Abstract
Objectives
To survey by measuring patient’s functional status which is crucial when end-stage renal disease patients begin a dialysis program. The influence of the disease on patients can be examined by the measurement of Karnofsky Performance Status (KPS) scores, together with a quality of life survey, and clinical variables.
Methods
The details for the dataset in the study were collected from patients receiving regular hemodialysis (HD) in one hospital, which were available retrospectively for 1166 patients during the 5-year study period. KPS scores were applied for quantifying functional status. To identify risk factors for functional status, clinical factors including demographics, laboratory data, and HD vintage were selected. This study applied a classification and regression tree approach (CART) and logistic regression to determine risk factors on functional impairment among HD patients.
Results
Ten risk factors were identified by CART and regression model (age, primary kidney disease subclass, treatment years, hemoglobin, albumin, creatinine, phosphorus, intact parathyroid hormone, ferritin, and cardiothoracic ratio). The results of logistic regression with selected interaction models showed older age or higher hematocrit, blood urea nitrogen, and glucose levels could significantly increase the log-odds of obtaining low KPS scores at in-person visits.
Conclusions
In interaction results, the combination of older age with higher albumin level and higher creatinine level with longer HD treatment years could significantly decrease the log-odds of a low KPS score assessment during in-person visits. Age, hemoglobin, albumin, urea, creatinine levels, primary kidney disease subclass, and HD duration are the major determinants for functional status in HD patients.
Electronic supplementary material
The online version of this article (10.1186/s40001-017-0298-1) contains supplementary material, which is available to authorized users.
Keywords: Hemodialysis, Functional status, Karnofsky Performance Status, Classification and regression tree approach
Background
Dialysis therapy is the mainstay of treatment for end-stage renal disease (ESRD). In recent years, equal importance has been placed not only on dialysis adequacy, but also on quality of life (QoL) in order to reduce mortality in dialysis patients [1–6]. In addition, a functionally impaired state in ESRD patients at the start of dialysis therapy also contributes to unfavorable outcomes. A previous study reported that poor functional status scores were associated with mortality after a 3-year follow-up in hemodialysis (HD) patients [7]. Moreover, the study also revealed functional status scores and QoL scores were independently correlated with the risk of mortality in new dialysis patients [8]. A recent study retrospectively examined the prognostic significance of physical activity changes by conducting a 7-year follow-up on patients receiving HD. The results demonstrated that reductions in physical activity were significantly associated with poor prognoses independent of baseline physical activity [9]. Physical inactivity is a component of frailty. Frailty scores were reported as year-to-year variability in patients receiving HD. Markers of inflammation and hospitalization were independently reported as being associated with worsening frailty [10]. Thus, a survey of the patient’s functional status using a simple measure is crucial when ESRD patients begin a dialysis program.
The most common measure for functional status assessment is the Karnofsky Performance Status (KPS) scale. The measurement of KPS scores must be performed by trained observers to maintain reliability. A prior study demonstrated that KPS measurements can be a useful tool when used by trained observers [11]. KPS has been widely used for quantifying the functional status of cancer patients, and a poor KPS score predicts poor survival in cancer patients [11, 12]. Owing to the noted pitfalls in the original KPS scale, a modified version of the KPS was proposed in order to study the functional status of dialysis patients [7, 13]. It consists of 14 different levels of activity, ranging from < 30 (hospitalized, progressive disability process) to ≥ 96 (normal function). According to the modified assessment values of the KPS, a lower KPS score has been demonstrated to be associated with early mortality in HD patients [7]. In addition, KPS scores have shown a significant positive correlation with the domains of the Short Form (SF)-36 health survey in HD patients [14]. Based on the aforementioned background, a KPS measurement is frequently used to complement medical information, together with a QoL survey, and clinical variables to examine the influence of the disease on patients.
In the present study, we used the classification and regression tree approach (CART) approach to identify the risk factors related to functional status in HD patients. In addition, we compared the results yielded by the CART algorithm to those produced by a logistic regression model to obtain complementary evidence in the identification of risk factors for functional impairment in HD patients.
Methods
Data sources
For this retrospective cohort study, patients who received maintenance outpatient HD at Kaohsiung Chang Gung Memorial Hospital in Taiwan were enrolled. The follow-up period was from January 1, 2009, to December 31, 2013. Patients for whom data were incomplete and those lost to follow-up during the study period were excluded. A total of 1166 patients were eligible for inclusion in the KPS analysis. The protocol for the study was approved by the Committee on Human Research at the Kaohsiung Chang Gung Memorial Hospital (101-1595B) for data review, and was conducted in accordance with the principles of the Declaration of Helsinki.
Baseline measurements
Laboratory values for the blood analysis and dialysis parameters included the intact parathyroid hormone (iPTH), urea-Daugirdas estimation of fractional removal of urea per dialysis treatment (Kt/V) [3], and normalized protein catabolic rate (nPCR), all of which were measured at the initial rate after enrolment. The cardiothoracic ratio (CT ratio) was assessed upon completion of the first year after enrollment. Initial serum calcium (Ca) was corrected using the following equation for serum albumin level < 4.0 g/dL: measured total Ca (mg/dL) + 0.8 × [4.0 − serum albumin (g/dL)].
Modified KPS scores were recorded once yearly by trained nurses at the HD unit. KPS scores comprised 14 different levels of activity, ranging from < 30 (hospitalized, progressive fatal process) to ≥ 96 (normal function) [7]. A low KPS score was defined as KPS < 80 and a high score as KPS ≥ 80.
In-person visit measurements
To adhere to the in-person visit analysis design, laboratory values for blood analysis were measured monthly during the study period; exceptions were ferritin, which was measured every 3 months, and iPTH, Kt/V, and the nPCR, all of which were measured every 6 months. The CT ratio was assessed yearly. The KPS scores used were measured at each in-person visit between 2009 and 2013.
CART analysis
The CART is a binary recursive partitioning algorithm that represents a model-free and nonparametric method for exploring nonlinear associations with a low false-positive rate [15]. The CART algorithm has been successfully applied to a variety of biological studies, for example, survival analysis [16], gene-environment interactions [17, 18], and prognostic models of mortality [19]. The CART algorithm was implemented using Salford Predictive Modeler software (version 7.0). In the CART setting, tree splitting was performed until the terminal nodes reached a prespecified minimum size of 10 subjects. The optimal tree structure was evaluated by the one standard error (1-SE) rule and tenfold cross validation (CV). Subgroups of individuals with differential risk patterns were detected in the different orders of nodes, indicating the presence of associations of assigned variables.
Statistical analysis
The baseline characteristics were calculated using descriptive statistics (means, standard deviation [SD]), and percentages. The differences between both groups were evaluated by the χ 2 test, Fisher’s exact test, and independent two-sample t test. Clinical features of the study population were used to predict the KPS score at each in-person visit using the multivariate logistic regression model without interactions. The linked nodes from the decision tree were selected as interaction items to predict the KPS score level at each in-person visit using the logistic regression model with interactions estimation. AIC (Akaike Information Criterion) values were estimated for logistic regression models with and without interactions; the logistic model with a two-order interaction provided a better AIC value. The 95% confidence interval (95% CI) and a P value were used to determine statistical significance. All statistical analyses were performed using STATA software (version 11.1).
Results
Participant characteristics
A total of 1166 HD patients were enrolled. The mean age was 60.87 years. The mean HD duration was 7.87 years. The HD frequency approached thrice weekly and each HD time approached 4 h. Males and females were found with near-equal distribution. The majority was nondiabetic. Systemic diseases (diabetes mellitus [DM], systemic lupus erythematous, gout, liver cirrhosis, cardiac failure, etc.) (Additional file 1: Table S1) were the leading causes of kidney failure in HD patients. There were 143 deaths (12.26%) during the follow-up period (Table 1).
Table 1.
Baseline characteristics of the study population (N = 1166)
| Variables | Total | |
|---|---|---|
| N | SD | |
| Age (mean, SD) (years) | 60.87 | 12.05 |
| Treatment of year (mean, SD) | 7.87 | 5.78 |
| The number of HD a week (mean, SD) | 2.93 | 0.27 |
| Each dialysis time (mean, SD) (hours) | 3.91 | 0.24 |
| Dialyzer surface area (m2) | 2.16 | 0.25 |
| Gender (n, %) | ||
| Male | 572 | 49.06 |
| Female | 594 | 50.94 |
| DM (n, %) | ||
| No | 797 | 68.35 |
| Yes | 369 | 31.65 |
| Primary disease subclass (n, %) | ||
| Glomerulonephritis | 279 | 23.93 |
| Systemic diseases | 641 | 54.97 |
| Obstructive nephropathy | 14 | 1.20 |
| Hemolytic uremic syndrome | 2 | 0.17 |
| Polycystic kidney disease | 16 | 1.37 |
| Others | 51 | 4.37 |
| Unknown cause | 162 | 13.89 |
| Use of vitamin D (n, %) | ||
| No | 708 | 60.72 |
| Yes | 458 | 39.28 |
| Use of antihypertensive (n, %) | ||
| No | 573 | 49.14 |
| Yes | 593 | 50.86 |
| Use of iron (n, %) | ||
| No | 881 | 75.56 |
| Yes | 285 | 24.44 |
| Parathyroidectomy (n, %) | ||
| No | 982 | 84.22 |
| Yes | 184 | 15.78 |
| Death (n, %) | ||
| No | 1023 | 87.74 |
| Yes | 143 | 12.26 |
HD hemodialysis
P value for categorical variables were estimated by χ² test or Fisher's exact test
P value for continuous variables were estimated by independent two samples
Clinical features of the study population assessed by in-person visit
A total of 3509 in-person visits were analyzed: 812 had a low KPS score (< 80), 2697 a high KPS score (≥ 80). Patients with high KPS scores at in-person visits demonstrated significantly higher levels of hemoglobin (Hb), albumin, blood urea nitrogen (BUN), creatinine (Cr), phosphorus (P), sodium (Na), potassium (K), uric acid, aluminum, iPTH, Kt/V, nPCR, and higher ultrafiltration amounts at each HD session compared with patients with low KPS scores at in-person visits (Table 2). On the other hand, patients with high KPS scores at in-person visits demonstrated significantly lower leukocyte counts and lower Ca, glucose, alkaline phosphatase, ferritin, and CT ratios.
Table 2.
Clinical features of the study population in-personvisit (N = 3509)
| Variable | Karnofsky score | P | |||
|---|---|---|---|---|---|
| Low (N = 812) | High (N = 2697) | ||||
| Mean | SD | Mean | SD | ||
| Hb (g/dL) | 10.44 | 1.28 | 10.73 | 1.22 | < 0.001 |
| Hct (%) | 32.16 | 4.03 | 32.87 | 3.78 | < 0.001 |
| WBC (103/μL) | 6.28 | 1.90 | 6.11 | 1.70 | 0.014 |
| Albumin (g/dL) | 3.69 | 0.31 | 3.92 | 0.29 | < 0.001 |
| BUN (mg/dL) | 67.82 | 19.52 | 72.60 | 17.06 | < 0.001 |
| Creatinine (mg/dL) | 9.46 | 2.23 | 11.36 | 2.28 | < 0.001 |
| Ca (mg/dL) | 9.46 | 0.85 | 9.38 | 0.85 | 0.019 |
| P (mg/dL) | 4.78 | 1.45 | 5.11 | 1.43 | < 0.001 |
| Na (mEq/L) | 135.60 | 3.62 | 136.74 | 3.19 | < 0.001 |
| K (mEq/L) | 4.79 | 0.78 | 5.05 | 0.69 | < 0.001 |
| Glucose (mg/dL) | 164.26 | 62.92 | 131.15 | 52.04 | < 0.001 |
| AST (IU/L) | 19.41 | 7.21 | 19.00 | 7.05 | 0.149 |
| Alkaline P (IU/L) | 87.13 | 37.28 | 79.26 | 33.28 | < 0.001 |
| Cholesterol (mg/dL) | 172.13 | 37.97 | 174.69 | 37.29 | 0.088 |
| Triglyceride (mg/dL) | 153.68 | 91.33 | 150.11 | 91.50 | 0.329 |
| Uric acid (mg/dL) | 7.14 | 1.43 | 7.54 | 1.42 | < 0.001 |
| Ferritin (ng/mL) | 491.65 | 293.49 | 409.03 | 254.75 | < 0.001 |
| Al (μμg/dL) | 1.12 | 0.84 | 1.23 | 0.90 | 0.001 |
| iPTH (pg/mL) | 295.98 | 340.86 | 338.58 | 343.49 | 0.002 |
| Kt/V | 1.63 | 0.28 | 1.68 | 0.31 | < 0.001 |
| nPCR | 1.11 | 0.30 | 1.18 | 0.26 | < 0.001 |
| Ultrafiltration amount in each HD (L) | 1.92 | 1.21 | 2.23 | 1.29 | < 0.001 |
| Cardiothoracic ratio | 0.52 | 0.07 | 0.49 | 0.07 | < 0.001 |
P value for continuous variables were estimated by independent two samples
Karnofsky score: low < 80; high ≥ 80
Ferritin, Al, iPTH, Kt/V, nPCR, Cardiothoracic ratio expressed by medium values, others expressed by mean values
Hb hemoglobin, Hct hematocrit, Ca calcium, P phosphorus, Na sodium, K potassium, AST aspartate aminotransferase, Alkaline P alkaline phosphatase, Al aluminum, iPTH intact parathyroid hormone, nPCR normalized protein catabolic rate, HD hemodialysis
CART for predicting functional status measured by KPS scores
Figure 1 shows a “pruned” classification tree model for predicting functional status measured by KPS scores. The first variable chosen by the CART algorithm to split the data was age. Age ≤ 67.5 years was assigned to the left node, while age > 67.5 years was assigned to the right node. There were ten left nodes and five subgroups in the age ≤ 67.5 years node. In contrast, there were sixteen right nodes and eight subgroups in the age > 67.5 years node. In the age ≤ 67.5 years classification, the second variable chosen by the CART algorithm was the primary kidney disease subclass (Additional file 1: Table S1). The final variable chosen was the CT ratio. In the age > 67.5 years classification, the second variable chosen by the CART algorithm was albumin. The final variable chosen was treatment in years.
Fig. 1.
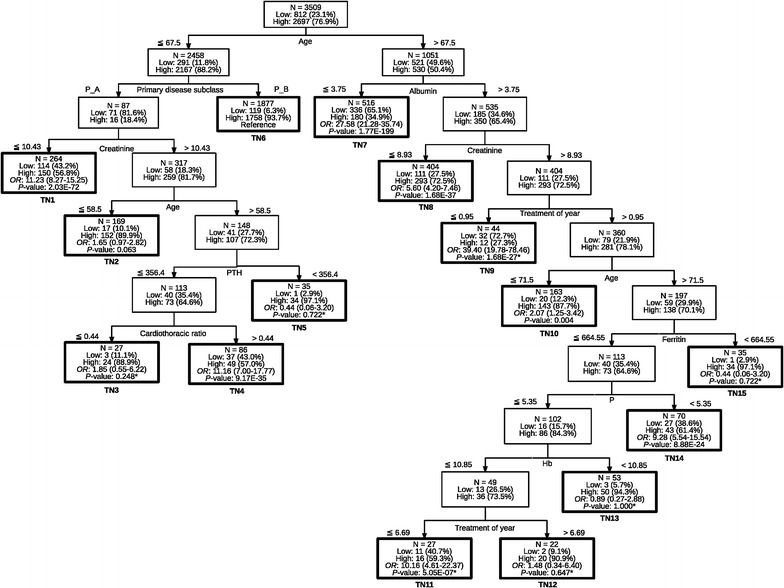
Classification tree for predicting Karnofsky Performance Status Scale in hemodialysis patients. TN: terminal node; *P value: P value for Fisher Chi Square test; P value: P value for Chi Square test; P_A, P_B items: see Additional file 1: Table S1
Logistic regression analyses
Table 3 presents the logistic regression analysis results without interactions to predict the KPS score level at each in-person visit. Older age, higher Hct, BUN, glucose, and ferritin levels significantly increased the log-odds of obtaining a low KPS score level at in-person visits. Primary kidney disease subclass in P_B (Additional file 1: Table S1), the use of vitamin D, anti-hypertensive agents, longer treatment years, a higher number of HD sessions per week, longer HD duration, larger dialyzer surface area, higher Hb, albumin, creatinine, Na, K, aspartate aminotransferase (AST) levels, and a higher Kt/V value significantly decreased the log-odds of measuring a low KPS score during an in-person visit.
Table 3.
Predicting Karnofsky score level at each in-person visit using the logistic regression model without interactions
| Variable | Estimate | SE | P value |
|---|---|---|---|
| Intercept | 13.19 | 2.79 | < 0.001 |
| Age | 0.08 | 0.01 | < 0.001 |
| Gender (female as Ref.) | 0.21 | 0.14 | 0.124 |
| DM | − 0.05 | 0.32 | 0.875 |
| Primary disease subclass (P_A as Ref.) | − 0.70 | 0.31 | 0.022 |
| Use of vitamin D | − 0.27 | 0.13 | 0.045 |
| Anti-hypertensive agent | − 0.40 | 0.11 | < 0.001 |
| Use of iron | − 0.14 | 0.17 | 0.417 |
| Parathyroidectomy | − 0.02 | 0.17 | 0.910 |
| Treatment of year | − 0.05 | 0.01 | < 0.001 |
| The number of times per week | − 0.57 | 0.18 | 0.002 |
| Each dialysis time | − 0.56 | 0.21 | 0.007 |
| Dialyzer surface area | − 0.95 | 0.22 | < 0.001 |
| Hb | − 0.93 | 0.18 | < 0.001 |
| Hct | 0.33 | 0.06 | < 0.001 |
| WBC | − 0.01 | 0.03 | 0.837 |
| Albumin | − 1.25 | 0.19 | < 0.001 |
| BUN | 0.01 | 4.27E−03 | 0.026 |
| Creatinine | − 0.21 | 0.03 | < 0.001 |
| Ca | 0.03 | 0.07 | 0.669 |
| P | 0.07 | 0.04 | 0.125 |
| Na | − 0.05 | 0.02 | 0.004 |
| K | − 0.42 | 0.08 | < 0.001 |
| Glucose | 2.48E−03 | 9.59E−04 | 0.010 |
| AST | − 0.02 | 0.01 | 0.013 |
| Alkaline P | 2.92E−03 | 1.65E−03 | 0.078 |
| Cholesterol | 1.76E−03 | 1.46E−03 | 0.226 |
| Triglyceride | 4.41E−04 | 6.54E−04 | 0.500 |
| Uric acid | − 0.05 | 0.04 | 0.281 |
| Ferritin | 4.05E−04 | 2.07E−04 | 0.050 |
| Al | − 0.05 | 0.06 | 0.437 |
| iPTH | 2.10E−04 | 1.97E−04 | 0.287 |
| Kt/V | − 0.70 | 0.22 | 0.001 |
| nPCR | 0.24 | 0.24 | 0.327 |
| Ultrafiltration dehydration amount | 0.07 | 0.04 | 0.118 |
| Cardiothoracic ratio | 1.08 | 0.80 | 0.178 |
Table 4 shows the results of predicting the KPS score level measured at each in-person visit using the logistic regression model including selected interactions. Older age, higher Hct, BUN, and glucose levels will significantly increase the log-odds of obtaining a low KPS score level. However, primary kidney disease subclass in P_B (Additional file 1: Table S1), the use of anti-hypertensive agents, a higher number of HD sessions per week, longer dialysis duration, larger dialyzer surface area, higher Hb, Na, K, AST levels, and a higher Kt/V value significantly decreased the log-odds of measuring a low KPS score level. For interaction results, the combination of older age with higher albumin level or longer treatment of years, and higher creatinine level with longer treatment years significantly decreased the log-odds of a low KPS score level, whereas for patients with the combination of primary kidney disease subclass (P_B) (Additional file 1: Table S1) and older age, and primary kidney disease subclass (P_B) and higher creatinine level, higher Hb levels and longer treatment years significantly increased the log-odds of having a low KPS score assessment.
Table 4.
Predicting Karnofsky score level at each in-person visit using the logistic regression model including selected interaction
| Variable | Estimate | SE | P value |
|---|---|---|---|
| Intercept | 2.54 | 6.89 | 0.712 |
| Age | 0.20 | 0.07 | 0.007 |
| Gender (female as Ref.) | 0.25 | 0.14 | 0.075 |
| DM | 0.09 | 0.32 | 0.776 |
| Primary disease subclass (P_A as Ref.) | − 6.18 | 1.12 | < 0.001 |
| Use of vitamin D | − 0.24 | 0.14 | 0.078 |
| Anti-hypertensive agent | − 0.35 | 0.11 | 0.002 |
| Use of iron | − 0.17 | 0.17 | 0.324 |
| Parathyroidectomy | 0.05 | 0.18 | 0.791 |
| Treatment of year | − 0.02 | 0.12 | 0.876 |
| The number of times per week | − 0.52 | 0.18 | 0.005 |
| Each dialysis time | − 0.54 | 0.21 | 0.010 |
| Dialyzer surface area | − 0.93 | 0.23 | < 0.001 |
| Hb | − 0.96 | 0.24 | < 0.001 |
| Hct | 0.32 | 0.06 | < 0.001 |
| WBC | − 0.01 | 0.03 | 0.691 |
| Albumin | 2.14 | 1.62 | 0.188 |
| BUN | 0.01 | 4.32E−03 | 0.039 |
| Creatinine | − 0.01 | 0.38 | 0.972 |
| Ca | 0.04 | 0.07 | 0.590 |
| P | 0.27 | 0.35 | 0.438 |
| Na | − 0.04 | 0.02 | 0.009 |
| K | − 0.42 | 0.08 | < 0.001 |
| Glucose | 2.29E−03 | 9.69E−04 | 0.018 |
| AST | − 0.02 | 0.01 | 0.040 |
| Alkaline P | 2.26E−03 | 1.70E−03 | 0.183 |
| Cholesterol | 1.07E−03 | 1.48E−03 | 0.470 |
| Triglyceride | 5.89E−04 | 6.61E−04 | 0.373 |
| Uric acid | − 0.05 | 0.04 | 0.279 |
| Ferritin | 8.62E−04 | 1.64E−03 | 0.600 |
| Al | − 0.02 | 0.06 | 0.704 |
| iPTH | − 8.04E−04 | 1.46E−03 | 0.581 |
| Kt/V | − 0.75 | 0.23 | 0.001 |
| nPCR | 0.29 | 0.24 | 0.241 |
| Ultrafiltration dehydration amount | 0.08 | 0.05 | 0.093 |
| Cardiothoracic ratio | − 0.10 | 1.07 | 0.923 |
| Age × albumin | − 0.04 | 0.02 | 0.043 |
| Primary disease subclass × age | 0.06 | 0.01 | < 0.001 |
| Primary disease subclass × creatinine | 0.15 | 0.05 | 0.004 |
| Albumin × creatinine | − 0.07 | 0.08 | 0.342 |
| Creatinine × age | 1.41E−03 | 2.85E−03 | 0.620 |
| Creatinine × treatment of year | − 0.02 | 0.01 | 0.004 |
| Age × iPTH | − 1.18E−05 | 1.63E−05 | 0.470 |
| iPTH × cardiothoracic ratio | 3.53E−03 | 2.29E−03 | 0.123 |
| Treatment of year × age | − 2.36E−03 | 1.15E−03 | 0.041 |
| Age × ferritin | − 1.23E−05 | 2.07E−05 | 0.553 |
| Ferritin × P | 7.21E−05 | 1.38E−04 | 0.602 |
| P × Hb | − 0.02 | 0.03 | 0.434 |
| Hb × treatment of year | 0.03 | 0.01 | 0.001 |
Discussion
In the present study, we used the CART algorithm and a logistic regression model to identify and predict the risk factors correlating with functional status during the maintenance of HD patients. The CART algorithm identified at-risk subgroups of functional impairment based on variable clinical parameters. The CART analysis results showed that the pattern of risk factors differed across the main node of age, and that outcomes were not related to dialysis adequacy, and were not strongly related to Hb levels. This raised an important issue regarding the relative importance of clinical variables versus variables that reflect individual factors, such as age or primary cause of renal disease. Nevertheless, the results did not reject the role of dialysis adequacy or treatment goal in clinical variables on the maintenance of functional status in HD patients. These results provide an alternative approach in the management of functional status in HD patients. The approach could include a rehabilitation program for the aged, continuous monitoring of primary renal disease, and prevention of relevant complications.
We also used logistic regression analysis as a complementary approach to identify risk factors for functional status in HD patients. Although the results illustrated that there was a slight difference between the two analyses, there was some overlap in the identification of risk factors. Variables such as age, primary renal disease, serum albumin and creatinine, treatment years stand as risk factors for functional impairment measured by KPS scores in HD patients. There is no definitive advantage or disadvantage when comparing classification tree analysis and logistic regression analyses. One approach may be better than the other in some situations. The advantages of CART analysis are (1) the flexibility to deal with numerous response types such as numerical data, categorical data, and ratings; (2) robustness of construction; (3) ease of interpretation; and (4) the ability to deal with missing values in response and explanatory variables. CART analysis complements many traditional statistical techniques, including logistic regression, loglinear models, and linear discriminant analysis [20]. Classification tree analysis captures sequential decision rules that may apply to subgroups of cohort based on variables having clinical utility or theoretical significance. In general, a regression analysis relatively weighs pervasiveness, while a classification tree analysis weighs specificity [21]. Based on our results, we propose that both methods can be complementary to explore the relevant clinical situation in dialysis patients. Nevertheless, additional clinical studies are required to further validate these methods.
When we compared the different results obtained from CART and logistic regression analyses, age was the main variable being able to predict low KPS scores in both analyses. Older age is associated with higher odds of frailty in patients receiving HD [22]. One component of frailty is low physical activity. Low KPS scores generally indicate low physical activity; however, measurements of physical activity in patients receiving HD could be biased if based on self-reported function. A comparison between self-reported function and a performance-based definition was reported in a well-designed study [23]. The researchers found that self-reported function achieved only 72.5% overall accuracy. In the present study, KPS scores were measured by trained HD nurses in the hemodialysis room, thereby preventing bias due to self-reported function from individual patients receiving HD. A previous study in a HD cohort with a cutoff age of 65 years demonstrated that physical aspect was independently associated with age and KPS score after 1 year of HD initiation [24, 25]. The authors also found elderly HD patients lost fewer QoL measures compared with younger HD patients. From these results, the authors concluded that elderly HD patients could make adaptation to HD with less difficulty and overcome most of the physical limitations induced by aging or dialysis therapy. Accordingly, our CART analysis revealed the cutoff age to be 67.5 years as the first split node. Furthermore, age demonstrated a powerful predictor of low KPS score by logistic regression analysis. Therefore, we conclude that age is the most important risk factor for functional status in HD patients. Nevertheless, we did not investigate the direct relationship between functional status and the physical or mental aspect in the study. A further well-designed cohort study is warranted to explore these relationships.
In the present study, we also examined the interaction effects on risk for the measurement of KPS scores by clinical variables. Some combinations were identified, which significantly correlated to KPS scores in HD patients. Indeed, older age with higher albumin level or longer treatment years, and higher creatinine level with longer treatment years significantly decreased the log-odds of obtaining a low KPS score in-person visit. In contrast, primary kidney disease with older age or higher creatinine level and higher Hb level with longer treatment years significantly increased the log-odds of measuring low KPS scores at an in-person visit. We recognized that higher albumin and creatinine levels indicated a better nutritional status. Nutritional status may reduce morbidity in dialysis patients [26, 27]. Consequently, good functional status should be expected in HD patients. Similarity, longer HD treatment years indicate better adaptation and clinical condition in HD patients. It also may preserve good functional status in HD patients. However, the interaction study demonstrated longer HD treatment years and higher Hb level would significantly increase the log-odds of measuring low KPS scores. A similar trend was also demonstrated in CART analysis in the older age subgroup. It seemed that the benefits of HD treatment years would be averted when combined with higher Hb levels. A clear explanation cannot be drawn from the present study. Similar results were also found in primary kidney disease with older age and higher creatinine levels by logistic regression analysis. In the CART analysis at a younger age node, primary kidney disease, creatinine, and age were the leading parameters that were able to split the data. It appeared that results obtained by both approaches showed a parallel comparison in the younger age subclass. Overall, the clinical variables by logistic regression interaction analysis, i.e., age, albumin, creatinine levels, and HD treatment years, indicated a favorable functional status in HD patients, which were similar to the selected clinical variables resulting from the CART algorithm.
Conclusions
We present two tools to investigate the clinical variables related to functional status measured by KPS in HD patients. The comparison of results yielded by the CART algorithm with those produced by a logistic regression analysis was used to obtain complementary evidence on important risk factors for functional status in HD patients. Although the weight of contribution to the functional status in each clinical variable was slightly different, the overall trend was very similar in the CART and logistic regression analyses. The results can be used as a reference for clinicians to determine therapeutic strategy for functional improvement in maintenance of HD patients.
Authors’ contributors
JBC and YDL designed the study, performed the analyses, and drafted the manuscript. WCL, BCC, and SHM participated in the design of the statistical analysis and CART analysis. CHY conceived of the study and participated in design. All the authors read and approved the final manuscript.
Acknowledgements
The authors would like to thank Mrs. Ching-Yi Yu and Miss Shu-Fen Su for their excellent secretarial assistance.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The datasets generated during and/or analyzed during the current study are not publicly available, but are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Disclaimer
The authors of this manuscript state they do not have any competing interests to disclose.
Disclosure
The content of present study was partly presented with abstract version in the 1st International Congress of Chinese Nephrologists-scientific Congress on Nephropathies, 2015, and at the annual meeting of American Society of Nephrology, 2015.
Ethics approval and consent to participate
The protocol for the study was approved by the Kaohsiung Chang Gung Memorial Hospital in Taiwan.
Funding
This study was partly supported by the National Science Council of Taiwan for Grant 106-2811-E-151-002-, 106-2221-E-151-009-MY2, and 105-2221-E-151-053-MY2.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- CART
classification and regression tree approach
- HD
hemodialysis
- KPS
Karnofsky Performance Status
- ESRD
end-stage renal disease
- QoL
quality of life
- SF
Short Form
- iPTH
intact parathyroid hormone
- nPCR
normalized protein catabolic rate
- CT ratio
cardiothoracic ratio
- Ca
calcium
- DM
diabetes mellitus
- Hb
hemoglobin
- BUN
blood urea nitrogen
- Cr
creatinine
- P
phosphorus
- AST
aspartate aminotransferase
Additional file
Additional file 1: Table S1. The primary kidney disease subclass.
Footnotes
Electronic supplementary material
The online version of this article (10.1186/s40001-017-0298-1) contains supplementary material, which is available to authorized users.
Contributor Information
Jin-Bor Chen, Email: chenjb1019@gmail.com.
Wen-Chin Lee, Email: leewenchin@gmail.com.
Ben-Chung Cheng, Email: benzmcl@yahoo.com.tw.
Sin-Hua Moi, Email: moi9009@gmail.com.
Cheng-Hong Yang, Phone: +886-3814586, Email: chyang@cc.kuas.edu.tw.
Yu-Da Lin, Phone: +886-3814586, Email: e0955767257@yahoo.com.tw.
References
- 1.Chen JB, Lam KK, Su YJ, Lee WC, Cheng BC, Kuo CC, Wu CH, Lin E, Wang YC, Chen TC, et al. Relationship between Kt/V urea-based dialysis adequacy and nutritional status and their effect on the components of the quality of life in incident peritoneal dialysis patients. BMC Nephrol. 2012;13:39. doi: 10.1186/1471-2369-13-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chow FY, Briganti EM, Kerr PG, Chadban SJ, Zimmet PZ, Atkins RC. Health-related quality of life in Australian adults with renal insufficiency: a population-based study. Am J Kidney Dis. 2003;41(3):596–604. doi: 10.1053/ajkd.2003.50121. [DOI] [PubMed] [Google Scholar]
- 3.Daugirdas JT. The post: pre-dialysis plasma urea nitrogen ratio to estimate Kt/V and NPCR: mathematical modeling. Int J Artif Organs. 1989;12(7):411–419. [PubMed] [Google Scholar]
- 4.Kalantar-Zadeh K, Kopple JD, Block G, Humphreys MH. Association among SF36 quality of life measures and nutrition, hospitalization, and mortality in hemodialysis. J Am Soc Nephrol: JASN. 2001;12(12):2797–2806. doi: 10.1681/ASN.V12122797. [DOI] [PubMed] [Google Scholar]
- 5.Merkus MP, Jager KJ, Dekker FW, De Haan RJ, Boeschoten EW, Krediet RT. Quality of life over time in dialysis: the Netherlands Cooperative Study on the adequacy of dialysis. NECOSAD Study Group. Kidney Int. 1999;56(2):720–728. doi: 10.1046/j.1523-1755.1999.00563.x. [DOI] [PubMed] [Google Scholar]
- 6.Owen WF, Jr, Lew NL, Liu Y, Lowrie EG, Lazarus JM. The urea reduction ratio and serum albumin concentration as predictors of mortality in patients undergoing hemodialysis. N Engl J Med. 1993;329(14):1001–1006. doi: 10.1056/NEJM199309303291404. [DOI] [PubMed] [Google Scholar]
- 7.Ifudu O, Paul HR, Homel P, Friedman EA. Predictive value of functional status for mortality in patients on maintenance hemodialysis. Am J Nephrol. 1998;18(2):109–116. doi: 10.1159/000013318. [DOI] [PubMed] [Google Scholar]
- 8.McClellan WM, Anson C, Birkeli K, Tuttle E. Functional status and quality of life: predictors of early mortality among patients entering treatment for end stage renal disease. J Clin Epidemiol. 1991;44(1):83–89. doi: 10.1016/0895-4356(91)90204-M. [DOI] [PubMed] [Google Scholar]
- 9.Shimoda T, Matsuzawa R, Yoneki K, Harada M, Watanabe T, Matsumoto M, Yoshida A, Takeuchi Y, Matsunaga A. Changes in physical activity and risk of all-cause mortality in patients on maintenance hemodialysis: a retrospective cohort study. BMC Nephrol. 2017;18(1):154. doi: 10.1186/s12882-017-0569-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Johansen KL, Dalrymple LS, Delgado C, Chertow GM, Segal MR, Chiang J, Grimes B, Kaysen GA. Factors associated with frailty and its trajectory among patients on hemodialysis. Clin J Am Soc Nephrol. 2017;12(7):1100–1108. doi: 10.2215/CJN.12131116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mor V, Laliberte L, Morris JN, Wiemann M. The Karnofsky Performance Status scale: an examination of its reliability and validity in a research setting. Cancer. 1984;53(9):2002–2007. doi: 10.1002/1097-0142(19840501)53:9<2002::AID-CNCR2820530933>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 12.Chamberlain MC, Johnston SK, Glantz MJ. Neoplastic meningitis-related prognostic significance of the Karnofsky Performance Status. Arch Neurol. 2009;66(1):74–78. doi: 10.1001/archneurol.2008.506. [DOI] [PubMed] [Google Scholar]
- 13.Hutchinson TA, Boyd NF, Feinstein AR, Gonda A, Hollomby D, Rowat B. Scientific problems in clinical scales, as demonstrated in the Karnofsky index of performance status. J Chronic Dis. 1979;32(9–10):661–666. doi: 10.1016/0021-9681(79)90096-1. [DOI] [PubMed] [Google Scholar]
- 14.Arogundade FA, Zayed B, Daba M, Barsoum RS. Correlation between Karnofsky Performance Status Scale and Short-Form Health Survey in patients on maintenance hemodialysis. J Natl Med Assoc. 2004;96(12):1661–1667. [PMC free article] [PubMed] [Google Scholar]
- 15.Breiman L, Friedman J, Stone CJ, Olshen RA. Classification and regression trees. Boca Raton: CRC Press; 1984. [Google Scholar]
- 16.Mitra AP, Pagliarulo V, Yang DY, Waldman FM, Datar RH, Skinner DG, Groshen S, Cote RJ. Generation of a concise gene panel for outcome prediction in urinary bladder cancer. J Clin Oncol. 2009;27(24):3929–3937. doi: 10.1200/JCO.2008.18.5744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ihsan R, Chauhan PS, Mishra AK, Yadav DS, Kaushal M, Sharma JD, Zomawia E, Verma Y, Kapur S, Saxena S. Multiple analytical approaches reveal distinct gene-environment interactions in smokers and non smokers in lung cancer. PLoS ONE. 2011;6(12):e29431. doi: 10.1371/journal.pone.0029431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhai RH, Chen F, Liu G, Su L, Kulke MH, Asomaning K, Lin XH, Heist RS, Nishioka NS, Sheu CC, et al. Interactions among genetic variants in apoptosis pathway genes, reflux symptoms, body mass index, and smoking indicate two distinct etiologic patterns of esophageal adenocarcinoma. J Clin Oncol. 2010;28(14):2445–2451. doi: 10.1200/JCO.2009.26.2790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Augustin S, Muntaner L, Altamirano JT, Gonzalez A, Saperas E, Dot J, Abu-Suboh M, Armengol JR, Malagelada JR, Esteban R, et al. Predicting early mortality after acute variceal hemorrhage based on classification and regression tree analysis. Clin Gastroenterol Hepatol. 2009;7(12):1347–1354. doi: 10.1016/j.cgh.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 20.De’ath G, Fabricius KE. Classification and regression trees: a powerful yet simple technique for ecological data analysis. Ecology. 2000;81(11):3178–3192. doi: 10.1890/0012-9658(2000)081[3178:CARTAP]2.0.CO;2. [DOI] [Google Scholar]
- 21.Piper ME, Loh WY, Smith SS, Japuntich SJ, Baker TB. Using decision tree analysis to identify risk factors for relapse to smoking. Subst Use Misuse. 2011;46(4):492–510. doi: 10.3109/10826081003682222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Johansen KL, Dalrymple LS, Delgado C, Kaysen GA, Kornak J, Grimes B, Chertow GM. Association between body composition and frailty among prevalent hemodialysis patients: a US Renal Data System special study. J Am Soc Nephrol. 2014;25(2):381–389. doi: 10.1681/ASN.2013040431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Johansen KL, Dalrymple LS, Delgado C, Kaysen GA, Kornak J, Grimes B, Chertow GM. Comparison of self-report—based and physical performance—based frailty definitions among patients receiving maintenance hemodialysis. Am J Kidney Dis. 2014;64(4):600–607. doi: 10.1053/j.ajkd.2014.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garcia-Mendoza M, Valdes C, Ortega T, Rebollo P, Ortega F. Differences in health-related quality of life between elderly and younger patients on hemodialysis. J Nephrol. 2006;19(6):808–818. [PubMed] [Google Scholar]
- 25.Balogun SA, Balogun R, Philbrick J, Abdel-Rahman E. Quality of life, perceptions, and health satisfaction of older adults with end-stage renal disease: a systematic review. J Am Geriatr Soc. 2017;65(4):777–785. doi: 10.1111/jgs.14659. [DOI] [PubMed] [Google Scholar]
- 26.Churchill DN, Taylor DW, Cook RJ, LaPlante P, Barre P, Cartier P, Fay WP, Goldstein MB, Jindal K, Mandin H, et al. Canadian hemodialysis morbidity study. Am J Kidney Dis. 1992;19(3):214–234. doi: 10.1016/S0272-6386(13)80002-9. [DOI] [PubMed] [Google Scholar]
- 27.Cooper BA, Penne EL, Bartlett LH, Pollock CA. Protein malnutrition and hypoalbuminemia as predictors of vascular events and mortality in ESRD. Am J Kidney Dis. 2004;43(1):61–66. doi: 10.1053/j.ajkd.2003.08.045. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available, but are available from the corresponding author on reasonable request.


