Introduction
Nephrotic syndrome (NS), a condition that may occur in the context of various glomerular diseases, has been associated with venous thromboembolism (VTE) and less commonly with arterial thromboembolism (ATE). Although the association of ATE with NS has been challenged in the past,1 there is increasing evidence supporting this hypothesis.2, 3 ATE in NS patients has been reported in several sites such as the coronary, peripheral, and cerebral circulations.3 Multiple factors have been associated with the risk of thromboembolism in NS patients. However, predicting and preventing these complications remain a challenge for nephrologists.3 In particular, little is known about the risk of ischemic stroke, which is a potential but rare complication of NS that has only been described in a limited number of case reports. Considering the substantial mortality and long-term disability associated with stroke, identifying NS patients at higher risk of ATE is an important issue.4, 5 Here we present a case of ischemic stroke in a patient with NS and a review of the literature with the objective of delineating patients at risk of stroke in this context (Table 1). We also reviewed the etiologic mechanism of stroke and classified it using the TOAST (Trial of Org 101172 in Acute Stroke Treatment) system.6
Table 1.
Teaching points
| Nephrotic syndrome is associated with a higher risk of both venous and arterial thrombosis than the general population. |
| Ischemic stroke can occur in young patients and be the initial event in nephrotic syndrome. |
| Traditional cardiovascular risk factors, especially smoking, seem to play a role in arterial thrombosis as in patients without nephrotic syndrome. They should be assessed and managed in all patients with nephrotic syndrome. |
| Severe hypoalbuminemia and proteinuria seem to play a role in venous thromboembolism, but their contribution to arterial thromboembolism is less clear. |
| Anticoagulation and antiplatelet agents seem to be protective for acute ischemic stroke in patients in NS but their role in primary and secondary prophylaxis has yet to be determined. |
Case Presentation
A 43-year-old woman was referred to our center for an acute ischemic stroke. Her medical history revealed that she had received a diagnosis of membranous nephropathy 10 years previously and had entered spontaneous remission with complete resolution of the proteinuria at that time. Five months before the current episode, she experienced a relapse of NS. She was then started on furosemide, perindopril, and atorvastatin. Laboratory values at relapse (Table 2) revealed a serum creatinine level of 57 μmol/l (reference range, 53–97 μmol/l), albuminemia of 22 g/l (reference range, 35–52 g/l), and a spot urine protein/creatinine ratio of 1.397 g/mmol. Of note, antimyeloperoxidase was detected by the automated Luminex-based immunoassay system Bio-Plex 200 (Bio-Rad, Hercules, CA) with the result that 7.7 arbitrary units (reference range, 0.0–0.9 arbitrary units) and antiproteinase 3 were negative.7 Serum antiphospholipase 2 receptor antibody were weakly positive using a semiquantitative immunofluorescence technique (negative to high positive). The patient was not taking any medication that has been associated with anti–neutrophil cytoplasmic antibody–positive vasculitis.8 A kidney biopsy was performed that revealed 11 glomeruli, 1 of which was sclerotic. All glomeruli showed glomerular basement membrane thickening without any inflammation, endocapillary proliferation, or crescent formation. Mild interstitial fibrosis was present. Arteries and arterioles were unremarkable. Immunofluorescence microscopy showed granular staining for IgG (3+) κ (3+) and λ (3+) light chains as well as focal granular deposits for C3 on the glomerular basement membrane. Only traces of IgM and C1q were detected. Electron microscopy showed scattered subepithelial deposits. The biopsy showed no evidence of vasculitis. Staining for anti–phospholipase A2 receptor was not performed. The diagnosis of membranous nephropathy was made.
Table 2.
Summary of laboratory results
| Laboratory test | Laboratory value at relapse of nephrotic syndrome | Reference range | Laboratory value at presentation for acute stroke | Reference range |
|---|---|---|---|---|
| Leukocyte (109/l) | 11.3 | 4.5–10.8 | 19 | 4.0–11.0 |
| Hb (g/l) | 138 | 117–157 | 117 | 120–160 |
| Ht (%) | 0.414 | 0.370–0.470 | 0.347 | 3.9–5.5 |
| Platelet (104/mm3) | 433 | 140–400 | 369 | 145–470 |
| Creatinine (μmol/l) | 57 | 53–97 | 56 | 42–89 |
| Glucose (mmol/l) | 4.1 | 4.0–6.2 | ||
| HbA1c | 0.0527 | 0.0440–0.0600 | ||
| Albumin (g/l) | 22 | 35–52 | 13 | 36–45 |
| Protein/creatinine spot ratio (g/mmol) | 1.397 | 0.828 | ||
| Proteinuria (g/24 h) | 4.84 | |||
| Cholesterol (mmol/l) | 9.33 | 8.25 | 3.40–7.30 | |
| HDL (mmol/l) | 1.03 | 0.95 | 0.90–2.38 | |
| LDL (mmol/l) | 7.2 | 5.68 | ||
| Non–HDL cholesterol (mmol/l) | 8.30 | 7.30 | ||
| Triglycerides (mmol/l) | 2.45 | 0.45–2.25 | 3.55 | 0.43–2.69 |
| C reactive protein (mg/l) | <5.0 | <9.99 | ||
| aPTT (s) | 23 | 22–31 | ||
| INR | 1.0 | 0.9–1.2 | ||
| Thrombin time (s) | 23 | 14–18 | ||
| Fg (g/l) | 5.39 | 2.00–4.50 | ||
| AT III (U/l) | 1.24 | 0.80–1.20 | ||
| Functional protein C (U/l) | 1.45 | 0.70–1.40 | ||
| Free protein S antigen (U/l) | 1.23 | 0.69–1.31 | ||
| Homocysteine (μg/l) | 10 | <12 | ||
| C3 (g/l) | 1.08 | 0.85–2.00 | ||
| C4 (g/l) | 0.23 | 0.10–0.50 | ||
| ANA | Negative | Negative | ||
| ENA | Negative | |||
| Anti-PR3 (AU) | <0.2 | 0.0–0.9 | <0.2 | 0.0–0.9 |
| Antimyeloperoxidase (AU) | 7.7 | 0.0–0.9 | 2.2 | 0.0–0.9 |
| Anti–glomerular basal membrane | <0.2 | <1.0 | ||
| Serum anti–PLAR2 antibody | Weakly positive | |||
| Factor V Leiden | Negative | |||
| Mutation factor II | Negative | |||
| Lupus anticoagulant | Negative | |||
| Anti–cardiolipins IgM (MPL-U/ml) | 0.3 | <12.5 | ||
| Anti–cardiolipin IgG (GPL-U/ml) | <1.6 | <15 |
aPTT, activated partial thromboplastin time; ANA, antinuclear antibody; AT III, antithrombin III; AU, arbitrary units; ENA, extractable nuclear antigens; Fg, fibrinogen; GPL-U, G phospholipids-unit; Hb, hemoglobin; HDL, high-density lipoprotein; Ht, hematocrite; INR, international normalized ratio; LDL, low-density lipoprotein; MPL-U, M phospholipids-unit; MPO, myeloperoxidase; PLAR2, phospholipase A2 receptor.
The patient was then started on a modified Ponticelli protocol.9 She had received 3 days of i.v. methylprednisolone at a dose of 1 g/d and 10 days of 0.5 mg/kg per day of oral prednisone when left hemiparesis developed. Her medication at the time was prednisone 30 mg/d, furosemide 20 mg/d, perindopril 4 mg/d, atorvastatin 20 mg/d, alendronate 70 mg every week, vitamin D, calcium, and ranitidine. She was an active smoker but had no previous episode of thrombosis and no family history of thrombosis. Head computed tomodensitometry performed at the referring center showed acute ischemic changes (ASPECT [Alberta Stroke Program Early Computed Tomography10] score of 6) in the right middle cerebral artery territory. She received a diagnosis of acute ischemic stroke at the referring center. Intravenous thrombolysis with recombinant tissue plasminogen activator was started, and she was then referred to our center for endovascular thrombectomy.
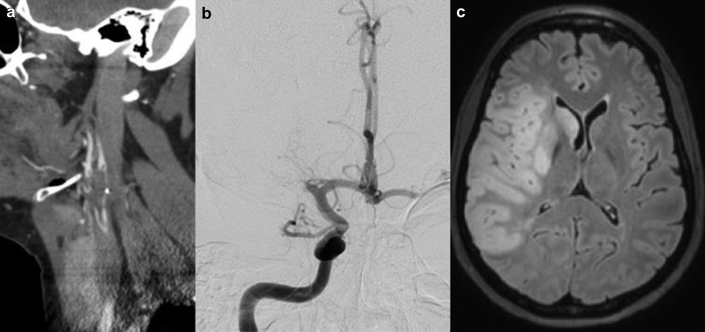
Upon transfer to our center, her physical examination still revealed left hemiplegia, hemianesthesia, hemianopsia, and heminegligence, with a National Institutes of Health Stroke Score of 20 (severe stroke).11 Her complete laboratory data can be found in Table 2. Notably, she had a serum creatinine level of 56 μmol/l (reference range, 42–89 μmol/l), a serum albumin level of 13 g/l (reference range, 36–45 g/l), a total cholesterol level of 8.25 mmol/l (reference range, 3.40–7.30 mmol/l), and a protein/creatinine spot ratio of 0.828 g/mmol, and 24-hour proteinuria was 4.84 g. The antimyeloperoxidase remained slightly elevated with 2.2 arbitrary units (reference range, 0–0.9 arbitrary units), in line with the results obtained from her referring center. Cerebral computed tomodensitometry angiography showed a thrombus in the right common carotid artery with extension in the internal and external carotid arteries as well as an occlusion of the right middle cerebral artery (Figure 1a). This large artery thrombus was considered a possible source of embolus to the cerebral circulation, although it could not be ruled out that the ischemic event resulted from total occlusion of this large vessel with subsequent recanalization. This patient would be considered to have large artery atherosclerosis per the TOAST system, which uses 5 subtypes to classify the etiology of ischemic stroke (large artery atherosclerosis, cardioembolic, small-vessel occlusion, stroke of other determined etiology, and stroke of undetermined etiology).6 The patient was brought to the angiography suite, and endovascular thrombectomy was performed (Figure 1b). Magnetic resonance imaging obtained on day 4 showed ischemic changes in the territory of the right middle cerebral artery (Figure 1c). Her ensuing clinical outcome was favorable with complete resolution of her neurologic symptoms.
Figure 1.
(a) Carotid computed tomodensitometry angiography showing a thrombus in the right external carotid bulb. (b) Angiography showing occlusion of the right middle cerebral artery. (c) Magnetic resonance imaging showing ischemic stroke in the territory of the right middle cerebral artery.
Further investigation for an underlying cause of ischemic stroke was negative. No arrhythmia was found during Holter monitoring. A transthoracic echocardiogram was normal, with no evidence of shunt or cardioembolic source. The carotid computed tomodensitometry angiography showed a thrombus in the right external carotid bulb but no atherosclerosis. The glycated hemoglobin level was normal. Further investigation for thrombophilia, including factor V Leiden, prothrombin mutation G20210A, protein S and C deficiencies, anticardiolipins, anti–β2-glycoprotein I, lupus anticoagulant, and hyperhomocysteinemia was negative. Considering her young age and a negative investigation for underlying cause, it was considered probable that the hypercoagulability associated with NS contributed to the pathogenesis of the ischemic stroke. Because of the presence of a thrombus in the external carotid artery, the decision was made to start anticoagulation therapy in this patient. Intravenous heparin treatment was initiated and later replaced by warfarin after transfer to the referring center. She completed the modified Ponticelli protocol and attained a partial remission with a proteinuria of 0.63 g/d.9
Discussion
NS is associated with a hypercoagulable state, which is thought to result from changes in platelet activation and aggregation as well as in the metabolism and concentration of coagulation proteins.12 Urinary loss of low molecular weight proteins such has plasminogen and antithrombin III could lead to impaired fibrinolysis and regulation of coagulation.12 Furthermore, hypoalbuminemia induces increased hepatic synthesis of clotting factors.13 Several studies exposed an increased risk of VTE in patients with NS.3, 14 Even though there are more data regarding the risk of VTE, NS also places patients at risk of ATE, including stroke. Notably, a single-center retrospective cohort study of 298 NS patients found an ATE incidence of 1.5% annually, which represents 8 times the risk of the age-matched population.3 In this study, the most common sites of ATE are myocardial infarction or unstable angina (44% and 14%, respectively) followed by peripheral arterial disease (14%) and ischemic stroke or transient ischemic attack (11.5% and 11.5%, respectively).3 Determining the factors that predict the risk of thrombosis in patients with NS is still an area of debate. The data available mainly come from retrospective observational studies and are conflicting. Furthermore, most of the data available describe the risk of VTE. Different factors have been suggested to influence the risk of ATE in NS patients. Those include the presence of classic cardiovascular risk factors, the severity of the hypoalbuminemia and proteinuria, the type of glomerular disease, the use of corticosteroids or diuretics, and urinary loss of high molecular weight molecules with antithrombotic properties.
In addition to our patient, only 21 cases of acute ischemic stroke in patients with NS were found in the English literature. The clinical information regarding these cases is summarized in Table 3.15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33
Table 3.
Review of the literature
| Case | Age, yr | Sex | Histology | Duration of NS, yr | Albumin, g/l | Proteinuria | Steroid use (Y/N) | Diuretic use (Y/N) | Presentation | Cerebral vascular territory involved | TOAST | Cardiovascular risk factors | Thrombophilia |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 (our case) |
43 | F | MN | 3 | 13 | 0.828 g/mmol | Y | Y | Left hemiplegia and hemianesthesia, hemianopsia, heminegligence | Right MCA | LAA | Smoking, HT, DLP | Fg ↑ |
| 215 | 55 | M | MN | 0.125 | 12 | 18 g/24 h | Y | Y | Death | Multiple | Possibly CE | Mild ASO | Fg ↑ |
| 316 | 23 | M | MCD | 0 | 7 | 10 g/24 h | N | N | Left hemiplegia, lower left limb ischemia | Right MCA | LAA | Alcohol | Fg ↑, AT III ↓, prot C ↓ |
| 417 | 36 | M | MPGN | 0 | 24 | 11.4 g/24 h | N | N | Right hemiparesis and hemianesthesia, global aphasia, right hemianopsia | Left MCA | LAA | — | Fg ↑, prot S ↓ |
| 517 | 34 | M | MN | 0 | 27 | 6.6 g/24 h | N | N | Left hemiparesis and hemianesthesia, dysarthria, left homonymous inferior visual field deficit | Right MCA | LAA | Smoking, cocaine | Fg ↑ |
| 618 | 28 | M | MN | 6 | 16 | 7.9 g/24 h | Y | N | Right hemiparesis, headache | Left basal ganglia and internal capsule | LAA | Smoking | Fg ↑, AT III ↓ |
| 718 | 21 | M | MCD | 1.5 | 15 | 5.6 g/24 h | Y | N | Loss of consciousness, aphasia | Left MCA | CE | Smoking | AT III ↓, prot C ↓ |
| 819 | 20 | M | MCD | 18 | 14 | >300 mg/dl | Y | N | Lethargy, left hemiparesia and hemianesthesis, hemineglect, left hemianopsia | Right MCA | LAA | — | Fg ↑, AT III ↓, prot S ↓ |
| 920 | 51 | M | No biopsy | 0 | 20 | 4.6 g/l | N | N | Headache | Right MCA | UND | Smoking | Fg ↑, ATII ↓ |
| 1021 | 37 | F | MN | 0 | 26 | NR | N | N | Right hemiparesis, expressive aphasia, right upper extremity ischemia | Right MCA | LAA | Obesity, smoking, HT | Fg ↑, prot S ↓ |
| 1122 | 39 | F | MPGN | 2 | 20 | 3.4 g/24 h | Y | Y | Left hemiparesis, dysarthria | Periventrical area of the right frontal lobe | UND | — | N Fg, prot S ↓ |
| 1223 | 29 | F | MPGN | 15 | 32 | 2.2 g/24 h | N | N | Left hemiparesis and hemianesthesia, aphasia | Right MCA, watershed right ACA-MCA | LAA | HT, DLP | Fg ↑ |
| 1324 | 36 | F | Db | NR | 11 | 2.8 g/24 h | NR | NR | Convulsions, left hemiplegia | Multiple | CE | Db | AT III ↓ |
| 1425 | 59 | M | MN | 0.038 | 17 | 12.0 g/24 h | Y | N | Convulsions | Bilateral occipital lobes | LAA | — | AT III ↓, |
| 1526 | 35 | F | IgA | 5 | 11 | 6.8 g/24 h | N | N | Right hemiparesis, dysarthria, dysphagia | Left MCA | Possibly CE | — | AT III ↓, prot C ↓ |
| 1627 | 53 | M | FSGS | 0.0625 | 12 | 7.8 g/24 h | Y | Y | Left hemiparesis, left hemianesthesia, | Right MCA | LAA | Smoking, ROH, cirrhosis | Prot S ↓ |
| 1728 | 28 | F | No biopsy | 0 | 26 | 3.5 g/24 h | N | N | Right hemiparesis, | Left MCA | UND | Smoking | — |
| 1829 | 61 | M | MN | 0 | 25 | 3.9 g/24 h | N | N | Left hemianesthesia | None identified on imaging | TIA | HT | — |
| 1930 | 72 | M | LCDD | 0 | 18 | 12.0 g/24 h | N | Y | Right hemiparesis, expressive aphasia | Left frontal lobe | UND | — | — |
| 2031 | 68 | M | MCD | 0 | 16 | 14.0 g/24 h | N | N | Left hemiparesis, left paresthesia, aphasia | Right MCA and PCA | Possibly CE | HT | — |
| 2123 | 71 | M | MN | 0 | 20 | 21.0 g/24 h | Y | N | Aphasia, right facial paralysis | Multiple | SAO | HT | Fg ↑, AT III ↓, prot S ↓ |
| 2233 | 35 | M | MN | 0 | 18 | 7.5 g/g | N | N | Right hemiparesis, dysarthria | Left MCA | LAA | Smoking | Fg ↑ |
ACA, anterior cerebral artery; ASO, antistreptolysin O; AT III, antithrombin III; CE, cardioembolic; Db, diabetic nephropathy; DLP, dyslipidemia; DLPx, F, female; Fg, fibrinogen; FSGS, focal and segmental glomerulosclerosis; HT, hypertension; LAA, large artery atherosclerosis; LCDD, light chain deposition disease; M, male; MCA, middle cerebral artery; MCD, minimal changes disease; MN, membranous nephropathy; MPGN, membranoproliferative glomerulonephritis; N, no; NI, not thoroughly investigated; NR, not reported; NS, nephrotic syndrome; PCA, posterior cerebral artery; prot C, protein C; prot S, protein S; ROH, SAO, small artery occlusion; TIA, transient ischemic attack; TOAST, Trial of Org 101172 in Acute Stroke TreatmenT; UND, undetermined; Y, yes.
We calculated and report 95% confidence intervals (CIs) for the estimated proportions of the different cardiovascular risk factors in patients with stroke and NS to provide an estimate of the uncertainty around that proportion and to allow for comparison with figures reported in the general stroke population. The median age at stroke presentation was 36 years. Fifty-nine percent (13 patients; 95% CI 36.4%–79.3%) were younger than 50 years of age compared with ∼10% in the general population diagnosed with stroke.34 Seven (32%; 95% CI 13.9%–54.9%) were women, and 15 (68%) were men. In the general population, stroke incidence rates are lower in women than in men in the younger age group but approximately equal in the older age group.34 Classic risk factors were found in 14 patients (63%) in the current review; 6 patients (27.3%; 95% CI 10.7%–50.2%) had previously diagnosed hypertension, 1 (4.5%; 95% CI 0–22.8%) had diabetes, and 9 (40.9%; 95% CI 20.7%–63.6%) were smokers. Hypertension was found in 77%, diabetes in 7.3%, and smoking 19.8% of patients with stroke in the general population.34 These data suggest that patients with NS diagnosed with stroke may be younger, with a lower prevalence of hypertension but more frequently smokers than patients with stroke in the general population. However, the number of patients reported and analyzed in this review is limited and may not be completely representative of all cases of stroke in NS.
Nine cases (40%) of stroke in NS were diagnosed in patients with membranous nephropathy, 4 (18%) had minimal change disease, 3 (14%) had membranoproliferative glomerulonephritis, 1 (5%) had focal segmental glomerulosclerosis, 1 (5%) had light chain deposition disease, 1 (5%) had diabetic nephropathy, and 1 (5%) had IgA nephropathy. Only 2 patients did not undergo kidney biopsy. Data regarding the link between ATE and the underlying type of glomerular disease are sparse. In the study by Mahmoodi et al.,3 membranous nephropathy histology was not found to be an independent risk factor for ATE. However, patients with membranous nephropathy tend to have more severe hypoalbuminemia and proteinuria than other histologies.14 Both hypoalbuminemia and proteinuria are markers of disease severity in NS and have been proposed to influence the risk of thrombosis.14, 35, 36, 37 In the current review of the literature, the mean serum albumin level was 18.4 g/l, and the mean proteinuria was 8.1 g/24 h. Serum albumin levels lower than 20 g/l and 15 g/l were seen in 15 patients (68.2%) and 7 patients (31.8%), respectively. Proteinuria >5 g/24 h was found in 13 patients (59.1%). Some authors suggested that diuretics and steroids may promote thrombosis in the setting of NS.14 Previous corticosteroid or diuretic treatment was found in 9 patients (41%) and 5 patients (23%), respectively. Eleven of the patients reported in the literature (50%) had ischemic stroke, while on neither diuretic nor corticosteroid treatment.
Certain studies observed that the risk of thrombosis is highest shortly after the diagnosis of NS.3 Similarly, in this review, the acute cerebral thrombosis was diagnosed within 6 months in 14 cases (63%) and within 1 year in 6 cases (29%). Furthermore, in 11 of the reported cases (50%) in the literature, acute stroke was the presenting symptom of NS. The clinical presentation was not specific, with most patients presenting with symptoms commonly associated with strokes, including hemiparesis, aphasia, and convulsions.
The etiology of the stroke was investigated in most patients in the current review. Using the TOAST system to classify the subtypes of ischemic stroke, 11 (50%) patients, including our case, were classified to have large-artery atherosclerosis (9 in the internal carotid artery, 1 in the middle cerebral artery, and 1 in the basilar artery).6 None of them had significant atherosclerotic changes in their vessels apart from the thrombus. As in the current case, it is impossible to determine whether the ischemia was a result of an embolus of the large artery thrombus in the cerebral circulation or occlusion of the large vessel with subsequent recanalization. A source of cardioembolism was identified in 2 patients (9%) and suspected in 3 additional patients (14%) on the basis of stroke in more than 1 vascular territory or evidence of systemic embolism. Only 1 patient (5%) had small artery occlusion. A total of 4 patients (18%) were classified as having a stroke of undetermined etiology because of an incomplete investigation, and 1 (5%) patient had a transient ischemic attack and could not be classified per the TOAST system. Hence, 23% of the patients in this review had an ischemic stroke of at least possible cardioembolic etiology compared with between 15% and 44% in the general population.38, 39, 40
In patients with NS and stroke, antithrombin III levels were decreased in 9 of the 17 patients (53%) for whom these data were available. Protein C and S levels were, respectively, decreased in 3 of 12 (25%) and in 5 of 13 (54%). Elevated fibrinogen levels are frequently seen in patients with NS, but do not appear to be correlated to the risk of VTE.13 An elevated fibrinogen level was observed in 12 of 17 patients (71%) including case 1. Because increased fibrinogen and deficiency in AT III, protein C, or protein S are common in NS patients,41 measurement of these proteins appears to be of little or no value for predicting the risk of thrombosis and stroke.
To what extent the use of anticoagulants and antiplatelet agents can prevent acute ischemic stroke in patients in NS is unknown. However, no patient found in the literature experienced such an event while on anticoagulation therapy, which might suggest a protective effect. One case occurred while the patient was on antiplatelet drug ticlopidine. Further data are needed to determine the role of anticoagulants and antiplatelet agents in secondary and possibly even primary prevention of ischemic stroke in patients with NS.
Conclusion
ATE and acute ischemic stroke are rare consequences of NS. Stroke can occur in young patients and be the initial event of NS. The data regarding the factors that predict the risk of ATE patients with NS are sparse and mostly extrapolated from data regarding VTE. The mechanism by which NS promotes ATE and VTE could be different and seems to be influenced by multiple factors because no set of conditions was universally observed in patients with NS and thrombosis. Traditional cardiovascular risk factors, especially smoking, seem to play a role in the occurrence of stroke in NS patients. We strongly advise that these risk factors be carefully assessed and addressed in patients with NS, even in young individuals. The role of anticoagulants and antiplatelet agents in primary and secondary prophylaxis of stroke in the setting of NS has yet to be determined.
Disclosure
YD received honorarium from Bayer and Bristol-Myers Squibb/Pfizer in the past 2 years. GB received honorarium from Genzyme, Alexion, and Otsuka. All the other authors declared no competing interests.
Acknowledgments
We thank Natacha Patey for her helpful comments on renal biopsy findings.
References
- 1.Wass V., Cameron J.S. Cardiovascular disease and the nephrotic syndrome: the other side of the coin. Nephron. 1981;27:58–61. doi: 10.1159/000182025. [DOI] [PubMed] [Google Scholar]
- 2.Ordonez J.D., Hiatt R.A., Killebrew E.J., Fireman B.H. The increased risk of coronary heart disease associated with nephrotic syndrome. Kidney Int. 1993;44:638–642. doi: 10.1038/ki.1993.292. [DOI] [PubMed] [Google Scholar]
- 3.Mahmoodi B.K., ten Kate M.K., Waanders F. High absolute risks and predictors of venous and arterial thromboembolic events in patients with nephrotic syndrome: results from a large retrospective cohort study. Circulation. 2008;117:224–230. doi: 10.1161/CIRCULATIONAHA.107.716951. [DOI] [PubMed] [Google Scholar]
- 4.Grysiewicz R.A., Thomas K., Pandey D.K. Epidemiology of ischemic and hemorrhagic stroke: incidence, prevalence, mortality, and risk factors. Neurol Clin. 2008;26:871–895. doi: 10.1016/j.ncl.2008.07.003. vii. [DOI] [PubMed] [Google Scholar]
- 5.Kelly-Hayes M., Beiser A., Kase C.S. The influence of gender and age on disability following ischemic stroke: the Framingham study. J Stroke Cerebrovasc Dis. 2003;12:119–126. doi: 10.1016/S1052-3057(03)00042-9. [DOI] [PubMed] [Google Scholar]
- 6.Adams H.P., Jr., Bendixen B.H., Kappelle L.J. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993;24:35–41. doi: 10.1161/01.str.24.1.35. [DOI] [PubMed] [Google Scholar]
- 7.Kaul R., Johnson K., Scholz H., Marr G. Performance of the BioPlex 2200 Autoimmune Vasculitis kit. Autoimmun Rev. 2009;8:224–227. doi: 10.1016/j.autrev.2008.07.033. [DOI] [PubMed] [Google Scholar]
- 8.Choi H.K., Merkel P.A., Walker A.M., Niles J.L. Drug-associated antineutrophil cytoplasmic antibody-positive vasculitis: prevalence among patients with high titers of antimyeloperoxidase antibodies. Arthritis Rheum. 2000;43:405–413. doi: 10.1002/1529-0131(200002)43:2<405::AID-ANR22>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 9.Ponticelli C., Altieri P., Scolari F. A randomized study comparing methylprednisolone plus chlorambucil versus methylprednisolone plus cyclophosphamide in idiopathic membranous nephropathy. J Am Soc Nephrol. 1998;9:444–450. doi: 10.1681/ASN.V93444. [DOI] [PubMed] [Google Scholar]
- 10.Pexman J.H., Barber P.A., Hill M.D. Use of the Alberta Stroke Program Early CT Score (ASPECTS) for assessing CT scans in patients with acute stroke. AJNR Am J Neuroradiol. 2001;22:1534–1542. [PMC free article] [PubMed] [Google Scholar]
- 11.Brott T., Adams H.P., Jr., Olinger C.P. Measurements of acute cerebral infarction: a clinical examination scale. Stroke. 1989;20:864–870. doi: 10.1161/01.str.20.7.864. [DOI] [PubMed] [Google Scholar]
- 12.Singhal R., Brimble K.S. Thromboembolic complications in the nephrotic syndrome: pathophysiology and clinical management. Thromb Res. 2006;118:397–407. doi: 10.1016/j.thromres.2005.03.030. [DOI] [PubMed] [Google Scholar]
- 13.Orth S.R., Ritz E. The nephrotic syndrome. N Engl J Med. 1998;338:1202–1211. doi: 10.1056/NEJM199804233381707. [DOI] [PubMed] [Google Scholar]
- 14.Lionaki S., Derebail V.K., Hogan S.L. Venous thromboembolism in patients with membranous nephropathy. Clin J Am Soc Nephrol. 2012;7:43–51. doi: 10.2215/CJN.04250511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kendall A.G., Lohmann R.C., Dossetor J.B. Nephrotic syndrome. A hypercoagulable state. Arch Intern Med. 1971;127:1021–1027. [PubMed] [Google Scholar]
- 16.Parag K.B., Somers S.R., Seedat Y.K. Arterial thrombosis in nephrotic syndrome. Am J Kidney Dis. 1990;15:176–177. doi: 10.1016/s0272-6386(12)80516-6. [DOI] [PubMed] [Google Scholar]
- 17.Marsh E.E., 3rd, Biller J., Adams H.P., Jr., Kaplan J.M. Cerebral infarction in patients with nephrotic syndrome. Stroke. 1991;22:90–93. doi: 10.1161/01.str.22.1.90. [DOI] [PubMed] [Google Scholar]
- 18.Fuh J.L., Teng M.M., Yang W.C., Liu H.C. Cerebral infarction in young men with nephrotic syndrome. Stroke. 1992;23:295–297. doi: 10.1161/01.str.23.2.295. [DOI] [PubMed] [Google Scholar]
- 19.Horowitz D.R., Tuhrim S., Rand J.H., Danque P.O. Stroke secondary to carotid occlusion in a young man with nephrotic syndrome: case description and review of the literature. J Stroke Cerebrovasc Dis. 1992;2:26–33. doi: 10.1016/S1052-3057(10)80031-X. [DOI] [PubMed] [Google Scholar]
- 20.Fritz C., Braune H.J. Cerebral infarction and nephrotic syndrome. Stroke. 1992;23:1380–1381. doi: 10.1161/01.str.23.9.1380. [DOI] [PubMed] [Google Scholar]
- 21.Chaturvedi S. Fulminant cerebral infarctions with membranous nephropathy. Stroke. 1993;24:473–475. doi: 10.1161/01.str.24.3.473. [DOI] [PubMed] [Google Scholar]
- 22.Song K.S., Won D.I., Lee A.N. A case of nephrotic syndrome associated with protein S deficiency and cerebral thrombosis. J Korean Med Sci. 1994;9:347–350. doi: 10.3346/jkms.1994.9.4.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Maruyama M.Y., Yanagimoto S., Maruyama T. Bilateral internal carotid artery occlusions in a young patient with nephrotic syndrome. Acta Med Biol. 1995;43:117–120. [Google Scholar]
- 24.Huang T.Y., Chau K.M. Biventricular thrombi in diabetic nephrotic syndrome complicated by cerebral embolism. Int J Cardiol. 1995;50:193–196. doi: 10.1016/0167-5273(95)02366-5. [DOI] [PubMed] [Google Scholar]
- 25.Ogawa M., Tsukahara T., Saisho H. Nephrotic syndrome with acute renal failure and cerebral infarction in a patient with myasthenia gravis. Am J Nephrol. 1999;19:622–623. doi: 10.1159/000013512. [DOI] [PubMed] [Google Scholar]
- 26.Lee C.H., Chen K.S., Tsai F.C. Concurrent thrombosis of cerebral and femoral arteries in a patient with nephrotic syndrome. Am J Nephrol. 2000;20:483–486. doi: 10.1159/000046204. [DOI] [PubMed] [Google Scholar]
- 27.Yun Y.W., Chung S., You S.J. Cerebral infarction as a complication of nephrotic syndrome: a case report with a review of the literature. J Korean Med Sci. 2004;19:315–319. doi: 10.3346/jkms.2004.19.2.315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kotani K., Kawano M. A young female with marked hyperlipoprotein(a)emia associated with nephrotic syndrome and stroke. J Atheroscler Thromb. 2005;12:234. doi: 10.5551/jat.12.234. [DOI] [PubMed] [Google Scholar]
- 29.Nandish S.S., Khardori R., Elamin E.M. Transient ischemic attack and nephrotic syndrome: case report and review of literature. Am J Med Sci. 2006;332:32–35. doi: 10.1097/00000441-200607000-00006. [DOI] [PubMed] [Google Scholar]
- 30.Yeh S.M., Lee J.J., Hung C.C., Chen H.C. Acute cerebral infarction in a patient with nodular glomerulopathy–atypical features and differential diagnosis. Kaohsiung J Med Sci. 2011;27:39–44. doi: 10.1016/j.kjms.2010.04.002. [DOI] [PubMed] [Google Scholar]
- 31.Babu A., Boddana P., Robson S., Ludeman L. Cerebral infarction in patient with minimal change nephrotic syndrome. Indian J Nephrol. 2013;23:51–53. doi: 10.4103/0971-4065.107203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gigante A., Barbano B., Liberatori M. Nephrotic syndrome and stroke. Int J Immunopathol Pharmacol. 2013;26:769–772. doi: 10.1177/039463201302600322. [DOI] [PubMed] [Google Scholar]
- 33.Sasaki Y., Raita Y., Uehara G. Carotid thromboembolism associated with nephrotic syndrome treated with dabigatran. Case Rep Nephrol Urol. 2014:442–452. doi: 10.1159/000362162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mozaffarian D., Benjamin E.J., Go A.S. Heart Disease and stroke statistics-2016 update: a report from the American Heart Association. Circulation. 2016;133:e38–e360. doi: 10.1161/CIR.0000000000000350. [DOI] [PubMed] [Google Scholar]
- 35.Barbour S.J., Greenwald A., Djurdjev O. Disease-specific risk of venous thromboembolic events is increased in idiopathic glomerulonephritis. Kidney Int. 2012;81:190–195. doi: 10.1038/ki.2011.312. [DOI] [PubMed] [Google Scholar]
- 36.Ismail G., Mircescu G., Ditoiu A.V. Risk factors for predicting venous thromboembolism in patients with nephrotic syndrome: focus on haemostasis-related parameters. Int Urol Nephrol. 2014;46:787–792. doi: 10.1007/s11255-013-0574-0. [DOI] [PubMed] [Google Scholar]
- 37.Bellomo R., Wood C., Wagner I. Idiopathic membranous nephropathy in an Australian population: the incidence of thromboembolism and its impact on the natural history. Nephron. 1993;63:240–241. doi: 10.1159/000187197. [DOI] [PubMed] [Google Scholar]
- 38.Newbury J., Kleinig T., Leyden J. Stroke Epidemiology in an Australian Rural Cohort (SEARCH) Int J Stroke. 2017;12:161–168. doi: 10.1177/1747493016670174. [DOI] [PubMed] [Google Scholar]
- 39.Saber H., Thrift A.G., Kapral M.K. Incidence, recurrence, and long-term survival of ischemic stroke subtypes: a population-based study in the Middle East. Int J Stroke. 2016 Jan doi: 10.1177/1747493016684843. 1747493016684843. [DOI] [PubMed] [Google Scholar]
- 40.Gulli G., Rutten-Jacobs L.C., Kalra L. Differences in the distribution of stroke subtypes in a UK black stroke population - final results from the South London Ethnicity and Stroke Study. BMC Med. 2016;14:77. doi: 10.1186/s12916-016-0618-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Miller R.B., Harrington J.T., Ramos C.P. Long-term results of steroid therapy in adults with idiopathic nephrotic syndrome. Am J Med. 1969;46:919–929. doi: 10.1016/0002-9343(69)90094-1. [DOI] [PubMed] [Google Scholar]