Figure 2. Intracellular Effects of Excess Cholesterol on Myeloid Cells and T Cells That Influence Atherosclerosis.
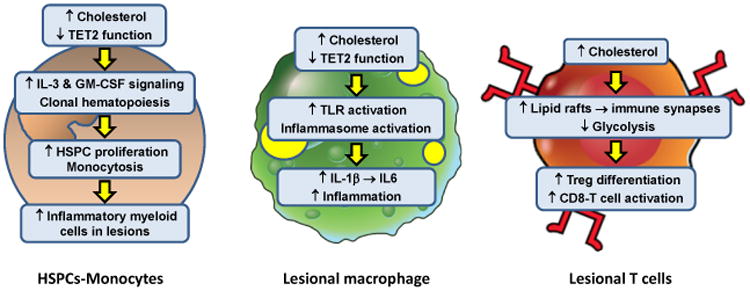
In the setting of hypercholesterolemia or defects in cholesterol efflux, hematopoietic stem and progenitor cells (HSPCs) accumulate excess cholesterol. The consequence is enhanced IL-3 and GM-CSF growth factor signaling, leading to HSPC proliferation and monocytosis. With aging, clonal hematopoiesis can occur owing to loss-of-function mutations in a number of genes, including TET2. This process also contributes to monocytosis. Monocytosis is associated with increased accumulation of inflammatory monocyte-derived macrophages in atherosclerotic lesions and higher risk of atherosclerotic vascular disease in humans. These lesional macrophages are also subject to intracellular cholesterol accumulation owing to their internalization of subendothelial apoB LPs. Excess cholesterol in macrophages has multiple effects that enhance lesion inflammation, including toll-like receptor (TLR) and inflammasome activation. The result is increased production of inflammatory chemokines and cytokines, including inflammasome-derived IL-1β and IL-1β-induced IL-6 production. Moreover, changes associated with clonal hematopoiesis, e.g., loss of TET2 function, can also activate the inflammasome in macrophages, further fueling lesional inflammation. T cells do not have the capacity to accumulate large amounts of excess cholesterol, but several studies have shown that perturbations of T cell cholesterol metabolism can affect T cell differentiation and activation. Impaired ABCG1 cholesterol efflux from T cells results in enhanced Treg differentiation which reduces atherosclerotic lesion development and inflammation. In contrast, impaired esterification of cholesterol by deficiency or inhibition of acyl-coenzyme A:cholesterol acyltransferase (ACAT) increases CD8 effector T cell lipid raft formation and thereby enhances immune synapse formation and killing functions of these cells. The net effect of increases in T cell cholesterol on lesion development and inflammation are likely to reflect changes in the Teff:Treg balance and the influence of Teff cells on lesional macrophages.
