Abstract
Background
Microvascular decompression (MVD) and partial sensory rhizotomy (PSR) provide longstanding pain relief in trigeminal neuralgia (TN). Given their invasiveness, complications can result from such posterior fossa procedures, but the impact of these procedures and their complications on patient-reported outcome measures (PROM), such as quality of life and distress, are not well established.
Method
Five years after surgery, patients who underwent first MVD or PSR for TN at one institution, between 1982 and 2002, were sent a self-completion assessment set containing a range of PROMs: the Short Form-12 (SF-12) questionnaire to assess quality of life, the Hospital Anxiety and Depression Scale (HADS) to assess distress, and a questionnaire containing questions about postoperative complications, their severity and impact on quality of life. These findings and demographic data were compared between MVD and PSR.
Results
One hundred and eighty-one of 245 (73.9%) patients after first MVD and 49 of 60 (81.7%) after PSR responded, and were included in analyses. The mean SF-12 scores of patients after MVD and PSR at five-year follow-up were significantly lower than English age-matched norms. Though there were no differences in SF-12 physical or mental component scores between the two procedures, patients after PSR were more likely to have case-level anxiety (RR = 3.3; 95% CI, 1.1–10.5; p = 0.03), had more postoperative complications, and of greater severity, including pain (RR = 2.52; 95% CI, 1.5–4.1; p < 0.001), numbness (RR = 5.9; 95% CI, 3.8–9.2; p < 0.001), burning sensations (RR = 3.0; 95% CI, 1.5–5.8; p = 0.001) and difficulty in eating (RR = 17.1; 95% CI, 5.6–53.1; p < 0.001), and these had a larger impact on quality of life for PSR compared to MVD.
Conclusions
The quality of life 5 years after MVD or PSR is poorer than in the general population and associated with postoperative complications such as pain, numbness, burning sensation and difficulty in eating. These complications are commoner after PSR than MVD, and this is associated with anxiety in PSR patients at five-year follow-up. However, these differences are not reflected by quality of life scores. Outcome measures need to incorporate patient experience after treatment for TN, and represent patient priorities for quality of life.
Electronic supplementary material
The online version of this article (10.1007/s00701-017-3350-6) contains supplementary material, which is available to authorized users.
Keywords: Trigeminal neuralgia, Posterior fossa surgery, PROMs
Introduction
Trigeminal neuralgia (TN) presents with severe, unilateral, electric shock-like facial pain provoked by light touch, resulting in significant social disability and associated anxiety and depression [1, 2]. Treatment with anticonvulsant drugs provides some patients with prolonged pain relief and acceptable quality of life. For other patients, the variety of surgical techniques available for TN provide the longest period of pain relief, with a low but significant morbidity and mortality [3–5]. These include invasive posterior fossa procedures, such as microvascular decompression (MVD), partial sensory rhizotomy (PSR), or more recently, internal neurolysis. If neurovascular compression (NVC) is found on magnetic resonance imaging (MRI), MVD is performed, with the aim of decompressing the trigeminal nerve in the region of the root entry zone, where pressure from major arterial vessels is most likely to occur. PSR or internal neurolysis are only carried out if no NVC is found. PSR entails the division of the lateral part of the sensory root, whereas internal neurolysis entails separating nerve fibres longitudinally, and it is postulated that the latter causes less injury to the nerve. PSR, therefore, has become less common [4].
Despite the range of surgical techniques on offer for TN, there are few randomised controlled trials of their efficacy, and existing cohort studies are usually retrospective and of poor quality [6]. These studies do not use standardised outcome measures, making results hard to interpret and compare, resulting in difficulty for patients in choosing between surgical techniques [7, 8]. The primary outcome measure used in many surgical studies for TN is pain relief with or without medication, with neglect of patient-reported outcome measures (PROMs) such as quality of life. Use of PROMS may highlight the potential benefits of posterior fossa procedures for TN over medical management [6]. PROMs to measure pain and its impact have been used in TN, but the impact of other factors on quality of life, such as the fear of return of pain after MVD, or the incidence of numbness after destructive procedures like PSR, is unexplored [9, 10]. A systematic assessment and comparison of the effect of these postoperative complications on quality of life is needed.
Aims and hypothesis
In this cross-sectional study, we aimed to establish long-term PROMs after first MVD or PSR for TN, including quality of life and distress. Since PSR results in a higher incidence of postoperative complications, while pain relief from MVD and PSR is equivalent [10], we expected to find poorer quality of life and distress in patients undergoing PSR than in patients undergoing their first MVD. We also aimed to determine the relative impact of common postoperative complications on PROMs, 5 years after MVD or PSR.
Methods
This cross-sectional study is reported in accordance with the STROBE checklist for observational studies [11]. Ethical approval was given by Frenchay Hospital Bristol LREC (Project Number 2001/60).
Participants and eligibility
Patients were drawn from one neurosurgeon’s entire posterior fossa surgery practice from 1982 to 2002, previously reported in terms of pain recurrence and satisfaction with outcome [12]. The inclusion criteria were: patients with primary idiopathic TN using the diagnostic criteria of International Classification for Headache Disorders [13], and patients who had been followed-up prospectively after MVD or PSR with an operation 6 months or longer before the survey had been carried out. The exclusion criteria were: patients with a secondary cause of TN, clinical evidence of multiple sclerosis, concurrent cranial nerve disorder such as hemifacial spasm, or significant co-morbidities that might influence quality of life or distress.
Data collection
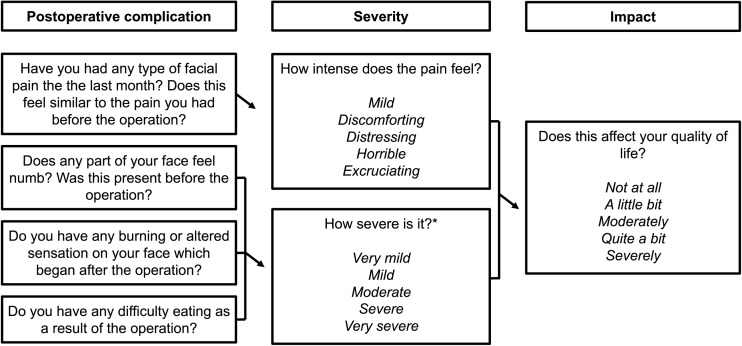
Patients were posted a self-completion assessment set relating to PROMs [14]. The Medical Outcomes Study Short Form-12 questionnaire (SF-12) and the Hospital Anxiety and Depression Scale (HADS) were included in the set. Included were questions about the most common postoperative complications, including their presence, severity and impact on quality of life reported by the patient (Fig. 1). Completed assessments were returned to independent observers, including one physician, one neurosurgeon and a patient from the Trigeminal Neuralgia Association UK. For patients who responded, electronic healthcare records were used to provide demographic data including age, gender, co-morbidities, previous surgery, mean duration of symptoms, side of pain, division(s) of the trigeminal nerve affected and current use of medication. The clinical outcomes for this population have already been reported [12]. The main outcome measures for both MVD and PSR patients were PROMs, including: questionnaire results for postoperative complications, quality of life and distress.
Fig. 1.
Self-assessment questionnaire. Questions and possible answers provided by the questionnaire as part of the self-assessment set. Severity and impact upon quality of life was asked separately for each postoperative complication. *Possible responses for severity of burning sensation or difficulty in eating differed from severity of numbness, and were: severe, quite a bit, moderate or a little bit
The Short-Form 36 (SF-36) and its shorter version, the SF-12, are broad health survey questionnaires used in large studies to assess quality of life across a range of mental and physical health conditions [15]. The SF-12 consists of 12 questions in eight domains: physical functioning, role physical, bodily pain, general health, vitality, social functioning, role emotional and mental health. A raw score for each of the eight domains is calculated, and these scores are weighted and transformed into SF-12 Physical Component Scores (PCS) and Mental Component Scores (MCS), corresponding to the physical and mental dimensions of quality of life, and scored from 0 to 100, where 0 is the lowest possible quality of life, 100 is the highest possible quality of life, and 50 represents the 1998 United States population average. SF-12 scores were calculated as standard [16]. For analyses, SF-12 scores for the MVD and PSR groups were compared with normative data from English 50– to 64-year-olds, collected in 2012 [17]. The HADS is designed for assessment of distress in patients with medical problems, and has been used to evaluate effects of surgery for TN [10, 14, 18]. It is a 14-item scale, with seven items corresponding to anxiety and seven to depression. The response to each item is scored from 0 (absence of symptom) to 3 (high level of symptom), giving scores of 0–21 for anxiety and depression. The cut of point of 8/21 is used for borderline anxiety or depression, and 11/21 for case-level anxiety or depression [19]. The HADS anxiety and HADS depression scores for each patient were then dichotomised either to borderline anxiety and depression or not, and case-level anxiety or depression or not.
Data analysis
Scores for the SF-12 PCS and MCS were compared for MVD and PSR groups using Mann-Whitney U tests. For the HADS, MVD and PSR groups were compared for the numbers of patients with borderline levels or case levels of anxiety or depression using χ2 tests. Where demographic factors were significantly different between groups, their effect on the SF-12 and HADS within each group was determined using Mann-Whitney U tests and χ2 or Fisher’s exact tests respectively. The numbers of patients with postoperative complications, and their severity and impact on quality of life, were compared using χ2 tests. Patients with surgery prior to MVD were separated from the cohort and their results reported separately.
All data were managed using Microsoft Excel 2016 (Microsoft, Redmond, VA, USA). Statistical analyses were performed using IBM SPSS Statistics for Macintosh, Version 24.0 (IBM, Armond, NY, USA). Mean values are reported with 95% confidence intervals (95% CI), and median values are reported with interquartile ranges (IQRs). For all statistics, p values less than or equal to 0.05 were considered statistically significant. Graphs were drawn using Prism for Macintosh, Version 7 (GraphPad Software, San Diego, CA, USA).
Results
Study participants
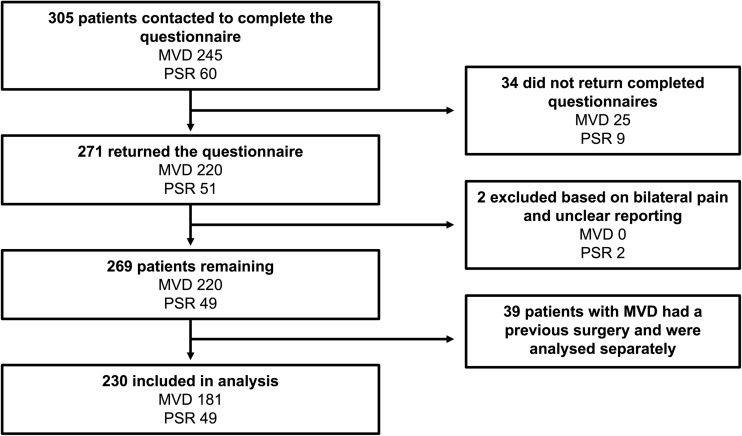
A mean of 5 years after their surgery, 305 patients, of whom 245 underwent MVD and 60 underwent PSR, were contacted and posted self-completion assessment sets (Fig. 2), of whom 271 returned them. Two patients who underwent a PSR experienced bilateral facial pain, and were excluded as the side of pain referred to in the PROMs was unknown. This left 269 (88.2%) from the original cohort. Thirty-nine of 220 (17.7%) responders with an MVD had previous Gasserian ganglion surgery; these were separated from the cohort, leaving 181 patients after MVD (59.2%) and 49 patients after PSR (81.7%) for main analyses.
Fig. 2.
Participant flow diagram
The characteristics of the 194 patients are described in Table 1. Some variables had missing data in either or both groups, indicated by the smaller denominators. There was a significant difference between the division of the trigeminal nerve affected between MVD and PSR groups (χ2 = 23.7; p = 0.001). There was also a higher proportion of medication use (χ2 = 8.52; p = 0.004) in patients who underwent a PSR compared to MVD. Patient co-morbidities were separated into those which were unlikely to have an impact upon quality of life, and those that were, after consensus between a clinician and a psychologist. Of those with co-morbidities, there was a higher proportion of patients with co-morbidities that were likely to impact quality of life after MVD (n = 38/73; 52.1%) compared to PSR (n = 4/15; 26.7%), but this was not significant (Fisher’s exact test; p = 0.092). Nineteen patients out of 49 (38.8%) had previous surgery before PSR. The characteristics of patients with a previous surgery before MVD are reported in Table 2.
Table 1.
Patient characteristics by group and differences between groups
| Patient characteristic | MVD group (n = 181) | PSR group (n = 49) | Test, p value |
|---|---|---|---|
| Mean age in years (± SD) | 59.3 (± 11.6) | 56.9 (± 12.2) | t = 1.24 n.s. |
| Gender: female (%) | 108 (57.9) | 34 (69.4) | χ2 = 1.54 n.s. |
| Other co-morbidities (%) | 78 (43.1) | 16 (32.7) | χ2 = 1.74 n.s. |
| Median duration of symptoms in years (IQR) | 5.0 (2.5–8.0) | 4.0 (2.0–9.0) |
U = 2,802.0 n.s. |
| Side of pain (%): right, left | 116/172 (67.4), 56/172 (32.6) | 27 (55.1), 22 (44.9) | χ2 = 2.54 n.s. |
| Division affected (%) | χ2 = 23.7 p = 0.001 |
||
| V1 | 11/164 (6.7) | 0/46 (0.0) | |
| V2 | 20/164 (12.2) | 10/46 (21.7) | |
| V3 | 36/164 (22.0) | 10/46 (21.7) | |
| V1 + V2 | 27/164 (16.5) | 0/46 (0.0) | |
| V2 + V3 | 13/164 (7.9) | 1/46 (2.2) | |
| V1 + V2 + V3 | 57/164 (34.8) | 25/46 (54.3) | |
| Currently taking medication for pain (%) | 14/180 (7.8) | 11/49 (22.4) | χ2 = 8.52 p = 0.004 |
| Occasional use | 2/14 (14.2) | 2/10 (20.0) | |
| Once daily use | 9/14 (64.3) | 6/10 (60.0) | |
| Use twice daily or more | 3/14 (21.4) | 2/10 (20.0) |
Table 2.
Patient characteristics of MVD patients with previous surgery
| Patient characteristic | MVD group with previous surgery (n = 39) |
|---|---|
| Mean age in years (± SD) | 59.3 (± 11.6) |
| Gender: female (%) | 23 (59.0) |
| Other co-morbidities (%) | 19 (48.9) |
| Median duration of symptoms in years (IQR) | 5.0 (2.5–8.0) |
| Side of pain (%): right, left | 22 (56.4), 17 (43.6) |
| Division affected (%) | |
| V1 | 4/37 (10.8) |
| V2 | 6/37 (16.2) |
| V3 | 11/37 (29.7) |
| V1 + V2 | 6/37 (16.2) |
| V2 + V3 | 2/37 (5.4) |
| V1 + V2 + V3 | 8/37 (21.6) |
| Currently taking medication for pain (%) | 10 (25.6) |
| Occasional use | 1/10 (10.0) |
| Once daily use | 9/10 (90.0) |
PROMs after MVD or PSR
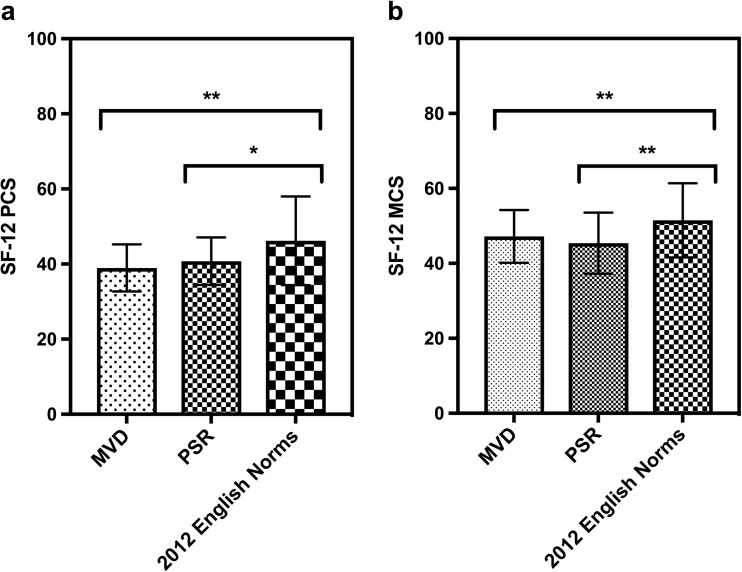
The SF-12 and HADS were used to compare patients 5 years after MVD or PSR (Supplementary Material 1). There were no significant differences between the median SF-12 physical component score or median mental component score of patients in the two groups, but mean SF-12 scores for both were significantly lower than contemporary English norms for 50– to 64-year-olds (n = 2,214): mean PCS, 46.2 (SD, 11.8) and mean MCS 51.5 (SD, 9.9) (Fig. 3). ANOVA with Tukey’s pairwise comparisons showed significantly lower PCS and MCS scores after MVD (F = 33.6; p < 0.0001 for both PCS and MCS) or PSR (F = 22.1; p < 0.0063 for PCS and p = 0.0002 for MCS). Using χ2 tests, it was found that patients after PSR were more likely to have case-level anxiety, scoring 11/21 on the HADS, than after MVD (relative risk; RR = 3.3; 95% CI, 1.1–10.5; χ2 = 4.71; p = 0.03).
Fig. 3.
Comparison of SF-12 PCS and MCS with UK norms from 2012. The mean SF-12 PCS (a) and MCS (b) scores of MVD and PSR patients and those of English norms of 50– to 64-year-olds in 2012 were compared using ANOVA and Tukey’s pairwise multiple comparisons. Error bars represent standard deviations; significance levels: * p < 0.05, ** p < 0.01
Of 39 patients with previous surgery after MVD, 31 completed the SF-12. Their median SF-12 PCS was 39.2 (IQR, 36.2–44.3) and median SF-12 MCS was 45.0 (IQR, 41.3–51.3). Thirty-eight of these patients completed the HADS, of whom 14 (36.8%) demonstrated borderline anxiety on the HADS, five (13.2%) case-level anxiety, nine (23.7%) borderline depression and three (8.9%) case-level depression. To determine the effect on PROMs of the differences in characteristics between groups, division of the trigeminal nerve affected and medication use were compared by PROMs within the MVD and PSR groups using Mann-Whitney U tests and contingency analyses. In the MVD group, patients on regular pain medication were more likely to have borderline depression (Fisher’s exact test; p = 0.043), whereas in the PSR group, medication use was also associated with case-level depression (Fisher’s exact test; p = 0.008). Also in the PSR group, the median SF-12 MCS of those on medication at follow-up (median, 39.1; IQR, 28.6–49.7) was significantly lower than that of those not taking medication (median, 49.4; IQR, 42.8–50.9; U = 81.0; p = 0.02).
PROMs by postoperative complication after MVD or PSR
The most common postoperative complications were pain, numbness, burning sensations and difficulty in eating. A comparison of the two surgical procedures in terms of postoperative complications was made (Table 3). Significantly greater proportions of patients experienced pain (RR = 2.52; 95% CI, 1.5–4.1), numbness (RR = 5.9; 95% CI, 3.8–9.2), burning sensation (RR = 3.0; 95% CI, 1.5–5.8) and difficulty in eating (RR = 17.1; 95% CI, 5.6–53.1) after PSR than after MVD. Patients directly rated the impact of each postoperative complication on their quality of life (Fig. 1). For both MVD and PSR, the severity rating for each postoperative complication correlated with the rating of impact on quality of life (p < 0.001). Data for the impact of pain on quality of life was not available. PSR patients reported greater impact on quality of life of numbness (χ2 = 81.1; p < 0.001), burning (χ2 = 26.0; p < 0.001) and difficulty in eating (χ2 = 39.5; p < 0.001) than MVD patients (Table 4). The presence of postoperative complications, their severity and impact on quality of life in patients who had surgery prior to their MVD are reported in Table 5.
Table 3.
Postoperative complications and severity after MVD or PSR
| Postoperative complication | MVD group | PSR group | Test, p value |
|---|---|---|---|
| Pain (%) | 25/157 (15.9) | 18/42 (42.9) | χ2 = 12.4, p < 0.001 |
| Excruciating | 2/25 (8.0) | 1/18 (5.6) | |
| Horrible | 5/25 (20.0) | 3/18 (16.7) | |
| Distressing | 7/25 (28.0) | 2/18 (11.1) | |
| Discomforting | 5/25 (20.0) | 10/18 (55.6) | |
| Mild | 6/25 (24.0) | 2/18 (11.1) | |
| Numbness (%) | 20/157 (14.6) | 6/48 (75.0) | χ2 = 71.7, p < 0.001 |
| Very severe | 2/20 (10.0) | 3/36 (8.3) | |
| Severe | 1/20 (5.0) | 9/36 (25.0) | |
| Moderate | 5/20 (25.0) | 10/36 (27.8) | |
| Mild | 5/20 (25.0) | 8/36 (22.2) | |
| Very mild | 7/20 (35.0) | 6/36 (16.7) | |
| Burning (%) | 14/157 (8.9) | 13/49 (26.5) | χ2 = 10.2, p = 0.0014 |
| Severe | 0/14 (0.0) | 2/13 (15.4) | |
| Quite a bit | 2/14 (14.3) | 5/13 (38.5) | |
| Moderate | 1/14 (7.1) | 2/13 (15.4) | |
| A little bit | 11/14 (78.6) | 4/13 (30.8) | |
| Difficulty in eating (%) | 3/157 (1.9) | 16/49 (32.7) | Fisher’s test, p < 0.001 |
| Severe | 0/3 (0.0) | 3/16 (18.8) | |
| Quite a bit | 0/3 (0.0) | 2/16 (12.5) | |
| Moderate | 1/3 (33.3) | 5/16 (31.3) | |
| A little bit | 2/3 (66.7) | 6/16 (37.5) |
Table 4.
Impact of postoperative complications on quality of life between MVD and PSR
| Impact of postoperative complications upon quality of life | MVD group | PSR group | Test, p value |
|---|---|---|---|
| Impact of numbness on quality of life (%) | χ2 = 81.1, p < 0.001 | ||
| Severe | 0/20 (0.0) | 2/36 (5.6) | |
| Quite a bit | 0/20 (0.0) | 5/36 (13.9) | |
| Moderately | 2/20 (10.0) | 7/36 (19.4) | |
| A little bit | 6/20 (30.0) | 11/36 (30.6) | |
| Not at all | 12/20 (60.0) | 11/36 (30.6) | |
| Impact of burning on quality of life (%) | χ2 = 26.0, p < 0.001 | ||
| Severe | 0/15 (0.0) | 3/15 (20.0) | |
| Quite a bit | 0/15 (0.0) | 2/15 (13.3) | |
| Moderately | 1/15 (6.7) | 4/15 (26.7) | |
| A little bit | 5/15 (33.3) | 4/15 (26.7) | |
| Not at all | 9/15 (60.0) | 2/15 (13.3) | |
| Impact of difficulty in eating on quality of life (%) | χ2 = 39.5, p < 0.001 | ||
| Severe | 0/7 (0.0) | 3/17 (17.6) | |
| Quite a bit | 0/7 (0.0) | 2/17 (11.8) | |
| Moderately | 0/7 (0.0) | 2/17 (11.8) | |
| A little bit | 3/7 (42.9) | 7/17 (41.2) | |
| Not at all | 4/7 (57.1) | 3/17 (17.6) |
Table 5.
Postoperative complications after MVD with prior surgery
| Postoperative complication | Severity | Impact on quality of life |
|---|---|---|
| Pain (%) | 17/39 (43.6) | |
| Excruciating 3/17 (17.6) | – | |
| Horrible 2/17 (11.8) | – | |
| Distressing 3/17 (17.6) | – | |
| Discomforting 5/17 (29.4) | – | |
| Mild 4/17 (23.5) | – | |
| Numbness (%) | 8/28 (28.6) | |
| Very severe 0/8 (0.0) | Severe 0/9 (0.0) | |
| Severe 1/8 (12.5) | Quite a bit 2/9 (22.2) | |
| Moderate 3/8 (37.5) | Moderately 0/9 (0.0) | |
| Mild 0/8 (0.0) | A little bit 3/9 (33.3) | |
| Very mild 4/8 (50.0) | Not at all 4/9 (44.4) | |
| Burning (%) | 4/29 (13.8) | |
| Severe 2/4 (50.0) | Severe 1/5 (20.0) | |
| Quite a bit 0/4 (0.0) | Quite a bit 1/5 (20.0) | |
| Moderate 0/4 (0.0) | Moderately 0/5 (0.0) | |
| A little bit 2/4 (50.0) | A little bit 1/5 (20.0) | |
| – | Not at all 2/5 (40.0) | |
| Difficulty in eating (%) | 3/29 (10.3) | |
| Severe 1/3 (33.3) | Severe 1/4 (25.0) | |
| Quite a bit 0/3 (0.0) | Quite a bit 1/4 (25.0) | |
| Moderate 1/3 (33.3) | Moderately 0/4 (0.0) | |
| A little bit 1/3 (33.3) | A little bit 0/4 (0.0) | |
| – | Not at all 2/4 (50.0) |
Discussion
This prospective study assessed all patients with TN a mean 5 years after MVD or PSR using a range of PROMs, including SF-12, the HADS and questions about postoperative complications. The results varied according to the PROM used. Patients’ responses supported our main hypothesis, that PSR, given its higher rate and severity of complications [12], is associated with poorer quality of life and that pain recurrence is not the only factor impacting on quality of life. The SF-12 showed that the physical and mental dimensions of quality of life were lower in patients 5 years after posterior fossa surgery than UK norms from 2012. The SF-12 did not significantly differ between MVD and PSR, though patients were over 3 times more likely to have case-level anxiety on the HADS after PSR than after MVD.
This is the first study to prospectively compare long-term PROMs between two posterior fossa procedures for the treatment of TN. Survey and data collection were carried out by a physician, neurosurgeon and patient, all of whom were independent of the surgical unit. Patients with co-morbidities unrelated to facial pain were also included, ensuring the cohort was as representative of the TN population as possible. However, our study is not without limitations. As PSR is less commonly performed than MVD, the number of patients in the PSR group is relatively small, and so the comparison between the two procedures should be interpreted with caution. Though the four most common postoperative complications are reported in this cohort, this does not accommodate for other complications, such as hearing loss, headaches, dizziness or visual problems, which may impact PROMs and account for the low SF-12 scores in both surgical groups [20, 21]. The HADS has come under increasing criticism for an unstable structure that affects interpretability [22]. The decision to score anxiety and depression separately and categorically was taken before these problems became clear, but made patient distress hard to interpret. Not all patients completed every item of the SF-12, which means for 22 patients after MVD and seven patients after PSR, the SF-12 PCS and MCS could not be calculated.
Other studies have also compared MVD and PSR, but comparison with these is difficult to make due to their focus on pain relief with or without presence of complications [23–29]. Some studies found that postoperative complications are more frequent after PSR than MVD, but others found that the destructive procedure yields similar pain outcomes and equivocal postoperative complications. Our results suggest that patients after PSR are more anxious and have more frequent postoperative complications, which have a greater severity and impact on quality of life than MVD. The high incidence of complications and numbness in this cohort is consistent with previous reports of PSR [30]. As demonstrated by patient responses, postoperative complications such as numbness and burning can seriously affect quality of life, and this is also reported after stereotactic radiosurgery (SRS) [31]. Though these complications are well reported after destructive procedures, their presence and frequency after MVD in our cohort was surprising. It was demonstrated that 14.6% of patients undergoing MVD may develop numbness, which is milder than after PSR, but can impact on quality of life. Previous studies have shown neurosensory disturbance after MVD, including numbness and change of taste [20, 21]. Only one study in a cohort of patients used the Japanese version of the SF-36 to assess quality of life after MVD [32]. In this study, the individual norm-based items: physical role, emotional role and social functioning were significantly lower than the Japanese population 2 years after MVD, similar to our comparison with the UK population sample. The only other two studies assessing quality of life after surgery for TN reported individual norm-based items of the SF-36 after SRS. Azar and colleagues [33] found improved SF-36 scores after SRS, with a mean follow-up of 54 months. Pan and colleagues [34] found higher scores for individual items of the SF-36 to be associated with lower pain severity, but did not report on other complications. Comparison to these studies is difficult, because of their use of individual items of the SF-36, rather than calculation of PCS and MCS.
Posterior fossa procedures aim to provide immediate pain relief for TN. MVD is considered the procedure of choice for classic TN with NVC, but in patients without NVC, with TN relapsing after MVD or in patients with a vascular anatomy that prohibits safe decompression, destructive procedures such as PSR have been considered a safe and effective alternative [30]. Although PSR has largely been discontinued in routine clinical practice, internal neurolysis is now advocated in those with no NVC but, as with PSR, the incidence of numbness after internal neurolysis is high [4]. Neurosensory disturbance of the trigeminal nerve results in poorer health-related quality of life [35], as found in our study. There have been no head-to-head comparisons of PSR against internal neurolysis, but as they are both destructive procedures, surgeons must warn patients of the increased risk of complications, such as numbness, and their impact on anxiety and wellbeing to ensure informed consent has been obtained. The difference between SF-12 scores of patients and English norms from 2012 is concerning. This may be due to postoperative complications and recurrence of pain, but may also be due to fear of the return of pain, which was not assessed in this study. In TN, there is risk of development of depressive, anxiety or sleep disorders [1], which also reduces quality of life compared to the general population, and there is no evidence to suggest that posterior fossa surgery alleviates these. Regardless of the reasons for poorer quality of life in these patients, these results suggest unmet needs after surgery, and in this respect and as previously suggested, that the current management of TN is suboptimal [36]. Future studies should seek to determine whether posterior fossa surgery reduces the incidence of anxiety, depression and fear of recurrence in TN more effectively than other modalities, such as medications and Gasserian ganglion surgery. In this cohort, though the patients’ responses revealed PSR to yield more common and severe complications that impact quality of life, this was not reflected by the SF-12. This may be because the SF-12 provides a generalised measure of the physical and mental dimensions of quality of life, and is not sensitive to the impact of these postoperative complications after surgery for TN. Therefore, the next step is to determine from patients themselves what outcomes they consider important, often divergent from those routinely assessed, and to estimate minimally important clinical differences, as has been done for SRS patients [37, 38]. A recent review identified over 116 quality of life scales that were categorised into 32 health domains [39]. Ideally a quality of life scale not only represents patients’ priorities but can also be translated into quality-adjusted life years, which in turn, can be represented in economic terms to guide treatment choices [40]. As TN is managed by a wide variety of medical and surgical interventions, arriving at a consensus for what constitutes the best quality of service would be of considerable value, as has been done for headaches [41].
Conclusions
In conclusion, PSR was associated with a higher risk of postoperative complications than MVD, which was associated with anxiety and impacted patients’ rated quality of life, although this was not detected by the SF-12. The quality of life of postoperative patients with TN is lower than that of the age-matched English population, regardless of the surgical procedure, suggesting that this disorder should be regarded as a long-term condition. Patient-centred outcomes and their assessment in TN remains an under-researched area, but one of considerable clinical and economic importance.
Electronic supplementary material
(DOCX 60 kb)
Acknowledgements
The authors would like to acknowledge Jillie Abbott, a patient from the Trigeminal Neuralgia Association support group, who entered the data and assisted with data interpretation and arbitration. Ben Lopez and Sung Kim were authors on previous publications and assisted with conception. Hugh Coakham was the neurosurgeon who operated on all the patients and Elizabeth Varian was the nurse who managed the database and contacted patients on an annual basis. The authors are also grateful to UCL AcaMedics for linking D.J. to this study. J.Z. is supported by the National Institute for Health Research University College London Hospitals Biomedical Research Centre.
Abbreviations
- HADS
Hospital Anxiety and Depression Scale
- MCS
mental component score
- NVC
neurovascular compression
- PCS
physical component score
- PROM
patient-reported outcome measure
- PSR
partial sensory rhizotomy
- RR
relative risk
- TN
trigeminal neuralgia
- SRS
stereotactic radiosurgery
Funding
No funding was received for this research.
Compliance with ethical standards
Conflict of interest
The authors have no known conflicts of interest to declare. All authors contributed to the design of the study. J.Z. designed and distributed the assessment set and organised its data entry. A.W. converted all the data into SPSS, and assisted with interpretation of psychometric data. D.J. analysed the data and wrote the first draft. All authors contributed equally to the subsequent revision and acceptance of the final manuscript, and are accountable for all aspects of the study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional committee and with the latest amendment of the Helsinki declaration.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Footnotes
Electronic supplementary material
The online version of this article (10.1007/s00701-017-3350-6) contains supplementary material, which is available to authorized users.
Comments
Patient reported outcome measures (PROM) provide a more holistic assessment of the impact of trigeminal neuralgia therapy than merely relying on the degree of pain relief. This is especially germane when we have a number of therapies that can be offered to patients. The present article reports on reduced quality of life measures undergoing otherwise successful MVD surgery for trigeminal neuralgia and following open partial sensory rhizotomy. A comparison with PROMs for patients with medical treatment only, percutaneous rhizolysis, and radiosurgical therapy could provide us with a more nuanced and rational basis for offering the appropriate therapeutic modality. This may well depend on patient variables such as age and comorbidities. Furthermore, the duration and characteristics of the symptomatology may well impact the patient outcome measures for the different treatment modalities.This article provides the incentive for similar comparative studies to our other treatment modalities in trigeminal neuralgia.
Zvi Harry Rappaport.
Petah Tiqva, Israel.
References
- 1.TH W, LY H, Lu T, Chen PM, Chen HJ, Shen CC, Wen CH. Risk of psychiatric disorders following trigeminal neuralgia: a nationwide population-based retrospective cohort study. J Headache Pain. 2015;16:64. doi: 10.1186/s10194-015-0548-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zakrzewska JM, Wu J, Williams MM, Phillips N, Pavitt SH. Evaluating the impact of trigeminal neuralgia. Pain. 2017;158:116–1174. doi: 10.1097/j.pain.0000000000000853. [DOI] [PubMed] [Google Scholar]
- 3.Gronseth G, Cruccu G, Alksne J, Argoff C, Brainin M, Burchiel K, Nurmikko T, Zakrzewska JM. Practice parameter: the diagnostic evaluation and treatment of trigeminal neuralgia (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology and the European Federation of Neurological Societies. Neurology. 2008;71:1183–1190. doi: 10.1212/01.wnl.0000326598.83183.04. [DOI] [PubMed] [Google Scholar]
- 4.Ko AL, Ozpinar A, Lee A, Raslan AM, McCartney S, Burchiel KJ. Long-term efficacy and safety of internal neurolysis for trigeminal neuralgia without neurovascular compression. J Neurosurg. 2015;122:1048–1057. doi: 10.3171/2014.12.JNS14469. [DOI] [PubMed] [Google Scholar]
- 5.Zakrzewska JM, Coakham HB. Microvascular decompression for trigeminal neuralgia: update. Curr Opin Neurol. 2012;25:296–301. doi: 10.1097/WCO.0b013e328352c465. [DOI] [PubMed] [Google Scholar]
- 6.Zakrzewska JM, Akram H. Neurosurgical interventions for the treatment of classical trigeminal neuralgia. Cochrane Database Syst Rev. 2011;7:CD007312. doi: 10.1002/14651858.CD007312.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Akram H, Mirza B, Kitchen N, Zakrzewska JM. Proposal for evaluating the quality of reports of surgical interventions in the treatment of trigeminal neuralgia: the Surgical Trigeminal Neuralgia Score. Neurosurg Focus. 2008;35:E3. doi: 10.3171/2013.6.FOCUS13213. [DOI] [PubMed] [Google Scholar]
- 8.Spatz AL, Zakrzewska JM, Kay EJ. Decision analysis of medical and surgical treatments for trigeminal neuralgia: how patient evaluations of benefits and risks affect the utility of treatment decisions. Pain. 2007;131:302–310. doi: 10.1016/j.pain.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 9.Lee JY. Measurement of trigeminal neuralgia pain: Penn Facial Pain Scale. Neurosurg Clin N Am. 2016;27:327–336. doi: 10.1016/j.nec.2016.02.003. [DOI] [PubMed] [Google Scholar]
- 10.Zakrzewska JM, Jassim S, Bulman JS. A prospective, longitudinal study on patients with trigeminal neuralgia who underwent radiofrequency thermocoagulation of the Gasserian ganglion. Pain. 1999;79:51–58. doi: 10.1016/S0304-3959(98)00145-6. [DOI] [PubMed] [Google Scholar]
- 11.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. STROBE Initiative. BMJ. 2007;20:806–808. doi: 10.1136/bmj.39335.541782.AD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zakrzewska JM, Lopez BC, Kim SE, Coakham HB. Patient reports of satisfaction after microvascular decompression and partial sensory rhizotomy for trigeminal neuralgia. Neurosurgery. 2005;56:1304–1311. doi: 10.1227/01.NEU.0000159883.35957.E0. [DOI] [PubMed] [Google Scholar]
- 13.Headache Classification Committee of the International Headache Society Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia. 1998;8:1–96. [PubMed] [Google Scholar]
- 14.Zakrzewska JM, Lopez BC, Kim SE, Varian EA, Coakham HB. Patient satisfaction after surgery for trigeminal neuralgia—development of a questionnaire. Acta Neurochir. 2005;147:925–932. doi: 10.1007/s00701-005-0575-6. [DOI] [PubMed] [Google Scholar]
- 15.Ware JE, Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473–483. doi: 10.1097/00005650-199206000-00002. [DOI] [PubMed] [Google Scholar]
- 16.Ware JE Jr, Kosinski M, Turner-Bowker DM, Gandek B (2002) User’s Manual for the SF-12v2® Health Survey. QualityMetric, Lincoln
- 17.Layte R, Jenkinson C. Normative Data for the SF-12 Health Survey In the Republic of Ireland with Comparisons to England. Ir J Psychol. 2012;22:63–72. doi: 10.1080/03033910.2001.10558264. [DOI] [Google Scholar]
- 18.Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67:361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 19.Bjelland I, Dahl AA, Haug TT, Neckelmann D. The validity of the Hospital Anxiety and Depression Scale. An Updated Literature Review. J Psychosom Res. 2002;52:69–77. doi: 10.1016/S0022-3999(01)00296-3. [DOI] [PubMed] [Google Scholar]
- 20.Barker FG, Jannetta PJ, Bissonette DJ, Larkins MV, Jho HD. The Long-Term Outcome of Microvascular Decompression for Trigeminal Neuralgia. N Engl J Med. 1996;334:1077–1084. doi: 10.1056/NEJM199604253341701. [DOI] [PubMed] [Google Scholar]
- 21.Ichida MC, de Almeida AN, da Nobrega JC, II, Teixeira MJ, de Siqueira JT, de Siqueira SR. Sensory abnormalities and masticatory function after microvascular decompression or balloon compression for trigeminal neuralgia compared with carbamazepine and healthy controls. J Neurosurg. 2015;112:1315–1323. doi: 10.3171/2014.9.JNS14346. [DOI] [PubMed] [Google Scholar]
- 22.Cosco TD, Doyle F, Ward M, McGee H. Latent structure of the Hospital Anxiety And Depression Scale: a 10-year systematic review. J Psychosom Res. 2012;72:180–184. doi: 10.1016/j.jpsychores.2011.06.008. [DOI] [PubMed] [Google Scholar]
- 23.Bederson JB, Wilson CB. Evaluation of microvascular decompression and partial sensory rhizotomy in 252 cases of trigeminal neuralgia. J Neurosurg. 1989;71:359–367. doi: 10.3171/jns.1989.71.3.0359. [DOI] [PubMed] [Google Scholar]
- 24.Campbell RL, Trentacosti CD, Eschenroder TA, Harkins SW. An evaluation of sensory changes and pain relief in trigeminal neuralgia following intracranial microvascular decompression and/or trigeminal glycerol rhizotomy. J Oral Maxillofac Surg. 1990;48:1057–1062. doi: 10.1016/0278-2391(90)90289-E. [DOI] [PubMed] [Google Scholar]
- 25.Gao J, Fu Y, Guo SK, Li B, ZX X. Efficacy and prognostic value of partial sensory rhizotomy and microvascular decompression for primary trigeminal neuralgia: a comparative study. Med Sci Monit. 2017;23:2284–2291. doi: 10.12659/MSM.901510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Klun B. Microvascular decompression and partial sensory rhizotomy in the treatment of trigeminal neuralgia: personal experience with 220 patients. Neurosurgery. 1992;30:49–52. doi: 10.1227/00006123-199201000-00009. [DOI] [PubMed] [Google Scholar]
- 27.Tatli M, Satici O, Kanpolat Y, Sindou M. Various surgical modalities for trigeminal neuralgia: literature study of respective long-term outcomes. Acta Neurochir. 2008;150:243–255. doi: 10.1007/s00701-007-1488-3. [DOI] [PubMed] [Google Scholar]
- 28.Wang DD, Raygor KP, Cage TA, Ward MM, Westcott S, Barbaro NM, Chang EF. Prospective comparison of long-term pain relief rates after first-time microvascular decompression and stereotactic radiosurgery for trigeminal neuralgia. J Neurosurg. 2017;24:1–10. doi: 10.3171/2016.9.JNS16149. [DOI] [PubMed] [Google Scholar]
- 29.Zorman G, Wilson CB. Outcome following microsurgical vascular decompression or partial sensory rhizotomy in 125 cases of trigeminal neuralgia. Neurology. 1984;34:1362–1365. doi: 10.1212/WNL.34.10.1362. [DOI] [PubMed] [Google Scholar]
- 30.Young JN, Wilkins RH. Partial sensory rhizotomy at the pons for trigeminal neuralgia. J Neurosurg. 1993;79:680–687. doi: 10.3171/jns.1993.79.5.0680. [DOI] [PubMed] [Google Scholar]
- 31.Little AS, Shetter AG, Shetter ME, Bay C, Rogers CL. Long-term pain response and quality of life in patients with typical trigeminal neuralgia treated with gamma knife stereotactic radiosurgery. Neurosurgery. 2008;63:915–923. doi: 10.1227/01.NEU.0000327689.05823.28. [DOI] [PubMed] [Google Scholar]
- 32.Shibahashi K, Morita A, Kimura T. Surgical results of microvascular decompression procedures and patient’s postoperative quality of life: review of 139 cases. Neurol Med Chir (Tokyo) 2013;53:360–364. doi: 10.2176/nmc.53.360. [DOI] [PubMed] [Google Scholar]
- 33.Azar M, Yahyavi ST, Bitaraf MA, Gazik FK, Allahverdi M, Shahbazi S, Alikhani M. Gamma knife radiosurgery in patients with trigeminal neuralgia: quality of life, outcomes, and complications. Clin Neurol Neurosurg. 2009;111:174–178. doi: 10.1016/j.clineuro.2008.09.020. [DOI] [PubMed] [Google Scholar]
- 34.Pan HC, Sheehan J, Huang CF, Sheu ML, Yang DY, Chiu WT. Quality-of-life outcomes after Gamma Knife surgery for trigeminal neuralgia. J Neurosurg. 2010;113(Suppl):191–198. doi: 10.3171/2010.8.GKS10879. [DOI] [PubMed] [Google Scholar]
- 35.Leung YY, McGrath C, Cheung LK. Trigeminal neurosensory deficit and patient reported outcome measures: the effect on quality of life. PLoS One. 2013;8:e77391. doi: 10.1371/journal.pone.0077391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tölle T, Dukes E, Sadosky A. Patient Burdern of Patient Burden of Trigeminal Neuralgia: Results from a Cross- Sectional Survey of Health State Impairment and Treatment Patterns in Six European Countries. Pain Pract. 2006;6:153–160. doi: 10.1111/j.1533-2500.2006.00079.x. [DOI] [PubMed] [Google Scholar]
- 37.Beale M, Cella M, Williams AC. Comparing patients’ and clinician-researchers’ outcome choice for psychological treatment of chronic pain. Pain. 2011;152:2283–2286. doi: 10.1016/j.pain.2011.06.007. [DOI] [PubMed] [Google Scholar]
- 38.Sandhu SK, Halpern CH, Vakhshori V, Mirsaeedi-Farahani K, Farrar JT, Lee JY. Brief pain inventory—facial minimum clinically important difference. J Neurosurg. 2015;122:180–190. doi: 10.3171/2014.8.JNS132547. [DOI] [PubMed] [Google Scholar]
- 39.Macefield RC, Jacobs M, Korfage IJ, Nicklin J, Whistance RN, Brookes ST, Sprangers MA, Blazeby JM. Developing core outcomes sets: methods for identifying and including patient-reported outcomes (PROs) Trials. 2014;15:49. doi: 10.1186/1745-6215-15-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sivakanthan S, Van Gompel JJ, Alikhani P, van Loveren H, Chen R, Agazzi S. Surgical management of trigeminal neuralgia: use and cost-effectiveness from an analysis of the Medicare Claims Database. Neurosurgery. 2014;75:220–226. doi: 10.1227/NEU.0000000000000430. [DOI] [PubMed] [Google Scholar]
- 41.Peters M, Jenkinson C, Perera S, Loder E, Jensen R, Katsarava Z, Gil GR, Broner S, Steiner T. Quality in the provision of headache care. 2: defining quality and its indicators. J Headache Pain. 2012;13:449–457. doi: 10.1007/s10194-012-0465-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 60 kb)