Abstract
During deep space missions, astronauts will be exposed to low doses of charged particle irradiation. The long-term health effects of these exposures are largely unknown. We previously showed that low doses of oxygen ion (16O) irradiation induced acute damage to the hematopoietic system, including hematopoietic progenitor and stem cells in a mouse model. However, the chronic effects of low dose 16O irradiation remain undefined. In the current study, we investigated the long-term effects of low dose 16O irradiation on the mouse hematopoietic system. Male C57BL/6J mice were exposed to 0.05 Gy, 0.1 Gy, 0.25 Gy and 1.0 Gy whole body 16O (600 MeV/n) irradiation. The effects of 16O irradiation on bone marrow (BM) hematopoietic progenitor cells (HPCs) and hematopoietic stem cells (HSCs) were examined three months after the exposure. The results showed that the frequencies and numbers of BM HPCs and HSCs were significantly reduced in 0.1 Gy, 0.25 Gy and 1.0 Gy irradiated mice compared to 0.05 Gy irradiated and non-irradiated mice. Exposure of mice to low dose 16O irradiation also significantly reduced the clongenic function of BM HPCs determined by the colony-forming unit assay. The functional defect of irradiated HSCs was detected by cobblestone area-forming cell assay after exposure of mice to 0.1 Gy, 0.25 Gy and 1.0 Gy of 16O irradiation, while it was not seen at three months after 0.5 Gy and 1.0 Gy of γ-ray irradiation. These adverse effects of 16O irradiation on HSCs coincided with an increased intracellular production of reactive oxygen species (ROS). However, there were comparable levels of cellular apoptosis and DNA damage between irradiated and non-irradiated HPCs and HSCs. These data suggest that exposure to low doses of 16O irradiation induces long-term hematopoietic injury, primarily via increased ROS production in HSCs.
Introduction
Deep space missions are associated with several risk factors, including the exposure to space radiation. Two main sources of space radiation are solar particle events (SPE) and galactic cosmic rays (GCR)[1]. Specifically, high atomic number and energy (HZE) particles from GCR, such as 56Fe, 28Si, 16O, and 12C, have higher energy and stronger toxicities to normal tissues than photon and proton radiation[2, 3]. Although exposure to space radiation usually occurs at low doses and dose rates, an extended exposure of the body to space radiation, particularly to HZE radiation, during long-term space missions might result in a dose accumulation of HZE radiation sufficient to cause health hazards[4, 5]. Fornace’s group reported that the value of relative biological effectiveness (RBE) in relation to γ-ray irradiation was 1.25 for 56Fe, 1.4 for 28Si and 0.99 for 12C using a mouse model[2, 6]. However, there is limited knowledge of the RBE of 16O ion exposure, especially regarding the long-term health effects, which limits the development of mitigating strategies against space radiation.
A plethora of studies including ours have reported that photon radiation (such as γ-rays) induced both acute and chronic bone marrow (BM) suppression, especially the long-term defect of hematopoietic stem cells (HSCs), resulting from radiation-induced production of reactive oxygen species (ROS), DNA damage, apoptosis, and cellular senescence[7–9]. Previous studies have demonstrated that exposure of mice to high energy 56Fe, 12C and neutron had detrimental effects on the hematopoietic system, including decreased peripheral blood counts and reduced colony forming ability of HSCs and hematopoietic progenitor cells (HPCs) [2, 10–12]. However, the hematopoietic effects of high energy 16O irradiation have been barely studied. In addition, several labs have demonstrated that exposure to protons at low doses significantly decreased the numbers of peripheral blood cells including white blood cells and platelets starting at 4 hours post exposure in porcine and mouse models[13–18]. The abnormal levels of white blood cells in the spleen still existed 110 days after proton exposure[19]. Consistent with these results, we recently reported that low doses of proton total body irradiation (TBI) resulted in long-term detrimental effects on the hematopoietic system[20]. However, we have recently shown that low doses of high HZE TBI in the form of 16O ions (16O TBI) also caused acute hematopoietic damage, when measured two weeks after TBI in C57BL/6J mice[21]. 16O TBI was not only associated with a decrease in peripheral blood cell counts, but also with a functional deficiency in HPCs and HSCs. The acute negative effects on HPC and HSC function coincided with induction of ROS production, DNA damage and apoptosis[21]. However, long-term effects of 16O TBI on the hematopoietic system remain unknown and therefore were investigated in the present study.
Materials and methods
Animals and irradiation
Male C57BL/6J mice were purchased from the Jackson Laboratory (Bar Harbor, ME) and housed at the University of Arkansas for Medical Sciences (UAMS) until 6 months of age. Mice were administered a standard soy-protein free rodent diet (2020X, Harlan Teklad, Indianapolis, IN) and water ad libitum and housed under a constant 12 h light: dark cycle throughout the study. Six-month old mice were shipped to Brookhaven National Laboratories (BNL) in Upton, NY. After a one-week acclimation period, the mice were either sham irradiated or received whole-body 16O irradiation (600 MeV/n; 0.05 Gy, 0.1 Gy, 0.25 Gy and 1.0 Gy, 0.25–0.26 Gy/min, n = 5) at the NASA Space Radiation Laboratory (NSRL). For each exposure, animals were individually placed into clear Lucite cubes (3 in x 1½ in x 1½ in) with breathing holes. Sham irradiated mice were placed into the same enclosures for the same amount of time, but were not exposed to radiation. One day after (sham-) irradiation, the mice were returned to UAMS. Mice were administered 2020X chow containing 150 ppm fenbendazole for the first 8 weeks after return, as a routine UAMS quarantine procedure. During the entire experiment, sham-irradiated mice were not housed together with irradiated mice. All procedures in this study were approved by the Institutional Animal Care and Use Committees of UAMS and BNL.
Dosimetry was performed by the NSRL physics group to ensure the quality of exposure. Briefly, the NSRL beamline recorded the charge delivered to a transmission ion chamber placed just in front of the animals. The transmission ion chamber was calibrated against a National Institute of Standards and Technology (NIST)-traceable 1.0 cm3 thimble ion chamber (EG&G, Inc.) which was placed at the target position. The doses indicated in the present study were thus the absorbed doses by the animals in the ion chamber at the target surface location on the beamline. The BM of mice is < = 1.0 mm beneath the skin of the animals so the total material between the skin surface and the BM is <2 mm. With particles of multi-centimeter ranges, there is no significant difference in dose as reported at the surface compared to ~1.5 mm depth for the BM. Therefore, the doses of 0.05 Gy, 0.1 Gy, 0.25 Gy and 1.0 Gy 16O TBI are the actual doses to the BM in our current study. In addition, we ran a GERM (GCR Event-based Risk Model) code simulation to determine that the LET (Linear Energy Transfer) of 600 MeV/n oxygen ions is 16.45 keV/micron. We applied the following formula to calculate fluence: Dose (cGy) = 1.602x10-7 x LET (keV/um) X fluence. The fluence at 0.1 Gy was 3.8E+06 particles/cm2. The diameter of hematopoietic stem and progenitor cells is 8.0 microns[22]. Assuming circular cells of 8.0 microns in diameter, a dose of 0.1 Gy oxygen ions (600 MeV/n) will result in an average of 1.91 ion traversals per cell. The average numbers of oxygen ion traversing each cell are 0.96 ion/cell for 0.05 Gy, 1.91 ions/cell for 0.1 Gy, 4.78 ions/cell for 0.25 Gy and 19.1 ions/cell for 1.0 Gy, respectively (Table 1). Based on the poisson distribution of particle traversal, 62% of cells will be traversed by one or more ions and 38% will not be traversed at a dose of 0.05 Gy. However, 99% of cells will be traversed at a dose of 0.25 Gy (Table 1).
Table 1. Properties of oxygen ion irradiation.
| Doses of 16O | Average hits/cell | % of traversed/non-traversed cells |
|---|---|---|
| 0.05 Gy | 0.96 | 62/38 |
| 0.1 Gy | 1.91 | 85/15 |
| 0.25 Gy | 4.78 | 99/1 |
| 1.0 Gy | 19.1 | 100/0 |
Total body γ-ray irradiation (γ-TBI) was performed at UAMS. Six-month old mice were put in a plastic box on a rotating platform and exposed to 0.5 Gy and 1.0 Gy of γ-TBI (n = 5) in a Mark I 137Ce γ-irradiator (JL Shepherd, Glendale, CA, USA) at a rate of 1.14 Gy/min.
Tissue and peripheral blood collection
Three months after irradiation, mice were anesthetized and sacrificed with continued exposure to 5% isoflurane for at least 5 min after respiratory arrest. A modified infusion set (27G with shortened tubing) was used to inject a single dose of heparin (30–40 U/kg) into the abdominal vena cava. Without changing the position of the infusion set, a blood sample was drawn, transferred into an EDTA coated tube, and stored on ice until further analysis. Peripheral blood cell counts were determined by a Vet ABC™ Hematological analyzer (SCIL Animal Care Co.). The femora and tibia were collected and immediately processed for cell isolation and analysis as described below.
Isolation of BM mononuclear cells (BM-MNCs), analysis of the frequencies and numbers of different hematopoietic cell populations by flow cytometry
The femora and tibia were immediately flushed with Hanks' Balanced Salt Solution (HBSS) containing 2% fetal bovine serum (FBS) using a 21-gauge needle and syringe to collect BM cells. BM samples were placed on ice and analyzed within the same day as described below.
For phenotypic analysis, BM cells were incubated with biotin-conjugated anti-CD3e, anti-CD45R/B220, anti-Gr-1, anti-CD11b, and anti-Ter-119 antibodies and with anti-CD16/32 (Fcγ II/III Receptor or FcγR) antibody to block the Fcγ receptors. Cells were then labeled with streptavidin-FITC, anti-Sca-1-PE-Cy7, anti-c-Kit-APC-Cy7, anti-CD150-APC and anti-CD48-Pacific blue for HPCs (Lin-Sca1-c-kit+ cells), LSK cells (Lin-Sca1+c-kit+ cells), and HSCs (Lin-Sca1+c-kit+CD150+CD48- cells). Dead cells were excluded by gating out the cells stained positive with propidium iodide (PI). The frequencies of HPCs and HSCs were analyzed with an Aria II cell sorter and presented as percentages of total living BM cells. The numbers of different hematopoietic cell populations in each mouse were calculated by multiplying the total numbers of BM cells harvested from the two hind legs of each mouse with the frequencies of each population in the cells. For each sample, approximately 8 x 105 to 1 x 106 BM cells were acquired and the data were analyzed using BD FACSDiva 6.0 (BD Biosciences) and FlowJo (FlowJo, Ashland, OR) software.
For the isolation of Lineage negative cells (Lin- cells), BM-MNCs were isolated by Histopaque 1083 separation solution (Sigma, St. Louis, MO). BM-MNCs were incubated with purified rat antibodies specific for murine CD3e, Mac-1, CD45R/B220, Ter-119, and Gr-1. The labeled mature lymphoid and myeloid cells were depleted by incubating with goat anti-rat IgG paramagnetic beads (Life Technologies, Grand Island, NY) at a bead:cell ratio of approximately 4:1. Cells binding the paramagnetic beads were removed with a magnetic field. Lin- cells were washed twice with 2% FBS/HBSS and resuspended in complete medium (RPMI1640 medium supplemented with 10% FBS, 2 mM L-glutamine, 10 μM HEPES buffer, and 100 U/mL penicillin and streptomycin) at 1x107 cells/mL. Lin- cells were subsequently used to analyze cellular apoptosis, ROS production, DNA damage and cell cycle in HPCs, LSK cells and HSCs as shown below. All flow antibodies were purchased from eBioscience (San Jose, CA).
Colony-forming unit (CFU) assay
The CFU assay was performed by culturing BM-MNCs in MethoCult GFM3434 methylcellulose medium (Stem Cell Technologies, Vancouver, BC). 2×104 BM-MNCs were seeded into each well of 12-well plates. The numbers of CFU-granulocyte macrophage (CFU-GM) and burst-forming unit-erythroid (BFU-E) were scored under microscopy on day 7 and those of CFU-granulocyte, -erythrocyte, -monocyte, and -megakaryocyte (GEMM) were counted on day 12 after cells were seeded.
Cobblestone area-forming cell (CAFC) assay
1×103 FBMD-1 stromal cells were seeded in each well of flat-bottomed 96-well plates (Falcon, Lincoln Park, NJ). One week later, BM-MNCs from irradiated and non-irradiated mice were suspended in CAFC medium (Iscove’s MDM supplemented with 20% horse serum, 10−5 M hydrocortisone, 10−5 M 2-mercaptoethanol, 100 units/ml penicillin, and 100 μg/ml streptomycin). Cells were then overlaid on the stromal layers in six dilutions and 3-fold apart. Twenty wells were plated for each dilution to allow limiting dilution analysis of the precursor cells forming hematopoietic clones under the stromal layer. Cells were fed weekly by changing one-half of the medium. Wells were scored positive if at least one phase-dark hematopoietic clone (containing 5 or more cells) was seen. The frequencies of CAFC were determined at week 5 to determine the clonogenic function of HSCs. The frequency of CAFC was then calculated by using Poisson statistics.
Apoptosis assay
Lin- cells were incubated with anti-CD16/32 at 4°C for 15 min to block the Fc-γ receptors and then stained with antibodies against various cell surface markers in the dark. After Annexin V staining with a kit from BD Pharmingen (San Diego, CA) according to the manufacturer’s instructions, apoptotic cells in different hematopoietic cell populations were analyzed with an Aria II cell sorter.
Analysis of the levels of intracellular ROS
After staining with the appropriate cell surface marker antibodies, Lin- cells (1 x 107/mL) were suspended in PBS supplemented with 5 mM glucose, 1 mM CaCl2, 0.5 mM MgSO4, and 5 mg/ml BSA and then incubated with 10 μM 2',7'-dichlorofluorescein diacetate (DCFDA) (Life Technologies, Grand Island, NY) for 30 minutes at 37°C. The levels of ROS in HPCs and HSCs were analyzed by measuring the mean fluorescence intensity (MFI) of 2',7'-dichlorofluorescein (DCF) with an Aria II cell sorter. For each sample, a minimum of 200,000 lineage negative cells was acquired and the data were analyzed as previously described [23].
DNA damage analysis
Lin- cells were first stained with antibodies against various cell-surface markers and fixed and permeabilized using the Fixation/Permeabilization Solution from BD Biosciences (San Diego, CA) followed by 0.2% Triton-X-100 incubation for 10 min. Cells were then stained with Alexa Fluor 488 conjugated anti-phospho-Histone 2AX (Ser139) (γH2AX, Cell Signaling Technology, MA) antibody for 1.5 hours at 4°C and analyzed by flow cytometry. The levels of DNA double strand break damage were expressed by the mean fluorescence intensity (MFI) of γH2AX with an Aria II cell sorter.
Cell cycle analysis
Lin- cells were first stained with antibodies against various cell-surface markers and fixed and permeabilized using the Fixation/Permeabilization Solution (BD Biosciences, CA). Subsequently, they were stained with anti-Ki67-FITC antibody (BD Biosciences, CA) and 7-aminoactinomycin (7-AAD, Sigma, St. Louis, MO) and then analyzed by flow cytometry.
Quantitative reverse transcription polymerase chain reaction (qRT-PCR)
HPCs and HSCs for 1.0 Gy 16O TBI and non-irradiated mice were sorted out. Total RNA of HPCs and HSCs was isolated using the Qiagen RNeasy Mini Kit (Valencia, CA) according to the manufacturers’ instructions. RNA yield and quality were determined by measuring absorbencies at 260 nm and 280 nm, respectively. First-strand cDNA was synthesized in a final volume of 20 μl using the Superscript III First-Strand Synthesis System (Invitrogen, Carlsbad, CA). qRT-PCR analyses were performed using a SYBR Green mix on an ABI StepOne Plus Real-Time PCR System (Applied Biosystems, Foster City, CA). Hypoxanthinephophoribosyltransferase (HPRT) transcripts were used as a housekeeping internal reference for mRNA. The expression of Catalase, GPX1 (Glutathione Peroxidase 1), GPX2, SOD1 (Superoxide Dismutase 1), SOD2 and SOD3 was calculated by the comparative CT method. The sequences for all the primers used in the qRT-PCR assays are available upon request.
Statistical analysis
All data are presented as mean ± standard deviation of three to five independent biological samples per radiation dose. The differences between sham-irradiated and irradiated groups were examined by one-way ANOVA, followed by Tukey-Kramer test for individual comparisons. Differences were considered significant at p < .05. Statistical analysis was performed using GraphPad Prism (GraphPad Software Inc. LaJolla, CA).
Results
16O TBI induced long-term BM suppression
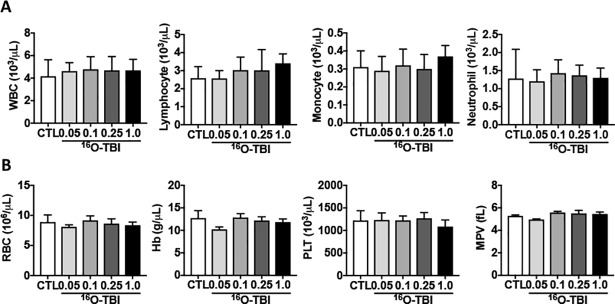
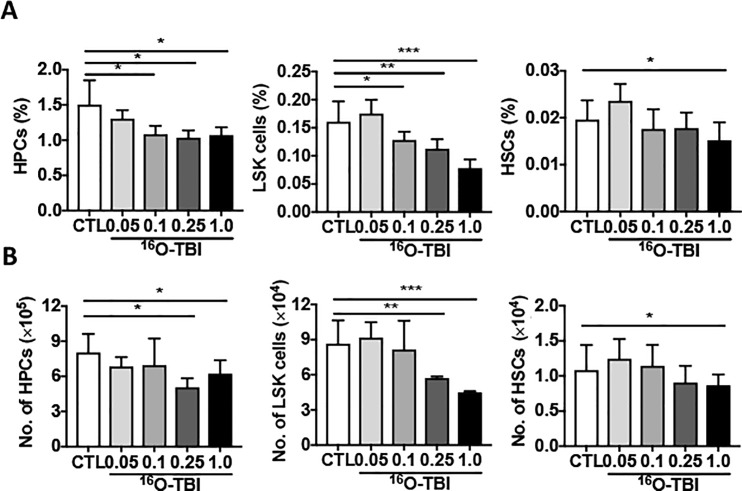
We have previously shown that 1.0 Gy of 16O TBI significantly decreased peripheral blood cell counts, particularly those of white blood cells (WBC) and platelets (PLT), in mice two weeks after TBI[21]. To assess the chronic effects of 16O TBI on peripheral blood cells, we collected peripheral blood at three months after 0.05 Gy, 0.1 Gy, 0.25 Gy and 1.0 Gy of 16O TBI. As shown in Fig 1, the numbers of all different types of peripheral blood cells including WBC, PLT and red blood cells had reached normal levels, regardless of radiation doses. Peripheral blood cells were also recovered at three months after 0.5 Gy and 1.0 Gy of γ-ray irradiation (S1 Fig). However, the data from peripheral blood cell counts could not infer potential changes in BM HPCs and HSCs after TBI as shown in our previous studies [21]. Therefore, the frequencies and numbers of different BM hematopoietic cell populations were analyzed by flow cytometry (S2 Fig). As shown in Fig 2A, compared to 0.05 Gy 16O TBI and non-irradiated controls, the frequency of HPCs (Lin-Sca1-c-kit+ cells) was significantly reduced in all other three doses of 16O-irradiated mice (p<0.05). Moreover, the frequency of LSK cells (Lin-Sca1+c-kit+cells) was decreased in 0.1 Gy-, 0.25 Gy- and 1.0 Gy-irradiated mice compared to that in 0.05 Gy irradiated and non-irradiated mice (p<0.05-p<0.001). While the frequency of HSCs (Lin-Sca1+c-kit+CD150+CD48-cells) in 1.0 Gy irradiated mice was decreased compared to that in 0.05 Gy-, 0.1 Gy-, 0.25 Gy- and non-irradiated mice(p<0.05), the frequencies of HSCs in 0.05 Gy-, 0.1 Gy-, 0.25 Gy-irradiated mice were comparable to those in non-irradiated mice. The total numbers of HPCs, LSK cells and HSCs showed trends similar to those of the frequencies of each of these cell populations (Fig 2B, p<0.05–0.001). There were increasing trends in both frequencies and numbers of HSCs from 0.5 Gy and 1.0 Gy γ-irradiated mice without reaching statistical significance (S3A and S3B Fig). This might be due to the variations in HSCs from non-irradiated mice. The frequencies and numbers of HPCs and LSK cells from γ-irradiated mice were comparable to those in non-irradiated controls at three months after 0.5 Gy and 1.0 Gy γ-ray exposure (S3A and S3B Fig).
Fig 1. Peripheral blood cell counts were comparable between non-irradiated and irradiated mice at three months after 16O exposure.
C57BL/6J mice were exposed to 0.05 Gy, 0.1 Gy, 0.25 Gy and 1.0 Gy doses of 16O irradiation or were sham irradiated as a control (CTL). The cell counts in peripheral blood were determined three months after radiation exposure. (A-C) The numbers of WBC, lymphocytes, monocytes, neutrophils, RBC, Hb, platelet (PLT) and mean platelet volume (MPV) in irradiated mice are presented as means ±SD (n = 5), and comparable to those in non-irradiated mice.
Fig 2. 16O TBI caused reductions in percentages and numbers of HPCs, LSK cells and HSCs at three months after exposure.
HPCs (Lin-Sca1-c-kit- cells), LSK cells (Lin-Sca1+c-kit+cells) and HSCs (Lin-Sca1+c-kit+CD150+CD48- cells) in BM were measured three months after 0.05 Gy, 0.1 Gy, 0.25 Gy, and 1.0 Gy 16O TBI. The frequencies (panel A) and numbers (panel B) of HPCs, LSK cells and HSCs from total bone marrow cells in each mouse are presented as means ±SD (n = 5). The statistical significance for differences between the control group (CTL) and each of the irradiated groups is indicated by asterisks. *p<0.05, **p<0.01, ***p<0.001 as determined by one-way ANOVA, followed by Tukey-Kramer test for individual comparisons.
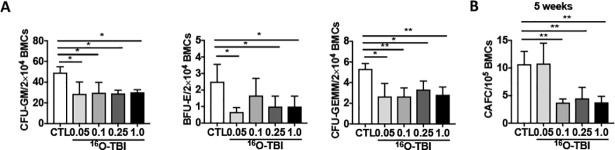
To further evaluate the effect of 16O TBI on BM HPCs and HSCs, a colony forming assay was used to examine their clonogenic function. The results showed that the frequencies of BFU-E, CFU-GMs, and CFU-GEMMs in BM cells were significantly reduced, regardless of radiation doses (Fig 3A, p<0.05–0.01), indicating that the clonogenic function of HPCs was dramatically impaired in all irradiated mice, while the decreased colony forming abilities of HPCs and HSCs were not seen three months after 0.5 Gy and 1.0 Gy γ-TBI (S3C Fig). In addition, we used another standard in vitro HSC functional assay named the cobblestone area forming assay (CAFC) to assess the function of HSCs. The data showed that the frequencies of 5-week CAFCs were significantly lower in BM cells from 0.1 Gy, 0.25 Gy and 1.0 Gy 16O-irradiated mice than those from 0.05 Gy 16O-irradiated mice and non-irradiated controls (Fig 3B, p<0.01). However, there were comparable numbers of CAFCs formed in 0.1 Gy, 0.25 Gy and 1.0 Gy 16O-irradiated mice. The frequencies of 5-week CAFCs in 0.05 Gy 16O-irradiated mice were equal to those in non-irradiated mice. These data indicate that exposure of mice to 16O TBI, especially higher than 0.05 Gy 16O TBI, causes long-term damage not only to HPCs but also to HSCs.
Fig 3. 16O TBI caused the reduction of the clonogenic function in HPCs and HSCs.
(A) BM-MNCs were isolated from irradiated and non-irradiated (CTL) mice three months after 16O TBI and a CFU assay was performed. Results are presented as mean CFUs per 1x105 BM-MNCs (n = 5). (B) Total BM cells (BMCs) were analyzed by CAFC assays, and the numbers of five-week CAFCs are expressed as means ± SD (n = 3 mice per group) per 1x105 BMCs. The statistical significance for differences between the control group and each of the irradiated groups is indicated by asterisks. *p<0.05, **p<0.01 as determined by one-way ANOVA, followed by Tukey-Kramer test for individual comparisons.
16O TBI induced oxidative stress in HSCs
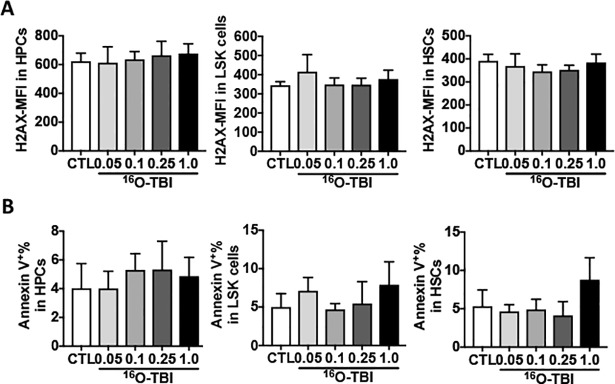
Exposure to ionizing radiation can cause oxidative stress, DNA damage and apoptosis or senescence [23]. To test these possibilities, we first used γH2AX immunostaining to assess DNA double strand breaks in HPCs and HSCs after 16O TBI. The immune fluorescence intensities (MFI) of γH2AX in HPCs, LSK cells and HSCs were comparable between non-irradiated and irradiated cells three months after 16O TBI (Fig 4A). We then tested apoptosis in different populations using Annexin V staining. As shown in Fig 4B, there were no differences in the percentage of apoptotic cells between irradiated and non-irradiated HPCs. Compared to 0.05 Gy-, 0.1 Gy-, 0.25 Gy- and non-irradiated groups, LSK cells and HSCs exposed to 1.0 Gy showed a trend of 5–10% increase in apoptosis without reaching a statistical significance.
Fig 4. No changes were detected in DNA damage and apoptosis in HPCs, LSK cells and HSCs at three months after 16O TBI.
(A) Lin- cells were stained with an anti-γH2AX antibody and analyzed by flow cytometry. Data are presented as mean fluorescence intensity (MFI). (B) Isolated Lin- cells were stained with Annexin V to determine cellular apoptosis. Percentages of Annexin V positive cells are presented as means ± SD (n = 5).
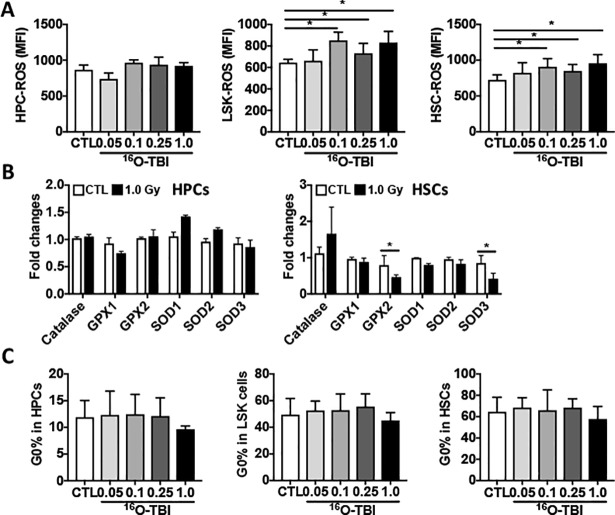
In addition to DNA damage and apoptosis, ionizing radiation can also affect cellular function through induction of oxidative stress and alterations in cell cycling [23]. To determine whether induction of oxidative stress may mediate the effects of 16O TBI on BM HPCs and HSCs, the intracellular production of ROS in HPCs and HSCs was assessed. As shown in S4 Fig, the distribution of ROS in non-irradiated and 1.0 Gy 16O-irradiated HPCs was similar, while the distribution of ROS in 1.0 Gy 16O-irradiated LSK cells and HSCs was different from that in non-irradiated LSK cells and HSCs. Consistently, ROS production in irradiated HPCs was comparable to that in cells from non-irradiated mice (Fig 5A). There were significantly higher levels of ROS production (up to 30% increase of normal levels) in LSK cells and HSCs from mice exposed to 0.1 Gy, 0.25 Gy and 1.0 Gy of 16O TBI compared to that in 0.05 Gy 16O TBI and non-irradiated cells (Fig 5A, p<0.05). However, the levels of ROS production in LSK cells and HSCs from mice exposed to different doses of 16O were not significantly different. To determine whether the increase in ROS production was attributable to a decreased expression of antioxidant enzymes, we sorted out HPCs and HSCs from 1.0 Gy 16O irradiated and non-irradiated mice to perform gene expression analysis. As shown in Fig 5B, there were significant lower levels of GPX2 and SOD3 expression in irradiated HSCs compared to those in non-irradiated HSCs (p<0.05). No difference was observed in the expression of catalase, GPX1, SOD1 and SOD2 between irradiated and non-irradiated HSCs. There were comparable expression levels of these antioxidant genes in irradiated and non-irradiated HPCs (Fig 5B). We also utilized Ki-67 and 7-AAD double immunostaining to measure the cell cycle status of HPCs and HSCs after 16O exposure. Comparable percentages of cells in G0 phase were detected in HPCs, LSK cells and HSCs from non-irradiated and irradiated mice (Fig 5B), suggesting that the status of the cell cycle in BM HPCs and HSCs was recovered at 3 months after 16O irradiation. However, no increased DNA damage, apoptosis, ROS and cell cycle in irradiated HPCs and HSCs were detected three months after 0.5 Gy and 1.0 Gy of γ-ray exposure (S5A and S5B Fig, data not shown).
Fig 5. 16O TBI caused an increase in ROS production in LSK cells and HSCs three months after the exposure.
(A) Lin- cells were used to measured ROS production by staining with DCFDA and analyzed by flow cytometry. The DCF mean fluorescence intensity (MFI) in BM HPCs and HSCs are presented as means ± SD (n = 5). (B) Fold changes in relative gene expression for several antioxidant genes in sorted HPCs (left panel) and HSCs (right panel) from 1.0 Gy of 16O TBI and non-irradiated mice. (C) Lin- cells were isolated and cell cycling was measured by cytometry using Ki-67 and 7-AAD double staining in HPCs and HSCs from control (CTL) and irradiated mice. The statistical significance for differences between the control group and each of the irradiated groups is indicated by asterisks. *p<0.05, **p<0.01, ***p<0.001 as determined by one-way ANOVA, followed by Tukey-Kramer test for individual comparisons.
Discussion
Based on measurements in the Mars Science Laboratory in 2011–2012, the exposure to GCR inside the space craft was estimated to be 481 ± 80 μGy per day or 176 ± 29 mGy per year [24]. To straddle this estimated annual dose, we selected doses from 0.05 Gy to 0.25 Gy in the range of expected doses for extended lunar and deep space missions. For a 600- to 900- day Mars mission, the contributions from GCR would be 0.33 to 0.49 Gy (neglecting protons from solar particle events which could bring total exposures close to 1.0 Gy). Therefore, 1.0 Gy high dose of 16O irradiation allows us to span the range of expected values and enables us to detect curvature in our dose responses. Because some doses used in the present study are higher than those exposed by astronauts during space flights, cautions should be taken when the findings from our mouse study are extrapolated to humans. During long-term space missions, astronauts will be exposed to space irradiation in a chronic or fractionated manner. To better mimic HZE exposure in deep space, chronic or fractionated irradiation should be used in the actual studies. However, these are not currently achievable because of radiation source limiting access. In the present report, the acute dose rate (0.25–0.26 Gy/min) was used with a limitation of current study, while these practical limitations may not invalidate the value of the study.
In our previous study, we evaluated the acute effects of 16O TBI on the hematopoietic system two weeks after irradiation in a mouse model and showed that 16O TBI acutely decreased various peripheral blood cell counts and the frequency and number of BM HPCs and HSCs[21]. The present study extended previous investigations using four different doses of 16O TBI to determine long-term effects of 16O TBI on the hematopoietic system. We are the first to demonstrate that exposure to low doses of 16O TBI induces long-term damage to BM HPCs and HSCs in mice, but without a significant effect on the numbers of WBC, RBC and PLT. These long-term effects of 16O irradiation on peripheral blood cells are similar to the effects of other types of ionizing radiation including currently used 0.5 Gy and 1.0 Gy of γ-TBI[25, 26]. For instance, the restoration of peripheral blood cell counts to normal levels could be seen two months after γ-ray exposure[27]. However, through analysis of BM HPCs and HSCs, we show that the seemingly complete recovery of peripheral blood cell counts may obscure the discovery of long-term BM suppression induced by radiation[25], which may cause this important pathology to be overlooked.
Studies from other labs and ours have demonstrated that sublethal doses of γ-rays (such as 6.5 Gy γ-TBI) and low doses of proton radiation resulted in long-term residual damage in the hematopoietic system[23, 26, 28, 29], as evidenced by a reduction in HSC reserves and a defect in HSC function. Consistent with these previous results, the current study showed that low doses of 16O TBI, especially higher than 0.05 Gy of 16O TBI, induced a significant decrease in the numbers and function of LSK cells and HSCs in BM three months after exposure. However, it was not seen at three months after 0.5 Gy and 1.0 Gy of γ-TBI. On the other hand, while the numbers of HPCs in irradiated mice reached normal levels two weeks after γ-ray and proton exposure, our data indicate that acute exposure to low doses of 16O TBI causes significant reductions in numbers of HPCs at 2 weeks and 3 months after the exposure [21]. These results suggest that 16O irradiated HPCs experience a slower recovery than proton- and photon-irradiated HPCs. This may be related to the higher RBE [30] and potential bystander effects of 16O irradiation. According to the calculation of the average numbers of oxygen ion traversing cells, each cell will experience 1.91 ions for 0.1 Gy exposure and 4.78 ions for 0.25 Gy exposure. After applying a poisson distribution (Table 1), we estimate that at a dose of 0.1 Gy 85% of cells will be traversed by one or more ions and 15% will not be traversed. Therefore, non-irradiated cells (15%) might produce irradiated effects because of the signals from irradiated cells (85%). On the other hand, a dose of 0.25 Gy would result in 99% of cells being traversed. Due to the poisson distribution of particle traversal, traversed cells will experience different doses at low doses of exposure, which might cause heterogenous responses in irradiated hematopoietic cells.
The sensitivity of HPCs and HSCs to ionizing radiation is usually examined by evaluating both cell numbers and their function. We have previously shown that HPCs, but not HSCs, showed high levels of apoptosis two weeks after 1.0 Gy of 16O TBI, together with a decrease in the HPC colony forming abilities as indicated by lower numbers of BFU-E, CFU-GM and CFU-GEMM compared to those in non-irradiated HPCs[21]. The current study shows that HPC colony forming abilities remained much lower compared to normal controls three months after 16O TBI. Moreover, the decreased colony forming abilities of irradiated HPCs were radiation dose-independent in the dose-range used here, which suggests “hit and damage”. However, we don’t know the underlying mechanisms of the decreased HPC function after low doses of 16O TBI, since apoptosis, ROS production and DNA damage in irradiated HPCs were comparable with non-irradiated controls.
Our in vitro CAFC assay demonstrated that 16O TBI reduced the long-term colony forming capacities of HSCs after 0.1 Gy, 0.25 Gy and 1.0 Gy exposure, again indicating “hit and damage”. However, there were comparable numbers of 5-week CAFCs between 0.05 Gy irradiated mice and non-irradiated controls, suggesting that HSC function was not reduced after exposure to 0.05 Gy 16O TBI. These long-term effects of 16O TBI on HSCs are similar to those of other types of ionizing radiation[20, 31, 32]. For example, 1.0 Gy of proton radiation decreased the CAFC forming ability in HSCs at 22 weeks in a mouse model[20]. A dose of 6.5 Gy of γ-rays reduced HSC colony forming ability 2 months after the exposure[7]. These findings indicate that long-term HSC suppression occurs upon low doses of ionizing radiation, including 16O, but 16O is more potent to induce the long-term HSC suppression than other types of ionizing radiation. Although the numbers and function of HSCs in BM were decreased upon 1.0 Gy of 16O TBI, the counts of peripheral blood cells were back to normal levels at 3 months after exposure. Proliferation of progenitors (such as myeloid, lymphoid and erythroid progenitors) might compensate for the decrement of irradiated HSCs, which would cause a transient decrease of these progenitors at a certain time point after irradiation. Notably, the decrease of HSC functions induced by 16O irradiation appeared in all irradiated mice in a dose-independent manner. These unusual dose-response curves have also been seen in the studies of 28Si irradiation, such as effects of 28Si irradiation on synaptic plasticity and contextual fear memory [33, 34].
Previously, we demonstrated that the acute adverse effects of 16O TBI on HSCs could be ascribed to induction of apoptosis, DNA damage, ROS production and enhanced cell cycling[21]. In this study, we extended these studies by measuring these parameters in HSCs three months after 16O TBI. While the levels of apoptosis, DNA damage and cell cycling were comparable to those in non-irradiated HSCs, there was a consistent increase in ROS production in irradiated HSCs from 16O TBI mice. Consistently, the expression of the antioxidant genes GPX2 and SOD3 was significantly decreased in 1.0 Gy 16O irradiated HSCs compared to non-irradiated HSCs. The long-term negative effects of ROS accumulation in HSCs have been studied extensively using proton radiation and γ-rays[20, 31, 35, 36]. For example, we have previously shown that both protons and γ-rays induced an increase in ROS in HSCs, together with a decrease in in vitro colony forming ability and in vivo engraftment capacity. Our recent data showed that γ-Tocotrienol (γ-GT3) treatment significantly increased the capacities of hematopoietic stem and progenitors to forming colonies at three months after 0.25 Gy of 16O TBI. This is because 16O TBI-induced oxidative stress in irradiated HSCs was significantly decreased after γ-GT3 treatment (unpublished data). These data suggest that oxidative stress may be a major detrimental player in abnormal HSC function in response to ionizing radiation, including 16O. In addition, several studies demonstrated that low doses of space ion radiation (such as proton, 56Fe and 28Si) induced chromosomal aberrations in human lymphocytes and mouse BM cells [37–40], which may predispose irradiated individuals to leukemia. Thus, it is necessary to further assess whether low doses of 16O radiation induce chromosomal aberrations in near future, which might be a potential risk factor of 16O irradiation-induced hematopoietic malignancies.
Our present study demonstrates long-term effects of 16O TBI on HSCs. To minimize the health effects of deep space travel, decreasing oxidative stress might be a good approach to mitigate the adverse effects of HZE particle exposure on the hematopoietic system.
Supporting information
C57BL/6J mice were exposed to 0.5 Gy and 1.0 Gy doses of γ–ray irradiation (γ-TBI) or were sham irradiated as a control (CTL). The cell counts in peripheral blood were determined three months after radiation exposure. (A-C) The numbers of WBC, lymphocytes, monocytes, neutrophils, RBC, Hb, platelet (PLT) and mean platelet volume (MPV) in irradiated mice are presented as means ±SD (n = 5), and comparable to those in non-irradiated mice.
(TIF)
Representative gating strategy of flow cytometric analysis for HPCs (Lin-Sca1-c-kit- cells), LSK cells (Lin-Sca1+c-kit+cells) and HSCs (Lin-Sca1+c-kit+CD150+CD48- cells) in bone marrow is shown from 1.0 Gy of 16O-TBI and sham-irradiation (CTL).
(TIF)
(A and B) HPCs, LSK cells and HSCs in BM were measured three months after 0.5 Gy and 1.0 Gy γ-TBI. The frequencies (panel A) and numbers (panel B) of HPCs, LSK cells and HSCs from total bone marrow cells in each mouse are presented as means ±SD (n = 5). (C) BM-MNCs were isolated from irradiated and non-irradiated (CTL) mice three months after γ-TBI and a CFU assay was performed. Results are presented as mean CFUs per 2x104 BM-MNCs (n = 5). The statistical significance for differences between the control group and each of the irradiated groups was determined by one-way ANOVA, followed by Tukey-Kramer test for individual comparisons.
(TIF)
Lin- cells were stained with the probe DCFDA and various surface markers, and analyzed by flow cytometry. The distribution and mean fluorescence intensity (MFI) of ROS in non-irradiated and irradiated HPCs, LSK cells and HSCs were presented.
(TIF)
(A) Lin- cells were used to measured ROS production by staining with DCFDA and analyzed by flow cytometry three months after 0.5 Gy and 1.0 Gy γ-TBI. The DCF mean fluorescence intensity (MFI) in BM HPCs, LSK cells and HSCs are presented as means ± SD (n = 5). (B) Isolated Lin- cells were stained with Annexin V to determine cellular apoptosis. Percentages of Annexin V positive cells are presented as means ± SD (n = 5).
(TIF)
Acknowledgments
The study was supported in part by the National Space Biomedical Research Institute through NCC 9–58 (MB), Centers of Biomedical Research Excellence (P20, GM109005, MHJ), and R01CA122023 (DZ).
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
The study was supported in part by the National Space Biomedical Research Institute through NCC 9-58 (MB), Centers of Biomedical Research Excellence (P20, GM109005, MHJ), and National Institutes of Health (R01CA122023, DZ).
References
- 1.Hellweg CE and Baumstark-Khan C. Getting ready for the manned mission to Mars: the astronauts' risk from space radiation. Naturwissenschaften. 2007; 94:517–526. doi: 10.1007/s00114-006-0204-0 [DOI] [PubMed] [Google Scholar]
- 2.Datta K, Suman S, Trani D, Doiron K, Rotolo JA, Kallakury BV, et al. Accelerated hematopoietic toxicity by high energy (56)Fe radiation. Int J Radiat Biol. 2012; 88:213–222. doi: 10.3109/09553002.2012.639434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boerma M, Sridharan V, Mao XW, Nelson GA, Cheema AK, Koturbash I, et al. Effects of ionizing radiation on the heart. Mutat Res. 2016; 770:319–327. doi: 10.1016/j.mrrev.2016.07.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hamilton SA, Pecaut MJ, Gridley DS, Travis ND, Bandstra ER, Willey JS, et al. A murine model for bone loss from therapeutic and space-relevant sources of radiation. J Appl Physiol (1985). 2006; 101:789–793. [DOI] [PubMed] [Google Scholar]
- 5.Cucinotta FA, Wu H, Shavers MR and George K. Radiation dosimetry and biophysical models of space radiation effects. Gravit Space Biol Bull. 2003; 16:11–18. [PubMed] [Google Scholar]
- 6.Suman S, Datta K, Trani D, Laiakis EC, Strawn SJ and Fornace AJ Jr. Relative biological effectiveness of 12C and 28Si radiation in C57BL/6J mice. Radiat Environ Biophys. 2012; 51:303–309. doi: 10.1007/s00411-012-0418-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang Y, Schulte BA, LaRue AC, Ogawa M and Zhou D. Total body irradiation selectively induces murine hematopoietic stem cell senescence. Blood. 2006; 107:358–366. doi: 10.1182/blood-2005-04-1418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shao L, Sun Y, Zhang Z, Feng W, Gao Y, Cai Z, et al. Deletion of proapoptotic Puma selectively protects hematopoietic stem and progenitor cells against high-dose radiation. Blood. 2010; 115:4707–4714. doi: 10.1182/blood-2009-10-248872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yu H, Shen H, Yuan Y, XuFeng R, Hu X, Garrison SP, et al. Deletion of Puma protects hematopoietic stem cells and confers long-term survival in response to high-dose gamma-irradiation. Blood. 2010; 115:3472–3480. doi: 10.1182/blood-2009-10-248278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hagan MP, Holahan EV and Ainsworth EJ. Effects of heavy ions on cycling stem cells. Adv Space Res. 1986; 6:201–211. [DOI] [PubMed] [Google Scholar]
- 11.Ainsworth EJ, Afzal SM, Crouse DA, Hanson WR and Fry RJ. Tissue responses to low protracted doses of high LET radiations or photons: early and late damage relevant to radio-protective countermeasures. Adv Space Res. 1989; 9:299–313. [DOI] [PubMed] [Google Scholar]
- 12.Phillips TL, Ross GY, Goldstein LS, Ainsworth J and Alpen E. In vivo radiobiology of heavy ions. Int J Radiat Oncol Biol Phys. 1982; 8:2121–2125. [DOI] [PubMed] [Google Scholar]
- 13.Kajioka EH, Andres ML, Li J, Mao XW, Moyers MF, Nelson GA, et al. Acute effects of whole-body proton irradiation on the immune system of the mouse. Radiat Res. 2000; 153:587–594. [DOI] [PubMed] [Google Scholar]
- 14.Gridley DS, Rizvi A, Luo-Owen X, Makinde AY, Coutrakon GB, Koss P, et al. Variable hematopoietic responses to acute photons, protons and simulated solar particle event protons. In Vivo. 2008; 22:159–169. [PubMed] [Google Scholar]
- 15.Ware JH, Sanzari J, Avery S, Sayers C, Krigsfeld G, Nuth M, et al. Effects of proton radiation dose, dose rate and dose fractionation on hematopoietic cells in mice. Radiat Res. 2010; 174:325–330. doi: 10.1667/RR1979.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maks CJ, Wan XS, Ware JH, Romero-Weaver AL, Sanzari JK, Wilson JM, et al. Analysis of white blood cell counts in mice after gamma- or proton-radiation exposure. Radiat Res. 2011; 176:170–176. doi: 10.1667/RR2413.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Luo-Owen X, Pecaut MJ, Rizvi A and Gridley DS. Low-dose total-body gamma irradiation modulates immune response to acute proton radiation. Radiat Res. 2012; 177:251–264. [DOI] [PubMed] [Google Scholar]
- 18.Sanzari JK, Cengel KA, Wan XS, Rusek A and Kennedy AR. Acute Hematological Effects in Mice Exposed to the Expected Doses, Dose-rates, and Energies of Solar Particle Event-like Proton Radiation. Life Sci Space Res (Amst). 2014; 2:86–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gridley DS and Pecaut MJ. Whole-body irradiation and long-term modification of bone marrow-derived cell populations by low- and high-LET radiation. In Vivo. 2006; 20:781–789. [PubMed] [Google Scholar]
- 20.Chang J, Feng W, Wang Y, Luo Y, Allen AR, Koturbash I, et al. Whole-body proton irradiation causes long-term damage to hematopoietic stem cells in mice. Radiat Res. 2015; 183:240–248. doi: 10.1667/RR13887.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chang J, Luo Y, Wang Y, Pathak R, Sridharan V, Jones T, et al. Low Doses of Oxygen Ion Irradiation Cause Acute Damage to Hematopoietic Cells in Mice. PLoS One. 2016; 11:e0158097 doi: 10.1371/journal.pone.0158097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gao DY, Chang Q, Liu C, Farris K, Harvey K, McGann LE, et al. Fundamental cryobiology of human hematopoietic progenitor cells. I: Osmotic characteristics and volume distribution. Cryobiology. 1998; 36:40–48. doi: 10.1006/cryo.1997.2060 [DOI] [PubMed] [Google Scholar]
- 23.Shao L, Luo Y and Zhou D. Hematopoietic stem cell injury induced by ionizing radiation. Antioxid Redox Signal. 2014; 20:1447–1462. doi: 10.1089/ars.2013.5635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zeitlin C, Hassler DM, Cucinotta FA, Ehresmann B, Wimmer-Schweingruber RF, Brinza DE, et al. Measurements of energetic particle radiation in transit to Mars on the Mars Science Laboratory. Science. 2013; 340:1080–1084. doi: 10.1126/science.1235989 [DOI] [PubMed] [Google Scholar]
- 25.Chang J, Wang Y, Shao L, Laberge RM, Demaria M, Campisi J, et al. Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat Med. 2016; 22:78–83. doi: 10.1038/nm.4010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xu G, Wu H, Zhang J, Li D, Wang Y, Zhang H, et al. Metformin ameliorates ionizing irradiation-induced long-term hematopoietic stem cell injury in mice. Free Radic Biol Med. 2015; 87:15–25. doi: 10.1016/j.freeradbiomed.2015.05.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Testa NG, Hendry JH and Molineux G. Long-term bone marrow damage in experimental systems and in patients after radiation or chemotherapy. Anticancer Res. 1985; 5:101–110. [PubMed] [Google Scholar]
- 28.Farres J, Martin-Caballero J, Martinez C, Lozano JJ, Llacuna L, Ampurdanes C, et al. Parp-2 is required to maintain hematopoiesis following sublethal gamma-irradiation in mice. Blood. 2013; 122:44–54. doi: 10.1182/blood-2012-12-472845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chua HL, Plett PA, Sampson CH, Joshi M, Tabbey R, Katz BP, et al. Long-term hematopoietic stem cell damage in a murine model of the hematopoietic syndrome of the acute radiation syndrome. Health Phys. 2012; 103:356–366. doi: 10.1097/HP.0b013e3182666d6f [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Habermehl D, Ilicic K, Dehne S, Rieken S, Orschiedt L, Brons S, et al. The relative biological effectiveness for carbon and oxygen ion beams using the raster-scanning technique in hepatocellular carcinoma cell lines. PLoS One. 2014; 9:e113591 doi: 10.1371/journal.pone.0113591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li H, Wang Y, Pazhanisamy SK, Shao L, Batinic-Haberle I, Meng A, et al. Mn(III) meso-tetrakis-(N-ethylpyridinium-2-yl) porphyrin mitigates total body irradiation-induced long-term bone marrow suppression. Free Radic Biol Med. 2011; 51:30–37. doi: 10.1016/j.freeradbiomed.2011.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Epperly MW, Wang H, Jones JA, Dixon T, Montesinos CA and Greenberger JS. Antioxidant-chemoprevention diet ameliorates late effects of total-body irradiation and supplements radioprotection by MnSOD-plasmid liposome administration. Radiat Res. 2011; 175:759–765. doi: 10.1667/RR2398.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Raber J, Marzulla T, Stewart B, Kronenberg A and Turker MS. 28Silicon Irradiation Impairs Contextual Fear Memory in B6D2F1 Mice. Radiat Res. 2015; 183:708–712. doi: 10.1667/RR13951.1 [DOI] [PubMed] [Google Scholar]
- 34.Raber J, Rudobeck E, Campbell-Beachler M, Allen AR, Allen B, Rosi S, et al. (28)Silicon radiation-induced enhancement of synaptic plasticity in the hippocampus of naive and cognitively tested mice. Radiat Res. 2014; 181:362–368. doi: 10.1667/RR13347.1 [DOI] [PubMed] [Google Scholar]
- 35.Chai X, Li D, Cao X, Zhang Y, Mu J, Lu W, et al. ROS-mediated iron overload injures the hematopoiesis of bone marrow by damaging hematopoietic stem/progenitor cells in mice. Sci Rep. 2015; 5:10181 doi: 10.1038/srep10181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cho J, Yusuf R, Kook S, Attar E, Lee D, Park B, et al. Purinergic P2Y(1)(4) receptor modulates stress-induced hematopoietic stem/progenitor cell senescence. J Clin Invest. 2014; 124:3159–3171. doi: 10.1172/JCI61636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rithidech KN, Honikel LM, Reungpatthanaphong P, Tungjai M, Golightly M and Whorton EB. Effects of 100MeV protons delivered at 0.5 or 1cGy/min on the in vivo induction of early and delayed chromosomal damage. Mutat Res. 2013; 756:127–140. doi: 10.1016/j.mrgentox.2013.06.001 [DOI] [PubMed] [Google Scholar]
- 38.George KA, Hada M and Cucinotta FA. Biological Effectiveness of Accelerated Protons for Chromosome Exchanges. Front Oncol. 2015; 5:226 doi: 10.3389/fonc.2015.00226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rithidech KN, Honikel LM, Reungpathanaphong P, Tungjai M, Jangiam W and Whorton EB. Late-occurring chromosome aberrations and global DNA methylation in hematopoietic stem/progenitor cells of CBA/CaJ mice exposed to silicon ((28)Si) ions. Mutat Res. 2015; 781:22–31. doi: 10.1016/j.mrfmmm.2015.09.001 [DOI] [PubMed] [Google Scholar]
- 40.Ritter S and Durante M. Heavy-ion induced chromosomal aberrations: a review. Mutat Res. 2010; 701:38–46. doi: 10.1016/j.mrgentox.2010.04.007 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
C57BL/6J mice were exposed to 0.5 Gy and 1.0 Gy doses of γ–ray irradiation (γ-TBI) or were sham irradiated as a control (CTL). The cell counts in peripheral blood were determined three months after radiation exposure. (A-C) The numbers of WBC, lymphocytes, monocytes, neutrophils, RBC, Hb, platelet (PLT) and mean platelet volume (MPV) in irradiated mice are presented as means ±SD (n = 5), and comparable to those in non-irradiated mice.
(TIF)
Representative gating strategy of flow cytometric analysis for HPCs (Lin-Sca1-c-kit- cells), LSK cells (Lin-Sca1+c-kit+cells) and HSCs (Lin-Sca1+c-kit+CD150+CD48- cells) in bone marrow is shown from 1.0 Gy of 16O-TBI and sham-irradiation (CTL).
(TIF)
(A and B) HPCs, LSK cells and HSCs in BM were measured three months after 0.5 Gy and 1.0 Gy γ-TBI. The frequencies (panel A) and numbers (panel B) of HPCs, LSK cells and HSCs from total bone marrow cells in each mouse are presented as means ±SD (n = 5). (C) BM-MNCs were isolated from irradiated and non-irradiated (CTL) mice three months after γ-TBI and a CFU assay was performed. Results are presented as mean CFUs per 2x104 BM-MNCs (n = 5). The statistical significance for differences between the control group and each of the irradiated groups was determined by one-way ANOVA, followed by Tukey-Kramer test for individual comparisons.
(TIF)
Lin- cells were stained with the probe DCFDA and various surface markers, and analyzed by flow cytometry. The distribution and mean fluorescence intensity (MFI) of ROS in non-irradiated and irradiated HPCs, LSK cells and HSCs were presented.
(TIF)
(A) Lin- cells were used to measured ROS production by staining with DCFDA and analyzed by flow cytometry three months after 0.5 Gy and 1.0 Gy γ-TBI. The DCF mean fluorescence intensity (MFI) in BM HPCs, LSK cells and HSCs are presented as means ± SD (n = 5). (B) Isolated Lin- cells were stained with Annexin V to determine cellular apoptosis. Percentages of Annexin V positive cells are presented as means ± SD (n = 5).
(TIF)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.