Abstract
Background
Regular fish and omega-3 consumption may have several health benefits and are recommended by major dietary guidelines. Yet, their intakes remain remarkably variable both within and across populations, which could partly owe to genetic influences.
Objective
To identify common genetic variants that influence fish and dietary eicosapentaenoic acid plus docosahexaenoic acid (EPA+DHA) consumption.
Design
We conducted genome-wide association (GWA) meta-analysis of fish (n = 86,467) and EPA+DHA (n = 62,265) consumption in 17 cohorts of European descent from the CHARGE (Cohorts for Heart and Aging Research in Genomic Epidemiology) Consortium Nutrition Working Group. Results from cohort-specific GWA analyses (additive model) for fish and EPA+DHA consumption were adjusted for age, sex, energy intake, and population stratification, and meta-analyzed separately using fixed-effect meta-analysis with inverse variance weights (METAL software). Additionally, heritability was estimated in 2 cohorts.
Results
Heritability estimates for fish and EPA+DHA consumption ranged from 0.13–0.24 and 0.12–0.22, respectively. A significant GWA for fish intake was observed for rs9502823 on chromosome 6: each copy of the minor allele (FreqA = 0.015) was associated with 0.029 servings/day (~1 serving/month) lower fish consumption (P = 1.96x10-8). No significant association was observed for EPA+DHA, although rs7206790 in the obesity-associated FTO gene was among top hits (P = 8.18x10-7). Post-hoc calculations demonstrated 95% statistical power to detect a genetic variant associated with effect size of 0.05% for fish and 0.08% for EPA+DHA.
Conclusions
These novel findings suggest that non-genetic personal and environmental factors are principal determinants of the remarkable variation in fish consumption, representing modifiable targets for increasing intakes among all individuals. Genes underlying the signal at rs72838923 and mechanisms for the association warrant further investigation.
Introduction
Consumption of fish (including finfish and shellfish) and long-chain omega-3 fatty acids is linked to lower risk of several chronic diseases, in particular fatal coronary heart disease [1]. These beneficial associations in observational studies are supported by randomized controlled trials demonstrating favorable effects of fish or fish oil on numerous chronic disease risk factors and on cardiac mortality [1,2]. As a result, regular fish consumption is recommended by all major national and international dietary guidelines [1].
In contrast to these guidelines and in comparison to many other foods, remarkable variation exists in the amount of fish consumption within and across populations. In many Western nations, approximately one-third of individuals consume no fish at all, approximately one-third consume fish but relatively rarely (up to once per week), and approximately one-third consume fish more frequently [3]. While some of this wide variation in fish consumption is undoubtedly due to personal and environmental factors (e.g., culture, geographic residence, family habits, socioeconomic status), the potential contribution of intrinsic biologic factors, such as genetic variation, is not well established. In one analysis among Danish twins, the estimated heritability of fish consumption was 17% in men and 61% in women, based on additive genetic effects [4]. For example, potential heritability could relate to differences in genes related to taste, digestion, fatty acid metabolism, or other unknown processes related to food preferences. Yet, the potential genetic variants underlying this estimated heritability are unknown; and such heritability estimates also require further replication.
A basic concept underlying “personalized nutrition” is that a person’s genes can influence their behaviors and responses to the environment. Dietary habits, including the consumption of fish, are among the most relevant factors that influence the development of chronic diseases. Elucidating whether, and in what manner, specific genes alter fish and long-chain omega-3 fatty acid consumption would have implications for understanding influences on variation in fish intake within populations and the biology of partiality to foods. Furthermore, identification of such variants could also inform the development of personalized nutrition—dietary recommendations based on genetic preferences for consumption.
As has been seen with other characteristics such as physiologic risk factors, genome-wide association (GWA) studies may lead to discovery of novel genes and biologic pathways that influence the individual characteristic of interest. Although such studies have been performed for major macronutrients (e.g., fat, carbohydrate, protein) [5,6], few analyses have been done for specific foods[7], whose intakes may be influenced by complex characteristics of tastes, textures, aromas, and nutrient contents. The ability to undertake food-specific genetic analyses has been limited by the modest sample sizes of individual cohorts having both dietary and genetic information and the potential lack of reproducibility of genetic findings discovered in any single cohort.
We therefore performed a collaborative investigation to estimate heritability of and assess how common genetic variation relates to dietary consumption of both fish and long-chain omega-3 fatty acids (eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)) as part of the of the CHARGE (Cohorts for Heart and Aging Research in Genomic Epidemiology) Consortium Nutrition Working Group, bringing together investigators and data from 17 US and European population-based cohort studies totaling 86,467 participants of European descent.
Subjects and methods
Cohorts
The present work was a collaboration among 17 US and European population-based cohort studies participating in the Nutrition Working Group of the CHARGE Consortium (S1 Table). These included the Atherosclerosis Risk in Communities Study (ARIC); Cardiovascular Health Study (CHS); Dietary, Lifestyle, and Genetic Determinants of Obesity and Metabolic Syndrome (DILGOM); Estonian Study; Family Heart Study (FamHS); Framingham Heart Study (FHS); Helsinki Birth Cohort Study (HBCS); Health 2000 survey (H2000); Health, Aging, and Body Composition (HealthABC) Study; Health Professionals Follow-up Study (HPFS); Invecchiare [Aging] in Chianti Area (InCHIANTI); Multi-Ethnic Study of Atherosclerosis (MESA); Nurses’ Health Study (NHS); Rotterdam Study; The Hellenic Study of Interactions between SNPs and Eating in Atherosclerosis Susceptibility (THESIAS); Women’s Genome and Health study (WGHS); and Young Finns Study (YFS). Additional details on these cohorts have been published previously [5,6] and are provided in S1 Table. All persons studied were of European descent, consented to genetic research, and provided written informed consent. For each study, examination protocols were approved by local institutional review boards at Johns Hopkins University (ARIC, MESA), University of Washington (CHS), Epidemiology and Public Health of the Hospital District of Helsinki and Uusimaa (DILGOM), Washington University (FamHS), Boston University (FHS), University of Pittsburgh (HealthABC), Harvard University (HPFS, NHS), National Public Health Institute of Finland (HBCS), Epidemiology and Public Health of the Hospital District of Helsinki and Uusimaa (H2000), Italian National Institute of Research and Care of Aging (InCHIANTI), Erasmus Medical Center and The Netherlands Ministry of Health, Welfare and Sports (Rotterdam), Harokopio University (THESIAS), Brigham and Women’s Hospital (WGHS), University of Helsinki (YFS), and procedures were in accordance with the ethical standards of the responsible institutional or regional committee on human subject research.
Assessment of fish and omega-3 fatty acid consumption
Usual dietary intake was assessed in each cohort using detailed food frequency questionnaires designed to capture the dietary habits of the population under study (S2 Table). Typically, participants were asked to indicate how often, on average, they had consumed various foods and beverages over the past year according to multiple frequency categories (e.g., 9 categories ranging from <1/month to 6+/day), with usual portion sizes specified on the questionnaire or by the participant. Fish intake was generally assessed using multiple questions, such as on consumption of tuna fish; dark meat fish such as salmon or sardines; other white fish; shellfish; and fried fish or fish sandwiches. For each question, the midpoint of each frequency category was used to estimate usual intake which was then multiplied by the specified portion size; these intakes were summed across all questions on fish. For this analysis, we standardized fish consumption in each cohort to 100g servings/day. In 12 cohorts, total dietary consumption of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) was estimated by linking the dietary assessment tool to a food composition table specific to the cohort (e.g., the USDA food composition database in the US). For each of the types of foods consumed, the frequency and average portion size were multipled by the content of EPA/DHA in the food. The total was calculated by summing across all foods in the questionnaire. For cohorts that included nutrients from supplements, the portion of EPA+DHA from supplements was excluded from our analysis.
Heritability estimates
To evaluate potential heritability of fish and EPA+DHA consumption, we estimated heritability using family-based methods in two family-based cohorts (FamHS and FHS) using the variance components method in Sequential Oligogenic Linkage Analysis Routines (SOLAR; Texas Biomedical Research Institute; San Antonio, TX), and adjusting for age and sex. Briefly, heritability is calculated using a maximum likelihood method using the ratio of the genetic variance to total phenotypic variance.[8]
Genotyping and analysis
Genome-wide genotyping was conducted in each cohort using Affymetrix or Illumina platforms. Each study performed quality control for genotyped single nucleotide polymorphisms (SNPs) based on minor allele frequency (MAF), call rate, and departure from Hardy-Weinberg equilibrium (S3 Table). Phased haplotypes from HapMap CEU were used to impute ~2.5 million autosomal SNPs using a Hidden Markov Model algorithm implemented in MACH, IMPUTE, or BimBam. Study-specific GWA analyses were conducted within each cohort using genotyped and imputed SNP dosages assuming an additive genetic model. Fish and EPA+DHA consumption were separately evaluated as the dependent variable using linear regression with robust standard error, adjusted for age, sex, energy intake (kcal/d), study-specific centers where applicable, and population stratification principal components when the cohort lambda was >1. SNPs with low MAF (<1%), low imputation quality (MACH: R2<0.3; or IMPUTE: proper info <0.4), were excluded. Quality control for cohort-level GWAS results was performed to ensure correct specification of the minor allele and agreement in frequencies with the reference population (HapMap CEU), consistent distribution of effect sizes and standard error, and examination of QQ plots to assess any large inflation of test statistics. Results across studies were combined using fixed-effect meta-analysis with inverse variance weights (METAL software)[9]. The association results from individual studies as well as meta-analyses were adjusted for genomic control. To explore potential heterogeneity by demographic region, a meta-analysis within cohorts from Europe and USA was performed. Genome-wide significance was considered at the Bonferroni-corrected threshold of P<5x10-8. Statistical power to detect a true association at various effect sizes (heritability) was calculated using GWAPower software for the analysis of 1 million independent SNPs for both fish and EPA+DHA (Feng S, 2011 BMC Genetics), assuming linkage disequilibrium (r2) of 0.5 between a SNP and putative causal variant and 10% variance explained by three covariates.
Exploratory analysis of plasma phospholipid EPA and DHA
Result from genome-wide association analyses of circulating EPA and DHA are publically available (http://faculty.washington.edu/rozenl/files/) [10]. These databases were mined to test whether the top SNPs from the fish and EPA+DHA intake GWAS are associated with circulating levels of EPA and DHA.
Results
The 17 cohorts were from the US, Estonia, Finland, Greece, Italy, and the Netherlands and included 86,467 participants with information on fish consumption and 62,265 with information on EPA+DHA consumption. Across participating cohorts, mean fish consumption ranged from 0.19 servings/day (FHS) to 0.75 servings/day (THISEAS) (Table 1). Mean intake of EPA+DHA consumption ranged from 89 (Rotterdam) to 563 (HBCS) mg/d and was generally consistent with findings on fish intake, except in THISEAS (Greece) which had relatively higher intakes of fish than EPA+DHA, suggesting predominant consumption of white (non-oily) fish. In general, participants in European cohorts had higher fish consumption than those in US cohorts.
Table 1. Characteristics of the cohorts included in this analysis of the genetics of fish consumption.
| Study | Maximum N |
Age | % Female | Total Fish Intake (serv/day) Median (5-95th%) | Dietary EPA+DHA (mg/d) Median (5-95th%) | ||
|---|---|---|---|---|---|---|---|
| ARIC | 9557 | 54.3 ± 5.7 | 53 | 0.21 | (0.0–0.9) | 180 | (10–730) |
| CHS | 3190 | 72.3 ± 5.4 | 61 | 0.29 | (0.1–0.8) | 191 | (27–569) |
| DILGOM_METABO | 3467 | 51.5 ± 13.4 | 55 | 0.42 | (0.1–1.3) | 431 | (119–1277) |
| DILGOM_GWA | 604 | 52.4 ± 13.5 | 52 | 0.44 | (0.1–1.2) | 444 | (107–1233) |
| ESTONIAN Study | 9920 | 48.8 ± 20.1 | 53 | 0.21 | (0.0–0.6) | - | - |
| FamHS | 3640 | 52.2 ± 13.7 | 53 | 0.14 | (0.0–0.7) | 170 | (1–680) |
| FHS | 7044 | 47.3 ± 11.8 | 54 | 0.13 | (0.0–0.6) | 200 | (40–640) |
| Health ABC | 1494 | 74.8 ± 2.9 | 48 | 0.20 | (0.2–0.3) | - | - |
| Health 2000 | 1935 | 50.5 ± 10.9 | 51 | 0.39 | (0.1–1.1) | 505 | (102–1477) |
| HBCS | 1701 | 61.5 ± 2.9 | 57 | 0.44 | (0.1–1.3) | 563 | (145–1895) |
| HPFS | 4133 | 58.6 ± 8.7 | 0 | 0.29 | (0.1–0.9) | - | - |
| InCHIANTI | 1194 | 68.3 ± 15.4 | 55 | 0.19 | (0.0–0.5) | - | - |
| MESA | 2305 | 62.7 ± 10.2 | 52 | 0.17 | (0.0–0.7) | 100 | (20–300) |
| NHS | 6776 | 54.4 ± 6.7 | 100 | 0.28 | (0.1–0.8) | - | - |
| Rotterdam | 4606 | 67.6 ± 7.7 | 59 | 0.07 | (0.0–0.5) | 89 | (8–443) |
| THISEAS | 395 | 59.4 ± 13.0 | 41 | 0.59 | (0.0–2.0) | 137 | (4–479) |
| WGHS | 22691 | 54.7 ± 7.1 | 100 | 0.20 | (0.0–0.7) | 150 | (30–470) |
| YFS | 1815 | 37.8 ± 5.0 | 56 | 0.34 | (0.1–0.9) | 357 | (92–902) |
Abbreviations: Atherosclerosis Risk in Communities Study (ARIC); Cardiovascular Health Study (CHS); Dietary, Lifestyle, and Genetic Determinants of Obesity and Metabolic Syndrome (DILGOM); Estonian Study; Family Heart Study (FamHS); Framingham Heart Study (FHS); Helsinki Birth Cohort Study (HBCS); Health 2000 survey (H2000); Health, Aging, and Body Composition (HealthABC) Study; Health Professionals Follow-up Study (HPFS); Invecchiare [Aging] in Chianti Area (InCHIANTI); Multi-Ethnic Study of Atherosclerosis (MESA); Nurses’ Health Study (NHS); Rotterdam Study; The Hellenic Study of Interactions between SNPs and Eating in Atherosclerosis Susceptibility (THESIAS); Women’s Genome and Health study (WGHS); and Young Finns Study (YFS).
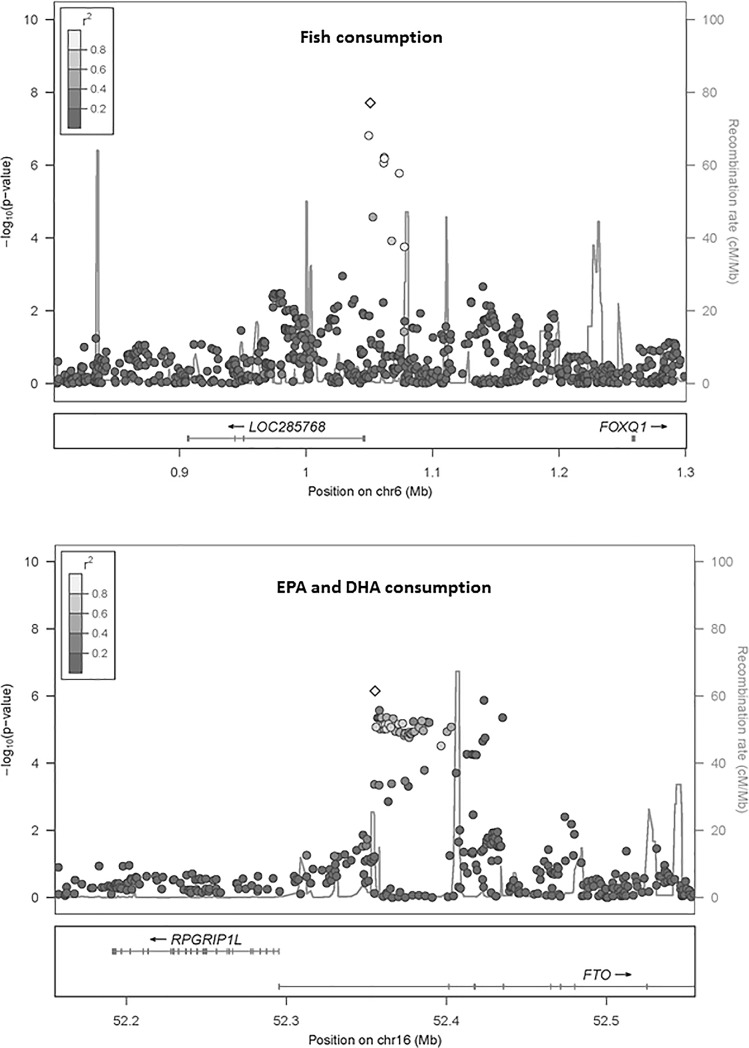
The heritability estimates for fish intake were 0.13±0.03 (FamHS) and 0.24±0.02 (FHS); and for EPA+DHA intake, 0.12±0.03 (FamHS) and 0.22±0.02 (FHS). In GWA meta-analyses of fish (17 cohorts) and EPA+DHA (11 cohorts) consumption, the genomic control lambda values were 1.07 and 0.99 respectively (S1 and S2 Figs). A genome-wide significant association was observed for fish intake on chromosome 6 for rs9502823 (Table 2). The minor allele (FreqA = 0.015) was associated with 0.029 servings/day lower fish consumption (P = 1.96x10-8). This SNP was mapped to LOC285768 gene of unknown function (Fig 1, top panel); and was not identified in NHGRI-EBI GWAS catalogue (http://www.ebi.ac.uk/gwas/, search on Feb 23, 2017). The second top hit was rs17396472 on chromosome 3, not achieving genome-wide statistical significance (P = 5.62x10-8).
Table 2. The most significant associations from genome-wide association meta-analysis of fish and EPA+DHA consumption.
| Trait | SNPID | Chr | Position | N | Effect/ Non-effect | Freq (Effect) | Effect | StdErr | P-value |
|---|---|---|---|---|---|---|---|---|---|
| Fish Intake | rs9502823 | 6 | 1050674 | 71910 | A/G | 0.02 | -0.029 | 0.005 | 1.96E-08 |
| (serving/day) | rs17396472 | 3 | 68469758 | 63102 | A/T | 0.98 | 0.031 | 0.005 | 5.62E-08 |
| rs1860343 | 12 | 4583970 | 78153 | T/C | 0.52 | -0.006 | 0.001 | 4.30E-07 | |
| rs1562806 | 15 | 35038800 | 61909 | T/C | 0.94 | 0.022 | 0.004 | 1.41E-06 | |
| rs16834168 | 1 | 150754770 | 77898 | A/G | 0.02 | -0.020 | 0.004 | 1.58E-06 | |
| EPA+DHA | rs11877506 | 18 | 49176846 | 52909 | A/G | 0.96 | 16.313 | 3.091 | 1.18E-07 |
| (mg/day) | rs2456163 | 19 | 1427432 | 36937 | T/C | 0.04 | -15.560 | 3.087 | 4.20E-07 |
| rs7476409 | 10 | 24809042 | 52909 | T/C | 0.05 | -17.907 | 3.589 | 5.50E-07 | |
| rs7206790 | 16 | 52355409 | 56099 | C/G | 0.55 | -7.000 | 1.420 | 7.44E-07 |
Fig 1. Regional association plot.
The figures display–log10 p-values for SNPs within the locus of highest significance for the analysis of fish consumption (top panel) and eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) consumption (bottom panel). The degree of linkage disequilibrium (r2) is displayed in gradients of gray from dark (low linkage) to light (high linkage).
No genome-wide significant association was observed for EPA+DHA consumption (S1 and S2 Figs). The top association for EPA+DHA consumption was observed for rs11877506 (P = 1.18x10-7) (Table 2). Additionally, rs7206790 in the obesity-associated FTO gene was among the top SNPs for EPA+DHA intake: the body mass index-raising G allele was associated with 7mg/day greater EPA+DHA intake (Fig 1, bottom panel; P = 7.44x10-7).
To obtain more information on the locus associated with fish consumption on chromosome 6, we investigated data from the ENCODE project. Using CEU 1000genomes data, we calculated the LD within the region 250kb upstream and downstream from rs9502823. Mapping the SNPs found in the rs9502823 LD block to ENCODE regulatory regions, we identified rs72838923 (in complete LD with rs9502823) as a functional candidate. rs72838923 falls within a experimentally determined H3k27Ac region, identified in several cell types in ENCODE. H3k27Ac regions are thought to be markers of active enhancer activity. In addition, rs72838923 falls within DNAse Hypersensitivity Peak which were identified experimentally across 65 cell types from the ENCODE project. There is further evidence of transcription factor binding sites for FOXA1, among others in this region, from ENCODE CHIP-Seq experiments. In addition, mapping rs72838923 on the UCSC genome browser suggested that this SNP is found within a region of conservation across mammals.
Due to the varying ranges of average fish consumption in US versus European studies, we performed exploratory subgroup GWA meta-analysis stratified by geographic location. No significant associations were identified in USA nor European studies (S3 Fig) for fish or EPA+DHA consumption (S4 Fig).
A prior consortium analysis including several of these same cohorts reported on genome-wide association of SNP variants with plasma phospholipid EPA and DHA, the concentrations of which are determined by both dietary intake and endogenous metabolism regulation.[11] In exploratory analysis, we evaluated whether the top 5 hits for fish consumption and the top 4 hits for estimated dietary EPA+DHA consumption identified in the present analysis were associated with plasma phospholipid concentrations of EPA or DHA in that prior analysis [10], adjusting for multiple comparisons (9 SNPs x 2 fatty acids = Bonferroni-corrected alpha of 0.05/18 = 0.0028). No significant associations were identified (S4 Table).
Discussion
In this large GWA meta-analysis of 17 US and European cohorts totaling 86,467 participants, we found evidence that common genetic variation may be associated with consumption of fish. We found no genome-wide significant association of common variants with EPA+DHA intake. While the sample size for the analysis of EPA+DHA was smaller than the fish intake analysis, with 62,265 individuals the analysis had 95% power to detect an effect size (heritability) of 0.08%.
We identified one locus on chromosome 6 in association with fish consumption. The SNP was mapped to LOC285768 with unknown function. The next closest gene is forkhead box Q1 (FOXQ1) which is a member of the cancer-associated forkhead-box (FOX) gene family [12]. Our evaluation of data from ENCODE, taken together, identified a functional candidate, rs72838923, that appears to lie within a transcriptionally active region of the genome. While the association was statistically significant, the magnitude of effect was small, with the minor allele being associated with a difference of 0.03 servings/day or approximately 1 serving/month of fish. This finding is more likely to be relevant for understanding the biology of food preferences than for influencing clinical outcomes, although even small differences in fish consumption, over a lifetime, could influence health. The nonsignificant top associations identified in chromosomes 1, 3, and 12 each represent intragenic regions of genes highly expressed in the brain, but these associations did not achieve genome-wide significance.
In heritability analyses, we found evidence for modest heritability of fish (0.13 to 0.24) and EPA+DHA (0.12 to 0.22). Our GWA results identified one locus in association with fish intake that cannot fully account for this observed heritability, suggesting that observed heritability might be due to remarkably small effects across a large number of SNPs, other types of genetic variation such as copy number variants, epigenetic modifications, or multiple unobserved genetic interactions with unknown environmental factors. This challenge of “missing” or unaccounted for heritability is a frequent finding in GWA analyses of common diseases and traits [13]. Heritability analyses may overestimate heritability due to unmeasured shared environmental influences, for example from in utero/placental influences through childhood and adult life. In this light, our heritability findings are lower than those previously reported [4] and represent an additional important new contribution. Our findings support the need for future investigations of the possible explanations for the modest but as yet missing heritability of fish and EPA+DHA consumption.
This investigation had several strengths. Our pooling of multiple large, well-established cohorts provided a very large sample of participants for investigating our research questions. Our post-hoc power calculations demonstrate 95% statistical power to detect a genetic variant associated with an effect size of 0.05% for fish consumption and 0.08% for EPA+DHA consumption. We adjusted for total reported energy intake, which helps to address any systematic over- or under-reporting by individuals and also real differences in total food consumed (i.e., due to differences in age, sex, body size, or physical activity), facilitating evaluation of dietary composition. All the studies in the meta-analysis used comparable dietary assessment tools that were appropriate for the population under study, providing the highest quality data that can be reasonably collected across multiple large epidemiological studies.
Limitations should be considered. While dietary intakes assessed by food frequency questionnaire represent a reasonably valid method to collect data on usual dietary habits in large populations [14], such data also include measurement error, which could limit the ability to detect true associations. However, many validation studies have demonstrated that fish and EPA+DHA consumption are measured reasonably well by food frequency questionnaires, whether compared with multiple diet records or with objective circulating or tissue biomarkers [15,16,17,18]. Indeed, because biomarker levels also represent imperfect measures of “true” habitual consumption with uncorrelated errors compared to questionnaire estimates, the actual correlations of estimated fish or EPA+DHA consumption with “true” consumption are likely much higher, in the range of 0.8 or more. Compared with a candidate gene approach, GWA has lower statistical power to detect small genetic effects. Yet, candidate gene approaches for evaluating fish consumption would be strongly limited by imperfect knowledge of which genes affect known systems and biologic processes related to food preferences and, even more so, which genes may affect currently unknown systems and biologic influences on food preferences.
In summary, this large pooling project across 17 established cohorts identified modest heritability of fish and omega-3 fatty acid consumption and one genetic locus associated with fish consumption. These findings suggest that genetic variation may have small effects on fish consumption and, by extension, that other modifiable factors–for example, childhood diet, culture, education, income, and local availability–are the main determinants of the remarkable differences in fish consumption within and across populations, representing targets for increasing fish intake among all individuals.
Supporting information
(TIFF)
Genome-wide association meta-analysis results of fish (A) and eicosapentanoic acid and docasahexanoic acid (B) with ~2.5 million SNPs graphed by chromosome position and–log10 p-value.
(TIFF)
Genome-wide association meta-analysis results of fish consumption in European cohorts (A) and cohorts from the United States (B) for ~2.5 million SNPs graphed by chromosome position and–log10 p-value.
(TIFF)
Genome-wide association meta-analysis results of EPA+DHA consumption in European cohorts (A) and cohorts from the United States (B) for ~2.5 million SNPs graphed by chromosome position and–log10 p-value.
(TIFF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
Disclaimer: The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the other funders.
The Atherosclerosis Risk in Communities (ARIC) study is carried out as a collaborative study supported by National Heart, Lung, and Blood Institute contracts N01‐HC‐55015, N01‐HC‐55016, N01‐HC‐55018, N01‐HC‐55019, N01‐HC‐55020, N01‐HC‐55021, N01‐HC‐55022, R01HL087641, R01HL59367 and R01HL086694; National Human Genome Research Institute contract U01HG004402; and National Institutes of Health contract HHSN268200625226C. The authors thank the staff and participants of the ARIC study for their important contributions. Infrastructure was partly supported by Grant Number UL1RR025005, a component of the National Institutes of Health and NIH Roadmap for Medical Research. Dr. Nettleton was supported by a K01 from the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases (5K01DK082729-02).
The Cardiovascular Health Study (CHS) research reported in this article was supported by contracts HHSN268201200036C, HHSN268200800007C, N01HC55222, N01HC85079, N01HC85080, N01HC85081, N01HC85082, N01HC85083, N01HC85086, and grant U01HL080295 from the National Heart, Lung, and Blood Institute, with additional contribution from the National Institute of Neurological Disorders and Stroke. Additional support was provided by R01AG023629 from the National Institute on Aging. A full list of principal CHS investigators and institutions can be found at CHS-NHLBI.org. DNA handling and genotyping was supported in part by National Center for Research Resources grant M01RR00069 to the Cedars‐Sinai General Clinical Research Center Genotyping core and National Institute of Diabetes and Digestive and Kidney Diseases grant DK063491 to the Southern California Diabetes Endocrinology Research Center. Dr. Mozaffarian was supported by R01 HL085710 from the National Heart, Lung, and Blood Institute.
The DILGOM study has been funded by the Academy of Finland (grant numbers 139635, 129494, 118065, 129322, 250207, 136895, 141005), the Orion-Farmos Research Foundation, the Finnish Foundation for Cardiovascular Research, and the Sigrid Jusélius Foundation. We thank the many colleagues who contributed to collection and phenotypic characterization of the clinical samples, and DNA extraction and genotyping of the data, especially Eija Hämäläinen, Minttu Jussila, Outi Törnwall, Päivi Laiho, and the staff from the Genotyping Facilities at the Wellcome Trust Sanger Institute. We would also like to acknowledge those who agreed to participate in the DILGOM study.
EGCUT received financing by FP7 grants (278913, 306031, 313010), Center of Excellence in Genomics (EXCEGEN) and University of Tartu (SP1GVARENG). We acknowledge EGCUT technical personnel, especially Mr V. Soo and S. Smit. Data analyzes were carried out in part in the High Performance Computing Center of University of Tartu.
The Family Heart Study (FamHS) work was supported in part by NIH grants 5R01 HL08770003, 5R01 HL08821502 (Michael A. Province) from NHLBI, and 5R01 DK07568102, 5R01 DK06833603 from NIDDK (Ingrid B. Borecki), and by the National Heart, Lung, and Blood Institute cooperative agreement grants U01 HL 67893, U01 HL67894, U01 HL67895, U01 HL67896, U01 HL67897, U01 HL67898, U01 HL67899, U01 HL67900, U01 HL67901, U01 HL67902, U01 HL56563, U01 HL56564, U01 HL56565, U01 HL56566, U01 HL56567, U01 HL56568, and U01 HL56569. The investigators thank the staff and participants of the FHS for their important contributions.
The Framingham Offspring Study and Framingham Third Generation Study (FHS) were conducted in part using data and resources from the Framingham Heart Study of the National Heart Lung and Blood Institute of the National Institutes of Health and Boston University School of Medicine. The analyses reflect intellectual input and resource development from the Framingham Heart Study investigators participating in the SNP Health Association Resource (SHARe) project. This work was partially supported by the National Heart, Lung and Blood Institute's Framingham Heart Study (Contract No. N01‐HC‐25195) and its contract with Affymetrix, Inc. for genotyping services (Contract No. N02‐HL‐6‐4278). A portion of this research utilized the Linux Cluster for Genetic Analysis (LinGA‐II) funded by the Robert Dawson Evans Endowment of the Department of Medicine at Boston University School of Medicine and Boston Medical Center. Also supported by National Institute for Diabetes and Digestive and Kidney Diseases (NIDDK) R01 DK078616 to Drs. Meigs, Dupuis and Florez, NIDDK K24 DK080140 to Dr. Meigs, and a Massachusetts General Hospital Physician Scientist Development Award and a Doris Duke Charitable Foundation Clinical Scientist Development Award to Dr. Florez. Dr. Hivert was supported by the Centre de Recherche Medicale de l’Universite de Sherbrooke (CRMUS) and a Canadian Institute of Health Research (CHIR) Fellowships Health Professional Award. Dr. Nicola McKeown is supported by the USDA agreement No. 58-1950-7-707.
We thank all study participants as well as everybody involved in the Helsinki Birth Cohort Study. Helsinki Birth Cohort Study has been supported by grants from the Academy of Finland, the Finnish Diabetes Research Society, Folkhälsan Research Foundation, Novo Nordisk Foundation, Finska Läkaresällskapet, Signe and Ane Gyllenberg Foundation, University of Helsinki, European Science Foundation (EUROSTRESS), Ministry of Education, Ahokas Foundation, Emil Aaltonen Foundation.
The Health, Aging and Body Composition (Health ABC) study was supported in part by the Intramural Research Program of the NIH, National Institute on Aging contracts N01AG62101, N01AG62103, and N01AG62106. The genome-wide association study was funded by NIA grant R01 AG032098 to Wake Forest University Health Sciences and genotyping services were provided by the Center for Inherited Disease Research (CIDR). CIDR is fully funded through a federal contract from the National Institutes of Health to The Johns Hopkins University, contract number HHSN268200782096C.
The use of Health 2000 data in this study has been financially supported by the Academy of Finland (grant 250207) and Orion-Farmos Research Foundation. The authors would like to thank the many colleagues who contributed to collection and phenotypic characterization of the clinical samples, and DNA extraction and genotyping of the data, especially Eija Hämäläinen, Minttu Jussila, Outi Törnwall, Päivi Laiho, and the staff from the Genotyping Facilities at the Wellcome Trust Sanger Institute. They would also like to acknowledge those who agreed to participate in the H2000 study.
Invecchiare in Chianti (aging in the Chianti area, InCHIANTI) study investigators thank the Intramural Research Program of the NIH, National Institute on Aging who are responsible for the InCHIANTI samples. Investigators also thank the InCHIANTI participants. The InCHIANTI study baseline (1998‐2000) was supported as a “targeted project” (ICS110.1/RF97.71) by the Italian Ministry of Health and in part by the U.S. National Institute on Aging (Contracts: 263 MD 9164 and 263 MD 821336).
The Multi-Ethnic Study of Atherosclerosis (MESA) and MESA SHARe project are conducted and supported by contracts N01-HC-95159 through N01-HC-95169 and RR-024156 from the National Heart, Lung, and Blood Institute (NHLBI). Funding for MESA SHARe genotyping was provided by NHLBI Contract N02‐HL‐6‐4278. The authors thank the participants of the MESA study, the Coordinating Center, MESA investigators, and study staff for their valuable contributions. A full list of participating MESA investigators and institutions can be found at http://www.mesa-nhlbi.org.
The NHS and HPFS are supported by the National Cancer Institute (NHS: UM1 CA186107, HPFS:UM1 CA167552) with additional support for genotyping. The NHS Breast Cancer GW scan was performed as part of the Cancer Genetic Markers of Susceptibility initiative of the NCI (R01CA40356, U01-CA98233). The NHS/HPFS type 2 diabetes GWAS (U01HG004399) is a component of a collaborative project that includes 13 other GWAS funded as part of the Gene Environment-Association Studies (GENEVA) under the NIH Genes, Environment and Health Initiative (GEI) (U01HG004738, U01HG004422, U01HG004402, U01HG004729, U01HG004726, 01HG004735, U01HG004415, U01HG004436, U01HG004423, U01HG004728, AHG006033) with additional support from individual NIH Institutes (NIDCR: U01DE018993, U01DE018903; NIAAA: U10AA008401, NIDA: P01CA089392, 01DA013423; NCI: CA63464, CA54281, CA136792, Z01CP010200). Assistance with phenotype harmonization and genotype cleaning, as well as with general study coordination, was provided by the GENEVA Coordinating Center (U01HG004446).Genotyping was performed at the Broad Institute of MIT and Harvard, with funding support from the NIH GEI (U01HG04424), and Johns Hopkins University Center for Inherited Disease Research, with support from the NIH GEI (U01HG004438) and the NIH contract "High throughput genotyping for studying the genetic contributions to human disease”(HHSN268200782096C). The NHS/HPFS CHD GWAS was supported by Merck/Rosetta Research Laboratories, North Wales, PA. The NHS/HPFS Kidney GWAS was supported by NIDDK: 5P01DK070756.
The generation and management of GWAS genotype data for the Rotterdam Study is supported by the Netherlands Organization of Scientific Research NWO Investments (nr. 175.010.2005.011, 911‐03‐012), the Research Institute for Diseases in the Elderly (014‐93‐015; RIDE2),EUROSPAN (European Special Populations Research Network;LSHG‐CT‐2006‐01947),the Netherlands Organization for Scientific Research (Pionier, 047.016.009, 047.017.043;050‐060‐810), Erasmus Medical Center and the Centre for Medical Systems Biology (CMSB I and II and Grand; National Genomics Initiative) of the Netherlands Genomics Initiative (NGI); The Rotterdam Study is further funded by Erasmus Medical Center and Erasmus University, Rotterdam, Netherlands Organization for the Health Research and Development (ZonMw), the Research Institute for Diseases in the Elderly (RIDE), the Ministry of Education, Culture and Science, the Ministry for Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam. We thank Pascal Arp, Mila Jhamai, Dr Michael Moorhouse, Marijn Verkerk, and Sander Bervoets for their help in creating the GWAS database. The authors are grateful to the study participants, the staff from the Rotterdam Study and the participating general practitioners and pharmacists.
The Hellenic study of Interactions between SNPs and Eating in Atherosclerosis Susceptibility (THISEAS) study thanks the Genotyping Facility at the Wellcome Trust Sanger Institute for typing the THISEAS samples and in particular Sarah Edkins and Cordelia Langford. PD is supported by the Wellcome Trust.
The WGHS is supported by HL043851 and HL080467 from the National Heart, Lung, and Blood Institute and CA047988 from the National Cancer Institute, the Donald W. Reynolds Foundation and the Fondation Leducq, with collaborative scientific support and funding for genotyping provided by Amgen.
The Young Finns Study has been financially supported by the Academy of Finland: grants 126925, 121584, 124282, 129378 (Salve), 117787 (Gendi), and 41071 (Skidi), the Social Insurance Institution of Finland, Kuopio, Tampere and Turku University Hospital Medical Funds (grant 9M048 for TeLeht), Juho Vainio Foundation, Paavo Nurmi Foundation, Finnish Foundation of Cardiovascular Research and Finnish Cultural Foundation, Tampere Tuberculosis Foundation and Emil Aaltonen Foundation (T.L). The expert technical assistance in the statistical analyses by Irina Lisinen, Ville Aalto and Mika Helminen are gratefully acknowledged.
Abbreviations
- EPA
eicosapentaenoic acid
- DHA
docosahexaenoic acid
- LD
linkage disequilibrium
- GWAS
genome-wide association study
Data Availability
The meta-analysis results from this study are available at dbGAP (accession number phs000930).
Funding Statement
The Atherosclerosis Risk in Communities (ARIC) study is carried out as a collaborative study supported by National Heart, Lung, and Blood Institute contracts N01‐HC‐55015, N01‐HC‐55016, N01‐HC‐55018, N01‐HC‐55019, N01‐HC‐55020, N01‐HC‐55021, N01‐HC‐55022, R01HL087641, R01HL59367 and R01HL086694; National Human Genome Research Institute contract U01HG004402; and National Institutes of Health contract HHSN268200625226C. The authors thank the staff and participants of the ARIC study for their important contributions. Infrastructure was partly supported by Grant Number UL1RR025005, a component of the National Institutes of Health and NIH Roadmap for Medical Research. Dr. Nettleton was supported by a K01 from the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases (5K01DK082729-02). The Cardiovascular Health Study (CHS) research reported in this article was supported by contracts HHSN268201200036C, HHSN268200800007C, N01HC55222, N01HC85079, N01HC85080, N01HC85081, N01HC85082, N01HC85083, N01HC85086, and grant U01HL080295 from the National Heart, Lung, and Blood Institute, with additional contribution from the National Institute of Neurological Disorders and Stroke. Additional support was provided by R01AG023629 from the National Institute on Aging. A full list of principal CHS investigators and institutions can be found at CHS-NHLBI.org. DNA handling and genotyping was supported in part by National Center for Research Resources grant M01RR00069 to the Cedars‐Sinai General Clinical Research Center Genotyping core and National Institute of Diabetes and Digestive and Kidney Diseases grant DK063491 to the Southern California Diabetes Endocrinology Research Center. Dr. Mozaffarian was supported by R01 HL085710 from the National Heart, Lung, and Blood Institute. The DILGOM study has been funded by the Academy of Finland (grant numbers 139635, 129494, 118065, 129322, 250207, 136895, 141005), the Orion-Farmos Research Foundation, the Finnish Foundation for Cardiovascular Research, and the Sigrid Jusélius Foundation. We thank the many colleagues who contributed to collection and phenotypic characterization of the clinical samples, and DNA extraction and genotyping of the data, especially Eija Hämäläinen, Minttu Jussila, Outi Törnwall, Päivi Laiho, and the staff from the Genotyping Facilities at the Wellcome Trust Sanger Institute. We would also like to acknowledge those who agreed to participate in the DILGOM study. EGCUT received financing by FP7 grants (278913, 306031, 313010), Center of Excellence in Genomics (EXCEGEN) and University of Tartu (SP1GVARENG). We acknowledge EGCUT technical personnel, especially Mr V. Soo and S. Smit. Data analyzes were carried out in part in the High Performance Computing Center of University of Tartu. The Family Heart Study (FamHS) work was supported in part by NIH grants 5R01 HL08770003, 5R01 HL08821502 (Michael A. Province) from NHLBI, and 5R01 DK07568102, 5R01 DK06833603 from NIDDK (Ingrid B. Borecki), and by the National Heart, Lung, and Blood Institute cooperative agreement grants U01 HL 67893, U01 HL67894, U01 HL67895, U01 HL67896, U01 HL67897, U01 HL67898, U01 HL67899, U01 HL67900, U01 HL67901, U01 HL67902, U01 HL56563, U01 HL56564, U01 HL56565, U01 HL56566, U01 HL56567, U01 HL56568, and U01 HL56569. The investigators thank the staff and participants of the FHS for their important contributions. The Framingham Offspring Study and Framingham Third Generation Study (FHS) were conducted in part using data and resources from the Framingham Heart Study of the National Heart Lung and Blood Institute of the National Institutes of Health and Boston University School of Medicine. The analyses reflect intellectual input and resource development from the Framingham Heart Study investigators participating in the SNP Health Association Resource (SHARe) project. This work was partially supported by the National Heart, Lung and Blood Institute's Framingham Heart Study (Contract No. N01‐HC‐25195) and its contract with Affymetrix, Inc. for genotyping services (Contract No. N02‐HL‐6‐4278). A portion of this research utilized the Linux Cluster for Genetic Analysis (LinGA‐II) funded by the Robert Dawson Evans Endowment of the Department of Medicine at Boston University School of Medicine and Boston Medical Center. Also supported by National Institute for Diabetes and Digestive and Kidney Diseases (NIDDK) R01 DK078616 to Drs. Meigs, Dupuis and Florez, NIDDK K24 DK080140 to Dr. Meigs, and a Massachusetts General Hospital Physician Scientist Development Award and a Doris Duke Charitable Foundation Clinical Scientist Development Award to Dr. Florez. Dr. Hivert was supported by the Centre de Recherche Medicale de l’Universite de Sherbrooke (CRMUS) and a Canadian Institute of Health Research (CHIR) Fellowships Health Professional Award. Dr. Nicola McKeown is supported by the USDA agreement No. 58-1950-7-707. We thank all study participants as well as everybody involved in the Helsinki Birth Cohort Study. Helsinki Birth Cohort Study has been supported by grants from the Academy of Finland, the Finnish Diabetes Research Society, Folkhälsan Research Foundation, Novo Nordisk Foundation, Finska Läkaresällskapet, Signe and Ane Gyllenberg Foundation, University of Helsinki, European Science Foundation (EUROSTRESS), Ministry of Education, Ahokas Foundation, Emil Aaltonen Foundation. The Health, Aging and Body Composition (Health ABC) study was supported in part by the Intramural Research Program of the NIH, National Institute on Aging contracts N01AG62101, N01AG62103, and N01AG62106. The genome-wide association study was funded by NIA grant R01 AG032098 to Wake Forest University Health Sciences and genotyping services were provided by the Center for Inherited Disease Research (CIDR). CIDR is fully funded through a federal contract from the National Institutes of Health to The Johns Hopkins University, contract number HHSN268200782096C. The use of Health 2000 data in this study has been financially supported by the Academy of Finland (grant 250207) and Orion-Farmos Research Foundation. The authors would like to thank the many colleagues who contributed to collection and phenotypic characterization of the clinical samples, and DNA extraction and genotyping of the data, especially Eija Hämäläinen, Minttu Jussila, Outi Törnwall, Päivi Laiho, and the staff from the Genotyping Facilities at the Wellcome Trust Sanger Institute. They would also like to acknowledge those who agreed to participate in the H2000 study. Invecchiare in Chianti (aging in the Chianti area, InCHIANTI) study investigators thank the Intramural Research Program of the NIH, National Institute on Aging who are responsible for the InCHIANTI samples. Investigators also thank the InCHIANTI participants. The InCHIANTI study baseline (1998‐2000) was supported as a “targeted project” (ICS110.1/RF97.71) by the Italian Ministry of Health and in part by the U.S. National Institute on Aging (Contracts: 263 MD 9164 and 263 MD 821336). The Multi-Ethnic Study of Atherosclerosis (MESA) and MESA SHARe project are conducted and supported by contracts N01-HC-95159 through N01-HC-95169 and RR-024156 from the National Heart, Lung, and Blood Institute (NHLBI). Funding for MESA SHARe genotyping was provided by NHLBI Contract N02‐HL‐6‐4278. The authors thank the participants of the MESA study, the Coordinating Center, MESA investigators, and study staff for their valuable contributions. A full list of participating MESA investigators and institutions can be found at http://www.mesa-nhlbi.org. The NHS and HPFS are supported by the National Cancer Institute (NHS: UM1 CA186107, HPFS:UM1 CA167552) with additional support for genotyping. The NHS Breast Cancer GW scan was performed as part of the Cancer Genetic Markers of Susceptibility initiative of the NCI (R01CA40356, U01-CA98233). The NHS/HPFS type 2 diabetes GWAS (U01HG004399) is a component of a collaborative project that includes 13 other GWAS funded as part of the Gene Environment-Association Studies (GENEVA) under the NIH Genes, Environment and Health Initiative (GEI) (U01HG004738, U01HG004422, U01HG004402, U01HG004729, U01HG004726, 01HG004735, U01HG004415, U01HG004436, U01HG004423, U01HG004728, AHG006033) with additional support from individual NIH Institutes (NIDCR: U01DE018993, U01DE018903; NIAAA: U10AA008401, NIDA: P01CA089392, 01DA013423; NCI: CA63464, CA54281, CA136792, Z01CP010200). Assistance with phenotype harmonization and genotype cleaning, as well as with general study coordination, was provided by the GENEVA Coordinating Center (U01HG004446).Genotyping was performed at the Broad Institute of MIT and Harvard, with funding support from the NIH GEI (U01HG04424), and Johns Hopkins University Center for Inherited Disease Research, with support from the NIH GEI (U01HG004438) and the NIH contract "High throughput genotyping for studying the genetic contributions to human disease”(HHSN268200782096C). The NHS/HPFS CHD GWAS was supported by Merck/Rosetta Research Laboratories, North Wales, PA. The NHS/HPFS Kidney GWAS was supported by NIDDK: 5P01DK070756. The generation and management of GWAS genotype data for the Rotterdam Study is supported by the Netherlands Organization of Scientific Research NWO Investments (nr. 175.010.2005.011, 911‐03‐012), the Research Institute for Diseases in the Elderly (014‐93‐015; RIDE2),EUROSPAN (European Special Populations Research Network;LSHG‐CT‐2006‐01947),the Netherlands Organization for Scientific Research (Pionier, 047.016.009, 047.017.043;050‐060‐810), Erasmus Medical Center and the Centre for Medical Systems Biology (CMSB I and II and Grand; National Genomics Initiative) of the Netherlands Genomics Initiative (NGI); The Rotterdam Study is further funded by Erasmus Medical Center and Erasmus University, Rotterdam, Netherlands Organization for the Health Research and Development (ZonMw), the Research Institute for Diseases in the Elderly (RIDE), the Ministry of Education, Culture and Science, the Ministry for Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam. We thank Pascal Arp, Mila Jhamai, Dr Michael Moorhouse, Marijn Verkerk, and Sander Bervoets for their help in creating the GWAS database. The authors are grateful to the study participants, the staff from the Rotterdam Study and the participating general practitioners and pharmacists. The Hellenic study of Interactions between SNPs and Eating in Atherosclerosis Susceptibility (THISEAS) study thanks the Genotyping Facility at the Wellcome Trust Sanger Institute for typing the THISEAS samples and in particular Sarah Edkins and Cordelia Langford. PD is supported by the Wellcome Trust. The WGHS is supported by HL043851 and HL080467 from the National Heart, Lung, and Blood Institute and CA047988 from the National Cancer Institute, the Donald W. Reynolds Foundation and the Fondation Leducq, with collaborative scientific support and funding for genotyping provided by Amgen. The Young Finns Study has been financially supported by the Academy of Finland: grants 126925, 121584, 124282, 129378 (Salve), 117787 (Gendi), and 41071 (Skidi), the Social Insurance Institution of Finland, Kuopio, Tampere and Turku University Hospital Medical Funds (grant 9M048 for TeLeht), Juho Vainio Foundation, Paavo Nurmi Foundation, Finnish Foundation of Cardiovascular Research and Finnish Cultural Foundation, Tampere Tuberculosis Foundation and Emil Aaltonen Foundation (T.L). The expert technical assistance in the statistical analyses by Irina Lisinen, Ville Aalto and Mika Helminen are gratefully acknowledged. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the other funders.
References
- 1.Mozaffarian D, Wu JH (2011) Omega-3 fatty acids and cardiovascular disease: effects on risk factors, molecular pathways, and clinical events. J Am Coll Cardiol 58: 2047–2067. doi: 10.1016/j.jacc.2011.06.063 [DOI] [PubMed] [Google Scholar]
- 2.Rizos EC, Ntzani EE, Bika E, Kostapanos MS, Elisaf MS (2012) Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: a systematic review and meta-analysis. JAMA 308: 1024–1033. doi: 10.1001/2012.jama.11374 [DOI] [PubMed] [Google Scholar]
- 3.Papanikolaou Y, Brooks J, Reider C, Fulgoni VL, 3rd (2014) U.S. adults are not meeting recommended levels for fish and omega-3 fatty acid intake: results of an analysis using observational data from NHANES 2003–2008. Nutr J 13: 31 doi: 10.1186/1475-2891-13-31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hasselbalch AL, Heitmann BL, Kyvik KO, Sorensen TI (2008) Studies of twins indicate that genetics influence dietary intake. J Nutr 138: 2406–2412. doi: 10.3945/jn.108.087668 [DOI] [PubMed] [Google Scholar]
- 5.Tanaka T, Ngwa JS, van Rooij FJ, Zillikens MC, Wojczynski MK, Frazier-Wood AC, et al. (2013) Genome-wide meta-analysis of observational studies shows common genetic variants associated with macronutrient intake. Am J Clin Nutr 97: 1395–1402. doi: 10.3945/ajcn.112.052183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chu AY, Workalemahu T, Paynter NP, Rose LM, Giulianini F, Tanaka T, et al. (2013) Novel locus including FGF21 is associated with dietary macronutrient intake. Hum Mol Genet 22: 1895–1902. doi: 10.1093/hmg/ddt032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Amin N, Byrne E, Johnson J, Chenevix-Trench G, Walter S, Nolte IM, et al. (2012) Genome-wide association analysis of coffee drinking suggests association with CYP1A1/CYP1A2 and NRCAM. Mol Psychiatry 17: 1116–1129. doi: 10.1038/mp.2011.101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Almasy L, Blangero J (1998) Multipoint quantitative-trait linkage analysis in general pedigrees. Am J Hum Genet 62: 1198–1211. doi: 10.1086/301844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Willer CJ, Li Y, Abecasis GR (2010) METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics (Oxford, England) 26: 2190–2191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lemaitre RN, Tanaka T, Tang W, Manichaikul A, Foy M, Kabagambe EK, et al. (2011) Genetic loci associated with plasma phospholipid n-3 fatty acids: a meta-analysis of genome-wide association studies from the CHARGE Consortium. PLoS genetics 7: e1002193 doi: 10.1371/journal.pgen.1002193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lemaitre RN, Tanaka T, Tang W, Manichaikul A, Foy M, Kabagambe EK, et al. (2011) Genetic loci associated with plasma phospholipid n-3 fatty acids: a meta-analysis of genome-wide association studies from the CHARGE Consortium. PLoS Genet 7: e1002193 doi: 10.1371/journal.pgen.1002193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Katoh M (2004) Human FOX gene family (Review). Int J Oncol 25: 1495–1500. [PubMed] [Google Scholar]
- 13.Zaitlen N, Kraft P (2012) Heritability in the genome-wide association era. Hum Genet 131: 1655–1664. doi: 10.1007/s00439-012-1199-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Willett W (1998) Nutritional Epidemiology. New York: Oxford University Press. [Google Scholar]
- 15.Salvini S, Hunter DJ, Sampson L, Stampfer MJ, Colditz GA, Rosner B, et al. (1989) Food-based validation of a dietary questionnaire: the effects of week-to-week variation in food consumption. Int J Epidemiol 18: 858–867. [DOI] [PubMed] [Google Scholar]
- 16.Kaaks R, Slimani N, Riboli E (1997) Pilot phase studies on the accuracy of dietary intake measurements in the EPIC project: overall evaluation of results. European Prospective Investigation into Cancer and Nutrition. Int J Epidemiol 26 Suppl 1: S26–36. [DOI] [PubMed] [Google Scholar]
- 17.Mozaffarian D, Lemaitre RN, Kuller LH, Burke GL, Tracy RP, Siscovick DS (2003) Cardiac benefits of fish consumption may depend on the type of fish meal consumed: the Cardiovascular Health Study. Circulation 107: 1372–1377. [DOI] [PubMed] [Google Scholar]
- 18.Serra-Majem L, Nissensohn M, Overby NC, Fekete K (2012) Dietary methods and biomarkers of omega 3 fatty acids: a systematic review. Br J Nutr 107 Suppl 2: S64–76. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(TIFF)
Genome-wide association meta-analysis results of fish (A) and eicosapentanoic acid and docasahexanoic acid (B) with ~2.5 million SNPs graphed by chromosome position and–log10 p-value.
(TIFF)
Genome-wide association meta-analysis results of fish consumption in European cohorts (A) and cohorts from the United States (B) for ~2.5 million SNPs graphed by chromosome position and–log10 p-value.
(TIFF)
Genome-wide association meta-analysis results of EPA+DHA consumption in European cohorts (A) and cohorts from the United States (B) for ~2.5 million SNPs graphed by chromosome position and–log10 p-value.
(TIFF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
The meta-analysis results from this study are available at dbGAP (accession number phs000930).