Key Points
Patients with cGVHD develop antibodies targeting cell membrane antigens.
Targeted antigens are broadly expressed in acute myeloblastic leukemia cells and in tissues affected by cGVHD.
Abstract
Chronic graft-versus-host disease (cGVHD) after allogeneic hematopoietic stem cell transplant reflects a complex immune response resulting in chronic damage to multiple tissues. Previous studies indicated that donor B cells and the antibodies they produce play an important role in the development of cGVHD. To understand the pathogenic role of antibodies in cGVHD, we focused our studies on posttransplant production of immunoglobulin G antibodies targeting cell surface antigens expressed in multiple cGVHD affected tissues, due to their potential functional impact on living cells in vivo. Using plate-bound cell membrane proteins as targets, we detected a significantly higher level of antibodies reactive with these membrane antigens in patients who developed cGVHD, compared with those who did not and healthy donors. Plasma-reactive antibody levels increased significantly prior to the clinical diagnosis of cGVHD and were reduced following cGVHD therapies including prednisone, interleukin-2, or extracorporeal photophoresis. Using cell-based immunoprecipitation with plasma from cGVHD patients and mass spectrometry, we identified 43 membrane proteins targeted by these antibodies. The presence of antibodies in cGVHD patients’ plasma that specifically target 6 of these proteins was validated. Antibodies reactive with these 6 antigens were more frequently detected in patients with cGVHD compared with patients without cGVHD and healthy donors. These results indicate that antibodies that target membrane antigens of living cells frequently develop in cGVHD patients and further support a role for B cells and antibodies in the development of cGVHD.
Visual Abstract
Introduction
Chronic graft-versus-host disease (cGVHD) is a major complication of allogeneic hematopoietic stem cell transplant (HSCT) and remains a leading cause of long-term morbidity and mortality.1-4 The clinical manifestations of cGVHD can affect multiple organ systems and often resemble autoimmune diseases.3 Recent studies in murine models and patients have provided new insights into the complex immunopathologic mechanisms that contribute to immune dysregulation and the development of cGVHD months to years after allogeneic HSCT.4,5 These studies highlight the complexity of immune responses in cGVHD and interactions between adaptive and innate immune systems resulting in chronic inflammation and tissue damage.6
In this context, many studies have demonstrated an important role for B-cell immune responses in cGVHD. Donor B cells can function as antigen-presenting cells to facilitate the development of cGVHD.7-10 In these studies, antigen presentation by donor B cells promotes the survival and proliferation of pathogenic CD4+ T cells and is essential for the development of cGVHD caused by donor T cells. In addition to their antigen-presentation function, recent studies revealed that the antibodies produced by B cells also play an important role in the pathogenesis of cGVHD. Antibodies against HY minor histocompatibility antigens frequently develop in male recipients with female stem cell donors and the development of these antibodies is significantly increased in patients with cGVHD.11-13 Notably, anti-HY antibodies can be detected as early as 3 months after transplant and precede the clinical onset of cGVHD.14 In murine models, antibodies produced by donor B cells are required for the development of cGVHD. Immunoglobulin G (IgG)-containing donor serum leads to deposition of IgG in thymus and skin, resulting in tissue damage and perpetuation of cGVHD. Interventions that prevent B-cell recovery or inhibit B-cell function prevent or delay the onset of cGVHD.15-17
In addition to antibodies against minor histocompatibility antigens, studies have identified antibodies specific for other antigens in patients with cGVHD. However, most antibodies previously identified in this setting are specific for intracellular antigens.11,18,19 Considering the important role that antibodies play in the development of cGVHD, we hypothesized that patients with cGVHD also develop antibodies that target surface antigens on live cells. Antibodies directed against surface membrane antigens would have a direct functional impact on target cells in affected tissues and could help explain the mechanism whereby antibodies contribute to the pathogenesis of cGVHD. To address this issue, we developed specific assays to monitor the development of antibodies that target membrane antigens after allogeneic HSCT. We further investigated the specific targets of these anti-membrane antibodies and validated the specificity of antibodies against 6 membrane antigens in patients with cGVHD. Our goals were to characterize the generation of anti-membrane antibodies in cGVHD and correlate the levels of anti-membrane antibodies with the onset of cGVHD and response to immune-suppressive therapy.
Materials and methods
Patient characteristics
All samples were collected from patients and healthy donors enrolled in clinical research protocols approved by the Human Subjects Protection Committee of the Dana-Farber/Harvard Cancer Center. Written informed consent was obtained prior to sample collection, in accordance with the Declaration of Helsinki. Experiments were performed with plasma samples from patients who underwent allogeneic HSCT at the Dana-Farber Cancer Institute and Brigham and Women’s Hospital (Boston, MA). Clinical characteristics of these patients are summarized in Tables 1 and 2. Patients in the cGVHD cohort had active cGVHD, which was defined by the presence of ongoing cGVHD symptoms but these patients were not receiving intensive immune-suppressive therapy at the time of sample collection. Ninety-three patients were receiving prednisone <20 mg daily, sirolimus up to 5 mg per day, or tacrolimus up to 4 mg per day. Seven patients were on topical steroids, 6 patients had new onset cGVHD and were treated after the samples were collected, and 5 were not on treatment. Patients in the cGVHD-T cohort were receiving more intensive immune-suppressive treatment including prednisone at 20 mg per day or higher dose, extracorporeal photopheresis (ECP), or low-dose interleukin-2 (IL-2). cGVHD patients who received cord blood transplants or rituximab after transplant were excluded from this study. Samples were not included if patients received immunoglobulin infusion within 3 months prior to sample collection.
Table 1.
Characteristics of HSCT patients
| Characteristics | No cGVHD, n = 85 | cGVHD, n = 106 | cGVHD-T, n = 93 | |
|---|---|---|---|---|
| Age, y | Medium | 56 | 61 | 58 |
| Range | 22-75 | 30-77 | 26-75 | |
| Sex, % | F | 47 (55) | 56 (53) | 40 (43) |
| M | 38 (45) | 50 (47) | 53 (57) | |
| Disease, % | AML/MDS | 65 (76) | 85 (80) | 63 (68) |
| Others | 20 (24) | 21 (20) | 30 (32) | |
| Conditioning regimen, % | MAC | 35 (41) | 36 (34) | 32 (34) |
| RIC | 50 (59) | 70 (66) | 61 (66) | |
| Donor type, % | MRD | 37 (44) | 29 (27) | 31 (33) |
| MUD | 42 (49) | 58 (55) | 49 (53) | |
| MMUD | 6 (7) | 19 (18) | 13 (14) | |
| GVHD prophylaxis, % | Tac-MTX | 35 (41) | 41 (39) | 31 (33) |
| Tac-Sir | 12 (14) | 14 (13) | 25 (27) | |
| Tac-Sir-MTX | 27 (32) | 41 (39) | 30 (32) | |
| others | 11 (13) | 10 (9) | 7 (8) | |
| Months from HSCT to sample collection | Median | 22 | 24 | 20 |
| Range | 12-164 | 6-116 | 6-140 |
AML, acute myeloid leukemia; cGVHD, chronic GVHD patients as described in “Materials and methods”; cGVHD-T, chronic GVHD patients receiving intensive treatments described in “Materials and methods”; F, female; M, male; MAC, myeloablative conditioning; MDS, myelodysplastic syndrome; MMUD, mismatched unrelated donor; MRD, matched related donor; MTX, methotrexate; MUD, matched unrelated donor; RIC, reduced-intensity conditioning; Sir, rapamycin; Tac, tacrolimus.
Table 2.
Characteristics of HSCT patients for posttransplant follow-up study
| Characteristics | No cGVHD, n = 19 | cGVHD, n = 30 | |
|---|---|---|---|
| Age at transplant, y | Medium | 51 | 60 |
| Range | 21-69 | 29-71 | |
| Sex, % | F | 12 (63) | 13 (43) |
| M | 7 (37) | 17 (57) | |
| Disease, % | AML/MDS | 12 (63) | 24 (80) |
| Others | 7 (37) | 6 (20) | |
| Conditioning regimen, % | MAC | 6 (32) | 10 (33) |
| RIC | 13 (68) | 20 (67) | |
| Donor type, % | MRD | 8 (42) | 9 (30) |
| MUD | 9 (47) | 19 (63) | |
| MMUD | 2 (11) | 2 (7) | |
| GVHD prophylaxis, % | Tac-MTX | 11 (57) | 11 (37) |
| Tac-Sir | 3 (16) | 2 (7) | |
| Tac-Sir-MTX | 3 (16) | 13 (43) | |
| Others | 4 (21) | 4 (13) | |
Abbreviations are explained in Table 1.
Antibodies, cell lines, and reagents
Goat anti-human IgG, Fc specific (SouthernBiotech), was conjugated with Alexa Fluor A680 using the Alexa Fluor A680 conjugation kit (Invitrogen, Carlsbad, CA). Acute myeloblastic leukemia (AML; Kasumi-1), skin keratinocyte cell line CCD1106 (originated from human embryo), skin keratinocyte cell line HEK001 (originated from adult human), and skin fibroblast cell line (Detroit 551) were purchased from American Type Culture Collection (ATCC; Manassas, VA). Cell culture media and supplies were purchased from Invitrogen. Unconjugated antibodies were purchased from BioLegend (San Diego, CA). Fluorescence-conjugated antibodies for flow cytometry were purchased from BD Biosciences (San Jose, CA) and BioLegend.
Membrane protein extract preparation
Single-cell suspensions of cultured cell lines in Dounce buffer (10 mM Tris, pH 7.4, 0.5 mM MgCl2) were irrupted in a nitrogen pressure vessel (Parr Instrument Co., Moline, IL) twice under 1500 PSI to 1800 PSI.20 The cell lysate was centrifuged 3000 rpm for 20 minutes. The pellet was resuspended with Dounce buffer and centrifuged in the same conditions. The pellet was resuspended in EBC250 buffer (250 mM NaCl, 10 mM Tris, pH 7.4, protease inhibitors) containing 3% 3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate (CHAPS) and 1% Mega-10.21 The lysate was centrifuged at 14 000 rpm for 15 minutes to remove any insoluble material. The membrane protein containing supernatant was collected and dialyzed against phosphate-buffered saline (PBS) overnight. The final product was stored at −80°C.
Fluorescence-linked immunosorbent assay
High-binding half-area 96-well plates (Greiner BioOne) were coated with membrane protein extracts at 0.6 mg per plate. Plates were washed with cold PBS and blocked with 1% bovine serum albumin (BSA) in PBS. Plasma samples from healthy donors or patients were diluted 1/4 with 1% BSA in PBS. The dilution factor of 1/4 was determined from titration experiments to optimize the range and sensitivity of the assay. Forty microliters of diluted samples per well were loaded as duplicates onto fluorescence-linked immunosorbent assay (FLISA) plates. After 2-hour incubation and wash, 50 μL of secondary antibody solution, which contains 10 μg/mL Alexa 680–conjugated (Invitrogen) goat anti-human IgG, Fc specific (SouthernBiotech, Birmingham, AL) in PBS containing 1% BSA, was added to each well and incubated for 1 hour. Plates were washed, scanned, and analyzed on a Li-Cor Odyssey Imaging System (Lincoln, NE).
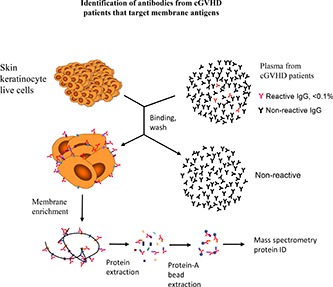
Cell-based immunoprecipitation and antigen identification
Forty million cells in single-cell suspension in 10 mL of PBS were incubated with 40 mL of pooled plasma from 4 cGVHD patients on a rotator for 3 hours at 4°C. Cells were washed 3 times with cold PBS and processed for membrane protein extraction as described earlier in the section. The IgG-antigen complexes in the freshly prepared membrane protein extracts were isolated with protein-A beads (RepliGen, Waltham, MA) (supplemental Figure 1, available on the Blood Web site). Protein-IgG complexes attached to the protein-A beads were eluted with 30 μL of 1× protein gel Laemmli Sample Buffer (Bio-Rad, Hercules, CA) and 5 minutes in boiling water. For initial immunoprecipitation confirmation, 2 μL of the eluted proteins were analyzed through capillary protein gel electrophoresis by loading onto a chip from the Protein 230 kit (Agilent Technologies, Santa Clara, CA) and analyzed on an Agilent 2100 Bioanalyzer. The remaining eluted proteins were loaded onto a 4% to 15% protein gel for gradient gel electrophoresis. The gel was stained with Brilliant Blue and stained gel was dissected into sections and sent to the Mass Spectrometry (Proteomics/Metabolomics) Core at Beth Israel Deaconess Medical Center for peptide identification.
Fluorescence-activated cell sorter–based fluorescent antigen trap bead assay
QuantumPlex beads with carboxyl (COOH) surface and internal allophycocyanin were purchased from Bangs Laboratories (Fishers, IN) and conjugated with antibodies for specific antigens, following the manufacturer’s instructions. The antibody-conjugated beads for different antigens were pooled, incubated with membrane protein extracts from CCD1106. After wash, the antigen-loaded beads were incubated with 50 μL of plasma 1/4 diluted with 1% BSA in PBS for 2 hours. The beads were washed and incubated with phycoerythrin (PE)-conjugated mouse anti-human IgG, Fc specific (BioLegend), and analyzed on the LSRFortessa (BD Biosciences Franklin Lakes, NJ) (supplemental Figure 2). The data were analyzed with FlowJo software (Ashland, OR).
Statistical analysis
Patient baseline characteristics and antibody reactivity data were analyzed primarily descriptively. Levels of antibody reactivity were compared using the Wilcoxon rank-sum test for group comparison. A high-level producer for antibody reactive against each antigen was defined as a value above the 95th percentile of healthy donor samples. All tests were 2-sided at the significance level of 0.05 and multiple comparisons were not considered. All statistical analyses were performed using SAS version 9.2 (SAS Institute, Cary, NC) and R version 3.1.3 (the CRAN project). All graphs were made using Prism (GraphPad, La Jolla, CA) or Microsoft Excel software.
Results
Increased anti-membrane antibodies in patients with cGVHD
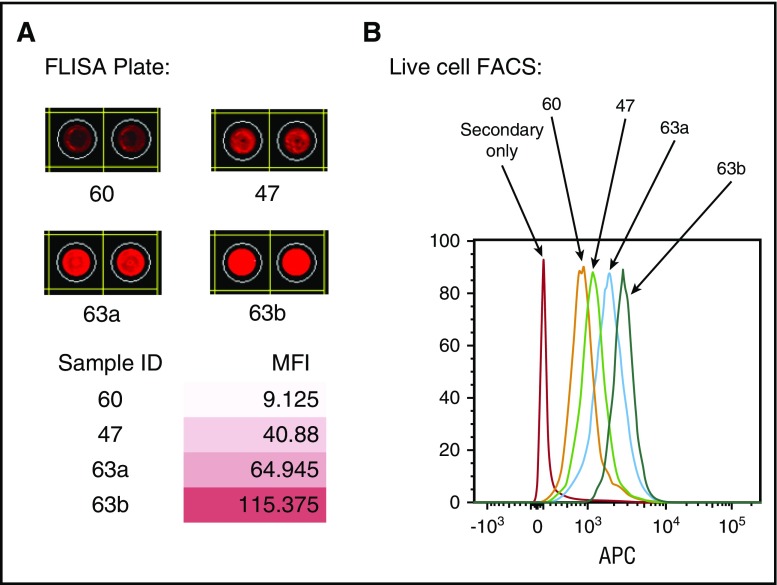
To detect antibodies that target membrane antigens, we developed a FLISA using membrane proteins prepared from different cell lines. To confirm that this assay detected antibodies reactive with membrane antigens on live cells, we compared results of FLISA with conventional flow cytometry. Figure 1 compares results using plasma samples from 4 different patients tested for reactivity against membrane proteins from Kasumi-1, an acute myeloblastic leukemia cell line. Antibody reactivity detected by FLISA (Figure 1A) shows different levels of binding to plate-bound antigens measured by mean fluorescence intensity. The level of FLISA reactivity correlated very closely with the level of antibody reactivity measured by flow cytometry using the same samples and target cells (Figure 1B).
Figure 1.
Comparison of antibody reactivities to plate-bound antigens detected by FLISA with antigens on live cells by FACS. (A) Antibody detection by FLISA in 4-patient samples against plate-bound membrane antigens from the AML cell line (Kasumi-1). (B) Antibody reactivities of the same patient samples against live cells (Kasumi-1) detected by FACS. APC, allophycocyanin; FACS, fluorescence-activated cell sorter; MFI, mean fluorescence intensity.
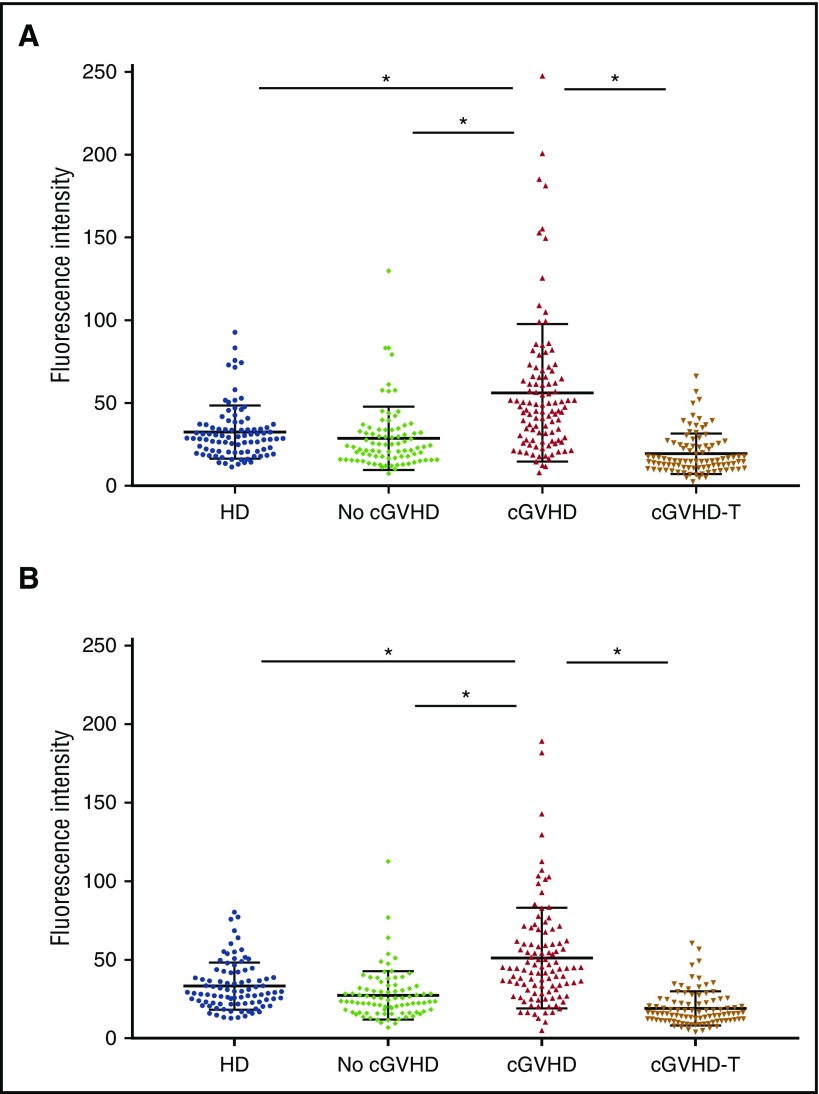
Because the skin is a common target organ in cGVHD, we used FLISA to detect antibodies against membrane proteins from a human skin keratinocyte cell line (CCD1106), as well as a human fibroblast cell line of skin origin (Detroit 551). Plasma samples from 284 allogeneic HSCT patients were studied (Table 1); 106 patients with active cGVHD, 93 patients with active cGVHD receiving intensive therapy (prednisone dose ≥20 mg/day, IL-2, or ECP), and 85 patients who did not develop cGVHD. In these groups, the median time from transplant to sample collection was 20 to 24 months. Although some samples in the 2 cGVHD groups were collected as early as 6 months after transplant, samples in the no cGVHD group were collected at least 12 months after transplant. Plasma samples from 90 healthy donors were also analyzed. Compared with patients who did not develop cGVHD and healthy donors, patients who developed cGVHD had significantly higher levels of IgG reactive with cell surface antigens from both skin keratinocytes and fibroblasts (cGVHD vs healthy donors, P < .0001; cGVHD vs no GVHD, P < .0001) whereas intensive treatment of cGVHD significantly reduced levels of these reactive antibodies (cGVHD vs cGVHD-T; P < .0001) (Figure 2). Total IgG levels were measured in 64% of no cGVHD patients, 79% of cGVHD patients, and 70% of cGVHD-T patients. We did not find a significant correlation between serum IgG and anti-membrane antibody reactivity in 84 patients in the cGVHD group (r = 0.2978 for IgG vs anti-skin keratinocyte antibodies and r = 0.2957 for IgG vs anti-skin fibroblast antibodies).
Figure 2.
Antibody reactivities against skin cell membrane antigens. Healthy donor (n = 90). No cGVHD indicates patients with no cGVHD (n = 85). cGVHD indicates patients with active cGVHD (n = 106) as described in “Materials and methods” cGVHD-T indicates patients with active cGVHD and receiving intensive treatments as described in “Materials and methods” (n = 93). *P < .0001. (A) Antibody reactivity was tested with FLISA using membrane proteins from the skin keratinocyte cell line (CCD1106) as antigens. (B) Antibody reactivity was tested with FLISA using membrane proteins from the skin fibroblast cell line (Detroit 551) as antigens. HD, healthy donor.
The development of membrane-reactive antibodies precedes clinical onset of cGVHD
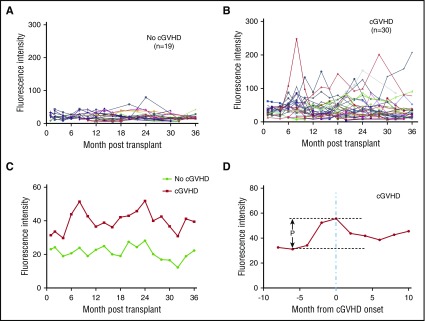
Previous studies in male recipients of stem cell transplants from female donors demonstrated that HY antibodies can be detected 3 months after transplant and precede the diagnosis of cGVHD.22 The development of anti-HY antibodies was significantly associated with the development of cGVHD.14 To examine the development of anti-membrane antibodies, we examined serial samples from 49 HSCT patients from the time of transplant to up to 3 years after transplant. Thirty patients in this cohort developed cGVHD during this time and 19 patients did not (Table 2). Although the level of anti-membrane antibodies varied in different patients (Figure 3A-B), patients who developed cGVHD had higher levels of anti-membrane antibodies throughout the 3-year period of sampling (Figure 3C). When the measurements of anti-membrane antibodies in patients with cGVHD were synchronized by the time of clinical diagnosis of cGVHD, we observed a significant increase in levels of anti-membrane antibodies beginning 4 months prior to diagnosis. Following diagnosis, the levels of anti-membrane antibodies began to recede, presumably reflecting a response to immune-suppressive therapy (Figure 3D).
Figure 3.
Antibody reactivities in HSCT patient plasma against skin keratinocyte antigens. Antibody reactivities in patient plasma were tested against membrane antigens from the skin keratinocyte cell line (CCD1106). (A) Antibody reactivities from individual patients who did not develop cGVHD after HSCT (n = 19). (B) Antibody reactivities from individual patients who developed cGVHD during the 3 years after HSCT (n = 30). (C) The mean fluorescence intensity from cGVHD group and No cGVHD group. (D) The mean fluorescence intensity of the cGVHD group after the time points of all patients within the group were synchronized to the time of cGVHD clinical onset. P = .03.
Anti-membrane antibodies target common cell surface antigens that are expressed on skin keratinocytes
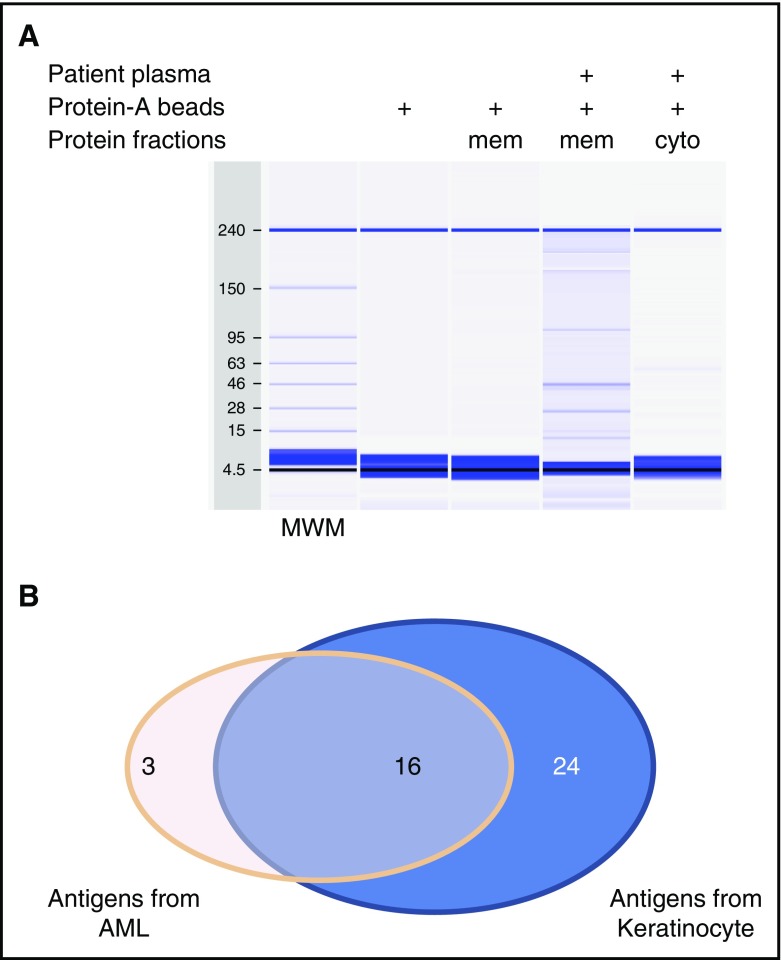
To identify specific antigens targeted by anti-membrane antibodies, we developed a method for immunoprecipitation using live cells. Pooled plasma from patients with cGVHD was incubated with either CCD1106 or Kasumi-1 cells to allow the binding of reactive antibodies to their target cell surface antigens. Cells, with antibodies bound to the surface, were solubilized and antibody-antigen complexes were isolated using protein-A beads (supplemental Figure 1). Protein gel capillary electrophoresis of these solubilized products shown in Figure 4A shows that complexes are only isolated when both cells and patient plasma were used. Moreover, complexes were only retained in the membrane extracts and were not present in the cytoplasmic fraction. Isolated proteins were separated by polyacrylamide gel electrophoresis and peptides extracted from the gel bands were identified using mass spectrometry. This approach identified 43 distinct membrane proteins, 40 using skin keratinocytes (CCD1106) as targets for cell-based immunoprecipitation, and 19 membrane proteins using AML cells (Kasumi-1). Sixteen antigens were shared among both groups (Figure 4B). All but 1 of the proteins (ANXA2) we identified are predicted to have plasma membrane localization (www.proteinatlas.org). These proteins are often expressed in other cellular compartments as well. Most of the proteins we identified also have tissue-staining images in the protein atlas database showing cell membrane staining. Seven are known CD antigens. ANXA2 is an intracellular protein that forms a heterodimer with other proteins that subsequently translocate to the cell surface.23
Figure 4.
Identification of targeted antigens by cell-based immunoprecipitation. (A) Analysis of the immunoprecipitation product with cGVHD patient plasma and keratinocyte cells by capillary protein gel electrophoresis. (B) Summary of the unique antigens identified when cell lines indicated were used. cyto, cytoplasmic fraction; mem, membrane fraction; MWM, molecular weight marker.
Antibody-targeted antigens are widely expressed on human tissues and cell lines
Examination of data from the Human Protein Atlas (www.proteinatlas.org) reveals that most of the identified membrane antigens are expressed at the protein level in multiple tissues often affected by cGVHD such as lung, liver, and skin, with only 3 exceptions due to the lack of available data (Figure 5B). Although most of these membrane proteins are expressed at high levels in the gastrointestinal tract, the majority are also expressed at medium or high levels in hematopoietic cells, lung, liver, and skin. Thus, antibodies generated in patients with cGVHD target a wide range of membrane antigens expressed on many tissues affected by cGVHD.
Figure 5.
Tissue protein expression of identified targeted antigens. Proteins are listed by their gene name. (A) The antigen source from which they were identified is highlighted by the green color. (B) The level of protein expression in human tissues for each gene is indicated by the height of the bars as high, medium, or low, based on data from Human Protein Atlas (www.proteinatlas.org). *Antigens selected for confirmation study. N/A, data not available.
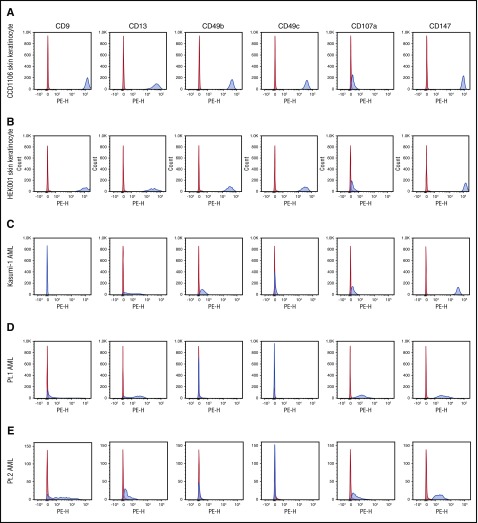
Based on the availability of monoclonal antibodies specific for membrane antigens in our panel, we focused validation studies on 6 of the 43 identified antigens. These antigens are CD9, CD13 (ANPEP), CD49b (ITGA2), CD49c (ITGA3), CD107a (LAMP1), and CD147 (BSG). All 6 antigens were identified using skin keratinocytes as the antigen source and 2 (CD107a and CD147) were also identified using AML cells as the antigen source (Figure 5A). To independently confirm the expression of these 6 target antigens on skin keratinocytes and AML cells, we used well-defined monoclonal antibodies and conventional flow cytometry to examine the expression of these antigens on 2 skin keratinocyte cell lines, the Kasumi AML cell line and circulating CD34+ blasts from 2 patients with AML. As shown in Figure 6A-B, both skin keratinocyte cell lines expressed all 6 antigens. CD107a was expressed at relatively low levels but the other 5 antigens were very highly expressed. Although data from the Human Protein Atlas show low expression of CD147 and no data for CD13 in skin tissue, our fluorescence-activated cell sorter (FACS) data showed that both antigens are highly expressed in skin keratinocytes. Expression of the 6 antigens on AML cells was more variable. However, at least 4 of the 6 antigens were expressed on the AML cell line and primary AML cells (Figure 6C-E), suggesting that AML cells could also be potential targets for these antibodies.
Figure 6.
Expression of targeted antigens on different cell types. The expression of the 6 antigens targeted by antibodies was examined by flow cytometry. The expression on 2 skin keratinocyte cell lines (A-B), 1 AML cell line (C), and CD34+ circulation AML cells from 2 patients (D-E) is shown. Red indicates isotype control–PE; blue, anti-antigen–PE. Pt., patient.
Validation of antibodies directed against 6 membrane antigens in patients with cGVHD
To confirm the production of antibodies against these 6 antigens, commercially available murine monoclonal antibodies specific for these antigens were used to develop FACS-based antigen-trapping bead assays. This assay was used to validate the presence of endogenous human antibodies to the same targets in patient samples. In this assay, fluorescent beads coated with murine monoclonal antibodies against specific antigens were incubated with solubilized membrane protein extracts from CCD1106 to allow binding of each antigen to specific beads. Antigen-loaded beads were then used as bait to detect human antibody in patient plasma (supplemental Figure 2). Using a panel of beads with different fluorescent markers, we could simultaneously detect antibodies against each of the 6 CD antigens in each patient sample. A total of 75 samples from transplant patients were analyzed, including 40 from patients with cGVHD and 35 patients without cGVHD. Thirty-eight samples from healthy donors were also tested.
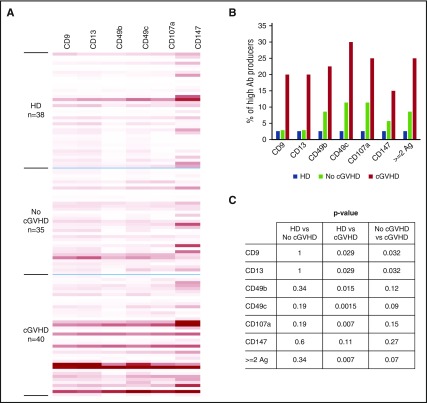
Results in Figure 7 summarize the detection of antibodies against all 6 antigens in stem cell transplant patients and healthy donors. It is apparent that antibodies directed against all 6 antigens are detected more frequently in patients with cGVHD compared with patients without cGVHD or healthy donors (Figure 7A). High-level producers for antibody reactive with CD9, CD13, CD49b, CD49c, and CD107a, defined by florescence intensity values for the specific antigen greater than the 95th percentile of healthy donor samples, were detected in 20% to 30% of patients with cGVHD (Figure 7B). Similar levels of antibodies to these same antigens were only seen in 2.9% to 11.4% of patients without cGVHD and 2.6% of healthy donors. Antibodies specific for CD147 were detected more frequently and at higher levels than antibodies to the other antigens in healthy donors, and this raised the threshold for identification of high-level producers. However, even with this increased threshold, there are more high producers in patients with cGVHD (15%) compared with healthy donors (2.6%) and patients without cGVHD (5.7%). The frequency of patients who are high-level producers of antibodies against >1 antigen is also significantly higher in cGVHD patients (25%) compared with healthy donors (2.6%, P = .007) and patients without cGVHD (8.6%, P = .07) (Figure 7A-B). Overall, 5 patients (12.5%) in the cGVHD group are high producers of antibodies against all 6 antigens, compared with 1 healthy donor (2.6%) and none of the patients (0%) without cGVHD.
Figure 7.
Antibody reactivity against 6 antigens. (A) Antibody reactivity in plasma of HSCT patients and healthy donors (HD) against the 6 antigens listed were tested with the FACS-based antigen-trapping bead assay. Fluorescence intensities are displayed as heatmap with Excel conditional formatting by equalizing the highest intensity of each antigen. (B) The percentages of high producers for each antigen. High producers were defined as having florescence intensity values for the specific antigen greater than the 95th percentile of HD. (C) The P values between groups in panel B for each antigen. Ab, antibody; Ag, antigen.
Discussion
In this study, we developed specific assays to detect antibodies that target membrane antigens after allogeneic HSCT. We found that levels of anti-membrane antibodies were significantly higher in patients with cGVHD and preceded the development of clinical symptoms. Our further investigation into the specific targets of these antibodies revealed 43 cell membrane targets and we confirmed that antibodies against 6 of these antigens are significantly increased in patients with cGVHD.
It is well documented that antibodies to alloantigens develop in patients with cGVHD and in animal models of cGVHD. In patients who receive HLA-matched stem cells, the best-characterized antibodies are directed against minor histocompatibility antigens such as HY antigens in male recipients who receive stem cell grafts from female donors.11,12,14,24,25 Antibodies to autosomal minor histocompatibility antigens have also been documented as well as antibodies against autoantigens commonly detected in patients with autoimmune diseases.19,26 In almost all cases, these antibodies are directed against intracellular proteins and the role of these antibodies as mediators of tissue injury is not known. In contrast, antibodies targeting membrane antigens may have a direct functional impact on live cells. However, except for antibodies specific for B-cell maturation antigen, platelet-derived growth factor receptor, and angiotensin type 1 receptor, few studies have identified anti-membrane antibodies in patients with cGVHD.27-29 The lack of antibodies targeting membrane antigens in patient samples is partly due to technical challenges. In widely used protein microarrays, membrane proteins are either presented in peptide format or expressed in nonmammalian expression systems. The resulting antigens presented on the arrays either lack tertiary structure or necessary posttranslational modifications, and are insufficient to capture antibodies that target native membrane proteins. With the techniques we have developed, we are starting to reveal the broad spectrum of anti-membrane antibodies that develop in patients with cGVHD.
Unlike anti-HY antibodies in patients who receive sex-mismatched stem cell transplants, the antigens we identified, especially the 6 antigens we validated, are common antigens expressed on various tissues and cells and are not known to be associated with genetic polymorphisms that distinguish donors and transplant recipients. Like antibodies against intracellular autoantigens that develop in patients with cGVHD, these anti-membrane antibodies target autoantigens and resemble the autoantibodies found in autoimmune diseases. In previous studies, it was shown that anti-HY antibodies are generated primarily de novo following sex-mismatched HSCT, and only the de novo development of HY antibodies after HSCT predicts cGVHD development.24,25 Patients with cytomegalovirus reactivation after transplant were more likely to develop antibodies against multiple HY antigens.14 These observations suggest that allogeneic immune responses posttransplant, or other immunological events such as viral reactivation, may lead to activation of a broader immune response and the generation of antibodies against autoantigens as well as alloantigens.
Recent studies in animal models and patients have shown that B cells play an important role in the development of cGVHD.30 B cells exhibit significant abnormalities during immune reconstitution including aberrant B-cell activation, reduced frequency of regulatory B cells, and higher proportion of memory B cells.31-34 Antibodies against both alloantigens and autoantigens are often detected after HSCT and deposition of alloantibodies on target tissues leads to the development of cGVHD in mouse models.11,15,17,19,35 Therapies that target B cells, such as anti-CD20 antibody rituximab and Bruton tyrosine kinase inhibitor ibrutinib, have proven to be effective in mouse models and clinically beneficial in human trials.36-40 These observations suggest that B cells play an important role in the development of cGVHD and that antibodies produced by B cells contribute to the immune pathology of cGVHD. We identified a wide range of membrane-expressed antigens that are targeted by antibodies generated in patients after HSCT, especially in patients who developed cGVHD. Moreover, these antigens are expressed on various tissues affected by cGVHD. Our observation that the generation of anti-membrane antibodies appears to increase 4 months prior to onset of clinical symptoms of cGVHD provides further evidence that the generation of these antibodies was associated with the development of cGVHD. The requirement of antibodies in the development of cGVHD and deposition of antibodies in tissues affected by cGVHD in murine models suggest that antibodies able to bind cell surface antigens on cells such as skin keratinocytes may contribute to the development of cGVHD in humans.
The development of cGVHD is also associated with the desirable graft-versus-leukemia (GVL) effect of stem cell transplantation.41-43 Notably, the majority of membrane antigens targeted in patients with cGVHD were shared between AML and skin keratinocytes. Antibody binding can lead to antibody-dependent cell-mediated cytotoxicity of targeted cells. With overlapping expression of target antigens on both AML cells and skin keratinocytes, these anti-membrane antibodies can potentially induce both cGVHD and GVL. In addition, binding of antibodies to their specific target may have specific functional consequences that may affect the survival, growth, or migration of leukemia cells. For example, CD9 is upregulated in B-cell acute lymphoblastic leukemia, glioblastomas, and other cancers. CD9 is involved in cell migration and its expression has been correlated with cancer metastasis.44-46 Anti-CD9 antibody was shown to inhibit tumor progression in mouse models.47 Similarly, blocking CD147 reduced tumor metastasis and prolonged survival in mouse models.48-52 CD49b, CD49c, and CD49f (ITGA6) are cell surface receptor integrins known to play important roles in controlling the organization of cell cytoskeletons, cancer cell migration, and invasion.53,54 Targeting these surface proteins has been shown to inhibit cancer metastasis.53-56 CD13 has also been investigated as a target for cancer therapy.57
These anti-membrane antibodies generated in cGVHD patients can not only target affected tissues such as skin keratinocytes but can also target leukemia cells, due to the overlapping expression of the targeted antigens on leukemia cells. In addition to T cells targeting antigens expressed on leukemia cells and normal tissues, these findings provide another potential mechanism by which cGVHD is associated with GVL. Further studies to investigate the full scope of the anti-membrane antibodies and characterization of the functional impact of these antibodies on disease tissues in cGVHD, as well as on cancer cells, will enhance our understanding of the role played by donor B cells and antibodies in the development of cGVHD, their impact on GVL, and provide new potential biomarkers for predicting cGVHD development as well as new potential strategies for cGVHD therapy.
Supplementary Material
The online version of this article contains a data supplement.
Acknowledgments
The authors thank Alexander Lee, Lauren Gaffny, and Doreen Hearsey in the Pasquarello Tissue Bank for their support in obtaining patient samples.
This work was supported by National Institutes of Health, National Cancer Institute grants P01CA142106, CA183559, and CA183560.
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: K.S.W. performed experiments; K.S.W. and J.R. designed the study, analyzed data, and wrote the manuscript; H.T.K. analyzed data; C.S.C., S.N., J.K., V.T.H., P.A., E.P.A., B.R.B., R.J.S., and J.H.A. provided samples and ideas for the study; A.T.H. assisted with experiments; and all authors contributed to editing and reviewed the manuscript.
Conflict-of-interest disclosure: J.K. received research funding from Prometheus Labs and Takeda Oncology, served on advisory boards for Kadmon Corp and Takeda Pharmaceuticals, and consulted for Amgen. The remaining authors declare no competing financial interests.
Correspondence: Jerome Ritz, Dana-Farber Cancer Institute, 450 Brookline Ave, Boston, MA 02215; e-mail: jerome_ritz@dfci.harvard.edu.
References
- 1.Wingard JR, Majhail NS, Brazauskas R, et al. Long-term survival and late deaths after allogeneic hematopoietic cell transplantation. J Clin Oncol. 2011;29(16):2230-2239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Martin PJ, Counts GW Jr, Appelbaum FR, et al. Life expectancy in patients surviving more than 5 years after hematopoietic cell transplantation. J Clin Oncol. 2010;28(6):1011-1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arai S, Jagasia M, Storer B, et al. Global and organ-specific chronic graft-versus-host disease severity according to the 2005 NIH Consensus Criteria. Blood. 2011;118(15):4242-4249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Socié G, Ritz J. Current issues in chronic graft-versus-host disease. Blood. 2014;124(3):374-384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cooke KR, Luznik L, Sarantopoulos S, et al. The biology of chronic graft-versus-host disease: a task force report from the National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease. Biol Blood Marrow Transplant. 2017;23(2):211-234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.MacDonald KP, Hill GR, Blazar BR. Chronic graft-versus-host disease: biological insights from preclinical and clinical studies. Blood. 2017;129(1):13-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang C, Todorov I, Zhang Z, et al. Donor CD4+ T and B cells in transplants induce chronic graft-versus-host disease with autoimmune manifestations. Blood. 2006;107(7):2993-3001. [DOI] [PubMed] [Google Scholar]
- 8.Blazar BR, Murphy WJ, Abedi M. Advances in graft-versus-host disease biology and therapy. Nat Rev Immunol. 2012;12(6):443-458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tivol E, Komorowski R, Drobyski WR. Emergent autoimmunity in graft-versus-host disease. Blood. 2005;105(12):4885-4891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Young JS, Wu T, Chen Y, et al. Donor B cells in transplants augment clonal expansion and survival of pathogenic CD4+ T cells that mediate autoimmune-like chronic graft-versus-host disease. J Immunol. 2012;189(1):222-233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Miklos DB, Kim HT, Miller KH, et al. Antibody responses to H-Y minor histocompatibility antigens correlate with chronic graft-versus-host disease and disease remission. Blood. 2005;105(7):2973-2978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Porcheray F, Miklos DB, Floyd BH, et al. Combined CD4 T-cell and antibody response to human minor histocompatibility antigen DBY after allogeneic stem-cell transplantation. Transplantation. 2011;92(3):359-365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zorn E, Miklos DB, Floyd BH, et al. Minor histocompatibility antigen DBY elicits a coordinated B and T cell response after allogeneic stem cell transplantation. J Exp Med. 2004;199(8):1133-1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nakasone H, Tian L, Sahaf B, et al. Allogeneic HY antibodies detected 3 months after female-to-male HCT predict chronic GVHD and nonrelapse mortality in humans. Blood. 2015;125(20):3193-3201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Srinivasan M, Flynn R, Price A, et al. Donor B-cell alloantibody deposition and germinal center formation are required for the development of murine chronic GVHD and bronchiolitis obliterans. Blood. 2012;119(6):1570-1580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cutler C, Kim HT, Bindra B, et al. Rituximab prophylaxis prevents corticosteroid-requiring chronic GVHD after allogeneic peripheral blood stem cell transplantation: results of a phase 2 trial. Blood. 2013;122(8):1510-1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jin H, Ni X, Deng R, et al. Antibodies from donor B cells perpetuate cutaneous chronic graft-versus-host disease in mice. Blood. 2016;127(18):2249-2260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pastano R, Dell’Agnola C, Bason C, et al. Antibodies against human cytomegalovirus late protein UL94 in the pathogenesis of scleroderma-like skin lesions in chronic graft-versus-host disease. Int Immunol. 2012;24(9):583-591. [DOI] [PubMed] [Google Scholar]
- 19.Quaranta S, Shulman H, Ahmed A, et al. Autoantibodies in human chronic graft-versus-host disease after hematopoietic cell transplantation. Clin Immunol. 1999;91(1):106-116. [DOI] [PubMed] [Google Scholar]
- 20.Simpson RJ. Disruption of cultured cells by nitrogen cavitation. Cold Spring Harb Protoc. 2010;2010(11):pdb.prot5513. [DOI] [PubMed] [Google Scholar]
- 21.Churchward MA, Butt RH, Lang JC, Hsu KK, Coorssen JR. Enhanced detergent extraction for analysis of membrane proteomes by two-dimensional gel electrophoresis. Proteome Sci. 2005;3(1):5-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sahaf B, Yang Y, Arai S, Herzenberg LA, Herzenberg LA, Miklos DB. H-Y antigen-binding B cells develop in male recipients of female hematopoietic cells and associate with chronic graft vs. host disease. Proc Natl Acad Sci USA. 2013;110(8):3005-3010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Deora AB, Kreitzer G, Jacovina AT, Hajjar KA. An annexin 2 phosphorylation switch mediates p11-dependent translocation of annexin 2 to the cell surface. J Biol Chem. 2004;279(42):43411-43418. [DOI] [PubMed] [Google Scholar]
- 24.Miklos DB, Kim HT, Zorn E, et al. Antibody response to DBY minor histocompatibility antigen is induced after allogeneic stem cell transplantation and in healthy female donors. Blood. 2004;103(1):353-359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nakasone H, Sahaf B, Tian L, et al. Center for International Blood and Marrow Transplantation. Presensitization to HY antigens in female donors prior to transplant is not associated with male recipient post-transplant HY antibody development nor with clinical outcomes. Haematologica. 2016;101(1):e30-e33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Patriarca F, Skert C, Sperotto A, et al. The development of autoantibodies after allogeneic stem cell transplantation is related with chronic graft-vs-host disease and immune recovery. Exp Hematol. 2006;34(3):389-396. [DOI] [PubMed] [Google Scholar]
- 27.Svegliati S, Olivieri A, Campelli N, et al. Stimulatory autoantibodies to PDGF receptor in patients with extensive chronic graft-versus-host disease. Blood. 2007;110(1):237-241. [DOI] [PubMed] [Google Scholar]
- 28.Chiron A, Bouaziz JD, Carmagnat M, et al. Anti-Angiotensin type 1 receptor antibodies in chronic graft-versus-host disease. Transplantation. 2014;98(4):470-474. [DOI] [PubMed] [Google Scholar]
- 29.Bellucci R, Alyea EP, Chiaretti S, et al. Graft-versus-tumor response in patients with multiple myeloma is associated with antibody response to BCMA, a plasma-cell membrane receptor. Blood. 2005;105(10):3945-3950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sarantopoulos S, Blazar BR, Cutler C, Ritz J. B cells in chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2015;21(1):16-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sarantopoulos S, Stevenson KE, Kim HT, et al. Altered B-cell homeostasis and excess BAFF in human chronic graft-versus-host disease. Blood. 2009;113(16):3865-3874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kuzmina Z, Krenn K, Petkov V, et al. CD19(+)CD21(low) B cells and patients at risk for NIH-defined chronic graft-versus-host disease with bronchiolitis obliterans syndrome. Blood. 2013;121(10):1886-1895. [DOI] [PubMed] [Google Scholar]
- 33.de Masson A, Socié G, Bagot M, Bensussan A, Bouaziz JD. Deficient regulatory B cells in human chronic graft-versus-host disease. OncoImmunology. 2015;4(7):e1016707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bohmann EM, Fehn U, Holler B, et al. Altered immune reconstitution of B and T cells precedes the onset of clinical symptoms of chronic graft-versus-host disease and is influenced by the type of onset. Ann Hematol. 2017;96(2):299-310. [DOI] [PubMed] [Google Scholar]
- 35.Kier P, Penner E, Bakos S, et al. Autoantibodies in chronic GVHD: high prevalence of antinucleolar antibodies. Bone Marrow Transplant. 1990;6(2):93-96. [PubMed] [Google Scholar]
- 36.Jacobson CA, Ritz J. B-cell-directed therapy for chronic graft-versus-host disease. Haematologica. 2010;95(11):1811-1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Miklos D, Cutler CS, Arora M, et al. Multicenter open-label phase 2 study of ibrutinib in chronic graft versus host disease (cGVHD) after failure of corticosteroids [abstract]. Blood. 2016;128(22). Abstract LBA-3. [Google Scholar]
- 38.Dubovsky JA, Flynn R, Du J, et al. Ibrutinib treatment ameliorates murine chronic graft-versus-host disease. J Clin Invest. 2014;124(11):4867-4876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Arai S, Pidala J, Pusic I, et al. A randomized phase II crossover study of imatinib or rituximab for cutaneous sclerosis after hematopoietic cell transplantation. Clin Cancer Res. 2016;22(2):319-327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ryan CE, Sahaf B, Logan AC, et al. Ibrutinib efficacy and tolerability in patients with relapsed chronic lymphocytic leukemia following allogeneic HCT. Blood. 2016;128(25):2899-2908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Horowitz MM, Gale RP, Sondel PM, et al. Graft-versus-leukemia reactions after bone marrow transplantation. Blood. 1990;75(3):555-562. [PubMed] [Google Scholar]
- 42.Baron F, Labopin M, Niederwieser D, et al. Impact of graft-versus-host disease after reduced-intensity conditioning allogeneic stem cell transplantation for acute myeloid leukemia: a report from the Acute Leukemia Working Party of the European group for blood and marrow transplantation. Leukemia. 2012;26(12):2462-2468. [DOI] [PubMed] [Google Scholar]
- 43.Storb R, Gyurkocza B, Storer BE, et al. Graft-versus-host disease and graft-versus-tumor effects after allogeneic hematopoietic cell transplantation. J Clin Oncol. 2013;31(12):1530-1538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Arnaud MP, Vallée A, Robert G, et al. CD9, a key actor in the dissemination of lymphoblastic leukemia, modulating CXCR4-mediated migration via RAC1 signaling. Blood. 2015;126(15):1802-1812. [DOI] [PubMed] [Google Scholar]
- 45.Huan J, Gao Y, Xu J, et al. Overexpression of CD9 correlates with tumor stage and lymph node metastasis in esophageal squamous cell carcinoma. Int J Clin Exp Pathol. 2015;8(3):3054-3061. [PMC free article] [PubMed] [Google Scholar]
- 46.Podergajs N, Motaln H, Rajčević U, et al. Transmembrane protein CD9 is glioblastoma biomarker, relevant for maintenance of glioblastoma stem cells. Oncotarget. 2016;7(1):593-609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nakamoto T, Murayama Y, Oritani K, et al. A novel therapeutic strategy with anti-CD9 antibody in gastric cancers. J Gastroenterol. 2009;44(9):889-896. [DOI] [PubMed] [Google Scholar]
- 48.Monteiro LS, Delgado ML, Ricardo S, et al. EMMPRIN expression in oral squamous cell carcinomas: correlation with tumor proliferation and patient survival. Biomed Res Int. 2014;2014:905680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yin H, Shao Y, Chen X. The effects of CD147 on the cell proliferation, apoptosis, invasion, and angiogenesis in glioma. Neurol Sci. 2017;38(1):129-136. [DOI] [PubMed] [Google Scholar]
- 50.Sato M, Nakai Y, Nakata W, et al. EMMPRIN promotes angiogenesis, proliferation, invasion and resistance to sunitinib in renal cell carcinoma, and its level predicts patient outcome. PLoS One. 2013;8(9):e74313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bovenzi CD, Hamilton J, Tassone P, et al. Prognostic indications of elevated MCT4 and CD147 across cancer types: a meta-analysis. Biomed Res Int. 2015;2015:242437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang Y, Yuan L, Yang XM, et al. A chimeric antibody targeting CD147 inhibits hepatocellular carcinoma cell motility via FAK-PI3K-Akt-Girdin signaling pathway. Clin Exp Metastasis. 2015;32(1):39-53. [DOI] [PubMed] [Google Scholar]
- 53.Melchiori A, Mortarini R, Carlone S, et al. The alpha 3 beta 1 integrin is involved in melanoma cell migration and invasion. Exp Cell Res. 1995;219(1):233-242. [DOI] [PubMed] [Google Scholar]
- 54.Seguin L, Desgrosellier JS, Weis SM, Cheresh DA. Integrins and cancer: regulators of cancer stemness, metastasis, and drug resistance. Trends Cell Biol. 2015;25(4):234-240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ali MRK, Wu Y, Tang Y, et al. Targeting cancer cell integrins using gold nanorods in photothermal therapy inhibits migration through affecting cytoskeletal proteins. Proc Natl Acad Sci USA. 2017;114(28):E5655-E5663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kwan BH, Zhu EF, Tzeng A, et al. Integrin-targeted cancer immunotherapy elicits protective adaptive immune responses. J Exp Med. 2017;214(6):1679-1690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Grieger E, Gresch G, Niesen J, et al. Efficient targeting of CD13 on cancer cells by the immunotoxin scFv13-ETA′ and the bispecific scFv [13xds16]. J Cancer Res Clin Oncol. 2017;143(11):2159-2170. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.