Abstract
Objective
Little is known about the long-term effects of renin–angiotensin system inhibitors (RASI) on cardiovascular events in patients after acute myocardial infarction (AMI) with ischaemic mitral regurgitation (IMR). The purpose of this study was to investigate the association of RASI with the incidence of adverse cardiac events in patients with or without IMR after AMI.
Methods
We reviewed charts of 1208 consecutive patients admitted with AMI who underwent emergency coronary angiography between 2000 and 2012. After excluding patients who died within 30 days, 551 patients were diagnosed to have mild or greater MR by transthoracic echocardiography (patients with IMR); the remaining 505 patients had no or trivial MR (non-IMR patients).
Results
Of the study patients, 395 (72%) patients with IMR and 403 (80%) non-IMR patients received RASI. Survival analysis showed that freedom from cardiac death and the composite of cardiac death and heart failure (HF) was significantly higher in patients with IMR receiving RASI than in those not receiving RASI (P<0.001 and P<0.001, respectively). Moreover, adjusted survival analysis using the inverse probability treatment weighting method showed a significant association of RASI therapy with reduced cardiac death (P=0.010) and the composite of cardiac death and HF (P=0.044) in patients with IMR. However, in non-IMR patients, there were no significant associations between RASI therapy and the outcome measures.
Conclusions
RASI therapy was associated with a lower incidence of adverse cardiac events in patients with IMR after AMI, but not in patients without IMR.
Keywords: mitral regurgitation, renin-angiotensin system, heart failure treatment
Key questions.
What is already known about this subject?
Renin–angiotensin system inhibitors (RASI) are useful in reducing mortality in patients with left ventricular dysfunction after acute myocardial infarction (AMI).
What does this study add?
Little is known about the long-term effect of RASI in patients with or without ischaemic mitral regurgitation (IMR) after AMI. This study investigated the effect of RASI with the long-term outcomes in patients with or without IMR after AMI in the reperfusion setting.
How might this impact on clinical practice?
Our results suggest that patients with IMR might receive clinical benefit from RASI therapy after AMI regardless of MR severity and left ventricular dysfunction. Introduction at a low dose and uptitration of RASI should always be considered even in patients with IMR who seem to be intolerant to RASI therapy.
Introduction
Mitral regurgitation (MR) complicates the course of acute myocardial infarctions (AMI) in 13% to 50% of patients.1 2 Postinfarction ischaemic MR (IMR) portends a poor prognosis during long-term follow-up and has been identified as an independent predictor of heart failure (HF) and reduced long-term survival.3–6 It has been recognised that vasodilator therapy such as nitroprusside could improve the severity of IMR by decreasing left ventricular (LV) after load afterload and increasing forward stroke volume.7 8
Previous studies have shown that ACE inhibitors (ACEIs) may improve MR severity in patients with IMR.9 10 In addition, a study in animal model demonstrated that long-term ACEI therapy prevented the worsening of IMR by inhibiting progression of LV dilatation.11 The favourable effects of ACEIs on mortality of patients with LV dysfunction after AMI are well established by previous large-scale randomised controlled trials.12–14 Also, angiotensin-receptor blockers (ARBs) were shown to be non-inferior to ACEIs in patients with previous MI or HF.15–17 However, the clinical outcome in the presence or absence of IMR has not been previously reported. Although guideline-directed medical therapy for HF including β-blockers and ACEIs/ARBs is recommended for all patients with LV dysfunction and IMR,18 little is known about the long-term effect of medical therapy in patients with IMR. Moreover, there is controversy whether ACEI improves clinical outcomes in patients with preserved LV ejection fraction (EF) after AMI,19 stable coronary artery disease.20 The purpose of this study was to investigate the association of rennin–angiotensin system inhibitor (RASI) with the long-term outcomes in patients with or without IMR after AMI in the reperfusion setting.
Methods
Patient characteristics
Between January 2000 and December 2012, 1208 consecutive patients were admitted to our hospital for AMI and underwent emergency coronary angiography. Patients who died within 30 days (n=66) and those who did not undergo transthoracic echocardiography during index hospitalisation (n=85) were excluded from the study. Thus, 551 patients diagnosed to have mild or greater MR (patients with IMR) and 505 patients diagnosed to have no or trivial MR (non-IMR patients) by transthoracic echocardiography were analysed. No patient had significant mitral valve prolapse as a cause of MR. A diagnosis of AMI was made if the patient fulfilled at least two of the following three criteria: (1) history of central chest pressure, pain or tightness lasting 30 min; (2) typical ECG changes (ST segment elevation ≥0.1 mV in ≥1 standard limb lead or ≥2 precordial leads, ST segment depression ≥0.1 mV in ≥2 leads, abnormal Q waves or T-wave inversion in two leads); and (3) an increase in serum creatine kinase (CK) levels of twice the upper normal limit. A total of 936 patients underwent primary percutaneous coronary intervention (PCI), and the remaining 120 patients were treated with coronary artery bypass grafting (CABG; n=53) or standard medical therapy including thrombolytic and antiplatelet/anticoagulant therapy (n=67).
Baseline characteristics were assessed retrospectively from the medical records. Hypertension was defined as systolic blood pressure (BP) ≥140 mm Hg or diastolic BP ≥90 mm Hg, or both. Diabetes mellitus was defined as a fasting glucose level ≥126 mg/dL, a non-fasting glucose level ≥200 mg/dL, an HbA1c level ≥6.5% and/or a previous medical diagnosis of diabetes. Dyslipidaemia was defined as low-density lipoprotein (LDL) cholesterol level ≥140 mg/dL, high-density lipoprotein (HDL) cholesterol level <40 mg/dL and/or a triglycerides level ≥150 mg/dL. Patients with hypoalbuminaemia (<35 g/L) and low body mass index (BMI<18.5) was defined as frail.21 22 Estimated glomerular filtration rate was assessed as previously reported.23
This study was approved by the Institutional Review Board of Kobe City Medical Center General Hospital. Waiver of informed consent was obtained given the nature of the study.
Echocardiographic evaluation
All patients who underwent echocardiography during index hospitalisation were identified, and the results of all echocardiogram were reviewed by the principal investigator without knowledge of the patients’ outcomes and histories. When echocardiography demonstrated that the main cause of MR was leaflet tethering by displacement of papillary muscles as a consequence of regional or global LV dysfunction (not the pathology of mitral valve apparatus including leaflets and chordae tendineae), we defined MR as IM. If patients had both leaflet tethering and degenerative changes such as leaflet thickening or mitral annular calcification, we classified these patients as having IMR. The severity of MR was evaluated semiquantitatively from the area of the regurgitant jet by colour Doppler as previously reported.4 MR was classified as absent or trivial, mild, and moderate or severe. EF was calculated by the biplane Simpson method. Diastolic function was evaluated by the deceleration time of the early mitral inflow.
Clinical follow-up
Cardiac death and the composite of cardiac death and HF were analysed. All patients were followed at the outpatient clinic every 4 to 8 weeks after discharge. Further follow-up was done by the referring physician. The long-term follow-up was performed by chart review and telephone contact. Of the 1056 patients, 538 underwent complete follow-up at 5 years. The median follow-up duration was 5.1 years (IQR 2.4–7.2 years). Cardiac death was defined as death related to AMI, arrhythmia, cardiogenic shock, end-stage HF or sudden death. HF was defined as new or worsening symptoms of dyspnoea and fatigue with the presence of pulmonary congestion or peripheral oedema which required hospitalisation. We reviewed all available data to ascertain the immediate cause of death. Only incident episodes of HF after the index MI were considered in the analysis of the association between MR and HF after MI.
Statistical analysis
Categorical variables are presented as number and percentage and compared by the χ2 test or Fisher’s exact test as appropriate. Continuous variables are presented as mean±SD and were compared by unpaired t tests. Survival analysis was performed by Kaplan-Meier analysis, and differences in survival between groups was examined with the log-rank test. Survival rates are expressed as percentage with mean±SE. Cox proportional hazards models were constructed to evaluate the risk of cardiac death or HF according to the presence or absence of RASI therapy. Variables included in the proportional hazards models were chosen from those known to be of clinical interest in post-MI risk stratification, with the goal of keeping the models as parsimonious as possible. Inverse probability of treatment weighted (IPTW) methods based on the propensity score was used to adjust for baseline differences between the IMR patients with and without RASI therapy. The included covariates were age, sex, coronary risk factors (hypertension, diabetes mellitus, dyslipidaemia and smoking), frailty, BMI (for non-IMR patients), history of prior MI, Killip classification, moderate or severe MR, anterior MI, revascularisation procedure (PCI or CABG), peak CK level, serum creatinine, β-blocker therapy and receiving statins. Data analyses were performed with SPSS software (V.22; SPSS) and R software (V.3.1.1).
Results
Patient characteristics and echocardiographic results in patients with IMR
A total of 551 patients were diagnosed as having mild or greater MR. Among these patients, 23 had both leaflet tethering and degenerative changes. There were no patients with mitral valve prolapse resulting from severe myxomatous degeneration with a redundancy of leaflets known as Barlow’s valve or chordal rupture. The median time interval between the index MI and the echocardiogram was 11 days. Of the 551 patients, 395 (72%) patients received RASI before hospital discharge. None of the patients were deprived of RASI therapy because of low BP (systolic BP <80 mm Hg). Baseline characteristics and echocardiographic results of the patients with and without RASI therapy are shown in table 1 and table 2, respectively. Patients without RASI therapy were older and more likely to have smaller body mass index, higher creatinine level, ST segment elevation MI, higher Killip classification on admission, CABG, moderate or severe MR, and less likely to be taking β-blockers or statins, compared with patients with RASI therapy. Prevalence of prior MI, peak CK level, LV end-diastolic volume and end-systolic volume were comparable between the two groups.
Table 1.
Baseline patient characteristics stratified by the presence or absence of RASI therapy in patients with IMR
| Characteristics | Unadjusted data | IPTW | ||
| RASI (+) (n=395) |
RASI (−) (n=156) |
P value | P value | |
| Age, years, mean±SD | 67±11 | 71±10 | <0.001 | 0.15 |
| Gender, male/female | 301/94 | 121/35 | 0.73 | 0.47 |
| Cardiovascular risk factors | ||||
| Hypertension, n (%) | 207 (52) | 82 (53) | 0.97 | 0.65 |
| Diabetes mellitus, n (%) | 130 (33) | 49 (31) | 0.73 | 0.52 |
| Dyslipidaemia, n (%) | 157 (40) | 54 (35) | 0.26 | 0.70 |
| Current smoker, n (%) | 129 (33) | 41 (26) | 0.14 | 0.84 |
| Frailty | 9 (2) | 963) | 0.04 | 0.71 |
| BMI, kg/m2, mean±SD | 23.7±3.2 | 22.9±3.3 | 0.01 | 0.50 |
| ST segment elevation MI, n (%) | 333 (84) | 117 (75) | 0.01 | 0.46 |
| Killip classification, n (%) | 1.2±0.6 | 1.5±1.0 | <0.001 | 0.85 |
| Prior MI, n (%) | 32 (8) | 12 (8) | 0.87 | 0.57 |
| Primary PCI, n (%) | 366 (93) | 123 (79) | <0.001 | 0.24 |
| CABG, n (%) | 8 (2) | 24 (15) | <0.001 | 0.79 |
| Peak CK, IU/L | 3119±2727 | 3037±3967 | 0.78 | 0.73 |
| Serum creatinine, mg/dL | 1.07±0.83 | 1.66±1.65 | <0.001 | 0.06 |
| eGFR, mL/min/1.73 m2 | 52±20 | 40±22 | <0.001 | 0.08 |
| ACEI, n (%) ARB, n (%) |
288 (73) 109 (27) |
0 (0) 0 (0) |
<0.001 <0.001 |
<0.001 <0.001 |
| β-blocker, n (%) | 324 (82) | 101 (65) | <0.001 | 0.61 |
| Statins, n (%) | 282 (71) | 85 (54) | <0.001 | 0.73 |
| Initial TIMI grade flow 3, n(%) | 61 (15) | 23 (15) | 0.84 | 0.05 |
| Anterior MI, n (%) | 155 (39) | 66 (42) | 0.51 | 0.35 |
ACEI, ACE inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index; CABG, coronary artery bypass grafting; CK, creatine kinase; eGFR, estimated glomerular filtration rate; IMR, ischaemic mitral regurgitation; IPTW, inverse probability of treatment weighted; MI, myocardial infarction; PCI, percutaneous coronary intervention; RASI, renin–angiotensin system inhibitor; TIMI, Thrombolysis In Myocardial Infarction.
Table 2.
Baseline echocardiographic findings in patients with IMR
| RASI (+) (n=395) |
RASI (−) (n=156) |
P value | |
| LV EDV, mL | 89±28 | 86±33 | 0.24 |
| LV ESV, mL | 45±20 | 46±27 | 0.66 |
| LVEF, % | 51±10 | 49±12 | 0.03 |
| LA dimension, cm | 3.7±0.6 | 3.7±0.6 | 0.40 |
| Moderate/severe MR, n(%) | 57 (14) | 52 (33) | <0.001 |
| Mitral deceleration time, ms | 205±59 | 206±63 | 0.86 |
| E/A ratio | 1.03±0.53 | 1.01±0.61 | 0.70 |
EDV, end-diastolic volume; EF, ejection fraction; ESV, end-systolic volume; IMR, ischaemic mitral regurgitation; LA dimension, left atrial dimension; LV, left ventricular; MR, mitral regurgitation; RASI, renin–angiotensin system inhibitor.
Survival analysis in patients with IMR
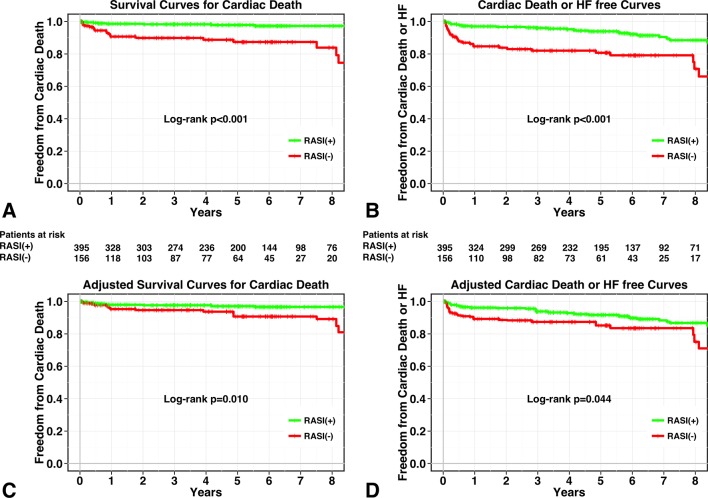
During the follow-up periods, 73 deaths occurred in patients with IMR. Of these, 31 were cardiac deaths (21 HFs, 7 sudden deaths, 2 MIs, 1 ventricular arrhythmia). Details of the remaining 42 non-cardiac deaths were as follows: cancer (n=17), infectious disease (n=10), stroke (n=4), renal failure (n=1), respiratory failure (n=1), pancreatitis (n=1), trauma (n=2) and unknown cause (n=6). Five-year survival from cardiac death was 98%±1% for patients receiving RASIs versus 87%±3% for patients not receiving RASIs (log-rank test, P<0.001; see figure 1A). In addition, 5-year survival from all-cause death was 93%±1% for patients receiving RASIs versus 76%±4% for patients not receiving RASIs (log-rank test, P<0.001). After the adjustment with IPTW, survival analysis showed a significant association between RASI therapy and a lower risk of all-cause death and cardiac death (log-rank test, P=0.005 for all-cause death and P=0.010 for cardiac death; see figure 1C).
Figure 1.
Kaplan-Meier curves for patient groups with RASI therapy (green line) and without RASI therapy (red line). Unadjusted curves for cardiac death (A) and composite of cardiac death and HF (B) and inverse probability of treatment-weighted survival curves for cardiac death (C) and composite of cardiac death and HF (D). HF, heart failure; RASI, renin–angiotensin system inhibitor.
Similarly, during the follow-up periods, 47 episodes of HF occurred. Five-year survival from cardiac death and HF was 94%±1% for patients receiving RASIs versus 81%±3% for patients not receiving RASIs (log-rank test, P<0.001; figure 1B). IPTW-adjusted event-free survival curves from cardiac death and HF showed a significant association between RASI therapy and a lower risk of cardiac death or HF (log-rank test, P=0.044; figure 1D).
Univariate predictors of cardiac death and composite outcome are shown in table 3. In addition, clinical outcomes according to RASI therapy using Cox regression analysis are shown in table 4.
Table 3.
Univariate predictors of cardiac death and composite outcome in patients with IMR
| Cardiac death | Cardiac death and heart failure | |||
| HR (95% CI) | P value | HR (95% CI) | P value | |
| Age | 1.04 (1.01 to 1.07) | 0.02 | 1.06 (1.04 to 1.09) | <0.001 |
| Male | 1.00 (0.44 to 2.29) | 0.99 | 1.47 (0.88 to 2.44) | 0.14 |
| Hypertension | 1.06 (0.55 to 2.02) | 0.87 | 1.62 (1.02 to 2.57) | 0.04 |
| Dyslipidaemia | 0.97 (0.50 to 1.86) | 0.92 | 1.01 (0.64 to 1.57) | 0.98 |
| Diabetes mellitus | 1.52 (0.79 to 2.94) | 0.21 | 1.69 (1.09 to 2.65) | 0.02 |
| Smoking | 1.22 (0.63 to 2.35) | 0.56 | 0.96 (0.61 to 1.53) | 0.87 |
| Killip classification | 2.54 (1.95 to 3.30) | <0.001 | 2.16 (1.77 to 2.64) | <0.001 |
| Peak CK | 1.15 (1.08 to 1.23) | <0.001 | 1.11 (1.05 to 1.17) | <0.001 |
| Creatinine | 1.30 (1.19 to 1.42) | <0.001 | 1.23 (1.14 to 1.34) | <0.001 |
| Prior MI | 2.37 (1.04 to 5.42) | 0.04 | 1.92 (1.04 to 3.54) | 0.04 |
| Anterior MI | 0.80 (0.42 to 1.54) | 0.51 | 1.23 (0.79 to 1.91) | 0.36 |
| PCI | 1.05 (0.37 to 2.96) | 0.93 | 0.70 (0.38 to 1.29) | 0.25 |
| Moderate or severe MR | 14.90 (7.74 to 28.68) | <0.001 | 5.13 (3.19 to 8.25) | <0.001 |
| β-blocker | 0.38 (0.20 to 0.73) | 0.004 | 0.41 (0.26 to 0.64) | <0.001 |
| Statins | 0.74 (0.39 to 1.43) | 0.37 | 0.62 (0.39 to 0.96) | 0.03 |
Per 1000 IU/L increments.
CK, creatine kinase; IMR, ischaemic mitral regurgitation; MI, myocardial infarction; PCI, percutaneous coronary intervention.
Table 4.
Clinical outcome according to RASI therapy in patients with IMR
| Treatment | Univariate analysis | Multivariate analysis | IPTW | |||||
| RASI(+) (n=395) |
RASI(−) (n=156) |
Unadjusted (95% CI) |
P value | Adjusted HR (95% CI) |
P value | Adjusted HR (95% CI) |
P value | |
| All-cause death | 34 (9) | 39 (25) | 0.27(0.17 to 0.43) | <0.001 | 0.44(0.27 to 0.72) | 0.001* | 0.43(0.25 to 0.74) | 0.002 |
| Cardiac death | 10 (3) | 21 (13) | 0.15(0.07 to 0.32) | <0.001 | 0.33(0.14 to 0.76) | 0.009* | 0.28(0.12 to 0.66) | 0.004 |
| Composite outcome† | 29 (7) | 31 (20) | 0.30(0.18 to 0.50) | <0.001 | 0.53(0.30 to 0.92) | 0.025‡ | 0.51(0.28 to 0.95) | 0.033 |
*Covariates include peak CK, Killip classification, serum creatinine, prior MI, moderate/severe MR and β-blocker.
†Cardiac death or heart failure.
‡Adjusted with hypertension, peak CK, Killip classification, serum creatinine, prior MI, moderate/severe MR and β-blocker.
CK, creatine kinase; IMR, ischaemic mitral regurgitation; IPTW, Inverse probability of treatment weighted; MI, myocardial infarction; RASI, renin–angiotensin system inhibitor.
Patient characteristics and echocardiographic results in non-IMR patients
Of the 505 patients with no or trivial MR, 403 (80%) patients received RASI before hospital discharge. Baseline characteristics and echocardiographic results of the patients with and without RASI therapy are shown in table 5 and table 6, respectively. Patients without RASI therapy were less likely to have hypertension, anterior MI and β-blocker, and more likely to have smaller LV end-diastolic volume and end-systolic volume compared with patients with RASI therapy. Serum creatinine level and peak CK level were comparable between the two groups.
Table 5.
Baseline patient characteristics stratified by the presence or absence of RASI therapy in non-IMR patients
| Characteristics | Unadjusted data | IPTW | ||
| RASI (+) (n=403) |
RASI (−) (n=102) |
P value | P value | |
| Age, years, mean±SD | 62±12 | 65±12 | 0.11 | 0.94 |
| Gender, male/female | 334/69 | 87/15 | 0.55 | 0.26 |
| Cardiovascular risk factors | ||||
| Hypertension, n (%) | 230 (57) | 46 (45) | 0.03 | 0.83 |
| Diabetes mellitus, n (%) | 131 (33) | 26 (25) | 0.17 | 0.48 |
| Dyslipidaemia, n (%) | 181 (45) | 48 (47) | 0.70 | 0.83 |
| Current smoker, n (%) | 173 (43) | 36 (35) | 0.16 | 0.34 |
| Frailty | 8 (2) | 5 (5) | 0.10 | 0.80 |
| BMI, kg/m2, mean±SD | 24.5±3.6 | 22.9±3.3 | <0.001 | 0.16 |
| ST segment elevation MI, n (%) | 316 (78) | 76 (75) | 0.40 | 0.99 |
| Killip classification, n (%) | 1.2±0.5 | 1.2±0.6 | 0.56 | 0.91 |
| Prior MI, n (%) | 32 (8) | 6 (6) | 0.47 | 0.63 |
| Primary PCI, n (%) | 366 (91) | 81 (79) | 0.003 | 0.55 |
| CABG, n (%) | 7 (2) | 14 (14) | <0.001 | 0.93 |
| Peak CK, IU/L | 2380±2641 | 2194±2417 | 0.52 | 0.96 |
| Serum creatinine, mg/dL | 1.05±0.97 | 1.07±0.39 | 0.85 | 0.67 |
| eGFR, mL/min/1.73 m2 | 52±18 | 49±21 | 0.10 | 0.01 |
| ACEI, n (%) | 296 (73) | 0 (0) | <0.001 | <0.001 |
| ARB, n (%) | 107 (27) | 0 (0) | <0.001 | <0.001 |
| β-blocker, n (%) | 315 (78) | 66 (65) | 0.01 | 0.96 |
| Statins, n (%) | 266 (66) | 58 (57) | 0.09 | 0.78 |
| Initial TIMI grade flow 3, n(%) | 81 (20) | 21 (21) | 0.91 | 0.68 |
| Anterior MI, n (%) | 216 (54) | 40 (39) | 0.01 | 0.69 |
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index; CABG, coronary artery bypass grafting; CK, creatine kinase; eGFR, estimated glomerular filtration rate; IMR, ischaemic mitral regurgitation; MI, myocardial infarction; PCI, percutaneous coronary intervention; RASI, renin–angiotensin system inhibitor; TIMI, Thrombolysis In Myocardial Infarction.
Table 6.
Baseline echocardiographic findings in non-IMR patients
| RASI (+) (n=403) |
RASI (−) (n=102) |
P value | |
| LV EDV, mL | 86±28 | 75±24 | 0.001 |
| LV ESV, mL | 41±21 | 35±18 | 0.02 |
| LVEF, % | 55±10 | 56±12 | 0.41 |
| LA dimension, cm | 3.5±0.6 | 3.5±0.6 | 0.98 |
| Moderate/severe MR, n(%) | 0 (0) | 0 (0) | – |
| Mitral deceleration time, ms | 217±55 | 213±56 | 0.54 |
| E/A ratio | 0.91±0.34 | 0.95±0.35 | 0.35 |
EDV, end-diastolic volume; EF, ejection fraction; ESV, end-systolic volume; IMR, ischaemic mitral regurgitation; LA dimension, left atrial dimension; LV, left ventricular; MR, mitral regurgitation; RASI, renin–angiotensin system inhibitor.
Survival analysis in non-IMR patients
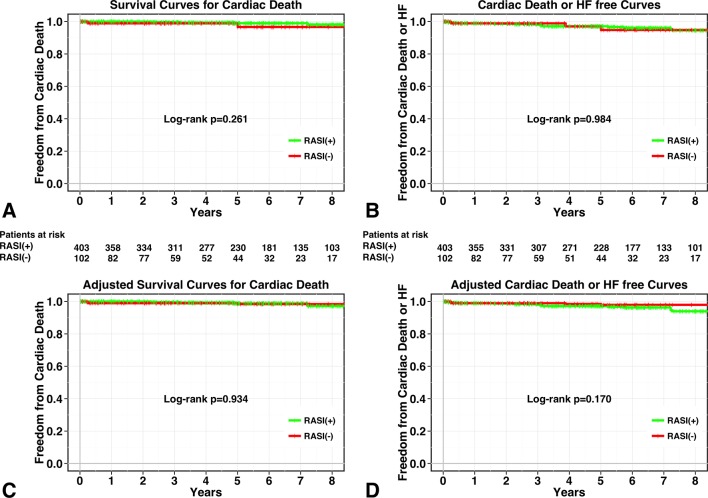
During the follow-up periods, 34 deaths occurred in non-IMR patients. Of these, six were cardiac deaths (two 2 HFs, three 3 sudden deaths and one 1 ventricular arrhythmia). Details of the remaining 28 non-cardiac deaths were as follows: cancer (n=9), infectious disease (n=7), stroke (n=4), liver failure (n=2), respiratory failure (n=1), ruptured aortic aneurysm (n=1), pancreatitis (n=1), unknown cause (n=3). Five-year survival from cardiac death was 99%±1% for patients receiving RASIs versus 99%±1% for patients not receiving RASIs (log-rank test, P=0.261; see figure 2A). In addition, 5-year survival from all-cause death was 95%±1% for patients receiving RASIs versus 95%±2% for patients not receiving RASIs (log-rank test, P=0.037). After the adjustment with IPTW, survival analysis showed no significant association between RASI therapy and a lower risk of all-cause death and cardiac death (log-rank test, P=0.362 for all-cause death and P=0.934 for cardiac death; see figure 2C).
Figure 2.
Kaplan-Meier curves for patient groups with RASI therapy (green line) and without RASI therapy (red line). Unadjusted curves for cardiac death (A) and composite of cardiac death and HF (B) and inverse probability of treatment-weighted survival curves for cardiac death (C) and composite of cardiac death and HF (D). HF, heart failure; RASI, renin–angiotensin system inhibitor.
Similarly, during the follow-up periods, 15 episodes of HF occurred. Five-year survival from cardiac death and HF was 97%±1% for patients receiving RASIs versus 97%±2% for patients not receiving RASIs. There were no significant differences in the rate of the composite of cardiac death and HF between patients with and without RASI therapy (log-rank test, P=0.984; figure 2B). In addition, IPTW-adjusted survival-free event-free survival curves from cardiac death and the composite of cardiac death and HF did not show significant associations between RASI therapy and these endpoints (log-rank test, P=0.170; figure 2D)
Discussion
The main findings of this study were as follows. (1) RASI therapy was associated with a lower incidence of adverse cardiac events in patients with IMR after AMI. (2) There were no significant differences in the incidence of cardiac events according to the use of RASI in patients without IMR.
It has been widely recognised that patients with IMR after AMI have poorer prognosis than those without IMR. IMR occurs as a consequence of global or regional LV remodelling despite a structurally normal valve, which cause the leaflet tethering by displacement of papillary muscles.24–26 Accordingly, the therapeutic approaches should be aimed at the underlying LV dysfunction. Medical therapy including β-blockers and RASI for LV dysfunction and chronic HF is the essential therapy for patients with IMR. Cardiac resynchronisation therapy and coronary revascularisation are also considered in patients with an appropriate indication. Since mitral valve surgery can correct MR severity and improve symptoms of HF, surgical repair of IMR combined with coronary revascularisation has been thought to be a reasonable option. However, it is unclear whether it could improve long-term survival in patients with IMR who do not require CABG.27 28 Considering the uncertain survival benefit and operative risk in patients with IMR, medical therapy may currently be the only reasonable first-line strategy for the patients with IMR after AMI.
Previous studies reported that ACEI was efficacious in reducing IMR in patients with HF and LV dysfunction9 10; however, these studies did not assess the effect of ACEI on cardiovascular events. In a study that investigated the long-term prognosis of medically treated patients with functional MR and LV dysfunction receiving current standard pharmacological therapy, medical therapies including ACEI and β-blocker were not independent predictors of mortality.6 However, a more recent study demonstrated that the use of RASIs was associated with better long-term prognosis in patients with MI and significant (>moderate) IMR.29 Our present study is consistent with those results.
Although ACEIs (or ARB when an ACEI was not tolerated) are recommended in all patients after AMI, a considerable portion of patients after AMI do not receive RASIs in the real-world clinical practice mainly because of intolerance to RASI therapy such as hypotension, renal insufficiency and hyperkalemia. The prescription rate of RASIs at discharge after AMI was approximately 60% in 1998 and 80% in 2010 in a post-MI registry in Japan.30 According to a Japanese registry study that included patients who underwent PCI or CABG, about 70% of patients with history of MI received RASI therapy at discharge, which means almost 30% of patients did not receive RASI therapy.31 Our results suggest that introduction at a low dose and uptitration of RASI should always be considered even in patients with IMR who seem to be intolerant to RASI therapy.
This study did not find a significant association between RASI therapy and reduced cardiac events in patients who did not have IMR after AMI. A possible explanation for these results may be the different patient characteristics in this study compared with previous studies. Many of the non-IMR patients who did not receive RASI therapy had non-dilated LV with preserved EF; in addition, most of the patients in this study were revascularised by primary PCI or emergent CABG during acute phase of MI. In the PREAMI study,19 which investigated whether ACEIs show beneficial effects in elderly postinfarction patients with preserved LVEF, ACEI therapy was associated with reduced LV remodelling, but not with better clinical outcomes. Because non-IMR patients in this study seemed to have a lower risk of cardiovascular events compared with patients in previous trials of ACEIs who had reduced LVEF or chronic HF, this might lead to a failure to find the benefit of RASI therapy in cardiovascular prevention in this patient population. Our results suggest that patients with IMR might receive clinical benefit from RASI therapy after AMI regardless of MR severity and LV dysfunction in the reperfusion setting.
Our study has several limitations. First, since our study was based on a retrospective analysis, initiation of ACEI therapy depended on the physician’s discretion. Although we performed adjustment with the inverse probability treatment weighting to minimise the selection bias, unmeasured confounding factors might be present. Second, we evaluated the long-term effect of RASI based on the medications at discharge. However, the dose and class of RASI, and discontinuation after discharge were not assessed. Thus, patients with and without RASI therapy were compared in an intention-to-treat fashion based on the initial treatment assignment. Duration of RASI therapy may have a significant impact on clinical outcomes in patients with IMR. Third, the peak CK levels may be less accurate than sigma CK levels to evaluate the infarct size. However, since there were wide varieties in the timing and frequency of the assessments of CK levels, we could not calculate the sigma CK levels in all patients. Finally, we assessed IMR semiquantitatively and did not evaluate it quantitatively. The use of colour Doppler for determining MR severity might not be very accurate because of technical and haemodynamic limitations,32 but is part of the routine echocardiographic examination. In addition, although we defined IMR with rigorous echocardiographic criteria, we could not completely exclude patients with pre-existing MR.
Footnotes
Contributors: KK: acquisition of data, analysis and interpretation of data, drafting of manuscript; SK: study concept and design, analysis and interpretation of data; MK, RM, YS, TK, TY, NE, AK, MK: acquisition of data; YF: critical revision.
Competing interests: None declared.
Ethics approval: Institutional Review Board of Kobe City Medical Center General Hospital.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: There are no additional data available for this paper.
References
- 1. Bursi F, Enriquez-Sarano M, Nkomo VT, et al. Heart failure and death after myocardial infarction in the community: the emerging role of mitral regurgitation. Circulation 2005;111:295–301. 10.1161/01.CIR.0000151097.30779.04 [DOI] [PubMed] [Google Scholar]
- 2. Pellizzon GG, Grines CL, Cox DA, et al. Importance of mitral regurgitation inpatients undergoing percutaneous coronary intervention for acute myocardial infarction: the Controlled Abciximab and Device Investigation to Lower Late Angioplasty Complications (CADILLAC) trial. J Am Coll Cardiol 2004;43:1368–74. 10.1016/j.jacc.2003.11.046 [DOI] [PubMed] [Google Scholar]
- 3. Grigioni F, Enriquez-Sarano M, Zehr KJ, et al. Ischemic mitral regurgitation: long-term outcome and prognostic implications with quantitative Doppler assessment. Circulation 2001;103:1759–64. 10.1161/01.CIR.103.13.1759 [DOI] [PubMed] [Google Scholar]
- 4. Aronson D, Goldsher N, Zukermann R, et al. Ischemic mitral regurgitation and risk of heart failure after myocardial infarction. Arch Intern Med 2006;166:2362–8. 10.1001/archinte.166.21.2362 [DOI] [PubMed] [Google Scholar]
- 5. Amigoni M, Meris A, Thune JJ, et al. Mitral regurgitation in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both: prognostic significance and relation to ventricular size and function. Eur Heart J 2007;28:326–33. 10.1093/eurheartj/ehl464 [DOI] [PubMed] [Google Scholar]
- 6. Agricola E, Ielasi A, Oppizzi M, et al. Long-term prognosis of medically treated patients with functional mitral regurgitation and left ventricular dysfunction. Eur J Heart Fail 2009;11:581–7. 10.1093/eurjhf/hfp051 [DOI] [PubMed] [Google Scholar]
- 7. Weiland DS, Konstam MA, Salem DN, et al. Contribution of reduced mitral regurgitant volume to vasodilator effect in severe left ventricular failure secondary to coronary artery disease or idiopathic dilated cardiomyopathy. Am J Cardiol 1986;58:1046–50. 10.1016/S0002-9149(86)80036-4 [DOI] [PubMed] [Google Scholar]
- 8. Hamilton MA, Stevenson LW, Child JS, et al. Sustained reduction in valvular regurgitation and atrial volumes with tailored vasodilator therapy in advanced congestive heart failure secondary to dilated (ischemic or idiopathic) cardiomyopathy. Am J Cardiol 1991;67:259–63. 10.1016/0002-9149(91)90556-Z [DOI] [PubMed] [Google Scholar]
- 9. Seneviratne B, Moore GA, West PD. Effect of captopril on functional mitral regurgitation in dilated heart failure: a randomised double blind placebo controlled trial. Br Heart J 1994;72:63–8. 10.1136/hrt.72.1.63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Levine AB, Muller C, Levine TB. Effects of high-dose lisinopril-isosorbide dinitrate on severe mitral regurgitation and heart failure remodeling. Am J Cardiol 1998;82:1299–301. 10.1016/S0002-9149(98)00623-7 [DOI] [PubMed] [Google Scholar]
- 11. Shimoyama H, Sabbah HN, Rosman H, et al. Effects of long-term therapy with enalapril on severity of functional mitral regurgitation in dogs with moderate heart failure. J Am Coll Cardiol 1995;25:768–72. 10.1016/0735-1097(94)00426-Q [DOI] [PubMed] [Google Scholar]
- 12. Pfeffer MA, Braunwald E, Moyé LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 1992;327:669–77. 10.1056/NEJM199209033271001 [DOI] [PubMed] [Google Scholar]
- 13. Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. Lancet 1993;342:821–8. [PubMed] [Google Scholar]
- 14. Køber L, Torp-Pedersen C, Carlsen JE, et al. A clinical trial of the angiotensin-converting-enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. Trandolapril Cardiac Evaluation (TRACE) Study Group. N Engl J Med 1995;333:1670–6. 10.1056/NEJM199512213332503 [DOI] [PubMed] [Google Scholar]
- 15. Dickstein K, Kjekshus J. OPTIMAAL Steering Committee of the OPTIMAAL Study Group. Effects of losartan and captopril on mortality and morbidity in high-risk patients after acute myocardial infarction: the OPTIMAAL randomised trial. Optimal trial in myocardial infarction with angiotensin II antagonist losartan. Lancet 2002;360:752–60. [DOI] [PubMed] [Google Scholar]
- 16. Pfeffer MA, McMurray JJ, Velazquez EJ, et al. Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N Engl J Med 2003;349:1893–906. 10.1056/NEJMoa032292 [DOI] [PubMed] [Google Scholar]
- 17. Pfeffer MA, Swedberg K, Granger CB, et al. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the charm-overall programme. Lancet 2003;362:759–66. 10.1016/S0140-6736(03)14282-1 [DOI] [PubMed] [Google Scholar]
- 18. Nishimura RA, Otto CM, Bonow RO, et al. AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American college of cardiology/American heart association task force on practice guidelines. J Am Coll Cardiol 2014;2014:e57–185. [DOI] [PubMed] [Google Scholar]
- 19. Ferrari R. Perindopril and Remodeling in Elderly with Acute Myocardial Infarction Investigators. Effects of angiotensin-converting enzyme inhibition with perindopril on left ventricular remodeling and clinical outcome: results of the randomized perindopril and remodeling in elderly with acute myocardial infarction (PREAMI) Study. Arch Intern Med 2006;166:659–66. 10.1001/archinte.166.6.659 [DOI] [PubMed] [Google Scholar]
- 20. Braunwald E, Domanski MJ, Fowler SE, et al. PEACE Trial Investigators. Angiotensin-converting-enzyme inhibition in stable coronary artery disease. N Engl J Med 2004;351:2058–68. 10.1056/NEJMoa042739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Corti MC, Guralnik JM, Salive ME, et al. Serum albumin level and physical disability as predictors of mortality in older persons. JAMA 1994;272:1036–42. 10.1001/jama.1994.03520130074036 [DOI] [PubMed] [Google Scholar]
- 22. Bandeen-Roche K, Xue QL, Ferrucci L, et al. Phenotype of frailty: characterization in the women’s health and aging studies. J Gerontol A Biol Sci Med Sci 2006;61:262–6. 10.1093/gerona/61.3.262 [DOI] [PubMed] [Google Scholar]
- 23. Matsuo S, Imai E, Horio M, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis 2009;53:982–92. 10.1053/j.ajkd.2008.12.034 [DOI] [PubMed] [Google Scholar]
- 24. Otsuji Y, Handschumacher MD, Schwammenthal E, et al. Insights from three-dimensional echocardiography into the mechanism of functional mitral regurgitation: direct in vivo demonstration of altered leaflet tethering geometry. Circulation 1997;96:1999–2008. 10.1161/01.CIR.96.6.1999 [DOI] [PubMed] [Google Scholar]
- 25. Otsuji Y, Handschumacher MD, Liel-Cohen N, et al. Mechanism of ischemic mitral regurgitation with segmental left ventricular dysfunction: three-dimensional echocardiographic studies in models of acute and chronic progressive regurgitation. J Am Coll Cardiol 2001;37:641–8. 10.1016/S0735-1097(00)01134-7 [DOI] [PubMed] [Google Scholar]
- 26. Kumanohoso T, Otsuji Y, Yoshifuku S, et al. Mechanism of higher incidence of ischemic mitral regurgitation in patients with inferior myocardial infarction: quantitative analysis of left ventricular and mitral valve geometry in 103 patients with prior myocardial infarction. J Thorac Cardiovasc Surg 2003;125:135–43. 10.1067/mtc.2003.78 [DOI] [PubMed] [Google Scholar]
- 27. Wu AH, Aaronson KD, Bolling SF, et al. Impact of mitral valve annuloplasty on mortality risk in patients with mitral regurgitation and left ventricular systolic dysfunction. J Am Coll Cardiol 2005;45:381–7. 10.1016/j.jacc.2004.09.073 [DOI] [PubMed] [Google Scholar]
- 28. Mihaljevic T, Lam BK, Rajeswaran J, et al. Impact of mitral valve annuloplasty combined with revascularization in patients with functional ischemic mitral regurgitation. J Am Coll Cardiol 2007;49:2191–201. 10.1016/j.jacc.2007.02.043 [DOI] [PubMed] [Google Scholar]
- 29. Okura H, Kataoka T, Yoshida K. Renin-angiotensin system inhibitors in patients with myocardial infarction and secondary mitral regurgitation. Heart 2016;102:694–700. 10.1136/heartjnl-2015-308536 [DOI] [PubMed] [Google Scholar]
- 30. Hara M, Sakata Y, Nakatani D, et al. Comparison of 5-year survival after acute myocardial infarction using angiotensin-converting enzyme inhibitor versus angiotensin II receptor blocker. Am J Cardiol 2014;114:1–8. 10.1016/j.amjcard.2014.03.055 [DOI] [PubMed] [Google Scholar]
- 31. Nishino T, Furukawa Y, Kaji S, et al. Distinct survival benefits of angiotensin-converting enzyme inhibitors/angiotensin II receptor blockers in revascularized coronary artery disease patients according to history of myocardial infarction. Circ J 2013;77:1242–52. 10.1253/circj.CJ-12-0963 [DOI] [PubMed] [Google Scholar]
- 32. Zoghbi WA, Enriquez-Sarano M, Foster E, et al. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr 2003;16:777–802. 10.1016/S0894-7317(03)00335-3 [DOI] [PubMed] [Google Scholar]