Abstract
Gene therapy is a promising strategy for musculoskeletal tissue repair and regeneration where local and sustained expression of proteins and/or therapeutic nucleic acids can be achieved. However, the musculoskeletal tissues present unique engineering and biological challenges as recipients of genetic vectors. Targeting specific cell populations, regulating expression in vivo, and overcoming the harsh environment of damaged tissue accompany the general concerns of safety and efficacy common to all applications of gene therapy. In this review, we will first summarize these challenges and then discuss how biomaterial carriers for genetic vectors can address these issues. Second, we will review how limitations specific to given vectors further motivate the utility of biomaterial carriers. Finally, we will discuss how these concepts have been combined with tissue engineering strategies and approaches to improve the delivery of these vectors for musculoskeletal tissue regeneration.
Keywords: : gene therapy, musculoskeletal, biomaterials, controlled release
Introduction
Musculoskeletal (MS) tissue repair and regeneration remain a challenging, costly, and relatively underachieved therapeutic goal in healthcare despite recent medical advances. The MS tissues provide support, stability, and movement to our body, and given the number of critical roles these tissues provide in our daily life routines, it is easy to envision that MS injuries are among the most common causes of disability. The intrinsic healing capacity for MS tissues depends on the tissue type and the severity of the injury. While bone and muscle display reasonable capacity for tissue regeneration, cartilage lacks intrinsic regeneration regardless of the type of injury or insult. Traditional treatments for the MS tissues include autografts, allografts, and synthetic materials to replace damaged tissue, but these options are limited by integration, material supply, and donor-site morbidity. Tissue engineering strategies represent a clinically appealing alternative for guiding MS tissue regeneration, and gene delivery can be a tool to augment such strategies.
Tissue engineering is a multidisciplinary field that synergizes advances in medical, biological, and engineering sciences toward the development of biological substitutes to restore, maintain, or improve the functionality of mammalian tissues. MS tissue engineering strategies provide functions including the following: (1) deliver cells or soluble and insoluble bioactive cues to desired body sites; (2) provide mechanical support, while maintaining tissue shape and integrity; and (3) provide a template for conducting tissue regeneration and repair. Most strategies have focused on delivering instructive or inductive protein formulations. However, providing sufficient local protein concentrations over the timeframes necessary for enabling full MS tissue regeneration can be expensive and problematic. Promoting the production of these factors in situ by gene delivery circumvents these issues.
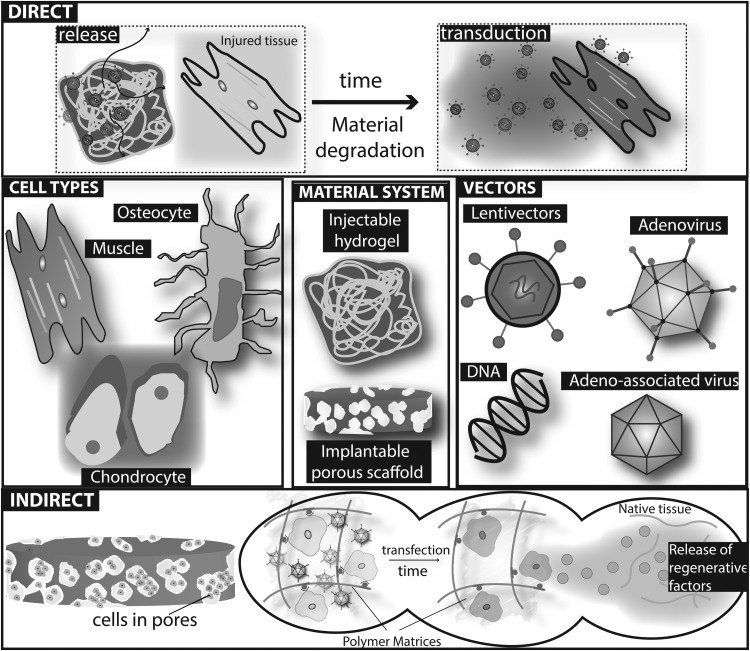
Gene therapy can yield sustained local production and secretion of proteins. This allows one to bypass the limitations of protein delivery related to short half-life and makes possible the regulation of bioactive cue timing at a tissue site. Accordingly, gene therapy is a promising avenue for MS regeneration, but important considerations remain for safely and efficiently delivering nucleic acids to MS tissues. To address this, several routes of administration, classes of vectors, and biomaterial carriers are in development (Fig. 1). In general, gene therapy can be performed (1) directly in vivo or (2) indirectly ex vivo. In the indirect approach, cells are modified or manipulated in vitro before being readministered to the patient. This approach holds the advantage of safely controlling the genetic modification of selected cell populations, but requires extensive cellular procurement and manipulation. In the direct approach, the technical complexity of handling cells is diminished, but there is also less control over cellular targeting and dosing. Therefore, the direct strategy requires engineering design of the vector and/or carrier to achieve cellular targeting and dosing of genetic payloads.
FIG. 1.
Schematic illustration of biomaterial approaches for gene delivery in musculoskeletal tissues. Nonviral or viral vectors can be incorporated into material systems for use in either direct in vivo or indirect ex vivo gene delivery.
This review will cover the combinatorial strategies of biomaterial carriers and gene delivery used for MS tissue repair and regeneration. The challenges of gene delivery to MS tissue will be briefly overviewed to provide a context in which these vectors and biomaterial carriers are being developed. The general strategies and forms in which biomaterials are used for gene delivery will be discussed. Following this, a few of the more prominent gene therapy vectors will be introduced, their limitations will be identified, and the strategies for using biomaterials to enhance their potential will be examined. Finally, gene therapy examples within representative tissues of the MS system will be discussed with specific emphasis given to applications of biomaterial-mediated gene delivery. In depth descriptions of the introduced vectors are outside the scope of this review. In a similar manner, the expansive literature on tissue engineering and gene therapy will be condensed to where these two fields meet for MS tissue applications.
Barriers for MS Gene Therapy
More than 1800 clinical protocols have been approved for human gene therapy.1 Small-scale trials have documented clinical success; however, no gene therapy has yet been approved by the FDA for clinical use. These disappointing results could be related with intrinsic biological barriers and engineering challenges of gene transfer.
Biological barriers to gene transfer
The introduction of foreign genetic payloads is interpreted as an abnormal situation by the cells and therefore the extracellular environment and cell machinery are designed to be resistant to gene transfer. The biological barriers to gene transfer can be summarized with four distinct challenges, including (1) reaching target cells, (2) achieving cellular internalization, (3) nuclear targeting, and (4) regulating gene expression. First, to reach target cells or tissues, genetic vectors need to bypass extracellular hurdles such as avoiding nonspecific uptake by nontarget cells or tissues and the recognition and/or degradation by the host immune system. While clearance is typically mediated by both renal (6–8 nm in hydrodynamic diameter [HD]) and biliary excretion, nonspecific uptake occurs by the cellular elements of the reticuloendothelial system. Vector size is an important parameter to ensure appropriate tissue targeting as it has been well recognized that colloidal particles >250 nm in HD are rapidly removed by the liver, lungs, and spleen.2 For example, the size of the viral vectors commonly used for MS tissue engineering strategies typically range from 29 to 166 nm (Table 1). In addition, the vector needs to survive protective mechanisms, including binding to the antigalactose antibodies present in human serum and exonuclease degradation.3 Second, after reaching target cells, the vector needs to cross the cell membrane. While viral vectors are highly effective at cellular uptake, nonviral vectors display poor efficiency to overcome this biological barrier.4 Third, nuclear targeting represents an essential condition for gene therapy and in the opposite of viral vectors, nonviral vectors strive to be transported efficiently to the cell nucleus where transcription occurs. Finally, the regulation of gene expression can be challenged by the inability to precisely control the integration of the transgene into the host genome, by gene silencing and the long-term regulation of transcription.
Table 1.
Vector Physical Characteristics
| Diameter (nm) | ||||||
|---|---|---|---|---|---|---|
| Vector | DLS | cryo-EM | Particle surface | ξ-potential (mV) | Diffusivity (μm2/s) | Affinities and interactions |
| Ad | 112159 | 9028 | Icosahedral capsid Nonenveloped Elongated fiber projections |
−18160 | 4.5161,a | Integrins—αvβ3, αvβ5, αvβ1, α3β1, α5β1CAR Heparan sulfate proteoglycan CD46 CD80 and CD86 Sialic acid162 |
| AAV | 29163 | 2641 | Icosahedral capsid Nonenveloped |
−9164 | 7.5165,b | Sialic acid Galactose Heparan sulfate proteoglycan Fibroblast growth factor receptor Hepatoctye growth factor receptor Laminin receptor166 |
| LV | 166167 | 12564 | Spherical Enveloped |
−18168,c | 3.6169,d | Low-density lipid receptor170 CD46 and SLAM receptor171 Poly-l-Lysine15,172 Chitosan13,88 Hydroxyapatite14,89,90 Phosphatidylserine173 |
| Plasmid | Radius of gyration (nm) bp 2686–6000 Linear 115–235 Supercoil 86–146174 |
NA | NA | bp 2686–9416 Linear ∼0.1–2 Supercoil ∼1–3174,e |
Cationic polymers Cationic lipids |
|
Photon-correlation spectroscopy.
Single molecule tracking.
MoMLV VSV-G.
Approximated with Stokes-Einstein equation
Fluorescence recovery after photobleaching.
AAV, adeno-associated virus; Ad, adenovirus; cryo-EM, cryo-electron microscopy; DLS, dynamic light scattering; LV, lentivirus; VSV-G, vesicular stomatitis virus glycoprotein.
Engineering challenges to gene transfer
Alongside the biological perspective, issues of low efficiency and variable gene expression can be viewed as engineering challenges to be addressed. These engineering challenges relate to the physical and chemical barriers that could influence the outcome of gene transfer. In general, these barriers are derived from the specific diffusion limitations of the vector of interest and/or from the physical and chemical characteristics of extracellular matrix (ECM). Indeed, the success of gene transfer depends on the rate and the extent to which vectors can move through the MS tissues until reaching the target cells. Increased transport heightens the risk of off-target effects, while too little transport can fail to cover sufficient tissue area. Vector transport through MS tissues is a complex process that is not readily modeled by simple mathematical considerations derived from Fick's law. Vectors will move toward locations with lower concentrations and this flux of vectors is directly proportional to the concentration gradient of vectors present locally, but transport in MS tissue is far from idealized. The physical size and spatial arrangement of extracellular proteins significantly contribute to the kinetics of vector diffusion.5,6 The physical and chemical characteristics of ECM within the MS tissues are highly diverse, and the rates of diffusive transport of vectors could vary dramatically among these tissues. For example, gene transfer with DNA in skeletal muscle tends to be relatively confined (i.e., only 100 fibers or less are transfected per injection site).7 In a merely engineering and more reductionist view, the ECM can be represented as a hydrogel of proteins and polysaccharides. The meshwork of the ECM provides strong resistance to the flux of vectors and it is essentially due to a combination of steric (i.e., size-selective molecular sieves dictated by the three-dimensional fiber mesh of the ECM) and hydrodynamic interactions between the vectors and the ECM. Therefore, the development of mathematical models could be extremely useful to predict the flux of vectors for a given concentration gradient and type of ECM and to better design the appropriate delivery strategy to enhance vector transfer.8
Biomaterial Carriers of Genetic Vectors
Central to most MS tissue engineering strategies, and emerging in gene delivery strategies, is the incorporation of a biomaterial system. These biomaterials provide a combination of mechanical structure, cellular support, and biochemical signaling control. To achieve this, biomaterial constructs are designed to mimic the structures of native ECM, which have evolved to provide favorable combinations of cellular receptors, mechanical signals, and presentation of growth factors and cytokines. The ECM varies considerably between the myriad MS tissue types, and to recapitulate these forms, various biomaterial constructs have been developed. For example, mechanically stiff materials, such as hydroxyapatite, and sturdy three-dimensional porous scaffolds are suitable for bone regenerative applications.9 Conversely, the hydrated polymer meshwork of hydrogels becomes suitable for the treatment of soft tissues such as muscle.10
Additional design consideration becomes important when these tissue engineering strategies are extended to gene delivery, and will vary between two broad classifications of gene delivery route: indirect and direct delivery. These routes are delineated by whether cells are introduced to genetic vectors ex vivo or in vivo, respectively. In this study, the appropriate design will employ different levels of two important parameters: cellular adhesivity and vector retention. The adhesion of cells to a biomaterial is mediated by interactions between cell surface receptors and specific amino acid sequences presented by the polymer. These sequences are either naturally present on the polymer or can be coupled through chemical modification.11 Vector retention is directly correlated with the outward vector transport from the polymer system. Analogous to transport in tissue, the vector size, steric hindrances, and affinity interactions will serve to slow or quicken release from the polymer system. Each class of vector contains intrinsic affinities for specific proteins, sugars, and proteoglycans involved in their cellular uptake.12 In addition, nonspecific interactions with charged polymers and particles have also been described.13–15 These affinities can be harnessed to promote vector interaction with a biomaterial carrier. Alternatively, vectors can be modified to promote interactions or covalent binding with a biomaterial to restrict the release of the vector.16–18
Indirect gene delivery strategies aim to provide a robust environment for a given cell type and a confining volume for a genetic vector. To isolate gene delivery to the seeded cells, these constructs are engineered to support cellular adhesion and maximize the retention of the genetic vector used. Direct delivery constructs encapsulate vectors within a carrier and can be engineered to either (1) deliver genetic material to cells that infiltrate the construct in vivo, or (2) deliver genetic material to cells in the immediate vicinity of the construct. Accordingly, these approaches can vary from cell-supportive and vector-retaining to cell-neutral and vector-releasing. In the latter approach, the spatiotemporal control in release becomes critical to providing safe and efficient doses to nearby cells.
For both indirect and direct gene delivery strategies, a variety of naturally occurring and synthetic polymers have been successfully used as delivery vehicles. Naturally occurring polymers are isolated from plant, animal, or human tissues. Often, delivery vehicles formulated from these polymers have the advantage of recapitulating key structural and/or biochemical properties of the ECM due to their natural derivation. Examples of such polymers include fibrin19 and collagen.20 However, some naturally occurring polymers are not inherently recognized by the human body, but can be easily modified with some adhesive ligands, such as the arginine-glycine-aspartic acid (RGD) peptide, to obtain cellular interactivity. Examples of these polymers include alginate21 and chitosan.22 In general, naturally occurring polymers hold the advantages of low immunogenicity, relative abundance, and ease of processing. The formation of hydrogels and scaffolds can be carried out in gentle manners allowing for the encapsulation of more sensitive genetic vectors and cells. In contrast to delivery vehicles fabricated from natural sources, several other promising polymers are obtained through chemical synthesis. These synthetic polymers hold the distinct advantage of reproducible and controlled production with the option for precise manipulations. Typically, these polymers do not have inherent cellular recognition, but these characteristics can be added through chemical modification. Commonly used synthetic polymers for gene therapy include poly(ethylene glycol) (PEG),23 poly(lactic-co-glycolic) acid (PLGA),24 and polycaprolactone (PCL).25
Types of Vectors and Associated Biomaterial Carriers
The choice of gene delivery vector for MS tissue repair and regeneration is an important parameter. This selection must consider several variables, including the cellular target, the efficiency of delivery, the required duration of expression, toxicity, immunotolerance, and safety. Each choice of vector has inherent strengths and weaknesses in these categories, but the incorporation of a biomaterial carrier can provide additional levels of design to bolster deficiencies.
Numerous vectors have been tested for gene therapy, including viral and nonviral vectors. In general, viral vectors are highly effective for gene transduction, but carry potential safety issues. On the other hand, nonviral vectors attract lower safety concerns, but display poor or low gene transfection. Viral vectors differ in their size, morphology, and surface characteristics and display varied diffusion characteristics in solution or tissue environments (Table 1).
With appropriate consideration of vector and tissue-target characteristics, some of the inherent limitations of each vector can be addressed by incorporating a carrier system. These carriers serve to localize and concentrate vectors within a localized area, while simultaneously providing protection against inactivation by the host immune response. This approach provides increased transduction and transfection efficiencies with lower doses and minimized off-target effects. Furthermore, these systems can be tailored for either ex vivo (indirect) or in vivo (direct) gene delivery. These approaches are overviewed in the following sections in the context of commonly used vectors (Table 2).
Table 2.
Carrier Designs for Gene Delivery Vectors
| Vector | Biomaterial | Physical formulation | Reference | |
|---|---|---|---|---|
| Ad | Naturally occurring | Agarose | Hydrogel | 175 |
| Alginate | Hydrogel | 175,36 | ||
| Chitosan | Microsphere | 36,176,177 | ||
| Collagen | Sponge | 150,178 | ||
| Fibrin | Scaffold | 150,179,180 | ||
| Gelatin | Hydrogel | 175 | ||
| Sponge | 181 | |||
| Synthetic | PEG | Coating | 160,182–186 | |
| PCL/PEG | Scaffold | 187 | ||
| PLGA/PEG | Microsphere | 188 | ||
| AAV | Naturally occurring | Fibrin | Hydrogel | 189,190 |
| Gelatin | Sponge | 156 | ||
| Heparin | Nanoparticle | 191 | ||
| Polypeptide | Hydrogel | 192 | ||
| Synthetic | Iron oxide | Nanoparticles | 191 | |
| PCL | Scaffold | 116,118 | ||
| PEI | Surface patterning | 193,194 | ||
| PEG | Hydrogel | 195,196 | ||
| PEO/PPO | Micelles | 197 | ||
| PLGA | Scaffold | 156 | ||
| Polystyrene | Microparticle | 198 | ||
| LV | Naturally occurring | Alginate | Hydrogel | 87 |
| Microsphere | 199 | |||
| Collagen | Hydrogel | 14 | ||
| Fibrin | Hydrogel | 157,159,200 | ||
| Polypeptide | Hydrogel | 201 | ||
| Synthetic | PCL | Scaffold | 15 | |
| PEG | Hydrogel | 88,172,202,203 | ||
| Coating | 167 | |||
| PLGA | Scaffold | 13,14,89,90,173,204,205 | ||
| Polyester | Scaffold | 206 | ||
PCL, polycaprolactone; PEG, poly(ethylene glycol); PEO, poly(ethylene oxide); PPO, poly(propylene oxide); PLGA, poly(lactic-co-glycolic) acid.
Adenovirus
Adenovirus (Ad) is a species from the Adenoviridae family consisting of a double-stranded DNA genome of 36 kilobases (kb).26,27 The virus is a nonenveloped, icosahedral, 90 nm diameter particle encapsulating an inner nucleoprotein core.27,28 The most studied serotype is Ad5, but 7 subgroups and 57 serotypes have been identified.27,29 Ad5 infects cells through a fiber knob that binds the cellular receptor CAR (coxsackie and adenovirus receptor). Subsequently, integrins present in the cell membrane interact with the arginine-glycine-aspartate (RGD) peptide motif in the penton base (the capsid protein at the base of the fiber) and facilitate cell entry by endocytosis.30
As a gene therapy vector, Ad has the ability to transduce a variety of dividing and nondividing cells with strong, but transient, gene expression.12 Ad vectors remain the most prevalent vectors in clinical trials.1,31 There are 521 approved clinical trials for Ad, predominantly for cancer treatment (70%), with two protocols currently in phase IV.31 In 2003, Ad formed the basis for the first therapeutic gene therapy medicine approved for commercialization in China. The Shenzhen SiBiono GenTech developed the Geneticine, a modified Ad vector encoding the p53 tumor suppressor gene, which gained approval for treating head and neck cancer.32,33 The results involving this therapy are still controversial due to a lack of available information on the clinical outcomes.32
Despite its prevalence in clinical trials, Ad has a few key limitations: (1) the activation of a strong immune response by proteins present in the viral capsid34; (2) the dependence of the CAR receptor to transduce host cells, which can lead to preferential transduction of liver cells34; and (3) a limited in vivo biological activity over time.35 These limitations motivated some of the earliest attempts to incorporate biomaterial carriers with viral vectors.36 A variety of strategies for minimizing immunogenicity and controlling local concentrations have been implemented with biomaterials derived from natural and synthetic sources; prominent among these approaches is the masking of Ad from the host immune response through surface coatings and/or hydrogel encapsulation.
Adeno-associated virus
Adeno-associated virus (AAV) was first discovered in 1965 following observations of “virus-like” particle contaminants in Ad stocks.37,38 These contaminants proved to be a poorly replicating member of the Parvoviridae family that required Ad as a coinfecting helper for replication. AAV is a small simple virus composed of a 4.7-kb single-stranded DNA genome carried in a nonenveloped, 26 nm, icosahedral protein capsid.39–41 The AAV genome comprised two open reading frames, rep and cap, flanked on each end by inverted terminal repeat sequences (ITR). Together, rep and cap produce the structural and nonstructural proteins necessary for viral replication, transcription, integration, assembly, and nuclear localization.42–47 For recombinant AAV, a gene of interest is inserted between the ITRs in place of the rep and cap genes, which are provided during production as separate constructs. DNA delivered by rAAV exists in a predominantly episomal form with low frequencies of chromosomal integration.48–51
As a gene therapy vector, AAV has many distinct advantages that have led to its use for in vivo gene delivery in a growing number of clinical trials. AAV is nonpathogenic and due to high episomal stability, can achieve long-term expression in nondividing somatic cells with low risk of insertional mutagenesis.52–55 Twelve serotypes and several variations occur naturally, which result in wide tissue tropisms.56 Pseudotyped vectors have been developed by integrating the prototypic AAV2 genome with a variety of capsids from different serotypes. Furthermore, the diversity of capsid proteins has been expanded using natural and artificial evolution, allowing a number of specific cells and tissues to be targeted.57–59 There are currently 162 clinical trials approved for AAV, with 10 protocols in phase III.31
While these characteristics of AAV vectors have spurred research and development, several key hurdles remain to be addressed. AAV is widespread among human population and a majority of people harbor neutralizing antibodies from prior natural exposure.60 Furthermore, exposure to AAV therapy can lead to the buildup of anti-AAV antibodies, which can lower the efficiency of readministrations if they are required.61 A few approaches for using biomaterials to address limitations in AAV delivery have recently been investigated. These strategies rely on controlled release from hydrogels, scaffolds, and nanoparticles–microparticles.62
Lentivirus
Lentivirus (LV) is a genus of the viral family Retroviridae, derived from the human immunodeficiency virus, which possesses a diploid (+) RNA genome of ∼9 kb with a particle diameter of 125 nm.12,26,63,64 Three important modifications have increased the safety profile of the current generation of LVs. (1) A nonpathogenic and replication-deficient lentivector was generated by removing the viral genes gag, pol, env, and rev from the LV genome. During packaging, these sequences are provided to human embryonic kidney 293T cells as separate constructs.65–67 (2) The infectivity spectrum was increased by pseudotyping with vesicular stomatitis virus glycoprotein (VSV-G) as an envelope. (3) Self-inactivating (SIN) vectors were developed by deleting the enhancer/promoter sequences of the 3′ long terminal repeat.68,69
Several features of lentivectors make them attractive for gene therapy. Lentivectors can transduce quiescent cells,70,71 harbor an expression cassette capacity up to 8 kb, elicit a minimal immune response, and produce long-term expression of transgenes.33 They have been widely used in gene therapy preclinical protocols to express genes and shRNAs to treat genetic diseases, including mucopolysaccharidosis type I,72,73 Duchenne Muscular Dystrophy,74,75 severe combined immune deficiencies,76,77 and also to treat others diseases such as cancer,78,79 peripheral artery disease,80,81 and Alzheimer's disease.82 There are currently 135 clinical trials using lentivectors, including three in phase III to treat thalassemia Major and X-linked adrenoleukodystrophy.31
Limitations of LV in gene therapy center on safety concerns associated with DNA integration and gene expression timing due to silencing. Vector integration poses the risk of disturbing active genes83,84 or activating proto-oncogenic genes.69 LV integration has also been related with histone modifications, including H3 acetylation, H4 acetylation, and H3K4 methylation.85 This contributes to the silencing of gene expression using lentivectors, which presents an obstacle to obtaining long-term therapeutic benefit.86 The safety concerns of LV have motivated the use of biomaterial carriers to control the spatiotemporal presentation of these vectors. For example, we recently demonstrated sustained murine hindlimb GFP expression achieved with a reduced viral insert copy number.87 Separate studies have pursued gene delivery within hydrogel and scaffold carriers. As such, modifications with polycationic polymers13,88 and hydroxyapatite14,89,90 have been incorporated into these hydrogels and scaffolds to increase retention of LV within the carrier.
Nonviral
Plasmid DNA (pDNA) is a circular, double-stranded DNA that replicates independent of chromosomal DNA. pDNA can be easily engineered to express one gene or multiple genes of interest depending on the specific application. pDNA offers several advantages over viral delivery systems, including the ability of plasmids to accommodate large genes, such as the 14-kb dystrophin Cdna,91 the relative ease and lower cost of large-scale production and storage, the more amenable pharmaceutical formulation, and lower toxicity.92,93 There are currently 422 clinical trials using pDNA to treat several diseases, including 14 phase III trials (www.wiley.com/legacy/wileychi/genmed/clinical).
pDNA presents some limitations, including low transduction efficiency and short expression of the transgene. pDNA has an anionic charge and is electrostatically repelled from the cell surface, which reduces cellular uptake. Furthermore, the transgene product usually declines to very low or undetectable levels in a period of days, even though the pDNA is not lost. In this study, DNA silencing has been attributed to the presence of CpG islands.94,95 To address these problems, different types of pDNA have been developed such as minicircles96–99 and nonviral integrative vectors such as Sleeping Beauty Transposon and phiC31 integrase.100
Due to the low efficiency of cellular uptake, a large body of research has emerged using physical methods such as electroporation and sonoporation associated or not with biomaterial strategies for nonviral gene delivery. Electroporation is the use of electrical field to mediate gene transfer and sonoporation is the formation of small pores in cell membranes by using ultrasound for the transfer of nucleic acid materials.101,102 Several studies have combined biomaterials such as gold nanoparticles, fibrin, collagen, and PLGA with physical methods to treat MS injuries.103–108 Other methods of gene delivery, including lipoplexes, polyplexes, and lipid–polymer systems, are outside the scope of this review, but have been thoroughly described in several excellent reviews covering the myriad forms of pDNA vehicles.109–111
Biomaterial-Guided Gene Delivery to MS Tissue
Bone
Bone is a tissue that provides a versatile and critical function. In terms of mechanics, bone is involved in skeletal stability, locomotion, and the protection of internal organs. Bone also serves as a mineral reservoir and harbors a large portion of the bodies' bone marrow. Unsurprisingly, bone is a dynamic, highly vascularized complex tissue with a unique ability to regenerate and heal. This regenerative ability is critical as fractures are the most common large-organ traumatic injury in humans. Separate informative reviews have extensively covered the healing process of bone.112 This process is sufficient to properly heal roughly 90% of fractures that occur, and does so with minimal scarring. However, ∼10% of fractures result in nonunion, wherein the natural regenerative mechanisms are insufficient, and full healing does not occur. This insufficient healing is compounded in anatomical locations with low blood supply such as the scaphoid and radius. Currently, autologous bone grafting represents the gold standard for treatment of nonunion fractures. However, this treatment is subject to donor-site morbidity and a limited supply of autologous bone. Bone tissue engineering and gene therapy are emerging as potential alternative strategies.113,114
Tissue engineering scaffolds are promising alternatives to bone grafts as they can provide mechanical strength and a matrix for cell remodeling and growth factor delivery. These scaffolds can also serve as carriers for genetic vectors. The concept of a gene-activated matrix (GAM) was first introduced for bone regeneration and was a pivotal point for the use of carriers for gene delivery. In its original description, the GAM consisted of a collagen matrix with immobilized pDNA encoding for bone morphogenetic protein-2 (BMP-2) and represented an in vivo approach capable of promoting bone formation.115 This concept has now been expanded to include new materials and alternative vectors, and have employed both ex vivo and in vivo approaches (Table 3). For example, PCL scaffolds coated with a self-complimentary AAV encoding BMP-2 demonstrated an ability to promote bone formation in a critical-sized rat femoral defect.116 This study employed both an ex vivo approach, with mesenchymal stem cells (MSCs) preseeded within the scaffold, and an in vivo approach, with a cell-free scaffold. Interestingly, the cell-free constructs outperformed the preseeded constructs in terms of bone bridging, ingrowth, and ultimate mechanical integrity. GAMs can also be used to deliver multiple genes in vivo from a single matrix. In this study, a collagen scaffold modified with nano-hydroxyapatite demonstrated how dual delivery of vascular endothelial growth factor (VEGF) and BMP-2 pDNA can synergistically promote bone regeneration.117
Table 3.
Representative Examples of Biomaterial-Guided Gene Delivery to Musculoskeletal Tissue
| Gene therapy | Vector | Gene | Biomaterials | References | |
|---|---|---|---|---|---|
| Bone | Ex vivo | AAV | BMP-2 | PCL | 116 |
| Ad | VEGF | Chitosan | 178 | ||
| Ad | BMP-2 | PEG-DA | 207 | ||
| In vivo | AAV | BMP-2 | PCL | 116 | |
| Ad | PDGF | Collagen | 180 | ||
| Ad | BMP-2 | Collagen | 179 | ||
| Plasmid | VEGF/BMP-2 | Collagen HA | 117 | ||
| In vitro | AAV | BMP-2/IGF | Chitosan | 208 | |
| Muscle | Ex vivo | Plasmid | VEGF-165/SDF-1 | Collagen | 131 |
| In vivo | AAV | VEGF165 | Collagen-GAG | 209 | |
| Ad | BMP-7 | Fibrin, collagen | 133 | ||
| LV | Luciferase | Alginate | 126 | ||
| Plasmid | FGF4 | GHG | 132 | ||
| In vitro | LV | RNAi for integrin α7 | PDO | 210 | |
| Cartilage | Ex vivo | Plasmid | BMP-2 | Fibrin glue | 211 |
| Plasmid | BMP-4 | PLLGA | 212 | ||
| Plasmid | TGF-β1 | PLGA/fibrin | 142 | ||
| Plasmid | TGF-β1 | PLGA/fibrin | 140 | ||
| In vivo | No results | No results | No results | No results | |
| In vitro | LV | TGF-β3 | PCL | 15 | |
| LV | IL-1Ra | PCL | 143 | ||
| Plasmid | TGF-β1 | HA/chitosan | 142 | ||
| Plasmid | TGF-β3 | Alginate | 213 | ||
| Plasmid | TGF-β1 | Chitosan | 214 | ||
| Plasmid | BMP-6 | Chitosan | 215 | ||
| Ligament, tendon, and intervertebral disc | Ex vivo | Ad | TGF-β1 | Chitosan/collagen | 150 |
| LV | Luciferase | PEG-LM111 | 155 | ||
| Plasmid | PDGF-B | Chitosan/coral | 216 | ||
| Plasmid | BMP-2 | Alginate | 217 | ||
| Plasmid | BMP-2/Smad8 | Collagen | 151 | ||
| In vivo | Plasmid | RNAi for TGF-β1 | PLGA | 152 | |
| Plasmid | RNAi for TGF-β1 | PLGA | 153 | ||
| In vitro | LV | TGF-β3 | CHS | 108 | |
| Plasmid | BMP-2 | Chitosan | 218 | ||
| Plasmid | BMP-2 | PEI/PLGA | 219 | ||
| Plasmid | PDGF | Chitosan/collagen | 149 | ||
| Plasmid | Luciferase | Hyaluronan-PAMAM | 154 |
CHS, chondroitin sulfate-hyaluronate acid-silk fibroin; HA, hyaluronic acid; PAMAM, polyamidoamine.
The combination of biomaterials and physical methods of gene delivery such as electroporation and sonoporation has also been studied for bone reconstruction. Collagen sponge has been used as a matrix for cells electroporated with BMP-9 to induce fracture repair.105 PLGA scaffold was used successfully for differentiation of adipose stem cells electroporated with osteogenic genes.118 The high efficiency of transfection produced by physical methods combined with scaffolds may be an alternative for bone grafts.
Muscle
The U.S. healthcare system spends $150 billion to treat >20 million muscle injuries such as sprains, fractures, and contusions each year.119 Muscle injuries can be caused by direct trauma such as lacerations, contusions, and strains or by indirect trauma that results from medical conditions such as ischemia, genetic disorders, or neurologic dysfunction.120 Although muscles can undergo regeneration after injury, the healing process is slow and often culminates in incomplete functional recovery and fibrosis.121 The formation of dense scar tissue can impair muscle function and lead to muscle contracture and chronic pain.121 Despite the considerable medical need, there has been relatively little progress in the development of therapeutic approaches to enhance healing following muscle injury. The treatments for muscle contusions are based on conventional therapies such as RICE (rest, ice, compression, and elevation), immobilization, anti-inflammatories, and physiotherapy.121,122 Nonetheless, these methods have a lower efficacy for prevention or treatment of complications such as atrophy, contracture, pain followed by functional loss, and fibrosis.123,124
The gold standard method for severe injuries, particularly laceration, is the suture of the muscle, which can reduce the formation of dense scar tissue and help in the recovery of muscle strength.125 However, this therapy does not provide complete injury regeneration and a new treatment alternative is needed. Gene and cellular therapies, or the combination of both, appear as the main future therapies for muscle injury treatment.126
Biomaterials have been used in muscle injuries for cell and growth factor delivery.127 These biomaterials increase the survival and migration of the cells after transplantation in the injured muscle.128 In cell-free approaches, complete functional recovery of ischemic injured skeletal muscle has been reported using alginate hydrogels to deliver VEGF and insulin-like growth factor (IGF).129 However, the delivery of proteins relies on large, expensive bolus doses even with controlled release strategies. Gene delivery using these same biomaterial approaches has seen some utility for muscle regeneration, but the examples are limited (Table 3). In one ex vivo approach, a collagen hydrogel was used to deliver VEGF- and SDF-1-transfected myoblasts and led to vascularization of muscle defects in rats.130 In vivo approaches are also under early investigation. Biodegradable gelatin hydrogels were shown to prevent degradation of pDNA encoding for fibroblast growth factor-4 (FGF4) and relieved muscle necrosis in a rabbit hindlimb ischemia model.131 Hydrogels have also shown potential for protecting and extending expression within muscle tissue for Ad and LV vectors, although these studies have not yet addressed regenerative applications.87,132
Articular cartilage
Articular cartilage (AC) is an avascular connective tissue formed by a sparse population of chondrocytes, which is frequently injured as a result of trauma (joint dislocation, ligament tear, meniscus tear, and fall/impact), infection, and degenerative joint diseases such as osteoarthritis.133,134 The AC has a low capacity to self-repair due to slow cell turnover and minimal vascular supply.133,134 Current treatments are limited, according with the severity of the symptoms. Typically, the clinical treatments for pain rely on opioids, analgesics, and nonsteroidal inflammatory drugs (NSAIDs) combined with physical therapy and/or exercise. The viscosupplementation using hyaluronic acid (HA) or collagen is also an option for pain management and repair. The last option is surgical intervention that often fails or causes further tissue injury.135–137
Gene therapy is emerging as a promising alternative to the current treatment options for cartilage.138 The dense matrix present in cartilage has made direct in vivo gene delivery inefficient as poor vector transport becomes a barrier. This motivates the use of indirect ex vivo approaches for gene delivery to cartilage. These procedures have predominantly used porous sponges and hydrogels for cellular encapsulation.139 These approaches can also be combined. For example, PLGA porous sponges and fibrin hydrogels are used simultaneously to encapsulate MSCs and pDNA complexes encoding for transforming growth factor (TGF)-β1.140,141 Several promising in vitro examples are also in development for delivery of pDNA and LV. Chitosan scaffolds embedded with hybrid HA/chitosan(CS)/pDNA nanoparticles were used to transfect (TGF)-β1 with sustained pDNA release and expression for over 120 days.142 Recently, PCL woven scaffolds, which can be shaped to fit anatomical structures, were loaded with poly-l-lysine-immobilized LV and seeded with MSCs. The controlled production of TGF-β3 or interleukin (IL)-1RA led to chondrogenesis and resistance to cytokine-induced degradation.15,143
Ligaments, tendons, and intervertebral disc
Ligaments and tendons are MS soft tissue components made of dense fiber structures responsible for connecting bone to bone and muscle to bone, respectively. They play a critical role in the MS system by transferring mechanical loads to guide joint motions, while maintaining the appropriate anatomic alignment. Ligaments and tendons are essentially composed of type I collagen (∼80% of dry weight), proteoglycans, elastin, glycolipids, and cells.144 The sparse density of cells, mainly fibroblasts (termed ligamentocytes and tenocytes, respectively), is a key factor for the limited regenerative capacity of ligaments and tendons. Ligament injuries usually occur in human joints during a traumatic event and initiates a degenerative cycle leading to osteoarthritis, whereas tendon injuries occur by repeated motion and degeneration over time.145,146 Currently, treatments include rest, physical therapy, splinting and orthosis, medications, and reconstructive surgeries using biological grafts (autografts and allografts).147 However, the healing of connective tissue injuries is usually incomplete, leaving the patient with residual pain, joint dysfunction, and functional disabilities.147 In the case of surgeries, there is still a risk of disease transmission, lack of appropriate donors, immune response, and high costs.148
Gene and cellular therapies, or the combination of both, emerge as possible future therapies for ligament and tendon treatment. The anterior cruciate ligament has been the focus of recent in vitro work. In one approach, a chondroitin sulfate-hyaluronate acid-silk fibroin (CHS) scaffold was fabricated with lentivectors encoding TGF-β3 immobilized within the scaffold using phosphatidylserine. These scaffolds were then seeded with MSCs to generate cartilage-like tissue in vitro. The TGF-β3 released by transfected MSCs could induce the chondral differentiation of MSCs, while the silk significantly increased the tensile strength of the scaffold to meet mechanical requirements.108 Several ex vivo studies have been also developed to reconstitute the periodontal ligament. In this study, chitosan/collagen scaffolds containing genetically modified human periodontal ligament cells were used to create a three-dimensional microenvironment.149,150 Work in tendon injury is also being undertaken. An ex vivo study showed the differentiation of MSCs into tenocytes after transfection with pDNA expressing Smad8 and BMP-2. The transfected cells were seeded onto a collagen sponge and then placed within the tendon defect. The transfected MSCs survived and induced tendon tissue regeneration.151 PLGA nanoparticles have also been used to release plasmids expressing RNAi for (TGF)-β1, with the downregulation of this gene leading to reduced inflammation, necrosis, and fibrosis in the injured tendon.152,153
The intervertebral disk (IVD) represents a heterogeneous fibrocartilaginous tissue located between each vertebrae of the spinal column. Structurally, the IVD is composed of three distinct parts, including the nucleus pulposus (NP), the annulus fibrosus (AF), and the cartilaginous endplates. Functionally, the IVD is fundamental to absorb and disperse energy and transfer loads within the column. The IVD is avascular and hypoxic and, therefore, IVD regeneration poses great challenges for the tissue engineering field. Critically, a major challenge facing IVD gene therapy is the poor transfection of NP and AF cells. HA chemically cross-linked using polyamidoamine (PAMAM) has been tested to address this limitation. The resultant particles complexed DNA efficiently and exhibited minimal cytotoxicity toward IVD cells at normal working concentrations.154 In addition, a recent study described a high transduction efficiency using LV expressing the reporter gene luciferase to transduce NP cells. An injectable laminin-111-functionalized PEG (PEG-LM111) hydrogel was developed as a biomaterial carrier for NP-transduced delivery to the IVD. The presence of biomaterial was responsible for a significant increase of cell retention in the IVD.155
Conclusions and Future Directions
Gene therapy is a promising route forward for MS repair and regeneration, but efficient and safe delivery remain limitations. Biomaterials as carriers for genetic vectors represent a versatile tool where advancements in tissue engineering strategies can be utilized to address these limitations. There is a wealth of research dedicated to the design of suitable biomaterials for use in MS tissues and the design of increasingly efficacious gene delivery vectors. Recent work has demonstrated that MS regeneration can be enhanced by combining gene delivery vectors with these biomaterial systems. This approach is useful for circumventing biological and engineering barriers to MS gene delivery. Biomaterial–vector interactions can be manipulated to preserve or enhance the vector functionality, avoid potential immune response, and provide a localized presentation of vectors and modified cells to MS tissue.
The potential for developments in tissue engineering material strategies and genetic vector design to complement each other is beginning to be explored. However, there is also an opportunity for engineering systems that are designed to be complimentary. In one interesting approach for immobilizing AAV within a scaffold, a FLAG-tagged chimeric mutant of AAV2 was created, which allowed for the immobilization to polymeric scaffolding covalently modified with anti-FLAG antibodies. The chimeric AAV2 retained transduction efficiency, and localized and efficient transduction was observed in vitro.156 In a similar manner, it has also been demonstrated that immobilizing lentivectors can be achieved by modifying the viral envelope. Peptide sequences recognized by factor XIII and plasmin were fused into VSV-G and this modified viral envelope was used for lentivector pseudotyping. The resulting lentivector covalently binds to fibrin hydrogels with release that is dependent upon enzymatic degradation.157,158 This “bottom-up” approach can create a substantial level of control in how vectors are presented to cells ex vivo and in vivo. Future directions should look at biomaterials that have been optimized for a given tissue and seek to incorporate vectors with this level of engineering design to further improve on the safety and efficacy concern with gene delivery.
Disclosure Statement
No competing financial interests exist.
References
- 1.Ginn S.L., et al. Gene therapy clinical trials worldwide to 2012—an update. J Gene Med 15, 65, 2013 [DOI] [PubMed] [Google Scholar]
- 2.Nel A.E., et al. Understanding biophysicochemical interactions at the nano-bio interface. Nat Mater 8, 543, 2009 [DOI] [PubMed] [Google Scholar]
- 3.Cejka P. DNA end resection: nucleases team up with the right partners to initiate homologous recombination. J Biol Chem 290, 22931, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wolff J.A., and Budker V. The mechanism of naked DNA uptake and expression. In: Huang L., Hung M.-C., Wagner E., eds. Advances in Genetics. Cambridge, Massachusetts: Academic Press, 2005, pp. 1–20 [DOI] [PubMed] [Google Scholar]
- 5.Shorten P.R., and Sneyd J. A Mathematical analysis of obstructed diffusion within skeletal muscle. Biophys J 96, 4764, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Papadopoulos S., Jürgens K.D., and Gros G. Protein diffusion in living skeletal muscle fibers: dependence on protein size, fiber type, and contraction. Biophys J 79, 2094, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Davis H.L., et al. Plasmid DNA is superior to viral vectors for direct gene transfer into adult mouse skeletal muscle. Hum Gene Ther 4, 733, 1993 [DOI] [PubMed] [Google Scholar]
- 8.Cu Y. and Saltzman, W.M. Mathematical modeling of molecular diffusion through mucus. Adv Drug Deliv Rev 61, 101, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Leukers B., et al. Hydroxyapatite scaffolds for bone tissue engineering made by 3D printing. J Mater Sci Mater Med 16, 1121, 2005 [DOI] [PubMed] [Google Scholar]
- 10.Drury J.L., and Mooney D.J. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 24, 4337, 2003 [DOI] [PubMed] [Google Scholar]
- 11.Delaittre G., et al. Chemical approaches to synthetic polymer surface biofunctionalization for targeted cell adhesion using small binding motifs. Soft Matter 8, 7323, 2012 [Google Scholar]
- 12.Waehler R., Russell S.J., and Curiel D.T. Engineering targeted viral vectors for gene therapy. Nat Rev Genet 8, 573, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Thomas A.M., and Shea L.D. Polysaccharide-modified scaffolds for controlled lentivirus delivery in vitro and after spinal cord injury. J Control Release 170, 421, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shin S., and Shea L.D. Lentivirus immobilization to nanoparticles for enhanced and localized delivery from hydrogels. Mol Ther 18, 700, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Brunger J.M., et al. Scaffold-mediated lentiviral transduction for functional tissue engineering of cartilage. Proc Natl Acad Sci U S A 111, E798, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Doukas J., et al. Delivery of FGF genes to wound repair cells enhances arteriogenesis and myogenesis in skeletal muscle. Mol Ther 5, 517, 2002 [DOI] [PubMed] [Google Scholar]
- 17.Hobson D.A., Pandori M.W., and Sano T. In situ transduction of target cells on solid surfaces by immobilized viral vectors. BMC Biotechnol 3, 4, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chorny M., et al. Adenoviral gene vector tethering to nanoparticle surfaces results in receptor-independent cell entry and increased transgene expression. Mol Ther 14, 382, 2006 [DOI] [PubMed] [Google Scholar]
- 19.Brown A.C., and Barker T.H. Fibrin-based biomaterials: modulation of macroscopic properties through rational design at the molecular level. Acta Biomater 10, 1502, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chattopadhyay S., and Raines R.T. Review collagen-based biomaterials for wound healing. Biopolymers 101, 821, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee K.Y., and Mooney D.J. Alginate: properties and biomedical applications. Prog Polym Sci 37, 106, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Raftery R., O'Brien F.J., and Cryan S.A. Chitosan for gene delivery and orthopedic tissue engineering applications. Molecules 18, 5611, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bakaic E., Smeets N.M.B., and Hoare T. Injectable hydrogels based on poly(ethylene glycol) and derivatives as functional biomaterials. RSC Adv 5, 35469, 2015 [Google Scholar]
- 24.Gentile P., et al. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int J Mol Sci 15, 3640, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dash T.K., and Konkimalla V.B. Poly-ε-caprolactone based formulations for drug delivery and tissue engineering: a review. J Control Release 158, 15, 2012 [DOI] [PubMed] [Google Scholar]
- 26.Chira S., et al. Progresses towards safe and efficient gene therapy vectors. Oncotarget 6, 30675, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Appaiahgari M.B., and Vrati S. Adenoviruses as gene/vaccine delivery vectors: promises and pitfalls. Expert Opin Biol Ther 15, 337, 2015 [DOI] [PubMed] [Google Scholar]
- 28.Stewart P.L., et al. Image reconstruction reveals the complex molecular organization of adenovirus. Cell 67, 145, 1991 [DOI] [PubMed] [Google Scholar]
- 29.Fields B.N., Knipe D.M., and Howley P.M. Fields Virology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins, 2013, pp. 1733–1762 [Google Scholar]
- 30.Mathias P., et al. Multiple adenovirus serotypes use alpha v integrins for infection. J Virol 68, 6811, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gene Therapy Trials Worldwide. 2016; Available from: www.wiley.com/legacy/wileychi/genmed/clinical Last accessed September15, 2016
- 32.Li Y., et al. Key points of basic theories and clinical practice in rAd-p53 (Gendicine) gene therapy for solid malignant tumors. Expert Opin Biol Ther 15, 437, 2015 [DOI] [PubMed] [Google Scholar]
- 33.Sheridan C. Gene therapy finds its niche. Nat Biotechnol 29, 121, 2011 [DOI] [PubMed] [Google Scholar]
- 34.Leen A.M., et al. Identification of hexon-specific CD4 and CD8 T-cell epitopes for vaccine and immunotherapy. J Virol 82, 546, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Choi J.W., et al. Local sustained delivery of oncolytic adenovirus with injectable alginate gel for cancer virotherapy. Gene Ther 20, 880, 2013 [DOI] [PubMed] [Google Scholar]
- 36.Kalyanasundaram S., et al. Coacervate microspheres as carriers of recombinant adenoviruses. Cancer Gene Ther 6, 107, 1999 [DOI] [PubMed] [Google Scholar]
- 37.Atchison R.W., Casto B.C., and Hammon W.M. Adenovirus-associated defective virus particles. Science 149, 754, 1965 [DOI] [PubMed] [Google Scholar]
- 38.Hoggan M.D., Blacklow N.R., and Rowe W.P. Studies of small DNA viruses found in various adenovirus preparations: physical, biological, and immunological characteristics. Proc Natl Acad Sci U S A 55, 1467, 1966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Srivastava A., Lusby E.W., and Berns K.I. Nucleotide sequence and organization of the adeno-associated virus 2 genome. J Virol 45, 555, 1983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Samulski R.J., and Muzyczka N. AAV-mediated gene therapy for research and therapeutic purposes. Annu Rev Virol 1, 427, 2014 [DOI] [PubMed] [Google Scholar]
- 41.DiMattia M.A., et al. Structural insight into the unique properties of adeno-associated virus serotype 9. J of Virol 86, 6947, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Im D.S., and Muzyczka N. The AAV origin binding protein Rep68 is an ATP-dependent site-specific endonuclease with DNA helicase activity. Cell 61, 447, 1990 [DOI] [PubMed] [Google Scholar]
- 43.King J.A., et al. DNA helicase-mediated packaging of adeno-associated virus type 2 genomes into preformed capsids. EMBO J 20, 3282, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Im D.S., and Muzyczka N. Partial purification of adeno-associated virus Rep78, Rep52, and Rep40 and their biochemical characterization. J Virol 66, 1119, 1992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sonntag F., et al. Adeno-associated virus type 2 capsids with externalized VP1/VP2 trafficking domains are generated prior to passage through the cytoplasm and are maintained until uncoating occurs in the nucleus. J Virol 80, 11040, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sonntag F., Schmidt K., and Kleinschmidt J.A. A viral assembly factor promotes AAV2 capsid formation in the nucleolus. Proc Natl Acad Sci U S A 107, 10220, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Grieger J.C., Snowdy S., and Samulski R.J. Separate basic region motifs within the adeno-associated virus capsid proteins are essential for infectivity and assembly. J Virol 80, 5199, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Flotte T.R., Afione S.A., and Zeitlin P.L. Adeno-associated virus vector gene expression occurs in nondividing cells in the absence of vector DNA integration. Am J Respir Cell Mol Biol 11, 517, 1994 [DOI] [PubMed] [Google Scholar]
- 49.Penaud-Budloo M., et al. Adeno-associated virus vector genomes persist as episomal chromatin in primate muscle. J Virol 82, 7875, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nakai H., et al. Extrachromosomal recombinant adeno-associated virus vector genomes are primarily responsible for stable liver transduction in vivo. J Virol 75, 6969, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Schnepp B.C., et al. Genetic fate of recombinant adeno-associated virus vector genomes in muscle. J Virol 77, 3495, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Flannery J.G., et al. Efficient photoreceptor-targeted gene expression in vivo by recombinant adeno-associated virus. Proc Natl Acad Sci U S A 94, 6916, 1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Peel A.L., et al. Efficient transduction of green fluorescent protein in spinal cord neurons using adeno-associated virus vectors containing cell type-specific promoters. Gene Ther 4, 16, 1997 [DOI] [PubMed] [Google Scholar]
- 54.Snyder R.O., et al. Efficient and stable adeno-associated virus-mediated transduction in the skeletal muscle of adult immunocompetent mice. Hum Gene Ther 8, 1891, 1997 [DOI] [PubMed] [Google Scholar]
- 55.Acland G.M., et al. Long-term restoration of rod and cone vision by single dose rAAV-mediated gene transfer to the retina in a canine model of childhood blindness. Mol Ther 12, 1072, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu Z., Asokan A., and Samulski R.J. Adeno-associated virus serotypes: vector toolkit for human gene therapy. Mol Ther 14, 316, 2006 [DOI] [PubMed] [Google Scholar]
- 57.Lisowski L., Tay S.S., and Alexander I.E. Adeno-associated virus serotypes for gene therapeutics. Curr Opin Pharmacol 24, 59, 2015 [DOI] [PubMed] [Google Scholar]
- 58.Yang L., Li J., and Xiao X. Directed evolution of adeno-associated virus (AAV) as vector for muscle gene therapy. Methods Mol Biol 709, 127, 2011 [DOI] [PubMed] [Google Scholar]
- 59.Buning H., et al. Engineering the AAV capsid to optimize vector-host-interactions. Curr Opin Pharmacol 24, 94, 2015 [DOI] [PubMed] [Google Scholar]
- 60.Boutin S., et al. Prevalence of serum IgG and neutralizing factors against adeno-associated virus (AAV) types 1, 2, 5, 6, 8, and 9 in the healthy population: implications for gene therapy using AAV vectors. Hum Gene Ther 21, 704, 2010 [DOI] [PubMed] [Google Scholar]
- 61.Manno C.S., et al. AAV-mediated factor IX gene transfer to skeletal muscle in patients with severe hemophilia B. Blood 101, 2963, 2003 [DOI] [PubMed] [Google Scholar]
- 62.Rey-Rico A., and Cucchiarini M. Controlled release strategies for rAAV-mediated gene delivery. Acta Biomater 29, 1, 2016 [DOI] [PubMed] [Google Scholar]
- 63.Blessing D., and Deglon N. Adeno-associated virus and lentivirus vectors: a refined toolkit for the central nervous system. Curr Opin Virol 21, 61, 2016 [DOI] [PubMed] [Google Scholar]
- 64.Briggs J.A.G., et al. The mechanism of HIV-1 core assembly: insights from three-dimensional reconstructions of authentic virions. Structure 14, 15, 2006 [DOI] [PubMed] [Google Scholar]
- 65.Dull T., et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 72, 8463, 1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Naldini L., et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272, 263, 1996 [DOI] [PubMed] [Google Scholar]
- 67.Page K.A., Landau N.R., and Littman D.R. Construction and use of a human immunodeficiency virus vector for analysis of virus infectivity. J Virol 64, 5270, 1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yu S.F., et al. Self-inactivating retroviral vectors designed for transfer of whole genes into mammalian cells. Proc Natl Acad Sci U S A 83, 3194, 1986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sakuma T., Barry M.A., and Ikeda Y. Lentiviral vectors: basic to translational. Biochem J 443, 603, 2012 [DOI] [PubMed] [Google Scholar]
- 70.Freed E.O., and Martin M.A. HIV-1 infection of non-dividing cells. Nature 369, 107, 1994 [DOI] [PubMed] [Google Scholar]
- 71.Bukrinsky M.I., et al. A nuclear localization signal within HIV-1 matrix protein that governs infection of non-dividing cells. Nature 365, 666, 1993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kobayashi H., et al. Neonatal gene therapy of MPS I mice by intravenous injection of a lentiviral vector. Mol Ther 11, 776, 2005 [DOI] [PubMed] [Google Scholar]
- 73.Di Domenico C., et al. Gene therapy for a mucopolysaccharidosis type I murine model with lentiviral-IDUA vector. Hum Gene Ther 16, 81, 2005 [DOI] [PubMed] [Google Scholar]
- 74.Meng J., et al. Autologous skeletal muscle derived cells expressing a novel functional dystrophin provide a potential therapy for Duchenne Muscular Dystrophy. Sci Rep 6, 19750, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Quenneville S.P., et al. Autologous transplantation of muscle precursor cells modified with a lentivirus for muscular dystrophy: human cells and primate models. Mol Ther 15, 431, 2007 [DOI] [PubMed] [Google Scholar]
- 76.Carbonaro D.A., et al. Preclinical demonstration of lentiviral vector-mediated correction of immunological and metabolic abnormalities in models of adenosine deaminase deficiency. Mol Ther 22, 607, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Montiel-Equihua C.A., Thrasher A.J., and Gaspar H.B. Gene therapy for severe combined immunodeficiency due to adenosine deaminase deficiency. Curr Gene Ther 12, 57, 2012 [DOI] [PubMed] [Google Scholar]
- 78.Liu A., et al. Lentivirus-mediated shRNA interference of ghrelin receptor blocks proliferation in the colorectal cancer cells. Cancer Med 5, 2417, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.de Melo S.M., et al. The anti-tumor effects of adipose tissue mesenchymal stem cell transduced with HSV-Tk gene on U-87-driven brain tumor. PLoS One 10, e0128922, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.da Cunha F.F., et al. Comparison of treatments of peripheral arterial disease with mesenchymal stromal cells and mesenchymal stromal cells modified with granulocyte and macrophage colony-stimulating factor. Cytotherapy 15, 820, 2013 [DOI] [PubMed] [Google Scholar]
- 81.Beegle J.R., et al. Preclinical evaluation of mesenchymal stem cells overexpressing VEGF to treat critical limb ischemia. Mol Ther Methods Clin Dev 3, 16053, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Revilla S., et al. Lenti-GDNF gene therapy protects against Alzheimer's disease-like neuropathology in 3xTg-AD mice and MC65 cells. CNS Neurosci Ther 20, 961, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schroder A.R., et al. HIV-1 integration in the human genome favors active genes and local hotspots. Cell 110, 521, 2002 [DOI] [PubMed] [Google Scholar]
- 84.Mitchell R.S., et al. Retroviral DNA integration: ASLV, HIV, and MLV show distinct target site preferences. PLoS Biol 2, E234, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wang G.P., et al. HIV integration site selection: analysis by massively parallel pyrosequencing reveals association with epigenetic modifications. Genome Res 17, 1186, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pannell D., and Ellis J. Silencing of gene expression: implications for design of retrovirus vectors. Rev Med Virol 11, 205, 2001 [DOI] [PubMed] [Google Scholar]
- 87.Stilhano R.S., et al. Injectable alginate hydrogel for enhanced spatiotemporal control of lentivector delivery in murine skeletal muscle. J Control Release 237, 42, 2016 [DOI] [PubMed] [Google Scholar]
- 88.Thomas A.M., et al. Heparin-chitosan nanoparticle functionalization of porous poly(ethylene glycol) hydrogels for localized lentivirus delivery of angiogenic factors. Biomaterials 35, 8687, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Boehler R.M., et al. A PLG/HAp composite scaffold for lentivirus delivery. Biomaterials 34, 5431, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Tuinstra H.M., et al. Multifunctional, multichannel bridges that deliver neurotrophin encoding lentivirus for regeneration following spinal cord injury. Biomaterials 33, 1618, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Bertoni C., et al. Enhancement of plasmid-mediated gene therapy for muscular dystrophy by directed plasmid integration. Proc Natl Acad Sci U S A 103, 419, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Essner J.J., McIvor R.S., and Hackett P.B. Awakening gene therapy with Sleeping Beauty transposons. Curr Opin Pharmacol 5, 513, 2005 [DOI] [PubMed] [Google Scholar]
- 93.Aronovich E.L., et al. Prolonged expression of a lysosomal enzyme in mouse liver after Sleeping Beauty transposon-mediated gene delivery: implications for non-viral gene therapy of mucopolysaccharidoses. J Gene Med 9, 403, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Paillard F. CpG: the double-edged sword. Hum Gene Ther 10, 2089, 1999 [DOI] [PubMed] [Google Scholar]
- 95.Chen Z.Y., et al. Silencing of episomal transgene expression by plasmid bacterial DNA elements in vivo. Gene Ther 11, 856, 2004 [DOI] [PubMed] [Google Scholar]
- 96.Chen Z.Y., He C.Y., and Kay M.A. Improved production and purification of minicircle DNA vector free of plasmid bacterial sequences and capable of persistent transgene expression in vivo. Hum Gene Ther 16, 126, 2005 [DOI] [PubMed] [Google Scholar]
- 97.Osborn M.J., et al. Minicircle DNA-based gene therapy coupled with immune modulation permits long-term expression of alpha-L-iduronidase in mice with mucopolysaccharidosis type I. Mol Ther 19, 450, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Lijkwan M.A., et al. Short hairpin RNA gene silencing of prolyl hydroxylase-2 with a minicircle vector improves neovascularization of hindlimb ischemia. Hum Gene Ther 25, 41, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wang Q., et al. In vivo electroporation of minicircle DNA as a novel method of vaccine delivery to enhance HIV-1-specific immune responses. J Virol 88, 1924, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ehrhardt A., et al. A direct comparison of two nonviral gene therapy vectors for somatic integration: in vivo evaluation of the bacteriophage integrase phiC31 and the Sleeping Beauty transposase. Mol Ther 11, 695, 2005 [DOI] [PubMed] [Google Scholar]
- 101.Gehl J. Electroporation: theory and methods, perspectives for drug delivery, gene therapy and research. Acta Physiol Scand 177, 437, 2003 [DOI] [PubMed] [Google Scholar]
- 102.Tomizawa M., et al. Sonoporation: gene transfer using ultrasound. World J Methodol 3, 39, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Zu Y., et al. Gold nanoparticles enhanced electroporation for mammalian cell transfection. J Biomed Nanotechnol 10, 982, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Torio-Padron N., et al. Implantation of VEGF transfected preadipocytes improves vascularization of fibrin implants on the cylinder chorioallantoic membrane (CAM) model. Minim Invasive Ther Allied Technol 16, 155, 2007 [DOI] [PubMed] [Google Scholar]
- 105.Kimelman-Bleich N., et al. Targeted gene-and-host progenitor cell therapy for nonunion bone fracture repair. Mol Ther 19, 53, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Li Y.H., et al. Targeted delivery of biodegradable nanoparticles with ultrasound-targeted microbubble destruction-mediated hVEGF-siRNA transfection in human PC-3 cells in vitro. Int J Mol Med 31, 163, 2013 [DOI] [PubMed] [Google Scholar]
- 107.Nomikou N., et al. Ultrasound-mediated gene transfer (sonoporation) in fibrin-based matrices: potential for use in tissue regeneration. J Tissue Eng Regen Med 10, 29, 2016 [DOI] [PubMed] [Google Scholar]
- 108.Sun L., et al. Immobilized lentivirus vector on chondroitin sulfate-hyaluronate acid-silk fibroin hybrid scaffold for tissue-engineered ligament-bone junction. Biomed Res Int 2014, 816979, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Rezaee M., et al. Progress in the development of lipopolyplexes as efficient non-viral gene delivery systems. J Control Release 236, 1, 2016 [DOI] [PubMed] [Google Scholar]
- 110.Yin H., et al. Non-viral vectors for gene-based therapy. Nat Rev Genet 15, 541, 2014 [DOI] [PubMed] [Google Scholar]
- 111.Jones C.H., et al. Overcoming nonviral gene delivery barriers: perspective and future. Mol Pharm 10, 4082, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Einhorn T.A., and Gerstenfeld L.C. Fracture healing: mechanisms and interventions. Nat Rev Rheumatol 11, 45, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Phillips J.E., Gersbach C.A., and García A.J. Virus-based gene therapy strategies for bone regeneration. Biomaterials 28, 211, 2007 [DOI] [PubMed] [Google Scholar]
- 114.Bose S., Roy M., and Bandyopadhyay A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol 30, 546, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Fang J., et al. Stimulation of new bone formation by direct transfer of osteogenic plasmid genes. Proc Natl Acad Sci U S A 93, 5753, 1996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Dupont K.M., et al. Synthetic scaffold coating with adeno-associated virus encoding BMP2 to promote endogenous bone repair. Cell Tissue Res 347, 575, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Curtin C.M., et al. Combinatorial gene therapy accelerates bone regeneration: non-viral dual delivery of VEGF and BMP2 in a collagen-nanohydroxyapatite scaffold. Adv Healthc Mat 4, 223, 2015 [DOI] [PubMed] [Google Scholar]
- 118.Lee S., et al. Electrospun nanofibrous scaffolds for controlled release of adeno-associated viral vectors. Acta Biomater 7, 3868, 2011 [DOI] [PubMed] [Google Scholar]
- 119.Evans C.H., and Huard J. Gene therapy approaches to regenerating the musculoskeletal system. Nat Rev Rheumatol 11, 234, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Prisk V., and Huard J. Muscle injuries and repair: the role of prostaglandins and inflammation. Histol Histopathol 18, 1243, 2003 [DOI] [PubMed] [Google Scholar]
- 121.Jarvinen T.A., et al. Muscle injuries: biology and treatment. Am J Sports Med 33, 745, 2005 [DOI] [PubMed] [Google Scholar]
- 122.Bedair H.S., et al. Angiotensin II receptor blockade administered after injury improves muscle regeneration and decreases fibrosis in normal skeletal muscle. Am J Sports Med 36, 1548, 2008 [DOI] [PubMed] [Google Scholar]
- 123.Nozaki M., et al. Improved muscle healing after contusion injury by the inhibitory effect of suramin on myostatin, a negative regulator of muscle growth. Am J Sports Med 36, 2354, 2008 [DOI] [PubMed] [Google Scholar]
- 124.Worrell T.W. Factors associated with hamstring injuries. An approach to treatment and preventative measures. Sports Med 17, 338, 1994 [DOI] [PubMed] [Google Scholar]
- 125.Menetrey J., et al. Suturing versus immobilization of a muscle laceration. A morphological and functional study in a mouse model. Am J Sports Med 27, 222, 1999 [DOI] [PubMed] [Google Scholar]
- 126.Stilhano R.S., et al. Gene and cell therapy for muscle regeneration. Curr Rev Musculoskelet Med 8, 182, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Qazi T.H., et al. Biomaterials based strategies for skeletal muscle tissue engineering: existing technologies and future trends. Biomaterials 53, 502, 2015 [DOI] [PubMed] [Google Scholar]
- 128.Hill E., Boontheekul T., and Mooney D.J. Designing scaffolds to enhance transplanted myoblast survival and migration. Tissue Eng 12, 1295, 2006 [DOI] [PubMed] [Google Scholar]
- 129.Borselli C., et al. Functional muscle regeneration with combined delivery of angiogenesis and myogenesis factors. Proc Natl Acad Sci U S A 107, 3287, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Zhou W., et al. Angiogenic gene-modified myoblasts promote vascularization during repair of skeletal muscle defects. J Tissue Eng Regen Med 9, 1404, 2015 [DOI] [PubMed] [Google Scholar]
- 131.Kasahara H., et al. Biodegradable gelatin hydrogel potentiates the angiogenic effect of fibroblast growth factor 4 plasmid in rabbit hindlimb ischemia. J Am Coll Cardiol 41, 1056, 2003 [DOI] [PubMed] [Google Scholar]
- 132.Schek R.M., Hollister S.J., and Krebsbach P.H. Delivery and protection of adenoviruses using biocompatible hydrogels for localized gene therapy. Mol Ther 9, 130, 2004 [DOI] [PubMed] [Google Scholar]
- 133.Vinatier C., and Guicheux J. Cartilage tissue engineering: from biomaterials and stem cells to osteoarthritis treatments. Ann Phys Rehabil Med 59, 139, 2016 [DOI] [PubMed] [Google Scholar]
- 134.Bernhard J.C., and Vunjak-Novakovic G. Should we use cells, biomaterials, or tissue engineering for cartilage regeneration? Stem Cell Res Ther 7, 56, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Embree M.C., et al. Exploiting endogenous fibrocartilage stem cells to regenerate cartilage and repair joint injury. Nat Commun 7, 13073, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Merkle D., and McDonald D.D. Use of recommended osteoarthritis pain treatment by older adults. J Adv Nurs 65, 828, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ondresik M., et al. Management of knee osteoarthritis. Current status and future trends. Biotechnol Bioeng 114, 717, 2016 [DOI] [PubMed] [Google Scholar]
- 138.Steinert A.F., Nöth U., and Tuan R.S. Concepts in gene therapy for cartilage repair. Injury 39, 97, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Chung C., and Burdick J.A. Engineering cartilage tissue. Adv Drug Deliv Rev 60, 243, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Wang W., et al. In vivo restoration of full-thickness cartilage defects by poly(lactide-co-glycolide) sponges filled with fibrin gel, bone marrow mesenchymal stem cells and DNA complexes. Biomaterials 31, 5953, 2010 [DOI] [PubMed] [Google Scholar]
- 141.Li B., et al. Fabrication of poly(lactide-co-glycolide) scaffold filled with fibrin gel, mesenchymal stem cells, and poly(ethylene oxide)-b-poly(L-lysine)/TGF-β1 plasmid DNA complexes for cartilage restoration in vivo. J Biomed Mater Res A 101, 3097, 2013 [DOI] [PubMed] [Google Scholar]
- 142.Lu H., et al. Porous chitosan scaffolds with embedded hyaluronic acid/chitosan/plasmid-DNA nanoparticles encoding TGF-beta1 induce DNA controlled release, transfected chondrocytes, and promoted cell proliferation. PLoS One 8, e69950, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Moutos F.T., et al. Anatomically shaped tissue-engineered cartilage with tunable and inducible anticytokine delivery for biological joint resurfacing. Proc Natl Acad Sci 113, E4513, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Subramanian A., and Schilling T.F. Tendon development and musculoskeletal assembly: emerging roles for the extracellular matrix. Development 142, 4191, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Fleming B.C., et al. Ligament injury, reconstruction and osteoarthritis. Curr Opin Orthop 16, 354, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Lin T.W., Cardenas L., and Soslowsky L.J. Biomechanics of tendon injury and repair. J Biomech 37, 865, 2004 [DOI] [PubMed] [Google Scholar]
- 147.Brown M.N., Shiple B.J., and Scarpone M. Regenerative approaches to tendon and ligament conditions. Phys Med Rehabil Clin N Am 27, 941, 2016 [DOI] [PubMed] [Google Scholar]
- 148.Petrigliano F.A., McAllister D.R., and Wu B.M. Tissue engineering for anterior cruciate ligament reconstruction: a review of current strategies. Arthroscopy 22, 441, 2006 [DOI] [PubMed] [Google Scholar]
- 149.Peng L., et al. Novel gene-activated matrix with embedded chitosan/plasmid DNA nanoparticles encoding PDGF for periodontal tissue engineering. J Biomed Mater Res A 90, 564, 2009 [DOI] [PubMed] [Google Scholar]
- 150.Zhang Y., et al. Novel chitosan/collagen scaffold containing transforming growth factor-β1 DNA for periodontal tissue engineering. Biochem Biophy Res Commun 344, 362, 2006 [DOI] [PubMed] [Google Scholar]
- 151.Hoffmann A., et al. Neotendon formation induced by manipulation of the Smad8 signalling pathway in mesenchymal stem cells. J Clin Invest 116, 940, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Zhou Y., et al. Nanoparticle-mediated delivery of TGF-beta1 miRNA plasmid for preventing flexor tendon adhesion formation. Biomaterials 34, 8269, 2013 [DOI] [PubMed] [Google Scholar]
- 153.Zhou Y., et al. Effective modulation of transforming growth factor-beta1 expression through engineered microRNA-based plasmid-loaded nanospheres. Cytotherapy 17, 320, 2015 [DOI] [PubMed] [Google Scholar]
- 154.Srivastava A., et al. Improved gene transfection efficacy and cytocompatibility of multifunctional polyamidoamine-cross-linked hyaluronan particles. Macromol Biosci 15, 682, 2015 [DOI] [PubMed] [Google Scholar]
- 155.Francisco A.T., et al. Injectable laminin-functionalized hydrogel for nucleus pulposus regeneration. Biomaterials 34, 7381, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Li H., et al. Immobilization of FLAG-tagged recombinant adeno-associated virus 2 onto tissue engineering scaffolds for the improvement of transgene delivery in cell transplants. PLoS One 10, e0129013, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Padmashali R.M., and Andreadis S.T. Engineering fibrinogen-binding VSV-G envelope for spatially- and cell-controlled lentivirus delivery through fibrin hydrogels. Biomaterials 32, 3330, 2011 [DOI] [PubMed] [Google Scholar]
- 158.Raut S.D., et al. Fibrin-mediated lentivirus gene transfer: implications for lentivirus microarrays. J Control Release 144, 213, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Cotten M., and Weber J.M. The adenovirus protease is required for virus entry into host cells. Virology 213, 494, 1995 [DOI] [PubMed] [Google Scholar]
- 160.Moon C.Y., et al. Dual tumor targeting with pH-sensitive and bioreducible polymer-complexed oncolytic adenovirus. Biomaterials 41, 53, 2015 [DOI] [PubMed] [Google Scholar]
- 161.Oliver C.J., Shortridge K.F., and Belyanin G. Diffusion coefficient and molecular weight of type 5 adenovirus by photon-correlation spectroscopy. Biochimica et Biophysica Acta 437, 589, 1976 [DOI] [PubMed] [Google Scholar]
- 162.Zhang Y., and Bergelson J.M. Adenovirus receptors. J Virol 79, 12125, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Wright J.F., et al. Identification of factors that contribute to recombinant AAV2 particle aggregation and methods to prevent its occurrence during vector purification and formulation. Mol Ther 12, 171, 2005 [DOI] [PubMed] [Google Scholar]
- 164.Le H.T., et al. Utility of PEGylated recombinant adeno-associated viruses for gene transfer. J Control Release 108, 161, 2005 [DOI] [PubMed] [Google Scholar]
- 165.Seisenberger G., et al. Real-time single-molecule imaging of the infection pathway of an adeno-associated virus. Science 294, 1929, 2001 [DOI] [PubMed] [Google Scholar]
- 166.Nonnenmacher M., and Weber T. Intracellular transport of recombinant adeno-associated virus vectors. Gene Ther 19, 649, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Croyle M.A., et al. PEGylation of a vesicular stomatitis virus G pseudotyped lentivirus vector prevents inactivation in serum. J Virol 78, 912, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.You J.-O., and Peng C.-A. Phagocytosis-mediated retroviral transduction: co-internalization of deactivated retrovirus and calcium-alginate microspheres by macrophages. J Gene Med 7, 398, 2005 [DOI] [PubMed] [Google Scholar]
- 169.Lai B.E., et al. Transport theory for HIV diffusion through in vivo distributions of topical microbicide gels. Biophys J 97, 2379, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Finkelshtein D., et al. LDL receptor and its family members serve as the cellular receptors for vesicular stomatitis virus. Proc Natl Acad of Sci 110, 7306, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Frecha C., et al. Advances in the Field of lentivector-based transduction of T and B lymphocytes for gene therapy. Mol Ther 18, 1748, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Skoumal M., et al. Localized lentivirus delivery via peptide interactions. Biotechnol Bioeng 113, 2033, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Shin S., et al. Phosphatidylserine immobilization of lentivirus for localized gene transfer. Biomaterials 31, 4353, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Shen H., Hu Y., and Saltzman W.M. DNA diffusion in mucus: effect of Size, topology of DNAs, and transfection reagents. Biophys J 91, 639, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Neumann A.J., et al. Enhanced adenovirus transduction of hMSCs using 3D hydrogel cell carriers. Mol Biotechnol 53, 207, 2013 [DOI] [PubMed] [Google Scholar]
- 176.Park H., et al. Fabrication of cross-linked alginate beads using electrospraying for adenovirus delivery. Int J Pharm 427, 417, 2012 [DOI] [PubMed] [Google Scholar]
- 177.Sailaja G., et al. Encapsulation of recombinant adenovirus into alginate microspheres circumvents vector-specific immune response. Gene Therapy 9, 1722, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Koc A., et al. Evaluation of adenoviral vascular endothelial growth factor-activated chitosan/hydroxyapatite scaffold for engineering vascularized bone tissue using human osteoblasts: in vitro and in vivo studies. J Biomater Appl 29, 748, 2014 [DOI] [PubMed] [Google Scholar]
- 179.Sonobe J., et al. Osteoinduction by bone morphogenetic protein 2-expressing adenoviral vector: application of biomaterial to mask the host immune response. Hum Gene Ther 15, 659, 2004 [DOI] [PubMed] [Google Scholar]
- 180.Chang P.C., et al. Adenovirus encoding human platelet-derived growth factor-B delivered to alveolar bone defects exhibits safety and biodistribution profiles favorable for clinical use. Hum Gene Ther 20, 486, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Hu W.W., Wang Z., and Krebsbach P.H. Virus immobilization on biomaterial scaffolds through biotin-avidin interaction for improving bone regeneration. J Tissue Eng Regen Med 10, E63, 2016 [DOI] [PubMed] [Google Scholar]
- 182.Yao X., et al. Systemic administration of a PEGylated adenovirus vector with a cancer-specific promoter is effective in a mouse model of metastasis. Gene Ther 16, 1395, 2009 [DOI] [PubMed] [Google Scholar]
- 183.Fan G., et al. Bio-inspired polymer envelopes around adenoviral vectors to reduce immunogenicity and improve in vivo kinetics. Acta Biomater 30, 94, 2016 [DOI] [PubMed] [Google Scholar]
- 184.Leggiero E., et al. PEGylated helper-dependent adenoviral vector expressing human Apo A-I for gene therapy in LDLR-deficient mice. Gene Ther 20, 1124, 2013 [DOI] [PubMed] [Google Scholar]
- 185.Yao H., et al. Functional characterization of a PEI-CyD-FA-coated adenovirus as delivery vector for gene therapy. Curr Med Chem 20, 2601, 2013 [DOI] [PubMed] [Google Scholar]
- 186.Singarapu K., Pal I., and Ramsey J.D. Polyethylene glycol-grafted polyethylenimine used to enhance adenovirus gene delivery. J Biomed Mater Res A 101, 1857, 2013 [DOI] [PubMed] [Google Scholar]
- 187.Liao I.C., et al. Sustained viral gene delivery through core-shell fibers. J Control Release 139, 48, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Mok H., Park J.W., and Park T.G. Microencapsulation of PEGylated adenovirus within PLGA microspheres for enhanced stability and gene transfection efficiency. Pharm Res 24, 2263, 2007 [DOI] [PubMed] [Google Scholar]
- 189.Lee H.H., et al. Release of bioactive adeno-associated virus from fibrin scaffolds: effects of fibrin glue concentrations. Tissue Eng Part A 17, 1969, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Schmidt C., et al. A slow-release fibrin matrix increases adeno-associated virus transduction of wound repair cells in vivo. J Biomater Appl 28, 1408, 2014 [DOI] [PubMed] [Google Scholar]
- 191.Hwang J.-H., et al. Heparin-coated superparamagnetic nanoparticle-mediated adeno-associated virus delivery for enhancing cellular transduction. Int J Pharm 421, 397, 2011 [DOI] [PubMed] [Google Scholar]
- 192.Rey-Rico A., et al. Effective and durable genetic modification of human mesenchymal stem cells via controlled release of rAAV vectors from self-assembling peptide hydrogels with a maintained differentiation potency. Acta Biomater 18, 118, 2015 [DOI] [PubMed] [Google Scholar]
- 193.Kim E., et al. Sticky “delivering-from” strategies using viral vectors for efficient human neural stem cell infection by bioinspired catecholamines. ACS Appl Mater Interfaces 6, 8288, 2014 [DOI] [PubMed] [Google Scholar]
- 194.Kim E., et al. Drawing sticky adeno-associated viruses on surfaces for spatially patterned gene expression. Angew Chem Int Ed Engl 51, 5598, 2012 [DOI] [PubMed] [Google Scholar]
- 195.Zeng Y.F., et al. Controlled delivery of recombinant adeno-associated virus serotype 2 using pH-sensitive poly(ethylene glycol)-poly-L-histidine hydrogels. Biomaterials 33, 9239, 2012 [DOI] [PubMed] [Google Scholar]
- 196.Tseng S.J., et al. Environment acidity triggers release of recombinant adeno-associated virus serotype 2 from a tunable matrix. J Control Release 170, 252, 2013 [DOI] [PubMed] [Google Scholar]
- 197.Rey-Rico A., et al. PEO-PPO-PEO micelles as effective rAAV-mediated gene delivery systems to target human mesenchymal stem cells without altering their differentiation potency. Acta Biomaterialia 27, 42, 2015 [DOI] [PubMed] [Google Scholar]
- 198.Mah C., et al. Improved method of recombinant AAV2 delivery for systemic targeted gene therapy. Mol Ther 6, 106, 2002 [DOI] [PubMed] [Google Scholar]
- 199.Madrigal J.L., et al. Microfluidic generation of alginate microgels for the controlled delivery of lentivectors. J Mater Chem B 4, 6989, 2016 [DOI] [PubMed] [Google Scholar]
- 200.Kidd M.E., Shin S., and Shea L.D. Fibrin hydrogels for lentiviral gene delivery in vitro and in vivo. J Control Release 157, 80, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Rodriguez A.L., et al. Tailoring minimalist self-assembling peptides for localized viral vector gene delivery. Nano Res 9, 674, 2016 [Google Scholar]
- 202.Shepard J.A., et al. Hydrogel macroporosity and the prolongation of transgene expression and the enhancement of angiogenesis. Biomaterials 33, 7412, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Thomas A.M., Palma J.L., and Shea L.D. Sponge-mediated lentivirus delivery to acute and chronic spinal cord injuries. J Control Release 204, 1, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Shin S., Salvay D.M., and Shea L.D. Lentivirus delivery by adsorption to tissue engineering scaffolds. J Biomed Mater Res A 93, 1252, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Aviles M.O., and Shea L.D. Hydrogels to modulate lentivirus delivery in vivo from microporous tissue engineering scaffolds. Drug Deliv Transl Res 1, 91, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Jen M.C., et al. Sustained, localized transgene expression mediated from lentivirus-loaded biodegradable polyester elastomers. J Biomed Mater Res A 101, 1328, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Bikram M., et al. Endochondral bone formation from hydrogel carriers loaded with BMP2-transduced cells. Ann Biomed Eng 35, 796, 2007 [DOI] [PubMed] [Google Scholar]
- 208.Kim S., et al. Sequential delivery of BMP-2 and IGF-1 using a chitosan gel with gelatin microspheres enhances early osteoblastic differentiation. Acta Biomater 8, 1768, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Moimas S., et al. AAV vector encoding human VEGF165-transduced pectineus muscular flaps increase the formation of new tissue through induction of angiogenesis in an in vivo chamber for tissue engineering: a technique to enhance tissue and vessels in microsurgically engineered tissue. J Tissue Eng 6, 2041731415611717, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.McClure M.J., et al. Role of integrin alpha7beta1 signaling in myoblast differentiation on aligned polydioxanone scaffolds. Acta Biomater 39, 44, 2016 [DOI] [PubMed] [Google Scholar]
- 211.Schillinger U., et al. A fibrin glue composition as carrier for nucleic acid vectors. Pharm Res 25, 2946, 2008 [DOI] [PubMed] [Google Scholar]
- 212.Shi J., et al. Nanoparticle delivery of the bone morphogenetic protein 4 gene to adipose-derived stem cells promotes articular cartilage repair in vitro and in vivo. Arthroscopy 29, 2001, 2013 e2 [DOI] [PubMed] [Google Scholar]
- 213.Gonzalez-Fernandez T., et al. Gene delivery of TGF-beta3 and BMP2 in an MSC-laden alginate hydrogel for articular cartilage and endochondral bone tissue engineering. Tissue Eng Part A 22, 776, 2016 [DOI] [PubMed] [Google Scholar]
- 214.Guo T., et al. Porous chitosan-gelatin scaffold containing plasmid DNA encoding transforming growth factor-beta1 for chondrocytes proliferation. Biomaterials 27, 1095, 2006 [DOI] [PubMed] [Google Scholar]
- 215.Kayabasi G.K., Aydin R.S., and Gumusderelioglu M. In vitro chondrogenesis by BMP6 gene therapy. J Biomed Mater Res A 101, 1353, 2013 [DOI] [PubMed] [Google Scholar]
- 216.Zhang Y., et al. A platelet-derived growth factor releasing chitosan/coral composite scaffold for periodontal tissue engineering. Biomaterials 28, 1515, 2007 [DOI] [PubMed] [Google Scholar]
- 217.Wang C.J., et al. pCMV-BMP-2-transfected cell-mediated gene therapy in anterior cruciate ligament reconstruction in rabbits. Arthroscopy 26, 968, 2010 [DOI] [PubMed] [Google Scholar]
- 218.Li D.D., et al. Characterization and cytocompatibility of thermosensitive hydrogel embedded with chitosan nanoparticles for delivery of bone morphogenetic protein-2 plasmid DNA. J Mater Sci Mater Med 27, 134, 2016 [DOI] [PubMed] [Google Scholar]
- 219.Xie Q., et al. Fabrication of core-shell PEI/pBMP2-PLGA electrospun scaffold for gene delivery to periodontal ligament stem cells. Stem Cells Int 2016, 5385137, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]