Abstract
Objective
To investigate the effects of prostate cancer (PCa) surgery on the stress system and to identify potential independent factors associating with stress recovery.
Methods
The design of the study was prospective and PCa surgery included robot assisted radical prostatectomy (RARP) or retropubic radical prostatectomy (RRP). Between February 2013 to December 2014, 315 consecutive patients were evaluated. The effects of PCa surgery on the stress system were measured by cortisol serum levels before and after surgery on postoperative day (POD) 0, 1, 3, 5 and 45. Cortisol variations in the population and subpopulation (RARP vs. RRP) of patients were investigated by statistical methods. Factors associating with stress recovery were assessed by simple linear regression (SLR) and multiple linear regression (MLR) analysis.
Results
RARP was performed in 75.9% of cases. In the patient population, there were wide serum cortisol perioperative variations. PCa surgery triggered the stress system which immediately (POD 0) responded by cortisol overproduction which induced the negative feedback mechanism that started on POD 1, continued on POD 3, was still ongoing on POD 5 and completely settled on POD 45 (stress recovery). In the subpopulation of patients, significantly lower cortisol serum levels were detected on POD 3–5 in RARP cases in whom cortisol levels were close to preoperative levels (stress recovery) on POD 5. Independent predictive factors of serum cortisol on POD 5 (stress recovery) were preoperative cortisol (p = 0.02), cortisol levels on POD 3 (p < 0.0001) and RARP (p = 0.03) in which the association was negative (stress recovery faster than RRP).
Conclusion
Our study shows that PCa surgery immediately (POD 0) triggers the stress system which respond by overproduction of cortisol which induces the negative feedback mechanism that starts on POD 1, is still ongoing on POD 5, but is completely settled on POD 45. Moreover, after surgical trauma, our study gives evidence that the RARP procedure associates with stress recovery faster than RRP. Further confirmatory studies are required.
Keywords: Prostate cancer, Robot assisted radical prostatectomy, Retropubic radical prostatectomy, Stress system, Hypothalamus–pituitary–adrenal axis, Cortisol hormone
1. Introduction
Prostate cancer (PCa) is the second most frequent solid tumor after lung cancer in adult males [1] and associates with a mortality rate of 1.2% [2]. Radical prostatectomy (RP) is the most performed procedure for treating clinically localized PCa in men who have life expectancy more than 10 years, associates with extended lymph node dissection (LND) in high risk patients [3], and aims to eradicate cancer disease, to preserve urinary continence, and potency as well [3], [4]. RP may be performed by the open retropubic approach (RRP) or by minimally invasive laparoscopic techniques which include the laparoscopic RP (LRP) and robot assisted RP (RARP) techniques [3], [4], [5]. Actually, RARP is increasingly displacing RRP all over the world [3], [4], and associates with lesser blood loss than RRP. However, postoperative complications related to RARP and RRP appear similar. Although there are no prospective randomized trials comparing RRP vs. RARP, experts consider RARP a valuable therapeutic option for treating clinically localized PCa [5].
It has been shown that major surgery trauma induces profound immunological dysfunction [6] by triggering the stress system which is represented by the hypothalamic–pituitary–adrenal (HPA) axis [7], [8], [9]. The HPA axis is a neuroendocrine system that regulates the circulating levels of glucocorticoid hormones (mainly cortisol) that are important for homeostasis and play a critical role in stress response [7], [8], [9].
The hormones of the stress system include the corticotrophin-releasing hormone (CRH) which is released by the hypothalamus, the adrenocorticotropic hormone (ACTH) which is produced by the pituitary and cortisol which is the main glucocorticoid hormone secreted by the adrenals. Approximately, 95% of plasma cortisol is bound to proteins that include the cortisol binding globulin (CBG, 95%) and albumin (10%–15%); moreover, the remaining 5% is unbound, free to cross cell membranes and binds to glucocorticoid and mineralocorticoid receptors [9].
Major surgery is a significant challenge to homeostasis because it alters the steady state of neuroendocrine systems of which the most pivotal is the HPA axis [9]. Clinical models and studies investigating on the stress system after major surgery are actually unknown. Although it has been shown that RARP is less invasive than RRP [10], studies dealing with the effects of RARP and RRP related to the stress system are missing. Actually, the pathophysiology of the HPA axis after PCa surgery is an unexplored field because we are missing clinical models which are important for progressing and improving care. The present study explores the pathophysiology of the stress system by RARP and RRP as well as complications related to the HPA axis by the two procedures.
2. Patients and methods
The study relates to a period that ranges from February 2013 to October 2014 and includes 315 consecutive patients who underwent RP by RARP or RRP.
Exclusion criteria were as follows: (i) sarcoma of the prostate, (ii) preceding focal therapies, (iii) androgen blockade, (iv) history of glucocorticoid medications or adrenal surgery. The choice of the procedure, RARP or RRP, was based on a joint decision by urologists and patients who were sharply informed and provided written consent to surgery as well as to the use of their data. Robot assisted RP was delivered by the three-arm da Vinci Robot System (Intuitive Surgical, Inc, Sunnyvale, CA, USA) and performed by two experienced surgeons who were experienced in both techniques. The approach was transperitoneal with prostatic dissection in all cases [11]. RRP was performed by experienced surgeons according to the Walsh technique [12]. According to the D'Amico class risk classification [13], LND was planned in all high risk patients and intermediate risk cases with a risk of metastases ≥5% according to the Briganti's nomogram [14]. The protocol after surgery was standardized for both procedures and patients were discharged on postoperative day (POD) 4–6 when there were no further complications. The peri-operative surgical risk of patients was evaluated by the American Society of Anesthesiologists (ASA) score system [15]. The complications were classified and coded according to Clavien–Dindo system (CDS) [16].
The study is part of a larger trial of PCa biobanking which has been already approved by the local Ethical Committee. The design was prospective and the data of each case were collected in one data base. The evaluated variables included age (years), body mass index (BMI, kg/m2), ASA score, baseline total prostate specific antigen (PSA) serum levels (ng/mL), proportion of positive cores (P+), prostate volume (PV, mL), operating time (OT, min), blood lost (BL, mL), clinical stage (cT), biopsy Gleason score (bGS), pathology Gleason score (pGS), pathologic tumor stage (pT), lymph node status (pN) and surgical margin status (R, 1 positive, 0 negative). PV was estimated by the ellipsoid method, using prostate diameters calculated from TRUS. In patients undergoing RARP, OT was calculated from the insertion of the Veress needle to the suture of the last laparoscopic port, whereas in RRP it was computed from skin incision to suture. Patients were classified according to primary tumor stage, lymph node and metastatic status, using the TNM categories recommended by the 1997 International Union against Cancer TNM classification system [17]. Finally, the discharge day (DD, days) was measured as a continuous variable and reported as median and range, while readmission to the hospital (R-AD) was coded as a categorical variable.
Serum cortisol concentrations were measured on an Immulite 2000 automated immunoassay analyzer (Siemens Healthcare, Marburg, Germany) by the Immulite 2000 Cortisol assay. The Immulite 2000 Cortisol is a solid-phase, competitive chemiluminescent enzyme immunoassay, displaying a within-run imprecision of 4.9%–6.7% and 5.3%–10.8%, respectively [18]. Cortisol venous blood samples were taken 20–30 days before surgery at 8:00 a.m. (Tb), 4 h after surgery (T0), on POD 1 (T1), 3 (T3), 5 (T5) and 45 (T45). Serum levels of cortisol were expressed by ng/mL (normal range: 4.0–25.0).
Statistical methods. It was used the advanced SPSS statistical software (IBM, Chicago, IL, USA). Data on continuous variables are reported as medians with their respective ranges, and differences between groups analyzed with the Mann–Whitney U test. Data on categorical variables are presented as proportions, and differences between groups analyzed with Pearson's chi-squared test or Fisher exact test as appropriate.
To generate logistic simple logistic regression models of predictors associating with CDS ≥ 3a, all continuous variables were entered as continuous variables unless otherwise stated. Only variables that were significant in univariate models were included in the multivariable analysis. To generate simple linear regression models of covariates associating with DD, all continuous variables were entered as continuous variables unless otherwise stated. Only variables that were significant in univariate models were included in the multivariable analysis. Where significant co-linearity existed between independent variables, the one with the most predictive power was used in preference in the multivariate model. Multivariate models were generated using a simple stepwise model.
All tests were two-sided with p < 0.05 considered to indicate statistical significance.
3. Results
After excluding criteria, 290 patients were evaluated. Surgery included RARP in 220 patients (75.9%) and RRP in 70 cases (24.1%). Table 1 shows the demographics of the cohort study. Patients who underwent RARP were younger (p = 0.002), had lower median PSA levels (p < 0.0001) and showed higher ASA 2 score rates (p = 0.009). Patients who underwent RRP had higher median BL (p < 0.0001), showed higher perioperative complications (p < 0.0001) and R-AD (p < 0.0001) rates than RARP cases who had lower median values of length of hospital stay (p < 0.0001).
Table 1.
Preoperative and perioperative variables related to the cohort study.
| Variable | Population n = 290 |
Subpopulations |
p-Value | |
|---|---|---|---|---|
| RARP n = 220 (75.9%) |
RRP n = 70 (24.1%) |
|||
| Age (year) | 65 (43–76) | 64 (43–76) | 67 (53–76) | 0.002 |
| PSA (ng/mL) | 6.3 (0.8–94.3) | 5.8 (0.8–37.2) | 8.3 (2.3–94.3) | <0.0001 |
| BMI (kg/m2) | 26.1 (18.5–35.4) | 26.0 (18.5–35.4) | 26.2 (20.3–32.9) | 0.236 |
| PV (mL) | 36 (15–130) | 35 (15–120) | 40 (17–130) | 0.733 |
| ASA score | 0.009 | |||
| 1 | 17 (5.9) | 15 (6.8) | 2 (2.9) | |
| 2 | 239 (82.4) | 187 (85.0) | 52 (74.3) | |
| 3 | 31 (10.7) | 17 (7.7) | 14 (20.0) | |
| 4 | 2 (0.7) | 1 (0.5) | 1 (1.4) | |
| 5 | 1 (0.3) | 0 (0) | 1 (1.4) | |
| cT | 0.014 | |||
| 1c | 181 (62.4) | 146 (50.3) | 35 (12.1) | |
| 2 | 109 (37.6) | 74 (25.5) | 35 (12.1) | |
| bGS | <0.0001 | |||
| 6 | 153 (52.8) | 139 (47.9) | 14 (4.8) | |
| 7 | 108 (37.2) | 67 (23.1) | 41 (14.1) | |
| >7 | 29 (10.0) | 14 (4.8) | 15 (5.2) | |
| LND | <0.0001 | |||
| Yes | 96 (33.1) | 33 (11.4) | 63 (21.7) | |
| No | 194 (66.9) | 187 (64.5) | 7 (2.4) | |
| pGS | <0.0001 | |||
| 6 | 68 (23.4) | 61 (21) | 7 (2.4) | |
| 7 | 164 (56.6) | 128 (44.1) | 36 (12.4) | |
| >7 | 58 (20) | 31 (10.7) | 27 (9.3) | |
| pT | <0.0001 | |||
| 2 | 208 (71.7) | 176 (60.7) | 32 (11) | |
| 3a | 39 (13.4) | 24 (8.3) | 15 (5.2) | |
| 3b | 43(14.8) | 20 (6.9) | 23 (7.9) | |
| pN | <0.0001 | |||
| 0 | 77 (26.6) | 29 (10) | 48 (16.6) | |
| 1 | 20 (6.9) | 5 (1.7) | 15 (5.2) | |
| x | 193 (66.6) | 186 (64.1) | 7 (2.4) | |
| OT (min) | 180 (75–410) | 185 (75–400) | 177.5 (100–400) | 0.054 |
| BL (mL) | 400 (100–3500) | 400 (100–3500) | 500 (200–1600) | <0.0001 |
| Clavien–Dindo | <0.0001 | |||
| 0 | 220 (75.9) | 185 (63.8) | 35 (12.1) | |
| 1c | 32 (11.0) | 18 (6.2) | 14 (4.8) | |
| 2 | 21 (7.2) | 14 (4.8) | 7 (2.4) | |
| 3a | 8 (2.8) | 2 (0.7) | 6 (2.1) | |
| 3b | 7 (2.4) | 1 (0.3) | 1 (0.3) | |
| 4a | 1 (0.3) | 0 (0) | 1 (0.3) | |
| 5 | 1 (0.3) | 0 (0) | 1 (0.3) | |
| R-AD | <0.0001 | |||
| Yes | 15 (5.2) | 3 (1.0) | 12 (4.1) | |
| No | 275 (94.8) | 217 (74.8) | 58 (20) | |
| LOHS (DD), day | 6 (3–17) | 6 (3–12) | 7 (4–17) | <0.0001 |
Continuous variables are reported as median (range) and categorical as n (%).
ASA, American Society of Anesthesiologists; bGS, biopsy Gleason score; BL, blood lost; BMI, body mass index; cT, clinical stage; DD, discharge day; LND, lymph node dissection; LOHS, length of hospital stay; OT, operating time; pGS, pathology Gleason score; pN, lymph node status; PSA, prostate specific antigen, pT: pathologic tumor stage; PV, prostate volume; R-AD, readmission to the hospital; RARP, robot assisted radical prostatectomy; RRP, retropubic radical prostatectomy.
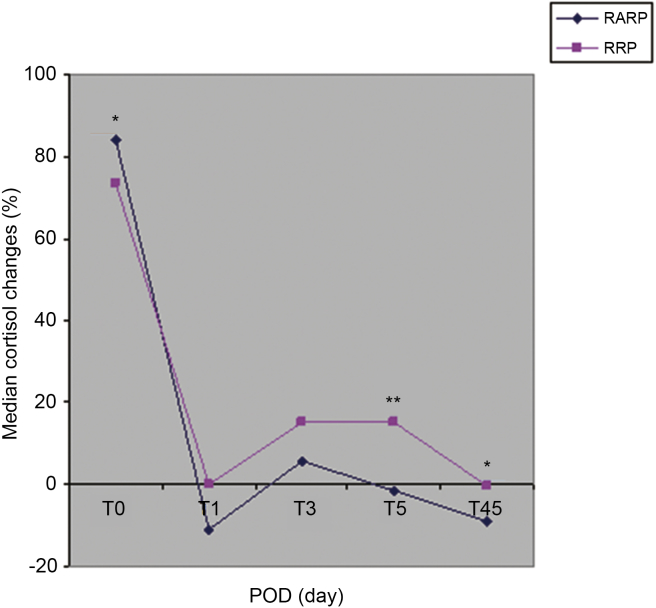
Table 2 shows the perioperative kinetics of cortisol plasma levels. Preoperative basal levels of cortisol did not differ between RARP and RRP cases. Median cortisol plasma levels detected 4 h after surgery were significantly lower in RRP cases than RARP patients who however in the subsequent perioperative period showed lower median cortisol plasma levels on POD 5 (p < 0.0001) and 45 (p < 0.0001). Fig. 1 depicts the variations (%) of cortisol plasma levels as follows: basal, perioperative period and POD 45. As reported, significant variations were detected at T0, T5 and T45.
Table 2.
Basal and perioperative cortisol plasma levels in PCa patients undergoing surgery (median (range)).
| Cortisol (ng/mL) | RARP | RRP | p-Value |
|---|---|---|---|
| T | 20 (4–46) | 15 (4–27) | 0.33 |
| T0 | 29.1 (3.9–54.1) | 26.2 (2.8–46.5) | 0.002 |
| T1 | 14.1 (1.2–31.4) | 14.6 (0.9–48.1) | 0.777 |
| T3 | 16.7 (1.1–30.9) | 17.4 (0.9–27.4) | 0.05 |
| T5 | 15.6 (6.2–28.7) | 16.9 (9.6–31.1) | <0.0001 |
| T45 | 14.4 (5.6–33.8) | 15.0 (8.5–22.5) | <0.0001 |
PCa, prostate cancer; POD, postoperative day; RARP, robot assisted radical prostatectomy; RRP, retropubic radical prostatectomy; T, basal cortisol; T0, cortisol 4 h after surgery; T1, cortisol on POD 1; T3, cortisol on POD 3; T5, cortisol on POD 5; T45, cortisol on POD 45.
Figure 1.
Median cortisol perioperative changes (as percentage) from basal preoperative levels. *p < 0.05, **p< 0.001. RARP, robot assisted radical prostatectomy; RRP, retropubic radical prostatectomy; T0, cortisol 4 h after surgery; T1, cortisol on POD 1; T3, cortisol on POD 3; T5, cortisol on POD 5; T45, cortisol on POD 45.
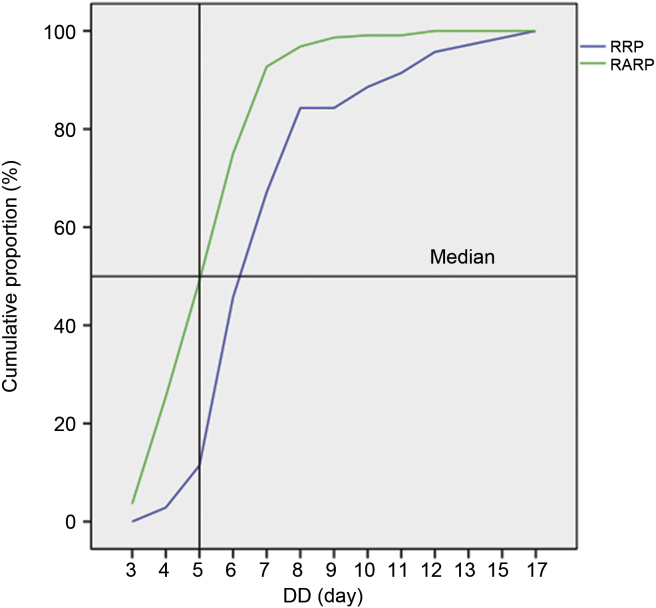
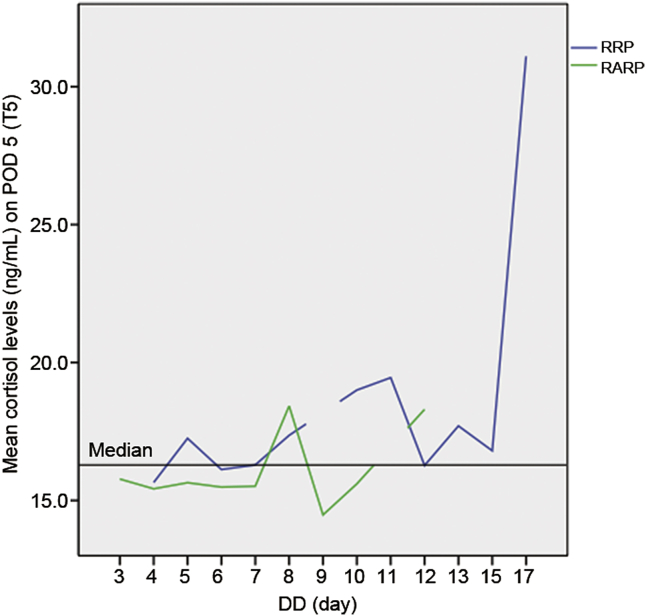
Table 3 shows the logistic regression models predicting a Clavien–Dindo complication score ≥ 3a. The multivariate logistic regression model showed that only DD (p < 0.0001) and BL (p < 0.0001) were independent predictors associating with CDS ≥ 3a; however, the former showed a stronger association (OR = 1.425; 95%CI: 1.168–1.739) than the latter (OR = 1.002; 95%CI: 1.001–1.003). Because of the strong association of LOHS with the CDS, we computed a linear regression analysis in order to detect potential covariates predicting DD. As shown in Table 4, analysis of simple models showed that DD directly associated with LND (p < 0.0001), age (p = 0.015), ASA ≥ 3, cortisol levels on POD 1 (p = 0.023), and cortisol plasma levels on POD 5 (p < 0.0001), but inversely with RARP (p < 0.0001). However, analysis of the multivariate model showed that independent covariates predicting DD were ASA ≥ 3 (b = 1.22; p < 0.0001), CDS ≥ 3a (b = 1.253; p < 0.0001), cortisol plasma levels on POD 5 (b = 0.097; p = 0.002), and RARP (b = –0.901; p = 0.005). As depicted in Fig. 2, the median length of DD was significantly shorter in RARP cases (5 days) than RRP patients (6 days) who showed, as illustrated in Fig. 3, significantly higher median plasma levels of T5 cortisol.
Table 3.
Covariates associating with Clavien–Dindo score ≥ 3a by logistic regression analysis
| Variable | Univariate analysis |
Multivariate analysis |
||||||
|---|---|---|---|---|---|---|---|---|
| OR | 95%CI of OR |
p-Value | OR | 95%CI of OR |
p-Value | |||
| Lower | Upper | Lower | Upper | |||||
| RARP | 0.190 | 0.096 | 0.400 | <0.0001 | 0.459 | 0.141 | 1.492 | 0.195 |
| LND | 2.915 | 1.400 | 5.84 | 0.003 | 0.93 | 0.300 | 2.919 | 0.909 |
| Age | 1.086 | 1.022 | 1.160 | 0.003 | 1.038 | 0.971 | 1.109 | 0.277 |
| BMI | 1.044 | 0.928 | 1.170 | 0.473 | ||||
| PV | 1.011 | 0.992 | 1.030 | 0.273 | ||||
| ASA 3 | 1.500 | 0.576 | 3.900 | 0.406 | ||||
| cT2 | 1.003 | 0.997 | 1.010 | 0.381 | ||||
| BL | 1.000 | 1.001 | 1.000 | <0.0001 | 1.002 | 1.001 | 1.003 | <0.0001 |
| LOHS (DD) | 1.521 | 1.277 | 1.810 | <0.0001 | 1.425 | 1.168 | 1.739 | <0.0001 |
| T | 0.934 | 0.867 | 1.000 | 0.072 | ||||
| T0 | 0.997 | 0.954 | 1.000 | 0.89 | ||||
| T1 | 1.035 | 0.983 | 1.090 | 0.192 | ||||
| T3 | 0.967 | 0.894 | 1.040 | 0.389 | ||||
| T4 | 1.116 | 1.008 | 1.240 | 0.035 | 1.004 | 0.967 | 1.012 | 0.281 |
| T45 | 0.993 | 0.870 | 1.130 | 0.927 | ||||
ASA, American Society of Anesthesiologists; BL, blood lost; BMI, body mass index; cT, clinical stage; DD, discharge day; LND, lymph node dissection; LOHS, length of hospital stay; PV, prostate volume; RARP, robot assisted radical prostatectomy; T, basal cortisol; T0, cortisol 4 h after surgery; T1, cortisol on POD 1; T3, cortisol on POD 3; T5, cortisol on POD 5; T45, cortisol on POD 45.
Table 4.
Covariates predicting LOHS (DD) by linear regression analysis.
| Variable | Univariate analysis |
Multivariate analysis |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| b | SD | 95%CI of b |
p-Value | b | SD | 95%CI of b |
p-Value | |||
| Lower | Upper | Lower | Upper | |||||||
| RARP | −1.767 | 0.24 | −2.240 | −1.294 | <0.0001 | −0.905 | 0.32 | −1.532 | −0.274 | 0.005 |
| LND | 1.277 | 0.23 | 0.833 | 1.722 | <0.0001 | 0.366 | 0.27 | −0.183 | 0.915 | 0.209 |
| AGE | 0.042 | 0.02 | 0.008 | 0.076 | 0.015 | −0.003 | 0.02 | −0.034 | 0.028 | 0.857 |
| BMI | 0.005 | 0.04 | −0.072 | 0.082 | 0.904 | |||||
| ASA ≥ 3 | 1.564 | 0.34 | 0.093 | 2.225 | <0.0001 | 1.220 | 0.30 | 0.615 | 1.824 | <0.0001 |
| Clavien–Dindo ≥ 3a | 1.872 | 0.31 | 1.256 | 2.489 | <0.0001 | 1.251 | 0.30 | 0.664 | 1.840 | <0.0001 |
| cT2 | 0.230 | 0.23 | −2.225 | 0.684 | 0.339 | |||||
| BL | 0.000 | 0.00 | 0.000 | 0.001 | 0.811 | |||||
| T | −0.019 | 0.02 | −0.063 | 0.025 | 0.397 | |||||
| T0 | 0.008 | 0.02 | −0.021 | 0.036 | 0.602 | |||||
| T1 | 0.040 | 0.02 | 0.006 | 0.075 | 0.023 | 0.097 | 0.01 | −0.001 | 0.060 | 0.06 |
| T3 | 0.047 | 0.03 | 0.067 | 0.097 | 0.067 | |||||
| T5 | 0.145 | 0.04 | 0.076 | 0.214 | <0.0001 | 0.097 | 0.03 | 0.034 | 0.590 | 0.002 |
| T45 | 0.066 | 0.04 | −0.019 | 0.150 | 0.127 | |||||
RARP, robot assisted radical prostatectomy; POD, postoperative day; LND, lymph node dissection; BMI, body mass index; ASA, American Society of Anesthesiologists' risk; cT, clinical stage; BL, blood lost; T, basal cortisol; T0, cortisol 4 h after surgery; T1, cortisol on POD 1; T3, cortisol on POD 3; T5, cortisol on POD 5; T45, cortisol on POD 45; b, regression coefficient; SD, standard deviation.
Figure 2.
Cumulative proportion (%) of length of hospital day (DD, days) in RARP and RRP patients. DD, discharge day; RARP, robot assisted radical prostatectomy; RRP, retropubic radical prostatectomy.
Figure 3.
Mean cortisol plasma levels (ng/mL) on POD 5 (T5) as a function of DD (days) in RARP and RRP cases. Patients who underwent RARP show significantly lower median levels of cortisol plasma levels than RRP cases. DD, discharge day; POD, postoperative day; RARP, robot assisted radical prostatectomy; RRP, retropubic radical prostatectomy; T5, cortisol on POD 5.
4. Discussion
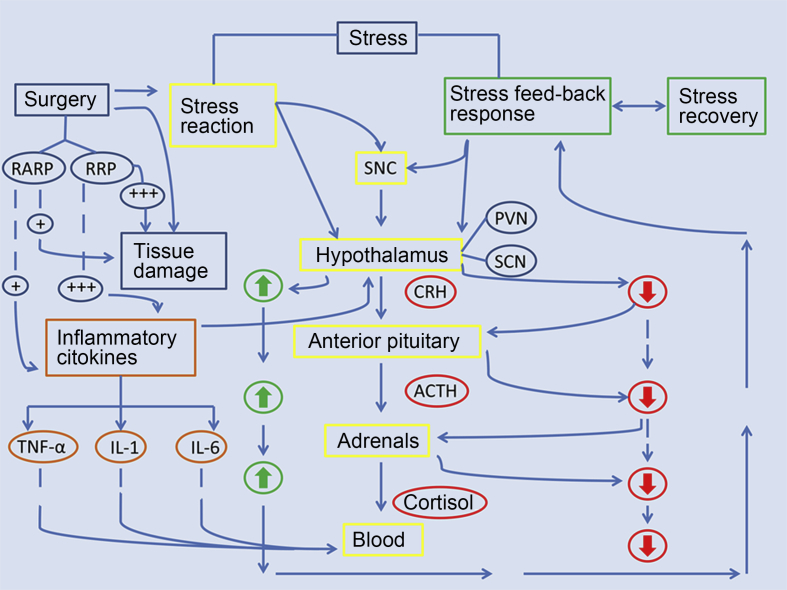
Major surgery trauma is a significant challenge to homeostasis because it activates compensatory autonomic and neuro-endocrine systems, of which the HPA axis is the most pivotal one [9]. Initially, production of pro-inflammatory cytokines; tumor necrosis factor α (TNF-α) and interleukin-1 (IL-1) and interleukin-6 (IL-6) cause direct stimulation of CRH and ACTH by the hypothalamus and pituitary respectively. After stress stimulation, along the HPA axis, adrenal glands undergo cortisol overproduction; as a result, there is a significant increase in total serum levels of cortisol which is the main hormone in the pathway of the neuro-endocrine stress response. In the present study, we showed that the patient population recovered from stress on POD 5 (sample F); moreover, stress recovery was more effective in RARP patients compared to RRP cases (Table 2 and Fig. 3). Interestingly, as shown in Table 2, significant higher mean levels of cortisol were detected in RARP patients on T0 (few hours after surgical stress). An explanation of this finding might be related to the surgical injury and stretching of the peritoneum related to the laparoscopic procedure. The present study shows that RARP, along the HPA axis, is major surgical procedure that triggers lower stress levels than RRP, as shown by MLR analysis in which RARP showed a negative independent association with F cortisol (Table 3 and Fig. 3). As a theory, the negative feedback mechanism of cortisol serum levels on the stress system were faster in patients who underwent RARP because of the negative association of the procedure with stress recovery. Indeed, as shown by F cortisol serum levels, the negative feedback was almost concluded in RARP patients while it was still ongoing in RRP cases.
It has been shown that RARP induces lower tissue trauma than RRP because of the more limited increase in acute-phase inflammatory mediators, such as IL-6 and C reactive protein (CRP) [10]. As a consequence, the stress system, along the HPA axis, is triggered to a lesser degree, according to the physiopathology of the stress system [6], [7], [8], [9]. The present study shows that RARP accelerates surgical stress recovery along the neuroendocrine system. Our results are confirmed by an experimental study that compared the stress response to robotic technology during thoracoscopic mobilization and anastomosis of the esophagus vs. the conventional open approach [19]. As a theory, RRP increased the hyper inflammatory response followed by cell-mediated immunosuppression which triggered higher stress levels along the HPA axis (Fig. 4). Our study showed that acute stretching of the peritoneum by insufflation of carbon dioxide, as a trigger of cortisol release along the HPA axis, did not play an important role in RARP. Moreover, the lesser pain levels, as a theory, might explain the faster stress recovery following RARP. These different cortisol stress related kinetics could explain why, in RARP cases, cortisol levels on POD 4 to 5 were significantly lower than those detected in RRP patients (Table 3 and Fig. 3).
Figure 4.
Pathways of post-operative stress kinetics along the neuroendocrine system after acute surgical trauma (see Discussion section for details). ACTH, adrenocorticotropic hormone; CRH, corticotrophin-releasing hormone; RARP, robot assisted radical prostatectomy; RRP, retropubic radical prostatectomy; SNC, central nervous system; PVN, paraventricular nucleus; SCN, suprachiasmatic nucleus.
There are limits in our study. First, the lack of randomized allocation of the patients might impact for the differences in the baseline characteristics of the two subpopulations, as shown in Table 1. However, now a days very few patients are willing to undergo randomization to RARP vs. RRP. As a result, the choice of surgical approach for RP is effectively based on the request and preference of the patient. Consequently, the present study reflects what's daily clinical practice in a urologic unit. Second, factors triggering the stress system as well as the measurement of both CRH and ACTH were not investigated. Third, although significant differences between RARP and RRP related to age, PSA and ASA, the multivariate independent regression models (logistic and linear) showed that RRP strongly associated with major postoperative surgical complications (Clavien–Dindo score ≥ 3a) that delayed LOHS. It would be interesting to compare the acquired data to studies that describe the cortisol level from other surgical procedures which are lacking at the moment. However, the present study confirms definitively the preliminary results of our trial [20] and provides a useful framework for planning further trials for investigating PCa surgery feed-backs on the stress system. As a theory, in high risk PCa surgery, the probability of disease progression, such as loco-regional and systemic migration of malignant cells, should be less likely when the stress system is triggered to lower degree levels and to a shorter perioperative period.
Therefore we conclude that our study shows that PCa surgery immediately (POD 0) triggers the stress system which respond by overproduction of cortisol which induces the negative feedback mechanism that starts on POD 1, is still ongoing on POD 5, but is completely settled on POD 45. Moreover, our study gives evidence that, after surgical trauma, the RARP procedure associates with stress recovery fester than RRP. Further confirmatory studies are required.
Conflicts of interest
The authors declare no conflict of interest.
Footnotes
Peer review under responsibility of Second Military Medical University.
References
- 1.Siegel R., Niashadham D., Jemal A. Cancer statistic, 2012. CA Cancer J Clin. 2012;62:10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- 2.Ferlay J., Autier P., Boniol M., Heanue M., Colombet M., Boyle P. Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol. 2007;18:581–592. doi: 10.1093/annonc/mdl498. [DOI] [PubMed] [Google Scholar]
- 3.Heidenreich A., Bellmunt J., Bolla M., Joniau S., Mason M., Matveev V., European Association of Urology EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and treatment of clinically localized disease. Eur Urol. 2011;59:61–71. doi: 10.1016/j.eururo.2010.10.039. [DOI] [PubMed] [Google Scholar]
- 4.Mullins J.K., Feng Z., Trock B.J., Epstein J.I., Walsh P.C., Loeb S. The impact of anatomical radical prostatectomy on cancer control: the 30-year anniversary. J Urol. 2012;188:2219–2224. doi: 10.1016/j.juro.2012.08.028. [DOI] [PubMed] [Google Scholar]
- 5.Montorsi F., Wilson T.G., Rosen R.C., Ahlering T.E., Artibani W., Carrol P.R., Pasadena Consensus Panel Best practices in robot-assisted radical prostatectomy: recommendations of the Pasadena Consensus Panel. Eur Urol. 2012;62:368–381. doi: 10.1016/j.eururo.2012.05.057. [DOI] [PubMed] [Google Scholar]
- 6.Ni Choileain N., Redmond H.P. Cell response to surgery. Arch Surg. 2006;141:1132–1140. doi: 10.1001/archsurg.141.11.1132. [DOI] [PubMed] [Google Scholar]
- 7.Chrousos G.P., Gold P.W. The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. JAMA. 1992;267:1244–1252. [PubMed] [Google Scholar]
- 8.Tsigos C., Chrousos G.P. Hypothalamic–pituitary–adrenal axis, neuroendocrine factors and stress. J Psychosom Res. 2002;53:865–871. doi: 10.1016/s0022-3999(02)00429-4. [DOI] [PubMed] [Google Scholar]
- 9.Gibbison B., Angelini G.D., Lightman L. Dynamic output and control of the hypothalamic–pituitary–adrenal axis in critical illness and major surgery. Br J Anesth. 2013;111:347–360. doi: 10.1093/bja/aet077. [DOI] [PubMed] [Google Scholar]
- 10.Fracalanza S., Ficarra V., Cavalleri S., Galfano A., Novara G., Mangano A. Is robotically assisted laparoscopic radically prostatectomy less invasive than retropubic radical prostatectomy? Results from a prospective, unrandomized, comparative study. BJU Int. 2008;101:1145–1149. doi: 10.1111/j.1464-410X.2008.07513.x. [DOI] [PubMed] [Google Scholar]
- 11.Menon M., Tewari A., Peabody J., The VIP Team Vattikuti Institute prostatectomy: technique. J Urol. 2003;169:2289–2292. doi: 10.1097/01.ju.0000067464.53313.dd. [DOI] [PubMed] [Google Scholar]
- 12.Walsh P.C. Anatomic radical retropubic prostatectomy. In: Walsh P.C., Retik A.B., Vaughan E.D., Wein A.J., editors. Campbell's urology. 8th ed. WB Saunders; Philadelphia: 2002. pp. 3107–3129. [Google Scholar]
- 13.D'Amico A.V., Whittington R., Malkowicz S.B., Shultz D., Blank K., Broderick G.A. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280:969–974. doi: 10.1001/jama.280.11.969. [DOI] [PubMed] [Google Scholar]
- 14.Briganti A., Larcher A., Abdollah F., Capitanio U., Gallina A., Suardi N. Updated nomogram predicting lymph node invasion in patients with prostate cancer undergoing extended pelvic lymph node dissection: the essential importance of percentage of positive cores. Eur Urol. 2012;61:480–487. doi: 10.1016/j.eururo.2011.10.044. [DOI] [PubMed] [Google Scholar]
- 15.Dripps R.D., Lamont A., Eckenhoff J. The role of anesthesia in surgical mortality. JAMA. 1961;178:261–263. doi: 10.1001/jama.1961.03040420001001. [DOI] [PubMed] [Google Scholar]
- 16.Dindo D., Demartines N., Clavien P.A. Classification of surgical complications. A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–213. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Flemming I.D., Cooper J.S., Henson D.E., Hutte R.V.P., Kennedy B.J., Murphy G.P., editors. American Joint Committee on Cancer Staging Manual. 5th ed. JP Lippincott; Philadelphia: 1997. pp. 219–222. [Google Scholar]
- 18.Roberts F.S., Roberts W.L. Performance characteristics of five automated serum cortisol immunoassays. Clin Biochem. 2004;37:489–493. doi: 10.1016/j.clinbiochem.2004.01.009. [DOI] [PubMed] [Google Scholar]
- 19.Eisold S., Mehrabi A., Konstantinidis L., Mieth M., Hinz H., Kashfi A. Experimental study of cardiorespiratory stress factors in esophageal surgery using robot-assisted thoracoscopic or open thoracic approach. Arch Surg. 2008;143:156–163. doi: 10.1001/archsurg.2007.56. [DOI] [PubMed] [Google Scholar]
- 20.Porcaro A.B., Molinari A., Terrin A., De Luyk N., Bldassarre R., Brunelli M. Robotic-assisted radical prostatectomy is less stressful than the open approach: results of a contemporary prospective study evaluating pathophysiology of cortisol stress-related kinetics in prostate cancer surgery. J Robot Surg. 2015;9:249–255. doi: 10.1007/s11701-015-0522-3. [DOI] [PubMed] [Google Scholar]