Abstract
Patellar tendinopathy is very common in patients complaining of anterior knee pain. Its aetiology is still unclear, but neovascularisation seems to play a role. Different treatments have been proposed overtime, from rehabilitation to platelet-rich-plasma injections, but there is no agreement on the best treatment protocol. The final stage of patellar tendinopathy is patellar tendon rupture. In these cases surgical treatment is often required. The aim of this literature review is to focus on the aetiology, diagnosis, and treatment of both patellar tendinopathy and rupture. We report the conservative treatments proposed for patellar tendinopathy and the surgical techniques described for its rupture.
Keywords: patella, reconstruction, repair, rupture, tendinopathy
Patellar tendinopathy
The most common complaint of patients with patellar tendinopathy is pain around the patella. Different authors have demonstrated that the disease has a degenerative aetiology rather than an inflammatory aetiology.1
Blazina was the first to define this pathology as “Jumper's knee”, and to divide it into four different stages: (1) pain after sport activity; (2) pain during sport activity without performance limitation; (3) pain during or after sport activity with performance limitation; and (4) patellar rupture.2 The last stage will be further defined in the next section.
Patellar tendinopathy can be further divided into acute: onset in < 2 weeks; subacute: between 2 weeks and 6 weeks; and chronic: > 6 weeks.3 Repetitive micro trauma was demonstrated to be the major cause of patellar tendinopathy; for this reason it is also defined as “overuse pathology”. It is more frequent in volleyball, basketball, and soccer players, because of changes of direction, jumping, and running activities.4
Patellar tendinopathy is disabling, most of all for athletes. About 30% of athletes affected by patellar tendinopathy do not return to sport for 6 months, with 50% of these having anterior knee pain 15 years after diagnosis.5
Different authors have proposed that an excessive mechanical loading placed on the tendon as the mechanism for the development of patellar tendinopathy. Recently Zhang et al6 tried to investigate tendon mechanic-biological responses through an in vivo (mouse treadmill running model) and an in vitro model. In the in vivo study they found that treadmill running elevated the expression of mechanical growth factors and enhanced the proliferative potential of tendon stem cells (TSC). Besides, moderate treadmill running upregulated different tenocyte-related genes (i.e., collagen type I), but did not affect nontenocyte-related genes. On the contrary, intensive treadmill running played a role in the upregulation of both tenocyte and nontenocyte related genes. In the in vitro part of the study they demonstrated that low mechanical stretching of TSCs from both patellar and Achilles tendon increased the expression of only tenocyte-related genes. However, high mechanical stretching increased both the expression of tenocyte and nontenocyte related genes. The authors concluded that excessive mechanical loading may cause anabolic changes in the tendons, inducing the differentiation of TSC into nontenocytes, which may lead to degenerative tendinopathy development.
Ferretti et al7 evaluated 18 patellar tendon fragments from surgeries and observed that the histological alterations were localised in the tendon-bone junctions, while the tendon itself was healthy.
From a histological point of view there is a “disorganisation” in the collagen fibres associated with fibrosis, increased cellularity, and vascularity. Inflammation is poor in a chronic setting, while it plays a fundamental role in the first phase of the pathology.8 Because of the continuous stress on the tendon, the tenocytes produce more cytokines and interleukins, causing some micro fractures in the collagen fibres.8
The tendon alterations include an increase in collagen type III fibres and the deposition of glycosaminoglycans. There is a different kind of regulation of the homeostasis of the matrix enzymes, which are involved in collagen, metalloproteinase, and their inhibitor degradation. Macroscopically the healthy tendon is not a neural structure; during the process, however, the innervations increase, with chronic pain due to cytokines, pain mediators, hypoxia, and pH changes.9
Neovascularisation seems to be involved in pain generation, but this is still a debated issue. There are different studies on shoulder rotator cuff, but diminished vascularisation seems to be a more credible theory. However there is no certain aetiology of the pain in this pathology.10
Recently some genetic studies tried to better understand the aetiology of this pathology by analysing the DNA sequences which can be involved in tendon alterations. Some polymorphisms have been recently described for the collagen V gene,11 as well as some genetic variations on type II metalloproteinase tissue inhibitor and some other genes (i.e., ADAMTS14). Those genes seemed to be involved in increasing or decreasing Achilles tendinopathy.12
Diagnosis of patellar tendinopathy
Patients with patellar tendinopathy usually describe anterior knee pain, which is exacerbated by activity or sometimes by prolonged knee flexion. The onset of pain is often insidious, sometimes related to a period of increased activity, and most of all referred to the inferior pole of the patella. In severe cases patients can experience pain also during daily activities. In the evaluation of tendon overuse, different scales were proposed, but they often fail to discriminate symptomatic differences.2 Recently, a more accurate scale was developed: the Victorian Institute Sports tendon Assessment (VISA) score. This scale was studied specifically to assess symptoms and functionality in patellar tendinopathy, with good inter and intra-observer reliability and stability.13 Physical findings are fairly straightforward in patellar tendinopathy; the most consistent factor is localised tenderness at the inferior pole of the patella, but with low sensitivity and specifity.14 The most important differential diagnoses with patellar tendinopathy are patello-femoral pain syndrome and Hoffa impingement. Plain X-rays are useful to assess patella position, associated pathologies (i.e., Osgood-Schlatter), or intratendinous calcifications.15 Ultrasonoghraphic (US) evaluation is typically the second line imaging, showing the typical echogenicity of the fibrillar structure of the patellar tendon.16 In patellar tendinitis there is degradation and disorganisation of collagen fibres that is depicted as a well-defined hypoechogenic zone often associated with tendon thickening.17 In advanced cases small foci of hyper reflectivity (i.e., calcifications) or intratendinous clefts can be found. In association with US, colour and Power Doppler can be useful to assess intratendinous vascularisation.18 Magnetic resonance imaging (MRI) can also be useful to assess areas of increased signal intensity, especially on short echo time images. The detection of partial tears is difficult, but it is better evaluated on T2-weighted images with high signal intensity.19 Figure 1 shows an MRI image of patellar tendinopathy (in the red circle).
Figure 1.
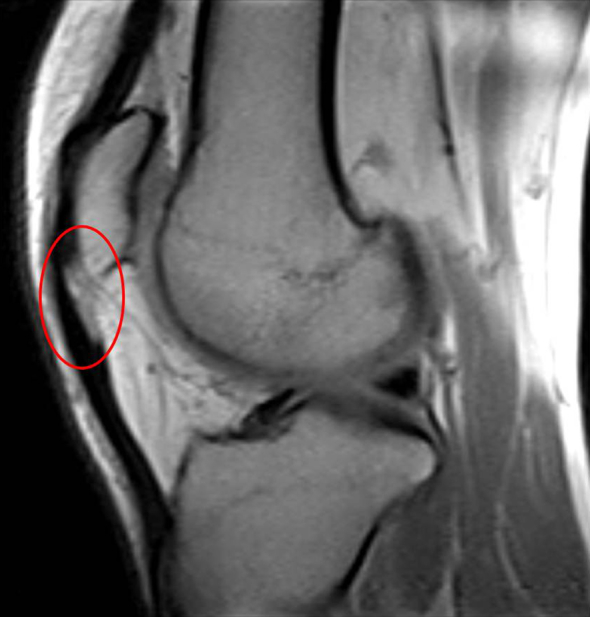
Magnetic resonance imaging caption showing insertional patellar tendinopathy (red circle).
Treatment options and results of patellar tendinopathy
The lack of basic knowledge on the aetiology and pain mechanisms associated with patellar tendinopathy is reflected in the existence of many different treatment protocols.15 The aim of this section is to analyse the different possible treatments described in recent literature.
Eccentric exercises
Rehabilitation using eccentric exercises plays a central role in patellar tendinopathy therapy. The exercises can be performed with single leg squat at 25° of inclination or with weight-bearing squat using leg press. These exercises seem to promote the collagen fibres' cross-linkage and tendinous reshaping, and so tendinopathy prevention. In the literature, decreased pain and neovascularisation in patellar tendinopathy treated with this rehabilitation protocol were demonstrated with good long-term results.5
Extracorporeal shock wave therapy
Extracorporeal shock wave therapy (ESWT) is a procedure that has good results on chronic tendinopathy and a positive effect on pain and functionality. Different studies demonstrated good results with an overall improvement from 73.5% to 87.5% on patients' functionality.20 A recent systematic review evaluated the effect of ESWT on athletes affected by patellar tendinopathy without interrupting their sport activity. The authors included seven studies, but only two of them had good quality methodology. They concluded good clinical outcomes in athletes treated with ESWT, with an improvement in about 74.7% of patients.21
Zwerver et al22 performed the VISA questionnaire at 1 week, 12 weeks, and 22 weeks after ESWT treatment and they concluded that, despite the athletes continued training and competitions, no worsening of the symptoms could be detected. The same authors in a randomised study in 201123 reported their results using ESWT in 62 athletes, concluding that after 22 weeks no differences in outcomes could be detected compared with the control group (placebo). However, this poor result can be explained by the short time for functional recovery the patients had during the competitive season.23
Patellar tendinopathy has been described also in patients who underwent anterior cruciate ligament reconstruction using bone-tendon-bone graft. In this population Wang et al20 and Peers et al24 in two different studies described good results using ESWT, with good patients' satisfaction, improved tendon vascularization, and reduced tendon thickness at the US compared with the control group.
Despite the demonstrated utility of ESWT in a chronic setting, there is a limited evidence to recommend a specific treatment protocol for in-season athletes in an acute setting.21 However, in literature there is great nonuniformity on treatment methodology with ESWT. ESWT can be divided into localised and radial based on the physical characteristics of the wave. Most of the studies reporting on ESWT used focalised waves, and the results on patellar tendinopathy are unclear.25
In conclusion, a definitive answer on the efficacy of ESWT therapy is difficult. Based on the literature, an ESWT treatment using low frequencies and low energy waves, without anaesthesia, after an initial period of rest, seems to be reasonable. However there is a lack of evidence regarding the type of waves to use (focalised or radial) and their efficacy.25
Percutaneous technique
There are different percutaneous and injective therapies described in literature to treat patellar tendinopathy: platelet-rich-plasma (PRP), autologous blood, needling, corticosteroid injections, and sclerosant therapy. Recently some authors demonstrated the inefficacy of topical nitroglycerin in association with eccentric exercises,26 and for this reason we will not focus on this treatment.
PRP
PRP is an autologous preparation of platelets in a small volume of plasma. In the α cells of platelets there are different growth factors [transforming growth factor-β1, transforming growth factor-β2, platelet-derived growth factor (PDGF)-AA, PDGF-BB, PDGF-AB, growth factor-1, epidermal growth factor, hepatocyte growth factor], that work together to improve wound healing, to reduce inflammatory response, and to improve regeneration of damaged tissues.
Using PRP to improve tendon healing has been recently proposed. Volpi et al27 in 2007 reported their results on eight patients treated with a single PRP injection in association with a rehabilitation protocol to treat chronic tendinopathy. One hundred and twenty days after the injection the patients reported a significant improvement in pain, with associated improvement in MRI imaging in 80% of patients.27
Kon et al28 in 2009 described their results on 20 patients treated with three PRP injections (1 every 15 days containing about 6.8 million platelets) at a minimum follow-up of 6 months. The authors concluded that there was a significant improvement in all the scores, but there was no control group. Filardo et al29 performed a similar study comparing patients treated with PRP injections (3 injections, 1 every 15 days) with those treated with physical therapy only. No differences were detected between the two groups in pain scores, satisfaction, or recovery time, but the activity level significantly improved in the PRP-group. De Vos et al30 published a randomised control trial (RCT) on 54 patients affected by Achilles tendinopathy and randomly allocated into a PRP treatment group or placebo group. The mean VISA-A score significantly improved in both the groups, without any statistically significant differences. The authors concluded that in patients with chronic Achilles tendinopathy treated with eccentric exercises, a PRP injection compared with a saline injection did not result in greater improvement in pain and activity.
van Ark et al31 in a systematic review analysed three studies with good clinical outcomes using PRP, concluding that all the studies had some bias and limitations. Recently Gonsens et al32 reported their results on 36 patients, underlying that one single PRP injection had positive results on pain and functionality, most of all on patients who had not undergone any previous percutaneous treatment. Vetrano et al33 compared patients treated with PRP injections or ESWT in a RCT. In the first group, two US-guided autologous PRP injections every 2 weeks were performed while the second group of patients underwent three ESWT sessions every 48–72 hours. In association with these treatments all the patients underwent a standardised rehabilitation protocol for 2 weeks after the last injections-physical therapy session. The authors concluded on a significant improvement in both groups at 2 months, 6 months, and 12 months of follow-up, with better results at the last follow-up in the PRP group.
Lane et al,34 in a rabbit model, demonstrated increased cellular activity at 7 days in the PRP-group, confirming that PRP factors are “short-acting”. Other authors confirmed that tumour necrosis factor-α and vascular endothelial growth factor levels returned to basal levels 24 hours after the injection.35
Recently Charousset et al36 reported on 28 athletes (17 professional, 11 semiprofessional) with chronic patellar tendinopathy refractory to nonoperative management and treated with three US-guided pure PRP injections into the site of the tendinopathy. After 2 years of follow-up the authors concluded that three consecutive US-guided PRP injections significantly improved symptoms and function in this population, with MRI 3 months after the last injection showing normal tendon architecture.
In conclusion there are still different debates in the literature regarding PRP injections in patellar tendinopathy treatment, mainly due to the variability of the treatment protocol reported. Recently a systematic review was published in the Cochrane database, underling the limitations of the published studies and concluding that there is low evidence on the efficacy of PRP injection in patellar tendinopathy treatment.37 In 2014 Andia et al38 published a systematic review on PRP treatment of painful tendinopathy, including 13 prospective controlled studies. The authors concluded that PRP treatment can achieve better intermediate to long-term pain control compared with control interventions, but further studies circumventing heterogeneity are needed to reach firm conclusions.
Wang39 recently published a review in which he described the different factors potentially influencing the conflicting outcomes of PRP, dividing them into PRP-related and patient-related factors. The PRP-related factors may include: (1) preparation (i.e., containing white blood cells); (2) platelet concentration; (3) activated or nonactivated PRP; (4) method of PRP activation (i.e., calcium or thrombin); (5) mode of application (injection or implantation); and (6) frequency of PRP application. The patient-associated factors include: (1) age; (2) timing of tendon injury; (3) type of tissue injured (i.e., tendon, soft tissue); (4) patient activity level; (5) treatment history; and (6) postrecovery rehabilitation.
Autologous blood and needling
The aim of these techniques is to increase bleeding in the pathologic tendon and to induce granulation tissue formation and subsequent healing.
James et al40 compared the results of needling alone, autologous blood alone, and an association of both techniques, with the latter technique obtaining the best clinical outcomes. Their study had some limitations, including the absence of a control group.
Sclerosing injections
Tendinous vascularisation seems to play a role in pain pathogenesis; for this reason using some substances limiting neovascularisation can be a therapeutic option.
In a recent RCT, Willberg et al41 compared the results obtained with sclerosing injection or arthroscopic shaving. The authors concluded that good outcomes are obtained with both treatments, but in the group with arthroscopic shaving, pain reduction and patient satisfaction were greater. Hoksrud et al42 evaluated the efficacy of sclerosing injections obtaining some improvement in pain and knee function, but most of the patients still complained about knee pain at 2 years follow-up.
Corticosteroid injections
The anti-inflammatory properties of corticosteroid injections are well known; however there is some concern about using them in tendinopathy because of some collateral effects, like tendon rupture.
There are three clinical trials in the literature describing the results of corticosteroid injections in patellar tendinopathy. All the studies described a short-term improvement in pain, but with some cases of atrophy and tendon rupture, and poor long-term outcomes.43, 44, 45 Finally, Zhang et al46 demonstrated that one single corticosteroid injection stimulated tendinous cell differentiation into cartilage or adipose tissue, reducing the number of cells and increasing the risk of tendon rupture.
Surgical treatment
After 6 months of ineffective conservative treatment in Blazina stage III disease, a surgical indication may be necessary. In this chapter we will review only surgical treatment without tendon rupture; surgical treatment of tendon rupture will be described later.
The aims of surgical treatment are: removing fibrous adherences, re-establishing vascularisation, and trying to improve the biological stimulus for healing. If the pathology is localised in the centre of the tendon, there is some agreement in the efficacy of longitudinal tenotomies and degenerated tissue removal.47
In cases of insertional tendinopathy there is no agreement in the literature on the best surgical treatment. Arthroscopic treatment allows the patients a quicker recovery with a low complication rate. In these cases, treatment options are shaving,41, 48 peritenonion release in association with osseous denervation,49 and inferior patellar pole resection.50
It is still debated if the association between arthroscopic shaving and bone procedures is correct. To date there is no agreement in the literature and there is limited evidence on the best surgical treatment choice for insertional tendinopathy.51
Patellar tendon rupture
Extensor apparatus injuries have an incidence rate of 0.5–6%, but they are serious injuries because the extensor mechanism is essential for normal human gait, and can lead to great disability.52
Patellar tendon ruptures are more likely caused by direct or indirect trauma in young people; however they can be the extreme consequence of patellar tendinopathy, micro traumas, or overuse pathology.53
There are few data regarding patellar tendon ruptures in active people, because they are more frequent after total knee arthroplasty. However, in the literature there are a lot of case reports with small numbers with the focus being placed on different repairing techniques despite the aetiology or diagnostic process used.
Typically these lesions occur in active young people (younger than 40 years) and they are usually caused by an indirect trauma to the knee, like a sudden contraction of the quadriceps with the knee in slight flexion (sudden impulsion, sprint, avoiding a fall, etc.).54 Some authors also reported on traumatic patellar tendon lesions due to rapid dynamic loading conditions occurring in many sports situations; however, injuries in maximum tensile stress during static test conditions are less common.55
Strain forces on the patellar tendon are greater at the osteo-tendinous insertion than in the mid-substance: for this reason mid-substance lesions are much rarer than avulsions in normal tendons. Because of the great forces needed to break a healthy tendon, there is some agreement in the literature about the involvement of a degenerated tendon in patellar tendon ruptures, most of all in cases of a mid-substance lesion.56
Different authors have described the possible predisposing conditions for patellar tendon rupture, such as: corticosteroids local injections57; diabetes58; micro trauma2; rheumatic diseases59; metabolic disorders60; iatrogenic lesions (anterior cruciate ligament reconstruction)61; and fluoroquinolone systemic use.62
Diagnosis of patellar tendon rupture
To correctly evaluate patients affected by patellar tendon rupture, a good patient history is the first step. Typically the patients describe unexpected pain and subsequent knee impotence that can be sometimes associated to a fall, during a fast movement. A history of previous patellar tendinopathy should be assessed, and the surgeon should ask for other comorbidities or potential risk factors, as mentioned above. At clinical inspection the knee is usually swollen and, frequently, the patella cannot be recognised because of the oedema. Palpating the knee, a defect in the patellar tendon and a patella alta can be identified, especially in complete ruptures. Intra-articular haematoma is usually considerable and should be evacuated in emergency. The active knee extension may be impossible, both in some partial tendon rupture it is conserved. However in cases of complete tendon rupture the surgeon should search for extension lag sign.
A plain X-ray evaluation, both in antero-posterior and lateral views, is the first line imaging. A patellar avulsion, a patella alta, or a tibial tuberosity fracture can be suggestive of extensor mechanism rupture. In an emergency, a US can be useful to assess complete patellar tendon ruptures. The main advantage of this technique is the possibility to perform a dynamic evaluation; on the contrary its main limitation is poor reproducibility.
The gold standard for tendon ruptures is an MRI, but it is considered to be more expensive and less available.63 Computed tomography scan can be useful in cases of suspected tendon avulsion, but it is not useful in mid-substance lesions.
Treatment options and outcomes of patellar tendon rupture
Acute setting
Incomplete ruptures with intact function may be treated nonoperatively with 2–3 weeks of full extension immobilisation.64
In cases of incomplete lesions with functional deficits or complete ruptures, surgical treatment is required. In the acute setting the repair should be performed as soon as possible in order to have the best chance to achieve a tension-free suture.54 In the treatment of acute patellar tendon repair, isolated suture repair is not recommended because of the high failure rate. Besides, most of the lesions occur in the osteo-tendinous junctions, facilitating a trans-osseous suture.65
Normally the anterior approach through a midline incision is the best choice; then the surgeon should perform an accurate dissection of the retinacular tears and the tendon ends should be debrided. The inferior pole of the patella is accurately decorticated to improve bleeding and, consequently, healing. A pair of continuous, locking heavy (No. 2 or 5), nonabsorbable sutures are placed in the tendon using a Krakow or similar suture technique. Three parallel bone tunnels are drilled with a 2.5 mm drill from distal to proximal in the patella, a suture passer is used to pass the free suture ends in the bone tunnels, with the central one containing two sutures.
Once the correct patellar position and tracking are checked the strands are tensioned with the knee fully extended to approximate the tendon to the inferior pole of the patella. The retinacula are now closed with an absorbable suture and the knee is flexed at 90° to test the repair.64 Some authors described using suture anchors to repair patellar tendon ruptures, concluding that it can result in a low-profile construct and better recreation of tendon insertion into the inferior pole of the patella.66 Other authors reported on a lower gap formation after cyclic loading in suture anchors compared with the transpatellar technique.67, 68
In rare cases of mid-substance ruptures, continuous locking stitches can be used and patellar bone tunnels can be performed to hold the proximal fragment.69 Alternatively a neutralisation nonabsorbable wire or heavy sutures can be placed transversely through the patella and through another bone tunnel in the proximal tibia; this technique has been used to provide additional stability to the primary repair.70
Some authors have suggested different surgical techniques to augment a primary repair if it is too tenuous, using tension wire anchored to the patella or strenuous PDS augmentation.71 However the most commonly described augmentation is the one performed using semitendinosus autograft. In this technique, a tibial tunnel is normally drilled from medial to lateral to the anterior tibial tuberosity and the tendon is passed through it. A second tunnel is than drilled in the patella and the tendon is passed through it from lateral to medial. At this point the end of the tendon is sutured back to its tibial insertion with nonabsorbable stitches.72, 73 However, it is not clear if an augmentation is really needed in acute patellar tendon repair.74
Chronic setting
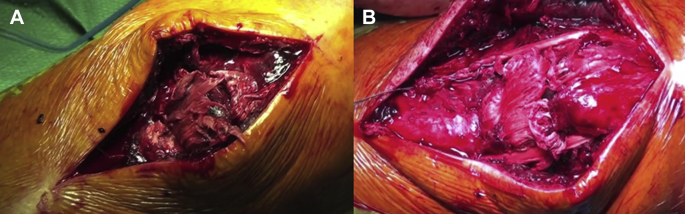
In a chronic setting, primary repair is difficult because of tendon retraction, tendon quality, and adhesion formation.64 Despite some authors describing primary repair using fascia lata or hamstring augmentation, sometimes the reconstruction is the only suitable option.75 In chronic patellar tendon rupture, hamstrings can be used to perform a reconstruction similar to the one described for augmentation. The tendons are normally left inserted, the tibial tunnel is then drilled, and the tendons are passed all around the patella in a circular or figure-of-eight fashion and then sutured back to their insertion.76, 77, 78 Different variations of the original technique were described, as a percutaneous one or a technique in which the hamstrings were passed through a longitudinal bone tunnel and fixed to the superior pole of the patella with an Endobutton (Smith & Nephew, London, UK).79, 80 However, in cases of chronic rupture, the augmentation is always suggested, even in cases in which a primary repair is possible. Figure 2 shows the results of a patellar tendon reconstruction using hamstrings.
Figure 2.
(A) Patellar tendon rupture to the proximal insertion. (B) Final results of chronic patellar tendon rupture reconstruction using hamstring.
Some authors have described the use of a contra-lateral central third of patellar tendon with proximal and distal bone plugs in cases of chronic patellar tendon rupture.81, 82 Good clinical outcomes were also reported using a Y-shaped flap folded back from the vastus lateralis fascia.83
Recently different authors suggested using allograft tissue, commonly Achilles tendon, in patellar tendon reconstruction. The bone plugs of the allograft are normally fixed to the tibial tubercle after a squared trench is performed. The tendon is split into three parts: the central one is passed through a longitudinal patellar tunnel and sutured to the superior pole of the patella and the other two parts are sutured to the lateral and medial retinaculum with nonabsorbable stitches.84 Also synthetic grafts were proposed in low-demand patients because of the low biological profile of synthetic grafts.85
Table 1 shows a literature review of outcomes in the surgical treatment of both acute and chronic patellar tendon rupture.
Table 1.
Description of studies reporting on acute or chronic patellar tendon ruptures outcomes.
| Study | No. | Median F-UP |
Technique | Results |
|---|---|---|---|---|
| Siwek & Rao75 | 31 patients (25 acute/6 chronic) | 2–11 y | Acute: Bunnell pull-out wire augmented with a Steinman Chronic: initially treated with traction |
Acute repair: 20 excellent results, 4 good, & 1 rerupture Chronic repair: 2 excellent results, 3 good, & 1 unsatisfactory result |
| Dejour et al82 | 13 (chronic) | 8.6 mo | Contra lateral bone-patellar-bone tendon | Correction of the extensor lags in all the cases & mean postoperative flexion of 91° |
| Hsu et al69 | 35 patients (all acute) | Not reported | Primary repair with a neutralisation wire | 57% of outcomes excellent; 28.6% good; & 14.2% unsatisfactory. No rerupture of the tendon |
| Marder & Timmerman74 | 14 patients (acute) | 2.6 y | Primary repair with Krackow & patella tunnels | 86% excellent results with patients resuming their previous activity level |
| Kasten et al71 | 32 patients | 8.1 (1–18) y | End-to-end suture augmented with wire cerclage or PDS | 2 of 22 patients had an extension lag in Group A (wire cerclage) compared with no extension lag in Group B (PDS cerclage). The average Hospital for Special Surgery Knee Score was 92 (SD 17) in Group A & 96 (SD 12) in Group B. 3 patients were dissatisfied |
| Bhargava et al86 | 11 patients (acute) | 26 (14–38) mo | Suture repair of the patella tendon & retinacula protection with a cerclage wire | No patients had fixed flexion deformities or demonstrated an extensor lag & the average range of motion was 0–137°. Mean loss of power of 6% (range, 2–11%) when measuring concentric extension & a mean loss of 7% power in concentric flexion |
| Ramseier et al65 | 19 | 57 mo | Suture repair associated with McLaughlin cerclage | No difference between the range of motion & muscle strength when the injured leg was compared to the noninjured leg |
| Bushnell et al67 | 14 | 29 mo | Suture anchors | No intraoperative complication, 3 failures (21%), mean extension gap 1° |
| West et al87 | 30 | 4 (1–12) y | Trans-patellar repair with nonabsorbable wire augmentation | No complication. No evidence of shortening or lengthening of the tendon. At 6 months, 40 had full extension, & 10 lacked 3–10° of active extension (not specified if quadriceps or patellar tendon) |
| Massoud88 | 12 | 45 (24–48) mo | Tran patellar suture reinforced with a “suture line tension-regulating suture” | The active knee movement averaged 0–154.6° compared with 0–156.7° in the contra lateral knee |
| Wiegand et al83 | 16 | 28 (16–36) mo | Y-shaped flap folded back from the vastus lateralis fascia | The mean Knee Society pain & function scores as well as the average range of motion of the knee increased significantly after the operations |
| Maffulli et al76 | 19 | 5.8 (4–7.8) y | Ipsilateral hamstring tendon graft reconstruction | The mean modified Cincinnati & Kujala scores were notably improved. All patients had returned to ordinary daily activities. 14 patients were very satisfied, 3 were satisfied, 1 was moderately satisfied, & 1 was unsatisfied |
| Jain et al79 | 9 | 4.5 y | Percutaneos semintendinosus reconstruction | At Lysholm score & Siwek & Rao grading were good to excellent in all patients, & at 12 months, all patients showed quadriceps strength ≥ 80% of opposite quadriceps |
Conclusion
Chronic patellar tendinopathy is frequent, most of all in athletes. The physiopathology of tendinopathy is still unclear, despite the amount of studies published, as well as the role of different inflammatory pathways. There is no standardised treatment protocol in patellar tendinopathy, with few high-level studies. Rehabilitation plays a fundamental role in patellar tendinopathy treatment, but other different techniques, such as PRP injection, were recently proposed.
For patellar tendon rupture, both in an acute setting and chronic setting, treatment indications are clearer.
In an acute setting, a direct repair using transpatellar bone tunnels is the gold standard treatment, and it can be associated with an augmentation. In a chronic setting, patellar tendon reconstruction is often the best choice. However the best graft choice or surgical technique is still unclear.
Conflicts of interest
The authors have no conflicts of interest relevant to this article.
Funding/support
No financial or material support of any kind was received for the work described in this article.
References
- 1.Maffulli N., Khan K.M., Puddu G. Overuse tendon conditions: time to change a confusing terminology. Arthroscopy. 1998;14:840–843. doi: 10.1016/s0749-8063(98)70021-0. [DOI] [PubMed] [Google Scholar]
- 2.Blazina M.E., Kerlan R.K., Jobe F.W., Carter V.S., Carlson G.J. Jumper's knee. Orthop Clin North Am. 1973;4:665–678. [PubMed] [Google Scholar]
- 3.el Hawary R., Stanish W.D., Curwin S.L. Rehabilitation of tendon injuries in sport. Sports Med. 1997;24:347–358. doi: 10.2165/00007256-199724050-00006. [DOI] [PubMed] [Google Scholar]
- 4.Lian O.B., Engebretsen L., Bahr R. Prevalence of jumper's knee among elite athletes from different sports: a cross-sectional study. Am J Sports Med. 2005;33:561–567. doi: 10.1177/0363546504270454. [DOI] [PubMed] [Google Scholar]
- 5.Gaida J.E., Cook J. Treatment options for patellar tendinopathy: critical review. Curr Sports Med Rep. 2011;10:255–270. doi: 10.1249/JSR.0b013e31822d4016. [DOI] [PubMed] [Google Scholar]
- 6.Zhang J., Wang J.H. The effects of mechanical loading on tendons–an in vivo and in vitro model study. PLoS One. 2013;8:e71740. doi: 10.1371/journal.pone.0071740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ferretti A., Ippolito E., Mariani P., Puddu G. Jumper's knee. Am J Sports Med. 1983;11:58–62. doi: 10.1177/036354658301100202. [DOI] [PubMed] [Google Scholar]
- 8.Lavagnino M., Arnoczky S.P., Elvin N., Dodds J. Patellar tendon strain is increased at the site of the jumper's knee lesion during knee flexion and tendon loading: results and cadaveric testing of a computational model. Am J Sports Med. 2008;36:2110–2118. doi: 10.1177/0363546508322496. [DOI] [PubMed] [Google Scholar]
- 9.Morrey M.E., Dean B.J.F., Carr A.J., Morrey B.F. Tendinopathy: same disease different results-why? Oper Tech Orthop. 2013;23:39–49. [Google Scholar]
- 10.Dean B.J., Gwilym S.E., Carr A.J. Why does my shoulder hurt? A review of the neuroanatomical and biochemical basis of shoulder pain. Br J Sports Med. 2013;47:1095–1104. doi: 10.1136/bjsports-2012-091492. [DOI] [PubMed] [Google Scholar]
- 11.Abrahams Y., Laguette M.J., Prince S., Collins M. Polymorphisms within the COL5A1 3'-UTR that alters mRNA structure and the MIR608 gene are associated with Achilles tendinopathy. Ann Hum Genet. 2013;77:204–214. doi: 10.1111/ahg.12013. [DOI] [PubMed] [Google Scholar]
- 12.El Khoury L., Posthumus M., Collins M., Handley C.J., Cook J., Raleigh S.M. Polymorphic variation within the ADAMTS2, ADAMTS14, ADAMTS5, ADAM12 and TIMP2 genes and the risk of Achilles tendon pathology: a genetic association study. J Sci Med Sport. 2013;16:493–498. doi: 10.1016/j.jsams.2013.02.006. [DOI] [PubMed] [Google Scholar]
- 13.Visentini P.J., Khan K.M., Cook J.L., Kiss Z.S., Harcourt P.R., Wark J.D. The VISA score: an index of severity of symptoms in patients with jumper's knee (patellar tendinosis). Victorian Institute of Sport Tendon Study Group. J Sci Med Sport. 1998;1:22–28. doi: 10.1016/s1440-2440(98)80005-4. [DOI] [PubMed] [Google Scholar]
- 14.Cook J.L., Khan K.M., Kiss Z.S., Purdam C.R., Griffiths L. Reproducibility and clinical utility of tendon palpation to detect patellar tendinopathy in young basketball players. Victorian Institute of Sport tendon study group. Br J Sports Med. 2001;35:65–69. doi: 10.1136/bjsm.35.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Peers K.H., Lysens R.J. Patellar tendinopathy in athletes: current diagnostic and therapeutic recommendations. Sports Med. 2005;35:71–87. doi: 10.2165/00007256-200535010-00006. [DOI] [PubMed] [Google Scholar]
- 16.Rasmussen O.S. Sonography of tendons. Scand J Med Sci Sports. 2000;10:360–364. doi: 10.1034/j.1600-0838.2000.010006360.x. [DOI] [PubMed] [Google Scholar]
- 17.Campbell R.S., Grainger A.J. Current concepts in imaging of tendinopathy. Clin Radiol. 2001;56:253–267. doi: 10.1053/crad.2000.0653. [DOI] [PubMed] [Google Scholar]
- 18.Ohberg L., Lorentzon R., Alfredson H. Neovascularisation in Achilles tendons with painful tendinosis but not in normal tendons: an ultrasonographic investigation. Knee Surg Sports Traumatol Arthrosc. 2001;9:233–238. doi: 10.1007/s001670000189. [DOI] [PubMed] [Google Scholar]
- 19.Weatherall P.T., Crues J.V., 3rd Musculotendinous injury. Magn Reson Imaging Clin N Am. 1995;3:753–772. [PubMed] [Google Scholar]
- 20.Wang C.J., Ko J.Y., Chan Y.S., Weng L.H., Hsu S.L. Extracorporeal shockwave for chronic patellar tendinopathy. Am J Sports Med. 2007;35:972–978. doi: 10.1177/0363546506298109. [DOI] [PubMed] [Google Scholar]
- 21.van Leeuwen M.T., Zwerver J., van den Akker-Scheek I. Extracorporeal shockwave therapy for patellar tendinopathy: a review of the literature. Br J Sports Med. 2009;43:163–168. doi: 10.1136/bjsm.2008.050740. [DOI] [PubMed] [Google Scholar]
- 22.Zwerver J., Dekker F., Pepping G.J. Patient guided piezo-electric extracorporeal shockwave therapy as treatment for chronic severe patellar tendinopathy: a pilot study. J Back Musculoskelet Rehabil. 2010;23:111–115. doi: 10.3233/BMR-2010-0257. [DOI] [PubMed] [Google Scholar]
- 23.Zwerver J., Hartgens F., Verhagen E., van der Worp H., van den Akker-Scheek I., Diercks R.L. No effect of extracorporeal shockwave therapy on patellar tendinopathy in jumping athletes during the competitive season: a randomized clinical trial. Am J Sports Med. 2011;39:1191–1199. doi: 10.1177/0363546510395492. [DOI] [PubMed] [Google Scholar]
- 24.Peers K.H., Lysens R.J., Brys P., Bellemans J. Cross-sectional outcome analysis of athletes with chronic patellar tendinopathy treated surgically and by extracorporeal shock wave therapy. Clin J Sport Med. 2003;13:79–83. doi: 10.1097/00042752-200303000-00003. [DOI] [PubMed] [Google Scholar]
- 25.van der Worp H., van den Akker-Scheek I., van Schie H., Zwerver J. ESWT for tendinopathy: technology and clinical implications. Knee Surg Sports Traumatol Arthrosc. 2013;21:1451–1458. doi: 10.1007/s00167-012-2009-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Steunebrink M., Zwerver J., Brandsema R., Groenenboom P., van den Akker-Scheek I., Weir A. Topical glyceryl trinitrate treatment of chronic patellar tendinopathy: a randomised, double-blind, placebo-controlled clinical trial. Br J Sports Med. 2013;47:34–39. doi: 10.1136/bjsports-2012-091115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Volpi P., Marinoni L., Bait C., De Girolamo L., Schoenhuber H. Treatment of chonic patellar tendinosis with buffered platelet rich plasma: a preliminary study. Med Sport. 2007;60:595–603. [Google Scholar]
- 28.Kon E., Filardo G., Delcogliano M. Platelet-rich plasma: new clinical application: a pilot study for treatment of jumper's knee. Injury. 2009;40:598–603. doi: 10.1016/j.injury.2008.11.026. [DOI] [PubMed] [Google Scholar]
- 29.Filardo G., Kon E., Della Villa S., Vincentelli F., Fornasari P.M., Marcacci M. Use of platelet-rich plasma for the treatment of refractory jumper's knee. Int Orthop. 2010;34:909–915. doi: 10.1007/s00264-009-0845-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.de Vos R.J., Weir A., van Schie H.T. Platelet-rich plasma injection for chronic Achilles tendinopathy: a randomized controlled trial. JAMA. 2010;303:144–149. doi: 10.1001/jama.2009.1986. [DOI] [PubMed] [Google Scholar]
- 31.van Ark M., Zwerver J., van den Akker-Scheek I. Injection treatments for patellar tendinopathy. Br J Sports Med. 2011;45:1068–1076. doi: 10.1136/bjsm.2010.078824. [DOI] [PubMed] [Google Scholar]
- 32.Gosens T., Den Oudsten B.L., Fievez E., van't Spijker P., Fievez A. Pain and activity levels before and after platelet-rich plasma injection treatment of patellar tendinopathy: a prospective cohort study and the influence of previous treatments. Int Orthop. 2012;36:1941–1946. doi: 10.1007/s00264-012-1540-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Vetrano M., Castorina A., Vulpiani M.C., Baldini R., Pavan A., Ferretti A. Platelet-rich plasma versus focused shock waves in the treatment of jumper's knee in athletes. Am J Sports Med. 2013;41:795–803. doi: 10.1177/0363546513475345. [DOI] [PubMed] [Google Scholar]
- 34.Lane J.G., Healey R.M., Chase D.C., Amiel D. Use of platelet-rich plasma to enhance tendon function and cellularity. Am J Orthop (Belle Mead NJ) 2013;42:209–214. [PubMed] [Google Scholar]
- 35.Volpi P., Quaglia A., Schoenhuber H. Growth factors in the management of sport-induced tendinopathies: results after 24 months from treatment. A pilot study. J Sports Med Phys Fitness. 2010;50:494–500. [PubMed] [Google Scholar]
- 36.Charousset C., Zaoui A., Bellaiche L., Bouyer B. Are multiple platelet-rich plasma injections useful for treatment of chronic patellar tendinopathy in athletes? A prospective study. Am J Sports Med. 2014;42:906–911. doi: 10.1177/0363546513519964. [DOI] [PubMed] [Google Scholar]
- 37.Moraes V.Y., Lenza M., Tamaoki M.J., Faloppa F., Belloti J.C. Platelet-rich therapies for musculoskeletal soft tissue injuries. Cochrane Database Syst Rev. 2014;4:CD010071. doi: 10.1002/14651858.CD010071.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Andia I., Latorre P.M., Gomez M.C., Burgos-Alonso N., Abate M., Maffulli N. Platelet-rich plasma in the conservative treatment of painful tendinopathy: a systematic review and meta-analysis of controlled studies. Br Med Bull. 2014;110:99–115. doi: 10.1093/bmb/ldu007. [DOI] [PubMed] [Google Scholar]
- 39.Wang J.H. Can PRP effectively treat injured tendons? Muscles Ligaments Tendons J. 2014;4:35–37. [PMC free article] [PubMed] [Google Scholar]
- 40.James S.L., Ali K., Pocock C. Ultrasound guided dry needling and autologous blood injection for patellar tendinosis. Br J Sports Med. 2007;41:518–521. doi: 10.1136/bjsm.2006.034686. discussion 522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Willberg L., Sunding K., Forssblad M., Fahlstrom M., Alfredson H. Sclerosing polidocanol injections or arthroscopic shaving to treat patellar tendinopathy/jumper's knee? A randomised controlled study. Br J Sports Med. 2011;45:411–415. doi: 10.1136/bjsm.2010.082446. [DOI] [PubMed] [Google Scholar]
- 42.Hoksrud A., Torgalsen T., Harstad H. Ultrasound-guided sclerosis of neovessels in patellar tendinopathy: a prospective study of 101 patients. Am J Sports Med. 2012;40:542–547. doi: 10.1177/0363546511433012. [DOI] [PubMed] [Google Scholar]
- 43.Fredberg U., Bolvig L., Pfeiffer-Jensen M., Clemmensen D., Jakobsen B.W., Stengaard-Pedersen K. Ultrasonography as a tool for diagnosis, guidance of local steroid injection and, together with pressure algometry, monitoring of the treatment of athletes with chronic jumper's knee and Achilles tendinitis: a randomized, double-blind, placebo-controlled study. Scand J Rheumatol. 2004;33:94–101. doi: 10.1080/03009740310004126. [DOI] [PubMed] [Google Scholar]
- 44.Capasso G.T.V., Maffulli N. Aprotinin, corticosteroids and normosaline in the management of patellar tendinopathy in athletes: a prospective randomized study. Sports Exerc Injury. 1997;3:111–115. [Google Scholar]
- 45.Kongsgaard M., Kovanen V., Aagaard P. Corticosteroid injections, eccentric decline squat training and heavy slow resistance training in patellar tendinopathy. Scand J Med Sci Sports. 2009;19:790–802. doi: 10.1111/j.1600-0838.2009.00949.x. [DOI] [PubMed] [Google Scholar]
- 46.Zhang J., Keenan C., Wang J.H. The effects of dexamethasone on human patellar tendon stem cells: implications for dexamethasone treatment of tendon injury. J Orthop Res. 2013;31:105–110. doi: 10.1002/jor.22193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cucurulo T., Louis M.L., Thaunat M., Franceschi J.P. Surgical treatment of patellar tendinopathy in athletes. A retrospective multicentric study. Orthop Traumatol Surg Res. 2009;95:S78–S84. doi: 10.1016/j.otsr.2009.09.005. [DOI] [PubMed] [Google Scholar]
- 48.Willberg L., Sunding K., Ohberg L., Forssblad M., Alfredson H. Treatment of Jumper's knee: promising short-term results in a pilot study using a new arthroscopic approach based on imaging findings. Knee Surg Sports Traumatol Arthrosc. 2007;15:676–681. doi: 10.1007/s00167-006-0223-6. [DOI] [PubMed] [Google Scholar]
- 49.Ogon P., Maier D., Jaeger A., Suedkamp N.P. Arthroscopic patellar release for the treatment of chronic patellar tendinopathy. Arthroscopy. 2006;22:462.e461–462.e465. doi: 10.1016/j.arthro.2005.06.035. [DOI] [PubMed] [Google Scholar]
- 50.Lorbach O., Diamantopoulos A., Paessler H.H. Arthroscopic resection of the lower patellar pole in patients with chronic patellar tendinosis. Arthroscopy. 2008;24:167–173. doi: 10.1016/j.arthro.2007.08.021. [DOI] [PubMed] [Google Scholar]
- 51.Marcheggiani Muccioli G.M., Zaffagnini S., Tsapralis K. Open versus arthroscopic surgical treatment of chronic proximal patellar tendinopathy. A systematic review. Knee Surg Sports Traumatol Arthrosc. 2013;21:351–357. doi: 10.1007/s00167-012-2100-9. [DOI] [PubMed] [Google Scholar]
- 52.Sharma P., Maffulli N. Tendon injury and tendinopathy: healing and repair. J Bone Joint Surg Am. 2005;87:187–202. doi: 10.2106/JBJS.D.01850. [DOI] [PubMed] [Google Scholar]
- 53.Raschke D., Schuttrumpf J.P., Tezval M., Sturmer K.M., Balcarek P. Extensor-mechanism-reconstruction of the knee joint after traumatic loss of the entire extensor apparatus. Knee. 2014;21:793–796. doi: 10.1016/j.knee.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 54.Saragaglia D., Pison A., Rubens-Duval B. Acute and old ruptures of the extensor apparatus of the knee in adults (excluding knee replacement) Orthop Traumatol Surg Res. 2013;99:S67–S76. doi: 10.1016/j.otsr.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 55.Zernicke R.F., Garhammer J., Jobe F.W. Human patellar-tendon rupture. J Bone Joint Surg Am. 1977;59:179–183. [PubMed] [Google Scholar]
- 56.Kelly D.W., Carter V.S., Jobe F.W., Kerlan R.K. Patellar and quadriceps tendon ruptures–jumper's knee. Am J Sports Med. 1984;12:375–380. doi: 10.1177/036354658401200508. [DOI] [PubMed] [Google Scholar]
- 57.Clark S.C., Jones M.W., Choudhury R.R., Smith E. Bilateral patellar tendon rupture secondary to repeated local steroid injections. J Accid Emerg Med. 1995;12:300–301. doi: 10.1136/emj.12.4.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ramirez-Castillo H.D., Carbajal-Contreras R., Gonzalez-Morales D.D. Acute bilateral lesion of the patellar tendon associated to diabetes mellitus. Case report. Acta Ortop Mex. 2010;24:23–27. [PubMed] [Google Scholar]
- 59.Lu M., Johar S., Veenema K., Goldblatt J. Patellar tendon rupture with underlying systemic lupus erythematosus: a case report. J Emerg Med. 2012;43:e35–38. doi: 10.1016/j.jemermed.2009.08.054. [DOI] [PubMed] [Google Scholar]
- 60.Sullivan R.L. Traumatic bilateral patellar tendon rupture with chronic renal disease. Wis Med J. 1986;85:12–13. [PubMed] [Google Scholar]
- 61.Kartus J., Movin T., Karlsson J. Donor-site morbidity and anterior knee problems after anterior cruciate ligament reconstruction using autografts. Arthroscopy. 2001;17:971–980. doi: 10.1053/jars.2001.28979. [DOI] [PubMed] [Google Scholar]
- 62.Maffulli N., Wong J. Rupture of the Achilles and patellar tendons. Clin Sports Med. 2003;22:761–776. doi: 10.1016/s0278-5919(03)00009-7. [DOI] [PubMed] [Google Scholar]
- 63.Heyde C.E., Mahlfeld K., Stahel P.F., Kayser R. Ultrasonography as a reliable diagnostic tool in old quadriceps tendon ruptures: a prospective multicentre study. Knee Surg Sports Traumatol Arthrosc. 2005;13:564–568. doi: 10.1007/s00167-004-0576-7. [DOI] [PubMed] [Google Scholar]
- 64.Lee D., Stinner D., Mir H. Quadriceps and patellar tendon ruptures. J Knee Surg. 2013;26:301–308. doi: 10.1055/s-0033-1353989. [DOI] [PubMed] [Google Scholar]
- 65.Ramseier L.E., Werner C.M., Heinzelmann M. Quadriceps and patellar tendon rupture. Injury. 2006;37:516–519. doi: 10.1016/j.injury.2005.12.014. [DOI] [PubMed] [Google Scholar]
- 66.Capiola D., Re L. Repair of patellar tendon rupture with suture anchors. Arthroscopy. 2007;23:906.e901–906.e904. doi: 10.1016/j.arthro.2006.10.023. [DOI] [PubMed] [Google Scholar]
- 67.Bushnell B.D., Byram I.R., Weinhold P.S., Creighton R.A. The use of suture anchors in repair of the ruptured patellar tendon: a biomechanical study. Am J Sports Med. 2006;34:1492–1499. doi: 10.1177/0363546506287489. [DOI] [PubMed] [Google Scholar]
- 68.Ettinger M., Dratzidis A., Hurschler C. Biomechanical properties of suture anchor repair compared with transosseous sutures in patellar tendon ruptures: a cadaveric study. Am J Sports Med. 2013;41:2540–2544. doi: 10.1177/0363546513500633. [DOI] [PubMed] [Google Scholar]
- 69.Hsu K.Y., Wang K.C., Ho W.P., Hsu R.W. Traumatic patellar tendon ruptures: a follow-up study of primary repair and a neutralization wire. J Trauma. 1994;36:658–660. [PubMed] [Google Scholar]
- 70.Giles S.N., Morgan-Jones R., Brown M.F. The use of hinged Kirschner wires for fixation of patellar tendon rupture. Injury. 1999;30:539–540. doi: 10.1016/s0020-1383(99)00147-3. [DOI] [PubMed] [Google Scholar]
- 71.Kasten P., Schewe B., Maurer F., Gosling T., Krettek C., Weise K. Rupture of the patellar tendon: a review of 68 cases and a retrospective study of 29 ruptures comparing two methods of augmentation. Arch Orthop Trauma Surg. 2001;121:578–582. doi: 10.1007/s004020100298. [DOI] [PubMed] [Google Scholar]
- 72.Kinmont J.C., Walter E., Curtis M.J. Augmentation of patellar tendon repair with poly-p-dioxannone cord. Injury. 2002;33:263–264. doi: 10.1016/s0020-1383(00)00197-2. [DOI] [PubMed] [Google Scholar]
- 73.Larson R.V., Simonian P.T. Semitendinosus augmentation of acute patellar tendon repair with immediate mobilization. Am J Sports Med. 1995;23:82–86. doi: 10.1177/036354659502300114. [DOI] [PubMed] [Google Scholar]
- 74.Marder R.A., Timmerman L.A. Primary repair of patellar tendon rupture without augmentation. Am J Sports Med. 1999;27:304–307. doi: 10.1177/03635465990270030601. [DOI] [PubMed] [Google Scholar]
- 75.Siwek C.W., Rao J.P. Ruptures of the extensor mechanism of the knee joint. J Bone Joint Surg Am. 1981;63:932–937. [PubMed] [Google Scholar]
- 76.Maffulli N., Del Buono A., Loppini M., Denaro V. Ipsilateral hamstring tendon graft reconstruction for chronic patellar tendon ruptures: average 5.8-year follow-up. J Bone Joint Surg Am. 2013;95:e1231–1236. doi: 10.2106/JBJS.L.01462. [DOI] [PubMed] [Google Scholar]
- 77.Harris J.D., Fazalare J.J., Phieffer L.S., Flanigan D.C. Patellar tendon reconstruction with semitendinosus-gracilis autograft. J Knee Surg. 2013;26:S19–S24. doi: 10.1055/s-0031-1280972. [DOI] [PubMed] [Google Scholar]
- 78.Chen B., Li R., Zhang S. Reconstruction and restoration of neglected ruptured patellar tendon using semitendinosus and gracilis tendons with preserved distal insertions: two case reports. Knee. 2012;19:508–512. doi: 10.1016/j.knee.2011.07.007. [DOI] [PubMed] [Google Scholar]
- 79.Jain J.K., Vidyasagar J.V., Chabra R. Percutaneous reconstruction of patellar tendon using semitendinosus tendon in chronic patellar tendon injury–case series and outcome. Knee. 2014;21:726–730. doi: 10.1016/j.knee.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 80.Gokce A., Ekici H., Erdogan F. Arthroscopic reconstruction of a ruptured patellar tendon: a technical note. Knee Surg Sports Traumatol Arthrosc. 2008;16:581–584. doi: 10.1007/s00167-008-0511-4. [DOI] [PubMed] [Google Scholar]
- 81.Milankov M.Z., Miljkovic N., Stankovic M. Reconstruction of chronic patellar tendon rupture with contralateral BTB autograft: a case report. Knee Surg Sports Traumatol Arthrosc. 2007;15:1445–1448. doi: 10.1007/s00167-007-0365-1. [DOI] [PubMed] [Google Scholar]
- 82.Dejour H., Denjean S., Neyret P. Treatment of old or recurrent ruptures of the patellar ligament by contralateral autograft. Rev Chir Orthop Reparatrice Appar Mot. 1992;78:58–62. [PubMed] [Google Scholar]
- 83.Wiegand N., Naumov I., Vamhidy L., Warta V., Than P. Reconstruction of the patellar tendon using a Y-shaped flap folded back from the vastus lateralis fascia. Knee. 2013;20:139–143. doi: 10.1016/j.knee.2012.05.008. [DOI] [PubMed] [Google Scholar]
- 84.Burks R.T., Edelson R.H. Allograft reconstruction of the patellar ligament. A case report. J Bone Joint Surg Am. 1994;76:1077–1079. doi: 10.2106/00004623-199407000-00019. [DOI] [PubMed] [Google Scholar]
- 85.Naim S., Gougoulias N., Griffiths D. Patellar tendon reconstruction using LARS ligament: surgical technique and case report. Strategies Trauma Limb Reconstr. 2011;6:39–41. doi: 10.1007/s11751-010-0101-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bhargava S.P., Hynes M.C., Dowell J.K. Traumatic patella tendon rupture: early mobilisation following surgical repair. Injury. 2004;35:76–79. doi: 10.1016/s0020-1383(03)00069-x. [DOI] [PubMed] [Google Scholar]
- 87.West J.L., Keene J.S., Kaplan L.D. Early motion after quadriceps and patellar tendon repairs: outcomes with single-suture augmentation. Am J Sports Med. 2008;36:316–323. doi: 10.1177/0363546507308192. [DOI] [PubMed] [Google Scholar]
- 88.Massoud E.I. Repair of fresh patellar tendon rupture: tension regulation at the suture line. Int Orthop. 2010;34:1153–1158. doi: 10.1007/s00264-009-0879-x. [DOI] [PMC free article] [PubMed] [Google Scholar]