, and, probably, of the regular 5/28 days regimen. mEHT is applicable also as a single treatment if chemotherapy is impossible, and as a salvage treatment after the failure of chemotherapy.
Keywords: recurrent glioblastoma, modulated electro-hyperthermia (meht), oncothermia, dose-dense temozolamide (ddtmz, effect-to-treatment analysis (eta), cost-effectiveness analysis
Strengths and limitations of this study.
The study first introduces the application of a novel clinical analysis called effect-to-treatment analysis.
The study applies a systematic comparator in the form of the pooled average of a meta-analysis of a systematic review of comparable trials.
The study includes comprehensive economic evaluation, comprising consistent costs analysis, cost-effectiveness analysis, budget-impact analysis and cost-benefit analysis.
Because the study is based on a single retrospective trial, future studies are needed to confirm its findings.
Background
Glioblastoma multiforme (GBM) is a common and aggressive primary brain tumour, accounting for 45%–54% of all adult gliomas.1 2 Despite the recent treatment advances, GBM prognosis remains dismal, with the median survival time (MST) limited to 15–18 months.3 The prognosis for patients with recurrent GBM remains poor, with the MST between 3 and 6 months.4 As 20 years ago, treatment of recurrent GBM can be considered successful if the stable disease is achieved.5
Standards of care are not yet defined for recurrent GBM.6 Treatment options at recurrence include surgical resection, re-irradiation and chemotherapy (CTX),7 although all of these options have significant limitations.8 The standard CTX treatment for recurrent GBM, based on the milestone European Organisation for Research and Treatment of Cancer/National Cancer Institute of Canada Clinical Trials Group (EORTC/NCIC CTG) trial,9 10 includes oral DNA-alkylating agent temozolomide (TMZ) given daily at 150–200 mg/m2 for 5 days in each 28-day cycle (5/28 d) (Stupp regimen).3 Unfortunately, TMZ adds only about 2.5 months to the MST compared with RT alone at first-line treatment.9 10 Given that >50% of patients fail to respond to TMZ treatment over 6–9 months, and the majority (60%–75%) of patients with GBM who do not have a methylated O6-methylguanine-DNA methyltransferase (MGMT) promoter derive limited benefit from TMZ treatment,11 and 15%–20% of patients treated with TMZ develop clinically significant toxicity,8 TMZ should be considered a modestly effective chemotherapy. Attempts to improve the Stupp regimen involve, among others, the increased TMZ dosage, known as dose-dense TMZ (ddTMZ) regimens.12
The rationale for ddTMZ is based on the known role of specific DNA repair enzyme 7MGMT in tumour resistance to alkylating agents such as TMZ. MGMT effectively recovers TMZ-related DNA damage. Methylation of the promoter region of the MGMT gene suppresses MGMT expression. A methylated MGMT promoter is observed in 30%–60% of GBMs.13 Because MGMT is a suicide enzyme and requires resynthesis for recovery of its enzymatic activity,14 it can be depleted by continuous alkylating pressure. Therefore, prolonged exposure and higher cumulative doses of TMZ could sensitise tumours to the alkylating damage, with toxicity as a natural limiter of such dose escalation. Some ddTMZ regimens were applied versus the standard 5/28 d regimen, including the 7/14 d (7 days on/7 days off), 21/28 d and continuous administration (7/7 d or 28/28 d) regimes.12 15 Multiple single-armed and retrospective studies of ddTMZ at recurrent GBM showed progression-free survival at 6 months (PFS-6m) ranging from 19% to 44% and an MST of 7–10 months.12 However, a recent phase III randomised controlled trial (RTOG 0525)16 of ddTMZ 21/28 d versus the standard 5/28 d adjuvant regimen for newly diagnosed patients with GBM after completion of concurred chemoradiotherapy (CRT), failed to show an advantage of ddTMZ in MST (14.9 vs 16.6 months in the standard arm, p=0.63), although it did show an improvement of PFS-6m (6.7 vs 5.5 months) with borderline significance (p=0.06), with somewhat higher toxicity in the ddTMZ arm. Therefore, the efficacy of ddTMZ regimens remain unproven.12
Finally, it should be noted that the modern chemotherapies like TMZ, bevacizumab and other antiangiogenic agents are not cost-effective.17–20 In fact, there remains a significant unmet need for more effective treatments of high-grade gliomas,21 and the poor outcomes of the current treatment of recurrent GBM requires novel approaches.5
There is a physical technology called modulated electrohyperthermia (mEHT, oncothermia), the effectiveness of which was demonstrated in many phase I/II trials in recurrent brain gliomas,22–26 and also in cancer of lung,27–30 liver,31–33 pancreas,34 35 cervix,36 37 breast,38oesophagus,39 colorectal cancer,40–43 malignant ascites44 and soft tissue sarcomas.45 46 Clinically, mEHT is typically used as an enhancer of radiation27 36 and chemotherapy, although it possesses its own effectiveness of at least a similar magnitude to these treatments.23 40 47
mEHT is a novel method of treatment of solid malignant tumours by the local application of a high-frequency electromagnetic field (13.56 MHz), modulated by 0–5 kHz flicker noise, by virtue of impedance-coupled functionally asymmetric electrodes.48 mEHT is positioned as a next-generation hyperthermic technology based on the selective heating of intercellular compartments of tumour tissue and cell membranes, instead of the heating of a bulk volume of the tissue, as the conventional temperature-dependent hyperthermia (HT) does.49–53
Unlike the old HT technologies, mEHT transfers the focus from the dielectric heating (field effect) to the Joule (electric) heating in order to improve focusing and penetration depth. Since the current has a known ability to concentrate in areas with a higher conductance,54 and the increased conductance is one of the basic properties of malignant tissue,55 a tumour is a natural concentrator of electrical current. This feature has long been used for electrical impedance scanning56 and current-density imaging.57 58 The penetration depth of current in the impedance-matched system is 20–25 cm59 vs 14–18 cm only60 in the regular capacitive HT at 13.56 MHz. Therefore, the emphasis on the current allows transferring energy selectively to the tumour for any depth and with minimal losses. ‘Electrohyperthermia’ means predominantly electric heating.61
A combined set of technical solutions is used to achieve maximal electrical heating: namely, the impedance matching based on the phase angle between voltage and current; functionally asymmetric electrodes, providing the necessary stability of the field and size difference-dependent amplification of the current; physiologic skin cooling, minimising skin losses at energy transfer and a ‘skin sensor’ concept, which allows for refuse thermometry without detriment to safety.48 ‘Free of thermometry’ use is a great advantage of mEHT, abolishing the labour-intensive thermometry planning, installation and control, thus drastically reducing time and costs, minimising side effects and significantly improving the perception of the treatment by a patient.62
The electric heating creates quasi-stable local thermal gradients at the nano level (eg, transmembrane thermal gradient63), which are maintained by the balance of continuous delivery of energy by external field and energy dissipation by natural cooling mechanisms, mainly by a blood flow.64 65 Thus, the nanoheating, depending on the field power applied and physiological cooling power displayed, can develop even without macroscopic heating.66 It was shown ex vivo that a 42°C temperature in mEHT is only responsible for 25%–30% of the total antitumour effect and a slightly smaller effect was shown in the case of normothermia.67 Thus, the effect of mEHT is thermally induced but not temperature-dependent.68
The clinical value of the not temperature-dependent effects can no longer be questioned after the Food and Drug Administration approval69 of tumour-treating fields (TTF), an athermal technology using continuous impact of a low-intensity (0.7–1 V/cm) alternating electromagnetic field with a frequency of 100–200 kHz through insulated scalp cross-sectional electrodes.70–75 In a phase III study,76 TTF displayed the same efficacy at recurrent GBM as the best physician choice CTX (MST 6.6 vs 6.0 months, respectively (p=0.27)) with better quality of life.
Nevertheless, mEHT usually causes hyperthermia-range heating77–80 in accordance with a classical maxima of Schwan on the impossibility to reach significant ‘non-thermal’ effects without substantial heating.81 The effect of mEHT is power-dependent but not signal-dependent. It is not connected with multiple tiny and questionable processes such as demodulation and molecular energy uptake82 (although we cannot completely exclude these possibilities). The power range of mEHT (0.2–2 W/cm2) is far above the ‘thermal noise limit’ of 0.01 W/cm2.83
Fractal modulation is a specific feature of mEHT. The carrying frequency is amplitude-modulated by ‘pink noise’ (1/f),84 which is typically emitted by all self-organised living systems and reflects their fractal organisation.85 Since a malignancy always losses organisation, it more or less emits ‘red’ or Brownian noise (1/f2)86 (correctly speaking, its noise spectrum is more ‘reddish’). Fractal modulation allows for increasing specific absorption of modulated field energy in the ‘red noise’ sites, selectively amplifying the effect of mEHT.87 Also, the noise can amplify cancer-specific frequencies88 by ‘stochastic resonance’.89 It is reported in vitro that modulation can amplify the effect of mEHT by 20%–50%.87
An important feature of mEHT is its selectivity, both macroscopic and cellular. Macroscopic selectivity of tumour heating is based on the automatic impedance-based autofocusing of electric current in the tumour.54 The cellular selectivity of mEHT, based on the membrane selectivity and modulation, was demonstrated in vitro using a mixed culture of cancerous and normal cells. mEHT selectively destroyed malignant cells without damage to the normal cells, and the extent of the damage was proportional to the degree of malignancy.90
The exact mechanism of mEHT action is unknown. Both temperature-dependent and -independent mechanisms are among possible options. Temperature-dependent mechanisms include disorder of tumour blood flow, oxygen and glucose deprivation, depletion of intracellular ATP, the influx of sodium and depolarisation of cellular membrane91–93 and acidification.94–96 Since these effects are present in all HT applications, and they do not lead to results characteristic for mEHT, we propose that there must be other mEHT-specific mechanisms of action. Many not temperature-dependent (so-called ‘non-thermal’) effects are reported to have a peak at about 10 MHz, namely direct bactericidal effect and enhancement of antibiotics action (bioelectric effect), both in bacterial films97 and planktonic phase98; dielectrophoresis,99 damage of mitochondrial function100 and destruction of lysosomes.101
Although the frequency and field strength (2–5 V/cm) applied in mEHT cannot cause a significant change in the membrane potential,102 there are many reasons to suggest a specific membrane-acting effect of mEHT. The 10 MHz is a relaxation frequency of the β-dispersion range (0.1–100 MHz) caused by Maxwell-Wagner relaxation of cell membranes,103 which means a peak of membrane dielectric loss and selective membrane excitation (heating) at this frequency104 (reorientation of protein-bound water molecules, the motion of polar protein subgroups, the Maxwell-Wagner relaxation of the cell interior or the additional Maxwell-Wagner relaxations due to the non-spherical cell shape, also contribute to the β-dispersion103), and also a peak of phase shift of membrane polarisation under the effect of the external alternative field, which nearly reaches a quadrature (−80°).102 The relaxation frequency of the reorientational proton motion of water-bound proteins also peaks at about 10 MHz (range 1–100 MHz).105
Another possible effect of mEHT is an arrest of cell division with possible mitotic catastrophe,98 attributable to a subcellular ponderomotoric effect (dielectrophoretic forces suppress the assembly of the mitotic spindle71), to membrane polarisation (cell division phases are associated with changes in membrane potential, and non-linear processes of hyperpolarisation and depolarisation, under the effect of radiofrequency (RF) field, suppress proliferation72) or to resonance phenomena.106 Also, effects on the cytoskeleton107 108 and selective activation of some enzymes, both conformational and voltage-dependent (in the case of membrane enzymes),109 are reported.
The overall effect of mEHT is connected with an extracellular expression of intracellular signalling molecules of cellular stress (eg, heat shock proteins (HSP) and p53 protein),110 which unmask cancer cells and initiate the immune response and apoptosis.111 It has been shown in vivo and in vitro that the antitumour effect of mEHT is mainly connected with significant activation of apoptosis, which develops over 72 hours after a single impact.111–113 Some immune-dependent effects are reported, namely the abscopal effect,114 115 which is considered as a basis for a ‘RF vaccination’.116 117 Expression of many immune-specific pathways has been reported in vitro in mEHT.111 118–120 Overexpression of cell-junction proteins with the significant restoration of intercellular junctions, which can contribute to the induction of apoptosis,121 122 and reorganisation of cytoskeleton107 are reported for mEHT.
Taking into account the extensive and long-term (since 1996) successful application without any negative report, a systematic review of results of mEHT is possible and necessary. Collecting the data for the systematic review and meta-analysis on the mEHT treatment of brain gliomas, we asked for raw data whenever possible. The raw data of the trial by Sahinbas et al 23 including 155 patients with high-grade gliomas (HGG) were obtained on request. After analysis of the data, some shortcomings were revealed, namely duplications, incorrect grouping by histology and incorrect calculation of survival function in view of incorrect processing of censoring. After corrections and recalculation, the results of this trial appeared so interesting that we believe they deserved to be republished. In this retrospective analysis, we report the result of the systematic clinical comparison and economic evaluation of mEHT concurrent to the ddTMZ 21/28 d regimen in the treatment of recurrent GBM. No change to the raw data was made.
Materials and methods
Objectives
The objective of this study is to assess the efficacy and cost-effectiveness of mEHT concurrent to ddTMZ 21/28 d regimen versus ddTMZ 21/28 d alone in patients with recurrent GBM.
Questions of the study
Does mEHT significantly enhance the ddTMZ 21/28 d regimen?
Is the addition of mEHT to ddTMZ 21/28 d regimen cost-effective?
Trial design
This retrospective clinical and economic evaluation is based on a systematic comparison and effect-to-treatment analysis (ETA) of a retrospective, single-arm study23 (study of interest (SOI)) performed in two German centres (the Gronemeyer Institute of Microtherapy at the University of Bochum and the clinic ‘Closter Paradise’, Soest) between 2000 and 2005.
Inclusion and exclusion criteria
Patients with relapsed or progressed after incomplete resection or progressive inoperable histologically confirmed GBM or gliosarcoma (WHO IV), having undergone a complete conventional first-line and second-line pretreatment were selected. From those, patients treated with ddTMZ 21/28 d in combination with mEHT (with or without supportive therapy but without re-irradiation, resurgery or other chemotherapy), were selected. No exclusion criteria were applied.
Outcomes
Survival was the main outcome of the study:
MST is the time from the initial event to the moment when the value of cumulative survival function (Kaplan-Meier estimate (KME)) reaches 50%. Here, the term MST is applied to survival since relapse/progression or the date of the first mEHT session, while survival since the date of diagnosis is defined as median overall survival time.
Overall survival (OS) is the value of cumulative survival function (KME) at the set time moments from the date of the initial event.
OS time is the time from the initial event to the death of any reason.
No surrogate outcomes were used.
Intervention
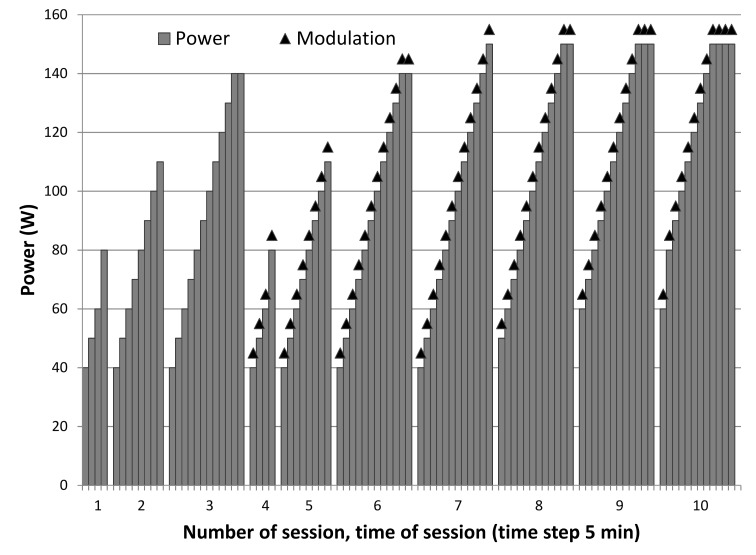
The studied intervention was a combination of ddTMZ 21 days on, 7 days off regimen (100 mg/m2/day) with concurrent mEHT as an enhancer (ddTMZ+mEHT). MEHT (the intervention of interest (IOI)) was applied using an EHY2000 device (Oncotherm Kft, Hungary) with 2 days intervals between sessions (on each third day) concurrent with TMZ and afterwards, for up to 3 months. A dose-escalating scheme was used with a gradual increase of power from 40 to 150 W and increase of time from 20 to 60 min, during 2 weeks, adding modulation from the second week (figure 1). Then, a step-up heating was applied, increasing the power from 60 to 150 W during 60 min sessions, to ensure tumour temperature of >40°C during 90% of the treatment time. Dose escalation was limited by patient’s individual tolerance. The mEHT course was considered low-dose (LD-mEHT), if did not exceed eight complete 60 min sessions. Supportive and alternative treatments (SAT) included Boswellia caterii extract 6 g/day three times daily to be taken orally, mistletoe extract 15 ng/day subcutaneously 3 times/week, and selenium 300 µg/day orally, for 3 months.
Figure 1.
Dose-escalating scheme of modulated electrohyperthermia. The tenth session attains the maximum escalation, the further sessions are the same.
Response and survival assessment
The objective response was assessed according to the MRI McDonald criteria.123 Survival function was assessed by the Kaplan-Meier estimate. Survivors were right-censored on the date of completion of the study (30 May 2005), lost patients were censored on the date of the last contact and excluded patients were left-censored on the date of diagnosis/enrolment.
Statistical methods
Statistical analysis was performed using the built-in Excel 2016 analysis package using the methods of descriptive statistics, correlation and regression analysis. Normality of distribution was estimated by the Kolmogorov-Smirnov test. CIs of medians were calculated according to Conover,124 relative risks (RR) and ORs according to Altman,125 risk difference (RD) according to Newcomb and Altman,126 product of means according to Goodman,127 ratio of means according to Fieller128 129 for independent means, and by Taylor approximation130 for dependent means, and the ratio of two independent lognormally distributed estimates by Newcomb’s MOVER-R algorithm.131 Inverse-variance weighting was used.132 The significance of differences in parametric criteria was estimated by the two-sample Student’s t-test or Welch t-test for unequal variance133; and for paired non-parametric criteria (proportions) by the Pearson’s χ2 test according to Campbell-Richardson.134 The significance of rates and proportions with known 95% CI was estimated according to Altman and Bland,135 and the significance of the difference of two independent estimates by the two-sample z-test. All p values are two-sided. A 95% probability (α=0.05) was used for significance testing. Since log-transformation significantly inflates CIs (up to 40-times in some cases136), 90% probability (α=0.1) is considered applicable for the significance of the difference of estimates based on log-transformed parameters in some cases.
Survival analysis was performed using the Excel-based software package GRISA (Galenic Research Institute, 2015) by KME of the cumulative probability of survival.137 SEs and CIs of KME were estimated by Greenwood’s formula,138 and the significance of differences by the log-rank test.139 The hazard function was estimated by the Cox proportional hazards regression model.140
Meta-analysis was performed using the Excel-based software package GRIMA (Galenic Research Institute, 2015) according to Borenstein et al 132 and statistical algorithms of the Cochrane Collaboration.141 The heterogeneity of studies was assessed by the I2 criterion.142 In view of the significant heterogeneity of the cohorts, a random effects model was applied.
Effect-to-treatment analysis
ETA was performed according to our own algorithm143 with the following settings: a unit of treatment is a 28-day cycle, and the parameter of comparison is the mean survival time (mST) after relapse. Here, we use mST for mean survival time and MST for median survival time. Medians were transformed into means with 95% CI using the algorithm by Hozo et al 144 for medians with range and our own simplified algorithm (see online supplementary 1) for medians with 95% CI. The life months gained (LMG) parameter was calculated by subtracting the expected mST (emST). Effect-to-treatment ratio (ETR) was calculated by dividing the LMG by the mean number of cycles (mNC). Life quality adjustment was not possible due to significant initial differences between the cohorts. The median ETR (METR) was estimated by attenuation of the ETR according to the formula: , where CA is a coefficient of attenuation. The dependence of mST from mNC was estimated by the function , where NC is a serial number of cycle; the extremum of the function is a maximal attainable survival time (MAST), the abscissa of the extremum is a peak number of cycle (PNC). Cost-effective number of cycles (CENC) was estimated as abscissa of cost-effective survival time value (CEST=95% MAST). Cycles needed to treat per LMG (CNTM) was estimated as the reciprocal of the difference of . The effect enhancement ratio () was estimated as an auxiliary parameter for calculation of CI and significance of CNTM: since EER and CNTM use the same parameters with the same null hypothesis [], their CIs and significance are the same, and these parameters can be easily calculated for EER according to Altman and Bland.135
bmjopen-2017-017387supp001.pdf (55.7KB, pdf)
Economic evaluation
For economic evaluation, cost-effectiveness analysis (CEA) with sensitivity analysis, budget impact (BIA) and cost-benefit (CBA) analyses were performed.145–149 CEA and BIA were performed from the perspective of a health provider. CEA was based on the cost-utility ratio (CUR) and incremental cost-effectiveness ratio (ICER). The ratio of CURs (CURR) and increment of CURs (ICUR) were used to compare CURs. The proportion of cost-effective cases (%CE) was estimated by one-tailed directional integral z-test with the null hypothesis [], where CET is a cost-effectiveness threshold. To estimate a sensitivity of CEA, a multiparametric equal cost-effectiveness test was performed exploring the value of a key parameter in which the value of CURR equals 1.0 (or ICUR=0). The BIA estimated the difference of costs for treatment of 1000 patients per year. CBA estimated the total economic effect (saving and earnings before interest and taxes (EBIT)) from the perspective of a healthcare facility.
Reporting
SOI is reported according to the STROBE (Strengthening the Reporting of Observational studies in Epidemiology) statement for reporting observational studies.150 Economic evaluation is reported according to the CHEERS (Consolidated Health Economic Evaluation Reporting Standards) standards.151
Results
Patients’ flow
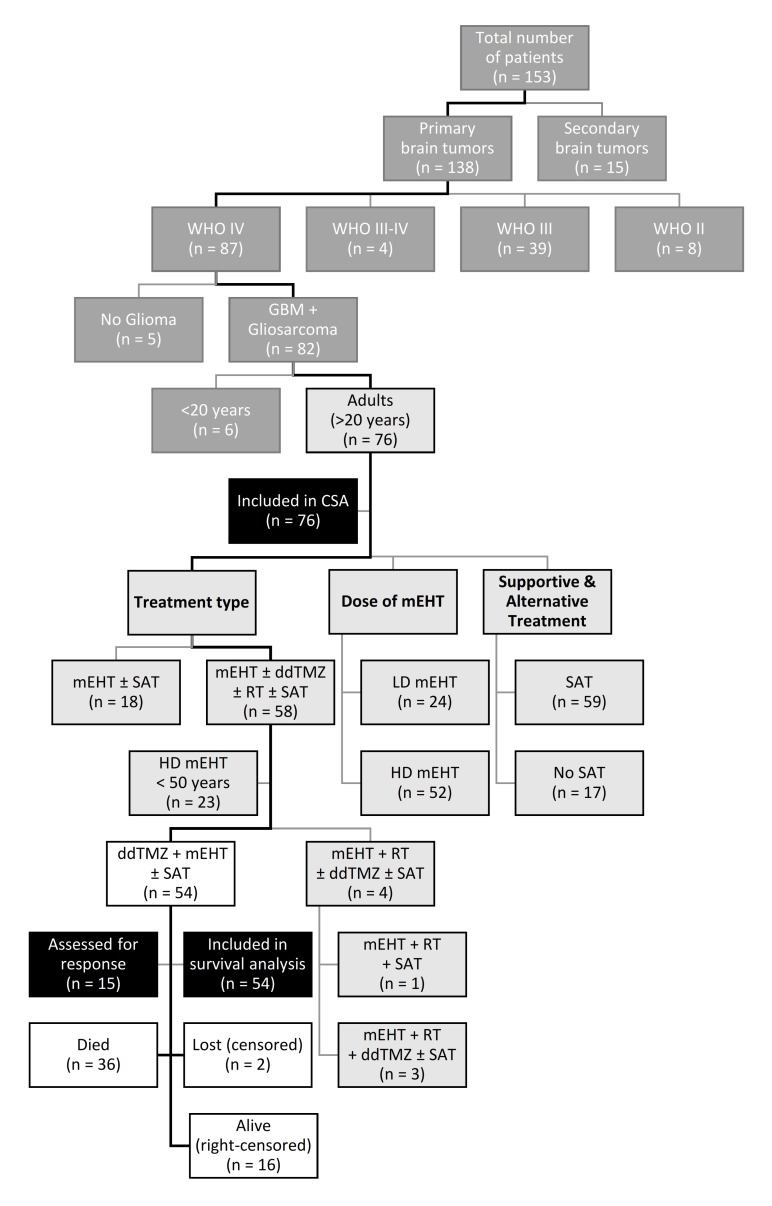
A total of 153 patients with different brain tumours (box) were enrolled in the two centres between 2000 and 2005 (figure 2). Of those, 138 patients had primary brain tumours, and 87 were graded as WHO IV, including 81 GBM and one gliosarcoma (n=82). Of those, 76 patients were adults (>20 years). Fifty-eight adult patients with GBM received a combination treatment (mEHT±ddTMZ±RT±SAT), other 18 patients with GBM were treated with mEHT only (with or without SAT). Twenty-three patients of the combination cohort were younger than 50 years and received high-dose (HD) mEHT (HD-mEHT). The cohort of interest (COI) included 54 patients who received mEHT+ddTMZ (with or without SAT). Four other patients of the combination cohort received RT in addition to mEHT, either alone (n=1) or with ddTMZ (n=3) (with or without SAT). Of the adult patients with GMB (n=76), 24 received LD-mEHT and 52 received HD-mEHT; 59 received SAT vs 17 who did not.
Box. Histological types of brain tumours (SOI).
Total patients: 153
-
(C71) Malignant neoplasm (MN) of brain: 137
-
WHO II: 8
Astrocytoma: 4
Mixed glioma: 4
-
WHO III: 39
Astrocytoma: 34
Mixed glioma: 3
Ependimoma: 1
Oligodendroglioma: 1
-
WHO III–IV: 4
Astrocytoma: 3
Infratentorial glioma: 1
-
WHO IV: 87
-
Glioblastoma: 81
Age >20 years: 75
Age <20 years: 6
Gliosarcoma: 1
Medulloblastoma: 3
Primitive neuroectodermal tumour: 1
-
-
(D43.1) Neoplasm of uncertain behaviour of brain, infratentorial: 1
-
(C79.3) Secondary MN of brain and cerebral meninges: 15
-
Adenocarcinoma: 12
MN of breast: 7
MN of bronchus and lung: 3
MN of colon: 1
MN of pancreas: 1
Ewing sarcoma: 1
Malignant rhabdoid tumour: 1
Cancer of unknown primary: 1
-
Figure 2.
CONSORT flow chart. White: COI, cohort of interest; light grey: CSA, cohorts of covariate survival analysis; dark grey: cohorts out of analysis; black: analyses; ddTMZ, dose-dense temozolomide; GBM, glioblastoma; mEHT, modulated electrohyperthermia; SAT, supportive and alternative treatments.
Patients’ characteristic
Fifty-four adult patients with WHO IV GBM (n=53) and gliosarcoma (n=1) matched the inclusion criteria (COI). The mean age was 48.7±1.5 years (median, 49.8 years; range 25.9–68.2; 95% CI 42.2 to 52.8), including 2 (4%) elderly patients (≥68 years) and 26 patients (48%) over 50 years. Thirty-three patients were males and 21 females (table 1).
Table 1.
Patients’ characteristic
| Parameter | All GBM | mEHT±SAT | Combination treatment | ddTMZ+mEHT | LD-mEHT | HD-mEHT | HD-mEHT <50 years |
|||||||
| (1) | (2) | (3) | (4) | (5) | (6) | (7) | ||||||||
| Value | % | Value | % | Value | % | Value | % | Value | % | Value | % | Value | % | |
| NOP | 76 | 18 | 58 | 54 | 24 | 52 | 23 | |||||||
| Male | 46 | 61% | 10 | 56% | 36 | 62% | 33 | 61% | 16 | 67% | 30 | 58% | 11 | 48% |
| Female | 30 | 39% | 8 | 44% | 22 | 38% | 21 | 39% | 8 | 33% | 22 | 42% | 12 | 52% |
| Earliest born | 24 February 1932 | 24 February 1932 | 19 September 1935 | 19 September 1935 | 24 February 1932 | 18 June 1932 | 31 October 1954 | |||||||
| Latest born | 03 April 1975 | 10 March 1971 | 03 April 1975 | 03 April 1975 | 03 April 1975 | 21 August 1973 | 21 August 1973 | |||||||
| Earliest diagnosed | 01 August 1993 | 01 September 2000 | 01 August 1993 | 01 August 1993 | 12 July 1999 | 01 August 1993 | 01 August 1993 | |||||||
| Latest diagnosed | 15 March 2005 | 03 July 2004 | 15 March 2005 | 30 August 2004 | 08 July 2004 | 15 March 2005 | 15 March 2005 | |||||||
| Age (years) | ||||||||||||||
| Mean | 50.2±1.3 | 55.1±2.8 | 48.7±1.4 | 48.7±1.5 | 50.9±2.6 | 49.9±1.,5 | 39.9±1.2 | |||||||
| Median | 50.4 | 59.1 | 49.8 | 49.8 | 50.8 | 50.2 | 41.0 | |||||||
| Range | 25.9–71.9 | 30.9–71.9 | 25.9–68.2 | 25.9–68.2 | 25.9–68.9 | 27.0–71.9 | 27.0–49.1 | |||||||
| 95% CI | 44.8–53.9 | 44.4–64.9 | 42.7–52.3 | 42.2–52.8 | 42.2–59.8 | 44.4–55.8 | 36.7–43.0 | |||||||
| p Value (t-test) | 0037 | <0.0001* | ||||||||||||
| Elderly (over 68 years) | 4 | 5% | 2 | 11% | 2 | 3% | 2 | 4% | 2 | 8% | 2 | 4% | 0 | 0% |
| Mature (over 50 years) | 40 | 53% | 12 | 67% | 28 | 48% | 26 | 48% | 13 | 54% | 27 | 52% | 0 | 0% |
| Adults (over 20 years) | 76 | 100% | 18 | 100% | 58 | 100% | 54 | 100% | 24 | 100% | 52 | 100% | 23 | 100% |
| Pretreatment: | ||||||||||||||
| Surgery+chemoradiation | 57 | 75% | 13 | 72% | 44 | 76% | 42 | 78% | 15 | 63% | 42 | 81% | 20 | 87% |
| Chemoradiation | 2 | 3% | 1 | 6% | 1 | 2% | 1 | 2% | 1 | 4% | 1 | 2% | 0 | 0% |
| Surgery+radiation | 7 | 9% | 2 | 11% | 5 | 9% | 4 | 7% | 4 | 17% | 3 | 6% | 2 | 9% |
| Surgery+chemotherapy | 5 | 7% | 0 | 0% | 5 | 9% | 4 | 7% | 1 | 4% | 4 | 8% | 1 | 4% |
| Radiaton only | 5 | 7% | 2 | 11% | 3 | 5% | 3 | 6% | 3 | 13% | 2 | 4% | 0 | 0% |
| Chemotherapy total | 64 | 84% | 14 | 78% | 50 | 86% | 47 | 87% | 17 | 71% | 47 | 90% | 21 | 91% |
| Radiation total | 71 | 93% | 18 | 100% | 53 | 91% | 50 | 93% | 23 | 96% | 48 | 92% | 22 | 96% |
| Surgery total | 69 | 91% | 15 | 83% | 54 | 93% | 50 | 93% | 20 | 83% | 49 | 94% | 23 | 100% |
*Versus all GBM sample.
ddTMZ, dose-dense temozolomide; GBM, glioblastoma; HD, high dose; LD, low dose; mEHT, modulated electrohyperthermia; NOP, no. of patients; SAT, supportive and alternative treatments.
Forty-two (78%) patients underwent complete trimodal pretreatment including surgery and chemoradiation, four (7%) received previous surgery and radiation, four (7%) received surgery and chemotherapy, three (6%) received only radiation and one (2%) received only chemoradiation. By modalities, 50 (93%) patients underwent previous surgery, 50 (93%) radiation and 47 (87%) chemotherapy (mainly TMZ). The characteristics of the other cohorts are given in table 1.
Details of treatment
All patients (100%) in the COI received ddTMZ+mEHT treatment, and 43 (80%) patients received concurrent SAT (table 2).
Table 2.
Details of treatment
| Parameter | All GBM | mEHT±SAT | Combination treatment | ddTMZ+mEHT | LD-mEHT | HD-mEHT | HD-mEHT <50 years |
|||||||
| (1) | (2) | (3) | (4) | (5) | (6) | (7) | ||||||||
| Value | % | Value | % | Value | % | Value | % | Value | % | Value | % | Value | % | |
| Time to first mEHT since diagnosis (months) | ||||||||||||||
| Mean | 12.1±1.6 | 11.2±2.3 | 12.3±1.9 | 12.9±2.1 | 13.3±2.4 | 11.5±2.0 | 12.7±4.2 | |||||||
| Median | 8.5 | 8.0 | 9.3 | 9.5 | 9.9 | 8.2 | 5.9 | |||||||
| Range | 0.2–94.2 | 2.3–44.1 | 0.2–94.2 | 0.2–94.2 | 1.6–49.1 | 0.2–94.2 | 1.0–94.2 | |||||||
| 95% CI | 6.7 to 10.6 | 6.1 to 15.2 | 5.8 to 10.7 | 5.9 to 10.7 | 6.1 to 11.6 | 5.1 to 10.0 | 4.1 to 10.0 | |||||||
| Earliest mEHT | 01 March 2001 | 07 May 2001 | 01 March 2001 | 01 March 2001 | 07 June 2001 | 01 March 2001 | 01 March 2001 | |||||||
| Latest mEHT | 20 May 2005 | 19 May 2005 | 20 May 2005 | 20 May 2005 | 28 April 2005 | 20 May 2005 | 20 May 2005 | |||||||
| Treatment combinations | ||||||||||||||
| mEHT+CRT+SAT | 2 | 3% | 0 | 0% | 2 | 3% | 0 | 0% | 0 | 0% | 2 | 4% | 0 | 0% |
| mEHT+chemoradiation | 1 | 1% | 0 | 0% | 1 | 2% | 0 | 0% | 0 | 0% | 1 | 2% | 1 | 4% |
| mEHT+chemotherapy+SAT | 43 | 57% | 0 | 0% | 43 | 74% | 43 | 80% | 12 | 50% | 31 | 60% | 13 | 57% |
| mEHT+radiation+SAT | 1 | 1% | 0 | 0% | 1 | 2% | 0 | 0% | 0 | 0% | 1 | 2% | 1 | 4% |
| mEHT+chemotherapy | 11 | 14% | 0 | 0% | 11 | 19% | 11 | 20% | 6 | 25% | 5 | 10% | 3 | 13% |
| mEHT+SAT | 13 | 17% | 13 | 72% | 0 | 0% | 0 | 0% | 4 | 17% | 9 | 17% | 5 | 22% |
| mEHT only | 5 | 7% | 5 | 28% | 0 | 0% | 0 | 0% | 2 | 8% | 3 | 6% | 0 | 0% |
| Treatment by modality | ||||||||||||||
| Radiation total | 4 | 5% | 0 | 0% | 4 | 7% | 0 | 0% | 0 | 0% | 4 | 8% | 2 | 9% |
| SAT total | 59 | 78% | 13 | 72% | 46 | 79% | 43 | 80% | 16 | 67% | 43 | 83% | 19 | 83% |
| Chemotherapy total | ||||||||||||||
| NOP | 57 | 75% | 0 | 0% | 57 | 98% | 54 | 100% | 18 | 75% | 39 | 75% | 17 | 74% |
| No. of cycles | 89 | 0 | 89 | 84 | 18 | 71 | 32 | |||||||
| Mean | 1.5±0.1 | 0 | 1.6±0.1 | 1.6±0.1 | 1.0±0.0 | 1.8±0.1 | 1.8±0.2 | |||||||
| Median | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.5 | 2.0 | |||||||
| Range | 1.0–5.0 | 1.0–3.0 | 1.0–5.0 | 1.0–5.0 | 1.0–1.0 | 1.0–5.0 | 1.0–5.0 | |||||||
| 95% CI | 1.0 to 1.0 | 1.0 to 2.0 | 1.0 to 1.0 | 1.0 to 1.0 | 1.0 to 1.0 | 1.0 to 2.0 | 1.0 to 2.0 | |||||||
| mEHT total | ||||||||||||||
| NOP | 76 | 100% | 18 | 100% | 58 | 100% | 54 | 100% | 24 | 100% | 52 | 100% | 23 | 100% |
| No. of sessions | 1367 | 292 | 1075 | 995 | 169 | 1198 | 545 | |||||||
| Mean | 18.0±0.3 | 16.2±0.6 | 18.5±0.4 | 18.4±0.4 | 7.0±0.1 | 23.0±0.4 | 23.7±0.6 | |||||||
| Median | 14.0 | 13.5 | 14.0 | 14.0 | 7.0 | 18.0 | 23.0 | |||||||
| Range | 3.0–65.0 | 4.0–43.0 | 3.0–65.0 | 3.0–65.0 | 3.0–9.0 | 10.0–65.0 | 10.0–65.0 | |||||||
| 95% CI | 11.0 to 16.0 | 7.0 to 23.0 | 11.0 to 17.0 | 10.0 to 17.0 | 6.0 to 9.0 | 15.0 to 26.0 | 15.0 to 27.0 | |||||||
| Low-dose mEHT | 24 | 32% | 6 | 33% | 18 | 31% | 18 | 33% | 24 | 100% | 0 | 0% | 0 | 0% |
| Time of treatment (months) | ||||||||||||||
| Mean | 2.5±0.4 | 1.6±0.4 | 2.8±0.5 | 2.7±0.6 | 0.5±0.0 | 3.4±0.6 | 3.4±0.7 | |||||||
| Median | 1.1 | 1.0 | 1.1 | 1.1 | 0.5 | 1.9 | 1.9 | |||||||
| Range | 0.0–26.4 | 0.2–6.4 | 0.0–26.4 | 0.0–26.4 | 0.0–0.8 | 0.2–26.4 | 0.5–12.2 | |||||||
| 95% CI | 0.8 to 1.5 | 0.5 to 2.1 | 0.8 to 1.6 | 0.8 to 1.6 | 0.4 to 0.6 | 1.2 to 2.8 | 1.2 to 4.6 | |||||||
| p Value (t-test) | 0.233 | 0.001 | ||||||||||||
| Terminated (NOP) | 9 | 12% | 1 | 6% | 8 | 14% | 8 | 15% | 9 | 38% | 0 | 0% | 0 | 0% |
| p Value (χ2) | 0.35 | <0.0001 | 0.085* | |||||||||||
*Versus all GBM sample.
ddTMZ, dose-dense temozolomide; GBM, glioblastoma; HD, high dose; LD, low dose; mEHT, modulated electrohyperthermia; NOP, no. of patients; SAT, supportive and alternative treatments.
In total, 84 ddTMZ cycles were performed for 54 patients, an average of 1.6±0.1 cycles per patient (median 1.0 cycles; range 1.0–5.0; 95% CI 1.0 to 1.0). The average duration of the treatment was 2.7±0.6 months (median 1.1 months; range 1 day to 26.4 months; 95% CI 0.8 to 1.5 months). In eight (15%) cases, the treatment was terminated because of progressive disease. The average time elapsed since primary diagnosis to the first mEHT session was 12.9±2.1 months (median 9.5 months; range 0.2–94.2; 95% CI 5.9 to 10.7). A total of 995 mEHT sessions were performed, with a mean of 18.4±0.4 per patient (median 14; range 3–65; 95% CI 10 to 17). There were 18 (33%) patients with LD-mEHT.
Response
Fifteen patients (28%) in the COI were assessed for a response (figure 2). One patient (7%) showed a complete response (CR) and two (13%) showed a partial response (PR) so that the objective response rate was 20% (table 3).
Table 3.
Survival and response rates (COI)
| Parameter | All GBM | mEHT±SAT | Combination treatment | ddTMZ+mEHT | LD-mEHT | HD-mEHT | HD-mEHT <50 years |
|||||||
| (1) | (2) | (3) | (4) | (5) | (6) | (7) | ||||||||
| Value | % | Value | % | Value | % | Value | % | Value | % | Value | % | Value | % | |
| Response | ||||||||||||||
| NOP estimated | 22 | 29% | 7 | 39% | 15 | 26% | 15 | 28% | 9 | 38% | 13 | 25% | 7 | 30% |
| CR | 1 | 5% | 0 | 0% | 1 | 7% | 1 | 7% | 1 | 11% | 0 | 0% | 0 | 0% |
| PR | 2 | 9% | 0 | 0% | 2 | 13% | 2 | 13% | 0 | 0% | 2 | 15% | 2 | 29% |
| OR | 3 | 14% | 0 | 0% | 3 | 20% | 3 | 20% | 1 | 11% | 2 | 15% | 2 | 29% |
| SD | 9 | 41% | 4 | 57% | 5 | 33% | 5 | 33% | 2 | 22% | 7 | 54% | 4 | 57% |
| BRR | 12 | 55% | 4 | 57% | 8 | 53% | 8 | 53% | 3 | 33% | 9 | 69% | 6 | 86% |
| PD | 10 | 45% | 3 | 43% | 7 | 47% | 7 | 47% | 6 | 67% | 4 | 31% | 1 | 14% |
| p Value (χ2) | 0.77 | 0.003 | 0.007* | |||||||||||
| Exitus | 49 | 64% | 12 | 67% | 37 | 64% | 36 | 67% | 18 | 75% | 31 | 60% | 11 | 48% |
| Censored | 27 | 36% | 6 | 33% | 21 | 36% | 18 | 33% | 6 | 25% | 21 | 40% | 12 | 52% |
| Lost | 2 | 3% | 0 | 0% | 2 | 3% | 2 | 4% | 1 | 4% | 1 | 2% | 1 | 4% |
| Right-censored | 25 | 33% | 6 | 33% | 19 | 33% | 16 | 30% | 5 | 21% | 20 | 38% | 11 | 48% |
| Overall survival (since diagnosis)† | ||||||||||||||
| MST (months) | 20.0 | 14.8 | 20.7 | 20.8 | 18.5 | 20.4 | 23.9 | |||||||
| (95% CI)† | (14.7 to 23.6) | (12.2 to 28.3) | (15.0 to 25.0) | (15.2 to 25.1) | (11.8 to 23.0) | (14.6 to 25.7) | (13.0 to NR) | |||||||
| Range | 1.4–141.5 | 4.4–48.9 | 1.4–141.5 | 1.4–141.5 | 3.2–53.8 | 1.4–141.5 | 2.4–141.5 | |||||||
| 5-Year survival (%) | 13.5 | 0.0 | 13.3 | 13.5 | 0.0 | 16.1 | 31.0 | |||||||
| (95% CI) | (2.8 to 24.2) | (0.0 to 0.0) | (1.0 to 25.6) | (1.0 to 26.0) | (0.0 to 0.0) | (2.0 to 30.1) | (5.1 to 56.8) | |||||||
| p Value (log-rank) | 0.436 | 0.350 | 0.32* | |||||||||||
| Survival since first mEHT (months)† | ||||||||||||||
| MST (months) | 7.6 | 6.4 | 7.7 | 7.7 | 4.4 | 8.3 | 12.8 | |||||||
| (95% CI)† | (5.8 to 9.3) | (3.1 to 9.9) | (5.8 to 9.5) | (5.7 to 9.4) | (2.2 to 8.8) | (6.7 to 12.3) | (8.2 to 48.1) | |||||||
| Range | 0.3–47.3 | 0.3–13.6 | 0.7–47.3 | 0.7–47.3 | 0.3–14.9 | 1.0–47.3 | 1.0–47.3 | |||||||
| 1-Year survival (%) | 28.8 | 22.6 | 30.2 | 29.5 | 8.7 | 36.6 | 56.9 | |||||||
| (95% CI) | (16.5 to 41.0) | (0.0 to 47.9) | (16.1 to 44.2) | (15.5 to 43.6) | (0.0 to 24.5) | (21.3 to 51.9) | (33.3 to 80.5) | |||||||
| 2-Year survival (%) | 16.8 | 0.0 | 19.2 | 18.8 | 0.0 | 23.3 | 32.5 | |||||||
| (95% CI) | (6.0 to 27.5) | (0.0 to 0.0) | (6.8 to 31.6) | (6.5 to 31.1) | (0.0 to 0.0) | (9.0 to 37.5) | (7.7 to 57.4) | |||||||
| p Value (log-rank) | 0.403 | 0.007 | 0.047* | |||||||||||
| Survival time after the last mEHT (follow-up) (months) | ||||||||||||||
| Mean | 5.0±0.8 | 3.8±0.8 | 5.3±1.0 | 5.6±1.1 | 3.9±0.7 | 5.5±1.1 | 7.4±2.4 | |||||||
| Median | 3.3 | 2.9 | 3.4 | 3.5 | 2.4 | 3.4 | 3.3 | |||||||
| Range | 0.0–46.4 | 0.0–12.1 | 0.1–46.4 | 0.1–46.4 | 0.0–14.3 | 0.1–46.4 | 0.2–46.4 | |||||||
| 95% CI | 2.2 to 4.6 | 0.8 to 5.5 | 2.2 to 5.0 | 2.2 to 5.3 | 1.5 to 5.3 | 2.5 to 5.0 | 1.3 to 7.3 | |||||||
*Versus all GBM sample.
†Kaplan-Meier estimation.
CR, complete response; ddTMZ, dose-dense temozolomide; GBM, glioblastoma; HD, high dose; LD, low dose; mEHT, modulated electrohyperthermia; MST, median survival time; NOP, no. of patients; NR, not reached; PD, progressive disease; PR, partial response; SAT, supportive and alternative treatments; SD, stable disease.
Five patients (33%) showed stable disease and seven (47%) were in progressive disease status, giving a beneficial response rate (BRR) of 53% (see the section ’Bias assessment and limitations of the study').
Survival
All of the patients of the COI were included in the survival analysis (figure 2). Average follow-up since the first mEHT session was 8.4±1.2 months (median 6.0 months; range 0.7–47.3 months; 95% CI 4.6 to 7.5 months). Average follow-up since the last mEHT session (table 3) was 5.6±1.1 months (median 3.5 months; range 1 day to 46.4 months; 95% CI 2.2 to 5.3 months). For that period, 36 (67%) patients died, 2 (4%) were lost (censored) and 16 (30%) were alive at the end of the follow-up period (right-censored). The MST since the first diagnosis was 20.8 months (95% CI 15.2 to 25.1) and the 5-year OS was 13.5% (95% CI 1.0% to 26.0%). The MST since the first mEHT session was 7.7 months (95% CI 5.7 to 9.4). Survival since the first mEHT session at 12 and 24 months was 29.5% (95% CI 15.5% to 43.6%) and 18.8% (95% CI 6.5% to 33.1%), respectively (figure 3) (see the section ’Bias assessment and limitations of the study').
Figure 3.
Kaplan-Meier survival function of the patients treated with ddTMZ+mEHT (n=54) since diagnosis (A) and since first mEHT session (A1). C, censored; ddTMZ, dose-dense temozolomide; mEHT, modulated electrohyperthermia; S, survival function.
Safety
Unfortunately, the raw data presented does not contain safety data, so we rely on the safety data of the 140 patients reported in the primary paper.23 No grade III–IV toxicity was reported. Short-term (<2 hour) asthenia after treatment was encountered in 10% of the cases, rubor of the skin in 8%, oedema of fresh scars in <1%, subcutaneous fibrosis in 1%, burning blisters grade I–II in 2% and headache, fatigue and nausea (1–2 days) in 12% (see the section ’Bias assessment and limitations of the study').
Analysis of the results
Covariates survival analysis
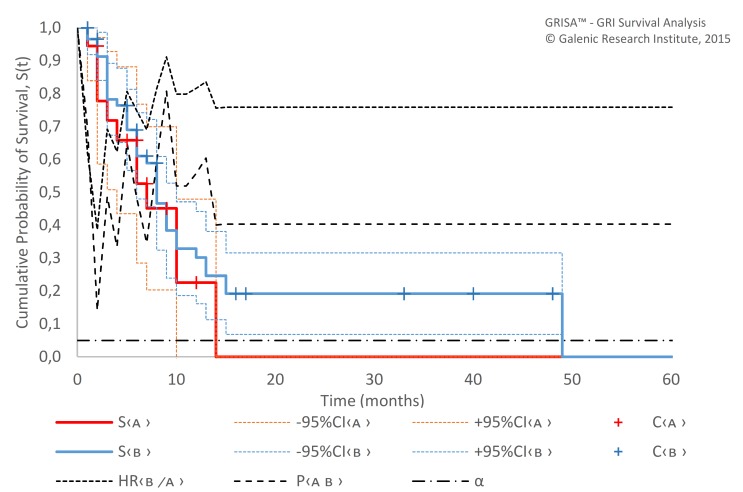
There was no difference in survival between patients treated with mEHT only (with or without SAT) and with the combination treatment (table 3, figure 4), neither by survival (MST since first mEHT 6.4 months (95% CI 3.1 to 9.9) vs 7.7 months (5.8 to 9.5), p=0.403) or by response (BRR 57% vs 53%, p=0.77), although the mEHT-only regimen was applied to significantly older patients (median 59.1 vs 49.8 years in the combination treatment sample, p=0.037) with KPS <60% unfit for chemotherapy and radiation.
Figure 4.
Survival (Kaplan-Meier estimate) since first mEHT session of ‘mEHT-only’ (A, n=18) and combination treatment (B, n=58) samples. α, probability of type I error; C, censored; mEHT, modulated electrohyperthermia; P, p value; S, survival function.
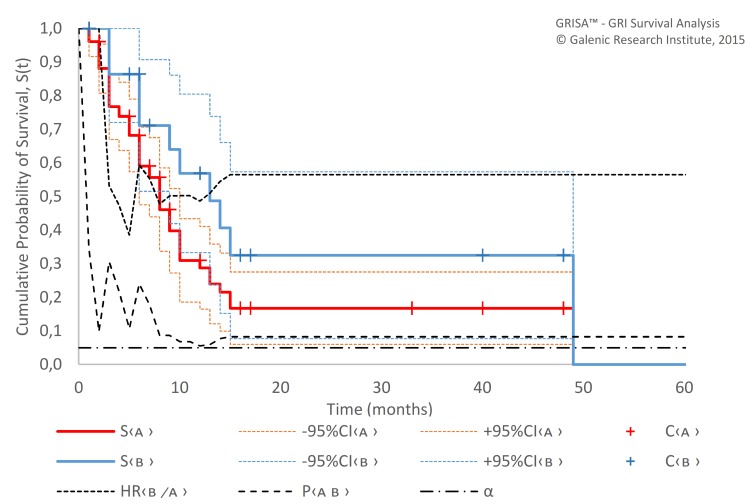
However, we did detect a significant difference between samples with LD-mEHT and HD-mEHT, both in survival since first mEHT (p=0.007; HR 2.19; 95% CI 1.21 to 3.95) and response (p=0.003) (table 4, figure 5). A similar pattern was shown in the analysis of the sample treated with SAT versus the sample without SAT (figure 6): the MST since first mEHT was 8.7 months (95% CI 7.2 to 11.4) with SAT vs 2.9 months (95% CI 2.3 to 5.5) only without SAT (p=0.004, HR 0.40 (95% CI 0.36 to 0.45)) (see the section ’Discussion').
Table 4.
Comparison of dose-dense temozolomide trials: patients’ characteristic
| Study (enrolment) |
NOP | Country | Study design | Inclusion | Med age | KPS | Pretreatment | MTAD | Other | Current treatment | |||
| SRG | RT | TMZ | Regimen | NOC | |||||||||
| Brandes et al153 | 33 | Italy | Phase II Prospective cohort uncontrolled |
Recurrent/progressive GBM in chemo-naïve patients with KPS ≥60 in SCC; 45% of met-MGMT | 57 | 90% (60–100) | 100% | 100% | 0% | N/A | R1: 100%: met 45.5%; re-op. 3% | 75 mg/m2/day qd X21/28 d | 153 ccls: mean 4.6, med 3 (1–15)* |
| Strik155
(2005–2007) |
18 | Germany | Recurrent/progressive GBM, KPS ≥50 in SCC: 1st relapse: 78%, 2nd relapse: 22% | 54.8 | 60% (50–100) | 100% | 100% | 100% (≥1 adj TMZ ccls) | 7.5 m† | R1/2: 77.8/22.2%; met. 46.2%; re-op. 33.3% | 100 mg/m2/day qd X21/28 d | 154 ccls, mean 7.3, med 5 (2–18)* | |
| Abacioglu156
(2006–2008) |
16 | Turkey | Recurrent/progressive GBM, KPS ≥70 in SCC | 50 | 80% (50–100) | 100% | 100% | 100% (med 6 ccls) | 13 (6–105)* | med 2 (1–8)* | |||
| Berrocal157 | 47 | Spain | Recurrent/progressive HGG with KPS ≥60 in SCC; WHO IV GBM 57%, WHO III 43% | 50 | (70%–80%) ECOG 1 | 81% | 100% | 100% (med 6 ccls) | 14 m (6–126)* | 85 mg/m2/day qd X21/28 d | med 2 (1–13)* | ||
| Norden154 | 55 | USA | Recurrent/progressive GBM with KPS ≥60 in SCC, standard (Stupp) pretreatment with ≥2 adjuvant cycles) | 57 | 90% (60–100) | 100% | 100% | 100% (≥2 adj TMZ ccls) (med six ccls (12–16)) | N/A | R1: 100%; R/P: 48%/52%, met. 65% | 100 mg/m2/day qd X21/28 d×12 ccls or until PD | N/A | |
| Sahinbas23
(2000–2005) |
54 | Germany | Retrospective cohort uncontrolled | Recurrent/progressive GBM, KPS ≥40 | 49.8 | 60% (40–100)‡ | 93% | 93% | 87% | 9.5 m (5.9–10.7)§ | 100 mg/m2/day qd X21/28 d+mEHT | 84 ccls, mean 1.6±0.1, med 1 (1–5)* | |
*Range.
†Corrected data (the originally reported survival in months is derived from weeks by division to 4 (eg, 32.8 weeks=8.2 months), which overprices survival for 9%).
‡Estimated.
§95% CI.
ccls, cycles; GBM, glioblastoma multiforme; ECOG, Eastern Cooperative Oncology Group; HGG, high-grade glioma; KPS, Karnofsky performance score; met, methylated MGMT promoter gene; MGMT, O6-methylguanine DNA methyltransferase; MTAD, median time after diagnosis; NOC, number of cycles;
SCC, stable clinical condition; qd, daily; N/A, not available; PD, progressive disease; R/P, relapse/progression; R1, first relapse/progression; R1/2, first/second relapse; re-op, re-operation; TMZ, temozolomide.
Figure 5.
Survival (Kaplan-Meier estimate) since first mEHT session of patients treated with low-dose mEHT (A, n=24) and high-dose mEHT (B, n=52). α, probability of type I error; C, censored; mEHT, modulated electrohyperthermia; P, p value; S, survival function.
Figure 6.
Survival (Kaplan-Meier estimate) since first mEHT session of patients with SAT (A, n=59) and without SAT (B, n=17). α, probability of type I error; C, censored; mEHT, modulated electrohyperthermia; P, p value; S, survival function; SAT, supportive and alternative treatments.
The sample of younger patients (<50 years) with HD-mEHT treatment showed the best results (figure 7): an MST since diagnosis of 23.9 months (95% CI 13.0 to not attained); a 5-year OS of 31.0% (95% CI 5.1 to 56.8); an MST since first mEHT session of 12.8 months (95% CI 8.2 to 48.1) and a BRR of 85.7%. Although the OS did not differ significantly from the complete sample (p=0.32), the survival since first mEHT and BRR were significantly better (p=0.047 and p=0.007, respectively).
Figure 7.
Survival (Kaplan-Meier estimate) since first mEHT session of all patients with GBM (A, n=76) and younger (<50 years) patients with high-dose mEHT (B, n=23). α, probability of type I error; C, censored; mEHT, modulated electrohyperthermia; P, p value; S, survival function.
Systematic comparator
Based on a systematic review152 and a narrative review12 of different ddTMZ regimens, five phase II, cohort, uncontrolled clinical trials addressing the ddTMZ 21/28 d regime were identified (table 4).
The Italian trial of Brandes et al 153 studied a highly selected group of CTX-naïve patients with good performance status (median KPS=90%). This was a specific design aimed to study the efficacy of TMZ at GBM recurrent in TMZ-naïve patients, and, due to this specificity, the results of Brandes et al are incomparable to both the current trial and the all other four ddTMZ trials, all made on TMZ-pretreated patients with KPS 60%–80%. The US trial by Norden et al 154 is another standalone trial with a median KPS of 90% and an extremely high share (65%) of patients with a methylated MGMT promoter (excluded from the comparison, see the section ’Bias assessment and limitations of the study'). The German trial by Strik et al 155 also stands alone: despite the worst patients’ performance status (median KPS=60%, which is usually considered unfit for CTX), the patients received the extensive course of ddTMZ (a median of five cycles; mean 7.3) with a modest toxicity. Two other studies, a Turkish study by Abacioglu et al 156 and a Spanish study by Berrocal et al 157 were the real-world19 studies without an obvious difference from everyday practice, although the trial by Berrocal et al claims to have selected TMZ-resistant patients, its findings do not differ from those of the trial by Abacioglu et al both by extent of TMZ pretreatment (median of six cycles) or by the time elapsed since diagnosis (14 vs 13 months).
The details of patients’ characteristic and treatment schedules are presented in table 4. The response and survival data are presented in table 5.
Table 5.
Comparison of dose-dense temozolomide trials: response and survival
| Study | NOP | Response | OS | Survival since relapse | |||||
| Total | EFR | CR | ORR | BRR | MST mo (95% CI) | MST mo (95% CI) | 1-Year OS (95% CI) | MTTP (95% CI) | |
| Brandes153 | 33 | 33 | 3% | 9% | 61% | N/A | 9.1 (7.1 to 14.5) | 38% | 3.7 (2.8 to 6.3) |
| Strik155 | 18 | 18 | 17% | 22% | 61% | 16.4* (17.9†) | 8.35* (9.1†) (N/A) | N/A | N/A |
| Abacioglu156 | 16 | 14 | 0% | 7% | 57% | N/A | 7 (5.7 to 8.2) | 0% | 3.0 (1.8 to 4.2) |
| Berrocal157 | 47 | 27 | 0% | 7% | 38%* | N/A | 5.1 (3.7 to 8.5)‡ | N/A | 2.0 (0.9 to 3.1) |
| Norden154 | 55 | 54 | 0% | 13% | 48% | 11.7 (8.1 to 16.2) | N/A | N/A | 1.8 (1.8 to 2.8) |
| Sahinbas23 | 54 | 15 | 7% | 20% | 53% | 20.8 (15.2 to 25.1) | 7.7 (5.7 to 9.4)§ | 29.5% (15.5–43.6) | N/A |
*Corrected data (the originally reported survival in months is derived from weeks by division to 4 (eg, 32.8 weeks=8.2 months), which overprices survival for 9%).
†Originally reported data (without correction).
‡For the complete sample of 47 patients, including 27 GBM and 20 WHO III tumours.
§Since first mEHT (not since relapse).
CR, complete response; BRR, beneficial response rate (ORR+stable disease); EFR, estimated for response; MST, median survival time (Kaplan-Meier estimation); MTTP, median time to progression; N/A, not available; NOP, number of patients; ORR, objective response rate (CR+partial response); OS, overall survival.
The survival data by Strik et al were corrected because the originally reported survival in months was derived from weeks by the division to 4 (eg, 32.8 weeks=8.2 ‘chemo months’), which overrated survival by an average of 9%.
Effect-to-treatment analysis
We used ETA to compare the trials according to the principles described in the ‘Statistical Methods’ section. The mST after relapse in patients receiving standard modern treatment (which can be defined as trimodal first-line and second-line treatment approximately equal to Stupp protocol9 was the parameter of comparison. Since the expected (reference) value of mST is absent in the literature, we deducted it from the available data as 4.775 months (95% CI 3.9 to 5.6) (see online supplementary 2). Taking into account the worst MST of the study by Berrocal et al (5.1 months (95% CI 3.7 to 8.5)), this MST expectancy seems reasonable. For the further analysis, we considered this parameter as emST since relapse (in view of supposed normal distribution according to central limit theorem). For further comparisons, meta-analysis and economic evaluations, the median parameters of all trials (MST and number of cycles) were translated into means according to the ‘Statistical methods’ section.
bmjopen-2017-017387supp002.pdf (35.6KB, pdf)
The results of ETA show the advantage of the mEHT+ddTMZ regimen. The main comparator was the weighted average of three ddTMZ trials with comparable samples (weighted average (WA) (2–4)) (table 6).
Table 6.
Effect-to-treatment analysis: basic parameters
| No | Study | NOP | mST | p Value | Rank | LMG | p Value | mNC | p Value | ETR (95% CI) | p Value | Rank |
| 1 | Brandes153 | 33 | 9.95 (7.73–12.17) | 0.070 | 1 | 5.18 (2.79–7.56) | 0.104 | 4.60 (3.87–5.33) | <0.001 | 1.13 (0.72 to 1.80) | 0.273 | 2 |
| 2 | Strik155 | 18 | 8.35 (7.67–9.03) | 0.416 | 2 | 3.58 (1.98–5.17) | 0.506 | 7.30 (6.05–8.55) | <0.001 | 0.49 (0.31 to 0.70) | 0.001 | 6 |
| 3 | Abacioglu156 | 16 | 6.98 (6.23–7.73) | 0.345 | 6 | 2.20 (1.05–3.35) | 0.486 | 3.33 (2.43–4.22) | 0.004 | 0.66 (0.38 to 1.05) | 0.022 | 3 |
| 4 | Berrocal157 | 47 | 5.60 (4.16–7.04) | 0.031 | 7 | 0.83 (−0.86–2.51) | 0.073 | 4.55 (3.94–5.16) | <0.001 | 0.18 (−0.05 to 0.44) | <0.001 | 7 |
| 5 | WA (1–4) | 114 | 7.27 (6.30–8.24) | 0.638 | 4 | 2.50 (1.20–3.80) | 0.718 | 4.20 (3.82–4.57) | <0.001 | 0.59 (0.39 to 0.85) | 0.006 | 4 |
| 6 | WA (2–4)* | 81 | 7.16 (6.25–8.08) | 0.531 | 5 | 2.39 (1.13–3.65) | 0.633 | 4.13 (3.68–4.57) | <0.001 | 0.58 (0.37 to 0.83) | 0.005 | 5 |
| 7 | Sahinbas23 | 54 | 7.63 (6.52–8.74) | 1.000 | 3 | 2.85 (1.44–4.26) | 1.000 | 1.56 (1.31–1.81) | 1.000 | 1.83 (1.04 to 4.20) | 1.000 | 1 |
*Main comparator.
LMG, life months gained; NOP, number of patients; WA, weighted average; mNC, mean number of cycles treated; mST, mean survival time since relapse.
The WA of all ddTMZ studies (WA (1–4)) and standalone studies by Brandes et al and Strik et al were the additional comparators.
The mST in the mEHT+ddTMZ sample (7.625±0.57 m) was ranked third after the cohorts by Brandes et al and Srtik et al, and was significantly better than in the trial by Berrocal et al (5.6±0.73 m, p=0.031) and worse than in the sample by Brandes et al, with borderline significance (9.95±1.13 m, p=0.070); other differences were not significant (table 6). The differences by LMG were not significant. The mNC in the mEHT+ddTMZ sample (1.56±0.13) was significantly less compared with all cohorts and WAs (p≤0.004). The relative survival gain changes the ranking: ddTMZ+mEHT provided significantly better ETR (ETR=1.83 LMG/ccl (95% CI 1.04 to 4.20)) compared with all other cohorts and WAs (p<0.022), except the cohort by Brandes et al (ETR=1.13 LMG/ccl (95% CI 0.72 to 1.80), p=0.273).
To make ETRs comparable, the common denominator was estimated as a median of the mean number of cycles of all of the cohorts: median number of cycles (MNC)=4.2 cycles. To lead ETRs to the common denominator, attenuation modelling was performed in the range of CA 10%–25%×ccl-1 (table 7).
Table 7.
Effect-to-treatment analysis: 15% attenuation model estimation
| No | Study | MAST | p Value | PNC | CEST | CENC | METR | EER | p Value | CNTM | ||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | ||||||||||
| 1 | Brandes153 | 10.15 (9.24–11.06) | 0.943 | 6 | 9.64 | 4 | 1.20 (0.74–1.95) | 1.01 | 0.979 | ∞ | 2.56 | 1.59 | 0.99 | 1.65 | 1.59 | 91 |
| 2 | Strik155 | 8.40 (7.52–9.29) | 0.015 | 6 | 7.98 | 4 | 0.81 (0.44–1.48) | 0.68 | 0.302 | −2.56 | ∞ | 4.22 | 1.62 | 4.63 | 4.19 | −2.64 |
| 3 | Abacioglu156 | 7.34 (6.46–8.22) | <0.001 | 6 | 6.98 | 4 | 0.57 (0.37–0.89) | 0.48 | 0.016 | −1.59 | −4.22 | ∞ | 2.62 | −47.9 | 592 | −1.62 |
| 4 | Berrocal157 | 5.63 (4.76–6.51) | <0.001 | 6 | 5.35 | 3 | 0.19 (0.08–0.49) | 0.16 | <0.001 | −0.99 | −1.62 | −2.62 | ∞ | −2.48 | −2.63 | −1.00 |
| 5 | WA (1–4) | 7.44 (6.56–8.31) | <0.001 | 6 | 7.07 | 4 | 0.59 (0.40–0.88) | 0.50 | 0.015 | −1.65 | −4.63 | 47.9 | 2.48 | ∞ | 44.3 | −1.68 |
| 6 | WA (2–4)* | 7.34 (6.46–8.21) | <0.001 | 6 | 6.97 | 4 | 0.57 (0.39–0.85) | 0.48 | 0.011 | −1.59 | −4.19 | −592 | 2.63 | −44.3 | ∞ | −1.62 |
| 7 | Sahinbas23 | 10.10 (9.10–11.10) | 1.000 | 6 | 9.5 | 4 | 1.19 (0.59–2.40) | 1.00 | 1.000 | −91 | 2.64 | 1.62 | 1.00 | 1.68 | 1.62 | ∞ |
*Main comparator.
CA, coefficient of attenuation; CENC, cost-effective number of cycles; CEST, cost-effective survival time; EER, effect enhancement rate; MAST, maximal attainable survival time; METR, median effect-to-treatment ratio; WA, weighted average; PNC, peak number of cycles.
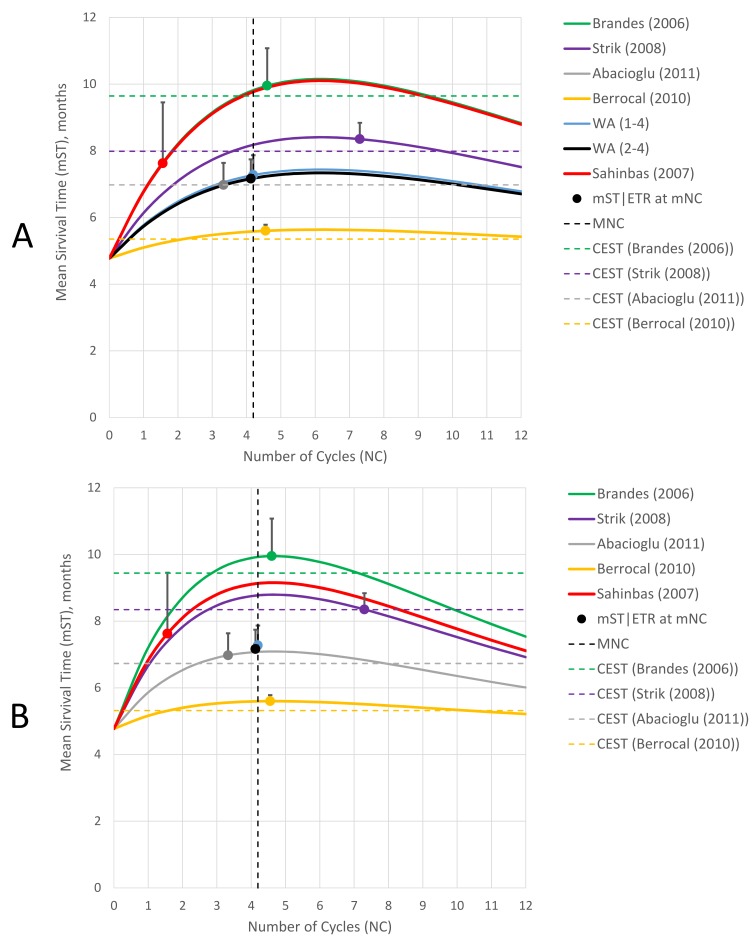
A CA level of 15% was chosen for the following analysis as an optimal prognosis (figure 8A). According to this scenario, the median ETR (METR) of the ddTMZ+mEHT cohort is 1.19 LMG/ccl (95% CI 0.59 to 2.40), which is significantly more than the METR of the main comparator (METR=0.57 LMG/ccl (95% CI 0.39 to 0.85), p=0.011) and other cohorts (p≤0.016), except that of the cohorts by Brandes et al (METR=1.20 LMG/ccl (95% CI, 0.74 to 1.95), p=0.979) and Strik et al (METR=0.81 LMG/ccl (95% CI 0.44 to 1.48), p=0.302). This scenario means that the ddTMZ+mEHT cohort would have to reach the maximal attainable survival time (MAST) of 10.10 months (95% CI 9.10 to 11.10) at the sixth cycle, which is significantly more than the MAST of the main comparator (7.34 months (95% CI 6.46 to 8.21), p<0.001) and other cohorts (p≤0.015), except the cohort by Brandes et al (10.15 months (95% CI 9.24 to 11.06), p=0.943).
Figure 8.
Effect-to-treatment analysis, attenuation modelling. (A) CA=15.0%; (B) CA=19.3%. CA, coefficient of attenuation; MNC, median number of cycles; mNC, mean number of cycles; mST | ETR: dot, mean survival time, ETR, line segment effect-to-tretament ratio.
Based on the CNTM criterion (table 7), the ddTMZ+mEHT regimen displayed strong and significant benefit versus the cohorts by Berrocal et al and Abacioglu et al and both WAs (CNTM=1.00–1.68 ccls/LMG, p<0.016), moderate and insignificant benefit versus cohort by Strik et al (CNTM=2.64 ccls/LMG, p=0.302) and no effect versus the cohort by Brandes et al (CNTM=−90.98 ccls/LMG, p=0.979).
Thus, our ETA suggests a strong and significant enhancement of the ddTMZ 21/28 d regimen by concurrent mEHT.
Sensitivity analysis
Sensitivity analysis was completed to validate the robustness of the ETA results. For this purpose, the lower and upper limits of CA were estimated (figure 8, table 8).
Table 8.
Effect-to-treatment analysis: sensitivity analysis
| No | Study | mST | CA=15% | CA=19.3% | ||||||
| CEST | METR | CNTM | p Value | CEST | METR | CNTM | p Value | |||
| 1 | Brandes153 | 9.95 (7.73–12.17) | 9.64 | 1.20 (0.74–1.95) | 90.98 (48.52 ─ 170.60) | 0.979 | 9.44 | 1.23 (0.75–2.01) | 5.30 (2.97 ─ 9.47) | 0.585 |
| 2 | Strik155 | 8.35 (7.67–9.03) | 7.98 | 0.81 (0.44–1.48) | −2.64 (−5.43 ─ −1.28) | 0.302 | 8.35 | 0.95 (0.49–1.86) | −11.73 (−24.39 ─ −5.64) | 0.830 |
| 3 | Abacioglu156 | 6.98 (6.23–7.73) | 6.98 | 0.57 (0.37–0.89) | −1.62 (−2.94 ─ −0.89) | 0.016 | 6.73 | 0.55 (0.36–0.83) | −2.04 (−3.43 ─ −1.22) | 0.016 |
| 4 | Berrocal157 | 5.60 (4.16–7.04) | 5.35 | 0.19 (0.08–0.49) | −1.00 (−2.77 ─ −0.36) | <0.001 | 5.32 | 0.20 (0.08–0.51) | −1.19 (−3.22 ─ −0.44) | 0.001 |
| 5 | WA (1–4) | 7.27 (6.30–8.24) | 7.07 | 0.59 (0.40–0.88) | −1.68 (−2.93 ─ −0.96) | 0.015 | 6.91 | 0.59 (0.40–0.88) | −2.26 (−3.70 ─ −1.38) | 0.027 |
| 6 | WA (2–4)* | 7.16 (6.25–8.08) | 6.97 | 0.57 (0.39–0.85) | −1.62 (−2.84 ─ −0.92) | 0.011 | 6.82 | 0.57 (0.38–0.85) | −2.14 (−3.52 ─ −1.30) | 0.018 |
| 7 | Sahinbas23 | 7.63 (6.52–8.74) | 9.6 | 1.19 (0.59–2.40) | ∞ | 1.000 | 8.69 | 1.04 (0.77–1.41) | ∞ | 1.000 |
*Main comparator.
CA, coefficient of attenuation; CEST, cost-effective survival time; CNTM, cycles needed to treat per life month gained; METR, median effect-to-treatment ratio; mST, mean survival time; WA, weighted average.
The lower limit of CA=15% is defined by the cohort by Abacioglu et al, in which the ascending mST reaches a CEST level (6.98 months) with other cohorts being between CEST and MAST (figure 8A); the upper limit at CA=19.3% is defined by the cohort by Strik et al, in which the descending mST reaches CEST=8.35 months (figure 8B). The CNTM of the ddTMZ+mEHT cohort versus the main comparator attenuates from strong to moderate from the lower to the upper limit (from 1.62 to 2.14 ccls/LMG), but remains significant (p=0.011–0.018). The extremum modelling shows that the CNTM of the ddTMZ+mEHT cohort versus the main comparator remains significant (p≤0.05) up to CA=24.4%. Thus, the result of the ETA is robust.
Safety comparison
The ddTMZ+mEHT regimen did not display any grade II–IV toxicity, whereas the ddTMZ regimens generated such toxicity events at a rate of 45%–92%, the difference was always highly significant (p<0.001) (table 9).
Table 9.
Comparison of dose-dense temozolomide trials: adverse events
| Adverse event | Grade | Brandes153 | Strik155 | Abacioglu156 | Berrocal157 | Norden154 | Sahinbas23 |
| NOP | 33 | 18 | 16 | 47 | 55 | 140 | |
| Total events | I–II | 122% | N/A | 44% | 194% | N/A | 34% |
| III–IV | 76% | 49% | 92% | 45% | 60% | 0% | |
| χ2 | 123 721 | 72 196 | 141 308 | 70 654 | 100 593 | ||
| p Value | <0.00001 | <0.00001 | <0.00001 | <0.00001 | <0.00001 | ||
| Lymphopoenia | I–II | 21% | 12% | 55% | 0% | ||
| III–IV | 24% | 14% | 80% | 28% | 38% | 0% | |
| Leucopenia | I–II | 21% | 20% | 28% | 0% | ||
| III–IV | 24% | 14% | 4% | 2% | 5% | 0% | |
| Neutropaenia | I–II | 9% | 17% | 0% | |||
| III–IV | 12% | 2% | 4% | 0% | |||
| Thrombocytopenia | I–II | 3% | 8% | 19% | 0% | ||
| III–IV | 3% | 5% | 8% | 11% | 4% | 0% | |
| Anaemia | I–II | 26% | 4% | 0% | |||
| III–IV | 3% | 2% | 0% | ||||
| Nausea/vomiting | I–II | 6% | 26% | 4% | |||
| III–IV | 3% | 2% | 2% | 0% | |||
| Fatigue | I–II | 4% | |||||
| III–IV | 5% | 0% | |||||
| Constipation/diarrhoea | I–II | 24% | 15% | 0% | |||
| III–IV | 3% | 0% | |||||
| Infection | I–II | 12% | 0% | ||||
| III–IV | 3% | 5% | 0% | ||||
| Headache | I–II | 4% | |||||
| Skin reactions | I–II | 12% | |||||
| Asthenia | I–II | 17% | 10% | ||||
| Gastrointestinal | I–II | 17% | 0% | ||||
| III–IV | 10% | 0% |
N/A, not available.
Grade I–II toxicity in the ddTMZ+mEHT cohort was mild. Since 4% of grade I nausea can be attributed to TMZ, total 30% of the mEHT-related events encountered. The main of them are grade I–II skin reactions (12%) and grade I short-term (<2 hours) post-treatment asthenia (10%).
Economic evaluation
Cost-effectiveness analysis
CEA was performed from the perspective of a health provider with a lifetime horizon. The goal of the CEA was to evaluate the cost-effectiveness of the ddTMZ+mEHT regimen versus ddTMZ only, so that only the direct costs for these two modalities were analysed. It was considered by default that other costs are dispensed proportionally and do not affect the estimation based on the direct costs (see the section ‘Bias assessment and limitations of the study’).
Two costs models were used for the CEA: conditionally termed ‘German’ and ‘US’ (see the section ‘Discussion’). The German model has lower costs and less variance compared with the US model. For both the models, end user prices for TMZ were estimated based on open sources (as at 21 January 2017): mean US$1.70/mg (95% CI 1.44 to 1.95) in the USA158 and €1.14/mg (95% CI 1.12 to 1.17) in Germany.159
The cost of the single mEHT session varies between countries, from $100 in Russia to $500 in Israel and South Korea (as at 2016). In the European Union, it varies in the range from €145.14 per session in Germany to €300–€400 in private clinics outside Germany. From the perspective of a health provider, this cost is limited by national regulations: for example, one deep HT session is reimbursed at a rate of €173 in Italy (National tariff nomenclature code 99.85.2) and €145.14 in Germany (GOA code 5854). In those countries where HT is not reimbursed by the health insurance system (eg, Spain and Austria), the median private cost is about €300.
Thus, from the perspective of a health provider, the mean cost of a single mEHT session in Germany was estimated as €145.14 with zero variance (95% CI €145.14 to €145.14), whereas in the USA the estimated mean is US$300 (95% CI US$234 to US$366) (table 10).
Table 10.
Calculated prices for economic evaluation
| Parameter | US model | German model | ||
| TMZ | mEHT | TMZ | mEHT | |
| US$/mg | US$/session | €/mg | €/session | |
| Mean (95% CI) | 1.70 (1.44 to 1.95) | 300 (234 to 366) | 1.14 (1.12 to 1.17) | 145 (145 to 145) |
| Median (range) | 1.77 (0.59–4.42) | 300 (150–500) | 1.14 (0.88–1.55) | 145 (145–300) |
mEHT, modulated electrohyperthermia; TMZ, temozolomide.
The results of the CEA are presented in table 11 (German model) and table 12 (US model).
Table 11.
Cost-effectiveness analysis (German model)
| Study | Costs, €mean (95% CI) | p Value | CUR, €/QALY (95% CI) | ICUR, €/QALY (95% CI) |
CURR, (95% CI) |
p Value | %CE25k | %CE30k | ICER €/QALYG (95% CI) |
∆C1000
€ |
∆E1000
QALYG |
| Brandes153 | 14 905 (14 586 to 15 225) | <0.001 | 24 292 (20 263 to 28 321) | 4421 (2090 to 6752) | 1.22 (1.10 to 1.35) | 0.061 | 53.57% | 76.5% | 28 706 (−5529 to 62 940) | 5 561 695 | 193.8 |
| Strik155 | 31 539 (30 863 to 32 215) | <0.001 | 61 250 (53 939 to 68 561) | 41 379 (37 491 to 45 267) | 3.08 (2.83 to 3.34) | <0.001 | 0.00% | 0.0% | 367 368 (−710 070 to 1 444 806) | 22 195 135 | 60.4 |
| Abacioglu156 | 14 379 (14 071 to 14 687) | <0.001 | 33 429 (30 717 to 36 141) | 13 558 (11 791 to 15 325) | 1.68 (1.57 to 1.80) | <0.001 | 0.12% | 1.8% | −92 957 (−352 869 to 166 956) | 5 035 150 | −54.2 |
| Berrocal157 | 16 721 (16 362 to 17 079) | <0.001 | 48 419 (39 174 to 57 665) | 28 548 (23 705 to 33 391) | 2.44 (2.16 to 2.71) | <0.001 | 0.31% | 0.7% | −43 717 (−91 130 to 3697) | 7 377 172 | −168.8 |
| WA (1–4) | 17 922 (17 538 to 18 306) | <0.001 | 39 967 (35 985 to 43 949) | 20 096 (17 787 to 22 405) | 2.01 (1.86 to 2.16) | <0.001 | 0.04% | 0.3% | −291 167 (−1 869 626 to 1 287 291) | 8 577 947 | −29.5 |
| WA (2–4) | 18 043 (17 657 to 18 430) | <0.001 | 40 845 (36 926 to 44 763) | 20 973 (18 692 to 23 255) | 2.06 (1.90 to 2.21) | <0.001 | 88.8% | 99.2% | −226 212 (−1 153 427 to 701 004) | 8 699 523 | −38.5 |
| WA (2–3)* | 18 138 (17 750 to 18 527) | <0.001 | 40 424 (36 758 to 44 091) | 20 553 (18 384 to 22 722) | 2.03 (1.89 to 2.18) | <0.001 | 0.02% | 0.2% | −302 629 (−1 934 133 to 1 328 875) | 8 794 882 | −29.1 |
| Sahinbas23 | 9344 (9199 to 9488) | 1.000 | 19 871 (17 719 to 22 024) | 0 | 1.00 | 1.000 | 88.8% | 99.2% | 0 | 0 | 0.0 |
*Main comparator.
∆C1000, costs difference per 1000 patients; %CE30k, %CE at CET €30 000; CUR, cost-utility ratio; ∆E1000, effect difference per 1000 patients (QALY gained); ICER, incremental cost-effectiveness ratio; mEHT, modulated electrohyperthermia; RCUR, relative CUR; TMZ, temozolomide; QALY, quality-adjusted life-year; QALYG, QALY gained.
Table 12.
Cost-effectiveness analysis (US model)
| Study | Costs, US$mean (95% CI) | p Value | CUR, US$/QALY (95% CI) | ICUR, US$/QALY (95% CI) |
CURR, (95% CI) |
p Value | %CE30k | %CE50k | ICER US$/QALYG (95% CI) |
∆C1000
$ |
∆E1000
QALYG |
| Brandes153 | 22 106 (18 799 to 25 413) | 0.003 | 36 028 (28 866 to 43 189) | 3324 (−1280 to 7927) |
1.10 (0.96 to 1.25) |
0.472 | 3.01% | 84.02% | 34 727 (−12 095 to 81 549) |
6 728 332 | 193.8 |
| Strik155 | 46 775 (39 779 to 53 772) | <0.001 | 90 841 (76 123 to 105 558) | 58 136 (50 122 to 66 151) | 2.78 (2.45 to 3.11) |
<0.001 | 0.02% | 0,21% | 519 683 (−1009,423 to 2 048 790) |
31 397 527 | 60.4 |
| Abacioglu156 | 21 325 (18 135 to 24 515) | 0.007 | 49 579 (42 820 to 56 338) | 16 875 (12 433 to 21 317) | 1.52 (1.35 to 1.68) |
<0.001 | 0.17% | 51,27% | −109 798 (−426 187 to 206 591) |
5 947 408 | −54.2 |
| Berrocal157 | 24 799 (21 089 to 28 508) | <0.001 | 71 811 (56 003 to 87 619) | 39 107 (30 569 to 47 644) | 2.20 (1.89 to 2.51) |
<0.001 | 0.26% | 1,56% | −55 827 (−122 100 to 10 445) |
9 420 880 | −168.8 |
| WA (1–4) | 26 580 (22 604 to 30 555) | <0.001 | 59 276 (50 498 to 68 053) | 26 571 (21 289 to 31 853) | 1.81 (1.61 to 2.02) |
<0.001 | 0.08% | 2,34% | −380 229 (−2 447 832 to 1 687 373) |
11 201 761 | −29.5 |
| WA (2–4) | 26 760 (22 757 to 30 763) | <0.001 | 60 577 (51 756 to 69 398) | 27 873 (22 572 to 33 174) | 1.85 (1.64 to 2.06) |
<0.001 | 0.06% | 1,96% | −295 965 (−1 515 454 to 923 523) |
11 382 070 | −38.5 |
| WA (2–3)* | 26 901 (22 877 to 30 925) | <0.001 | 59 954 (51 427 to 68 481) | 27 249 (22 075 to 32 423) |
1.83 (1.63 to 2.04) |
<0.001 | 0.06% | 2,04% | −396 520 (−2 540 572 to 1 747 533) |
11 523 498 | −29.1 |
| Sahinbas23 | 15 378 (12 703 to 18 052) | 1.000 | 32 704 (27 215 to 38 193) | 0 | 1.00 (1.00 to 1.00) |
1.000 | 4.45% | 94,60% | 0 | 0 | 0.0 |
*Main comparator.
∆C1000, costs difference per 1000 patients; %CE50k, %CE at CET $50 000; CUR, cost-utility ratio; ∆E1000, effect difference per 1000 patients (QALY gained); ICER, incremental cost-effectiveness ratio; mEHT, modulated electrohyperthermia; RCUR, relative CUR; TMZ, temozolomide; QALY, quality-adjusted life-year; QALYG, QALY gained.
Along with four single cohorts of comparison, three WA were assessed. WA (1–4) combines all the cohorts, WA (2–4) excludes the cohort by Brandes et al as a selected cohort (selection bias-free average), WA (2–3) also excludes the cohort by Berrocal et al in view of its very low survival gain, which significantly affected the final results (low-result bias-free average, the main comparator).
The mean costs of ddTMZ+mEHT regimen both in the German (€9344 (95% CI 9199 to 9488)) and US (US$15 378 (12 703 to 18 052)) models were significantly less versus all cohorts and WAs (p<0.05 in all cases). The cohort by Abacioglu et al displayed the lowest costs (€14379 (95% CI14071 to 14687)) and US$21 325 (95% CI 18 135 to 24 515), respectively) and the cohort by Strik et al the highest (€31 539 (95% CI 30 863 to 32 215) and US$46 775 (95% CI 39 779 to 53 772)); the main comparator WA (2–3) costs were calculated to be €18 138 (95% CI 17 750 to 18 527) and US$26 901 (95% CI 22 877 to 30 925)).
For estimation of the CUR, we used the weighted average index of health-related quality of life of all five cohorts (0.74 quality-adjusted life year (QALY)/LY) to counterweight the initial difference of the samples (range of median KPS 60%–90%) not connected with the treatment (table 1).
The CUR of the ddTMZ+mEHT regimen, both in the German (€19 871/QALY (95% CI 17 719 to 22 024)) and the US (US$32 704/QALY (95% CI 27 215 to 38 193)) models was also less versus all comparators. The difference was highly significant (p≤0.001), except for the cohort by Brandes et al (€24 292/QALY (95% CI 20 263 to 28 321)), p=0.061 and US$36 028/QALY (95% CI 28 866 to 43 189), p=0.472). The main comparator WA (2–3) was calculated as €40 424/QALY (95% CI 36 758 to 44 091) and US$59 954/QALY (95% CI 51 427 to 68 481), p<0.001 for both.
In the German model, versus CET €25 000/QALY (%CE25k) and €30 000/QALY (%CE30k), the %CE for the ddTMZ+mEHT regimen was 88.8% (%CE25k) and 99.2% (%CE30k) (ie, it was cost-effective vs both CETs). All the other comparators showed negligible %CE (0%–2.5%), except the cohort by Brandes et al, which was also mainly cost-effective at both CETs (%CE25k = 53.6% and %CE30k = 76.5%). In the US model, versus CETs US$30 000/QALY (%CE30k) and US$50 000/QALY (%CE50k), the %CE for the ddTMZ+mEHT regimen was 4.5% (%CE30k) and 94.6% (%CE50k) (ie, it was cost-effective vs CET=US$50 000 only). Two other cohorts were also mainly cost-effective versus CET=US$50 000: namely the cohorts by Brandes et al (%CE50k = 84%) and Abacioglu et al (%CE50k = 51.3%); the %CE50k of all of the WAs was negligible (2.0%–2.3%).
As for comparative cost-effectiveness, only the cohort by Brandes et al showed an ICER of less than the applied CETs (€28 706/QALY (95% CI −5 529 to 62 940) and US$34 727/QALY (95% CI −12 095 to 81 549). All of the other cohorts and WAs were not cost-effective with the ICER ranging from €43 717/QALY/US$55 827/QALY to €367 368/QALY/US$519 683/QALY.
Sensitivity analysis
The sensitivity of the CEA was analysed by using an equal cost-effectiveness test, that is, by exploring the value of a key parameter in which the value of the relative CUR (CURR) of the ddTMZ+mEHT regimen and the main comparator (WA (2–3)) equals to 1.0 (or ICUR=0). For this purpose, the following variables were tested: the price of the mEHT session; the number of TMZ application days (days on) over a 28 days cycle; the price of TMZ; the number of cycles of ddTMX+mEHT.
The equivalent price of the mEHT session is €683 in the German model, and US$1013 in the US model and the coefficient of reliability of the CEA result (CR, the ratio of a key parameter of CE-equivalent model and the standard model) is 3.4/4.7 (table 13).
Table 13.
Cost-effectiveness analysis: sensitivity analysis
| Parameter | US model | German model | ||||||||
| TMZ | mEHT US$/sess |
mNC | CR | TMZ | mEHT €/sess |
mNC | CR | |||
| Price, US$/mg | Days on | Price, €/mg | Days on | |||||||
| Standard regimen | 1.70 (1.44–1.95) | 21 | 300 (234–366) | 1.60 | 1.14 (1.12–1.17) | 21 | 145.14 (145–145) | 1.60 | ||
| Maximal mEHT price | NC | NC | 1013.47 | NC | 3.38 | NC | NC | 683.65 | NC | 4.71 |
| Minimal TMZ days on | NC | 6,21 | NC | NC | 3,38 | NC | 4.46 | NC | NC | 4.71 |
| Minimal TMZ price | 0,50 | NC | NC | NC | 3.38 | 0.24 | NC | NC | NC | 4.71 |
| Maximal TMZ+mEHT cycles | NC | NC | NC | 2.86 | 1.79 | NC | NC | NC | 3.17 | 2.05 |
CR, coefficient of reliability; mEHT, modulated electrohyperthermia; mNC, mean number of cycles; NC, no change; TMZ, temozolomide.
The equivalent price of TMZ is US$0.50/mg in the US model and €0.24/mg in the German model; once again with CR=3.4/4.7. Since these key parameters (prices) do not affect the treatment efficacy, their equivalent values do not need any size-dependent correction. The result means that the ddTMZ+mEHT regimen is cost-effective in the entire range of possible prices with double to quadruple redundancy.
The equivalent number of TMZ ‘days on’ is 4.46 days in the German model and 6.21 days in the US model, once again with CR=3.4/4.7. This time, the key parameter affects the treatment efficacy, because the diminished dose (days) of ddTMZ can decrease the effectiveness and, therefore, can increase the ddTMZ+mEHT/ddTMZ CURR and cause an offset of the equivalence point to the lower values of ‘days on’. This means that the ddTMZ+mEHT regimen, most probably, keeps the cost-effectiveness up to the standard 5/28 d regimen and below it, and the cost-effectiveness of mEHT could be generalised for the entire range of TMZ treatment of recurrent gliomas.
The maximal equivalent number of ddTMZ+mEHT cycles is 2.86 in the US model and 3.17 cycles in German model (CR=1.8/2.1). This key parameter also affects the treatment efficacy, because, with an increase of cycle number of the ddTMZ+mEHT regimen, the treatment efficacy and CUR will rise with an offset of the equivalence point towards the longer course. At the least, this result means that the length of the ddTMZ+mEHT regimen can be doubled without loss of cost-effectiveness.
Thus, the sensitivity analysis confirms that the results of the CEA are remarkably stable, with double to quadruple redundancy.
Budget impact analysis
We estimated a budget impact of the treatment of 1000 patients per year (tables 11 and 12) with a time horizon of 1 year; versus the main comparator, the saving (ΔC1000) is €8 794 882/US$11 523 498 per year (German/US model) with 29.1 years of survival gain (∆E1000). The average saving ranged from €8 577 947/US$11 201 761 to €8 794 882/US$11 523 498 with 29.1–38.5 QALY gained. To extrapolate the economic results to a larger time horizon, the depreciation rate of 20% per year must be applied.
Cost-benefit analysis
CBA was performed from the perspective of a large neurooncology centre treating >150 patients with recurrent GBM per year (table 14, table 15).
Table 14.
Cost-benefit analysis (US model)
| Parameter | Rate | Year | Total | |||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |||
| Number of patients per year | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 1200 | |
| Mean sessions per patient | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | ||
| Sessions per year | 2691 | 2691 | 2691 | 2691 | 2691 | 2691 | 2691 | 2691 | ||
| Sessions per day | 11 | 11 | 11 | 11 | 11 | 11 | 11 | 11 | ||
| Number of units | 1 | 1 | ||||||||
| Capital costs* | 400 000 | 400 000 | ||||||||
| Service costs | 12%† | 48 000 | 48 000 | 48 000 | 48 000 | 48 000 | 48 000 | 288 000 | ||
| Depreciation | 15% | 60 000 | 60 000 | 60 000 | 60 000 | 60 000 | 60 000 | 60 000 | 420 000 | |
| Reimbursement per session | 30 000 | 30 000 | 30 000 | 30 000 | 30 000 | 30 000 | 30 000 | 30 000 | ||
| Reimbursement per year | 807 300 | 807 300 | 807 300 | 807 300 | 807 300 | 807 300 | 807 300 | 807 300 | 6 458 400 | |
| Operational costs per year | 50%‡ | 538 200 | 538 200 | 538 200 | 538 200 | 538 200 | 538 200 | 538 200 | 538 200 | 4 305 600 |
| Economy per patient | 20% | 11 523 | 9219 | 7375 | 5900 | 4720 | 3776 | 3021 | 2417 | 47 951 |
| Economy per year | 1 728 525 | 1 382 820 | 1 106 256 | 885 005 | 708 004 | 566 403 | 453 122 | 362 498 | 7 192 632 | |
| Earnings per year | 2 535 825 | 2 190 120 | 1 913 556 | 1 692 305 | 1 515 304 | 1 373 703 | 1 260 422 | 1 169 798 | 13 651 032 | |
| Total costs per year | 938 200 | 598 200 | 646 200 | 646 200 | 646 200 | 646 200 | 646 200 | 646 200 | 5 413 600 | |
| Economy and EBIT | 1 597 625 | 1 591 920 | 1 267 356 | 1 046 105 | 869 104 | 727 503 | 614 222 | 523 598 | 8 237 432 | |
| EBIT | −130 900 | 209 100 | 161 100 | 161 100 | 161 100 | 161 100 | 161 100 | 161 100 | 1 044 800 | |
| Cumulative EBIT | −130 900 | 78 200 | 239 300 | 400 400 | 561 500 | 722 600 | 883 700 | 1 044 800 | ||
*Acquisition price+shipment + installation+training.
†Share of capital cost per year.
‡Profit rate.
§Annual depreciation rate of the saving.
EBIT, earnings before interest and taxes.
Table 15.
Cost-benefit analysis (German model)
| Parameter | Rate | Year | Total | |||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |||
| Number of patients per year | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 1200 | |
| Mean sessions per patient | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | 17.9 | ||
| Sessions per year | 2691 | 2691 | 2691 | 2691 | 2691 | 2691 | 2691 | 2691 | ||
| Sessions per day | 10.8 | 10.8 | 10.8 | 10.8 | 10.8 | 10.8 | 10.8 | 10.8 | ||
| Number of units | 1 | 1 | ||||||||
| Capital costs* | 300 000 | 300 000 | ||||||||
| Service costs | 12.0%† | 36 000 | 36 000 | 36 000 | 36 000 | 36 000 | 36 000 | 216 000 | ||
| Depreciation | 15.0% | 45 000 | 45 000 | 45 000 | 45 000 | 45 000 | 45 000 | 45 000 | 315 000 | |
| Reimbursement per session | 145.14 | 145.14 | 145.14 | 145.14 | 145.14 | 145.14 | 145.14 | 145.14 | ||
| Reimbursement per year | 390 572 | 390 572 | 390 572 | 390 572 | 390 572 | 390 572 | 390 572 | 390 572 | 3 124 574 | |
| Operational costs per year | 50%‡ | 260 381 | 260 381 | 260 381 | 260 381 | 260 381 | 260 381 | 260 381 | 260 381 | 2 083 049 |
| Economy per patient | 20%§ | 8795 | 7036 | 5629 | 4503 | 3602 | 2882 | 2306 | 1844 | 36 597 |
| Economy per year | 1 319 232 | 1 055 386 | 844 309 | 675 447 | 540 358 | 432 286 | 345 829 | 276 663 | 5 489 509 | |
| Earnings per year | 1 709 804 | 1 445 958 | 1 234 880 | 1 066 019 | 930 929 | 822 858 | 736 401 | 667 235 | 8 614 083 | |
| Total costs per year | 560 381 | 305 381 | 341 381 | 341 381 | 341 381 | 341 381 | 341 381 | 341 381 | 2 914 049 | |
| Economy and EBIT | 1 149 423 | 1 140 576 | 893 499 | 724 637 | 589 548 | 481 477 | 395 019 | 325 854 | 5 700 034 | |
| EBIT | −169 809 | 85 191 | 49 191 | 49 191 | 49 191 | 49 191 | 49 191 | 49 191 | 2 10 525 | |
| Cumulative EBIT | −169 809 | −84 619 | −35 428 | 13 762 | 62 953 | 112 143 | 161 334 | 210 525 | ||
*Acquisition price+shipment+installation+training.
†Share of capital costs per year.
‡Profit rate.
§Annual depreciation rate of the economy.
EBIT, earnings before interest and taxes.
The main assumptions of the CBA are as follows: mean sessions per patient is equal to that of SOI; the mEHT device does not generate revenues other than healthcare system reimbursement for the treatment of those patients; the mEHT device operates in 12 hours/day mode; the capital costs including acquisition costs, shipment, installation and training are €300 000 in the German model and US$400 000 in the US model; the service costs rate is 12% of the capital costs per year with 2-year free of charge guarantee service; the depreciation of the mEHT equipment at a rate of 15% per year; the norm of profit of the healthcare provider is 50% (operational costs are 67% of revenues); the saving obtained as a result of the introduction of the ddTMZ+mEHT regimen depreciates at a rate of 20% per year; the saving is not included in EBIT; no price discount/inflation rate is used; the time horizon is 8 years.
Our CBA shows that use of an mEHT device is profitable with the above parameters and generates the total revenues in amount of €3 124 574/US$6 458 400 with EBIT €210 525/US$1 044 800 per mEHT device over 8 years, provided that operational costs are €2 083 049/US$4 305 600 for that period (€260 381/US$538 200 per year). With respect to the saving due to the use of the ddTMZ+mEHT regimen instead of ddTMZ only, the total economic effect (saving+EBIT) over the 8-year period is €5 700 034/US$8 237 432 per mEHT device.
Discussion
Clinical evaluation
In a general comparison, the ddTMZ+mEHT cohort has revealed a non-significantly better mean survival time (mST=7.63 months (95% CI 6.52 to 8.74)) compared with the main comparator, the pooled mST of three trials on TMZ-pretreated patients (7.16 months (95% CI 6.25 to 8.08), p=0.531).
Covariates survival analysis has revealed the comparable efficacy of mEHT and ddTMZ, at least in weakened patients (figure 4), suggesting the feasibility of mEHT as a single treatment in those patients, for which CTX is impossible in view of toxicity or bad performance. The advantage of mEHT over chemotherapy was shown elsewhere in GBM22 and other cancers.30 33 41 44
Despite the shown significant dependence of survival from mEHT dose (p=0.007), it is difficult to say how the difference in the mEHT dose actually affects the response and survival because the LD-mEHT sample included weakened patients with longer time since diagnosis to first mEHT (median 9.9 months (95% CI 6.1 to 11.6)), shortest treatment time (median 0.5 months (95% CI 0.4 to 0.6) vs 1.9 months (95% CI 1.2 to 2.8) in the HD-mEHT sample, p=0.0001) and highest rate of treatment termination (38% vs 0% in the HD-mEHT sample, p<0.0001) (table 2). More correctly, the LD-mEHT was rather a sequence of poor patient states, which likely accounts for the decrease in survival. In other words, the impossibility to reach an adequate mEHT dose for weakened patients made their prognosis dismal.
The dependence of survival on SAT use is questioned. The extremely low survival in the ‘No SAT’ sample (2.9 months (95% CI 2.3 to 5.5), almost twofold lower than the expected value) undisputedly indicates for the selection of patients with bad prognosis and small life expectancy. Comparison of the samples showed that ‘No SAT’ includes patients with significantly less TMZ cycles (mean 1.1±0.1 cycles vs 1.7±0.1, p=0.017) and mEHT sessions (mean, 11.2±0.5; median, 10 vs 19.9±0.4; median, 15, p=0.013) with a higher proportion of LD-mEHT (47% vs 27%, RR=1.74 (0.90–3.34), p=0.12). Therefore, this survival difference shows a tendency to not apply SAT to patients with a bad prognosis, and that these patients were heavily undertreated.
The shown significantly reduced toxicity of ddTMZ+mEHT is, in our opinion, caused by the short course of TMZ in the COI (median one cycle only). TMZ is known as a relatively safe alkylating drug. Its toxicity appears after two to three cycles and a development of the grade III–IV lymphopoenia (the main adverse event) becomes virtually inevitable after six cycles. Thus, the data presented here allow us to conclude that mEHT per se is safe, but do not allow us to estimate the modifying effect of mEHT on TMZ toxicity (if such an effect exists).
Effect-to-treatment analysis
Direct comparison of the ddTMZ+mEHT results with the other ddTMZ studies is impossible because the ddTMZ+mEHT treatment in the participating tertiary centres was not continued up to the maximal attainable course (MAC). The median number of cycles was just one, and only 15% of treatments were stopped in view of the disease progression, without limiting toxicity. In tertiary centres, the end of treatment is caused either by the physician’s decision, by the patient’s personal decision, economic reasons, by an applied protocol or because of a combination of these reasons. Therefore, the treatment is typically limited by one to three cycles only, whereas in clinics the median duration of MAC of recurrent GBM is five cycles.18 Therefore, ETA was used for the comparison.143
The idea of ETA is simple and based on the ETR, that is, life months gained per a typical 28 days treatment cycle, which is considered a unit of a CTX treatment. By ETR, we identified ddTMZ+mEHT as the uncontested leader, with 1.83 LMG/ccl vs 1.13 LMG/ccl of the nearest competitor (cohort by Brandes et al) and 0.58 LMG/ccl of the main comparator (WA 2–4) (table 6), although in terms of conventional MST-based comparison, ddTMZ+mEHT was ranked third (behind the cohorts by Brandes et al and Strik et al).
The next step of the ETA follows from the idea of attenuation of the treatment effect. This is a typical feature of all cancer treatments because of the ability of cancer cells to rapidly develop multiple mechanisms of acquired resistance to an applied treatment. This is especially correct for diseases such as GBM, which almost inevitably progresses, and for TMZ, for which many distinct mechanisms of acquired resistance are available,160–162 so that virtually all patients develop resistance to TMZ. As a result, the effectiveness of any cancer treatment decays (attenuates).
The offered equation of the attenuation is based on ETR and CA. It is suggested that CA is common for all the ddTMZ cohorts. The maximum value of CA corresponds to the assumption that the treatments have almost reached MAST, which equals the extremum of the function. In this case, CA=15%/ccl exactly matches this assumption (table 7A). Although the cohort by Strik et al is located after the maximum of the function, it is acceptable because this cohort is likely overtreated (mNC=7.3 ccls vs 3–4.5 ccls in other ddTMZ cohorts).
The natural sequence of the attenuation idea is incomparability of ETRs obtained in a different number of cycles. This is because an early ETR with the lower impact of attenuation is higher than a later one. For the correct comparison, ETRs should be led to the common denominator. The best common denominator is the MNC, which equals 4.2 cycles. The resulting parameter METR allows us to correctly compare the different treatments. In this comparison, COI (METR=1.19 LMG/ccl (95% CI 0.59 to 2.40)) significantly surpasses the main comparator WA (2–4) (METR=0.57 LMG/ccl (95% CI 0.39 to 0.85), p=0.011) and all other comparators (METR=0.19–0.59, p=0.00–0.016), except the cohorts by Brandes et al (METR=1.20 LMG/ccl (0.74–1.95), p=0.979) and Strik et al (METR=0.81 LMG/ccl (0.44–1.48), p=0.302) (table 7). In other words, the efficacy of IOI in CTX-pretreated patients with a median KPS of 60%–70% is the same as in the selected cohort of CTX-naïve patients with a median KPS of 90%, and significantly better compared with the TMZ-pretreated cohorts.
With CA 15%/ccl, the COI reach a MAST of 10.10 months (95% CI 9.10 to 11.10) at the sixth cycle, which is significantly more than the MAST of the main comparator (7.34 months (95% CI 6.46 to 8.21), p<0.001) and other cohorts, except the cohort by Brandes et al (10.15 months (95% CI 9.24 to 11.06), p=0.943). The next assumption is that the CA of the ddTMZ+mEHT regimen is lower than that of the ddTMZ-only regimen. Actually, the mechanisms of resistance to the RF field have to differ substantially from those of CTX. Little is known about such acquired resistance. TTF reports a possibility of selection or development of giant-cell GBM with syncytial-type cells,163 which is reasonable adaptation for 100 kHz range, where the large size of a cell improves the shielding from the external field, although it is a single-case observation, and it is hardly applicable to high-frequency range (HFR), where size difference is not decisive. Taking into account the results of long-term (6 months to 3 years) mEHT treatments,33 45 47 especially in patients with multiple liver metastases, which is a similarly lethal condition as GBM, where mEHT displayed the ability to support PFS up to 3 years, and even to revert the progression after stopping mEHT33 (ie, mEHT does not lose its efficacy over years), the assumption that the CA of mEHT is lower than that of TMZ looks reasonable. If we assume that the CA=12.5%/ccl, the ddTMX+mEHT cohort can attain a MAST of 10.84 months, or of 12.13 months with a CA=10.0%.
The last parameter of ETA, called ‘cycles needed to treat per one life month gained’ (CNTM), is an analogue of the known parameter ‘number needed to treat’ (NNT). The CNTM shows the number of cycles of the compared treatments, at which the difference in their MST reaches 1 month. Positive CNTM means a benefit, negative means detriment, and the value of CNTM characterises the strength of the effect (figure 9). In this comparison, all of the cohorts displayed strong to moderate detriment versus the ddTMZ+mEHT regimen (table 7), except the cohort by Brandes et al (no effect).
Figure 9.
Cycles needed to treat per one life-month gained (CNTM) scale.
Thus, the ETA has allowed us to uncover the real efficacy of the ddTMZ+mEHT treatment, which was impossible to assess with the conventional comparison by general end points, and has suggested that mEHT strongly and significantly enhances the efficacy of the ddTMZ 21/28 d regimen with significantly less toxicity.
Economic evaluation
We studied two options for the mEHT application. The first, so-called German option, is specific for a high-income country with rigid governmental regulation of the medical market, which leads to relatively low prices for pharmaceuticals with low variance (mean price of TMZ is €1.14/mg (95% CI 1.12 to 1.17)) and fixed and low enough prices for medical procedures (in this case, €145.14/sess with zero variance (95% CI 145.14 to 145.14)). The second, so-called US option, is specific for a high-income country with lower governmental regulation, which leads to relatively high prices for pharmaceuticals with higher variance (mean price of TMZ US$1.70/mg (95% CI 1.44 to 1.95)) and variable and high enough prices for medical procedures (in this case, US$300/sess (95% CI 234 to 366)).
First, the adequacy of our costs estimation (€18 138 (95% CI, 17 750 to 18 527)) and US$26 901 (95% CI 22 877 to 30 925) in the main comparator) have to be assessed (tables 11 and 12). For this purpose, the result was compared with a recent study by Ray et al,19 where expenditures for cancer drugs (without supportive drugs like antiemetics, pain killers, neutropaenia related, etc) for a 6-month period were assessed as US$13 555–US$17 204. Since the study was devoted to TMZ treatment and taking into account the difference in price of TMZ and other cancer drugs, 95%–99% of these ‘cancer drugs’ costs can be attributed to TMZ. Although the reported range of US$13 555–US$17 204 appears to be much less than the average US$27 000 displayed in the current assessment, it should be noted that the general practice of recurrent GBM treatment is based almost exclusively on the standard TMZ 5/28 d regimen9, with 100–150 mg/m2/day. The current regimen ddTMZ 21/28 d 75–100 mg/m2/day consumes 2.1–4.2 times more TMZ per course. Therefore, it is at least two to three times more expensive. Thus, the estimated costs range for the ddTMZ 21/28 d regimen is US$27 000–US$50 000, and the cost estimation of the current trial is adequate. It also corresponds to other estimations.17 18
The result suggests the significant advantage of the ddTMZ+mEHT regimen over all the comparators (p<0.003) (except the cohort by Brandes et al, against which the advantage was not significant (p=0.061–0.472)). In the German model (table 11), the ddTMZ+mEHT regimen was cost-effective versus both the €25 000/QALY and €30 000/QALY cost-effectiveness thresholds (CET) (88.8% and 99.2% of cost-effective cases, respectively), whereas the main comparator was not cost-effective (%CE of 0.0% and 0.2%). ICER versus ddTMZ+mEHT varied from €43 717/QALY to €367 368/QALY (except for the cohort by Brandes et al, which displayed an ICER of €28 706/QALY).
In the US model (table 12), the pattern was the same with more pronounced differences. The ddTMZ+mEHT regimen was not cost-effective versus CET=US$30 000/QALY (%CE=4.5% only), and only CET US$50 000 /QALY provides cost-effectiveness (%CE=94.6%), whereas the main comparator showed a negligible cost-effectiveness (%CE50k = 2.0%). ICER versus ddTMZ+mEHT varied from US$55 827/QALY to US$519 683/QALY (except for the cohort by Brandes et al, which displayed an ICER of US$34 727/QALY).
The CET (or willingness-to-pay (WTP)) is set by the National Institute for Health and Care Excellence (NICE) at £20 000–£30 000/QALY,164 although studies show that the acceptable limit can be lower (up to £13–£14 000).165 In high-income countries, a CET of €/US$/£30 000 is considered standard. The CET for low-income and middle-income countries is suggested by WHO at the level of their triple GDP per capita for each disability-adjusted life-year,166 which is typically close to the above NICE WTP. For end-of-life applications, where the QALY increase could be negligible, a CET of £50 000 is supposed by NICE.167 Finally, for some orphan diseases, the third CET of about £100 000 is offered.168 Since a treatment of the recurrent GBM can be considered an end-of-life application, a CET of US$50 000/QALY is applicable in the US model.
Thus, the economic evaluation suggests that the inclusion of mEHT in the ddTMZ 21/28 d regimen makes it cost-effective versus the applicable CET levels, whereas the ddTMZ 21/28 d alone is not cost-effective. The sensitivity analysis suggests that this estimation is highly reliable, with double to quadruple redundancy. The sensitivity analysis also suggests that the advantage of ddTMZ+mEHT in cost-effectiveness remains true throughout the entire applicable range of prices for TMZ and the mEHT procedure, as well as for the TMZ intercycle variances (ie, up to the lowest 5/28 d regimen). It also suggests that the ddTMZ+mEHT course can be at least doubled without loss of cost-effectiveness. Since the CENC (ie, the number of cycles at which MST reaches 95% of MAST) for the ddTMZ+mEHT regimen equals 3.0 (table 7), this means the all-range cost-effectiveness of the regimen.
The BIA suggests significant savings from the introduction of mEHT, which can be estimated as about €8 794 882 per year per 1000 patients in the German model and US$11 523 498 per year per 1000 patients in the US model, with an additional 29.1–38.5 QALY gained per 1000 patients.
Finally, the CBA shows that the mEHT, from the perspective of a single neurooncology centre, is profitable in both of the tested models (tables 14 and 15).
Thus, the introduction of mEHT generates savings for budget and healthcare providers and significant profit for the latter.
Applicability of mEHT in GBM treatment
The result obtained in this study looks promising, although a single retrospective trial does not provide the necessary grounds for generalisation. Nevertheless, if the result is confirmed in a further meta-analysis, it will provide an excellent ground for generalisation. At the least, it means that mEHT can be recommended as an enhancer of all ddTMZ regimens in the treatment of recurrent GBM, and, probably, for the regular 5/28 d regimen too. Next, as shown by the covariates survival analysis (figure 5), mEHT is feasible as a single treatment in those patients for which chemotherapy is impossible because of toxicity or bad performance. Thus, mEHT has a capacity as a salvage treatment after the failure of chemotherapy. With respect to the known low toxicity of mEHT22–26 and its possibility to restore the performance and chemosensitivity,33 45 47 this salvage treatment can, in some cases, provide an opportunity to continue chemotherapy in previously failed patients.
Bias assessment and limitations of the study
Only 15 patients (28%) in the COI were assessed for response. Although natural selection is supposed, selection bias is not excluded. Consequently, the response rate was excluded from the analysis.
Although follow-up period was short enough (median 6.0 months; range 0.7–47.3 months; 95% CI 4.6 to 7.5 months), it is close to the MST since the first mEHT session (7.7 months, 95% CI 5.7 to 9.4), and the mean of the follow-up (8.4±1.2 months) exactly fits the CI of the MST. Thus, the MST value is robust. Although 1-year and 2-year survivals since first mEHT are less robust in view of the short follow-up, they are also well within the range of the follow-up time (0.7–47.3 months) and, therefore, are reliable enough. Nevertheless, in view of their lower reliability, the 1-year and 2-year survivals were excluded from the comparison, which was based solely on the robust MST value.
The absence of the safety data matched to the COI is not a serious limitation because the absence of severe toxicity in the whole sample also excludes it for the subsamples. So, the absence of grade III–IV toxicity and limited grade I–II toxicity (up to 30%) findings are relevant and robust, although the rate and distribution of the mild toxicity in the COI are approximate.
We excluded the trial by Norden et al 154 from the ETA because of a lack of information on the number of cycles and some uncertainties (eg, survival definition and some statistical uncertainties). The modest effect shown would not affect the comparison.
The main possible bias of a retrospective study is a selection bias. We consider the probability of the selection bias as minimal in the SOI because, in addition to the assurances of the authors of no exclusions from the sample, 153 patients with HGG is consistent with such patients in the enrolling centres, which are small tertiary centres not specialised in neurooncology (and, in the case of the Institute of Microtherapy, in cancer care at all), for the 5-year period. Thus, we consider the sample as consecutive patients with HGG enrolled for the stated period without exclusions or selection. The declared inclusion criteria (recurrence/progression of HGG with KPS ≥40%) rather describe the sample than limit it in any way. The absence of exclusion criteria confirms this suggestion.
At the same time, some compared ddTMZ studies showed an obvious selection bias. First, the study by Brandes et al, in which the selection of CTX-naïve patients is presumed by the protocol, but the selection of patients with good performance (median KPS=90%) also seems to be present (although this might be a natural sequence of the inclusion criteria). The same extremely favourable KPS is shown in the excluded trial by Norden et al, which also showed an extremely high share of MGMT methylated patients (65% vs 45%–46% in the other trials, which exceeds the highest historical level of about 60%13 (table 6). Also, the large share of re-operations in the study by Strik et al (33.3%) might significantly improve the observed survival, making it hardly attributable to the applied ddTMZ treatment.
The difference in dosage between the ddTMZ regimens was not analysed in the ETA (although it was considered in the economic evaluation). As many studies had displayed, there is no or negligible difference in efficacy of different doses of ddTMZ regimens, and sometimes lower doses were preferable.169 Moreover, the possibility of dose reduction/escalation in all of the protocols makes such an analysis impossible. The average dose is never reported and cannot be retrieved from the reported data. We do not exclude the possibility that the actual doses were similar to each other.
There is an unequal MST starting point bias because the MST in the ddTMZ+mEHT cohort was calculated since the first session of mEHT, rather than since relapse/progression in the other cohorts. Since the SOI was carried out in tertiary centres, it is normal that mEHT was applied not just after relapse but rather as the second-line treatment of the relapse. Based on the median time of 9.0 months elapsed since diagnosis to the first mEHT treatment, and estimated 7.5 months MPFS in GBM, the delay of mEHT since relapse can be 1–1.5 months. This could significantly change the results in favour of the ddTMZ+mEHT cohort (eg, estimated MST since relapse can reach 9 months instead of 7.6 months, as in the best ddTMZ studies). At the same time, due to this delay, probably some first-line treatments of relapse in the SOI were not included in the assessment. Based on the delay, the median one treatment cycle is supposed to be added, increasing the mean CTX cycles number to 2–2.5, which can somewhat change the economic results in favour of concurrent ddTMZ studies. Thus, the bias of not equal MST starting point rather distorts the comparison in favour of ddTMZ studies, although economically it is somewhat counterbalanced.
It should also be noted that the two ‘real-life’ studies by Abacioglu et al and Berrocal et al displayed the longest time from initial diagnosis to enrolment (13 and 14 months, respectively), which is responsible for the low MST values in these trials. We consider that, in the weighted average assessment, this difference is counterbalanced by early enrolment in the trials by Brandes et al and Strik et al and the median position of the SOI (table 7). It is also counterbalanced (and even outbalanced) by the unequal histology bias, since the trials by Abacioglu et al and Berrocal et al included WHO III tumours (28% and 43%, respectively) with much longer survival, which can be, in turn, the reason for the delayed relapse.
Nevertheless, there is a reciprocal dependence between the time to enrolment (relapse) and the MST since the enrolment (the SOI displays the medium-power correlation, Pearson’s correlation 0.35), which is not considered in the ETA but seems counterbalanced or even outbalanced in favour of the ddTMZ cohorts.
It is worth noting that all of the ‘real-life’ studies (ie, studies by Sahinbas et al, Berrocal et al and Abaciouglu et al) showed the same median age of 50 years, whereas the supposedly selection-biased trials included the older patients (55–57 years).
MEHT required additional visits to the hospital (two to three times a week), which means additional transportation costs and influences cost-effectiveness from the patient’s perspective, although this does not affect the assessment from the health provider perspective. At the same time, since a planned mEHT session typically does not require the physician’s involvement (a nursing procedure), we do not assume a better treatment control. Moreover, such control seems much more extensive in the compared prospective trials, where the follow-up included weekly complete blood counts,154 155 physical and neurological examinations every 4 weeks,153 155 or even biweekly155 and brain imaging with MRI every 8 weeks154 or earlier if indicated.153 To compare, only 28% of patients in the SOI underwent brain imaging (the specificity of small tertiary centres). Better treatment control could significantly improve the treatment results.
Finally, all of the compared ddTMZ studies recruited only patients in a stable condition, whereas there was no such limitation in the SOI.
In general, although the assessment is distorted in favour of the ddTMZ studies, it still allows us to make an unambiguous conclusion on the advantage of the combination of mEHT and TMZ.
Also, on completion of the paper, we have identified one additional ddTMZ 21/28 d cohort in phase III randomised trial of Brada et al.169 The result of this cohort (MST since relapse 6.6 months after median four ddTMZ cycles, which results in METR ≤0.5 LMG/ccl) would not in any way affect the results obtained.
Generalisability of the results
The results of the sensitivity analysis of the CEA supposes the generalisability of the CEA results to the entire range of application of TMZ at recurrent GBM. There is a probability of similar enhancement of TMZ efficacy and cost-efficiency by mEHT can also be achieved in the treatment of the newly diagnosed GBM, although, to the best our knowledge, mEHT has never been studied in such a setting.
Since TMZ is considered the current most effective CTX treatment of GBM, the results of the covariate survival analysis (figure 4) can be generalised to CTX. Thus, mEHT as a single treatment can be considered in those patients for which CTX is impossible because of toxicity or bad performance, and mEHT has a capacity as a salvage treatment after the failure of CTX.
Perspectives of research
This study creates a good basis for the further research on mEHT enhancement of the GBM treatments with the possibility to develop a cost-effective alternative. First, we will estimate the other existing mEHT cohort trials, followed by a systematic review with meta-analysis. Second, a new cohort and randomised trials at recurrent and newly diagnosed GBM are warranted.
Verifiability of the results
To provide the possibility to verify the results obtained, raw data of the study are available in online supplementary 3.
bmjopen-2017-017387supp003.pdf (36.6KB, pdf)
Conclusions
Our ETA suggests that mEHT strongly and significantly enhances the efficacy of the ddTMZ 21/28 d regimen (p=0.011), with a maximum attainable MST of 10.10 months (95% CI 9.10 to 11.10). The ddTMZ+mEHT cohort has displayed significantly less toxicity than the ddTMZ 21/28 d cohorts (no grade III–IV toxicity vs 45%–92%, respectively) because of the shorter TMZ course. mEHT per se displays high safety with a mild grade I–II toxicity (30% of events), mainly of mild skin reactions (12%) and short (<2 hours) post-treatment asthenia (10%). Our CEA suggests that the ddTMZ+mEHT regimen is cost-effective compared with the applicable cost-effectiveness thresholds €$25 000–50 000/QALY, whereas ddTMZ 21/28 d only is not cost-effective, with ICER versus ddTMZ+mEHT ranging from €43 717/QALY to €367 368/QALY. This CEA result is highly reliable with double to quadruple redundancy. Our BIA suggests a significant saving from the introduction of mEHT, which can be estimated from €8 577 947 to $11 523 498 with 29.1–38.5 QALY gained per 1000 patients. The CBA, from the perspective of a single neurooncology centre, suggests that mEHT is profitable and will generate a total revenue of €3 124 574–$6 458 400 with total economic effect (economy +EBIT) of €5 700 034–$8 237 432 per mEHT device over an 8-year period. After confirmation of these findings, mEHT can be recommended as an enhancer for all ddTMZ regimens in the treatment of recurrent GBM, and, probably, for the regular 5/28 d regimen. mEHT can be applied as a single treatment in those patients for which chemotherapy is impossible because of its toxicity or bad performance, and as a salvage treatment after the failure of chemotherapy, with a possibility to restore the patient’s performance and chemosensitivity and subsequently continue chemotherapy.
bmjopen-2017-017387supp004.doc (88KB, doc)
Supplementary Material
Acknowledgments
The authors would like to thank Professor Andras Szasz from Szent István University (Godollo, Hungary), who provided the primary data for the study. The authors would like to thank all the other authors of the original study,23 namely Dr Hüseyin Sahinbas and Professor Dietrich HW Grönemeyer from the Institute of Microtherapy of University Witten-Herdecke (Bochum, Germany) and Dr Eckhard Böcher from Clinic ‘Closter Paradise’ (Soest, Germany) for conducting this remarkable trial, although they may not agree with all the interpretations and conclusions of this paper. The authors would also like to thank proof-reading-service.com for proofreading of the paper.
Footnotes
Contributors: SVR is the only contributor and guarantor, solely responsible for its writing and contents. No other persons or professional writers are involved.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: Patient level data are available in the online supplementary 3. Consent for data sharing was not obtained but the presented data are completely anonymised, and risk of identification is absent.
References
- 1. Ostrom QT, Bauchet L, Davis FG, et al. The epidemiology of glioma in adults: a “state of the science” review. Neuro Oncol 2014;16:896–913. 10.1093/neuonc/nou087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Dolecek TA, Propp JM, Stroup NE, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2005–2009. Neuro Oncol 2012;14(Suppl 5):v1–49. 10.1093/neuonc/nos218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Venur VA, Peereboom DM, Ahluwalia MS. Current medical treatment of glioblastoma. Cancer Treat Res 2015;163:103–15. 10.1007/978-3-319-12048-5_7 [DOI] [PubMed] [Google Scholar]
- 4. Reithmeier T, Graf E, Piroth T, et al. BCNU for recurrent glioblastoma multiforme: efficacy, toxicity and prognostic factors. BMC Cancer 2010;10:30 10.1186/1471-2407-10-30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Brandes AA, Fiorentino MV. The role of chemotherapy in recurrent malignant gliomas: an overview. Cancer Invest 1996;14:551–9. [DOI] [PubMed] [Google Scholar]
- 6. Weller M, Cloughesy T, Perry JR, et al. Standards of care for treatment of recurrent glioblastoma–are we there yet? Neuro Oncol 2013;15:4–27. 10.1093/neuonc/nos273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Seystahl K, Wick W, Weller M. Therapeutic options in recurrent glioblastoma–An update. Crit Rev Oncol Hematol 2016;99:389–408. 10.1016/j.critrevonc.2016.01.018 [DOI] [PubMed] [Google Scholar]
- 8. Hau P, Baumgart U, Pfeifer K, et al. Salvage therapy in patients with glioblastoma: is there any benefit? Cancer 2003;98:2678–86. 10.1002/cncr.11845 [DOI] [PubMed] [Google Scholar]
- 9. Stupp R, Mason WP, van den Bent MJ, et al. National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005;352:987–96. [DOI] [PubMed] [Google Scholar]
- 10. Stupp R, Hegi ME, Mason WP, et al. EORTC Brain Tumour and Radiation Oncology Groups; NCIC Clinical Trials Group. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 2009;10:459–66. [DOI] [PubMed] [Google Scholar]
- 11. Chamberlain MC. Temozolomide: therapeutic limitations in the treatment of adult high-grade gliomas. Expert Rev Neurother 2010;10:1537–44. [DOI] [PubMed] [Google Scholar]
- 12. Nagane M. Dose-dense temozolomide: is it still promising? Neurol Med Chir 2015;55:38–49. 10.2176/nmc.ra.2014-0277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hegi ME, Diserens AC, Gorlia T, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 2005;352:997–1003. 10.1056/NEJMoa043331 [DOI] [PubMed] [Google Scholar]
- 14. Pegg AE, Dolan ME, Moschel RC. Structure, function, and inhibition of O6-alkylguanine-DNA alkyltransferase. Prog Nucleic Acid Res Mol Biol 1995;51:167–223. [DOI] [PubMed] [Google Scholar]
- 15. Hegi ME, Liu L, Herman JG, et al. Correlation of O6-methylguanine methyltransferase (MGMT) promoter methylation with clinical outcomes in glioblastoma and clinical strategies to modulate MGMT activity. J Clin Oncol 2008;26:4189–99. 10.1200/JCO.2007.11.5964 [DOI] [PubMed] [Google Scholar]
- 16. Gilbert MR, Wang M, Aldape KD, et al. Dose-dense temozolomide for newly diagnosed glioblastoma: a randomized phase III clinical trial. J Clin Oncol 2013;31:4085–91. 10.1200/JCO.2013.49.6968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Wu B, Miao Y, Bai Y, et al. Subgroup economic analysis for glioblastoma in a health resource-limited setting. PLoS One 2012;7:e34588 10.1371/journal.pone.0034588 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wasserfallen JB, Ostermann S, Leyvraz S, et al. Cost of temozolomide therapy and global care for recurrent malignant gliomas followed until death. Neuro Oncol 2005;7:189–95. 10.1215/S1152851704000687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ray S, Bonafede MM, Mohile NA. Treatment Patterns, Survival, and Healthcare Costs of Patients with Malignant Gliomas in a Large US Commercially Insured Population. Am Health Drug Benefits 2014;7:140–9. [PMC free article] [PubMed] [Google Scholar]
- 20. Kovic B, Xie F. Economic Evaluation of Bevacizumab for the First-Line Treatment of Newly Diagnosed Glioblastoma Multiforme. J Clin Oncol 2015;33:2296–302. 10.1200/JCO.2014.59.7245 [DOI] [PubMed] [Google Scholar]
- 21. Chamberlain MC. Temozolomide: therapeutic limitations in the treatment of adult high-grade gliomas. Expert Rev Neurother 2010;10:1537–44. 10.1586/ern.10.32 [DOI] [PubMed] [Google Scholar]
- 22. Wismeth C, Dudel C, Pascher C, et al. Transcranial electro-hyperthermia combined with alkylating chemotherapy in patients with relapsed high-grade gliomas: phase I clinical results. J Neurooncol 2010;98:395–405. 10.1007/s11060-009-0093-0 [DOI] [PubMed] [Google Scholar]
- 23. Sahinbas H, Grönemeyer DHW, Böcher E, et al. Retrospective clinical study of adjuvant electro-hyperthermia treatment for advanced brain-gliomas. Dtsch Z Onkol 2007;39:154–60. [Google Scholar]
- 24. Fiorentini G, Giovanis P, Rossi S, et al. A phase II clinical study on relapsed malignant gliomas treated with electro-hyperthermia. In Vivo 2006;20:721–4. [PubMed] [Google Scholar]
- 25. Hager ED, Dziambor H, Popa C, et al. Clinical Response and Overall Survival of Patients with Recurrent Gliomas Grade III-IV Treated with RF Deep Hyperthermia – An Update Program & Abstracts: ICHS 2004 (26th ICHS Conference): Shenzhen (China): Chinese Medical Association, 2004:50–1. [#CHO-13]. [Google Scholar]
- 26. Hager ED, Sahinbas H, Groenemeyer DH, et al. Prospective phase II trial for recurrent high-grade malignant gliomas with capacitive coupled low radiofrequency (LRF) deep hyperthermia. ASCO 2008 Annual Meeting Proceedings. J Clin Oncol 2008;26:2047 http://meeting.ascopubs.org/cgi/content/abstract/26/15_suppl/2047 [Google Scholar]
- 27. Yeo SG. Definitive radiotherapy with concurrent oncothermia for stage IIIB non-small-cell lung cancer: A case report. Exp Ther Med 2015;10:769–72. 10.3892/etm.2015.2567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Lee DY, Park JS, Jung HC, et al. The Outcome of the Chemotherapy and Oncothermia for Far Advanced Adenocarcinoma of the Lung: Case Reports of Four Patients. Adv Lung Cancer 2015;4:1–7. [Google Scholar]
- 29. Szasz A. Current status of oncothermia therapy for lung cancer. Korean J Thorac Cardiovasc Surg 2014;47:77–93. 10.5090/kjtcs.2014.47.2.77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lee DY, Haam SJ, Kim TH, et al. Oncothermia with Chemotherapy in the Patients with Small-Cell Lung Cancer. Conference Papers in Medicine. 2013;2013:7[#910363]. [Google Scholar]
- 31. Gadaleta-Caldarola G, Infusino S, Galise I, et al. Sorafenib and locoregional deep electro-hyperthermia in advanced hepatocellular carcinoma: A phase II study. Oncol Lett 2014;8:1783–7. 10.3892/ol.2014.2376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ferrari VD, de Ponti S, Valcamonico F, et al. Deep electro-hyperthermia (EHY) with or without thermo-active agents in patients with advanced hepatic cell carcinoma: phase II study. ASCO 2007 Annual Meeting Proceedings, 29 Jun 1905. J Clin Oncol 2007;25:15168 http://meeting.ascopubs.org/cgi/content/abstract/25/18_suppl/15168 [Google Scholar]
- 33. Lorencz P, Csejtei A. Experience in the treatment of liver metastases with special reference to the consequences of interruption of long-run treatments. ICHS 2012 (31st ICHS Conference & 2nd Int Oncothermia Symp), Oct 12-14, 2012, Budapest, Hungary. Oncothermia J 2013;7:292–4. http://www.oncothermia-journal.com/journal/page/2013/Vol7.ENG/ [Google Scholar]
- 34. Volovat C, Volovat SR, Scripcariu V, et al. Second-line chemotherapy with gemcitabine and oxaliplatin in combination with loco-regional hyperthermia (EHY-2000) in patients with refractory metastatic pancreatic cancer - preliminary results of prospective trial. Romanian Reports in Physics 2014;66:166–74. [Google Scholar]
- 35. Hager ED, Süße B, Popa C, et al. Complex therapy of the not in sano respectable carcinoma of the pancreas – a pilot study. 21 National Cancer Congress of the German Cancer Society, Mar 7-11, 1994, Hamburg, Germany. J Cancer Res Clin Oncol 1994;120:#P1.04.15. [Google Scholar]
- 36. Strauss CA, Kotzen JA, Baeyens A, et al. Oncothermia in HIV-Positive and -Negative Locally Advanced Cervical Cancer Patients in South Africa. Conference Papers in Medicine. 2013:3[#293968] 10.1155/2013/293968 [DOI]
- 37. Minnaar CA, Kotzen JA, Bayens A. Hyperthermia combined with radiation in cervical cancer. presented at ICHS 2016 (34th Annual Conference of ICHS). Pesaro (Italy), 2016. [Google Scholar]
- 38. Lee SY, Lee NR. Positive response of a primary leiomyosarcoma of the breast following salvage hyperthermia and pazopanib. Korean J Intern Med 2016. dx.doi.org/10.3904/kjim.2015.242 [Epub ahead of print 15 Apr 2016]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Akutsu Y. A phase I / II study of EHY-2000 oncothermia therapy for advanced esophageal cancer. ACHO 2014: 6th Asian Congress of Hyperthermic Oncology (ACHO) & 31st Japanese Congress of Thermal Medicine (JCTM), Sep 5-6, 2014, Fukui City, Japan. Therm Med 2014;30:#LS1-2. [Google Scholar]
- 40. Pang CLK. Clinical Research on Integrative Treatment of Colon Carcinoma with Oncothermia and Clifford TCM Immune Booster. Oncothermia J 2012;5:24–41. http://www.oncothermia-journal.com/journal/2012/Clinical_research_on_integrative_treatment_of_colon_carcinoma.pdf [Google Scholar]
- 41. Hager ED, Dziambor H, Hohmann D, et al. Deep hyperthermia with radiofrequencies in patients with liver metastases from colorectal cancer. Anticancer Res 1999;19:3403–8. [PubMed] [Google Scholar]
- 42. Fiorentini G, Milandri C, Dentico P, et al. Deep electro-hyperthermia with radiofrequencies combined with thermoactive drugs In patients with liver metastases from colorectal cancer (CRC): A phase II clinical study. ICHS 2012 (31st ICHS Conference & 2nd Int Oncothermia Symp), Oct 12-14, 2012, Budapest, Hungary. Oncothermia J 2013;7:358 http://www.oncothermia-journal.com/journal/page/2013/Vol7.ENG/ [Google Scholar]
- 43. Kovaliov AA, Mel’nichuk MP. [Locoregional electrohyperthermia in complex treatment of resectable rectal cancer], 2. [In Russian]. Promeneva diagnostika I promeneva terapiia 2008. [Google Scholar]
- 44. Pang CLK, Zhang X, Wang Z, et al. Local modulated electro-hyperthermia in combination with traditional Chinese medicine versus intraperitoneal chemoinfusion in the treatment of peritoneal carcinomatosis with malignant ascites: a phase II randomized trial. Mol Clin Oncol. 2016;6:723–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Jeung T, Ma S, Choi J, et al. Results of Oncothermia Combined with Operation, Chemotherapy and Radiation Therapy for Primary, Recurrent and Metastatic Sarcoma. Case Rep Clin Med 2015;4:157–68. [Google Scholar]
- 46. Volovat SR, Volovat C, Scripcariu V, et al. The Results of Combination of Ifosfamid and Locoregional Hyperthermia (EHY 2000) in Patients with advanced Abdominal Soft-Tissue Sarcoma after Relapse of First Line Chemotherapy. Romanian Reports in Physics 2014;66:175–81. [Google Scholar]
- 47. Jeung TS, Sy M, Yu J, et al. Cases That Respond to Oncothermia Monotherapy. Conference Papers in Medicine. 2013:12[#392480]. [Google Scholar]
- 48. Szasz A, Szasz N, Szasz O. Oncothermia: Principles and Practices. Springer Netherlands, 2011. 565 стр. ISBN: 978-90-481-9497-1. [Google Scholar]
- 49. Fiorentini G, Szasz A. Hyperthermia today: electric energy, a new opportunity in cancer treatment. J Cancer Res Ther 2006;2:41–6. https://tspace.library.utoronto.ca/bitstream/1807/5441/1/cr06010.pdf [DOI] [PubMed] [Google Scholar]
- 50. Szasz A, Morita T. [Heat Therapy in oncology. New paradigm in Hyperthermia]. Tokyo: Nippon Hvoronsha; 2012:208 ISBN: 978-4-535-98377-9. [In Japanese]. [Google Scholar]
- 51. Szigeti GP, Hegyi G, Szasz O. Hyperthermia versus Oncothermia: Cellular Effects in Cancer Therapy. Conference Papers in Medicine. 2013;2013:274687–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Andocs G, Rehman MU, Zhao Q-L, et al. Comparison of biological effects of modulated electro-hyperthermia and conventional heat treatment in human lymphoma U937 cells. Cell Death Dis 2016;2:16039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Wang YS. Different cytotoxic effect from different hyperthermia devices. Comparison of the oncotherm-labehy and the thermotron RF-8 in an in vitro model. 6th Asian Congress of Hyperthermic Oncology (ACHO) & 31st Japanese Congress of Thermal Medicine (JCTM), 5-6 Sep 2014, Fukui City, Japan. Therm Med 2014;30:54(S2-1). [Google Scholar]
- 54. Salengke S, Sastry SK. Experimental investigation of ohmic heating of solid-liquid mixtures under worst-case heating scenarios. J Food Eng 2007;83:324–36. [Google Scholar]
- 55. Miklavcic D, Pavselj N, Hart FX. Electric properties of tissues Wiley Encyclopedia of Biomedical Engineering. 2006:12. [Google Scholar]
- 56. Scholtz B, Anderson R. On Electrical Impedance Scanning - Principles and Simulations. Electromedica. [Internet] 2000;68:35–44. http://www.biophysicssite.com/Documents/Siemens_EIT.pdf [Google Scholar]
- 57. Mikac U, Demsar F, Beravs K, et al. Magnetic resonance imaging of alternating electric currents. Magn Reson Imaging 2001;19: 845–56. http://citeseerx.ist.psu.edu/viewdoc/download?doi = 10.1.1.538.9683&rep = rep1&type = pdf [DOI] [PubMed] [Google Scholar]
- 58. Wang D. Three dimensional radio frequency current density imaging [Doctor of Philosophy dissertation]. Toronto: Department of Electrical and Computer Engineering, University of Toronto, 2010:167 https://tspace.library.utoronto.ca/bitstream/1807/26388/6/Wang_Dinghui_201011_PhD_thesis.pdf (accessed 16 Feb 2017). [Google Scholar]
- 59. Szasz O, Szigeti GP, Szasz A. Hyperthermia dosing and depth of effect. Private communication, 2017. [Google Scholar]
- 60. Polk C, Postow E. Handbook of Biological Effects of Electromagnetic Fields. Boca Raton, New York, London, Tokyo: CRC Press, 1996:15. [Google Scholar]
- 61. Szasz A, Szasz O, Szasz N. Electrohyperthermia: a new paradigm in cancer therapy. Deutsche Zeitschrift für Onkologie 2001;33:91–9. [Google Scholar]
- 62. Roussakow S, Szasz A, Szasz O, et al. [A method of treatment of solid tumors by oncothermia (medical technology)] Способ лечения солидных злокачественных опухолей методом онкотермии (медицинская технология). Moscow, 2011:96 [In Russian]. [Google Scholar]
- 63. Szasz A, Vincze G, Szasz O, et al. An energy analysis of extracellular hyperthermia. Electromagn Biol Med 2003;22:103–15. [Google Scholar]
- 64. Szasz O, Heating SA. Efficacy and Dose of Local Hyperthermia. Open J Biophys 2016;6:10–18. [Google Scholar]
- 65. Szasz O, Szasz A. Oncothermia – Nano-Heating Paradigm. J Cancer Sci Ther 2014;6:117–21. [Google Scholar]
- 66. Szasz O, Szigeti G, Szasz A. Connections between the Specific Absorption Rate and the Local Temperature. Open J Biophys 2016;6:53–74. [Google Scholar]
- 67. Andocs G, Renner H, Balogh L, et al. Strong synergy of heat and modulated electromagnetic field in tumor cell killing. Strahlentherapie und Onkologi.e 2009;185:120–6. [DOI] [PubMed] [Google Scholar]
- 68. Andocs G, Galfi P, Renner H, et al. Thermally induced but temperature independent cell-destruction by modulated electrohyperthermia in nude-mice xenograft model In: Abstract book: STM 2009 Annual Meeting: “Expanding the Frontiers of Thermal Biology, Medicine and Physics”. 2009:49 [#OS11]. [Google Scholar]
- 69. FDA approval P100034 – NovoTTF-100A System. http://www.accessdata.fda.gov/cdrh_docs/pdf10/p100034a.pdf (accessed 25 Feb 2017).
- 70. Fonkem E, Wong ET. NovoTTF-100A: a new treatment modality for recurrent glioblastoma. Expert Rev Neurother 2012;12:895–9. [DOI] [PubMed] [Google Scholar]
- 71. Kirson ED, Dbaly V, Tovarys F, et al. Alternating electric fields arrest cell proliferation in animal tumor models and human brain tumors. Proc Natl Acad Sci U S A 2007;104:10152–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Vodovnik L, Miklavcic D, Sersa G. Modified cell proliferation due to electrical currents. Med Biol Eng Comput 1992;30:CE21–8. [DOI] [PubMed] [Google Scholar]
- 73. Stupp R, Wong ET, Kanner AA, et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer 2012;48:2192–202. [DOI] [PubMed] [Google Scholar]
- 74. FDA approval P100034 – NovoTTF-100A System. http://www.accessdata.fda.gov/cdrh_docs/pdf10/p100034a.pdf (accessed 25 Feb 2017).
- 75. Kanner AA, Wong ET, Villano JL, et al. EF-11 investigators. Post Hoc analyses of intention-to-treat population in phase III comparison of NovoTTF-100A™ system versus best physician’s choice chemotherapy. Semin Oncol 20014;41(Suppl. 6):S25–34. [DOI] [PubMed] [Google Scholar]
- 76. Stupp R, Wong ET, Kanner AA, et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer 2012;48:2192–202. [DOI] [PubMed] [Google Scholar]
- 77. Nagy G, Meggyeshazi N, Szasz O. Deep Temperature Measurements in Oncothermia Processes; Conf. Papers in Med 2013:ID 6852646. [Google Scholar]
- 78. Balogh L, Polyák A, Pöstényi Z, et al. Temperature increase induced by modulated electrohyperthermia (oncothermia®) in the anesthetized pig liver. J Cancer Res Ther 2016;12:1153–9. [DOI] [PubMed] [Google Scholar]
- 79. Herzog A. [Measurement of the temperature distribution on the model of non-perfused pig liver in local hyperthermia with short waves at 13.56 MHz] Messung der Temperaturverteilung am Modell der nicht perfundierten Schweineleber bei lokaler Hyperthermie mit Kurzwellen mit 13,56 MHz. Forum Hyperthermie. [Internet]. 2008;14:30 http://www.forum-medizin.de/download/977/ [Google Scholar]
- 80. Balogh L, Cs K, Gyongy M. Tumor-temperature by oncothermia in real-animal. Report presented at ICHS 2015 (33rd Annual Conference of ICHs). Nidda (Germany), 2015. [Google Scholar]
- 81. Schwan HP, Piersol GM. The absorption of electromagnetic energy in body tissues; a review and critical analysis. Am J Phys Med 1955;34:425–48. [PubMed] [Google Scholar]
- 82. Giuliani L, Soffritti M. Non-Thermal Effects and Mechanisns of Interaction Between Electromagnetic Fields and Living Matter. ICEMS Monograph: National Institute for the Study and Control of Cancer and Environmental Diseases “Bernardino Ramazzini”, Bologna, Italy. Eur J Oncol Library 2010;5:200. [Google Scholar]
- 83. Gy V, Szasz A, Szasz N. On the thermal noise limit of cellular membranes. Bioelectromagnetics 2005;26:28–35. [DOI] [PubMed] [Google Scholar]
- 84. Szasz A, Szasz O, Szasz N. Radiofrequency hyperthermia device with target feedback signal modulation. Oncotherm Kft 2010. Pat. No.: WO2010043372 A1. [Google Scholar]
- 85. Szendro P, Vincze G, Szasz A. Pink-noise behaviour of bio-systems. Eur Biophys J 2001;30:227–31. [DOI] [PubMed] [Google Scholar]
- 86. Lovelady DC, Richmond TC, Maggi AN, et al. Distinguishing cancerous from noncancerous cells through analysis of electrical noise. Phys Rev E Stat Nonlin Soft Matter Phys 2007;76:041908. [DOI] [PubMed] [Google Scholar]
- 87. Kiss E. The role of modulation in modulated electro-hyperthermia (mEHT) in colorectal allograft tumor model. XXXIII Conference of the International Clinical Hyperhtermia Society (ICHS) & 3rd. Int Oncothermia Symp 2015:10-12 Nidda, Germany. [Google Scholar]
- 88. Zimmerman JW, Pennison MJ, Brezovich I, et al. Cancer cell proliferation is inhibited by specific modulation frequencies. Br J Cancer 2012;106:307–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Astumian RD, Adair RK, Weaver JC. Stochastic resonance at the single-cell level. Nature 1997;388:632–3. [DOI] [PubMed] [Google Scholar]
- 90. Brunner G. [Electro hyperthermia of skin cancer cells: Recent findings on potential molecular mechanisms of action]. Hyperthermie Symposium 2007. Cologne. [Google Scholar]
- 91. Calabresi P, Pisani A, Mercuri NB, et al. On the mechanisms underlying hypoxia-induced membrane depolarization in striatal neurons. Brain 1995;118:1027–38. [DOI] [PubMed] [Google Scholar]
- 92. Calabresi P, Marfia GA, Centonze D, et al. Sodium Influx Plays a Major Role in the Membrane Depolarization Induced by Oxygen and Glucose Deprivation in Rat Striatal Spiny Neurons. Stroke 1999;30:171–9. [DOI] [PubMed] [Google Scholar]
- 93. Calabresi P, Marfia GA, Amoroso S, et al. Pharmacological Inhibition of the Na+/Ca2+ Exchanger Enhances Depolarizations Induced by Oxygen/Glucose Deprivation but Not Responses to Excitatory Amino Acids in Rat Striatal Neurons. Stroke 1999;30:1687–94. [DOI] [PubMed] [Google Scholar]
- 94. Ardenne Mv. [Theoretical and Experimental Basis of Cancer Multistep Therapy] Theoretische u. experimentelle Grundlagen der Krebs-Mehrschritt-Therapie. 2nd ed Berlin: Volk u. Gesundheit, 1970–71:963. [Google Scholar]
- 95. Song CW, Kang MS, Rhee JG, et al. Vascular damage and delayed cell death in tumours after hyperthermia. Br J Cancer 1980;41:309–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Dewhirst MW, Vujaskovic Z, Jones E, et al. Re-setting the biologic rationale for thermal therapy. Int J Hyperthermia 2005;21:779–90. [DOI] [PubMed] [Google Scholar]
- 97. Caubet R, Pedarros-Caubet F, Chu M, et al. A radio frequency electric current enhances antibiotic efficacy against bacterial biofilms. Antimicrob Agents Chemother 2004;48:4662–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Giladi M, Porat Y, Blatt A, et al. Microbial growth inhibition by alternating electric fields. Antimicrob Agents Chemother 2008;52:3517–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Asami K, Takahashi Y, Takashima S. Dielectric properties of mouse lymphocytes and erythrocytes. Biochim Biophys Acta 1989;1010:49–55. [DOI] [PubMed] [Google Scholar]
- 100. Curley SA, Palalon F, Lu X, et al. Noninvasive radiofrequency treatment effect on mitochondria in pancreatic cancer cells. Cancer 2014;120:3418–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Ponne CT, Balk M, Hancioglu Ömre, et al. Effect of radio frequency energy on biological membranes and microorganisms : Ponne CT, Interaction of electromagnetic energy with vegetabls food constituents. [Internet]. Eindhoven: Eindhoven University of Technology, Doctoral dissertation, 1996;29:41–8. http://alexandria.tue.nl/extra3/proefschrift/PRF11B/9504917.pdf 10.1006/fstl.1996.0006 [DOI] [Google Scholar]
- 102. Kotnik T, Miklavcic D. Second-order model of membrane electric field induced by alternating external electric fields. IEEE Trans Biomed Eng 2000;47:1074–81. 10.1109/10.855935 [DOI] [PubMed] [Google Scholar]
- 103. Wolf M, Gulich R, Lunkenheimer P, et al. Broadband dielectric spectroscopy on human blood. Biochim Biophys Acta 2011;1810:727–40. 10.1016/j.bbagen.2011.05.012 [DOI] [PubMed] [Google Scholar]
- 104. Kotnik T, Miklavčič D. Theoretical evaluation of the distributed power dissipation in Biological cells exposed to electric fields. Bioelectromagnetics 2000;21:385–94. [DOI] [PubMed] [Google Scholar]
- 105. Wolf M, Gulich R, Lunkenheimer P, et al. Relaxation dynamics of a protein solution investigated by dielectric spectroscopy. Biochim Biophys Acta 1824;2011:723–30. [DOI] [PubMed] [Google Scholar]
- 106. Zimmerman JW, Jimenez H, Pennison MJ, et al. Targeted treatment of cancer with radiofrequency electromagnetic fields amplitude-modulated at tumor-specific frequencies. Chin J Cancer 2013;32:573–81. 10.5732/cjc.013.10177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Vincze G, Szasz A, Szigeti GP. Reorganization of the cytoskeleton. J Adv Biol 2016;9:1872–82. [Google Scholar]
- 108. Taghi M, Gholamhosein R, Saeed R-Z. Effect of radio frequency waves of electromagnetic field on the tubulin. Recent Pat Endocr Metab Immune Drug Discov 2013;7:252–6. 10.2174/18722148113079990007 [DOI] [PubMed] [Google Scholar]
- 109. Markin VS, Tsong TY. Frequency and concentration windows for the electric activation of a membrane active transport system. Biophys J 1991;59:1308–16. 10.1016/S0006-3495(91)82345-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Andocs G, Meggyeshazi N, Balogh L, et al. Upregulation of heat shock proteins and the promotion of damage-associated molecular pattern signals in a colorectal cancer model by modulated electrohyperthermia. Cell Stress Chaperones 2015;20:37–46. 10.1007/s12192-014-0523-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Meggyeshazi N, Andocs G, Balogh L, et al. DNA fragmentation and caspase-independent programmed cell death by modulated electrohyperthermia. Strahlentherapie und Onkologie 2014;190:815–22. 10.1007/s00066-014-0617-1 [DOI] [PubMed] [Google Scholar]
- 112. Andocs G, Balogh L, Meggyeshazi N, et al. Apoptosis induction with modulated radiofrequency (RF) hyperthermia (oncothermia) in immuno-defficient mice xenograft tumors (Review). 1st Int Oncothermia Symp, Cologne 22-23, 2010. Oncothermia J 2010;1:32–3. [Google Scholar]
- 113. Cha J, Jeon TW, Lee CG, et al. Electro-hyperthermia inhibits glioma tumorigenicity through the induction of E2F1-mediated apoptosis. Int J Hyperthermia 2015;31:784–92. 10.3109/02656736.2015.1069411 [DOI] [PubMed] [Google Scholar]
- 114. Fiorentini GM, Yoon SM, Okamoto Y, et al. Abscopal effect: new perspectives in Oncothermia. XXXI Conference of the International Clinical Hyperhtermia Society (ICHS) & 2nd Int Oncothermia Symp, 12-14 Oct 2012 Budapest, Hungary. Oncothermia J 2013:278–81. [Google Scholar]
- 115. Yoon SM, Lee JS. Case of Abscopal effect with Metastatic Non-Small-Cell Lung Cancer. Oncothermia J 2012;5:53–7. [Google Scholar]
- 116. Andosc G, Kovago C, Meggyeshazi N, et al. Oncothermia treatment induced immunogenic cancer cell death — New possibilities for therapeutic cancer vaccine. 6th Asian Congress of Hyperthermic Oncology (ACHO) & 31st Japanese Congress of Thermal Medicine (JCTM), 5-6 Sep 2014, Fukui City, Japan. Therm Med 2014;30:110 (GSE17). [Google Scholar]
- 117. Akutsu Y, Tamura Y, Murakami K, et al. Can modulated electro-hyperthermia (mEHT) elicit immune reaction? – From basic and clinical research. 6th Asian Congress of Hyperthermic Oncology (ACHO) & 31st Japanese Congress of Thermal Medicine (JCTM), 5-6 Sep 2014, Fukui City, Japan. Therm Med 2014;30:62(WS1-1-3). [Google Scholar]
- 118. Jeon TW, Yang H, Lee CG, et al. Electro-hyperthermia up-regulates tumour suppressor Septin 4 to induce apoptotic cell death in hepatocellular carcinoma. Int J Hyperthermia 2016;7:1–9. [DOI] [PubMed] [Google Scholar]
- 119. Tsang Y-W, Huang C-C, Yang K-L, et al. Improving immunological tumor microenvironment using electro-hyperthermia followed by dendritic cell immunotherapy. BMC Cancer 2015;15:708 10.1186/s12885-015-1690-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. QIN WEI, Akutsu Y, Andocs G, et al. Modulated electro-hyperthermia enhances dendritic cell therapy through an abscopal effect in mice. Oncol Rep 2014;32:2373–9. 10.3892/or.2014.3500 [DOI] [PubMed] [Google Scholar]
- 121. Kim K, Pang KM, Evans M, et al. Overexpression of β-Catenin Induces Apoptosis Independent of Its Transactivation Function with LEF-1 or the Involvement of Major G1 Cell Cycle Regulators. Mol Biol Cell 2000;11:3509–23. 10.1091/mbc.11.10.3509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Krutovskikh VA, Piccoli C, Yamasaki H, et al. Gap junction intercellular communication propagates cell death in cancerous cells. Oncogene 2002;21:1989–99. 10.1038/sj.onc.1205187 [DOI] [PubMed] [Google Scholar]
- 123. Macdonald DR, Cascino TL, Schold SC, et al. Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 1990;8:1277–80. 10.1200/JCO.1990.8.7.1277 [DOI] [PubMed] [Google Scholar]
- 124. Conover WJ. Practical nonparametric statistics. 3rd ed New York: Wiley, 1980:592 ISBN 978-0-471-16068-7. [Google Scholar]
- 125. Altman DG. Practical statistics for medical research. London: Chapman and Hall, 1991:611. [Google Scholar]
- 126. Newcombe RG, Altman DG. Proportions and their differences In: Altman DG, Machin D, Bryant TN, eds Statistics with confidence. 2nd ed, 2000. BMJ Books. [Google Scholar]
- 127. Goodman LA. On the exact variance of products. J Am Stat Assoc 1960;55:708–13. 10.1080/01621459.1960.10483369 [DOI] [Google Scholar]
- 128. Fieller EC. Some problems in interval estimation. J Royal Stat Soc, Series B 1954;16:175–85. JSTOR 2984043. [Google Scholar]
- 129. PubH 7470. Statistics for translational & clinical research: Use of Fieller’s theorem for the estimation of ratios. http://www.biostat.umn.edu/~chap/S08-EstimateRatios.pdf (accessed 2 Feb 2017).
- 130. Casella G, Berger RL. Statistical inference. 2nd ed Pacific Grove, CA, USA: Duxbury, Thomson Learning Inc., 2002:240–5. ISBN 0-534-24312-6. [Google Scholar]
- 131. Newcombe RG. MOVER-R confidence intervals for ratios and products of two independently estimated quantities. Stat Methods Med Res 2016;25:1774–8. 10.1177/0962280213502144 [DOI] [PubMed] [Google Scholar]
- 132. Borenstein M, Hedges LV, Higgins JPT, et al. Introduction to meta-analysis, John Wiley & Sons, Ltd, 2009:413 ISBN: 978-0-470-05724-7. [Google Scholar]
- 133. Welch BL. The generalisation of student’s problems when several different population variances are involved. Biometrika 1947;34:28–35. [DOI] [PubMed] [Google Scholar]
- 134. Campbell I. Chi-squared and Fisher-Irwin tests of two-by-two tables with small sample recommendations. Stat Med 2007;26:3661–75. 10.1002/sim.2832 [DOI] [PubMed] [Google Scholar]
- 135. Altman DG, Bland JM. How to obtain the P value from a confidence interval. BMJ 2011;343:d2304 10.1136/bmj.d2304 [DOI] [PubMed] [Google Scholar]
- 136. Lui KJ. Interval estimation of risk ratio in the simple compliance randomized trial. Contemp Clin Trials 2007;28:120–9. 10.1016/j.cct.2006.05.005 [DOI] [PubMed] [Google Scholar]
- 137. Hosmer DW, Lemeshow S. Applied Survival Analysis: Regression Modeling of Time to Event Data. NewYork: John Wiley and Sons, 1999. [Google Scholar]
- 138. Greenwood M. The Natural Duration of Cancer. Reports of Public Health and Related Subjects. London: HMSO, 1926;33. [Google Scholar]
- 139. Crawley MJ. Statistics: an Introduction using R. 2nd ed Chichester: John Wiley & Sons, 2015:339 ISBN 978-1-118-94109-6 http://www.imperial.ac.uk/bio/research/crawley/statistics/ [Google Scholar]
- 140. Cox DR, Oakes D. Analysis of Survival Data. London – New York: Chapman and Hall, 1984:201. [Google Scholar]
- 141. Deeks JJ, Higgins JPT. Statistical algorithms in Review Manager 5. Cochrane Collaboration. 2010. http://ims.cochrane.org/revman/documentation/Statistical-methods-in-RevMan-5.pdf (accessed 22 Aug 2016).
- 142. Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ 2003;327:557–60. 10.1136/bmj.327.7414.557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Roussakow S. Effect-to-treatment analysis: a clinically useful instrument for study and comparison of a treatment effect. BMJ 2017. [Google Scholar]
- 144. Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 2005;5:13 10.1186/1471-2288-5-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Pradelli L, Wertheimer A, Pharmacoeconomics: Principles and Practice. SEEd, 2012:125 http://public.eblib.com/choice/publicfullrecord.aspx?p=3034683 [Google Scholar]
- 146. Rascati KL. Essentials of pharmacoeconomics. 2nd ed Baltimore, MD: Lippincott Williams & Wilkins, 2014:295. [Google Scholar]
- 147. Arnold RJG, Pharmacoeconomics: from theory to practice. Drug discovery series. Boca Raton: CRC Press/Taylor & Francis, 2010;13:243. [Google Scholar]
- 148. Bootman JL, Townsend RJ, McGhan WF, Principles of pharmacoeconomics. 3rd ed Cincinnati, OH: Harvey Whitney Books Co, 2005:409. [Google Scholar]
- 149. Walley T, Haycox A, Boland A, Pharmacoeconomics. Edinburgh; New York: Churchill Livingstone, 2004:203. [Google Scholar]
- 150. von Elm E, Altman DG, Egger M, et al. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 2007;335:806–8. 10.1136/bmj.39335.541782.AD [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Husereau D, Drummond M, Petrou S, et al. ISPOR Health Economic Evaluation Publication Guidelines-CHEERS Good Reporting Practices Task Force. Consolidated Health Economic Evaluation Reporting Standards (CHEERS) – explanation and elaboration: a report of the ISPOR Health Economic Evaluation Publication Guidelines Good Reporting Practices Task Force. Value Health 2013;16:231–50. [DOI] [PubMed] [Google Scholar]
- 152. Wei W, Chen X, Ma X, et al. The efficacy and safety of various dose-dense regimens of temozolomide for recurrent high-grade glioma: a systematic review with meta-analysis. J Neurooncol 2015;125:339–49. 10.1007/s11060-015-1920-0 [DOI] [PubMed] [Google Scholar]
- 153. Brandes AA, Tosoni A, Cavallo G, et al. Temozolomide 3 weeks on and 1 week off as first-line therapy for recurrent glioblastoma: phase II study from gruppo italiano cooperativo di neuro-oncologia (GICNO). Br J Cancer 2006;95:1155–60. 10.1038/sj.bjc.6603376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Norden AD, Lesser GJ, Drappatz J, et al. Phase 2 study of dose-intense temozolomide in recurrent glioblastoma. Neuro Oncol 2013;15:930–5. 10.1093/neuonc/not040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Strik HM, Buhk JH, Wrede A, et al. Rechallenge with temozolomide with different scheduling is effective in recurrent malignant gliomas. Mol Med Rep 2008;1:863–7. 10.3892/mmr_00000042 [DOI] [PubMed] [Google Scholar]
- 156. Abacioglu U, Caglar HB, Yumuk PF, et al. Efficacy of protracted dose-dense temozolomide in patients with recurrent high-grade glioma. J Neurooncol 2011;103:585–93. 10.1007/s11060-010-0423-2 [DOI] [PubMed] [Google Scholar]
- 157. Berrocal A, Perez Segura P, Gil M, et al. Extended-schedule dose-dense temozolomide in refractory gliomas. J Neurooncol 2010;96:417–22. 10.1007/s11060-009-9980-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. GoodPx Web-site. https://www.goodrx.com/temozolomide? = &form = capsule&dosage = 140mg&quantity = 15&days_supply = &label_override = temozolomide (accessed 21 Jan 2017).
- 159. Medizinfuchs Web-site. https://www.medizinfuchs.de/wirkstoff/temozolomid-2582.html (accessed 21 Jan 2017).
- 160. Happold C, Roth P, Wick W, et al. Distinct molecular mechanisms of acquired resistance to temozolomide in glioblastoma cells. J Neurochem 2012;122:444–55. 10.1111/j.1471-4159.2012.07781.x [DOI] [PubMed] [Google Scholar]
- 161. Pan Q, Yang XJ, Wang HM, et al. Chemoresistance to temozolomide in human glioma cell line U251 is associated with increased activity of O6-methylguanine-DNA methyltransferase and can be overcome by metronomic temozolomide regimen. Cell Biochem Biophys 2012;62:185–91. 10.1007/s12013-011-9280-7 [DOI] [PubMed] [Google Scholar]
- 162. Lee SY. Temozolomide resistance in glioblastoma multiforme. Genes Dis 2016;3:198–210. 10.1016/j.gendis.2016.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163. Turner SG, Gergel T, Wu H, et al. The effect of field strength on glioblastoma multiforme response in patients treated with the NovoTTF™-100A system. World J Surg Oncol 2014;12:162 10.1186/1477-7819-12-162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Appleby J, Devlin N, Parkin D. NICE’s cost effectiveness threshold. BMJ 2007;335:358–9. https://doi.org/ 10.1136/bmj.39308.560069.BE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Claxton K, Martin S, Soares M, et al. Methods for the Estimation of the NICE Cost Effectiveness Threshold: Final Report: Centre for Health Economics, The University of York, 2013:436. [Google Scholar]
- 166. WHO Commission on Macroeconomics and Health. Macroeconomics and health: investing in health for economic development. Report of the Commission on Macroeconomics and Health. Geneva: World Health Organization, 2001. [Google Scholar]
- 167. Collins M, Latimer N. NICE’s end of life decision making scheme: impact on population health. BMJ 2013;346:f1363 https://doi.org/ 10.1136/bmj.f1363 [DOI] [PubMed] [Google Scholar]
- 168. Barham L. Three NICE thresholds for cost-effectiveness: does that make sense? Pharmaphorum 2016;25 http://pharmaphorum.com/views-and-analysis/three-nice-thresholds-for-cost-effectiveness-does-that-make-sense/ [Google Scholar]
- 169. Brada M, Stenning S, Gabe R, et al. Temozolomide versus procarbazine, lomustine, and vincristine in recurrent high-grade glioma. J Clin Oncol 2010;28:4601–8. 10.1200/JCO.2009.27.1932 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2017-017387supp001.pdf (55.7KB, pdf)
bmjopen-2017-017387supp002.pdf (35.6KB, pdf)
bmjopen-2017-017387supp003.pdf (36.6KB, pdf)
bmjopen-2017-017387supp004.doc (88KB, doc)