Abstract
The World Health Organization (WHO) African Region has approximately 100 million people with chronic hepatitis B virus (HBV) infection. This review describes the status of hepatitis B control in the Region. We present hepatitis B vaccine (HepB) coverage data and from available data in the published literature, the impact of HepB vaccination on hepatitis B surface antigen (HBsAg) prevalence, a marker of chronic infection, among children, HBsAg prevalence in pregnant women, and risk of perinatal transmission. Lastly, we describe challenges with HepB birth dose (HepB-BD) introduction reported in the Region, and propose strategies to increase coverage. In 2015, regional three dose HepB coverage was 76%, and 16(34%) of 47 countries reported ≥ 90% coverage. Overall, 11 countries introduced HepB-BD; only nine provide universal HepB-BD, and of these, five reported ≥ 80% coverage. From non-nationally representative serosurveys among children, HBsAg prevalence was lower among children born after HepB introduction compared to those born before HepB introduction. However, some studies still found HBsAg prevalence to be above 2%. From limited surveys among pregnant women, the median HBsAg prevalence varied by country, ranging from 1.9% (Madagascar) to 16.1% (Niger); hepatitis B e antigen (HBeAg) prevalence among HBsAg-positive women ranged from 3.3% (Zimbabwe) to 28.5% (Nigeria). Studies in three countries indicated that the risk of perinatal HBV transmission was associated with HBeAg expression or high HBV DNA viral load. Major challenges for timely HepB-BD administration were poor knowledge of or lack of national HepB-BD vaccination guidelines, high prevalence of home births, and unreliable vaccine supply. Overall, substantial progress has been made in the region. However, countries need to improve HepB3 coverage and some countries might need to consider introducing the HepB-BD to help achieve the regional hepatitis B control goal of < 2% HBsAg prevalence among children < 5 years old by 2020. To facilitate HepB-BD introduction and improve timely coverage, strategies are needed to reach both facility-based and home births. Strong political commitment, clear policy recommendations and staff training on HepB-BD administration are also required. Furthermore, high quality nationally representative serosurveys among children are needed to inform decision makers about progress towards the regional control goal.
Keywords: Hepatitis B control, vaccination, hepatitis B prevalence
Introduction
About 100 million persons in the World Health Organization (WHO) AfricanRegion have chronic hepatitis B virus (HBV) infection, and all countries in the Region have an intermediate (2%–7%) or high (≥ 8%) population prevalence of chronic HBV infection [1, 2]. Chronically infected individuals have a 15%–25% estimated lifetime risk of developing liver cancer or cirrhosis, dependent upon age at infection [1]. About 70%–90% of infants infected before 1 year of age will develop chronic HBV infection, compared with 20%–50% of those infected between 1-5 years of age, and with 5%–10% of those infected after 5 years of age [1]. About 90% of babies born to hepatitis B surface antigen (HBsAg) positive (a serologic marker of chronic HBV infection) and hepatitis B e antigen (HBeAg) positive (a marker of infectivity) mothers become chronically infected, compared with about 35% of babies born to HBeAg-negative chronically infected mothers [1]. In areas of intermediate or high endemicity, the majority of chronic HBV infections in the population are attributable to mother-to-child (perinatal) and early childhood transmission [1]. Childhood transmission is effectively prevented by administration of hepatitis B vaccine (HepB) in the routine childhood vaccination schedule, and perinatal transmission is effectively prevented by the timely administration of a HepB birth dose (HepB-BD). WHO recommends that all infants receive hepatitis B vaccine at birth, preferably within 24 hours, followed by two or three additional doses with a minimum interval of four weeks [1].
In November 2014, the WHO African Regional Committee endorsed a resolution for a hepatitis B control goal to reduce chronic HBV infection prevalence to < 2% in children less than 5 years of age in all Members States by 2020 [3]. In this paper, we present the status of hepatitis B control in the Region, including national policies for routine childhood hepatitis B vaccination and HepB-BD, coverage estimates for the HepB series and the HepB-BD, available HBsAg prevalence data among children pre- and post-HepB introduction, and available data on the risk of mother-to-child-transmission of HBV. We also describe common challenges associated with HepB-BD introduction, and propose strategies to facilitate increased HepB-BD coverage in the African Region.
Methods
For each country in the WHO African Region, we compiled data on hepatitis B vaccination (WHO-UNICEF coverage estimates) [4, 5], the number of annual births [6], the proportion of institutional births, the proportion of births attended by a skilled birthing assistant (SBA), and an estimate of at least one antenatal care (ANC) visit [7].
We also conducted a review of published literature from January 1995 to October 2016 using MEDLINE with the search criteria “(Country Name) AND hepatitis B”. The search time frame accounted for women of child bearing age and the exponential rise of HIV cases in the 1980s and early 1990s, which could have affected the risk of HBV transmission if individuals were HBV/HIV co-infected, before introduction of HIV antiviral treatment [8, 9]. Articles were initially selected by title, then by abstract review prior to complete article review. Any article that reported HBsAg prevalence among pregnant women and children, or transmission of HBV from mother to child was considered for inclusion. Articles which included prisoners, commercial sex workers, members of the armed forces, or blood donors were excluded as these groups might have higher or lower HBsAg prevalence compared to the general population, resulting in biased results. We also excluded studies that only tested infant cord blood (as this might have been contaminated with the mother’s blood) as well as studies that only included known HIV-positive women. The latter for two reasons; HIV and HBV share transmission pathways and many HIV positive women in the studies were provided anti-retroviral treatment (which also lowers HBV DNA levels and subsequently decreases the risk of perinatal transmission of HBV). Articles with fewer than 100 participants or that did not specify the laboratory test used to determine HBsAg status were also excluded. We identified additional articles cited in manuscripts identified in the literature search. From over 2,000 articles identified, 99 articles were selected for inclusion following full paper review. For The Gambia, where HepB had been introduced during the early 1990s, and in Cameroon, and Tanzania, where HBsAg prevalence among children pre-HepB introduction were unavailable after 1995, we identified five relevant articles for inclusion that were published from 1990 to1995. Median HBsAg and HBeAg prevalence estimates among pregnant women were calculated for each country using data from relevant studies. Minimum and maximum estimates are also presented.
We also summarized findings from HepB-BD post-introduction assessments conducted in Botswana (Feb 2016), Namibia (May 2016), Nigeria (Sept 2015), and Sao Tome and Principe (Jul 2015) (unpublished reports). For Mauritania, a joint Centers for Disease Control and Prevention (CDC)-WHO trip report (Dec 2015) was used to provide information on the current status of the HepB-BD implementation. Further information about country experiences with HepB-BD was gathered from the literature search described and from the reports of a Hepatitis B Birth Dose Training Workshop (Feb 2015) and the Regional Consultation on Viral Hepatitis Control in the WHO African Region (Nov 2016).
Current status of knowledge
Current status of Hepatitis B control in the African region
Childhood hepatitis B vaccination
All 47 countries in the WHO Africa Region have introduced HepB into the routine infant immunization schedule; 44 (94%) countries use pentavalent vaccine (diphtheria, tetanus, pertussis, Haemophilus influenza type B and hepatitis B vaccines) and 33 (70%) countries follow a three-dose schedule at 6, 10, and 14 weeks of age (Table 1). As of December 2016, nine countries, representing 28% of the regional birth cohort, have introduced auniversal HepB-BD policy (Table 2). Two countries, Sao Tome and Principe and Mauritius, only provide HepB-BD for babies born to HBsAg-positive mothers [10].
Table 1.
childhood hepatitis B vaccine 3-dose (HepB3) coverage by country in the World Health Organization African Region, 2011–2015
| Country1 | Year Introduced2 | HepB3 Coverage %2 | ||||
|---|---|---|---|---|---|---|
| 2011 | 2012 | 2013 | 2014 | 2015 | ||
| Algeria | 2001 | 95 | 95 | 95 | 95 | 95 |
| Angola | 2006 | 72 | 75 | 77 | 64 | 64 |
| Benin | 2005 | 75 | 81 | 74 | 75 | 79 |
| Botswana | 1995 | 95 | 95 | 95 | 95 | 95 |
| Burkina Faso | 2006 | 91 | 90 | 88 | 91 | 91 |
| Burundi | 2004 | 96 | 96 | 96 | 95 | 94 |
| Cabo Verde | 2002 | 90 | 94 | 93 | 95 | 93 |
| Cameroon | 2005 | 82 | 85 | 89 | 87 | 84 |
| Central African Republic | 2008 | 47 | 47 | 23 | 47 | 47 |
| Chad | 2008 | 33 | 45 | 48 | 46 | 55 |
| Comoros | 2003 | 83 | 86 | 83 | 80 | 80 |
| Congo | 2007 | 80 | 79 | 85 | 90 | 80 |
| Cote d’Ivoire | 2001 | 62 | 82 | 80 | 76 | 83 |
| DR of Congo | 2007 | 74 | 75 | 74 | 80 | 81 |
| Equatorial Guinea | 2013 | - | - | - | 20 | 16 |
| Eritrea | 2002 | 96 | 94 | 94 | 94 | 95 |
| Ethiopia | 2007 | 65 | 69 | 72 | 77 | 86 |
| Gabon | 2004 | 75 | 82 | 79 | 70 | 80 |
| Gambia | 1990 | 96 | 98 | 97 | 96 | 97 |
| Ghana | 2002 | 91 | 92 | 90 | 98 | 88 |
| Guinea | 2006 | 63 | 62 | 63 | 51 | 51 |
| Guinea-Bissau | 2008 | 80 | 80 | 80 | 80 | 80 |
| Kenya | 2002 | 96 | 94 | 93 | 92 | 89 |
| Lesotho | 2003 | 96 | 95 | 93 | 93 | 93 |
| Liberia | 2008 | 77 | 80 | 76 | 50 | 52 |
| Madagascar | 2002 | 73 | 70 | 74 | 73 | 69 |
| Malawi | 2002 | 97 | 96 | 89 | 91 | 88 |
| Mali | 2003 | 66 | 68 | 71 | 77 | 68 |
| Mauritania | 2005 | 75 | 80 | 80 | 84 | 73 |
| Mauritius | 1997 | 98 | 98 | 98 | 97 | 97 |
| Mozambique | 2001 | 76 | 76 | 78 | 79 | 80 |
| Namibia | 2009 | 82 | 84 | 89 | 88 | 92 |
| Niger | 2008 | 75 | 71 | 67 | 68 | 65 |
| Nigeria | 2004 | 46 | 42 | 45 | 49 | 56 |
| Rwanda | 2002 | 97 | 98 | 98 | 98 | 98 |
| Sao Tome and Principe | 2003 | 96 | 96 | 97 | 95 | 96 |
| Senegal | 2004 | 92 | 91 | 92 | 89 | 89 |
| Seychelles | 1995 | 99 | 99 | 99 | 99 | 98 |
| Sierra Leone | 2007 | 89 | 91 | 92 | 83 | 86 |
| South Africa | 1995 | 76 | 73 | 65 | 74 | 71 |
| South Sudan | 2014 | - | - | - | - | 31 |
| Swaziland | 1996 | 91 | 95 | 98 | 98 | 98 |
| Togo | 2008 | 85 | 84 | 84 | 87 | 88 |
| Uganda | 2002 | 82 | 78 | 78 | 78 | 78 |
| United Republic of Tanzania | 2002 | 90 | 92 | 91 | 97 | 98 |
| Zambia | 2005 | 81 | 78 | 79 | 86 | 90 |
| Zimbabwe | 2000 | 94 | 97 | 95 | 91 | 87 |
All countries provide pentavalent (DTwPHibHepB) vaccine, except for Algeria and Mauritius which provide monovalent hepatitis B vaccine and South Africa which provides monovalent and hexavalent (DTaPIPVHibHepB) vaccines. All countries follow a 3-dose schedule at 6, 10, and 14 weeks of age, except for Algeria (0, 1, 5 months), Angola (0, 2, 4, 5 months), Botswana (0, 2, 3, 4 months), Burkina Faso (8, 12, 16 weeks), Cabo Verde (0, 2, 4, 6, 18 months), Congo (8, 12, 16 weeks), Gambia (0, 2, 3, 4 months), Mauritania (0, 6, 10, 14 weeks), Namibia (0, 6, 10, 14 weeks), Nigeria (0, 6, 10, 14 weeks), Sao Tome and Principe (0, 6, 10, 14 weeks), Senegal (0, 6, 10, 14 weeks), Seychelles (3, 4, 5 months), and South Africa (6, 10, 14 weeks, 18 months for hexavalent vaccine).
Vaccine introduction year and annual coverage estimates were derived from WHO UNICEF Joint Reporting (updated July 2016) http://apps.who.int/immunization_monitoring/globalsummary/timeseries/tswucoveragehepb3.html.
Table 2.
Hepatitis B vaccine birth dose (HepB-BD) coverage, institutional births, and antenatal care visits by country in the World Health Organization African Region
| Country | Year HepB-BD introduced | HepB-BD Coverage %1 | Annual Births (1000s)2 | Institutional deliveries %3 | Births attended by SBA %3 | >1 ANC visit %3 | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2011 | 2012 | 2013 | 2014 | 2015 | ||||||
| Algeria | 2004 | 99 | 99 | 99 | 99 | 99 | 936 | 97 | 97 | 93 |
| Angola4 | 2015 | - | - | - | - | 19 | 1,128 | 46 | 47 | 80 |
| Botswana | Pre 2000 | 99 | 99 | 99 | 99 | 99 | 55 | 100 | 95 | 94 |
| Cabo Verde | 2002 | 99 | 99 | 94 | 99 | 93 | 11 | 76 | 78 | 98 |
| Gambia | 1990 | 90 | 97 | 93 | 96 | 98 | 83 | 63 | 57 | 86 |
| Mauritania | 2013 | - | - | - | - | 51 | 134 | 65 | 65 | 84 |
| Mauritius5 | n.a. | - | - | - | - | - | 14 | 99 | 100 | - |
| Namibia | 2014 | - | - | - | 1 | 87 | 72 | 87 | 88 | 97 |
| Nigeria | 2004 | 31 | 32 | 32 | 32 | 32 | 7,133 | 36 | 38 | 61 |
| Sao Tome and Principe5 | 2002 | - | - | - | - | - | 6 | 91 | 93 | 98 |
| Senegal | 2016 | - | - | - | - | - | 567 | 77 | 59 | 96 |
| No birth dose | ||||||||||
| Benin | 388 | 87 | 77 | 83 | ||||||
| Burkina Faso | 717 | 66 | 66 | 94 | ||||||
| Burundi | 488 | 60 | 60 | 99 | ||||||
| Cameroon | 847 | 61 | 65 | 83 | ||||||
| Central African Republic | 164 | 53 | 54 | 68 | ||||||
| Chad | 630 | 22 | 24 | 53 | ||||||
| Comoros | 26 | 76 | 82 | 92 | ||||||
| Congo | 167 | 92 | 94 | 93 | ||||||
| Cote d’Ivoire | 838 | 57 | 59 | 91 | ||||||
| DR of Congo | 3,217 | 80 | 80 | 88 | ||||||
| Equatorial Guinea | 29 | 67 | 68 | 91 | ||||||
| Eritrea | 175 | 34 | 34 | 89 | ||||||
| Ethiopia | 3,176 | 16 | 16 | 41 | ||||||
| Gabon | 51 | 90 | 89 | 95 | ||||||
| Ghana | 884 | 73 | 71 | 91 | ||||||
| Guinea | 460 | 40 | 45 | 85 | ||||||
| Guinea-Bissau | 68 | 44 | 45 | 92 | ||||||
| Kenya | 1,571 | 61 | 62 | 96 | ||||||
| Lesotho | 61 | 77 | 78 | 95 | ||||||
| Liberia | 156 | 56 | 61 | 96 | ||||||
| Madagascar | 831 | 38 | 44 | 82 | ||||||
| Malawi | 665 | 89 | 87 | 96 | ||||||
| Mali | 758 | 45 | 49 | 70 | ||||||
| Mozambique | 1,087 | 55 | 54 | 91 | ||||||
| Niger | 983 | 59 | 40 | 83 | ||||||
| Rwanda | 363 | 91 | 91 | 99 | ||||||
| Seychelles | 2 | - | - | - | ||||||
| Sierra Leone | 229 | 54 | 60 | 97 | ||||||
| South Africa | 1,111 | 95 | 94 | 97 | ||||||
| South Sudan | 446 | - | 19 | 62 | ||||||
| Swaziland | 38 | 88 | 88 | 99 | ||||||
| Togo | 256 | 73 | 59 | 73 | ||||||
| Uganda | 1,665 | 57 | 57 | 93 | ||||||
| United Republic of Tanzania | 2,064 | 50 | 49 | 88 | ||||||
| Zambia | 645 | 67 | 64 | 96 | ||||||
| Zimbabwe | 539 | 80 | 80 | 94 | ||||||
Coverage estimates are derived from WHO UNICEF Joint Reporting (updated July 2016) http://apps.who.int/immunization_monitoring/globalsummary/timeseries/tswucoveragehepb_bd.html.
Annual birth data is derived from the WHO Immunization Monitoring System (updated May 2016) http://apps.who.int/immunization_monitoring/globalsummary.
Data derived from UNICEF (updated February 2016) www.data.unicef.org. SBA-skilled birth attendant, ANC-Antenatal care.
2015 coverage data reported to SAGE 2016 in WHO review of hepatitis B birth dose.
Sao Tome and Principe and Mauritius do not offer the birth dose universally, but follow a selective policy where infants of mothers that test HBsAg are offered vaccine.n.a.-not available.
Regional reported coverage with 3 doses of HepB (HepB3) increased from 5% in 2000 to 76% in 2015. However, coverage has plateaued at 70%-75% since 2009 (Figure 1) [11]. This is below the 2015 global HepB3 coverage of 84%. Country-specific HepB3 coverage estimates for 2015 ranged from 16% in Equatorial Guinea to 98% in Rwanda, The Seychelles, Swaziland, and United Republic of Tanzania; 16 (34%) countries reported national HepB3 coverage of at least 90% (Table 1) [4]. Regional reported HepB-BD coverage increased from 0% in 2000 to 10% in 2015, although coverage has plateaued at 10% since 2010 [12]. This is below the 2015 global HepB-BD coverage of 39% (Figure 1). Among countries that have introduced the birth dose, HepB-BD coverage ranged from 19% in Angola to 99% in Algeria and Botswana (Table 2) [5]. Algeria, Botswana, Cabo Verde, and The Gambia, all of which had introduced the birth dose over a decade ago, reported at least 90% national HepB-BD coverage (Table 2).
Figure 1.
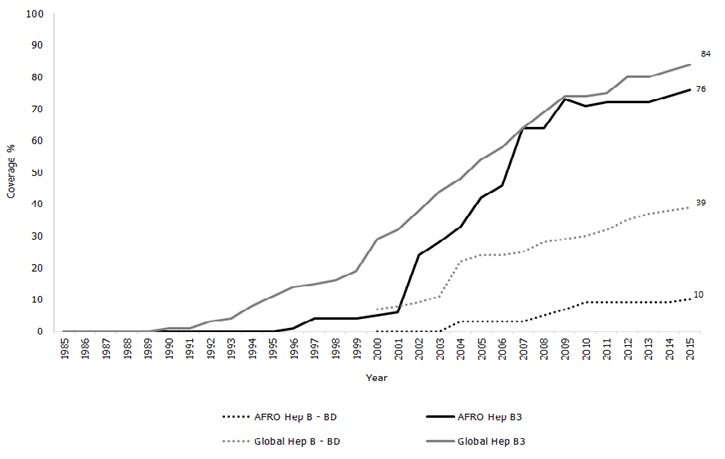
Childhood hepatitis B vaccination coverage in the World Health Organization African Region compared with global coverage, 1985-2015
A recent situational report of the WHO African Region indicated HepB-BD introduction has been recommended or is under consideration in Cameroon, Cote d’Ivoire, Guinea Bissau, Mozambique, Niger, the Republic of Congo, Sierra Leone, South Africa, and Uganda [10]. In Ethiopia and Gabon, HepB-BD introduction has been proposed for the next comprehensive multi-year plan. In Rwanda, the national Expanded Programme on Immunization (EPI) reported that it has received approval from the Ministry of Health but is waiting for endorsement from the Interagency Coordination Committee (ICC). Ghana has included HepB-BD introduction in its comprehensive multi-year strategic plan for immunization and the National Viral Hepatitis Control Plan, but so far, HepB-BD introduction has been postponed due to competing priorities [10]. Countries have reported multiple barriers to HepB-BD introduction, including lack of financial support from Gavi, the vaccine alliance (10 countries), the need for evidence on the burden of chronic HBV infection and the risk of perinatal transmission in Africa (6 countries), insufficient cold chain storage (3 countries), lack of trained healthcare workers (HCWs) to attend births or conduct post-natal visits (2 countries), and a high proportion of home births (2 countries) [10].
Impact of hepatitis B vaccination in children
To document the impact of HBV vaccination on chronic HBV infection prevalence in the African Region, we identified HBsAg serosurveys conducted pre- and post-HepB introduction among children in seven countries: the Gambia, Nigeria, Cameroon, Ghana, Senegal, South Africa, and Tanzania (Table 3) [13-36]. All studies were limited to a few areas, districts, villages, hospitals, or clinics, resulting in HBsAg prevalence estimates that were not representative of the true burden of chronic HBV infection among children in those countries. Some studies that were conducted post-HepB introduction, only included children that had three documented doses of HepB [17, 18, 21, 28, 33]. These studies reported HBsAg prevalence among fully vaccinated children, but they do not reflect the true burden among these age groups.
Table 3.
Hepatitis B surface antigen seroprevalence among children pre- and post-vaccine introduction by country --- World Health Organization African Region
| Pre-Hepatitis B vaccine introduction | Post- Hepatitis B vaccine introduction | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Country | Year of study | Sample size | Study population | Age groups | HBsAg prevalence% | Year of study | Sample size | Study population | Age groups | HBsAg prevalence% | % with HepB history1 | HepB3 coverage |
| Countries with universal birth dose at the time of the surveys | ||||||||||||
| Gambia2 | 1988 | 313 | 2 sets of villages on the north bank of the river[13] | 3-8yrs | 8 | 1998-99 | 235 | Manduar and Kenebavillages [17] | 1-<5yrs | 1.3 | 100 | 100 |
| 1988 | 959 | 7 neighboring villages on the south bank of the river[14] | 6mo-5yrs | 14.6 | 2003 | 236 | Manduar and Kenebavillages[18] | 1-4yrs | 0 | 100 | 100 | |
| 1990-91 | 816 | 4 areas, 1 from each ecological zone in the country [15] | 3-4yrs | 12.6 | 2007-08 | 1921 | 40 villages in the Central River region [19] | 1-5yrs | 0.2 | n.a. | - | |
| 1995-96 | 823 | 4 areas, 1 from each ecological zone in the country[16] | 9yrs | 12 | ||||||||
| Nigeria3 | 1997 | 200 | Daycares and vaccination centers in Calabar, Ibadan, Warri cities [20]4 | 1-3yrs | 20 | 2001 | 223 | Sabondigga-Ora town[21] | 1-4yrs | 1.3 | 1005 | 100 |
| 2001 | 219 | Sabondigga-Ora town [21] | 1-4yrs | 4.6 | 2006 | 449 | Sabondigga-Ora town [22] | 6-8yrs | 2 | 1006 | - | |
| 2006 | 373 | Sabondigga-Ora town[22] | 6-8yrs | 11.8 | 2011 | 142 | 1 hospital; Benin city [23]5 | 2mo-<10yrs | 14.1 | 277 | - | |
| 2011 | 192 | 1 hospital; Ibadan[24] | <10yrs | 0.5 | 818 | - | ||||||
| Countries without universal birth dose at the time of the surveys | ||||||||||||
| Cameroon | 1989 | 172 530 |
6 primary schools in Kumba city [25] | 4-6yrs 7-14yrs | 17.4 25.1 |
2009-10 | 763 | 2 hospitals in Yaounde[26] | 3mo-6yrs | 0.7 | 239 | - |
| Ghana | 1994 | 140 503 |
14 communities in the rural district of Ashanti- Akim North [27]10 | 1-5yrs <11yrs |
9.3 21.7 |
2009-10 | 104 | KissenaNankana district [28] | 1-10yrs | 1.9 | 100 | 100 |
| 2013 | 433 | 11 health districts in Offin River Valley [29] | ≤11yrs | 1.8 | n.a. | - | ||||||
| Senegal | 1993 | 211 | Kolda[30] | 12-24mo | 10.9 | 1993 | 229 | Kolda[30] | 12-24mo | 3.9 | 86 | 40 |
| 1999 | 2009 | Health centers, nurseries, pre-schools, orphanages in Dakar &Thies[31] |
0-<5yrs | 13.5 | 2009-10 | 485 | 2 hospitals in Dakar [26] | 3mo-6yrs | 0.2 | 43 11 | - | |
| South Africa | 1995-96 | 2288 | 7 rural and urban clinics in Mdantsane district [32] | 0-6yrs | 10.4 | 1999 | 756 | rural clinics, all provinces [33] | 18mo | 0.13 | 100 | 100 |
| 2011-12 | 209 154 | 1 hospital, Pretoria [34] | 1-5yrs 6-10yrs | 0.5 1.3 | n.a. | - | ||||||
| Tanzania | 1991-92 | 199 | General hospital in 1 urban, 1 rural area, Dodom[35] | 1-5yrs | 2.1 | 2006-08 | 110 | Vaccination clinics at 2 hospitals, Moshi [36] | 0-<5yrs | 0 | n.a. | - |
HBsAg-Hepatitis B surface antigen, %-percent, HepB-hepatitis B vaccine, HepB3-3 doses of HepB, yrs-years, mo-months, n.a.-not available.
Document verified vaccination history.
Hepatitis B vaccine was introduced to Gambia progressively during the Gambian Hepatitis Intervention Study between 1986 and 1990.
Vaccine was widely available in Nigeria from 2004, but had been available in limited areas beforehand.
Information on the time period in which the study was conducted was unavailable, year of publication is presented.
52% (n=116) of children had also received the hepatitis B vaccine birth dose (HepB-BD).
All participants received ≥2 doses of HepB; 42% (n=188) of children had received the HepB-BD and an additional 3 doses of HepB.
All of the participants with documented vaccination history had received the HepB-BD, most (>68%) after 7 days post-birth. The proportion that received additional doses of HepB was not reported.
All of the participants with documented vaccination history had received HepB, but the number of HepB doses received was not specified.
99% of participants with documented vaccination history had received HepB, but the number of doses received was not specified.
Included compounds were selected based on the presence of anHBsAg-positive child from a previous school base serosurvey, therefore HBsAg prevalence estimates are likely to be over-estimates.
100% of participants with documented vaccination history had received HepB, but the number of doses received was not specified.
Countries that have introduced the birth dose
In The Gambia, surveys conducted between 1990 and 2008, most of which were associated with the Gambia Hepatitis Intervention Study (GHIS) an initiative which progressively introduced HepB into the Gambian routine immunization program during 1986–1990, showed that HBsAg prevalence decreased from 8%-14.6% among 6 month–9 year old children to < 2% among 1–5 year old children post-HepB introduction (Table 3) [13-19]. However, two of the three post-HepB introduction studies were conducted among children that had received at least 3 doses of HepB verified by vaccination records [17, 18]. Therefore, the true HBsAg prevalence may be higher. Available data from convenience and non-representative samples in Nigeria also found a decrease in HBsAg prevalence among children after vaccine introduction (Table 3) [20-24]. However, in three of four post-HepB introduction studies that found HBsAg prevalence to be < 2%, the majority of participants had documented HepB receipt [21, 22, 24]. In the remaining study, hepatitis B vaccination history was only available for 27% of the participants, thus some of the participants without vaccination history may not have received HepB, which is likely given national HepB3 coverage was 46% in 2011 in Nigeria (Table 1), resulting in the higher HBsAg prevalence of 14.1% [23].
Countries that have NOT introduced the birth dose
Studies conducted in Cameroon, Ghana, Senegal, South Africa and Tanzania, countries that did not have a HepB-BD in their schedule at the time of the surveys, reported a drop in HBsAg prevalence among children born after HepB introduction (Table 3) [25-36]. However, none of the studies were nationally representative. More specifically, estimated HBsAg prevalence among children born post-HepB introduction was less than 2% in Cameroon, Ghana, South Africa, and Tanzania [25-29, 32-36]. These estimates were derived from studies that only included children with document verified 3 doses of HepB (Ghana, South Africa), where documented vaccination history was only available for a small proportion of participants (Cameroon), or vaccination history was not reported (Ghana, South Africa, Tanzania). In Senegal, HBsAg prevalence was > 2% in a 1993 post-HepB introduction study [30]. This study was a cluster survey conducted a year after HepB introduction in a pilot region; hepatitis B vaccination history was available for 86% of participants, of which only 40% had received 3 doses of HepB. In comparison, another study conducted in 2009 reported a HBsAg prevalence of 0.2% among hospitalized children at one hospital in Dakar; hepatitis B vaccination history was available for 43% of participants and all had received HepB [26]. Since at the time of the latter study, national HepB3 coverage would have been around 90% (Table 1) it is likely that most of the participants without vaccination history were vaccinated.
Evidence for perinatal transmission of HBV infection in Africa
HBsAg and HBeAg prevalence among pregnant women
Understanding the prevalence of HBsAg and HBeAg among pregnant women or women of child-bearing age helps to assess the risk of perinatal HBV transmission and the need for a HepB-BD. From 1995 to 2016, we identified 75 studies from 18 countries that reported HBsAg prevalence among pregnant women (Table 4) [37-111]. Of these, 24 reported HBeAg prevalence [37-39, 45-47, 61, 70-72, 85, 88-90, 96, 100-102, 104-106, 108, 109, 111]. Over one third of the studies were conducted in Nigeria. Nearly all studies were cross-sectional or cohort designs that recruited participants through convenience sampling, many times at a single study site, and were subject to selection bias. Reported median HBsAg prevalence by country varied widely, from 1.9% in Madagascar to 16.2% in Niger (Table 4). Where reported, median HBeAg prevalence among HBsAg positive pregnant women ranged from 3.3% in Zimbabwe to 28.5% in Nigeria (Table 4). For most of the countries with multiple studies, HBsAg and HBeAg prevalence estimates varied widely, reflecting the different HBV infection risks in different parts of each country and among distinct population groups, e.g. groups of differing ethnicities, socio-economic status and education levels. Although none of the studies were nationally representative, they highlight that the prevalence of chronic HBV infection among the pregnant women surveyed was intermediate to high, and that the proportion of chronically-infected mothers who were HBeAg-positive varied widely by country.
Table 4.
Hepatitis B surface antigen and e antigen prevalence among pregnant women by country within the World Health Organization African Region, 1995-2016
| Country | No. of studies | Year of studies | Study site (no. of studies) | Study settingi 1(no. of studies) | Median No. participants per study (min, max) | Median % HBsAg prevalence (min %, max %) | Median % HBeAg prevalence among HBsAg positive women (min %, max%) | ||
|---|---|---|---|---|---|---|---|---|---|
| (n=75) | Single | Multiple | Urban | Rural | |||||
| Western Africa | (n=46) | ||||||||
| Benin | 1 | 2011 | 1 | - | - | 1 | 283 | 15.5 [37] | 11.4 [37] |
| Burkina Faso | 7 | 1995-2009 | 4 | 3 | 7 | 1 | 321 (129, 917) | 8.1 (5.8, 17.1) [38-44] | 21.2 (18.2, 24.1) [38, 39] |
| Cote D'Ivoire | 3 | 1995-2002 | 1 | 2 | 3 | - | 498 (395, 4385) | 8.0 (8.0, 18.2) [45-47] | 14.5 (7.5, 15.3) [45-47] |
| Ghana | 4 | 2000-2013 | 3 | 1 | 2 | 2 | 772 (168, 1368) | 12.0 (9.5, 14.5) [48-51] | |
| Mali | 2 | 1994-2009 | 1 | 1 | 2 | - | 2244 (829, 3659) | 11.8 (8.0, 15.6)(52, 53] | - |
| Mauritania | 1 | 2008-2009 | - | 1 | 1 | - | 1020 | 10.7 [54] | - |
| Niger | 1 | 2008 | - | 1 | 1 | 1 | 495 | 16.2 [55] | - |
| Nigeria | 26 | 1997-2015 | 20: | 6 | 23 | 3 | 358 (150, 5760) | 6.9 (1.4, 16.5)[56-81] | 28.5 (6.5, 36.4) [61, 70-72] |
| Sierra Leone | 1 | 2005 | - | 1 | 1 | - | 302 | 6.3[82] | |
| Central Africa | (n=8) | ||||||||
| Cameroon | 7 | 2000-2015 | 1 | 6 | 4 | 5 | 349 (176, 7069) | 7.7 (4.4, 20.4) [83-89] | 12.1(0, 22.7) [85, 88, 89] |
| Gabon | 1 | 2005 | - | 1 | 1 | - | 1186 | 9.2 [90] | 10.1 [90] |
| Eastem and Southern Africa | (n=21) | ||||||||
| Ethiopia | 9 | 2002-2015 | 6 | 3 | 8 | 1 | 269 (165,493) | 4.4 (3.0, 7.8) [91-99] | 12.5 [96] |
| Kenya | 1 | 2001-2002 | - | 1 | 1 | 1 | 2241 | 9.3 [100] | 8.8 [100] |
| Madagascar | 1 | 2012 | 1 | - | 1 | - | 1050 | 1.9 [101] | 5 [101] |
| South Africa | 4 | 1999-2013 | 1 | 3 | 4 | 3 | 1124 (294, 1882) | 3.9 (0.4, 5.8) [102-105] | 17.1(0, 37.5) [102, 104, 105] |
| Uganda | 1 | 2012-2013 | - | 1 | 1 | - | 397 | 11.8[106] | 14.9[106] |
| Tanzania | 3 | 1995-2010 | 2 | 1 | 3 | 1 | 434 (310, 980) | 4.2 (3.9, 6.3) [107-109] | 12 (O, 24)(108, 109] |
| Zimbabwe | 2 | 1996-2005 | 1 | 1 | 2 | 1 | 701 (418, 984) | 14.2 (3.3, 25.0)[110, 111] | 3.3 [111] |
No.–number; min–minimum; max–maximum; %-percent; HBsAg–Hepatitis B surface antigen; HBeAg–Hepatitis B e antigen.
If multi-site studies included both urban and rural settings they were recorded in both columns.
Perinatal transmission of HBV in the African region
In addition to serosurveys conducted among pregnant women, we identified four studies that assessed perinatal transmission of HBV infection [38,45,49,108]. Among women of unknown or negative HIV status in Burkina Faso, Cote d’Ivoire, and Ghana, perinatal transmission of HBV was more frequent when mothers expressed HBeAg or had a high HBV DNA viral load (≥104 IU/ml) [38, 45, 49]. In Cote d’Ivoire, 9 (38%) of 24 infants born to HBsAg-positive/HBeAg-positive mothers tested HBsAg-positive at six weeks of age, compared with none (0%) of 142 infants born to HBsAg-positive/HBeAg-negative mothers [45]. In Ghana, 5 (5.2%) of 97 infants born to HBsAg-positive mothers tested HBV DNA positive at two weeks of age; the relative risk of perinatal transmission associated with high maternal HBV DNA viral load (≥104 IU/ml) compared with low maternal HBV DNA viral load was 2.4 (95% CI:1.1–5.4, p=0.048) [49]. One study from Burkina Faso reported that 7 (32%) of 22 infants born to HBsAg-positive/HBeAg-negative and 2 (29%) of 7 infants born to HBsAg-positive/HBeAg-positive mothers tested HBsAg-positive within 24 hours of birth [38].
Challenges and strategies for improving hepatitis B vaccine birth dose coverage in Africa
Despite the introduction of HepB by all countries in the Region, for 31 countries (66%) HepB3 coverage is below the 90% recommended coverage level. Given the high chronic HBV infection prevalence throughout the Region, particularly among pregnant women, and the importance of perinatal and early childhood transmission in intermediate and high endemicity settings, countries need to improve HepB3 coverage and those without a birth dose might need to consider introducing the HepB-BD to reach the regional hepatitis B control goal by 2020. In African countries that have already introduced the HepB-BD, several challenges, including timely administration of the HepB-BD, high prevalence of home births, the lack of services available to reach infants born at home and unreliable vaccine supply have limited HepB-BD implementation. In this section we present those challenges and list some strategies that could help overcome them to improve HepB-BD coverage.
Challenges associated with birth dose implementation in Africa
HepB-BD assessments conducted in the Region have consistently identified timely HepB-BD administration (within 24 hours of birth) as a challenge (BD workshop report, Regional Consultation on Viral Hepatitis Control, Mauritania CDC trip report, Botswana & Namibia BD assessments, unpublished reports). National policy recommendations for HepB-BD administration varied from within 24-hours in Nigeria to up to two weeks after birth in Namibia (Nigeria & Namibia BD assessments, unpublished reports). Restricting HepB-BD administration time to within 24 hours after birth, as recommended in Nigeria in February 2015, might limit coverage by preventing vaccination of infants born outside a health facility. In Nigeria, median timely HepB-BD administration was 1% (range: 0%–20%) and total HepB-BD coverage was 4% (range: 0%–22%) among health facilities visited prior to the birth dose assessment conducted in September 2015 (Nigeria BD assessment, unpublished report). In an assessment among children admitted to an emergency room in Nigeria, the mean age at HepB-BD receipt was 28 days, and only 13 (32%) of 41 infants received a HepB-BD within 7 days of birth [23]. In the Gambia, a review of 10 years of district HepB-BD vaccination coverage data showed that only 1% of infants were vaccinated within 1 day of birth, 5% were vaccinated within 7 days, and 58% were vaccinated within 28 days [112]. In Botswana, 50% of the facilities visited during a birth dose assessment administered the vaccine within 24 hours of birth (Botswana BD Assessment, unpublished report).
Other challenges identified included high prevalence of home births and the lack of services available to reach infants born at home, unreliable vaccine supply and inappropriate forms to document HepB vaccination (BD workshop report, Regional Consultation on Viral Hepatitis Control, Mauritania CDC trip report, Botswana & Namibia BD assessments, unpublished reports). In The Gambia and Nigeria, where the proportion of home births was high, cultural factors such as waiting until after a child’s naming day (around 7 days) to bring him/her to a healthcare facility delayed vaccination (BD workshop report, Botswana & Nigeria BD assessments, unpublished reports). Vaccine stock outs or limited vaccination sessions hindered the provision of timely HepB-BD in Botswana, The Gambia, Mauritania, Namibia, and Nigeria (BD workshop report; Botswana, Namibia, & Nigeria BD assessment, Mauritania CDC trip report, unpublished reports) [112]. In Botswana, despite daily HepB immunization sessions, HepB stock outs lasting over one month were reported at two of 16 visited facilities; and in Namibia, two events of HepB stock outs were reported in the six months before the assessment visit. Monitoring HepB-BD coverage is dependent upon having appropriate documentation tools to record both timely and total HepB-BD coverage. For all assessed countries, documentation of HepB-BD administration was suboptimal (BD workshop report; Botswana, Namibia, Nigeria, Sao Tome and Principe BD assessments; unpublished reports). In Nigeria, only doses administered within 24 hours could be recorded, while in most other countries there was no place to record timely HepB-BD.
Strategies for improving birth dose coverage
Many of the challenges identified in the HepB-BD assessments in Africa can be overcome based on the experiences in the WHO Western Pacific Region (WPR) and South East Asian Region (SEAR), where several countries had high HBsAg endemicity and high home birth rates. By implementing hepatitis B vaccination strategies, including HepB-BD administration, the WPR decreased chronic HBV infection prevalence among children at least 5 years of age from 8.3% in 1990 to 0.9% in 2012 [113]. The strategies described below could help to improve HepB-BD coverage and promote the achievement of the hepatitis B control goal in the Africa Region.
Advocate for strong political commitment
Strong political commitment is essential to identify resources in the country’s budget or to seek financial support from donors to introduce HepB-BD. In order to engage decision makers, initial steps need to be taken, including gathering the evidence on the prevalence of chronic HBV infection in pregnant women and risk of perinatal transmission, to present to the national immunization technical advisory group or equivalent technical bodies within each country to review [114]. All potential decision makers and opinion leaders from a wide variety of organizations should be engaged, including Ministries of Health and Finance, professional societies, medical associations, donor agencies, non-governmental organizations, as well as community and religious leaders [114]. Advocacy with partners in different but related sectors, such as cancer prevention, chronic disease prevention, safe motherhood and essential newborn care, might also strengthen political commitment [114]. It will be important to highlight that monovalent HepB is affordable, varying from US $0.16 per dose for a 10-dose vial to US $0.38 per dose for a single dose vial [115]. In addition, the vaccine is 95% effective in preventing infection in newborns [1].
Develop clear policy recommendations
HepB-BD guidelines and policies should be consistent with the WHO Strategic Advisory Group of Experts (SAGE) recommendations. All infants should receive their first dose of HepB as soon as possible after birth, preferably within 24 hours and up until the time of the first primary dose, since vaccination up until 7 days after birth can still be effective at preventing perinatal HBV transmission [116]. Infants born to HBsAg-positive mothers vaccinated after 7 days post-birth, compared with those vaccinated 1-3 days after birth, had an increased risk of HBV infection (OR = 8.6) [1]. HepB-BD administration is needed for all infants, because selective vaccination of infants born to HBsAg-positive mothers identified by screening as is the current policy for Sao Tome and Principe and Mauritius, has been found to miss at-risk babies [117].
In China, an effective policy to ensure timely administration of the HepB-BD was to assign responsibility for vaccine administration to whoever delivered the infant (“Who delivers the infant gives the birth dose”) [118].
All countries providing or considering HepB-BD introduction should ensure that staff at health facilities, hospitals, and public health departments, including MCH and perinatal care staff, are well-trained on the policy recommendations and reporting requirements to help address challenges that have been identified during the HepB-BD assessments.
Ensure relevant documentation for monitoring birth dose administration is available
To appropriately monitor HepB-BD coverage, countries need to record separately birth dose administered within 24 hours of birth and birth dose provided after 24 hours. Countries in the Africa Region that have introduced the birth dose and were recently assessed, had insufficient tools for documenting both timely and total birth dose. Therefore, all immunization reporting tools, including immunization cards, EPI registers, and electronic data management systems, should have a place to record the date of HepB-BD administration and to track whether it was provided within 24 hours of birth. Synergy of data collection forms across EPI and Maternal and Child Health (MCH) programs could also improve data monitoring.
Maximize HepB-BD coverage among health facility births
In just under one third of countries (14 out of 47) in the Region, ≥ 80% of births occurred in health facilities (Table 2). In these countries, high HepB-BD coverage can be achieved among children born in health facilities. Utilizing ANC visits to promote health facility deliveries could improve post-natal care and maternal health as well as facilitate administration of HepB-BD. In 81% of countries (38 out of 47) in the WHO African Region, ≥ 80% of pregnant women had at least one ANC visit and in 57% of countries (27 out of 47), ≥ 90% of pregnant women had at least one ANC visit (Table 2). Also, engaging local community and religious leaders to promote hospital births may be helpful. In China, increasing hospital deliveries resulted in improving timely HepB-BD coverage and contributed to decreasing maternal mortality, and eliminating maternal and neonatal tetanus [118].
Appropriate training for MCH and EPI staff is also essential and can improve timely HepB-BD coverage. In the Philippines, assessment and correction of HCWs through on-the-spot training increased timely HepB-BD coverage among hospital births [119]. In China, strong collaboration between MCH and EPI programs, supervision of low performing sites, and HCW training led to high timely HepB-BD coverage among hospitals [118]. Further improvements could be made through well managed vaccine delivery and procurement to avoid vaccine stock outs at health facilities. Availability of HepB-BD policies on-site and standing orders for birth dose vaccination led to higher HepB-BD coverage in the Philippines [119].
Reach children born at home within 24 hours of birth
Across Africa, one of the major challenges for timely administration of the HepB-BD is the large proportion of home births. In nine countries, institutional births accounted for ≤ 50% of births (Table 2). In settings where births are attended by an SBA, the HepB-BD can be administered by SBAs who have been trained to administer monovalent HepB and provided access to the vaccine. In the African Region, SBAs attend > 70% of births in 18 countries (Table 2).
In settings where births are not attended by SBAs, activities that could improve timely HepB-BD coverage include tracking pregnancies to increase HCW awareness of potential births in their community, educating pregnant women about the importance of timely HepB-BD administration during antenatal care visits and training community health workers (CHWs) or volunteers to organize outreach visits to vaccinate the newborn in a timely manner. Birth notification using village health volunteers was piloted in Lao PDR and resulted in more health facility deliveries and improved HepB-BD coverage [120]. Training CHWs or traditional birth attendants (TBAs) to vaccinate newborns with compact single-dose pre-filled auto-disable injection devices (CPAD) of monovalent HepB (Uniject™) has been conducted in Indonesia to provide HepB-BD during outreach activities for home births, improving timely access to the vaccine [121].
Storage of HepB outside the cold chain
A strategy for reaching newborn infants in areas that lack or have unreliable cold chain is to store monovalent Hep B outside the cold chain (OCC). Data from several HepB manufacturers indicate that the vaccine is thermostable for at least 4 weeks at temperatures of 37°C and 40°C–45°C and that HepB stored OCC induces a similar level of protection and seroconversion as vaccine stored between 2°C–8°C. Recently, WHO SAGE issued recommendations that support countries that choose to pursue an OCC policy for monovalent HepB vaccination [116, 122]. Storing monovalent HepB OCC has been shown to significantly improve HepB-BD coverage in Indonesia, China, Lao, and Vietnam [123-126]. Therefore, countries in the African Region, where the proportion of home births is high and/or cold chain storage might not be optimal, might want to consider storing monovalent HepB OCC to promote timely HepB-BD administration and subsequently prevent perinatal infections.
Conclusion
Substantial progress has been made to introduce routine infant hepatitis B vaccination in the WHO African Region; however, in 2015 the Region had the lowest HepB3 coverage estimate (76%) compared with other WHO Regions [11]. Only 11 (23%) countries have introduced the Hep B-BD; of these, five had total HepB-BD coverage ≥ 80%.Limited data from non-representative serosurveys conducted to date suggest hepatitis B vaccination has decreased HBsAg prevalence among children born after HepB introduction in some countries. However, several studies reported > 2% HBsAg prevalence among children born after vaccination introduction. Given the HBsAg prevalence data among pregnant women and children is limited and not nationally representative, the prevalence estimates presented for each country may not reflect the true situation. High quality nationally representative serosurveys among children born after implementation of routine infant hepatitis B vaccination are needed to inform local decision makers about progress towards and actions needed to achieve the Africa Region hepatitis B control goal. National estimates of HBsAg prevalence among children could be obtained by conducting nationally representative hepatitis B serosurveys or by incorporating HBsAg testing into Demographic and Health Surveys, Multiple Indicator Cluster Surveys, or HIV/AIDS surveys. Available data suggest that a substantial burden of chronic HBV infection among children might be occurring from perinatal and early childhood HBV transmission in the African Region. These findings reinforce the need to improve HepB3 coverage and that some countries might need to consider introducing a HepB-BD to achieve the regional hepatitis B control goal. Ultimately, all countries will need the HepB-BD to make further progress to eliminate mother to child HBV transmission by 2030 [127]. To facilitate HepB-BD introduction and improve timely HepB-BD coverage, strategies are needed to reach both facility-based and home births. Strong political commitment, clear policy recommendations, and training of staff on HepB-BD administration and recording are also required
What is known about this topic
There is a high prevalence of chronic hepatitis B virus (HBV) infection in the African Region, resulting in 100 million people being at risk of death from liver cancer and cirrhosis;
The Africa Region has established a hepatitis B control goal to reduce the prevalence of hepatitis B surface antigen (HBsAg, a marker of chronic HBV infection) to < 2% among children aged < 5 years by 2020;
Administering a hepatitis B vaccine birth dose within 24 hours of birth and three additional doses during infancy protects children from acquiring chronic HBV infection through perinatal transmission and during early childhood when the risk of developing chronic infection is highest.
What this study adds
All countries in the Africa Region have introduced hepatitis B vaccine in the routine childhood vaccination schedule, but only 11 have introduced the birth dose;
Childhood hepatitis B vaccination has reduced chronic disease among children; however, several studies documented > 2% HBsAg prevalence among children born after vaccine introduction, highlighting the need for countries to improve HepB3 coverage and to consider introducing HepB-BD to prevent perinatal and early childhood HBV transmission;
High quality nationally representative serosurveys among children born after the implementation of routine infant hepatitis B vaccination are needed to inform decision makers about progress towards and actions needed to achieve the African Region hepatitis B control goal.
Competing interests
The authors declare no competing interest.
Authors’ contributions
Dr Breakwell designed the study, reviewed the literature, summarized the data and wrote the manuscript; Dr Tevi Benissan reviewed the manuscript; Ms Childs contributed to the data abstraction and analysis, and reviewed the manuscript; Dr Mihigo reviewed the manuscript; Dr Tohme contributed to the study design and reviewed the manuscript. All the authors have read and agreed to the final manuscript.
References
- 1.World Health Organization Hepatitis B vaccines. Weekly Epidemiological Record. 2009;84(40):405–20. [Google Scholar]
- 2.World Health Organization . Regional Office for Africa, editor. 2016. Hepatitis Fact Sheet. [Google Scholar]
- 3.World Health Organization . Sixty-fourth session of the WHO Regional Committee for Africa. (cited 17 february 2017) [Google Scholar]
- 4.World Health Organization . WHO-UNICEF estimates of HepB3 coverage. 2016. (updated 03/03/2017; cited 2017 04/3/2017) [Google Scholar]
- 5.World Health Organization . WHO-UNICEF estimates of HepB_BD coverage. 2016. (updated 03/03/2017; cited 2017 04/03/2017) [Google Scholar]
- 6.World Health Organization . WHO Vaccine-Preventable Diseases: Monitoring System. 2016 Global Summary. 2016. (updated 12/1/2016; cited 2016 10/3/2016) [Google Scholar]
- 7.UNICEF . UNICEF Data: Monitoring the Situation of Children and Women. 2016. (updated 02/1/2016; cited 2016 10/3/2016) [Google Scholar]
- 8.UNAIDS . HIV estimates with uncertainty bounds 1990-2015. 2016. (November 14, 2016) [Google Scholar]
- 9.Kourtis AP, Bulterys M, Hu DJ, Jamieson DJ. HIV-HBV coinfection-a global challenge. N Engl J Med. 2012 May 10;366(19):1749–52. doi: 10.1056/NEJMp1201796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Health Organization . A review. Global compliance with Hepatitis B vaccine birth dose and factors related to timely schedule. (cited 2016 November 14, 2016) [Google Scholar]
- 11.World Health Organization . Hepatitis B (HepB3) immunization coverage estimates by WHO region. 2016. (accessed 12/14/2016) [Google Scholar]
- 12.WHO/UNICEF . Coverage estimates for 1980-2015: Regional Global Coverage. 2016. (cited 04/03/2017) [Google Scholar]
- 13.Chotard J, Inskip HM, Hall AJ, Loik F, Mendy M, Whittle H, et al. The Gambia Hepatitis Intervention Study: follow-up of a cohort of children vaccinated against hepatitis B. J Infect Dis. 1992 Oct;166(4):764–8. doi: 10.1093/infdis/166.4.764. [DOI] [PubMed] [Google Scholar]
- 14.Vall Mayans M, Hall AJ, Inskip HM, Chotard J, Lindsay SW, Coromina E, et al. Risk factors for transmission of hepatitis B virus to Gambian children. Lancet. 1990 Nov 3;336(8723):1107–9. doi: 10.1016/0140-6736(90)92580-b. [DOI] [PubMed] [Google Scholar]
- 15.Fortuin M, Chotard J, Jack AD, Maine NP, Mendy M, Hall AJ, et al. Efficacy of hepatitis B vaccine in the Gambian expanded programme on immunisation. The Lancet. 1993 May 1;341(8853):1129–31. doi: 10.1016/0140-6736(93)93137-p. [DOI] [PubMed] [Google Scholar]
- 16.Viviani S, Jack A, Hall AJ, Maine N, Mendy M, Montesano R, et al. Hepatitis B vaccination in infancy in The Gambia: protection against carriage at 9 years of age. Vaccine. 1999 Aug 6;17(23-24):2946–50. doi: 10.1016/s0264-410x(99)00178-4. [DOI] [PubMed] [Google Scholar]
- 17.Whittle H, Jaffar S, Wansbrough M, Mendy M, Dumpis U, Collinson A, et al. Observational study on vaccine efficacy 14 years after trial of hepatitis B vaccination in Gambian children. BMJ. 2002 Sep 14;325(7364):569. doi: 10.1136/bmj.325.7364.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van der Sande MAB, Waight P, Mendy M, Rayco-Solon P, Hutt P, Fulford T, et al. Long-Term Protection against Carriage of Hepatitis B Virus after Infant Vaccination. The Journal of Infectious Diseases. 2006 Jun 1;193(11):1528–35. doi: 10.1086/503433. Epub 2006 Apr 25. [DOI] [PubMed] [Google Scholar]
- 19.Peto TJ, Mendy ME, Lowe Y, Webb EL, Whittle HC, Hall AJ. Efficacy and effectiveness of infant vaccination against chronic hepatitis B in the Gambia Hepatitis Intervention Study (1986-90) and in the nationwide immunisation program. BMC Infectious Diseases. 2014;14(7):1471–2334. doi: 10.1186/1471-2334-14-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Akenami FOT, Koskiniemi M, Ekanem EE, Bolarin DM, Vaheri A. Seroprevalence and coprevalence of HIV and HBsAg in Nigerian children with/without protein energy malnutrition. Acta Tropica. 1997;(64):167–74. doi: 10.1016/s0001-706x(96)00629-8. [DOI] [PubMed] [Google Scholar]
- 21.Odusanya OO, Alufohai FE, Meurice FP, Wellens R, Weil J, Ahonkhai VI. Prevalence of hepatitis B surface antigen in vaccinated children and controls in rural Nigeria. International Journal of Infectious Diseases. 2005 May;9(3):139–43. doi: 10.1016/j.ijid.2004.06.009. [DOI] [PubMed] [Google Scholar]
- 22.Odusanya OO, Alufohai E, Meurice FP, Ahonkhai VI. Five-year post vaccination efficacy of hepatitis B vaccine in rural Nigeria. Human Vaccines. 2011;7(6):625–9. doi: 10.4161/hv.7.6.14990. [DOI] [PubMed] [Google Scholar]
- 23.Sadoh AE, Ofili A. Hepatitis B infection among Nigerian children admitted to a children's emergency room. African Health Sciences. 2014 Jun;14(2):377–83. doi: 10.4314/ahs.v14i2.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Okonko I, Okerentugba P, Innocient-Adiele H. Detection of Hepatitis B Surface Antigen (HBsAg) among Children in Ibadan, Southwestern Nigeria. The Internet Journal of Tropical Medicine. 2012;10(1) [Google Scholar]
- 25.Rapicetta M, Stroffolini T, Ngatchu T, Chionne P, Ciccaglione AR, Lantum D, et al. Age- and sex-related study of HBV-DNA in HBsAg asymptomatic children from an endemic area (Cameroon) Annals of Tropical Paediatrics. 1991 doi: 10.1080/02724936.1991.11747523. [DOI] [PubMed] [Google Scholar]
- 26.Rey-Cuille MA, Njouom R, Bekondi C, Seck A, Gody C, Bata P, et al. Hepatitis B virus exposure during childhood in Cameroon, Central African Republic, and Senegal after the integration of HBV vaccine in the Expanded Program on Immunization. The Pediatric Infectious Disease Journal. 2013 Oct;32(10):1110–5. doi: 10.1097/INF.0b013e31829be401. [DOI] [PubMed] [Google Scholar]
- 27.Martinson FE, Weigle KA, Royce RA, Weber DJ, Suchindran CM, Lemon SM. Risk factors for horizontal transmission of hepatitis B virus in a rural district in Ghana. Am J Epidemiol. 1998 Mar 1;147(5):478–87. doi: 10.1093/oxfordjournals.aje.a009474. [DOI] [PubMed] [Google Scholar]
- 28.Dassah S, Sakyi SA, Frempong MT, Luuse AT, Ephraim RK, Anto EO, et al. Seroconversion of Hepatitis B Vaccine in Young Children in the Kassena Nankana District of Ghana: A Cross-Sectional Study. PLoS One. 2015;10(12):e0145209. doi: 10.1371/journal.pone.0145209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ampah KA, Pinho-Nascimento CA, Kerber S, Asare P, De-Graft D, Adu-Nti F, et al. Limited Genetic Diversity of Hepatitis B Virus in the General Population of the Offin River Valley in Ghana. PLoS One. 2016;11(6):e0156864. doi: 10.1371/journal.pone.0156864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Excler JL, Yvonnet B, Gaye Y, Monnereau A, Mangin JL, Schlumberger M, et al. Inclusion de la vaccination contre l'hépatite B dans le Programme élargi de vaccination: étude de faisabilité dans la région médicale de Kolda (Sénégal) Cahiers Santé. 1995;5:37–42. [PubMed] [Google Scholar]
- 31.Sall Diallo A, Sarr M, Fall Y, Diagne C, Kane MO. Hepatitis B infection in infantile population in Senegal. Dakar Medical. 2004;49(2):136–42. [PubMed] [Google Scholar]
- 32.Vardas E, Mathai M, Blaauw D, McAnerney J, Coppin A, Sim J. Preimmunization epidemiology of hepatitis B virus infection in South African children. Journal of Medical Virology. 1999 Jun;58(2):111–5. [PubMed] [Google Scholar]
- 33.Schoub BD, Matai U, Singh B, Blackburn NK, Levin JB. Universal immunization of infants with low doses of a low-cost, plasma-derived hepatitis B vaccine in South Africa. Bulletin of the World Health Organization. 2002;80(4):277–81. [PMC free article] [PubMed] [Google Scholar]
- 34.Amponsah-Dacosta E, Lebelo RL, Rakgole JN, Burnett RJ, Selabe SG, Mphahlele MJ. Evidence for a change in the epidemiology of hepatitis b virus infection after nearly two decades of universal hepatitis B vaccination in South Africa. Journal of Medical Virology. 2014 Jun;86(6):918–24. doi: 10.1002/jmv.23910. [DOI] [PubMed] [Google Scholar]
- 35.Pellizzer G, Ble C, Zamperetti N, Stroffolini T, Upunda G, Rapicetta M, et al. Serological survey of hepatitis B infection in Tanzania. Public Health. 1994 Nov;108(6):427–31. doi: 10.1016/s0033-3506(94)80100-2. [DOI] [PubMed] [Google Scholar]
- 36.Muro FJ, Fiorillo SP, Sakasaka P, Odhiambo C, Reddy EA, Cunningham CK, et al. Seroprevalence of Hepatitis B and C Viruses Among Children in Kilimanjaro Region, Tanzania. J Pediatric Infect Dis Soc. 2013 Dec;2(4):320–6. doi: 10.1093/jpids/pit018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.De Paschale M, Ceriani C, Cerulli T, Cagnin D, Cavallari S, Ndayake J, Zaongo D, Priuli G, Vigano P, Clerici P. Prevalence of HBV, HDV, HCV and HIV Infection During Pregnancy in Northern Benin. Journal of Medical Virology. 2014;86:1281–7. doi: 10.1002/jmv.23951. Epub 29 April 2014. [DOI] [PubMed] [Google Scholar]
- 38.Sangare L, Sombie R, Combassere AW, Kouanda A, Kania D, Zerbo O, et al. Antenatal transmission of hepatitis B virus in an area of HIV moderate prevalence, Burkina Faso. Bull Soc Pathol Exot. 2009 Oct;102(4):226–9. [PubMed] [Google Scholar]
- 39.Nacro B, Dao B, Dahourou H, Hien F, Charpentier-Gautier L, Meda N, et al. HBs antigen carrier state in pregnant women in Bobo Dioulasso (Burkina Faso) Dakar Med. 2000;45(2):188–90. [PubMed] [Google Scholar]
- 40.Collenberg E, Ouedraogo T, Ganame J, Fickenscher H, Kynast-Wolf G, Becher H, et al. Seroprevalence of six different viruses among pregnant women and blood donors in rural and urban Burkina Faso: A comparative analysis. J Med Virol. 2006 May;78(5):683–92. doi: 10.1002/jmv.20593. [DOI] [PubMed] [Google Scholar]
- 41.Ilboudo D, Karou D, Nadembega WMC, Savadogo A, Djeneba O, Pignatelli S, Pietra V, Bere A, Simpore J, Traore AS. Prevalence of Human Herpes Virus-8 and Hepatitis B Virus among HIV Seropositive Pregnant Women Enrolled in the Mother-to-child HIV Transmission Prevention Program at Saint Camille Medical Centre in Burkina Faso. Pakistan Journal of Biological Sciences. 2007;10(17):2831–7. doi: 10.3923/pjbs.2007.2831.2837. [DOI] [PubMed] [Google Scholar]
- 42.Ouermi D, Simpore J, Belem AM, Sanou DS, Karou DS, Ilboudo D, et al. Co-infection of Toxoplasma gondii with HBV in HIV-infected and uninfected pregnant women in Burkina Faso. Pak J Biol Sci. 2009 Sep 1;12(17):1188–93. doi: 10.3923/pjbs.2009.1188.1193. [DOI] [PubMed] [Google Scholar]
- 43.Simpore J, Granato M, Santarelli R, Nsme RA, Coluzzi M, Pietra V, et al. Prevalence of infection by HHV-8, HIV, HCV and HBV among pregnant women in Burkina Faso. J Clin Virol. 2004 Sep;31(1):78–80. doi: 10.1016/j.jcv.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 44.Simpore J, Savadogo A, Ilboudo D, Nadambega MC, Esposito M, Yara J, et al. Toxoplasma gondii, HCV and HBV seroprevalence and co-infection among HIV-positive and -negative pregnant women in Burkina Faso. J Med Virol. 2006 Jun;78(6):730–3. doi: 10.1002/jmv.20615. [DOI] [PubMed] [Google Scholar]
- 45.Ekra D, Herbinger KH, Konate S, Leblond A, Fretz C, Cilote V, et al. A non-randomized vaccine effectiveness trial of accelerated infant hepatitis B immunization schedules with a first dose at birth or age 6 weeks in Cote d'Ivoire. Vaccine. 2008 May 23;26(22):2753–61. doi: 10.1016/j.vaccine.2008.03.018. [DOI] [PubMed] [Google Scholar]
- 46.Lohoues-Kouacou MJ, Toure M, Hillah J, Camara BM, N'Dri N, Kouame KJ, et al. Materno-fetal transmission of hepatitis B virus in Ivory Coast - Plea for mass vaccination. Sante. 1998 Nov-Dec;8(6):401–4. [PubMed] [Google Scholar]
- 47.Rouet F, Chaix ML, Inwoley A, Msellati P, Viho I, Combe P, et al. HBV and HCV Prevalence and Viraemia in HIV-Positive and HIV-Negative Pregnant Women in Abidjan, Cote d'Ivoire: The ANRS 1236 Study. Journal of Medical Virology. 2004 Sep;74(1):34–40. doi: 10.1002/jmv.20143. [DOI] [PubMed] [Google Scholar]
- 48.Apea-Kubi KA, Yamaguchi S, Sakyi B, Ofori-Adjei D. HTLV-1 and other viral sexually transmitted infections in antenatal and gynaecological patients in Ghana. West African Journal Medicine. 2006;25(1) doi: 10.4314/wajm.v25i1.28239. [DOI] [PubMed] [Google Scholar]
- 49.Candotti D, Danso K, Allain JP. Maternofetal transmission of hepatitis B virus genotype E in Ghana, west Africa. Journal of General Virology. 2007 Oct;88(Pt 10):2686–95. doi: 10.1099/vir.0.83102-0. [DOI] [PubMed] [Google Scholar]
- 50.Cho Y, Bonsu G, Akoto-Ampaw A, Nkrumah-Mills G, Nimo JA, Park JK, et al. The prevalence and risk factors for hepatitis B Surface Ag Positivity in Pregnant Women in Eastern Region of Ghana. Gut and Liver. 2012;6(2):235–40. doi: 10.5009/gnl.2012.6.2.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ephraim R, Donko I, Sakyi SA, Ampong J, Agbodjakey H. Seroprevalence and risk factors of Hepatitis B and Hepatitis C infections among pregnant women in the Asante Akim North Municipality of the Ashanti region, Ghana; a cross sectional study. Afr Health Sci. 2015 Sep;15(3):709–13. doi: 10.4314/ahs.v15i3.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sidibe S, Sacko BY, Traore I. Prevalence of serologic markers of the hepatitis B virus in pregnant women of Bamako, Mali. Bull Soc Pathol Exot. 2001 Nov;94(4):339–41. [PubMed] [Google Scholar]
- 53.MacLean B, Hess RF, Bonvillain E, Kamate J, Dao D, Cosimano A, et al. Seroprevalence of hepatitis B surface antigen among pregnant women attending the Hospital for Women & Children in Koutiala, Mali. S Afr Med J. 2011 Dec 14;102(1):47–9. [PubMed] [Google Scholar]
- 54.Mansour W, Malick FZ, Sidiya A, Ishagh E, Chekaraou MA, Veillon P, et al. Prevalence, risk factors, and molecular epidemiology of hepatitis B and hepatitis delta virus in pregnant women and in patients in Mauritania. J Med Virol. 2012 Aug;84(8):1186–98. doi: 10.1002/jmv.23336. [DOI] [PubMed] [Google Scholar]
- 55.Mamadou S, Ide M, Maazou AR, Aoula B, Labo S, Bozari M. HIV infection and hepatitis B seroprevalence among antenatal clinic attendees in Niger, West Africa. HIV AIDS (Auckl). 2012;4:1–4. doi: 10.2147/HIV.S27881. Epub 2012 Jan 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Buseri FI, Seiyaboh E, Jeremiah ZA. Surveying Infections among Pregnant Women in the Niger Delta Nigeria. J Glob Infect Dis. 2010;2(3):203–11. doi: 10.4103/0974-777X.68525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Eke AC, Eke UA, Okafor CI, Ezebialu IU, Ogbuagu C. Prevalence, correlates and pattern of hepatitis B surface antigen in a low resource setting. Virol J. 2011 Jan 12;8:12. doi: 10.1186/1743-422X-8-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Eze M, Buseri FI, Wachukwu CK, Nnatuanya IN. Effects of Hepatitis B Infection on Haemtological Parameters in Pregnancy in Port Harcourt, Nigeria. Research Journal of Medical Sciences. 2009;3(6):194–7. [Google Scholar]
- 59.Ikeako L, Ezegwui H, Ajah L, Dim C, Okeke T. Seroprevalence of Human Immunodeficiency Virus, Hepatitis B, Hepatitis C, Syphilis, and Co-infections among Antenatal Women in a Tertiary Institution in South East, Nigeria. Ann Med Health Sci Res. 2014 Nov;4(6):954–8. doi: 10.4103/2141-9248.144925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kolawole OM, Wahab AA, Adekanle DA, Sibanda T, Okoh AI. Seroprevalence of hepatitis B surface antigenemia and its effects on hematological parameters in pregnant women in Osogbo, Nigeria. Virol J. 2012 Dec 27;9:317. doi: 10.1186/1743-422X-9-317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mbaawuaga EM, Enenebeaku MNO, Okopi JA, Damen JG. Hepatitis B Virus (HBV) Infection among Pregnant Women in Makurdi, Nigeria. African Journal of Biomedical Research. 2008;11:155–9. [Google Scholar]
- 62.Olokoba AB, Salawu FK, Danburam A, Olokoba LB, Midala JK, Badung LH, et al. Hepatitis B virus infection amongst pregnant women in North-eastern Nigeria - a call for action. Niger J Clin Pract. 2011 Jan-Mar;14(1):10–3. doi: 10.4103/1119-3077.79232. [DOI] [PubMed] [Google Scholar]
- 63.Omalu ICJ, Jibrin A, Olayemi IK, Hassan SC, Mgbemena C, Mgbemena A, et al. Seroprevalence of Malaria and Hepatitis B (HBsAg) with Associated Risk Factors among Pregnant Women Attending Antenatal Clinic in General Hospital Minna, North-Central Nigeria. Annual Review & Research in Biology. 2012;2(4):83–8. [Google Scholar]
- 64.Onakewhor JU, Offor E, Okonofua FE. Maternal and neonatal seroprevalence of hepatitis B surface antigen (HBsAg) in Benin City, Nigeria. J Obstet Gynaecol. 2001 Nov;21(6):583–6. doi: 10.1080/01443610120085528. [DOI] [PubMed] [Google Scholar]
- 65.Osazuwa F, Obinna OV, Chika AF. Sero-epidemiology of Human Immunodeficiency Virus, Hepatitis B and C among Pregnant Women in Rural Communities of Abaji Area Council, Nigeria. TAF Preventive Medicine Bulletin. 2012;11(4) [Google Scholar]
- 66.Pennap GR, Muazu IF, Fatima M. Parallel and Overlapping Hepatitis B and C Virus Infection among Pregnant Women Attending Antenatal in a Rural Clinic in Northern Nigeria. International Journal of Current Microbiology and Applied Sciences. 2015;4(5):16–23. [Google Scholar]
- 67.Pennap GR, Osanga ET, Ubam A. Seroprevalence of Hepatitis B Surface Antigen among Pregnant Women Attending Antenatal Clinic in Federal Medical Center Keffi, Nigeria. Research Journal of Medical Sciences. 2011;5(2):80–2. [Google Scholar]
- 68.Ugbebor O, Aigbirior M, Osazuwa F, Enabudoso E, Zabayo O. The prevalence of hepatitis B and C viral infections among pregnant women. N Am J Med Sci. 2011 May;3(5):238–41. doi: 10.4297/najms.2011.3238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Usanga V, Abia-Bassey L, Inyang-etoh P, Udoh S, Ani F, Archibong E. Prevalence of Sexually Transmitted Diseases in Pregnant and Non-Pregnant Women in Calabar, Cross River State, Nigeria. The Internet Journal of Gynecology and Obstetrics. 2009;14(2) [Google Scholar]
- 70.Aba HO, Aminu M. Seroprevalence of hepatitis B virus serological markers among pregnant Nigerian women. Ann Afr Med. 2016 Jan-Mar;15(1):20–7. doi: 10.4103/1596-3519.172555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Adegbesan-Omilabu MA, Okunade KS, Gbadegesin A, Olowoselu OF, Oluwole AA, Omilabu SA. Seroprevalence of hepatitis B virus infection among pregnant women at the antenatal booking clinic of a Tertiary Hospital in Lagos Nigeria. Nigerian Journal of Clinical Practice. 2015;18(6) doi: 10.4103/1119-3077.163283. [DOI] [PubMed] [Google Scholar]
- 72.Anaedobe CG, Fowotade A, Omoruyi CE, Bakare RA. Prevalence, sociodemographic features and risk factors of Hepatitis B virus infection among pregnant women in Southwestern Nigeria. Pan Afr Med J. 2015;20:406. doi: 10.11604/pamj.2015.20.406.6206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Faleye TO, Adewumi MO, Ifeorah IM, Omoruyi EC, Bakarey SA, Akere A, et al. Detection of hepatitis B virus isolates with mutations associated with immune escape mutants among pregnant women in Ibadan, southwestern Nigeria. Springerplus. 2015;4:43. doi: 10.1186/s40064-015-0813-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Iklaki CU, Emechebe CI, Ago BU, Njoku CO. Sero-prevalence of Hepatitis B infection and its risk factors among women admitted for delivery in Ucth, Calabar, Nigeria. British Journal of Medicine & Medical Research. 2015;8(4):324–33. [Google Scholar]
- 75.Olaleye OA, Kuti O, Makinde NO, Ujah AO, Olaleye OA, Badejoko OO, et al. Perinatal transmission of hepatitis B virus infection in Ile-Ife, South Western, Nigeria. J Neonatal Perinatal Med. 2013;6(3):231–6. doi: 10.3233/NPM-1366412. [DOI] [PubMed] [Google Scholar]
- 76Opaleye OO, Igboama MC, Ojo JA, Odewale G. Seroprevalence of HIV, HBV, HCV and HTLV among pregnant women in Southwestern Nigeria. Journal of Immunoassay and Immunochemistry. 2016;37(1):29–42. doi: 10.1080/15321819.2015.1040160. [DOI] [PubMed] [Google Scholar]
- 77.Utoo BT Hepatitis B surface antigenemia (HBsAg) among pregnant women in southern Nigeria. Afr Health Sci. 2013 Dec;13(4):1139–43. doi: 10.4314/ahs.v13i4.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Adeyemi AB, Enabor OO, Ugwu IA, Abraham OA, Bello FA, Olayemi O. Prevalence of antenatal hepatitis B infection in tertiary and non-tertiary health facilities in Ibadan, Nigeria. Niger J Med. 2014 Jul-Sep;23(3):248–53. [PubMed] [Google Scholar]
- 79.Akani CI, Ojule AC, Opurum HC, Ejilemele AA. Sero-prevalence of hepatitis B surface antigen (HBsAg) in pregnant women in Port Harcourt, Nigeria. Niger Postgrad Med J. 2005 Dec;12(4):266–70. [PubMed] [Google Scholar]
- 80.Baba MM, Onwuka IS, Baba SS. Hepatitis B and C virus infections among pregnant women in Maiduguri, Nigeria. Central European Journal of Public Health. 1999;7(2):60–2. [Google Scholar]
- 81.Onwere S, Chigbu B, Aluka C, Kamanu CI, Okoro O, Ndukwe PE, et al. Risk Factors for Hepatitis B Virus Infection during Pregnancy in South Eastern Nigeria. East Afr Med J. 2012 Mar;89(3):89–93. [PubMed] [Google Scholar]
- 82.Wurie IM, Wurie AT, Gevao SM. Sero-prevalence of hepatitis B virus among middle to high socio-economic antenatal population in Sierra Leone. West Afr J Med. 2005 Jan-Mar;24(1):18–20. doi: 10.4314/wajm.v24i1.28156. [DOI] [PubMed] [Google Scholar]
- 83.Abongwa LM, Clara AM, Edouard NA, Ngum NH. Sero-Prevalence of Human Immunodeficiency Virus (HIV) and Hepatitis B Virus (HBV) Co-Infection among Pregnant Women Residing in Bamenda Health District, Cameroon. International Journal of Current Microbiology and Applied Sciences. 2015;4(12):473–83. [Google Scholar]
- 84.Dionne-Odom J, Mbah R, Rembert NJ, Tancho S, Halle-Ekane GE, Enah C, et al. Hepatitis B, HIV, and Syphilis Seroprevalence in Pregnant Women and Blood Donors in Cameroon. Infect Dis Obstet Gynecol. 2016;2016:4359401. doi: 10.1155/2016/4359401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ducancelle A, Abgueguen P, Birguel J, Mansour W, Pivert A, Le Guillou-Guillemette H, et al. High endemicity and low molecular diversity of hepatitis B virus infections in pregnant women in a rural district of North Cameroon. PLoS One. 2013;8(11):e80346. doi: 10.1371/journal.pone.0080346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Fomulu NJ, Morfaw FL, Torimiro JN, Nana P, Koh MV, William T. Prevalence, correlates and pattern of Hepatitis B among antenatal clinic attenders in Yaounde-Cameroon: is perinatal transmission of HBV neglected in Cameroon? BMC Pregnancy Childbirth. 2013 Aug 08;13:158. doi: 10.1186/1471-2393-13-158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Frambo AA, Atashili J, Fon PN, Ndumbe PM. Prevalence of HBsAg and knowledge about hepatitis B in pregnancy in the Buea Health District, Cameroon: a cross-sectional study. BMC Res Notes. 2014 Jun 25;7:394. doi: 10.1186/1756-0500-7-394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kfutwah AKW, Tejiokem MC, Njouom R. A low proportion of HBeAg among HBsAg- positive pregnant women with known HIV status could suggest low perinatal transmission of HBV in Cameroon. Virology Journal. 2012;9(62) doi: 10.1186/1743-422X-9-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Noubiap JJ, Nansseu JR, Ndoula ST, Bigna JJ, Jingi AM, Fokom-Domgue J. Prevalence, infectivity and correlates of hepatitis B virus infection among pregnant women in a rural district of the Far North Region of Cameroon. BMC Public Health. 2015 May 02;15:454. doi: 10.1186/s12889-015-1806-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Makuwa M, Caron M, Souquiere S, Malonga-Mouelet G, Mahe A, Kazanji M. Prevalence and genetic diversity of hepatitis B and delta viruses in pregnant women in Gabon: molecular evidence that hepatitis delta virus clade 8 originates from and is endemic in central Africa. J Clin Microbiol. 2008 Feb;46(2):754–6. doi: 10.1128/JCM.02142-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Awole M, Gebre-Selassie S. Seroprevalence of HBsAg and its risk factors among pregnant women in Jimma, Southwest Ethiopia. Ethiop J Health Dev. 2005;19(1) [Google Scholar]
- 92.Desalegn Z, Wassie L, Beyene HB, Mihret A, Ebstie YA. Hepatitis B and human immunodeficiency virus co-infection among pregnant women in resource-limited high endemic setting, Addis Ababa, Ethiopia: implications for prevention and control measures. Eur J Med Res. 2016 Apr 14;21:16. doi: 10.1186/s40001-016-0211-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Metaferia Y, Dessie W, Ali I, Amsalu A. Seroprevalence and associated risk factors of hepatitis B virus among pregnant women in southern Ethiopia: a hospital-based cross-sectional study. Epidemiol Health. 2016;38:e2016027. doi: 10.4178/epih.e2016027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Molla S, Munshea A, Nibret E. Seroprevalence of hepatitis B surface antigen and anti HCV antibody and its associated risk factors among pregnant women attending maternity ward of Felege Hiwot Referral Hospital, northwest Ethiopia: a cross-sectional study. Virol J. 2015 Dec 02;12:204. doi: 10.1186/s12985-015-0437-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ramos JM, Toro C, Reyes F, Amor A, Gutierrez F. Seroprevalence of HIV-1, HBV, HTLV-1 and Treponema pallidum among pregnant women in a rural hospital in Southern Ethiopia. Journal of Clinical Virology. 2011 May;51(1):83–5. doi: 10.1016/j.jcv.2011.01.010. [DOI] [PubMed] [Google Scholar]
- 96.Tegegne D, Desta K, Tegbaru B, Tilahun T. Seroprevalence and transmission of Hepatitis B virus among delivering women and their new born in selected health facilities, Addis Ababa, Ethiopia: a cross sectional study. BMC Res Notes. 2014 Apr 15;7:239. doi: 10.1186/1756-0500-7-239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Yohanes T, Zerdo Z, Chufamo N. Seroprevalence and Predictors of Hepatitis B Virus Infection among Pregnant Women Attending Routine Antenatal Care in Arba Minch Hospital, South Ethiopia. Hepat Res Treat. 2016;2016:9290163. doi: 10.1155/2016/9290163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Zenebe Y, Mulu W, Yimer M, Abera B. Sero-prevalence and risk factors of hepatitis B virus and human immunodeficiency virus infection among pregnant women in Bahir Dar city, Northwest Ethiopia: a cross sectional study. BMC Infect Dis. 2014 Mar 01;14:118. doi: 10.1186/1471-2334-14-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Tiruneh M. Seroprevalence of multiple sexually transmitted infections among antenatal clinic attendees in Gondar Health Center, northwest Ethiopia. Ethiop Med J. 2008;46(4):359–66. [PubMed] [Google Scholar]
- 100.Okoth F, Mbuthia J, Gatheru Z, Murila F, Kanyingi F, Mugo F, et al. Seroprevalence of hepatitis B markers in pregnant women in Kenya. East Afr Med J. 2006 Sep;83(9):485–93. doi: 10.4314/eamj.v83i09.46771. [DOI] [PubMed] [Google Scholar]
- 101.Randriamahazo TR, Raherinaivo AA, Rakotoarivelo ZH, Contamin B, Rakoto Alson OA, Andrianapanalinarivo HR, et al. Prevalence of hepatitis B virus serologic markers in pregnant patients in Antananarivo, Madagascar. Med Mal Infect. 2015 Jan-Feb;45(1-2):17–20. doi: 10.1016/j.medmal.2014.10.008. [DOI] [PubMed] [Google Scholar]
- 102.Andersson MI, Maponga TG, Ijaz S, Barnes J, Theron GB, Meredith SA, et al. The epidemiology of hepatitis B virus infection in HIV-infected and HIV-uninfected pregnant women in the Western Cape, South Africa. Vaccine. 2013 Nov 12;31(47):5579–84. doi: 10.1016/j.vaccine.2013.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Burnett RJ, Ngobeni JM, Francois G, Hoosen AA, Leroux-Roels G, Meheus A, et al. Increased exposure to hepatitis B virus infection in HIV-positive South African antenatal women. Int J STD AIDS. 2007 Mar;18(3):152–6. doi: 10.1258/095646207780132523. [DOI] [PubMed] [Google Scholar]
- 104.Diale Q, Pattinson R, Chokoe R, Masenyetse L, Mayaphi S. Antenatal screening for hepatitis B virus in HIV-infected and uninfected pregnant women in the Tshwane district of South Africa. S Afr Med J. 2015 Dec 16;106(1):97–100. doi: 10.7196/SAMJ.2016.v106i1.9932. [DOI] [PubMed] [Google Scholar]
- 105.Thumbiran NV, Moodley D, Parboosing R, Moodley P. Hepatitis B and HIV co-infection in pregnant women: indication for routine antenatal hepatitis B virus screening in a high HIV prevalence setting. S Afr Med J. 2014 Apr;104(4):307–9. doi: 10.7196/samj.7299. [DOI] [PubMed] [Google Scholar]
- 106.Bayo P, Ochola E, Oleo C, Mwaka AD. High prevalence of hepatitis B virus infection among pregnant women attending antenatal care: a cross-sectional study in two hospitals in northern Uganda. BMJ Open. 2014 Nov 11;4(11):e005889. doi: 10.1136/bmjopen-2014-005889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Croce F, Fedeli P, Dahoma M, Deho L, Ramsan M, Adorni F, et al. Risk factors for HIV/AIDS in a low HIV prevalence site of sub-Saharan Africa. Trop Med Int Health. 2007 Sep;12(9):1011–7. doi: 10.1111/j.1365-3156.2007.01880.x. [DOI] [PubMed] [Google Scholar]
- 108.Menendez C, Sanchez-Tapias JM, Kahigwa E, Mshinda H, Costa J, Vidal J, et al. Prevalence and mother-to-infant transmission of hepatitis viruses B, C and E in Southern Tanzania. J Med Virol. 1999 Jul;58(3):215–20. doi: 10.1002/(sici)1096-9071(199907)58:3<215::aid-jmv5>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 109.Rashid S, Kilewo C, Aboud S. Seroprevalence of hepatitis B virus infection among antenatal clinic attendees at a tertiary hospital in Dar es Salaam, Tanzania. Tanzan J Health Res. 2014 Jan;16(1):9–15. doi: 10.4314/thrb.v16i1.2. [DOI] [PubMed] [Google Scholar]
- 110.Mavenyengwa RT, Moyo SR, Nordbo SA. Streptococcus agalactiae colonization and correlation with HIV-1 and HBV seroprevalence in pregnant women from Zimbabwe. European Journal of Obstretrics & Gynecology and Reproductive Biology. 2009;150:34–8. doi: 10.1016/j.ejogrb.2010.02.021. [DOI] [PubMed] [Google Scholar]
- 111.Madzime S, Adem M, Mahomed K, Woelk GB, Mudzamiri S, Williams MA. Hepatitis B virus infection among pregnant women delivering at Harare Maternity Hospital, Harare Zimbabwe, 1996 to 1997. Cent Afr J Med. 1999 Aug;45(8):195–8. doi: 10.4314/cajm.v45i8.8414. [DOI] [PubMed] [Google Scholar]
- 112.Miyahara R, Jasseh M, Gomez P, Shimakawa Y, Greenwood B, Keita K, et al. Barriers to timely administration of birth dose vaccines in The Gambia, West Africa. Vaccine. 2016 Jun 17;34(29):3335–41. doi: 10.1016/j.vaccine.2016.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wiesen E, Diorditsa S, Li X. Progress towards hepatitis B prevention through vaccination in the Western Pacific, 1990-2014. Vaccine. 2016 May 27;34(25):2855–62. doi: 10.1016/j.vaccine.2016.03.060. [DOI] [PubMed] [Google Scholar]
- 114.World Health Organization . Preventing perinatal Hepatitis B Virus Transmission: A guide for Introducing and Strengthening Hepatitis B birth dose vaccination. 2015. (14 December 2016) [Google Scholar]
- 115.UNICEF . HepB-UNICEF. 2016. (updated 4/14/2016; cited 2016 12/15/2016) [Google Scholar]
- 116.World Health Organization . Summary of the October 2016 meeting of the Strategic Advisory Group of Experts on immunization (SAGE) 2016. (cited 2016 12/14/2016) [Google Scholar]
- 117.Borresen ML, Koch A, Biggar RJ, Ladefoged K, Melbye M, Wohlfahrt J, et al. Effectiveness of the targeted hepatitis B vaccination program in Greenland. Am J Public Health. 2012 Feb;102(2):277–84. doi: 10.2105/AJPH.2011.300239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Hutin Y, Hennessey K, Cairns L, Zhang Y, Li H, Zhao L, et al. Improving hepatitis B vaccine timely birth dose coverage: lessons from five demonstration projects in China, 2005-2009. Vaccine. 2013 Dec 27;31(Suppl 9):J49–55. doi: 10.1016/j.vaccine.2013.03.025. [DOI] [PubMed] [Google Scholar]
- 119.Hennessey K, Mendoza-Aldana J, Bayutas B, Lorenzo-Mariano KM, Diorditsa S. Hepatitis B control in the World Health Organization's Western Pacific Region: targets, strategies, status. Vaccine. 2013 Dec 27;31(Suppl 9):J85–92.. doi: 10.1016/j.vaccine.2012.10.082. [DOI] [PubMed] [Google Scholar]
- 120.Xeuatvongsa A, Datta SS, Moturi E, Wannemuehler K, Philakong P, Vongxay V, et al. Improving hepatitis B birth dose in rural Lao People's Democratic Republic through the use of mobile phones to facilitate communication. Vaccine. 2016 Nov 11;34(47):5777–84. doi: 10.1016/j.vaccine.2016.09.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sutanto A, Suarnawa IM, Nelson CM, Stewart T, Soewarso TI. Home delivery of heat-stable vaccines in Indonesia: outreach immunization with a prefilled, single-use injection device. Bull World Health Organ. 1999;77(2):119–26. [PMC free article] [PubMed] [Google Scholar]
- 122.World Health Organization . Evidence presented to SAGE. 2016. Oct, A systematic review of monovalent hepatitis B vaccine thermostability. (cited 2016 11/14/2016) [Google Scholar]
- 123.Creati M, Saleh A, Ruff TA, Stewart T, Otto B, Sutanto A, et al. Implementing the birth dose of hepatitis B vaccine in rural Indonesia. Vaccine. 2007 Aug 10;25(32):5985–93. doi: 10.1016/j.vaccine.2007.05.055. [DOI] [PubMed] [Google Scholar]
- 124.Wang L, Li J, Chen H, Li F, Armstrong GL, Nelson C, et al. Hepatitis B vaccination of newborn infants in rural China: evaluation of a village-based, out-of-cold-chain delivery strategy. Bull World Health Organ. 2007 Sep;85(9):688–94. doi: 10.2471/BLT.06.037002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kolwaite AR, Xeuatvongsa A, Ramirez-Gonzalez A, Wannemuehler K, Vongxay V, Vilayvone V, et al. Hepatitis B vaccine stored outside the cold chain setting: a pilot study in rural Lao PDR. Vaccine. 2016 Jun 14;34(28):3324–30. doi: 10.1016/j.vaccine.2016.03.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Huong VM, Hipgrave D, Hills S, Nelson C, Hien DS, Cuong NV. Out-of-Cold-Chain Delivery of the Hepatitis B Birth Dose in Four Districts of Vietnam. PATH (Program for Appropriate Technology in Health), 2006. 2006 Oct; Report No. [Google Scholar]
- 127.World Health Organization . Regional Office for Africa, editor. 2016. Prevention, Care and Treatment of Viral Hepatitis in the African Region: Framework for Action, 2016-2020. (cited 17 february 2017) [Google Scholar]


