Abstract
Introduction
A growing body of evidence suggests even small rises in serum creatinine (SCr) are of considerable clinical relevance. Given that participants in endurance events are exposed to potential (repeated) renal insults, a systematic review was undertaken to collate current evidence for acute kidney injury (AKI), complicating such events.
Methods
A systematic review of studies and case reports meeting inclusion criteria on Medline and EMBASE (inception to October 2015). Included: studies with markers of renal function before and after endurance or ultraendurance events; case reports of severe AKI. Two reviewers assessed risk of bias using the Newcastle-Ottawa scale.
Results
Eleven case report publications (n=27 individuals) of severe AKI, were retrieved, with risk factors including systemic illness or nephrotoxic medications usually identified. From 30 studies of endurance and ultraendurance events, mean rise in SCr was 29 (±12.3) µmol/L after marathon or ultramarathon (17 studies, n=568 participants) events. Where follow-up tests were conducted, SCr returned to baseline within 48 hours. Rises in biomarkers suggest potential parenchymal insult, rather than simply muscle breakdown. However, evidence of long-term deleterious effects is lacking.
Conclusions
Raised levels of SCr are reported immediately after endurance events. It is not clear whether this is either clinically significant, or if repeated participation predisposes to long-term sequelae. The aetiology of severe exercise-associated AKI is usually multifactorial, with risk factors generally identified in the rare cases reported. On-site biochemistry, urine analysis and biomarkers of AKI may help identify collapsed runners who are at significant short-term risk and allow suitable follow-up.
Keywords: acute kidney injury, marathon, endurance events, non-steroidal anti-inflammatories, biomarkers
What are the new findings.
Twenty-seven cases of acute kidney injury (AKI) requiring hospital treatment after an endurance or ultraendurance event have been reported, with rhabdomyolysis often associated alongside at least one other identifiable risk factor.
Across 21 studies (n=800), including half-marathons, where pre-event and postevent serum creatinine (SCr) levels were available, mean SCr increase was 25.7 (±11.6) Gmol/L.
Of the 17 marathons or ultramarathons studied (n=568), mean SCr increase was 29 (±12.3) Gmol/L.
Non-steroidal anti-inflammatory agents appear to be a significant risk factor for AKI, though few studies in this area have been published.
Use of renal biomarkers is of growing interest and in collapsed runners, may help identify those at high risk of significant AKI.
Introduction
Endurance events continue to increase in popularity. For example, marathon participation in the USA has risen from 25 000 in 1976 to over half a million in 2011.1 Participation in ultramarathons (a running event involving a distance longer than the traditional marathon length)2 is also rapidly growing. Although individuals who exercise regularly have lower rates of disability and a mean life expectancy some 6 years longer than that of their less active contemporaries, there remain some concerns regarding potential adverse sequelae.3–5 Although sudden death among athletes attracts much publicity, it is rare and often due to a pre-existing cardiomyopathy.6–9 More commonly, less severe organ damage may occur during strenuous exercise, which, if repeated over time, could have long-term significance, including renal damage, as evidenced by animal studies10 and human studies investigating cardiac imaging and enzyme markers.11–19 Though long-term evidence of harm remains scarce, recent studies have suggested an attenuation of the benefits of exercise, among those performing the most strenuous exercise, with a J-shaped or U-shaped association between all-cause mortality and running dose.20–22
Quantifying renal damage and defining renal failure has, until recently, suffered from a lack of consensus definition. However this has been addressed, in part, by the adoption of the concept of acute kidney injury (AKI) which classifies renal injury on the basis of changes in measured serum creatinine (SCr) compared with baseline values and/or changes in monitored urine output (see Appendix for AKI definitions).23 24 This has allowed classification of patients, particularly in acute settings such as the critical care environment, allowing comparison between groups, as well as defining renal injury, which may not progress to renal failure requiring extracorporeal support. Of particular relevance is the observation that what was previously viewed as relatively trivial increases in SCr, carries an increase in acute mortality. It is now clear that renal injury, as defined by AKI, puts patients at longer-term risks of both morbidity and mortality.25–27
The earliest studies reporting urinary changes on extreme exercise coincided with the seminal work by Bywaters and Beall describing the biochemical and histopathological features of crush injury observed in air raid victims during World War II which we now describe as rhabdomyolysis.28 The first documented study in athletes was in 1941 and described myoglobinuria, haematuria and albuminuria in marathon runners with the first case reports of acute renal failure complicating any sporting event reported in the 1960s.29 30 Within the clinical arena there are numerous potential specific causes of AKI but a number of factors alone, or in combination, may exacerbate the condition. These include volume depletion secondary to inadequate fluid intake and excessive losses, rhabdomyolysis and the use of drugs such as non-steroidal anti-inflammatory drugs (NSAIDs). It follows that these factors may be common to individuals competing in endurance events with the further potential complications of climate and exercise-associated heat stroke (EHS).31–35 Therefore, AKI may be a more common complication of endurance events than anticipated. While this may seem a relatively harmless ‘side-effect’ of endurance events, there is now a considerable body of evidence that points to the fact that an episode of AKI complicating critical illness, for example, carries a significant risk of longer-term mortality and morbidity. Could this observation be relevant to those exhibiting an SCr rise after endurance events?
Research questions
To systematically review the frequency of AKI) associated with endurance (and ultraendurance) events; to quantify changes in SCr as a marker of renal dysfunction in endurance event studies recording values pre-event and postevent. Furthermore, the review will explore mechanisms of renal dysfunction due to exercise-associated rhabdomyolysis and use of NSAIDs; finally future studies, including the use of renal biomarkers, will be addressed.
Rationale and objectives
Endurance event participation is increasing and emerging evidence suggests extreme exercise could potentially attenuate some of the benefits seen with regular exercise. AKI is an independent contributor to mortality in a large number of clinical settings. There have been case reports of significant AKI requiring hospital care following endurance events and also prospectively studied markers of renal dysfunction following endurance events, but to date, there has been no systematic review of this field. Thus, the rationale for this review was to collate all empirical evidence for AKI in endurance (and ultraendurance) events, fitting the prespecified eligibility criteria for inclusion, to answer the research questions. Explicit, systematic methods were selected with a view to minimising bias and provide reliable findings from which conclusions can be drawn.36 37
Methods
Inclusion criteria
Adult human studies in the English language in peer-reviewed journals, of endurance or ultraendurance events, examining markers of renal function; to quantify a change in SCr, studies would be included where an SCr value pre-event and immediately post the event was reported, though studies without both values would also be reported; case report studies of participants with AKI requiring hospitalisation following an endurance event.
Exclusion criteria
Research involving paediatric patients (<18 years old) and non-human studies; studies involving events not considered endurance (eg, <1 hour), such as track and field or football.
Identification, screening, risk of bias
We searched Medline and EMBASE for English language articles from inception to October 2015 with the text words and related Medical Subject Headings terms: ‘AKI,’ ‘acute kidney injury,’ ‘Acute renal failure,’ ‘Renal’ and ‘Marathon’ or ‘Endurance events’ as the initial set of search terms; to ensure a comprehensive literature search, reference lists were examined from retrieved articles (see Appendix for search string and PRISMA flow diagram). Titles and abstracts were screened and full articles reviewed if thought to be eligible for inclusion. Data extraction (using a pretested form), study design and quality were performed by two investigators. Discrepancies in any data extraction were discussed and assessment of a third reviewer was sought for resolution. Two reviewers assessed the risk of bias of each study using the Newcastle-Ottawa Scale.38 This scale awards a maximum of nine stars across three domains (selection, comparability of cohorts on the basis of the design and analysis, and outcome assessment). Studies assigned at least seven stars were deemed to be of good quality.39 For analysis of the magnitude of change in SCr we included observational studies in adults in which renal function was measured before and within hours after an endurance or ultraendurance event. Case reports of significant renal dysfunction requiring medical attention were included where the primary diagnosis was AKI.
Data extraction
Year of publication, data collection methods (retrospective or prospective), event (such as a marathon) setting (country or countries), number included, primary outcome, pre-SCr and post-SCr (where available), urine analysis, biomarkers, creatine kinase (CK) in the case reports of severe AKI; number who collapsed; and use of NSAIDs. Eleven case report studies reported a total of 27 cases of AKI. An additional 30 studies assessing markers of renal function including SCr changes, urine analysis and biomarkers (total number of subjects 1724) following endurance or ultraendurance events were found, of which 21 reported pre-event and postevent SCr levels.
Results
In 11 case report publications, 27 cases of AKI following endurance events were described (table 1). Of the events, five were marathons33 40–44 and six ultramarathons.30 31 45–47 Eight participants had collapsed during the event. The majority (21/27) presented to medical attention in a delayed fashion—sometimes over a week after the event. In two-thirds of cases (18/27), a history of NSAID before and/or during the event was elicited. Other risk factors included recent illness and vomiting during the events. Fifteen cases required temporary dialysis. A renal biopsy was performed on only four occasions, with features of acute tubular necrosis (ATN) in all the biopsies, suggesting an ischaemic aetiology. A diagnosis of rhabdomyolysis was made, or presumed, in 85% (n=23) of the cases. Where reported, follow-up renal function was reported to be normal, with no requirement for long-term renal replacement therapy.
Table 1.
Summary of 11 case reports of 27 athletes with AKI following a marathon or ultramarathon
| Study | Cases | Collapse | Delayed* | Type | NSAIDs | Dialysis | Risk factors | SCr (µmol/L) | CK (U/L) | Urine | Follow-up | Biopsy | Diagnosis / comments |
| Dancaster, 196930 | 2 | 2 | 2 | Ultramarathon | Both took an analgesic | – | Diarrhoea (2) Vomiting (1) |
– | – | Case 1: moderate albumin; Case 2: 2+ protein, RBCs and granular casts; spectral exam -ve for blood and myoglobin; occult test for blood +ve |
Case 1: 1 year CrCl 115 mL/min | Rhabdomyolysis. Case 2 had markedly elevated AST, ALT, LDH |
|
| Pollard 197040 | 1 | 1 | Marathon | - | Reported recurrent haematuria postexercise |
1193 72 hours post | 3+ RBCs, granular casts; spectroscopy –ve for free haemoglobin and myoglobin | Renal function ‘normal’ at 4/52 | ATN | March Haemoglobinuria? Bilateral flank pain | |||
| MacSearraigh, 197945 | 8 | 3 | 3 | Ultramarathon | 5 | 5 | Diarrhoea (1), Vomiting (3), EHS (1), recent illness (3) Hypertension (2) |
– | Case 4: 28 000 | Case 3: 6 years normal CrCl. 4: normal 15/12 7: CrCl normal 9: ‘normal’ |
Case 4 ATN Case 8: ATN and old fibrosis | n=7: severe muscle cramps, dark urine | |
| Bar-Sela, 197941 | 1 | 1 | Marathon | 1 | Recent flu-like illness | 751 | 10 400 | Proteinuria, RBCs, granular casts; -ve for myoglobin | normal CrCl 6/12 | Rhabdomyolysis: severe muscle cramps in race, dark urine | |||
| Goldsmith, 198442 | 1 | 1 | Marathon | 1 | – | 253 1/52 post | - | Weakly +ve for RBCs | Full recovery 6/52 | Rhabdomyolysis (presumed)—stiff leg muscles, lumbar pain to groins, polydipsia | |||
| Vitting,198633 | 1 | 1 | Marathon | 1 | 1 | Hypertension | 1025 48 hours | 83 | 3+ protein, 1+ RBC, scattered renal tubular cells, few erythrocytes | Day 26 SCr 106 | |||
| Seedat,198931 | 4 | 1 | 3 | Ultramarathon | 4 | 3 | Recent UTI Influenza |
Case 1: 930 2: 604 3: 593 4: 1263 |
1: 39 800 2: 29 800 3: 24 120 4: 2220 |
Not reported | |||
| Le Meurl, 199843 | 1 | 1 | Marathon | 1 | Recurrent haematuria on exercise; frank haematuria in race | 1,070 48 hours post | 233 | RBC count 4×106/mL; haemoglobinuria slightly +ve; myoglobinuria -ve | Full recovery 2/12 | ATN | Authors speculated haematuria of glomerular origin from prolonged exercise leading to intraluminal obstruction or toxic effect of haemoglobin on tubular cells similar to IgA nephropathy. | ||
| van Zyl-Smit, 200046 | 1 | 1 | Ultramarathon | – | 713, 10 days post | – | 2+ protein 1+ blood, moderate granular casts | full recovery | Rhabdomyolysis (presumed) | ||||
| Bruso,201044 | 3 | 3 | Ultramarathon | 3 | – | Case 1: vomiting, Case 1 and 3 required intravenous fluid post race |
Case 1 911 Case 2 433 Case 3 283 |
Case 1: 7 85 250 Case 2: 38 218 Case 3:>40 000 |
Case 1: 3+ blood, 1+ protein Case 2: 3+ blood, trace protein Case 3: 1+ blood |
All rhabdomyolysis Case 1: 12 days in hospital; 2: 3 days in hospital 3: 1 day in hospital 2 presented<24 hours; 1 presented 48 hours; all hyponatraemic (range 129–134) |
|||
| Boulter,201147 | 4 | 4 | Ultramarathon | 4 | 4 | All taken supplement; Vomiting (3) | Case 1: 656 2: 264 3: 551 4: 1139 |
Case 1: 48 934 2: N/A 3: 54 231 4: 5718 |
Rhabdomyolysis (4)—cases presented 1–4 days postrace. | ||||
| Total | 27 | 8 | 21 | 18 | 15 | 4 |
AKI, acute kidney injury; ATN, acute tubular necrosis; CK, creatine kinase; CrCl, creatinine clearance; EHS, exertional heat stroke; RBCs, red blood cells, SCr, serum creatinine; *delayed presentation to medical services.
A total of 30 studies (n=1724 participants) on renal function in endurance events were identified (table 2). These include 13 marathons,35 48–59 10 ultramarathons,60–69 2 half-marathons,70 71 an Ironman,72 a 24-hour marathon relay,73 a 60-km mountain run74 and 2 ultramarathon cycle races.75 76 Of 21 foot race events (n=800 participants) reporting pre-event and postevent SCr levels, 17 were studies of marathons or ultramarathons (n=568 participants). For risk of bias, 26 of the studies were deemed of good quality with a Newcastle-Ottawa Score of at least seven stars (see supplementary table for full details).
Table 2.
Summary of 30 studies looking at renal function in endurance events (those in grey did not report SCr prior to the event)
| Study | Year | Event | n | Collapse | NSAID | Prerace SCr (µmol/L) | Postrace SCr | Repeat SCr | Mean SCr rise | Additional markers and notes |
| Riley48 | 1975 | Marathon | 5 | 2 | 94 (±2) | 136 (±6) | 42 | |||
| Noakes64 | 1976 | Ultramarathon | 13 | 97 | 128 | 31 | ||||
| Neviackas49 | 1981 | Marathon | 6 | 84 (range 70–97) | 6 hours: 104 (70–159) | 1 week: 86 (62–97) | 20 | |||
| Irving35 | 1986 | Marathon | 6 | 89 (±3) | 96 (±5) | 24 hours: 86 | 7 | |||
| Kraemer50 | 1986 | Marathon | 20 | 80 (±17) | 120 (±20) | 40 | ||||
| Irving73 | 1989 | 24hour relay marathon | 2 | 81, 86 | 101, 119 | 48 hours: 92 and 92 | 20, 33 | |||
| Nelson58 | 1989 | Marathon | 38 | 81 | 123 | 42 | ||||
| Borkowski57 | 1990 | Marathon | 6 | Urine PCR x 2.6 higher postrace; increase trypsin inhibitor: creatinine ratio at 6–18 hours | ||||||
| Irving65 | 1990a | Ultramarathon | 5 | 1 | Collapse 42; normal 63 (±3) |
55 collapsed, normal 69 (±10) | 66 (±6); 53 | 6, 13 (collapsed) | Urine flow rate, CrCl unchanged (non-collapsed); urine creatinine excretion increase | |
| Irving60 | 1990b | Ultramarathon | 8 | 91 | 110 | 24 hours: 'normal' | 19 | |||
| Irving61 | 1991 | Ultramarathon | 26 | 8 | 3 | Collapsed 88.9 (±8), Controls 104 (±5) | Collapsed 68, Controls 78 | |||
| Holtzhausen51 | 1994 | Marathon | 96 | 36 | Collapsed 141 (±26), Controls 123 (±23) | 24 hours: Collapsed 104 (±13), Control 95 (±13) | ||||
| Kratz52 | 2002 | Marathon | 37 | 88 | 115 | 24 hours: 106 | 27 | |||
| Neumayr75 | 2003 | Ultramarathon cycle | 38 | 88 | 106 | 24 hours: 106 | 18 | |||
| Reid53 | 2004 | Marathon | 134 | 18 | M 110, F 100 (50–160) | NSAID group had higher SCr (p=0.01) | ||||
| Neumayr76 | 2005 | Ultramarathon cycle | 16 | 84 (±15) | 111 (±19) | 83 (±15) | 26 | |||
| Wharam72 | 2006 | Ironman triathalon | 333 | 100 | NSAID 130, controls 120 | Significant increase in NSAID group versus no NSAID (p=0.01) | ||||
| Page74 | 2007 | 60-km mountain run | 123 | 43 | 110 (±2) | F/U<1week: normal | No difference in NSAID users | |||
| Dumke | 2007 | Ultramarathon | 54 | 29 | NSAID 85 (±12), controls: 91 (±15) |
NSAID 118 (±25), controls 120 (±27) |
33 versus 29 | No significant difference between groups | ||
| Lippi70 | 2008 | Half-marathon | 17 | 99 (90–113) | 115 (112–130) | 24 hours: 101 (92–113) | 16 | eGFR decreased by 14 | ||
| Mingels54 | 2009 | Marathon | 70 | 86* | 132* | 24 hours: 96 | Cystatin C elevated in 26%—mean increase half as much as creatinine | |||
| McCullough55 | 2011 | Marathon | 25 | 80 (±9) | 106 (±18) | 24 hours: 71 (±9) | 27 | Rise: cystatin C, urine NGAL and kidney injury molecule-1 levels. Resolution<24 hours. | ||
| Mohseni71 | 2011 | Half-marathon | 195 | 78 | 80 | 97 | 17 | |||
| Mydlik56 | 2012 | Half or marathon | 49 | 93 (±10), 87 (±8) | 111 (±15), 112 (±9) | 2–6 days post 103 (±11) | 18, 25 | |||
| Lippi62 | 2012 | Ultramarathon | 16 | 68 (58–76) | 98 (76–118) | 30 | sNGAL x1.6, uNGAL x7.7, uNGAL/creatinine ratio x2.9 increase. 31% eGFR decrease | |||
| Hoffman63 | 2013 | Ultramarathon | 207 | 70 | 9 (4%) AKI ‘Injury’; 62 risk | Urine 1+ protein, 3+ blood and specific gravity>1.025 predicted those with AKI criteria | ||||
| Lipman66 | 2014 | Ultramarathon | 30 | 88 (±18) | 124 (±115) | 35 | ||||
| Christensen68 | 2014 | Ultramarathon | 10 | 60 | 80 | 20 | 25% increase in SCr at 6 hours | |||
| Hou67 | 2015 | Ultramarathon | 26 | 2 | 83 (±10) | 138 (±35) | 72.5 (64–81) | 55 | ||
| Hewing59 | 2015 | Marathon | 167 | 73 (66–82) | 90 (78–111) | 17 | Cystatin C: prerace 0.68 mg/dL, post 0.85 and f/u 0.66 Cystatin C-eGFR prerace 125 ml/min, post 94, f/u 130 |
Mean values (±SD), unless stated, *Median (IQR).
AKI, acute kidney injury; CrCl, creatinine clearance; eGFR, estimated glomerular filtration rate; F, female; M, male; NGAL, neutrophil gelatinase-associated lipocalin; NSAID, non-steroidal anti-inflammatory drug; PCR, protein:creatinine ratio; sNGAL, serum NGAL; SCr, serum creatinine; uNGAL, urine NGAL.
Creatinine production and clearance
A proportion of the SCr rise observed on extreme exercise reflects increases in muscle breakdown, with an enhanced solute load, rather than a fall in glomerular filtration rate (GFR).60 Hence the use of SCr to estimate GFR (eGFR) under conditions of extreme exercise should be avoided. Interestingly, a study on six marathon runners demonstrated that on the 1st day after the race, creatinine clearance actually increased, peaking on day 3 with increased urine flow rates on all 5 days postrace.35 This reflects the concept of augmented renal clearance, which is in fact a normal response to increased tubular flow. Rates of excretion and production of creatinine were closely matched on all study days.35 These results are in contrast to a study which showed no change in creatinine clearance, although inulin clearance reduced by 50%60 and further studies in ultramarathon cyclists where a decline in creatinine clearance of 18–25% was observed.75 76 Mean fractional sodium and uric acid excretion also fell, which may indicate reduced renal perfusion, and SCr rose by 20–36%, returning to normal at 24 hours.
Rises in SCr
Across 21 studies (n=800) where pre-event and post-event SCr levels were available, mean SCr increase was 25.7 (±11.6) µmol/L. Of the 17 marathons or ultramarathons described (n=568), mean SCr increase was 29 (±12.3) µmol/L, above the threshold for AKI stage 1.24 Though most studies demonstrated an increase in SCr immediately following the event, in the 13 studies reporting recovery of function, as described by changes in SCr, all had returned to baseline or were improving within 48 hours (table 2). Studies involving half-marathons reported smaller increases in SCr levels, with eGFR recovering within 6 hours in one study.70 In two recent extended ultraendurance races over weeks to months, no contestants developed AKI, despite significant rises in CK, suggesting repetitive, relatively lower intensity exercise, despite the large total distance covered, may mean that cumulative damage does not occur in these events.77 78 A recent study by Lipman et al 66 also supports this, documenting rises in SCr, with 45–67% meeting RIFLE criteria ‘Risk’ and 7–13% ‘Injury’ (see Appendix for RIFLE criteria) between stages of a multistage ultramarathon race, but found a rapid reduction back to baseline prior to the following stage. Christensen et al 68 investigated renal function up to 48 hours following a 78-km ultramarathon at moderate altitude (~2400 m) by 10 runners from the Mexican Tarahumara tribe, who have maintained the once widespread tradition of ultradistance running, from a young age. The authors found immediate postrace SCr was not significantly elevated, though an increase of 25% was noted by 6 hours, with a subsequent fall-back to baseline at 48 hours. Follow-up for the 30 studies did not extend beyond more than a few days, thus conclusions around the effects of recurrent participation in such events cannot be made.
Studies examining the use of NSAIDs
In the studies where NSAID use was reported, 23% (n=312/1359) of participants admitted to NSAID usage. Two studies, one of marathon runners53 and another of 330 Ironman participants,72 reported a significant increase in SCr in the participants using an NSAID. In contrast, a prospective ultramarathon study Dumke et al 69 studied a group taking an NSAID (n=29) and a group not (n=25) with no significant difference in SCr between groups.
Studies on collapsed runners
Two studies specifically reported on SCr levels in runners who had collapsed.51 61 In the first, 36 collapsed runners had mean postrace SCr of 141 (±26) vs 123 (±23) µmol/L in a control group of 60 runners (p<0.01).51 Within 24 hours, mean SCr had fallen to 104 (±13) µmol/L compared with 95 (±13) µmol/L in the control group (p<0.05). Irving et al included eight ultra-marathon runners who had collapsed with hyponatraemia (<130 mmol/L) requiring hospital admission. Their postrace mean SCr level was 89 µmol/L vs 104 µmol/L in a control group (n=18). A repeat test on ‘recovery’ showed mean SCr levels had reduced to 68 µmol/L (p<0.05) and 78 µmol/L (p<0.01), respectively.61
Other markers of possible renal dysfunction
The SCr provides the basis by which chronic kidney disease (CKD) is defined, with the eGFR allowing stratification of patients with CKD. However, the use of SCr, and derived eGFR equations implies that creatinine production rate is at a steady state and hence, more accurately reflects the GFR. This is not the case in many acute clinical scenarios and clearly not during and after, extreme exercise. The use of other candidate molecules as potential markers of renal dysfunction is emerging as a potential avenue to investigate the true significance of endurance events on renal function—particularly in the higher-risk group of participants who have collapsed. Any potentially useful candidate AKI biomarker(s) should ideally fulfil several criteria. These include, the provision of information above that currently available in determining abnormal renal function and also distinguish prerenal azotaemia from intrinsic renal disease. This is of particular relevance in extreme exercise where a prerenal component may predominate. If biomarkers could aid prognostication regarding potential renal recovery this also would be beneficial. Most recently identified biomarkers of AKI can be broadly classified into three groups: low molecular weight proteins which undergo glomerular filtration and hence reflect glomerular function, or markers of tubule damage, such as enzymes released into the urine after tubular cell injury, or inflammatory mediators either released by renal cells, or by infiltrating inflammatory cells.
Cystatin C is an effective biomarker of glomerular filtration, correlates well with GFR and is independent of muscle mass.79 Indeed, studies on renal function following strenuous exercise demonstrated that the mean cystatin C rise was half that of the mean SCr rise in runners following a marathon.54 Further studies demonstrated a correlation between cystatin C and SCr rise immediately after a marathon, with 40% of participants reaching the creatinine criteria for stage 1 AKI.55 Participants also demonstrated a rise in urinary neutrophil gelatinase-associated lipocalin (NGAL) and urinary kidney injury molecule-1 (KIM-1), suggesting early tubular dysfunction, although all biomarkers returned to baseline within 24 hours. Hewing59 in a Berlin marathon study (n=167), found cystatin-based GFR was significantly decreased by RIFLE criteria, with 30% demonstrating a decrease of >25% (risk) and 8% a decrease >50% (injury). All parameters returned to baseline ranges within a 2-week follow-up period.
NGAL is a 25 kDa acute phase protein largely produced by the kidney tubule, though it is also upregulated in other pathological conditions. In a study of 16 ultramarathon runners62 mean increase in SCr was 38%, with a significant increase in serum NGAL, urinary NGAL, as well as the urinary NGAL/creatinine ratio. Other studies have demonstrated increases in the urine protein:creatinine ratio 6–18 hours postrace.57 In a study on 152 runners in a 161 km ultramarathon, 34% met AKI criteria, with those demonstrating more significant degrees of injury having a significantly higher CK concentration. In addition, urinalysis tests that were positive for at least 1+ protein, 3+ blood and specific gravity ≥1.025 predicted those meeting the injury criteria with high sensitivity. The authors commented that using this simple test could be a useful screening test to identify those at highest rise.63
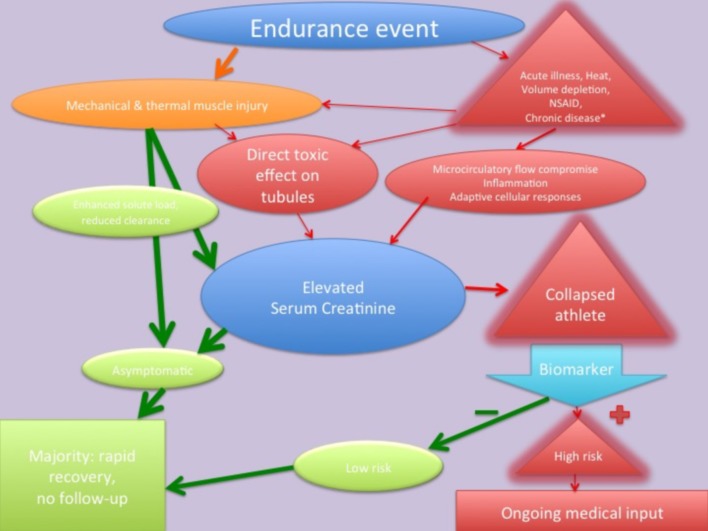
The diagnostic utility of most of the biomarkers described to date to diagnose AKI prior to elevations in SCr is variable. This is due, in part, to clinical studies being hampered by diverse patient populations with differing comorbidities, the timing of the measurements and chosen cut-off values. More recently, studies have emerged using two biomarkers associated with cell cycle arrest, namely insulin-like growth factor-binding protein 7 and tissue inhibitor of metalloproteinases-2.80 81 The concentrations of these two proteins was found to be in the urine of patients at high risk of AKI from a variety of aetiologies. Initial studies seem to suggest that these perform better than other known markers within this patient cohort; the mechanism of which may be an ‘alarm signal’ raised by the renal tubules, in response to stress. Furthermore, recent data imply that the presence of these biomarkers in the context of AKI may predict a worse prognosis in the critically ill, so perhaps they may be able to identify a group of athletes who may require closer follow-up (see figure 1 for a pathway outlining the aetiology of endurance exercise related renal dysfunction and potential utility of a biomarker to risk stratify participants).82
Figure 1.
Pathway outlining the aetiology of endurance exercise related renal dysfunction and potential utility of a biomarker to risk stratify participants. *Chronic disease such as hypertension, diabetes, chronic kidney disease. NSAID, non-steroidal anti-inflammatory drug.
Discussion
Pathophysiological effects of exercise on renal function
Increased blood flow to the muscles during exercise is accompanied by decreased renal blood flow proportional to extent and duration of the physical effort.83 In moderate exercise, renal blood flow may fall to 25% of the resting value, although GFR is preserved through an increase in the filtration fraction, which may double, limiting the transfer of metabolites or substances through the glomeruli and reducing the extent of exercise proteinuria.84–88 Extreme exercise however, may decrease GFR by up to 50%, through increased release of catecholamines, AVP and activation of the renin-angiotensin-aldosterone system.89–91 Whether prolonged reduction of renal blood flow is associated with intrinsic renal injury is uncertain and the clarification of this issue to date has been limited by the lack of a reliable biomarker. Despite evidence that AKI may progress to CKD, of the reported cases of AKI following an endurance event, we could find no documented case of progression to CKD. From the studies involving endurance events, SCr increases significantly, although whether this represents a significant insult to the kidney is not clear.
Extreme exercise—specific risk factors for AKI
Despite rises in measured SCr, the evidence for AKI requiring medical intervention is sparse. However, there are several conditions whereby the effects of extreme exercise on the kidney can be exacerbated. These include the development of pigment nephropathy, the use of NSAIDs and exertional heat stroke (EHS).
Pigment nephropathy
Rhabdomyolysis causes AKI through the direct toxic effect of myoglobin on the renal tubule. Severe exertion causes the syndrome through a combination of mechanical and thermal muscle injuries. Both intensity and duration of exercise are related to muscle damage in a dose-response manner.92–94 CK is the most sensitive marker of myocyte injury with levels at least five times above the upper limit of normal required to fulfil the diagnosis of rhabdomyolysis, although absolute levels do not correlate well with disease severity, nor risk of renal failure.95 96 AKI is common where rhabdomyolysis complicates critical illness, whereas in exercise-related rhabdomyolysis, AKI is rare. For example, Skenderi et al,97 found markedly raised CK levels in 39 runners after an ultra-marathon, yet none required medical assistance, nor developed AKI. Rhabdomyolysis with AKI has been observed following exercise when extreme physical exertion has been performed under conditions of high temperature, dehydration with, or without acute illness.98 99
Non-steroidal anti-inflammatory drugs
NSAIDs inhibit cyclo-oxygenase, thereby preventing prostaglandin synthesis (PGs). PGs enhance renal perfusion through vascular dilatation and promote diuresis and natriuresis through inhibition of sodium transport in the thick ascending limb of loop of Henle and the collecting ducts.100 101 PGE1 tends to antagonise the action of vasopressin and PGI2 in concert with PGE2, serves to maintain the GFR.102 103 In healthy subjects, basal renal vasoconstrictor tone is low and the vasodilation produced by PGs is not required for normal renal function.104 105 In disease states however, where increased sympathetic outflow to the kidney may be found, PGs vasodilate the renal vascular bed and ensure adequate renal blood supply. Therefore inhibition of PG synthesis may result in the unopposed vasoconstrictive action of leukotrienes, angiotensin II, vasopressin, endothelin and catecholamines. Indeed, NSAIDs have been shown to decrease renal blood flow with exercise, although this did not translate into significant differences in GFR (measured by inulin clearance) when subjects exercised at 80% VO2max for 30 min.106 Olsen et al studied healthy volunteers undergoing submaximal exercise with and without indomethicin.107 This NSAID significantly inhibited renal PG synthesis and renin release. Overall renal functional response to exercise remained unchanged, possibly because of inhibition of renin-mediated angiotensin II generation and cyclo-oxygenase-1 mediated thromboxane A2 synthesis.
Given that NSAIDs may have deleterious effects on renal blood flow, does the use of these agents translate into a higher risk of AKI in subjects undergoing extreme exercise? To date, few studies have addressed the impact of NSAID use on renal function after an endurance event, compared with those not taking NSAIDs. Reid et al found a significant mean increase in creatinine in the NSAID group compared with those not taking an NSAID in marathon runners.53 Wharam et al 72 in an Ironman study, reported a statistically significant difference in SCr of 10 µmol/L among the NSAID group, though this may not suggest a clinically significant difference. In contrast, Dumke et al, in a prospective study, found no significant difference in markers of renal function between those who did, and did not, use NSAIDs.69 However, our review data presented here show that 18 of the 27 case reports (67%) of renal failure (requiring hospital treatment) following an endurance event had taken an NSAID, which is in keeping with the widespread use of these agents. With regard to pigment nephropathy, conflicting results are found with studies showing an increase, no effect or a decrease in markers of muscle damage.108–110
Exertional heat stroke
AKI is often found in patients with EHS, which can be fatal.111–114 In addition to rhabdomyolysis and volume depletion, alterations in circulating vasoactive mediators are implicated.115 Subsequent multiorgan injury results from a complex interplay between the cytotoxic effect of the heat and the inflammatory and coagulation responses of the host.116 There have been recent guidelines developed to reduce the risk of EHS particularly in warm environments from the perspective of the athlete (including heat acclimatisation, initiate competition in an euhydrated state and minimise dehydration) and event organisers (provide shaded areas, cooling and rehydration facilities, and suitably schedule events).117
Exercise-induced AKI developing after anaerobic exercise
Exercise-induced AKI developing after anaerobic exercise (coined ALPE) is an uncommon syndrome differing from rhabdomyolysis by normal or slightly elevated serum muscle enzymes associated with muscle damage and the presence of severe loin pain. Vasoconstriction of intrarenal arteries within the kidney is thought to be the cause and studies using CT demonstrate multiple wedge-shaped areas of contrast enhancement in keeping with patchy vasoconstriction of the renal vessels. It is thought that vascular spasm accounts for the pain reported.118–121 Interestingly, a recent review of 118 cases of ALPE demonstrated a high percentage of subjects had hypouricaemia which can be associated with exercise-induced AKI.118
Limitations of the study
To the authors' knowledge this is the first systematic review to address case reports of AKI in association with endurance events and provides evidence from a number of observational studies, as to the effects of endurance events on SCr. The studies included were heterogenous (though most scored highly on a risk of bias assessment) and none included a significant time of follow-up, nor was any study found looking at the effect of performing multiple endurance events, over a period of years. Newer studies with renal biomarkers have suggested an increased SCr may be due to more than just an enhanced solute load and reduced clearance. However, firm conclusions as yet cannot be drawn from the available evidence. The case reports of significant renal injury by their retrospective nature have limitations, though the relatively large number with similar histories (acute illness, collapse, nephrotoxic drug use and delayed presentations) allows some interesting information to be drawn.
Conclusions
AKI requiring medical attention associated with extreme exercise is rare, with only 27 cases identified in this review, usually thought to be primarily due to rhabdomyolysis. Most cases had a number of risk factors for AKI, including the use of NSAIDs and an intercurrent illness or gastrointestinal upset. The majority presented in a delayed fashion, emphasising the need for awareness of this rare complication, particularly among participants who have become unwell during an event. It is likely that less severe cases of AKI with rhabdomyolysis go unreported and self-resolve. In contrast, an SCr rise after a marathon or ultra-marathon is common, with an average rise of 29 (±12.3) µmol/L, which appears to improve rapidly. However, there are few studies of collapsed runners with deranged renal function and their longer-term follow-up. Furthermore, in light of gathering evidence that strenuous exercise could have some adverse effects, it is not clear whether repeated participation in such events may eventually lead to significant long-term renal dysfunction. New biomarkers may be able to differentiate between an increased SCr due to an enhanced solute load and reduced clearance, rather than direct tubular damage. As such, their use in collapsed runners, who represent a higher-risk group, may be of future interest, as an addition to routine point-of-care testing.
Recommendations
Below are our pragmatic recommendations:
All marathon runners (or other endurance runners) should be given advice regarding the avoidance of NSAIDs, monitoring of urine volume and colour and the importance of adequate hydration.
Participants with pre-existing risk factors (for example, pre-existing renal dysfunction, hypertension and diabetes) should be made aware of the potential dangers of NSAIDs and systemic upset (such as vomiting) around the time of the event.
Runners who have collapsed during or at the end of a marathon, should have their observations recorded and basic investigations performed (including a urine dip and basic biochemistry); abnormal results should be followed up within a suitable time frame, according to local resources.
Future research could focus on biomarkers that more specifically reflect renal dysfunction, to help identify those at most risk and allow follow-up, where dysfunction has been found.
bmjsem-2015-000093supp001.pdf (405.9KB, pdf)
bmjsem-2015-000093supp002.pdf (258KB, pdf)
Footnotes
Contributors: LH: conception, design, literature search, risk of bias assessment, write-up. EW: conception, design, literature search, write-up. RV: conception, design, literature search, write-up. RG: conception, literature search, write-up. YP: conception design, write-up. FS: literature search, write-up. LF: conception design, literature search, risk of bias assessment, write-up.
Competing interests: None declared.
Provenance and peer review: Not commissioned; internally peer reviewed.
References
- 1. RunningUSA. Annual Marathon Report, Running USA. 2014.
- 2. Zaryski C, Smith DJ. Training principles and issues for ultra-endurance athletes. Curr Sports Med Rep 2005;4:165–70. 10.1097/01.CSMR.0000306201.49315.73 [DOI] [PubMed] [Google Scholar]
- 3. Chakravarty EF, Hubert HB, Lingala VB, et al. . Reduced disability and mortality among aging runners: a 21-year longitudinal study. Arch Intern Med 2008;168:1638–46. 10.1001/archinte.168.15.1638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Sarna S, Sahi T, Koskenvuo M, et al. . Increased life expectancy of world class male athletes. Med Sci Sports Exerc 1993;25:237–44. 10.1249/00005768-199302000-00013 [DOI] [PubMed] [Google Scholar]
- 5. Schnohr P, Marott JL, Lange P, et al. . Longevity in male and female joggers: the Copenhagen City Heart Study. Am J Epidemiol 2013;177:683–9. 10.1093/aje/kws301 [DOI] [PubMed] [Google Scholar]
- 6. Pugh A, Bourke JP, Kunadian V. Sudden cardiac death among competitive adult athletes: a review. Postgrad Med J 2012;88:382–90. 10.1136/postgradmedj-2011-130215 [DOI] [PubMed] [Google Scholar]
- 7. Sharma S, Whyte G, McKenna WJ. Sudden death from cardiovascular disease in young athletes: fact or fiction? Br J Sports Med 1997;31:269–76. 10.1136/bjsm.31.4.269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kim JH, Malhotra R, Chiampas G, et al. ; Race Associated Cardiac Arrest Event Registry (RACER) Study Group. Cardiac arrest during long-distance running races. N Engl J Med 2012;366:130–40. 10.1056/NEJMoa1106468 [DOI] [PubMed] [Google Scholar]
- 9. Corrado D, Basso C, Thiene G. Sudden cardiac death in athletes: what is the role of screening? Curr Opin Cardiol 2012;27:41–8. 10.1097/HCO.0b013e32834dc4cb [DOI] [PubMed] [Google Scholar]
- 10. Benito B, Gay-Jordi G, Serrano-Mollar A, et al. . Cardiac arrhythmogenic remodeling in a rat model of long-term intensive exercise training. Circulation 2011;123:13–22. 10.1161/CIRCULATIONAHA.110.938282 [DOI] [PubMed] [Google Scholar]
- 11. Scharhag J, George K, Shave R, et al. . Exercise-associated increases in cardiac biomarkers. Med Sci Sports Exerc 2008;40:1408–15. 10.1249/MSS.0b013e318172cf22 [DOI] [PubMed] [Google Scholar]
- 12. Middleton N, George K, Whyte G, et al. . Cardiac troponin T release is stimulated by endurance exercise in healthy humans. J Am Coll Cardiol 2008;52:1813–4. 10.1016/j.jacc.2008.03.069 [DOI] [PubMed] [Google Scholar]
- 13. Rifai N, Douglas PS, O'Toole M, et al. . Cardiac troponin T and I, echocardiographic [correction of electrocardiographic] wall motion analyses, and ejection fractions in athletes participating in the Hawaii Ironman Triathlon. Am J Cardiol 1999;83:1085–9. 10.1016/S0002-9149(99)00020-X [DOI] [PubMed] [Google Scholar]
- 14. Neilan TG, Januzzi JL, Lee-Lewandrowski E, et al. . Myocardial injury and ventricular dysfunction related to training levels among nonelite participants in the Boston marathon. Circulation 2006;114:2325–33. 10.1161/CIRCULATIONAHA.106.647461 [DOI] [PubMed] [Google Scholar]
- 15. Shave R, Baggish A, George K, et al. . Exercise-induced cardiac troponin elevation: evidence, mechanisms, and implications. J Am Coll Cardiol 2010;56:169–76. 10.1016/j.jacc.2010.03.037 [DOI] [PubMed] [Google Scholar]
- 16. Neumayr G, Gaenzer H, Pfister R, et al. . Plasma levels of cardiac troponin I after prolonged strenuous endurance exercise. Am J Cardiol 2001;87:369-–71. [DOI] [PubMed] [Google Scholar]
- 17. Scherr J, Braun S, Schuster T, et al. . 72-h kinetics of high-sensitive troponin T and inflammatory markers after marathon. Med Sci Sports Exerc 2011;43:1819–27. 10.1249/MSS.0b013e31821b12eb [DOI] [PubMed] [Google Scholar]
- 18. Neilan TG, Yoerger DM, Douglas PS, et al. . Persistent and reversible cardiac dysfunction among amateur marathon runners. Eur Heart J 2006;27:1079–84. 10.1093/eurheartj/ehi813 [DOI] [PubMed] [Google Scholar]
- 19. Trivax JE, Franklin BA, Goldstein JA, et al. . Acute cardiac effects of marathon running. J Appl Physiol 2010;108:1148–53. 10.1152/japplphysiol.01151.2009 [DOI] [PubMed] [Google Scholar]
- 20. Lee DC, Pate RR, Lavie CJ, et al. . Leisure-time running reduces all-cause and cardiovascular mortality risk. J Am Coll Cardiol 2014;64:472–81. 10.1016/j.jacc.2014.04.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Schnohr P, O'Keefe JH, Marott JL, et al. . Dose of jogging and long-term mortality: the Copenhagen City Heart Study. J Am Coll Cardiol 2015;65:411–9. 10.1016/j.jacc.2014.11.023 [DOI] [PubMed] [Google Scholar]
- 22. Mons U, Hahmann H, Brenner H. A reverse J-shaped association of leisure time physical activity with prognosis in patients with stable coronary heart disease: evidence from a large cohort with repeated measurements. Heart 2014;100:1043–9. 10.1136/heartjnl-2013-305242 [DOI] [PubMed] [Google Scholar]
- 23. Bellomo R, Ronco C, Kellum JA, et al. ; Acute Dialysis Quality Initiative workgroup. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 2004;8:R204–12. 10.1186/cc2872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. KDIGO clinical practice guideline for acute kidney injury. Kidney Int 2012;2(Suppl 1):1–136. 10.1038/kisup.2012.1 [DOI] [Google Scholar]
- 25. Chertow GM, Burdick E, Honour M, et al. . Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol 2005;16:3365–70. 10.1681/ASN.2004090740 [DOI] [PubMed] [Google Scholar]
- 26. Lassnigg A, Schmid ER, Hiesmayr M, et al. . Impact of minimal increases in serum creatinine on outcome in patients after cardiothoracic surgery: do we have to revise current definitions of acute renal failure? Crit Care Med 2008;36:1129–37. 10.1097/CCM.0b013e318169181a [DOI] [PubMed] [Google Scholar]
- 27. Newsome BB, Warnock DG, McClellan WM, et al. . Long-term risk of mortality and end-stage renal disease among the elderly after small increases in serum creatinine level during hospitalization for acute myocardial infarction. Arch Intern Med 2008;168:609–16. 10.1001/archinte.168.6.609 [DOI] [PubMed] [Google Scholar]
- 28. Bywaters EG, Beall D. Crush Injuries with Impairment of renal Function. Br Med J 1941;1:427–32. 10.1136/bmj.1.4185.427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ra B. Urinary findings before and after a marathon race. N Engl J Med 1941;225:801–2. [Google Scholar]
- 30. Dancaster CP, Duckworth WC, Roper CJ. Nephropathy in marathon runners. S Afr Med J 1969;43:758–60. [PubMed] [Google Scholar]
- 31. Seedat YK, Aboo N, Naicker S, et al. . Acute renal failure in the "Comrades Marathon" runners. Ren Fail 1989;11:209–12. [DOI] [PubMed] [Google Scholar]
- 32. Schiff HB, MacSearraigh ET, Kallmeyer JC. Myoglobinuria, rhabdomyolysis and marathon running. Q J Med 1978;47:463–72. [PubMed] [Google Scholar]
- 33. Vitting KE, Nichols NJ, Seligson GR. Naproxen and acute renal failure in a runner. Ann Intern Med 1986;105:144. [DOI] [PubMed] [Google Scholar]
- 34. Whitworth JA, Wolfman MJ. Fatal heat stroke in a long distance runner. Br Med J 1983;287:948 10.1136/bmj.287.6397.948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Irving RA, Noakes TD, Irving GA, et al. . The immediate and delayed effects of marathon running on renal function. J Urol 1986;136:1176–80. [DOI] [PubMed] [Google Scholar]
- 36. Antman EM, Lau J, Kupelnick B, et al. . A comparison of results of meta-analyses of randomized control trials and recommendations of clinical experts. treatments for myocardial infarction. JAMA 1992;268:240–8. [PubMed] [Google Scholar]
- 37. Oxman AD, Guyatt GH. The science of reviewing research. Ann N Y Acad Sci 1993;703:125–33. 10.1111/j.1749-6632.1993.tb26342.x [DOI] [PubMed] [Google Scholar]
- 38. Wells G, Shea B, Peterson J, et al. . The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses.
- 39. Patra J, Bhatia M, Suraweera W, et al. . Exposure to second-hand smoke and the risk of tuberculosis in children and adults: a systematic review and meta-analysis of 18 observational studies. PLoS Med 2015;12:e1001835 10.1371/journal.pmed.1001835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Pollard TD, Weiss IW. Acute tubular necrosis in a patient with march hemoglobinuria. N Engl J Med 1970;283:803–4. 10.1056/NEJM197010082831509 [DOI] [PubMed] [Google Scholar]
- 41. Bar-Sela S, Tur-Kaspa R, Eliakim M. Rhabdomyolysis and acute renal failure in a marathon runner in israel. Isr J Med Sci 1979;15:464–6. [PubMed] [Google Scholar]
- 42. Goldsmith HJ. Acute renal failure after a marathon run. Lancet 1984;1:278–9. 10.1016/S0140-6736(84)90149-1 [DOI] [PubMed] [Google Scholar]
- 43. Le Meur Y, Paraf F, Szelag JC, et al. . Acute renal failure in a marathon runner: role of glomerular bleeding in tubular injury. Am J Med 1998;105:251–2. [DOI] [PubMed] [Google Scholar]
- 44. Bruso JR, Hoffman MD, Rogers IR, et al. . Rhabdomyolysis and hyponatremia: a cluster of five cases at the 161-km 2009 western States endurance Run. Wilderness Environ Med 2010;21:303–8. 10.1016/j.wem.2010.06.012 [DOI] [PubMed] [Google Scholar]
- 45. MacSearraigh ET, Kallmeyer JC, Schiff HB. Acute renal failure in marathon runners. Nephron 1979;24:236–40. [DOI] [PubMed] [Google Scholar]
- 46. van Zyl-Smit R, Mills P, Vogelpoel L. Unrecognized acute renal failure following the comrades marathon. S Afr Med J 2000;90:39–40. [PubMed] [Google Scholar]
- 47. Boulter J, Noakes TD, Hew-Butler T. Acute renal failure in four comrades marathon runners ingesting the same electrolyte supplement: coincidence or causation? S Afr Med J 2011;101:876–8. [PubMed] [Google Scholar]
- 48. Riley WJ, Pyke FS, Roberts AD, et al. . The effect of long-distance running on some biochemical variables. Clin Chim Acta 1975;65:83–9. [DOI] [PubMed] [Google Scholar]
- 49. Neviackas JA, Bauer JH. Renal function abnormalities induced by marathon running. South Med J 1981;74:1457–60. [DOI] [PubMed] [Google Scholar]
- 50. Kraemer RR, Brown BS. Alterations in plasma-volume-corrected blood components of marathon runners and concomitant relationship to performance. Eur J Appl Physiol Occup Physiol 1986;55:579–84. [DOI] [PubMed] [Google Scholar]
- 51. Holtzhausen LM, Noakes TD, Kroning B, et al. . Clinical and biochemical characteristics of collapsed ultra-marathon runners. Med Sci Sports Exerc 1994;26:1095–101. [PubMed] [Google Scholar]
- 52. Kratz A, Lewandrowski KB, Siegel AJ, et al. . Effect of marathon running on hematologic and biochemical laboratory parameters, including cardiac markers. Am J Clin Pathol 2002;118:856–63. 10.1309/14TY-2TDJ-1X0Y-1V6V [DOI] [PubMed] [Google Scholar]
- 53. Reid SA, Speedy DB, Thompson JM, et al. . Study of hematological and biochemical parameters in runners completing a standard marathon. Clin J Sport Med 2004;14:344–53. [DOI] [PubMed] [Google Scholar]
- 54. Mingels A, Jacobs L, Kleijnen V, et al. . Cystatin C a marker for renal function after exercise. Int J Sports Med 2009;30:668–71. 10.1055/s-0029-1220733 [DOI] [PubMed] [Google Scholar]
- 55. McCullough PA, Chinnaiyan KM, Gallagher MJ, et al. . Changes in renal markers and acute kidney injury after marathon running. Nephrology 2011;16:194–9. 10.1111/j.1440-1797.2010.01354.x [DOI] [PubMed] [Google Scholar]
- 56. Mydlík M, Derzsiová K, Bohus B. Renal function abnormalities after marathon run and 16-kilometre long-distance run. Przegl Lek 2012;69:1–4. [PubMed] [Google Scholar]
- 57. Borkowski J, Sobiech KA. Protein: creatinine and trypsin inhibitor: creatinine ratios in the urine of marathon runners. Eur J Appl Physiol Occup Physiol 1990;61:124–7. 10.1007/BF00236705 [DOI] [PubMed] [Google Scholar]
- 58. Nelson PB, Ellis D, Fu F, et al. . Fluid and electrolyte balance during a cool weather marathon. Am J Sports Med 1989;17:770–2. [DOI] [PubMed] [Google Scholar]
- 59. Hewing B, Schattke S, Spethmann S, et al. . Cardiac and renal function in a large cohort of amateur marathon runners. Cardiovasc Ultrasound 2015;13:13:13 10.1186/s12947-015-0007-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Irving RA, Noakes TD, Burger SC, et al. . Plasma volume and renal function during and after ultramarathon running. Med Sci Sports Exerc 1990;22:581–7. [DOI] [PubMed] [Google Scholar]
- 61. Irving RA, Noakes TD, Buck R, et al. . Evaluation of renal function and fluid homeostasis during recovery from exercise-induced hyponatremia. J Appl Physiol 1991;70:342–8. [DOI] [PubMed] [Google Scholar]
- 62. Lippi G, Sanchis-Gomar F, Salvagno GL, et al. . Variation of serum and urinary neutrophil gelatinase associated lipocalin (NGAL) after strenuous physical exercise. Clin Chem Lab Med 2012;50:1585–9. 10.1515/cclm-2011-0954 [DOI] [PubMed] [Google Scholar]
- 63. Hoffman MD, Stuempfle KJ, Fogard K, et al. . Urine dipstick analysis for identification of runners susceptible to acute kidney injury following an ultramarathon. J Sports Sci 2013;31:20–31. 10.1080/02640414.2012.720705 [DOI] [PubMed] [Google Scholar]
- 64. Noakes TD, Carter JW. Biochemical parameters in athletes before and after having run 160 kilometres. S Afr Med J 1976;50:1562–6. [PubMed] [Google Scholar]
- 65. Irving RA, Noakes TD, Raine RI, et al. . Transient oliguria with renal tubular dysfunction after a 90 km running race. Med Sci Sports Exerc 1990;22:756–61. [DOI] [PubMed] [Google Scholar]
- 66. Lipman GS, Krabak BJ, Waite BL, et al. . A prospective cohort study of acute kidney injury in multi-stage ultramarathon runners: the Biochemistry in endurance Runner Study (BIERS). Res Sports Med 2014;22:185–92. 10.1080/15438627.2014.881824 [DOI] [PubMed] [Google Scholar]
- 67. Hou SK, Chiu YH, Tsai YF, et al. . Clinical Impact of Speed Variability to identify Ultramarathon Runners at Risk for acute Kidney Injury. PLoS One 2015;10:e0133146 10.1371/journal.pone.0133146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Christensen DL, Espino D, Infante-Ramírez R, et al. . Normalization of elevated cardiac, kidney, and hemolysis plasma markers within 48 h in Mexican Tarahumara runners following a 78 km race at moderate altitude. Am J Hum Biol 2014;26:836–43. 10.1002/ajhb.22607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Dumke CL, Nieman DC, Oley K, et al. . Ibuprofen does not affect serum electrolyte concentrations after an ultradistance run. Br J Sports Med 2007;41:492–6. 10.1136/bjsm.2006.033597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Lippi G, Schena F, Salvagno GL, et al. . Acute variation of estimated glomerular filtration rate following a half-marathon run. Int J Sports Med 2008;29:948–51. 10.1055/s-2008-1038745 [DOI] [PubMed] [Google Scholar]
- 71. Mohseni M, Silvers S, McNeil R, et al. . Prevalence of hyponatremia, renal dysfunction, and other electrolyte abnormalities among runners before and after completing a marathon or half marathon. Sports Health 2011;3:145–51. 10.1177/1941738111400561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Wharam PC, Speedy DB, Noakes TD, et al. . NSAID use increases the risk of developing hyponatremia during an ironman triathlon. Med Sci Sports Exerc 2006;38:618–22. 10.1249/01.mss.0000210209.40694.09 [DOI] [PubMed] [Google Scholar]
- 73. Irving RA, Noakes TD, van Zyl Smit R. Metabolic and renal changes in two athletes during a world 24 hour relay record performance. Br J Sports Med 1989;23:227–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Page AJ, Reid SA, Speedy DB, et al. . Exercise-associated hyponatremia, renal function, and nonsteroidal antiinflammatory drug use in an ultraendurance mountain run. Clin J Sport Med 2007;17:43–8. 10.1097/JSM.0b013e31802b5be9 [DOI] [PubMed] [Google Scholar]
- 75. Neumayr G, Pfister R, Hoertnagl H, et al. . The effect of marathon cycling on renal function. Int J Sports Med 2003;24:131–7. 10.1055/s-2003-38205 [DOI] [PubMed] [Google Scholar]
- 76. Neumayr G, Pfister R, Hoertnagl H, et al. . Renal function and plasma volume following ultramarathon cycling. Int J Sports Med 2005;26:2–8. 10.1055/s-2004-815717 [DOI] [PubMed] [Google Scholar]
- 77. Reid SA, King MJ. Serum biochemistry and morbidity among runners presenting for medical care after an Australian mountain ultramarathon. Clin J Sport Med 2007;17:307–10. 10.1097/JSM.0b013e31804c77da [DOI] [PubMed] [Google Scholar]
- 78. Schütz UH, Schmidt-Trucksäss A, Knechtle B, et al. ; The TransEurope FootRace Project. longitudinal data acquisition in a cluster randomized mobile MRI observational cohort study on 44 endurance runners at a 64-stage 4486 km transcontinental ultramarathon. BMC Med 2012;10:78 10.1186/1741-7015-10-78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Dharnidharka VR, Kwon C, Stevens G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis. Am J Kidney Dis 2002;40:221–6. 10.1053/ajkd.2002.34487 [DOI] [PubMed] [Google Scholar]
- 80. Kashani K, Al-Khafaji A, Ardiles T, et al. . Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit Care 2013;17:R25 10.1186/cc12503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Bihorac A, Chawla LS, Shaw AD, et al. . Validation of cell-cycle arrest biomarkers for acute kidney injury using clinical adjudication. Am J Respir Crit Care Med 2014;189:932–9. 10.1164/rccm.201401-0077OC [DOI] [PubMed] [Google Scholar]
- 82. Koyner JL, Shaw AD, Chawla LS, et al. ; Sapphire Investigators. Tissue Inhibitor Metalloproteinase-2 (TIMP-2)⋅IGF-Binding Protein-7 (IGFBP7) Levels Are Associated with Adverse Long-Term Outcomes in Patients with AKI. J Am Soc Nephrol 2015;26:1747–54. 10.1681/ASN.2014060556 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Castenfors J, Mossfeldt F, Piscator M. Effect of prolonged heavy exercise on renal function and urinary protein excretion. Acta Physiol Scand 1967;70:194–206. 10.1111/j.1748-1716.1967.tb03615.x [DOI] [PubMed] [Google Scholar]
- 84. Poortmans JR. Exercise and renal function. Sports Med 1984;1:125–53. [DOI] [PubMed] [Google Scholar]
- 85. Castenfors J. Renal function during prolonged exercise. Ann N Y Acad Sci 1977;301:151–9. [DOI] [PubMed] [Google Scholar]
- 86. Grimby G. Renal clearances during prolonged supine exercise at different loads. Journal of Applied Physiology 1965;20:1294–8. [DOI] [PubMed] [Google Scholar]
- 87. Köhler H. Fluid metabolism in exercise. Kidney Int Suppl 1987;21:S93–6. [PubMed] [Google Scholar]
- 88. Poortmans JR, Vanderstraeten J. Kidney function during exercise in healthy and diseased humans. an update. Sports Med 1994;18:419–37. 10.2165/00007256-199418060-00006 [DOI] [PubMed] [Google Scholar]
- 89. Röcker L, Kirsch KA, Heyduck B, et al. . Influence of prolonged physical exercise on plasma volume, plasma proteins, electrolytes, and fluid-regulating hormones. Int J Sports Med 1989;10:270–4. 10.1055/s-2007-1024914 [DOI] [PubMed] [Google Scholar]
- 90. Kosunen KJ, Pakarinen AJ. Plasma renin, angiotensin II, and plasma and urinary aldosterone in running exercise. J Appl Physiol 1976;41:26–9. [DOI] [PubMed] [Google Scholar]
- 91. McAllister RM. Adaptations in control of blood flow with training: splanchnic and renal blood flows. Med Sci Sports Exerc 1998;30:375–81. [DOI] [PubMed] [Google Scholar]
- 92. Armstrong RB. Muscle damage and endurance events. Sports Med 1986;3:370–81. [DOI] [PubMed] [Google Scholar]
- 93. Newham DJ, Jones DA, Edwards RH. Large delayed plasma creatine kinase changes after stepping exercise. Muscle Nerve 1983;6:380–5. 10.1002/mus.880060507 [DOI] [PubMed] [Google Scholar]
- 94. Noakes TD. Effect of exercise on serum enzyme activities in humans. Sports Med 1987;4:245–67. [DOI] [PubMed] [Google Scholar]
- 95. Brody SL, Wrenn KD, Wilber MM, et al. . Predicting the severity of cocaine-associated rhabdomyolysis. Ann Emerg Med 1990;19:1137–43. [DOI] [PubMed] [Google Scholar]
- 96. Ward MM. Factors predictive of acute renal failure in rhabdomyolysis. Arch Intern Med 1988;148:1553–7. [PubMed] [Google Scholar]
- 97. Skenderi KP, Kavouras SA, Anastasiou CA, et al. . Exertional Rhabdomyolysis during a 246-km continuous running race. Med Sci Sports Exerc 2006;38:1054–7. 10.1249/01.mss.0000222831.35897.5f [DOI] [PubMed] [Google Scholar]
- 98. Demos MA, Gitin EL, Kagen LJ. Exercise myoglobinemia and acute exertional rhabdomyolysis. Arch Intern Med 1974;134:669–73. [PubMed] [Google Scholar]
- 99. Bosch X, Poch E, Grau JM. Rhabdomyolysis and acute kidney injury. N Engl J Med 2009;361:62–72. 10.1056/NEJMra0801327 [DOI] [PubMed] [Google Scholar]
- 100. Murray MD, Brater DC. Renal toxicity of the nonsteroidal anti-inflammatory drugs. Annu Rev Pharmacol Toxicol 1993;33:435–65. 10.1146/annurev.pa.33.040193.002251 [DOI] [PubMed] [Google Scholar]
- 101. Kramer HJ, Stinnesbeck B, Klautke G, et al. . Interaction of renal prostaglandins with the renin-angiotensin and renal adrenergic nervous systems in healthy subjects during dietary changes in sodium intake. Clin Sci 1985;68:387–93. 10.1042/cs0680387 [DOI] [PubMed] [Google Scholar]
- 102. Stokes JB. Effect of prostaglandin E2 on chloride transport across the rabbit thick ascending limb of Henle. selective inhibitions of the medullary portion. J Clin Invest 1979;64:495–502. 10.1172/JCI109487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Anderson RJ, Berl T, McDonald KD, et al. . Evidence for an in vivo antagonism between vasopressin and prostaglandin in the mammalian kidney. J Clin Invest 1975;56:420–6. 10.1172/JCI108108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Whelton A. Nephrotoxicity of nonsteroidal anti-inflammatory drugs: physiologic foundations and clinical implications. Am J Med 1999;106:S13–24. 10.1016/S0002-9343(99)00113-8 [DOI] [PubMed] [Google Scholar]
- 105. Sedor JR, Davidson EW, Dunn MJ. Effects of nonsteroidal anti-inflammatory drugs in healthy subjects. Am J Med 1986;81:58–70. 10.1016/0002-9343(86)90908-3 [DOI] [PubMed] [Google Scholar]
- 106. Walker RJ, Fawcett JP, Flannery EM, et al. . Indomethacin potentiates exercise-induced reduction in renal hemodynamics in athletes. Med Sci Sports Exerc 1994;26:1302–6. [PubMed] [Google Scholar]
- 107. Olsen NV, Jensen NG, Hansen JM, et al. . Non-steroidal anti-inflammatory drugs and renal response to exercise: a comparison of Indomethacin and nabumetone. Clin Sci 1999;97:457–65. 10.1042/cs0970457 [DOI] [PubMed] [Google Scholar]
- 108. Donnelly AE, Maughan RJ, Whiting PH. Effects of ibuprofen on exercise-induced muscle soreness and indices of muscle damage. Br J Sports Med 1990;24:191–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Bourgeois J, MacDougall D, MacDonald J, et al. . Naproxen does not alter indices of muscle damage in resistance-exercise trained men. Med Sci Sports Exerc 1999;31:4–9. [DOI] [PubMed] [Google Scholar]
- 110. Donnelly AE, McCormick K, Maughan RJ, et al. . Effects of a non-steroidal anti-inflammatory drug on delayed onset muscle soreness and indices of damage. Br J Sports Med 1988;22:35–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Carter R. 3rd, Cheuvront SN, Williams JO, et al. Epidemiology of hospitalizations and deaths from heat illness in soldiers. Med Sci Sports Exerc 2005;37:1338–44. [DOI] [PubMed] [Google Scholar]
- 112. Pattison ME, Logan JL, Lee SM, et al. . Exertional heat stroke and acute renal failure in a young woman. Am J Kidney Dis 1988;11:184–7. [DOI] [PubMed] [Google Scholar]
- 113. Holman ND, Schneider AJ. Multi-organ damage in exertional heat stroke. Neth J Med 1989;35:38–43. [PubMed] [Google Scholar]
- 114. Roberts WO. Exertional heat stroke during a cool weather marathon: a case study. Med Sci Sports Exerc 2006;38:1197–203. 10.1249/01.mss.0000227302.80783.0f [DOI] [PubMed] [Google Scholar]
- 115. Walter E, Venn R, Stevenson T. Exertional Heat Stroke – The Athlete's Nemesis. Journal of the Intensive Care Society 2012;13:304–8. 10.1177/175114371201300408 [DOI] [Google Scholar]
- 116. Bouchama A, Knochel JP. Heat stroke.N Engl J Med 2002;346:1978–88. [DOI] [PubMed] [Google Scholar]
- 117. Racinais S, Alonso JM, Coutts AJ, et al. . Consensus recommendations on training and competing in the heat. Sports Med 2015;45:925–38. 10.1007/s40279-015-0343-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Ishikawa I. Acute renal failure with severe loin pain and patchy renal ischemia after anaerobic exercise in patients with or without renal hypouricemia. Nephron 2002;91:559–70. doi:65013 [DOI] [PubMed] [Google Scholar]
- 119. Izumi M, Yokoyama K, Yamauchi A, et al. . A young man with acute renal failure and severe loin pain. Nephron 1997;76:215–7. 10.1159/000190171 [DOI] [PubMed] [Google Scholar]
- 120. Han JS, Kim YG, Kim S, et al. . Bone scintigraphy in acute renal failure with severe loin pain and patchy renal vasoconstriction. Nephron 1991;59:254–60. [DOI] [PubMed] [Google Scholar]
- 121. Mehta RL, Kellum JA, Shah SV, et al. ; Acute Kidney Injury Network. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 2007;11:R31 10.1186/cc5713 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjsem-2015-000093supp001.pdf (405.9KB, pdf)
bmjsem-2015-000093supp002.pdf (258KB, pdf)